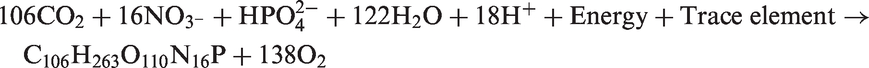Abstract
This work provides a simple and convenient method to manufacture the sorbent of Al-eggshell. The influence of AlCl3 concentration and pH values as well as the dosage of sorbent and their interactions on adsorption of phosphate was investigated. Therefore, Box–Behnken design coupled with response surface method was adopted to explore the empirical model for phosphate species removal. It was observed that there is an optimal point, C(AlCl3)(0.29 mol/l)–pH(6.12)–dosage(6.72 g/l), for the goal of maximizing phosphate species removal. The second-order polynomial model for phosphate reduction was given as Removal(%) = 96.43 +10.82X1 + 4.29X2 − 0.70X3 + 2.06X1X2 − 1.72X1X3 +8.24X2X3 − 13.10X12 − 17.26X22 − 1.72X32. Contour pictures implied that the interaction between pH values and sorbent dosage was the strongest, followed by C(AlCl3) versus dosage and C(AlCl3) versus pH. The adsorption of phosphate data had a good agreement with the Freundlich isotherm equation at 313 and 323 K. Otherwise, Langmuir–Freundlich model described the best fitness at the temperature of 293, 298, and 303 K. The process of adsorption of phosphate on Al-eggshell fitted a pseudo-second-order kinetic equation, which indicates the exothermic reaction. In conclusion, the present work suggests Al-eggshell as an efficient and environmental friendly sorbent for phosphate species adsorption from aqueous solutions.
Introduction
Phosphorus is a key limiting nutrient for the growth of aquatic plants and algae in water bodies, which can be confirmed by the following reaction
Previous research suggests that calcite, aluminum hydroxides/oxides, and iron hydroxides/oxides are significant phosphate sorbents (Karageorgiou et al., 2007; Lai et al., 2016). Waste eggshells—generally from residences, restaurants, and bakeries—consist primarily of calcium carbonate (Francis and Abdel Rahman, 2016; Guo et al., 2017) and are considered as a waste disposal and landfill problem. In recent years, different studies have explored useful applications for eggshells (Elabbas et al., 2016; Francis and Abdel Rahman, 2016). Those studies concluded that eggshell may be useful as a fertilizer and a feed additive for livestock. Eggshells have an estimated 7000–17,000 pores (William and Owen, 1995). The porous nature of eggshells makes them an attractive material to employ as an adsorbent. Eggshell appears to effectively adsorb certain heavy metals and organic compounds (Elabbas et al., 2016; Park et al., 2007); however, the application of discarded eggshells in the removal of phosphate by adsorption has received very little focus. Especially the use of AlCl3 doping waste eggshell was seldom studied.
In this work, we reuse waste eggshell as a sorbent of phosphate and simultaneously reusing a conventional waste. The prepared eggshell was cationized by applying AlCl3 to activate its phosphate sorption capacity. The eggshell powder before and after modification was characterized using N2-BET, X-ray diffraction (XRD), and morphological characterization (scanning electron microscope (SEM)) analyzer. The model of Design Expert was used to determine the optimum conditions for phosphate removal from polluted aqueous solutions. The effects of three key parameters in adsorption of phosphate including AlCl3 concentration, pH values, and sorbent dosage and their interactions on light phosphate removal were investigated using the response surface method. For this purpose, a Box–Behnken design was used to obtain statistical model for phosphate species removal percentage. Adoption equilibrium and kinetics isotherms experiments were performed to assess the behaviors and mechanisms of sorption.
Materials and methods
Materials
Eggshell samples were collected from the canteen of Shandong University of Science and Technology, Qingdao, China. To remove impurity and pollutants, the particles were rinsed with deionized water. Then the samples were dried at 100℃ overnight in the dry oven.
For the preparation of the Al-eggshell, 2.0 g eggshell powder was immersed in 100 ml solution of AlCl3 (0.29 mol/l) for 24 h with stirring speed 150 r/min. The mixture was agitated on a shake table at 30℃ at equilibrium. Then, the particles were centrifuged and dried, screened through a sieve of 100 mesh, and subsequently used for sorption experiments.
Stock solutions were prepared by dissolving the anhydrous potassium dihydrogen phosphate (KH2PO4) in distilled water to give a concentration of 1000 mg PO43−/l and diluted when necessary.
Design of experiments using Box–Behnken
In order to obtain the largest amount of information from a small number of experiments, the Box–Behnken method was used (Ahmad et al., 2016; Moghddam et al., 2016). Three key parameters including AlCl3 concentration, pH, and dosage were measured to generate a second-order polynomial model. The objective is selected to be increasing percentage of phosphate removal.
Predictor variables and their coded levels and actual values applied for experimental design.

