Abstract
In the past few decades, different efforts have been made toward chromium (VI) removal from wastewater. In this study, a Ti3C2Tx/PANI composite was synthesized via in situ polymerization from (PANI) and Mxene (Ti3C2Tx). The composite was characterized using XRD, FTIR, SEM–EDS, XPS, and Raman spectroscopy. FTIR spectra revealed characteristic functional groups of both Ti3C2Tx and PANI, with significant shifts and intensity changes upon Cr(VI) adsorption. EDS and XPS also showed the presence of Cr after adsorption. Raman spectroscopy further verified PANI polymerization and Cr(VI) adsorption. The adsorption performance for Cr(VI) is markedly influenced by the pH level of the solution, with optimal adsorption occurring at an acidic pH. The experimental findings showed good agreement with the Langmuir–Freundlich and Khan isotherm models, while the pseudo‐second‐order and MESO kinetic models best describe the adsorption process. The Ti3C2Tx/PANI composite exhibits a high adsorption capacity of 342.5 mg g−1 and good stability, maintaining its adsorption efficiency over seven cycles. The adsorption mechanism involves electrostatic attraction, surface complexation, and physical adsorption. Notably, the developed Ti3C2Tx/PANI composite exhibits a considerably higher Cr(VI) adsorption capacity than most previously reported MXene (Ti3C2Tx) and polyaniline (PANI)‐based adsorbents. This superior performance is attributed to the synergistic interaction between the conductive polymer PANI and the high surface reactivity of Ti3C2Tx layers, which enhances active site accessibility and electron transfer during adsorption. The findings demonstrate the composite’s potential as an efficient and robust adsorbent for Cr(VI) adsorption.
1. Introduction
The expansion of global populations and the pace of industrialization are primarily responsible for the escalation in water contamination levels [1]. Various studies have highlighted the detrimental impacts of water pollution emerging from the direct release of industrial effluents from industries such as textile, food, dye, and paint. These effluents are laden with a mix of different substances, posing a risk to living things, including humans, at a low level [2]. Such pollutants not only harm aquatic organisms but also lead to the degradation of aquatic ecosystems by escalating biological oxygen demand. Widely used in numerous industrial processes, chromium becomes a significant pollutant when discharged into water bodies, potentially leading to Cr(VI) contamination [3]. This contamination poses severe environmental and health risks [4]. To combat these challenges, research has explored various nanoparticles known for their pollutant adsorption capabilities, thanks to their expansive surface area and porosity. Among these are carbon materials [5], zeolites [6], metal oxides ferrous oxide (Fe3O2), manganese dioxide (MnO2), zinc oxide (ZnO), titanium dioxide (TiO2) [7–10], biochar [11], graphene and its derivatives [12], organometallic frameworks [13], carbon nanotubes (CNTs) [14], conductive polymers polyaniline (PANI), polypyrrole (Ppy), polythiophene (PTh) [15–17], and MXenes [18]. Researchers have shown that these materials, with their unique surface properties and affinity for hexavalent chromium ions, are particularly effective in removing chromium from wastewater. As a type of a two‐dimensional inorganic compound with thin layers of carbonitrides, transition metal nitrides or carbides. MXenes show promise for use in water treatment because they have a lot of surface area, are good at conducting electricity, and are hydrophilic [19–21]. They are identified by the formula Mn+1XnTx, where T is the surface termination group (Cl, OH, and F), and X is the number of terminations [22–24]. They are synthesized by etching MAX phases (Mn+1AXn), where M is the