Abstract
Background:
Inflammatory cytokines are associated with the pathophysiology of major depressive disorder (MDD). Nevertheless, whether they are trait or state biomarkers remains unclear. We aimed to assess and compare selected cytokines, miRNAs, and oxidative stress biomarker levels between patients with MDD, first-degree relatives (FDRs), and unrelated healthy controls (UHCs).
Methods:
Using a cross-sectional design, we recruited patients with MDD, their FDRs and age, gender, body mass index-matched UHCs. Serum levels of IL-6, IL-1β, TNF-α, hs-CRP, and IL-10 were evaluated using the Enzyme-Linked Immunosorbent Assay (ELISA) method. We quantified the gene expressions of plasma miR-16, miR-132, and miR-1202 using Quantitative Real Time-Polymerase Chain Reaction (qRT-PCR). We estimated the total oxidant status and total antioxidant status levels in the serum by ELISA. We used sequential multiple linear regression analysis to find the association between MDD vs. UHCs (model 1), FDRs vs. UHCs (model 2), and MDD vs. FDRs (model 3) after adjusting for age, gender, and BMI as covariates.
Results:
The study involved 50 patients with MDD (mean age = 34.7 ± 10.1 years), 31 FDRs (mean age = 32.7 ± 12.2 years), and 50 UHCs (mean age = 34.1 ± 10.3 years). Patients with MDD had higher levels IL-6 (β = 6.04; 95% CI; 1.60 to 10.49; p = 0.008) and downregulated miR-1202 (β = –0.38; 95% CI: –0.70 to –0.06; p = 0.02) compared to FDRs with statistically significant differences.
Conclusion:
The higher levels of serum IL-6 and downregulated plasma miR-1202 expression in patients with MDD compared to FDRs may be a state biomarkers in MDD. However, an adequately powered study can answer these questions with certainty.
Keywords
This study assessed IL-6 as trait or state biomarker in patients with MDD. We observed elevated IL-6 and downregulated miR-1202 in patients with MDD. IL-6 and miR-1202 may be state biomarkers patients in with MDD. Biomarkers may aid in diagnosis, severity prediction, and treatment monitoring in MDD.Key Messages:
Major depressive disorder (MDD) is a common mental illness that affects about 350 million people globally. In India, about 46 million individuals experience depressive symptoms. 1 MDD is a complex disorder influenced by familial, psychological, chronic diseases, genetic, epigenetic, and immune systems. Recent meta-analyses of controlled clinical trials indicate antidepressants have remission rates ranging from 30% to 50%, with one-third of patients showing no response. Subjective diagnosis of MDD increases the risk of misdiagnosis, ineffective treatment, and higher healthcare costs, impacting work, social responsibilities, and the well-being of individuals and their family members or caregivers.2,3 Identifying biomarkers for objective diagnosis is crucial for clinical practice and research.
A biomarker (e.g., diagnostic, prognostic, predictive, trait, and state biomarkers) indicates a disease or medical condition that can be measured. Trait biomarkers exhibit augmented levels before onset, during progression, and after disease remission. Measuring trait biomarkers helps to identify those susceptible to disease development, predict early diagnosis, minimize poor remission rates, and lessen the burden on the economy and quality of life. State biomarkers represent the status of clinical manifestations in patients, reflecting active changes in the disease course and alleviating after remission. Measuring these biomarkers can improve diagnostic accuracy, predict treatment responses, and manage treatment progression. 4
In recent years, the inflammatory hypothesis has gained popularity for explaining MDD. Studies have found that patients with MDD have higher levels of inflammatory cytokines such as interleukin-6 (IL-6), interleukin-1 beta (IL-1β), tumor necrosis factor-alpha (TNF-α), interferon-gamma (INF-γ), interleukin-2 (IL-2), and C-reactive protein (CRP) than healthy controls. Environmental factors (e.g., childhood trauma, chronic stress, loss of loved ones, and financial problems) and genetic factors (e.g., functional gene polymorphisms of IL-6, TNF-α, and IL-1β) are involved in the activation of inflammatory pathways, elevated oxidative stress, and cytokine production, which ultimately lead to MDD. 5 Furthermore, data from previous meta-analyses suggest that antidepressants can potentially lower levels of these inflammatory cytokines. 6 The changes in molecular levels owing to treatment allow us to understand the pathophysiological changes in patients with MDD.
