Abstract
Background:
Clozapine has well-documented inter-ethnic variations in pharmacokinetics. There is a paucity of data about clozapine use and associated adverse events such as seizures, obsessive compulsive symptoms, neutropenia, and agranulocytosis, from India.
Methods:
This retrospective cohort study followed up 228 patients initiated on clozapine in a tertiary care referral center in India for an average of 10 years. We calculated incidence rates of new-onset seizures, new-onset obsessive compulsive symptoms, agranulocytosis, and neutropenia. We collected data on doses of clozapine used and serum assays and calculated concentration-to-dose (C/D) ratios. We also collected relevant clinical details about clozapine-induced seizures.
Results:
In the sample, 16.8% had new-onset seizures, 12.3% had new-onset OC symptoms, 2.7% had neutropenia, and 0.9% had agranulocytosis. The mean C/D ratio was 2.09 (SD = 1.8). Almost half (46.3%) of available serum assays were in the supra-therapeutic range. Seizures were associated with a higher clozapine dose at one year (OR = 1.003; 95%CI = 1.000–1.006; P value = 0.045) and the presence of positive psychotic symptoms at one year (OR = 4.214; 95%CI = 1.894–9.373; P < 0.001).
Conclusion:
Compared to existing literature, Indians have a higher rate of clozapine- related seizures and need lower doses to reach therapeutic serum levels.
Indians seem to have a higher serum clozapine concentration to dose ratio than reported in Western populations. They may need lower doses of clozapine to reach therapeutic serum levels and may be more vulnerable to dose-related adverse events like seizures.Key Messages:
Clozapine is an atypical antipsychotic with proven efficacy in drug- resistant psychotic disorders.1, 2 It also has unpredictable pharmacokinetics that varies with ethnicity.3–7 Recent studies have suggested that Indians need lower doses of clozapine to reach therapeutic serum levels than Western populations.8–10
Clozapine is associated with various adverse effects that have led to its underutilization in several settings.5, 11–13 The most serious adverse effects are agranulocytosis and neutropenia.1, 14 Clozapine usage is also associated with an increased risk of seizures in a dose-dependent manner. 15 Psychiatric adverse effects like clozapine-induced obsessive-compulsive symptoms are also well documented. 16 Incidence rates of these adverse effects vary in the literature, and there is a paucity of data from India.17–19 This study aimed to add to the limited observational data about clozapine pharmacokinetics and specific adverse effects of seizures, de novo obsessive compulsive symptoms, neutropenia, and agranulocytosis in an Indian sample.
Materials and Methods
Setting and Design
The retrospective cohort study was conducted at a tertiary care mental health facility in Vellore, Tamil Nadu, India. It has 122 inpatient beds and an average outpatient attendance of 400–500 patients per day. The Institutional Review Board approved the study.
Participants
All adult patients who filled a prescription of clozapine in the institution pharmacy on an outpatient or inpatient basis in January 2013 were identified using online pharmacy records and included in the study. Patients who were lost to follow-up within one year of initiating clozapine were excluded. All patients were diagnosed using ICD-10 diagnostic guidelines. As an institutional policy, all patients were reviewed by consultant psychiatrists regularly to establish the validity of diagnoses and review the treatment strategies.
Data Sources
Data were extracted from a standardized pro forma used for all index visits to the institution, inpatient and outpatient clinical records, and online laboratory records. Patients were followed up from initiation of their clozapine trial till 2021, or till they were lost to follow-up. Data that was unavailable or unclear in patient records were coded as missing.
Variables
Primary outcome variables were: new-onset seizures at follow-up, new-onset obsessive compulsive symptoms at follow-up, serum clozapine levels, and calculated serum concentration to dose ratio (C/D ratio). Secondary outcome variables were: dose at one year and dose at the last follow-up.