Characterization
The chemical components of sorbents were estimated by energy dispersive X-ray spectroscopy (EDX, JB-750). The particle morphology of the eggshell and the Al-eggshell was characterized by powder XRD, which was obtained on a Shimadzu XRD-6000 diffractometer, using Cu Kα radiation at 40 kV, 30 mA, a scanning rate of 10°/min, a step size of 0.02°/s, and a 2θ angle ranking from 20° to 70°. The surface and matrix morphologies of the natural eggshell and the Al-eggshell were obtained on a KYKY-2800B SEM. The specific surface area of the eggshell samples was measured by the BET nitrogen gas sorption method. The evaluation of the porosity of natural waste eggshell and Al-eggshell sample was carried out using physical adsorption of N2 at the temperature of −196℃ (Quantachrome, Autosorb-1 C).
Adsorption procedure
Equilibrium studies
Adsorption equilibrium studies were conducted at initial phosphate solution pH of 6.12. Equilibrium data were obtained by adding 0.672 g of prepared Al-eggshell into a series of Erlenmeyer flasks each filled with 100 ml of phosphate solution. The initial phosphate concentration ranges from 0.5 to 200 mg/l. The Erlenmeyer flasks were covered with aluminum foil and then placed in a thermostatic shaker for specified time. During the adsorption, the temperature of the system was kept at 293, 298, 303, 313, and 323 K, respectively.
Kinetic studies
Adsorption experiments were carried out by adding a fixed amount of sorbent (0.672 g) to a series of conical flasks filled with 100 ml diluted solutions (3 and 50 mg/l). The conical flasks were placed in a thermostatic shaker at 298 K and a 6.12 pH.
Results and discussion
Response surface analysis of phosphate removal
Model equation construction
The design matrix and experimental results adopting Box–Behnken method.

Analysis of variance for phosphate removal.

In this case, the related factor or interaction is significant. Therefore, parameters X1, X2, and X3 and their interactions (i.e. X1 × X2, X1 × X3, X2 × X3, X12, X22, and X32) for the percentage of phosphate removal are statistically significant. Meanwhile, the second-order polynomial model for phosphate reduction given by OPTIMIZATION of “Design Expert” is presented in equation (1)