MicroRNAs (miRNAs) function as endogenous controllers that precisely regulate and control gene expression activation and inactivation. Recent clinical studies suggest that miRNAs also impact responses to pharmacotherapy. Multiple pieces of evidence indicate that miRNAs play a crucial role in its development, progression, and response to antidepressants. Research reveals a significant downregulation of miR-16, miR-132, and miR-1202 in patients with MDD, which normalizes in response to antidepressant treatment. Additionally, miRNAs regulate genes associated with cytokine secretion, controlling the regulation of IL-6, TNF-α, and IFN-γ and producing inflammatory cytokines, which may influence the condition or onset of disease.7,8
Oxidative stress is a state in which the production of reactive oxygen species exceeds the ability of cells to eliminate them and is significant among individuals suffering from MDD. Previous studies indicate that patients with MDD have higher total oxidant status (TOS) levels and decreased total antioxidant status (TAOS) than healthy controls. 9 Few studies have found no differences in oxidative stress markers or antioxidant levels after 4 to 8 weeks of antidepressant treatment in patients with MDD. Researchers have discovered a connection between oxidative stress and inflammation. Increased oxidative stress may activate the hypothalamic-pituitary-adrenal (HPA) axis and immune-inflammatory pathway, thereby increasing the IL-6, TNF-α, and IL-1β levels. Elevated IL-6 concentrations may decrease the total antioxidant capacity (albumin, zinc, and high-density lipoproteins), triggering depressive symptoms. 10
Against this background, in the present study, we compared and contrasted the levels of inflammatory cytokines (IL-6, IL-1β, TNF-α, hs-CRP, and IL-10) in drug-naïve, first-episode depressed outpatients with first-degree relatives (FDRs) and unrelated healthy controls (UHCs). Other Indian studies, tried to find the association between inflammatory cytokines and major depression compared with healthy controls. However, no other studies have attempted to find an influence of genetics on inflammatory cytokines and MDD by adding FDRs group in their studies.11-13 Despite extensive research on these biomarkers in patients with MDD, the results remain uncertain. Then, we assessed the role of selected miRNAs (miR-16, miR-132, and miR-1202) in the pathophysiology of MDD. We also explored the roles of TOS and TAOS in patients with MDD and compared them with healthy controls. 14 We hypothesized that inflammatory cytokine levels, miRNA expression, and oxidative stress markers would differ between patients with MDD and healthy controls.
Materials and Methods
Design and Settings
This cross-sectional study was conducted in the psychiatric outpatient department, JIPMER, Puducherry, a tertiary care hospital, from October 2021 to December 2022. The study commenced after receiving approval from the Institutional Ethics Committee for Human Studies (JIP/IEC/2019/108). Written informed consent was obtained from all participants following a detailed explanation of the procedures in their preferred language (Tamil, English, or Hindi). The Strengthening the Reporting of Observational Studies in Epidemiology guidelines were used to ensure optimal reporting.
Patient Population
We recruited 50 consecutive drug-naïve first-episode patients diagnosed with MDD as per the Diagnostic and Statistical Manual of Mental Disorders, 5th edition (DSM-5) criteria, 15 aged between 18 and 65 years. The diagnosis of MDD was further confirmed using the Structured Clinical Interview for the DSM-5-Research Version (SCID-5-RV). 16 Severity of depression in these patients was assessed utilizing the Hamilton Depression Rating Scale (HDRS) 17 ; we included individuals with baseline HDRS scores of ≥14. Patients with concurrent psychiatric disorders, steroid use, hyperthyroidism or hypothyroidism, nonsteroidal anti-inflammatory drugs, other immune modulators, substance use disorders, and pregnant or lactating women were excluded from the study. Thirty-one FDRs accompanying patients diagnosed with MDD to the psychiatry outpatient department were recruited as control 1. We utilized the modified mini screen (MMS), a screening tool designed to identify and exclude anxiety, mood, and psychotic symptoms, 18 and other medical comorbidities through personal interviews. Inclusion criteria comprised parents, siblings, sons, and daughters of patients with MDD aged 18 years and above. Exclusion criteria involved a family history of hereditary neurological disorders, hypertension, diabetes, liver or kidney impairment, substance use disorders, and pregnant and lactating women. Matching with cases was not performed. We enrolled 50 UHCs matched in terms of age (±5 years), gender, and body mass index (BMI) (±3 kg/m²) as control 2. These controls included hospital staff, friends, and attendees from other outpatient departments, ensuring that they had no blood relation to the cases. The screening method and exclusion criteria were identical to those for FDRs. We computed the sample size for all five biomarkers (IL-6, IL-1β, TNF-α, hs-CRP, and IL-10) based on the expected mean difference and standard deviations between patients with MDD and healthy controls. Calculations indicated the highest values for IL-6, assuming a mean difference of 12, with standard deviations 1 = 28; standard deviations 2 = 13; α-error = 5%; power = 80%. 19 This resulted in approximately 50 individuals per group. Consequently, the final sample size was set at 50 patients with MDD, 50 healthy FDRs, and 50 UHCs.
Assessments Done
The study assessed the serum levels of IL-6, IL-1β, TNF-α, IL-10, and hs-CRP using commercially available Enzyme-Linked Immunosorbent Assay (ELISA) kits. Reagent preparation, standardization, and control procedures followed the manufacturer’s instructions. The values for inflammatory cytokine were expressed in pg/ml and for hs-CRP in mg/L. Total RNA was extracted from plasma samples for gene expression analysis using commercially available kits. The RNA was then converted to cDNA via polymerase chain reaction. The absolute quantification of miR-16, miR-132, and miR-1202 was done using the quantitative real-time polymerase chain reaction method, and the quantified miRNAs were expressed in copy numbers. Serum TOS and TAOS were estimated using the ELISA method. Reagent preparation, standardization, and control procedures followed the manufacturer’s instructions. TOS results were expressed in µmol H2O2 Equiv./L, while TAOS results were expressed in mmol Trolox Equiv./L.
