Abstract
Nutraceutical agents and food supplements are commonly used as treatment adjuncts in neuropsychiatric disorders. Curcumin, a bioactive agent obtained from the rhizome of Curcuma longa, with its antioxidant and anti-inflammatory properties, has gained much research attention in the last few decades. In this narrative review, we intend to summarize the evidence available for curcumin as an add-on agent in the management of schizophrenia. We searched PubMed/EBSCO for both human and animal trials utilizing curcumin in the management of schizophrenia. We obtained ten articles (five preclinical and five clinical) from the focused literature search. Clinical research utilizing curcumin in schizophrenia is limited to negative and cognitive symptoms. Available preclinical studies suggest curcumin’s utility in ameliorating extrapyramidal and metabolic side effects when given as an adjunct with antipsychotics. Curcumin, as an add-on agent, appears promising to improve the negative and cognitive symptoms of schizophrenia. Notably, curcumin was tolerable and safe in all the randomized human clinical trials. The poor oral bioavailability is, however, a limiting factor in its widespread use.
Nutraceuticals and food supplements are commonly used as either stand-alone treatments or as add-on agents with ongoing psychopharmacological management. Different supplementary food agents tried include S-Adenosyl Methionine, l-methyl folate, omega-3-fatty acids, and vitamins like B complex, D, and E.1,2 The indications for such use include augmenting drugs for symptom reduction, reducing side effects, and cognitive enhancement. Curcumin is diferuloylmethane, an active ingredient present in the Indian spice turmeric. 3 Turmeric is widely used in the cuisines of India and other Asian countries.3,4 Curcumin is a nonflavanoid polyphenolic molecule derived from Curcuma longa. In traditional medicine, turmeric is commonly used as an anti-inflammatory and antiseptic agent.2,4 Its antioxidant property and low toxicity potential are utilized in physical disorders like rheumatioid arthritis, cardiovascular disorders, asthma, and neuropsychiatric disorders like Alzheimer’s dementia and Parkinsonism.3–8 There are no available reports of toxicity in humans following long-standing use of curcumin. 2 Despite its cost-effectiveness and favorable tolerability profile, poor absorption and high first-pass metabolism, with overall low bioavailability, have hindered its clinical utility.4,6
Schizophrenia is a severe mental disorder with disturbances in thought, emotion, perception, and behavior. 9 Genetic, early environmental, and psychological and social factors are conceptualized to play an important role in the development and progression of schizophrenia. Oxidative stress deregulation and related neuroplasticity changes are key contributors in the etiopathogenesis of this neurodevelopmental disorder.10,11 The life expectancy of schizophrenia patients is less by 10–15 years in comparison with the general population. 12 Increased substance use, cardiovascular morbidity, sedentary lifestyle, side effects of psychotropic drugs, and suicides contribute to this early mortality.13,14 Available antipsychotic drugs are often criticized for targeting preferentially the positive symptoms of schizophrenia, while the functional recovery depends on the improvement in negative and cognitive symptom domains as well.15,16 Repurposing the available drugs and utilizing the emerging adjunctive nutraceuticals may be beneficial in better understanding the pathophysiology of this complex disorder as well as in providing potential safe and tolerable treatment options.17,18 Such novel pharmacological options could be utilized to reduce the burden of negative and cognitive symptoms of schizophrenia. 19
The role of curcumin in the management of depression and bipolar disorder has been proposed recently. 20 Curcumin as an add-on to antidepressant drugs has been convincingly shown to reduce depressive symptoms, compared to placebo add-on. A recent meta-analysis supported the utility of adjunctive curcumin in the management of depression and anxiety disorders. 21 Notably, curcumin was tolerable and safe in all those randomized human clinical trials. In this narrative review, we aim to discuss the mechanism of action of curcumin, followed by a review of available evidence and its implications for clinical use of curcumin in schizophrenia management.
