Abstract
Beagle dogs are a key nonrodent species in nonclinical safety evaluation of new biomedical products. The Society of Toxicologic Pathology (STP) has published “best practices” recommendations for nervous system sampling in nonrodents during general toxicity studies (Toxicol Pathol 41[7]: 1028-1048, 2013), but their adaptation to the Beagle dog has not been defined specifically. Here we provide 2 trimming schemes suitable for evaluating the unique neuroanatomic features of the dog brain in nonclinical toxicity studies. The first scheme is intended for general toxicity studies (Tier 1) to screen test articles with unknown or no anticipated neurotoxic potential; this plan using at least 7 coronal hemisections matches the STP “best practices” recommendations. The second trimming scheme for neurotoxicity studies (Tier 2) uses up to 14 coronal levels to investigate test articles where the brain is a suspected or known target organ. Collection of spinal cord, ganglia (somatic and autonomic), and nerves for dogs during nonclinical studies should follow published STP “best practices” recommendations for sampling the central (Toxicol Pathol 41[7]: 1028-1048, 2013) and peripheral (Toxicol Pathol 46[4]: 372-402, 2018) nervous systems. This technical guide also demonstrates the locations and approaches to collecting uncommonly sampled peripheral nervous system sites.
Keywords
Introduction
Beagle dogs (Canis lupus familiaris) are the most common nonrodent species used in Europe and North America for nonclinical safety assessment. The primary advantages of Beagle dogs as nonrodent models for biomedical research are the relatively greater anatomic and physiologic equivalency to humans in comparison to rodents and rabbits; their greater availability and reduced cost compared to nonhuman primates (NHPs); and their smaller size compared to adult minipigs, which reduces the amounts of food, space, and test article needed to perform a study in dogs.
Evaluation of the central (CNS) and peripheral (PNS) nervous systems in all test species is required for routine toxicity testing in current guidance by regulatory agencies1,2 and international consortia.3,4 Based on this guidance, the Society of Toxicologic Pathology (STP) has produced “best practices” recommendations that cover sampling, trimming, and processing of CNS 5 and PNS 6 organs for rodents (based on Rao et al7,8) and nonrodents (based on Pardo et al 9 ) during general toxicity studies (Tier 1, performed to screen agents with unknown or no anticipated neurotoxic potential) as well as PNS sampling for neurotoxicity studies 6 (Tier 2, performed to better characterize test articles with suspected or known neurotoxic activity). Distinct neuroanatomic features of nonrodent species have required that these generic recommendations be adjusted to some degree for brain sampling among species. Detailed nervous system sampling protocols focusing on effective screening of principal brain domains during nonclinical toxicity studies have been prepared for major nonrodent species including rabbits, 10 minipigs, 11 and NHPs. 9 A prior sampling protocol for large-sized animal brains 12 employed dog specimens to illustrate a suitable trimming plan. However, the approach in this prior paper was published before the STP “best practices,” focused on the brain sampling, and did not include spinal cord or PNS sampling.
The current project was designed to fulfill 3 objectives. The primary goal was to provide detailed trimming protocols for the Beagle dog CNS and PNS that conform to the STP “best practices” recommendations for general toxicity studies and neurotoxicity studies. A second objective was to offer well-annotated images of hematoxylin and eosin (H&E)-stained dog brain sections produced using these trimming protocols to facilitate lesion localization by study pathologists. The final objective was to offer a detailed technical guide for identifying and sampling common ganglia and nerves (somatic and autonomic) in the dog.
Materials and Methods
Animals
All procedures were approved in advance by the Institutional Animal Care and Use Committee in keeping with U.S. federal regulations 13 and the Guide for the Care and Use of Laboratory Animals, 8th edition. 14 In accordance with “replace, reduce, refine” (3Rs) principles for animal research, preparation of this technical guide was performed as a satellite project using extra tissue from animals used for other investigations preapproved by the Pfizer, Inc. Institutional Animal Care and Use Committee (IACUC).
Naive, young adult, male and female Beagle dogs were obtained from Marshall BioResources (North Rose, NY, USA). Animals were housed individually in indoor 1.8 × 1.2 m runs. All housing units were cleaned daily. Dogs were given Certified Purina Diet 5L66 (PMI Feeds, Inc.) once daily and municipal tap water (treated by reverse osmosis) ad libitum. Animals were maintained in biologically clean rooms with HEPA (high-efficiency particulate air)–filtered air and a 12-hour light/dark cycle. Room temperature and relative humidity were maintained at 22°C ± 3°C and approximately 30% to 70%, respectively. Bedding and food were autoclaved before use, and enrichment devices were regularly provided.
Tissue Acquisition and Processing
Dogs were sedated with dexmedetomidine (Dexdomitor®, 0.5 mg/mL) via intramuscular (IM, ~1 mL) or intravenous (IV, ~0.75 mL) injection and then euthanized humanely using sodium pentobarbital IV at a target dose of 50 mg/kg to induce deep anesthesia, followed by exsanguination. Brains were exposed by removing the dorsal calvarium and reflecting the dura mater and falx cerebri. Brain attachments (cranial nerves [CNs] and spinal cord) were severed, and the brain was removed and fixed while intact by immersion for at least 48 to 72 hours at room temperature in at least 10 times greater volume of neutral buffered 10% formalin (NBF, pH 7.4) containing approximately 1% methanol as a stabilizing agent.
Coronal trimming planes were positioned using prominent landmarks on the lateral (Figure 1A), ventral (Figure 1B), and/or dorsal (Figure 1C) surfaces of the brain. The fixed brain was positioned in a proprietary, purpose-made matrix tool (developed in-house at Pfizer for obtaining reproducible trimming planes for brain sampling in nonrodent species10,11) with the following dimensions: 15-cm long, 8-cm wide, with a 6-cm wide central channel bordered by 2 outer raised edges that are 1-cm wide and 3.2 mm in height (Figure 2). Using the rostral margin of the pons as a landmark (Figure 1B), an initial cut at a rostrally slanted angle of 10° to 20° relative to the ventral surface of the brain stem was made with a Tissue Slicer Blade (Cat. No. 6727C18; Thomas Scientific, Swedesboro, NJ) to divide the fixed brain in half. Using this matrix, each brain was divided into multiple full-coronal, 3.2-mm thick slices having 2 flat surfaces as well as unleveled rostral and caudal ends that are not trimmed further (Figure 3A and B). For animals in routine toxicity studies, 17 or 18 coronal sections may be obtained from younger animals, while up to 23 coronal sections as depicted in this paper may be made for older (and larger) males; for a given study, the trimming planes should be approximately comparable for age- and sex-matched animals in concurrent control and treated groups. For most full-coronal slices, one half (i.e., a coronal hemisection) was placed in its own tissue cassette(s); the cerebellum was placed intact (i.e., full-coronal sections) in tissue cassettes. For very large brains, a coronal hemisection might be divided and the dorsal and ventral halves placed in 2 cassettes (Figure 3A and B). Tissues were processed routinely into paraffin with the rostral face of each slice placed “down” in the embedding mold (so that the rostral face was sampled during sectioning). A 5-µm-thick section was obtained from each block and stained with H&E.
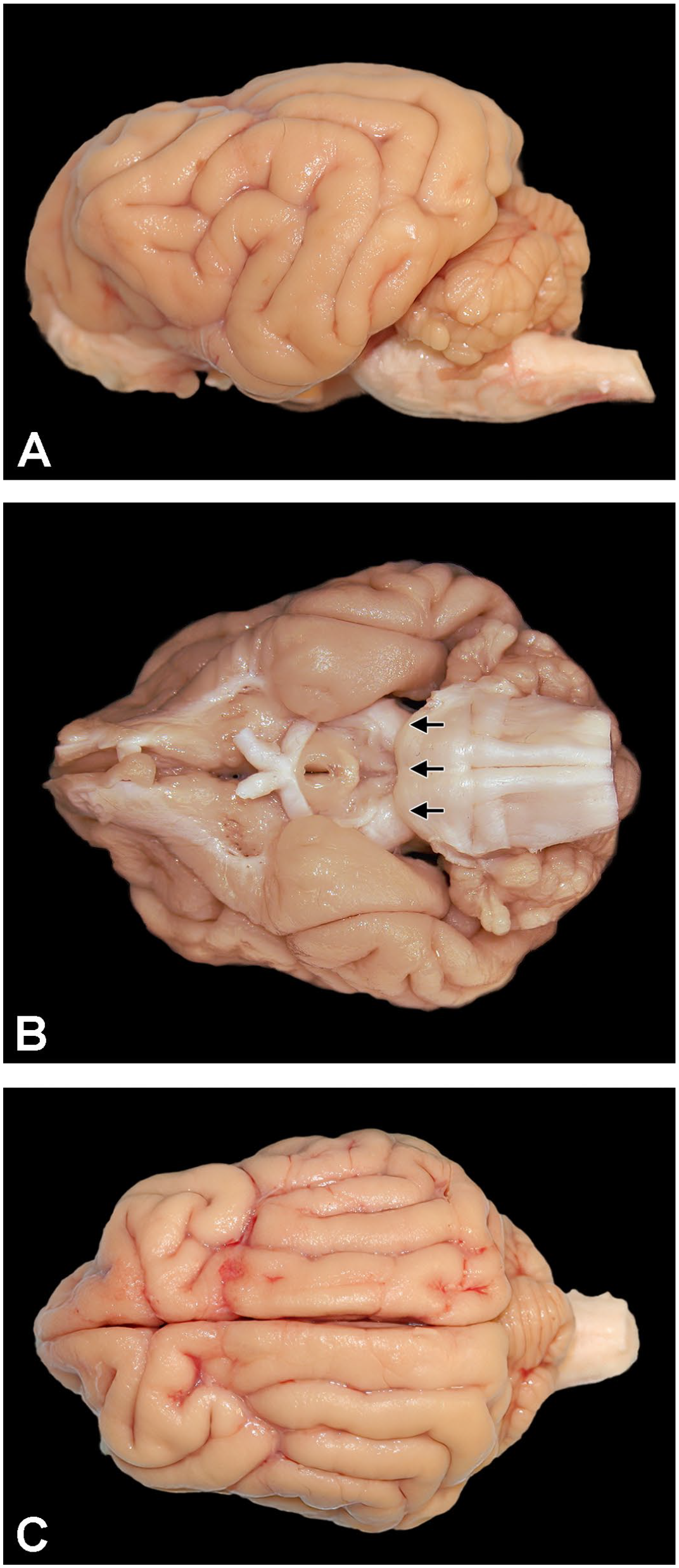
Macroscopic surface anatomy of a Beagle dog brain. Montage view of a fixed Beagle dog brain showing lateral (A), ventral (B), and dorsal (C) surfaces. The rostral border of the pons on the ventral surface (arrows in B) shows the location where the brain is divided in half in preparation for sampling.
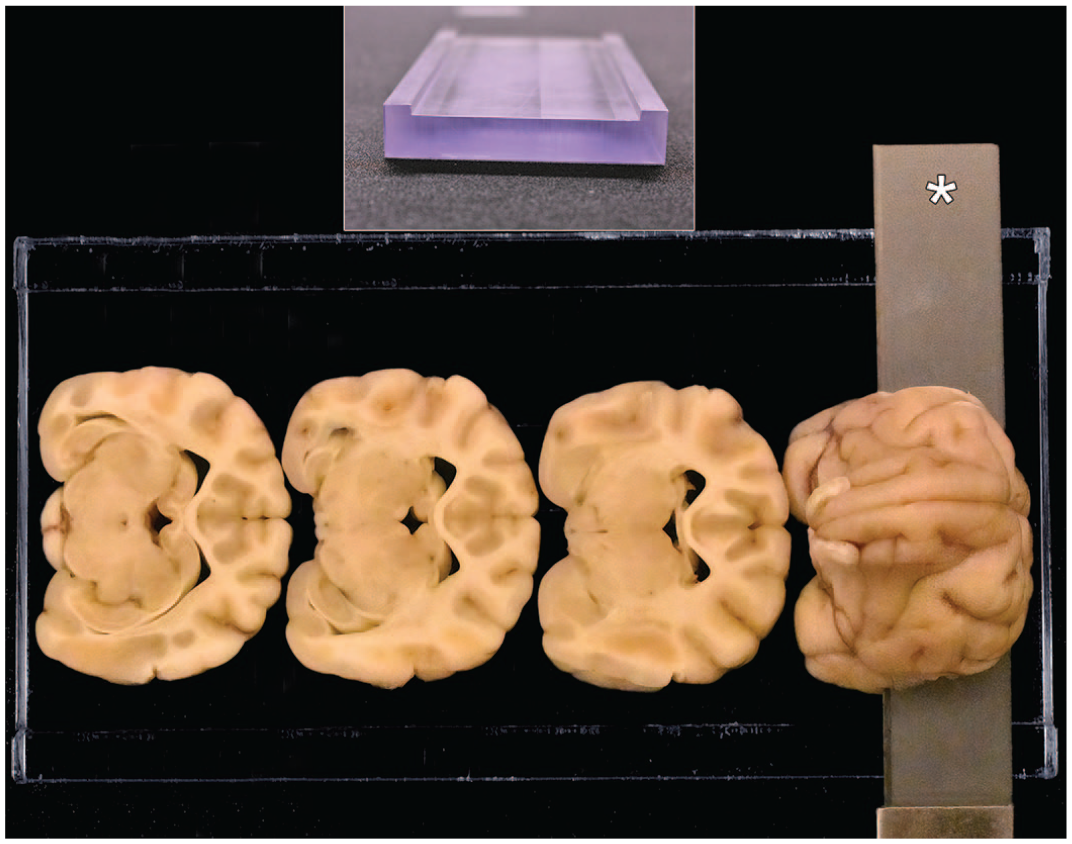
Purpose-made matrix tool for nonrodent brain sampling (developed at Pfizer Inc. for rabbits, 10 dogs, minipigs, 11 and nonhuman primates 9 ) used to obtain reproducible trimming planes. The matrix dimensions are 15-cm long and 8-cm wide, with a 6-cm wide central channel bordered by 2 outer raised edges that are 1-cm wide and 3.2 mm in height. Using this matrix, each half brain (rostral and caudal, separated using an initial coronal [transverse] cut at the rostral border of the pons [i.e., arrows in Figure 1B] may be divided into multiple full-coronal, 3.2-mm-thick slices using a tissue slicer blade (asterisk). Trimming of the rostral brain half is shown here.
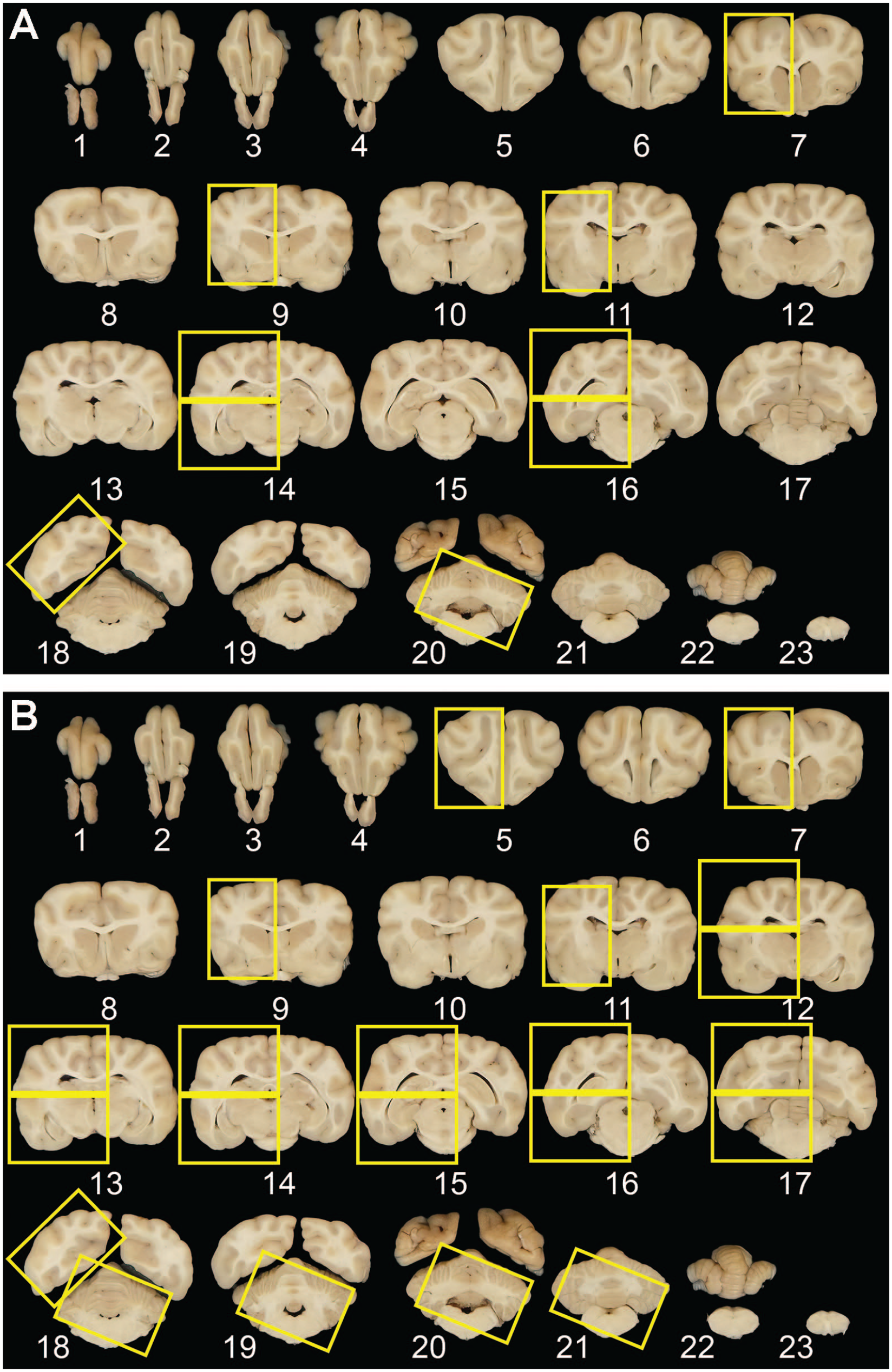
Coronal sectioning and recommended sampling of Beagle dog brain for toxicity studies. Set of 23 serial coronal brain slices obtained from a large adult Beagle dog using the purpose-made matrix tool (Figure 2). The yellow rectangles represent, respectively, the recommended trimming procedure for routine general toxicity studies (A) and a more extensive trimming approach suitable for neurotoxicity studies of test articles with suspected or known neurotoxic effects (B). Note that for the 7-level procedure (A), which is designed to meet current “best practice” recommendations for routine brain sampling (Toxicol Pathol 41[7]: 1028-1048, 20135), extra cassettes may be needed for certain levels of very large brains because some coronal hemisections (e.g., levels 14, 16) may need to be divided into dorsal and ventral halves to permit tissue sections to be applied to standard-sized glass slides.
Neuroanatomic Annotation
The figures included in the current guide are representative images for features that are typically visible in sections made from the 23 slices of a large dog brain (Figure 3B). Brain structures were annotated by comparison of dog neuroanatomic features to those of equivalent domains in other dog brain atlases15-18 as well as atlases for other major nonrodent test species—rabbits,10,19-21 minipigs,11,22-24 and NHPs.9,25-27
To minimize confusion, the neuroanatomic terminology in the current atlas conforms to the nomenclature conventions in a recent dog neuroanatomic atlas 17 (which is based on the human medical designations) but includes cross-referencing as needed to accepted Nomina Anatomica Veterinaria (NAV) standard anatomic terms for quadruped animals. 28
Results
As in all species, nervous system sampling in the dog for nonclinical toxicity testing should address both CNS and PNS structures. Harvesting the CNS (brain and spinal cord) as well as PNS (somatic and autonomic ganglia and nerves, as appropriate to address the study objective) typically should conform to the published STP “best practice” recommendations for CNS 5 and PNS 6 sampling and processing because these recommendations have been designed to address existing guidance by regulatory agencies (e.g., the U.S. Environmental Protection Agency [EPA] and U.S. Food and Drug Administration [FDA]) and global consortia seeking to standardize test methodology (e.g., International Council on Harmonisation of Technical Requirements for Pharmaceuticals for Human Use [ICH] and the Organisation for Economic Co-operation and Development [OECD]).
Brain
General descriptive anatomy of the brain of the Beagle dog
The brain of the Beagle dog weighs, on average, between 65 and 91 g (75 g on average in males [0.80% of body weight] and 71 g on average in females [0.92% of body weight]). These historical brain weight data (from Pfizer, Inc., Groton, CT, over the years 2010-2020) were obtained from Beagle dogs (N = 222 males and N = 237 females) aged more than 8 months (approximately 240 days) when enrolled in routine or investigative toxicity studies and then necropsied at ages ranging from 285 to 793 days. Brain weights can vary with age and sex of the animals. Brain weights of male and older animals are greater than those of female (age-matched) and younger animals.
Compared to primate brains, the Beagle dog brain has rounded frontal poles with well-developed olfactory lobes, ample ventricles, less-prominent gyri and sulci, smaller occipital poles, and a well-developed auditory system. The dog brain has an elongated rostrocaudal dimension compared to humans and common NHP test species (e.g., cynomolgus macaques [Macaca fascicularis]) in which the brain stem is redirected ventrally.
Collection, sampling, and sectioning of the Beagle dog brain for general toxicity studies
For general toxicity studies and in alignment with the approach used in the cynomolgus monkey,5,9 the technique described below allows for consistent sampling of many important neuroanatomic regions of the Beagle dog brain using a limited number of blocks and slides. This technique is easy to implement in general toxicity studies and requires minimal neuroanatomic knowledge. Based on specific study needs, in particular target expression as well as test article (chemical or vector or transgene) biodistribution, additional samples may be added to the core list. Although perfusion fixation is ideal for neurohistological evaluation, it is not required for general toxicity studies.
The dorsal calvarium is exposed by removing the skin, connective tissue, and muscle from the dorsal aspect of the skull extending from the top of the orbits and ears to the atlanto-occipital area. An autopsy saw (e.g., Model #100000, 1.5 amps, 115 volts AC, Hz50/60, Lipshaw, Pittsburgh, PA or Model 810, 120V-60 Hz 2.0A, Stryker, Kalamazoo, MI) is used to cut and remove the bone of the dorsal calvarium. This step necessitates technical skill to avoid damaging the underlying brain parenchyma. The dura mater is exposed and reflected, and the falx cerebri that descends vertically in the longitudinal fissure on the midline between the cerebral hemispheres is removed. This latter step is mandatory to remove the brain from the skull without tearing the neural tissue. When removing the brain, special care is needed at the levels of the pituitary gland (which may remain entrapped in the sella turcica on the base of the skull) and olfactory bulbs and tract (which will require careful dissection of these protruding brain regions from the surrounding bone). The CNs should be cut as far from the brain surface as possible because the nerve roots are important anatomical landmarks to identify appropriate levels during trimming.
The intact brain is fixed by immersion in NBF for at least 48 hours before trimming at a fixative: tissue volume ratio of at least 10:1. Using the most rostral portion of the pons as a landmark (Figure 1A and B), the entire fixed brain can be held firmly in the matrix tool (described above) and cut dorsoventrally with a tissue slicer blade at a 10° to 20° angle (Figure 2). This technique produces approximately 17 to 18 full-coronal slices of the brain in young dogs and up to 23 slices in older males (Figure 3A and B). All slices are 3.2 mm in thickness with the exception of the most rostral and most caudal slices, which may be thicker because they include the most rostral and caudal uncut surfaces of the brain. The rostral aspect of each slice will be placed “down” in the cassette for embedding so that side of the slice will be sectioned.
Knowledge of the test article for potential neurotoxicity (based on in vitro and early non-GLP in vivo single-dose toxicokinetic studies as well as published literature) will drive the required sampling to include potentially susceptible neuroanatomical areas even if they are not included in the routine recommended 7 levels. 5 For test articles with unknown susceptibility to neurotoxicity, STP “best practices” for general toxicity studies recommend at least 5 levels in nonrodents as a minimal approach for screening the brain. 5 This evaluation may be performed in coronal hemisections because test articles are presumed to affect both sides of the brain equally. For our scheme, 7 different brain levels (identified by squares in Figure 3A) containing frontal pole, anterior [rostral] commissure, rostral thalamus, substantia nigra and hippocampus, caudal thalamus, occipital lobe, and the middle of the cerebellum with brain stem) are trimmed minimally at the edges to fit into standard tissue cassettes. Two hemisections (rostral thalamus and caudal thalamus) are often too large to fit intact into a single cassette and so may need to be divided in half (dorsal and ventral), with each half placed in a separate cassette (Figure 3A). This trimming scheme will yield 7 to 9 cassettes depending on the brain size. Sections will fit on standard glass slides (75 × 25 mm Micro Slides, Superfrost, White clipped corner for Leica IP Ink Jet Printer, Catalog #89078-500, VWR International, LLC, Radnor, PA). In general, sampling is performed unilaterally, but residual slices from the contralateral hemisphere should be retained in fixative (if not used for biochemical or expression assays) for future evaluation if needed. Although this technique provides a very consistent way to sample the brain, comparable sections from different animals will not be perfectly identical because differences in brain sizes of animals will lead to modest shifts in the positions of coronal trimming planes that might miss some neuroanatomic areas with small dimension. This minimal anatomic variation is acceptable as long as the major neuroanatomical structures targeted by this sampling approach (Table 1) together with the functions, neural connections, and principal neurotransmitters they serve are obtained consistently.
Rationale for selecting major neural structures to sample during nonclinical general toxicity and neurotoxicity studies in dogs.
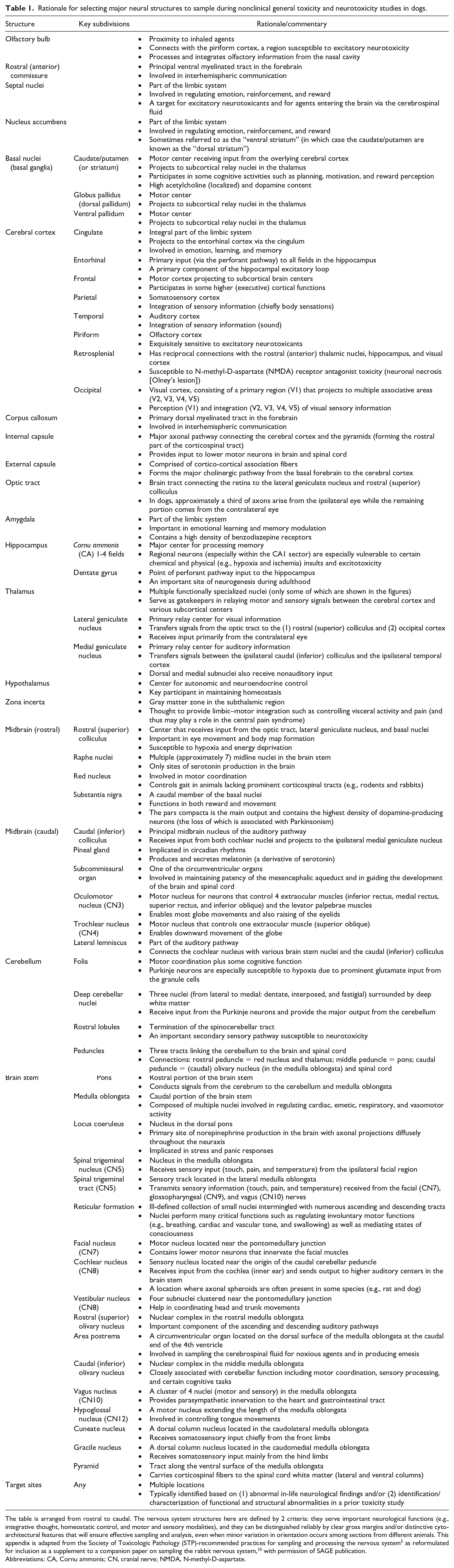
The table is arranged from rostral to caudal. The nervous system structures here are defined by 2 criteria: they serve important neurological functions (e.g., integrative thought, homeostatic control, and motor and sensory modalities), and they can be distinguished reliably by clear gross margins and/or distinctive cytoarchitectural features that will ensure effective sampling and analysis, even when minor variation in orientation occurs among sections from different animals. This appendix is adapted from the Society of Toxicologic Pathology (STP)-recommended practices for sampling and processing the nervous system 5 as reformulated for inclusion as a supplement to a companion paper on sampling the rabbit nervous system, 10 with permission of SAGE publication.
