Abstract
Certain biopharmaceutical products consistently affect dorsal root ganglia, trigeminal ganglia, and/or autonomic ganglia. Product classes targeting ganglia include antineoplastic chemotherapeutics, adeno-associated virus-based gene therapies, antisense oligonucleotides, and anti-nerve growth factor agents. This article outlines “points to consider” for sample collection, processing, evaluation, interpretation, and reporting of ganglion findings; these points are consistent with published best practices for peripheral nervous system evaluation in nonclinical toxicity studies. Ganglion findings often occur as a combination of neuronal injury (e.g., degeneration, necrosis, and/or loss) and/or glial effects (e.g., increased satellite glial cell cellularity) with leukocyte accumulation (e.g., mononuclear cell infiltration or inflammation). Nerve fiber degeneration and/or glial reactions may be seen in nerves, dorsal spinal nerve roots, spinal cord, and occasionally brainstem. Interpretation of test article (TA)-associated effects may be confounded by incidental background changes or experimental procedure-related changes and limited historical control data. Reports should describe findings at these sites, any TA relationship, and the criteria used for assigning severity grades. Contextualizing adversity of ganglia findings can require a weight-of-evidence approach because morphologic changes of variable severity occur in ganglia but often are not accompanied by observable overt in-life functional alterations detectable by conventional behavioral and neurological testing techniques.
Keywords
This “Points to Consider” article is a product of a working group of the Scientific and Regulatory Policy Committee (SRPC) of the Society of Toxicologic Pathology (STP). It has been reviewed and approved by the SRPC and Executive Committee of the STP, but it does not represent a formal Best Practice recommendation of the Society; rather, it is intended to provide key “Points to Consider” in designing nonclinical studies or interpreting data from toxicity and safety studies intended to support regulatory submissions. This article has been reviewed and endorsed by the European Society of Toxicologic Pathology and the British Society of Toxicological Pathology. The points expressed in this document are those of the authors and do not reflect the views or policies of the employing institutions. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to the Editor.
Introduction
Ganglia of the peripheral nervous system (PNS) including dorsal root ganglia (DRG), spiral ganglia (SG), trigeminal ganglia (TG), and autonomic ganglia have been identified as target organs for some classes of pharmaceutical products. (Abbreviations are defined in the article at the point where they are first mentioned, but a full list is available in Supplemental Table 1.) Agents that impact ganglia include conventional small molecule drugs such as aminoglycoside antibiotics1,2 and many antineoplastic chemotherapeutics3-5 as well as novel biomolecule-based therapies such as adeno-associated virus (AAV) vectors,6-10 antisense oligonucleotides (ASO) (unpublished data), and nerve growth factor inhibitors.11-13 Sensory polyneuropathy associated with antineoplastic chemotherapy is a common dose-limiting toxicity in human patients.14,15 Neurological deficits have not been confirmed in human patients administered gene therapies although symptoms reported in cases of patients with advanced neurodegenerative disease have been suggested as potential evidence for toxicity.16,17 Taken together, these outcomes suggest that nonclinical evaluation of ganglion structure and function is warranted when developing new test articles (TAs) for some therapeutic modalities.
Although evaluation of the nervous system is an essential element of nonclinical toxicity testing, existing guidance for nonclinical testing either mentions brain, spinal cord, and sciatic nerve as neural organs to sample routinely 18 or does not specify which neural tissues to collect.19-23 Initial industry “best practices” designed to address such guidance 24 did not include DRG, TG, or other sensory and autonomic ganglia among the standard list of tissues recommended for microscopic evaluation in routine nonclinical general toxicity studies, but more recent “best practice” recommendations have provided flexible options regarding collection of ganglia, nerves, and connected neural elements for general toxicity and focused neurotoxicity studies.25,26 Additional nervous system sampling is recommended by these “best practice” publications when class or target-related neuronal findings may be expected. 26
Literature (both peer-reviewed papers and proprietary presentations and white papers) on toxicologic pathology of ganglia in animals is limited, which impedes the entry of promising therapeutic candidates with suspected or likely TA-related ganglion pathology into clinical development. Accordingly, the Scientific and Regulatory Policy Committee (SRPC) of the Society of Toxicologic Pathology (STP) convened a working group (WG) to discuss, formulate, and publish key “points to consider” for evaluating, interpreting, and reporting of microscopic findings in ganglia (emphasizing DRG as the prototypic target tissue) for nonclinical toxicity studies. The points discussed herein provide key concepts to consider in designing, performing, analyzing, and interpreting anatomic pathology endpoints from safety studies intended to support regulatory submissions for TAs from product classes that are suspected or known to injure sensory ganglia; these points do not represent formal “best practice” recommendations endorsed by the STP or other global societies of toxicologic pathology. The selected WG members were subject matter specialists with diverse backgrounds and extensive professional experience as pathologists and toxicologists in biopharmaceutical companies, contract research organizations, consulting practices, and/or academic medical centers. The SRPC charge for the WG was to (1) review basic ganglia (DRG, TG, SG, and autonomic) neurobiology related to their potential toxicant vulnerability; (2) define suitable diagnostic nomenclature for TA-related microscopic findings in ganglia; (3) assess any notable differences in species sensitivity among laboratory animals; and (4) discuss possible correlations between these microscopic findings and functional endpoints. The current article addresses topics (1) briefly as well as (3) and (4) in detail while providing major points to consider regarding the sampling, processing, evaluation, interpretation, and reporting of sensory and autonomic ganglion pathology in nonclinical toxicity studies. A companion paper 27 prepared by the WG provides a comprehensive review of the biology and pathology of sensory and autonomic ganglia in nonclinical species, thereby addressing topics (1) and (2) above. These paired papers provide information needed by toxicologic pathologists, toxicologists, and regulatory reviewers to ensure that ganglionic effects of TA are identified and characterized during nonclinical safety testing.
Biology Fundamentals for Ganglia
Ganglia harbor primary somatic and visceral afferent neurons, the cells responsible for transfer of sensory information from peripheral receptors to the central nervous system (CNS). This section reviews the basic neurobiology of ganglia using the DRG as a prototype, since these organs are readily found and sampled in common nonclinical species (Figure 1),26,28-31 injury to DRG neurons is known to augment nociception (i.e., the perception of pain), 32 and DRG neurons are recognized targets for certain classes of small molecule and biomolecule therapeutics.3-7,9,10
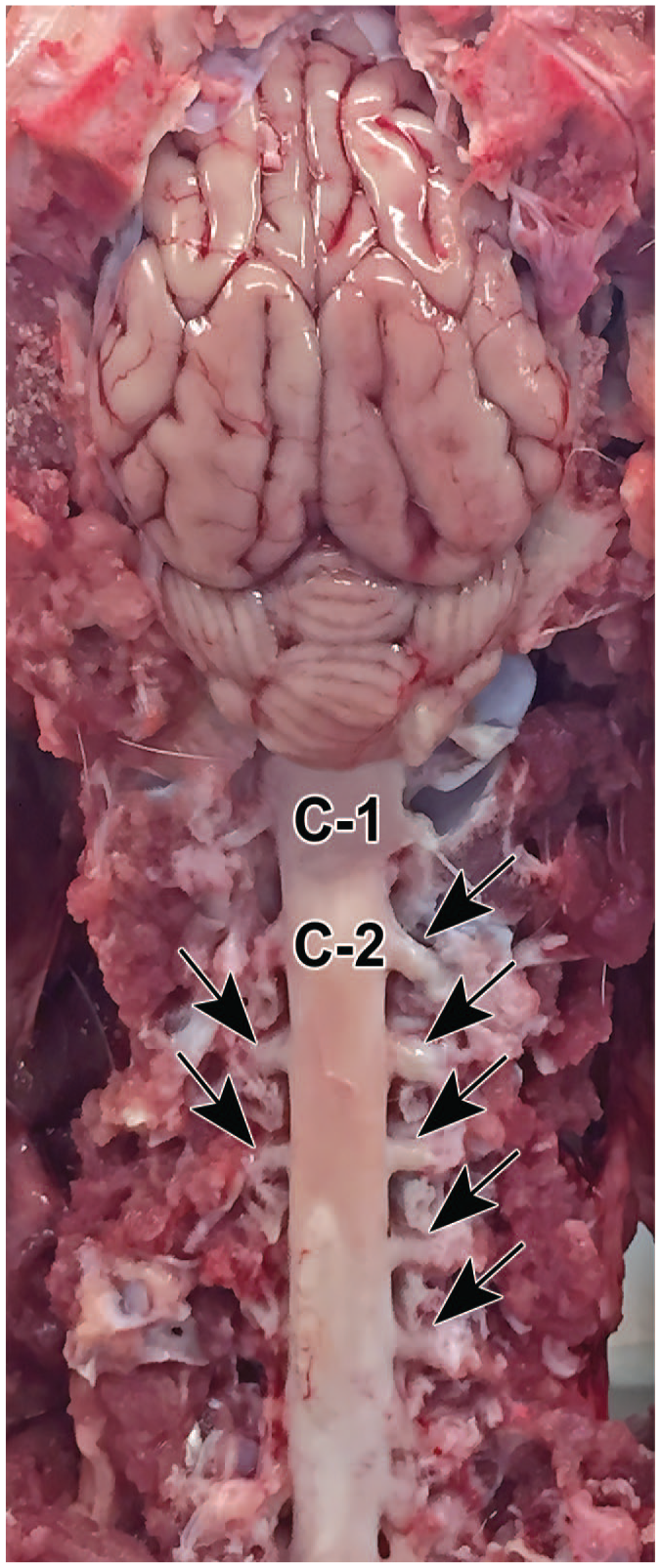
External landmarks used to identify bilaterally paired dorsal root ganglia (arrows) associated with the spinal cord. Segments in the upper cervical spinal cord are labeled (C1 and C2). Species: Göttingen minipig.
The DRG (also termed “spinal ganglia”) are paired organs associated with each spinal cord segment. Each DRG is positioned between a spinal nerve distally and a dorsal spinal nerve root proximally. The DRG neurons receive input via spinal nerves from sensory receptors throughout the body. Peripheral organs and tissues (e.g., bones, muscles, skin, tendons) are monitored by particular DRG associated with specific somatic nerve branches as specified by the pattern of axial segmentation (e.g., dermatomes, myotomes, sclerotomes) seen during early development 33 while viscera from the esophagus to the colon send sensory information to nearby DRG located in the cervical, thoracic, and upper lumbar segments. 34
The TG and SG are both functionally and structurally comparable with DRG. The TG provide sensory innervation to the face while the SG transfer auditory signals from the inner ear to the cochlear nucleus in the brainstem. Typically, TG (alternative designations: cranial nerve [CN] V, Gasserian, or “semilunar” ganglion 35 ) may be considered for retention in nonclinical toxicity studies because they are fairly easy to collect after the brain has been removed (Figure 2).28-31,36 The TG develop drug-induced ganglionic pathology equivalent to that reported in DRG.3,7 In contrast, the SG are sampled only in dedicated ototoxicity studies as their location encased within thick bone of the skull requires special tissue processing methods unsuitable for conventional nonclinical toxicity studies.37,38 In general, evaluation of DRG (± TG) but not SG is an acceptable approach for screening sensory ganglia for neurotoxicity in nonclinical studies.
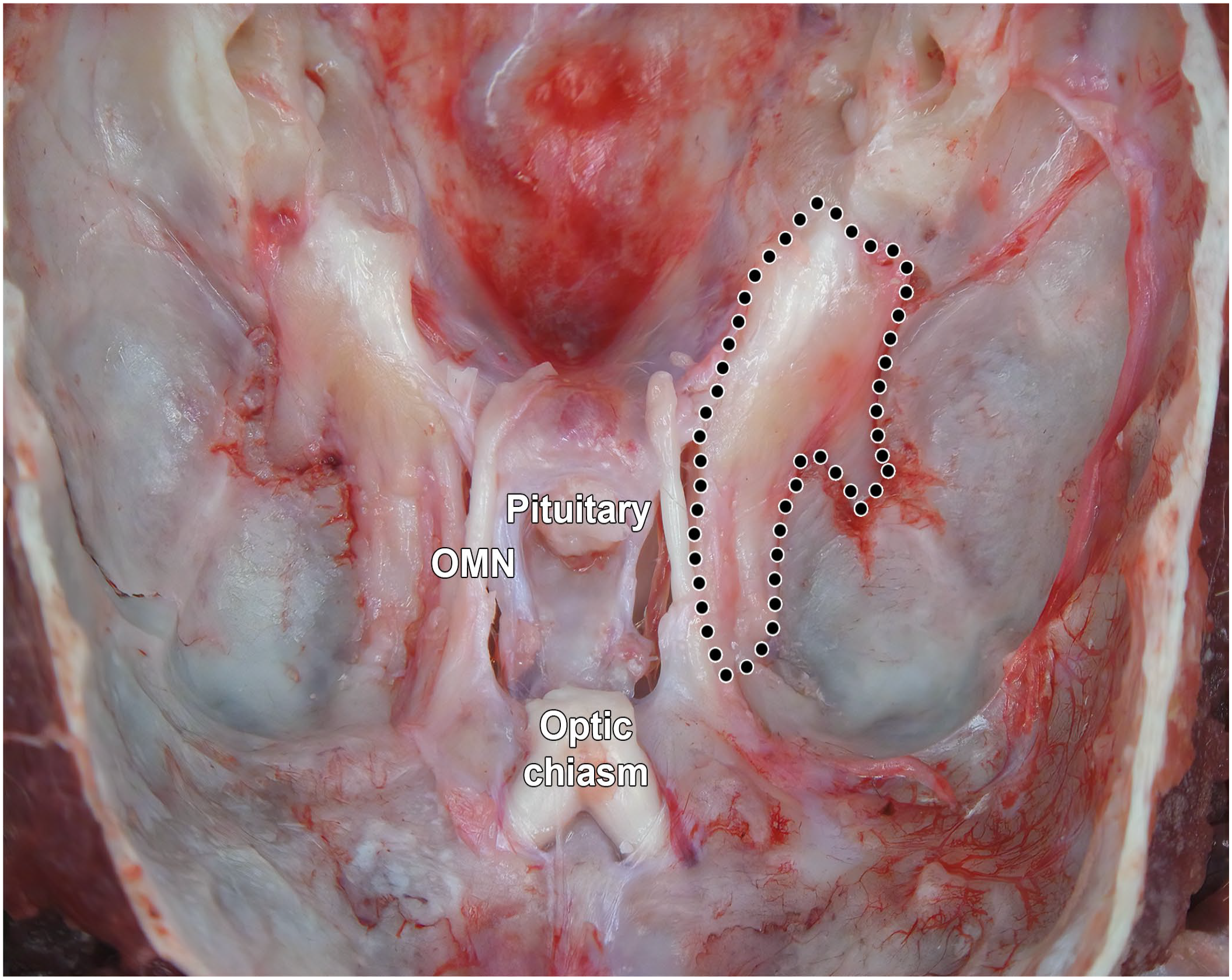
Trigeminal ganglia (somatic sensory) of a Beagle dog in situ after removal of the brain and dissection of the meninges (with the left ganglion delimited by black dots). OMN, oculomotor nerve (cranial nerve III).