Mechanism of Action
Oxidative-stress-dependent pathological changes are postulated to underlie the progression of neuropsychiatric disorders. 22 The proposed pharmacological actions of curcumin include antioxidant, anti-inflammatory, antimutagenic, antibacterial, antiprotozoal, antiobesity, antidiabetic, hepatoprotective, and procognitive properties.3,4,23,24 Curcumin inhibits the production of reactive oxygen species and reduces the production of inflammatory cytokines by inhibiting cyclooxygenase and lipoxygenase enzymes. 25 Curcumin is a lipophilic compound with the ability to penetrate the blood-brain barrier and exert its antioxidant, neuroprotective activity. 26 Generally, an oral dose of more than 2 g/day is reported to intensify curcumin’s antioxidant effects.27,28 Curcumin reduces the turnover of monoamines such as dopamine, serotonin, and norepinephrine and thereby prevents the accumulation of free radicals.23,25 Curcumin is also implicated as a free radicals scavenger that removes superoxide anion, hydroxyl, and singlet oxygen radicals. Curcumin upregulates the expression of genes related to the enzyme glutathione synthetase, ultimately leading to increased glutathione levels in the astrocytes and neurons. This enables better scavenging of the free radicals in the neuronal milieu. 29 Curcumin improves neuroplasticity and hippocampal neurogenesis through the enhancement of brain-derived neurotrophic factor (BDNF).30,31 Curcumin also improves the synthesis of docosahexaenoic acid (DHEA) through the activation of the enzymes involved. In animal models of depression, upregulation of DHEA levels is proposed to be neuroprotective. 32
Anti-obesity effects of curcumin are postulated to be secondary to the suppression of nuclear factor kappa B (NFkB) and suppression of tumor necrosis factor (TNF) expression.5,8,33 Curcumin regulates cellular enzymes like protein kinase C (PKC) and inhibits apoptosis. 5 In animal models of schizophrenia, curcumin protects against mitochondrial dysfunction by removing free radicals. 34 Inflammatory insults and oxidative stress since obstetric period cumulatively modulate the expression of neuroplasticity genes and ultimately result in “neuroprogression” of psychiatric disorders like schizophrenia.35,36 Deoxy-ribonucleic acid (DNA) methylation secondary to inflammatory insults silences gene expression and impairs neuroplasticity. Given its epigenetic regulatory properties, curcumin is postulated to reduce DNA methylation, histone deacetylation, and associated gene expression deficits.35,37–39 Importantly, curcumin inhibits monoamine oxidase A and B (MAO-A and MAO-B) enzymes and shows potent antidepressant action. 40 Also, in animal models, it reduces the expression of dopamine D1 receptor in the cerebellum and thereby probably reduces antipsychotic-induced depressive features. 41
To summarize, the possible mechanisms of curcumin’s action postulated to be useful in neuropsychiatric disorders include (but not limited to) regulation of oxidative stress and inflammation, reduction of monoaminergic and hypothalamo-pituitary-adrenal axis disturbances, addressing mitochondrial dysfunction, and hindering neuroprogression. 7 The current understanding of the etiopathogenesis of schizophrenia suggests all the above-mentioned mechanisms to be involved in the neuroprogression of the disorder.42–46 Hence, the role of adjunctive curcumin in the management of schizophrenia appears as a promising area of interest.
