Abstract
Dorsal root ganglia (DRG), trigeminal ganglia (TG), other sensory ganglia, and autonomic ganglia may be injured by some test article classes, including anti-neoplastic chemotherapeutics, adeno-associated virus-based gene therapies, antisense oligonucleotides, nerve growth factor inhibitors, and aminoglycoside antibiotics. This article reviews ganglion anatomy, cytology, and pathology (emphasizing sensory ganglia) among common nonclinical species used in assessing product safety for such test articles (TAs). Principal histopathologic findings associated with sensory ganglion injury include neuron degeneration, necrosis, and/or loss; increased satellite glial cell and/or Schwann cell numbers; and leukocyte infiltration and/or inflammation. Secondary nerve fiber degeneration and/or glial reactions may occur in nerves, dorsal spinal nerve roots, spinal cord (dorsal and occasionally lateral funiculi), and sometimes the brainstem. Ganglion findings related to TA administration may result from TA exposure and/or trauma related to direct TA delivery into the central nervous system or ganglia. In some cases, TA-related effects may need to be differentiated from a spectrum of artifactual and/or spontaneous background changes.
Keywords
This “Technical Review” article is a product of a working group of the Scientific and Regulatory Policy Committee (SRPC) of the Society of Toxicologic Pathology (STP). It has been reviewed and approved by the SRPC and Executive Committee of the STP, but it does not represent a formal Best Practice recommendation of the Society; rather, it is intended to provide basic knowledge to support a recent SRPC “Points to Consider” article on ganglion pathology (Bennet et al 21 ) that is focused on designing nonclinical studies or interpreting data from toxicity and safety studies intended to support regulatory submissions. This technical review has also been reviewed and endorsed by the European Society of Toxicologic Pathology (ESTP) and the British Society of Toxicologic Pathology (BSTP). The points expressed in this document are those of the authors and do not reflect the views or policies of the employing institutions or the endorsing societies. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to the Editor.
Introduction
Sensory and autonomic circuits of the nervous system include elements of both the central nervous system and peripheral nervous system (CNS and PNS, respectively). (Abbreviations are defined in the article at the point where they are first mentioned, but a full list is given in Supplemental Table 1 for reader convenience.) Principal PNS components include sensory receptors, sensory fibers in nerves, sensory neurons in ganglia (somatic and autonomic), and nerve roots linking ganglia to a CNS component (brain or spinal cord). Importantly, sensory ganglia (eg, dorsal root ganglia [DRG], spiral ganglia [SG], trigeminal ganglia [TG]) as well as various autonomic ganglia have been recognized as target organs for certain categories of pharmaceutical products. Agents that may affect somatic and/or autonomic ganglia include small molecule drugs like aminoglycoside antibiotics121,210 and some anti-neoplastic chemotherapeutics61,123,125 as well as some biomolecule-based, gene-specific therapies such as adeno-associated virus (AAV) vectors,95,99,165,179,226 antisense oligonucleotides (ASO), 11 and nerve growth factor (NGF) inhibitors.29,36,193 Accordingly, microscopic evaluation of ganglia should be considered as one aspect of screening for neurotoxicity during nonclinical development of new test articles (TAs) for some therapeutic modalities. Current best practices recommend that selected sensory ganglia and, where warranted, various autonomic ganglia should be collected at necropsy and submitted for histopathologic evaluation where TAs are predicted or known to produce neurological effects consistent with ganglionic damage. 28
Limited toxicologic pathology literature is available describing principal features of ganglion biology and pathology in laboratory animal species. The dearth of baseline information is an obstacle that impedes the efficient movement of promising therapeutic candidates with potential or likely TA-related ganglionic pathology through nonclinical toxicity testing. The Scientific and Regulatory Policy Committee (SRPC) of the Society of Toxicologic Pathology (STP) convened an expert working group (WG) to formulate and publish “points to consider” for evaluating, interpreting, and reporting of microscopic findings in ganglia (using DRG as the prototypic target tissue) for nonclinical toxicity studies. In addressing this charter, the WG considered that a key first step was to review neurobiology and diagnostic terminology for microscopic findings in PNS ganglia. The current paper reviews fundamental information on sensory and autonomic ganglion biology and pathology to ensure that toxicologic pathologists, toxicologists, and regulatory reviewers are equipped with a common core of basic knowledge needed to identify and characterize TA-related ganglionic effects during nonclinical safety testing. The “points to consider” regarding the evaluation, interpretation, and communication of pathologic findings in ganglia are available in a companion paper. 21
Comparative Neurobiology of Ganglia
Sensory ganglia contain the cell bodies of primary afferent neurons, the key links in the transfer of sensory information from the periphery to the CNS. The neurobiology of DRG is emphasized in this section since these organs are relatively easy to find and sample in common nonclinical species, damage to DRG is known to enhance nociception (ie, the perception of pain), 198 and the somatic sensory neurons in DRG are known targets for certain classes of small molecule and biomolecule therapeutics.61,95,99,123,125,179,226
The DRG (also termed “spinal ganglia”) are paired elements of the PNS associated with each spinal cord segment. The DRG neurons receive input via spinal nerves from sensory receptors throughout the body. Peripheral organs and tissues (eg, bones, muscles, skin, tendons) are monitored by specific DRG based on their locations relative to axial body segmentation (eg, dermatomes, myotomes, sclerotomes) expressed during development. 129 Many viscera from the esophagus to the colon supply sensory information to nearby DRG located in the cervical, thoracic, and upper lumbar regions. 44
The TG and SG are both microscopically and functionally similar to DRG. Typically, TG (also termed the cranial nerve [CN] V, Gasserian, or “semilunar” ganglion 249 ) may be considered for retention in nonclinical toxicity studies because they are relatively simple to collect after the brain has been removed. The TG have been demonstrated to exhibit drug-induced ganglionic pathology comparable with that reported in DRG.99,123 In contrast, the SG generally are collected only in special ototoxicity studies since their location buried within thick bone of the skull requires special tissue processing methods unsuitable for routine nonclinical studies.73,199 In general, evaluation of DRG (± TG) but not SG is a suitable approach for screening sensory ganglia for neurotoxicity in routine nonclinical studies.
Several autonomic nervous system (ANS) ganglia that include general visceral afferent (GVA) nerve fibers are assessed occasionally in nonclinical studies (on a case-by-case basis) to assess potential ANS effects of TAs. 28 The GVA nerve fibers transmit sensations from various internal organs and tissues; in general, GVA fibers co-localize with parasympathetic (“digest and rest”) or sympathetic (“fight or flight”) nerves even though the GVA components act as sensory elements rather than serving parasympathetic or sympathetic effector functions. 197 Principal ANS ganglia that harbor GVA nerve fibers include the geniculate ganglia of the facial nerve (CN VII); distal glossopharyngeal ganglia (CN IX, also termed the “petrosal ganglia”); and distal vagal ganglia (CN X, also designated the “nodose ganglia”). The cranial cervical ganglia (the proper name in quadrupeds 112 for the “superior cervical” ganglia [SCG] of bipeds) in the cranial neck and/or the cervicothoracic ganglia (the proper name in quadrupeds 112 for the “inferior cervical” or “stellate” ganglion of bipeds) at the cranial end of the thorax are large sympathetic ganglia that are fairly easy to collect. The distal vagal (nodose) ganglia and SCG are located adjacent to each other bilaterally in the deep neck tissues near the ventral surfaces of cervical vertebrae, so in many cases, institutions choose to harvest and process these ganglia together.28,178
General Organization of Sensory Ganglia
Anatomically, the DRG in vertebrates are paired PNS organs associated with each spinal cord segment. Each DRG is positioned between a centrally located dorsal spinal nerve root (comprised of multiple rootlets) and a somatic nerve (formed by the convergence of the dorsal spinal nerve root distal to the DRG and the corresponding ventral spinal nerve root) (Figure 1). 234 The portions of the dorsal spinal nerve roots near the DRG are myelinated by Schwann cells while the spinal nerve rootlets near the dorsal root entry zone (DREZ, where the spinal nerve root intersects with the surface of the spinal cord) are myelinated by oligodendrocytes. The DRG cohesiveness, size, and positioning relative to the intervertebral foramen varies with the location of the spinal cord segment. For instance, the DRG associated with the cervical and lumbar intumescences (ie, the main spinal cord segments connected to the forelimb and hind limb somatic nerve trunks, respectively) are larger than those of other spinal cord segments because they support sensation for larger masses of tissue compared with other spinal cord segments that do not contribute to limb nerves.84,129 The DRG sometimes may be small or even absent depending on the cord segment, species, and individual.84,225 In humans, individual DRG may be divided into multiple distinct parts. 84
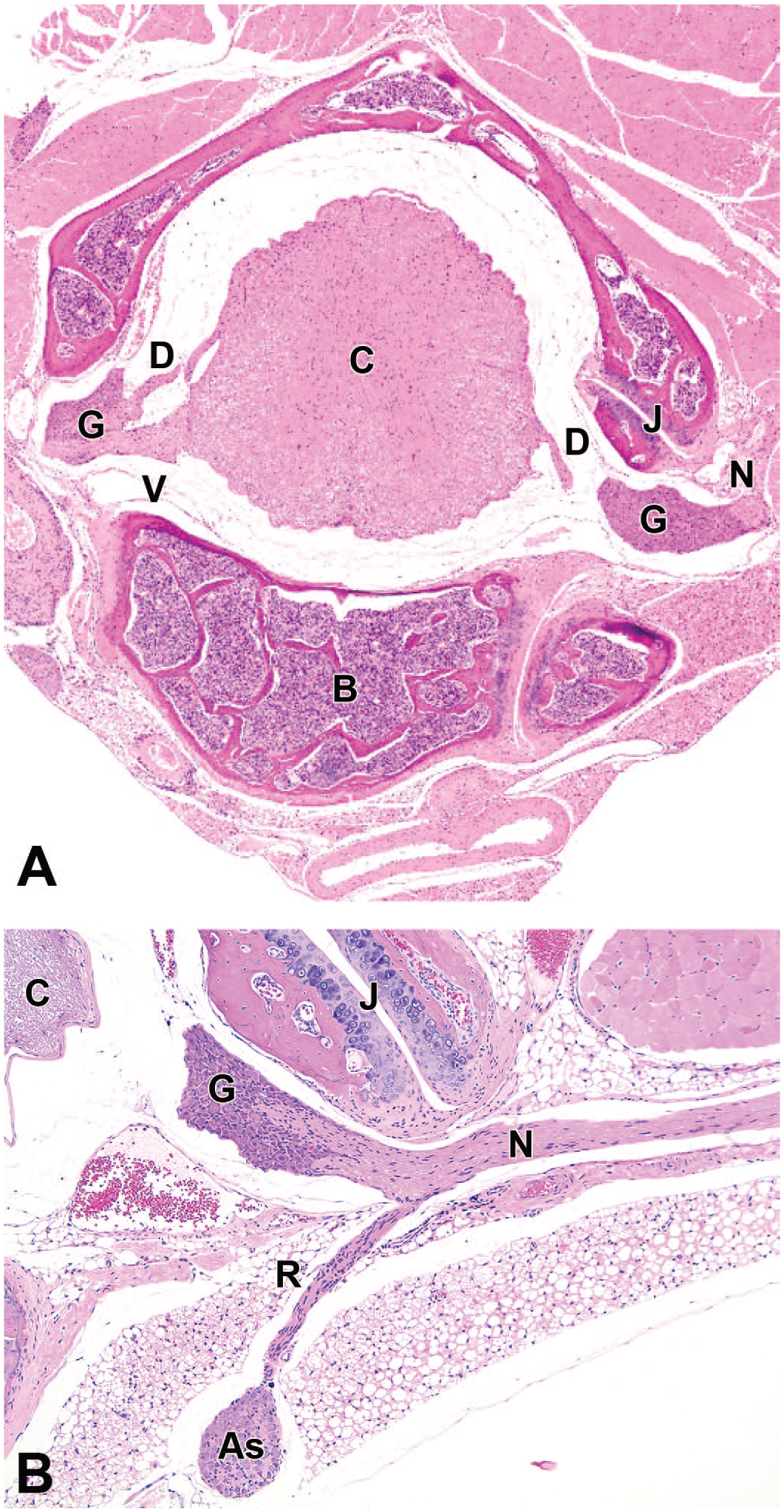
Regional neuroanatomy of a thoracic dorsal root ganglion (DRG) and connected portions of the peripheral nervous system. Each DRG (G) is connected proximally to the spinal cord (C) by dorsal (D) and ventral (V) spinal nerve roots and supports a somatic nerve (N) distally. Autonomic (sympathetic) nerve fibers depart the DRG via a communicating ramus (R) to enter a sympathetic chain ganglion (As). Other abbreviations: B = vertebral body, J = intervertebral joint. Species: adult C57BL/6 mouse. Hematoxylin and eosin (H&E). (Image in panel B is reproduced with adapted legend from Snyder et al, 208 by permission of Elsevier.)
The DRG consist chiefly of intermingled sensory neurons and supporting satellite glial cells (SGCs) enmeshed in a dense parenchyma of nerve fibers interspersed with fine connective tissue stroma. 84 The size of DRG varies with the spinal cord segment; in the rat, the lumbar (L) 4, L5, and L6 DRG contain approximately 12,000, 15,000, and 14,000 sensory neurons, respectively. 200 The size of DRG also varies across species (in accordance with body size) as human DRG have approximately 4- to 5-fold more neurons per ganglion compared with the same ganglia in rodents. 85 In general, sensory neurons are concentrated near the DRG periphery while the center contains intersecting columns of nerve fibers, although this appearance can vary depending on the plane of section.84,85 The numbers of sensory neurons may vary considerably in paired DRG for a given spinal cord segment. 246 Neurons in DRG range in size from 10 to over 100 µm in diameter (depending on age 172 and species), and neurons of all sizes are interspersed in the neuron-rich regions of sensory ganglia, with the bigger neurons frequently concentrated near the capsule. 159 Despite the large size of the neurons in the DRG, the bulk of the cytoplasm and cell support machinery (eg, endoplasmic reticulum and mitochondria) are distributed in the long axonal processes within the CNS and PNS rather than in the neuron cell bodies. 54 Other non-neuronal cells found in DRG include Schwann cells, endothelial cells, fibroblasts, and leukocytes (mononuclear cells [macrophages > lymphocytes] and, in rodents, scattered mast cells). In normal DRG (Figure 2), the numbers of sensory neurons (and their processes) and SGC dwarf other cell populations.