transition metal, A is the group IIIA/IVA element, and X is a C and/or N atom [25]. MXenes are known for having excellent characteristics such as high mechanical strength, excellent electrical conductivity, and broad hydrophilicity [26]. PANI is known for its nontoxicity, ease of synthesis, environmental stability, high conductivity, and cost‐effectiveness, making it suitable for various applications, including sensors, batteries, and anticorrosion coatings. Because it has many functional groups, a large surface area, and a variety of adsorption abilities, PANI is a good candidate for reducing agents. This is because it can be in a variety of oxidation states. However, the challenge of inadequate regeneration and recycling capabilities in bulk PANI necessitates the development of PANI‐based materials that are efficient in adsorbing Cr(VI) and capable of regeneration and reuse [27, 28]. Consequently, material composites combining MXenes and conductive polymers have emerged as potent solutions for water purification, leveraging their unique physicochemical properties [29]. This interest in composite materials stems from their innovative designs and potential to address water contamination effectively [30].
In this study, the Ti3C2Tx/PANI nanocomposite was synthesized via an intercalation of PANI and Mxene and then used for Cr(VI) absorption from the water environment. Characterization, synthesis, and batch adsorption investigations of the materials are all part of the systematic inquiry that makes up this experimental approach. The structural and compositional properties of the combined material have been elucidated through in‐depth physicochemical studies using techniques such as X‐ray diffractometer (XRD), Fourier transform infrared spectroscopy (FTIR), scanning electron microscopy–energy‐dispersive spectroscopy (SEM–EDS), X‐ray photoelectron spectroscopy (XPS) and Raman spectroscopy. Although various MXene‐ (Ti3C2Tx) and PANI‐based composites have been explored for heavy metal removal, most suffer from limited adsorption capacity, poor interfacial compatibility, or inadequate regeneration stability. In this study, a Ti3C2Tx/PANI composite was designed through a controlled in situ polymerization, ensuring strong interfacial interaction and enhanced electron transport. This significantly improves active site accessibility and redox reactivity toward Cr(VI) ions. The composite exhibits a notably higher adsorption capacity (342.5 mg g−1) and better reusability compared to many previously reported Ti3C2Tx‐ and PANI‐based adsorbents.
2. Materials and Methods
Titanium aluminum carbide (Ti3AlC2 99% purity) was used as the MAX phase precursor; dimethyl sulfoxide (DMSO 99.9%), potassium dichromate (K2Cr2O7 99.99%), hydrogen fluoride HF (99.98%), aniline (99.5%), APS (99.9%), and hydrogen chloride (HCl 35%) were purchased from Sigma‐Aldrich. All the chemicals for this study were analytical research grade.
Mxene (Ti3C2Tx) for this work was synthesized with a few modifications from a previous study. Typically, we added 1 g of Ti3AlC2 powders, pinch by pinch, to a 60 mL HF (35%) in a 100 mL polypropylene (PP) beaker, stirred continuously for 48 h, and centrifuged at approximately 8000 rpm for 10 min. The HF etching process was performed in a well‐ventilated fume hood using appropriate personal protective equipment including acid‐resistant gloves, face shield, and lab coat. HF waste was disposed following standard laboratory safety protocols. Then, the powder (Ti3C2Tx) underwent freeze‐drying for 24 h. The Ti3C2Tx product obtained through centrifugation was dispersed and subjected to ultrasonic treatment in DMSO to produce individual sheets of Ti3C2Tx that are not agglomerated. Finally, we used a freeze dryer to dry the Ti3C2Tx powders for 24 h [31]. Figure 1 depicts the exfoliation process of Mxene (Ti3C2Tx).

