Abstract
For general toxicity studies, a technique was designed to consistently sample the most important neuroanatomic regions of the brain, spinal cord, and peripheral nerve of cynomolgus monkeys using a limited number of blocks and slides. Using the most rostral portion of the pons as a landmark, the entire fixed brain was cut dorsoventrally into cross-sectional slabs 4 mm in thickness. For microscopic evaluation, six blocks of the brain at the levels of the frontal pole, anterior commissure, rostral thalamus, caudal thalamus, middle cerebellum with brainstem, and occipital lobe were trimmed to fit in standard tissue cassettes. Cross- and oblique sections of the spinal cord including the dorsal root ganglion and dorsal and ventral nerve roots were obtained at the levels of C1–C4, T10–T12, and L1–L4. Cross- and longitudinal sections of the sciatic nerve were also obtained. This technique offers a consistent and reliable method to routinely sample most of the important regions of the central and peripheral nervous system of monkeys using ten blocks. This method is readily adaptable to other species of nonhuman primates, dogs, and minipigs and can be quickly learned by the technicians performing the trimming procedures.
Keywords
Introduction
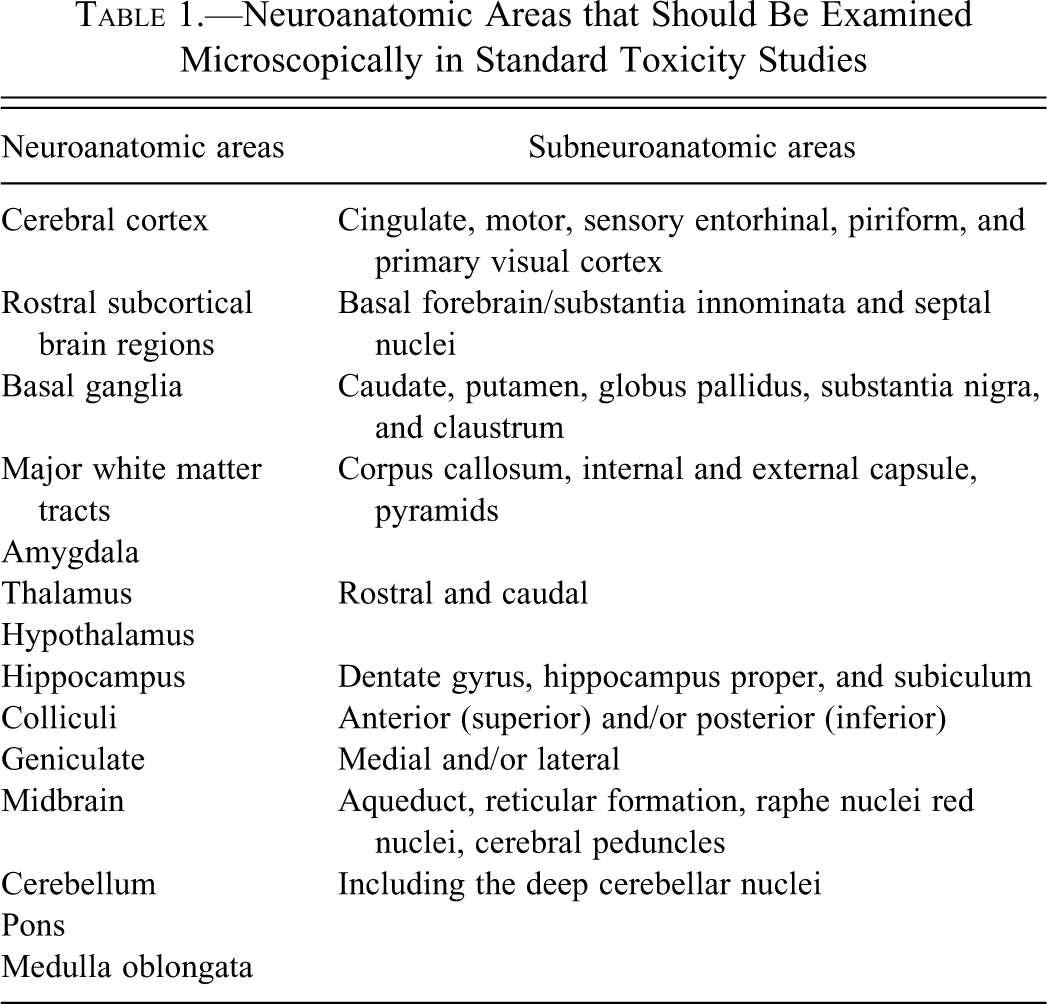
Evaluation of the brain, spinal cord, and nerves is important in general toxicity studies and in evaluation of animal models of neurodegenerative diseases and of brain or spinal cord injury. There is great variation in methods used to sample the brain for routine examination in toxicity studies and few references to guide toxicologic pathologists and technical staff in selection of appropriate regions for examination. Brain atlases of the rhesus monkey (Paxinos et al. 2000; Saleem and Logothetis 2007) are helpful in identifying brain regions in cynomolgus monkeys. In addition, Szabo and Cowan published in 1984 a stereotaxic atlas of the brain of the cynomolgus monkey. No atlas of the spinal cord anatomy in any macaque species is available; however, human (Alloway and Pritchard 2007; Crosby et al. 1962; Hendelman 2005) or domestic mammal (King 1999) textbooks generally may be used to define the neuroanatomy of the macaque spinal cord. It is recommended that a list of important brain regions be examined in general toxicity studies (Table 1). In this paper, a procedure for routine and consistent sampling of specific important neuroanatomic areas of the brain, spinal cord, and sciatic nerve of the cynomolgus monkey (Macaca fascicularis) is described. All procedures were approved by the animal care and use committee in accordance with federal regulations and the Guide for the Care and Use of Laboratory Animals.
Neuroanatomic Areas that Should Be Examined Microscopically in Standard Toxicity Studies
Neuroanatomic Areas Included in the Brain Section of the Frontal Pole (Figure 4)

The objective of this paper is to provide guidance on how to perform the evaluation of the central and peripheral system of cynomolgus monkeys for general toxicity studies, in addition to providing a brief review of the neuroanatomic regions present within the sections included in this technique. These methods have been illustrated in a CD-ROM presentation that is available from the authors (Pardo et al. 2010).