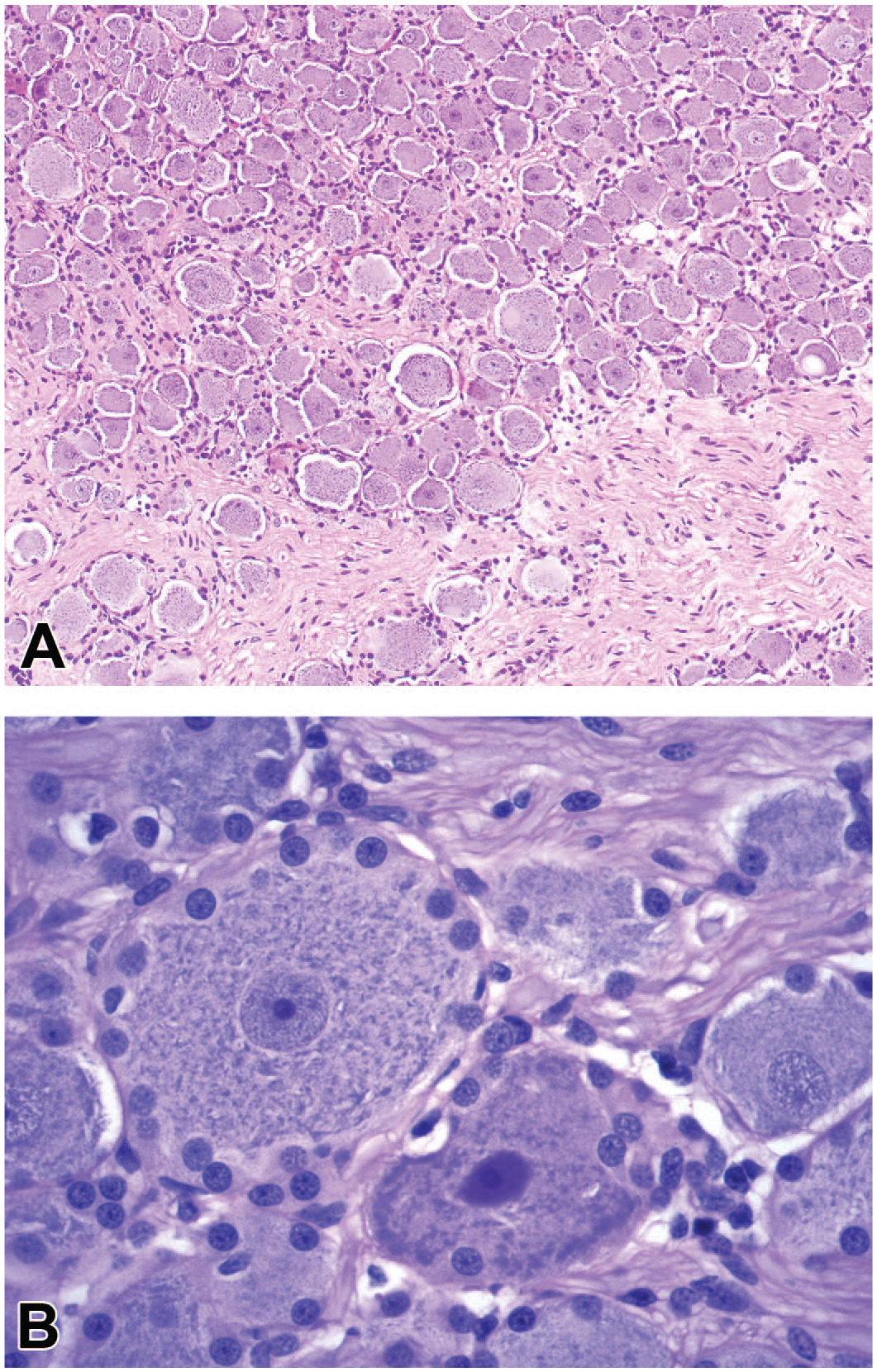
Structural features of a representative dorsal root ganglion (DRG). Panel A: Sensory neurons of various sizes are characterized by large central nuclei with large nucleoli and abundant cytoplasm. Panel B: Large sensory neurons contain abundant basophilic cytoplasmic granules (comprised of rough endoplasmic reticulum [“Nissl substance”]) and are encircled by an interrupted monolayer of small, round satellite glial cells. Both panels: Pale eosinophilic nerve fibers form tightly packed columns that traverse the peripherally located neuron fields. Site: lumbar DRG. Species: cynomolgus monkey. Hematoxylin and eosin (H&E). (Image in panel B reproduced with adapted legend from Pardo et al, 183 by permission of Sage.)
Neurons of the DRG are pseudo-unipolar cells with a single myelinated process that extends from the soma and soon thereafter bifurcates into centrally projecting afferent nerve fibers in the dorsal spinal nerve roots that enter the spinal cord at the DREZ while the peripherally projecting fibers populate the somatic nerves (Figure 3). The anatomic organization of the spinal nerve roots depends on their location relative to their DRG. The caudal spinal nerve roots (both dorsal and ventral) associated with the lumbar and sacral segments of the spinal cord are longer than more cranial spinal nerve roots because the vertebral canal is longer than the spinal cord; shortening of the cord during development results in species-specific cranial displacement of the lumbar and sacral segments, especially in rodents and primates compared with rabbits, dogs, and minipigs.21,28 The “cauda equina” is the designation for the collection of spinal nerve roots located caudal to the “conus medullaris” (ie, the tapering sacral spinal cord), and the nerve roots in this region are subject to procedure-related trauma when TAs are delivered directly into the “lumbar cistern” (ie, the cerebrospinal fluid [CSF]-filled subarachnoid space in which the cauda equina resides). The dorsal spinal nerve root is a pure sensory nerve and harbors most DRG-derived axons,53,129 but the ventral spinal nerve root (a predominantly motor nerve) does carry some afferent axons arising from DRG sensory neurons. 47 The number of afferent nerve fibers relative to the number of primary (“first order”) sensory neurons exhibits a 1:1 ratio in each DRG. 216 Subsequently, DRG axons enter the corresponding spinal cord segment and either turn cranially to ascend to the brain or form synapses with local “second order” interneurons and projection neurons at that level of the spinal cord (Figure 3). A subset of sensory neurons that convey touch and proprioception signals also send a collateral axon branch that turns cranially and ascends in ipsilateral white matter tracts of the dorsal funiculi of the spinal cord. 144 Ascending sensory nerve fibers in the dorsal funiculi are organized so that those arising from caudal (lower thoracic, lumbar, and sacral) DRG are located closest to the midline and synapse in the gracile nucleus while those contributed by more cranial (cervical to mid-thoracic) DRG are added in more lateral positions and synapse in the cuneate nucleus and lateral (accessory) cuneate nucleus; these two dorsal column nuclei reside in the medulla oblongata, the brain region that represents the rostral extension of the spinal cord.53,152 Additional collections of ascending nerve fibers with sensory information are found in the lateral funiculi (usually dorsolateral) and ventral funiculi; axons in these regions arise from projection neurons of the contralateral spinal cord gray matter that receive input from DRG and spinal interneurons (Figure 3).53,152 This arrangement means that evaluation of a spinal cord transverse section offers a relevant snapshot of the cumulative health of many DRG sensory neurons receiving input from proprioceptive and touch receptors (as described in more detail below) but does not effectively assess the integrity of primary nociceptive neurons, which terminate directly in the dorsal horn gray matter.
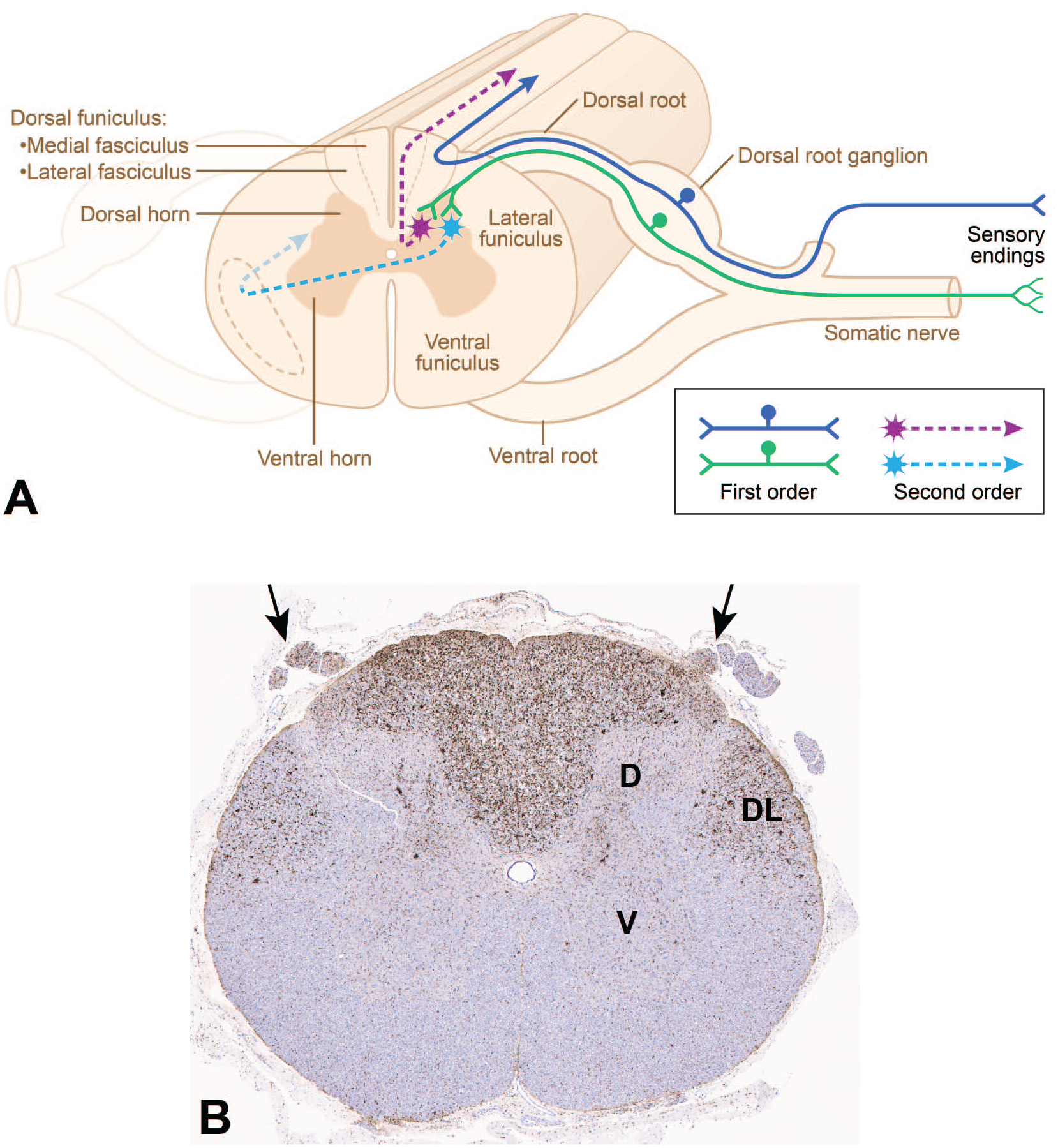
Neuroanatomic organization of the dorsal root ganglia (DRG) and their projections in the cervical spinal cord. Panel A: Sensory nerve fibers originate at peripheral receptors and extend to “first order” sensory neurons that reside in DRG. Some “first order” neurons send axons (solid dark blue fiber) through a dorsal spinal nerve root to directly enter the white matter of the ipsilateral dorsal funiculus (or posterior column) of the spinal cord, where they turn cranially to ascend to the brain. Other “first order” neurons (solid green) dispatch axons to the dorsal horn gray matter of the spinal cord where they synapse on “second order” interneurons. Axons from the “second order” neurons either ascend in the ipsilateral dorsal funiculus (dashed purple fiber) or project to the contralateral white matter to travel in the lateral funiculus (dashed turquoise fiber) or ventral funiculus. (The schematic diagram was prepared by Mr Tim Vojt.) Panel B: Microgliosis in the spinal cord white matter, shown by cell enlargement (nearly invisible at this magnification) with enhanced regional expression of the microglial marker IBA1 (ionized calcium-binding adaptor molecule 1), occurs as marked and diffuse labeling in the dorsal funiculi (due to degeneration of axons arising from “first order” DRG sensory neurons) and moderate and diffuse labeling in the lateral funiculi (resulting from degeneration of axons originating from “second order” interneurons in the dorsal horn [D]); axonal degeneration is also evident in the associated dorsal spinal nerve roots (arrows) secondary to DRG neuron degeneration and necrosis. Bilateral symmetry of IBA1 labeling is consistent with a TA-related effect while expression in both the medial and lateral fasciculi shows that neurons in both caudal and cranial DRG are affected. Scattered IBA1-positive microglial cells also are evident in the gray matter (mainly in the dorsal horn) in the interneuron layers that feed axons to the dorsolateral (DL) portion of the lateral funiculus. Study design: juvenile cynomolgus macaque given a single intrathecal injection in the lumbar cistern of an unspecified gangliotoxic agent. V indicates ventral (anterior) horn gray matter. (Figure reproduced with modified caption from Bolon, 27 by courtesy of Sage.)
Numerous blood vessels are present in DRG, and most are concentrated in neuron-rich rather than nerve fiber-rich regions. These blood vessels are lined by fenestrated endothelium, so the neurovascular interface (ie, “blood-ganglion barrier” or “blood-DRG barrier”) is porous and readily allows access to blood-borne molecules 192 ; the blood-ganglion barrier may be more permeable in neuron-rich regions in comparison with nerve fiber-rich domains, at least in the rat.97,148 Dense vascularization with permissive endothelium suggests that systemically administered low-molecular-weight and high-molecular-weight molecules are likely to have ready access to sensory neurons of the DRG.2,9,97,114,122 Blood vessels in TG are similarly lacking in an effective blood-ganglion barrier. 156 In contrast, vessels in the SG are reported to possess a conventional blood-brain barrier (BBB). 176 The BBB is a tight physical barricade comprised of non-fenestrated endothelium (joined by myriad tight junctions), pericytes (which are instrumental in maintaining the BBB), a continuous basement membrane surrounding both the endothelium and pericytes, and a dense network of astrocyte foot processes in close contact with the basement membrane.32,191,202,204 The blood-ganglion barrier in sympathetic ganglia resembles the conventional BBB and blood-SG barrier in that it is impervious due to the continuous (non-fenestrated) endothelium and numerous tight junctions between SGCs.40,217
The proximal portion of DRG (where the spinal nerve roots exit) and the entire TG are exposed to CSF as they reside partially (DRG) or completely (TG) within the subarachnoid space and are covered by leptomeninges. 190 There are cranial to caudal differences in the position of the DRG body relative to the intervertebral foramen, with cervical and cranial thoracic DRG located within the foramen while the more caudal DRG progressively lie more medial to the foramen. These regional differences appear to translate into a different extent of CSF proximity along different levels of the spinal cord and may influence the DRG exposure to TA administered into the CSF via the intracerebroventricular (ICV), intra-cisternal (intra-cisterna magna [ICM]), or lumbar intrathecal (IT) routes. 17 Moreover, it is reasonable to conjecture that administration of TA or viral particles directly into CSF may yield relatively higher cumulative exposure of the neurons in these sensory ganglia compared with intravenous (IV) systemic delivery. In vivo imaging studies have shown that CSF outflow in the cranial vault is removed via the arachnoid granulations and meningeal lymphatics and exits through the cribriform plate while caudally CSF flow follows the course of projecting somatic nerves to reach peripheral lymphatic vessels.149,187,212
The TG and SG vary from DRG chiefly in their locations. The main segments of the TG are located at the base of the brain on either side of bony ridges bordering the sella turcica (the central depression in the basal skull that harbors the pituitary gland). 105 The TG are also well-vascularized and surrounded by a fibrous capsule and meninges. Neurons in the TG are also pseudounipolar in conformation. The SG (also termed the “cochlear ganglia”) are encased in the dense bone (designated the “modiolus”) that forms the central axis of the cochlea in the inner ear. The dendrites of SG sensory neurons synapse with auditory hair cells in the spiral organ (of Corti) located at the center of the cochlea, after which the axons come together to form the cochlear nerve (CN VIII). 73 Neurons in the SG exhibit two conformations. 39 Approximately, 95% of SG neurons are bipolar (type I) cells with separate myelinated neurites (one axon and one dendrite) originating from two poles of the cell body; each type I SG neuron innervates a single inner hair cell (ie, the sensory receptor cells that transduce airborne vibrations into acoustic signals). The other 5% of SG neurons are pseudounipolar (type II) cells with one unmyelinated process that bifurcates to form a single dendrite and single axon shortly after leaving the cell body. Each type II SG neuron synapses with multiple outer hair cells (ie, sensory cells that sharpen frequency tuning for acoustic detection, especially for low-intensity sounds).
