Abstract
In this study, Co–Fe layered double hydroxides are prepared by a hydrothermal method. The Co–Fe layered double hydroxides are used as an adsorbent for the investigation of the thermodynamic parameters and adsorption kinetics of methyl orange from aqueous solution. The results show that adsorption is affected by adsorbent dosage, adsorption time, and temperature. The characteristics of samples are investigated using X-ray powder diffraction, scanning electron microscopy, Fourier-transform infrared spectroscopy, and N2 adsorption–desorption isotherms. The adsorption saturation level of Co–Fe layered double hydroxides on methyl orange is studied, with the results showing that the maximum uptake capacity for methyl orange is 10.21 mg g−1 based on Co–Fe layered double hydroxides. The adsorption kinetics of methyl orange is consistent with the Temkin isotherm equation and quasi-secondary kinetic model. Furthermore, separation is easily accomplished under the action of an applied magnetic field. The prepared Co–Fe layered double hydroxides can be applied as an effective adsorbent for decontamination of anionic dyes in industrial effluents.

Introduction
With rapid economic development, environmental problems such as air pollution, soil erosion, and water pollution are becoming more and more prominent. More than 7 × 105 tons of dyes are produced globally each year, of which about 10%–15% are released into the environment. 1 These dyes are non-biodegradable, potentially carcinogenic, teratogenic, and mutagenic and can pose serious risks to human health and the environment. 2
Among them, azo dyes are widely applied in the textile, printing, leather, food, and cosmetic industries and are the most used synthetic dyes.3–6 Currently, the commonly used methods for azo dye wastewater treatment include electrolytic precipitation,7,8 electrodialysis, 9 biochemical methods,10,11 and adsorption methods. 12 Among these, the adsorption method is widely used because of its high efficiency, low consumption, low cost, and simple operation.13–16 Therefore, the research and development of efficient and inexpensive adsorbents have become a current research hotspot. 6 According to the metal composition, layered double hydroxides (LDHs) can be divided into MgAl-LDHs, 17 NiAl-LDHs, 18 ZnAl-LDHs, 19 CoNi-LDHs, 20 XFe-LDHs (X: Co, Cu, Zn, Ni). 21 Among them, XFe-LDHs (X: Co, Cu, Zn, Ni) can be easily separated because of their magnetic properties. Meng et al. 22 synthesized three-dimensional porous carbon quantum dots (CQDs)/LDHs for the removal of boron(III) from water using CQDs as a structural guide. Meanwhile, Bo et al. 23 synthesized MgAl-LDHs by a hydrothermal method and investigated the mechanism for the removal of fluoride ions from water. Zhao et al. 24 prepared MgAl-LDH coated waste textile (MgAl-LDH@WT) by an in situ method for the separation of toxic metal ions such as As(V) and Cr(VI) from soil samples.
However, these applications suffered from several deficiencies, for example, a lack of procedures for how simple and rapid separation of adsorbents. Therefore, magnetic Co–Fe LDHs were applied to remove methyl orange (MO) in this study. The as-prepared samples were characterized by X-ray powder diffraction (XRD), scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR), and N2 adsorption–desorption isotherms, and their adsorption properties were investigated using MO simulation wastewater.
Results and discussion
The elemental analyses of CoxFe1-x-LDHs are shown in Table 1. The approximate chemical structural formulas of CoxFe1-x-LDHs are determined to be Co(0.58)Fe(0.42)-LDHs, Co(0.45)Fe(0.55)-LDHs, Co(0.67)Fe(0.33)-LDHs, and Co(0.77)Fe(0.23)-LDHs. The SEM images of the CoxFe1-x-LDHs samples are shown in Figure 1, with Figure 1(a) showing SEM images of Co0.45Fe0.55-LDHs. It can be seen from the images that the sample of Co0.45Fe0.55-LDHs is composed of different sized particles. The SEM images of Co0.58Fe0.42-LDHs are shown in Figure 1(b), and the sample appears as microcubes, with the microcube constructed by nanolayers. It can be seen that the Co0.58Fe0.42-LDHs are composed of an ordered stack of nanosheets of different sizes. The SEM images of Co0.67Fe0.33-LDHs show porous structured microspheres (Figure 1(c)), which increase the surface area and improve the adsorption performance. The SEM images of Co0.77Fe0.23-LDHs are shown in Figure 1(d). This sample occurs as nanoparticles. We can see from Figure 1 that the morphology of the samples changes with content of Fe.
Atomic percentage of Co and Fe in CoxFe1-x-LDHs.

