Abstract
Poly(acrylic acid-co-2-acrylamide-2-methyl-1-propanesulfonic acid)/kaolin hydrogel composite was synthesised using acrylic acid (AA), 2-acrylamide-2-methyl-1-propanesulfonic acid (AMPS) and kaolin (KL) as main materials. The composite was characterised and used to remove methyl orange (MO) from aqueous solutions. Effect of adsorption conditions, including initial concentration of MO, contact time, pH values and ionic strength, on the adsorption capacities was studied. Maximal adsorption capacity was 506 mg/g as the initial concentration of MO was 1000 mg/L. It showed that the adsorption process was spontaneous, and the isotherms and kinetics were in good agreement with the Freundlich isotherm model and pseudo-second-order equation, respectively.
Introduction
The dyeing and printing industry is of great importance in the development of national economy. However, large amounts of dye wastewater with high levels of chroma (500∼5000000) and toxicity are produced with the production and usage of dyes. Various methods have been tried to remove dyes from wastewater, including physical-chemical methods, biological methods, adsorption and electrochemical methods, etc. 1 Among them, adsorption seems to be superior to others in terms of its low investment, ease of operation, and renewable capability. 2 I'nal et al 3 have synthesised novel blend hydrogel beads to remove anionic textile dyes such as reactive red-120 (RR), cibacron brilliant red 3B-A (CBR) and remazol brilliant blue R (RBB) through adsorption. The maximum adsorption capacities are 116.8, 73.3 and 55.3 mg/g for RR, CBR and RBB, respectively. The adsorption is well described by pseudo-second-order kinetics and Langmuir isotherm. Chen et al 4 have studied the adsorption behaviour of both cationic and anionic dyes using a synthetic polymer. The highest adsorption capacities for victoria blue, crystal violet, neutral red and methylene blue are 5.821, 6.825, 6.135 and 5.381 mmol/g, respectively. Adsorption kinetics and isotherms analysis suggest that all the adsorption processes are exothermic and spontaneous.
Methyl orange (MO) is a kind of the p-aminoazobenzene (p-AAB) dyes. It is widely used in textile dyeing industry and chemical experiments as an acid-base indicator. However, the aqueous solution of MO is poisonous and irritating. Thus, removal of MO from aqueous solution is of great significance.
Kaolin (KL), whose chemical formula is Al4[Si4O10](OH)8, is one of the clay minerals with hexagonal flaky structures. Due to the asymmetry between the silicon oxygen tetrahedron and aluminium oxygen octahedrons, the interlayer adhesion of kaolin is extremely strong. Besides, owing to the numerous hydroxyl groups on its surface, kaolin is highly hydrophilic. As a result, kaolin is considered to be an alternative mineral resource with good stability and swelling abilities. 5 However, kaolin itself is impractical as an adsorbent due to its poor mechanical properties. By being incorporated into polymers or copolymers, the mechanical behaviour, thermal stability as well as the adsorption performance of the product can be improved greatly. Thus, great attention has been paid to the preparation and application of kaolin/polymer composites. 6 Chen et al 7 have prepared a novel urea-formaldehyde resin/ reactive kaolin composite by in situ polymerisation. It showed that the thermal stability was much better than that of pure urea-formaldehyde resin. Zhu et al 8 used kaolin/sodium alginate-grafted poly(acrylic acid-co-2-acrylamide-2-methyl -1-propanesulfonic acid) hydrogel composite as an adsorbent to remove lead, cadmium and zinc ions in aqueous solutions. Maximal adsorption capacities for Pb2+, Cd2+ and Zn2+ were 834.7, 69.9 and 139.8 mg/g.
In this paper, poly(acrylic acid-co-2-acrylamide-2-methyl-1-propanesulfonic acid)/kaolin (P(AA-co-AMPS)/KL) hydrogel composite was synthesised and characterised. The composite was used to remove MO from aqueous solutions by means of adsorption. Effect of adsorption conditions, including initial concentration of MO, contact time, pH values and ionic strength, on the adsorption capacities was studied in detail. The adsorption isotherms and kinetics were also discussed.
Experimental
Materials
Acrylic acid (AA, chemically pure, Sinopharm Chemical Reagent Co., Ltd, China), 2-acrylamide-2-methyl-1-propanesulfonic acid (AMPS, analytical reagent, Shandong Lianmeng Chemical Group Co., Ltd, China), Kaolin (KL, chemically pure, Shantou Xilong Chemical Co., Ltd, Guangdong, China). Ammonium persulfate (APS, analytical reagent, Tianjin Guangcheng Chemical Reagent Co., Ltd, Tianjin, China), N, N'-methylenebisacrylamide (MBA, analytical reagent, Chengdu kelong Chemical Reagent Factory, Sichuan, China), Methyl orange (MO, chemically pure, Shanghai Reagent Factory, China). The rest of the reagents were all analytical reagents.
Characterisation
IR spectra were recorded on a Nicolet 380 Fourier Transform Infrared Spectrometer using KBr pellets. XRD analysis was performed on an Ultima IV X-ray diffractometer (Rigaku Corporation). Morphology photos were obtained on a KYKY-2800B scanning electron microscope instrument with an accelerating voltage of 20 kV. TGA was carried out on a TGA/SDTA851e thermogravimetric analyser using nitrogen purge at a heating rate of 10°C/min. Absorbance of the samples was obtained from a WFG-7200 spectrophotometer (Shanghai Instruments Co., Ltd, China) at a wavelength of 560 nm using deionised water as a reference.
Synthesis of P(AA-co-AMPS)/KL Hydrogel Composite
7.2 g of AA was added in a beaker with 20 mL distilled water. NaOH solution (25 wt%, 12.8 g) was dropped into the beaker to neutralise AA to a degree of 80%. AMPS (1.44 g, 0.007 mol) was added to the neutralised AA solution. After stirring for 10 min the mixture was poured into a four-necked flask, which was equipped with a stirrer, a condenser, a nitrogen line and a thermometer. 1.57 g KL powder was dispersed into the mixture. After stirring for 20 min in a water bath at 40°C, MBA solution (0.005 g/mL, 2 mL) was added dropwise under nitrogen atmosphere. The mixture was slowly heated to 75°C and APS solution (0.1 g/mL, 2 mL) was dropped into it. The above mixture was allowed to react for 3 h at 75°C. The resulting gel was cut into small pieces and dried in an oven till constant weight.
Adsorption Experiments
A certain amount of the dry P(AA-co-AMPS)/KL hydrogel composite was immersed in a series of MO solution with different initial concentrations at room temperature. After adsorption for a certain period of time, the swollen samples were filtered and the filtrates were obtained for further determination of absorbency. Concentrations of MO in the filtrate were determined from the standard curve of MO. The adsorption capacity of MO on P(AA-co-AMPS)/KL hydrogel composite was calculated on the basis of Eq. (1):
9
IR Spectra
IR spectra of kaolin, P(AA-co-AMPS) copolymer and P(AA-co-AMPS)/KL hydrogel composite are presented in

