Abstract
Background:
Common mental disorders (CMDs) in older adults contribute to considerable disease burden and are associated with significant multimorbidity. With the increase in population ageing, the concerns related to multimorbidity are likely to increase. Cognitive-behavioural therapy (CBT) is an evidence-based approach for CMDs. More recently, transdiagnostic approaches to address shared mechanisms across disorders have been studied.
Objectives:
To examine the efficacy of a tCBT for older adults with CMDs.
Methods:
A two-group parallel-arm randomised controlled design will be adopted to examine the efficacy of tCBT in older adults with CMDs. A total of 120 older adults (
Expected Outcomes:
There is a dearth of systematic studies in India on psychotherapy in older adults, particularly those that target multimorbidity and transdiagnostic processes such as sleep and RNT. This psychotherapy trial will provide important empirical data on the efficacy of evidence-based tCBT in older adults with CMDs.
Keywords
This is the study protocol for a randomised controlled trial, whose primary objective is to examine the efficacy of transdiagnostic cognitive-behavioural therapy (C) for older adults in improving mood when compared to treatment as usual plus psychoeducation. The secondary objectives of the study are to examine the impact of the intervention on sleep, repetitive negative thinking, metabolic outcomes, and quality of life.Key Messages:
Global projections suggest that nearly 16% of the world’s population will be 60 years or older by 2050. 1 Correspondingly, the prevalence of mental disorders is also expected to rise. 2 In India, the lifetime and current prevalence of depressive disorders are reported to be higher in the elderly than in younger adults, with comparable rates for anxiety disorders. 3 Older adults often receive pharmacological interventions for depression and anxiety, which may be poorly tolerated. Since medication is not sufficient to address the complex health concerns of older adults, there is a growing demand for evidence-based psychological approaches capable of targeting co-morbid conditions. 4 However, there is a dearth of systematic research on the efficacy of evidence-based psychological interventions that address multimorbidity in older adults.
Literature Review
The CMDs, encompassing anxiety, depression, and medically unexplained symptoms, frequently co-occur, with comorbidity rates sometimes reaching 60% in clinical populations. 5 In older adults, CMDs are a significant cause of public health concern, and multimorbidity further impacts outcomes. Global estimates indicate that nearly one-quarter of the overall disease burden is attributable to conditions affecting older adults.6,7 According to the Longitudinal Ageing Study in India (LASI), NCDs, particularly cardiovascular disorders, were found to be highly prevalent in adults aged 60 and above, with the prevalence of depression at 8.3%. 8 High rates of multimorbidity, including non-communicable diseases (NCDs) and pre-existing mental health conditions, pose challenges to adequate health care among older adults, and this is compounded by other difficulties in older adults, such as poorer access to mental health, costs, and length of care. 2
Multimorbidity is a considerable concern in healthcare and suggests shared mechanisms across disorders. Transdiagnostic psychophysiological processes associated with mental health and other NCDs include physical activity levels, sleep patterns, repetitive negative thinking (RNT) (ruminations/worry), maladaptive coping styles, and reduced self-care. The interactions between these variables are well-documented, with older adults being at a greater risk for adverse outcomes, such as suicidality.4,9–12 Interactions between mental and physical disorders also pose a greater challenge as they may go unrecognised or attributed to processes of ageing, even by the older adults themselves, thereby delaying treatment seeking.