Statistical Analysis
Data were analyzed using Statistical Package for Social Sciences (SPSS) for Windows, version 22 (IBM Corp., Armonk, N.Y., USA). Categorical variables were expressed as frequencies and percentages. Continuous variables were expressed as mean ± standard deviation (SD) or median with interquartile range. The normality of the data was assessed using the Shapiro–Wilk test. Pairwise comparisons between the groups were made using a one-way ANOVA or Kruskal–Wallis one-way ANOVA test, depending on their normality.
We used multiple linear regression analysis to compare groups and ascertain the trait versus state marker status of inflammatory cytokines (IL-6, IL-1β, TNF-α, hs-CRP, and IL-10), miRNAs (miR-16, miR-132, and miR-1202), TOS, and TAOS levels while controlling for confounding factors such as age, gender, and BMI. We performed three sequential regression analyses for each biomarker; running successive models in the sequence was contingent upon the results of the previous model.
The initial regression model (model 1) compared patients with MDD versus UHCs, with FDRs excluded from the analysis. This regression analysis aimed to establish assay sensitivity and check whether patients with MDD separated from UHCs. We did not pursue further regression analyses for the marker under investigation if it did not exhibit discriminatory power between patients with MDD and UHCs. The covariates in this model were age, gender, and BMI.
The second regression analysis (model 2) intended to identify the selected biomarker’s trait marker status. To achieve this, we compared FDRs and UHCs; the MDD group was not included in this analysis. If FDRs separated from UHCs, then we concluded that the marker in question serves as a trait biomarker for patients with MDD. Age, gender, and BMI were included as covariates in this model.
The third regression analysis (model 3) intended to identify the selected biomarker’s state marker status. To achieve this, we compared patients with MDD versus FDRs; the UHC group was not included in this analysis. If patients with MDD separated from FDRs, we concluded that the marker in question serves as a state marker for MDD. Age, gender, and BMI were included as covariates in this model.