Methodology
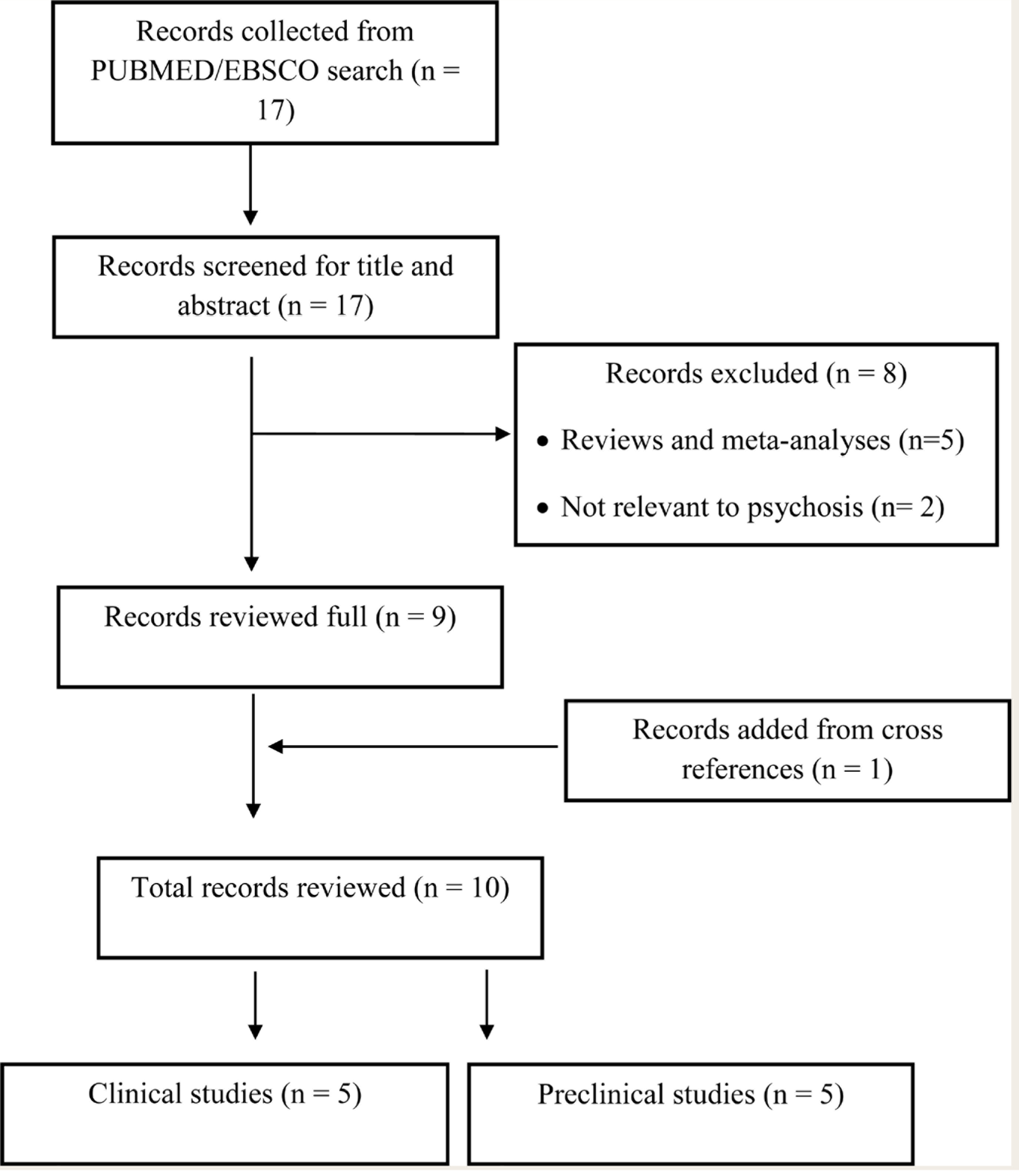
The authors (DD and VS) searched PubMed and EBSCO independently with the following search terms (as on April 2021): “Curcumin” “AND” “Schizophrenia,” “Psychosis,” “Delusional disorder,” “Catatonia,” “Cognition,” “adverse effects,” and “antipsychotic.” The authors identified 17 manuscripts. Both preclinical (animal studies) and clinical research studies that used curcumin in managing both the symptoms and antipsychotic-related side effects were included for this narrative review. The search was neither restricted to languages nor time. After removing review articles, commentaries or opinion letters, and manuscripts not relevant to psychosis, the total unique manuscripts identified for the final review were “10” (5-human clinical; 5-animal models). The selected studies are reviewed below and summarized in Tables 1 and 2. The outcome of the literature review is outlined in Figure 1.