Predictive variables included in the study were prognostic factors such as sex, clinical diagnosis, family history of psychotic illness, family history of seizures, and neuropsychiatric comorbidity at the time of initiation of clozapine, which could be associated with doses of clozapine required and, thus, influence rates of dose-related adverse effects. We also included possible confounders like the need for augmenting-antipsychotics at follow-up, persisting positive symptoms at one year, use of other psychotropics, age of onset of illness, months of follow-up, and time between the onset of psychiatric illness and initiation of clozapine trial, which could independently influence the outcome variables.
All serum clozapine assays were done 12 hours after the last clozapine dose on a steady dose of at least five days and were calculated using the high- performance liquid chromatography method. Serum clozapine levels were further categorized as subtherapeutic (<350 ng/ml), therapeutic (350–600 ng/ml), and supra-therapeutic (>600 ng/ml). 20 We calculated C/D ratios for all patients with the available data.
We collected additional observational data from records of patients with new-onset seizures after the initiation of clozapine. Patients with a past history of seizures before the clozapine trial were not included. Additional variables collected were: dose at the time of first seizure, time from initiation of clozapine trial, total duration of illness at first seizure, time to last adjustment of dose, use of augmenting antipsychotic at the time of first seizure, use of antiepileptic at the time of first seizure, additional psychotropic use at the time, medical comorbidity at the time of first seizure, the management strategy used for the seizure, and the total number of documented seizures.
Sample Size
Using an estimated seizure incidence from previously reported rates of 10%, the sample size for a power of 80% and an alpha of 0.05 was calculated to be 185. 19
Statistical methods
Continuous variables were described using mean, median, range, and standard deviation. Categorical variables were described using absolute numbers and percentages. Associations with categorical outcomes of interest (presence of new-onset seizures, presence of new-onset obsessive-compulsive symptoms) were tested, using the chi-square test, for categorical predictive variables like sex and family history of seizures. Student’s t-test or Mann–Whitney U test, as appropriate, were used for continuous predictive variables (months of follow-up, time between onset of symptoms and initiation of clozapine). Associations for continuous outcome variables (dose at one year, dose at last follow-up, C/D ratio) were tested using Spearman’s correlation coefficient for continuous predictive variables and t-test and Mann–Whitney U test for categorical predictive variables. Binary logistic regression was used to adjust for age and sex for significant binary associations with all primary outcome variables. Dose at one year, dose at the last follow-up, and positive symptoms at one year were also used as covariates for dose-related outcome variables of seizures and obsessive compulsive symptoms. We used SPSS version 16.0 for all analyses.
Results
Clozapine dose and non-seizure adverse effects
Table 1 details the characteristics of the sample, clozapine dose, and adverse effects. Two hundred and thirty-one patients were prescribed clozapine in January 2013. Three patients were lost to follow-up within one year and were excluded; 228 patients were included and followed up for a mean duration of 10.34 years (median = 9.33; SD = 5). The majority were male, with a diagnosis of schizophrenia, with the onset of illness in adulthood, with prolonged duration of illness, and without a family history of psychotic illness or seizures. One patient had organic psychosis related to a primary neurological diagnosis of Parkinson’s disease. A third of patients had persisting positive symptoms of psychosis after being on clozapine for one year, while one-sixth developed seizures and an eighth reported obsessive compulsive symptoms during follow-up. The mean clozapine dose at the last follow-up was lower than that recorded at one year. A very small minority developed agranulocytosis (0.9%) and neutropenia (2.7%) during the first year on clozapine.
Characteristics of the Study Sample (N = 228).