Each model met the assumptions of regression analysis. All statistical analyses were done at a two-sided 95% confidence interval, and P < .05 was considered statistically significant.
Results
Comparison of Sociodemographic and Clinical Parameters Between Cases and Controls
Table 1 illustrates the distribution of baseline sociodemographic data among cases and controls. Patients with MDD had a mean age of 34.7 ± 10 years, FDRs had a mean age of 32.1 ± 12 years, and UHCs had a mean age of 34.1 ± 10 years. Female participants constituted a higher proportion of the sample (29 [58%]). Patients with MDD had a BMI of 23.8 ± 4.1, FDRs had a BMI of 22.9 ± 3.3, and UHCs had a BMI of 24.3 ± 3.1.
Sociodemographic Data and Clinical Measures Between the Patients with Major Depressive Disorder and Healthy Controls.
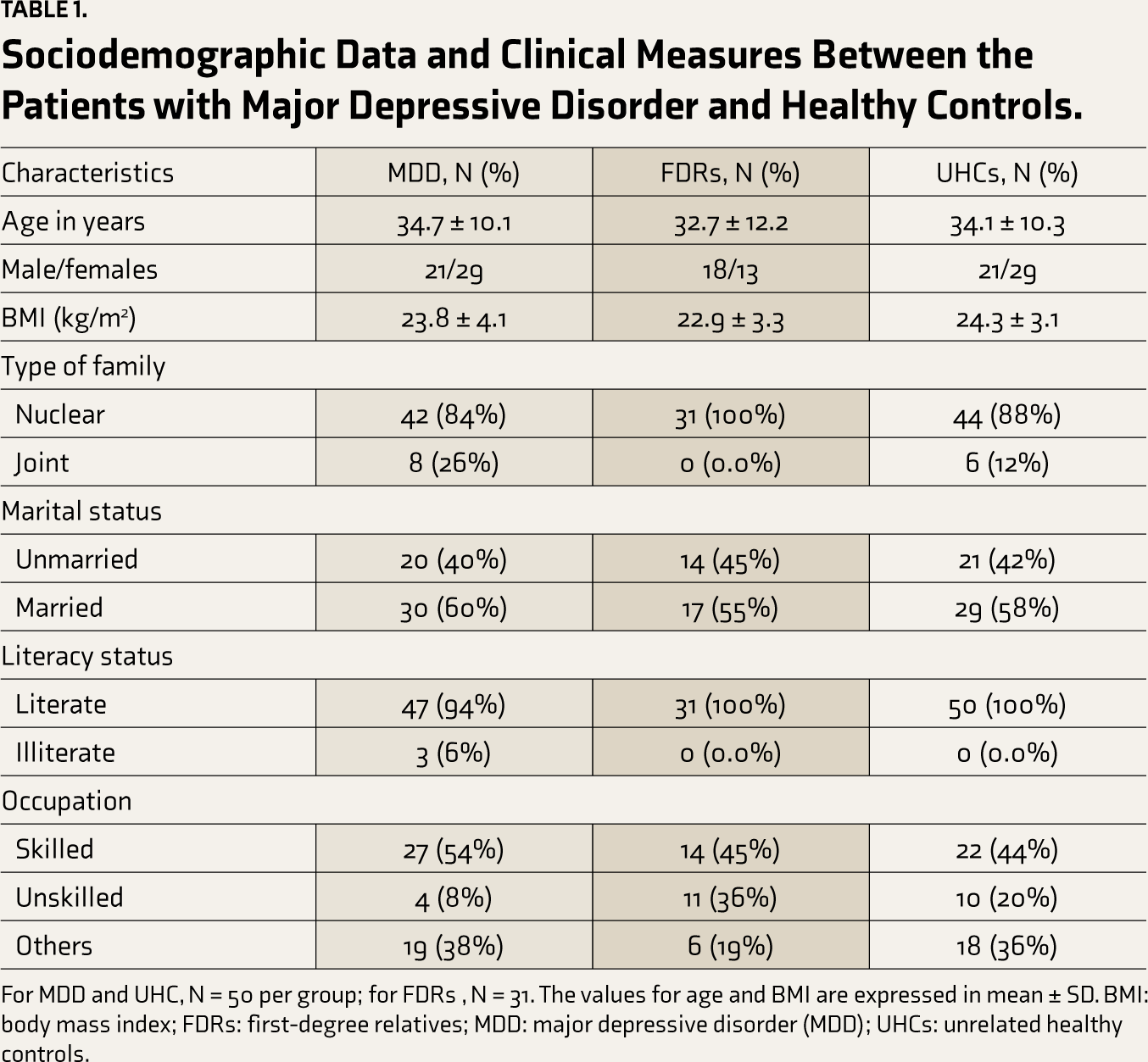
For MDD and UHC, N = 50 per group; for FDRs, N = 31. The values for age and BMI are expressed in mean ± SD. BMI: body mass index; FDRs: first-degree relatives; MDD: major depressive disorder (MDD); UHCs: unrelated healthy controls.
Estimation of Serum Inflammatory Cytokines Between the Cases and Controls
The study compared the levels of IL-6 in cases and controls, and the levels were higher in patients with MDD [3.5 (1.9, 6.7)] than FDRs [1.8 (1.2, 3.0 and UHCs [3.3 (1.9, 4.1)]. However, statistically significant differences were found between patients with MDD and FDRs (P < .001) and between FDRs and UHCs (P < .05). There was no significant difference between patients with MDD and UHCs. Additional details are provided in Table 2.
Estimating the Serum Levels of Inflammatory Cytokines, Plasma Micro RNAs and Total Oxidant and Total Antioxidant Status Among Patients with Major Depressive Disorder and Healthy Controls.
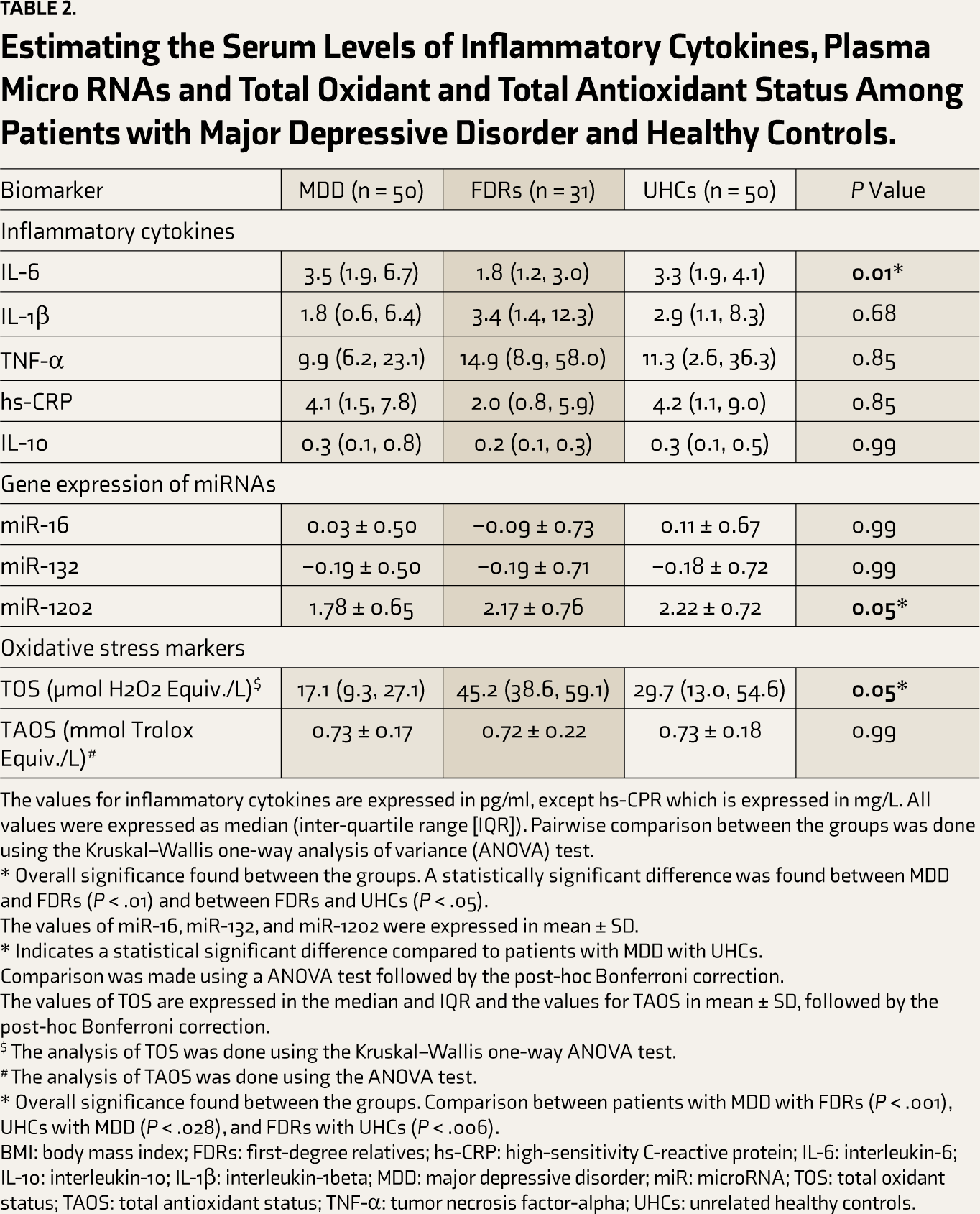
The values for inflammatory cytokines are expressed in pg/ml, except hs-CPR which is expressed in mg/L. All values were expressed as median (inter-quartile range [IQR]). Pairwise comparison between the groups was done using the Kruskal–Wallis one-way analysis of variance (ANOVA) test.