Autonomic sensory ganglia have multipolar neurons. 18 Such cells are characterized by multiple dendrites to gather sensory input and a single axon to transmit sensory output.
Resident Cell Types in Sensory Ganglia
With respect to risk assessment, consequential cell types in sensory ganglia include both neural and non-neural populations. Principal cells include the sensory neurons (and their processes) and SGC. The most important non-neural elements are those capable of mounting or regulating innate and acquired immune responses (ie, macrophages and lymphocytes). While other neural and non-neural components are found in ganglia, the cell types described here are emphasized in pathology evaluations of sensory ganglia because (1) they are needed to perform (neurons) or support (SGC) sensory function or (2) they have the capacity to disturb or destroy ganglionic function and/or structure (leukocytes).
Sensory neurons
The neurons of various sensory ganglia are similar but not identical in terms of morphology. Most sensory neurons are pseudo-unipolar in shape with a single axon “stem” that extends from the cell body and forms a T-junction with two major axon branches, one that courses toward the periphery and one that courses centrally toward the spinal cord. Signals are received at peripheral receptors on axon terminals and then transmitted in a retrograde fashion past the cell body and directly into the central axon branch to reach the spinal cord or brainstem. Sensory neurons in the DRG 84 and TG 166 as well as sensory neurons in the autonomic ganglia of cranial nerves VII, IX, and X adopt this pseudo-unipolar configuration. In contrast, in SG nearly all sensory neurons retain the embryonic bipolar conformation into adulthood in which an axon and a dendrite project from opposite ends of the cell body. 175 In DRG and TG, morphologically similar neurons that serve potentially distinct functions can be easily distinguished by their molecular signatures but less so by their cellular appearances.163,232
In terms of nonclinical toxicity studies, sensory neurons in DRG (and other somatic and autonomic PNS ganglia) exhibit a number of characteristic features when evaluated in a routine hematoxylin and eosin (H&E)-stained paraffin section. Sensory neurons typically have a large, slightly off-center nucleus with a single nucleolus and fairly abundant cytoplasm packed with basophilic granules of Nissl substance (rough endoplasmic reticulum) (Figure 2). Historically, neurons in sensory ganglia have been classified into subtypes using easily defined morphologic features: cell body size, axon diameter, and myelin sheath thickness (Table 1). 129 Using these criteria, A-alpha (Aα) and A-beta (Aβ) neurons have large cell bodies, large-diameter axons, and thick myelin sheaths; A-delta (Aδ) neurons have medium cell bodies, medium-diameter axons, and moderate myelin sheaths; and C neurons have small cell bodies, small-diameter axons, and thin myelin sheaths. The traditional structure-function correlates for this classification scheme are that Aα and Aβ neurons carry high-velocity sensations, such as proprioception and touch, while Aδ and C neurons transmit low-velocity sensations, such as nociception and thermoreception, and also serve a visceral sensory function. However, this classification scheme is an oversimplification as there are at least 15 different sensory neuron subtypes,141,256 and both touch and pain neurons can have Aβ, Aδ, or C fiber types. 49 Recent advances using alternative morphologic, neurophysiologic, and molecular criteria have shown that sensory neurons are far more diverse (Figure 4) than can be seen in conventional H&E-stained sections. Moreover, neurons with no histologic evidence of injury can exhibit molecular signatures associated with pain and contribute to hypersensitivity and pain. 92 Therefore, effort expended in trying to associate TA-related findings in ganglia to effects on a given morphologic subtype of sensory neurons using cytoarchitectural alterations on H&E-stained sections is not useful as a routine practice during nonclinical toxicity studies.
Historical morphology-based classification of sensory neurons and nerve fibers.
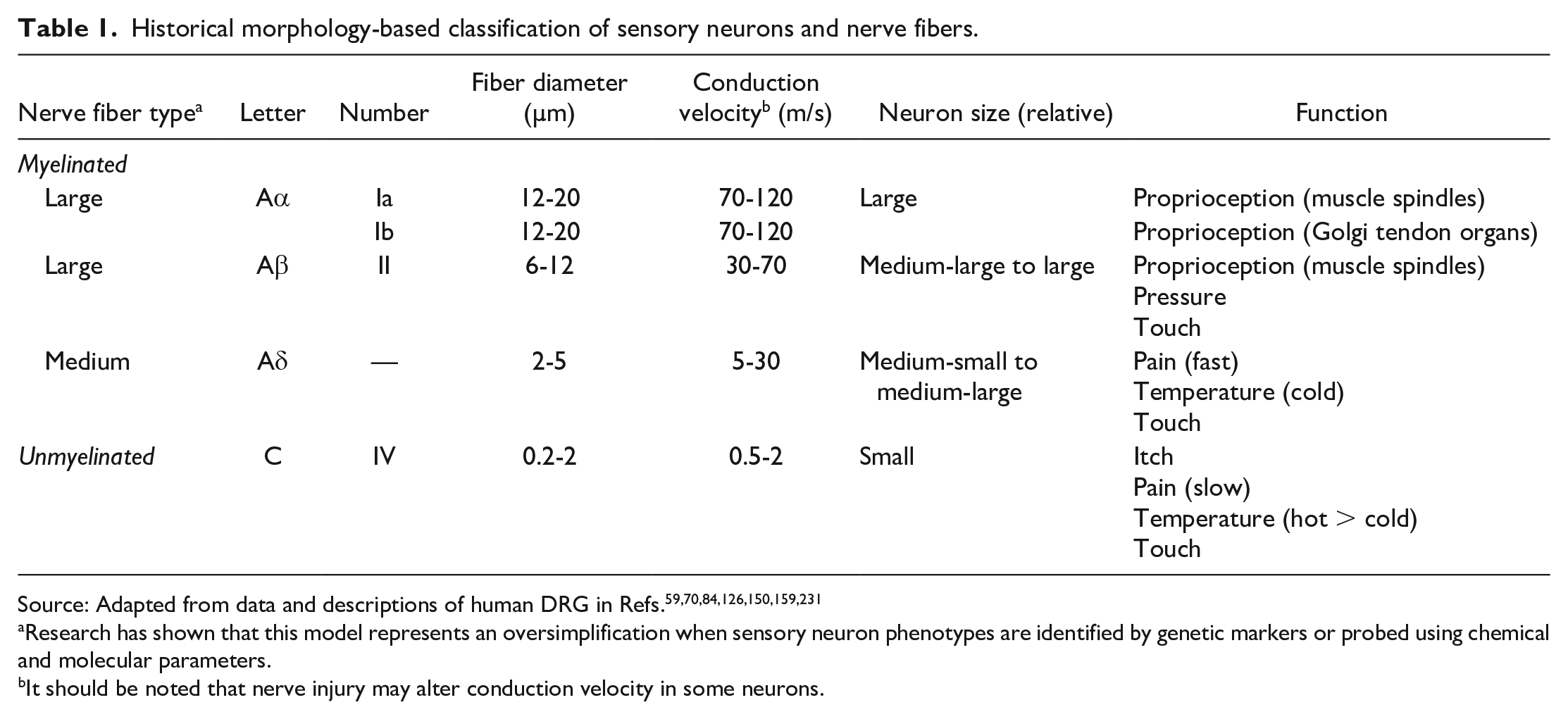
Research has shown that this model represents an oversimplification when sensory neuron phenotypes are identified by genetic markers or probed using chemical and molecular parameters.
It should be noted that nerve injury may alter conduction velocity in some neurons.
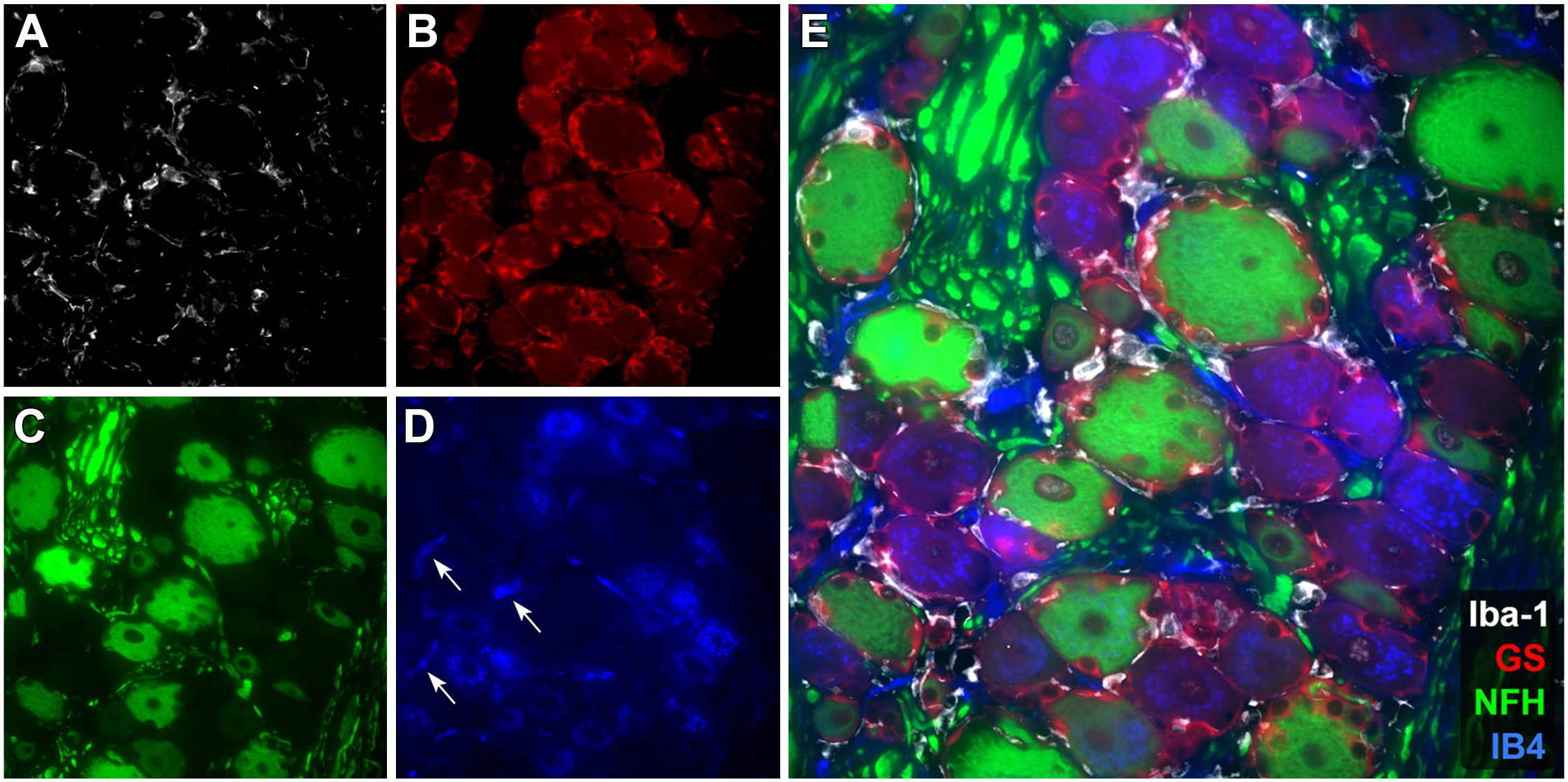
Multiplex pseudo-colored labeling by fluorescence immunohistochemistry (IHC) demonstrates several neuronal and non-neuronal cell types within the DRG. Panel A: Ionized calcium-binding adaptor molecule 1 (Iba-1, white) is used as a universal macrophage-lineage marker to identify resident macrophages within the DRG, adjacent to neuronal cell bodies and occasionally intermingled with satellite glial cells. Panel B: Glutamine synthetase (GS, red) demonstrates diffuse cytoplasmic labeling of satellite glial cells that form a ring or basket surrounding neurons. There is variable minimal labeling of neuronal somata and a notable lack of labeling of Schwann cells that are found within bundles of axons. Panel C: Neurofilament heavy chain (NFH, green) shows strong diffuse cytoplasmic labeling of the somata and axons of large-diameter neurons, including several subtypes of A-beta (Aβ) and A-delta (Aδ) sensory neurons. Tangential sections through large-diameter neurons are noted by the lack of an unlabeled central nucleus. Panel D: Griffonia simplicifolia lectin I, isolectin B4 (IB4, blue) reveals diffuse punctate cytoplasmic labeling of a subset of small-diameter nociceptors (pain-sensing neurons). IB4 also demonstrates variable labeling of occasional capillaries (white arrows). Panel E: A merged view of Panels A-D showing that co-localization of these four markers is scarce. Site: sacral DRG. Species: unaffected cynomolgus macaque.
A gradually expanding catalog of molecular markers (Table 2) may offer the opportunity in the future to explore the sensitivity of various sensory neuron subtypes to TA administration and/or examine physiological responses related to TA-associated ganglionic injury (eg, immune responses to sensory neurons altered by gene therapy).1,49,167,188,227,256,258 Details on emerging markers for various neuronal subtypes can be found in recent literature.141,195,215,256 For example, neurofilament heavy chain (NfH; 200 kD isotype) is expressed chiefly by large, moderately to heavily myelinated neurons (Aβ and Aδ subtypes) in many species, including macaques (Figure 4), marmosets, mice, rats, rabbits, and dogs (L.K. Crawford, unpublished data). 134 In contrast, NfH is pan-neuronal in sensory neurons of humans and horses (L.K. Crawford, unpublished data).84,174 Increased serum concentrations of neurofilament light chain (NfL) accompanied nerve fiber degeneration in sensory nerves of dogs with no functional (clinical or electrophysiological) abnormalities following long-term treatment with a neuroactive pyridazine derivative (branaplam), which suggests that NfL also might be a suitable biomarker for monitoring PNS toxicity. 218 Specific markers for subtypes of touch neurons are historically lacking, which has hindered evaluation of mechanisms underlying touch, paresthesia (ie, abnormal sensation), and allodynia (ie, pain experienced from a normally non-painful stimulus). Small-diameter pain neurons are easier to identify with histochemical stains, such as Griffonia simplicifolia lectin I, isolectin B4 (IB4) labeling or various immunohistochemistry (IHC) methods, though some markers (including IB4) can alter their labeling patterns, becoming less specific for pain neurons after nerve injury or inflammation (a phenomenon known as “phenotype switching”). 49 Molecular signatures of neuron damage and pain due to nerve injury may eventually help yield insights that cannot be attained on H&E-stained sections alone. For example, axotomized but restoration-competent (ie, still viable) sensory neurons may exhibit intra-nuclear expression of activating transcription factor 3 (ATF3) prior to the onset of overt structural changes in the neuronal cell bodies.108,206 The molecular responses to injury differ slightly between the DRG and TG 163 and so are likely to diverge for other sensory ganglia as well. At present, it is unknown if some sensory neuron subtypes in one or more PNS ganglia are more sensitive to small molecule or biomolecule therapies. 83 Therefore, advanced IHC and in situ hybridization (ISH) markers currently should be considered as an exploratory tool for possible use in investigational studies (similar to any non-validated soluble biomarker in blood or CSF) rather than as a routine procedure to implement during nonclinical toxicity testing. Finally, neurons may express unexpected molecules. For example, in response to immunization, DRG sensory neurons in mice have been shown to sequester antibodies (chiefly immunoglobulin G1 [IgG1]), suggesting that neurons may collaborate with the immune system in regulating antigen trafficking and antigen-mediated responses. 83
Routine and special stains that may be used for characterizing changes in ganglia.