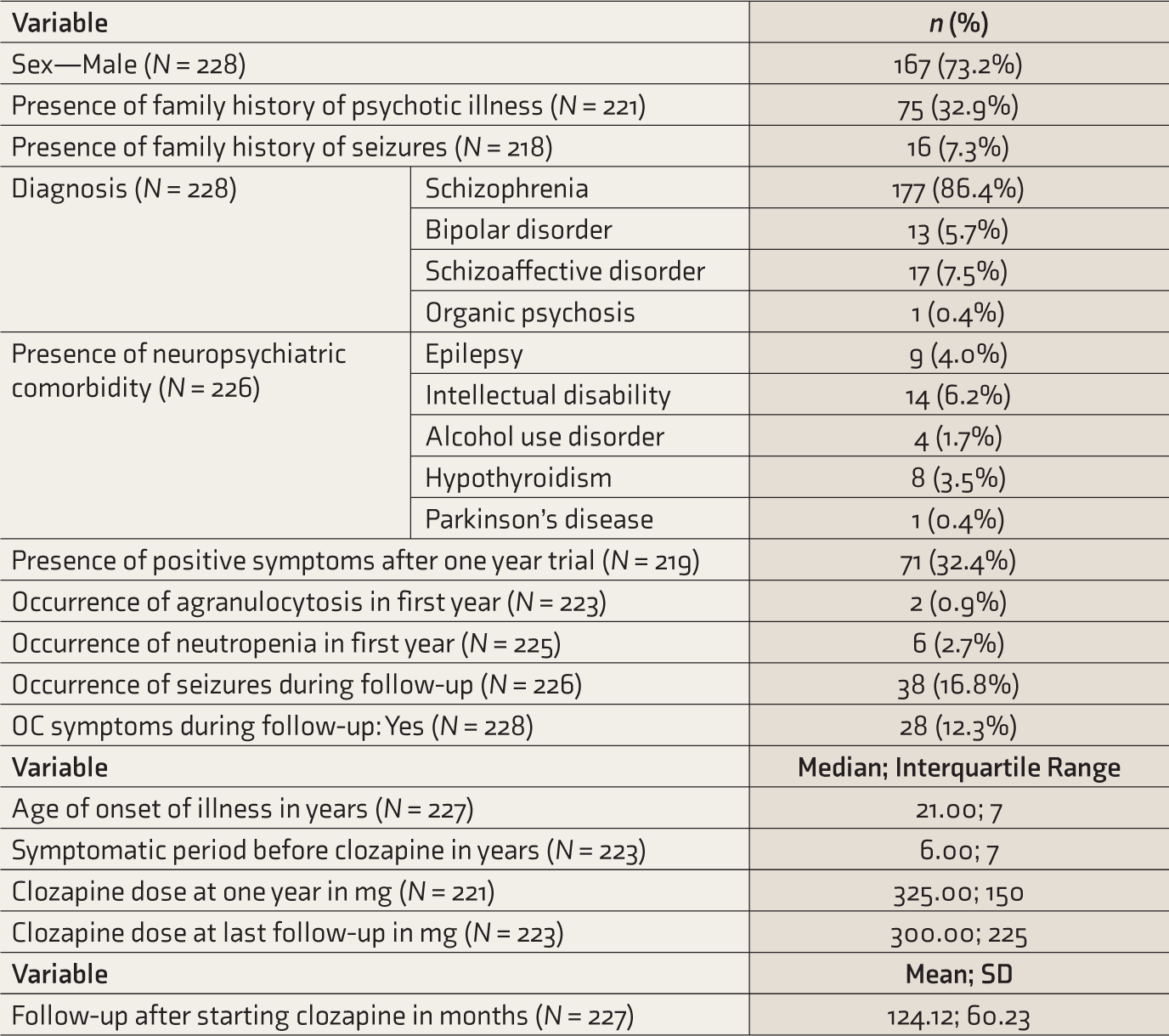
OC: Obsessive compulsive.
The 12-hour serum clozapine levels were available only for a small sub-sample (n = 54). The mean, median, standard deviation, and range were 719.19, 570.00, 554.43, and 74–3493 ng/ml, respectively. The mean, median, standard deviation, and range for the C/D ratio were 2.09, 1.62, 1.81, and 0.25–11.64, respectively. The serum clozapine levels were in the therapeutic range (350–600 ng/ml) for a third of the subjects (n = 17; 31.5%), while a little less than half were above the therapeutic range (n = 25; 46.3%) and about a fifth (n = 12; 22.2%), below the therapeutic range.