Here equation (1) is adopted to construct the contour plots where the third independent variable is in midpoint level. Equation (1) implies that phosphate removal efficiency improved with the AlCl3 usage (X1) and pH (X2) yet decreased dosage (X3).
The ANOVA was adopted to assess the significance of the developed model. As a valid statistical model, the adjusted R2 should be within R2 ± 0.2 (Bello and Ahmad, 2011b). In this present work, the difference between R2 and R2adj is 0.001 (R2 = 0.999 and R2adj = 0.9998). Meanwhile, the value of lack of fit is 0.0538, which is not significant relative to the pure error. These results indicate that this developed adsorption behavior could be described well by the predicted model.
3D response surface and contour plots
Interaction of each of the independent actors on the response variables in the whole design space is presented by the contour plots (Figure 1). The effects of AlCl3 concentration, pH values, and dosage on percentage reduction of phosphate are plotted in Figure 1.
3D response surface and contour plots of phosphate removal as the function: (a) C(AlCl3) and pH, (b) pH and dosage, and (c) C(AlCl3) and dosage.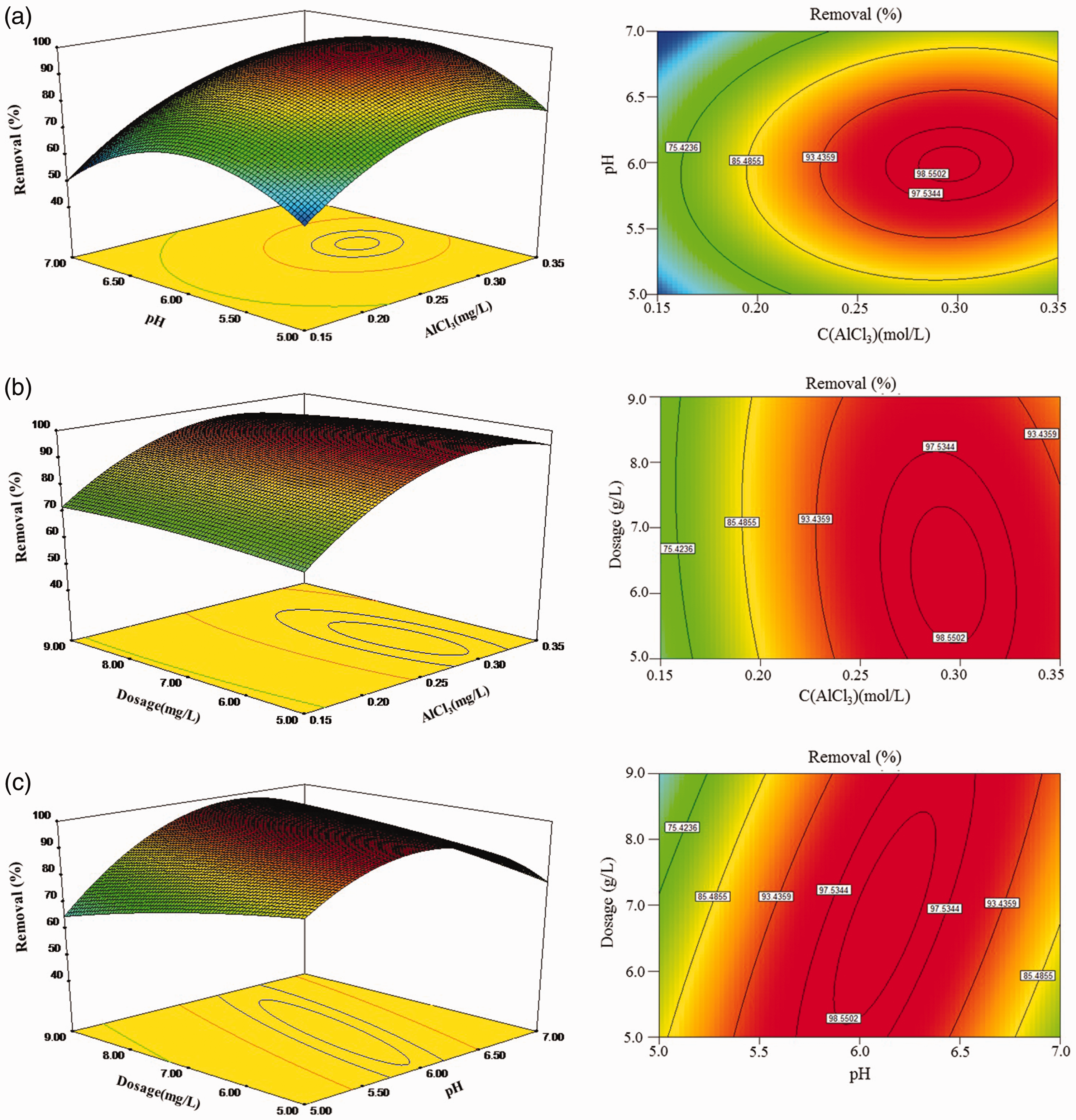
As per Figure 1(a), phosphate removal increases with the simultaneous behaviors of C(AlCl3) and pH value to 0.29 mol/l and 3.16, respectively. At this point, phosphate removal reaches 99.04%. The interaction between C(AlCl3) and dosage gives quite a different trend. As shown in Figure 1(b), the maximum phosphate removed is 98.94% when C(AlCl3) is 0.29 mol/l with a 6.14 g/l dose. Meanwhile, the effect of C(AlCl3) is much more obvious than the dose on the phosphate adsorption process. It has been found that removal of phosphate could reach more than 98.80% when C(AlCl3) ranges from 0.28 to 0.32 mol/l and dosage ranges from 5.54 to 6.78 g/l. Phosphate removal does not change significantly with a varying dose. As for the interaction effect between parameters of pH and dosage, the optimal point for phosphate removal (96.70%) is pH 6.12 with a 7.28 g/l dose.
In a combination of ANOVA and outcomes from the section of Model equation construction, the suggested solution was considered to be 0.29 mol/l C(AlCl3), pH 6.12, and a 6.72 g/l dose. At this point, phosphate species removal may reach 99.06%.
Moreover, the shape of the relationship of factors implies the interaction intensity between variables, that is the oval contours indicate a stronger interaction intensity than circular ones. Therefore, Figure 1 indicates that the interaction between pH and dose is stronger than that of both C(AlCl3) versus dose and C(AlCl3) versus pH.
Characteristics of the eggshell composite
Chemical components of natural eggshell and Al-eggshell.