Co–Fe LDHs: Co–Fe layered double hydroxides.

The SEM images of CoxFe1-x-LDHs: (a) Co0.45Fe0.55-LDHs, (b) Co0.58Fe0.42-LDHs, (c) Co0.67Fe0.33-LDHs, and (d) Co0.77Fe0.23-LDHs.
The XRD patterns of the CoxFe1-x-LDHs samples are shown in Figure 2(a). It can be seen that the CoxFe1-x-LDHs material showed a layered structure with symmetric basal reflections (006), (009), (015), (018), and (113) planes. 25 The cell parameters a and c are 0.306 and 2.271 nm, respectively, calculated from the equations a = 2d110 and c = 3/2(d003 + 2d006). 26

(a) The XRD patterns of CoxFe1-x-LDHs. (b) Nitrogen adsorption–desorption curves of Co0.67Fe0.33-LDHs. (c) The effect of the Co0.67Fe0.33-LDHs dosage on MO adsorption. (d) FTIR spectra of Co0.67Fe0.33-LDHs.
The N2 adsorption–desorption isotherm of Co0.67Fe0.33-LDHs is shown in Figure 2(b). It can be seen that the N2 adsorption–desorption isotherm of Co0.67Fe0.33-LDHs is the type IV nitrogen adsorption isotherm with H3-type hysteresis loops for the desorption isotherms. 27 It is noted that the adsorption isotherms present a plateau at high p/p0 values, showing that the physisorption of N2 was taking place in the holes and lamellar morphology of the materials. The surface area and pore size distribution were determined using the Brunauer–Emmett–Teller (BET) methods, and the results are shown in Table 2. The specific surface area of Co0.67Fe0.33-LDHs is 40.20 m2 g−1. The pore size is 9.23 cm3 g−1.
Specific surface area and pore size parameters.

The effect of the Co0.67Fe0.33-LDHs dosage on the MO adsorption is shown in Figure 2(c). As the dosage of Co0.67Fe0.33-LDHs is increased from 0.01 to 0.2 g, the adsorption of MO increases from 13.6% to 92.3%. But the adsorption of MO per unit mass of Co0.67Fe0.33-LDHs is followed by a decrease (from 33.80 to 7.86 mg g−1) because some pores do not reach saturation.
The FTIR spectrum of the Co0.67Fe0.33-LDHs is shown in Figure 2(d). The absorption band observed around 1355 cm−1 is due to CO32− in the interlayer. The absorption band around 3411 cm−1is the stretching vibration due to the hydrogen bond of the metal-hydroxy groups in the layers and hydrogen-bonded interlayer water molecules. The absorption band at 940 cm−1 is due to the bending vibration of O–H from interlayer water molecules. The absorption band around 1089 cm−1 is the stretching vibration of C–N. The sharp absorption bands around 851 and 736 cm−1 are the absorption peaks of Fe–OH, while that at 592 cm−1 is the absorption peak of Co–OH.
Figure 3(a) shows the effect of the adsorption time and temperature on the adsorption of MO by Co0.67Fe0.33-LDHs. As the temperature increases, the performance of Co0.67Fe0.33-LDHs decreases. The best adsorption performance occurs at 20 °C, indicating that the adsorption of MO by Co0.67Fe0.33-LDHs is an exothermic reaction. Furthermore, on increasing the adsorption time, the adsorption of MO gradually tended to equilibrium and basically reached an equilibrium state after 6 h.