* Overall significance found between the groups. A statistically significant difference was found between MDD and FDRs (P < .01) and between FDRs and UHCs (P < .05).
The values of miR-16, miR-132, and miR-1202 were expressed in mean ± SD.
Comparison was made using a ANOVA test followed by the post-hoc Bonferroni correction.
The values of TOS are expressed in the median and IQR and the values for TAOS in mean ± SD, followed by the post-hoc Bonferroni correction.
$ The analysis of TOS was done using the Kruskal–Wallis one-way ANOVA test.
# The analysis of TAOS was done using the ANOVA test.
* Overall significance found between the groups. Comparison between patients with MDD with FDRs (P < .001), UHCs with MDD (P < .028), and FDRs with UHCs (P < .006).
BMI: body mass index; FDRs: first-degree relatives; hs-CRP: high-sensitivity C-reactive protein; IL-6: interleukin-6; IL-10: interleukin-10; IL-1β: interleukin-1beta; MDD: major depressive disorder; miR: microRNA; TOS: total oxidant status; TAOS: total antioxidant status; TNF-α: tumor necrosis factor-alpha; UHCs: unrelated healthy controls.
Gene Expression of Plasma miR-16, miR-132, and miR-1202 Among MDD, FDRs, and UHCs
The miR-16, miR-132, and miR-1202 expression levels were compared between cases and controls and expressed in copy numbers (Table 2). The levels of miR-1202 were found to be downregulated in patients with MDD 1.78 ± 0.65 compared to FDRs 2.17 ± 0.76 and UHCs 2.22 ± 0.72, and these differences were statistically significant (P < .01). However, the comparison between patients with MDD and FDRs was not statistically significant, although there was trends towards significance in this comparison (P < .056). The levels of miR-16 and miR-132 did not show any significant differences between the groups.
Measurement of Serum Total Oxidant Status and Total Antioxidant Status in Cases and Controls
The serum levels of TOS were higher in FDRs (45.2 [38.6, 59.1]) than in patients with MDD (17.1 [9.3, 27.4]), and this difference was statistically significant (P < .01). Conversely, in UHCs (29.7 [13.0, 54.6]), the levels of TOS were higher and statistically significant than in patients with MDD (P < .01). The TOS levels significantly differed when comparing FDRs and UHCs (P < .01). However, the serum levels of TAOS were similar across the groups and were not statistically significant. Details are presented in Table 2.
Evaluation of IL-6 (Primary Outcome)
In model 1, we found a significant difference in IL-6 levels between patients with MDD and UHC groups (β = 1.92; 95% CI, 0.06 to 3.78; P < .04). In other words, compared to UHCs, levels of IL-6 were higher in MDD by 1.92 units, and this difference was statistically significant. In model 2, we did not find a significant difference in the IL-6 level between the FDR and UHC groups (β = –1.59; 95% CI, –3.87 to 0.67; P < .16). In other words, compared to UHCs, levels of IL-6 were lower in FDRs by 1.59 units, and this difference was not statistically significant. In model 3, we found a significant difference in IL-6 levels between the MDD and FDR groups (β = 6.04; 95% CI, 1.60 to 10.49; P < .05). In other words, compared to FDRs, levels of IL-6 were higher in patients with MDDs by 6.04 units, and this difference was statistically significant (Table 3).
Multiple Linear Regression Analysis to Compare Patients with Major Depressive Disorder, Their First-Degree Relatives, and Unrelated Healthy Controls on the Outcome Measures.