The Brain
General Descriptive Anatomy of the Brain of the Cynomolgus Monkey
The brain of the cynomolgus monkey weighs, on average, 74 g, ranging from 62 to 85 g (historical organ weight data from Pfizer, Inc., Groton, CT). This weight represents an average for cynomolgus monkeys of 2.5–5.0 years of age (the typical age selected for general toxicity studies). Brain weights can vary with age and sex of the animals. Brain weights of male and older animals are greater than those of female and younger animals. In contrast to dogs and rodents, the brain of the cynomolgus monkey has a relatively larger and more sharply pointed frontal pole, smaller olfactory lobes, narrow lateral and third ventricles, prominent gyri and sulci corresponding to a well-developed cerebral cortex, and extension of the occipital cortex over the brainstem and cerebellum. In addition, the horizontal plane of the brainstem is oriented at approximately 45° to the horizontal plane of the hemispheres, resulting in a wedge-shaped midbrain. Compared to humans, cynomolgus monkeys have a proportionally smaller frontal lobe, a wider interthalamic adhesion bridging the third ventricle, and a brainstem that is positioned at a less acute angle.
Collection, Sampling, and Sectioning of the Cynomolgus Monkey Brain
For general toxicity studies, the technique described below is recommended to consistently sample most of the important neuroanatomic regions of the brain using a limited number of blocks and slides. This technique is very simple and requires minimal neuroanatomic knowledge. Additional regions of interest can be easily added.
The brain is obtained by removing the skin, connective tissue, and muscle from the superior aspect of the skull (calvarium) that extend from the top of the orbits and ears to the atlanto-occipital area. The exposed calvarium is removed using a bone saw (Lipshaw Autopsy Saw Model #100000, 1.5 amps, 115 volts AC, Hz50/60, Pittsburgh, PA or Stryker Autopsy Saw, Model 810, 120V-60 Hz 2.0A, Kalamazoo, MI), being careful not to damage the brain parenchyma. The falx cerebri, a strong, arched fold of dura mater that descends vertically in the longitudinal fissure between the cerebral hemispheres, is removed at the midline, so that the brain can removed from the skull. Note that the pituitary gland will not come out with the brain, and it must be removed from within the sella turcica. Because the olfactory bulbs are small and embedded in the skull, collection of the olfactory tracts and bulbs requires care during the dissection of the brain. The brain is fixed in 10% neutral buffered formalin for at least forty-eight hours before trimming at a volume of about 10:1 to 20:1 (formalin: tissue).
Using the most rostral portion of the pons as a landmark, the entire fixed brain can be held firmly and cut (Tissue Slicer Blade, Thomas Scientific, Swedesboro, NJ, catalog #6727C18) dorsoventrally at a 45° angle (Figures 1A and 1B). A rectangular slicing matrix tool (fabricated in house) that is 15 cm in length and 8 cm in width and has two outer raised edges that are 1 cm in width and 4 mm in height is recommended to facilitate the brain slicing (Figure 2A) into multiple cross-sectional slabs 4 mm in thickness (Figure 2B). There are approximately fourteen cross-sections of the brain using this rectangular slicing matrix tool. All sections of brain are 4 mm in thickness with the exception of cross-sections 1 and 14, which are thicker because they are the rostral and caudal regions of the brain (Figure 3). Please note that the rostral aspect of each slice will be embedded downward in the paraffin boat and will be the side to be sectioned.
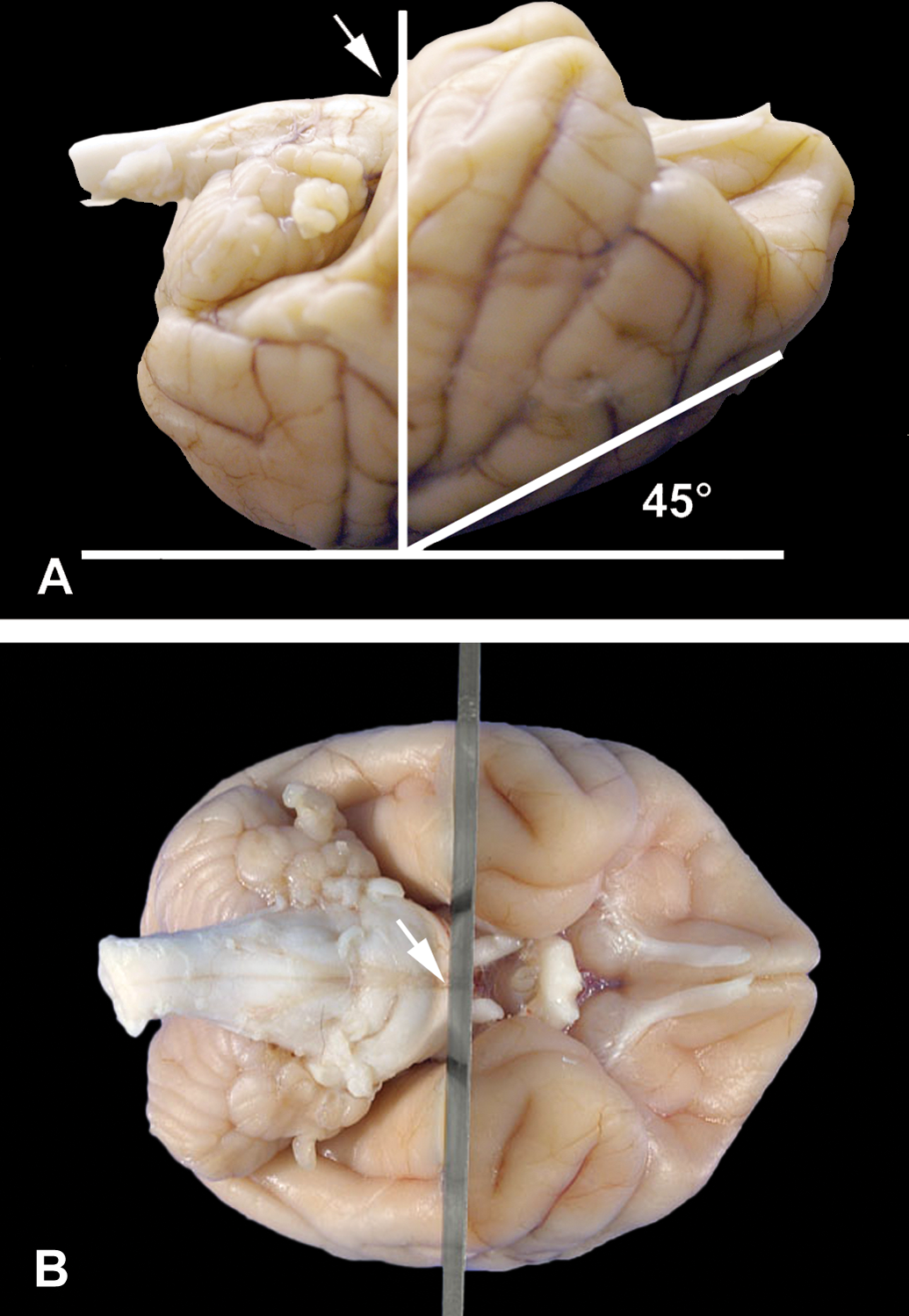
(A) Using the most rostral portion of the pons as landmark (arrow), the fixed brain is placed with the superior surface down and at approximately 45° angle. (B) The brain is sliced once vertically in cross-section at the level of the most rostral portion of the pons.