Psychosocial factors, including reduced mobility, poor physical health, loneliness, cognitive decline, adverse life events, and role transitions, further contribute to this relationship in older adults. 13 These findings suggest the need to integrate the management of mental health with other NCDs. 14 Pharmacological interventions in older adults are associated with greater side effects and are poorly tolerated, and combining psychological interventions that focus on behavioural activation, mood regulation, and coping skills has greater effects in the long run. 15
Evidence consistently supports cognitive-behavioural therapy (CBT) as an effective psychological treatment for mood disorders and certain chronic health conditions. 16 Meta-analytic reviews in the previous decade indicate that CBT is efficacious in older adults with depression and anxiety17,18 and more recently in insomnia. 19 However, these reviews highlight considerable variability in the quality of studies. Studies on CBT in older adults indicate heterogeneity in approach, delivery settings such as clinics, care homes, and community-based settings, and are disorder-specific without addressing multimorbidity.20–24 Only one controlled trial addressed both anxiety and depression. 25
Until recently, CBT has been studied as a disorder-specific intervention. 26 However, with the recognition of shared mechanisms, interactions between mood, physiological, and metabolic outcomes across disorders, and clinical realities of multimorbidity, transdiagnostic approaches have been proposed. 27
Transdiagnostic interventions, also known as “broadband interventions,” recognise and address multimorbidity. This is particularly relevant in older adults in whom multimorbidity contributes to lower life satisfaction, mobility, and morbidity. Transdiagnostic cognitive-behavioural (tCBT) therapy is a modified, flexible form of CBT designed to apply to patients across the range of anxiety and related emotional disorders. Transdiagnostic approaches to mental health have been considered over disorder-specific approaches based on the understanding of shared mechanisms across emotional disorders, the existence of comorbidities, and a standard elements treatment approach, which is a multi-problem, flexible and adaptable tCBT framework. This approach also acknowledges the high rates of comorbidity, poorer validity of many of the disorders included, and the overlap among disorders. 27 To address these shared mechanisms and underlying processes, tCBT has been considered suitable as it involves a multi-problem/component, flexible approach to problems that co-occur. Core elements of tCBT typically involve psychoeducation, strategies for arousal management, behavioural activation, exposure-based tasks, and cognitive restructuring that is guided by case formulation. These components are based on the understanding of shared mechanisms across emotional disorders/diagnoses and are designed for depression, anxiety, and associated symptoms.28,29 Initial data support the efficacy of tCBT and are particularly relevant for persons with co-morbid diagnoses. The components within tCBT address shared vulnerabilities such as arousal, skills in the regulation of emotions, exposure, and reappraisal or modification of unhelpful beliefs. Emerging research indicates that transdiagnostic methods hold considerable promise for effectively treating comorbidity in psychological disorders.26,29–31
Although substantial evidence supports psychological treatments such as CBT for CMDs in later life—and some findings suggest older adults may respond exceptionally well—systematic psychotherapy research within the Indian context remains limited. 32 There is a dearth of systematic research in India on psychotherapy for older adults. A few case reports, reviews, and preliminary empirical trials are reported from India,33–36 with only one controlled trial examining the prevention of depression among community-dwelling or assisted-living older adults using a brief psychological intervention (behavioural activation) focusing on mood and sleep, as well as education of self-care for physical conditions. 37 Given the multimorbidity and shared mechanisms among disorders, there is a need to establish evidence for transdiagnostic interventions that can be delivered parsimoniously and effectively in older adults in India.
Challenges in the delivery of psychological interventions among older adults remain unanswered, with a dearth of systematic trials on transdiagnostic approaches in higher-income settings as well. 38 Experts recommend that conventional psychological therapies must be adapted to address the needs of older adults, with particular attention to therapeutic processes. 39 Treatment satisfaction and acceptability that impact the uptake and adherence to therapy are less frequently examined.
In the context of these findings, we aim to address some of the research gaps in psychological interventions for older adults in the Indian setting and examine the efficacy of tCBT in this population.
Novelty
Transdiagnostic approaches to health are becoming increasingly relevant. While there is preliminary evidence for the integration of mental health interventions in primary care settings, the inclusion of metabolic outcomes in patients with CMDs is less researched. The key features of the intervention proposed include (a) a transdiagnostic modular approach, (b) a briefer format and option for online delivery, and (c) inbuilt strategies for adherence. The intervention will be co-designed by involving stakeholders and will be structured with consideration for the challenges of engaging older adults in psychological interventions.
Objectives
The primary objectives of this study are to examine the efficacy of a tCBT adapted for older adults in improving symptoms of depression and anxiety. The secondary objectives are to investigate the efficacy of tCBT in reducing RNT, improving sleep quality, quality of life, and metabolic parameters. In addition, treatment satisfaction and the acceptability of the intervention by older adults, as well as predictors of treatment response, will be examined.
Methodology
Study Design
The proposed study is a randomised controlled trial with parallel arms, comprising an intervention arm (tCBT + treatment as usual [TAU]) and a control arm (TAU + psychoeducation). The CONSORT flow diagram, with participant flow, is shown in Figure 1.
CONSORT Diagram.
