Abstract
Analysis of intraepidermal nerve fibers (IENFs) in skin biopsy samples has become a standard clinical tool for diagnosing peripheral neuropathies in human patients. Compared to sural nerve biopsy, skin biopsy is safer, less invasive, and can be performed repeatedly to facilitate longitudinal assessment. Intraepidermal nerve fiber analysis is also more sensitive than conventional nerve histology or electrophysiological tests for detecting damage to small-diameter sensory nerve fibers. The techniques used for IENF analysis in humans have been adapted for large and small animal models and successfully used in studies of diabetic neuropathy, chemotherapy-induced peripheral neuropathy, HIV-associated sensory neuropathy, among others. Although IENF analysis has yet to become a routine end point in nonclinical safety testing, it has the potential to serve as a highly relevant indicator of sensory nerve fiber status in neurotoxicity studies, as well as development of neuroprotective and neuroregenerative therapies. Recently, there is also interest in the evaluation of IENF via skin biopsy as a biomarker of small fiber neuropathy in the regulatory setting. This article provides an overview of the anatomic and pathophysiologic principles behind IENF analysis, its use as a diagnostic tool in humans, and applications in animal models with focus on comparative methodology and considerations for study design.
Keywords
Introduction
Measurement of intraepidermal nerve fiber (IENF) density in skin biopsies is considered a standard and sensitive clinical tool for diagnosing some of the most common and debilitating forms of peripheral neuropathy (PN) in human patients. 1,2 In an effort to emulate clinically relevant outcomes and enhance the translational capacity of animal models of PN, many academic research groups now routinely perform IENF analysis as the primary morphologic indicator of sensory nerve damage in both large and small animal models. 3 –12 Intraepidermal nerve fiber analysis also has the potential to serve as a highly sensitive, translationally relevant indicator of sensory nerve fiber injury in nonclinical neurotoxicity testing and drug development studies.
This article provides an overview of the anatomic and pathophysiologic principles of IENF analysis and its use as a diagnostic tool in human patients with certain types of PN. The methodology used for IENF analysis in humans is presented in detail, as it has served as the foundation for work in animal models. We then summarize the relevant literature wherein IENF analysis has been applied to primate and rodent models of PN, with focus on comparative methodology and considerations for IENF study design. The use of IENF analysis in less common laboratory species is also briefly discussed, along with adaptations of the basic skin biopsy technique for specialized analyses, including assessment of neuroregeneration.
Overview of Cutaneous Innervation
The skin is densely innervated by several subtypes of myelinated and unmyelinated somatosensory nerve fibers. These fibers are broadly classified based on their axonal diameter and conduction velocity (from largest and fastest to smallest and slowest) as Aβ, Aδ, or C fibers. 13,14 Large myelinated Aβ fibers conduct the sensations of vibration and light touch and terminate within the dermis. 15 –17 Temperature and pain signals are transmitted by thinly myelinated Aδ fibers and nonmyelinated C fibers, often referred to as nociceptors. Anatomically, the dermal Aβ and Aδ fibers are associated with Schwann cells that secrete a basal lamina and also produce myelin sheaths. The C fibers in the dermis organize as Remak bundles, which typically contain 2 to 3 axons and are enveloped by a continuous basal lamina produced by nonmyelinating Schwann cells. The majority of dermal nerve bundles comprise unmyelinated sensory fibers that form a horizontally oriented nerve fiber plexus in the superficial dermis, from which vertical branches extend toward the skin surface. At the dermal–epidermal junction, axons lose their Schwann cell sheaths, enter the epidermis as single unmyelinated axons, and pass between the basal keratinocytes, finally terminating as free epidermal nerve fiber endings (Figure 1A). 18 Damage to Aδ and/or C fibers due to systemic illness, neurotoxicity, or direct nerve trauma leads to centripetal axonal dieback and IENF loss. 19 –21
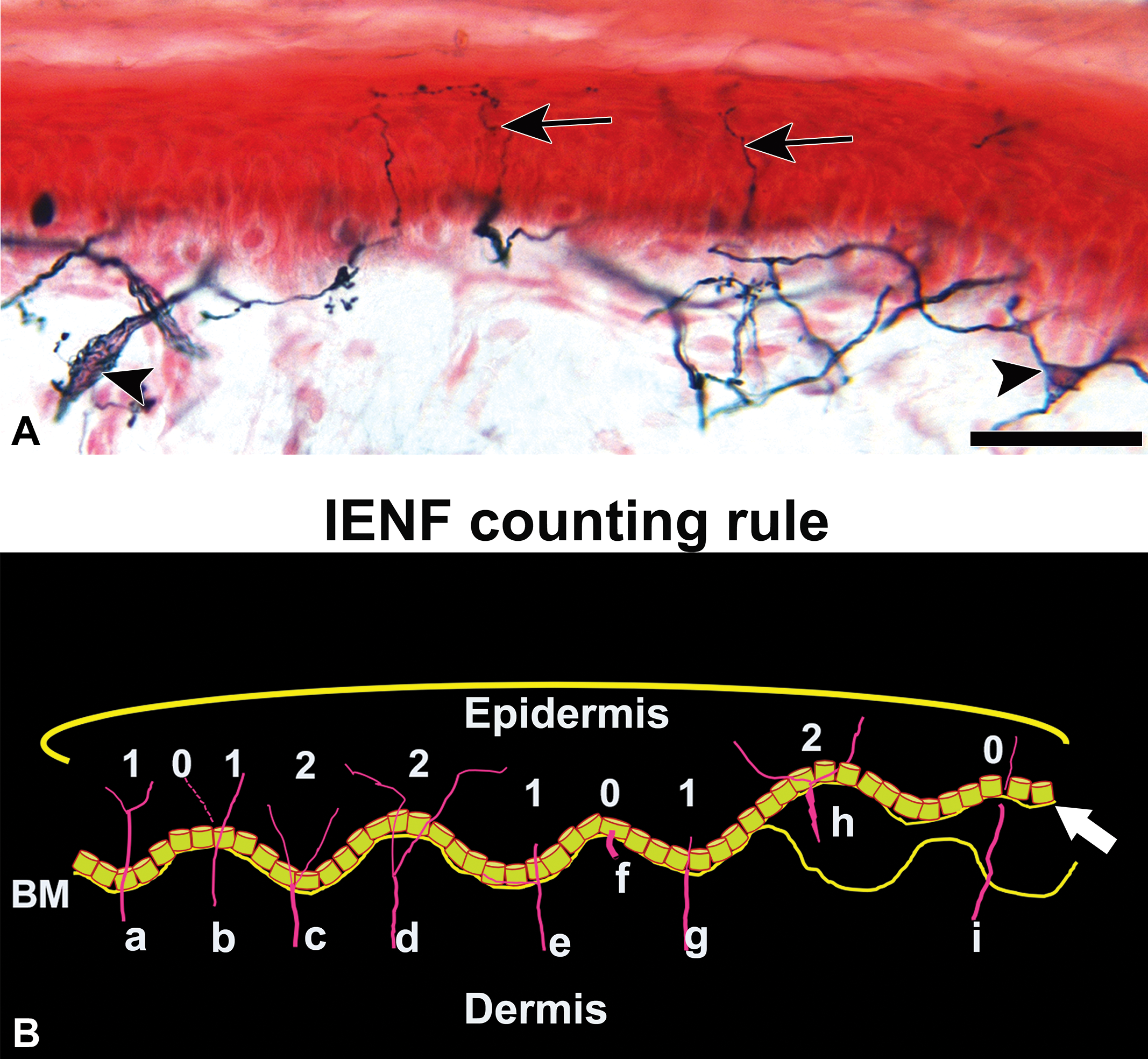