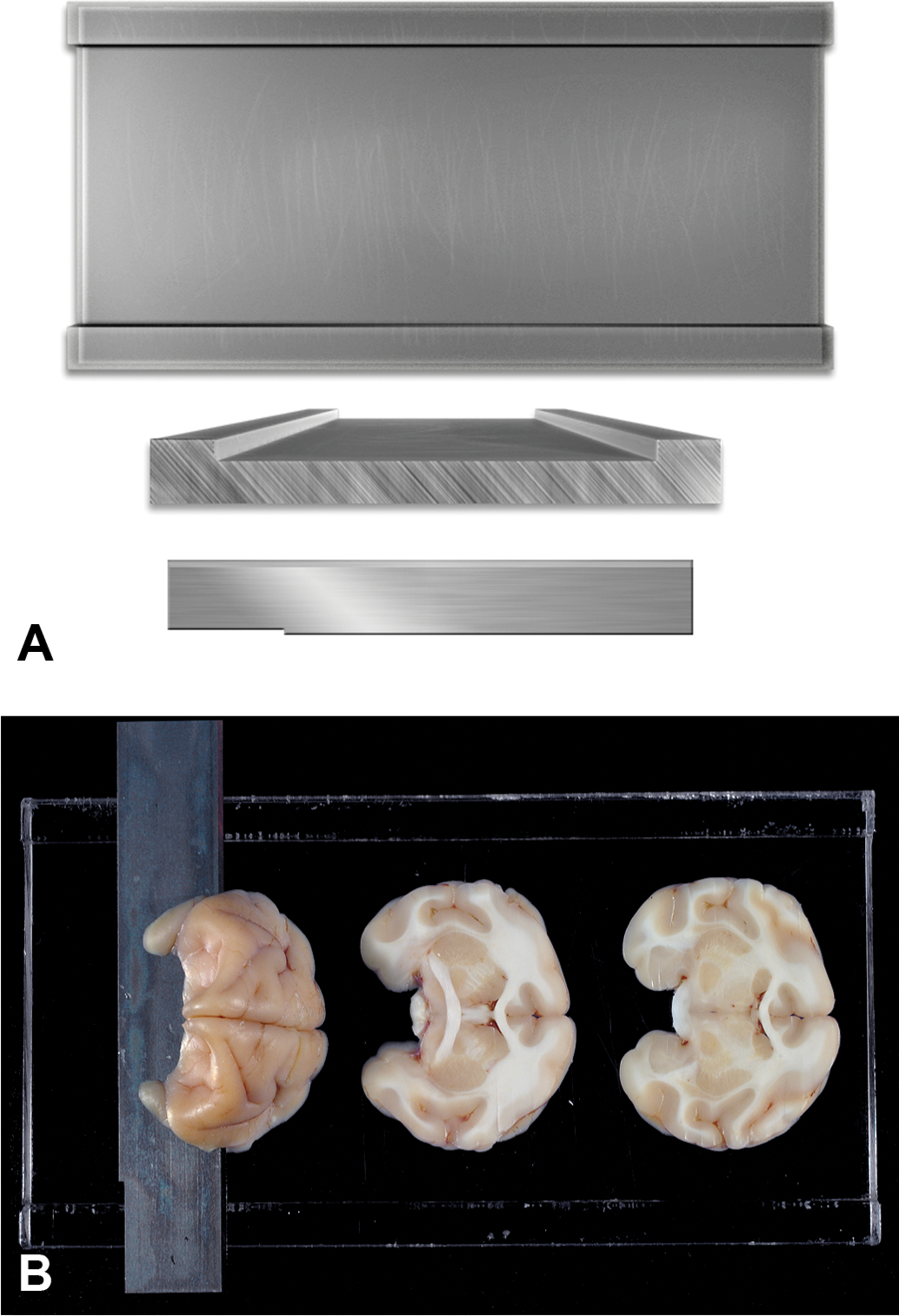
(A) A rectangular slicing matrix tool (fabricated in house).15 cm in length, 8 cm in width, with two outer raised edges that are 1 cm in width and 4 mm higher than the central trough, is used to slice the entire brain into cross-sectional slabs 4 mm in thickness. Fixed brain is cut with a Tissue Slicer Blade (Thomas Scientific, Swedesboro, NJ, catalog #6727C18). (B) The rostral half of the brain is placed cut surface down on the rectangular slicing matrix and is sectioned in multiple slices that are 4 mm thick. The caudal half of the brain is also sectioned in an identical manner.