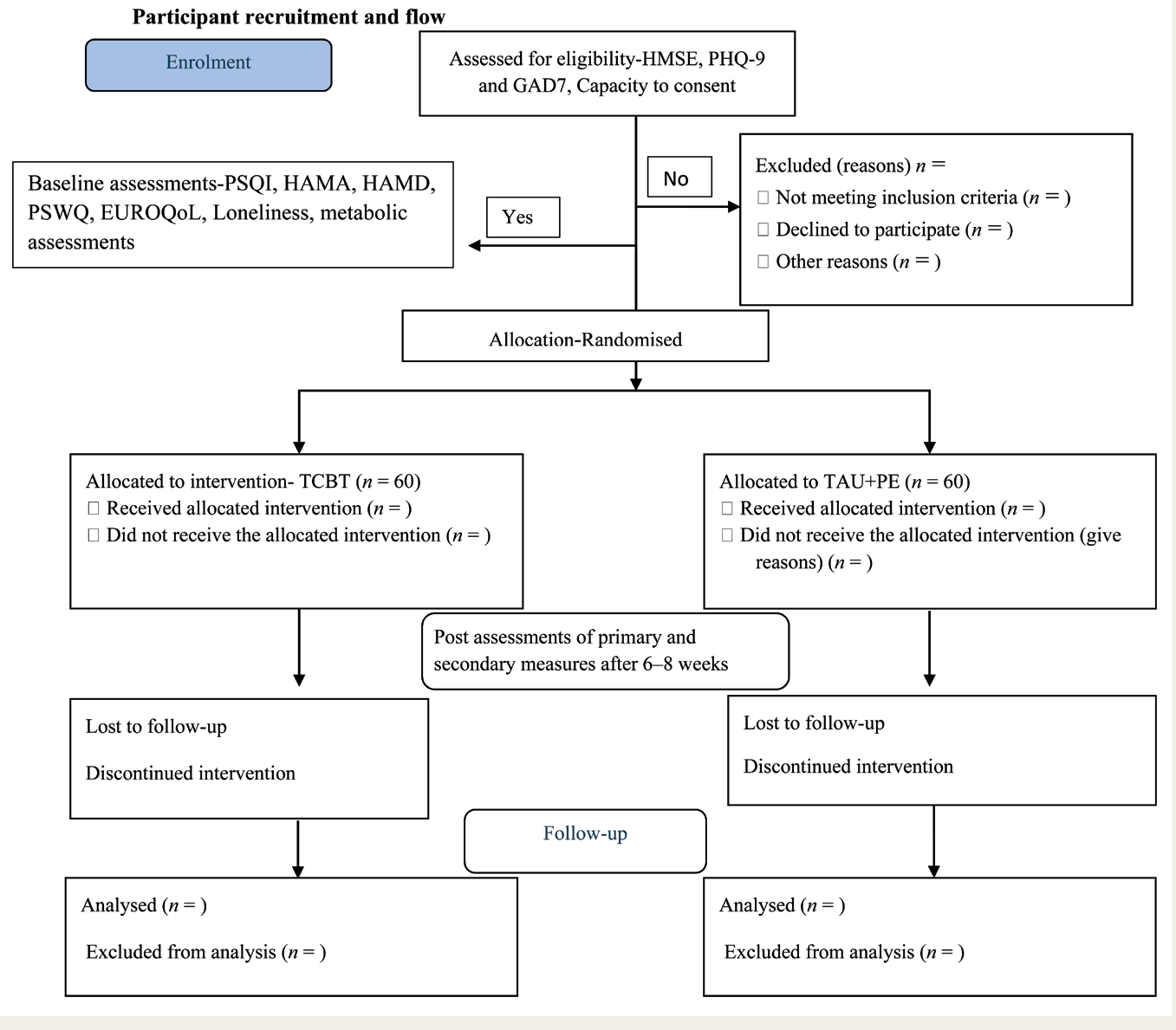
Study Duration and Study Site
The expected duration of the study is three years, encompassing both the adaptation and piloting phases of the intervention. The study will be conducted in the outpatient clinical services of a tertiary care hospital and in outreach settings associated with the hospital, including specialised units for geriatric psychiatry and clinical services that provide psychotherapeutic services within the hospital.
