Abstract
The adsorptive removal of methyl orange dye using a sugar beet bagasse (SBB) as agro-industrial by-product has been investigated. The adsorption potential of SBB was improved by grafting with FeCl3·6H2O. The dependency of removal efficiency (
Introduction
Colored sewage is as a consequence of dye usage in the several industries including dye manufacturing, textile industry, pulp and paper processing, leather tanning, battery production, and so on (Politi and Sidiras, 2012). Many dyes and their breakdown products have wide adverse effects for living organisms. Moreover, the colored wastewater reduces light penetration and then affects the photosynthetic activity of aquatic organisms (Yang et al., 2015). Azo dyes are an interesting class of compounds that are widely applied in chemical applications and industries. Although azo dyes are not strongly toxic, they can cause hypersensitivity and allergy; therefore, the presence of these dyes in water is highly undesirable (Deligeer et al., 2011; Ling et al., 2016). Hence, disposal of untreated effluents to the surrounding environment often leads problems for aquatic life and humans. To prevent any potential hazards, an effective treatment of these industrial wastewaters is needed before being discharged into the environment. Furthermore, removal of the azo dyes from the effluent is difficult because they are not easily degradable or removable by conventional wastewater treatments (Haris et al., 2010). Some methods like membrane separation (Saffaj et al., 2004), ion exchange (Wu et al., 2008), photocatalytic degradation (Lv et al., 2011), anodic oxidation (Tsantaki et al., 2012), ozone treatment (Zhang et al., 2014), biological treatment (Işık and Sponza, 2006), and adsorption (Ahmad and Hameed, 2010; Jiang et al., 2014; Liu and Zhang, 2007) have been widely applied in the process of removing dyes from aqueous solution. Among various treatment methods, adsorption is quite a promising technique for the removal of dyes due to its high efficiency, ease of handling, availability of different adsorbents, and its cost efficiency. In this regard, various adsorbents have been developed for the separation and removal of different dyes including activated carbons (Ahmad and Hameed, 2010; Oliveira et al., 2011), magnetic nanocomposites (Deligeer et al., 2011; Jiang et al., 2014; You et al., 2014), zeolites (Akgül, 2014; Hernández-Montoya et al., 2013), mesoporous materials (Lee et al., 2007; Punyapalakul and Takizawa, 2006), clays (Liu and Zhang, 2007), natural or agro-industrial by-products (Al-Ghouti et al., 2010; Haris et al., 2010; Saad et al., 2010; Subbaiah and Kim, 2016; Yu et al., 2012), and so on. In recent years, agricultural or agro-industry by-products are widely used as efficient adsorbents for improving the contaminated water and wastewaters. Sugar beet bagasse (SBB) is one of the primary agro-industrial by-products that are produced in large quantities in the world. Therefore, it can be a good candidate for developing cost-efficient adsorbents for removal of azo dyes. Response surface methodology (RSM) is a set of statistical and mathematical technique for designing experiments, building models, evaluating the relative significance of several independent variables, and determining optimum conditions for desired responses (Wantala et al., 2012). In literature, RSM based on central composite design (CCD) has been used in different applications of engineering including heavy-metal removal, dye removal, biofuel production, etc. (Auta and Hameed, 2011; Betiku and Taiwo, 2015; Dora et al., 2013; Skorupskaite et al., 2015). However, this is the first study on the optimization of methyl orange (MO) adsorption on SBB. Therefore, we decided to evaluate the MO removal potential of SBB under various operating conditions such as pH, initial MO concentrations, and sorbent dosage. The parameters for maximum removal efficiency are optimized by using CCD with RSM under (Design Expert. 7.0) software. The physicochemical characteristics of SBB are evaluated by Fourier transform infrared spectroscopy (FT-IR) and scanning electron microscopy (SEM). Moreover, Langmuir and Freundlich isotherms were applied to the equilibrium data to describe the main interactive mechanisms involved the removal process. Kinetic parameters were also calculated from the pseudo first-order and pseudo second-order models. Furthermore, thermodynamic parameters (enthalpy, entropy, and adsorption free energy), were calculated for better description of the adsorption mechanism.
Materials and methods
Chemicals
Analytical grade of hydrochloric acid (37%), sodium hydroxide (NaOH), and FeCl36H2O were all purchased from Aldrich and used without further purification. Deionized distilled water was used in the preparation of all solutions. MO that also known as C.I.ACID ORANGE 52 was purchased from Merck (Germany). The chemical formula for MO is C14H14N3NaO3S with molecular weights of 327.34 g.mol−1. MO molecular structure (single azo class) is shown in Scheme 1. All other reagents were of analytical grade and all glassware were cleaned by HNO3 and rinsed with double distilled water.
Molecular structures of methyl orange (MO).
Sorbent preparation
SBB was collected from local sugar factory of Kermanshah, West of Iran. First, SBB pulverized and then washed with tap water to remove dirt and impurities and dried in the oven at 70 ℃ for 12 h. Five grams of FeCl3·6H2O were dissolved in 100 mL deionized water and 10 g of dry SBB was added and stirred for 4 h and kept overnight. Produced Fe-SBB was then separated by filtration and dried in an oven. Fe-SBB residue was heated in a muffle furnace at 450 ℃ for 6 h, than washed and dried. Finally, the primary SBB and Fe-SBB were tested and compared for MO removal.
Physicochemical characterization of adsorbent
FT-IR analysis was carried out on Bruker Vector 22 FT-IR Spectrometer in the range of 400–4000 cm−1 employing the KBr pellet method. The structure and morphology of SBB and Fe-SBB particles were examined by SEM (MIRA3TESCAN, Czech Republic).
MO adsorption studies
Batch adsorption experiments were performed at room temperature (20 ℃) to study the effect of solution pH, initial dye concentration, and sorbent dosage. Each experiment was carried out in Erlenmeyer flasks containing 100 mL MO solution by shaking the flasks at 200 r/min for period contact time of 120 min. Samples were withdrawn at predetermined time intervals (0, 5, 15, 30, 60, 90, and 120 min) and filtered through 0.45-µm filters. The residual MO concentration in the supernatant was determined using the spectrophotometer (UVIKON) at 475 nm. To evaluate the effect of temperature on the dye adsorption, optimal process parameters (pH, sorbent dosage, and MO concentration) which were determined by the software were applied at four temperatures (20, 30, 40, and 50 ℃). The removal efficiency of the MO molecules was calculated by equation (1):