Abbreviations: CA, Cornu ammonis; CN, cranial nerve; NMDA, N-methyl-D-aspartate.
Collection, sampling, and sectioning of the Beagle dog brain for dedicated neurotoxicity studies
For dedicated neurotoxicity studies where in-life clinical findings, chemical structure, known or suspected mode of action, or other factors suggest that brain toxicity is likely or certain, an enhanced brain-trimming strategy has been recommended to ensure that sufficient brain tissue has been evaluated.12,29 Although the choice for this case-by-case sampling is left to the discretion of each institution, several strategies for further sampling may be pursued. Typical options for selecting additional slices for evaluation are nontargeted (e.g., embed slices taken at regular spacing along the length of the brain) or targeted (e.g., embed slices with specific regions based on their functional neuroanatomy). Using the 23 slices as a template, 14 levels are suggested. These will include the 7 levels evaluated during general toxicity studies (Figure 3A) with an additional 7 levels that capture other major functional domains of the brain (Figure 3B).
Possible adjustments to the trimming schemes for sampling Beagle dog brain
The 7-level trimming scheme for general toxicity studies is the minimum number of dog brain hemisections needed to conform to the STP “best practice” recommendations for brain sampling in nonrodents during general toxicity studies. 5 Therefore, this scheme is already reduced as far as possible as the microscopic evaluation in dogs is performed on coronal hemisections rather than the full sections used for rodents 5 and rabbits. 10