Participants
A total of 120 older adults meeting study criteria will be recruited into the study. Common mental disorders (CMDs) include depression and anxiety disorders. The categories under these include depression, generalised anxiety disorder (GAD), panic disorder, specific phobias, obsessive compulsive disorder (OCD), and CMD not otherwise specified. 40
The inclusion criteria for this study are, aged 55 years and older, meeting criteria for diagnosis under the categories of depressive disorders, anxiety disorders, such as GAD, social anxiety disorder, panic disorder, somatic symptom and related disorders (excluding obsessive compulsive and related disorders) as per DSM-V, 41 PHQ-9 42 scores 5–19 and/or GAD-7 43 scores 5–14 (mild-moderate severity), adequate or corrected hearing and vision, capacity to consent, patients who are on medications for mood/anxiety will be recruited after stabilisation for at least 4 weeks and consent to keep the dose constant during the study (unless there is worsening). The treating clinician will review this. Clinical diagnosis will be made by the treating clinician at the mental health setting and confirmed by the research staff using the appropriate instruments described in the subsequent sections.
Patients with suicidal risk (as determined by screening questions on suicidality on DIAMOND-Diagnostic Interview for anxiety, mood, and OCD and related neuropsychiatric disorders), 44 severe depression, agitation, psychosis, bipolar disorder—currently symptomatic, current substance dependence (other than tobacco), or any other clinical condition which interferes with their participation in psychotherapy or requires specific treatment protocols will be excluded. Patients with significant cognitive impairments and/or any medical/neurological complications/disorders that may interfere with participation in therapy, clinically assessed intellectual or developmental disability, and those who have received CBT for at least 10 sessions or more in the last 6 months will be excluded. All patients will be assessed by the project staff for suitability to participate in the trial meeting.
Sample Size Estimation
The sample size of 120 (
Randomisation
Participants meeting the specified study criteria will be randomised to either a treatment arm or a control arm (tCBT + TAU arm,
Module Development for tCBT
The treatment modules for tCBT will be developed based on stakeholder interviews. These interviews will be used to determine the key areas of focus in older adults, identify treatment needs, and determine session frequency/duration, as well as caregiver involvement. The extent of blending of face-to-face and telephonic/online interventions will also be discussed. Stakeholders (
Interventions
Intervention Arm (tCBT + TAU)
The intervention will be a tCBT, designed to address shared symptom presentations and underlying mechanisms that include arousal, negative thought patterns, emotion-driven behaviours, low mood, and sleep/activity disturbances across CMDs (Table 1). 29 Components required to address medical morbidities that older adults present with, and problems such as pain and sleep disturbances, will be integrated. Cognitive-behavioural interventions for sleep, pain, and lifestyle disorders include arousal reduction, exposure, behavioural activation, and belief modification. Based on the case conceptualisation, the content of sessions will be modified to address these concerns, while retaining the overall framework and techniques. Sleep hygiene will be discussed in both groups. The PI is a senior clinical psychologist with a specialisation in CBT. Additionally, two of the coinvestigators are also senior clinical psychologists and experts in CBT, and the COPI is a faculty clinical psychologist specialising in geriatric mental health.
Psychoeducation will be contextualised based on a functional analysis. Common components that will be included are presented in Table 1. Treatment components will be delivered in a modular fashion and personalised based on case conceptualisation. The estimated range of sessions is six to eight sessions delivered over 2–2.5 months. The final range of sessions will be assessed following the discussions with stakeholders. As older adults may find it challenging to attend face-to-face sessions, requiring a caregiver/family member to accompany them, a blend of face-to-face and telephonic/online sessions will be offered. Online sessions will be considered for those patients who are unable to attend face-to-face sessions and will follow appropriate guidelines for online psychotherapy. Considering the potential challenges that older adults may face when using online platforms, therapists will first assess their comfort level with online platforms before recommending their use. Wherever necessary, caregivers/significant others will be requested for assistance. The details of how these sessions are delivered will be documented, and any other information regarding the online and in-person sessions will be noted. The intervention group will continue to be on pharmacotherapy, with stabilisation of a minimum of four weeks prior to recruitment.
Proposed Components of the Transdiagnostic CBT Programme.

Control Group (TAU + Psychoeducation)
TAU will include standard pharmacotherapy for CMDs and a psychoeducation session that will cover the nature of the problems being experienced, the importance of physical activity and its relation to mood, self-care, and information on sleep hygiene practices.