A, Skin biopsy from the distal leg of a healthy human. Dermal axon bundles are ensheathed by Remak Schwann cells (Schwann cell nucleus, arrowheads) and terminate as free epidermal nerve fibers (arrows). Tissue was fixed in Zamboni’s fixative for 12 hours at 4°C, sectioned at thickness of 50 μm using a freezing sliding microtome, and counterstained with nuclear fast red. Scale bar: 50 m. B, Line diagram depicting the epidermal nerve fiber counting rules that are widely used in human studies. (a, b) Count a nerve fiber as it crosses the basement membrane (BM) of the epidermis. (c) Nerves that branch at the BM and enter the stratum spinosum are counted as separate units. (d) Nerves that split below the BM are counted as separate units. (e, g) Nerve fragments that cross the BM and pass vertically a single layer of the basal keratinocytes are counted. (f) Nerve fibers that approach the BM but do not cross the basal keratinocyte are not counted. (h) When the BM shifts at different planes (white arrow) only the epidermal axons that have continuous connection with the dermal axons are counted. (i) Epidermal axons that have no continuous connection with the dermal axons are not counted. Detailed protocols are available in laboratories that train laboratory personnel for IENF counting. IENF indicates intraepidermal nerve fiber.
Skin Biopsy as a Diagnostic Tool for Small Fiber Sensory Neuropathies
Conditions that selectively or predominantly affect Aδ or C fibers are known as small fiber sensory neuropathies (SFSNs). The underlying causes of SFSN in human patients are diverse and include a number of common metabolic conditions (eg, diabetes mellitus, hypothyroidism, hyperlipidemia), infectious diseases (eg, HIV, hepatitis C), neurotoxic agents (eg, alcohol, antiretroviral medications, chemotherapeutics), and immune-mediated conditions (eg, celiac disease, sarcoidosis, Sjögren syndrome) as well as several rare hereditary conditions. 22,23 For many patients with SFSN—up to 30% in some reports—the condition is classified as idiopathic when no underlying cause is identified. 24,25 Although the possible clinical manifestations of SFSN are also heterogeneous, patients most commonly present with symmetric, length-dependent symptoms that are most severe in the distal leg and described as burning, stinging, and electric shock.
Clinical diagnosis of SFSN can be challenging. Patients with pure SFSN (ie, without concurrent involvement of motor or larger sensory nerve fibers) appear relatively normal on routine neurologic examination, with preserved ankle reflexes, vibratory thresholds at the toe, and toe proprioception. Distal pinprick sensation and temperature sensation may be reduced. 26,27 Other conventional techniques used to diagnose PN, including electromyography, nerve conduction studies, and sural nerve biopsy, may be useful for excluding damage to motor and large, myelinated sensory nerves but fail to capture the morphology and function of small sensory nerve fibers. Quantitative sensory testing (QST) is a widely used, noninvasive method that quantifies thermal and pain threshold functions of small nerve fibers by applying increasing intensities of vibration, cold, warm, or heat pain stimuli to the skin of the index finger or great toe. Quantitative sensory testing cannot, however, differentiate between peripheral and central causes of sensory deficits and is subject to bias as it relies on patient responses; it is, therefore, best used in combination with other modalities. 28,29
Over the past 2 decades, immunostaining and quantification of IENF in skin punch biopsies has emerged as a reliable tool for diagnosing and studying SFSN in human patients. Compared to sural nerve biopsies, which are invasive and carry the risk of a long-lasting distal sensory deficits and pain, 30,31 skin punches are minimally invasive and permit the skin to be sampled at several anatomical sites simultaneously and serially over time. 26,32 –43 In a large study comparing the results of distal leg skin biopsies and sural nerve biopsies, distal IENF density and sural nerve small fiber density were concordant in 73% of patients. 44 Furthermore, reduced IENF density was shown to be the only indicator of SFSN in 23% of cases. In patients with diabetic neuropathy (DN), IENF density was found to inversely correlate with results of QST, the strongest correlation being with warm threshold. 42,45 –48 When patients with SFSN present with painful feet, skin biopsies have shown a diagnostic efficiency of 88.4% compared to clinical examination (54.6%) and QST (46.9%). 49 Among neuropathy patients with positive sensory symptoms, there is good concordance between loss of pinprick sensitivity and abnormal IENF density in the foot. 50 Few studies did not find a significant correlation between QST results and IENF density. 26,36,51 Although this end point has rarely been used in the regulatory settings in the past, a recent submission to the Office of Biomarker Qualification within the Center for Drug Evaluation and Research at the Food and Drug Administration includes a letter of interest to qualify epidermal neurite density as a diagnostic biomarker. 52 Along with other clinical indicators, the intention would be to confirm a diagnosis of SFSN in drug development studies.