Schematics of the Ti3C2Tx synthesis: (a) Ti3AlC2 MAX phase; (b) exfoliated Ti3AlC2 MAX by HF; and (c) delaminated Ti3C2Tx MXene by DMSO.
The composite is produced by the in situ polymerization process, as previously reported [32]. Typically, 0.2 g of Ti3C2Tx powder was evenly mixed with a solution of 30 mL of 1 M HCl. After that, the mixture endured ultrasonic dispersion for 1 h in order to achieve a homogeneous solution. Subsequently, a volume of 100 μL of distilled aniline (ANI) was introduced into the previously indicated solution and subsequently dispersed with the application of ultrasonication for 1 hour. Afterward, ammonium persulfate solution (0.335 g APS in 30 mL of 1 M HCl) was prepared. Then, the solution was added to the Ti3C2Tx suspension Figure 2. The mixture was immersed in a container of ice and stirred vigorously at a temperature ranging from 5°C to 7°C for 6 h. Lastly, the Ti3C2Tx/PANI composite powder was obtained by freeze‐drying.

In situ polymerization of the Ti3C2Tx/PANI composite.
Mxene (Ti3C2Tx), Ti3C2Tx/PANI, and Cr(VI)@Ti3C2Tx/PANI were characterized by using XRD (Bruker D8 Advance x‐ray) with Cu Kα radiation (
The synthesized Ti3C2Tx/PANI composite was employed for the adsorption of Cr(VI) from simulated wastewater at room temperature. Adsorption experiments were conducted using the batch adsorption method in Erlenmeyer flasks. A 10 mM Cr(VI) stock solution was prepared in a 1000 mL flask. To investigate the effect of initial ion concentration, varying concentrations (10–50 ppm) were mixed with 50 mg of Ti3C2Tx/PANI and agitated in a shaking incubator at 200 rpm for 2 h. Similar experiments were performed for pH and adsorbent dosage optimization studies. After each experiment, the solutions were filtered using Whatman No. 1 filter paper. The adsorption capacity was determined by using equation (1). All experiments were conducted at room temperature
To comprehensively evaluate the adsorption mechanism and avoid bias toward a single theoretical framework, the experimental data were fitted to multiple isotherm and kinetic models. The fitting of the selected models was evaluated using
3. Results and Discussion
3.1. Material Characterization
The peaks shown in Figure 3 (a) for pristine Ti3C2Tx correspond to the crystallographic planes (002), (004), (006), (0010), (0012), and (110), with peak positions at 9.0, 18.3, 27.8, 35.9, 41.7, and 60.5, respectively. These peaks correspond to the distinctive features of Ti3C2Tx MXene, which aligns with previous research findings. There is an observed phenomenon that shows a wider peak and lower intensity [31]. The diffraction pattern of Ti3C2Tx/PANI is also depicted in Figure 3(b). The diffraction pattern of Ti3C2Tx/PANI exhibits a distinct weak peak at 25.2°, corresponding to the crystal faces (200) of PANI. This confirms the successful synthesis of the Ti3C2Tx/PANI composite, with aniline being polymerized onto the Ti3C2Tx surface. The outcome of our work aligns with the findings of the prior study [33]. The polymerization of aniline on Ti3C2Tx resulted in the disappearance of diffraction peaks at 9.0 and 18.3, which can be attributed to the amorphous nature of PANI, as depicted in Figure 3(b). The XRD diffraction pattern was also collected after the adsorption of Cr(VI), as shown in Figure 3(c). It divulges that the peak related to PANI is disappeared, which may be due to Cr(VI) adsorption.

XRD of (a) Ti3C2Tx, (b) Ti3C2Tx/PANI, and (c) Ti3C2Tx/PANI after Cr(VI) adsorption.
The FTIR spectra of Ti3C2Tx are shown in Figure 4(a). The peaks seen at 882 and 618 cm−1 are likely a result of the deformation vibration of the Ti–O–Ti linkage and Ti–O bonding, respectively [34]. The peak at 1102 cm−1 corresponds to the stretching vibration of the C–Cl bond. The spectra depicted in Figure 4(b) reveal that the peak observed at 1581 cm−1 corresponds to the C–C stretching of quinoid rings, whereas the peak at 1484 cm−1 corresponds to the C=C stretching of benzenoid rings in the Ti3C2Tx/PANI composite. The peak detected at 1147 cm−1 corresponds to the vibration mode of N = Q = N, which is an electronic band associated with the quinoid ring. This finding is consistent with previous reports [35]. The band observed at 1296 cm−1 represent the C−N stretching [36]. The FTIR spectra analysis performed after the adsorption of Cr(VI) (Figure 4(c)) shows some changes on the intensity and the position of peaks. The intensity of the benzoid groups of the Ti3C2Tx/PANI has increased after Cr(VI) adsorption, as PANI loses electron from the quinoid group to the Cr(VI) and reduces it to Cr(III) [37]. The prominent peak of Ti3C2Tx/PANI at 1147 cm−1 undergoes a shift to 1149 cm−1 with reduced intensity following the adsorption of Cr(VI). A new peak emerges at 1310 cm−1 upon adsorption, which was absent in the Ti3C2Tx/PANI composite. The observed alterations in the intensity and locations of peaks provide evidence that the presence of functional groups on our composite facilitates the formation of complexes with Cr(VI) [38].