(a) Effect of the adsorbent time and temperature. (b) Pseudo-first-order kinetic model for the adsorption of MO at different concentrations with equation fitting. (c) The second-order kinetic model fitting diagram of the adsorption of MO by Co0.67Fe0.33-LDHs at different temperatures. (d) The structure of MO.
The rate constants and linear-fitting equations of the pseudo-first-order kinetic model and their correlation coefficients are shown in Table 3 and Figure 3(b). The actual saturated adsorption capacities of Co0.67Fe0.33-LDHs for MO were 5.595, 7.911, and 10.205 mg g−1, at initial MO concentrations of 20, 30, and 40 mg L−1, respectively. The theoretical adsorption values calculated by the model are 6.48, 7.35, and 8.52 mg g−1. Although the correlation coefficient of the fitted linear equation is high, the theoretical value differs greatly from the experimental value, indicating that the adsorption of MO by Co0.67Fe0.33-LDHs does not conform to the quasi-first-order kinetic equation.
Different conditions of the pseudo-first-order kinetic model equation parameters for the adsorption MO.
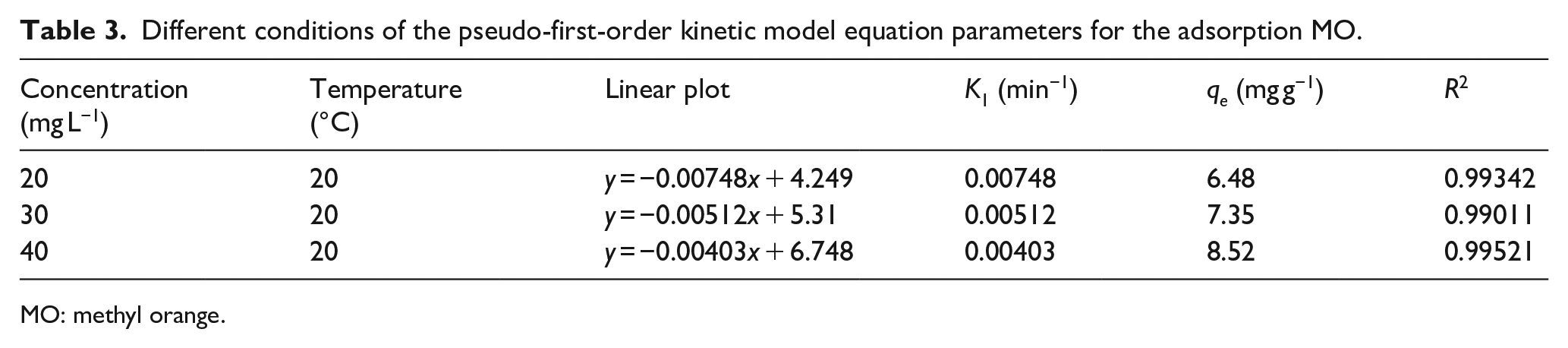
MO: methyl orange.
For further understanding the characteristics of the adsorption kinetics of MO, a pseudo second-order kinetic model was used to fit the experimental data

where k2 (g/(mg min)) is the pseudo second-order rate constant of adsorption, and qe and k2 can be obtained from a linear plot of t/qt versus t.
The linear fit equations of Co0.67Fe0.33-LDHs at different temperatures (20, 30, and 45 °C) are shown in Table 4 and Figure 3(c). The values of the correlation coefficients (R2) are higher than 0.99, and the actual saturation adsorption amounts are similar to those of the theoretical adsorption values calculated by the pseudo second-order kinetic model. Therefore, on the adsorption of MO by Co0.67Fe0.33-LDHs can be described by a pseudo second-order kinetic model. Figure 3(d) shows the structure of MO.
The equation parameters of the quasi-second-order kinetic adsorption of MO by Co0.67Fe0.33-LDHs at different temperatures.