Modest adjustments to the 14-level trimming scheme for dedicated neurotoxicity studies may be implemented at the discretion of the study design team and/or institution in certain cases, with appropriate scientific justification. For example, fewer slices might be evaluated by omitting the forebrain section if the test article is known to effect only in the cerebellum, while one or more additional rostral forebrain sections might be needed if toxicity to the olfactory system is expected.
Principal neuroanatomic features of the Beagle dog brain
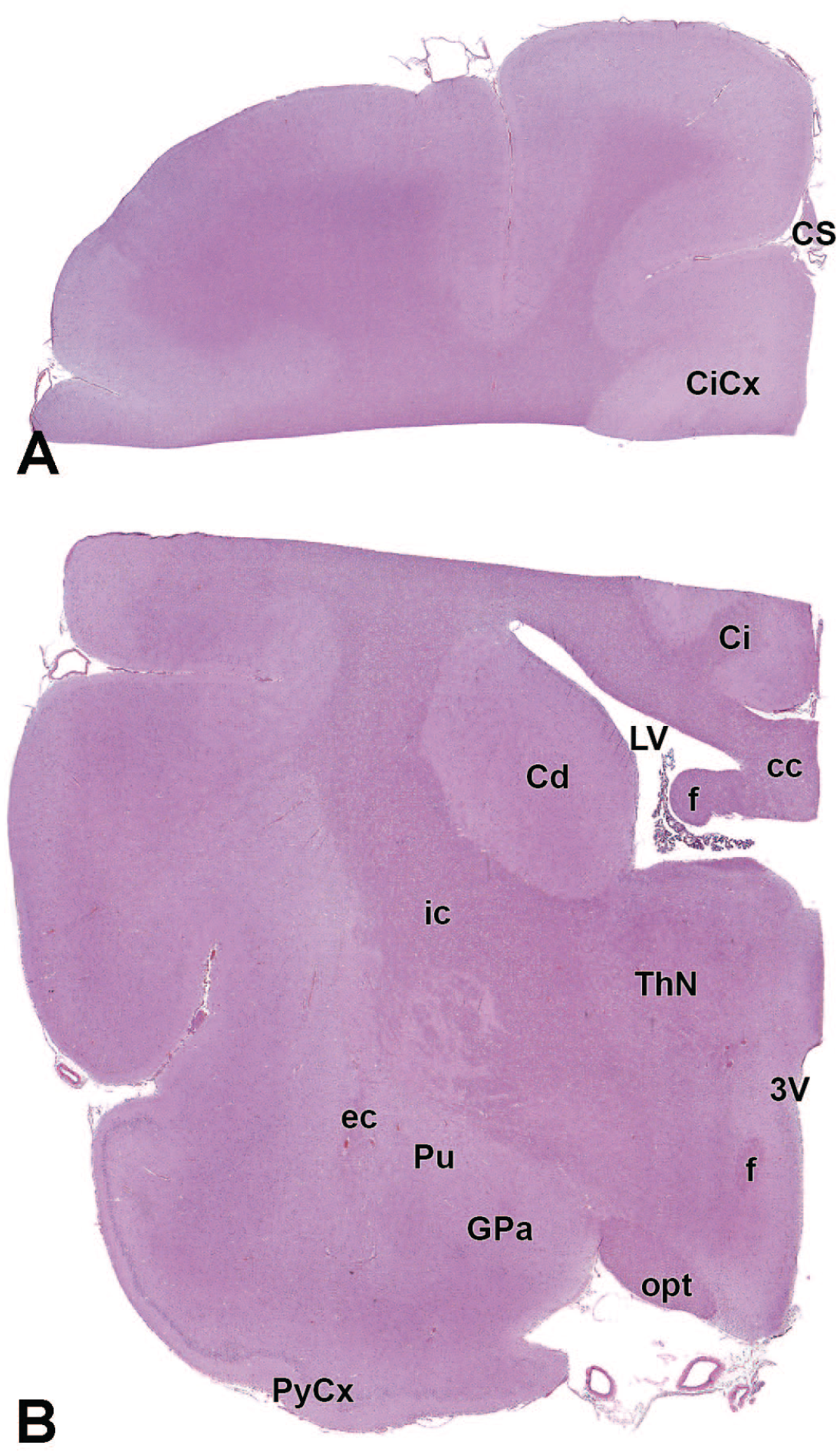
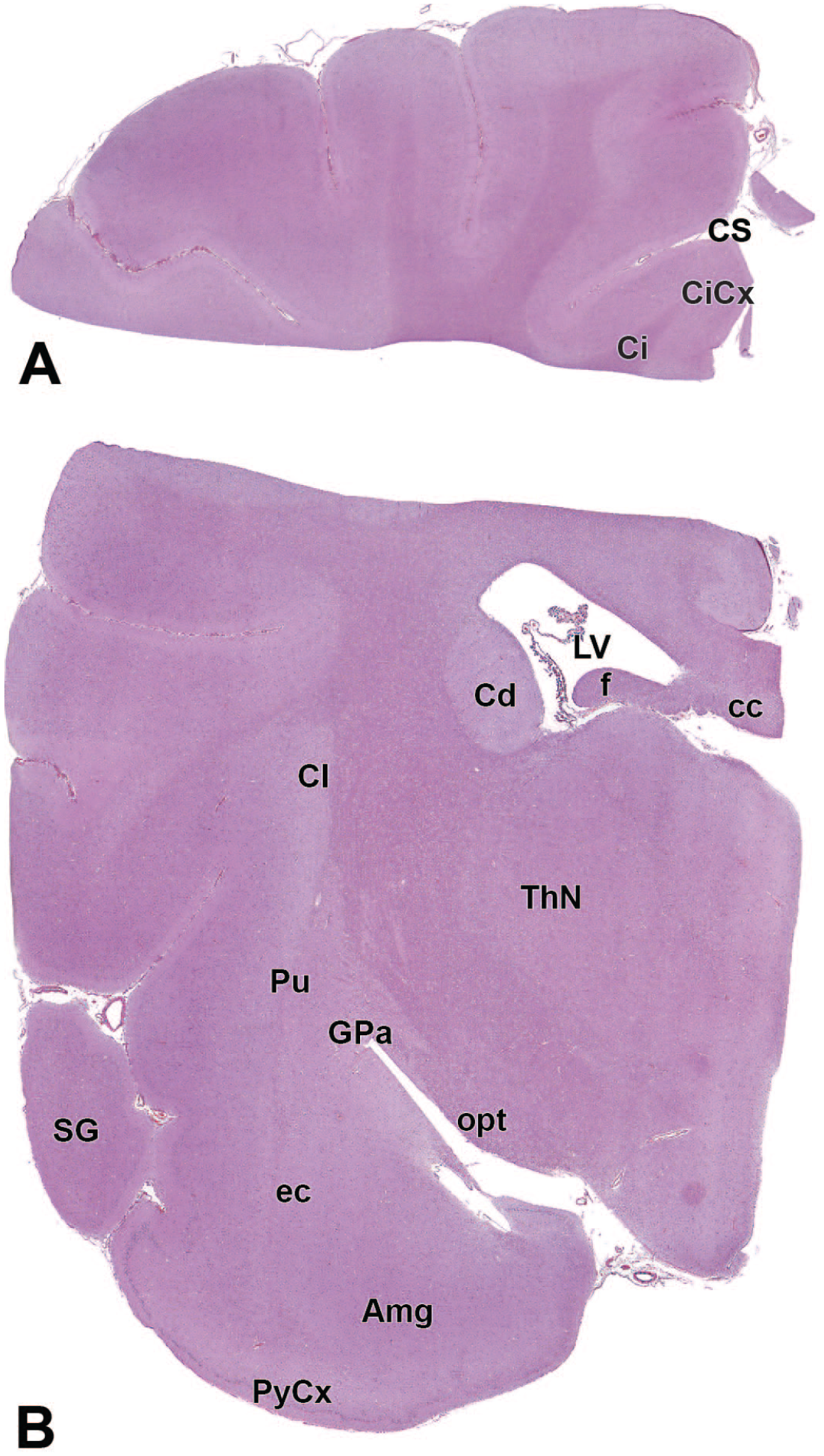
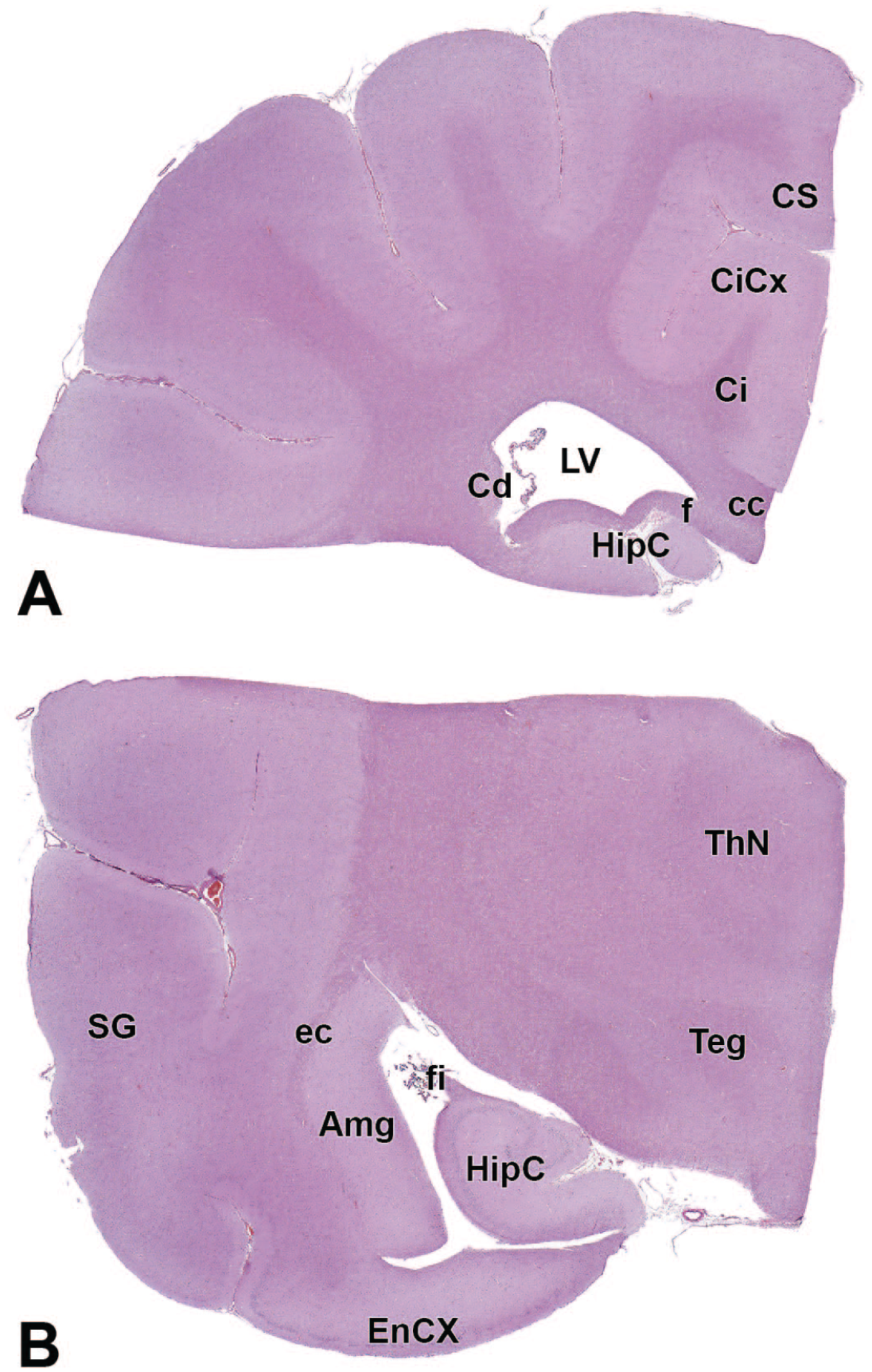
The primary neuroanatomic features in all brain hemisections from Beagle dogs are shown in Figures 4 to 25 (corresponding to levels 1-22 from Figure 3B). The main structures in each section are annotated. Owing to spatial constraints, annotations have been made only for the largest and most biologically and toxicologically significant structures (based on known regional function and known sensitivity to neurotoxicants [see Table 1]).
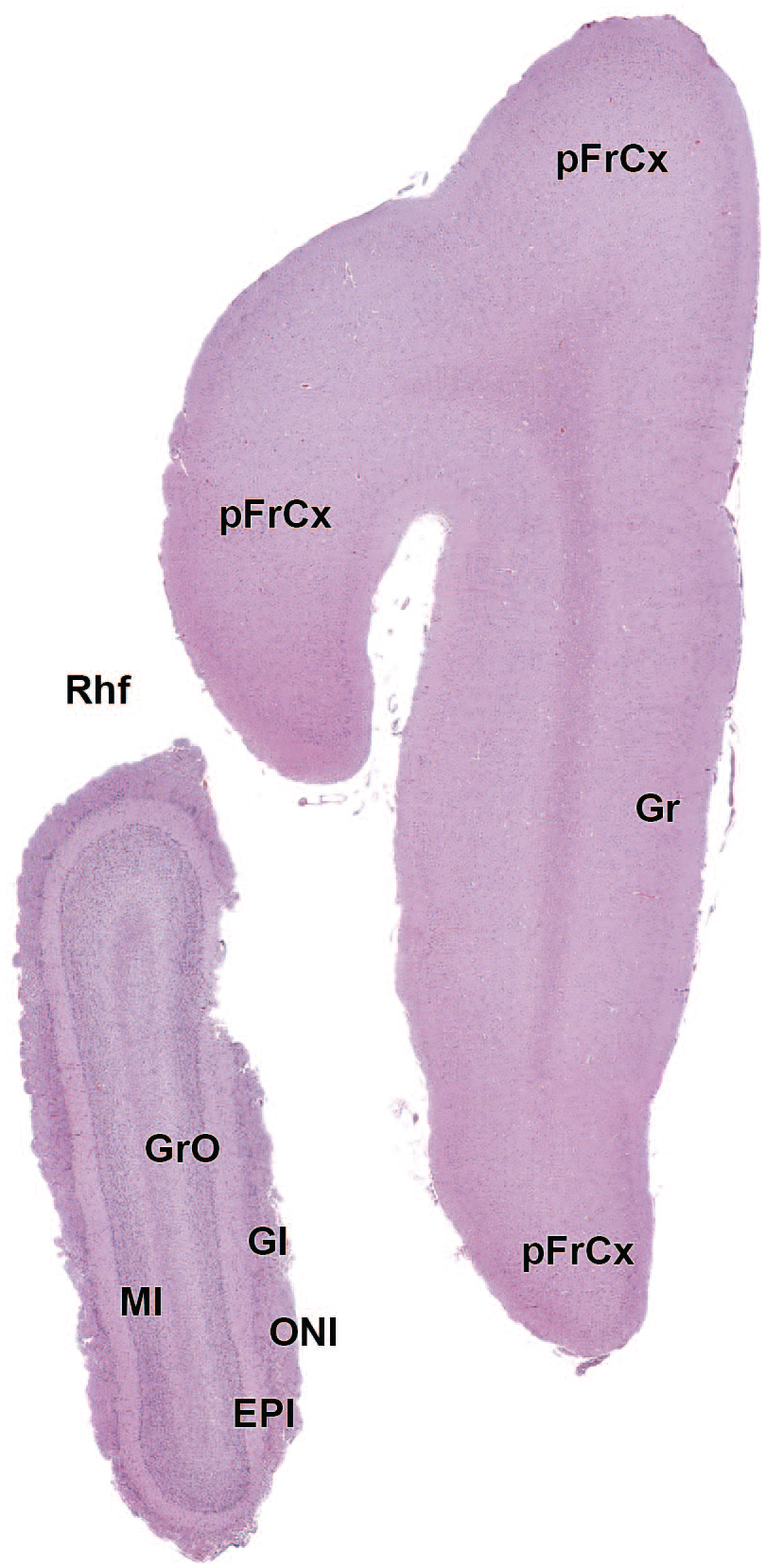
EPl: External plexiform layer of the olfactory bulb
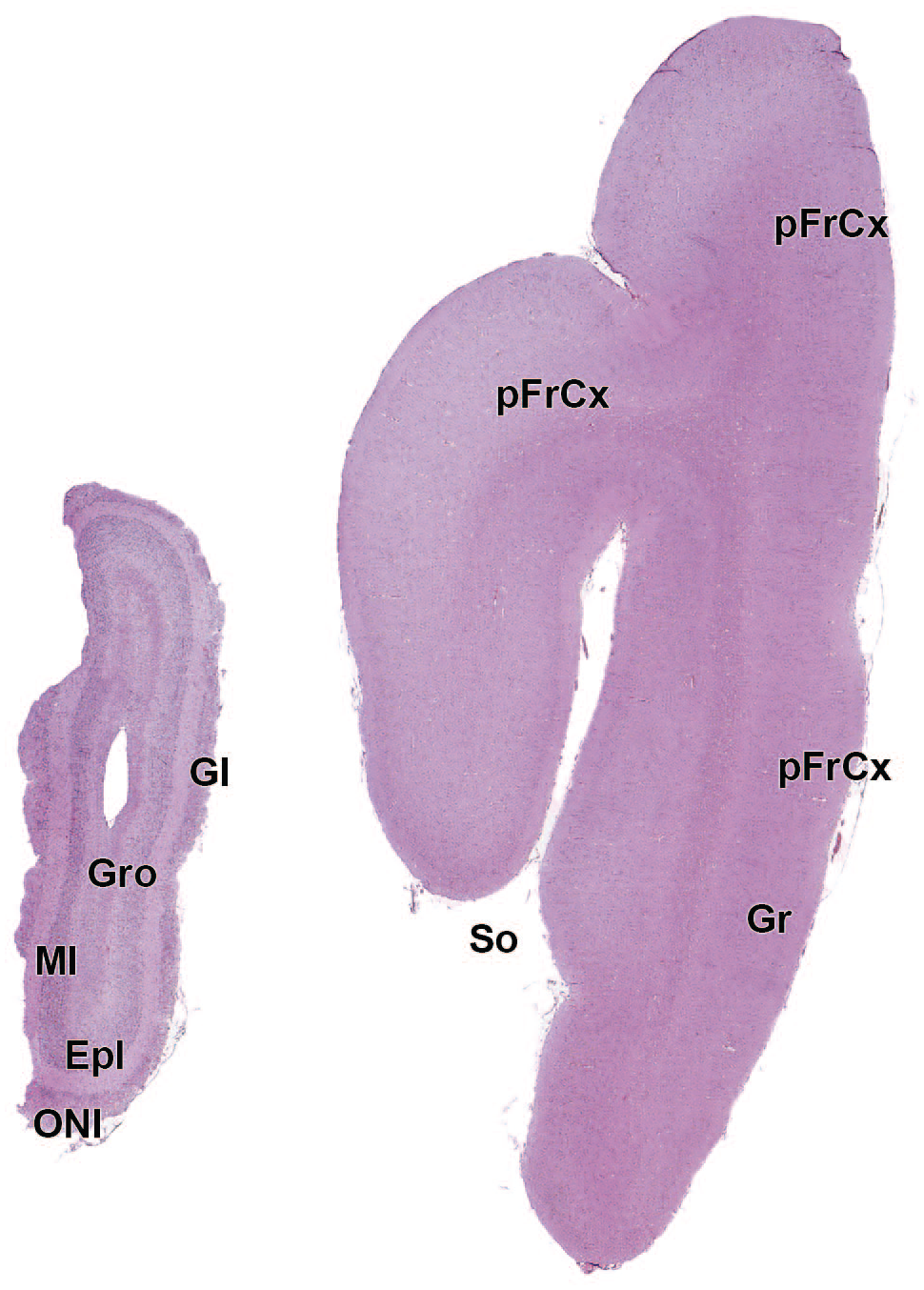
EPl: External plexiform layer of the olfactory bulb
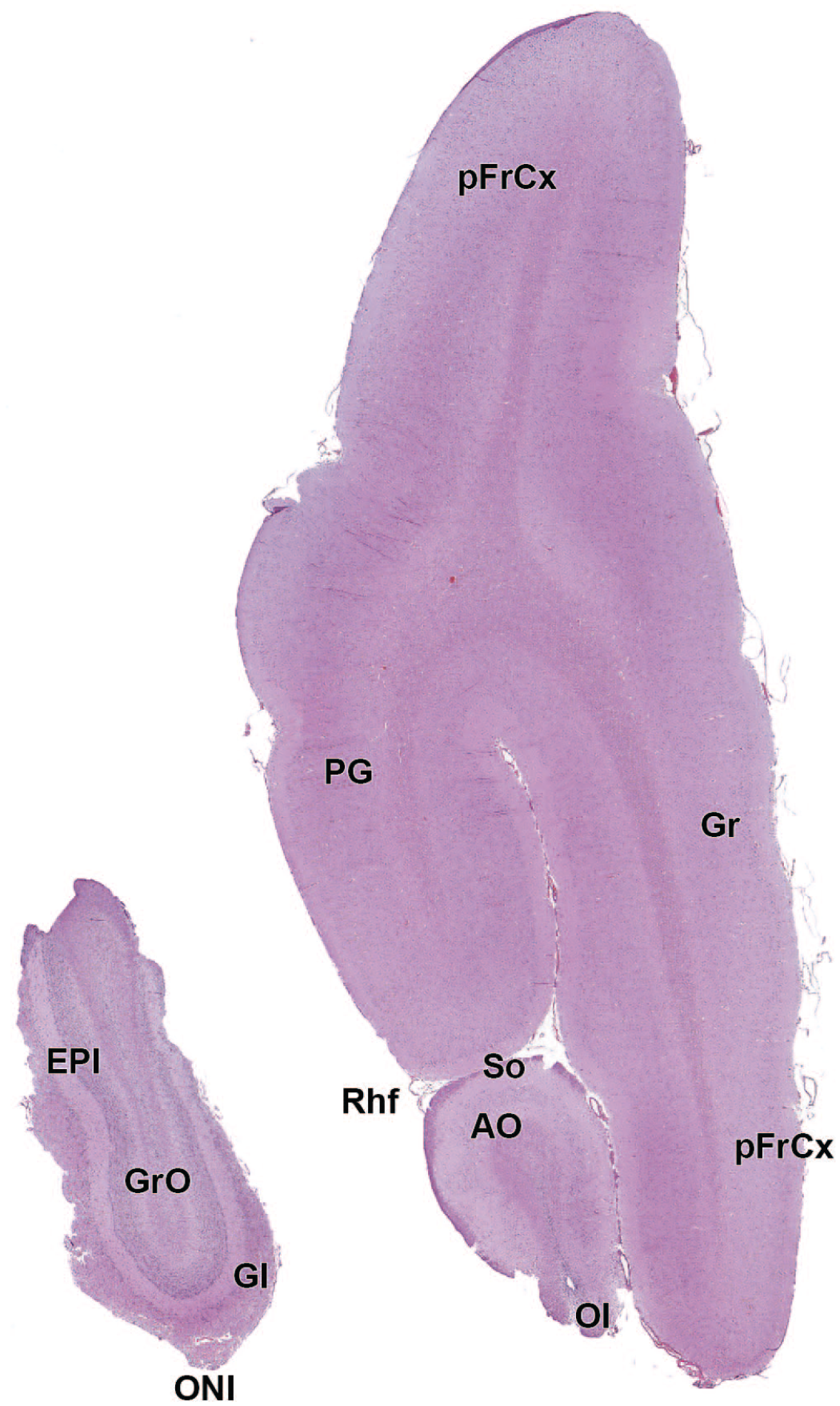
AO: Anterior (rostral) olfactory nucleus
