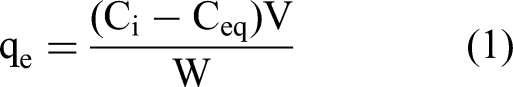Abstract
In this research, Casuarina equisetifolia fruit-based-activated carbon (CAC) was employed to investigate the simultaneous removal of crystal violet (CV), Naphthol Black Blue (NBB), and methyl orange (MO) in one mixture from aqueous solutions. The efficacy of adsorbent removal was studied concerning adsorption characteristics such as pH, dose of CAC, initial dye concentration, and adsorption time. Fourier transform infrared (FTIR), scanning electron microscope (SEM), and UV-visible analysis all showed that the dye mixture stuck to the CAC surface at the same time. Results revealed the best simultaneous adsorption efficiencies for the investigated dyes were attained in the pH range of 3–10, with a CAC dose of 0.08 g. The optimal initial concentration for the dye mixture was found to be 40 mg·L−1. The optimal contact time for CV, NBB, and MO was 45 min. Kinetic studies revealed a pseudo-second-order adsorption mechanism for the dye mixture. The Freundlich isotherm model best described the equilibrium data. The maximum adsorption capacities were CV (239.23 mg·g−1), NBB (134.95 mg·g−1), and MO (45.93 mg·g−1). Thermodynamic analysis suggests these adsorption processes to be endothermic and spontaneous. The findings of high adsorption capacities highlight the practical applicability of this approach in addressing complex dye pollution challenges.
Introduction
Water pollution happens when harmful substances such as chemicals, plastics, industrial waste, and wastewater contaminate bodies of water such as rivers, lakes, oceans, and groundwater. This pollution disrupts aquatic ecosystems, posing serious threats to marine life and human health. In addition, polluted water supplies can spread waterborne diseases, making them unsafe for drinking, bathing, and agriculture (Bashir et al., 2020). Addressing water pollution requirements coordinated global action, involving dropping waste disposal, adopting sustainable agricultural methods, and enforcing stricter regulations on industrial emissions to protect our vital water resources. Wastewater treatment is the process of removing pollutants from sewage or industrial wastewater so that it is safe for use either for recycling or environmental release. This process typically involves the use of physical, chemical, and biological techniques to filter and break down pollutants (Nishat et al., 2023). Primary treatment focuses on the elimination of solid waste, whereas secondary treatment engages biological processes to analyze and degrade organic matter. There are several advanced treatment methods, such as filtration and disinfection, to remove remaining pollutants and pathogens. To eliminate the last traces of contaminants and bacteria, numerous sophisticated treatment techniques, including filtration and disinfection, exist. Protection of ecosystems, preservation of clean water supplies, and prevention of the spread of waterborne diseases depend on effective wastewater treatment; hence it supports environmental sustainability and public health (Silva, 2023).
One of the common methods that plays an important role in several industries, as well as in the environment, is adsorption. This phenomenon is crucial for the degradation of contaminants in air, water, and earth (Satyam and Patra, 2024). Adsorption aids in the removal of contaminated sites and polluted locations by capturing toxic substances within activated carbon and clay. It is also utilized in industrial procedures for the separation of valuable gaseous and liquid products and for improving the quality of products. Adsorption also plays a crucial role in gas separation and wastewater treatment by enhancing the removal of impurities and improving the efficiency of purification systems. Recognizing the mechanism of adsorption process can lead to more efficient and sustainable approaches in managing environmental and industrial problems (Badran et al., 2023). Adsorption using activated carbon is useful in solving pollution problems of air, water, and soil because of its wide range of contaminants removal efficiency. The porous activated carbon has a large surface area. This pores trap contaminants such as volatile organics, heavy metals, and odors. Water treatment plants use this type of carbon because it plays a crucial role in wastewater treatment and drinking water purification. Activated carbon is also useful in various industrial applications for the recovery of important compounds at lower costs. The efficiency of activated carbon in adsorption techniques renders it useful in environmental management (Kuśmierek et al., 2021).