MO: methyl orange; Co–Fe LDHs: Co–Fe layered double hydroxides.
Table 5 shows the different thermodynamic adsorption models and their linear expressions. The linear-fitting equation and related parameters of the Langmuir adsorption isothermal model for MO adsorption by Co0.67Fe0.33-LDHs are shown in Table 6 and Figure 4(a). As can be seen in Table 6, when the temperature of the system is 20 °C, the highest correlation coefficient R2 of the linear-fitting equation is 0.93, and the theoretical adsorption capacity at this temperature is 9.193 mg g−1, which is close to the real experimental value of 10.205 mg g−1. However, when the temperature of the system is 30 or 45 °C, the correlation coefficient R2 is relatively lower. At these temperatures, the theoretical adsorption capacity of the Langmuir equation is significantly different from the experimental real value, indicating that the adsorption process of MO by Co0.67Fe0.33-LDHs is not single molecule adsorption.
Different thermodynamic adsorption models and their linear expressions.
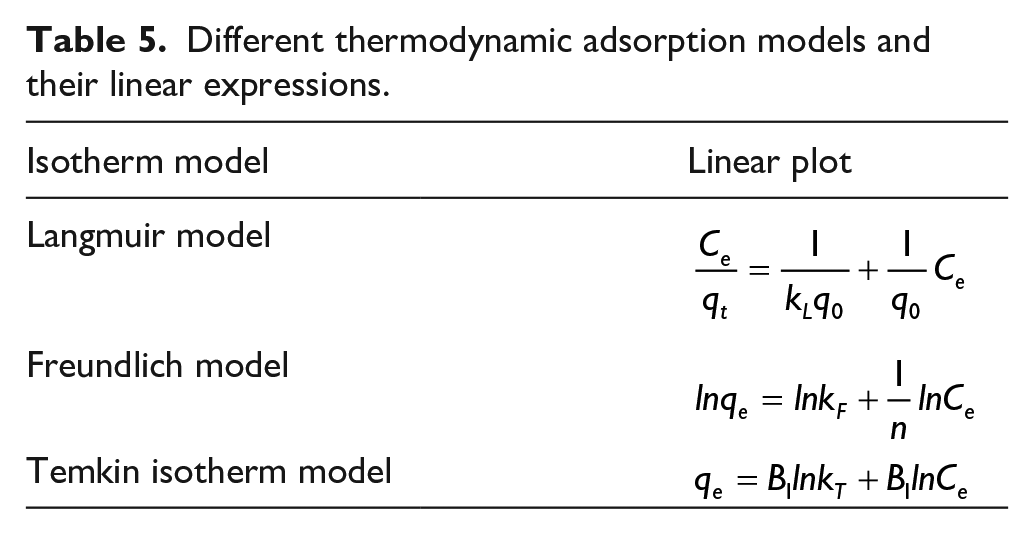
Langmuir isotherm parameters of MO adsorption.