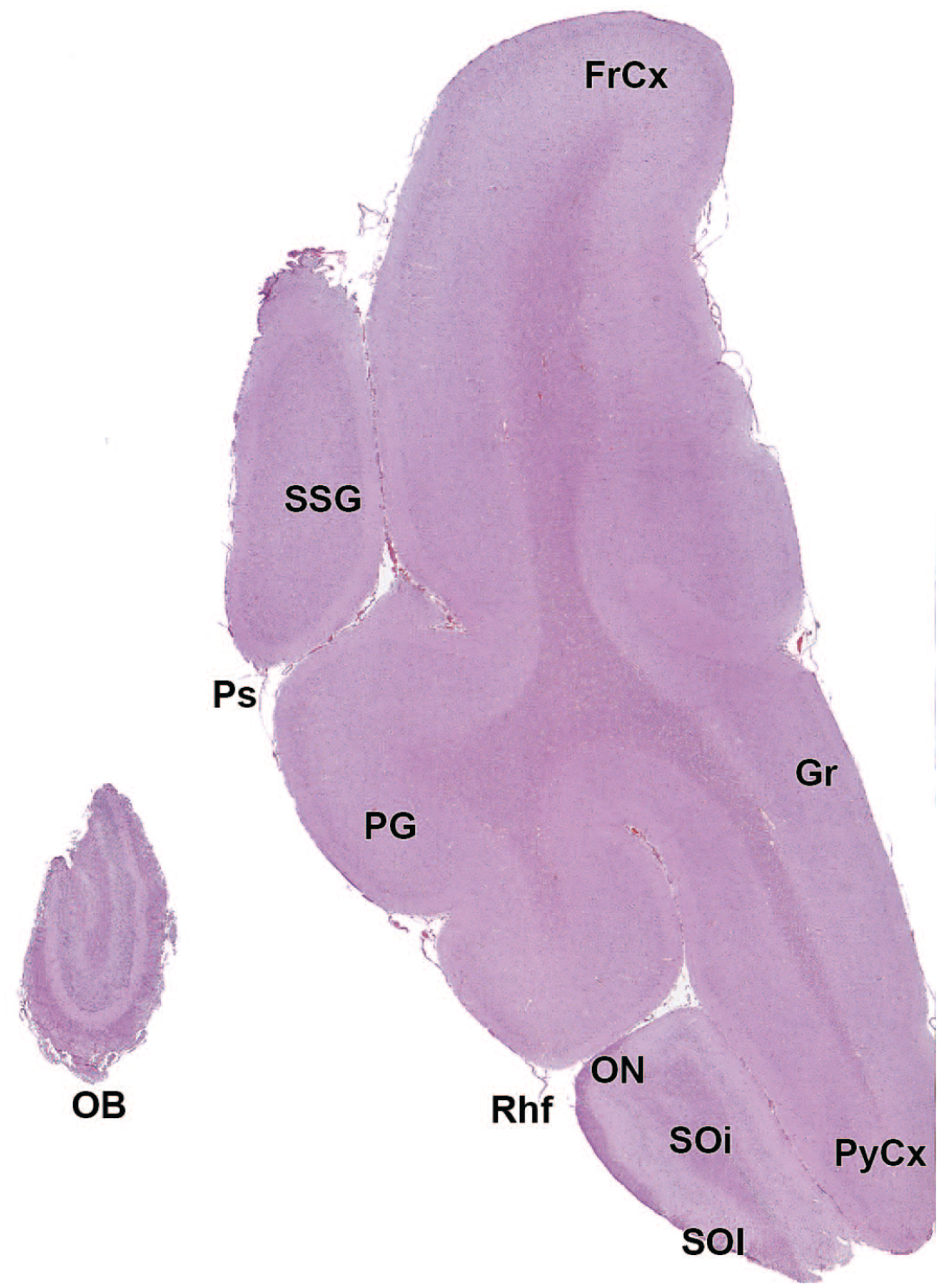
FrCx: Frontal cortex
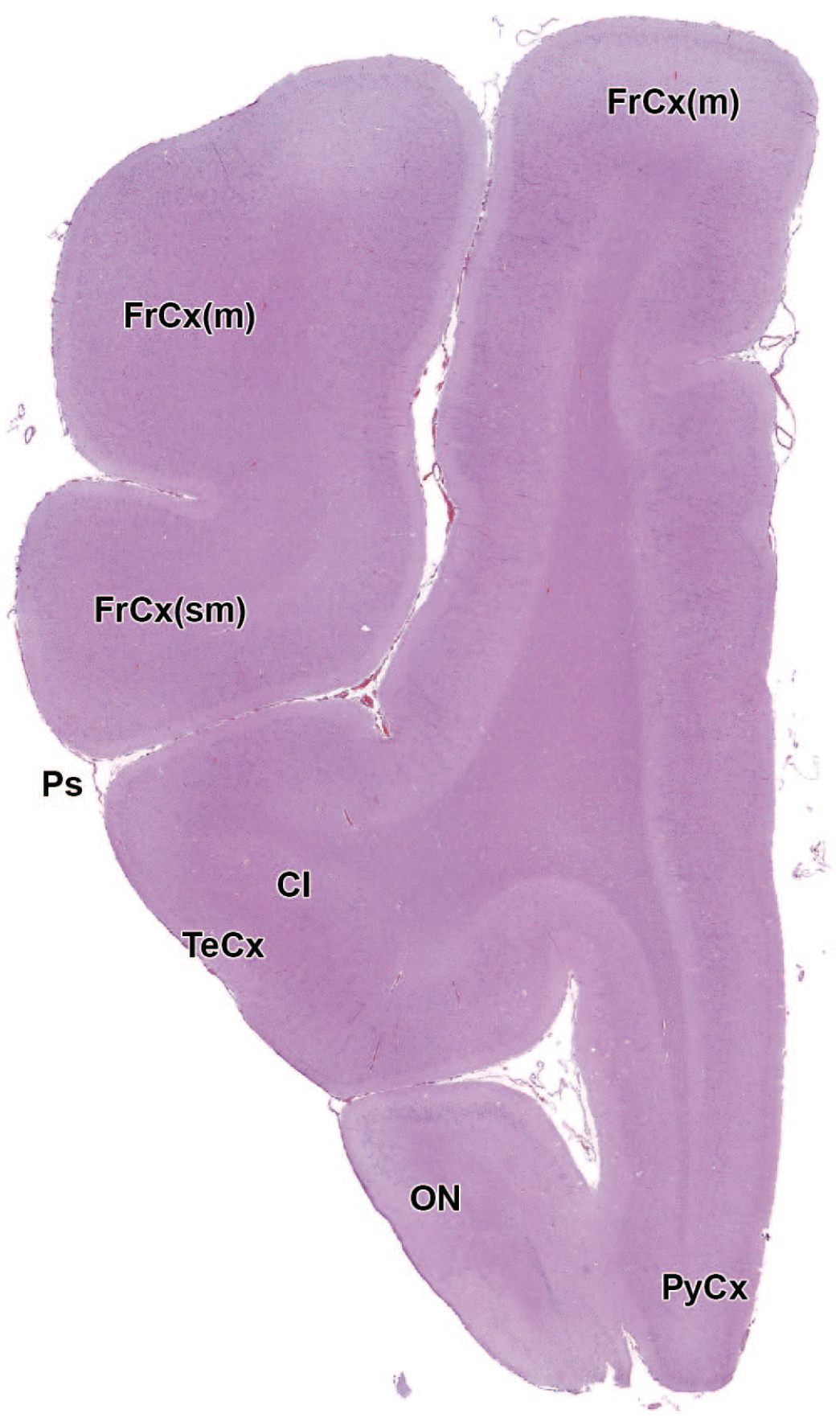
Cl: Claustrum
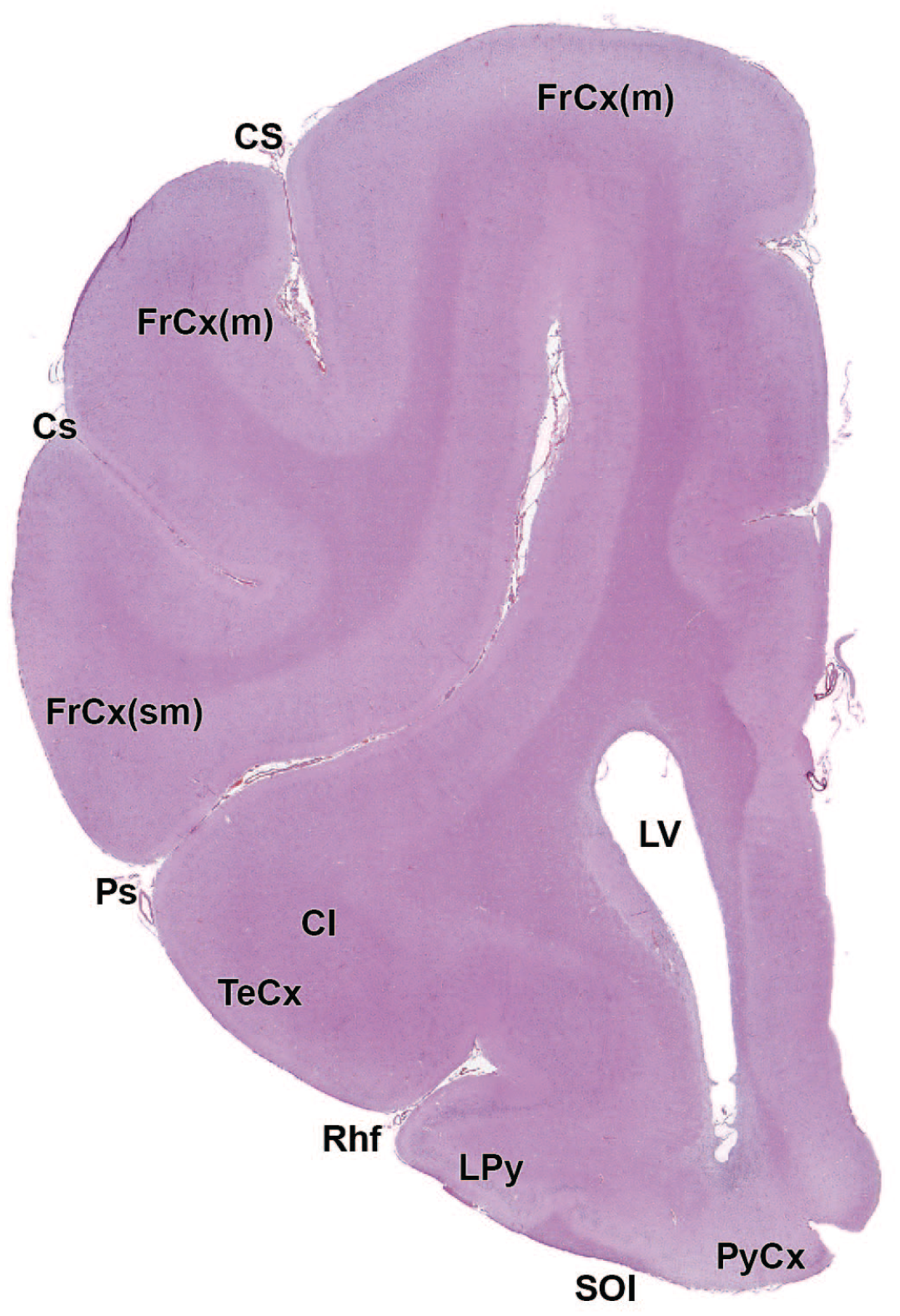
Cl: Claustrum
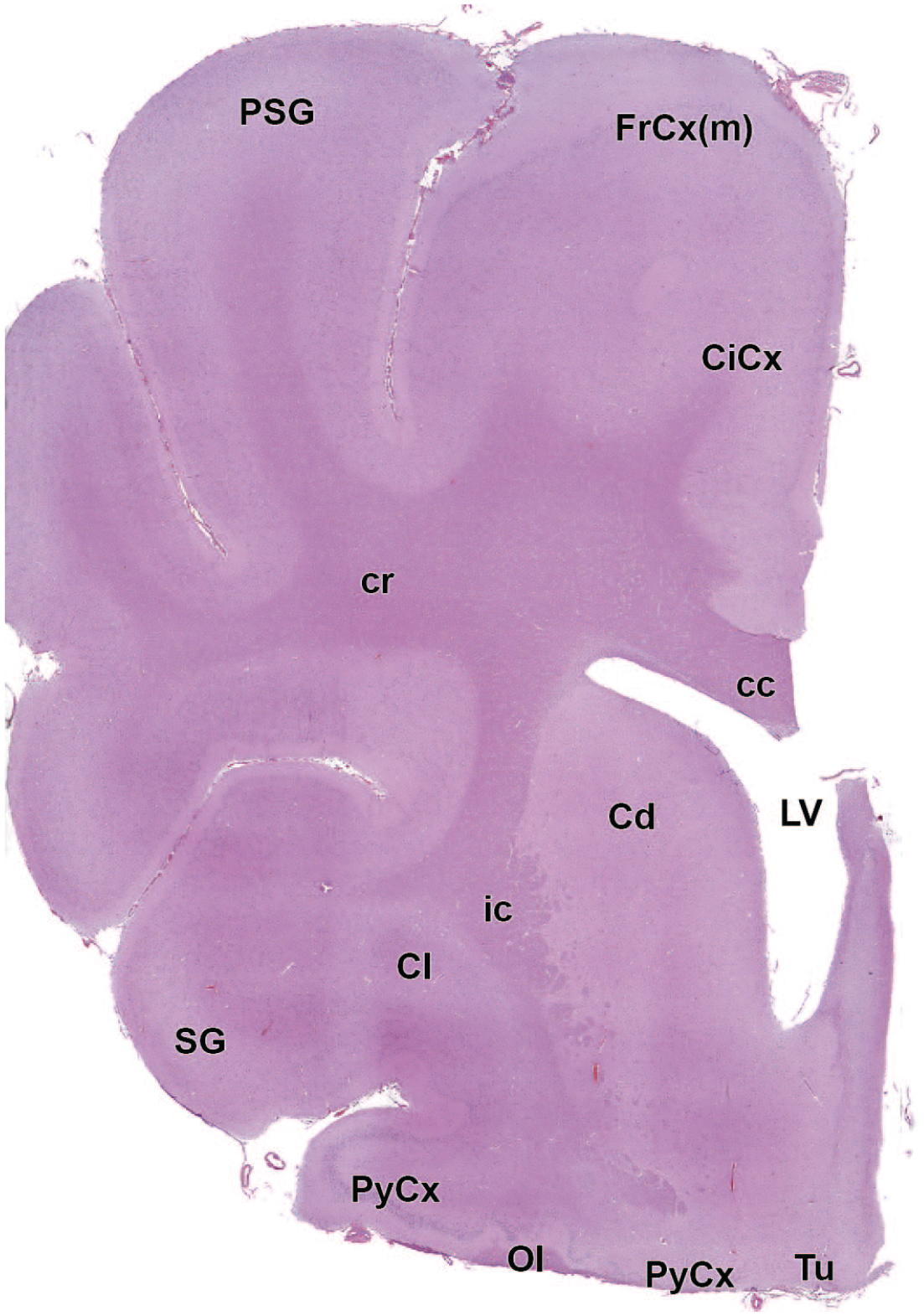
cc: Corpus callosum
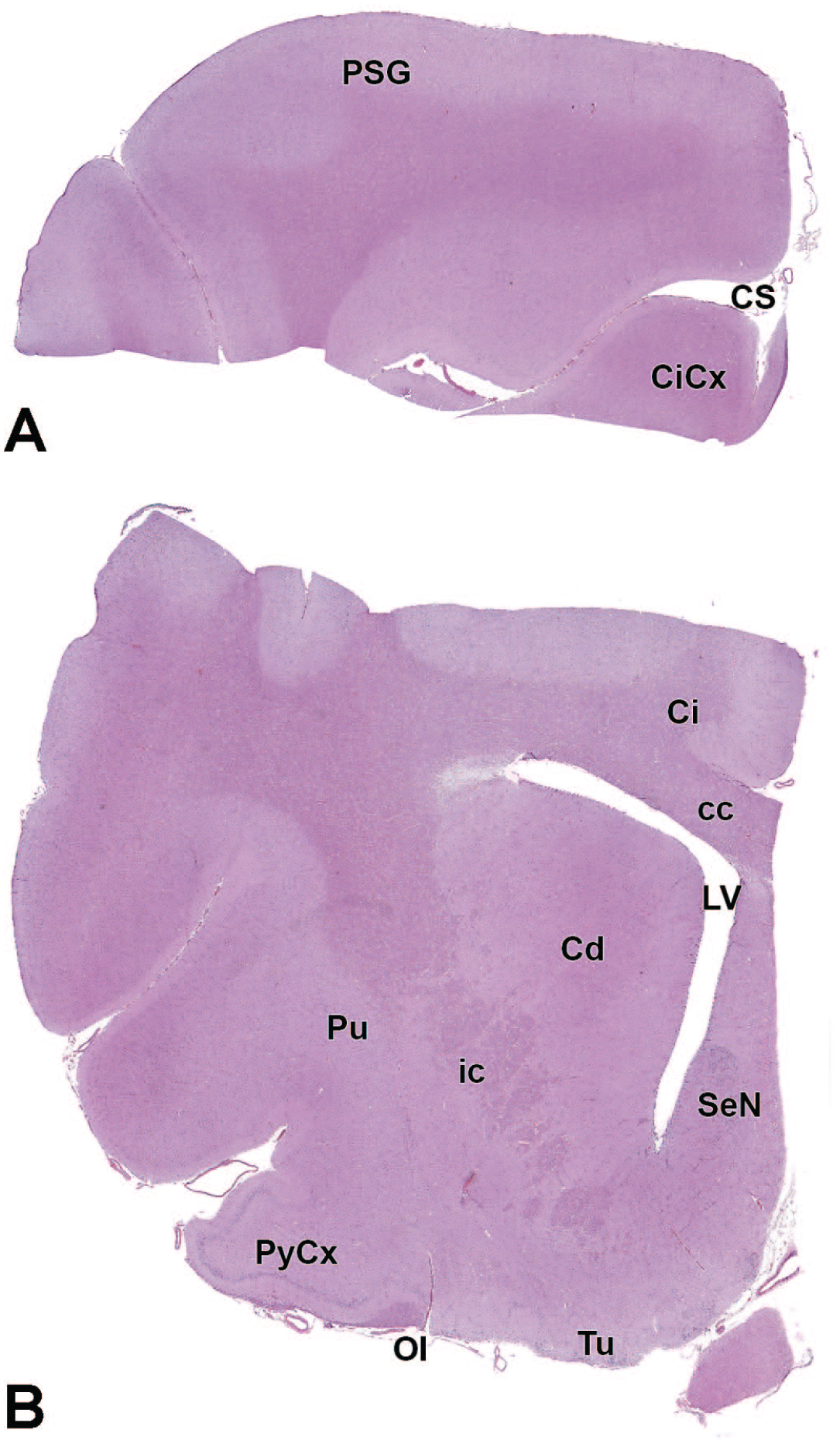
cc: Corpus callosum
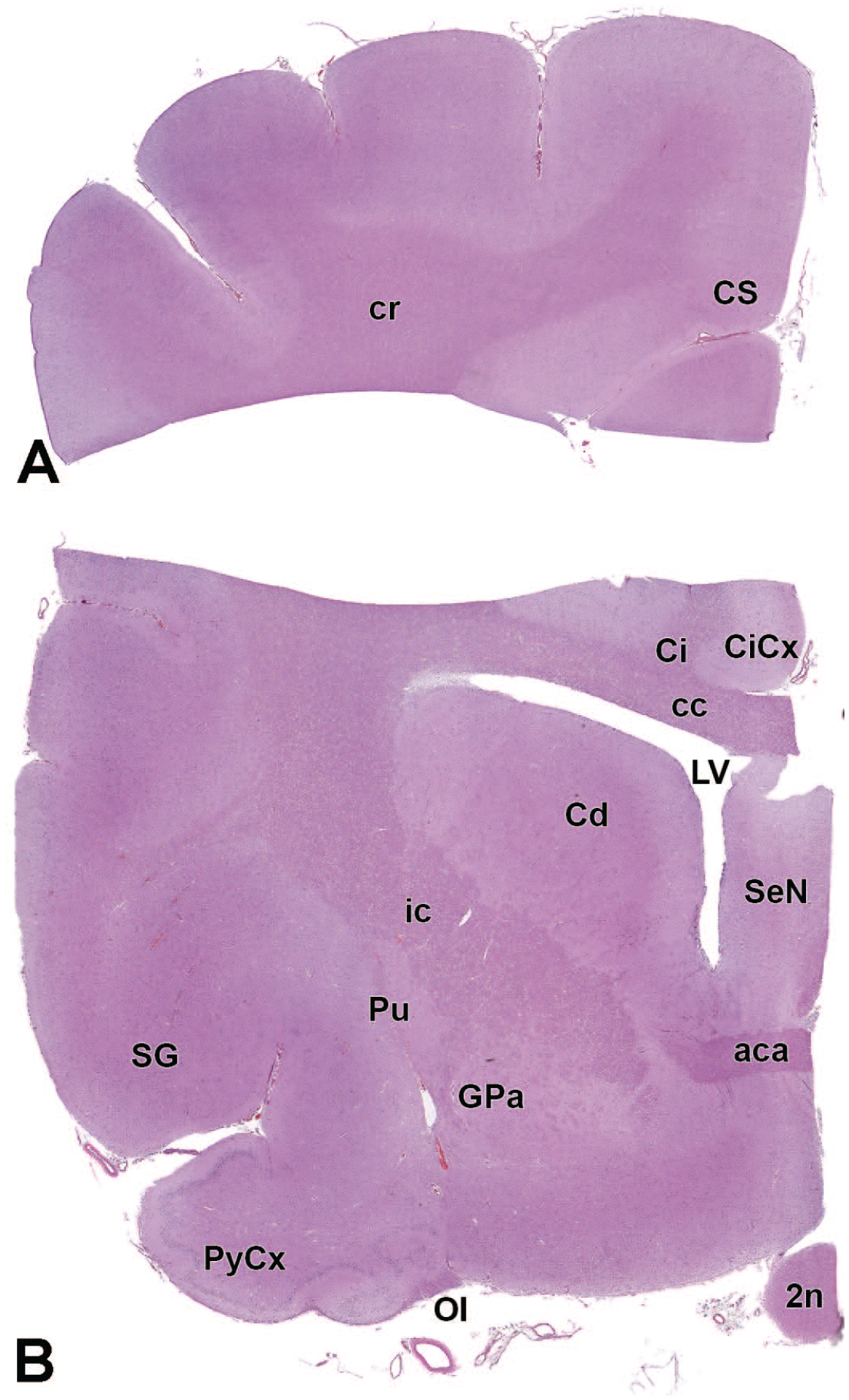
2n: Optic nerve (cranial nerve II)
3V: Third ventricle
Amg: Amygdala
Amg: Amygdala
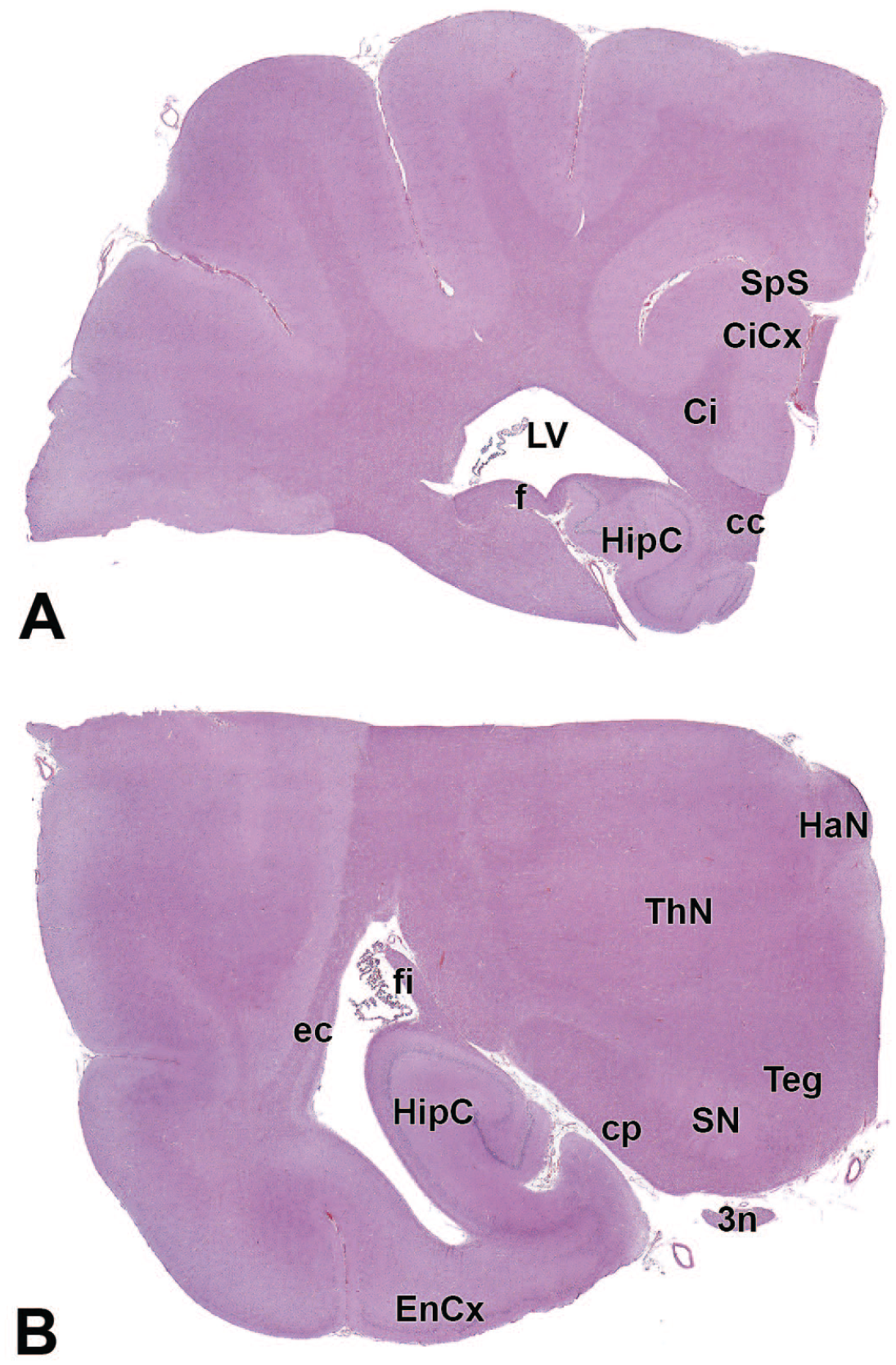
3n: Oculomotor nerve (cranial nerve III)
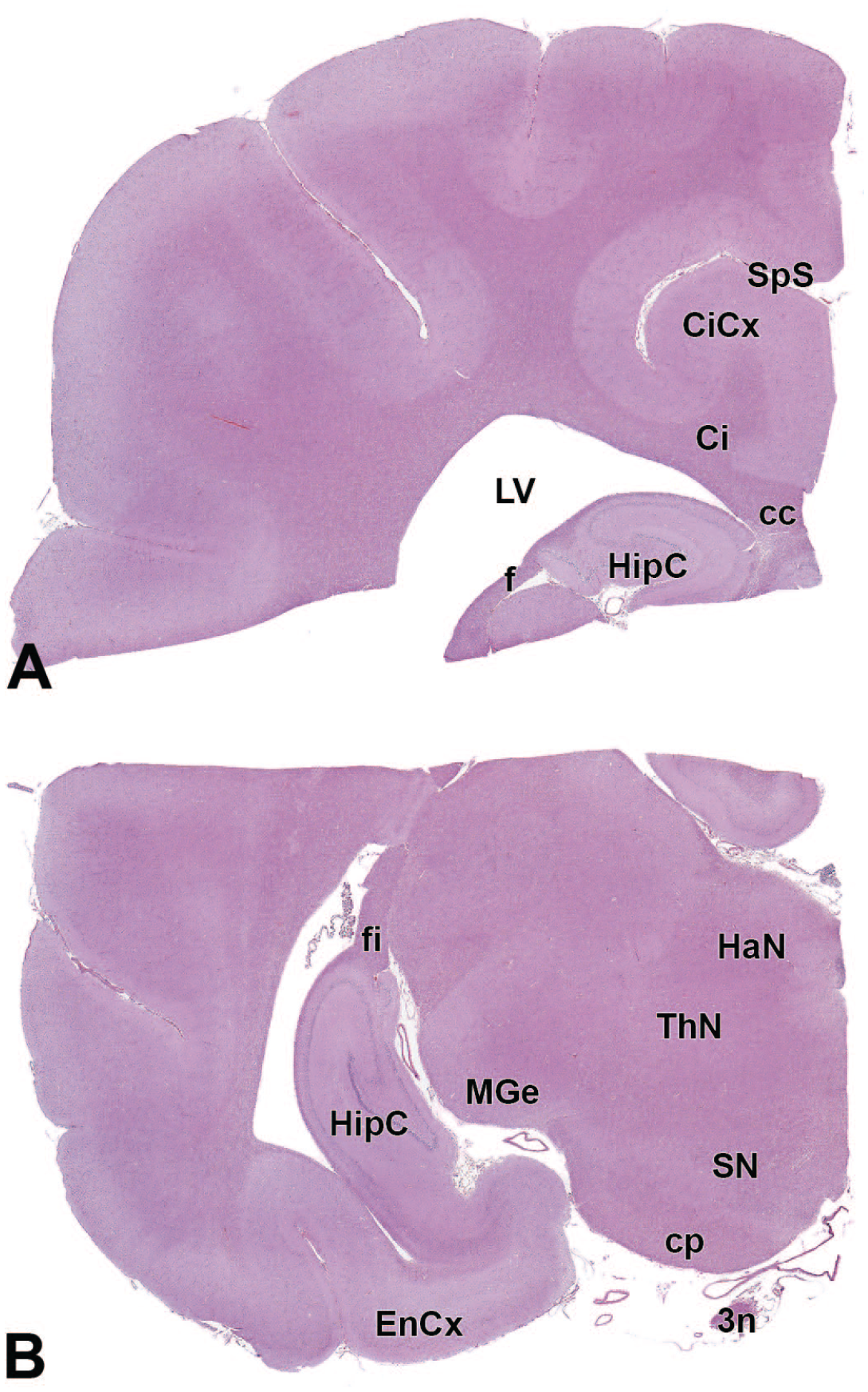
3n: Oculomotor nerve (cranial nerve III)
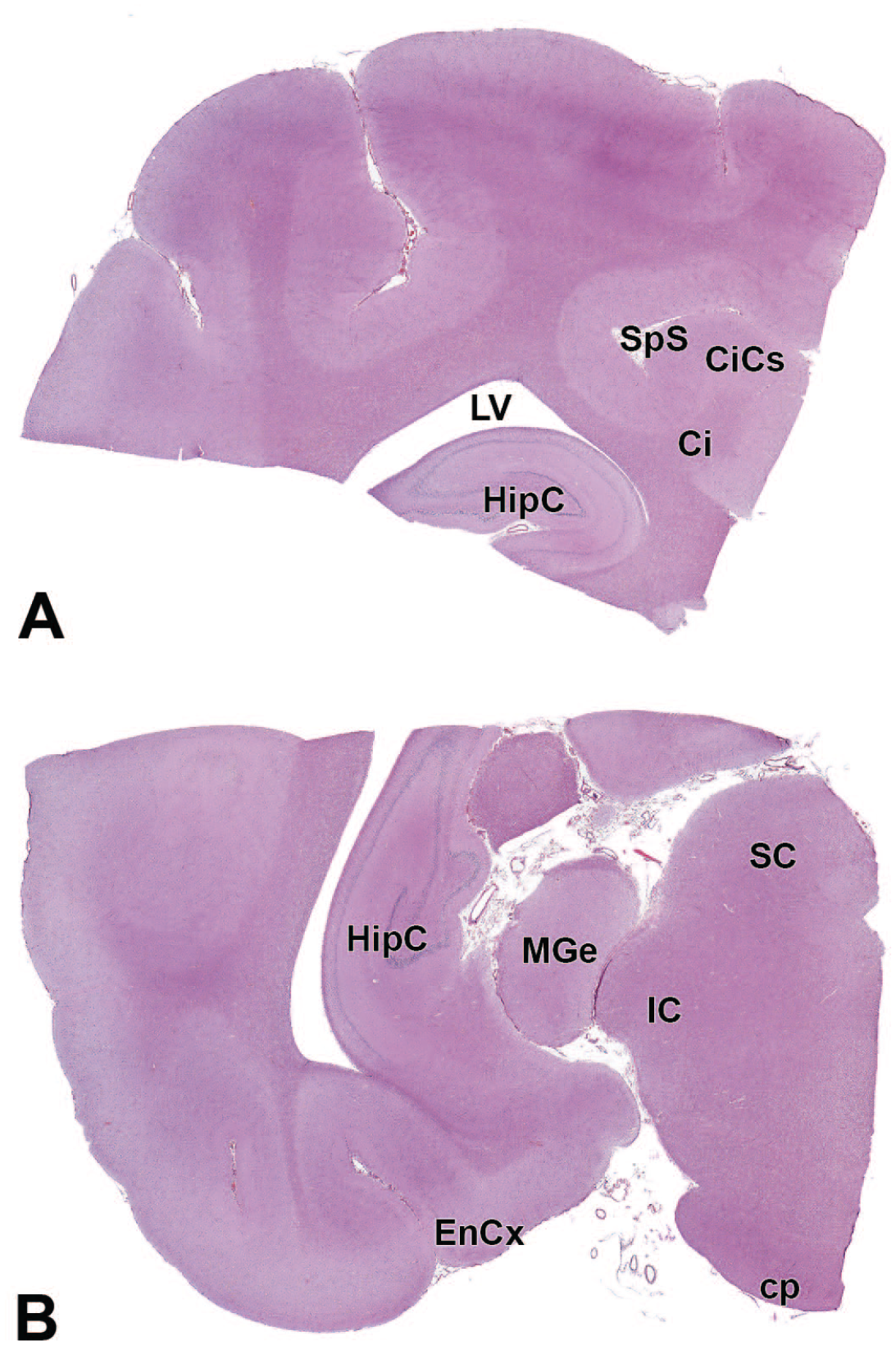
HipC: Hippocampus
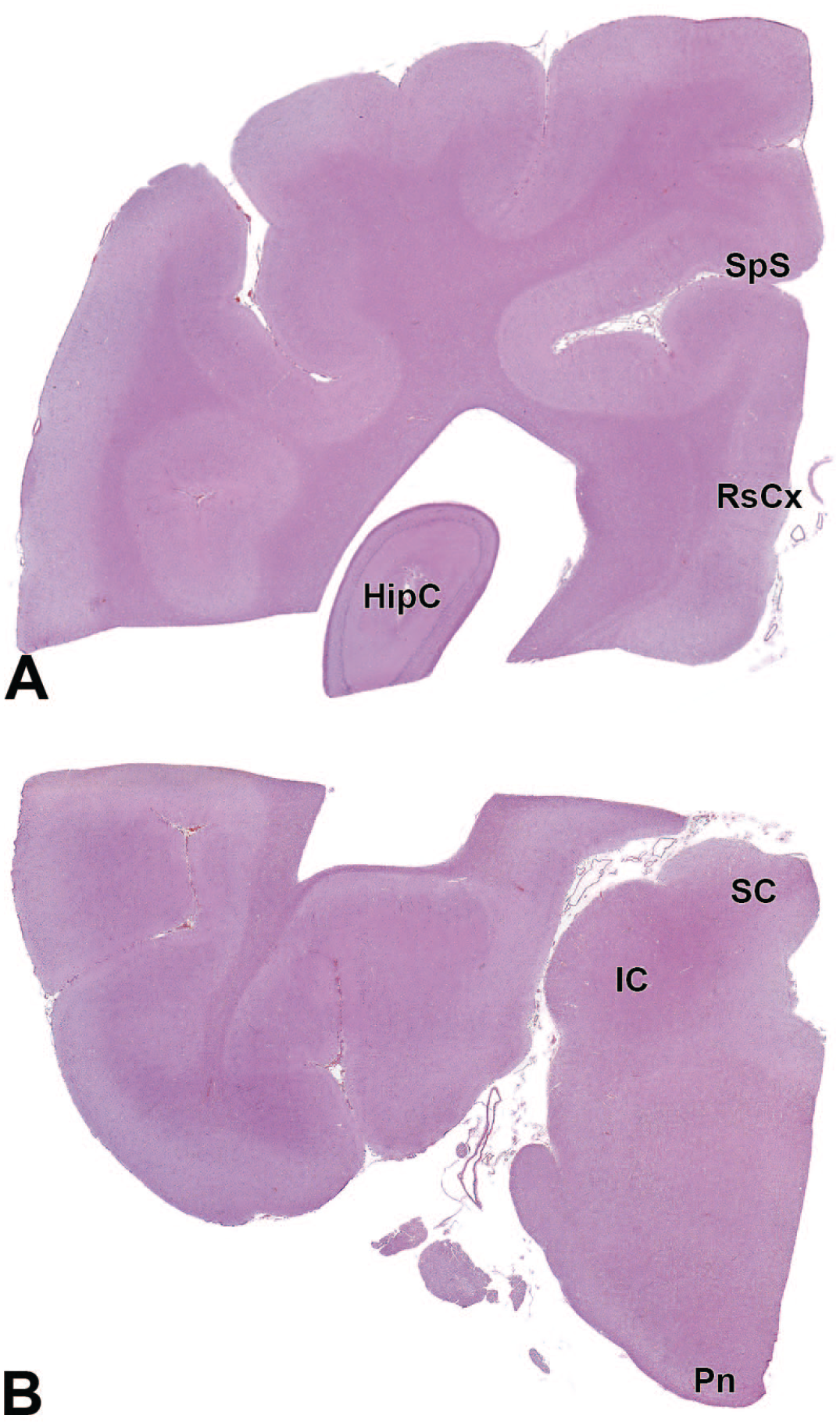
HipC: Hippocampus
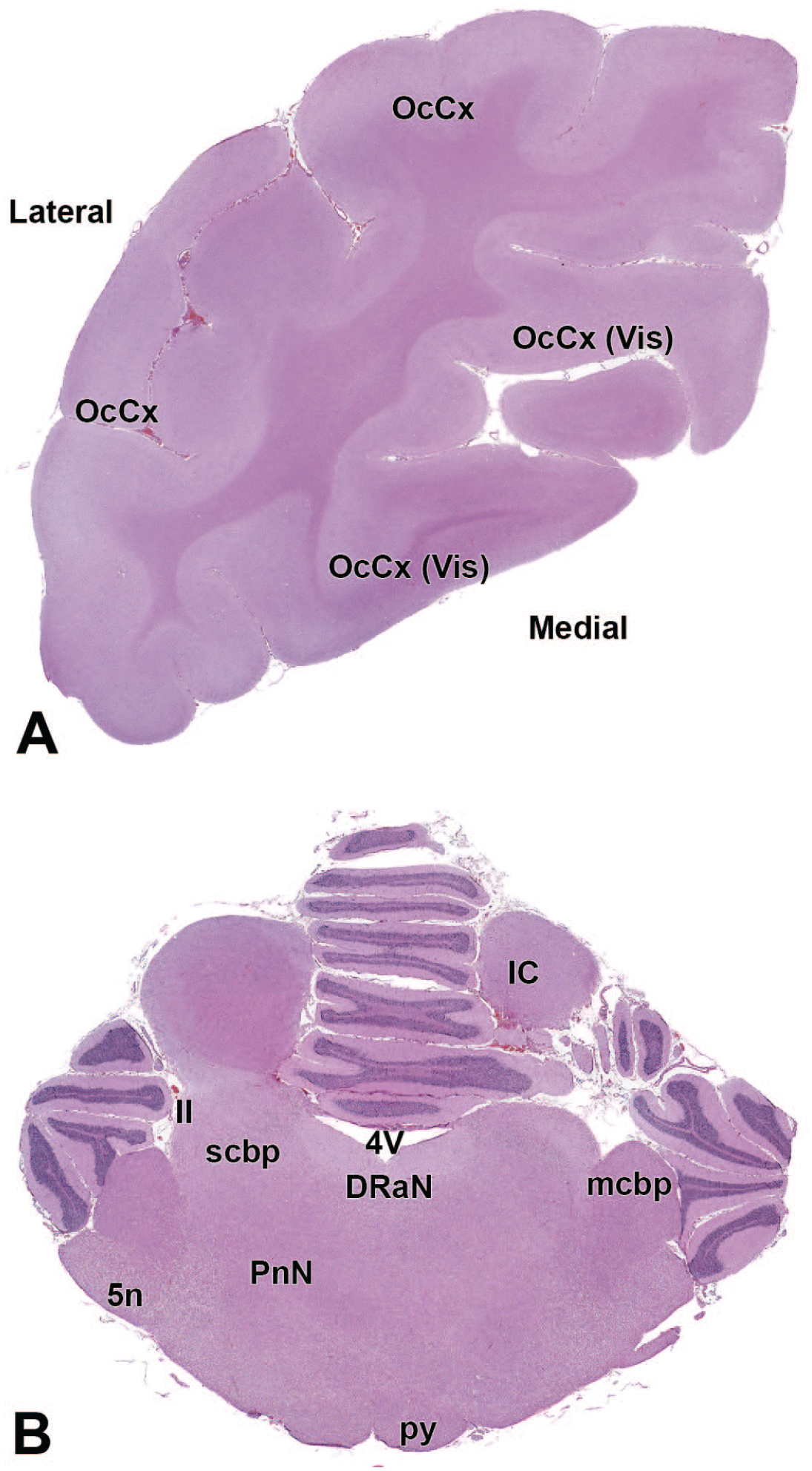
4V: Fourth ventricle
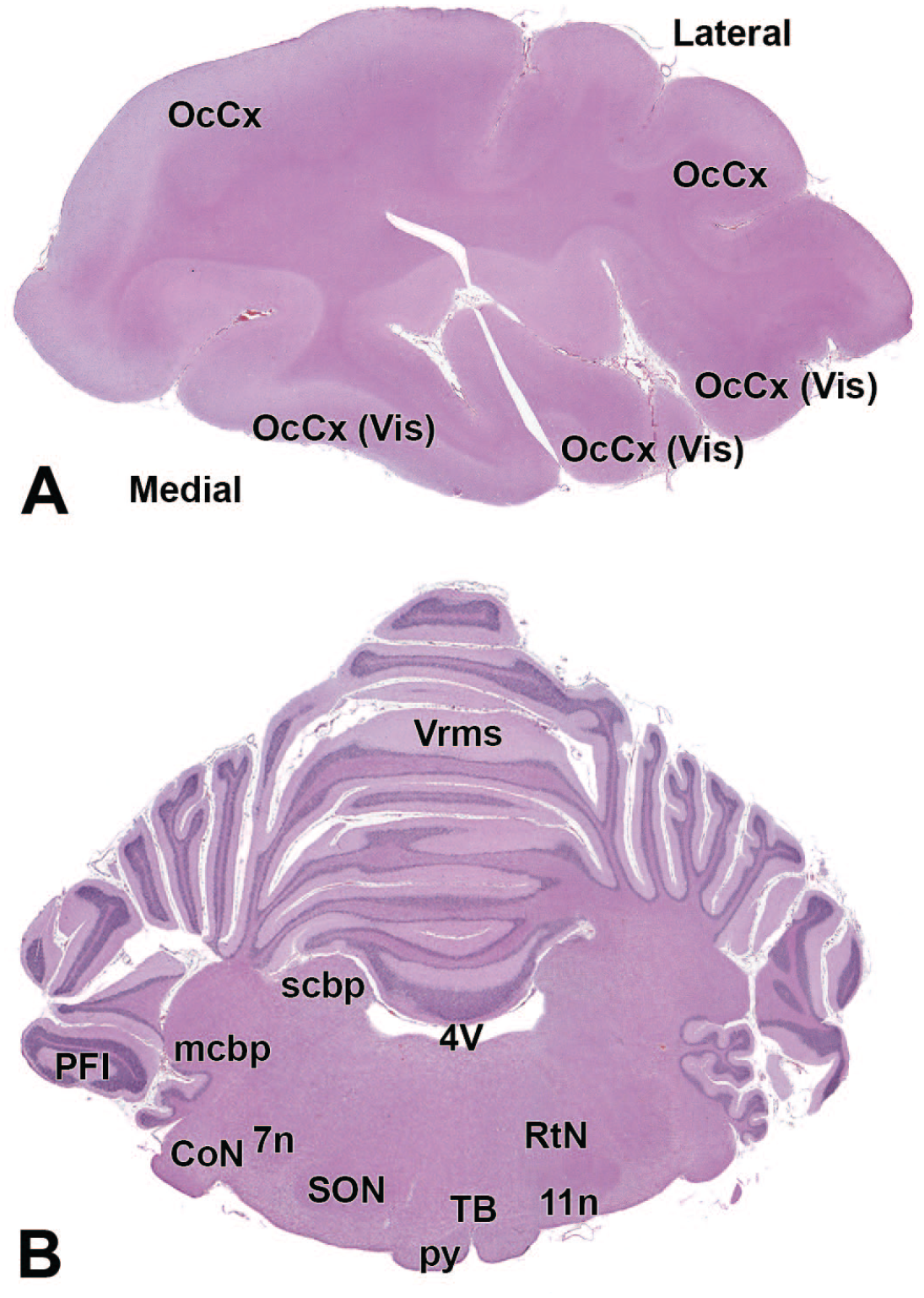
11n: Accessory nerve
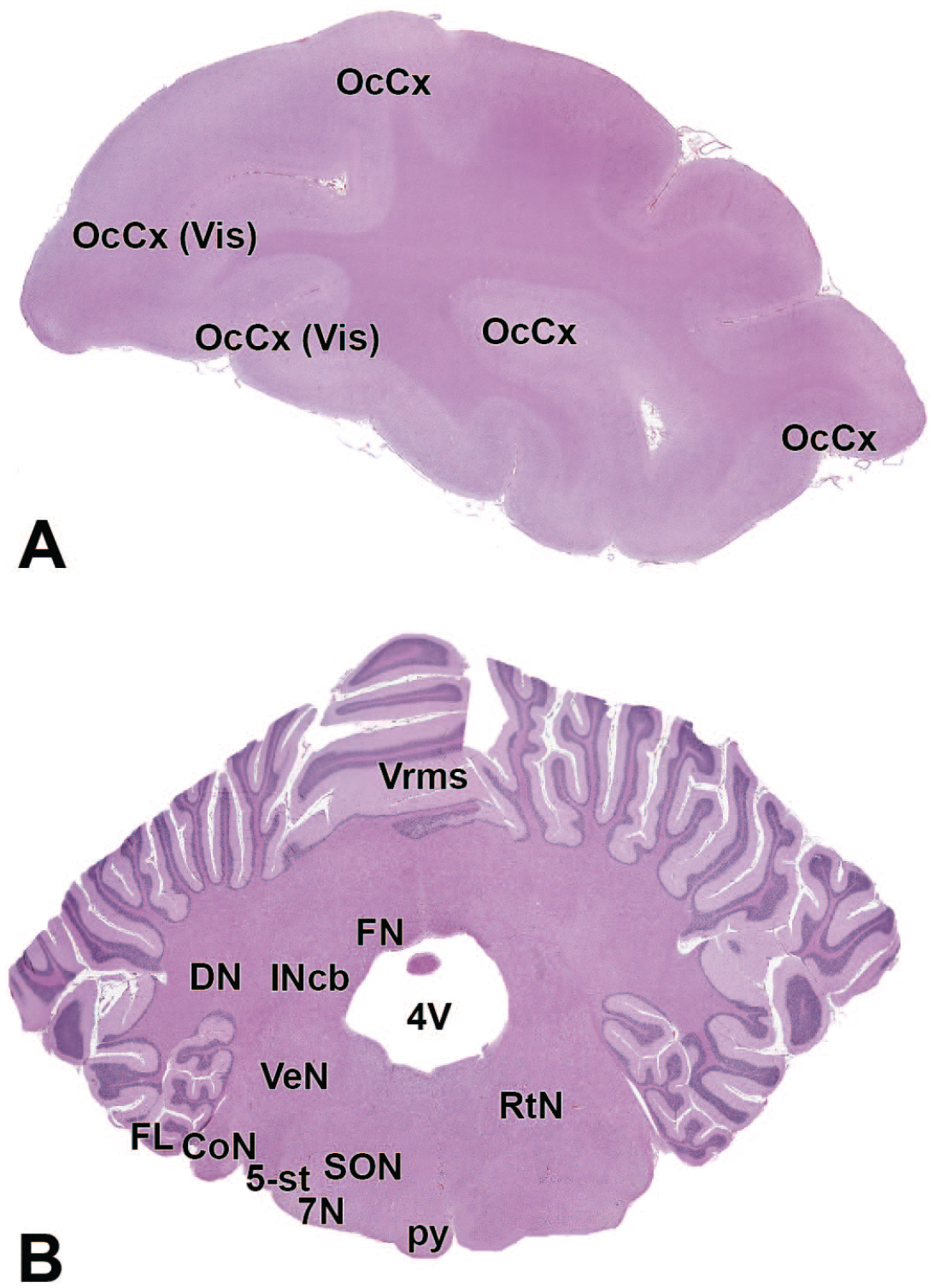
4V: Fourth ventricle
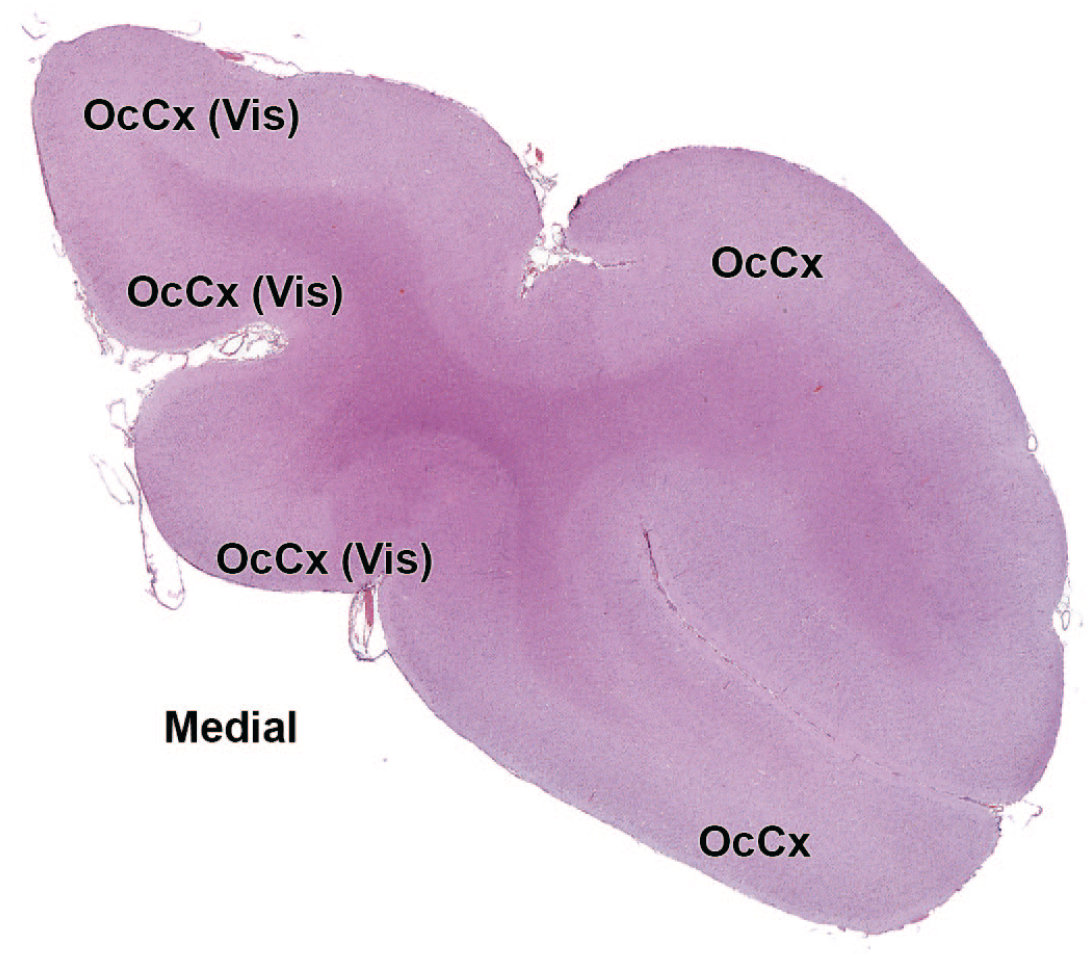
OcCx: Occipital cortex
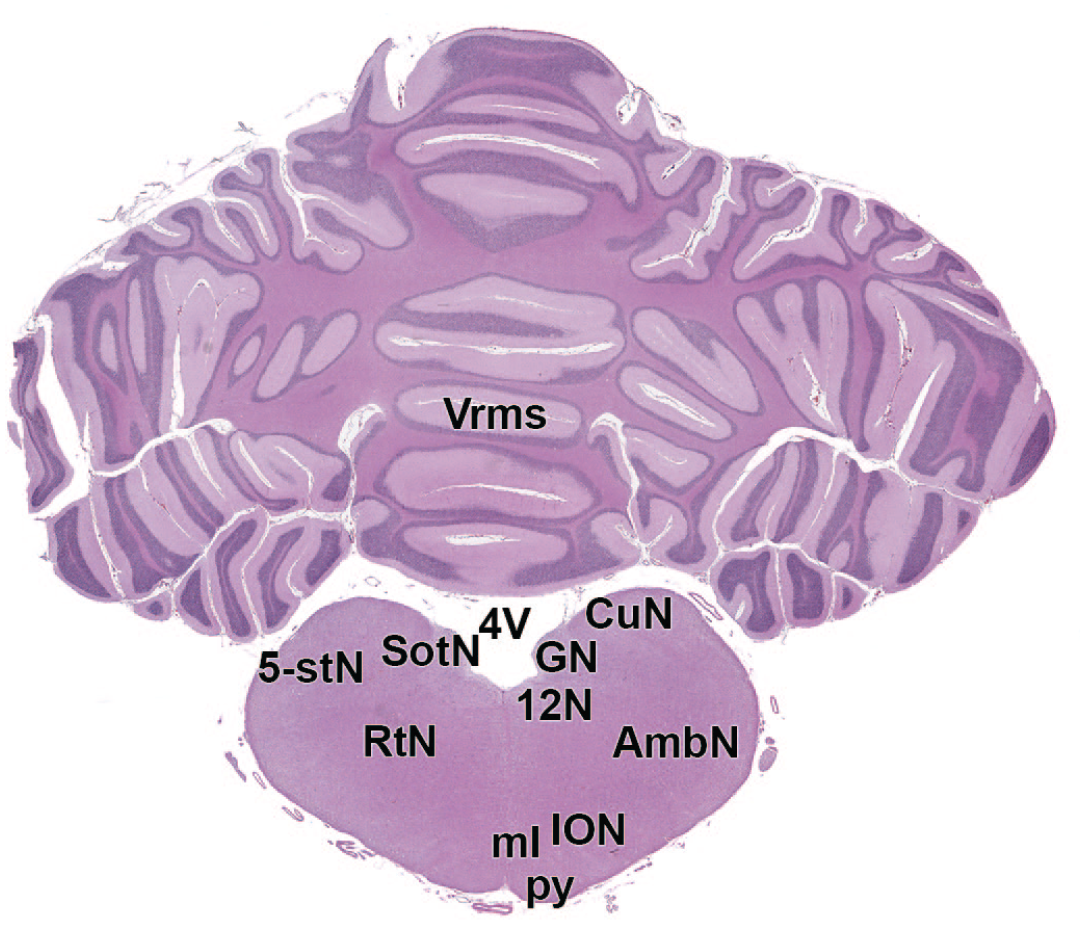
12N: Hypoglossus nucleus
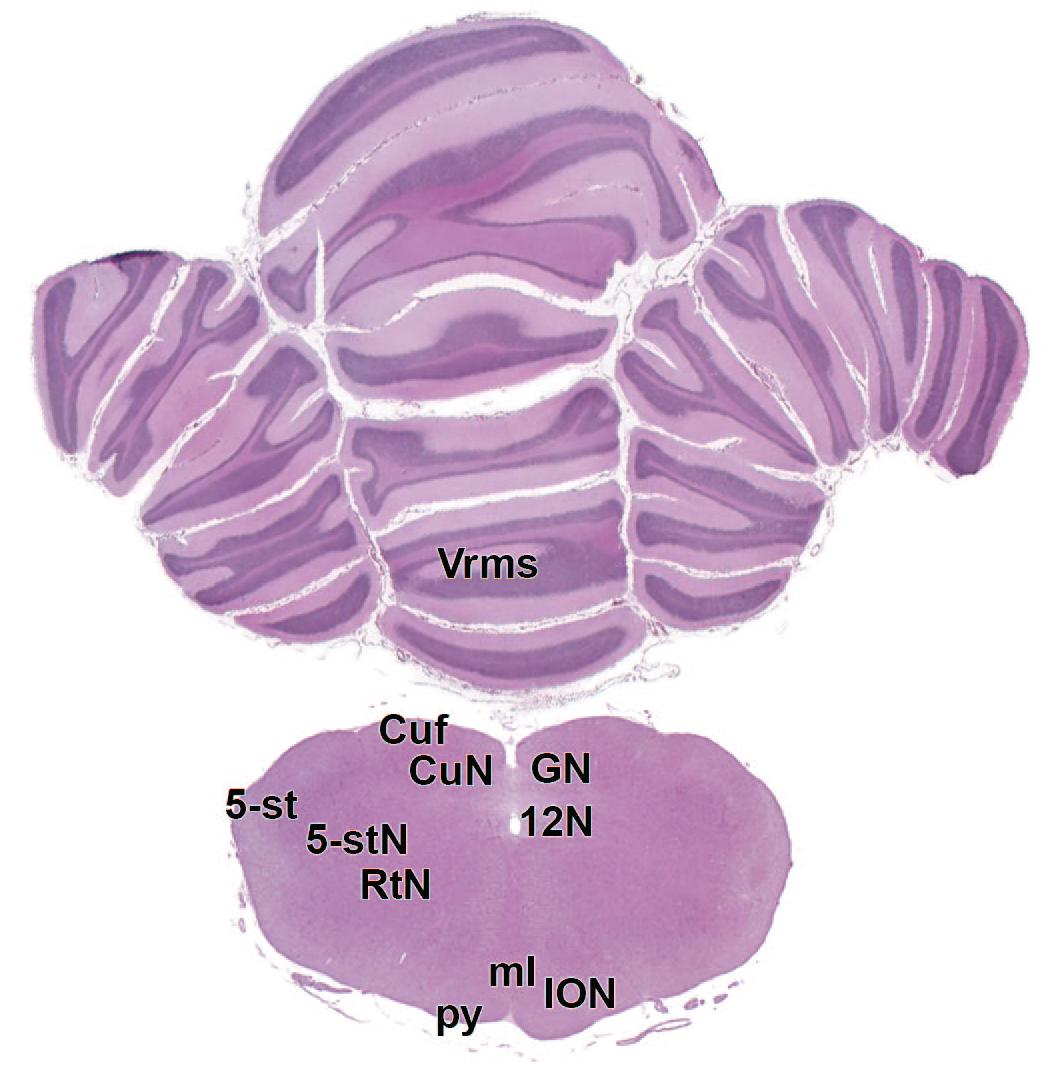
12N: Hypoglossus nucleus
Other neural organs: Spinal cord, ganglia, and nerves
The STP “best practice” recommendations for sampling, processing, and evaluation of the spinal cord5,6 and the PNS 6 depend on the expected pattern (or lack thereof) of nervous system findings. In general, neural structures outside the brain should be collected broadly and, if paired, bilaterally. The reason for this approach is that any tissues not obtained at necropsy will be lost permanently.