Autonomic nervous system (ANS) ganglia that incorporate general visceral afferent (GVA) nerve fibers are examined during some nonclinical studies (on a case-by-case basis) to assess potential ANS effects of TAs. 26 The GVA nerve fibers transmit sensations from various internal organs and tissues. 39 Principal autonomic ganglia that harbor GVA nerve fibers include the geniculate ganglia of the facial nerve (CN VII), distal glossopharyngeal ganglia (alternate designations: CN IX or petrosal ganglion), and distal vagal ganglia (alternate designations: CN X or nodose ganglion). Cranial cervical ganglia (the proper name in quadrupeds 40 for the “superior cervical” ganglia [SCG] of bipeds) in the cranial neck and/or the cervicothoracic ganglia (the proper name in quadrupeds 40 for the “inferior cervical” or “stellate” ganglia of bipeds) at the cranial end of the thorax may be sampled as a means of screening the sympathetic autonomic system. The distal vagal (nodose) ganglia and SCG reside next to each other bilaterally in the deep neck tissues near the ventral surfaces of cervical vertebrae so that, in most cases, institutions elect to acquire and process these ganglia together (Figure 3).26,28
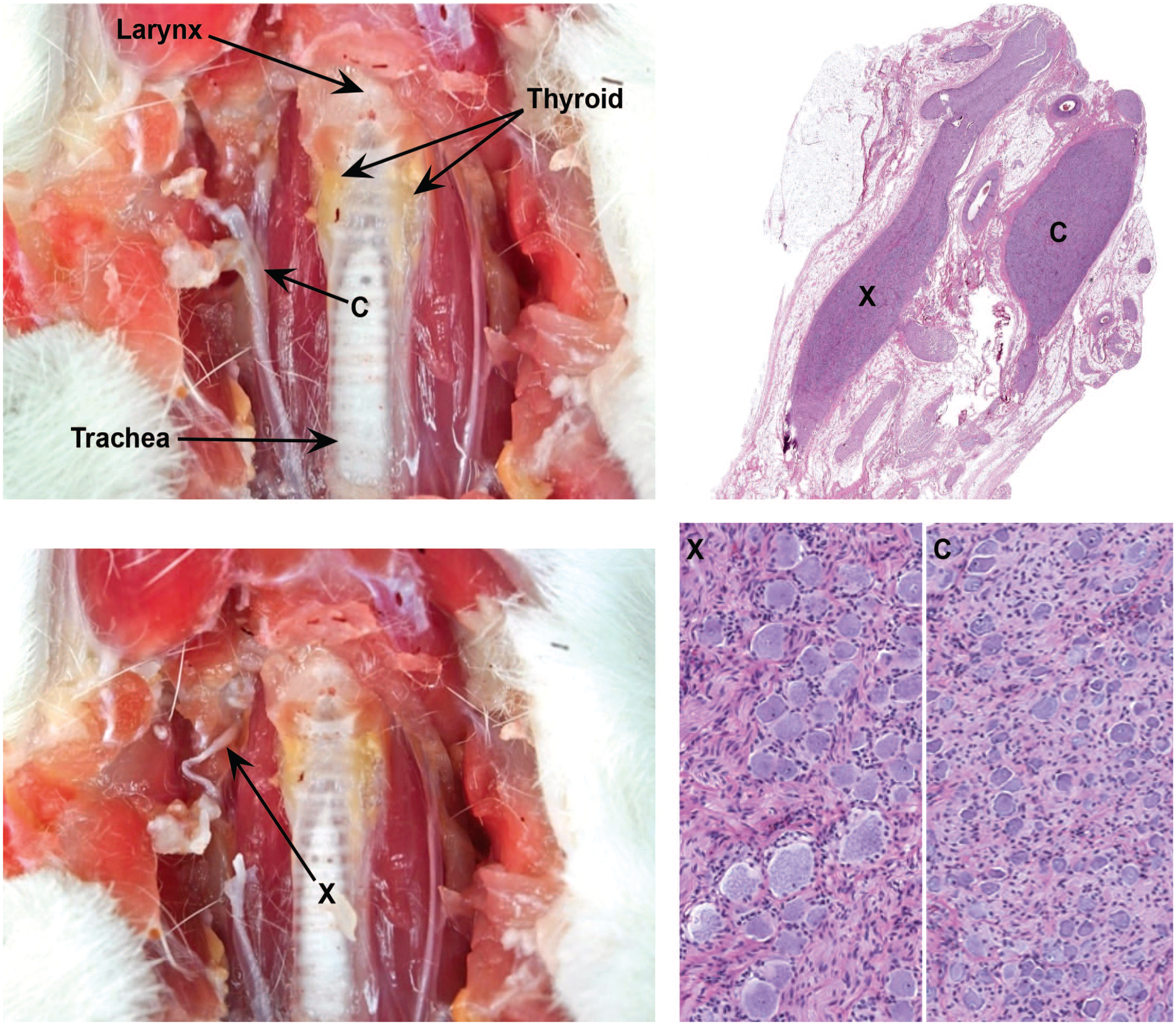
Location of key autonomic ganglia in the cervical region. Two cervical ganglia, the cranial (superior) cervical ganglion (C, a part of the sympathetic division) and the caudal vagal (nodose) ganglion (X, a visceral afferent [i.e., sensory] element), may be isolated lateral to the trachea and thyroid glands in the vicinity of the bifurcation of the carotid artery. In histologic sections, the two ganglia can be distinguished readily because the sympathetic neurons in the cranial cervical ganglion are much smaller than the sensory neurons in the caudal vagal ganglion. Samples: left column = adult rat (provided courtesy of Dr Magalie Boucher, by permission); right column = adult Beagle dog showing collection of both the cranial cervical ganglion (C) and caudal vagal ganglion (X) in the same histologic section and high-power depictions of these ganglia demonstrating their distinct features. Processing (right image): immersion fixation in neutral buffered 10% formalin, paraffin embedding, sectioning at 4 μm, hematoxylin and eosin staining.
Ganglia Structure
Screening for TA-related effects on sensory and/or autonomic ganglia (“gangliotoxicity”) requires familiarity with the structure and function of neural circuits at the macroscopic (gross) and microscopic levels. More detailed consideration of such ganglion anatomy may be obtained elsewhere,27,41-43 so this section provides a brief review of key elements relevant to toxicologic pathologists and toxicologists.
Macroscopic anatomy
The DRG occur as a series of bilaterally paired organs (Figure 1) located along the entire length of the vertebral column, with one pair associated with each spinal cord segment and its related vertebra. Each DRG forms a bridge between a spinal nerve distally and a dorsal spinal nerve root proximally. 26 The DRG for the spinal cord segments that comprise the cervical and lumbar intumescences (i.e., the primary spinal cord segments that serve the forelimb and hind limb somatic nerve trunks, respectively, Table 1) are bigger than those of other spinal cord segments because they contain a larger number of ventral horn motor neurons.42,52 The DRG may be tiny or even missing at some levels (usually C1) in some species.42,53
Approximate locations of spinal cord segments that give rise to large mixed somatic nerves.
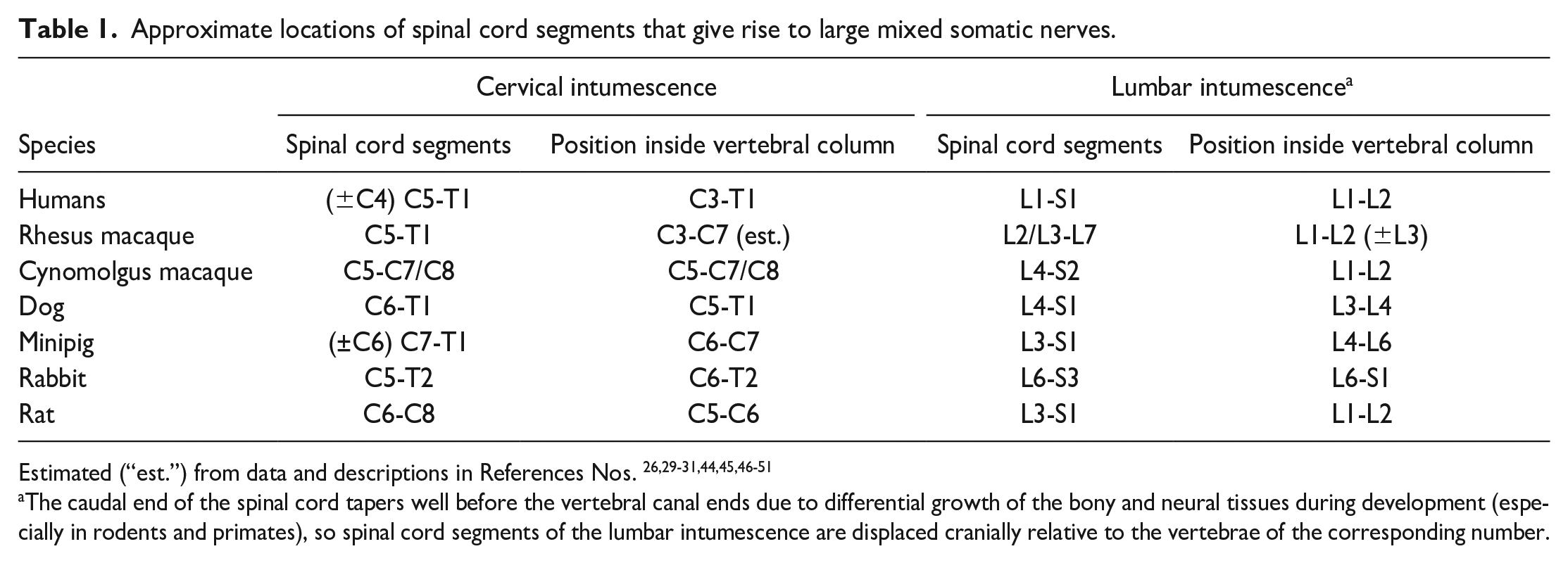
The caudal end of the spinal cord tapers well before the vertebral canal ends due to differential growth of the bony and neural tissues during development (especially in rodents and primates), so spinal cord segments of the lumbar intumescence are displaced cranially relative to the vertebrae of the corresponding number.
The TG are bilaterally paired ganglia found on the floor of the skull on either side of the pituitary gland (Figure 2). 36 The SG (alternative designation: cochlear ganglion) are encased in the dense bone (designated the “modiolus”) that forms the central axis of the cochlea in the inner ear.
In general, GVA fibers co-localize with parasympathetic (“digest and rest”) or sympathetic (“fight or flight”) nerves and ganglia even though the GVA components act as sensory elements rather than serving parasympathetic or sympathetic functions. 39 In routine nonclinical toxicity studies, parasympathetic autonomic ganglia generally are available for evaluation as clusters of neurons on the surface, in the walls, or scattered among parenchymal elements of viscera.26,54 Sympathetic autonomic ganglia are located bilaterally as neuronal clusters associated with the sympathetic chain, a series of interconnected ganglia connected by a nerve trunk located along the ventrolateral surface of the vertebral column. The cranial extension of the sympathetic chain into the cervical region starts at the cervicothoracic ganglion caudally and ends at the cranial cervical ganglion cranially.26,33,44
Microscopic anatomy
Ganglia consist of sensory or autonomic neurons and supporting satellite glial cells (SGCs) interspersed with nerve fibers and with fine connective tissue stroma. 42 Depending on the plane of section, DRG neurons are concentrated near the organ periphery while narrow intersecting columns of nerve fibers comprise the center. 42 Non-neuronal cells in DRG include SGC, which encircle sensory neurons; Schwann cells, which myelinate axons; endothelial cells, fibroblasts, and leukocytes (mainly T-lymphocytes and macrophages with fewer B-lymphocytes and, in rodents, mast cells). In normal DRG, sensory neurons and SGC outnumber other cell types.
Both spinal nerve roots contain axons arising from DRG. The dorsal root is a pure sensory nerve and carries most DRG-derived axons33,44 while the ventral root (a predominantly motor nerve) harbors just a few DRG-derived axons. 55 Upon entering the spinal cord, DRG axons form synapses with local interneurons and projection neurons in the same spinal cord segment while a subset of axons that transmit touch and proprioception signals supply branches that turn cranially and ascend in the ipsilateral spinal cord white matter in the dorsal (posterior) funiculi. 27 Ascending sensory nerve fibers in the dorsal funiculi are arranged so that axons originating from caudal (lower thoracic, lumbar, sacral) DRG are located in the medial fasciculus and synapse in the gracile nucleus while those arising from more cranial (cervical to mid-thoracic) DRG are added in the lateral funiculus and synapse in the cuneate nucleus and lateral (accessory) cuneate nucleus; the cuneate and gracile nuclei reside in the medulla oblongata, the brain region that represents a rostral extension of the spinal cord.44,56 Some ascending nerve fibers that transmit sensory information are found in the lateral funiculi (usually dorsolateral) and ventral funiculi; axons in these domains arise from projection neurons of the spinal cord gray matter that receive input from DRG and spinal interneurons.44,56 This ordered arrangement means that microscopic evaluation of a spinal cord transverse section offers a snapshot of axonal integrity for all DRG caudal to the level of that transverse section.
Each DRG has a rich vascular supply that is concentrated in the neuron-rich periphery rather than the nerve fiber-rich core. The DRG capillaries are lined by fenestrated endothelium, so the neurovascular interface (i.e., “blood-ganglion barrier”) readily allows access to molecules in the blood.57-62 Blood vessels in TG are similarly lacking in an effective blood-ganglion barrier, 63 while vessels in the SG are reported to possess a conventional blood-brain barrier (BBB). 64 In contrast to the blood-ganglion barrier, the BBB is a selectively permeable barricade formed by non-fenestrated endothelium (joined by myriad tight junctions), pericytes, a continuous basement membrane, and a dense network of astrocyte foot processes.65-68
Direct administration of TAs into the cerebrospinal fluid (CSF) results in exposure of the DRG and TG since the proximal DRG pole (where the dorsal spinal nerve root exits) and the entire TG reside inside the meninges and are bathed in CSF. 69 Central delivery of TAs into the CSF may reach ganglia outside the CNS axis since materials in the CSF do exit the neuraxis to reach peripheral organs. 70
Cytology
Sensory ganglia are composed of many cell types. This section briefly reviews key cell classes that normally reside in ganglia and that are responsible for ganglion function (i.e., neurons and their supporting SGC and Schwann cells) or that muster immune reactions in response to ganglion injury (i.e., macrophages).
Neurons have comparable but not identical morphologic features in tissue sections (reviewed in Bennet et al. 27 ). In standard formalin-fixed, paraffin-embedded, hematoxylin and eosin (H&E)-stained sections, neurons in sensory ganglia appear as cells of variable sizes (range, 10-100 µm in diameter) with a large, slightly off-center nucleus, single nucleolus, and abundant cytoplasm packed with basophilic granules of Nissl substance (rough endoplasmic reticulum). Historically, bigger neurons with large-diameter axons and thick myelin sheaths (Aα and Aβ classes) were thought to transmit high-velocity sensations, such as proprioception and touch, while smaller neurons with small-diameter axons and thin myelin sheaths (Aδ and C classes) carry lower-velocity sensations, such as nociception and thermoreception. 33 Recent work has shown that this classification scheme is too simplistic: at least 15 different sensory neuron subtypes have been identified,71,72 and neurons serving both touch (high-velocity) and pain (low-velocity) functions can have Aβ, Aδ, or C axon and myelin parameters. 43 Importantly, neurons with comparable cytoarchitectural features often serve different functions. In such cases, visually similar neurons may be differentiated by their molecular signatures but not their cell features.73,74 In practical terms, microscopic evaluation of neurons in sensory and autonomic ganglia during nonclinical toxicity studies usually is confined to examining features visible in H&E-stained sections (e.g., degeneration and/or vacuolation, necrosis, and reduced cell numbers or altered cell sizes).27,75-77 On occasion, special neurohistological methods may be applied to sensory ganglia to further characterize potential TA-related effects. For example, neuron death may be assessed using cleaved caspase 3 immunohistochemistry (IHC) or terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL) 27 ; Fluoro Jade, a useful cell death marker in the CNS, works inconsistently in DRG. 26 In the future, other markers may be useful for evaluating effects on various subtypes of sensory neurons in nonclinical toxicity studies, such as neurofilament heavy chain (NfH [200 kD isotype], a cytoskeletal protein that is expressed chiefly by large, moderately to heavily myelinated neurons in mice, rats, rabbits, dogs, and nonhuman primates [NHP]; L.K. Crawford, unpublished data) or activating transcription factor 3 (ATF3, a nuclear marker expressed by axotomized but viable [restoration-competent] sensory neurons prior to the advent of overt structural changes in the body78,79).