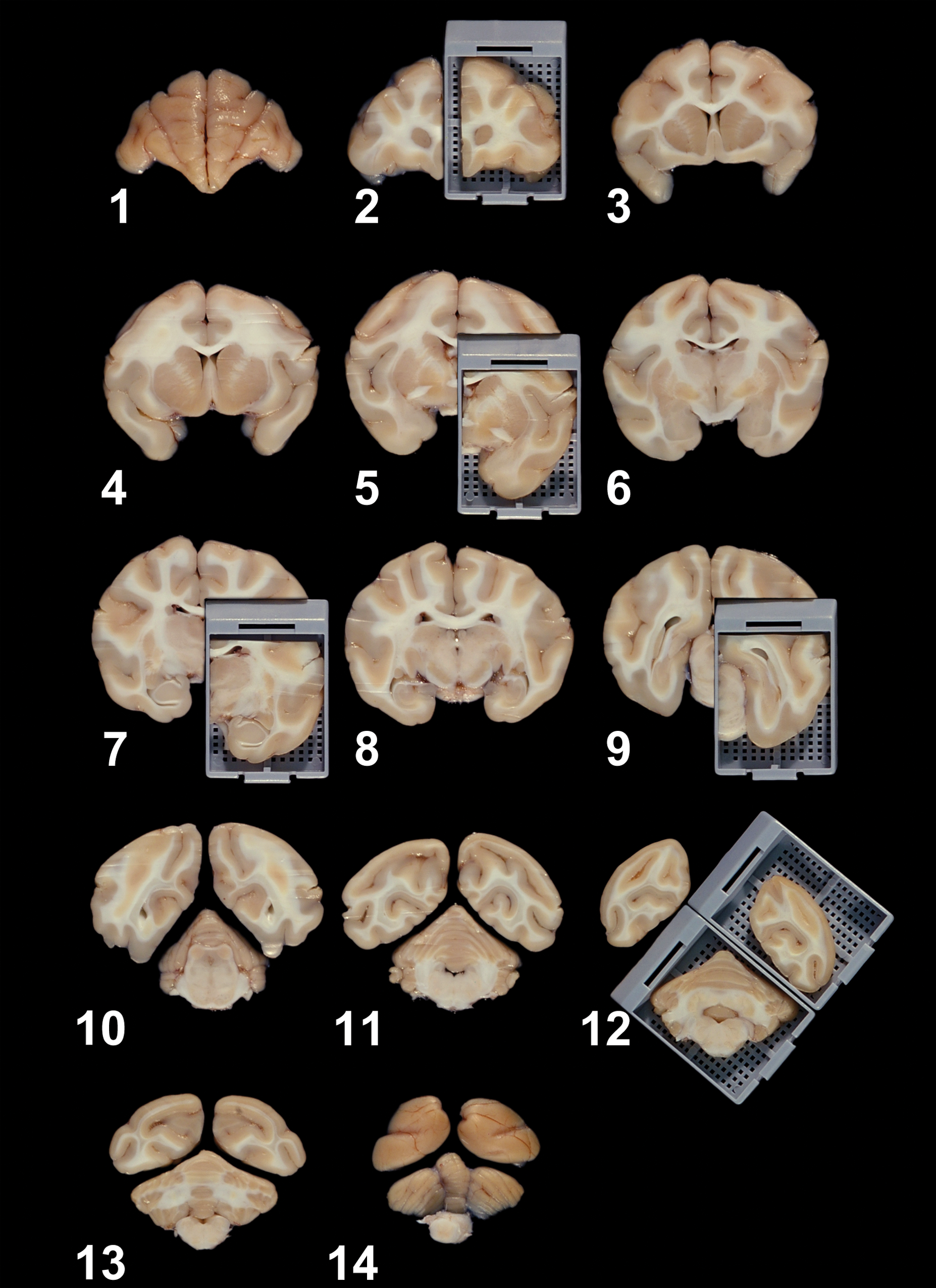
The fixed brain is sliced into fourteen cross-sections of 4 mm in thickness at periodic intervals. Cross-sections 2, 5, 7, 9, and 12 represent the frontal pole; anterior commissure; rostral thalamus; caudal thalamus; and occipital lobe, cerebellum, and brainstem, respectively. Please note that the rostral aspect of each slice is shown in this photo and it will be embedded downward in the paraffin boat to be the side to be sectioned.
For microscopic evaluation, six blocks of the brain at the levels of the frontal pole, anterior commissure, rostral thalamus, caudal thalamus, middle of the cerebellum with brainstem, and occipital lobe are trimmed to fit into standard tissue cassettes and slides (1 × 3-in., VWR Micro Slides Superfrost, White, Clipped corner for Leica IP Ink Jet Printer, Catalog #89078-500, VWR International, LLC, Radnor, PA). These sections are obtained unilaterally with the exception of the cerebellar/brainstem section, which is a full coronal slice. Sections of the cerebrum are taken unilaterally to maximize the numbers of neuroanatomic regions that are sampled, and since toxicologically induced lesions are typically bilateral in localization (Figure 3). However, contralateral slices of the sectioned brain should be preserved in formalin for future sampling. Although this technique provides a very consistent way to sample the brain, comparable sections from different animals will not be identical because of the small size of some neuroanatomic areas. The lack of a high degree of section homology is not considered to represent a problem in general toxicity studies, because having highly homologous sections results in the exclusion of some neuroanatomic regions from all of the sampled brains.
The neuroanatomic areas generally present at each level of the sectioned brain are described below. Selected sections of brain at the levels of the frontal pole, anterior commissure, rostral thalamus, caudal thalamus, middle of the cerebellum with brainstem, and occipital lobe can be processed and stained for light microscopic examination (Figures 4–9). In addition, Tables 2–7 briefly describe the functions, neural connections, and principal neurotransmitters present in a variety of important neuroanatomic areas within selected brain sections.
Figure 4. Hematoxylin and eosin cross section of the cynomolgus monkey brain at the level of the frontal pole (cross-section 2): 6 M, accessory motor cortex; cc, corpus callosum; Cd, caudate nucleus; Cl, claustrum; ec, external capsule; ic, internal capsule; Lv, lateral ventricle; Pir, piriform cortex; ProM, premotor cortex; Pu, putamen.
Figure 5. Hematoxylin and eosin cross section of the cynomolgus monkey brain at the level of the anterior commissure (cross-section 5): acp, anterior commissure; B, basal forebrain region; cc, corpus callosum; Cd, caudate nucleus; CG, cingulate cortex; Cl, claustrum; opc, optical chiasm; ec, external capsule; EGP, external globus pallidus; ER, entorhinal cortex; HT, hypothalamus; ic, internal capsule; IC, insular cortex; lf, lateral fissure; Pu, putamen; RA, rostral amygdala; rf, rhinal fissure; Rt, rostral thalamus; TC, temporal cortex; VP, ventral pallidum.