Spinal Cord
General descriptive anatomy of the Beagle dog spinal cord
The spinal cord of the Beagle dog has a long cylindrical shape and is composed of 8 cervical, 13 thoracic, 7 lumbar, 3 sacral, and 5 caudal segments. Each spinal cord segment is named based on the corresponding vertebra except that there is an extra cervical segment 8 (but only 7 cervical vertebrae), and there are only 5 caudal segments (but 6 caudal vertebrae). 30 The cervical and lumbar enlargements that give origin to the nerves that innervate the thoracic limb (brachial plexus) and pelvic limb (femoral and sciatic nerves) are located at the levels of C6 to T1 (especially C6–C7) and L4 to L7 spinal cord segments, respectively.
Collection, sampling, and sectioning of the Beagle dog spinal cord
The STP “best practices” recommend collection of 3 spinal cord divisions (cervical [C], thoracic [T], and lumbar [L]) for both general toxicity and neurotoxicity studies (Figure 26). To facilitate adequate fixation, spinal cord in nonrodents typically is removed from the vertebral column at necropsy. The dorsal and transverse vertebral processes and the vertebral arch are removed using medium-sized rongeurs or an autopsy saw. Subsequently, the exposed dura mater is grasped with forceps to elevate and remove the cord without touching or traumatizing (i.e., pulling or stretching) the delicate neural tissue. Some pathologists may choose to extract short segments of vertebral column (spanning 1-3 vertebral bodies) and then immerse the entire block in fixative, but this approach may lead to delayed fixation of the spinal cord at the center of the block due to slow penetration of fixative through the bone.
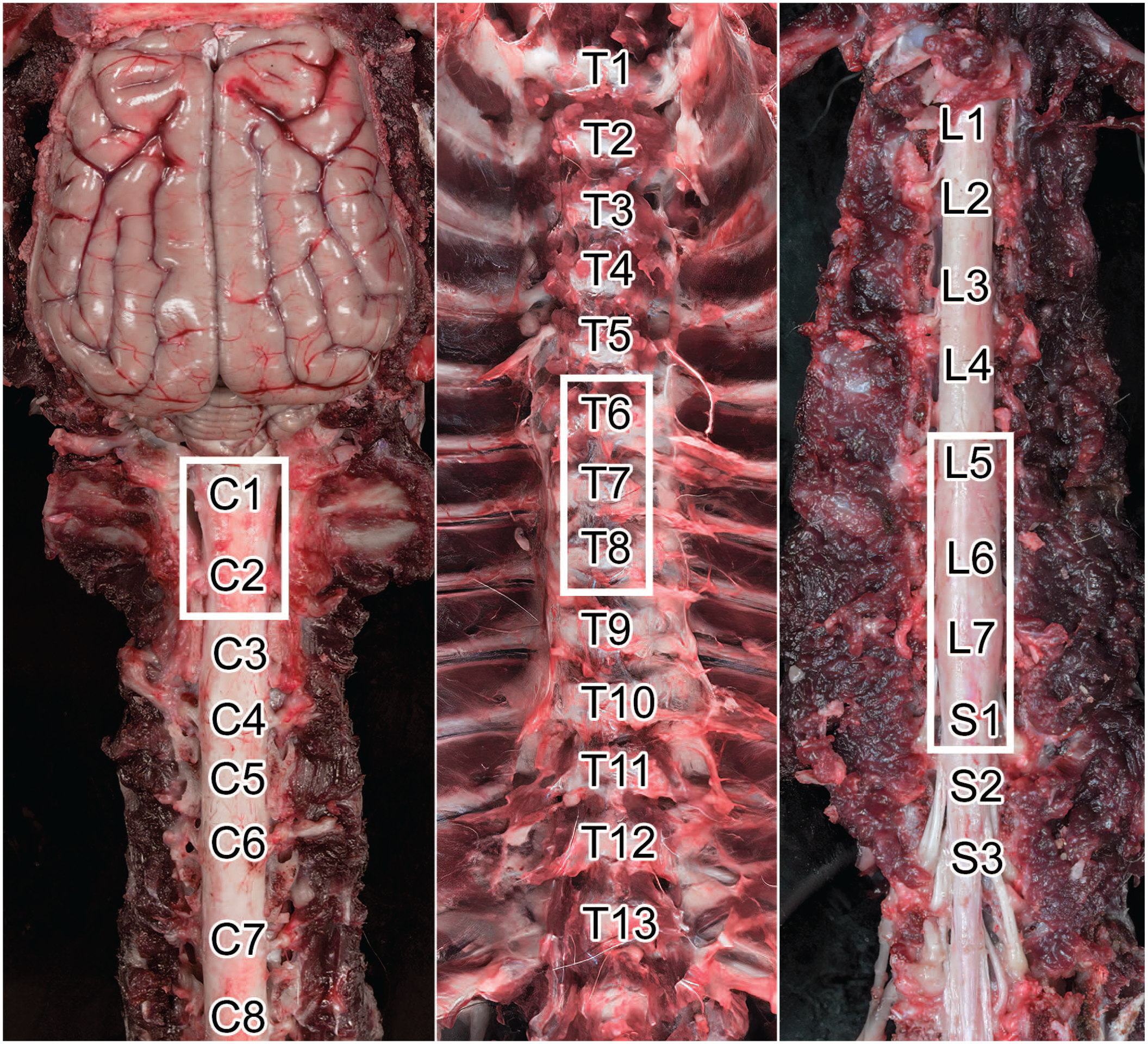
Anatomy and sampling approach to the Beagle dog spinal cord. Montage view of the cervical (C, left), thoracic (T, middle), and lumbosacral (L and S, respectively, right) regions in situ showing the locations of individual spinal cord segments. The white boxes denote typical sites for spinal cord sampling in the Beagle dog.
Specific spinal cord segments are harvested based on known structure–function correlates. A typical collection protocol for nonclinical toxicity studies should sample 3 spinal cord segments:
C1-C2—these segments have the largest diameter and contain the largest number of nerve fibers, thereby offering the best assessment of nerve fiber integrity in white matter tracts and providing an area of choice to assess “dying-back” sensory neuropathies.
T6-T8—these segments contain autonomic nervous system (ANS) neurons in the gray matter lateral columns.
L4-L5—these segments harbor neurons that supply nerve fibers to the hind limbs.
Importantly, lumbar cord segments L4 to L5 in the dog will be located within vertebrae L3 to L4. 30 Sacral (S) spinal cord or other functionally important segments (e.g., C6-C7 to capture the region with neurons supplying the forelimbs) may be added based on specific study objectives. Certain test articles induce lesions within the caudal spinal cord, suggesting that sections of the caudal lumbar and sacral spinal cord should be evaluated.31,32
At minimum, a transverse section of each spinal cord segment should be processed and examined. In general, however, more tissue should be evaluated. STP “best practices” recommend that a longitudinal or oblique section be examined for each segment as well (Figure 27),5,33 although analysis of multiple transverse sections per segment (taken at intervals and not as serial sections) is preferred by some pathologists.
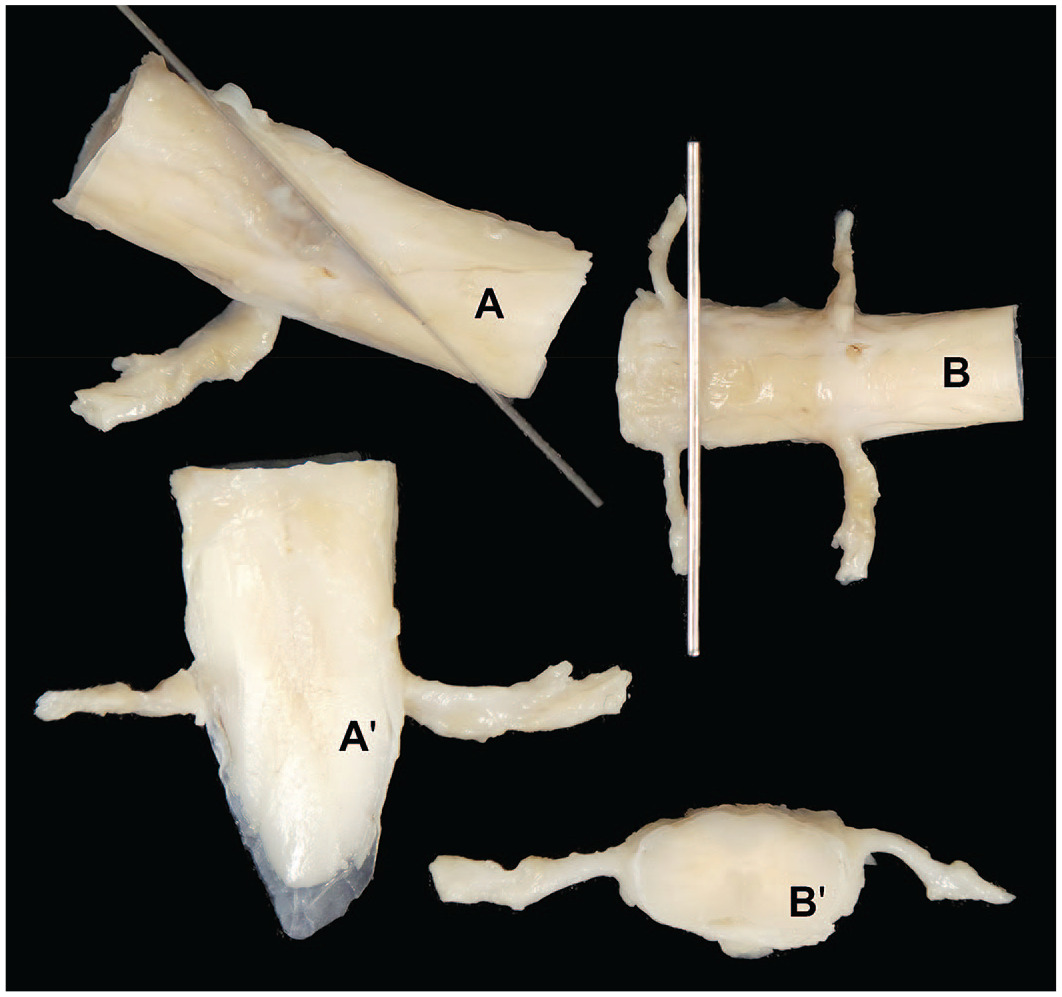
Postfixation trimming of the Beagle dog spinal cord. Montage view showing positioning of blades in trimming for a longitudinal oblique (upper left, A) and transverse (upper right, B) plane with resulting appearance of the trimmed oblique (bottom left, A’) and transverse (bottom right, B’) sections. Note the prominent spinal nerve roots for both trimming planes.
Ganglia and Nerves
Organization of the Beagle dog PNS
The PNS of the Beagle dog, like any other mammal, contains nerves and ganglia located outside the brain and spinal cord. The PNS is further divided into somatic and autonomic systems. The somatic system comprises all segmental nerves of intervertebral origin (i.e., spinal nerves) as well as 11 of 12 pairs of CNs; CN II (optic nerve) and the retina are actually components of the CNS. 34 In toxicity studies, somatic nerves originating from the brachial plexus (supplying the thoracic limb) and lumbosacral plexus (which feeds the pelvic limb) are of interest because most are mixed nerves fulfilling both motor and sensory functions.30,35 The ANS is responsible for involuntary functions involved in both reactions to emergencies and sustaining normal activities (e.g., peristalsis and vasoconstriction). The ANS is divided into sympathetic and parasympathetic systems: the main structural difference between these divisions is that most sympathetic ganglia are located just outside of the spinal cord while parasympathetic ganglia are situated in close proximity or within the innervated organs. 36 The enteric nervous system is another quasi-autonomous ANS division that performs various parasympathetic-like functions. 37
Approaches to sampling ganglia
Sampling of ganglia in the PNS depends on the study objective. For most toxicity studies, only somatic ganglia are obtained purposely, while autonomic ganglia are assessed if they are present within or near protocol-specified nonneural tissues (e.g., the walls of hollow organs). 6 In this regard, typical somatic ganglia collected at necropsy are the dorsal root ganglia (DRG) and sometimes the trigeminal (or CN V) ganglia (TG)6,38 These somatic sensory ganglia usually are evaluated if there is a possibility that a test article might target their neurons.39,40 Parasympathetic and enteric ganglia are usually evaluated as they are encountered during the evaluation of nonnervous system tissues. Sympathetic ganglia typically are collected and evaluated if the test article or a related material has been shown to produce effects on autonomic function39-42 or if insufficient information exists to rule out an effect on the sympathetic nervous system.
Specific emphasis has been placed recently on sampling ganglia due to their sensitivity to certain classes of biomolecules (e.g. adeno-associated virus [AAV] 32 and antisense oligonucleotide [ASO] gene therapies) and small molecules (antineoplastic chemotherapies 43 ). Key ganglia to evaluate in this regard are DRG and TG. These ganglia contain cell bodies of sensory neurons, and their function is essential in maintaining sensory and reflex motor circuits.
General descriptive anatomy of somatic ganglia in the Beagle dog
Dorsal root ganglia can be harvested by performing a partial laminectomy and using a dissecting microscope and microdissection tools. Various DRG levels may be required depending on the product development program and are dictated by the study protocol. The vertebral column is opened dorsally with rongeurs or an autopsy saw to expose the spinal cord, after which the spinous process and roof of the vertebrae are removed. Rongeurs are used to further clip away any bone covering the spinal cord and DRG, after which the DRG are removed (with the spinal nerve roots attached) using nontoothed forceps without directly grasping the ganglia. The DRG appear as bulbous-shaped and slightly tanner bulges relative to the adjacent spinal nerve roots and may be collected either attached to or separate from the spinal cord (Figure 28).
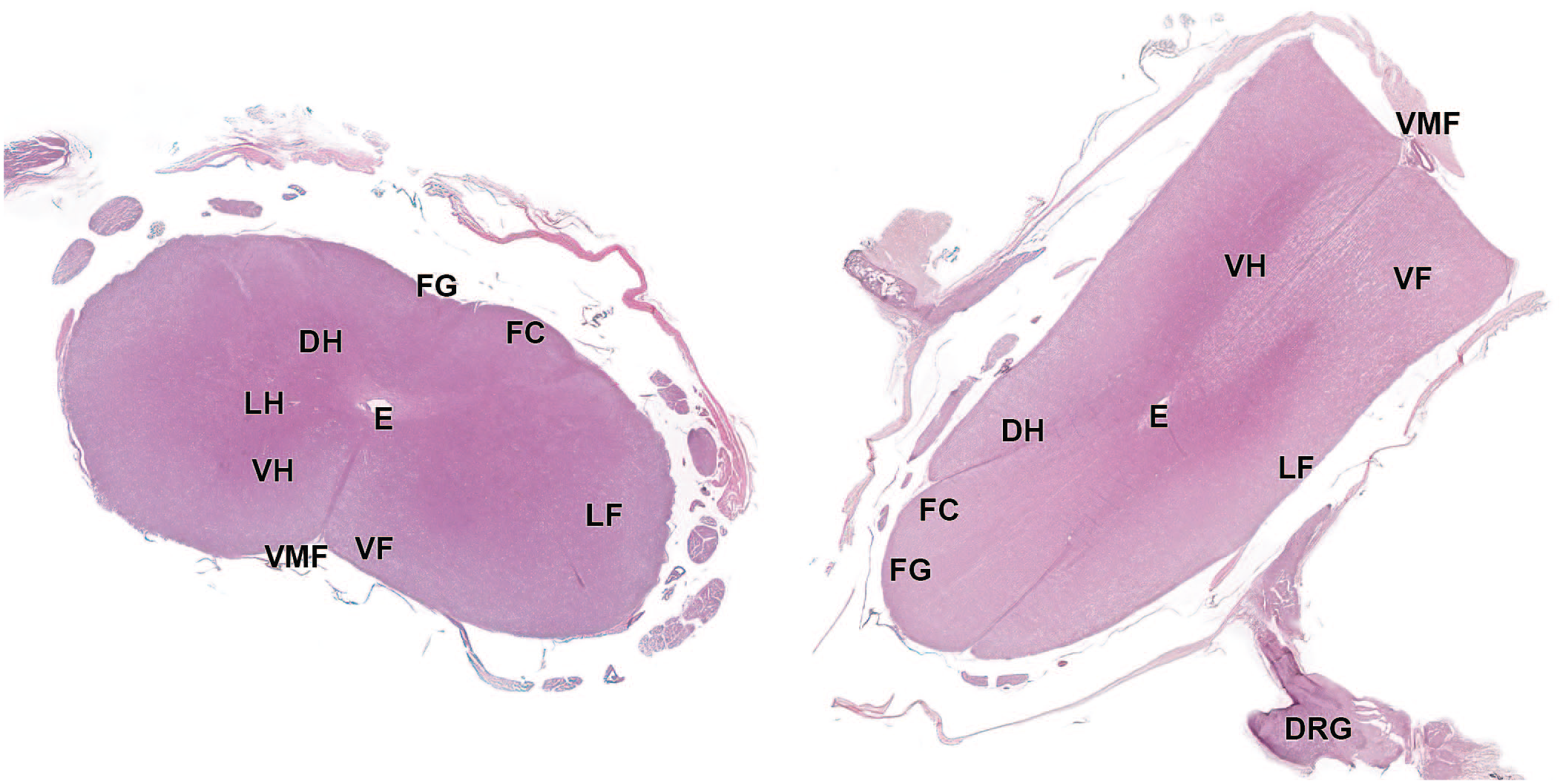
Photomicrograph (whole slide image) of the cervical spinal cord showing the corresponding microscopic appearance of the transverse (left) and longitudinal oblique (right) trimming planes. DH, dorsal horn; DRG, dorsal root ganglion; E, ependymal canal; FC, fasciculus cuneatus; FG, fasciculus gracilis; LF, lateral fasciculus; LH, lateral horn; VF, ventral fasciculus; VH, ventral horn; VMF, ventral median fissure.
The TG of nonrodent species are positioned on the base of the skull near the pituitary gland (Figure 29A and B). Approaches to sampling the TG in nonrodent species have been published by Howroyd. 38 Three trigeminal nerve (CN V) branches (ophthalmic, mandibular, and maxillary) innervate the face (Figure 29). After the brain has been removed, the TG may be located against the surface of the skull base diagonally and laterally from the pituitary gland and below the dura mater. Using rongeurs, the bony ridge of the adjacent petrous temporal bone is removed, and scissors are used to carefully remove the dura mater that covers the ganglia and associated nerve. Once exposed, the ophthalmic branch is located medially, the mandibular branch is seen laterally, and the maxillary branch is seen in the middle (Figure 29C). The tip of a #11 scalpel blade may be used to score along the outside edges of the nerve roots after which dissecting spring-scissors are employed to sever the 3 nerve branches as they enter foramina in the skull. The meninges are grasped with fine forceps, and the spring-scissors are used to carefully undermine the ganglia to free them from the skull surface (Figures 29A-C and 30).
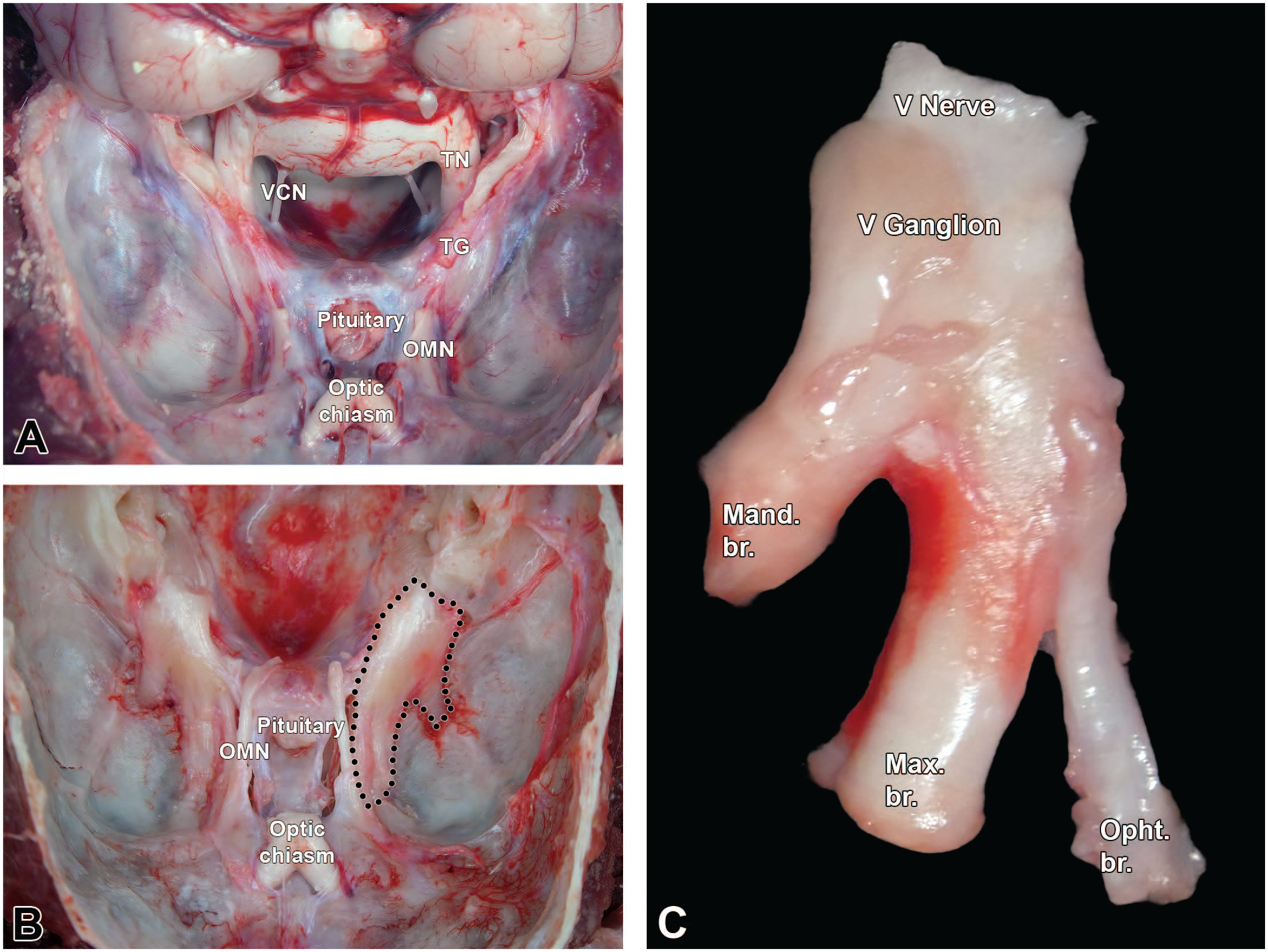
Trigeminal ganglia (somatic sensory) of a Beagle dog. Montage view showing (A) an unfixed Beagle dog skull (craniodorsal view). The brain is reflected caudally, exposing the trigeminal nerve (TN) and the trigeminal ganglion (TG). (B) Trigeminal ganglia in situ after removal of the brain and dissection of the meninges (with the left ganglion delimited by black dots). (C) Dissected right trigeminal ganglion containing the trigeminal nerve (cranial nerve V [V nerve]), the trigeminal ganglion (V ganglion) as well as the mandibular (Mand. br.), maxillary (Max. br.), and ophthalmic (Opht. br.) branches. OMN, oculomotor nerve; TG, trigeminal ganglion; TN, trigeminal nerve; VCN, vestibulocochlear nerve.