The SGCs are the main resident glia of sensory ganglia, and they fulfill many of the tasks in sensory ganglia that are relegated to distinct glial cell subtypes in other portions of the nervous system.27,80-83 These oval cells and the basement membrane they produce form a sheath that surrounds the neurons 84 ; in H&E-stained sections, SGC forms an intermittent cordon encircling and occasionally indenting the margins of sensory neurons. The narrow gap between SGC and neurons (~20 nm) allows direct two-way communication between the cells. The SGC barricade is porous, permitting ions, macromolecules, neurotransmitters, and toxicants to quickly and freely reach neurons.80,81,85 Various SGC subpopulations have been distinguished in somatic and sympathetic ganglia using molecular markers, 83 and different SGC subpopulations play distinct roles in nerve fiber repair. 86 The SGC in SG (but not other sensory ganglia) can maintain myelin sheaths. 81 In practical terms, microscopic evaluation of SGC during nonclinical toxicity studies typically is limited to examining cell numbers and their organization in H&E-stained sections (e.g., increased cellularity, residual nodules [of Nageotte]).27,75-77 In some instances, evaluation of SGC molecular markers may be helpful in evaluating SGC reactions to ganglion injury. For this purpose, SGC may be highlighted using IHC methods to demonstrate the expression of markers like connexin 43 (Cx43, a gap junction protein); glial fibrillary acidic protein (GFAP, a glial-specific intermediate filament); and glutamine synthetase (GS), an enzyme involved in metabolism of the neurotransmitters glutamate and gamma amino butyric acid (GABA).80-82,87 Activated SGCs in injured ganglia proliferate and may be identified by the increased intensity of GFAP labeling and higher gap junction numbers.88-95
Schwann cells are glial cells that surround neuronal processes in the PNS, including those in the core of sensory ganglia as well as somatic nerves and most cranial nerves and also the distal portions of spinal nerve roots. Schwann cell subtypes are classed as “myelinating” or “non-myelinating” depending on the degree to which they generate myelin.96-98 Schwann cells sustain axonal integrity in health and support axonal repair following injury to the axon or its neuron.99-102 In practical terms, microscopic evaluation of ganglion-associated Schwann cells during nonclinical toxicity studies generally is confined to examining cell numbers and their organization (e.g., increased cellularity) in the core of ganglia and adjacent nerve roots in H&E-stained sections. If necessary, Schwann cell reactions may be examined using IHC to evaluate the expression of markers like myelin basic protein (MBP), peripheral myelin protein 22 (PMP22), or S100 for myelinating Schwann cells103,104 or GFAP for non-myelinating Schwann cells. 105 Expression of matrix metalloproteinase-9 (MMP9) is increased in Schwann cells at sites of nerve injury. 106
Macrophages are reported to comprise 5% to 20% of resident cells in DRG. Macrophages are found in two locations within DRG: near the neuron-SGC units but outside the SGC basement membrane, where they provide trophic support to sensory neurons,107-109 and near blood vessels in ganglionic centers and nerve roots, where they function in immune surveillance. 110 Normal cell turnover in adult mice replaces most DRG macrophages (80%) every 3 months. 111 Upon acute ganglion injury, numbers of activated macrophages increase by 4- to 8-fold within a week.110,112 In practical terms, macrophages cannot be reliably distinguished from SGC using morphologic features in H&E-stained sections in nonclinical species (e.g., rodents, dogs, NHPs). Where necessary, these cell types may be differentiated using molecular markers. For example, SGCs express S100 while macrophages do not. 113 Microglia have been described in DRG based on the presence of cells that strongly express the marker ionized calcium-binding adaptor molecule 1 (IBA1), 114 but IBA1 and other microglial markers (e.g., CD11b, F4/80) are also expressed by macrophages.115,116 By convention, DRG cells that express macrophage markers are termed macrophages because microglial cells in the CNS have a different embryonic origin. 117
Species differences
In practical terms, the anatomy and histology of site-matched neural components (ganglia, nerves, and spinal cord) affected by gangliotoxic agents is comparable across common species of laboratory animals (i.e., “test systems” for toxicity studies) and humans. Nonetheless, some structural differences do exist among species, many of which are influenced by the organism’s size and sensory needs (e.g., heightened kinesthetic and tactile acuity in NHPs and humans). For instance, the lumbar segments giving rise to the bulk of the sciatic nerve vary slightly among species 26 : L3 to L5 for mice, 118 L4 to L6 in rats, 118 and L4 to L7 in NHPs (depending on the NHP species).119,120 Lumbar DRG in rats contain 12,000 to 15,000 sensory neurons 52 while lumbar DRG in humans harbor about 27,000 to 38,000 neurons. 121 Moreover, for humans, a larger body size is associated with larger lumbar DRG compared with individuals of average size. 121 The size of DRG neurons in rodents is smaller than those of NHPs and humans,42,119 and rodent DRG have relatively little connective tissue compared with human DRG. 42 Subtypes of DRG sensory neurons as defined by molecular expression of ion channels, neurotransmitters, and other signaling components are present in different proportions in rodent DRG compared with NHP (cynomolgus macaque) and human DRG.42,122 In particular, rodents and primates exhibit different patterns of nociceptor expression and function,42,122 indicating that bridging studies may be needed to select relevant nonclinical species for developing analgesic products for human use. The dorsal funiculi of the spinal cord are much larger in non-rodents compared with rodents (reaching approximately double the quantity as a percentage of total white matter in primates relative to rats 123 ), likely as a result of the higher dependence on fine sensation in the lower body needed in animals with prehensile digits.
Collection, Processing, and Evaluation of Ganglia for Nonclinical Studies
Regulatory guidance expects morphologic evaluation of the CNS and PNS for nonclinical toxicity testing of biopharmaceutical products.124,125 For example, general guidance by the U.S. Food and Drug Administration (FDA) specifically mentions brain, spinal cord, and sciatic nerve as tissues that should be considered for histopathologic evaluation. 126 In situations, where TA-related changes are anticipated in neural tissues, a need to consider further tissue sampling is implied in the existing FDA guidance recommending examination of “. . . samples representative of all major areas and elements of the brain, spinal cord, and peripheral nervous system”; 127 neither ganglia nor nerves are specified, so sponsors must decide what elements of the PNS should be evaluated. Similarly, guidance by multi-national consortia convened to standardize requirements for nonclinical studies (e.g., International Conference on Harmonisation [ICH]) generically mention the need for histopathology data but do not prescribe neural tissues to evaluate in compiling such data.19-21 Globally harmonized guidance by the Organisation for Economic Co-operation and Development (OECD) for general toxicity testing of chemicals—typically applied to environmental and occupational chemicals rather than small molecule pharmaceuticals—specifies evaluation of the brain, spinal cord, and nerve (sciatic or tibial) but not ganglia,128-130 but the OECD guidance for neurotoxicity testing includes DRG as a tissue for histopathologic evaluation. 131
Points to Consider in Sampling and Processing Sensory Ganglia for Nonclinical Toxicity Studies
Current industrial best practice for neuropathology evaluation of the PNS for nonclinical toxicity testing recommends collection and evaluation of DRG (Figure 1) and TG (Figure 2) in situations where somatic neurotoxicity is anticipated. 26 This best practice approach to neural tissue sampling and evaluation was deliberately constructed by an STP WG to address key guidance from FDA, ICH, OECD, and the U.S. Environmental Protection Agency (EPA). In addition, the best practices were conceived to improve identification and characterization of sensory changes induced by known “gangliotoxic” classes of biopharmaceutical agents while providing flexibility to sponsors in selecting a study design to best achieve their program objectives. A detailed technical explanation for collection and evaluation of DRG and TG in various species may be gleaned from other publications,26,29-31,36,45,132 but a brief consideration of ganglia sampling is given here to provide context for subsequent sections on tissue evaluation, diagnostic nomenclature, and data interpretation and reporting.
Briefly, the approach to collection and microscopic evaluation of ganglia during nonclinical toxicity testing differs with the likelihood of neurotoxicity (CNS or PNS) due to TA exposure. Four distinct scenarios are possible (Table 2) as detailed in ‘best practice” recommendations on PNS sampling, processing, and analysis produced previously by the STP. 26
STP “best practice” recommendations for collection and evaluation of sensory ganglia in nonclinical toxicity studies.
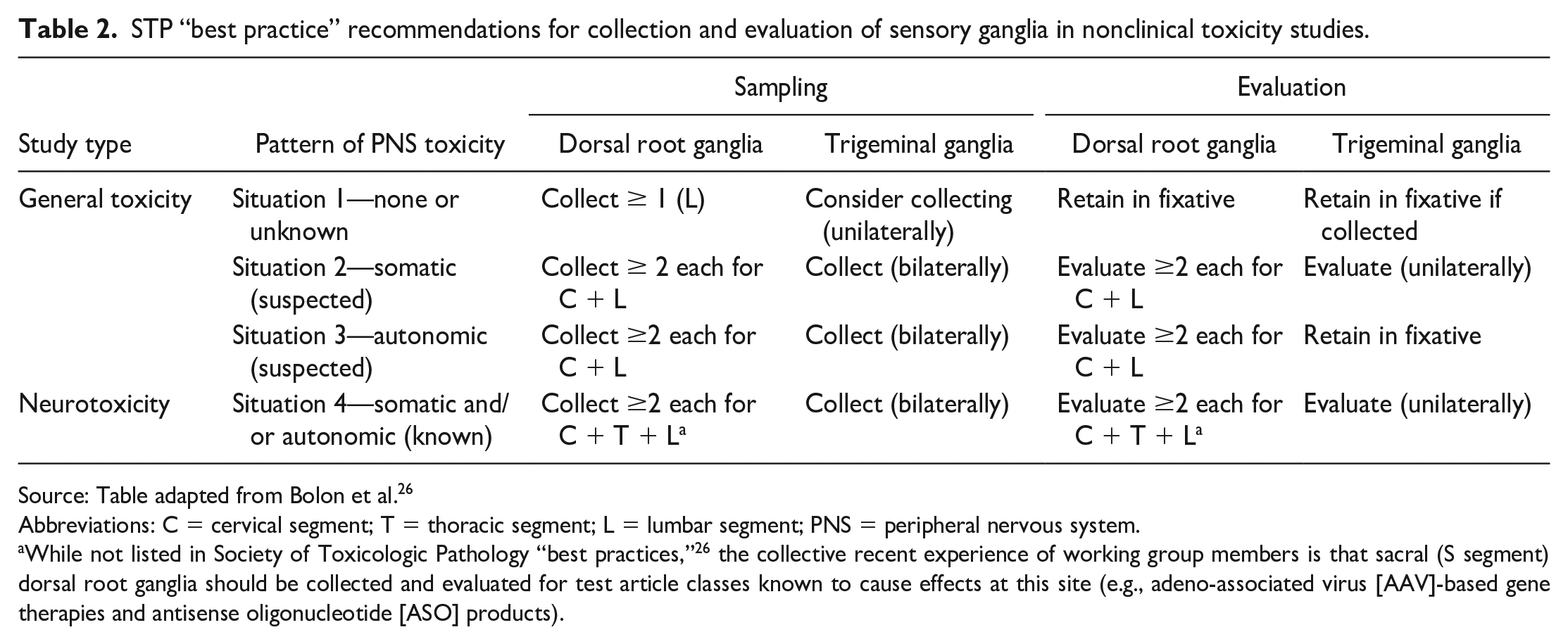
Source: Table adapted from Bolon et al. 26
Abbreviations: C = cervical segment; T = thoracic segment; L = lumbar segment; PNS = peripheral nervous system.
While not listed in Society of Toxicologic Pathology “best practices,” 26 the collective recent experience of working group members is that sacral (S segment) dorsal root ganglia should be collected and evaluated for test article classes known to cause effects at this site (e.g., adeno-associated virus [AAV]-based gene therapies and antisense oligonucleotide [ASO] products).
Situation 1 is applicable to general toxicity studies where the TA exhibits no or has unknown neurotoxic potential. In this scenario, STP best practices for PNS sampling 26 recommend that at least 1 DRG should be collected and retained in fixative just in case microscopic evaluation is necessary to explain findings in other neural tissues. For this purpose, a DRG corresponding to the lumbar intumescence (often acquired at L4-L5 in common nonclinical species) is typically chosen since this organ is associated with the hind limb nerve (usually sciatic but sometimes tibial) that is included in the list of protocol-specified tissues for routine general toxicity studies.24,128,129 Sciatic nerves need not be dissected all the way to the DRG for sampling; instead, sciatic nerve specimens are generally taken from one site (often middle or distal) or two sites (usually proximal and distal) to avoid potential trauma associated with having to remove vertebral bone that encases the junction of the nerve and DRG. In the collective experience of WG members, evaluation of DRG is rarely necessary for Situation 1. Accordingly, removal of DRG from the vertebral column at necropsy is not warranted for Situation 1 unless DRG are a protocol-specified tissue. A practical and rapid means of DRG retention for Situation 1 is to isolate short lengths of vertebral column (e.g., three vertebrae, which will span two intervening intervertebral disks and their pairs of DRG), remove extraneous soft tissues (e.g., hypaxial musculature) to facilitate tissue fixation, and leaving DRG in situ until needed. 26 For histological processing, rodent DRG may be left in situ as the vertebral column is decalcified and embedded while non-rodent DRG must be removed from the vertebral column and placed in tissue cassettes for processing. 26
Situation 2 is characterized by suspected somatic PNS neurotoxicity. In this setting, at least, 2 DRG each should be collected, processed, and evaluated for the spinal cord segments associated with the cervical and lumbar intumescences (i.e., those related to the origins of the forelimb and hind limb nerves 26 ). Locations at which these DRG may be harvested are given in Table 1.
Situation 3 is distinguished by clinical signs of potential autonomic PNS neurotoxicity. Collection and evaluation of key autonomic ganglia as well as at least 2 DRG each from cervical and lumbar segments is recommended to identify possible TA-related findings in the autonomic and somatic PNS. 26
Situation 4 is associated with expected or known neurotoxicity. In this setting, the best practice recommendations note that at least 2 DRG each should be collected and evaluated for cervical, thoracic, and lumbar segments 26 ; in the collective experience of WG members, it is also beneficial to collect and evaluate two DRG from the sacral segment as these ganglia are susceptible (and perhaps more sensitive compared with more cranial DRG for some TAs [e.g., AAV gene therapy vectors]) to some classes of gangliotoxic agents. Moreover, collective experience has shown that evaluation of more than 2 DRG per spinal cord segment is useful to avoid missing subtle or variable changes.133-135 For this reason, the WG suggests that more than 2 DRG be collected bilaterally for each spinal cord segment and either retained in fixative (in situ or isolated and held in tissue cassettes) or processed to blocks and evaluated if the TA comes from a biopharmaceutical product class with suspected or known gangliotoxic potential. Spinal nerve roots typically are collected and processed while remaining attached to their DRG.
Best practices call for collection of TG for Situations 2, 3, and 4 above (i.e., where some evidence of a TA-related neuroactive effect is suspected or evident), with evaluation of TG for Situations 2 and 4 (i.e., where the neuroactive effect is concentrated in the somatic nervous system). 26 In general, TG should be collected and evaluated for those TA known to induce findings in DRG. 26
Several factors are primary points to consider when devising a sampling protocol for sensory ganglia.
Control groups and group sizes. In studies with direct CNS delivery, inclusion of an appropriate concurrent control group (i.e., animals of the same genetic background [breed, stock, or strain from the same vendor as the experimental animals], sex, and age kept under the same husbandry conditions) is important as is provision of suitable group sizes to discriminate TA-related findings from procedure-related findings. This “sufficient group size” concept is also important for study designs with parenteral dosing (e.g., intravenous [IV] administration of AAV agents) due to the current paucity of historical control data for DRG (and TG) in nonclinical species as well as the wide range of inter-animal variability observed to date in nonclinical studies for AAV9-related gene therapy (GTx) products.