Synthetic dyes in particular are quite harmful to both humans and the environment, causing contamination of the soil and water supplies. Dyes are extensively used in many aspects of the industry, such as fabric, textile, printing, cosmetics, pharmaceuticals, and food. The negative impact of dyes appears in different forms, such as killing fish and algae, in addition to causing a decrease in biodiversity and an imbalance in ecosystems. Concerning human health, dyes may cause health issues such as skin irritation, respiratory problems, and cancer. Most dyes are not degradable in the environment, leading to long persistence. Authority in all countries must impose strict regulations, improved waste treatment procedures, and the development of safer, more ecologically friendly alternatives to decrease the toxicity of dyes and reduce the negative impact of dyes on humans and the planet (Yusuf, 2019).
The purpose of this study is to examine the potential of the activated carbon that is produced from Casuarina equisetifolia fruit (CAC) as an adsorbent for the simultaneous removal of dye mixture (crystal violet [CV], Naphthol Black Blue [NBB], and methyl orange [MO]). The chemical structure of the three dyes is shown in Scheme 1. This study also aims to indicate the best adsorption parameters for the simultaneous removal of the dye mixture. These parameters include the amount of CAC, the initial concentration of dyes, the contact time, and the pH value.

Chemical structure of (a) CV, (b) NBB, and (c) MO.
Materials and methods
Chemicals
Chemicals of CV (C25H30ClN3), NBB (C22H14N6Na2O9S2), MO (C14H14N3NaO3S), Sodium hydroxide (NaOH), and hydrochloric acid (HCl) were purchased from Sigma Aldrich. Double distilled water was used in the preparation of all solutions and all experiments.
The preparation of solutions
A 1000 mg·L−1 stock solution of CV, NBB, and MO was prepared by dissolving 1.0 g of each dye in 1.0 L volumetric flask. This stock solution was used to prepare the required concentrations related to the experiments by dilution with two-fold distilled water. Some experiments required a certain value of pH, so solutions of 0.1 M NaOH and HCl were used to achieve the desired value. A pH meter from Scientific Thermoficher was employed.
The preparation and characterization of CAC adsorbent
The CAC was collected from the campus of the University. Subsequently, the collected fruits were rinsed and dried at 70°C for 48 h. Next, they grounded, impregnated with a determined volume of 98% phosphoric acid, and finally heated to 450 °C (Alkhabbas et al., 2023). The prepared activated carbon from CAC was then rinsed with 5.0 M NaOH solution to achieve neutralization, which was confirmed by the eluent pH value. The CAC was pulverized and sieved to a particle size of 180 μm. Instrument of Fourier transform infrared (FT-IR), TENSOR model from BRUKER, Germany equipped with an ATR unit was used to identify the functional groups at the CAC surface. The measurements were conducted in the range of 4000–400 cm⁻¹ at ambient temperature. The instrument was operated with a resolution of 4 cm⁻¹, and each spectrum was obtained by averaging 32 scans to ensure a high signal-to-noise ratio. Baseline correction and spectral analysis were performed using Bruker's OPUS software. The surface of the adsorbent was depicted before and after adsorption using a scanning electron microscope (SEM) (Apreo 2 S LoVac, USA).
The experiment of batch adsorption
Numourous experiments were conducted by shaking a fixed amount of CAC with different concentrations of the three dyes (CV, NBB, and MO) to investigate the simultaneous adsorption. Adsorption parameters of temperature, shaking time, pH value, initial dye concentration, and CAC dosage were examined in this research. The concentrations of the three dyes were determined by using a UV-6100 PC double-beam spectrophotometer at wavelengths 581 nm, 610 nm, and 466 nm for CV, NBB, and MO, respectively. The adsorbed quantity of dyes (qe in mg·g−1) was computed by employing equation (1) (Al-Ma’abreh et al., 2024) in which Ci and Ceq (mg·L−1) are the initial and equilibrium concentrations, respectively:
where V and W are the volume (L) and weight of CAC (g), respectively.