Figure 6. Hematoxylin and eosin cross-section of the cynomolgus monkey brain at the level of the rostral thalamus (cross-section 7): cc, corpus callosum; Cd, caudate nucleus; CG, cingulate cortex; cp, cerebral peduncle; ec, external capsule; ER, entorhinal cortex; GP, globus pallidus; Hip, hippocampus; ic, internal capsule; IC, insular cortex; lf, lateral fissure; LG, lateral geniculate; Lv, lateral ventricle; opt, optic tract; Pu, putamen; rf, rhinal fissure; SN, substantia nigra; T, thalamus; TC, temporal cortex.
Figure 7. Hematoxylin and eosin cross-section of the cynomolgus monkey brain at the level of the caudal thalamus (cross-section 9): Aq, aqueduct (mesencephalic aqueduct or third ventricle); cc, corpus callosum; CG, cingulate cortex; DpMe, deep mesencephalic nuclei; eml, external medullary lamina; ER, entorhinal cortex; Hip, hippocampus; IC, insular cortex; Lv, lateral ventricle; PC, parietal cortex; Pi, pineal gland; PnO, pontine nuclei; pons, pons; Pul, pulvinar nucleus; Rn, raphe nuclei; Rt, reticular thalamic nuclei; Sc, superior colliculus; TC, temporal cortex.
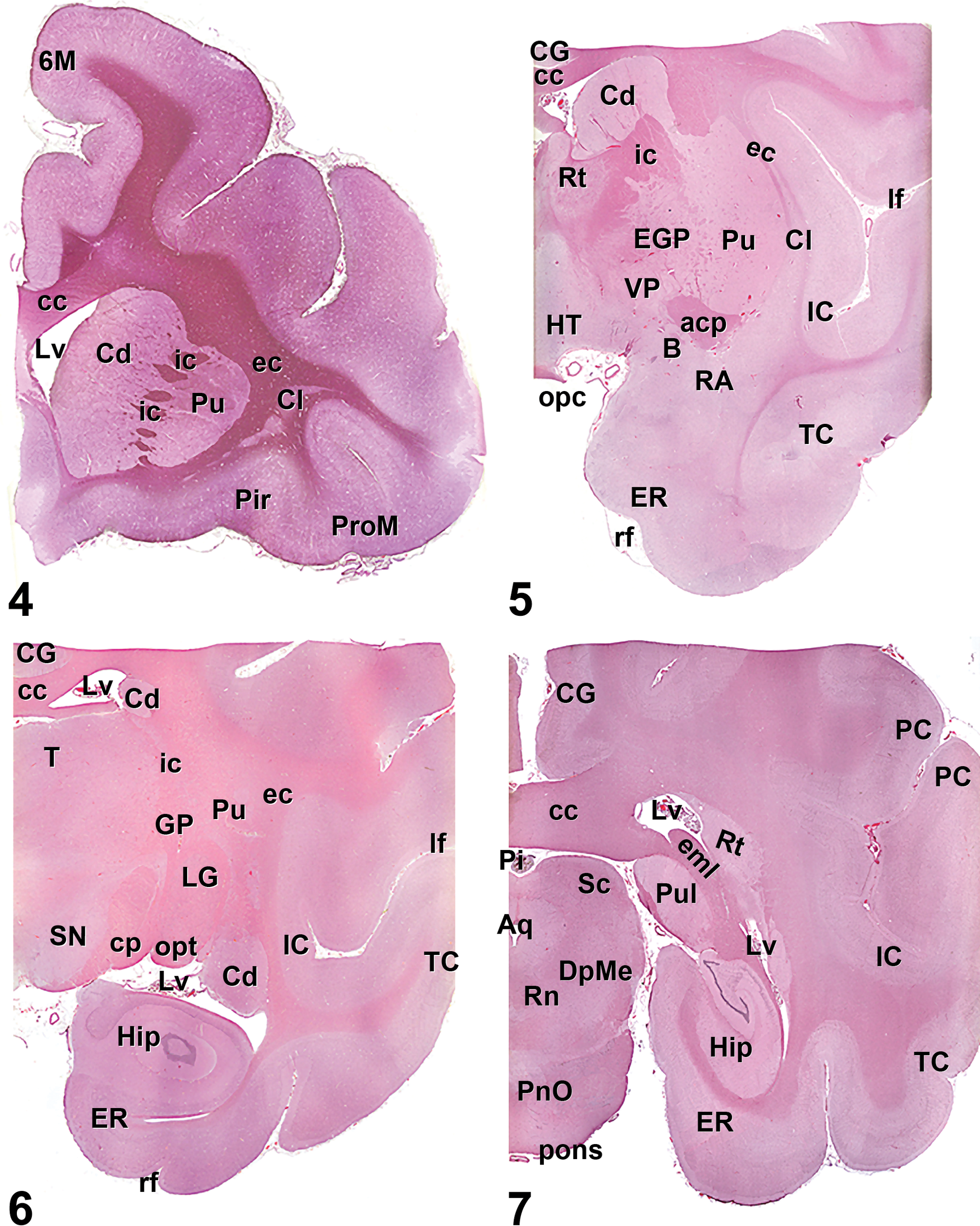
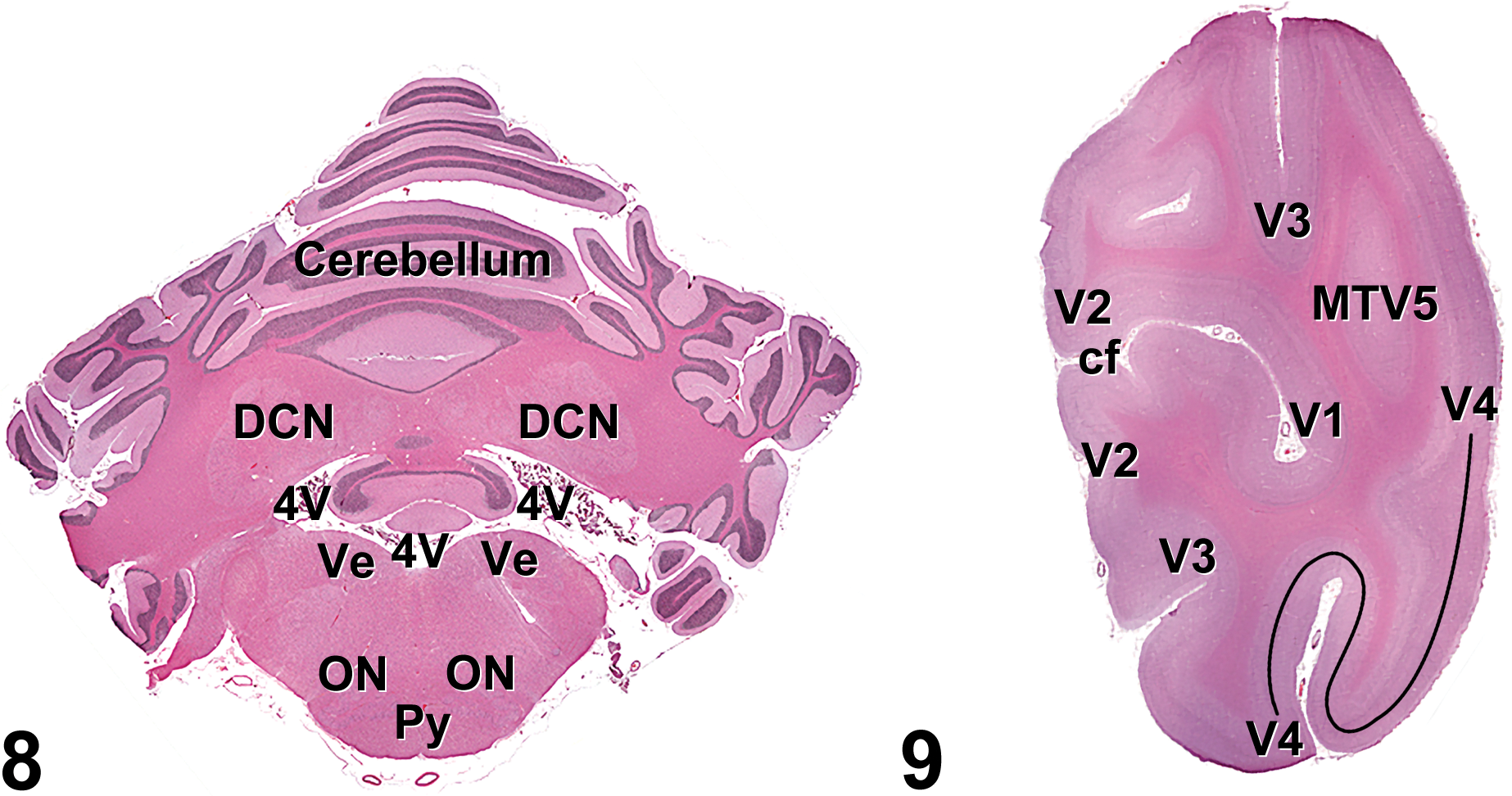
Figure 8. Hematoxylin and eosin cross-section of the cynomolgus monkey cerebellum (cross-section 13): DNC, deep cerebellar nuclei; ON, olivary nuclei; Py, pyramidal tracts; V4, fourth ventricle; Ve, vestibular nuclei.