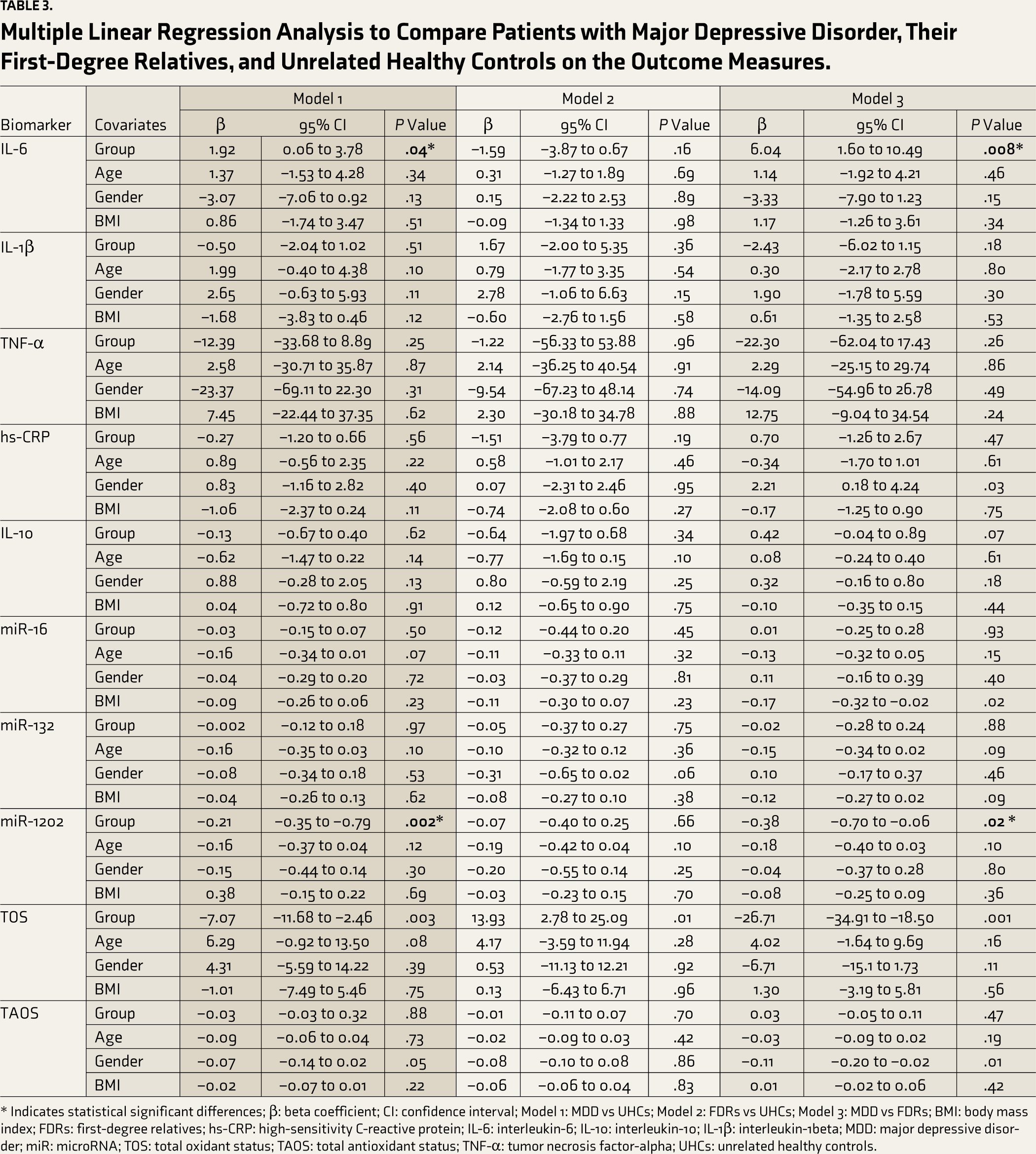
* Indicates statistical significant differences; β: beta coefficient; CI: confidence interval; Model 1: MDD vs UHCs; Model 2: FDRs vs UHCs; Model 3: MDD vs FDRs; BMI: body mass index; FDRs: first-degree relatives; hs-CRP: high-sensitivity C-reactive protein; IL-6: interleukin-6; IL-10: interleukin-10; IL-1β: interleukin-1beta; MDD: major depressive disorder; miR: microRNA; TOS: total oxidant status; TAOS: total antioxidant status; TNF-α: tumor necrosis factor-alpha; UHCs: unrelated healthy controls.
Evaluation of miR-1202 as a State or Trait Biomarker
In model 1, we found a significant difference in the miR-1202 levels between the patients with MDD and UHC groups (β = –0.21; 95% CI –0.35 to –0.79; P < .05). In other words, compared to UHCs, levels of miR-1202 were downregulated in MDD by 0.21 units, and this difference was statistically significant. In model 2, we did not find a substantial difference in the miR-1202 levels between the FDR and UHC groups (β = –0.07; 95% CI, –0.40 to 0.25; P < .66). In other words, compared to UHCs, levels of miR-1202 were downregulated in FDRs by 0.07 units, and this difference was not statistically significant. In model 3, we found a significant difference in miR-1202 levels between patients with MDD and FDR groups (β = –0.38; 95% CI, –0.70 to –0.06; P < .05). In other words, compared to FDRs, levels of miR-1202 were downregulated in patients with MDD by 0.38 units, which was statistically significant. Details are in Table 3.