Treatment Engagement Strategies
Between sessions, tasks (homework) will be provided, based on the module being addressed, along with worksheets and materials. Keeping in mind the heterogeneity in educational status and challenges in writing, the formats will be kept flexible and straightforward. Telephonic/online sessions will be introduced to enhance engagement and facilitate greater participation in the intervention. A caregiver will be involved when required, specifically for modules on enabling activities and functioning.
Treatment Fidelity/Quality and Adherence
The intervention will be provided by research staff recruited for the study. The research staff will comprise qualified clinical psychologists with an RCI-recognised Master’s degree in Clinical Psychology. A description of the components will be made available to ensure that the module delivery is uniform across all therapists. Therapists (research staff) will be supervised by senior members of the research team who are experts in CBT and ratings. Session ratings will be based on a random selection of recordings and notes. The fidelity checklist will be prepared based on the broad components of the final therapeutic programme, including providing psychoeducation rationale for procedures such as arousal reduction, use of self-monitoring, and homework. This will be further refined following the finalisation of the treatment programme.
Completers and Dropouts
Completers are defined as patients who complete 80% of the intervention and for whom post-intervention assessment is available. Patients for whom post-intervention data are not available after randomisation will be considered as dropouts.
Outcomes
Study Tools and Assessment Time Points (Table 2 SPIRIT Guidelines)
SPIRIT Diagram (Standard Protocol Items for Reporting Clinical Trials) for the Schedule of Enrolment, Interventions, and Assessments Schedule for Enrolment Into Study, Intervention, and Assessment Points.
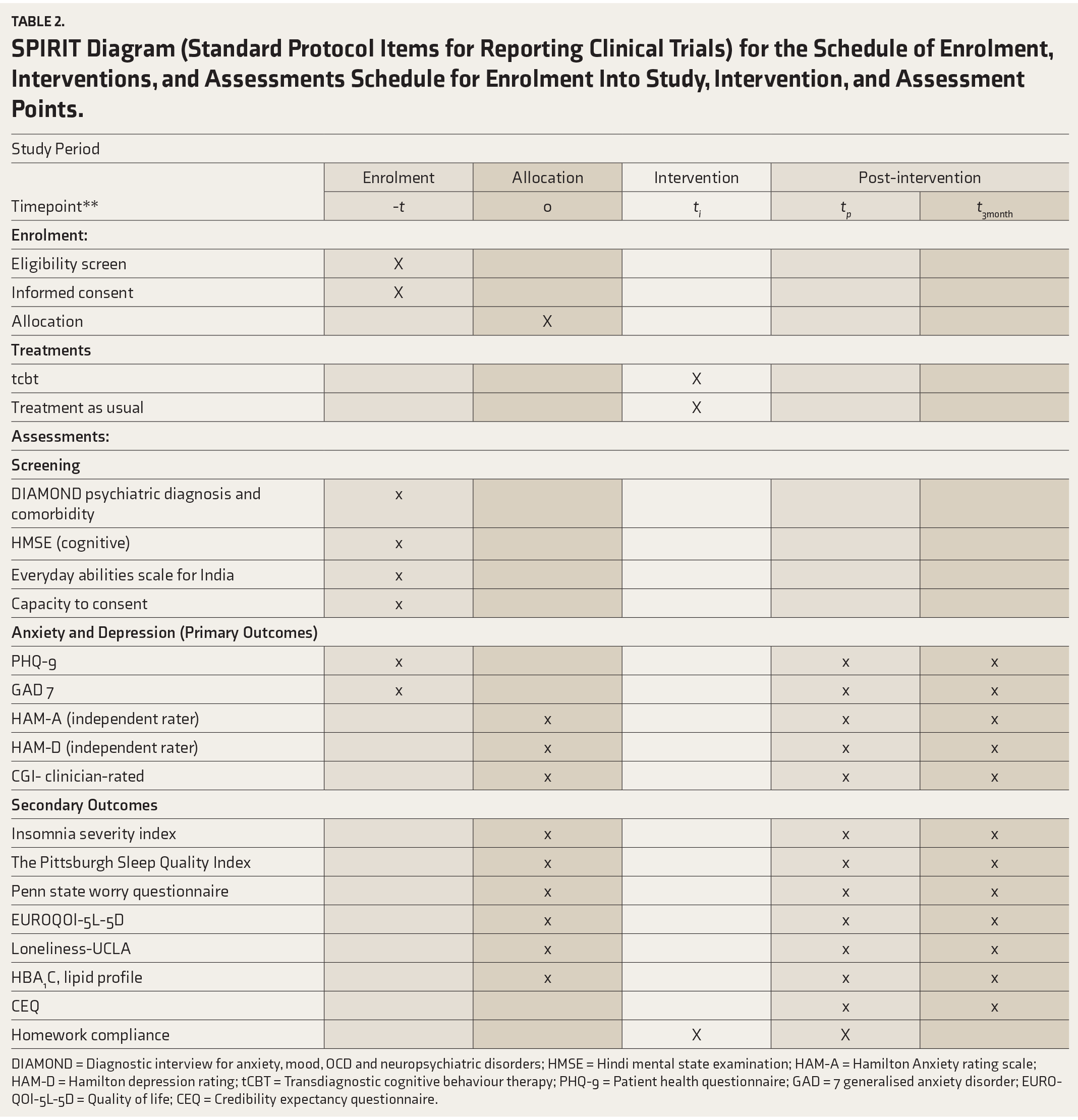
DIAMOND = Diagnostic interview for anxiety, mood, OCD and neuropsychiatric disorders; HMSE = Hindi mental state examination; HAM-A = Hamilton Anxiety rating scale; HAM-D = Hamilton depression rating; tCBT = Transdiagnostic cognitive behaviour therapy; PHQ-9 = Patient health questionnaire; GAD = 7 generalised anxiety disorder; EUROQOl-5L-5D = Quality of life; CEQ = Credibility expectancy questionnaire.
Initial screening: A structured clinical interview will be conducted to assess the patient, including their current psychiatric and medical status, comorbidities, and physical health parameters, based on the WHO STEP-wise approach to surveillance of non-communicable risk factors (STEPS). 45