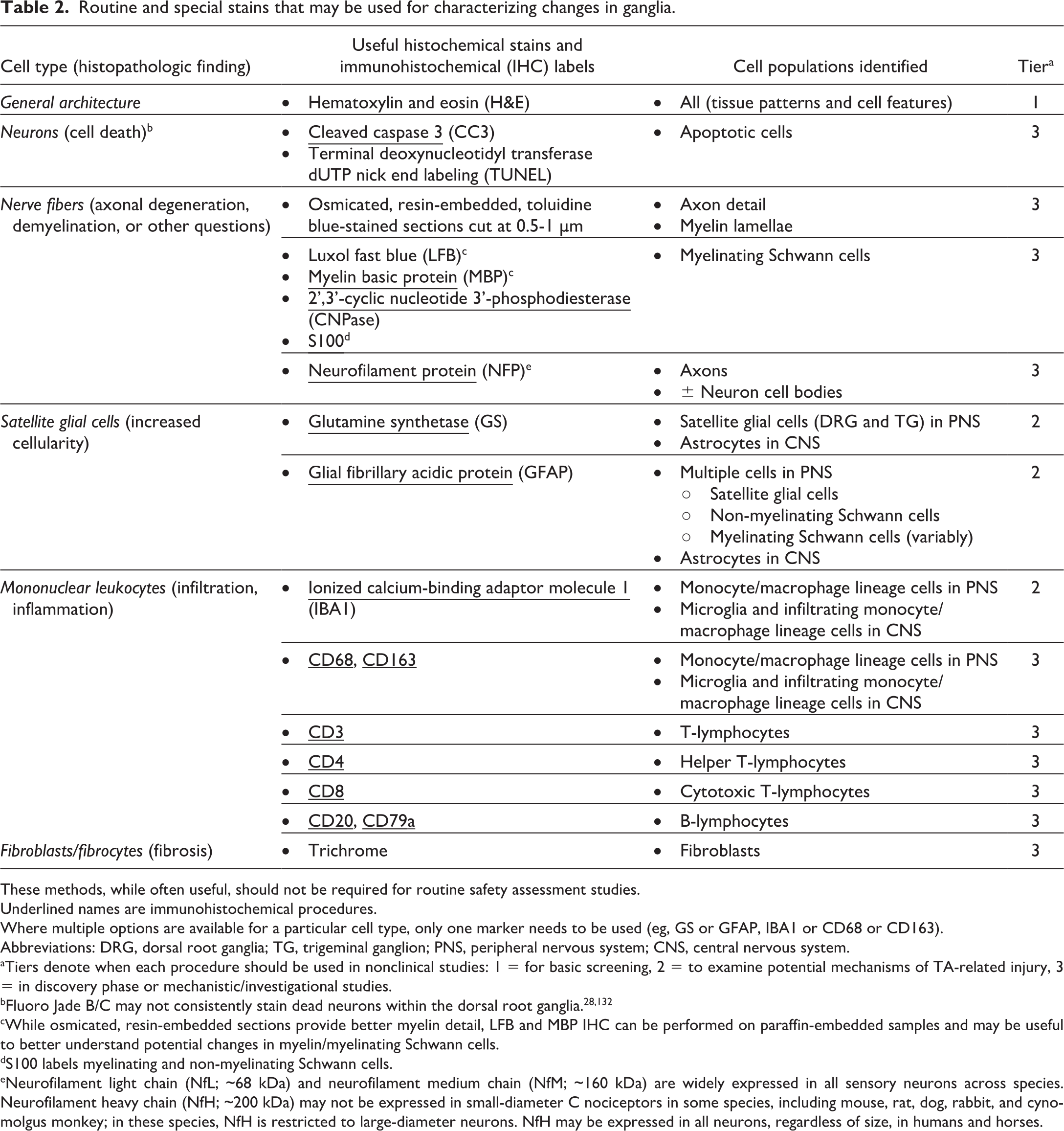
These methods, while often useful, should not be required for routine safety assessment studies. Underlined names are immunohistochemical procedures. Where multiple options are available for a particular cell type, only one marker needs to be used (eg, GS or GFAP, IBA1 or CD68 or CD163).
Abbreviations: DRG, dorsal root ganglia; TG, trigeminal ganglion; PNS, peripheral nervous system; CNS, central nervous system.
Tiers denote when each procedure should be used in nonclinical studies: 1 = for basic screening, 2 = to examine potential mechanisms of TA-related injury, 3 = in discovery phase or mechanistic/investigational studies.
While osmicated, resin-embedded sections provide better myelin detail, LFB and MBP IHC can be performed on paraffin-embedded samples and may be useful to better understand potential changes in myelin/myelinating Schwann cells.
S100 labels myelinating and non-myelinating Schwann cells.
Neurofilament light chain (NfL; ~68 kDa) and neurofilament medium chain (NfM; ~160 kDa) are widely expressed in all sensory neurons across species. Neurofilament heavy chain (NfH; ~200 kDa) may not be expressed in small-diameter C nociceptors in some species, including mouse, rat, dog, rabbit, and cynomolgus monkey; in these species, NfH is restricted to large-diameter neurons. NfH may be expressed in all neurons, regardless of size, in humans and horses.
Satellite glial cells
The SGC are the resident glia of sensory ganglia. These neural crest-derived, flattened cells and their basement membranes form a monolayer sheath that completely encircles, is intimately associated with, and sometimes indents the plasma membranes of their sensory neurons (Figures 2 and 4). 159 The separation between the extracellular membranes of SGC and their neurons approximates the width of a synapse (ie, ~20 nm). Numerous interdigitated microvilli increase the cell contact area by up to 40%.45,65,180 The close proximity permits regular and direct cross-talk between neurons and SGC so that each cell type influences the activity of the other, despite the absence of synapses between these two cell types. 91 Materials including ions, macromolecules, neurotransmitters, and toxicants rapidly and freely pass through the encircling SGC monolayer to reach neurons.88,91,107
Biologically, SGC fulfill many tasks in sensory ganglia that are relegated to distinct glial cell subtypes in other portions of the nervous system. The hybrid character of SGC is emphasized by their possession of functional, molecular, and/or structural properties that overlap with attributes of astrocytes, oligodendrocytes, macrophages, and Schwann cells. Key SGC functions similar to those of CNS astrocytes include metabolic support of neurons, microenvironmental regulation (including neurotransmitter clearance), and modulation of sensory input (especially pain).88,89,91,159 Like microglia, SGC serve as facultative (ie, inducible) phagocytes and antigen-presenting cells, and they can produce cytokines that activate nearby neurons and SGC as well as modulate local immune responses by resident and infiltrating leukocytes.84,160,230,238 Multiple SGC subpopulations have been identified in somatic and sympathetic ganglia using molecular markers. 158 Moreover, the distinct SGC subpopulations serve divergent functions; in DRG, some support nerve fiber restoration in peripheral nerves while others mediate nerve fiber repair in spinal nerve roots. 8 Interestingly, SGC in SG (but not in other sensory ganglia) are able to maintain myelin sheaths around neuronal processes. 91
The SGC express many proteins indicative of their status as multi-functional (hybrid) glia (Table 2). For instance, SGC display many astrocyte markers like glial fibrillary acidic protein (GFAP), an intermediate filament; glutamine synthetase (GS), an enzyme involved in metabolism of the neurotransmitters glutamate and gamma amino butyric acid (GABA); connexin 43 (Cx43), a gap junction protein; potassium channel-inwardly rectifying, subfamily J, member 10 (KCNJ10, colloquially known as Kir4.1); and S100B.88,91,92,181 In like manner, SGC express oligodendrocyte markers, such as the transcription factor sex-determining region Y (SRY)-related HMG-box 10 (SOX10) and the myelin-associated enzyme 2,’3’-cyclic nucleotide 3’-phosphodiesterase (CNPase); macrophage plasma membrane proteins, including major histocompatibility complex (MHC) I and MHC II as well as CD40 and CD54; and Schwann cell markers like cadherin 19 (CDH19) and S100.88,91,92,181 Detection of certain glial markers (eg, CNPase, GFAP, GS) in SGC by IHC varies across species. 222 In the face of injury to sensory neurons and/or their axons, activated SGC in DRG and TG increase the numbers of gap junctions to boost their cooperation with nearby SGC and enhance electrical coupling to the neurons they surround, upregulate their expression of such glial markers as GFAP and Kir4.1, and start to proliferate.25,26,58,87,90,224,236,238 In some conditions, activated SGC also may upregulate expression of non-glial proteins such as CD45 (a marker for circulating leukocytes) and CD163 (a marker for resident macrophages). 57
During postnatal development in rats, increases in sensory neuron numbers have been reported in DRG and TG, with the degree of proliferation varying with the sensory neuron subtype64,136 and location of the injury. 55 However, to date the supporting information for in vivo neuronogenesis in sensory ganglia remains controversial, due to both technical differences in quantitative methods and evidence that the lineage of regenerating cells (eg, glial or neural) appears to differ with the nature of the injury.50,157 A role for SGC in neuronogenesis in adult rodents has been suggested by cell proliferation in DRG explant cultures under neurotrophic conditions. 142 In young adult rats, unilateral transection of the sciatic nerve results in loss or delay in proliferation of DRG neurons on the transected side with a gradual post-operative increase in neuronal count in the contralateral control DRG consistent with normal development. 55 In chronic pain models induced in adult mice by unilateral injection of complete Freund’s adjuvant or transection of a distal (fibular, plantar, or tibial) nerve, SGC proliferation is associated with expression of stem cell genes such as nestin, platelet-derived growth factor receptor alpha (PDGFRα), and/or SRY-related HMG-box 2 (SOX2) in the ipsilateral DRG; interestingly, proliferating neurons are detected in the DRG contralateral to the site of injury. 254 Similar SGC responses have been reported in young adult and adult rodents within several months after physical (transection 55 or trauma82,170), inflammation-mediated, 254 or chemically induced (capsaicin51,74) damage to nerve fibers, suggesting that SGC-related neuronogenesis may have a limited role in postnatal replenishment of DRG sensory neurons. Consistent with these reports in rodents, SGC responses and intercellular induction of neuronal neurite formation have also been described in DRG explants of dogs. 222 These examples of inducible renewal of PNS ganglionic neurons is in contrast to most neuronal populations within the CNS, where neurons are not added beyond their respective developmental windows with only rare exceptions (eg, dentate gyrus of the hippocampus and olfactory bulbs). Regardless, it is unclear if neuronogenesis occurs in sensory ganglia of primates (including humans), if so to what extent depleted neuron populations may be restored, and for how long after individuals reach maturity that effectual neuronal replacement might occur.
Schwann cells
Schwann cells are specialized glial cells that surround axons in the PNS, including peripheral nerves and the distal portions of spinal nerve roots. Subtypes of this neural crest-derived cell population are defined as “myelinating” or “non-myelinating” depending on the extent to which they produce myelin to insulate PNS axons. Schwann cells are not found in the first two cranial nerves, the olfactory nerve (CN I) and the optic nerve (CN II). Axons in CN I are encased in Schwann cell-like elements termed olfactory ensheathing cells (OEC)16,241 while CN II is actually a peripherally located CNS domain where myelin is produced by oligodendrocytes. 171 Axons in the most proximal portions of spinal nerve roots (ie, just after exiting the spinal cord) also are encompassed by oligodendrocytes. 183 There are also non-myelinating terminal Schwann cells that surround and support certain types of sensory axon terminals in the skin and thus play a role in re-innervation after local injury. 143
Schwann cell function in axonal maintenance and regeneration is a reciprocal endeavor of the neuron (mediated by the axon) and the neighboring glial cell. Ligands released by the axon or within the extracellular matrix impel Schwann cells to adopt a myelinating or non-myelinating phenotype, and the resulting myelin sheath prepares the axon for signal transduction.43,78,120,128,196 Myelinating Schwann cells exhibit different phenotypes when involved with sensory vs. motor axons as indicated by the constellations of growth factors that they produce. 98 Nerve (axonal) injury diverts myelinating and non-myelinating Schwann cells from maintenance roles into nurturing roles to sustain damaged axons. To accomplish this objective, Schwann cells de-differentiate and adopt an alternative program where myelin-related genes are downregulated, existing myelin is removed, and genes for producing cytokines (to recruit and activate macrophages and Schwann cells to clear debris) and trophic factors (to support axon regrowth) are upregulated.16,117-119 Interestingly, the spectrum of trophic factors generated by Schwann cells following axon injury varies in denervated nerves versus spinal nerve roots. 33 Regeneration of axons in spinal nerve roots results in their extension into the superficial spinal cord, after which their growth halts and presynaptic axon terminals establish unproductive connections with non-neuronal cells. 56 Mechanistic details of axon-Schwann cell interactions during health and disease and phagocytic properties of Schwann cells are beyond the scope of this paper but may be explored in the published literature.16,24,48,79,81,116,128,145,213,228
Various markers have been used to evaluate Schwann cell morphology and function (Table 2). For example, S100 is present in nuclei and the perinuclear cytoplasm of myelinating and non-myelinating Schwann cells, but expression is proportional to myelin thickness and so is more prominent in myelinating cells. 161 Myelin basic protein (MBP) and peripheral myelin protein 22 (PMP22) are key myelin components in nerves, nerve roots, and sensory ganglia, and SOX10 is a transcription factor regulating expression of myelin-related genes in Schwann cells. 115 Occasional myelinating Schwann cells express both MBP and S100 75 ; intriguingly, PMP22 may be seen in some sensory neurons. 153 Non-myelinating Schwann cells express GFAP. 250 Matrix metalloproteinase-9 (MMP-9) is upregulated in Schwann cells at sites of nerve injury 66 and may be involved in initiating and sustaining pain in damaged nerves. 130 Another important marker for Schwann cells is CNPase, which is expressed in all peripheral myelin at levels inversely related to myelin thickness.220,233
Macrophages
Resident macrophages are reported to comprise 5% to 20% of nucleated cells in DRG, serving to provide both immune surveillance for the organ and trophic support to sensory neurons.62,131,257 Normal turnover in adult mice results in attrition and replacement of most macrophages (80%) in DRG over a 12-week period. 169 In general, in nonclinical species (eg, rodents, dogs, nonhuman primates [NHPs]) and humans, macrophages cannot be discriminated reliably from SGC using morphologic features that are visible in H&E-stained sections, so it is likely that their number is underestimated based on routine microscopic evaluation. Instead, molecular markers may be used to distinguish these morphologically similar cells; for example, SGC express S100 while macrophages do not. 146 Microglia have been described in DRG based on the presence of non-SGC cells that strongly express the microglial marker ionized calcium-binding adaptor molecule 1 (IBA1). 184 However, IBA1 and other microglial markers (eg, CD11b, F4/80) are also expressed by peripheral macrophages.4,76 Though historically somewhat controversial, by convention macrophage-lineage cells in the DRG are termed macrophages despite the resemblance to microglia of the spinal cord and brain because microglial cells in the CNS have a different embryonic origin. 237 Accordingly, use of the term “microglial cells” should be avoided in describing sensory ganglia but instead should be applied only to resident macrophages in the CNS.