Number of DRG for microscopic examination from each spinal cord segment. Findings for a particular test article are not necessarily consistent across all DRG in an individual in terms of findings or their severity. 26 For TAs with known or expected PNS toxicity, the previously published STP best practices recommended examination of at least 2 DRG from the cervical, thoracic, and lumbar segments of the spinal cord (Table 2). 26 However, the STP “Best Practice” paper 26 did not specifically address platforms with known DRG pathology where extensive DRG sampling is needed not only for microscopic evaluation but also to confirm biodistribution of the TA to sensory ganglia as a potential target tissue. In situations where gangliotoxic effects are expected, the collective experience of the WG members suggests that DRG be sampled from more than 2 segments from each spinal cord segment (Supplemental Table 2). Longitudinal bilateral samples of DRG containing the dorsal and ventral spinal nerve roots should be labeled to identify their location (see below for options).
Pre-existing knowledge regarding relative ganglion susceptibility. Certain TA classes are known to selectively damage some DRG to a greater degree than others. For instance, the DRG from lumbar and sacral segments appear to be more reproducibly affected, and to a greater degree, by AAV GTx.9,136 Accordingly, TAs of the AAV GTx class would benefit from a sampling scheme that includes several lumbar DRG (as stated in the existing STP best practices for PNS sampling 26 ) and also some sacral DRG (which are not mentioned in the STP best practices).
Unilateral vs bilateral DRG sampling. Like many other bilateral organs, xenobiotic-induced DRG findings typically manifest as bilateral changes. 26 Unilateral DRG sampling for microscopic evaluation conforms to STP best practice and is not uncommon for nonclinical general toxicity studies where gangliotoxic effects are not anticipated (Supplemental Table 3). However, bilateral DRG sampling (i.e., collecting both DRG for a given spinal cord segment) is helpful to explain intra-group variation (especially for studies with non-rodent species where group sizes are small) related to distinguishing among experimental procedure-related, TA-related, or spontaneous findings. Scientists may allocate each DRG from a pair for different approaches, such as one for morphologic evaluation and the other for quantitative molecular techniques where preserving the morphology is not essential (e.g., biodistribution, genomics [DNA], transcriptomics [RNA], proteomics, enzyme assays). This approach of parallel usage for paired DRG from the same animal can increase the confidence and rigor of data.
Study objectives. Sampling protocols for sensory ganglia and spinal cord may need to be modified from the published STP best practices 26 (Table 2) to effectively address the study objectives related to assessing possible gangliotoxic effects. Several options (for ganglionic sampling, scoring criteria, and data analysis) exist depending on the main purpose(s) of the study (Tables 3-5 and Supplemental Tables 2 and 3). The reason for modified sampling in such cases is that STP best practices are designed to optimize screening for structural effects, which may be managed with a few DRG. However, nonclinical studies for AAV and ASO products frequently include both efficacy and toxicity endpoints, and multiple DRG and spinal nerve roots may need to be allocated among microscopic evaluation and molecular assays to achieve other study aims (e.g., localization of TA biodistribution). In such instances, DRG for molecular assays may need to be collected and frozen first to limit RNA degradation, and DRG for in situ molecular pathology procedures (e.g., IHC and/or ISH [in situ hybridization]) may require specialized processing conditions that will necessitate collection of more than 2 DRG for each spinal cord segment.
Sample identification and embedding schemes. The manner for identifying the DRG location may need to be adjusted depending on the study objectives (Supplemental Tables 3 and 4). For routine safety assessment studies for regulatory submission, it is a common practice to place all DRG for a specific spinal cord segment in a single cassette because the knowledge of the exact segment and side (i.e., left or right) of each DRG is not required as long as sufficient DRG from each spinal cord segment are examined. 26 In this case, all DRG for the cervical segment would be placed in one cassette, all DRG from the thoracic segment would be in a second cassette, and all DRG from the lumbar segment would be held in a third cassette. In contrast, for studies with efficacy and quantitative molecular endpoints (biodistribution, etc.), each DRG may be placed in its own cassette, so that, the exact position it occupied in the animal (e.g., “L4” or “L5, left side”) is known. Individual identification is used if the knowledge of the exact level of DRG sampled is likely to be helpful in correlating the morphologic changes with the molecular endpoints, clinical signs, functional endpoints, and/or to findings in peripheral nerves. Placement of many DRG from all spinal cord segments (e.g., cervical, thoracic, and lumbar) in one cassette may be used on occasion to maximize the amount of tissue available for screening. 26 This latter approach is seldom employed for nonclinical studies since it limits the ability to correlate in-life clinical signs and biodistribution data linked to specific spinal cord segments with microscopic findings in DRG. An alternate approach for processing DRG from multiple spinal cord segments in a single block while retaining information regarding their precise location is to use a tissue microarray (TMA) mold (e.g., 4 mm, 15 core) in processing. In general, this TMA option is limited to investigative studies rather than nonclinical safety screens.
Route of administration. Sensory ganglia closest to sites of direct CNS delivery—cervical DRG and TG for intracerebroventricular (ICV) and intracisternal (i.e., intra-cisterna magna [ICM]) routes vs. lumbar and sacral DRG for lumbar intrathecal (IT) puncture—do not necessarily exhibit a higher incidence or increased severity of TA-related findings. With IT catheter delivery, it is useful to collect and examine multiple DRG (more than 2 per spinal cord segment) including at the catheter tip level (where the TA concentrations may be higher) to optimize the likelihood of identifying any ganglionic changes induced by the TA or the procedure. In particular, the possibility that DRG findings might be greater on the side where the catheter is placed is an argument for bilateral collection of DRG from a given spinal cord level.
Sampling spinal nerve roots and spinal cord. When gangliotoxic effects are suspected, evaluation of nerves and the spinal cord provides valuable insights regarding the potential TA-related findings within the DRG.25,26 The dorsal funiculi of the spinal cord contain ascending axons arising directly from DRG sensory neurons, so these white matter tracts may exhibit changes indicative of degeneration affecting DRG axons (assuming examination at appropriate time points). It should be noted, however, that procedure-related findings associated with direct IT delivery by catheter can confound the interpretation of TA-related findings in the spinal cord white matter tracts. When interpreting histopathologic effects, TA-related changes often are bilateral though not necessarily symmetrical and typically exhibit a dose response while procedure-related lesions usually are distributed randomly, often are asymmetric, and do not exhibit a dose response. Since DRG may have changes that would not necessarily be manifested in the spinal cord and/or nerves, examination of spinal cord should not be considered a replacement for examination of spinal nerve roots and DRG. The STP best practice recommendations for spinal cord sampling for nonclinical general toxicity studies suggest sampling of the cervical (C1-C2), thoracic (T6-T8) and lumbar (L3-L6) segment.25,26 These recommendations are aligned with the DRG and TG samplings proposed by the current STP WG except that the current WG also affirms the need to collect sacral spinal cord (S1-S3). Prosectors should recall that the level of lumbar and sacral spinal cord segments are displaced cranially such that the L4 to L5 spinal cord segments in rats and NHPs are found within vertebrae L1 to L2 (Table 1).25,26 For direct delivery studies with potential or expected DRG findings, spinal cord evaluation is not an acceptable substitute for a well-designed PNS assessment (e.g., DRG plus spinal nerve roots plus spinal cord) because DRG exhibit several changes that will not have counterparts that are visible in the spinal cord, especially at early time points.
Spinal cord evaluation is very important when TA-related findings are possible in DRG. Transverse spinal cord sections provide a summary view of axonal integrity for nerve fibers arising from all DRG caudal to that spinal cord level, and so offer an indirect means of screening more DRG than are included in the actual histopathologic assessment. Transverse and oblique or multiple transverse sections from cervical and lumbar spinal cord should be evaluated for all four situations defined above (Table 2), 26 and similar samples from thoracic spinal cord should be examined for Situations 2, 3, and 4 (Table 2). 26 Additional sections can be added to include macroscopic findings caused by IT delivery of xenobiotics. For AAV and ASO modalities, similar specimens from the sacral spinal cord may be sampled and evaluated if this location was employed as the “injection site” for IT delivery into the lumbar cistern.
Need for DRG isolation. The DRG of rodents may be examined in situ in the decalcified vertebral column, 25 ideally trimmed in the longitudinal plane, so that, multiple DRG are evident. If examined microscopically, the WG suggests that DRG slated for assessment include those at the lumbar intumescence (L4 to L5, which receives the largest complement of fibers for the sciatic nerve52,139,140), or alternatively the cervical intumescence (C4 to C7, which receives the largest complement of fibers from the nerves entering the brachial plexus), so that the organs examined are associated with the large nerves that are often affected in cases of TA-induced gangliotoxic injury.
Routine and special stains. The set of histological stains and markers to use in evaluating sensory ganglia is determined by both the objective(s) of the study and pathologist preference. The WG members employ a tiered approach in selecting the appropriate procedures for use in a given nonclinical study. In most cases, the microscopic evaluation of DRG and TG is weighted toward a basic survey (Tier 1). For routine nonclinical studies, H&E on 5-μm-thick paraffin sections is a serviceable stain for demonstrating cell density and cytoarchitectural detail. This single stain is often the only procedure used in initial screening during studies where no sensory ganglion pathology is anticipated. In some studies, other markers are applied to serial sections to further characterize TA-related injury in sensory ganglia (Tier 2). The choice of additional method(s) as well as their timing (i.e., contemporaneous with the H&E section or post hoc to explain findings seen in the H&E section) depends on the preference of the pathologist and institution. In the collective experience of the WG members, procedures undertaken to characterize TA-related effects may be applied to any cell population in DRG or TG. However, the most helpful methods typically assess the glial or inflammatory responses. SGCs usually are examined by an IHC method for GS although some institutions employ IHC for GFAP since this CNS marker often is already available in a histology laboratory. Leukocyte identities usually are examined using markers for lymphocytes—commonly for T-cells as a class (CD3) or for subsets that regulate (CD4) or carry out (CD8) immune responses against neurons—and macrophages (e.g., CD68 or IBA1). Additional glial and leukocyte markers, such as CD20 for B-lymphocytes, may be used occasionally although in most cases this cell type comprises a modest portion of TA-related ganglionic reactions. Death of ganglionic neurons in DRG and TG may be probed, typically in investigative studies (usually performed in rodents) rather than standard safety assessment studies, using special procedures, such as anti-cleaved caspase 3 IHC26,141 or TUNEL.141-144 Death detection (i.e., degeneration and necrosis) using Fluoro Jade (either the B or C variants, which are standard neuronal death markers in the CNS)145,146 is not consistently effective in sensory ganglia,26,146 and in the collective experience of WG members does not afford any additional sensitivity compared with what is attainable using routine H&E-stained sections. Moreover, DRG and TG (especially those of NHPs) also contain collagen and lipofuscin (i.e., “wear and tear” pigment granules in the cytoplasm of neurons), both of which autofluoresce. The potential to interpret false-positive autofluorescent signals as neuronal degeneration is an additional deterrent to using Fluoro Jade as a death-detecting stain (particularly in NHP ganglia). Additional histological procedures typically are performed during discovery phase or mechanistic/investigational studies (Tier 3). These markers are used to explore potential mechanisms of ganglionic injury but seldom are used during routine nonclinical toxicity studies because their inclusion is not essential for compiling the data set needed for lesion interpretation and human risk assessment.
Case Study 1—ganglion assessment via independent evaluation of multiple features.
Background: An AAV9-based test article in artificial CSF (aCSF) vehicle was delivered to cynomolgus macaques once at a single dose by intrathecal injection at the lumbar cistern. Control AAV-injected animals were necropsied at 45 and 120 days following test article delivery as part of a nonclinical toxicity study.
Methods: Two DRG each were evaluated for the cervical, thoracic, and lumbar spinal cord segments. Multiple features were evaluated and graded at both time points. Sample grading criteria are given for these components in descriptive terms (which may be replaced by specific numerical criteria at the discretion of the pathologist).
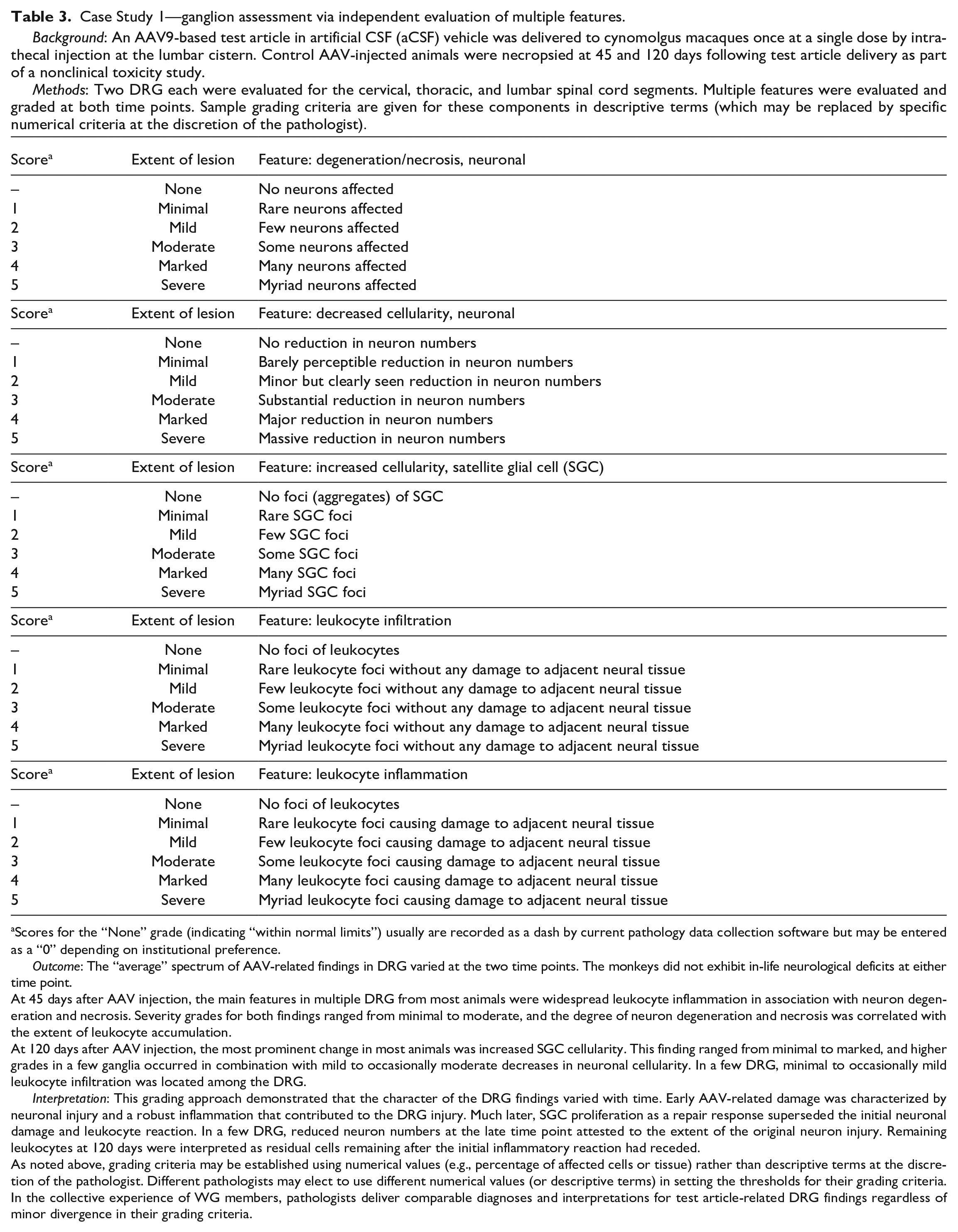
Scores for the “None” grade (indicating “within normal limits”) usually are recorded as a dash by current pathology data collection software but may be entered as a “0” depending on institutional preference.
Outcome: The “average” spectrum of AAV-related findings in DRG varied at the two time points. The monkeys did not exhibit in-life neurological deficits at either time point.
At 45 days after AAV injection, the main features in multiple DRG from most animals were widespread leukocyte inflammation in association with neuron degeneration and necrosis. Severity grades for both findings ranged from minimal to moderate, and the degree of neuron degeneration and necrosis was correlated with the extent of leukocyte accumulation.