MO: methyl orange.
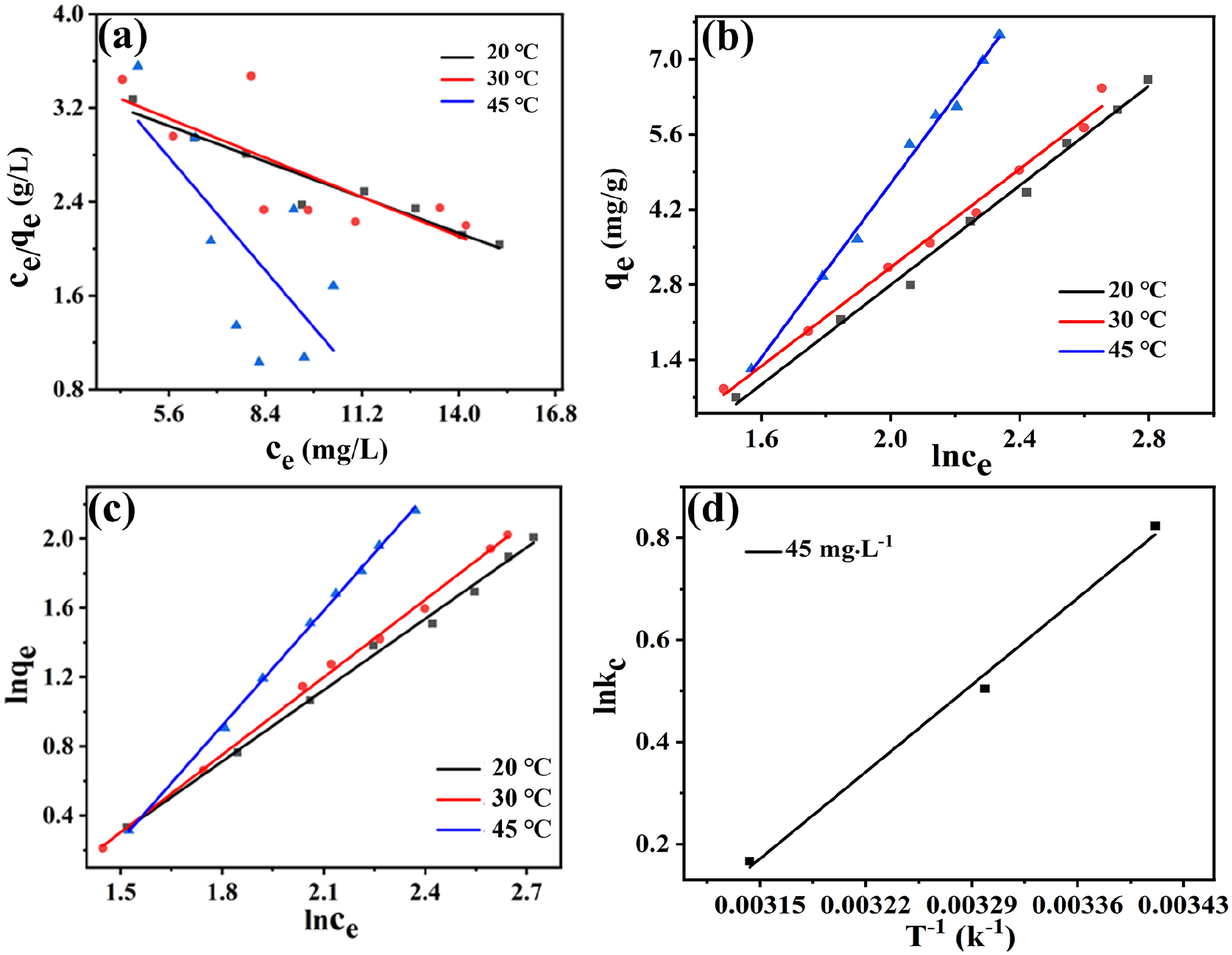
(a) The Langmuir isotherms of the adsorption of MO by Co0.67Fe0.33-LDHs. (b) The Temkin isotherms of Co0.67Fe0.33-LDHs-adsorbed MO. (c) The Freundlich isotherms of Co0.67Fe0.33-LDHs adsorbed MO. (d) The Van’t Hoff plot of adsorbed MO.
In order to understand the characteristics of the adsorption isotherms of MO adsorption by Co0.67Fe0.33-LDHs, the Temkin isotherm adsorption model was used to fit the experimental data (Figure 4(b))

where qe is the amount of substrate adsorbed by the sample when the adsorption reaches (mg g−1); Ce is the mass concentration of MO at the equilibrium of adsorption of Co0.67Fe0.33-LDHs (mg L−1); q0 is the maximum adsorption for the Langmuir model (mg g−1); KT is the Temkin isotherm rate constant of adsorption; B1 = RT·b−1, in which R is the gas constant and T is the absolute temperature.
The linear fit equations of Co0.67Fe0.33-LDHs at different temperatures (20, 30, and 45 °C) are shown in Figure 4(b) and Table 7. The values of the correlation coefficient (R2) are higher than 0.99. Hence, Co0.67Fe0.33-LDHs on the adsorption of MO can be described by the Temkin isotherm adsorption model.
The Temkin isotherm for the adsorption of MO by Co0.67Fe0.33-LDHs.