Park et al. (2007) stated that all eggshells have similar chemical components which mainly include CaCO3 and a few of other elements, i.e. Mg, S, Na, P, Al, K, Sr, and Si. Before modification, CaCO3 as limestone (97.025%) and MgO (0.842%) were the important inorganic compositions of eggshell. After modification, the dominant inorganic composition was identified as lime (96.883%) as well. The proportion of other elements decreased, excluding Cl. The result taken from Table 4 implies that the composition of the eggshell can be modified.
Physical properties of the natural and Al-eggshells.

BET: Brunauer,Emmett and Teller (BET), a surface area method named by these three famous persons.
Note: SBET is BET surface area (m2/g), Vmicro is micropore volume (cm3/g), Vmeso is mesopore volume (cm3/g), and VT is total pore volume.
Table 5 suggests that the Al-eggshell has improved SBET, Vmicro, Vmeso, and VT compared with natural eggshell sample. Figure 2 provides the pore size distribution of the natural eggshell and Al-eggshell samples. Both the natural eggshell and Al-eggshell exhibit one peak around 2–10 nm. As it is seen from Figure 2, these both samples include mesopores.
Pore size distribution of natural eggshell and Al-eggshell.
The XRD patterns of the natural eggshell and Al-eggshell were explored in Figure 3. The main peak appeared at 2θ = 29.5°. Simultaneously, Figure 3 also shows several other small peaks at 2θ = 23.2°, 39.5°, 47.3°, 48.7°, and 57.6° in both sorbents. This spectrum confirms the presence of calcium carbonate according to JCPDS Card No.82-1690, where the main characteristic peaks of calcium carbonate were investigated in the XRD graph of the eggshell powder. This outcome also confirms that the calcium carbonate is the main constituent in the eggshell powder (Elabbas et al., 2016; Li et al., 2016). The comparison of the peak information of both the natural eggshell and Al-eggshell implied that the main peak intensity increased; however, no aluminum hydroxide/oxide peak information was found. The reasons for this phenomenon maybe include (1) the mass of Al was too small and (2) aluminum hydroxide/oxide XRD diffraction peak is wide.
XRD spectra of natural eggshell and Al-eggshell.
SEM was used to examine the surface morphology of both the natural and Al-eggshells. SEM images of the eggshell sample before and after modification are shown in Figure 4(a) and (b), respectively. The natural eggshell has irregular granular crystal with 0.5–18 µm (Figure 4(a)). After modification, the surface of Al-eggshell is a relatively smooth sheet-like shape (Figure 4(b)).
SEM images of natural eggshell (a) and Al-eggshell (b).
Adsorption isotherms
The adsorption of phosphate onto the Al-eggshell at 293, 298, 303, 313, and 323 K was determined as a function of the equilibrium phosphate concentration and the corresponding adsorption plot, shown in Figure 5 (a) to (e). The analysis of equilibrium data is a requirement for the sign of adsorption systems. The experimental data were fitted to the Langmuir, Freundlich, Langmuir–Freundlich (Sips), and Temkin equations and the constant parameters of the isotherm equations were calculated. These model equations are expressed by equations (2) to (5) (Cheung et al., 2000; Chun et al., 2005; Mezenner and Bensmaili, 2009), respectively
Isotherm for phosphate removal at various solution temperatures on Al-eggshell (pH = 6.12, sorbent dosage=6.72 g/l): (a) 293 K, (b) 298 K, (c) 303 K, (d) 313 K, and (e) 323 K.