At 120 days after AAV injection, the most prominent change in most animals was increased SGC cellularity. This finding ranged from minimal to marked, and higher grades in a few ganglia occurred in combination with mild to occasionally moderate decreases in neuronal cellularity. In a few DRG, minimal to occasionally mild leukocyte infiltration was located among the DRG.
Interpretation: This grading approach demonstrated that the character of the DRG findings varied with time. Early AAV-related damage was characterized by neuronal injury and a robust inflammation that contributed to the DRG injury. Much later, SGC proliferation as a repair response superseded the initial neuronal damage and leukocyte reaction. In a few DRG, reduced neuron numbers at the late time point attested to the extent of the original neuron injury. Remaining leukocytes at 120 days were interpreted as residual cells remaining after the initial inflammatory reaction had receded.
As noted above, grading criteria may be established using numerical values (e.g., percentage of affected cells or tissue) rather than descriptive terms at the discretion of the pathologist. Different pathologists may elect to use different numerical values (or descriptive terms) in setting the thresholds for their grading criteria. In the collective experience of WG members, pathologists deliver comparable diagnoses and interpretations for test article-related DRG findings regardless of minor divergence in their grading criteria.
Case Study 2—ganglion assessment by emphasizing evaluation of one or two key features.
Background: An AAV9-based test article in phosphate-buffered saline vehicle was administered to rhesus macaques once by intravenous injection at a single dose. Control and AAV-injected animals were necropsied at 28 days following test article delivery as part of a nonclinical toxicity study.
Methods: Three DRG each were evaluated for the cervical, thoracic, and lumbar spinal cord segments. The primary diagnostic feature was the percentage of affected tissue (as defined by numbers of injured neurons and axons and/or the extent of leukocyte infiltration) in the most severely affected ganglion.
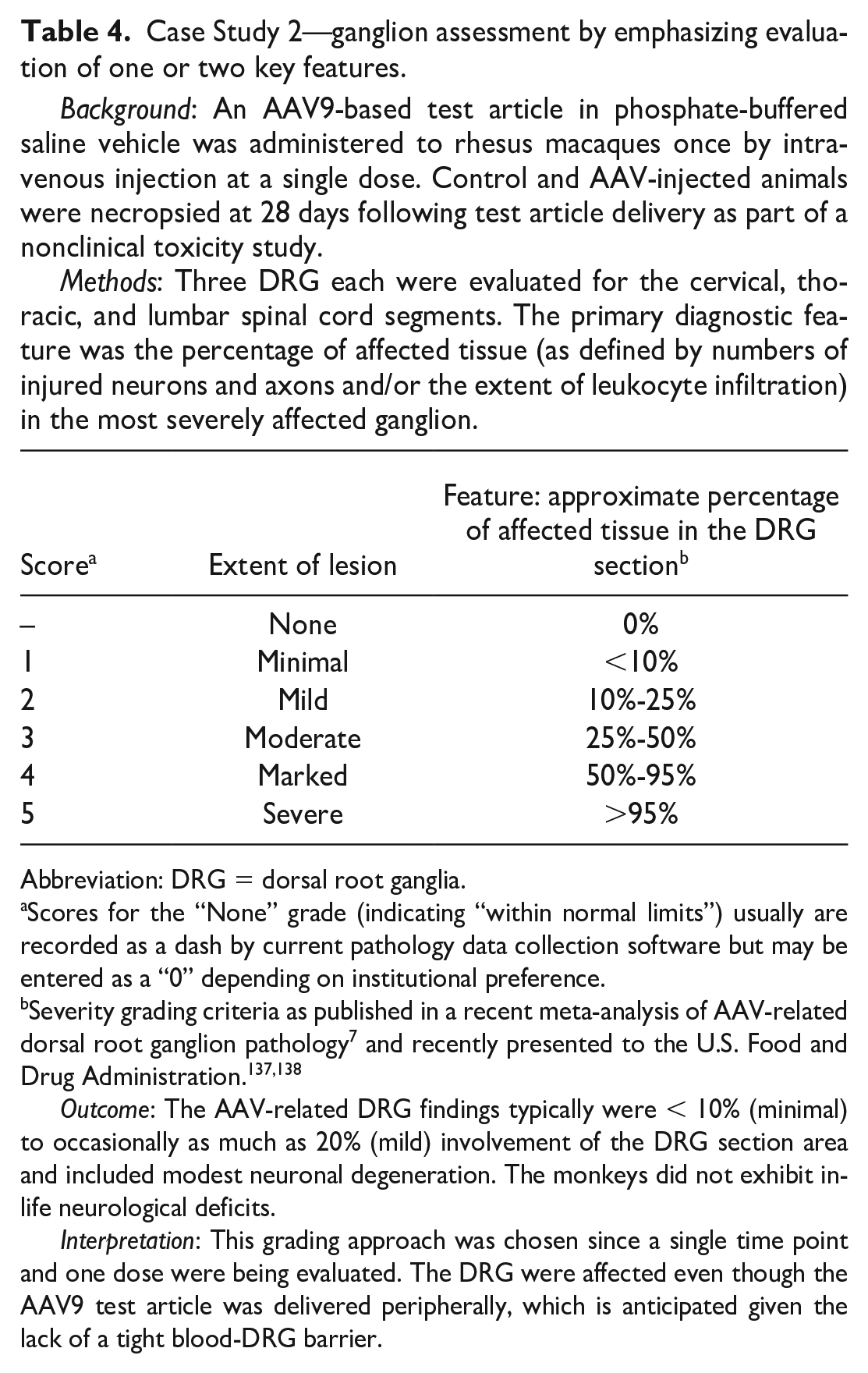
Abbreviation: DRG = dorsal root ganglia.
Scores for the “None” grade (indicating “within normal limits”) usually are recorded as a dash by current pathology data collection software but may be entered as a “0” depending on institutional preference.
Severity grading criteria as published in a recent meta-analysis of AAV-related dorsal root ganglion pathology 7 and recently presented to the U.S. Food and Drug Administration.137,138
Outcome: The AAV-related DRG findings typically were < 10% (minimal) to occasionally as much as 20% (mild) involvement of the DRG section area and included modest neuronal degeneration. The monkeys did not exhibit in-life neurological deficits.
Interpretation: This grading approach was chosen since a single time point and one dose were being evaluated. The DRG were affected even though the AAV9 test article was delivered peripherally, which is anticipated given the lack of a tight blood-DRG barrier.
Case Study 3—ganglion assessment via evaluation of features in each individual ganglion.
Background: Two AAV9-based test articles with the same transgene but different promoters were administered (each to one male cynomolgus macaque) once by intrathecal injection at the cisterna magna at the same dose level. Both animals were necropsied at 28 days following test article delivery as part of a discovery study to compare the transduction efficiency (transgene expression) and microscopic findings in DRG between the two AAV constructs.Methods: Four DRG each were evaluated for the cervical, thoracic, lumbar, and sacral spinal cord segments. For each segment, the DRG of each segment were assigned to microscopic evaluation or a molecular endpoint. The extent of DRG involvement (based on the percentage of tissue with mononuclear cell infiltration) was evaluated and graded for each individual DRG. Thereafter, weighted cumulative scores were calculated by adding all individual DRG severity grades, where the maximum cumulative score for a segment would be 20 (4 DRG × the highest severity grade of 5 in each segment) and the maximum cumulative score for the animal would be 80 (4 segments × the peak segmental score of 20).
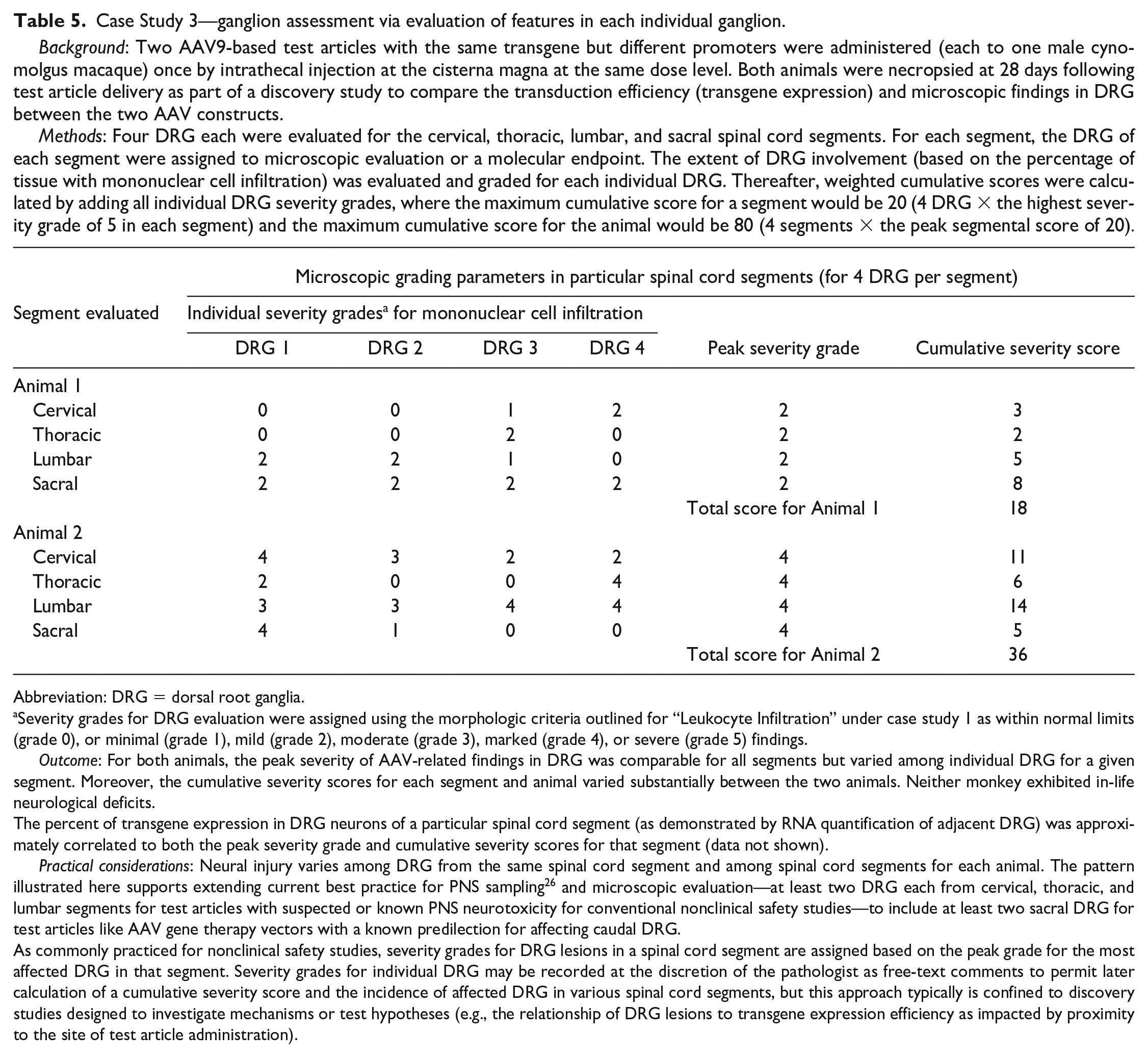
Abbreviation: DRG = dorsal root ganglia.
Severity grades for DRG evaluation were assigned using the morphologic criteria outlined for “Leukocyte Infiltration” under case study 1 as within normal limits (grade 0), or minimal (grade 1), mild (grade 2), moderate (grade 3), marked (grade 4), or severe (grade 5) findings.
Outcome: For both animals, the peak severity of AAV-related findings in DRG was comparable for all segments but varied among individual DRG for a given segment. Moreover, the cumulative severity scores for each segment and animal varied substantially between the two animals. Neither monkey exhibited in-life neurological deficits.
The percent of transgene expression in DRG neurons of a particular spinal cord segment (as demonstrated by RNA quantification of adjacent DRG) was approximately correlated to both the peak severity grade and cumulative severity scores for that segment (data not shown).
Practical considerations: Neural injury varies among DRG from the same spinal cord segment and among spinal cord segments for each animal. The pattern illustrated here supports extending current best practice for PNS sampling 26 and microscopic evaluation—at least two DRG each from cervical, thoracic, and lumbar segments for test articles with suspected or known PNS neurotoxicity for conventional nonclinical safety studies—to include at least two sacral DRG for test articles like AAV gene therapy vectors with a known predilection for affecting caudal DRG.
As commonly practiced for nonclinical safety studies, severity grades for DRG lesions in a spinal cord segment are assigned based on the peak grade for the most affected DRG in that segment. Severity grades for individual DRG may be recorded at the discretion of the pathologist as free-text comments to permit later calculation of a cumulative severity score and the incidence of affected DRG in various spinal cord segments, but this approach typically is confined to discovery studies designed to investigate mechanisms or test hypotheses (e.g., the relationship of DRG lesions to transgene expression efficiency as impacted by proximity to the site of test article administration).
Assigning Severity Grades for Sensory Ganglia Microscopic Findings in Nonclinical Studies
The severity grade is a frequent corollary component for a histopathologic diagnosis that helps differentiate TA-associated effects from incidental (“background”) findings and aids in defining no observed adverse effect levels (NOAEL) or equivalent cutoff values during hazard characterization.147,148 Such grades are used to sort morphologic findings into groups of similar degree for interpretation. For this reason, severity grades are essential tools for discriminating treatment-related effects from incidental background findings and establishing thresholds (e.g., highest non-severely toxic dose [HNSTD], lowest observed adverse effect level [LOAEL], or NOAEL) for risk assessment. 149 An STP “Points to Consider” paper on severity grading provides general guidance for defining relevant grading criteria. 147 Typically, severity grades are devised as non-linear, semi-quantitative, ascending levels identified using integers and terms. Common grades include “within normal limits” (represented in data collection software by the number 0 or more often a dash 147 ) and multiple tiers for findings, often rendered as 1 for “minimal,” 2 for “mild” (or “slight”), 3 for “moderate,” 4 for “marked,” and 5 for “severe.” Representative examples of a tiered severity grading approach for findings induced by AAV-based TAs are illustrated for DRG (Figure 4), spinal cord (Figure 5 [H&E] and Figure 6 [IBA1]), and peripheral nerves (Figure 7). Alternatively, the study pathologist may choose to develop a scale with fewer levels (e.g., by omitting the “severe” grade). The key point is that the grading scale should be so constructed that meaningful interpretations may be made for findings that are of a common severity.