The physical and physiological health parameters will include blood pressure, anthropometric data (height, weight, body mass index [BMI]), and the physical activity levels. They will be assessed by a staff nurse recruited for the study. All these will be measured at baseline, post-intervention, and follow-up points.
Assessment of Capacity and Cognitive Functioning
Hindi Mental Status Examination 46
A widely used global cognitive screen developed for older Indian adults will be administered to identify and screen patients for cognitive impairment. A cut-off score of 23 or below will be considered an indicator of possible cognitive impairment.
Capacity to Consent
The University of California, Irvine 47 (UCI) capacity to consent form will be administered to determine the capacity to consent to participate in the intervention.
Everyday Abilities Scale for India (EASI) 48
The EASI is a 12-item brief tool that assesses activities of daily living across four domains. These include self-care, mobility, social interactions, and cognitive function. The measure is suitable for use in evaluating older adults, particularly those with dementia, in India.
Primary Outcome
Patient Health Questionnaire-9 (PHQ-9) 42
The PHQ-9 is a self-report instrument that can be used for multiple purposes, such as screening, measuring, and monitoring the severity of depression, and includes an item identifying suicide ideation and major depression. The PHQ-9 is both sensitive and specific in its diagnoses and is used in clinical and research settings. In the present study, a cut-off of 5–19 (mild to moderate) will be considered as eligibility for recruitment into the trial. The PHQ-9 is available in local Indian languages.
The GAD Scale-7 (GAD-7) 43
The GAD-7 is a 7-item, self-rated instrument widely used for screening, measuring, and monitoring GAD severity. The scale has been evaluated in both primary care and psychiatric samples. A cut of 5-15 (mild to moderate severity) will be considered for the present study. The GAD-7 is available in local Indian languages.
Hamilton Anxiety Rating Scale (HAM-A) 49
The HAM-A is a clinician-rated scale, with 14 items used to assess the severity of anxiety. It is widely used in research to evaluate clinical outcomes. In the present study, the severity of anxiety symptoms will be measured by an independent blind rater.
Hamilton Depression Rating Scale (HAM-D) 50
The HAM-D is a 17-item clinician-rated scale used to assess the severity of depressive symptoms. It is widely used in research to determine the clinical outcome. In the present study, the severity of anxiety symptoms will be assessed by a blind rater.