Clinical Studies on Add-on Curcumin in Schizophrenia
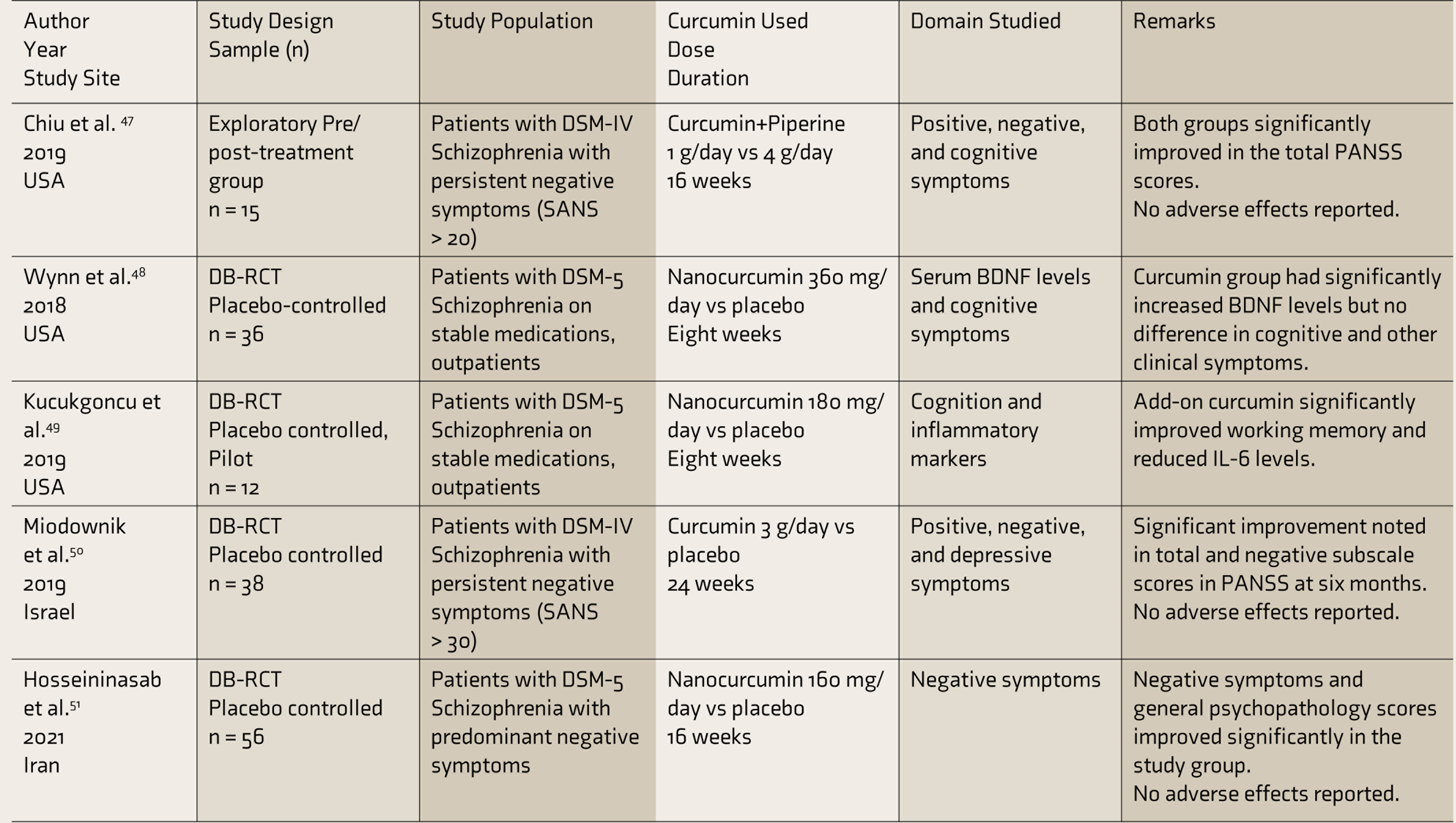
BDNF: brain-derived neurotropic factor, DB-RCT: double blind randomized controlled trial, DSM: Diagnostic and Statistic Manual, IL-6: interleukin-6, PANSS: positive and negative syndrome scale, SANS: Scale for the Assessment of Negative Symptoms.