MO: methyl orange; Co–Fe LDHs: Co–Fe layered double hydroxides.
The linear-fitting equation and related parameters of the Freundlich adsorption isothermal model of MO adsorbed by Co0.67Fe0.33-LDHs are shown in Figure 4(c) and Table 8. It can be seen from Table 8 that the correlation coefficient of each linear-fitting equation is above 0.99, which is higher than the correlation coefficient of the Langmuir isotherm equation and the Temkin isotherm equation. The results indicate that the adsorption process of MO by Co0.67Fe0.33-LDHs can be described by the Freundlich isothermal equation. In the Freundlich model, n represents a dimensionless parameter, the size of n can reflect the adsorption characteristics, and the size of 1/n can reflect the adsorption strength or surface uniformity. In this experiment, 1/n > 1 indicates that synergism occurs in the adsorption process, and there is a strong interaction between adsorbent molecules.
Freundlich isotherm parameters of MO adsorption.
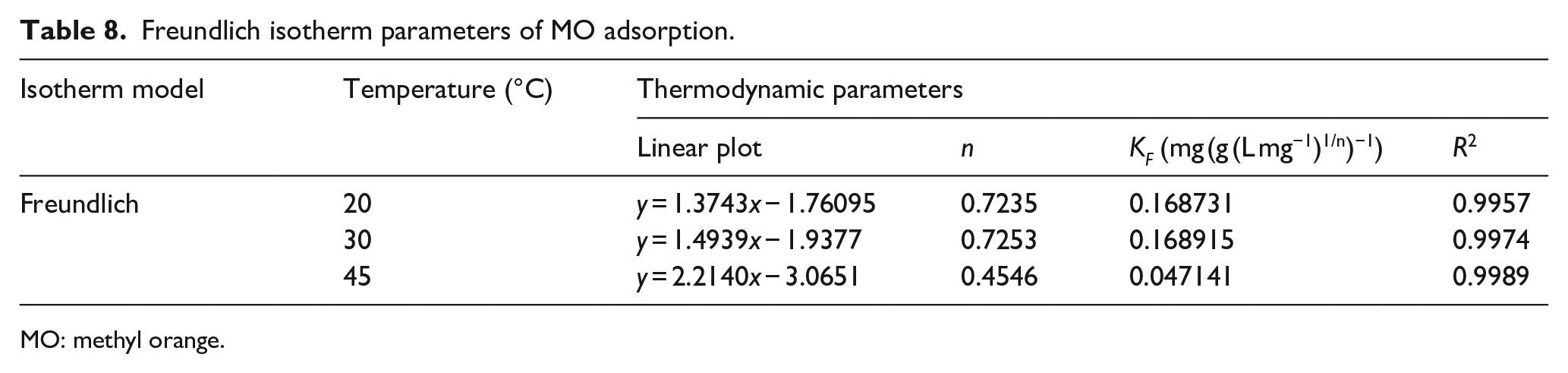
MO: methyl orange.
For an adsorption process, it can be deduced with the aid of thermodynamic parameters such as the change in Gibbs free energy (ΔG), the enthalpy (ΔH), and the entropy (ΔS), that the thermodynamic behavior of the adsorption reaction toward spontaneity can be evaluated. The parameters can be calculated using the following equation 27

The thermodynamic parameters of the MO adsorption process are shown in Table 9, and the Van’t Hoff plot of adsorbed MO is shown in Figure 4(d). Here, △G < 0, indicating that this adsorption process is spontaneous. When △S < 0, the adsorption process is spontaneous, and is a thermal entropy process.
Thermodynamic parameters of MO adsorption.

MO: methyl orange.
A recycling study using Co0.67Fe0.33-LDHs on MO adsorption/desorption is shown in Figure 5(a). The removal of MO by Co0.67Fe0.33-LDHs decreased from 94.5% to 74.8% after completing 10 cycles, which can be associated with incomplete desorption. The experimental results showed that Co0.67Fe0.33-LDHs have a good cycle stability. The magnetic behavior of Co0.67Fe0.33-LDHs is very important for practical applications. The room-temperature magnetization curves of Co0.67Fe0.33-LDHs are displayed in Figure 5(b). The hysteresis loop shows a coercivity (HC) value of 16.5 Oe, suggesting that Co0.67Fe0.33-LDHs possess magnetic properties. As shown in Figure 5(c), magnetic Co0.67Fe0.33-LDHs can be completely separated from the solution when the solution is subjected to an external magnetic field. It is immediately apparent that the Co0.67Fe0.33-LDHs have a rapid magnetic response ability at room temperature.