Figure 9. Hematoxylin and eosin cross-section of the cynomolgus monkey brainstem (cross-section 13): cf, calcarine fissure; MTV5, secondary visual cortex (medial temporal area); V1, primary visual cortex; V2, V3, V4, secondary visual cortex.

(A) The fixed spinal cord is sectioned coronally (cross-section) at the level of C1–C2. (B) The fixed spinal cord is sectioned obliquely at the level of T11–T12. (C) Cross- and oblique-fixed spinal cord sections at the level of C1–C2. Please note that the rostral aspect of each slice is shown in this photo, and it will be embedded downward in the paraffin boat to be the side to be sectioned.
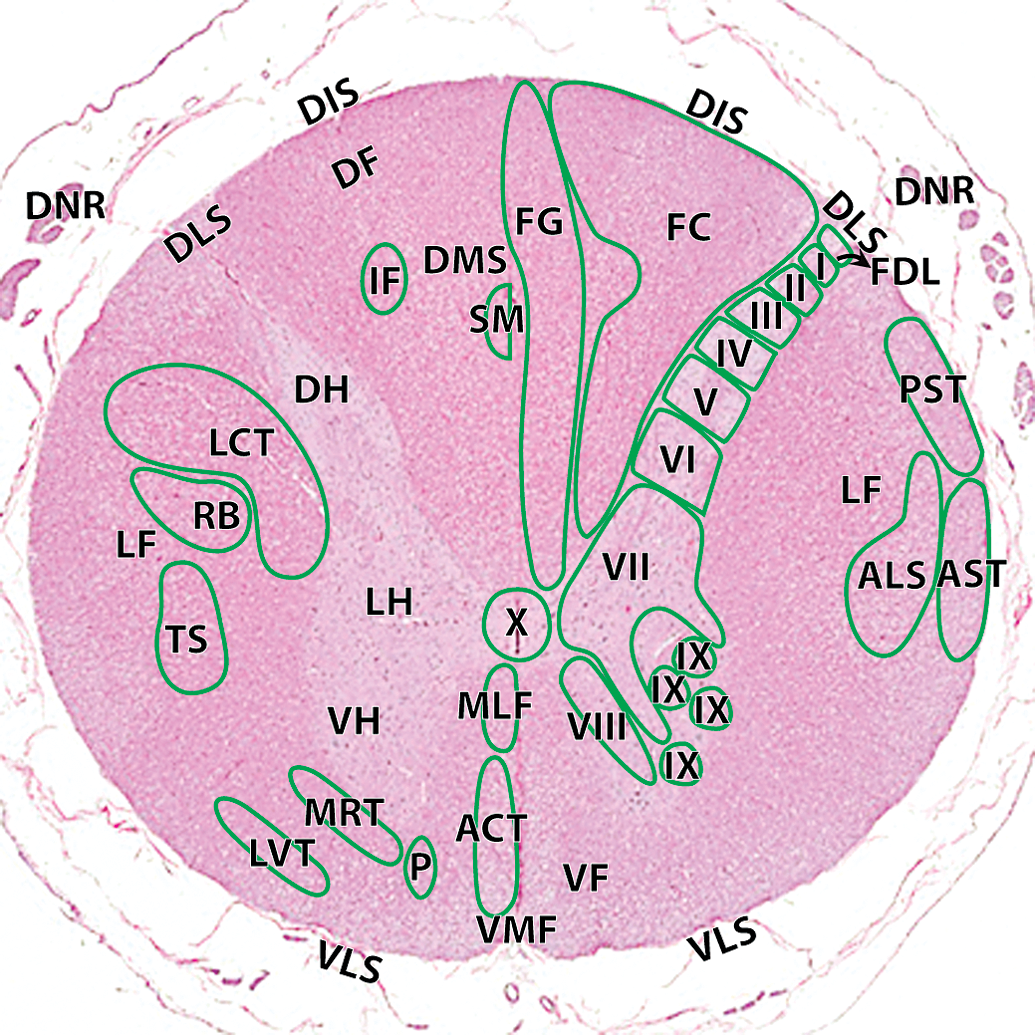
Hematoxylin and eosin cross–section of the cynomolgus money spinal cord at the level of C1–C2: ACT, anterior corticospinal tract; ALS, anteriolateral system; AST, anterior spinocerebellar tract; FC, fasciculus cuneatus; FDL, fasciculus dorsolateralis; FG, fasciculus gracilis; I, lamina 1; II, lamina II; III, lamina III; IV, lamina IV; V, lamina V; VI, lamina VI; VII, lamina VII; VIII, lamina VIII; IX, lamina IX; X, lamina X. Ascending tracts (right): MLF, medial longitudinal fasciculus; PST, posterior spinocerebellar tract. Descending tracts (left): DF, dorsal funiculi; DIS, dorsal intermediate septa; DH, dorsal horn; DLS, dorsolateral sulcus; DMS, dorsal median septum; DNR, dorsal nerve