FTIR spectra of (a) Ti3C2Tx, (b) Ti3C2Tx/PANI, and (c) Ti3C2Tx/PANI after Cr(VI) adsorption, where B and Q rings are benzenoid and quinoid rings.
The morphology and surface elemental composition of Ti3C2Tx/PANI and Cr(VI)@Ti3C2Tx/PANI are shown in Figure 5; the composite shows a porous structure [39]. The EDS analysis of the Ti3C2Tx/PANI composite revealed the presence of titanium (Ti), carbon (C), oxygen (O), fluorine (F), and nitrogen (N) in the material, confirming the successful incorporation of PANI onto the Ti3C2Tx surface. Specifically, the detection of nitrogen (N) is a clear indicator that PANI, which contains nitrogen in its structure, is chemically bound to the Ti3C2Tx surface, as shown in Figure 4(a). The presence of these elements is consistent with the formation of the Ti3C2Tx/PANI composite, with the fluorine (F) and oxygen (O) likely originating from the surface functional groups on Ti3C2Tx and carbon (C) from both Ti3C2Tx and PANI. After the adsorption of Cr(VI), the EDS spectra of the Ti3C2Tx/PANI composite (Figure 5(b)) revealed the appearance of chromium alongside the previously detected elements, demonstrating that Cr(VI) was successfully adsorbed onto the composite. The incorporation of Cr was observed in the form of Cr(VI), further confirming that the Ti3C2Tx/PANI composite material actively engages in the adsorption process and binds Cr ions onto its surface.

SEM–EDS of (a) Ti3C2Tx/PANI and (b) Cr(VI)@Ti3C2Tx/PANI.
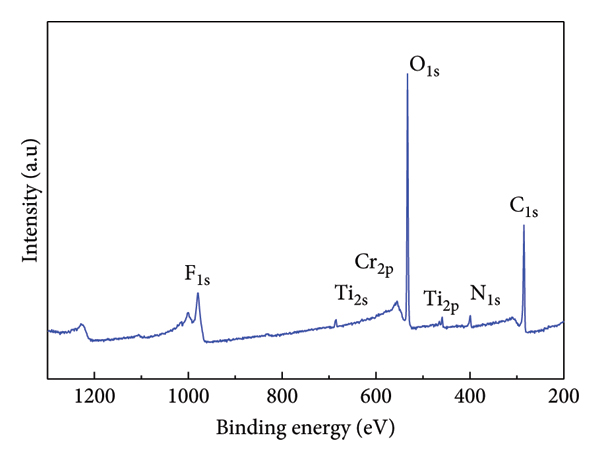
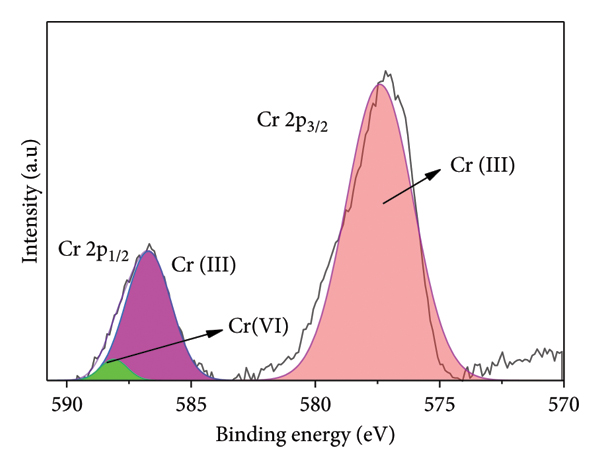
Additionally, analyzing the XPS spectrum allows for the identification and characterization of different species and their corresponding chemical states of Ti3C2Tx, Ti3C2Tx/PANI, and also after Cr adsorption. Figure 6 depicts the XPS scan spectrum for Ti3C2Tx, Ti3C2Tx/PANI, and after Cr(VI) adsorption, which shows that these materials are made from (Ti, C, O, and F), (Ti, C, O, F, and N), and (Ti, C, O, F, N, and Cr), respectively. The XPS of Ti 2p, Ti 2p1/2, and Ti 2s1/2 in Figure 6(b) can be fitted with binding energies at 460.13, 514.13, and 561.49 eV, respectively. The F 1s p3/2 peak centers at 686.03 eV, while the peaks at 286.88 and 533.05 correspond to C 1s and O, respectively. The peak centered at 401.13 eV, which corresponds to N 1s (Figure 6(b)), appeared after in situ polymerization with PANI, which indicates that Ti3C2Tx/PANI is formed successfully. The XPS survey scan of Cr@Ti3C2Tx/PANI is given in Figure 6(c), and the peak found at 580 eV is due to Cr (VI) [40]. We also used high‐resolution XPS to identify the oxidation number of Cr species present in Ti3C2Tx/PANI. An optimal match was achieved by decomposing the Cr 2p spectra into three distinct peaks by deconvolution in Figure 6(d). Cr (III) 2 p3/2 and 2 p1/2 are responsible for the peaks observed at 577.5 and 586.7 eV, respectively. On the other hand, the peaks at 588.2 eV are characteristic of Cr(VI) 2 p1/2 [41, 42]. The extent of Cr(VI) to Cr(III) reduction was not quantitively determined in this study.