The removal percentage of the dyes was computed based on equation (2):
Results and discussion
Characterization of adsorbent
Fourier transform infrared
The FT-IR spectroscopy results (Figure 1) provide important insights into the functional groups included in the CAC adsorbent and their role in the concurrent adsorption of a dye mixture. The existence of various functional groups on the surface of CAC was proven by the exhibition of distinct peaks in the FT-IR spectra. Peaks between 3340 and 3290 cm−1 corresponded to the stretching vibrations of the O-H group. Spectral peaks at 2924cm−1 correspond to the C-H stretching vibrations, which suggest the existence of cellulose and hemicellulose groups. The peaks seen at 1600–1660 cm−1 correspond to the stretching of the C=C bond. Significantly (Amran and Zaini, 2021), the change in absorbance intensity was detected when the dye mixture was adsorbed, suggesting a well-established interaction between the CAC surface and the dyes of CV, NBB, and MO. The obtained results offer insistent scientific support for the effective adsorption of the Dye mixture onto the CAC surface.

FT-IR spectra for the adsorbent (CAC) before and after adsorption of the dye mixture.
Scanning electron microscope analysis
Scanning electron microscopy analysis shows strong evidence for the successful simultaneous adsorption of the three dyes (CV, NBB, and MO) onto the CAC adsorbent. Figure 2(a) illustrates the surface of CAC before adsorption, showing a highly porous structure. The SEM image in Figure 2(b) illustrates the CAC surface after simultaneous adsorption of the three dyes. These images indicate a substantial decrease in pore visibility, which strongly implies that the moieties of CV, NBB, and MO have successfully adsorbed and covered the CAC surface pores. This visual confirmation further supports the adsorption process.

SEM analysis of (a) CAC and (b) CAC-dye mix.
Batch adsorption
The impact of CAC adsorbent dose
The impact of CAC dose on the simultaneous adsorption of CV, NBB, and MO was shown in this study. The dose of CAC was adjusted between 0.02 and 0.14 g while maintaining a fixed temperature of 298 ± 1 K. The ideal dose of the adsorbent CAC for each dye was determined to be 0.08 g, as shown in Figure 3. The simultaneous adsorption of the three dyes is significantly increased when the appropriate dose is used because it maximizes the availability of easily accessible active sites for adsorption. Nevertheless, adsorption capacity decreases when the system gets closer to equilibrium.

Impact of CAC dose on the simultaneous adsorption of CV, NBB, and MO.
The impact of the dye initial concentration
This study investigated the simultaneous dye adsorption onto CAC at a constant temperature of 298 ± 1 K concerning its initial concentration (10–80 mg·L−1). A dose of 0.08 g was shaken with different concentrations of the three dyes for a specified period. Findings (Figure 4) show removal percentages of 97.94%, 97.73%, and 96.96% for CV, NBB, and MO, respectively. Remarkably, 40 mg·L−1 was shown to be the ideal initial dose in all cases (Figure 4). There were enough accessible active sites on the CAC surface at this concentration, which significantly increased dye uptake. However, these sites became saturated as the concentration rose, leading to decreased adsorption efficiency as the system got closer to equilibriums.

Impact of dye initial concentration on the simultaneous adsorption of CV, NBB, and MO on CAC.
The impact of the adsorption time
To verify the optimal contact time required for maximum adsorption onto CAC, batch adsorption experiments were conducted for a mixture of the three dyes (CV, NBB, and MO). The experiments involved shaking 0.08 g CAC with a solution of 40 mg·L−1 CV, NBB, and MO at a constant temperature (298 ± 1 K) for varying durations (15–135 min). As illustrated in Figure 5, the maximum simultaneous uptake of the three dyes (CV, NBB, and MO) was achieved after 45 min.