MO adsorption capacity by SBB was determined according to equation (2):

Experimentation and optimization of adsorption process

Independent variables and their coded levels for the CCD.

CCD: central composite design; MO: methyl orange.
Experimental design in term of coded factors and results of the central composite design.

Results and discussion
Characterization of SBB
The morphology of the sorbent was studied by SEM. Figure 1 shows the SEM images of raw SBB, Fe-SBB and Fe-SBB after MO adsorption. It can be observed that the surface morphology of raw SBB was different from that of Fe-SBB and Fe-SBB after MO adsorption. Figure 1(a) shows the structure of raw SBB at a magnification of 59,500×. It is clear that SBB particles was an assemblage of fine particles, which did not have regular and fixed shape and size and exhibit fiber-like structures with low porosity. Figure 1(b) shows that the surface layer of Fe-SBB exhibit microstructure porosity by treatment with FeCl3·6H2O. Microporous active sites distinguished on the surface layer of Fe-SBB may increase the specific surface area and lead to faster action of adsorption. Johar et al. (2012) indicated that under the chemical treatment conditions almost all the components that bind the fibril structure of the biomass were removed thus enabling the fibers to be separate into an individual forms. Moreover, Figure 1(c) revealed that the smooth surface of Fe-SBB became thicker and coarser after MO adsorption, which providing further evidence that MO was loaded on the surface of the sorbent.
SEM images of (a) raw SBB, (b) Fe-SBB, and (c) Fe-SBB after MO adsorption.
FT-IR spectroscopy was used to detect the presence of binding groups in the SBB and Fe-SBB (Figure 2). The absorption band for H–O–H bending vibration in water is at around 1640 cm−1(An et al., 2010). The spectra show a broad band around 3000–3700 cm−1, which is due to adsorbed water molecules and also O–H stretching mode (Kalapathy et al., 2000). In the SBB spectra, the absorbance peak appears in 2920 cm−1 and was assigned to the C–H bond of biomass carbon structure. Moreover, the adsorption peaks aroused by other functional groups such as COOH (1740 cm−l), COC (1050 cm−1), and COH (1249 cm−1) can be observed in Figure 2 (Jiang et al., 2015). Based on the obtained results from the XRF analysis (not reported here), there are some metal oxides in the SBB structure, including SiO2 (1.3%) and Al2O3 (0.3%). Hence, the vibration signals around 462 and 1377 cm−1 present in SBB sample are typical Si–O–Si bands attributed to the bending and Al–O, respectively (Ghorbani et al., 2013). It is valuable to note that an absorption peak appears at 540 cm−1 after Fe species being grafted in SBB, but pure SBB shows no absorption at this region, indicating that the heteroatoms has been incorporated into the framework of sorbent and the Fe–O bonds have formed (Song et al., 2016).
FT-IR spectra of SBB and Fe-SBB.
Comparative MO adsorption study by SBB and Fe-SBB
As preliminary experiments, the MO removal capacities of the tow different produced sorbents (SBB and Fe-SBB) were compared. The results revealed that the 9:1 Fe-SBB showed the highest dye removal potential. Therefore, this sorbent was used in all the subsequent designed experiments. The dye removal kinetics of Fe-SBB was compared with those of SBB at 1 g.L−1 sorbent dosages, pH 5, and 100 mg.L−1 MO concentration. The results are presented in Figure 3.
Comparison of MO adsorption by SBB and Fe-SBB (sorbent dosage: 1g.L−1, pH: 5, and MO concentration: 100 mg.L−1).