XPS of (a) Ti3C2Tx, (b) Ti3C2Tx/PANI, (c) Cr@Ti3C2Tx/PANI, and (d) deconvoluted Cr.
(b)

(c)
(d)
From the Raman spectrum, the peaks found at 206 and 711 cm−1 in Figure 7(a) correspond to the A1g mode, which represents the out‐of‐plane vibrations of Ti and C, respectively. The peaks observed at 279, 409, and 616 cm−1 are attributed to Eg in the plane (shear) vibration of Ti, C, and terminal group atoms, respectively [43]. The peaks observed at 1162 and 1192 cm−1 correspond to the C–H stretching vibration of the B ring; 1492 cm−1 is due to the C–N stretching of the Q ring; and 1593 cm−1 corresponds to the C–C stretching of the B ring in Ti3C2Tx/PANI, which indicates that PANI has undergone polymerization on the surface of Ti3C2Tx [44]. A peak is around 580 cm−1, confirming the adsorption of Cr(IV) by the nanocomposite (Figure 7(b)). Additionally, the adsorption of Cr(VI) resulted in a reduction in intensity and a displacement in the peak position, as shown in Figure 7(b). Ti3C2Tx/PANI after adsorption exhibited a small peak about 911 cm−1 as shown in Figure 7(b), which is the distinctive peak associated with Cr(VI) [45, 46]. “B” is benzenoid, and “Q” is quinoid ring.

Raman spectra of (a) Ti3C2Tx/PANI and (b) Ti3C2Tx/PANI after Cr(VI) adsorption.
3.2. Optimization Study
pH significantly influences the adsorption process [47]. In the case of Cr(VI), different anionic forms exist, including Cr2O7 2−, HCr2O7 −, CrO4 2−, etc. Their form depends on their pH in aqueous solutions [48]. To see how pH changes the adsorption process, we did several studies with the same temperature, initial ion concentration (10 mg/L), and Ti3C2Tx/PANI mixture dosage (10 mg/L). We found a hexavalent chromium ion in the form of H2CrO4 at pH values less than 2 [49]. As pH increases to 2–9, it is predominantly detected in the form of Cr2O7 2− and HCrO4 −. Beyond the pH of 7, chromium (VI) transforms into the CrO4 2− form [50]. We determined point of zero charge (PZc) to further understand the behavior of the Ti3C2Tx/PANI composite, and found it to be 6.46, as shown in Figure 8(a). These findings support the notion that these composite surfaces exhibit positive charges at pH values below pHPZc. The existence of Cr(VI) in different forms, like HCrO4 − and Cr2O7 2−, has its own impact on their adsorbed capacity. Notably, a decrease in adsorption efficiency was seen at pH > 2 due to the existence of HCrO4 −. The strong competition between H2CrO4 and the proton for an adsorption site is the primary cause of this decrease. Acidic solutions also result in functional groups being protonated, which leads to strong electrostatic interaction between the Ti3C2Tx/PANI nanocomposite and Cr2O7 2− ions. Conversely, at higher pH levels (between 4.0 and 9.0), the dominant form of chromium is CrO4 [2–51]. At these levels, an excessive presence of the hydroxide ion (OH−) competes with the CrO4 2− ion for adsorption. It is found that the adsorption efficiency decreases as the pH increases [52]. The maximum removal efficiency obtained at pH = 2, but it significantly decreased as pH increased Figure 8(b).