Methods for IENF Analysis in Human Patients
Sample Collection
Skin biopsies for IENF analysis in human patients are most commonly obtained using local anesthesia (1% lidocaine HCl and epinephrine 1:1000 injected intradermally) and a 3-mm circular punch biopsy tool. A step-by-step video of this procedure is available online through the Johns Hopkins Cutaneous Nerve Laboratory. 53 Generally, 3 skin punches are taken along the lower limb, namely, proximal thigh, distal thigh, and distal leg. Hair follicles are avoided if possible since IENF are denser near follicles. These skin punches are easy to perform, cause minimum skin injury, and no sutures are required. It is critical to avoid crushing or pinching the biopsy tissue to avoid artifacts that may resemble focal IENF loss. A second method, known as the skin blister technique, makes use of a suction capsule that separates the epidermis from the dermis at the level of the basement membrane. 54 Although this technique is both painless and bloodless, the procedure requires 30 to 90 minutes for blister formation, and the relationship of dermal nerves to the epidermal nerves cannot be studied. However, good correlations have been reported between both sampling methods with regard to IENF density. 55
Fixation and Sectioning
Skin samples are immediately placed into fixative solution for 12 to 18 hours at 4°C. Zamboni (2% paraformaldehyde, picric acid), Lana (4% formaldehyde, picric acid), and PLP (paraformaldehyde, lysine, periodate) fixatives all preserve antigenic integrity and are routinely used. Standard 10% neutral-buffered formalin is suboptimal because it results in the epidermal nerves having a more fragmented appearance after staining. Following fixation, the biopsy tissue is transferred to a cryoprotectant solution composed of 20% glycerol in phosphate-buffered saline where it can remain for up to 1 month at 4°C or frozen at 20°C for longer periods.
To perform the standardized IENF counting technique described below, nerve fibers must be visible as continuous axons crossing the dermal–epidermal junction. Because of the wavy, 3-dimensional structure of the nerve fibers, this is best visualized in thick (>30 µm) sections. Conventional thin-section preparations primarily contain isolated nerve fiber fragments rather than full profiles. Although thick sections can be obtained from paraffin or soft plastic resin-embedded skin samples, frozen sections are generally preferred for this technique as they minimize tissue shrinkage and facilitate even antibody penetration through the entire depth of the tissue. 18,56,57 To produce frozen sections using a freezing sliding microtome, a drop of 30% sucrose first is applied to the microtome platform. The cryoprotected skin punch is appropriately oriented on the drop and covered with crushed dry ice to freeze prior to sectioning. Sections are then cut perpendicular to the epidermis at a thickness of 50 µm. A 3-mm punch yields approximately 45 to 50 sections per sample and of these, 3 to 4 sections are selected from roughly fixed intervals throughout the tissue (eg, 10th, 20th, and 30th section) in order to be representative of the entire biopsy and to avoid sampling bias.
Immunostaining for the Pan-Axonal Marker PGP9.5
Ubiquitin carboxyl-terminal esterase L1, also known as protein gene product 9.5 (PGP9.5), is a soluble enzyme expressed exclusively in the cytoplasm of neurons and is the most commonly targeted antigen for immunostaining all subtypes of IENF in skin biopsies. 58 The free-floating technique and use of a detergent are critical to achieve antibody penetration into the thick sections. All steps are performed in 96-well plates at room temperature. Although the process can be carried out by moving the sections from well to well using thin wire loops, the protocol is greatly facilitated by the use of filter-bottom plates and a vacuum manifold to remove solutions at each step. The next solution in the protocol can then simply be added to the wells using a multichannel pipette and the sections themselves do not need to be moved.
The sections are first bleached in 0.25% potassium permanganate for 5 minutes to remove melanin pigment, rinsed in 1X Tris-buffered saline (TBS; pH 7.4), and washed with 5% oxalic acid for 2 minutes. Nonspecific antibody binding is blocked by incubating for a minimum of 15 minutes with 4% normal goat serum, 1.0% Triton X-100, and 0.5% nonfat powdered milk in TBS. The sections are incubated overnight at 4°C with the primary anti-PGP9.5 antibody suspended in 0.5% Triton X-100 and 2% normal serum in TBS. The Johns Hopkins Cutaneous Nerve Laboratory currently uses mouse–antihuman PGP9.5 (Clone BH7, product # MCA 4750GA, Bio Rad, Kidlington, United Kingdom) at a dilution of 1:70 000. After washing in TBS, sections are incubated with a biotinylated secondary antibody directed against the Fc region of the primary antibody species (eg, goat anti-mouse) for 1 hour. Endogenous peroxidase is blocked for 1 hour with 0.5% H2O2 in methanol, and sections are then incubated with Avidin-Biotin Complex solution (Vector, Burlingame, California) for 1 hour followed by development using the SG peroxidase substrate kit (Vector). This kit produces dark blue-gray chromogenic staining that is preferable to 3,3′-diaminobenzidine since brown staining can be difficult to distinguish from residual melanin pigment.
The stained sections are mounted on chrome alum-gelatin subbed slides, air-dried for at least 30 minutes, counterstained with nuclear fast red (Vector) or dilute eosin (1%), and cover-slipped with Permount (Fisher Scientific, Pittsburgh, Pennsylvania). Final mounted epidermal thickness for sections prepared in this manner ranges from approximately 35 to 40 µm.
Intraepidermal Nerve Fiber Quantification
The manual method used to enumerate IENF in humans involves 2 distinct steps: counting the number of IENFs under light microscope at ×40 magnification and measurement of the length of the epidermis along the stratum corneum by using a computerized software or stereology program. 59 The linear IENF density is calculated and expressed as the number of fibers per millimeter of epidermal length IENF/mm. Two different counting rules have evolved over the past decade: (1) counting only the number of nerve fibers crossing the epidermal basement membrane and (2) counting that includes isolated nerve fragments in epidermis that do not cross the basement membrane. The European Federation of Neurological Studies task force has established guidelines and rules for counting and reporting IENF, 18,60 –62 and many laboratories follow the rule of counting only the number of single axons crossing the epidermal basement membrane (Figure 1B).