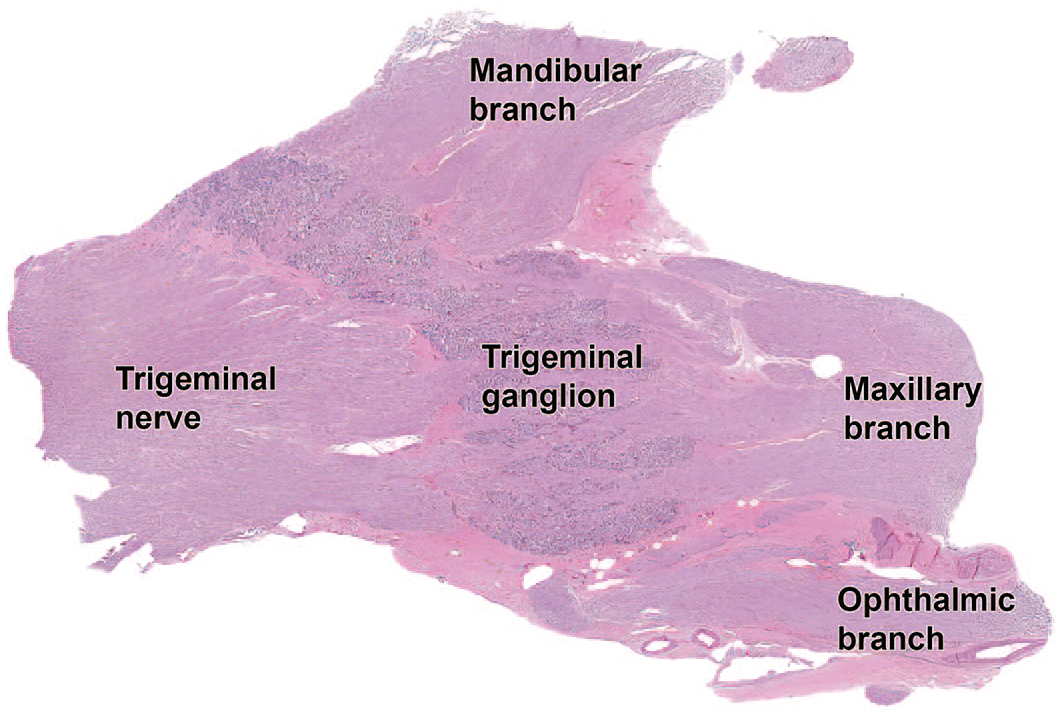
Low magnification photomicrograph of the trigeminal (somatic sensory) ganglion of a Beagle dog. Note that the ganglion is intermingled with nerve fibers of the trigeminal nerve, similar to the caudal (“nodose”) ganglion (somatic sensory) of the vagus nerve (cranial nerve X) of Figure 38. The trifurcation into ophthalmic, maxillary, and mandibular branches shown macroscopically in Figure 29 can be readily identified at this magnification.
Sampling somatic ganglia in the Beagle dog
For general toxicity studies, at least one caudal lumbar DRG should be collected and preserved in NBF to be processed later (if needed) for light microscopic examination. 6 In practical terms, DRG sampling often occurs at sites near the processed spinal cord segments (C1-C2, T6-T8, and L4-L5). For the caudal lumbar and sacral regions, technical staff may benefit from regular reminders that the DRG of a particular spinal cord segment may reside one or more intervertebral spaces caudal to the location of the actual spinal cord segment. These DRG generally should be procured even if no neurological signs are seen during in-life. In the absence of neurological signs, DRG may be left in situ (i.e., in their intervertebral locations) for fixation and archiving, and then removed and processed only if a decision is made to evaluate their microscopic structure. If neurological signs are observed during in-life, a minimum of 2 DRG each should be obtained for the cervical, thoracic, and lumbar divisions; collection of 2 or more sacral DRG is recommended for toxicity studies if the test article is a member of a therapeutic class known to target DRG neurons or if the test article is delivered directly into the neuroaxis (e.g., via intracerebroventricular, intracisternal, or intrathecal injection or infusion). 6 A TG should be collected (unilaterally is acceptable) and retained in fixative.
For dedicated neurotoxicity studies, 2 (or ideally more) DRG per spinal cord division as well as both TG should be sampled, and autonomic ganglia should be considered for collection. 6 The DRG are collected as noted above. Trigeminal ganglia and autonomic ganglia (e.g., cranial [superior] cervical, cervicothoracic, and cranial mesenteric) may be gathered using published procedures. 44
Approaches to sampling nerves
Collection of nerves from dogs depends primarily on the study objectives and secondarily on regulatory guidelines. For most general toxicity and neurotoxicity studies, only somatic nerves are sampled specifically. Autonomic nerves may be evaluated in routinely evaluated tissues in association with parasympathetic and enteric ganglia; autonomic nerves at these sites are not typically discernible without special staining. Alternatively, autonomic nerves may be observed away from organs in conjunction with sympathetic ganglia. In practice, autonomic nerves are usually not evaluated specifically unless autonomic signs are seen in-life or prior experience with a test article warrants their evaluation. Multiple large somatic nerve trunks offering ample tissue for microscopic evaluation may be accessed readily in dog limbs (Figures 31-37). Once removed, nerves are applied to rigid paper (index cards) or similar material to facilitate fixation of nerve fibers in a straight line. If desired, the orientation of the nerve (e.g., proximal and distal ends) and laterality may be noted simply by written notes on the card stock or using tissue marking dyes. Unfixed nerves should be handled with care to avoid trauma-related artifacts (e.g., crushing, pulling, and stretching). 31
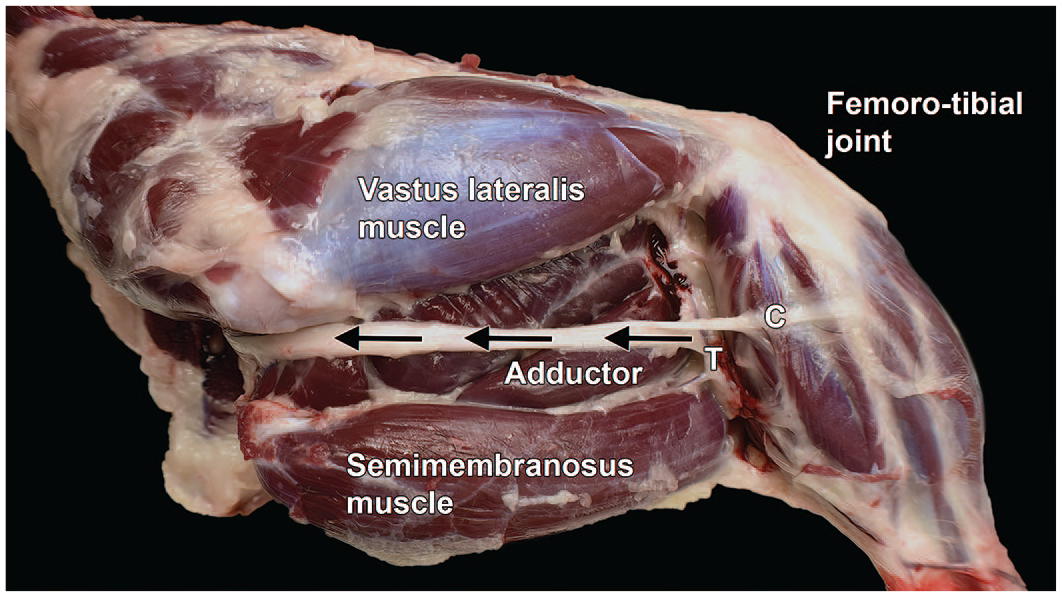
Dissection of the sciatic nerve from the right hind limb of a Beagle dog (lateral view). All skeletal muscles of the pelvic limb except the adductor muscles and cranial muscles of the thigh are innervated by the sciatic nerve (highlighted by arrows, with the arrowheads pointing proximally). After sending branches to the biceps femoris, semimembranosus, and semitendinosus muscles (collectively the “hamstring”), the sciatic nerve divides into 2 branches: the tibial nerve (T) to caudal muscles and the common fibular nerve (C) to cranial muscles. Femorotibial joint = “knee.”
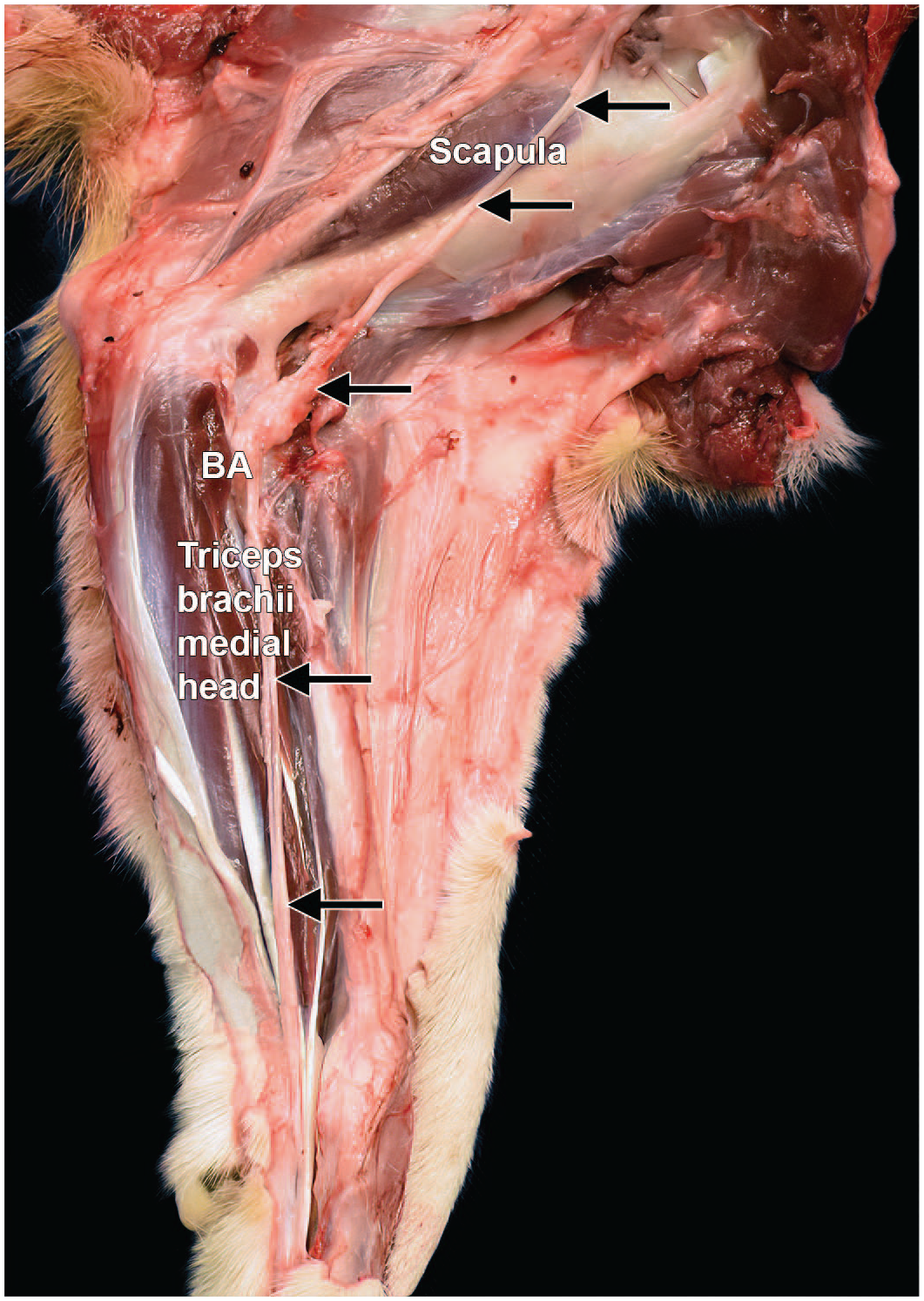
Dissection of the median nerve from the right forelimb of a Beagle dog (medial view). The median nerve (arrows) in the forearm lies caudal to the brachial artery (BA) and vein and is cranial in relation to the ulnar nerve. It crosses the flexor surface of the humeroradioulnar (“elbow”) joint cranial to the medial epicondyle of the humerus and gives muscular branches to the pronator and flexor muscles of the forelimb.
Dissection of the radial nerve from the left forelimb of a Beagle dog (lateral view). At the level of the lateral head of the triceps brachii muscle, the radial nerve sends a branch to the triceps and shortly thereafter bifurcates into deep and superficial branches. The deep branch supplies all the extensor muscles of the carpus and the digits while the superficial branch innervates the skin on the cranial surface of the antebrachium (“forearm”). “Elbow” = humeroradioulnar joint.
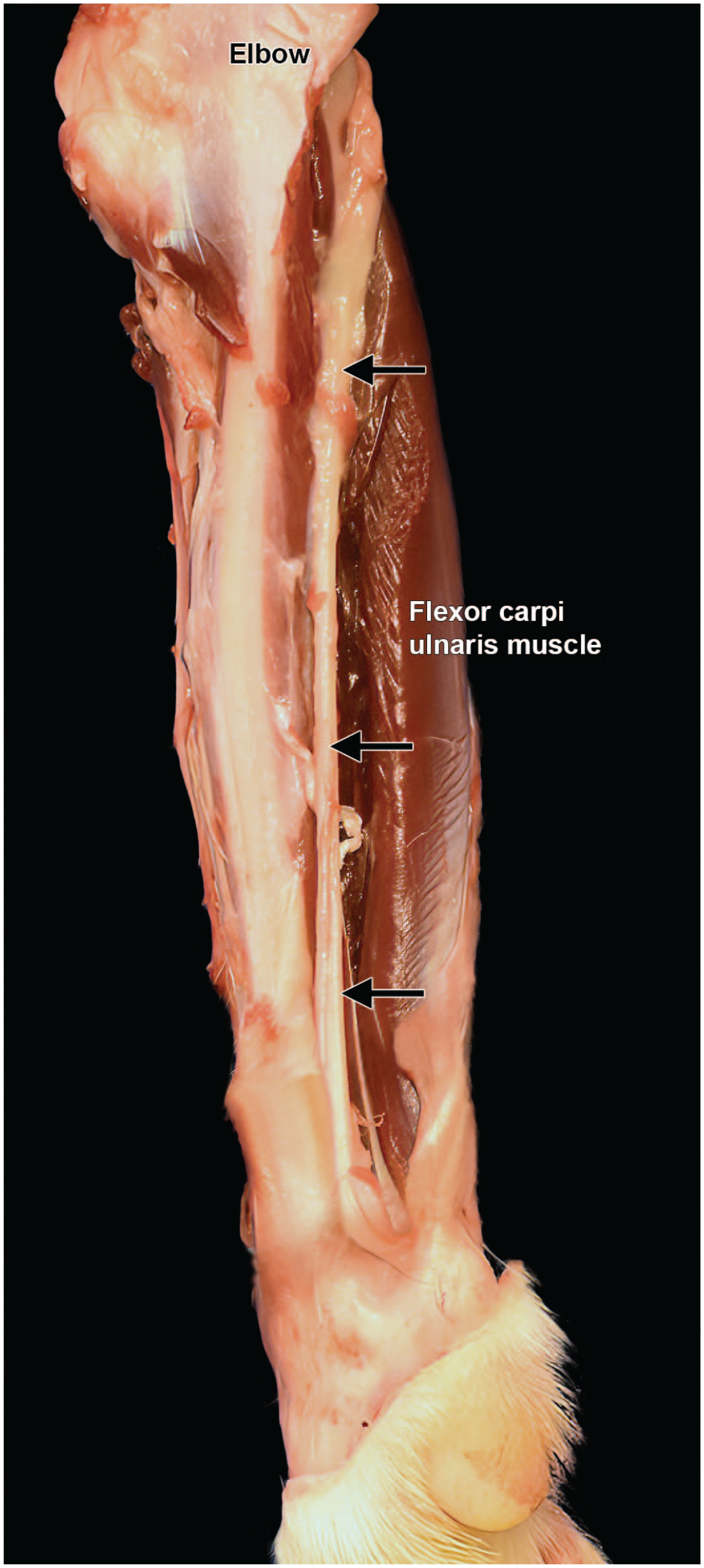
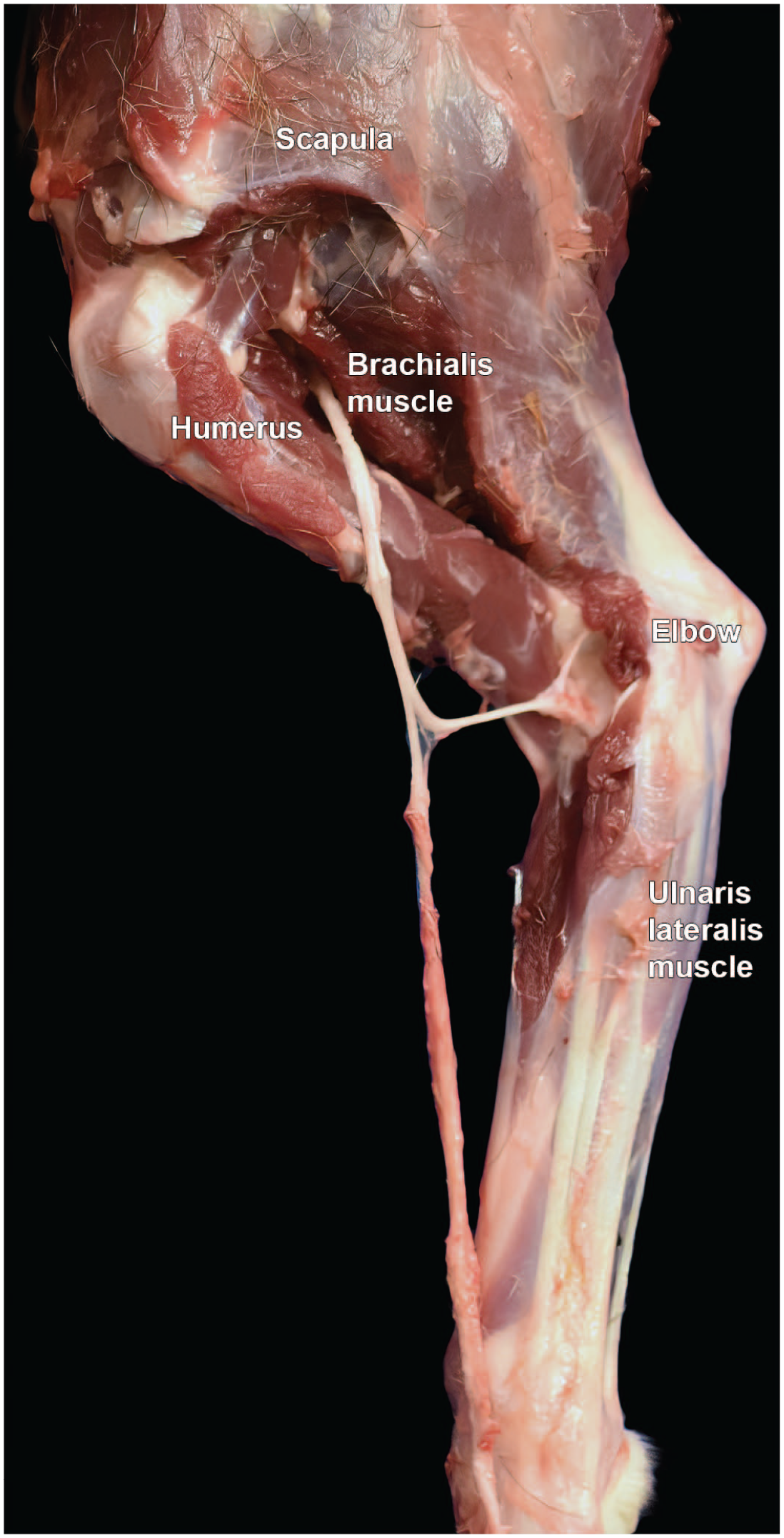
Dissection of the ulnar nerve from the left forelimb of a Beagle dog (medial view). Like the median nerve (Figure 32), the ulnar nerve in the antebrachium (“forearm”) lies caudal to the brachial artery and vein and supplies muscles on the caudomedial side of the forearm. “Elbow” = humeroradioulnar joint.
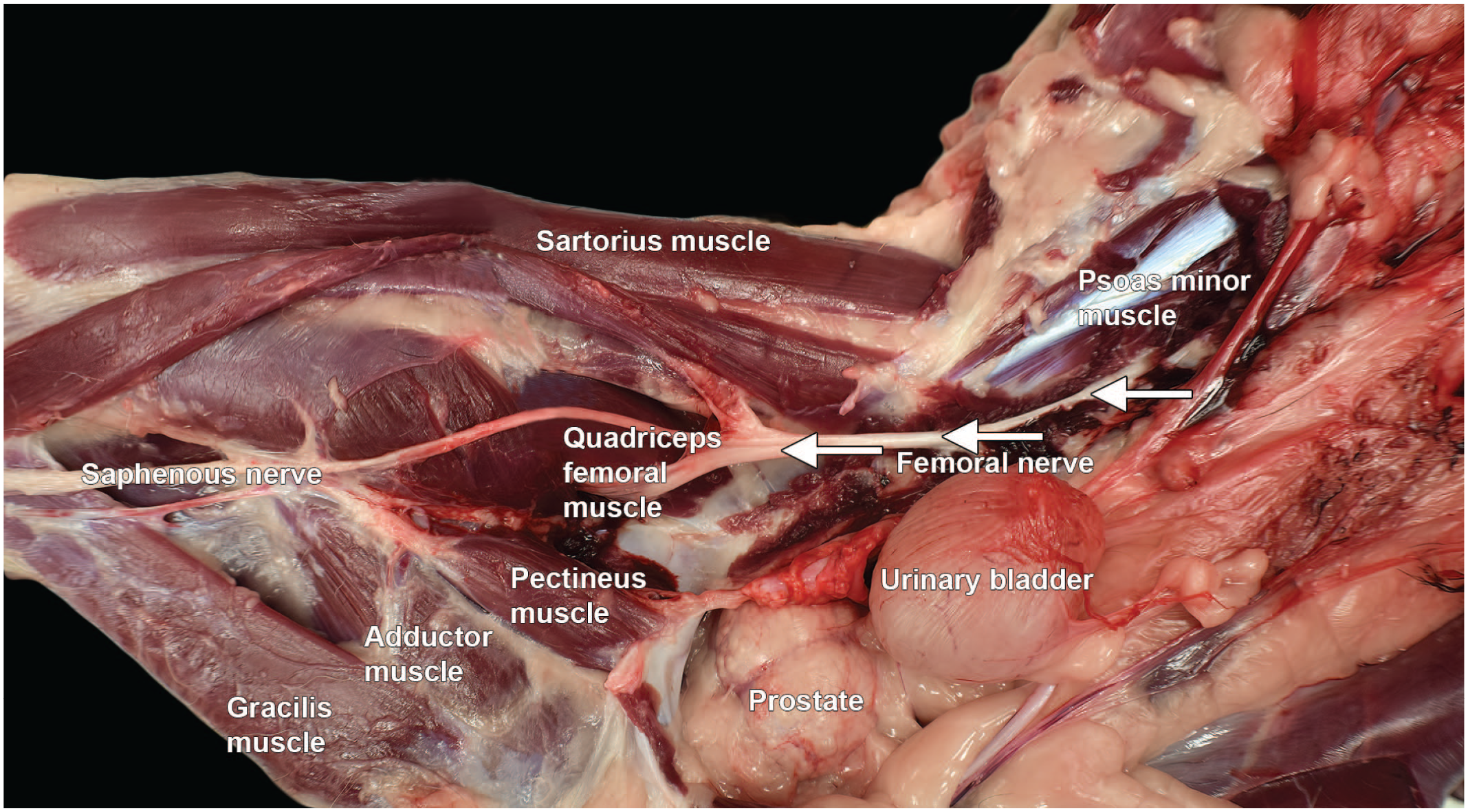
Dissection of the femoral nerve in the abdomen and right hind limb of a Beagle dog (dorso-medial view). The femoral nerve (arrows) innervates the cranial muscles of the thigh, while all remaining muscles of the limb are innervated by the sciatic nerve.
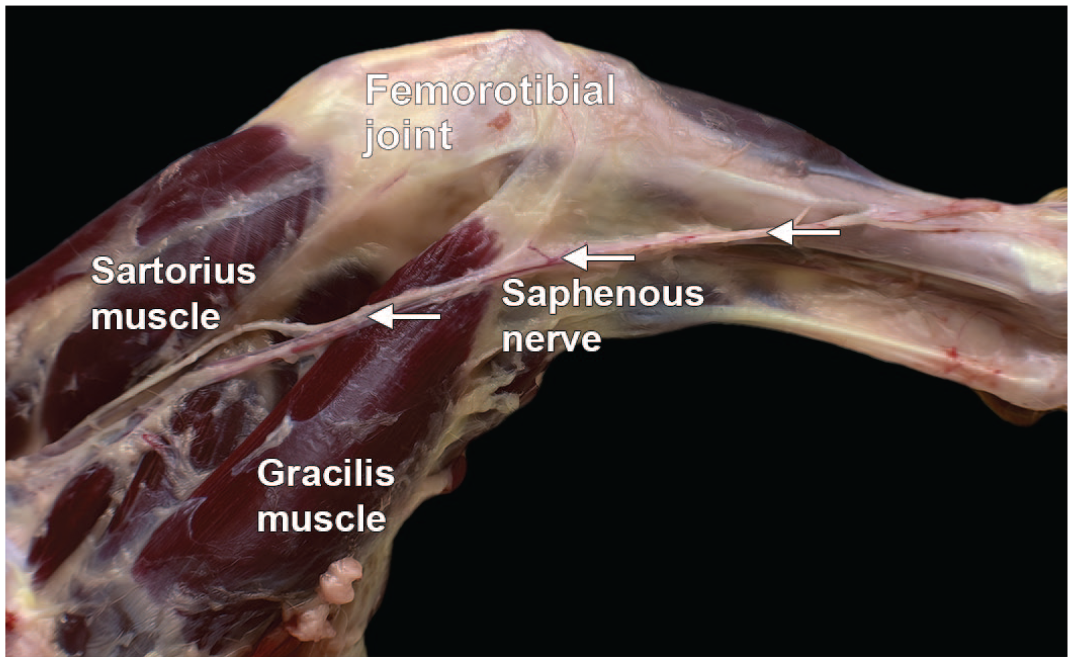
Dissection of the saphenous nerve from the right hind limb of a Beagle dog (lateral view). The saphenous nerve (highlighted by arrows with the arrowheads pointing proximally) is the continuation of the femoral nerve. It is a pure sensory nerve that descends into the quadriceps muscle. Below the femorotibial joint (“knee”), the branches of the saphenous nerve are distributed to the skin of the front and medial side of the leg.