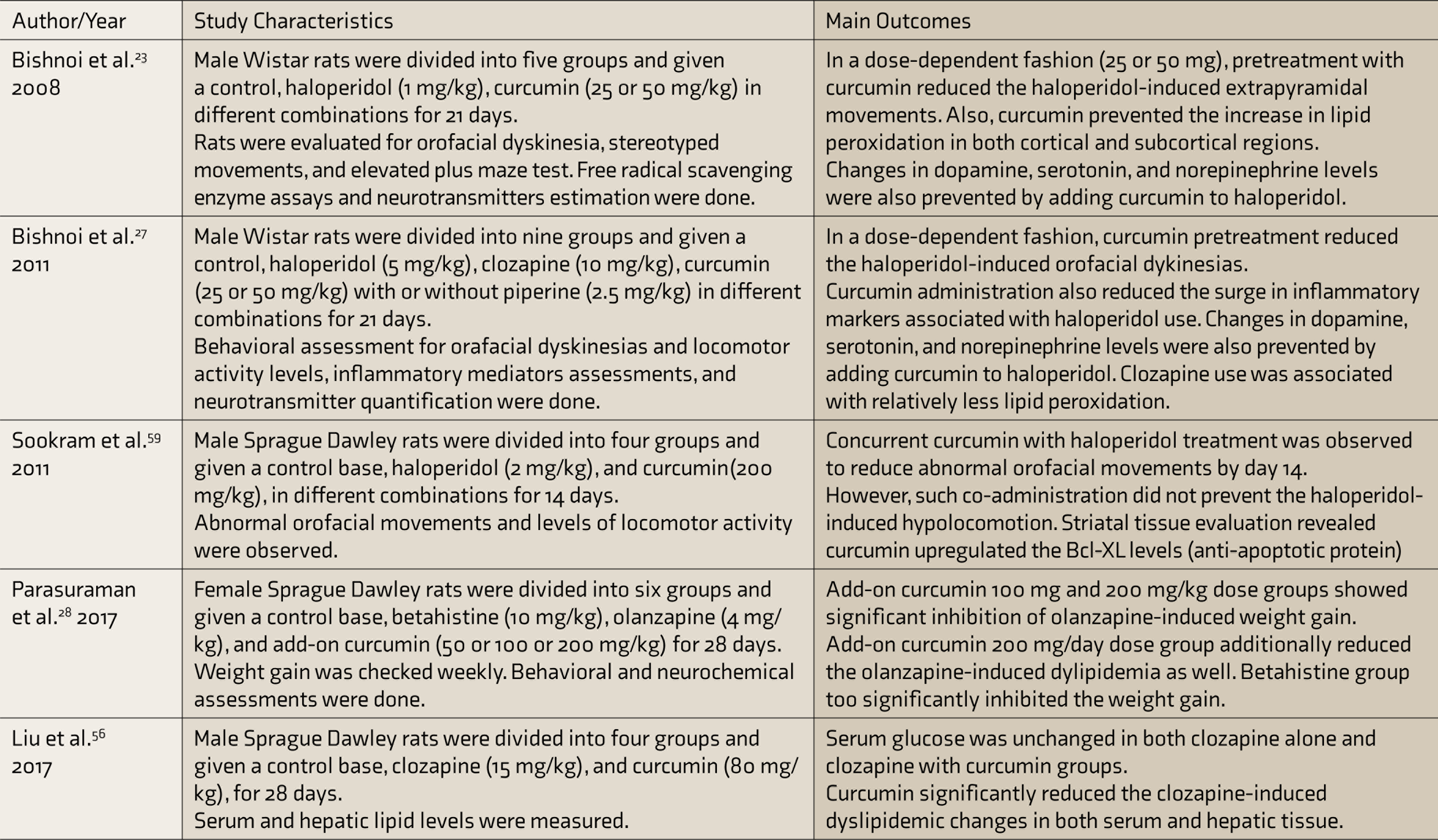
Preclinical Studies on Add-on Curcumin in Schizophrenia
Outcome of the Literature Review
Curcumin’s Role in the Management of Schizophrenia
An exploratory proof-of-concept study examined the effects of curcumin combined with piperine (from black pepper extract–used to improve the bioavailability of curcumin) as an add-on to the ongoing antipsychotic treatment. Fifteen chronic schizophrenia patients were randomized to receive 1 g vs 4 g of add-on curcumin and piperine compounds. 47 At the end of 16 weeks, both groups showed significant improvements in total scores and general psychopathology subscale of the Positive and Negative Syndrome Scale (PANSS). Both the doses were tolerated well without much adverse effects. 47
Another double-blind, randomized controlled trial examined the procognitive effects of curcumin (versus placebo) in stable, medicated schizophrenia patients. Despite a significant increase in the serum BDNF levels, 360 mg/day of a nanoparticle-based curcumin (Theracumin) preparation failed to improve cognitive performance or clinical variables at eight weeks. However, the study was underpowered, with a small sample size. 48 Another pilot study examined a lower dose of add-on curcumin (180 mg/day) in chronic stable schizophrenia patients. Curcumin significantly reduced pro-inflammatory cytokine (IL-6) levels and improved working memory performance at 12 weeks. 49
Curcumin’s effects on positive, negative, and depressive symptoms were explored, as an add-on to ongoing antipsychotic treatment, in a double-blind, randomized controlled trial. 50 At the end of six months, compared to placebo, curcumin significantly reduced negative symptoms and total scores on PANSS. Curcumin dose (3 g/day) was tolerated well by all the patients.
A double-blind randomized placebo-controlled study observed the effects of nanocurcumin soft gel capsules (160 mg/day) given for 16 weeks as an add-on to the ongoing antipsychotic drug regimen on the negative symptoms of chronic stable schizophrenia patients. 51 The nanocurcumin group showed significant improvements in negative symptoms, positive symptoms, general psychopathology scores, and total PANSS scores. Importantly, there were no significant differences in adverse effects between the groups, and none of the participants left the study citing adverse events. 51
Preclinical Studies
Management of Metabolic Adverse Effects