Assessment of IL-1β, TNF-α, hs-CRP, IL-10, miR-16, and miR-132 as State or Trait Biomarkers
In model 1, we did not find a significant difference in IL-1β (β = –0.50; 95% CI, –2.04 to 1.02; P < .51), TNF-α (β = –12.39; 95% CI, –33.68 to 8.89; P < .25), hs-CRP (β = –0.27; 95% CI, –1.20 to 0.66; P < .56), IL-10 (β = –0.13; 95% CI, –0.67 to 0.40; P < .62) and the gene expression of the miR-16 (β = –0.03; 95% CI, –0.15 to 0.07; P < .50) and miR-132 levels (β = –0.002; 95% CI, –0.12 to 0.18; P < .97) between the MDD and UHC groups. In other words, compared to UHCs, the levels of IL-1β by 0.50 units, TNF-α by 12.39 units, hs-CRP by 0.27 units, and IL-10 by 0.13 units were reduced, and gene expressions of miR-16 by 0.03 units and miR-132 by 0.002 units were downregulated in patients with MDDs, and all were statistically nonsignificant. As model 1 did not establish assay sensitivity for these biomarkers, we did not consider results from models 2 and 3 for interpretation (Table 3).
Assessment of Total Oxidant Status and Total Antioxidant State or Trait Biomarkers
In model 1, we did not find a significant difference in the TOS (β = –7.07; 95% CI, –11.68 to –2.46; P < .001) and TAOS levels (β = –0.03; 95% CI, –0.03 to 0.32; P < .88) between the MDD and UHC groups. In other words, compared to UHCs, levels of TOS were lower in patients with MDD by 7.07 units and TAOS by 0.03 units, both of which were statistically nonsignificant. As model 1 did not establish assay sensitivity for these biomarkers, we did not consider results from models 2 and 3 for interpretation (Table 3).
Discussion
The primary objective of the study was to assess the status of IL-6 as a trait or state biomarker in patients with MDD. Secondary outcome was to measure the levels of other inflammatory cytokines (IL-1β, TNF-α, hs-CRP, and IL-10), miRNAs (miR-16, miR-132, and miR-1202), and oxidative stress markers (TOS and TAOS). We documented significantly higher levels of IL-6 and downregulated expressions of miR-1202 in patients with MDD than FDRs, suggesting that these may be reflected as state biomarkers in MDD. In contrast, none of the other studied biomarkers emerged as trait or state biomarkers in MDD.
Evaluation of IL-6 as a State Biomarker in MDD
IL-6, a pleiotropic cytokine, is the most frequently investigated biomarker of inflammation concerning MDD. Data from the previous meta-analyses have shown that there is a positive relationship between MDD and IL-6 among children and adolescents, adults, and elderly patients with MDD when compared to healthy controls.19-22 Dowlati et al. 5 conducted a meta-analysis, excluding patients with other medical comorbidities, and found significantly higher levels of IL-6 in patients with MDD than controls. The studies mentioned above compared the levels of IL-6 between cases and controls; none of the studies were compared with FDRs, which will help to rule out the role of genetic influences on the cases.
Higher levels of IL-6 activated in response to genetic and environmental factors can affect various pathways, such as activating the HPA axis, impairing neuroplasticity, interfering with anti-oxidative mechanisms, disrupting neuroendocrine functions, neurotransmitter metabolism, and activating the serotonin transporter, which triggers depressive symptoms and leads to MDD. 23 Treatment with antidepressants can reverse these activities, thereby improving depressive symptoms and reducing IL-6 levels.24,25 We found that IL-6 levels in patients with MDD were significantly increased compared to FDRs after adjusting for confounders such as age, gender, and BMI. Hence, IL-6 can be considered as a state biomarker in MDD.
Expression of miR-1202 as a State Biomarker in MDD
The miR-1202, a primate-specific miRNA, is dysregulated in patients with MDD. A growing body of research states that miR-1202 targets the gene expression of metabotropic glutamate receptor 4 (GRM4), which in turn modulates the expression of glutamatergic, dopaminergic, GABAergic, and serotonergic neurotransmission and activation of the serotonin transporter, thereby leading to the severity of depressive symptoms.26,27 Data from the clinical trials documented that treatment with selective serotonin reuptake inhibitors and serotonin-norepinephrine reuptake inhibitors increased the expressions of miR-1202 after eight weeks in patients with MDD.28-30 We found that the expression of miR-1202 in patients with MDD was significantly downregulated compared with FDRs after adjusting for confounders, such as age, gender, and BMI. The studies of Lopez et al., 7 Gheysarzadeh et al., 31 and Zhou et al. 32 are in agreement with our findings. However, all these studies compared the expression of miR-1202 between patients with MDD (community- or hospital-based) and controls. No study, to date, has compared the miR-1202 expression between patients with MDD and their FDRs. The advantage of this comparison is that it addresses the possibility of unknown and unmeasured genetic confounders (MDD is not genetic in origin owing to the expression of genes) in the association being examined. In other words, this comparison helps confirm state marker status for a biomarker of interest as opposed to a comparison between patients with MDD and controls. Hence, miR-1202 can be considered as a state biomarker in MDD.