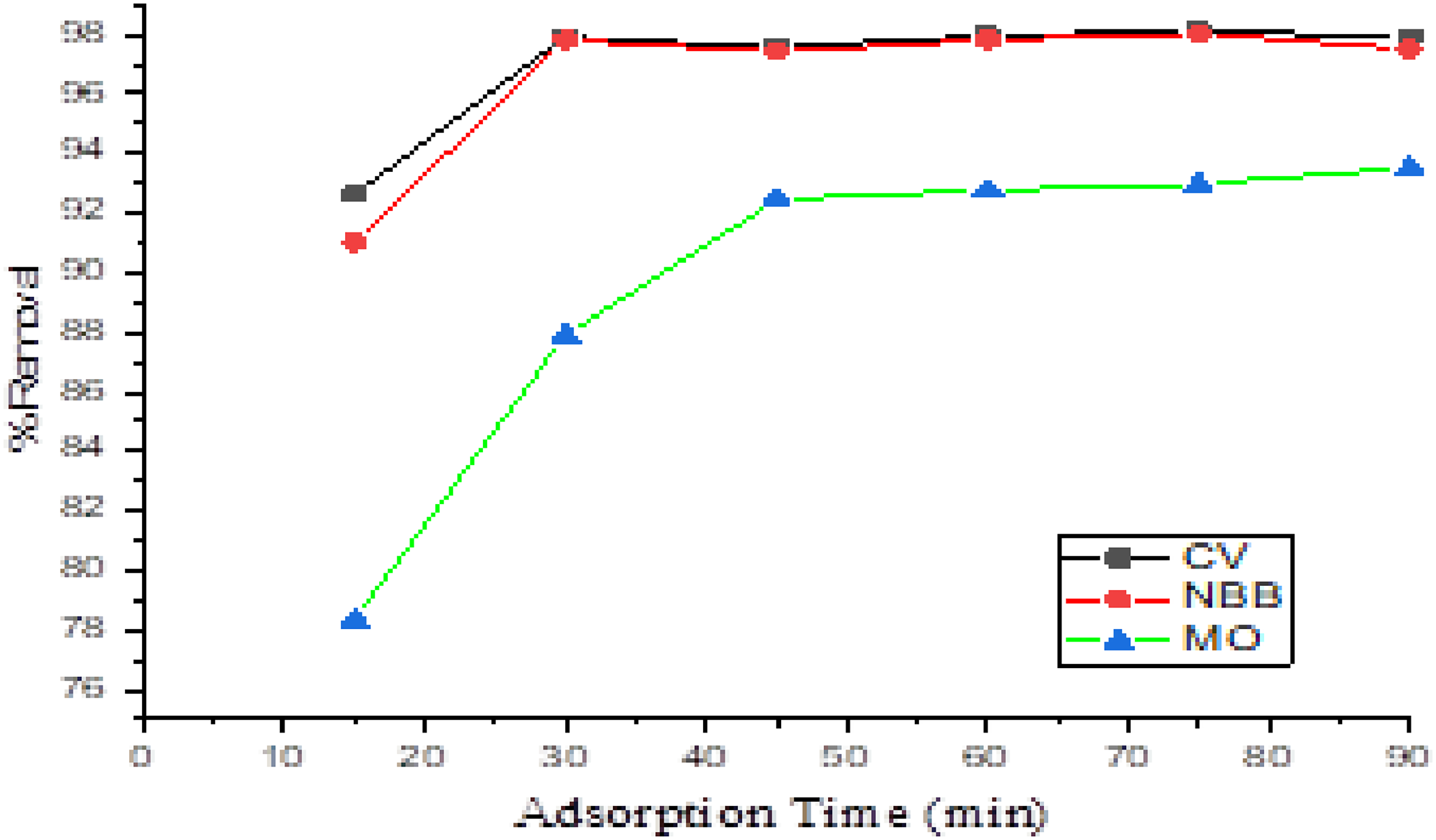
Impact of adsorption time for the simultaneous adsorption of CV, NBB, and MO on CAC.
The impact of the solution pH
The pHpzc of the CAC surface was determined to be 7.4 through an experiment. The adsorption of CV, NBB, and MO onto CAC was examined in this study concerning the pH of the solution (3–11). Batch adsorption experiments were conducted under conditions of 0.08 g CAC, a temperature of 298 ± 1 K, and 60 min shaking time. The adsorption of the three dyes in the pH range of 3–9 is only slightly influenced by pH, as illustrated in Figure 6. The percentage of removal decreases significantly after pH 9 is reached.

Impact of ph on the simultaneous adsorption of CV, NBB, and MO on CAC.
The column experiment
Following the results of the contact time impact, it was noticed that the removal of the dye mixture was greater than 70% in the first 15 min. As a result, a column experiment was conducted by packing 3.0 g of CAC adsorbent (4 cm height, 180–250 μm), then 50 ml of 40 mg·L−1 CV, NBB, and MO. Solution mixture was passed through the column for about 30 min. A non-colored eluent was obtained and subjected to absorbance measurements. Figure 7 illustrates the removal percentage of each dye. The column experiment conducted in this study provides valuable insights into the effectiveness of using CAC as an adsorbent for the removal of the three dyes. The high percentage of removal that was observed in a relatively short time suggests that this approach could be a promising solution for practical applications in the treatment of dye-contaminated wastewater (Solayman et al., 2023).