Patients with schizophrenia were on a significantly higher dose of clozapine at one-year follow-up (338 [SD = 113.6) mg versus 263 [SD = 145.2] mg; P value 0.01; t value = –3.22; df = 219) and at the last follow-up (296 [SD = 138.9] mg versus 193 [SD = 104.9) mg; P value < 0.001; t value = 3.96; df = 221) compared with those with other diagnoses. The association between dose and sex was not statistically significant. Nevertheless, the C/D ratio varied significantly with sex; males had a lower mean C/D ratio than women (1.65 versus 2.96; P value = 0.02; U value = 110; n = 42). These associations remained significant after adjusting for age and sex in multivariate analysis. Sex, diagnosis, family history of psychotic illness or seizures, age of onset of illness, neuropsychiatric morbidity/comorbidity, the time between the onset of illness and initiation on clozapine, need for augmentation with another antipsychotic, persisting positive symptoms, and duration of follow-up were not statistically significantly associated with agranulocytosis, neutropenia, or onset of obsessive-compulsive symptoms at follow-up.
Clozapine and seizures
Table 2 documents the details of seizures related to the use of clozapine. Thirty-eight patients (16.8%) had new-onset seizures during follow-up. The seizures seemed to occur late in the psychotic illness and at an average duration of being on clozapine for two years, on a stable dose of clozapine, about nine months after the last adjustment of clozapine dose. The majority were generalized tonic-clonic seizures. Most seizures (54.3%) occurred on clozapine monotherapy. Table 2 provides clinical details of patients with seizures along with details of concomitant medications at the time of the first seizure. None of the patients with new-onset seizures had a substance use disorder, concomitant antibiotic use, concomitant lithium use, or a clear triggering event like a febrile illness. No alternate etiology for the