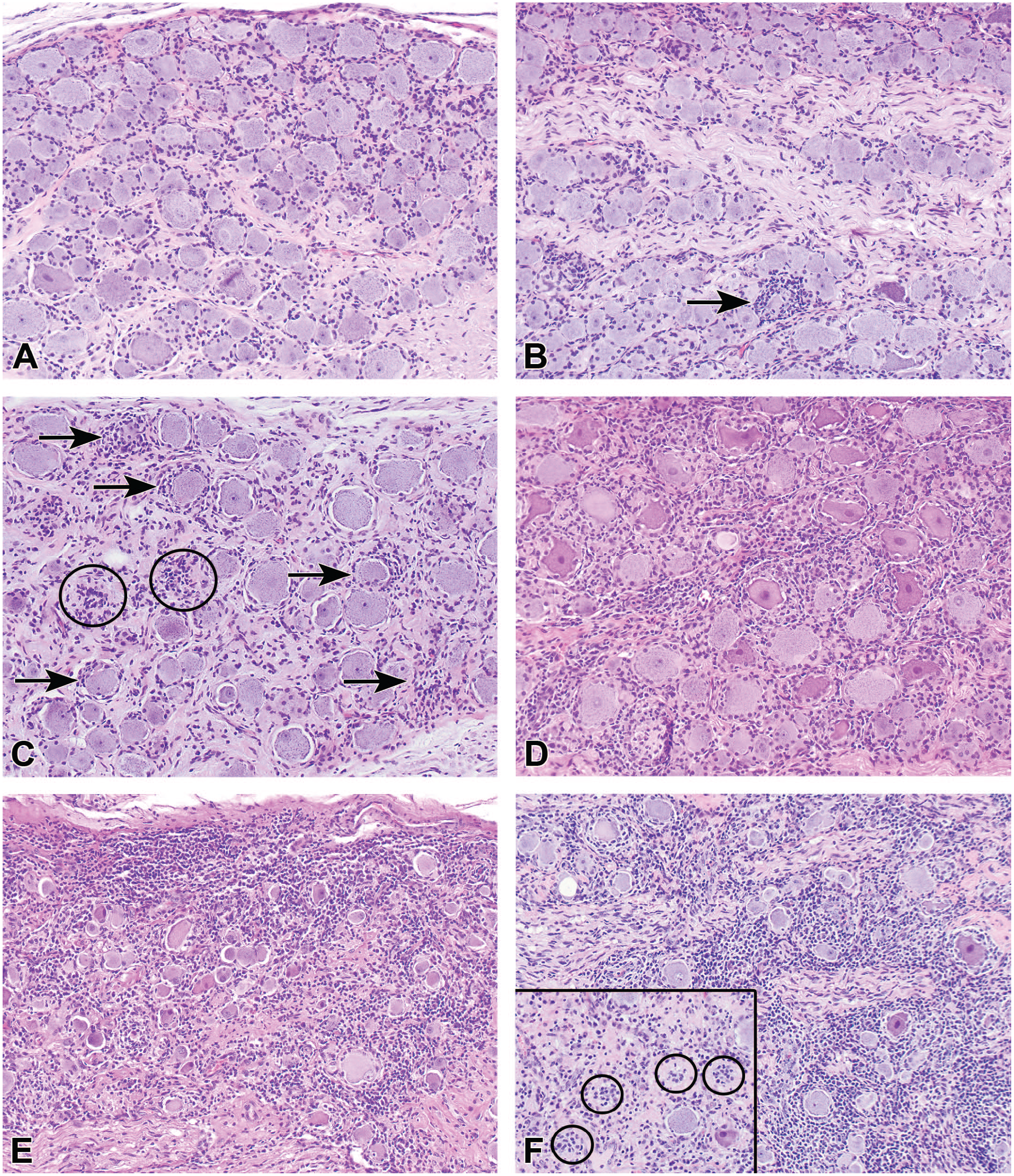
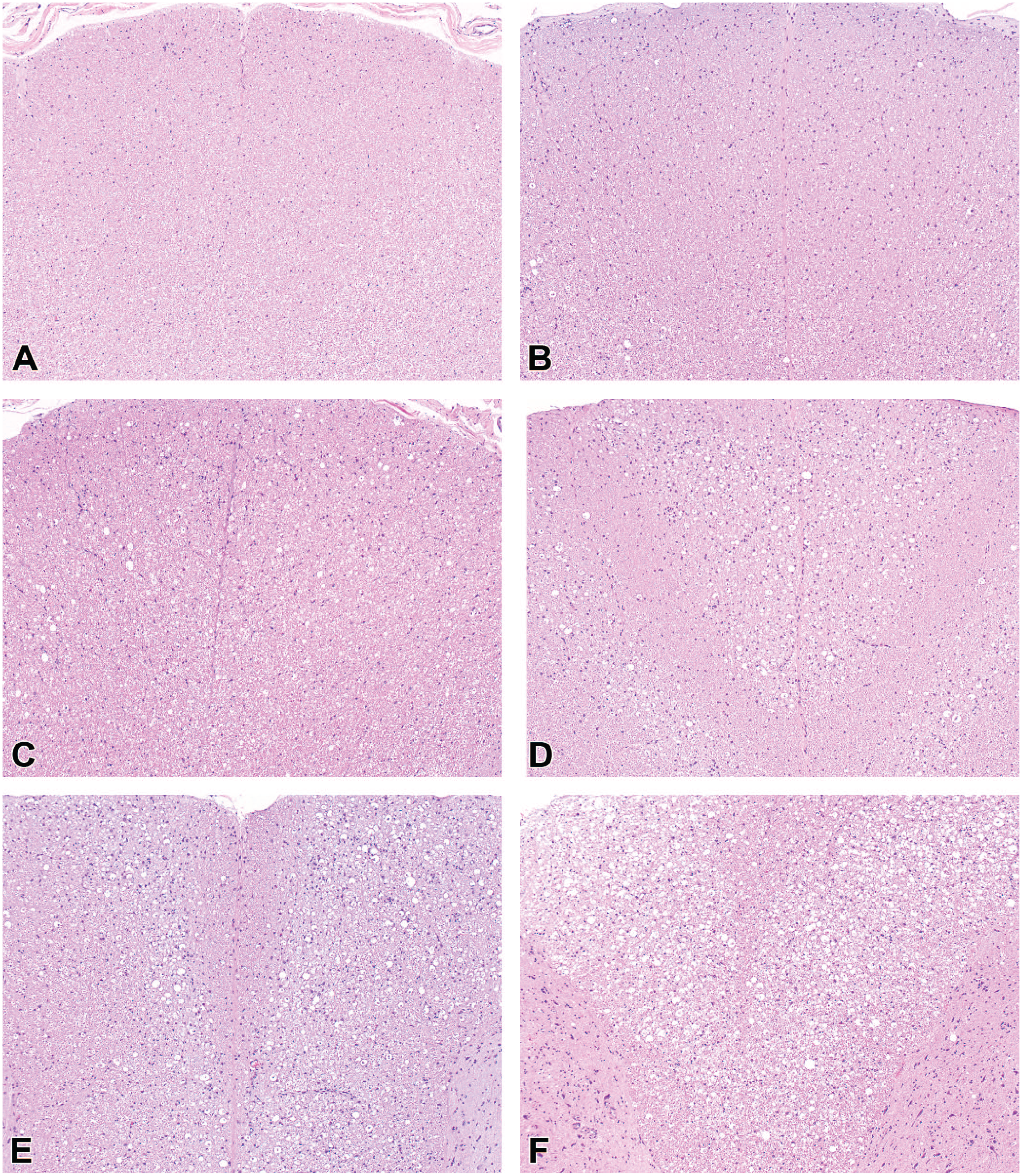
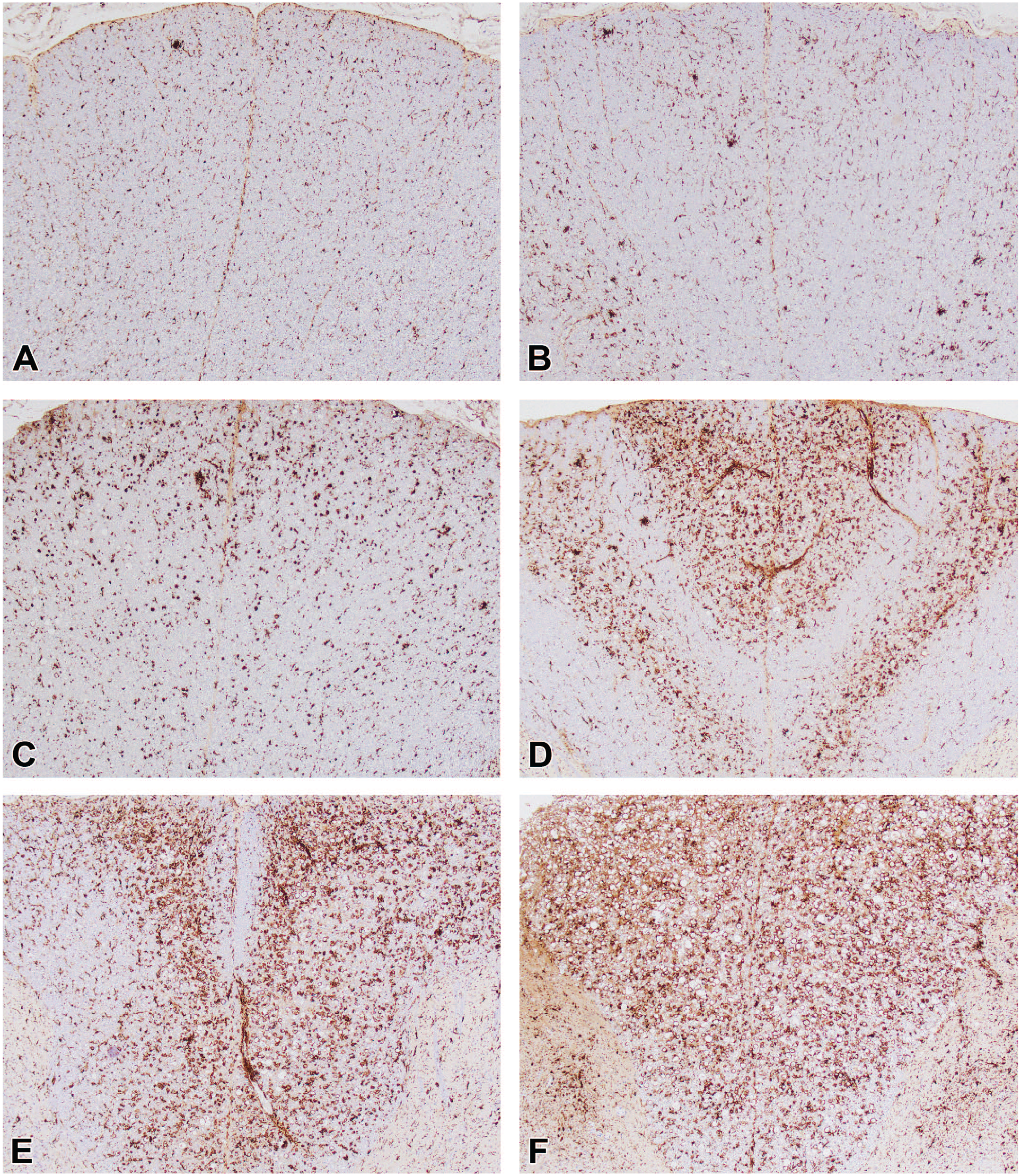
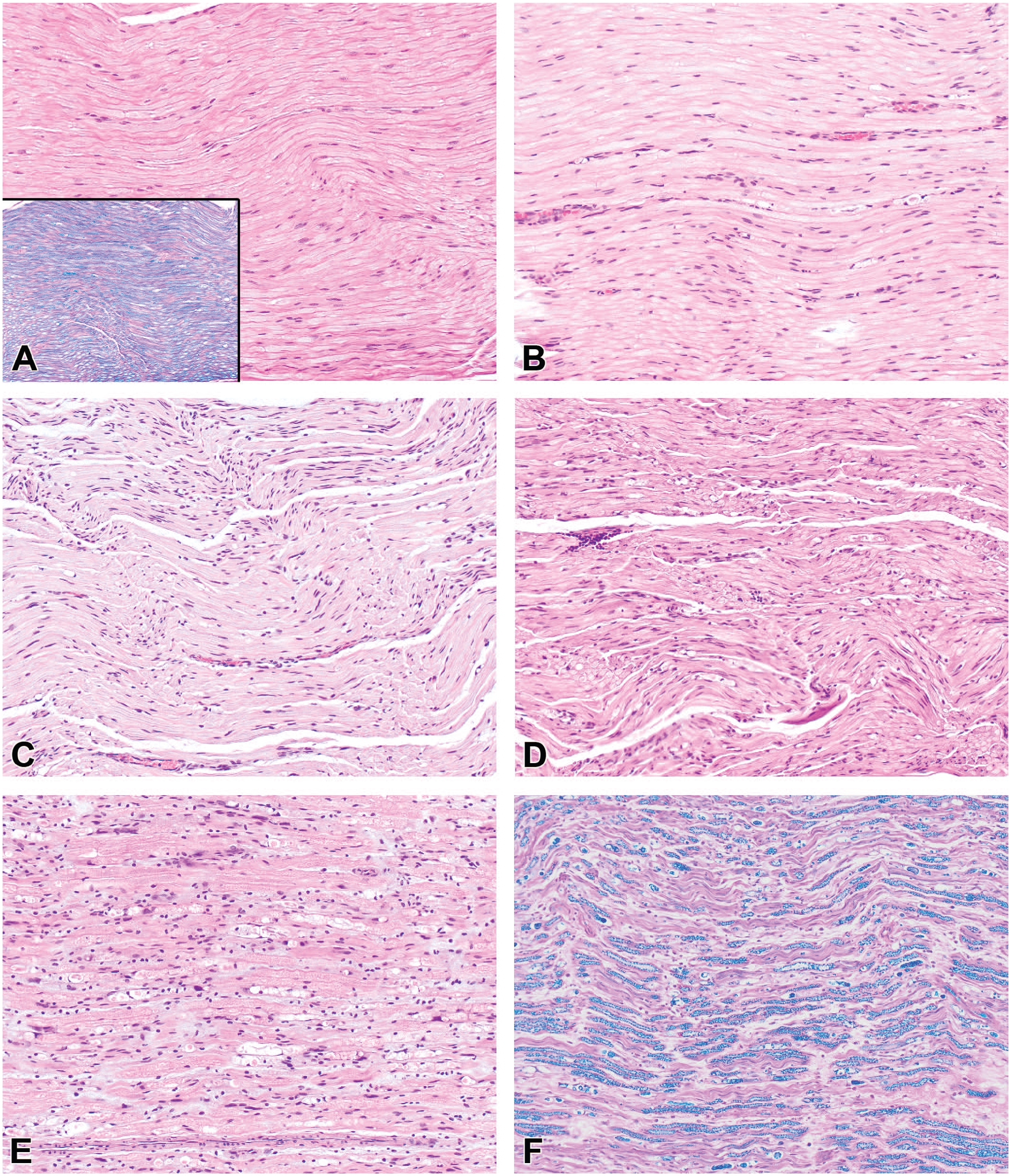
Severity grades typically will be based on such attributes as the distribution, extent, and/or severity of the microscopic findings rather than speculation regarding their clinical implications. The WG suggests that specific criteria be established for the various semi-quantitative grades for each diagnostic term as generally accepted grading approaches have yet to be defined for PNS findings associated with DRG neurotoxicity. Two general approaches may be used in selecting criteria for grading ganglionic and associated neural changes in nonclinical safety studies. The first approach incorporates independent grading criteria for multiple features. For instance, sensory neurons, SGC, and leukocytes in DRG as well as axonal changes in spinal cord and nerves may be categorized separately to assess the waxing and waning of effects concentrated on distinct cell populations (see Table 3, Case Study 1, as an example). The main advantage of a multi-component approach is that assigning the grades for specific cell types may capture changes of biological consequence in the lesion character over time, thereby permitting a nuanced interpretation of the evolving TA-related effects. For example, AAV-induced DRG injury in early phases often is indicated by the presence of neuronal degeneration and necrosis and/or substantial inflammation while in later phases effects on neurons may be detected as a reduced number of neurons and/or an increased number of SGC aggregates (residual nodules of Nageotte) with minimal or no inflammation. In the experience of the WG, assigning separate grades for specific findings often provides valuable insight into their pathogenesis and progression. The second approach is to emphasize one or two key features in the analysis. For example, a recent review and meta-analysis of AAV-induced DRG pathology in NHPs 7 used the percentage of the tissue exhibiting neuronal degeneration and/or leukocyte infiltration in an average high-power microscopic field as the criterion (see Table 4, Case Study 2, as an example). This approach allows correlation of chosen key microscopic features with other study parameters (e.g., biodistribution) and/or across multiple studies. The diagnostic approaches in Case Studies 1 and 2 follow the current convention for nonclinical toxicity studies of using the “peak” (most severe) response in a single DRG to set the severity grade for DRG findings in an affected spinal cord segment (if findings are collated for DRG in the cervical, thoracic, lumbar, and sacral segments separately) or for the animal as a whole (if findings are lumped under the single heading “DRG”). A third approach, employed infrequently for exploratory/investigative studies (see Table 5, Case Study 3, as an example), assigns grades to individual ganglia (using one or more features) and then combines these grades to produce a cumulative score for ganglia of a given spinal cord segment and/or the entire animal. This third approach is undertaken when the objective is to assess variations in the biological responses of individual ganglia rather than to survey for the range of effects that might develop in DRG as a group by screening a small subset of DRG. As screening is the usual objective of nonclinical toxicity studies intended for regulatory review, DRG evaluation typically will employ an approach consistent with that described for Case Study 1 (Table 3) or Case Study 2 (Table 4).