Optimization studies of (a) pHPZc, (b) pH, (c) dosage, and (d) initial concentration.
(b)

(c)

(d)

Additionally, we conducted a study to evaluate the impact of the Ti3C2Tx/PANI dosage on the removal of hexavalent chromium ions from wastewater, using adsorbent dosages ranging from 10 to 50 mg/L as depicted in Figure 8(c). An increase in the dose of Ti3C2Tx/PANI resulted in an increase in the removal efficiency of chromium adsorption. Elevated composite concentrations result in higher doses and an increase in the total surface area [53]. The increased surface area of Ti3C2Tx/PANI increases the removal efficiency. Ti3C2Tx/PANI has adsorption sites that promote the surface binding of the Cr(VI) ion. At the same time, adsorption capacity decreased because of the presence of excessive adsorption sites, which are left unsaturated.
Effect of the initial ion concentration was studied at pH = 2 for a time of 480 min and a dosage of 10 mg/L at ambient temperature. Figure 8(d) displays the findings of an empirical study examining the impact of varying concentrations of initial ions (10–50 mg/L). As the starting concentration of Cr(VI) went up, the removal efficiency increased from 66.6 mg g−1 to 228.56 mg/L. The adsorption capacity reached its maximum before the concentration of Cr(VI) exceeded 50 mg/L. At the same time, the percentage removal decreased due to surface saturation [54]. Due to higher concentration of Cr(VI), and saturated adsorbents, the removal efficiency decreased [55].
3.3. Kinetic Studies
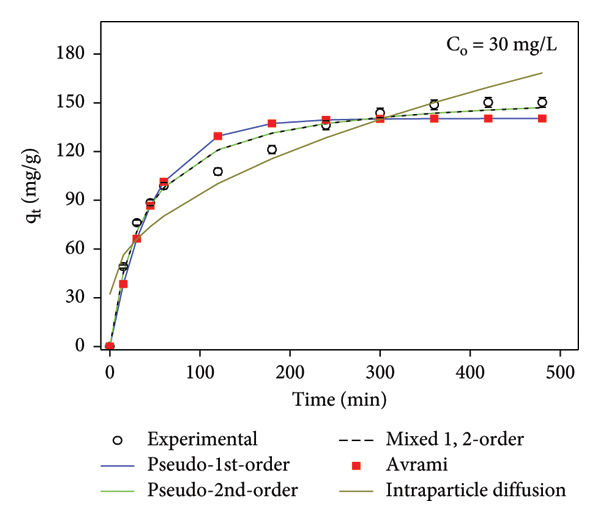
To investigate the adsorption kinetics, PFO, PSO, MFSO, IPD, and Avrami models were studied as shown in Figure 9 for hexavalent chromium adsorption at five initial concentrations over time at pH = 2 and 0.15 g/L of dosage. As the initial concentrations of hexavalent chromium increased, the maximum adsorption capacity also increased. The PSO and MESO models are found to be better to describe the Cr(VI)@Ti3C2Tx/PANI system. For PSO, the experimental values and correlation coefficients (

Fitting of the experimental data with kinetic model results of Cr(VI) adsorption onto Ti3C2Tx/PANI at initial concentrations (a) 10, (b) 20, (c) 30, (d) 40, and (e) 50 mg/L.
(b)

(c)
(d)

(e)

Variables used in the kinetic models by using the Ti3C2Tx/PANI nanocomposite.
3.4. Modeling of Adsorption Isotherms
The isotherms of adsorption are essential for studying the process and affinity of Ti3C2Tx/PANI toward hexavalent chromium ions. Nine models were used to accurately match the experimental results of the Ti3C2Tx/PANI nanocomposite’s adsorption of the hexavalent chromium ion at a temperature of 298 K. These results are represented as shown in Figure 10 and Table 2.