IR spectra of kaolin (A), P(AA-co-AMPS) copolymer (B) and P(AA-co-AMPS)/KL hydrogel composite (C)
XRD patterns for KL (A) and P(AA-co-AMPS)/KL hydrogel composite (B) are shown in
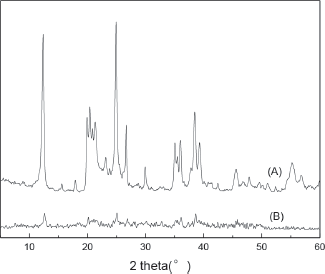
XRD patterns for KL (A) and P(AA-co-AMPS)/ KL (B)

SEM images for P(AA-co-AMPS)/KL hydrogel composite (A) and P(AA-co-AMPS) copolymer (B)
The morphologies of poly(AA-co-AMPS) copolymer and P(AA-co-AMPS)/KL hydrogel composite are displayed in
Thermogravimetric Analysis
TGA curves of P(AA-co-AMPS)/KL hydrogel composite (A) and P(AA-co-AMPS) copolymer (B) are shown in
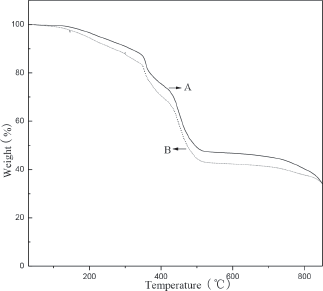
TGA curves of P(AA-co-AMPS)/KL hydrogel composite (A) and P(AA-co-AMPS) copolymer (B)
A series of methyl orange solutions with same volume (50 mL) but different initial concentrations are added into the corresponding 100 mL beakers. After the addition of 50 mg dried hydrogel composite to each beaker, respectively, the composites are allowed to adsorb MO for 24 h. The adsorbing capacities are calculated. Effects of initial concentrations of MO on the adsorption capacity are shown in

Effect of initial concentration of MO on the adsorption capacity (composite 50 mg, initial pH 7, contact time 24 h, temperature 15°C.)
It shows that the adsorption capacity is improved with the increase of the initial concentrations of MO. Maximal adsorption capacity is 506 mg/g as the initial concentration of MO reaches 1800 mg/L. Despite the interactions such as ion-exchange, electrostatic attraction and hydrogen bonds between the hydrogel composite and dye molecules,11,12 the good swelling performances of the hydrogel composite play an important role in the adsorption of dyes. It is known that dye molecules will distribute in the solution phase, hydrogel phase and the aqueous phase which is formed by the swelling of the composite. Thus, as the initial concentration of MO increases, the concentration of MO in both the hydrogel phase and the aqueous phase increases, and the adsorption capacities go up accordingly.
The effect of contact time on the adsorption capacity is shown in