seizures could be established in any patient. Most patients with seizures had no medical comorbidity (89.5%) or neuropsychiatric comorbidity (92.1%) at the time of the first seizure. All patients with partial seizures were referred to neurologists and were evaluated for other etiologies of seizures, which could not be established. Most patients with seizures were managed by lowering the clozapine dose and adding anticonvulsant medication, with good results.
Details of Seizures Related to Treatment with Clozapine (N = 38).
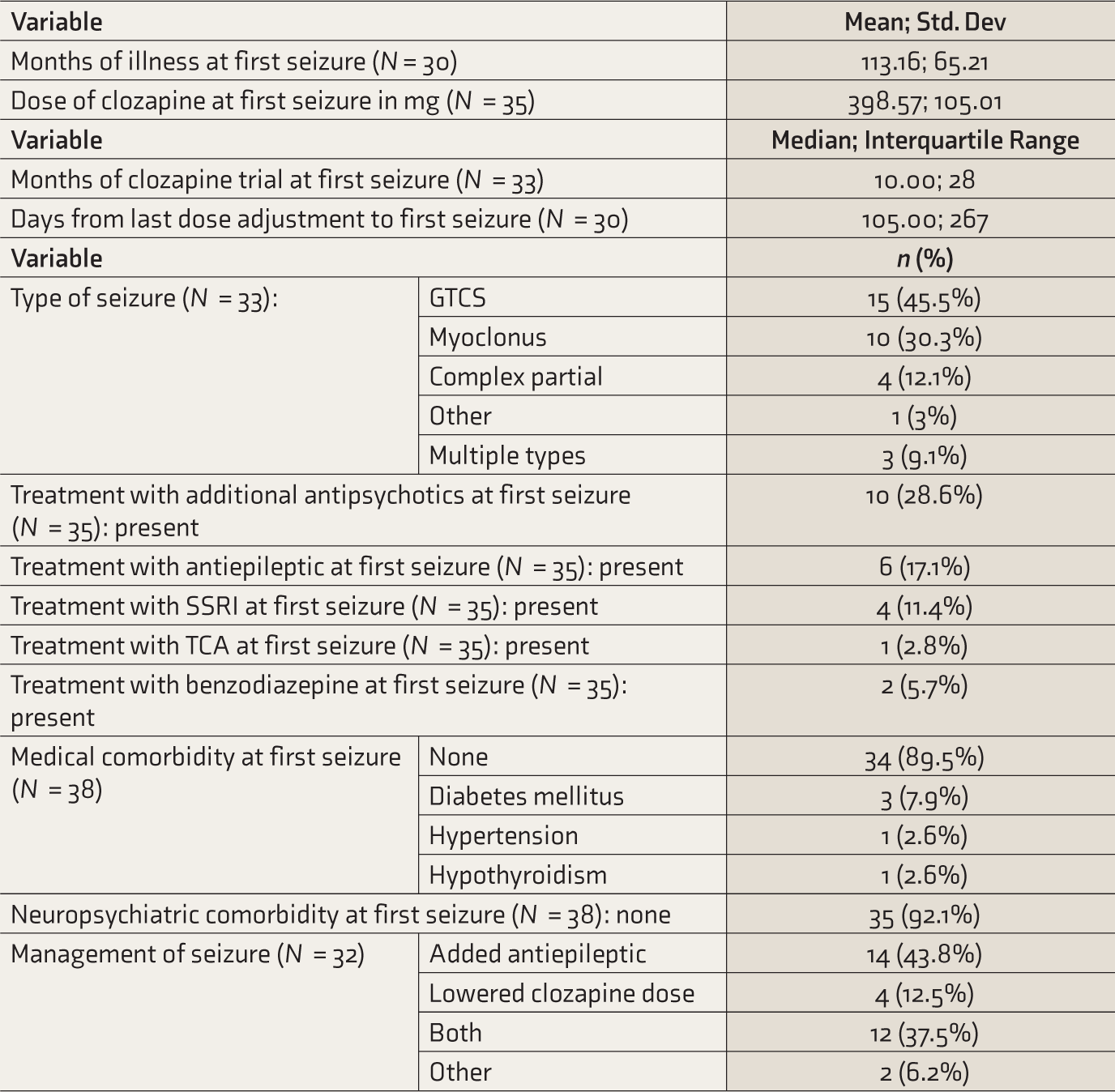
GTCS: generalized tonic-clonic seizure, SSRI: selective serotonin reuptake inhibitor, TCA: tricyclic antidepressant.
Patients with seizures had a significantly higher mean dose at one year (373 [SD = 119.2] mg versus 319 [SD = 119.8] mg; P value = 0.03; t value = –2.25, df = 217) and were more likely to have persisting psychotic symptoms at one year (OR = 3.46; 95%CI = 1.67–7.17; P value < 0.01). These associations remained significant after adjusting for age and sex in multivariate analysis. Further details of significant associations with seizures are provided in Table 3.
Significant Associations with Presence of Seizures at Follow-Up.

*Adjusted for age and gender.
#Adjusted for age, gender, dose at 1 year, and dose at last follow-up.
Discussion
This study fills in lacunae in available observational data about clozapine use in Indian patients. The most striking finding in this retrospective cohort is the significantly higher rate of seizures in the sample (16.8%) compared to other samples from India 18 that reported a rate of 6% and Western samples that reported far lower rates. 19 However, a smaller Japanese study had reported a higher rate of seizures (23.1%). 21 An important point to note while comparing these reported rates is that we classified, as clozapine-induced seizures, only de novo seizures after initiation of clozapine in patients with no past history of seizures.