Figure 5(a) to (e) suggests that the phosphate loading capacity of the Al-eggshell increased with the increase of temperature up to 323 K. This indicates that the adsorption process of phosphate was favored at higher temperatures and that the sorption course is endothermic.
The Temkin model had the poorest fit to the experimental data in comparison to other isotherm equations, with R2 ranging from 0.8565 at 293 K to 0.9154 at 298 K. A comparison of the experimental isotherms with the adsorption models described suggests that the Freundlich isotherm equation provides the best fit at 313 and 323 K (R2 = 0.9828 and 0.9945). The Langmuir–Freundlich equation gave the greatest overlap with the experimental data at 293, 298, and 303 K. This can be verified by a high correlation coefficient of 0.9917, 0.9951, and 0.9940.
The n parameter of the Freundlich equation ranging from 2.44 to 3.36 implies that adsorption sites with low energetic heterogeneity of the adsorption system (Chardon and Blaauw, 1998). The Freundlich parameter Kf reached the corresponding maximum value of 3.60 at 323 K and n of 3.36 in this work. This indicated that binding capacity reaches the highest value and that the affinity between the Al-eggshell and phosphate was also higher than other temperature values applied. The constant b represents affinity between the sorbent and sorbate, which increases from 0.038 to 0.284 l/g, respectively, with increasing temperature from 293 to 323 K.
Figure 5(a) to (e) suggests that the Langmuir–Freundlich model describes the adsorption of phosphate data with higher precision than the other fitted models. Generally, three-parameter models represent adsorption isotherm data well, because of high correlation coefficients and low percentage error values in comparison with the two-parameter models. In the course of the experiments, the value of K and β varied as a function of solution temperature. Accordingly, on increasing the temperature from 293 to 323 K, the value of K decreased from 22.33 to 8.96 mg/l and the value of β from 0.53 to 0.35.
Adsorption kinetics and effect of contact time
Kinetic studies can give further information on the adsorption process mechanisms. In this work, the experimental data were fitted to three of the most widely used kinetic models, which are pseudo-second-order, intraparticle diffusion, and Elovich models.
The pseudo-second-order model (Ho and McKay, 1999) is given as

The intraparticle diffusion model (Weber and Morris, 1963) is given as

The Elovich model (Inyang et al., 2016) has been accepted that the chemisorption process can be described below