Crushing skin biopsies with forceps, improper fixation and storage of skin sections, and inconsistent immunostaining techniques can yield variations in the staining of nerve fibers and lead to inaccurate IENF quantification. Strict quality control measures (eg, inclusion of control slides in each immunostaining run) and training of personnel to recognize signs of suboptimal staining, such as granular or patchy chromogen deposition, will help to minimize technical variation. Furthermore, a high degree of inter- and intrarater reliability can be achieved in IENF studies by implementing standardized counting rules and periodic quality control.
Normative Data and Variability in Human IENF Analysis
In healthy human patients, IENF density typically exhibits a normal, proximal-to-distal gradient with higher IENF density values in the upper thigh compared to the distal leg. Several groups have established normative series and a worldwide reference range has been published. 63 –65 Variation in IENF density among adjacent sections within the same site are minimal, and a high degree of association of IENF densities between 2 punches at the same anatomical site has been demonstrated. 2,66 Similarly, there is no significant IENF density variation between both distal legs and the diagnosis of SFSN can be reliably made from a single, unilateral skin biopsy at the distal leg. 67
The effect of gender, age, and height on distal leg innervation has been investigated by several investigators. In the first normative IENF study performed at the Johns Hopkins University, there was no significant effect of increasing age among patients aged 13 to 82 years, other than that of patients in the youngest decile having higher densities. 66 A recent study confirmed that epidermal innervation is denser in the pediatric population compared to healthy adults. 55 Other studies of healthy control patients have reported a mild inverse, gender-adjusted relationship between IENF density and age with a decrease in 0.6 to 1.8 fibers/mm/decade. Similarly, there has been variation in the role that gender plays with some studies reporting no effect while others finding mild increases in epidermal innervation among females. 42,66,68 –71 Comparative studies across different nationalities are necessary as a short study between Thai and US population has revealed that race, height, and weight affect IENF density. 72
Intraepidermal Nerve Fiber Analysis in Primate Models of PN
Among primate models, IENF analysis has primarily been used in SIV-infected macaque models of HIV-associated sensory neuropathy. Pigtailed macaques (Macaca nemestrina) inoculated with both an immunosuppressive SIV strain and a neurovirulent SIV clone develop striking IENF loss in the distal limbs that begins early in the course of infection (14 days postinoculation) and is associated with evidence of macrophage activation in the lumbar dorsal root ganglia. 3,73,74 Similar findings have also been reported in CD8+ T-cell–depleted rhesus macaques (Macaca mulatta) infected with SIVmac251. 7,8 In the dual SIV-inoculated pigtailed macaques, significant IENF loss was detectable earlier than decreased conduction velocity in the sural nerve, showing it to be a more sensitive indicator of small nerve fiber damage in this model. 73
Methodology
In macaques, 3-mm skin punch biopsies for IENF analysis are collected under general anesthesia or at necropsy from either glabrous skin of the metatarsal footpads or haired skin immediately distal to the lateral malleolus. 8,9 Using footpad skin avoids the dense follicles and adnexa in the haired skin of macaques, which can make microtomy more difficult and complicate downstream visualization and quantification of IENF. Once the punch has been made, it is critical to handle the sample using fine forceps to grasp only the deep dermal fat and avoiding the epidermis. As in humans, the biopsies are well tolerated by macaques and typically do not require post-op systemic antibiotics or analgesics. The protocols for tissue processing, sectioning, and immunostaining IENF in macaque skin samples are essentially identical to those described for human patients. 3,8,9 Trimming of the thick stratum corneum layer from the macaque footpad sections is an additional step that can help reduce folding of the final tissue sections during drying. This is performed after the sections have been mounted on slides (prior to air-drying and counterstaining) using a dissecting microscope and a razor blade.
Because of the extremely rich epidermal innervation in the macaque footpad, computed image analysis techniques for quantifying IENF are more practical and reproducible than manual counting in macaque studies. The most current method employs design-based stereology. Although stereology is somewhat time-consuming and requires specific software and equipment (eg, a motorized microscope stage), it is an unbiased approach for quantifying complex biologic structures in tissue sections, 75 and once a workflow is established, it can be carried out with relatively little specialized training.
The publications reporting stereological IENF data in macaques have all used Stereo Investigator software (MBF Biosciences, Williston, Vermont), 3,8,9 but other stereology programs and algorithms for estimating object length could also be used for this purpose. The general workflow for performing this analysis using Stereo Investigator is illustrated in Figure 2. Briefly, the user traces a region of interest (ROI) encompassing epidermis under low-power magnification. The software then places a grid of rectangular counting frames over the ROI and, after switching to high-power magnification (×60 oil), controls a motorized stage to move slide to random sampling sites, an approach known as systematic uniform random sampling. At each site (typically around 20 sites per section), the user scrolls through the z-axis of the tissue and marks intersections between the virtual hemispheric probe and PGP9.5-stained IENF. Results can be reported as total IENF length (in µm or mm) or a density value can be calculated by dividing the total IENF length by the estimated volume of the sampled epidermal tissue (mm/mm3). Alternative stereologic approaches for estimating object length, such as cycloids and isotropic virtual planes, could also be implemented depending on the capabilities of available software. 76 –79