Seizures at follow-up were significantly associated with higher doses of clozapine at one year of follow-up. Though doses at one year, at the last follow-up, and at the time of the first seizure were all within the defined therapeutic range, 46.3% of serum assays were in the supra-therapeutic range as defined by guidelines. 20 Clozapine doses, serum assay levels, and C/D ratios in this sample were similar to those recently reported in another Indian sample. 10 Seizures occurred in the absence of rapid titrations, augmenting antipsychotic agents, or concomitant psychotropic use. This higher seizure rate may be related to clozapine pharmacokinetics in the sample population.
Nevertheless, while the rate of seizures, a strongly dose-related adverse effect, was significantly higher than in Western data, the rates of idiosyncratic adverse effects, like agranulocytosis and neutropenia, were not. As clozapine-related seizures are known to have increasing cumulative risk, previous Indian studies with a shorter follow-up time may have underestimated seizure rates in this population. 15
All patients with seizures in this sample were continued on clozapine. The total number of seizures was not high, and all management strategies seemed to work equally well. Persisting positive psychotic symptoms at one year of clozapine trial were independently associated with the presence of seizures at follow-up in regression analysis, hinting at other interactions between predictors of clozapine response and clozapine-related seizures that could not be fully explored in this study.
Though this single retrospective study cannot comprehensively establish a causal relationship between clozapine and the documented adverse effects, particularly seizures, it strongly suggests it. Patients with seizures were on significantly higher doses of clozapine, thus establishing a gradient to this relationship. This study also reports higher C/D ratios in this population, providing a biological rationale for the higher rate of seizures reported. This pharmacokinetic data closely matches recent Indian literature, providing a measure of external validity and coherence. We only included patients with no prior history of seizures when calculating rates of clozapine-related seizures, in an effort to demonstrate a clear temporal sequence and exclude patients with a predisposition to seizures. Our data also rules out various alternate explanations for seizures, including a triggering medical event, concomitant medication, substance use, or alternate medical pathology. No alternate etiology for seizures was established in any of the patients.
In this sample, 12.3% of patients had de novo obsessive compulsive symptoms after starting clozapine. This is comparable with existing literature that reports an incidence of 3.5%–28.4%.16, 22 The only other Indian study has reported a rate of 6.8%. 23 Rates of agranulocytosis and neutropenia in this sample were well within the range of reported data.14, 24–26 The only other Indian study reported a rate of 0.6% for neutropenia. 26 This study adds to the very limited data in this area from India.
This study has significant strengths of adequate sample size and a long follow-up. However, it has several limitations due to its retrospective, single-center design and a small number of patients with serum clozapine levels. As data were collected retrospectively from outpatient records, there are inherent problems of missing data and a lack of a standardized pro forma to record adverse effects. We assumed no documentation of adverse events to mean no adverse events. However, it is important to note that such errors would tend to bias our results toward underestimating incidence rates of adverse events, making the high rates of seizures found in this cohort even more significant. This study focused on specific clozapine-related adverse effects, as there is little recent literature from India on these adverse effects, as well as due to limitations of available data. As the total number of participants who had seizures (n = 38) was small, the characteristics described may have limited generalizability. We also did not have data on participants’ smoking status, which is an important factor influencing clozapine levels. Details of concomitant medications and medical comorbidities were collected only for patients who had seizures, as part of ruling out other causes for the seizure. The smaller number of events per variable may have caused an overestimation of effects in the logistic regression. Other major limitations are the absence of a comparison group and the lack of structured causal analysis of these adverse events. However, we believe the study adds to very limited data in an area of immediate clinical relevance.
Conclusion
This study reports higher rates of dose-related adverse effects like seizures compared to existing literature. Pharmacokinetic data available for a subset of the sample adds to growing evidence that Indians need lower doses of clozapine to achieve a therapeutic serum level, compared to Western populations. Further, larger, prospective studies that better define a therapeutic dose range for the Indian population are a definite clinical need.
Footnotes
Acknowledgements
We thank Department of Psychiatry and Medical Records Department, Christian Medical College, Vellore for their support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