Secondary Outcomes
Clinical Global Impression Scale (CGI) 51
The CGI is a three-item, 7-point clinician-rated scale that assesses response to treatment. Items 1 and 2, namely, Severity of illness and Global Improvement, will be used in the study. The CGI-I will be used as a secondary outcome measure for improvement.
Assessment of Sleep and RNT
The Pittsburgh Sleep Quality Index 52 (PSQI)
The 19-item PSQI will be used to assess sleep quality in the past month. It has seven components, including subjective sleep quality, latency, duration of sleep, sleep efficiency, sleep disturbances, sleep medication usage, and daytime dysfunction. Higher scores on the PSQI indicate poorer overall sleep quality.
Insomnia Severity Index 53
This is a 7-item self-report measure of the nature, severity, and impact of insomnia, in the “last month.” The scale assesses parameters of insomnia and the problems associated with it, including daytime functioning.
Penn State Worry Questionnaire (PSWQ) 54
The PSWQ is a 16-item self-report measure and will be administered to assess the frequency, pervasiveness, and other features of ruminative/RNT.
Metabolic Outcomes
The glycated haemoglobin (HbA1c) and Lipid profiles will be assessed at baseline, 3 months (post-intervention), and 3-month follow-up points. Samples will be collected by a nurse who will be recruited for the study. The nurse will also carry out measurements of blood pressure, height, and weight.
UCLA 3-item Loneliness Scale 55
This scale consists of three items that measure three dimensions of loneliness: relational and social connectedness, and self-perceived isolation.
EUROQOl-5L-5D 56
It will be administered to assess the quality of life across various domains, including mobility, self-care, usual activities, pain/discomfort, depression, anxiety, and respondents will be asked to rate their best/worst health on a visual analogue scale. The measure is licensed and will be obtained for use in the study. The EUROQol-5L-5D is available in local Indian languages.
The Homework Compliance Scale 57
The HCS is a two-item scale that assesses the degree and quality of homework compliance in CBT and will be administered by the therapists.
The Credibility/Expectancy Questionnaire 58
The CEQ is a 6-item measure with two subscales: treatment rationale credibility (CEQ-C), which is a cognitive item, and treatment expectancy (CEQ-E), which is based on emotions. The CEQ demonstrates adequate psychometric properties of test-retest reliability and internal consistency.
Self-report measures that are not available in local languages will be translated and back-translated using standard procedures, and items will be read out to the patients. These measures have been utilised in clinical populations within the Indian setting.
Implementation Plan
Consecutive patients attending the geriatric services and other outpatient mental health settings of a tertiary mental health centre in southern India will be screened for eligibility. Those fulfilling the study criteria will be approached by the research staff for consent to participate, who will also provide therapy. For those consenting to participate, baseline assessments on primary and secondary outcome measures (Table 1) will be completed. Following this, participants will be allocated to either the intervention arm (tCBT + TAU) or the control group (TAU + psychoeducation), based on the randomisation sequence.
Outcomes
The primary endpoint of the study would be after the intervention. For both groups, the primary outcomes (PHQ-9, HAM-D, GAD-7, HAM-A) will be assessed at baseline, post-intervention, and at 1- and 3-month follow-ups. This would enable us to evaluate the treatment response over time and its durability. Independent assessors, blind to the group allocation, will conduct outcome assessments on primary outcomes. Secondary outcomes include RNT (characterised by worry and rumination), sleep quality and insomnia severity, metabolic outcomes (HbA1c and Lipid profile), and quality of life (Table 1). The feasibility and acceptability of the intervention would be determined based on rates of recruitment, retention, and treatment protocol completion. The patient’s report on the measure of CEQ and homework compliance would be assessed by the therapist using the Homework Compliance Scale.
The assessment of cognitive functions and suitability to participate will be carried out in person. Only if this is not feasible, it will be conducted telephonically/remotely by the independent assessor, who will be blind to the participant’s allocation to either study arm.
Data Analysis
Data will be entered for the application of statistical software packages (SPSS or R software). Baseline characteristics of the two groups will be compared using descriptive statistics,
Ethical Considerations
The Institute’s Ethics Committee has approved the study and is prospectively registered with the Clinical Trials Registry of the Indian Council of Medical Research (ICMR).
Informed consent will be obtained from participants by the research staff recruited for the study. Participants’ capacity to consent to the intervention will be assessed before recruitment. They will be briefed about the nature of the study and randomisation into either arm of the study. Those requiring medication changes during the intervention will be noted and analysed separately.