Different isotherm models for Cr(VI) adsorption by Ti3C2Tx/PANI at a temperature of 298 K.
The variables in the adsorption isotherm model for Cr(VI) by Ti3C2Tx/PANI.
The variables in the equation are as follows: qe represents the adsorption capacity at equilibrium (mg g−1). Ce stands for the concentration of adsorbate (Cr(VI)), and qmax is the maximum adsorption capacity (mg g−1). KL (L mg−1) is the Langmuir isotherm. KLF is the equilibrium constant for heterogeneous solids. KF (L mg−1) is the Freundlich isotherm coefficient, and n is a constant. There are several numbers that describe different types of things in a system: m1, m2, K1, and K2; ks is the Sips isotherm constant; ns is the Sips isotherm model exponent (also called the heterogeneity factor); KR and aR are the Redlich–Peterson constants; βR is an exponent that ranges from 0 to 1; KT (mg g−1) is a constant; aT (mg/L) is the Toth constant; and z is the degree of heterogeneity of the adsorption systems.
The tendency of Ti3C2Tx/PANI to bind hexavalent chromium depends a lot on how much of it is present at the start (Figures 8(a), 8(b), 8(c), 8(d)). The Ti3C2Tx/PANI nanocomposite can adsorb 66.6, 109.7, 150.3, 190.9, and 228.6 mg g−1 of Cr(VI) ions when the levels of Cr(VI) ions are 10, 20, 30, 40, and 50 mg/L, respectively. With increasing concentrations, the removal efficiencies decrease by 99.9%, 82.27%, 75.14%, 71.59%, and 68.57%, respectively. The fitted nonlinear Freundlich and Redlich–Peterson models have correlation coefficients (
4. Comparison Studies
The data in Table 3 show the adsorption capacity of the Ti3C2Tx/PANI composite adsorbent (this work) compared to other adsorbents from the literature, using Ti3C2Tx‐ and PANI‐based composites for Cr(VI). The capacity for adsorption of the adsorbents is determined by various elements, such as pH, time, temperature, dosage, initial ion concentration, etc.
Comparative analysis of the adsorption capacity for chromium (VI) by PANI‐ and Ti3C2Tx‐based composites based.
5. Reusability Study
The reusability of an adsorbent is a key factor in achieving a cost‐effective and sustainable adsorption process. To evaluate the recycling potential of the Ti3C2Tx/PANI composite, sequential batch adsorption–desorption experiments were performed. In each cycle, 10 mg of the Ti3C2Tx/PANI composite was mixed with a 10 mg L−1 of hexavalent chromium solution and stirred for 8 h. The composite was reused for seven consecutive cycles, as illustrated in Figure 11. The first cycle exhibited the good recovery efficiency (88.94%) in an acidic medium, indicating the composite’s excellent capability to adsorb and remove Cr(VI) from wastewater. The adsorbent stability tends to decrease with increasing contact time or repeated use due to the physical or chemical deterioration of the adsorbent surface or structure [65].
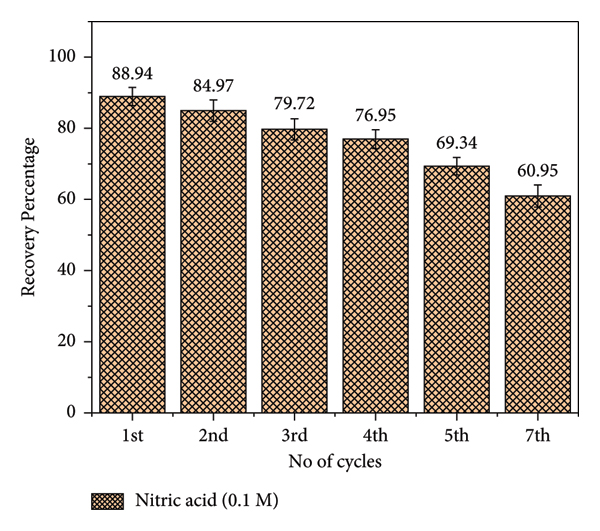
Recyclability studies.
6. Adsorption Mechanism