roots; IF, interfascicularis; LCT, lateral corticospinal tract; LH, lateral horn; LVT, lateral vestibulospinal tract; MRT, medullary (lateral) reticulospinal tract; P, pontine (medial) reticulospinal tract; RB, rubrospinal tract; SM, septomarginalis; VF, ventral funiculi; VH, ventral horn; VLS, ventrolateral sulcus; VMF, ventral median fissure.
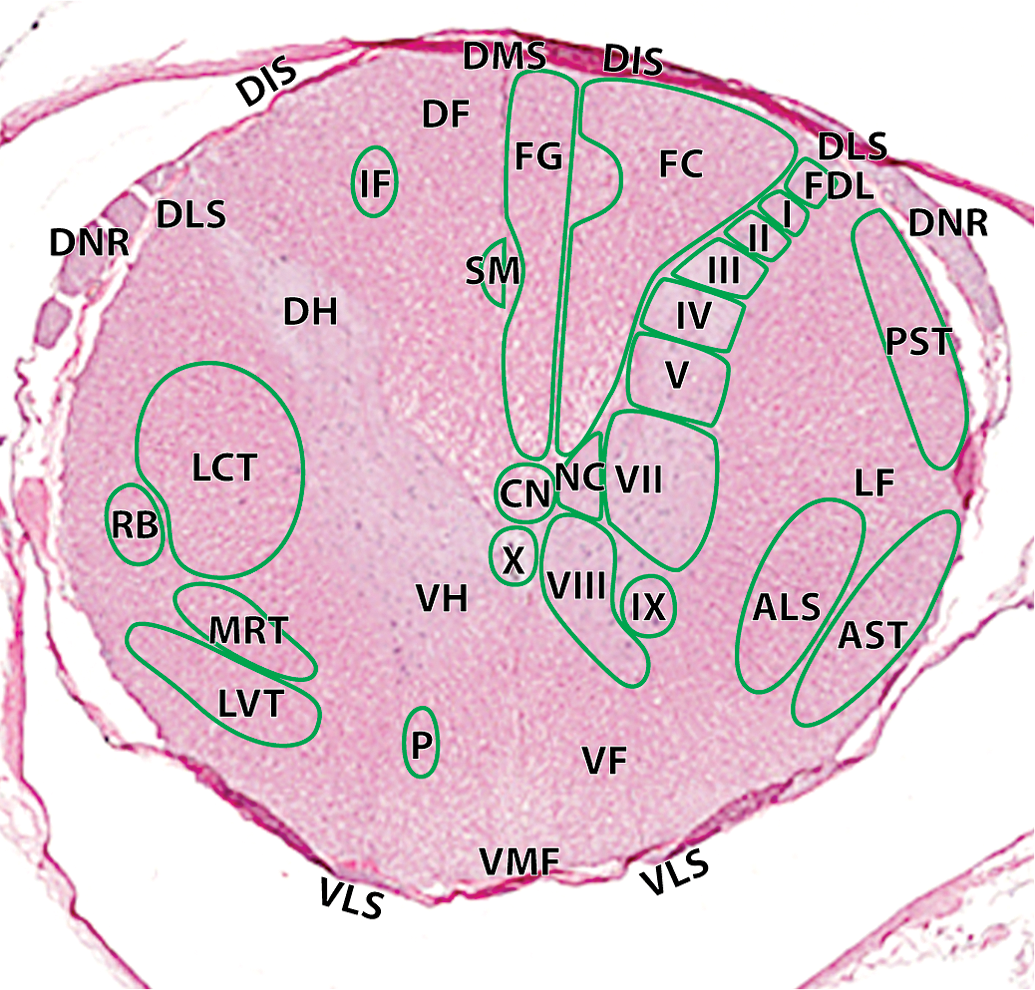
Hematoxylin and eosin cross-section of the cynomolgus money spinal cord at the level of T11–12: I, lamina I; II, lamina II; III, lamina III; IV, lamina IV; V, lamina V; VII, lamina VII; VIII, lamina VIII; IX, lamina IX; X, lamina X; NC, dorsal nucleus of Clarke. Ascending tracts (right): ALS, anteriolateral system; AST, anterior spinocerebellar tract; FC, fasciculus cuneatus; FDL, fasciculus dorsolateralis; FG, fasciculus gracilis; PST, posterior spinocerebellar tract. Descending tracts (left): CN, central canal; DF, dorsal funiculi; DH, dorsal horn; DIS, dorsal intermediate septa; DLS, dorsolateral sulcus; DMS, dorsal median septum; DNR, dorsal nerve roots; IF, interfascicularis; LCT, lateral corticospinal tract; LF, lateral funiculi; LVT, lateral vestibulospinal tract; MRT, medullary (lateral) reticulospinal tract; P, pontine (medial) reticulospinal tract; RB, rubrospinal tract; SM, septomarginalis; VF, ventral funiculi; VH, ventral horn; VLS, ventrolateral sulcus; VMF, ventral median fissure.