Since study pathologists generally147,151 use different cutoff values in establishing thresholds that separate severity grades, the criteria for severity grades in each Case Study are provided as examples and not to imply inherent superiority of one approach over another. For all three approaches, the grading criteria used by the study pathologist need to be clearly described in the pathology report, and ideally should be tied to specific morphologic attributes that can be semi-quantified to set thresholds for each severity grade. This point is of critical importance since the study pathologist is responsible for constructing the grading criteria they use in analyzing their study. Divergent professional experience among pathologists means that each individual will emphasize slightly different features in establishing their criteria and assigning their diagnostic grades. Such minor inter-individual differences are unavoidable and typically inconsequential. 147
Historical Control Data for Sensory Ganglia Microscopic Findings in Nonclinical Studies
Historical control data (HCD) of histopathologic findings in the DRG, TG, and other ganglia are not robust because these tissues traditionally have not been collected in Good Laboratory Practice (GLP)-compliant general toxicity studies. This historical practice has been evolving such that collection and evaluation of these tissues have increased within the last 5 years due to direct toxicity of these organs by some classes of xenobiotics (certain viral vectors for GTx and many anti-cancer chemotherapeutics). An additional complication when interpreting HCD for ganglionic findings are substantial variations in the kinds and numbers of ganglia collected across studies and across companies. However, in the collective experience of WG members both neuronal degeneration and necrosis are uncommon findings in the DRG, TG, and other ganglia in naïve/untreated nonclinical species, especially in cynomolgus monkeys. When neuronal degeneration and/or necrosis are present in ganglia as a TA-related finding, they are usually associated with multifocal or coalescing mononuclear cell infiltrates and increased numbers of SGC aggregates (i.e., collections of reactive [hyperplastic/hypertrophic] SGC). Importantly, neuronal autophagy (an incidental background finding that commonly occurs in the absence of leukocyte and SGC reactions in DRG and TG of control NHP77,150) has been diagnosed as “neuronal degeneration” or “neuronal necrosis” in many institutions until very recently, so HCD for these two findings in ganglia are likely unreliable in the near term. Inflammatory infiltrates in ganglia also may efface neuronal alterations since affected neurons are often obscured when numerous leukocytes are present.
Determining Adversity for Sensory Ganglia Microscopic Findings in Nonclinical Studies
A primary aspect of nonclinical toxicity studies is determining whether effects associated with TA exposure are adverse or non-adverse. This decision is necessary to set threshold values, such as the NOAEL and LOAEL, that are essential in defining the starting dose for subjects in Phase I clinical trials. The pathologist contributes to this effort by identifying findings (i.e., diagnoses), characterizing their extent (i.e., severity grading), and then interpreting their potential biological relevance. An STP “Best Practices” paper on determining, communicating, and using adverse effect data provides general guidance for making adversity decisions. 152 In accordance with these best practices, only TA-related effects that cause harm to the animal are considered to be adverse. Decisions regarding adversity are not linked solely to a specific severity grade152,153 but also depend on the context (the affected tissue and cell population, associated functional changes or lack thereof, etc.). Thus, the rationale for adversity decisions regarding TA-related findings should be given in the pathology report or study report. 152
Adversity determinations are influenced by the nature of the effect, the affected neural cell population(s), the constellation of associated tissue reactions, the clinical signs (if any) that might be attributed to such microscopic findings, and TA distribution (including the degree of transgene expression). Some tissue changes are deemed to be “inherently adverse,” including neuronal necrosis (as terminally differentiated neurons are not replaced in most neural tissues). 153 Thus, in principle, a single necrotic DRG sensory neuron in a test animal exhibiting no in-life neurological signs would be interpreted as adverse. Importantly, adversity decisions do not depend on the disease indication for which the TA is being developed; instead, adverse findings in nonclinical studies may be acceptable in clinical settings where the expected therapeutic benefit outweighs the potential risk. In practice, the study pathologist considers the degree of injury to ganglionic neurons (as well as along their central and peripheral projections) in the context of the other factors that might be contributing to or mitigating the injury (e.g., associated glial and leukocyte responses) in making adversity decisions.
A consideration in generating a pathology report for a nonclinical toxicity study is which threshold value to use when adverse findings are observed in ganglia at all dose levels in a study. In general, TA being developed for oncology indications20,22 or severe, life-threatening disorders 154 employ the HNSTD standard while TA being investigated for other indications utilize the NOAEL (or sometimes LOAEL) as the cutoff. In the experience of WG members, some classes of non-oncology TAs (especially AAV-based GTx) often induce non-dose-dependent ganglion effects at all dose levels, so that an NOAEL cannot be defined. For such instances, the non-oncology indication for which the TA is being developed is a severe terminal condition (e.g., a congenital or senescent neurodegenerative disease) for which no effective drug is available. In such cases, an HNSTD may be a more suitable approach for setting the threshold value in communicating adversity.
Points to Consider in the Study Design and Reporting of Sensory Ganglia Microscopic Findings in Nonclinical Studies
Data collection, interpretation, and reporting for findings in sensory ganglia (DRG, TG, and nerve roots) should be tailored to the study objectives, which often drive the overall study design. This section briefly discusses principal elements of several common study designs as well as adversity decisions as they apply to studies where DRG (or TG) are an expected target tissue.
Study Design Considerations
GLP-compliant or non-GLP nonclinical toxicity studies
Collection and microscopic evaluation of multiple DRGs per spinal cord segment (e.g., cervical, thoracic, lumbar, and sacral) for TA that are expected or known to produce DRG findings (e.g., AAV, antineoplastic agents, ASO) necessitated the development of multiple approaches regarding how to most effectively present the findings. One approach for reporting severity is assigning the highest severity grade seen in any DRG within that segment as the grade representing the maximal response (“peak grade”) for that spinal cord segment. This option may be used regardless of whether DRG for a given segment are processed in a single cassette or each DRG is processed in its own cassette. This “peak grade” approach for the segment is typically used where the reporting expectation is for a fast, simple and conservative interpretation (see Case Study 1 [Table 3] and Case Study 2 [Table 4] for examples). Another approach sometimes used in non-GLP nonclinical toxicity studies is to separately collect, process, and evaluate individually identified DRG (e.g., C5 or L4 in their own cassettes), record findings for each DRG individually, and then subcategorize the severity and incidence under the spinal cord segment of origin to assess the severity and extent within the spinal cord segment (see Case Study 3 [Table 5] for an example). A third possible approach, a “tissue comment” option, may be used to make notes on severity variation and/or distribution. The WG members typically advocate for using the “peak grade” approach when crafting pathology reports of nonclinical toxicity studies destined for regulatory review as it maintains some degree of important positional information (i.e., the maximal lesion for a particular spinal cord segment) while greatly reducing the time and labor needed to separately assess many ganglia per animal. More importantly, the complexity of the data tables is reduced to one entry (i.e., “peak grade”) per spinal cord segment, which often is sufficient for meaningful interpretation in making risk assessment decisions.
An alternative rapid screening-focused design that is used for collecting, processing, and reporting DRG findings in nonclinical toxicity studies where the TA has no or unknown gangliotoxic potential is to process a single cassette that contains several ganglia from all spinal cord segments. 26 This nonspecific limited information approach is useful when DRG assessment is desirable but there is no perceived need to identify the segment from which the ganglion was collected. In this setting, the data set typically is limited to the “peak” grade in any given DRG, though on occasion the number of affected DRG also may be tabulated.
Research/efficacy/exploratory/investigational studies
Many product development programs may benefit by having the pathologist interface with early discovery or research scientists to provide diagnoses and severity grades that facilitate understanding how TA biodistribution, the host immune response, or other factors might impact ganglionic function and structure. Non-pathologists should be reminded that pathology data are descriptive and that severity grades are ordinal data (whole integers) with no true zero and thus are not continuous or normally distributed. Therefore, it generally is inappropriate to apply parametric statistical tests based on a group mean severity grade. 147 If group means and statistical analysis are needed, nonparametric tests suited to ordinal data may be applied to severity grades or quantitative methods (e.g., morphometry or stereology) may be undertaken to permit digital image analysis. 155 In most cases, quantitative analyses of ganglia are not performed for routine nonclinical toxicity studies because these data do not offer additional value in terms of adversity decisions or risk assessment relative to their cost, labor-intensive nature, and length of additional time required to acquire such measurements.
As noted above for conventional GLP or non-GLP toxicity studies slated for regulatory review, several options may be used in reporting microscopic findings in DRG and other ganglia, their nerves, and spinal cord, but assigning straightforward “peak” severity grades is generally sufficient. In the research setting, the “individual DRG” approach (see Case Study 3 [Table 5]) may be useful since microscopic findings may be recorded for one member of a DRG pair while evaluating other endpoints (such as biodistribution analysis) in the other member of the pair. This “individual DRG” approach may also allow for evaluation of potential regional sensitivities of different ganglia across the spinal cord segment (e.g., L1 vs L5). The main disadvantages of this “individual DRG” approach are the greatly increased number of blocks per animal and the very complex data tables in which each ganglion is listed as a separate tissue. An adaptation of this individual ganglion-oriented approach to reduce the number of blocks would be to process all collected DRG for a particular spinal cord segment together in one cassette (e.g., L1, L4, L6), with the DRG having the lowest number per level being placed closest to the slide label (L1) and the DRG having the highest number (L6) positioned farthest from the label. The complexity of report text tables can be reduced by bringing forward only the “peak” severity grade for each spinal cord segment while keeping details regarding the severity grade for each DRG in the Individual Animal Data tables under comments (see Case Study 3 [Table 5]). Some WG members in discovery research areas have developed approaches that attempt to compare across animals in a study, for example by using a severity grade instead of the incidence for a key morphologic feature, where a “cumulative total grade” is generated for each spinal cord segment (or animal) by adding the severity grades for all DRG in that segment (or animal). This “cumulative total grade” approach for each segment or the whole animal has been helpful in generating a relative comparison across dose groups, in teasing out the average dose response from extreme outlier responses, in comparing the sensitivity of DRG for different spinal cord segments for an individual animal, and in comparing the responses for the same spinal cord segment among all animals within the same study. In addition, the individual severity grade range for each spinal cord segment may be communicated in the report narrative.
Reporting Considerations
Regardless of the tissue processing and reporting strategy, certain information with respect to the sensory ganglia evaluation should be considered for inclusion in the “Materials and Methods” section of the report. Detailed tissue collection and processing conditions are key for data interpretation and study comparability and reproducibility. Useful information will be the fixation procedure (e.g., immersion vs perfusion), embedding medium (e.g., paraffin vs epoxy resin), and possibly the approach to tissue identification (e.g., 1 DRG per cassette vs all DRG for each segment in a cassette). The diagnostic nomenclature (i.e., glossary) and grading criteria for unusual findings should be incorporated in the report, either within the “Materials and Methods” section or as appendices. Findings will need to be described; WG members typically arrange these descriptions by organ, starting with sites of primary TA-induced injury (e.g., DRG and TG) and then adding sites where secondary responses develop (e.g., spinal cord, spinal nerve roots, nerves). Statistical analysis generally is not warranted for pathology data, but if performed the ordinal severity grades (i.e., non-continuous, semi-quantitative data with no true zero value) must be tested using nonparametric statistics.147,156 Data visualization software may be useful in identifying major trends in such complex data sets. 157
A common challenge for interpreting changes in spinal nerve roots and spinal cord is to attempt to discriminate TA-related findings from experimental procedure-related findings (e.g., in the case of IT catheter placement/presence) and spontaneous background findings.158,159 In such cases, damage to neural tissue at or near the site of direct CNS administration typically is imputed to local physical trauma associated with the delivery device 70 while distant effects are considered to be TA-related or spontaneous findings. This simplistic approach is tempered by the facts that distant effects in neural tissues also may reflect radiation of neurite (axon and dendrite) degeneration outward from traumatized neurons at the site of administration while findings at the site of administration may represent TA-related cell injury. Successful interpretation thus relies on the availability of sufficient concurrent and historical control animals to characterize the kinds and severities of procedure-related and spontaneous findings.
Correlation Between Functional and Structural Findings in Sensory Ganglia During Nonclinical Toxicity Studies
Several methods may be used to probe the health of PNS circuits and structures, including the integrity of sensory ganglia. In principle, many of these may be performed in nonclinical laboratory animal species as well as humans. Practically, though, the labor-intensive nature and/or limited sensitivity of these methods preclude their routine use in nonclinical toxicity testing. This section briefly considers these methods, the rationale for their use, and their utility when applied to nonclinical laboratory animal species.
In-life neurological signs observed during nonclinical toxicity studies are a common starting point for assessing nervous system function. Injured sensory neurons may exhibit loss-of-function signs, gain-of function signs, or both. Principal loss-of-function signs related to DRG include reduced or absent reflexes (i.e., hyporeflexia or areflexia, respectively) and/or sensation (i.e., hypoalgesia or analgesia, respectively). Loss-of-function signs reflect sensory neuron dysfunction and/or axon damage that result in sensory denervation of the body periphery. Typical gain-of-function signs include pruritus, spontaneous pain behaviors (e.g., autotomy [self-amputation] 160 ), and increased or prolonged sensory responses to noxious stimuli (hyperalgesia), light static touch (static mechanical allodynia), moving light touch (dynamic mechanical allodynia), or cold stimuli (cold allodynia). Gain-of-function signs often are due to plasticity of viable neurons and synaptic inputs as well as fluctuations in the signaling molecule mélange within the DRG microenvironment. Signaling molecules that reach the DRG include blood-borne ligands, which easily gain entry through the fenestrated DRG blood vessels; paracrine ligands released by nearby hyperactive, damaged, or dying neurons, reactive SGC, resident macrophages cells, and/or infiltrating immune cells; or neurotransmitters released by central circuits anchored in the spinal cord and/or brain. Both gain- and loss-of-function can occur within the same disease process and in some cases may represent distinct points along a temporal progression of injury and shifting neurological signs.
Multiple methods can be used to assess in-life neurological signs during nonclinical toxicity studies. The simplest screen is a regular (e.g., daily) physical examination. More labor-intensive means of assessing neural function include the functional observational battery (FOB) and formal neurological examinations in larger species. Overt loss of function may be uncovered by quantitative electrophysiological tests, such as nerve conduction velocity (NCV) to detect delays in nerve impulse transmission (as can occur with loss of myelin) and Sensory Nerve Action Potential (SNAP) amplitude (to assess loss of sensory axons). Since most large-diameter nerves are mixed (i.e., containing motor and sensory nerve fibers) in laboratory animal species, NCV will provide an integrated signal that combines output for both motor nerve fibers and the A (large-diameter, heavily myelinated) subtypes of sensory nerve fibers. SNAP is likewise a measurement that is mediated mostly by large-diameter sensory neurons. Neither NCV nor SNAP are sufficiently sensitive methods for detecting damage to small-diameter nerve fibers,161,162 and decreased NCV is not a consistent feature of gain-of-function phenotypes mediated by large-diameter fibers, such as paresthesia or mechanical allodynia. Gain of function may be assessed by several techniques, including the von Frey assay (for static mechanical allodynia), hot plate- or infrared light-based assays (for thermal hyperalgesia), and similar methods. Incremental changes in cold sensation, dynamic mechanical allodynia, and proprioceptive deficits are much more difficult to detect than nociception, so these methods are not assessed in nonclinical toxicity studies though they may be used in specialized labs for investigative studies.