Under normal conditions, macrophages are located in specific positions relative to other ganglionic structures. Macrophages typically reside near the neuron-SGC units but outside the SGC basement membrane. Furthermore, macrophages often are found adjacent to blood vessels in ganglionic centers and nerve roots. 106 Upon acute injury to a nerve or ganglion, numbers of activated macrophages in the ganglion rise substantially, increasing by 4-fold to 8-fold within a week of nerve transection or induction of metabolic neuropathy (eg, streptozotocin-induced diabetes mellitus) in rats.106,221 This increase in DRG macrophages is known to play a crucial role in establishing chronic pain caused by nerve injury and chemotherapy-induced peripheral neuropathy.22,251,253 Removal of the inciting cause leads to a slow decline in both numbers and the activation state of macrophages in ganglia.
Lymphocytes
Resident lymphocytes are found in DRG in low numbers,10,62 where they comprise a portion of the mononuclear cells within the interstitium separating sensory neurons. In health, CD8-positive T cells appear to be the chief lymphocytic population in ganglia although fewer B cells also are present. 62 When warranted in cases of ganglionic damage, lymphocyte numbers may be supplemented by entry of immunocompetent T cells in response to damage-associated molecular patterns (DAMPs) or pathogen-associated molecular patterns (PAMPs).77,147,162 In vitro studies suggest that lymphocytes may contribute to regulating the neurotransmitter complement in sympathetic ganglia. 15 This effect may represent reciprocal feedback since autonomic neural activity has been suggested to alter both innate and acquired immune functions.20,247
Species Differences in Dorsal Root (Sensory) Ganglia Biology and Pathogenesis
Sensory ganglia are functionally and structurally equivalent across mammalian species, which permits animals to serve as meaningful models for basic investigations of PNS biology. That said, the growing catalog of subtle anatomic, genetic, and proteomic differences between humans and animals may have implications when translating nonclinical toxicity data for human risk assessment.84,124 This section will review selected features of sensory ganglia biology that differ among species, emphasizing DRG.
Ganglion size reflects body size and location. Therefore, human DRG are larger than rodent DRG. This difference reflects the greater numbers of neurons as well as the larger neuronal soma sizes necessary to appropriately cover the sensory needs of a larger body region. In contrast, the number of neurons in TG is reported to be higher in rats71,135 than humans, 137 perhaps because the vibrissae of the rodent snout make this body part a principal sensory organ in this species. The bimodal distribution of large and small neurons within human and rodent DRG is similar, but human DRG contain many more sensory neuron subtypes than are found in rodents. 84 The sizes of human sensory neurons are larger than those of rodents, likely because more protein synthesis machinery is necessary to sustain very long axons. 84 Human DRG contain more connective tissue fibers among the neurons and nerve fibers compared with rodents. 84 Interestingly, human DRG at some sites may be greatly reduced or entirely missing (cervical [C] 1, which is absent in about 70% of people) or divided into 2 or 3 small ganglia (usually associated with the L3 and L4 segments).84,205 Functional consequences of these variations (if any) are unknown. In particular, no reports have determined the functional reserve—the minimal number of neurons in sensory ganglia that are required to sustain normal function—for DRG or TG in either animals or humans. The challenges of these types of studies lie in the difficulty of detecting incremental loss of function—particularly in terms of sensory modalities, such as temperature, itch, touch, and proprioception—given the limited tools available to do so in conventional nonclinical studies. Differences in the transcriptome atlas for DRG have been documented among mice, guinea pigs, cynomolgus monkeys, and humans, 124 which highlights the potential for species-specific sensory neuron functions (especially in nociception) despite the conserved ganglion structure.
In general, one should also consider that rodents have higher metabolic rates than non-rodents and humans. Therefore, higher doses and/or longer dosing periods may be required in rodents to induce comparable degrees of peripheral neurotoxicity. 173 It is also important to note that mice are generally considered not very sensitive to peripheral neurotoxicity, and either parenteral TA administration of small molecule TAs and/or newborn animals may be needed to induce toxic peripheral neuropathy in mouse models.67,133,139 Whether a similar lack of sensitivity occurs for biomolecule TAs, such as AAV gene therapy vectors, is not known.
Class Effects of Selected TA Modalities on Sensory Ganglia
Ganglionic injury has been demonstrated to be a stereotypical effect for several TA classes. This section briefly discusses the key classes of TA that can damage sensory neurons (especially in DRG or TG) and reviews proposed pathogenic mechanisms by which they act.
Chemotherapy-induced peripheral neuropathy
Administration of anti-neoplastic chemotherapies is the longest known and best characterized variant of the TA-related peripheral neuropathies related to ganglionic injury. Many commonly used anticancer agents have been associated with peripheral neuropathy in nonclinical toxicity studies and/or in human cancer patients. Implicated agents include proteasome inhibitors (bortezomib, carfilzomib, and ixazomib); platinum-based compounds (cisplatin, carboplatin, and oxaliplatin); taxanes (paclitaxel, docetaxel, and ixabepilone); vinca alkaloids (vincristine, vinblastine, vinorelbine, and vindesine); alkylating agents (cyclophosphamide, hexamethylmelamine, ifosfamide, and procarbazine); and immunomodulatory agents (thalidomide, lenalidomide, and pomalidomide). Thus, molecules directed against a diverse array of molecular targets are capable of damaging ganglionic cells.3,7,61 Sensory neuron cell bodies (DRG) are considered the primary target of these xenobiotics. However, distal nerve terminals are affected by taxanes, vinca alkaloids, and immunomodulatory agents while nerve fibers are also affected by proteasome inhibitors and taxanes.7,61
Chemotherapy-induced peripheral neuropathy (CIPN) is known to have similar histopathologic features among humans, mice, and rats while fewer publications are available regarding non-rodent nonclinical species and their responses to these agents. The common feature in CIPN for affected species appears to be Wallerian-like retrograde (“dying back”) degeneration of nerve terminals and distal nerve fibers sustained by sensory neurons in DRG 61 ; the process is designated as “Wallerian-like” because true Wallerian degeneration is defined as retrograde nerve fiber fragmentation following physical trauma (eg, transection) to a distal axon. 194 Features of Wallerian-like degeneration have been reported with paclitaxel, vincristine, and thalidomide.113,164,207 At the subcellular level, toxic mechanisms evident in DRG neurons include altered energy metabolism, indicated by mitochondrial swelling (Figure 5) and small nuclear and/or cytoplasmic vacuoles, cytoskeletal disruption, DNA damage (evident over time as nuclear condensation and fragmentation), and lipid layer disorganization leading eventually to disintegration of organelle and plasma membranes.155,252 SGC activation and inflammation are also prominent features.154,238,252 Injury to individual ganglionic neurons has been linked to morphological alterations affecting entire ganglia. For example, increased ganglion volume (“hypertrophy”) has been documented using magnetic resonance imaging (MRI) in lumbosacral DRG in human patients with oxaliplatin-associated sensory neuronopathy. 6
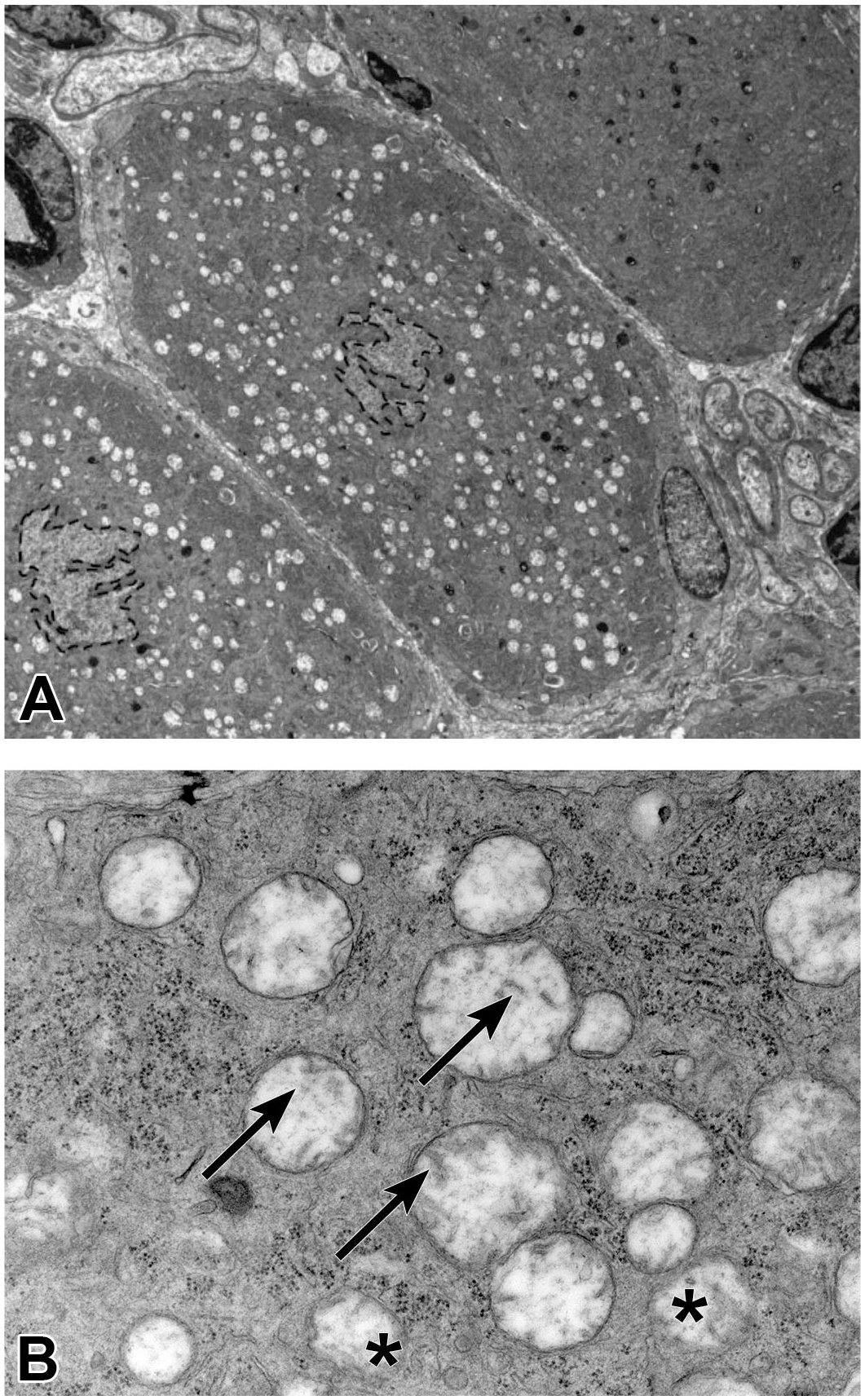
Mitochondrial swelling in large DRG sensory neurons is an early manifestation of chemotherapy-induced peripheral neuropathy (CIPN). Panel A: Compared with a partial unaffected neuron (upper right), two affected neurons (middle and lower left) exhibit multiple round, electron-lucent (white), cytoplasmic vacuoles encircling a central nucleus with jagged border and no heterochromatin or nucleolus. Panel B: The vacuoles represent swollen mitochondria with electron-lucent matrix and disorganized, fractured, and/or missing cristae (arrows) and sometimes a variably attenuated and/or indistinct double membrane (asterisk). These ultrastructural mitochondrial alterations correlate well with the light microscopic finding of sensory neuron degeneration. Site: thoracic DRG. Test article: unspecified gangliotoxic small molecule. Species: Wistar Han rat. Magnifications: Panel A = 2550X, Panel B = 26,500X. DRG indicates dorsal root ganglia.
Abnormal neurological signs accompany structural damage to ganglia in many species. Sensory polyneuropathy induced by anti-neoplastic chemotherapy is a common dose-limiting toxicity in human patients.5,235 Rodent CIPN models are reported to exhibit exaggerated reflex withdrawal responses, hypersensitivity in paw withdrawal in the presence of mechanical stimuli, electrophysiological abnormalities, and decreased nerve action potentials and nerve conduction velocity (NCV) as well as microscopic features similar to the condition in humans.61,86 In mice, treatment for 2 weeks with paclitaxel, eribulin mesylate, or ixabepilone is associated with changes in DRG including neuronal degeneration, characterized by dark perinuclear cytoplasmic inclusions and/or cytoplasmic vacuolation and/or swelling; degeneration of SGC; and axonal degeneration of small-diameter and large-diameter fibers in the sciatic nerves. 244 These axonal changes in nerves correlated with reduced caudal NCV, caudal nerve amplitude, and digital nerve amplitude for paclitaxel and ixabepilone but not for eribulin mesylate. 244 Similarly, in studies of up to 2-week duration with an oncology indication, a small molecule (non-platinum) TA administered via parenteral routes (IV infusion, IV bolus, or subcutaneous [SC] injection) in Wistar Han rats resulted in adverse clinical signs, including splayed posture, limited use of hind limbs, ataxia, abnormal gait, and related observations indicative of neurological dysfunction. Microscopic findings in the DRG and nerves of affected rats consisted of sensory neuron degeneration, peripherally displaced or absent nuclei, and axonal degeneration.