The FTIR, EDS, and XPS spectra confirmed that Cr species is adsorbed onto the surface of the Ti3C2Tx/PANI composite. The prominent N = Q = N peak at 1147 cm−1 of FTIR as depicted in Figure 4 indicates the presence of the quinoid ring on the Ti3C2Tx/PANI surface. After Cr(VI) uptake, the FTIR peak’s intensity red‐shifted to 1149 cm−1, indicating an interaction between the composite and Cr(VI) [22]. In addition, the EDS analysis of the Ti3C2Tx/PANI composite after adsorption showed the presence of Cr (Figure 5(b)). Besides, the XPS spectrum of Cr 2p of the Ti3C2Tx/PANI after Cr(VI) sorption clearly revealed the presence of Cr(VI) at 580 eV and the optimal match was achieved by decomposing the Cr 2p spectra into two distinct peaks, with Cr (III) 2p3/2 and 2 p1/2 responsible for 577.5 and 586.7 eV, respectively (Figure 6(d)). Based on FTIR, EDS, and XPS results, the Cr(VI) adsorption process by the Ti3C2Tx/PANI composite takes place by (i) electrostatic force between the negatively charged Cr(VI) and positively charged H+ or ‐NH2 species, generated from acid solutions and PANI oxidation, (ii) surface complexation/chelation of Cr(VI) and ‐OH, =O, and N–H functional groups, and (iii) physical adsorption of Cr(VI) on the composite surface [58]. The adsorption mechanism is shown in Figure 12.
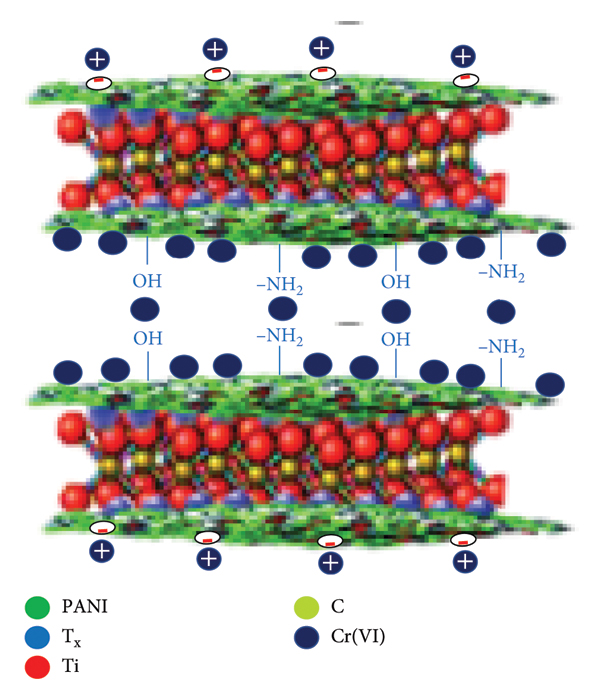
Adsorption mechanism of Cr (VI) by the Ti3C2Tx/PANI composite.
7. Conclusion
Using an in situ polymerization method, a Ti3C2Tx/PANI nanocomposite was made so that it could effectively remove Cr(VI) from simulated wastewater. The Ti3C2Tx/PANI composite was thoroughly studied using SEM–EDX, XRD, FTIR, Raman spectroscopy, and XPS methods. The material exhibited a significant Cr(VI) removal capability, influenced by parameters such as adsorbent dosage, initial Cr(VI) concentration, and pH. Notably, the interaction time and dosage required in this study were considerably lower than those reported previously for Cr(VI) removal. Under typical environmental conditions, the maximum adsorption capacity reached 342.5 mg g−1 at 2 pH.
The composite also showed excellent regeneration and reuse potential, making it a strong candidate for practical Cr(VI) remediation in aqueous systems. The Ti3C2Tx/PANI composite achieves efficient Cr(VI) removal through a combination of electrostatic attraction, surface complexation, and physical adsorption mechanisms. Overall, the higher adsorption capacity and reusability of the Ti3C2Tx/PANI composite demonstrate its novelty and efficiency compared to previously reported Cr(VI) adsorbents using Ti3C2Tx and PANI, mainly due to the synergistic interaction between the Ti3C2Tx layers and the conductive polymer PANI. Furthermore, the solution‐based, low‐temperature synthesis process makes the material amenable to scale‐up, and its rapid adsorption kinetics support potential integration into fixed‐bed adsorption columns, packed‐bed reactors, or point‐of‐use treatment cartridges. The material can be regenerated using mild acidic solutions, enabling multiple adsorption–desorption cycles with minimal performance loss. These features position the Ti3C2Tx/PANI composite as a practical and scalable adsorbent for Cr(VI) removal in real wastewater management systems.
Data Availability Statement
Data are available on request from the authors.
Conflicts of Interest
The authors declare no conflicts of interest.
Author Contributions
Hailemariam Assefa: conceptualization, methodology, data curation, and writing–original draft.
Sathiesh Kumar Subramaniam: visualization, investigation, supervision, editing, and revision of manuscript.
Funding
The author(s) received no specific funding for this work.