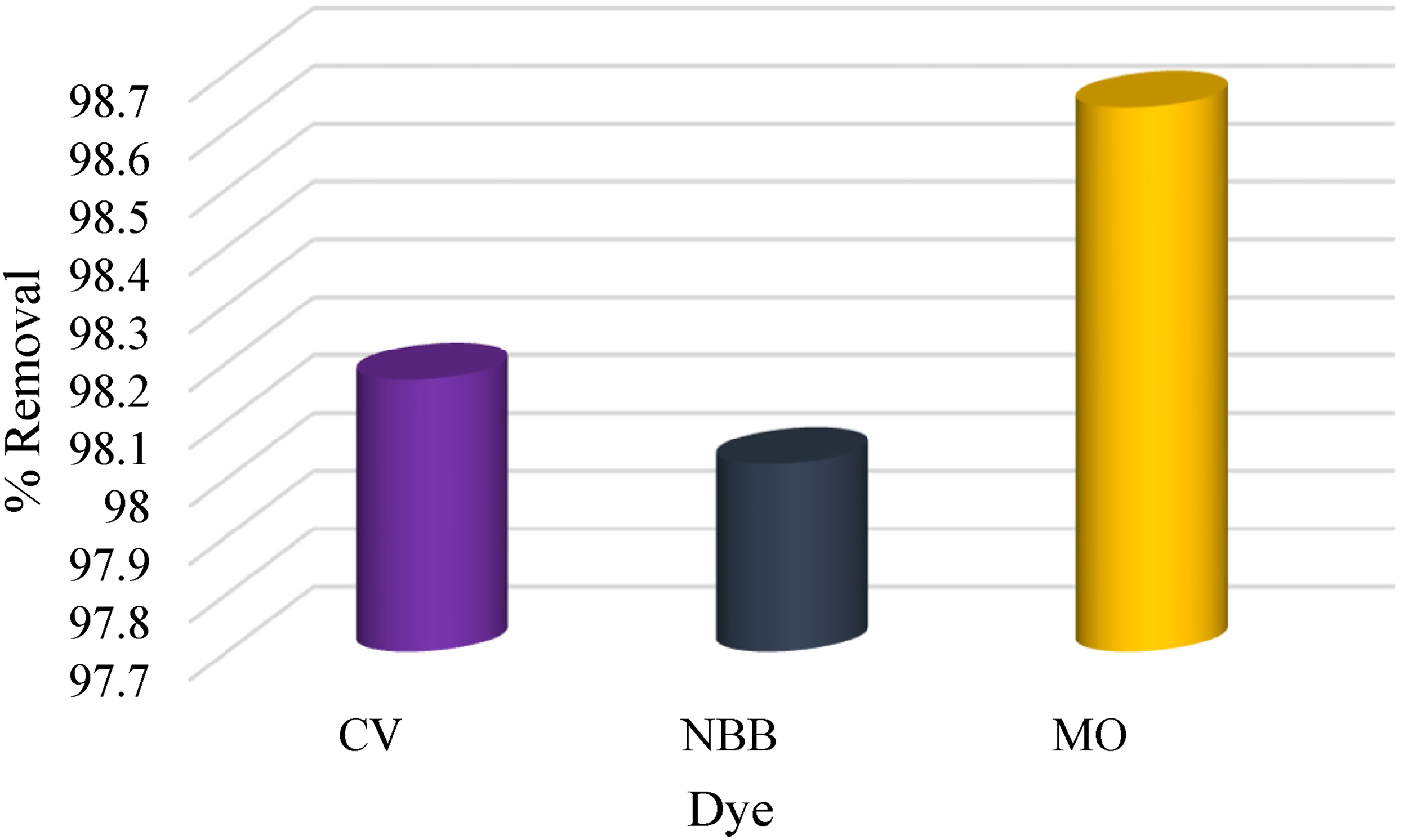
Column experiment results for the three dyes.