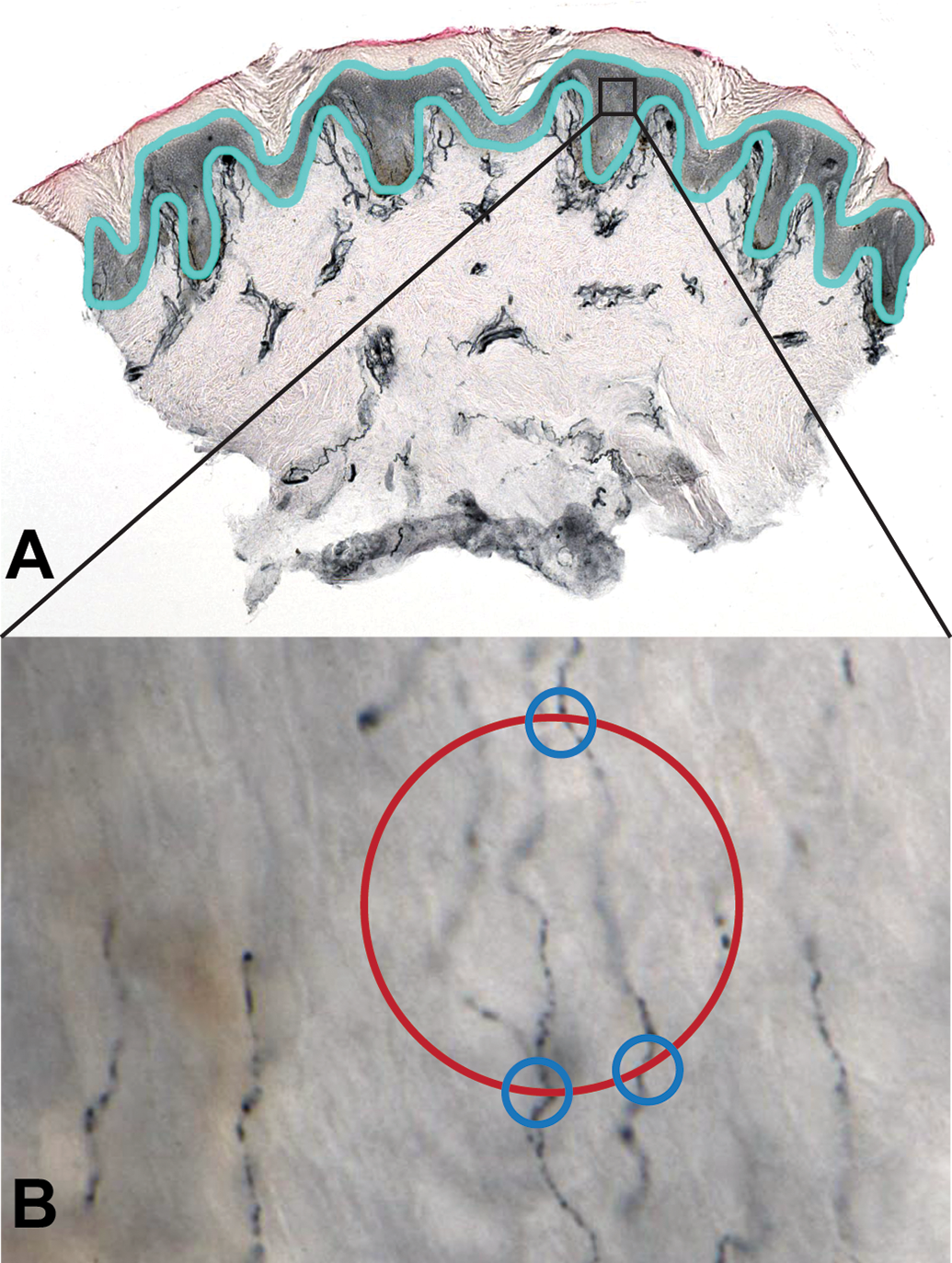
Illustration of the stereologic technique for measuring IENF length in a footpad skin biopsy. As depicted in (A), the epidermis is traced (light blue outline) in a PGP9.5-stained skin section under low magnification. This serves as the ROI. The Stereo Investigator program (or a comparable stereology software) then systematically selects a random subset of fields within the ROI for measuring IENF (represented by the black rectangle). After switching to high magnification, as shown in B (×60 original oil objective), the probe is virtually moved through the z-axis of the tissue by a motorized stage and points of intersection between IENF and a hemispheric probe (represented by red circle) are marked by the user (blue circles). Intersections are only counted when the nerve fiber is in sharp focus at the point of crossing. Tissue was fixed in Zamboni’s fixative for 12 hours at 4°C, sectioned at thickness of 50 μm using a freezing-sliding microtome, and counterstained with 1% eosin. Material is reproduced by permission of Sage Publications. 9 IENF indicates intraepidermal nerve fiber; ROI, region of interest.
Normative Data and Variability
At the time of writing, there have been no large-scale studies investigating normal age-, sex-, or species-related IENF variation in nonhuman primates. However, some important considerations for study design can be gleaned from SIV studies. For instance, the fact that SIV-infected rhesus macaques develop significant IENF decline when they have undergone CD8+ lymphocyte depletion, but not when dual-inoculated with immunosuppressive and neurotropic SIV strains highlights the importance of species-specific host immune responses in determining susceptibility to SIV-induced nerve damage. 7,9 When evaluating preinfection IENF data among a relatively large cohort of male pigtailed macaques (n = 50), animals that originated from Indonesia were found to have significantly lower IENF values than those born in the United States, suggesting that uniformity in animal origin may be critical for cross-sectional studies. 9 That same study found no significant difference in footpad IENF density when comparing a control group of male pigtailed macaques to healthy female rhesus macaques; however, group sizes were small and there was broader variation within the female group. Given the potential for unexpected variability, use of longitudinal, “change-from-baseline” studies, where skin biopsy is performed before, after, and, possibly, during treatment/infection, can be very helpful in accounting for baseline variation. 9
Intraepidermal Nerve Fiber Analysis in Rodent Models of PN
Intraepidermal nerve fiber quantification has been increasingly used as a primary morphologic outcome measure in rodent models of PN, often reported alongside behavioral and electrophysiologic data. In models of DN, significant declines in footpad IENF density have been observed in a number of mouse and rat models of both type 1 and type 2 diabetes mellitus. 4,10,80 –87 Interestingly, rodent models of obesity and prediabetes develop behavioral changes consistent with sensory neuropathy without concurrent IENF loss, indicating that functional alterations in cutaneous nerve fibers likely precede detectable morphologic changes. 88 –91 Intraepidermal nerve fiber analysis has also frequently been incorporated in rodent studies of chemotherapy-induced peripheral neuropathy (CIPN). As in patients with cancer, neuropathic signs and IENF loss have been documented in mice and rats treated with microtubule-directed compounds, such as paclitaxel, oxaliplatin, and vincristine. 92,93 Rodent CIPN studies that evaluated PNS pathology at multiple posttreatment time points have demonstrated significant declines in IENF density prior to the onset of pathologic changes in more proximal nerves (dermal nerve bundles, sciatic, saphenous) and sensory ganglia. 12,94,95 Other applications of IENF analysis in rodents include models of HIV and antiretroviral-associated neuropathy, ischemia, acrylamide neurotoxicity, mercury poisoning, and neuropathic pain following nerve injury. 6,11,96 –103
Methodology
There are currently no broadly standardized methods for IENF analysis in mice or rats—the technical details for each step in the process, from sample collection through nerve fiber staining and quantification, vary widely across the rodent literature. The vast majority of rodent IENF studies have examined glabrous skin from the hind footpads, although the exact anatomic location (eg, digital vs metatarsal pads), number, and size of the samples are rarely specified. In some studies, hind limbs are transected at the ankle following euthanasia and fixed in Zamboni’s or paraformaldehyde fixative en bloc prior to dissection of footpads or collection of 2- to 3-mm skin punches. 12,83,104 This method likely helps minimize crush artifact in the epidermis but may result in suboptimal fixation. Rodent skin samples have been sectioned using different types of microtomes at thicknesses ranging from 5 to 50 µm, and PGP9.5-labeled fibers have been visualized using an array of chromogenic and fluorescent markers. Notably, multiple groups working with rodent models have successfully used frozen sectioning and immunostaining protocols very similar to those described for human patients and primate models, 6,83,94,104,105 showing that some degree of technical standardization across species is attainable.