The contact time effect on the process of phosphate adsorption and the kinetics fitting lines is assessed in Figure 6. The parameters for each equation are listed in Table 6.
Contact time effect on phosphate adsorption by Al-eggshell and kinetics fitting lines: (a) 3 mg/l and (b) 50 mg/l. Parameters for kinetic equations.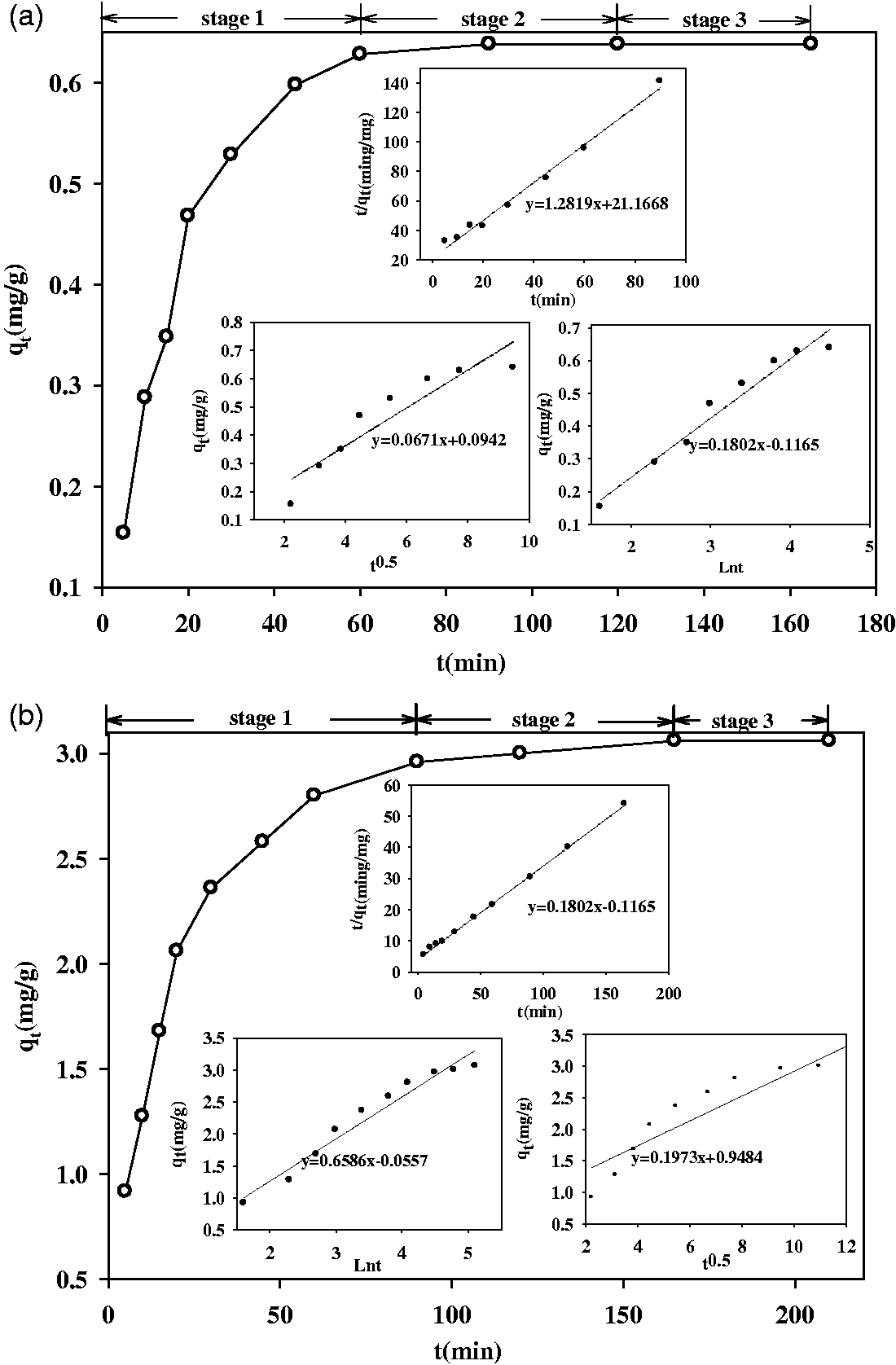

The phosphate uptake process onto the Al-eggshell appears to occur over three stages: (1) a first sharper reaction stage at 0–60 min for section of 3 mg/l and 0–90 min for 50 mg/l; (2) a low reaction stage at 60–120 min for phosphate concentration of 3 mg/l and 90–120 min for 50 mg/l; and, (3) an equilibrium stage after 120 min for both these sections (Figure 6). The first stage is similar to a report by Yang et al. (2013) who performed experiments applying Kanuma mud to adsorb phosphate. Phosphate was rapidly uptaken for 60 and 90 min for two phosphate solutions, possibly due to surface adsorption. Meanwhile, higher availability of vacant adsorption sites supplied advantageous effects in stage 1. Moreover, a higher driving force (Peleka and Deliyanni, 2009) provided by high concentration improved the contact chance between the sorbent and sorbate, thus, the higher phosphate adsorption efficiency by Al-eggshell can be achieved.
As seen from Figure 6 and Table 6, the pseudo-second-order kinetic equation with higher R2 values (0.9902 for 3 mg/l and 0.9992 for 50 mg/l) revealed better compliance with the experimental outcomes, implying that the adsorption is controlled by chemosorption. It can also be seen that equilibrium adsorption capacity (qe) increases from 0.7801 to 3.3389 mg/g as the initial phosphate concentration increases from 3 mg/l to 50 mg/l. Furthermore, the variations of the rate constant (k2) have a declining trend with improving initial phosphate concentration. The initial adsorption rate, h (mg/g min), is expressed as
The fraction h was calculated from the intercept of the line obtained by plotting t/qt versus t. There is a big increase in terms of h values of 0.202 mg/g min between 3 and 50 mg/l phosphate solutions.
Intraparticle diffusion model gave the poorest fitting efficiency, with correlation coefficients of 0.8631 and 0.8310 for these sections. The plot of qt versus t0.5 does not pass through the origin. This indicates some degree of boundary layer control and this also further implies that the intraparticle diffusion is not the only rate-limiting step, all of which could be happening simultaneously (Arami et al., 2008). It was obtained that intraparticle rate parameter values (kt) augmented with initial phosphate concentration. This can be described by the growing effect of driving force leading to a decrease in the diffusion of phosphate in the sorbent.
The Elovich equation coefficients might be calculated from the plot qt versus ln t. It can be seen that the values of ae and be varied as a function of the initial phosphate concentration. Hence, on increasing the initial phosphate concentration from 3 to 50 mg/l, the value of ae increases from 0.0944 to 0.6052 mg/gmin. Regarding the constant be, an increase of the initial phosphate concentration from 3 to 50 mg/l results in a decrease in the values of ae from 5.54939 to 1.5184 g/mg due to the decreased surface availability for phosphate.
Further discussion
Phosphate dissociation equilibrium in aqueous solution is pH dependent, presenting as (Yang et al., 2013)