Kinetic and mechanism
Linear kinetic models were employed to recognize the mechanisms of the CV, NBB, and MO adsorption on CAC surface. Pseudo-first-order, pseudo-second-order (PSO), and intra-particle diffusion (IPD) models were used to analyze the adsorption process and determine the mechanism. The relevant equations, presented in Table 1, quantified the adsorption behavior of the three dyes on CAC.
Kinetics models equations.

Figure 8 provides convincing evidence that the PSO kinetic model best describes the adsorption behavior of CV, NBB, and MO onto CAC surfaces for simultaneous removal.
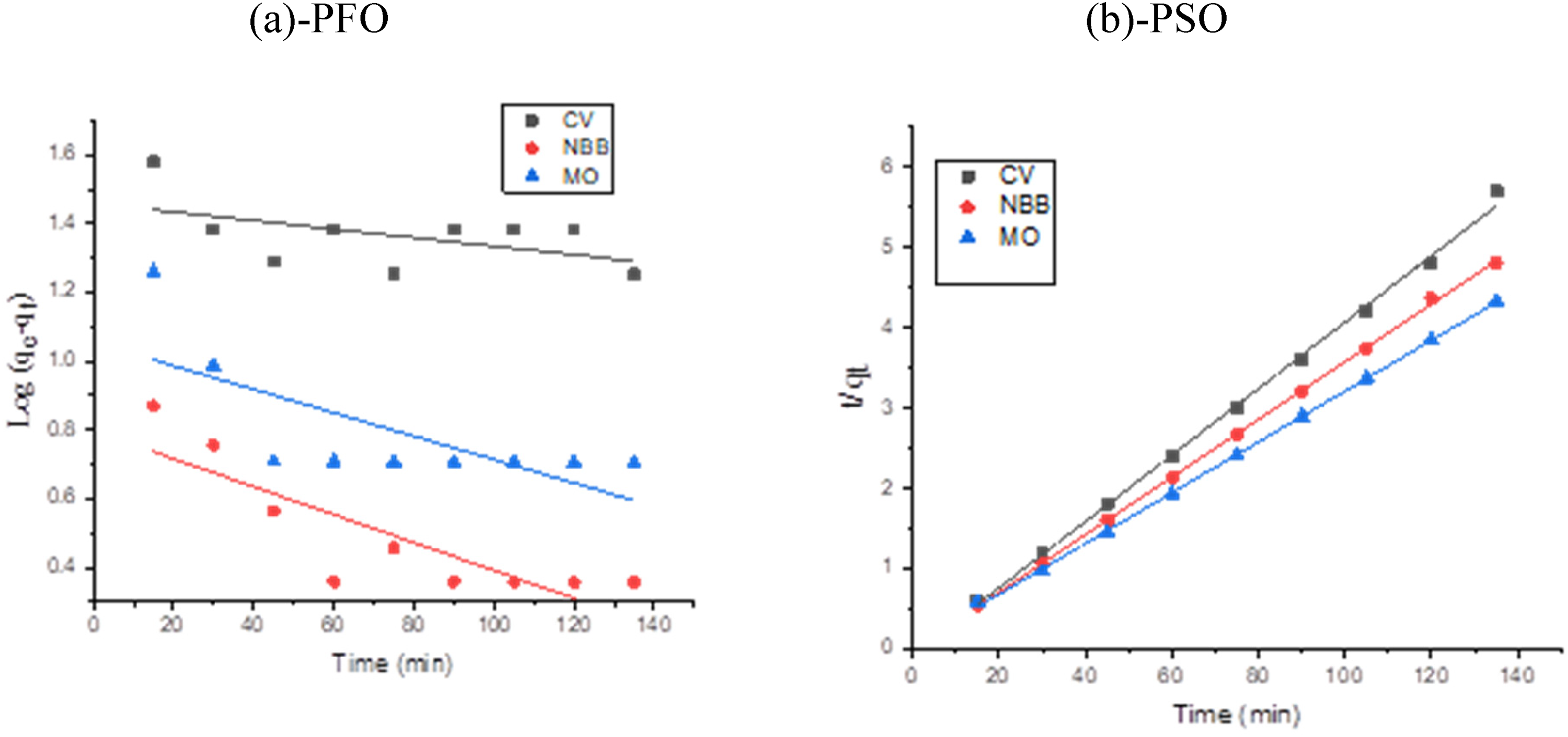
Linear kinetics studies of the simultaneous adsorption of the CV, NBB, and MO on CAC (a) PFO and (b) PSO.