Soluble biomarkers in fluid samples (e.g., blood, CSF, urine) obtained by minimally or non-invasive methods are an area of intense interest as a means of monitoring neural injury.163,164 Currently, neurofilament light chain (NfL) and less commonly neurofilament heavy chain (NfH) proteins are the most common biomarkers of neural injury evaluated in nonclinical species and human patients, including neurodegenerative diseases, nervous system infections, and nervous system neoplasia as well as neurotoxic conditions.165-167 For example, serum neurofilament concentrations are increased in animal models and cancer patients with chemotherapy-induced peripheral neuropathy (CIPN).168-171 Blood and CSF levels of NfL have been demonstrated to be a useful indicator of sensory neuron injury in DRG of asymptomatic rodents (Fader et al. 172 and unpublished data) and NHPs9,172 following a single AAV-based GTx administration, and these levels correlated well with the severity of microscopic changes in DRG and nerves up to 12 months post-injection.9,172 Several factors, such as biofluid kinetics, clearance, and degradation as well as the use of immunosuppressive therapies, stage of disease and/or injury,173,174 variations related to lifestyle, age, 175 and procedure-related/handling effects 176 can affect circulating NfL values. In contrast to NfL, fluid levels of NfH are not a specific biomarker of neural degeneration/necrosis because the majority of neurofilament protein is present in axons. 177 Other biomarkers, including vascular endothelial growth factor A (VEGF-A) and various microRNAs (e.g., miR124a, miR183, miR388) also have been proposed as suitable biomarkers for nerve injury in nonclinical species. 178 For the autonomic PNS, quantification of the norepinephrine metabolite 3-methoxy-4-hydroxyphenylethylene glycol (MHPG) in urine represents a potential non-invasive biomarker for autonomic sympathetic dysfunction associated with ganglion neuron depletion. 179 This premise has been tested using guanethidine, an anti-hypertensive agent that inhibits neurotransmitter (norepinephrine) release at sympathetic synapses and has been reported to induce neuronal necrosis. 180 The European Union’s Innovative Medicines Initiative (IMI) Translational Safety Biomarker NeuroDeRisk project is investigating safety biomarkers for peripheral neurotoxicity by performing nonclinical studies in rats with multiple agents known to induce peripheral neuropathy.178,181
Intraepidermal nerve fiber (IENF) density in glabrous (non-haired) skin has been suggested as a means for showing structural abnormalities in sensory nerve fibers in both nonclinical laboratory animal species and humans.182-185 Skin biopsy paired with IHC is used to quantify small-diameter unmyelinated sensory fibers in skin biopsies from human patients as a diagnostic biomarker for small fiber sensory neuropathy and is also seen in sensory ganglionopathies where pathology is centered on neurons within the DRG.186,187 Assessment of IENF density has been used to investigate rodent and NHP models of small fiber neuropathies including CIPN. 185 While IENF density has been examined in control dogs, 188 it remains to be validated in the context of toxicant-induced neuropathy. Decreased IENF density detected microscopically may be seen with either pain or numbness, and these phenotypes may represent a progressive spectrum of signs induced by a single disease process. However, the wide variation in IENF density among individuals for a given anatomic site as well as among different areas in the same individual in many cases may yield sensory alterations in the absence of significantly altered IENF density. For this reason, IENF analysis is not performed as a screening tool in routine toxicity studies. Instead, IENF quantification in the nonclinical setting typically is limited to investigational studies related to evaluating a particular mechanistic hypothesis with structural results considered in light of other functional assays or clinical signs. For such studies, consistent sampling and considerable attention to tissue handling and processing are essential to avoid technical artifacts that might preclude a meaningful microscopic evaluation. In DRG, uninjured neurons adjacent to injured neurons in the same ganglion or even adjacent ganglia can help drive gain-of-function signs, such as mechanical allodynia and hyperalgesia. 94 Thus, the type, distribution, and severity of microscopic findings in DRG (and TG) and IENF beds is not necessarily predictive of the potential for sensory dysfunction. As a general consideration, the site targeted by the behavioral stimulus or electrical recording is an important consideration for comparisons across studies because innervation patterns vary along the limb while glabrous and non-glabrous (haired) skin are innervated by distinct subtypes of sensory neurons.43,189 A lack of a sensory function phenotype based on altered IENF density should be interpreted with caution in the absence of functional tests that specifically assess multiple, relevant modalities of sensation.
Function-structure correlations may exist for endpoints relevant to sensory ganglia, but the extent of any correlation is unclear (Table 6). The degree of correlation depends on the class of biopharmaceutical product. For AAV GTx studies in NHPs, neurological exams and loss-of-function electrophysiology tests have yielded some insight regarding how DRG findings correlate with functional outcomes. In AAV studies, recordings have revealed no changes in NCV; however, SNAP amplitude has been found to be an informative test that correlates with more severe microscopic findings. Decreased SNAP amplitude alone indicates a loss of large-diameter sensory axons while decreased SNAP amplitude accompanied by prolonged signal duration shows altered myelin integrity.190,191 While SNAP amplitude recordings in NHPs lack the sensitivity to detect minimal to mild AAV-related pathology, increased severity grades for DRG findings (e.g., marked) have been associated with significant reduction in SNAP amplitude over time, with either decreases from baseline or complete loss in SNAP amplitude at times ranging from 28 days post-injection to terminal endpoints (typically 13 weeks or longer). Based on a recent meta-analysis, 7 DRG findings in rhesus macaques after AAV administration were produced by multiple neurotropic vectors, transgenes, and doses, but more severe microscopic pathology grades were observed with higher doses (> 1e13 genome copies [GCs] per animal) administered by the IT route. Clinical signs of ataxia and/or tremors were only observed in three NHPs (of 204 total) that were administered vectors encoding green fluorescent protein (GFP) at high doses (> 1e13 GC per animal). In a similar study of a therapeutic transgene that included SNAP assessment, two NHPs (of 22 tested) showed no abnormalities on the standard neurological exam but exhibited decreased median nerve SNAP amplitudes that were associated with median nerve axonal degeneration and endoneurial fibrosis, thereby suggesting that SNAP measurements may be more sensitive than neurological examination as a means of detecting clinical dysfunction associated with ganglionic injury. 137
Correlation between dorsal root ganglion-related clinical observations/functional deficits and microscopic findings.
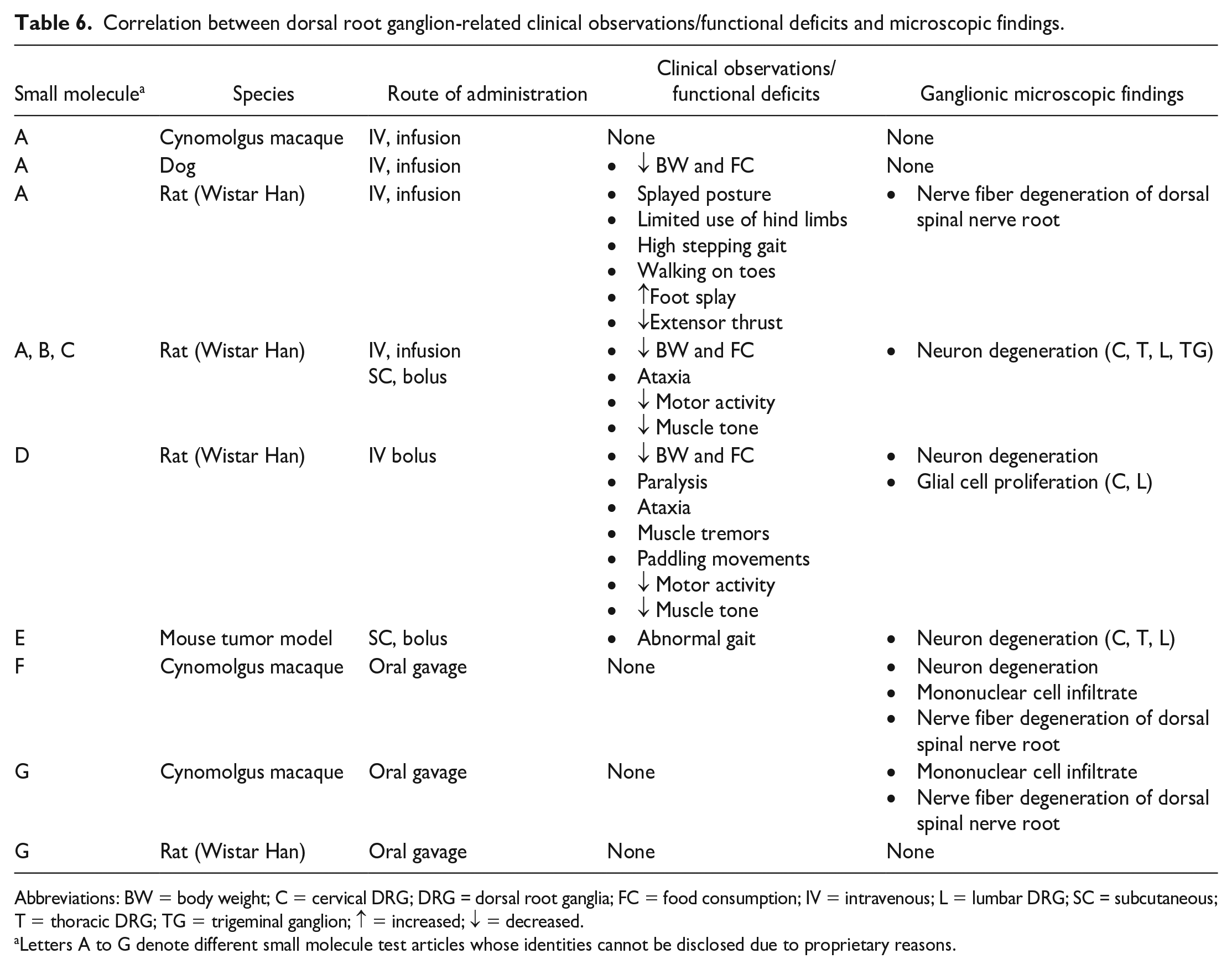
Abbreviations: BW = body weight; C = cervical DRG; DRG = dorsal root ganglia; FC = food consumption; IV = intravenous; L = lumbar DRG; SC = subcutaneous; T = thoracic DRG; TG = trigeminal ganglion; ↑ = increased; ↓ = decreased.
Letters A to G denote different small molecule test articles whose identities cannot be disclosed due to proprietary reasons.
DRG pathology findings in studies with other biopharmaceutical agents have identified only occasional links to altered sensory function, depending on the agent. AAV9 GTx 7 and ASOs (unpublished data) elicit a more consistent microscopic picture of DRG and TG changes, including neuronal degeneration (for AAV), increased SGC cellularity (for both TA classes), and mononuclear cell infiltration (especially for ASOs) or inflammation. These findings are seen in routine tissue preparations (i.e., formalin-fixed, paraffin-embedded, H&E-stained sections) for 1 or more ganglia in many animals for routine AAV GTx and ASO products, but these structural abnormalities are seldom accompanied by delayed NCV (reported for AAV) 7 and are only very rarely associated with any in-life neurological deficits in the canonical functional assays where they have been examined. 6 In comparison with AAV studies detailed above, when ASO-associated structural alterations are observed, the anatomic changes typically are much less severe, most often with only minimal to mild mononuclear cell infiltrates of spinal nerve roots, occasionally minimal increases in SGC cellularity, and no neuronal degeneration. All of these changes tend to be related to IT administration of more proinflammatory ASO sequences at very high doses (unpublished data). With repeated IT administration of ASOs in NHPs, TA-related microscopic findings have been reported in the spinal cord and associated ganglia that are most likely related to the degree of proinflammatory properties for individual ASO sequences (unpublished data); these findings may present as infiltration, mainly with mononuclear cells or macrophages containing basophilic granules (i.e., ingested ASO), or occasionally localized inflammation with associated nerve fiber degeneration and generally occur without any correlated clinical signs or gross observations referable to the microscopic findings. On occasion, however, high ASO doses may cause in-life neurological signs, such as ataxia; incoordination; hind limb weakness; increased hind limb muscle tone; hippus (rhythmic, spasmodic pupillary dilation and contraction); nystagmus (rapid, rhythmic eye movements); paralysis; loss of pain, grip, patellar reflex, and righting reflex; and/or proprioceptive deficits. 192
Antineoplastic chemotherapies with different molecular mechanisms induce dying-back degeneration of sensory nerve fibers and variable degrees of neuron degeneration, SGC activation, and inflammation in the DRG of humans, NHPs, and rodents.5,88,193,194 In addition, microscopic evaluation of sensory endings in skin biopsies shows a loss of IENF consisting of “dying-back” degeneration of nerve terminals and their distal axons. Functional correlates of the sensory lesions are well-documented in CIPN and include in-life gain-of-function neurological findings (e.g., bilaterally symmetrical paresthesia [distal burning and prickling sensations] and/or mechanical allodynia) and loss-of-function neurological findings (e.g., numbness, decreased NCV, and decreased SNAP amplitude).5,195-198 More specifically, clinical signs associated with taxanes, alkylating agents, and platinum compounds include symmetrical painful paresthesia, numbness in a “stocking-and-glove” distribution, and mechanical allodynia.5,195,199 Taxanes have also been associated with sensory loss and motor symptoms.5,195 Platinum compounds cause cold allodynia and hyperalgesia in earlier stages, shifting to loss of motor function and sensory neuropathy later. 5 Paclitaxel, docetaxel, and epothilones are associated with myalgia and myopathy. 195 Painful sensory neuropathy with neuropathic pain and paresthesia in distal limbs has been described with proteasome inhibitors and immunomodulatory agents.5,195 Muscle cramps have been reported for immunomodulatory agents. 5 Vinca alkaloids have been associated with painful sensorimotor neuropathy in a stocking-and-glove distribution, proprioceptive loss, areflexia, ataxia, tingling paresthesia, constipation, muscle weakness, and gait dysfunction.5,195 In addition to microscopic findings, rodents show gain-of-function phenotypes, including mechanical allodynia, decreased SNAP amplitudes and NCV, and electrophysiological abnormalities in recordings that target the neuronal cell body.5,199 Cisplatin, paclitaxel, and bortezomib reduce sensory or motor NCV along with microscopic evidence of axonal degeneration in mice after 4 weeks of treatment.5,200
Summary
Gangliotoxicity affecting sensory neurons is a biopharmaceutical class effect associated with certain products, most commonly cytotoxic chemotherapeutics and AAV-based gene therapies. Nonclinical toxicity studies for such products generally should incorporate minor adjustments in the design (e.g., collection of three or four spinal cord segments as well as many sensory ganglia and somatic nerves bilaterally), analysis (e.g., evaluation of multiple ganglia and nerves at least unilaterally), and reporting. Study pathologists and institutions retain considerable flexibility in choosing among various options as long as these organs are consistently examined. Morphologic changes in ganglia often occur in the absence of visible in-life functional alterations detectable by conventional behavioral and neurological testing techniques, so pathologists will be essential participants in the weight-of-evidence approach needed to determine whether ganglion changes represent the outcome of TA exposure, the experimental procedure, and/or background effects and whether or not they should be interpreted as adverse. This “Points to Consider” paper should be helpful in framing such factors when making product development and regulatory decisions.
Supplemental Material
sj-docx-1-tpx-10.1177_01926233231179707 – Supplemental material for Scientific and Regulatory Policy Committee Points to Consider: Sampling, Processing, Evaluation, Interpretation, and Reporting of Test Article-Related Ganglion Pathology for Nonclinical Toxicity Studies
Supplemental material, sj-docx-1-tpx-10.1177_01926233231179707 for Scientific and Regulatory Policy Committee Points to Consider: Sampling, Processing, Evaluation, Interpretation, and Reporting of Test Article-Related Ganglion Pathology for Nonclinical Toxicity Studies by Bindu M. Bennet, Ingrid D. Pardo, Basel T. Assaf, Elizabeth Buza, Sarah Cramer, LaTasha K. Crawford, Jeffery A. Engelhardt, Branka Grubor, James P. Morrison, Tanasa S. Osborne, Alok K. Sharma and Brad Bolon in Toxicologic Pathology
Footnotes
Acknowledgements
The authors thank Dr Elizabeth Galbreath for critical review of the manuscript and Ms Beth Mahler for her expertise in optimizing image quality.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