Although IENF density in rodent hind paws is high compared to human samples, 106,107 groups working with mice and rats have typically been able to employ manual counting methods generally similar to those described for humans—individual IENF are enumerated and the total is divided by the length or volume of epidermis sampled. However, the exact microscopic techniques and quantification approaches vary among studies. As with sectioning and immunostaining, many groups have applied the standardized counting rules used in human clinical practice to rodent models. 6,83,85,94,104,105 When such standardized protocols were followed in a study of healthy and neuropathic rats, a high interobserver agreement was obtained and IENF density significantly correlated with neurophysiologic changes. 85
Normative Data and Variability
Published normative data for rodents are scanty. 85 Inconsistencies in skin sampling, immunostaining techniques, and IENF counting methods across studies, as described above, have resulted in a wide range of IENF densities reported in healthy control animals. 10,94,103,108 –110 Until a time when standardized techniques are more widely adopted across different laboratories and large-scale normative data sets become available for mice and rats, inclusion of appropriate control groups will remain a requirement for each cross-sectional study. A small number of research groups have performed longitudinal analysis of cutaneous nerves in rodents, by collecting either 1 mm diameter biopsies from the plantar skin (in mice) or 3 mm biopsies from the dorsal surface of the hind paws (in rats) at sequential time points. 111,112
Sex and strain differences, both in baseline IENF density and susceptibility to IENF loss under experimental conditions, are other essential considerations for animal study design; however, reports that specifically address these factors in rodent models of PN are currently limited. A recent study investigating sex-based differences in the leptin-deficient (BTBR ob/ob) mouse model of type 2 diabetes found that male mice displayed a more extreme neuropathic phenotype with faster onset compared to females. 113 Although female mice did develop significant nerve conduction velocity deficits and IENF loss at 26 weeks of age (compared to 22 weeks in males), their IENF density was greater than that of males at study termination. Sex-based differences in susceptibly to neuropathy have also been observed in a genetically engineered mouse model of type 1 diabetes (Ins2+/Akita mice) 81 and a rat model of type 2 diabetes. 114 Background strain also strongly influences the manifestation of neuropathy in diabetic mouse models. 80 A study examining models of type 1 and type 2 diabetes found that background strain was a more important factor than hyperglycemia in the development of DN, with the commonly used C57BL/6J strain being relatively resistant to the development neuropathic signs compared to C57BKLS. 115 In a strain survey of paclitaxel-induced neuropathy, C57BL/6J mice were also found to be resistant to development neuropathic signs, whereas DBA/2 mice were highly susceptible; however, this study was based solely on behavioral responses and did not examine IENF density specifically. 116
Intraepidermal Nerve Fiber Analysis in Other Laboratory Species
Nine banded armadillos, which are used as a model of leprosy, possess dense epidermal innervation similar to that of humans (Figure 3); quantitative IENF analysis was recently reported as an outcome measure in an investigational Mycobacterium leprae vaccine study in the armadillo model. 117 Pigs have been proposed as potential models of sensory neuropathy since their body size, skin structure, and metabolism are similar those of humans. A recent article investigating proximal sciatic nerve irritation in as a porcine model of chronic neuropathic pain found significant reduction in fluorescently labeled IENF between 7 and 14 days postprocedure, whereas loss of dermal innervation was more gradual. 118 Marked reduction in distal IENF density has also been reported in an feline immunodeficiency virus infection model of HIV-associated neuropathy. 5
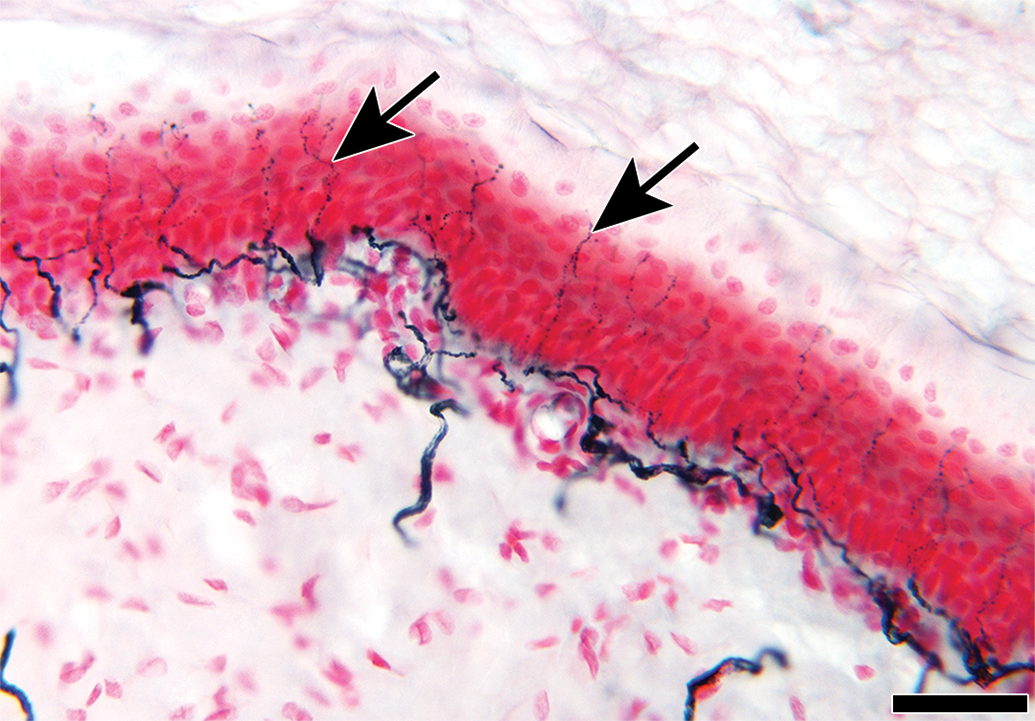
Dense cutaneous nerve bundles terminating as numerous epidermal nerve fibers (arrows) in a skin section from the abdomen of a nine-banded armadillo. Tissue was fixed in Zamboni’s fixative for 12 hours at 4°C, sectioned at thickness of 50 μm using a freezing sliding microtome, and counterstained with nuclear fast red. Scale bar, 10 μm.