Nonsignificant Differences in Other Biomarkers in Patients with MDD
Intriguingly, the levels of other inflammatory cytokines (IL-1β, TNF-α, hs-CRP, and IL-10) did not differ between the cases and controls; similar findings have been noted earlier by some investigators. 33 On a similar note, prior authors have found that the expression of miR-16 and that of miR-132 were not significantly different in cases and controls in our study.34-36 Regarding oxidative stress markers, our findings did not show any significant difference between cases and controls, which was in line with findings from prior studies. 14 In other words, none of these biomarkers emerged as trait or state biomarkers in MDD in the present study. Our nonsignificant findings may indicate two possibilities. First, our findings may be true in the population, which means these biomarkers are neither trait nor state biomarkers in MDD. Second, our study may have been underpowered, and the actual differences between patients with MDD and controls may be smaller in magnitude than what we found. Only an adequately powered study can answer these questions with certainty.
IL-6 and miR-1202 as State Biomarkers in Patients with MDD
Results from the unadjusted univariate analysis showed that the IL-6 levels were significantly higher in patients with MDD than the FDR and UHC groups. The miR-1202 levels were significantly downregulated in patients with MDD compared with UHCs. The levels of TOS were considerably higher in FDRs and UHCs than in patients with MDD. The higher levels of TOS in FDRs than that in MDD was an unusual finding in our study. Unadjusted analyses represent real-world associations between variables; hence, these findings may be of additional interest. Employing IL-6 and miR-1202 as state biomarkers has the following clinical implications. They may be used as diagnostic biomarkers (to confirm the presence of MDD), illness severity biomarkers (which reflect current illness severity), and prognostic biomarkers (which predict the MDD course). Thus, incorporating IL-6 and miR-1202 as state biomarkers in clinical practice could be a valuable advantage for clinicians in terms of accurate diagnosis, predicting the severity of MDD, and monitoring treatment response, potentially improving care and outcomes. Further, it also provides a basis for enhancing our understanding of the pathophysiology of MDD.
The Clinical Usage of Biomarkers in MDD
The clinical usage of biomarkers in MDD may pose certain challenges. MDD is a heterogeneous disorder that involves multiple biological systems and pathways. This complexity makes it difficult to pinpoint specific biomarkers. Most of the evidence-based biomarkers (e.g., cytokines, DNA, RNA, histones, proteins, and hormones) are variably expressed in individuals based on constitutional factors, such as age and gender, and lifestyle factors, such as stress and diet. Further routine estimation of biomarkers in MDD is hampered by cost, feasibility, and between-run variability.
Given the biological and clinical heterogeneity in MDD, further studies involving larger sample sizes and studying a biomarker panel comprising genetic, neurophysiological, neuroimaging, neuroendocrine, and blood biomarkers may have greater scientific relevance and practical utility. The advent of big data approaches has made it possible to explore the validity and clinical utility of biomarker panels and optimize them for better performance.
Strength of the Study
The strength of the study lies in the fact that we recruited drug-naïve, first-episode patients with MDD to minimize the confounding impacts of psychotropic exposure. We included an additional control group consisting of FDRs (blood relatives) to address genetic confounders in our analysis of associations. We used SCID-5-RV, a structured diagnostic tool for confirmation of MDD diagnosis, and MMS, a widely accepted screening tool, to rule out psychiatric illness in controls; both add rigor to the work.
Limitations
Out of 50 FDRs in the control group, we could recruit only 31 participants because many were not willing to participate. This might have affected the study’s strength. We excluded noninflammatory patients with MDD phenotypes that could have affected our findings.
Conclusion
IL-6 and miR-1202 may serve as state biomarkers in patients with MDD. As such, they may be useful in tracking illness severity over time and monitoring response to treatment. However, their greatest utility may lie in pointing to underlying pathophysiological mechanisms and helping develop treatments that target these putative pathways. Our study may have been underpowered to detect differences in other markers studied. Future studies should be conducted with larger sample sizes to strengthen our findings. Future work must also focus on developing biomarker panels for MDD that have optimal validity and utility for diagnosis and treatment. With the promise of big data approaches, this may be plausible.
Footnotes
Acknowledgements
The Indian Council of Medical Research (ICMR), New Delhi (Award No: 3/1/3/1/M/2022-NCD-II), is acknowledged for awarding the ICMR Senior Research Fellowship (SRF) to Mr. Rockson Christopher, PhD Research Scholar, Department of Pharmacology, JIPMER, Puducherry. We also thank Ms. Durga Devi, Medical Laboratory Technologist, Department of Biochemistry, JIPMER, Puducherry, for her technical support during the ELISA procedures.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration Regarding the Use of Generative AI
The authors declare that they did not use generative AI tools to collect or analyze data, produce images or graphs, or write this article.
Ethical Approval
This study commenced after receiving approval from the Institutional Ethics Committee for Human Studies (JIP/IEC/2019/108). Written informed consent was obtained from all participants.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Funding for this study was provided by ICMR Extramural ad-hoc grant from the Indian Council of Medical Research (ICMR), New Delhi (Grant No. 5/4-4/166/M/2020-NCD-II); the ICMR had no further role in study design; in the collection, analysis and interpretation of data; in the writing of the report; and in the decision to submit the paper for publication.