Metabolic disturbances are common even in drug naïve schizophrenia. Treatment with second-generation antipsychotics further accentuates lipid derangements and cardiovascular morbidity. 52 Clozapine is the ultimate treatment option in resistant schizophrenia. 53 Though increased appetite, food intake, and sedentary lifestyle may contribute to dyslipidemia, clozapine may affect lipid metabolism through other peripheral mechanisms as well. 54 Adenosine monophosphate-activated pro-tein kinase (AMPK) is a key regulator in hepatic energy and lipid metabolism. Clozapine and other antipsychotics suppress AMPK activity, thereby ultimately leading to dyslipidemia. 55 Curcumin, on the other hand, by facilitating AMPK activity, reduces lipogenesis. Recent evidence from animal studies suggests that curcumin mitigated clozapine-induced dyslipidemic changes through AMPK activation 56 and olanzapine-induced weight gain and dyslipidemia. 28 Curcumin as an add-on with antipsychotics appears a promising avenue to be explored to reduce lipid abnormalities.
Management of Extrapyramidal Adverse Effects
The imbalance between production and detoxification of free radicals putatively contributes to the development of antipsychotic-associated tardive extrapyramidal syndromes.57,58 Animal studies suggested that curcumin pretreatment protected haloperidol-exposed rats from developing tardive extrapyramidal movements. 23 Curcumin prevented the antipsychotic-associated increase in lipid peroxidation and reduction in levels of antioxidant enzymes such as catalase, superoxide dismutase, and glutathione. 23 Chronic administration of antipsychotics reduced the levels of monoamines such as dopamine, serotonin, and norepinephrine. Simultaneously, antipsychotics also increased oxidative stress parameters, TNF-alpha levels, caspase-3 activity, and NFKB activity in the striatal region. In animal models, pretreatment with curcumin (25–50 mg/kg—intraperitoneal) attenuated the negative oxidative effects of clozapine and haloperidol administration and prevented tardive movements. 27 Clozapine, however, produced less oxidative stress compared to haloperidol. 27 This protective effect of curcumin is also attributed to its antiapoptotic property. Concurrent curcumin administration with haloperidol is shown to upregulate the antiapoptotic protein Bcl-XL in the striatal region, thereby reducing apoptosis. 59
It is important to note that human clinical studies and translation of such evidence in managing several antipsychotic-related adverse effects are still pending. However, adjunctive curcumin appears a potential option in the management of antipsychotic-induced metabolic and extrapyramidal side effects.