As per the research from Karageorgiou et al. (2007), the dominant species are H2PO4− between a pH of 5–7; HPO42− from 7 to 12; and, PO43− when pH is higher than 12. The preferred pH of this study is 6.12. Thus, the dominant species is H2PO4− and is followed by HPO42−.
It is well known that metal oxides and hydroxides primarily existed bound to oxygen atoms and hydroxyl groups formed on the surface, which could provide an absorbing site for adsorption of phosphate (Yang et al., 2013).
The binding sites are protonated (MOH2+) when phosphate is at various pH solution systems. Therefore, the reaction of electrostatic adsorption, 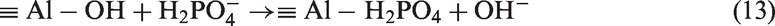

The proportion of Al increased sharply after it was modified by AlCl3 (Table 2), which enhanced the adsorption of phosphate efficiency effectively. Thus, this phenomenon seems to imply that for adsorption of phosphate onto the Al-eggshell, outer sphere complexes between the sorbent component surface and P, including electrostatic bonding mechanisms, potentially coexist with inner sphere surface complexes between the Al-eggshell and phosphate, which necessarily containing quantities of covalent bonding. Collins et al. (1999) reports that phosphate species are usually considered to be removed through an inner sphere ligand exchange mechanism (Collins et al., 1999). Meanwhile, a higher coulomb force between binding sites and phosphate in addition to chemical interaction results in higher phosphate loading, as presented in equations (13) and (14). The schematic diagram of phosphate adsorbed on Al-eggshell is explained in Figure 7.
Schematic diagram of phosphate adsorbed on Al-eggshell.
Reported sorption capacity of phosphate for various sorbents.

Conclusions and further research
The experimental and modeling results indicated that Al-eggshell could potentially be adopted as a sorbent for phosphate species.
As per Box–Behnken design, a quadratic model was developed for prediction of phosphate species adsorption by Al-eggshell. The prediction model exhibited that phosphate species removal efficiency increased by AlCl3 concentration and pH values and decreased with dosage. The optimum solution suggested by OPTIMIZATION of “Design Expert” was 0.29 mol/l C(AlCl3), pH 6.12, and a 6.72 g/l dose with 99.06% removal. Three-dimensional response surface and contour pictures reported that the interaction between pH and dosage was the strongest, followed by C(AlCl3) and dosage, and finally C(AlCl3) and pH.
Present work described that this adsorption process the Freundlich model at temperature 313 and 323 K provided good fit. On the other hand, the Langmuir–Freundlich equation agreement better fit with the data at other temperatures. The pseudo-second-order kinetic equation described the experimental results well, which implies that the adsorption is controlled by chemosorption.
In addition, Al-eggshell adsorbing phosphate species is friendly to the environment, requires no further treatment, and the absorbed sorbent can be used for soil fertilization.
Based on previous and present work, further research should focus on expanding the application of the Al-eggshell to heavy metals and organic contaminants, for example. The point of the selectivity of phosphorous over other ions also will be paid more attention in the following studies.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: publication of this article: Funding for this work by National Natural Science Foundation of China (51674161), Innovation Team Project of Shandong University of Science and Technology (2012KYTD102), Specialized Research Fund for the Doctoral Program of Higher Education (20133718110005), and Shandong Provincial Education Association for International Exchanges is gratefully acknowledged.