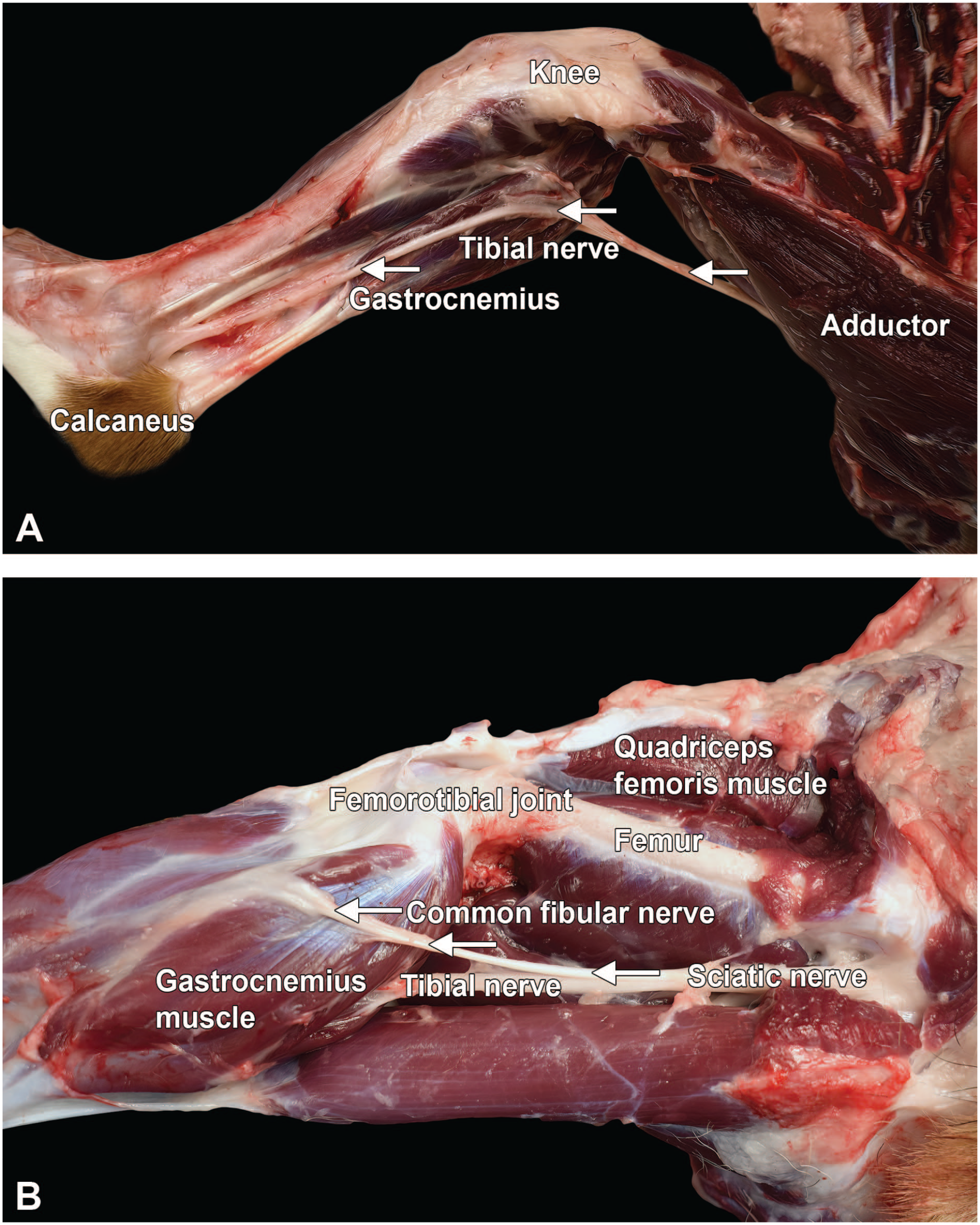
Dissection of distal nerves from the left hind limb of a Beagle dog: tibial (A, arrows) and common fibular (B, arrows). The tibial nerve is the larger terminal branch of the sciatic nerve, providing branches for the extensors of the tibiotarsal (“hock” or “ankle”) joint and flexors of the digits. The common fibular nerve shows a more lateral position than the tibial nerve in the lower hind limb and provides further branches below the femorotibial (“stifle” or “knee”) joint.
General descriptive anatomy of nerves in the Beagle dog
A number of somatic nerves may be sampled in dogs (Diagrams D1 and D2).6,35 The usual choice in the hind limb is the sciatic nerve (Figure 31). This trunk originates from neurons in spinal cord segments L4 and L5. The tibial nerve is a branch of the sciatic nerve that is supplied by neurons from spinal cord L7 to S1. This nerve is located by placing the animal in dorsal recumbency, reflecting the skeletal muscle from the medial aspect of the thigh to expose the sciatic nerve, and then removing the medial gastrocnemius muscle to find the tibial nerve (Figure 37A). The common fibular (or “common peroneal”) nerve arises from neurons in spinal cords L6 to L7 and is distinguished from the tibial nerve by its smaller size and more lateral position on the lower hind limb (Figure 37B). The common fibular nerve gives off superficial and deep branches just distal to the femorotibial (“stifle” or “knee”) joint. Some pathologists might prefer to sample and evaluate the femoral nerve (Figure 35), which arises from L4 to L6 and passes through the iliopsoas muscle to innervate all 4 heads of the quadriceps femoris muscle. The saphenous nerve is a sensory-predominant trunk in dogs that arises from the femoral nerve (Figures 35 and 36). The plantar nerve is another sensory-predominant trunk in dogs that arises from the distal end of the tibial nerve near the calcaneus. The medial branch of the plantar nerve supplies digits I, II, and III while the lateral branch supplies digits IV and V.
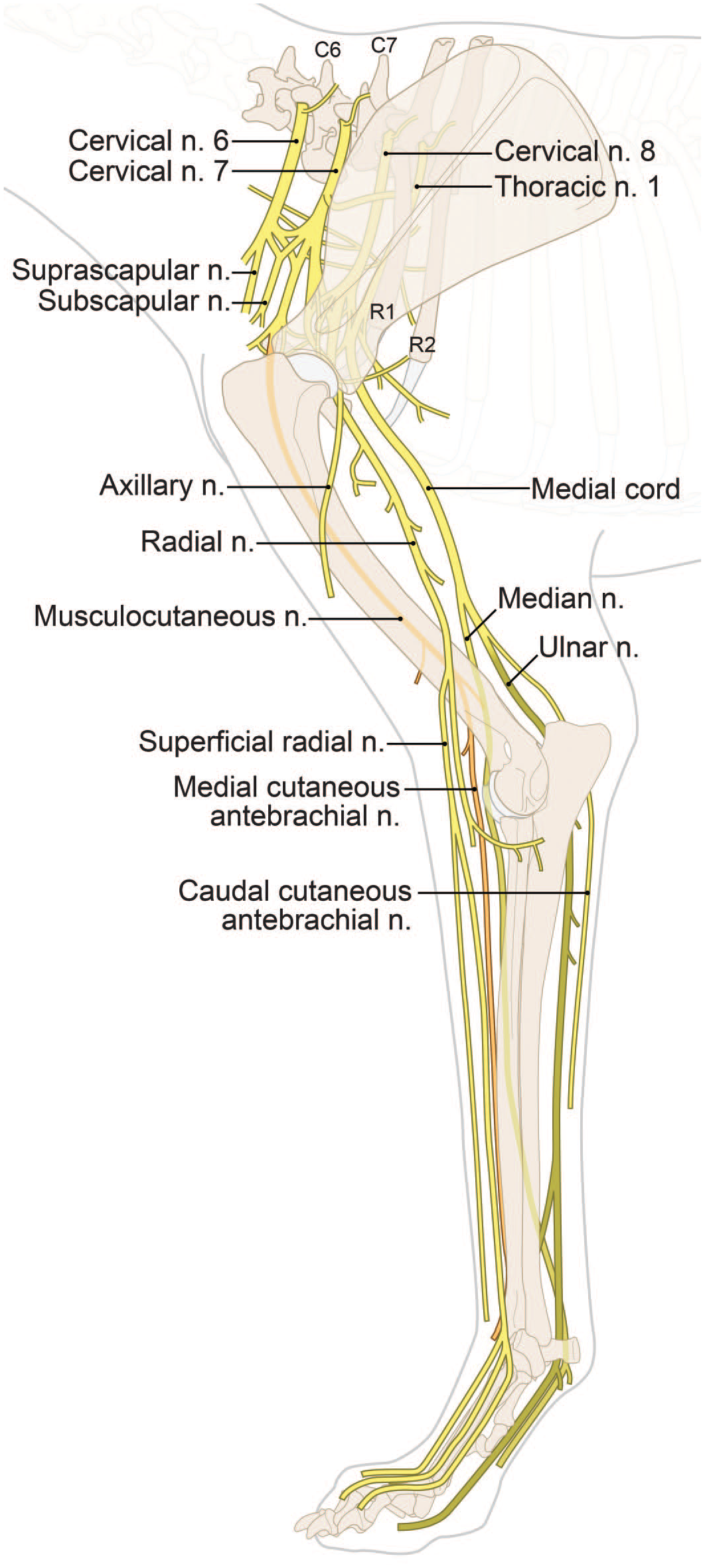
Anatomy of principal somatic nerves (n.) for the dog forelimb. [Diagram © 2022 by Mr. Tim Vojt and provided by courtesy of the author.]
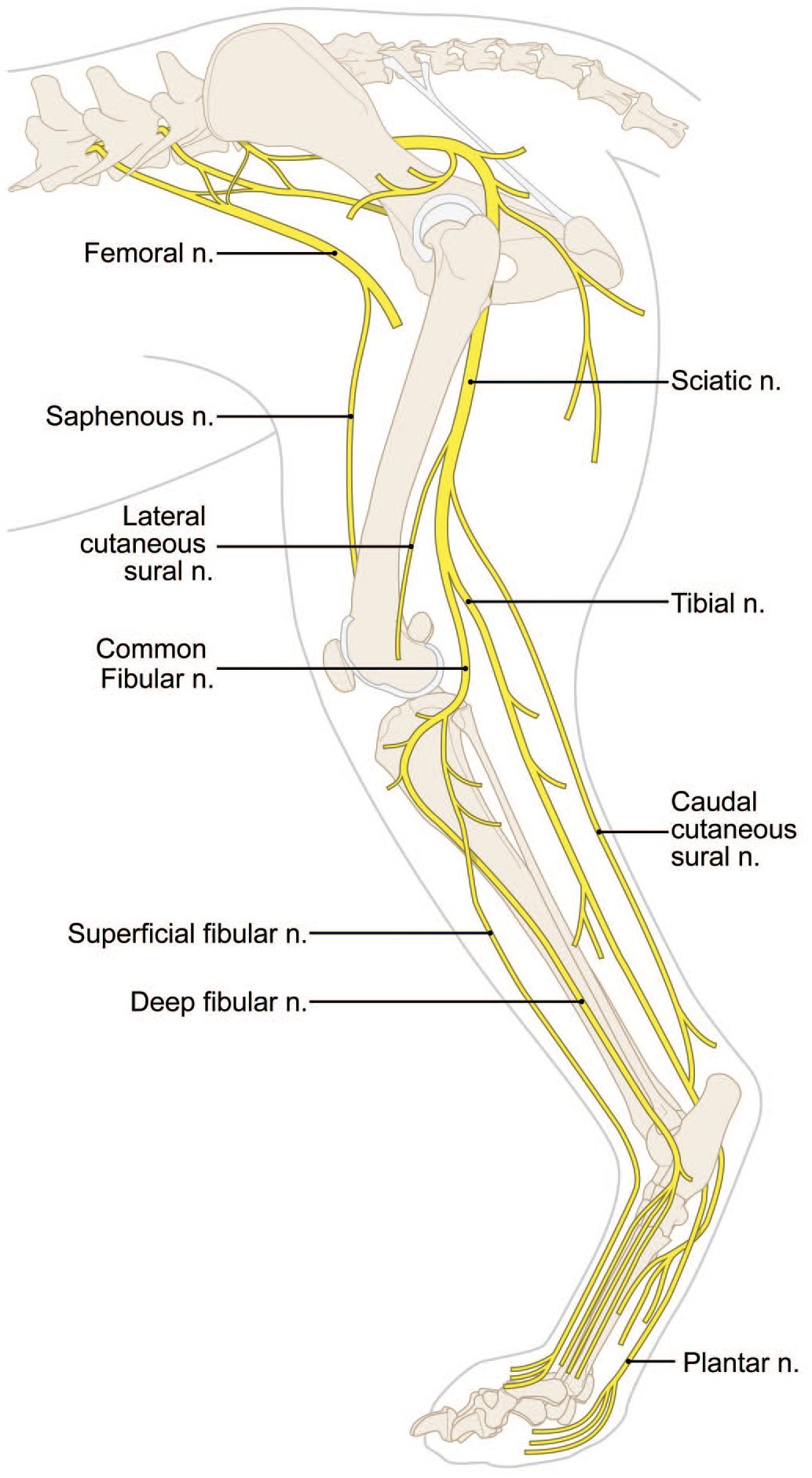
Anatomy of major somatic nerves (n.) for the dog hind limb. [Diagram © 2022 by Mr. Tim Vojt and provided by courtesy of the author.]
For the thoracic limb, a common nerve trunk originates from the combination of the C8 and T1 and T2 spinal nerves.30,35 This trunk may be found on the medial head of the triceps brachii muscle and between the brachial blood vessels. This trunk divides into 2, with the cranial division corresponding to the median nerve (Figure 32) and the caudal division to the ulnar nerve (Figure 34). The radial nerve arises from C7 to T2 spinal nerves, runs along the common median/ulnar trunk, and enters the triceps before spiraling around the humerus during its distal course (Figure 33).
Sampling somatic nerves in the Beagle dog
For general toxicity studies, many institutions sample one or more large mixed nerves, usually from the hind limb, to permit evaluation of the somatic motor and sensory systems simultaneously. In dogs, the sciatic nerve and one or more of its branches (most often the tibial and/or common fibular nerves) should be collected, but only the sciatic nerve is processed for microscopic evaluation. In general, nerves should be collected bilaterally to provide a reserve sample but are evaluated unilaterally because neurotoxic test articles typically do not induce unilateral effects. 6
For neurotoxicity studies, several mixed sensorimotor nerves (generally the sciatic, tibial, and common fibular at minimum) are sampled, processed, and evaluated. 6 In such studies, many institutions also collect at least one mixed sensorimotor nerve that serves the forelimb (e.g., median, radial, and/or ulnar) and/or a sensory-predominant nerve (e.g., plantar or saphenous from the hind limb 6 ) to increase the likelihood of identifying potential test article-related PNS effects. Length-dependent neuropathies may be missed if nerves are not sampled in the more distal portions of a limb.
Descriptive anatomy and sampling of the ANS in the Beagle dog
The parasympathetic ANS typically is sampled in general toxicity studies and neurotoxicity studies by evaluating ganglia (including enteric ganglia) and nerves present within the walls of various organs (e.g., intestine and urinary bladder). Where warranted, additional parasympathetic sampling may be done for neurotoxicity studies (Diagram D3). Vagus nerve (CN X) is the most widely distributed nerve in the body. Sampling CN X may include the distal (inferior) vagal ganglion (or “nodose” ganglion) and its associated cervical, thoracic, and even abdominal branches. The nerve and ganglion may be found shortly after CN X exits the jugular foramen of the skull, caudal to but near the cranial (superior) cervical ganglion in the cranial neck lateral to the thyroid gland (Figures 38, 39, and 41). 6
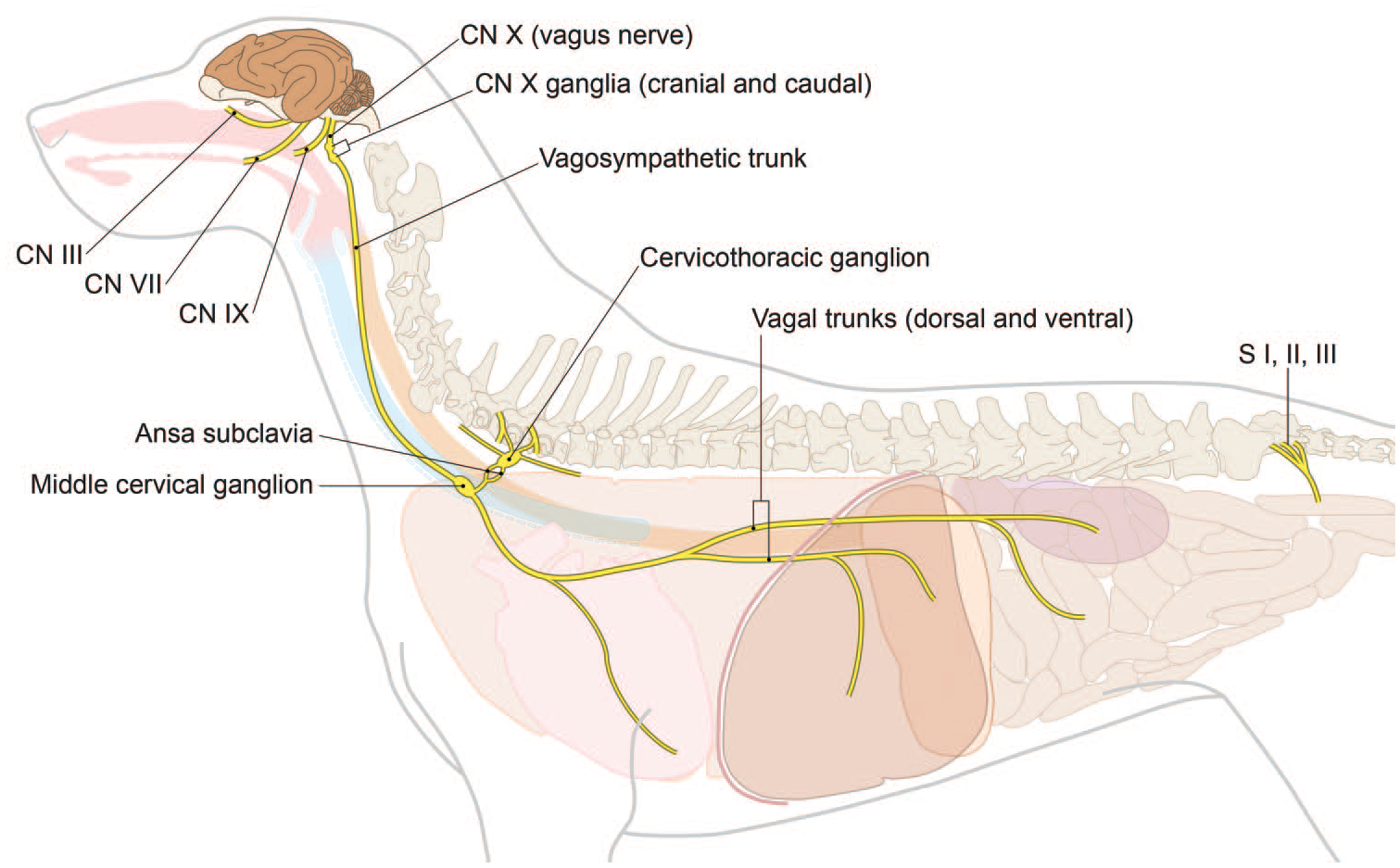
Anatomy of primary autonomic parasympathetic ganglia and nerves for the dog. CN, cranial nerve; S, sacral spinal nerve roots. [Diagram © 2022 by Mr. Tim Vojt and provided by courtesy of the author.]
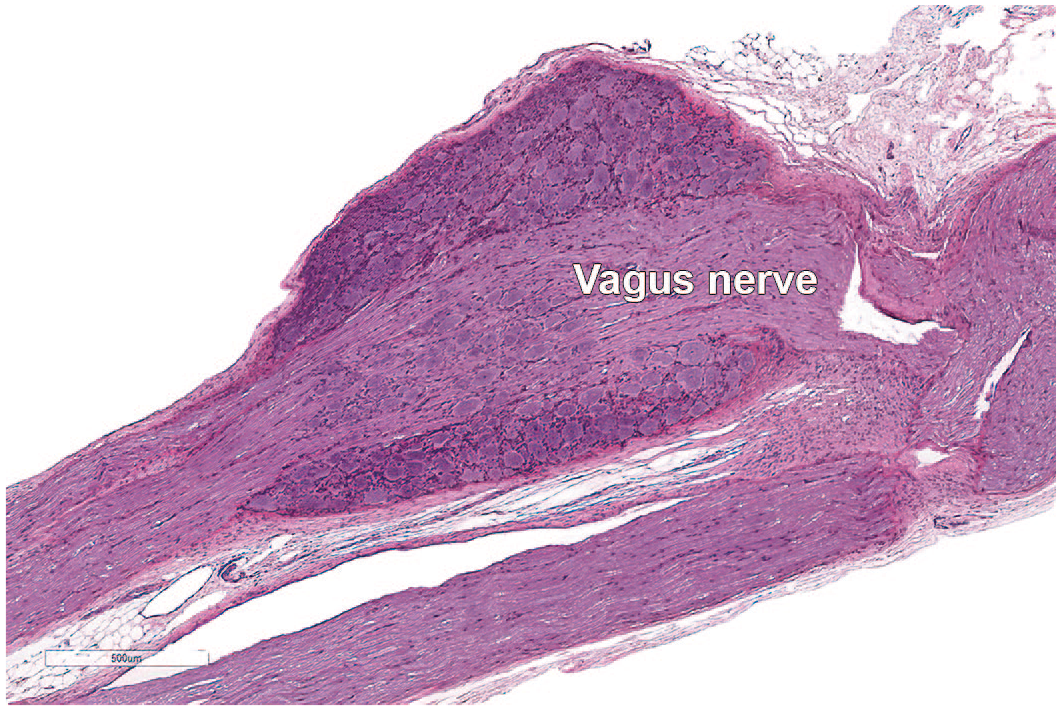
Microscopic appearance of the caudal (inferior or “nodose”) ganglion of the vagus nerve (cranial nerve X) of a Beagle dog. The nodose ganglion is found in the jugular foramen where the vagus nerve exits the ventral skull. This sensory ganglion holds unipolar or pseudo-unipolar neuronal cell bodies surrounded by satellite glial cells and some external connective tissue.
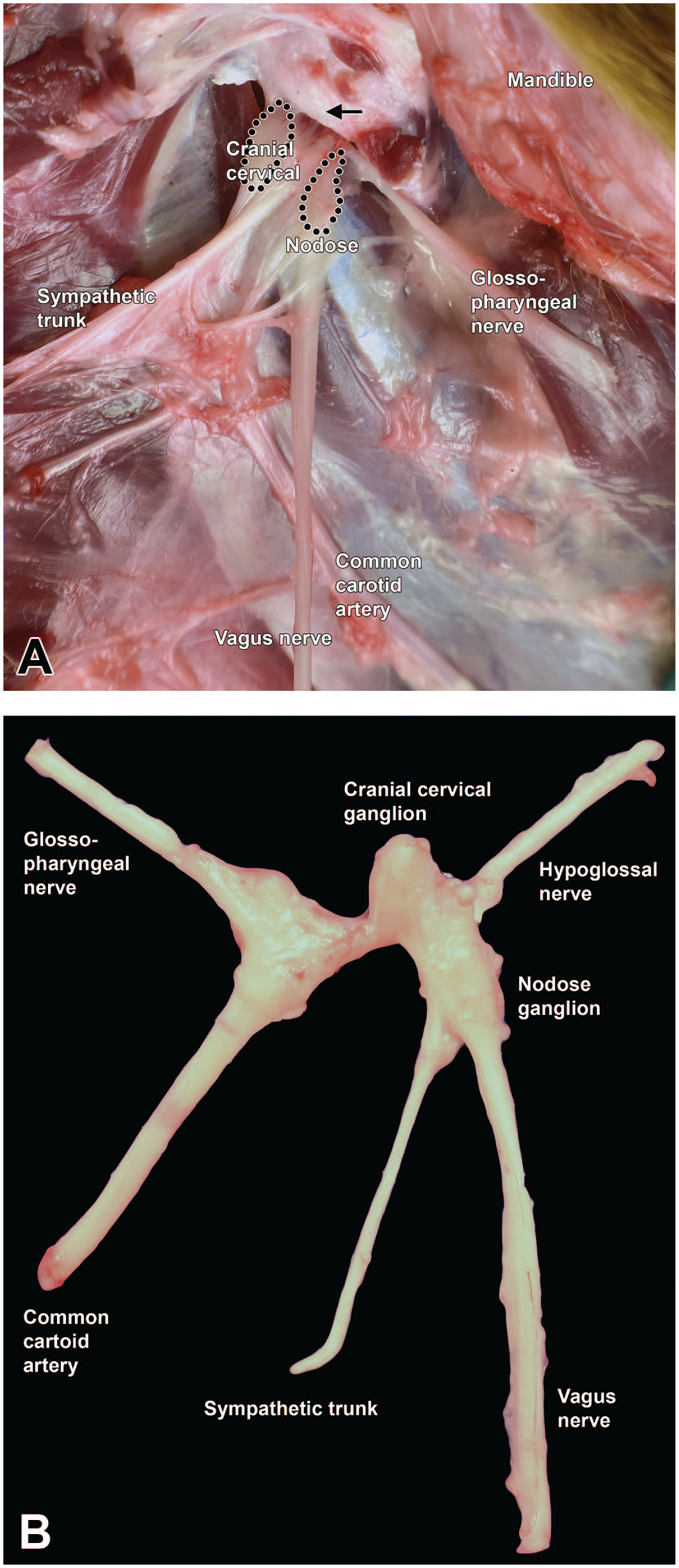
Dissection of key neural structures in the cervical region of the Beagle dog. Montage view showing in situ (A) and dissected (B) tissues below the jugular foramen (arrow), showing the vagus nerve (cranial nerve [CN] X) and its somatic sensory caudal (inferior or “nodose”) ganglion as well as the sympathetic trunk (an autonomic nerve) and its cranial (superior) cervical ganglion and the nearby common carotid artery and hypoglossal nerve (CN XII). The regions for sampling the cranial cervical and nodose ganglia are delimited by black dots (A); these structures typically can be placed in a single cassette for histopathologic evaluation.
The sympathetic ANS usually is sampled specifically only during neurotoxicity studies. Several elements may be collected (Diagram D4 and Figures 39–43). The sympathetic (paravertebral) trunk runs along the ventrolateral surfaces of the vertebral bodies (Figure 40). A sympathetic ganglion can be dissected from each intervertebral space, starting from the cervicothoracic (or “stellate”) ganglion. This ganglion is found at vertebra T1, and its alternate name derives from its large size and many branches (Figure 43). In the neck lateral (and cranial) to the thyroid gland and medial to the internal carotid artery, the cranial (superior) cervical ganglion is located cranial to and near the caudal (inferior or “nodose”) vagal ganglion; the 2 may be harvested and processed together. 6 The cranial cervical ganglion represents the most cranial group of sympathetic neurons (Figures 39 and 41). The celiac and cranial mesenteric ganglia are often adjacent to each other or fused and are located at the departure of the celiac (cranially) and cranial mesenteric (caudally) arteries from the aorta (Figure 42). A useful landmark for finding these structures consists of the left adrenal gland because these ganglia and the associated nerve plexus can be found just cranial to the left adrenal. These 2 ganglia provide sympathetic innervation to many abdominal viscera.