Adeno-associated virus (AAV) vector gene therapy
In nonclinical toxicity studies with AAV gene therapy vectors, TA-related microscopic findings have been reported in sensory ganglia (DRG and TG) of NHPs, including juvenile and adult cynomolgus and rhesus macaques23,63,95,96,99,100,103,104,165,179,226; piglets95,240; young adult rabbits46,219; and young adult rodents (rats and to a lesser extent mice).42,63,179,229 Similar changes have been detected in sympathetic ganglia of rats. 179 The main findings in sensory and (where examined) autonomic ganglia usually include neuronal degeneration and necrosis, nerve fiber degeneration (considered to result from secondary damage to axons following primary injury to neurons), SGC increased cellularity, and mononuclear cell infiltration or inflammation. Leukocytes enter DRG as early as 5 days after TA injection, with macrophages and certain lymphocyte classes (eg, natural killer [NK] cells, CD4+ helper T cells, B cells) entering first. 93 Inflammation participates in neuronal degeneration,93,99 but neuronal injury may develop in the absence of substantial leukocyte infiltration or inflammation (especially in mice). The findings are thought to arise from primary injury to ganglionic neurons since these cells readily take up AAV vectors and highly express transgenic DNA, RNA, and protein165,179,201,245,248; different AAV serotypes have been reported to target distinct neuron populations even when administered by the same route of delivery. 245 The pathogenesis of AAV-related microscopic findings in sensory ganglia of NHPs is complex due to the influence of multiple factors including dose, promoter strength, age of the animal, route of administration, and immunogenicity of the transgene product.99,179,201,240,242 Active neuronal injury appears as early as 4 days after dose administration by the IT route, and the incidence of active injury decreases to some extent some months after AAV administration (3-6 months, depending on the species and TA).41,42,99,179 With injection into the CSF (eg, ICV, cisterna magna [ICM] or lumbar cistern [IT] administration), the finding is generally most severe at the lumbar and sacral levels, followed by the cervical level, and least severe at the thoracic level. 243 Thus, DRG at any spinal cord level and TG may be affected by direct CNS delivery via any route (ICV, ICM, or IT) as well as by peripheral (eg, intraperitoneal [IP], IV > intramuscular [IM], SC) delivery, but caudal DRG appear to be more vulnerable.99,151,223,255 Transgene expression may be highly targeted and generally limited to sensory neurons in specific DRG by direct intra-ganglionic (“intraparenchymal”) injection,68,186,248 which is one approach to managing neuropathic pain.69,110,186 Morphologic changes in DRG are not accompanied by in-life neurological signs in nonclinical species, although changes in NCV have been reported in a few studies following direct CNS delivery of extremely high AAV doses.42,99
In general, mice are considered to be adequate test systems for efficacy and safety of AAV-based gene therapies, but age, route of administration, and/or other factors (transgene, AAV serotype) may influence the development of microscopic findings in sensory ganglia as much or more than the selection of the nonclinical species. In the experience of some WG members, mice seem to be less sensitive compared with NHPs for sensory ganglia microscopic findings induced by direct CNS or parenteral administration of similar doses (scaled by brain weight and body weight, respectively) of AAV (generally evaluated for AAV9) gene therapy vectors that carry human-origin transgenes. 99 Rats can develop ganglionic inflammation following exposure to AAV-based products to a degree comparable with that of NHPs, 42 but the degree of inflammation in some cases is modest or develops at a different pace compared with the more robust inflammatory changes that occur in NHPs. 179 Induction of TA-related findings may require higher AAV doses and longer duration studies. This possibility requires further investigation since nonclinical studies for AAV-based TA have only recently become more systematic in their collection and evaluation of DRG and other sensory ganglia, particularly in rodents. In selected circumstances where both rodent and NHP data were generated, the ages of the animals were not similar (eg, newborn rodents vs peri-pubescent NHPs) and/or routes of administration were not similar. The few reported cases of AAV9-related DRG findings in murine models involve direct CNS delivery of AAV9 vector genomes bearing the transgene for a highly immunogenic protein (eg, green fluorescent protein [GFP] 201 ) or that resulted in atypical accumulation of highly overexpressed DNA, RNA, or protein.179,229 Similarly, a single report of AAV-induced DRG pathology in three piglets (7-30 days old at the time of injection) was associated with IV injection of a very high TA dose. 95 It is the experience of some WG members that neuronal degeneration and necrosis in the DRG with no to minimal inflammation is the typical AAV-related finding in mice, and less often rats. Interestingly, the only current evidence of late-onset neurological signs following administration of an AAV9 TA was reported in a transgenic mouse model, in which sensorimotor deficits developed months after a single ICV injection due to long-term overexpression of a transgene. 229
The mechanism of AAV-associated sensory ganglia pathology is not completely understood and is likely multifactorial. AAV-associated sensory ganglia pathology has been hypothesized to result from transgene overexpression34,100,179,226 and/or an immune response mounted against the transgene product or vector capsid.60,185 AAV-mediated transgene overexpression with ensuing unfolded protein response (UPR)-mediated cell death is currently considered the leading hypothesis of primary AAV-associated DRG findings in NHPs.99,100 In a recent meta-analysis, the severity of AAV-associated findings in the sensory ganglia of NHPs was tightly associated with the transgene construct, implying a role for transgene expression in neuronal injury. 99 Using endogenous microRNA regulation of transgene expression through artificial target sequences, the DRG findings are attenuated by down-regulation of transgene expression (RNA and protein) in sensory neurons by incorporating a regulatory RNA guide sequence within the transgene derived mRNA that is targetable by microRNA 183 (miR183), which is highly expressed in sensory neurons.13,31,100 Unmodified vector (ie, possessing a transgene that lacks the miR183 targeting sequence) induced the expected DRG findings, and immunosuppression by systemic steroids failed to reduce such findings, indicating that DRG lesions for that particular study were primarily produced by presence of the transgene product and not by the immune response against the transgene product and/or vector elements. 100 Immune responses against the TA (either the transgene expression or the vector) may also play a primary or secondary toxicity-potentiating role in driving sensory ganglia findings.60,94,185 Reducing transgene expression in sensory neurons by including the miR183 targeting sequences minimized sensory ganglia pathology in nonclinical studies with NHPs. 100 With some transgenes, the severity of DRG pathology can be more than minimal or mild, and such increased severity is associated with more pronounced ganglionic inflammation, higher ELISpot responses (indicating substantial cytokine production), and on some occasions an NHP-specific MHC-1-restricted T cell response to a “non-self” transgene product. 243
In addition to overexpression of the transgene, immune mechanisms not related to a foreign protein may also contribute to the DRG findings. For example, the use of immunosuppression (mycophenolate mofetil [MMF] and rapamycin) did not eliminate or attenuate the findings related to administration of an AAV9 vector carrying an α-L-iduronidase (IDUA) transgene to NHPs.102,103 Therefore, further exploration is needed to evaluate and devise immunosuppressive regimens that more specifically target the suspected innate and adaptive (cytotoxic T cell) immune responses involved in producing ganglionic changes. An amyotrophic lateral sclerosis (ALS) clinical trial with an AAVrh10 vector expressing a microRNA transgene to suppress superoxide dismutase 1 (SOD1) expression reported neuropathic pain and loss of DRG sensory neurons in one patient without immunosuppression, consistent with the possibility that transgene protein is not required to induce DRG pathology. 168 However, a second ALS patient who received a strong immunosuppression protocol (prednisone, sirolimus, and rituximab) did not develop similar DRG findings, potentially supporting a role for the immune response in contributing to DRG injury. 168
Recombinant AAV vectors also carry the potential to induce innate immune responses through activation of several PAMPs, subsequently activating and boosting the potential for an adaptive immune response against the vector or its transgene product. 109 For instance, the presence of high levels of vector transcript (mRNA) and double-stranded RNA (detected by RNAscope ISH using the antisense and sense probes, respectively) in the affected DRG neurons of NHPs at 6 weeks post-administration may increase expression of intracellular innate immune response genes (such as retinoic acid-inducible gene I [RIG-I]-like receptor [RLR] family members). 226 Activation of this signaling pathway results in nuclear translocation of interferon regulatory transcription factor 3 (IRF3) and nuclear factor kappa B (NFκΒ), which culminates in transcription of pro-inflammatory cytokines including type I interferons.140,189,203 Increased expression of RLRs in affected DRG, specifically in neurons with degenerate microscopic features, is observed as early as 2 weeks post-dose and is correlated with the increased extent of T cell infiltrates in the DRG (K. Mansfield, personal communication).
Antisense oligonucleotides
TA-related findings in spinal nerves and their associated sensory ganglia have been observed in nonclinical toxicity studies conducted in NHPs with ASOs introduced directly into the CSF (eg, ICV, ICM, or IT). In the experience of WG members, the most common finding in such cases is TA-related, dose-dependent increases in small mononuclear cell aggregates (lymphocytes with some macrophages) in the meninges of the spinal cord and/or brain, the connective tissue around the DRG, and the spinal nerve roots. 12 This finding typically is of minimal to occasionally mild degree and of unknown functional significance. Macrophages in these foci often exhibit basophilic stippling of the cytoplasm (ie, ASO sequestered within lysosomes). Nerve fiber degeneration (especially in the spinal nerve roots, with ascending and descending effects in spinal cord and associated peripheral nerves) has been observed, with rare neuron degeneration in the DRG (unpublished data). Neuropathological findings are generally not evident following systemic (eg, IV or SC) ASO administration. 11
Nerve growth factor (NGF) inhibitors
Agents that block NGF are being investigated clinically as potential therapeutics for treating chronic pain disorders. 14 Such TA are associated with a number of ganglionic effects. For example, prenatal exposure to tanezumab (a humanized anti-NGF monoclonal antibody [MAb]) has been linked to persistently decreased neuron numbers in DRG and multiple sympathetic ganglia as well as reduced numbers of myelinated and unmyelinated nerve fibers in the sural nerve (a sensory-predominant trunk) in NHPs.29,36 These findings were visible by conventional (semi-quantitative) light microscopic evaluation as well as by stereology. Neither neurons nor nerve fibers exhibited degeneration or necrosis, indicating that the anti-NGF effect was not caused by cytotoxicity but was likely related to neuronal hypoplasia or atrophy. In contrast, adult NHPs exposed to tanezumab exhibit decreased sympathetic ganglia volume by stereology as a consequence of decreased neuron size (atrophy). 19 This change is present as early as 2 weeks following initial administration and peaks after 1 month with no further progression. The morphologic changes are fully reversible after TA withdrawal. Similar ganglionic findings have been reported for the NGF inhibitor fulranumab (a fully human MAb). 193
Aminoglycoside antibiotics
Aminoglycosides (eg, amikacin, gentamicin, kanamycin, tobramycin) are among the most effective treatments for severe Gram-negative bacterial infections. Aminoglycoside exposure is a known cause of hearing loss as a consequence of cochleotoxicity (ie, damage to the auditory sensory elements—hair cells [receptors], and to a modest degree sensory neurons of the SG) affecting the inner ear. Following systemic administration, aminoglycosides readily enter the cochlea and are taken up by hair cells in the spiral organ (of Corti) in the cochlea.52,121 Three parallel lines of outer hair cells (OHCs) accumulate more drug compared with the single line of inner hair cells (IHCs). 111 Aminoglycosides also accumulate to a modest degree in sensory neurons in the SG 111 as well as the DRG and TG. 52
Manifestations of aminoglycoside toxicity vary with both the targeted neural elements and the drug concentration. Toxicity at low doses develops first as degeneration and necrosis of OHCs at the cochlear base, but more apical OHCs and IHCs are also affected as the cumulative dose increases. 72 Depletion of SG neurons also occurs, presumably from primary degeneration of the ganglion neurons177,210 perhaps exacerbated by secondary attrition following loss of trophic factor support due to total ablation of IHCs that innervate the ganglion neurons. 111 Auditory dysfunction from aminoglycosides has also been attributed to cochlear synaptopathy, a condition where the drug disrupts synaptic interfaces between hair cells and their afferent SG neurons. 177
Evaluation of Primary Ganglia Microscopic Findings in Nonclinical Toxicity Studies
Different biopharmaceuticals elicit a spectrum of findings in ganglia. Both the types of findings and the affected ganglia are determined to some extent by the nature of the TA. For example, aminoglycoside antibiotics induce cell death leading to reduced cell density in SG. 121 CIPN classically is associated with Wallerian-like (“dying back”) degeneration of nerve terminals and distal sensory nerve fibers 61 and also leads in DRG to the expression of markers indicative of neuronal injury (ATF3 154 ) and SGC activation (GFAP 238 ); structural evidence of neuronal and/or SGC degeneration has been reported for some CIPN models (eg, mice administered paclitaxel 244 ). Direct CNS or IV administration of AAV-based gene therapies is associated with several consistent DRG findings: neuronal degeneration and/or necrosis, neuronal loss, increased SGC numbers, and leukocyte infiltration (cell influx without injury to the parenchyma) and/or inflammation (cell influx yielding tissue damage).34,95,99,101,103,179,226 Direct CNS administration of ASO has been associated with mononuclear cell infiltration, usually of minimal degree affecting DRG and/or spinal nerve roots. In general, neither the neurons in DRG nor their axons in the spinal cord dorsal funiculi exhibit morphologic changes indicative of ASO-related cell injury unless there is spinal root nerve fiber degeneration (unpublished data). The absence of degeneration or vacuolation in DRG neurons mirrors the absence of degenerative changes in CNS neurons with ASO-associated vacuolation. 138
Obviously, TA-related changes will only be seen in ganglia if these organs are included in the tissue list for histopathologic evaluation. These changes will need appropriate descriptions, diagnoses, and severity grades to permit effective interpretation and support data translation for human risk assessment.
Diagnostic nomenclature for ganglia in nonclinical studies
Current globally recognized INHAND (International Harmonization of Nomenclature and Diagnostic Criteria) terminology for CNS and PNS findings30,80,127,209 was designed for CNS and nerve findings but is readily adaptable to ganglia. A complete recapitulation of INHAND terms for nervous system findings is beyond the scope of this paper, but certain diagnoses are commonly applied to changes in DRG (and other ganglia) by WG members. The definition and interpretation of these particularly useful terms are reviewed here.