Data Monitoring and Safety
A data safety and monitoring board (DSMB) will be established for the trial. The DSMB will meet periodically to review the data and address safety concerns related to the trial.
Discussion
CMDs, such as depression and anxiety, contribute considerably to the worldwide burden of disease. 59 Findings from the National Mental Health Survey (NMHS) suggest a treatment gap of about 85% for mental disorders in India, due to both demand-side and supply-related barriers. 60 In older adults, the presence of CMDs is a public health concern that is further compounded by multimorbidity. CBT is considered to be the treatment of choice for CMDs. 16 The evidence for shared mechanisms across disorders has led to the development of transdiagnostic approaches to health.
Empirical research in India on psychotherapy for older adults, specifically transdiagnostic interventions, addressing multimorbidity, is limited. 15 Our review of relevant literature suggests that there are no systematic, published trials documenting evidence for psychotherapy in the Indian population, particularly the elderly. Research indicates that older adults respond better to psychological interventions than other age groups. 32 In this study, we will examine key transdiagnostic processes implicated in the risk and maintenance of CMDs, namely RNT, sleep, and loneliness. Addressing these targets is particularly relevant in older adults in whom physical health outcomes contribute to lower life satisfaction, mobility, and morbidity. While there is some evidence in primary care settings, evidence for similar approaches in mental health settings is lacking. 61 This study will be an essential contribution to building evidence for transdiagnostic psychological interventions for the older adult Indian population. Treatment satisfaction and acceptability of psychological interventions are essential process variables that we will be exploring, in addition to adherence.
The study has certain inherent limitations. These include the age range and clinical diagnosis, which may render the sample heterogeneous with respect to the concerns they present with. We do not expect a significant difference in response to and participation in psychotherapy, although processes may differ and this will be documented to the best possible extent. We considered this age range based on the definitions of older adults, and the understanding that many existing studies exclude people above 55, due to the same reasons. We have included assessment of cognitive functions and comorbidities and will report these as part of the study analysis. Clinical presentations of anxiety and depressive disorders suggest high rates of comorbidity and this trial attempts to address these using the transdiagnostic approach as recommended in recent literature. Although conceptualisations of tCBT vary, most frameworks emphasise five components: psychoeducation, emotion regulation strategies, activation tasks, exposure techniques, and cognitive restructuring.
These limitations notwithstanding, a significant contribution of this trial would be a therapy protocol for the effective replication and dissemination of the intervention, specifically designed for older adults. To our knowledge, systematic psychotherapy trials in older adults in the Indian context are limited. Research on psychological interventions for older adults indicates that many of these are often without significant adaptation or change. The study aims to address this research gap by employing a systematic method of adapting psychological interventions to the needs of older adults before empirical testing.
Expected Outcomes
We will be able to operationalise a tCBT module that can be delivered for older adults with CMDs and multimorbidity in the Indian setting. This is especially important as a large number of older adults present with multimorbidity, and there is a dearth of systematic studies on the efficacy and acceptability of transdiagnostic psychological interventions in this population. The deliverables will be documentation of the practical application and components of the intervention for replication and further testing. If we can demonstrate the effectiveness of this tCBT intervention, it can be tested further with full versions of the intervention and recommended as part of clinical care for both medical and psychiatric conditions.
Trial Status
Phase I of the trial, which involved stakeholder interviews and intervention adaptation, is now complete, and the trial is open to recruitment and randomisation of participants.
Supplemental Material
Supplemental material for this article available online.
Footnotes
Acknowledgements
We acknowledge the support of the Indian Council for Medical Research (ICMR), New Delhi, in carrying out this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Declaration Regarding the Use of Generative AI
Grammarly, an AI-based writing tool, was used to assist in grammar checking, spelling corrections, and improving the clarity of the manuscript. The authors assume full responsibility for the entire content of the manuscript, including the parts refined with the assistance of Grammarly.
Ethical Approval
The study has been approved by the Institute Ethics Committee and is registered with the Indian Council of Medical Research (ICMR) Clinical Trials Registry (CTRI/2024/03/064480).
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This protocol article describes the methodology of a project funded by the Indian Council for Medical Research (ICMR), New Delhi.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