Effect of contact time on adsorption capacity (composite 50 mg, initial pH 7, temperature 15°C)
The initial pH value of the solution plays an important role during adsorption because it influences the surface charges of both adsorbent and dye molecules. A series of HCl and NaOH solutions whose pH values range from 0.1 to 13.8 are used to adjust pH values, regardless of the volume changes of the solution. he effect of pH values on adsorption is shown in

Effect of initial pH on adsorption capacity (composite 50 mg, contact time 24 h, temperature 15°C)

The molecular structure of methyl orange
The effect of Na+ and Zn2+ concentration on adsorption is investigated by adding different amount of sodium chloride or zinc nitrate into a series of MO solutions, and the result is shown in

Effect of ionic strength on the adsorption capacity (composite 50 mg, initial concentration of MO 1000 mg/L, contact time 24 h, 15°C)
It is clear that both metal ions cause inhibition of adsorption. The effect of Zn2+ is particularly strong. On one hand, high concentration of metal ions will significantly reduce the solubility of MO, which brings about a decrease of adsorption capacities. 15 On the other hand, unlike Zn2+, certain amount of Na+ in the solution causes little impact to the swelling properties of the hydrogel composite for sodium polyacrylate is one of the main components of P(AA-co-AMPS)/KL. Thus, it is much more difficult for MO molecules to enter the inner part of the composite in the solution of Zn2+ than Na+.
Adsorption isotherms are conducive to the study of the interact forces between the adsorbate and the surface of adsorbent. Several equilibrium models are used to study the adsorption behaviour, which are the Langmuir, Freundlich and Redlich-Peterson equations, as shown in Eqs. (2), (3) and (4), respectively.
Langmuir isotherm equation:
In the Langmuir adsorption theory, the surface of the adsorbent is homogeneous, where each adsorption site has the same adsorptivity. The process is monolayer adsorption, and there is no interaction between adsorbed molecules. 16
Freundlich isotherm equation:
The Freundlich isotherm is an empirical equation which can be applied to both monolayer and heterogeneous adsorption, especially under the condition that the concentration of the adsorbate is low. 17
Redlich-Peterson (R-P) isotherm equation:
As the combination of both Langmuir and Freundlich systems, R-P model is always used to describe the adsorption on heterogeneous surfaces. 18
The data of
The calculated constants and correlation coefficients for adsorption of MO onto P(AA-co-AMPS)/KL hydrogel composite
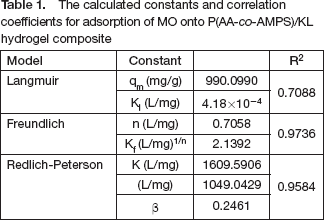
It is concluded that the adsorption process is in accordance with the Freundlich model from the values of R 2 .
The equilibrium constant obtained from the Freundlich equation are used to determine the change of the standard Gibbs free energy (ΔGθ) during adsorption according to Eq. (5):


Further study shows the adsorption kinetics during adsorption. Two models are adopted, which are the pseudo-first-order equation and pseudo-second-order equation,19,20 as shown in Eqs. (7) and (8), respectively.
Pseudo-first-order equation:

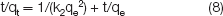
The data of
The calculated kinetic parameters and correlation coefficients for adsorption of MO onto P(AA-co-AMPS)/KL hydrogel composite

It is obvious that the adsorption of MO onto P(AA-co-AMPS)/KL conforms to the pseudo-second-order model, which indicates that the rate-determining step may be a chemical process by sharing or exchanging electrons between the adsorbent and adsorbate. 21
In this study, P(AA-co-AMPS)/KL hydrogel composite was synthesised and applied for the removal of MO from aqueous solutions. It was shown that neutral media were more effective to adsorb MO than acidic and basic media. Metal ions such as Zn2+ and Na+ restrained the composite from adsorbing MO. Maximal adsorption capacity was 506 mg/g under the conditions that the dry composite was 50 mg, initial concentration of MO was 1000 mg/L, contact time was 24 h and the temperature is 15°C. The adsorption was spontaneous and in good agreement with the Freundlich isotherm equation. A kinetic study showed that the adsorption conformed to the pseudo-second-order model.
Footnotes
Acknowledgment
The authors are grateful for support provided by a project of Shandong Province Higher Educational Science and Technology Program (J13LD07), the Specialised Research Fund for the Doctoral Program of Higher Education of China (20113718120006), the National Natural Science Fund-Coal Joint Fund to Support Key Projects (U1261205), and the key Science and Technology Project Development Plan of Qingdao Economic and Technological Development Zone (2013-1-66).