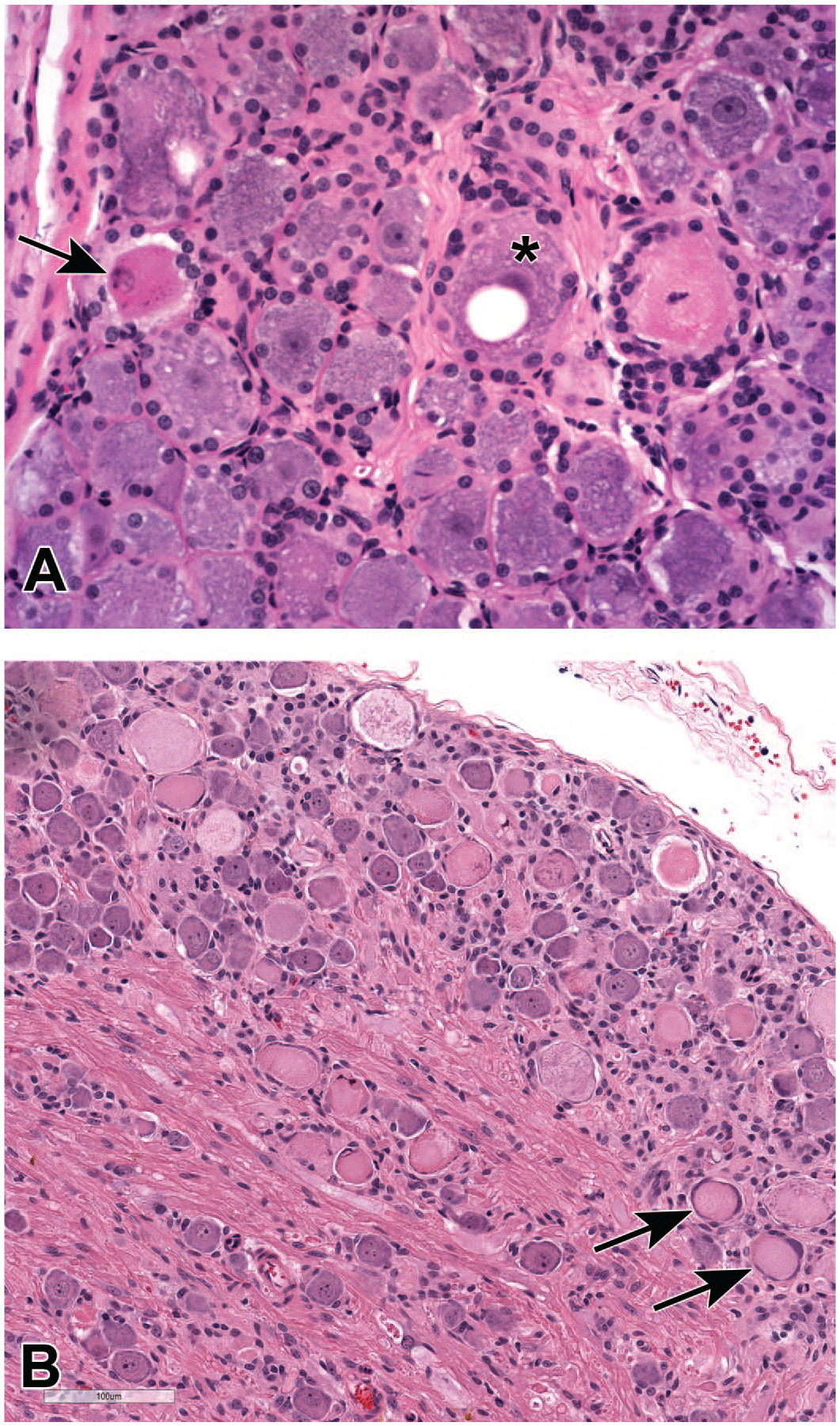
Neuronal findings common in dorsal root ganglia (DRG) following treatment with adeno-associated viral (AAV) vectors (Panel A) and anti-neoplastic chemotherapeutic small molecules (Panel B), where phrases in
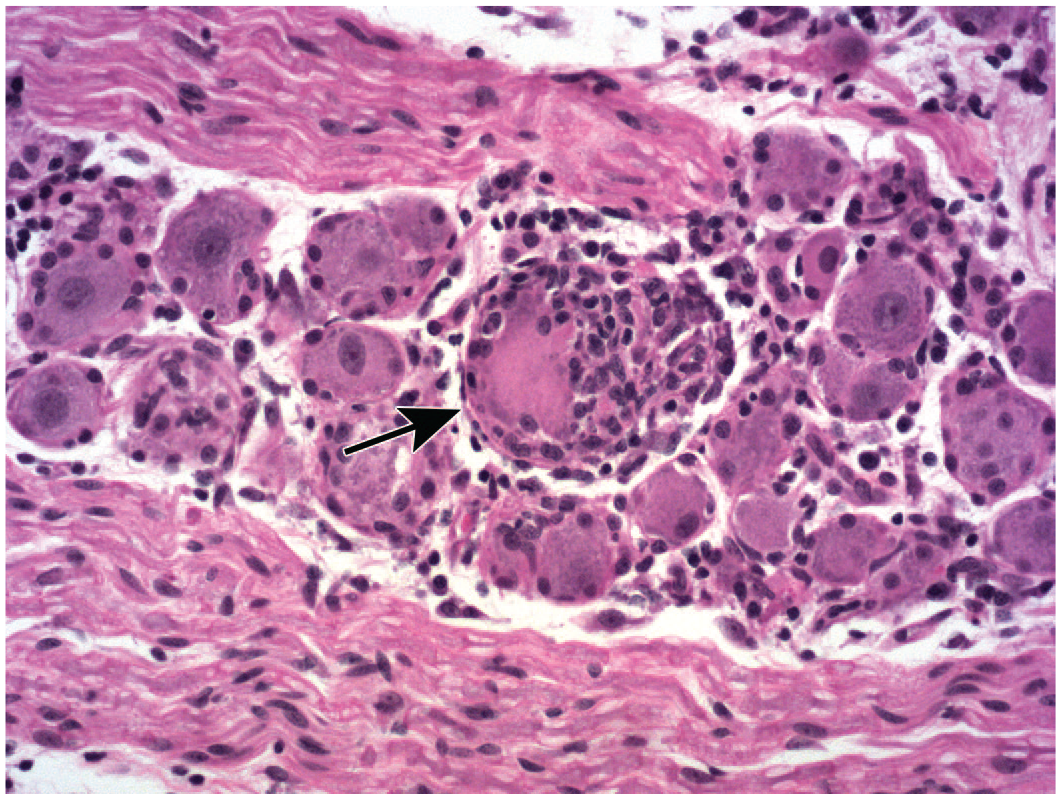
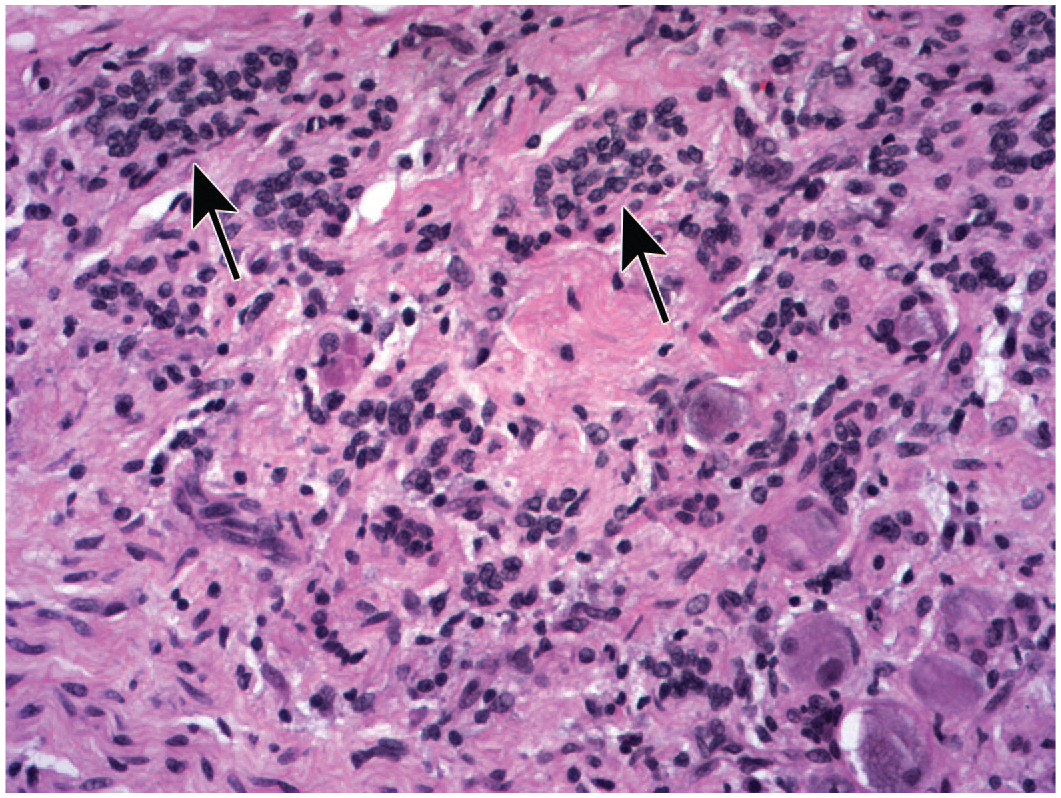
One of 3 spontaneous terms is used for neuron-specific changes, while 2 are findings that also occur with some frequency in other ganglion cell populations.
Additional diagnostic terms may be warranted for unusual changes, but this list of likely neuron findings should prove useful for most purposes and is suitable for inclusion in standard SEND (Standard for Exchange of Nonclinical Data)-compliant histopathology data sets expected in regulatory submissions to the US Food and Drug Administration (FDA).
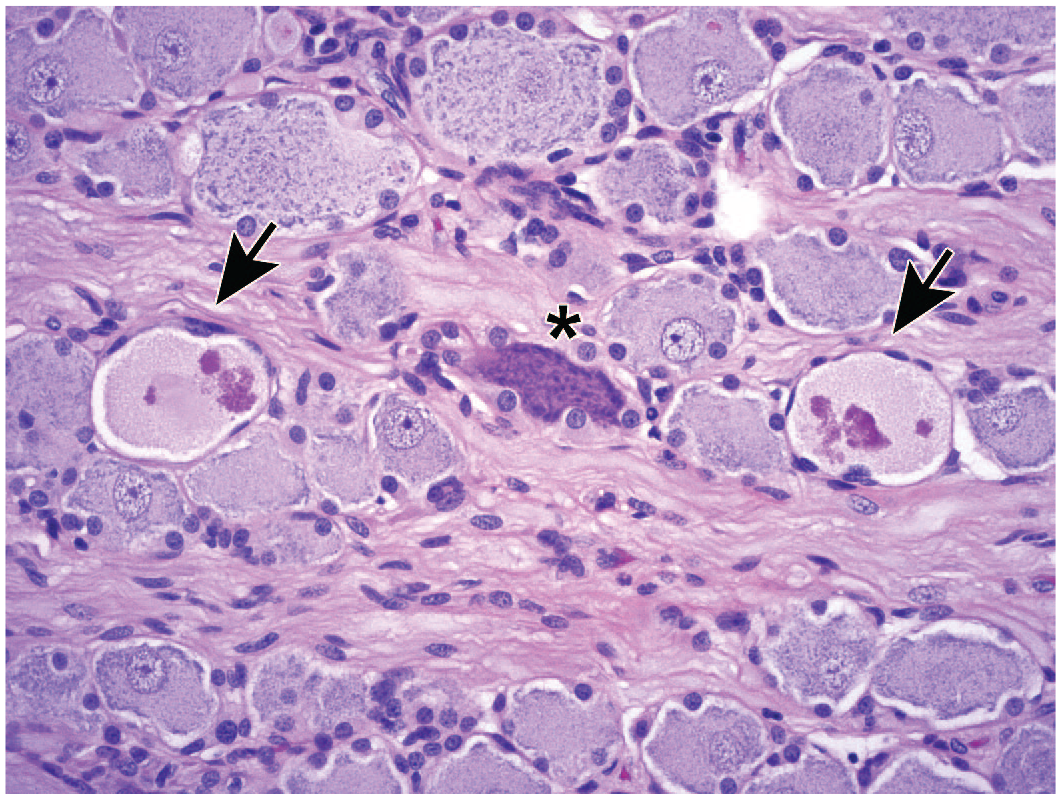
On occasion, structural changes to SGC may warrant microscopic terminology or findings commonly used with other cell types (eg, degeneration, necrosis, vacuolation, altered cell size), but these findings are uncommon compared with the proliferative responses noted above.
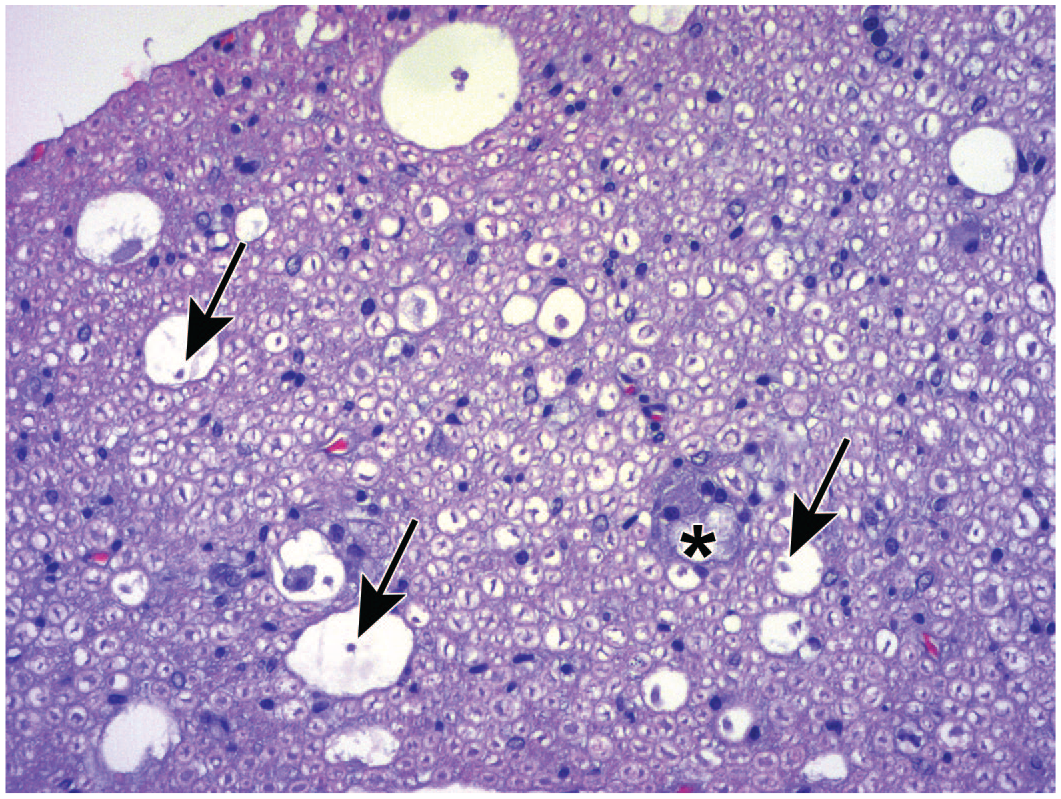
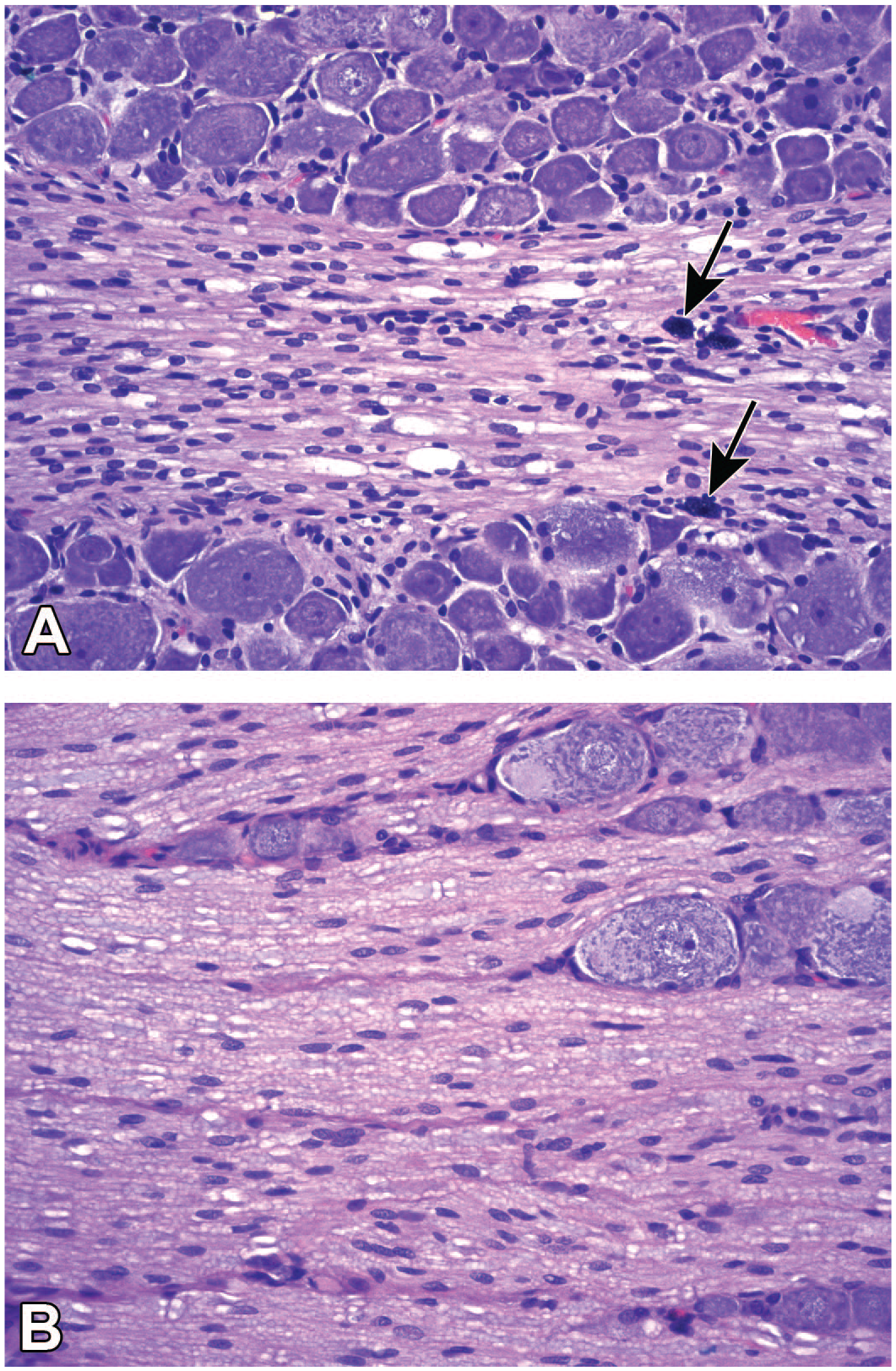
In general, the term “radiculoneuropathy” is seldom used during nonclinical toxicity studies less than 12 months in duration. 30 The other changes related to sensory ganglion injury may be recognized in nonclinical studies of any length.