It can be observed that the Fe-SBB sorbent has faster dye-removal kinetics and larger dye removal capacity when compared with SBB at the indicated concentration. This is attributed to the distribution of iron functional groups on the surface of SBB, thus making them kinetically more accessible for MO molecules, and increasing their binding sites available for adsorption. The adsorption can be defined as the attraction of Fe3+ ions to the adsorbent surface by chemical forces in addition to electrostatic forces (Hunter, 1988). Also, the formation of additional hydroxyl groups (Fe–OH) is another factor for increasing adsorption capacity.
Statistical analysis and the model fitting
RSM is more advantageous than the traditional single-parameter optimization in that it saves time, space, and raw material (Sadhukhan et al., 2016; Wu et al., 2014). There were a total of 20 runs for optimization of three individual parameters in the current CCD. By applying multiple regression analysis on the experimental data, the 
Also, the 
Regression analysis using response surface quadratic model for removal efficiency (
Regression analysis using response surface 2FI model for MO adsorption capacity (
2FI: two-factor interaction; MO: methyl orange.
Data given in Table 4 demonstrate that the regression of
Figure 4(a) and (b) shows the relationship between actual and the predicted values of removal efficiency ( Correlation of actual and predicted values for (a) removal efficiency (
Effect of process variables
In order to examine the interactions between the factors and responses, three-dimensional (3D) response surface plots are very helpful showing a function of two factors maintaining the other factor at a fixed level (Simsek et al., 2013). The influences of the three different process variables including initial solution pH, initial MO concentration, and sorbent dosage on removal efficiency ( 3D surface plots for MO removal efficiency ( 3D surface plots for MO adsorption capacity at 120 min showing the mutual effect of (a) pH and MO concentration, (b) pH and sorbent dosage, and (c) sorbent dosage and MO concentration. MO: methyl orange.

Effect of pH
According to the literature adsorption of MO usually is dependent on the pH of the solution (Mittal et al., 2007). Figure 5(a) and (b) show the interaction of pH with initial MO concentration and sorbent dosage on removal efficiency (
Effect of initial MO concentration
The mutual effect of initial MO concentration with pH and sorbent dosage was visualized in Figure 5(a) and (c). These diagrams pointed out that the removal efficiency increased with increase in initial MO concentration from 5 to 70 mg.L−1 and then relatively stable or slightly increased from 70 to 100 mg.L−1. It can be supposed that a certain amount of sorbent own limited number of active adsorption sites and a corresponding saturated adsorption capacity. When the sorbent reach saturated state, all active adsorption sites are occupied, so the sorbent could not adsorb much more dye molecules. A similar result was observed for the adsorption of acid orange by a mesoporous activated carbon (Noorimotlagh et al., 2014). Through the comparison of the results of MO adsorption capacity (
Effect of sorbent dosage
The mutual effect of sorbent dosage with pH and initial MO concentration based on the fitted quadratic models is presented in Figure 5(b) and (c). It can be observed that removal efficiency (
Optimization and model validation
One of the main aims of this study is to find the optimum process parameters to maximize the removal efficiency ( Desirability ramp for numerical optimization of five goals, namely the initial solution pH, initial MO concentration, sorbent dosage, MO adsorption capacity, and removal efficiency.
MO adsorption capacities of various adsorbents.