Discussion
Curcumin’s antioxidant, anti-inflammatory, and procognitive properties appear to be useful in managing neuropsychiatric disorders, especially schizophrenia. 7 Curcumin’s effects on monoamines turnover, monoamine oxidase enzyme inhibition, neurosteroid modulation, and upregulation of key neuroplastic markers like BDNF promise to be beneficial in hindering the detrimental neuroprogression in schizophrenia.32,49 Preliminary evidence for curcumin’s role in schizophrenia was suggested by the exploratory study by Chiu et al., 47 where participants in both groups (1 g vs 4 g) showed significant improvement in total PANSS scores. Further studies demonstrated its efficacy in reducing inflammatory markers and improving neuroplasticity markers and cognition in schizophrenia.48,49 Importantly, two double-blind, placebo-controlled trials reported add-on curcumin’s efficacy in reducing the negative symptoms of schizophrenia.50,51
Four out of five clinical studies reviewed utilized double-blind, randomized controlled designs with active placebo arms. One preliminary study utilized pre-post design in the same treatment group. All five studies recruited patients who were on stable antipsychotic medications. Three studies recruited patients with predominantly negative symptoms. The reviewed studies differed in duration of add-on curcumin treatment (8–24 weeks). Doses (160 mg/day–4 g/day) and preparations of curcumin differed across the studies. Nanocurcumin capsules were used in three studies.48,49,51 One study used a combination of curcumin and piperine. 47 All the doses were tolerated well, and none of the participants withdrew from these studies owing to intolerable adverse effects. Reportedly, three other clinical trials exploring the potential use of curcumin in schizophrenia are underway. 4
Further, preclinical studies support curcumin’s role in ameliorating the metabolic side effects of antipsychotics such as olanzapine 28 and clozapine. 56 Such positive metabolic changes are postulated to be secondary to the regulation of hepatic energy and lipid metabolism through AMPK activity. 56 Also, pretreatment with curcumin prevented the development of tardive extrapyramidal syndromes. In both haloperidol and clozapine exposed rats, pretreatment with curcumin successfully prevented the development to tardive orofacial movements.23,27,59 Curcumin’s antioxidant, free radical scavenging, and antiapoptotic properties are postulated to underlie this preventive mechanism. 59
To summarize, the evidence regarding curcumin’s efficacy in reducing the positive symptoms of schizophrenia is limited. But it has the potential to ameliorate negative symptoms and cognitive deficits. Preclinical studies also reported curcumin’s beneficial effects in preventing and reducing extrapyramidal and metabolic side effects.
Future Directions
Clinical trials evaluating curcumin’s efficacy against specific domains of psychopathology such as positive, negative, and cognitive symptoms are the need of the hour. Efforts to overcome the pharmacokinetic limitations of the compound are underway. Curcumin dose range, acceptability, and safety of the formulations are to be established for each indication.
Conclusion
Preclinical and clinical trials reported curcumin’s potential role in the management of schizophrenia. Clinical research utilizing curcumin in schizophrenia is limited to negative and cognitive symptoms. Preclinical studies reported its utility in ameliorating extrapyramidal and metabolic side effects when given adjunct with antipsychotics. The poor oral bioavailability is a limiting factor in its widespread use. Newer drug delivery models based on nanoparticles, phospholipids, and liposomes appear promising to improve the bioavailability.
Footnotes
Acknowledgements
GV acknowledges the support of Department of Biotechnology (DBT)—Wellcome Trust India Alliance (IA/CRC/19/1/610005) and Department of Biotechnology, Government of India (BT/HRD-NBA-NWB/38/2019-20(6).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