Equations shown in Table 1 are used to figure out how the IPD affected the way the three dyes attached to the CAC surface. The rate-limiting step is set by the intercept (C) value found during model fitting in the IPD model. The only factor impacting the adsorption rate when C is 0 is IPD. A non-zero C, on the other hand, means that surface adsorption, along with IPD, adds to the total rate. Figure 9 indicates that surface adsorption appears to be the phase that limits the rate at which the three dyes react, thereby controlling the process's total speed. The values of R2 and constants for all kinetics models are summarized in Table 2.

Linear and intra-particle model for the simultaneous adsorption of the CV, NBB, and MO on CAC.
Results of kinetics studies for CV, NBB, and MO simultaneous adsorption onto CAC.

The adsorption isotherms
Various adsorption isotherm models were employed to identify the nature and characteristics of the adsorption process for CV, NBB, and MO onto CAC. Some of these models, such as Langmuir, Freundlich, and Temkin (refer to Table 3 for details), helped to figure out how adsorbent (CAC) and the adsorbate (CV, NBB, and MO) interacted with each other. By analyzing the fit of these models, insights could be gained into whether the adsorption process is primarily physical or chemical.
Linear isotherms model equations.

Figure 10 (a–c) presents the linear form of the three isotherm models (Langmuir, Freundlich, and Temkin) for visual analysis. Table 4 summarizes the key parameters obtained from these plots, including correlation coefficients (R²) and relevant constants. Notably, the Freundlich isotherm exhibited the highest R² values for CV (0.9872), NBB (0.9912), and MO (0.9590) adsorption onto CAC. The high R² values point to a good fit for the Freundlich model. They show a non-uniform CAC surface with different adsorption sites for the three dyes (CV, NBB, and MO). The Temkin constant values show that the dye and the CAC adsorbent have a strong interaction.

The linear isotherms of (a) langmuir, (b) freundlich, and (c) temkin of CV, NBB, and MO removal by CAC.
Isotherms model constants.

Thermodynamics
To find out how well simultaneous removing of CV, NBB, and MO by CAC, thermodynamic parameters were determined under controlled conditions. Experiments were conducted at three constant temperatures (298 K ± 1.0, 308 K ± 1.0, and 318 K ± 1.0) with consistent agitation (1.0 h). Key thermodynamic parameters (ΔG°, ΔH°, and ΔS°) were calculated to reveal the nature of the adsorption process. The results are presented in Table 5. The standard Gibbs free energy change (ΔG°) was calculated using ΔG° = ΔH°-TΔS°. The Vant Hoff equation was used to determine the enthalpy change (ΔH°) and entropy change (ΔS°) from the slope and intercept of a plot of ln(K) vs. 1/T:
Thermodynamic parameters of simultaneous adsorption of dyes on CAC.

K corresponds to the adsorption equilibrium constant according to the best-fitted model, and R is the universal constant of ideal gases, 8.314 J·K−1 mol−1. The negative ΔG° value (Table 5) indicates that CAC simultaneously adsorbs the three dyes spontaneously. The positive ΔHo value (Table 5) means the endothermic nature of the dye mixture (Mahdavinia et al., 2015). If ΔSo is positive, it means that the adsorbent (CAC) is attached to the adsorbate (CV, NBB, and MO), and there are irregularities at the interface (Shah et al., 2021).