Applications of Skin Biopsies Beyond IENF Quantification
The basic skin biopsy technique described for IENF quantification in this review has been adapted to study many other components of cutaneous innervation, including dermal nerves, Schwann cells, autonomic (sweat gland and pilomotor) nerve fibers, and microvasculature. 83,119 Confocal microscopy, although labor-intensive, offers the advantage of visualizing different types of nerve fibers and their associated structures simultaneously using multiple fluorochromes (Figure 4). 120,121 Stacks of images acquired at progressive depths through the skin section can be compressed into a single, high-resolution image, allowing 3-dimensional morphology and spatial relationships among structures to be viewed in 2 dimensions. Skin biopsies can also serve as the starting material for ultrastructural examination and morphometry, 122 gene expression analyses, 123 and laser capture microdissection studies. 124
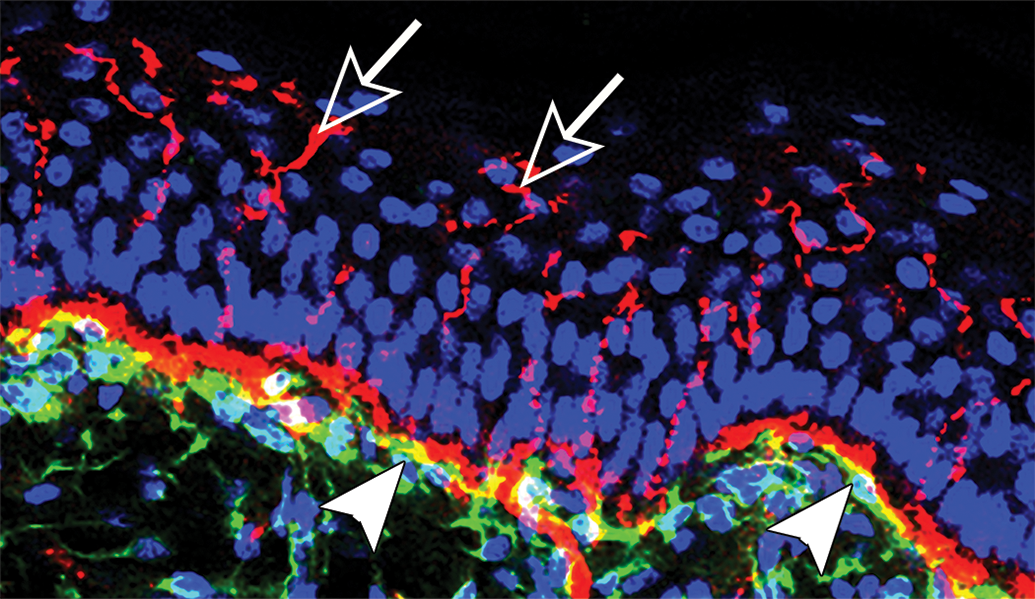
Confocal image from the skin of a macaque showing dense innervation of PGP 9.5 positive epidermal fibers (red, white open arrows). At the papillary dermis, bands of Schwann cells (p75, antinerve growth receptor, green, white arrowheads) enclose and colocalize with the dermal nerve bundles (solid red band beneath the epidermal basal lamina).