MO: methyl orange.
Kinetics, adsorption isotherms, and thermodynamic studies

Based on equilibrium adsorption, the pseudo-second-order kinetic equation is expressed as equation (8):

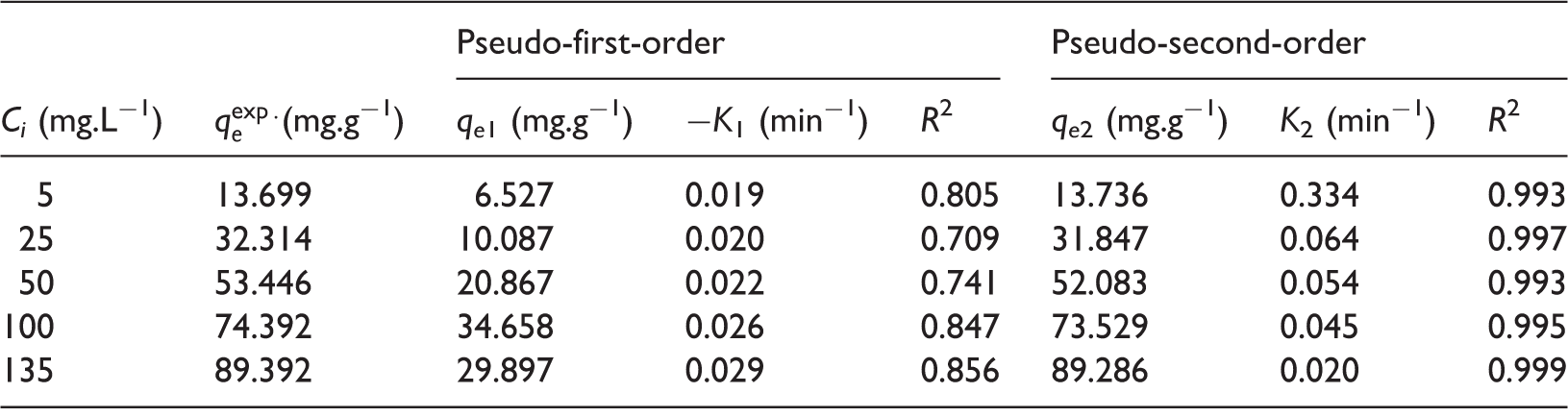
Kinetic parameters for adsorption rate expressions.
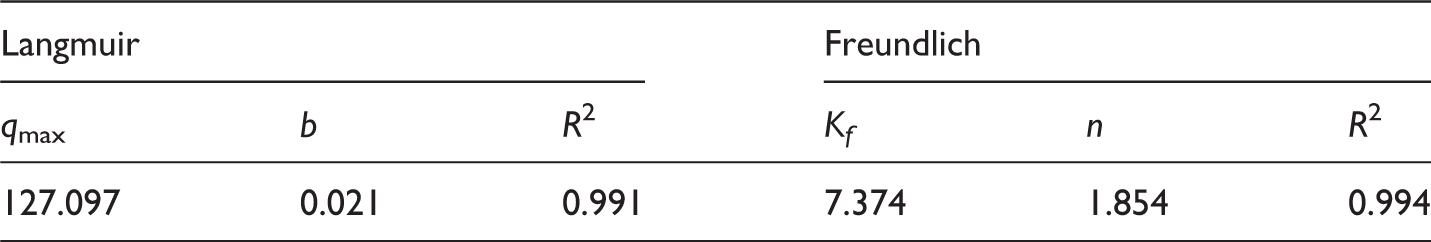
Langmuir and Freundlich isotherms for adsorption of MO on Fe-SBB with a sorbent dosage of 0.37 g.L−1 and pH level of 2.51.


Isotherm parameters for the adsorption of MO on Fe-SBB.



Thermodynamic parameters of MO adsorption on Fe-SBB at different temperatures in Kelvin (with initial MO concentration of 100 mg.L−1, sorbent dosage of 0.37 g.L−1, and pH level of 2.51).

Conclusion
The Fe-SBB prepared by pretreatment of SBB (as an agro-industry by-product) by FeCl3·6H2O was found to be an efficient adsorbent for the removal of MO molecules from aqueous solutions. FT-IR and SEM analysis were used for characterization of the adsorbent. These analyses showed that SBB particles exhibit non-spherical and fiber-like structures with low porosity. On the other hand, when the SBB treated by the FeCl3, iron ions were reacted by the surface of the sorbent and microporous active sites distinguished on the surface layer of Fe-SBB. The RSM was utilized to create optimum process condition for MO molecules adsorption. The operating parameters, initial pH of the solution, initial dye concentration, and sorbent dosage are recognized effective on the removal efficiency (
Footnotes
Acknowledgements
The authors wish to thank Mr Hoshyar Gavilian for his assistance (lab technicians of environment laboratory) at environmental science departments.
Declaration of Conflicting Interests
The author(s) declared no potential conflict of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