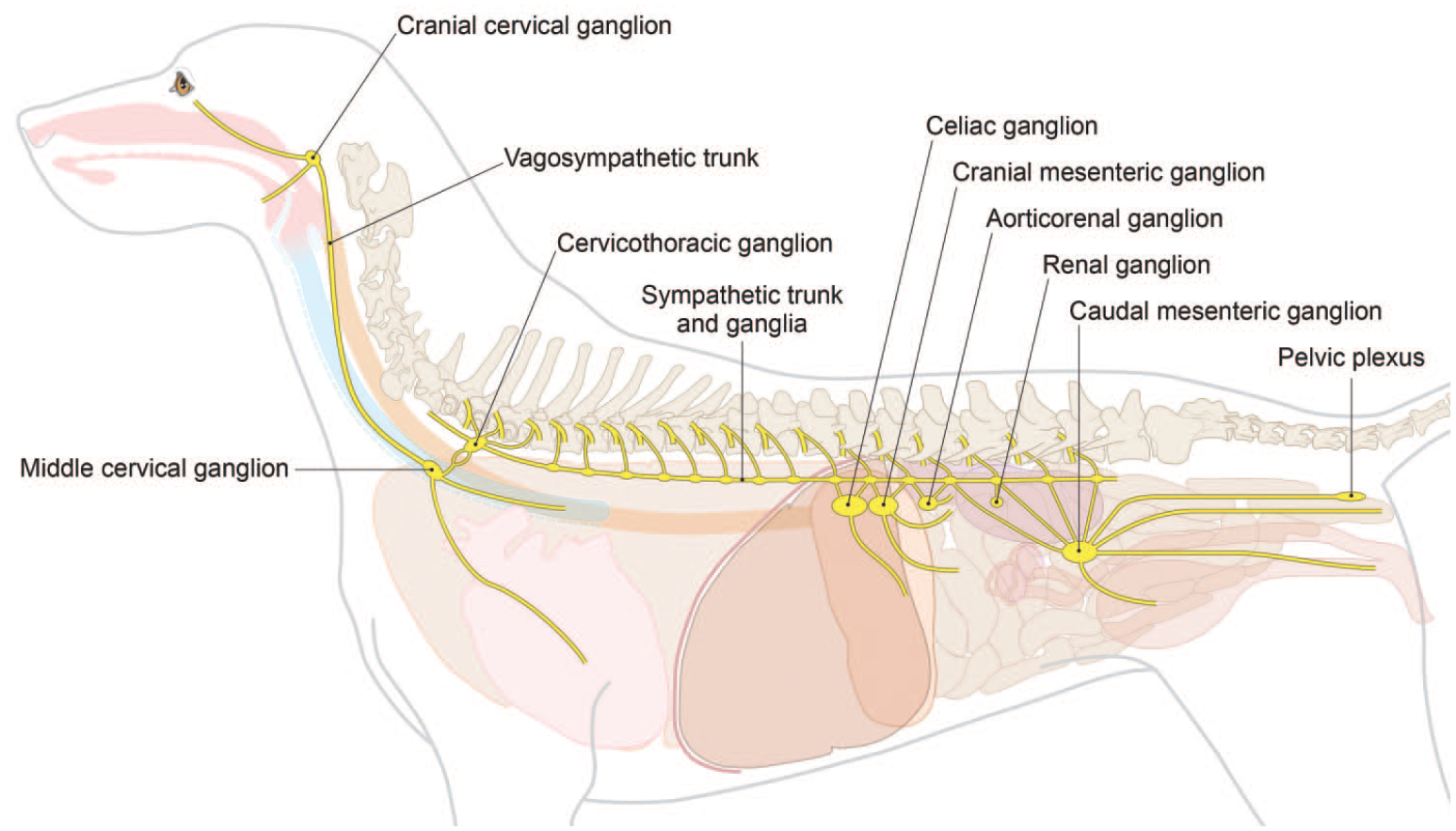
Anatomy of key autonomic sympathetic ganglia and nerves for the dog. [Diagram © 2022 by Mr. Tim Vojt and provided by courtesy of the author.]
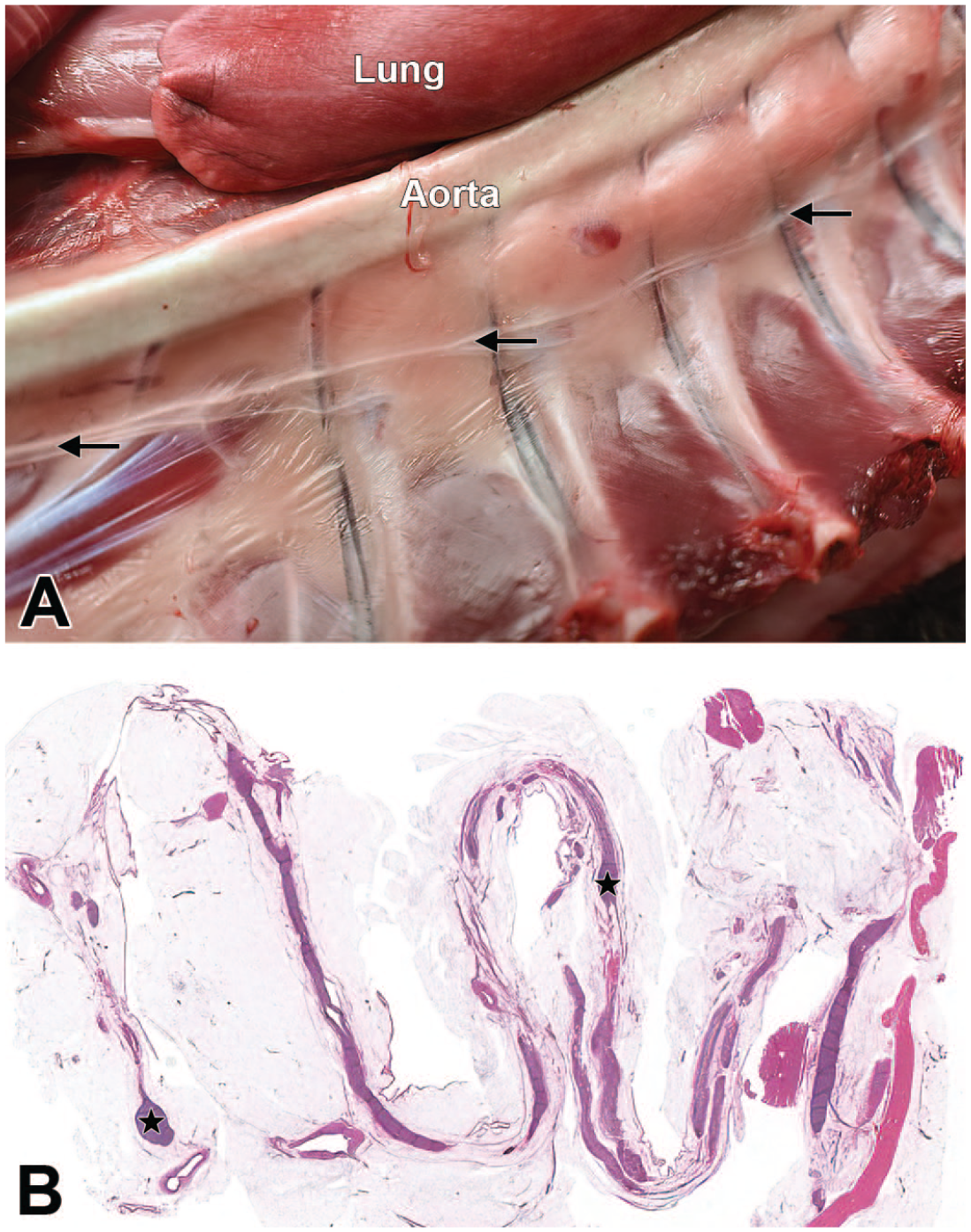
Sympathetic trunk of a Beagle dog. Montage view of the left hemithorax after lung and heart removal showing the paravertebral location (A, arrows) and low-magnification microscopic appearance (B) of the sympathetic trunk and its associated sympathetic chain ganglia (B, stars).
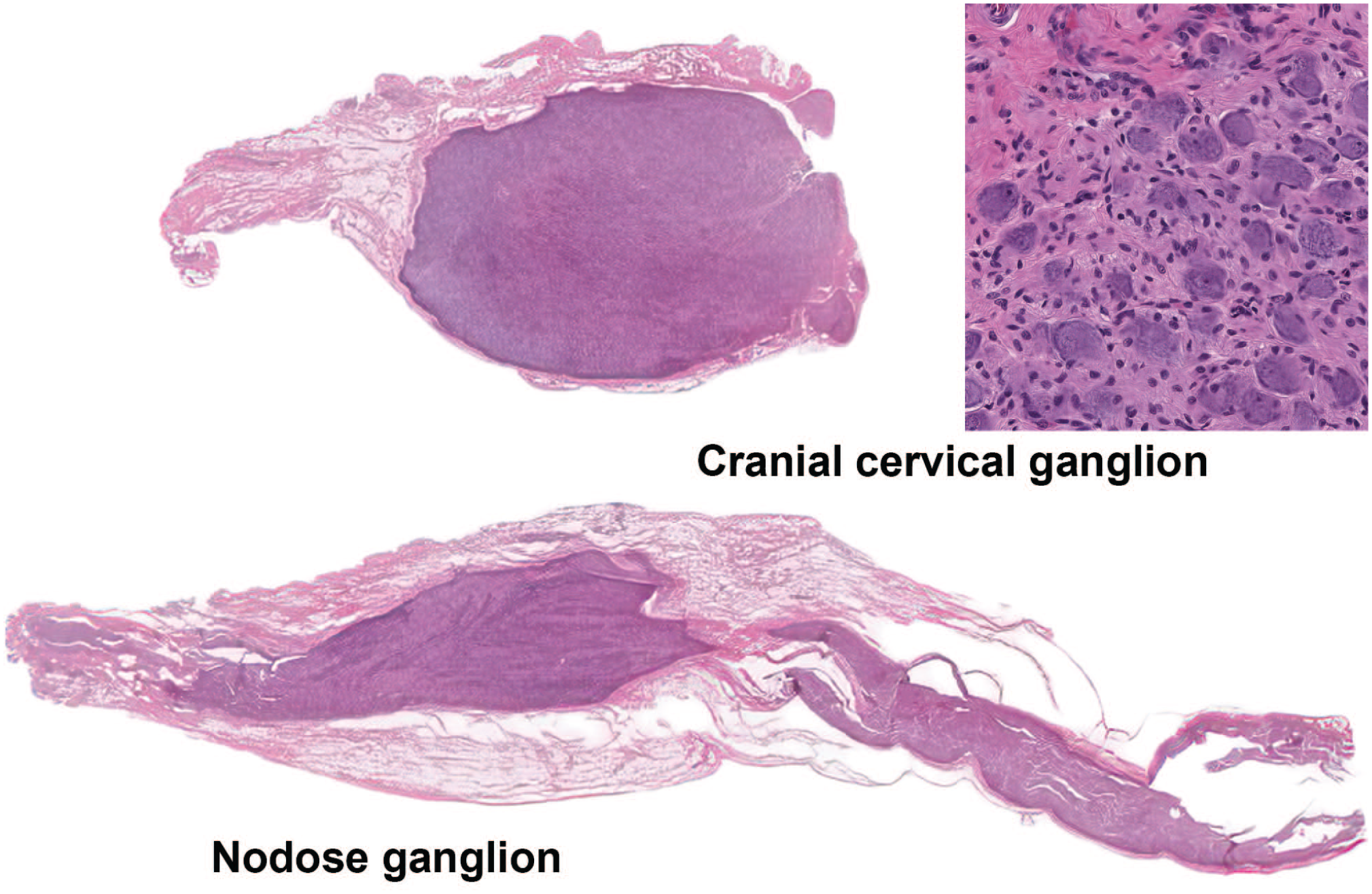
Differential microscopic appearance of the cranial cervical ganglion (autonomic) and caudal (inferior or “nodose”) ganglion of the vagus nerve (somatic sensory) of a Beagle dog. Low-magnification view of the cranial cervical ganglion as a well-individualized structure in comparison to the nodose ganglion that is interspersed among fibers of the vagus nerve. The insert shows a higher magnification of the cranial cervical ganglion demonstrating neuronal cell bodies with eccentric nuclei that are smaller and more homogeneous than the larger neuronal cell bodies with centrally located nuclei of somatic sensory ganglia (e.g., the nodose ganglion, figure 38).
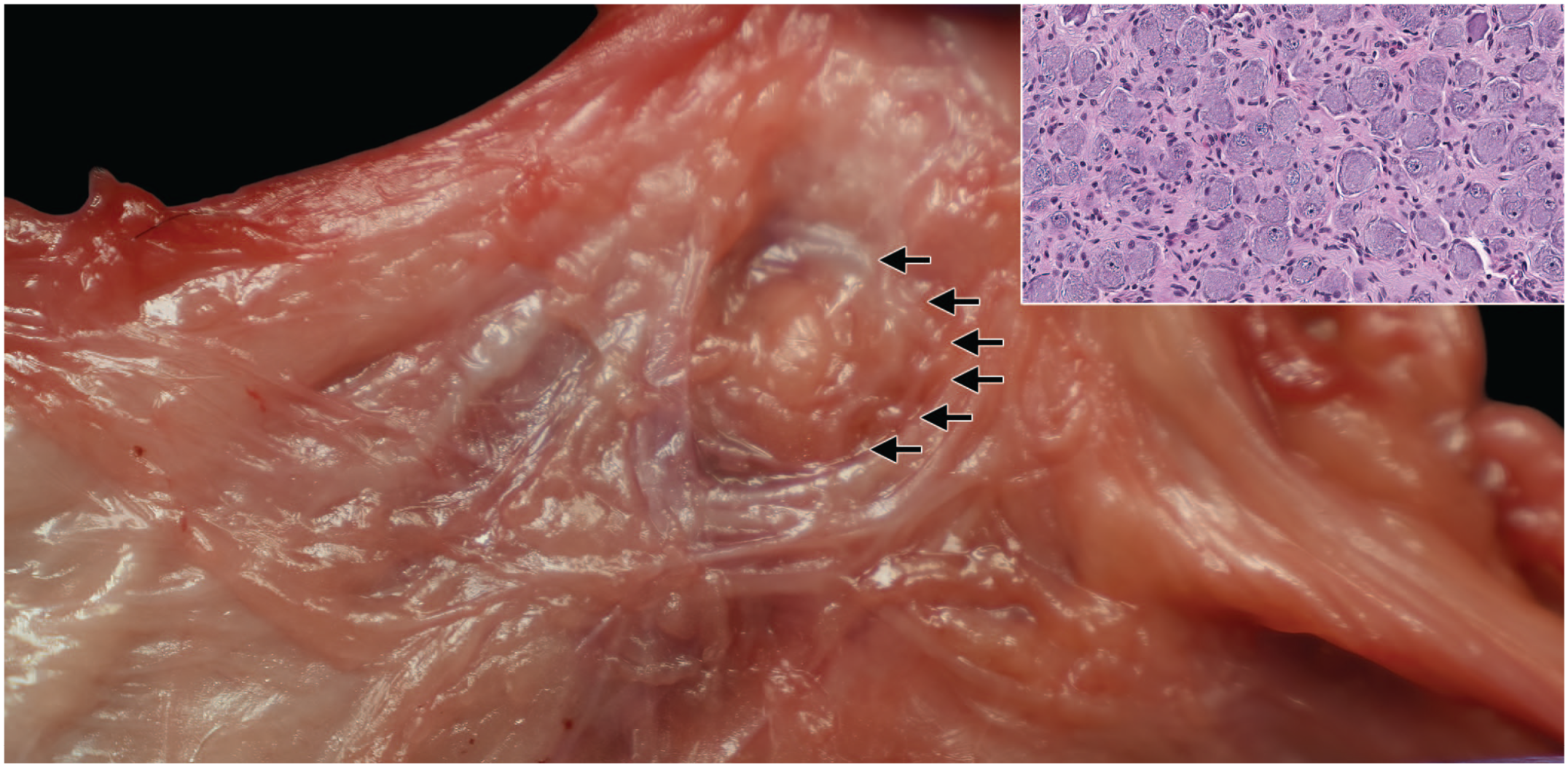
Sympathetic autonomic nervous system structures in the mesentery of a Beagle dog. Montage view showing a dissected cranial mesenteric ganglion (arrows) in the fatty tissue around the cranial mesenteric artery. This ganglion is adjacent to or variably fused with the celiac ganglion (with fused ganglia being referred to as the celiaco-mesenteric ganglion). The inset shows the microscopic correlate that is typical of an autonomic system (sympathetic) ganglion with eccentric nuclei within small and homogeneous basophilic neuronal cell bodies.
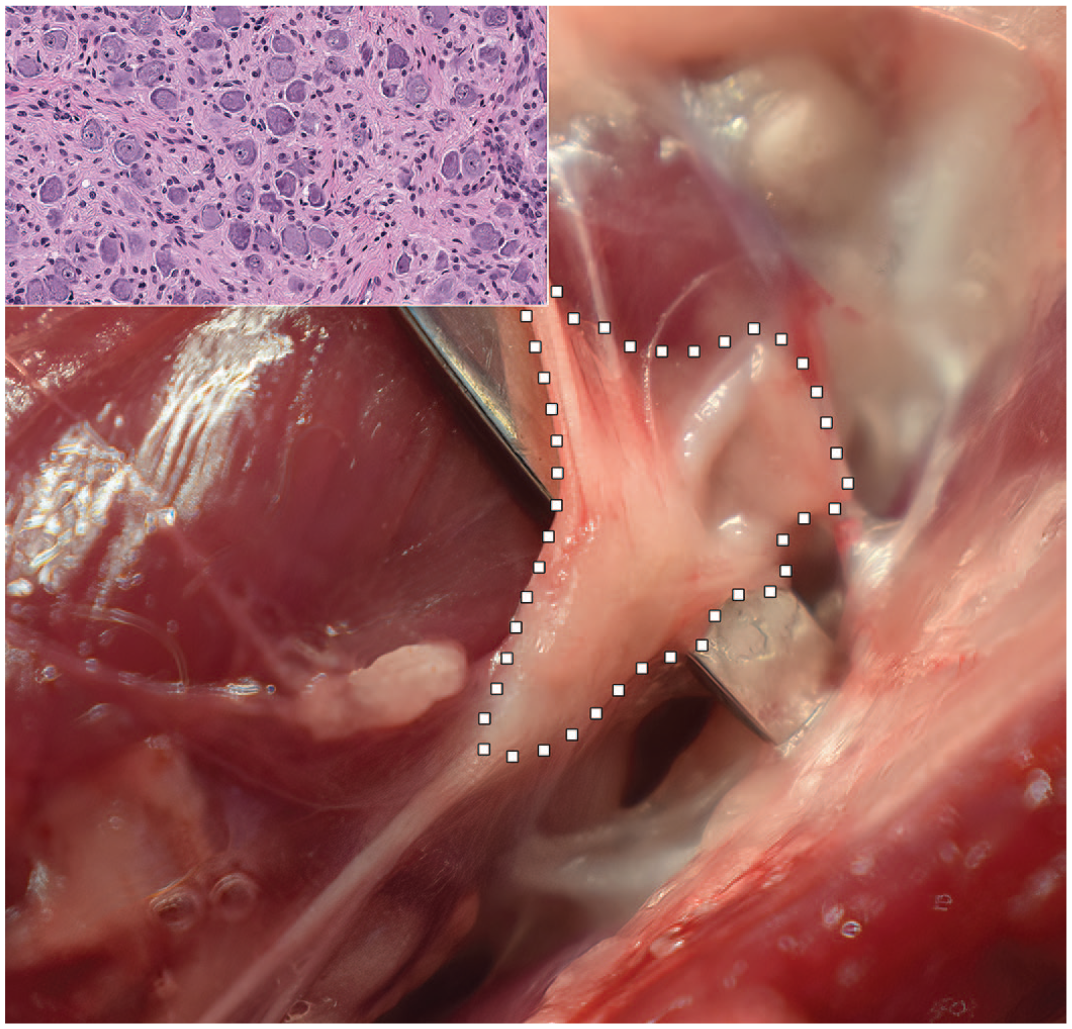
Cervicothoracic (or “stellate”) ganglion of the sympathetic autonomic nervous system of the Beagle dog. The ganglion in situ (delimited by white squares) is found at the level of thoracic vertebra 1 (T1), where it forms the cranial end of the thoracic portion of the sympathetic trunk. The inset shows the microscopic correlate that is typical of an autonomic system (sympathetic) ganglion with eccentric nuclei within small and homogeneous, basophilic neuronal cell bodies.
Care at necropsy is required to avoid damaging delicate neural structures in the cranial cervical region. In particular, the caudal vagal ganglion and the cranial cervical ganglion require meticulous dissection because they are located at the plane where cuts are made to remove the head. Accordingly, these ganglia should be removed prior to separating the head from the cervical region.
Special considerations for sampling the Beagle dog nervous system
Evaluation of the dog brain for nonclinical safety testing requires some adjustments compared to other species based on interspecies anatomic differences. First, the macrosmatic (i.e., relatively greater sense of smell) nature of dogs is reflected in large olfactory bulbs (0.31% of total brain volume) and prominent olfactory tracts relative to microsmatic primates (e.g., 0.01% of brain volume in humans). 45 The olfactory bulbs are embedded deeply in recesses within the rostroventral skull, so extra care must be taken while removing these lobes to ensure consistency in sampling. This care is necessary because olfactory bulbs represent approximately 2 g of brain weight, 46 which equates to about 2.5% of brain mass in dogs (based on the average weights noted in our series of dogs). Therefore, brain weights must be collected uniformly from all animals (i.e., always with or always without olfactory bulbs attached) to avoid generating excessive variation (bias) in brain weights. Second, the dog pituitary gland is enclosed within the sella turcica at the base of the skull and needs some particular attention at necropsy to be sampled together with the brain. Third, many adult Beagle dogs have asymptomatic ventriculomegaly (i.e., extensive dilation of the lateral ventricles), which should not be viewed as a test article-related effect. 47 This finding has been reported in 17 asymptomatic dogs using magnetic resonance imaging (MRI) 47 and is visible macroscopically at necropsy. 48 Functional and structural assessment including electron microscopy have demonstrated normal ependymal cells, white matter, and glia and microglial cells with normal cerebrospinal fluid flow. Finally, the dog brain possesses prominent gyri and sulci (gyrencephalic pattern) similar to other nonrodent test species (e.g., minipigs 11 and NHPs 9 ) and humans, though relative to NHPs the dog brain exhibits a conformation that is elongated along the rostrocaudal axis and the cerebrum and cerebellum are comparatively smaller.49,50
Discussion
This article provides practical tissue trimming schemes to ensure systematic and consistent sampling of the Beagle dog nervous system during nonclinical toxicity testing. Separate schemes are given for general toxicity studies and neurotoxicity studies. Both sampling and trimming protocols are consistent with existing regulatory guidance,1,2 globally harmonized testing expectations,3,4 and industrial “best practice” recommendations5,6 for evaluation of the CNS and PNS. Both schemes are suitable for use with current diagnostic nomenclature conventions for dogs. 48
For general toxicity studies, our 7-level brain trimming protocol (7 coronal hemisections on 7-9 slides [Figure 3A]) follows STP “best practice” recommendations for minimal brain trimming in nonrodent species. 5 Since the STP “best practices” paper does not specify the number of levels for dogs and provides a detailed illustration for NHPs only, we considered some species-specific differences in devising our 7-level brain-trimming protocol. The longer canine brain axis, the fact that the rostral portion of the cerebellum and brain stem are not partially covered by the occipital lobes in dogs (as they are in primates), and the need to survey various cerebrocortical regions effectively necessitated the addition of 2 more levels for dogs compared to NHPs. Our 7-level approach is aligned with but more detailed than previous recommendations for sampling the dog brain. 12 Importantly, the 7-section brain sampling approach for general toxicity studies using nonrodents generally should not be reduced further. The reason to avoid adjustments is that key neuroanatomic domains (defined in Table 1) need to be evaluated at least unilaterally, and our 7-level protocol provides the minimum means for performing this assessment in Beagle dogs.
For neurotoxicity studies, an expanded 14-level brain-trimming protocol (Figure 3B) allows for more extensive but still focused screening based on the evaluation of 11 coronal hemisections and 3 full-coronal sections (for the last 3 recommended levels containing caudal cerebellum and medulla oblongata). The more extensive 14-level brain-trimming protocol for neurotoxicity studies may be adjusted to some degree if existing knowledge has shown to the institution’s satisfaction that exclusion of one or a few levels will not impede hazard identification and characterization for a novel test article. Brain atlases for dogs may be used to develop more focused trimming schemes to access particular brain regions for neurotoxicity studies.17,18,51,52
Collection of spinal cord, ganglia (somatic and if warranted autonomic), and nerves should follow published species-agnostic recommendations for these tissues.5,6 For general toxicity studies in which nervous system toxicity is unknown or not expected, sampling may be limited to the cervical and lumbar spinal cord and sciatic nerve, although other neural tissues (e.g., thoracic spinal cord, DRG, and distal hind limb nerves) should be collected and archived in wet tissue so that they are not lost. For neurotoxicity, studies in which the nervous system is likely or known to be affected, all spinal cord segments (cervical, thoracic, lumbar, ±sacral); multiple DRG and TG; and several nerves (usually sciatic, tibial, and fibular) should be considered for evaluation.
Conclusion
The 2 trimming schemes defined in this paper describe methodical approaches for consistent sampling of the Beagle dog brain, spinal cord, and major somatic and autonomic ganglia and nerves during nonclinical toxicity studies. These trimming schemes are flexible by intent. The sampling requirements need to be defined by a pathologist at the stage of study planning to ensure that the scientific questions are answered. Success on the necropsy floor relies on basic neuroanatomic knowledge of the pathologist in charge as well as regular training of the technical team. While this guide illustrates commonly recommended structures, it is by no means exhaustive and the breadth of the tissue list may need to be adjusted to the context of the study. That said, for most of the studies and personnel involved, this current guide offers a succinct reference with sufficient detail to allow for the harvest and localization of morphologic changes throughout major critical regions of the nervous system. The 7-level trimming protocol for general toxicity studies follows the STP “best practices” recommendations for brain sampling in nonrodent species during general toxicity studies. 5 The 14-level trimming scheme for neurotoxicity studies provides for a more thorough brain sampling approach for detecting known or suspected neuroactive test articles.5,9,12
Supplemental Material
sj-docx-1-tpx-10.1177_01926233221099300 – Supplemental material for A Technical Guide to Sampling the Beagle Dog Nervous System for General Toxicity and Neurotoxicity Studies
Supplemental material, sj-docx-1-tpx-10.1177_01926233221099300 for A Technical Guide to Sampling the Beagle Dog Nervous System for General Toxicity and Neurotoxicity Studies by Xavier Palazzi, Ingrid D. Pardo, Hayley Ritenour, Deepa B. Rao, Brad Bolon and Robert H. Garman in Toxicologic Pathology
Footnotes
Acknowledgements
The authors would like to thank Mr. Casey Ritenour, Mr. Christopher Dubois, and Mrs. Laura Monarski in the global microscopic imaging group, histopathology laboratory, and morphology laboratory, respectively, at Pfizer, Inc. in Groton, CT for their help capturing macroscopic images and processing the tissues for light microscopic examination. The authors also commend the efforts of Mr. Tim Vojt for preparing the schematic diagrams as well as Mr. Walt Bobrowski (Lakeshore Digital Art, LLC) and Ms. Beth Mahler in optimizing the images.
Author Note
Part of Dr. Pardo’s contribution was performed while employed by Pfizer Inc.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