Intraepidermal Nerve Fiber Analysis in Studies of Neuroregeneration
Intraepidermal nerve fiber analysis has shown great promise as a tool for assessing neuroregeneration in humans and animal models. A technique known as cutaneous axotomy has been used to investigate the regenerative capacity of IENF in healthy human patients and patients with diabetes and HIV-associated neuropathy. 25,125 –127 In this method, cutaneous nerve fibers are transected by performing an initial 3 mm punch biopsy and either leaving the core of tissue in place (incisional model) or removing it (excisional model). The initial site is then allowed to heal prior to being collected at a later date using a 4- or 5-mm punch biopsy (Figure 5). These samples are then processed and immunostained as thick, frozen sections so that regrowth of IENF (as well as dermal nerve fibers and blood vessels) into the initial 3-mm biopsy site can be visualized and measured by stereology. 119,128 This technique has been employed in SIV studies of pigtailed macaques wherein all animals (uninfected and infected) showed relatively rapid and complete reinnervation of the axotomy site compared to humans. In SIV-infected animals, IENF regeneration was significantly slower compared to control animals and nerve fibers exhibited altered length and morphology. 119,128 In rodents, footpad IENF analysis is commonly used as a means to assess nerve regeneration and reinnervation patterns following experimental sciatic nerve injury. 98,102,109,129
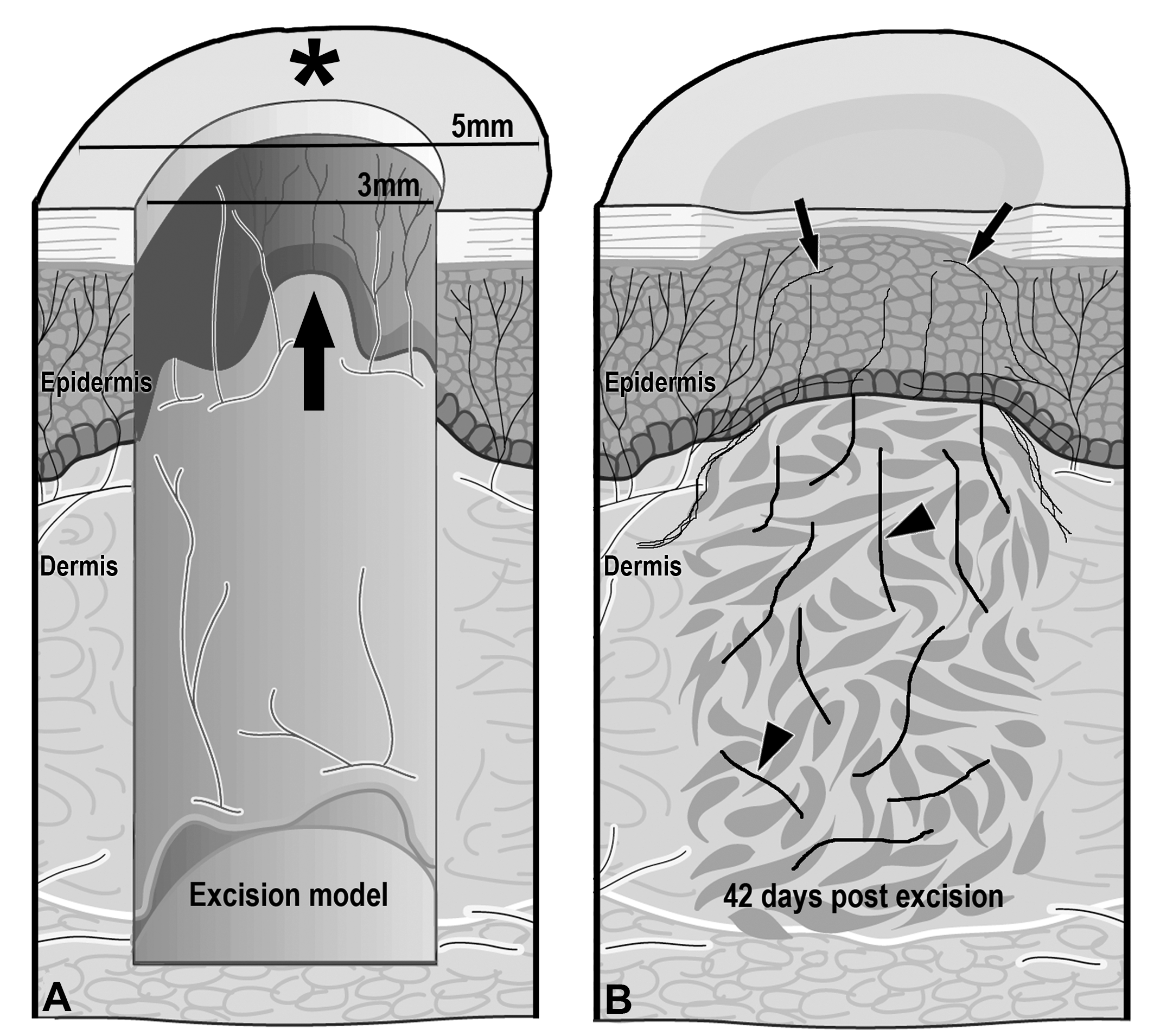
An intracutaneous excisional axotomy model in macaques. Circular cutaneous axotomies were performed at 2-week intervals on the dorsal interscapular skin using a 3-mm skin punch to transect epidermal axons (A). The central core containing epidermis and dermis was then removed (arrow) yielding an excisional axotomy. On the 70th day after the initial axotomy, a 5-mm circular biopsy punch (*) was used to harvest all of the previous 3-mm punch sites, providing samples containing axotomy sites that were 14, 28, 42, 56, and 70 days postaxotomy (A). By 42 days postaxotomy, collateral sprouts (arrows) and regenerating axons (arrowheads) had reinnervated the epidermis (B). Reprinted, by permission of John Wiley and Sons. 119
Summary and Conclusions
When working with any animal model, the likelihood of obtaining clinically relevant data is enhanced by the use of outcomes that are analogous, if not identical, to those measured in human patients. As described in this review, measurement of IENF density in skin biopsy material has proven to be an invaluable clinical tool for diagnosing and monitoring SFSNs in human patients. The technique has also been successfully adapted for use in animal models of PN where it is often found to be a more sensitive indicator of sensory nerve fiber damage than evaluation of more proximal nerve components, particularly at early time points. The methods used to achieve IENF quantification are somewhat specialized compared to conventional nerve histology, requiring the use of particular fixatives, thick frozen sections, and immunostaining of nerve fibers; however, the collection of skin samples requires very little expertise or extra time on the part of prosectors and a majority of the reagents and equipment are standard in any reference histology laboratory. Enumeration of IENF by manual counting or stereology entails a considerable amount of training and practice at the outset, but high rates of inter- and intraobserver agreement can be achieved. A review of the literature supports the premise that greater protocol standardization for IENF analysis is possible in animal models and would allow for comparisons to be made across studies as well as the creation of large-scale normative data sets for different species and strains. The basic skin biopsy technique can also serve as a platform for studying reinnervation, a feature that is especially vital in the development of new therapeutic strategies aimed at reversing nerve damage in PN and nerve injury.
Footnotes
Authors’ Note
All views reflect the views of the author (DR) and should not be construed as representing views or policies of her employer, the US Food and Drug Administration.
Acknowledgments
The authors would like to thank Michael Polydefkis and Joseph Mankowski for their professional guidance as well as Baohan Pan, Kelly Wagner, and Megan McCarron for their expert technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