Comparison
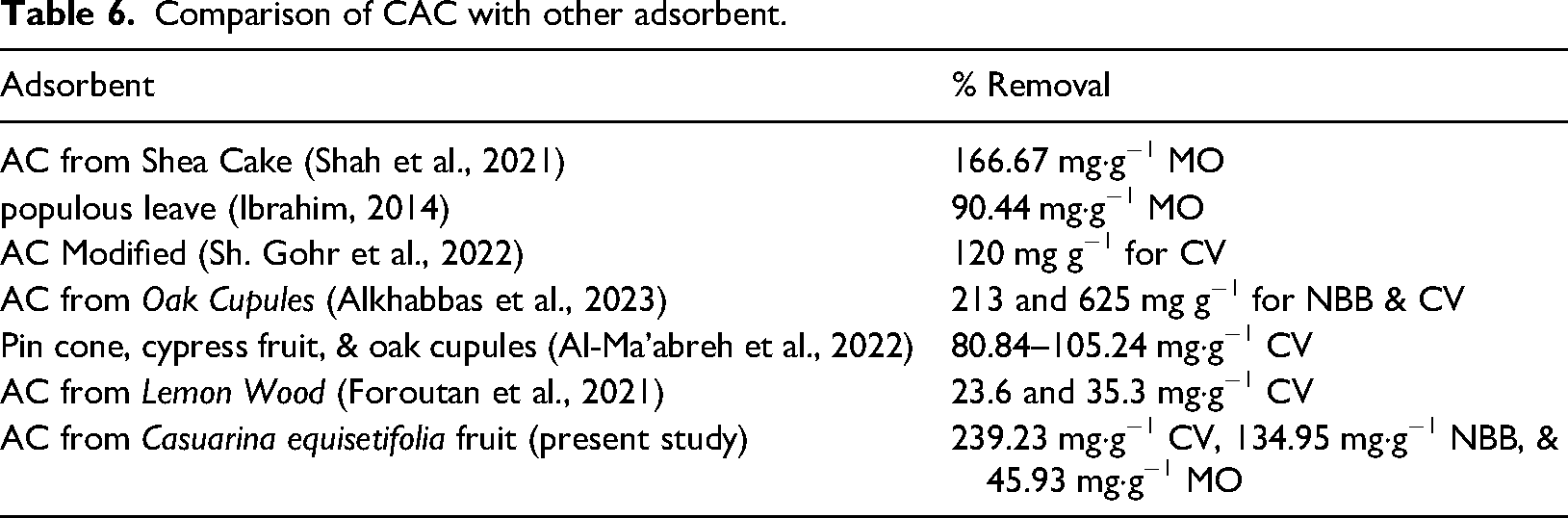
Currently, a great deal of research has been conducted to identify low-cost adsorbents with strong adsorption capability. Table 6 shows the findings of the current study, which looked at how well CAC-adsorbed dyes compare to other natural and activated carbon adsorbents.
Comparison of CAC with other adsorbent.
Conclusion
The study clearly shows that activated carbon from CAC is a highly effective adsorbent for the simultaneous removal of CV, NBB, and MO from aqueous solutions. Comprehensive batch and column adsorption experiments definitively identify the optimal parameters for maximizing the removal efficiency of the dye mixture using CAC. Findings reveal that the shaking time for CV and NBB was 45 min, while MO achieved maximum removal after 60 min. A 0.08 g is the best CAC dosage for the simultaneous removal of the three dyes. Furthermore, the adsorption process operates perfectly within a pH range of 3–11. Casuarina equisetifolia fruit exhibits a stronger affinity for CV compared to NBB and MO. Kinetic and isothermal studies have shown a strong correlation between the adsorption of these dyes by CAC and both the Freundlich isothermal model and the PSO kinetic model. Thermodynamic studies suggest that CAC has an endothermic and spontaneous process for the removal of dyes from the environment. In conclusion, this study provides compelling evidence of the efficacy of CAC as a promising adsorbent for the treatment of wastewater contaminated with dyes. The insights gained from this research can contribute to the development of more sustainable and effective water treatment strategies. Future research may modify the surface of the activated carbon to make it selective for specific pollutants.
Footnotes
Acknowledgements
The authors would like to express their appreciation to Isra University Innovation Center (IUIC) for its vital role in testing the samples. The state-of-the-art infrastructure at IUIC significantly contributed to the accuracy of our findings, and the authors are grateful for their commitment and professionalism.
Authorship
“All authors contributed to the study conception and design. Material preparation, data collection, and analysis were performed by all authors (Alaa Mahmoud Al-Ma’abreh, Dareen A Hmedat, Gada Edris, Fida’ F. Odeh, and Mariam A. Hamed). The first draft of the manuscript was written by Alaa Mahmoud Al-Ma’abreh, and all authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.”
Data availability
The article contains the data that substantiates the results of this investigation.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: “This research was funded by Isra University with grant number of [15-53/2023/2024]”.
Ethical statement
We confirm that this work is original and has not been published elsewhere nor is it currently under consideration for publication elsewhere.