(a) The recycling of Co0.67Fe0.33LDHs with respect to MO adsorption/desorption. (b) Magnetic hysteresis loops of Co0.67Fe0.33LDHs (the inset shows the enlarged magnetic hysteresis loops at low applied fields). (c) Photographs of (a): MO solution, (b): a Co0.67Fe0.33-LDHs dispersion in MO solution without a magnetic field, and (c): with magnetic field applied.
Conclusion
The Co–Fe-LDHs were prepared by a hydrothermal method and used for the adsorption of MO solution. The results showed that the maximum capacity for MO was 10.21 mg g−1 based on Co–Fe LDHs. The adsorption kinetics of MO are consistent with the Temkin isotherm equation and the quasi-secondary kinetic model. This indicates that the Co–Fe LDHs could be applied as an effective adsorbent for the decontamination of MO dyes in industrial effluents. Due to the magnetic property of Co–Fe-LDHs, they can be easily separated from solution after the adsorption process, which solves the problem of adsorbent separation and reuse. It is expected that the prepared complex will be suitable for the removal of organic pollutants for wastewater, and also used in biomedical fields such as biomolecular separations, targeted drug delivery, and magnetic resonance imaging.
Experimental
CoxFe1-x-LDHs composites were synthesized from CoSO4-7H2O, FeCl3, SDBS, and Na2CO3. First, CoSO4-7H2O and FeCl3 (molar mass ratio of CoSO4-7H2O to FeCl3 was 1:1, 2:1, 3:1) were dispersed in 50 mL of deionized water. Then, 0.2 g of SDBS and 1.5 g of Na2CO3 were added and mixture stirred for 2 h. The obtained mixed solution was transferred to a reaction kettle for the hydrothermal reaction (150 °C, 6 h), cooled to room temperature and filtered by suction with rinsing with deionized water, anhydrous ethanol. The obtained solid was dried in a drying oven at 60 °C for 8 h to give CoxFe1-x-LDHs, noted as CoxFe1-x-LDHs (X) (the value of X corresponds to the molar amount of cobalt sulfate). For comparison, blank samples were prepared by the same procedure, but without the addition of SDBS, and the samples obtained are denoted as CoxFe1-x-LDHs.
The adsorbent was added to MO aqueous solution for adsorption, and the adsorption capacity was recorded from the UV-Vis absorption spectrum. Polyethyleneimine (30% aqueous solution) was used as the desorption agent, and a magnet was used to separate the adsorbent after MO desorption.
The X-ray diffraction patterns of the CoxFe1-x-LDH composites were recorded on an X-ray diffractometer (Rigaku-Ultima IV, Japan) with Cu-Kα radiation operating at 40 kV and 40 mA. The pore structures, specific surface areas, and pore sizes of CoxFe1-x-LDHs were analyzed by N2 adsorption-desorption at 77 K on an automatic adsorption system (BelSorp II, Japan). FTIR spectra were recorded on a Nicolet iS 50 FTIR instrument. The morphologies and atomic percentage of CoxFe1-x-LDHs were characterized by field emission scanning electron microscopy (Sigma 500, Carl Zeiss, Germany) equipped with an energy dispersive X-ray (EDX). The hysteresis loop of Co0.67Fe0.33-LDHs in the range of −20 to 20 kOe was obtained by a vibrating sample magnetometer (VSM) analysis (LakeShore 7404, Lake Shore Cryotronics, Westerville, OH, USA). The concentrations of CoxFe1-x-LDHs were analyzed with a UV-3200PC spectrophotometer (Shanghai Yoke Instrument Co. Ltd., Shanghai, China). Finally, we performed reusability performance test of the adsorbent after adsorption.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Research Program of Application Foundation of Qinghai Province (no. 2020-ZJ-705) and the postgraduate courses of Qinghai Minzu University in 2021 (JK-2021-09).