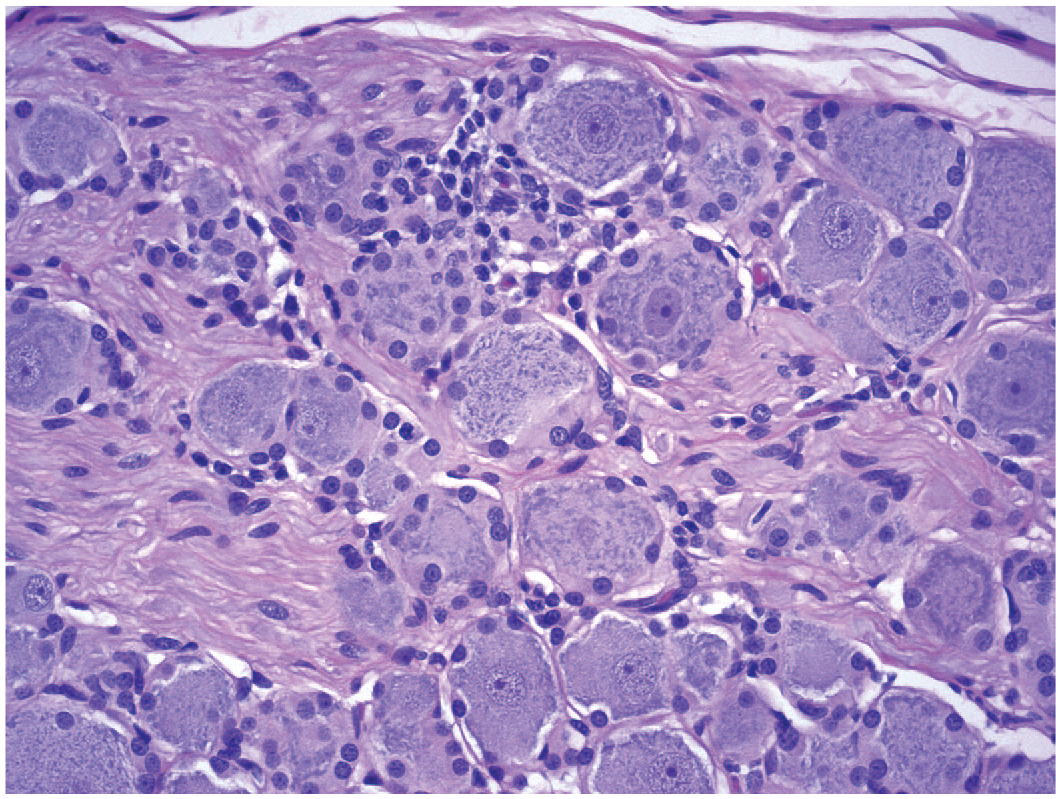
The generic form “mononuclear cell” typically is used as a modifier rather than the more specific terms “lymphocyte” and “macrophage” since this finding in DRG and TG usually represents a mixture of these cell types. IHC may be helpful for interpretation (eg, CD3, CD79a, and CD20 to discriminate lymphocyte populations and CD68 or IBA1 for monocyte/macrophage lineage). The modifier “mixed cell” may be employed instead in those cases where neutrophils and/or eosinophils (indicative of a more acute process) also are present within leukocyte aggregates.
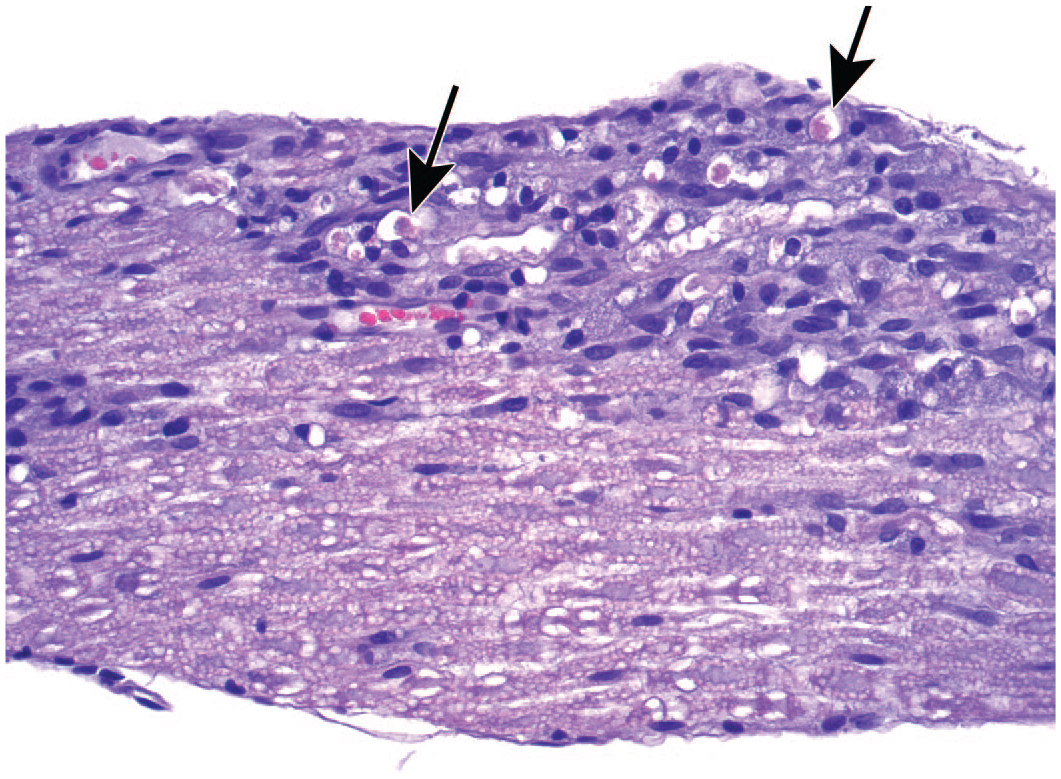
Two
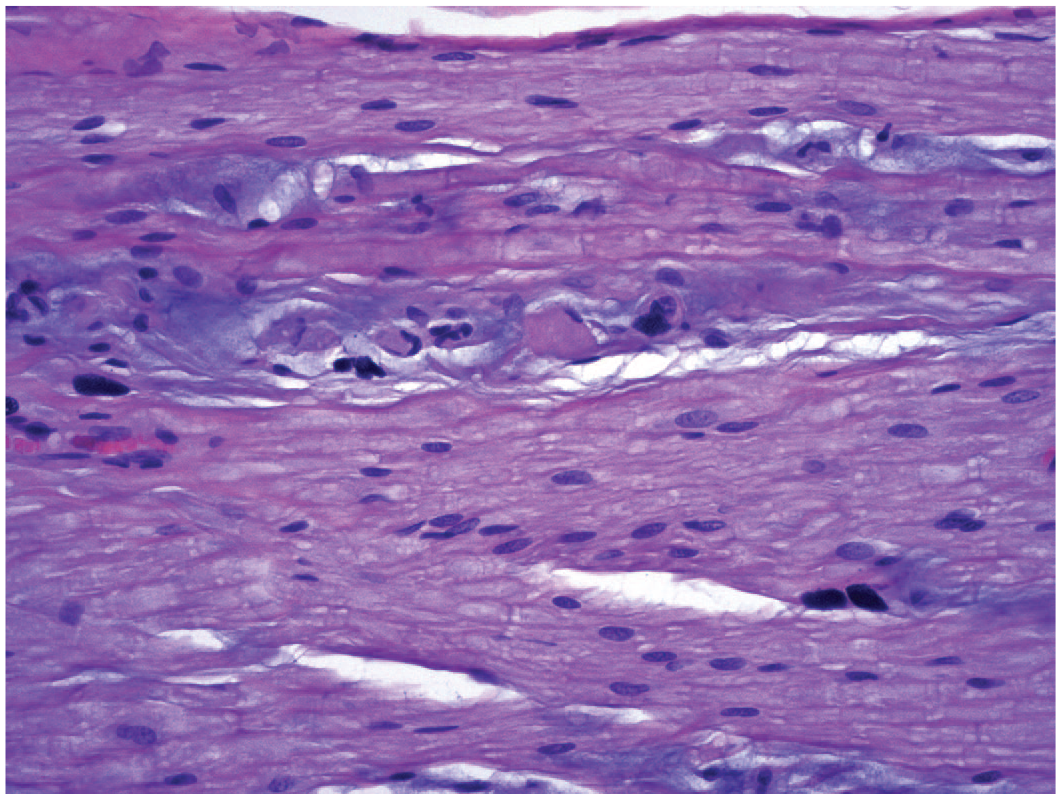
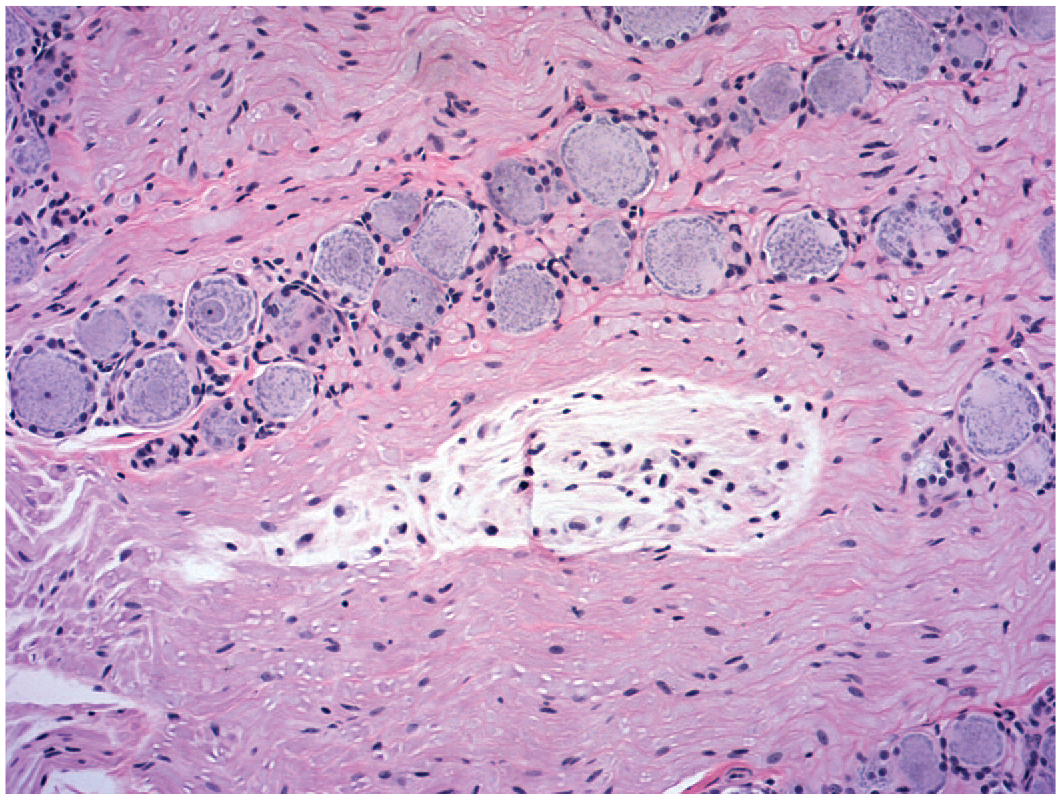
Both findings appear to be incidental background findings that are not affected by TA exposure. Neither is common in ganglia of control animals for all species, but matrix accumulation is seen more frequently. That said, matrix accumulation is unlikely to be present as a diagnosis in nonclinical historical control data as it is a very recent addition to the INHAND nomenclature. 30
As noted previously, the dorsal funiculi in spinal cord cross-sections represent a summation of axonal health for a large subset of sensory neurons, located in all of the DRG that are caudal to that plane of section. Accordingly, evaluation of spinal cord is essential when the DRG are anticipated to be a target for novel TA.
Summary
In conclusion, injury to sensory and autonomic ganglia (“gangliotoxicity”) affecting neurons is a potential effect associated with administration of certain biopharmaceutical products, most often anti-neoplastic chemotherapeutics and AAV-based gene therapies and also including ASOs, NGF inhibitors, and some antibiotics. Accordingly, ganglia (especially DRG) should be screened during nonclinical toxicity studies for TAs of these classes. Effective screens require carefully designed collection of these tissues at necropsy and that pathologists have familiarity with basic features of ganglionic biology and pathology.
This article reviews sensory and autonomic ganglion anatomy, cell biology, and pathology among common nonclinical species used in assessing product safety for such TA. Depending on institutional choices with respect to tissue orientation, the anatomy for specific sensory and autonomic ganglia exhibit fairly consistent microscopic changes when assessed in routine histological sections. Principal microscopic findings in ganglia include neuron degeneration, necrosis, and/or loss; altered cellularity (numbers) of SGCs and/or Schwann cells; and leukocyte infiltration and/or inflammation. Microscopic findings in other nervous system components that are connected to ganglia include nerve fiber degeneration and/or glial reactions in nerves, dorsal spinal nerve roots, spinal cord (dorsal and occasionally lateral funiculi), and sometimes the brainstem. Findings in both ganglia and associated CNS regions and nerves may wax and wane over the course of a given study. Ganglion findings may be caused by TA exposure (ie, toxicity) and/or TA administration (ie, procedure-associated trauma), and these commonly need to be differentiated from spontaneous background changes. Familiarity with such details regarding ganglion biology and pathology will help toxicologic pathologists, toxicologists, and regulatory reviewers design nonclinical studies that are capable of providing a suitable survey of TA effects in sensory ganglia and related CNS and PNS circuits.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233231213851 – Supplemental material for Scientific and Regulatory Policy Committee Technical Review: Biology and Pathology of Ganglia in Animal Species Used for Nonclinical Safety Testing
Supplemental material, sj-docx-1-tpx-10.1177_01926233231213851 for Scientific and Regulatory Policy Committee Technical Review: Biology and Pathology of Ganglia in Animal Species Used for Nonclinical Safety Testing by Bindu M. Bennet, Ingrid D. Pardo, Basel T. Assaf, Elizabeth Buza, Sarah D. Cramer, LaTasha K. Crawford, Jeffery A. Engelhardt, Elizabeth J. Galbreath, Branka Grubor, James P. Morrison, Tanasa S. Osborne, Alok K. Sharma and Brad Bolon in Toxicologic Pathology
Footnotes
Acknowledgements
The authors thank Ms Beth Mahler for her expertise in optimizing image quality.
Author Contribution
All authors contributed to the drafting and revision of this paper, and all approve and agree to be responsible for the final content. No external assistance (artificial intelligence software, ghost writers, etc) was used in preparing the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors express no conflict of interest in producing this technical review except that all are employed by organizations involved in the discovery and development of various biomedical products for treating human diseases (including AAV-based gene therapies, ASOs, other neuroactive biologics, and small molecules).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
ORCID iDs
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