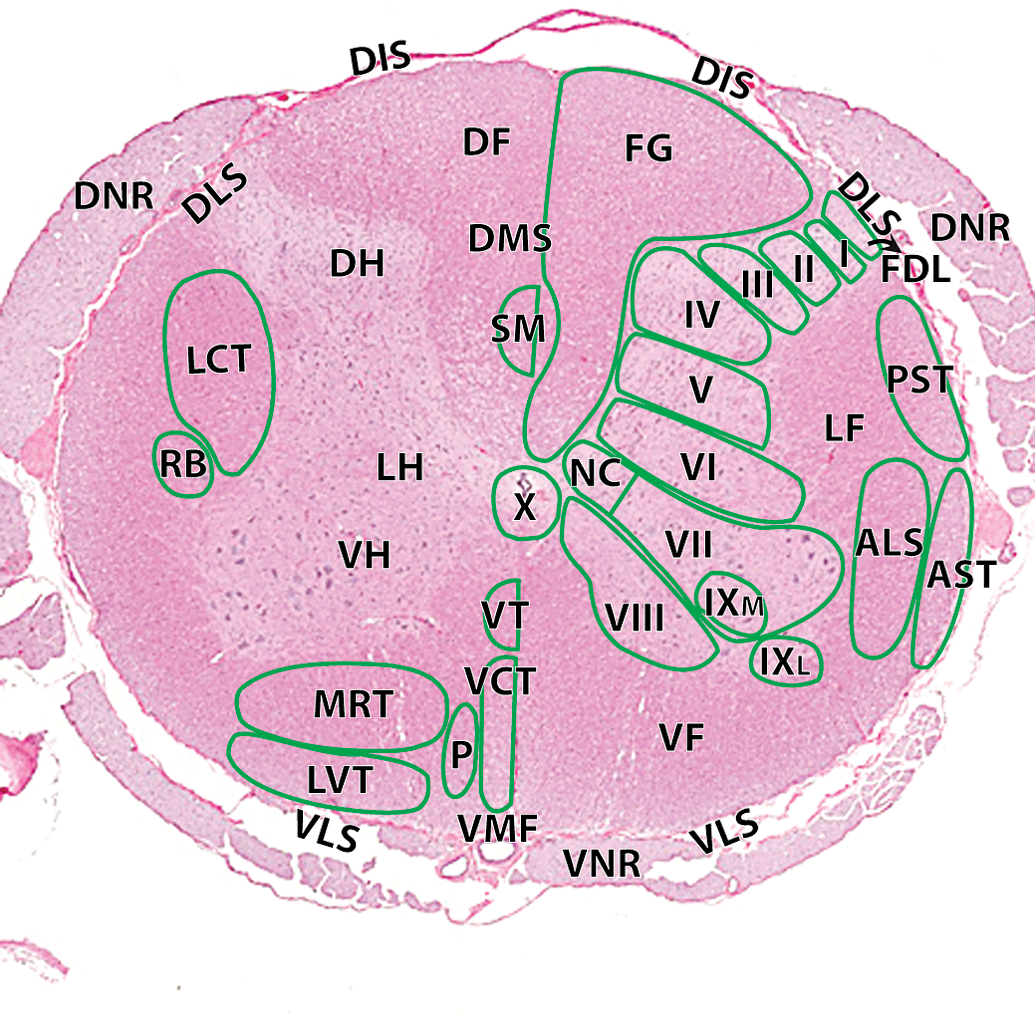
Hematoxylin and eosin cross-section of the cynomolgus monkey spinal cord at the level of L3–L4: I, lamina 1; II, lamina II; III, lamina III; IV, lamina IV; V, lamina V; VI, lamina VI; VII, lamina VII; VIII, lamina VIII; IXL, lamina IX lateral; IXM, lamina IX medial; X, lamina X. Ascending tracts (right): ALS, anteriolateral system; AST, anterior spinocerebellar tract; FG, fasciculus gracilis; FDL, fasciculus dorsolateralis; PST, posterior spinocerebellar tract. Descending tracts (left): DF, dorsal funiculi; DH, dorsal horn; DIS, dorsal intermediate septa; DLS, dorsolateral sulcus; DMS, dorsal median septum; DNR, dorsal nerve roots; LCT, lateral corticospinal tract; LF, lateral funiculi; LH, lateral horn; LVT, lateral vestibulospinal tract; MRT, medullary (lateral) reticulospinal tract; P, pontine (medial) reticulospinal tract; RB, rubrospinal tract; SM, septomarginalis; VCT, ventral corticospinal tract; VF, ventral funiculi; VH, ventral horn; VLS, ventrolateral sulcus; VMF, ventral median fissure; VNR, ventral nerve roots; VT, vestibulospinal tract.
Neuroanatomic Areas Included in the Brain Section at the Level of the Anterior Commissure (Figure 5)

Neuroanatomic Areas Included in the Brain Section at the Level of the Rostral Thalamus (Figure 6)

Neuroanatomic Areas Included in the Brain Section at the Level of the Caudal Thalamus (Figure 7)

Neuroanatomic Area Included in the Section at the Level of the Middle of the Cerebellum and Brainstem (Figure 8)

Neuroanatomic Area Included in the Section at the Level of the Occipital Lobe (Figure 9)

The Spinal Cord
General Descriptive Anatomy of the Cynomolgus Monkey Spinal Cord
The spinal cord in cynomolgus monkeys, as in humans, is composed of eight cervical, twelve thoracic, five lumbar, and five sacral segments, and one caudal (coccygeal) segment with their respective paired nerve roots. The spinal cord has two intumescences (one at each of the C5–T1 and L4–S2 levels) that correspond to larger numbers of neurons that innervate the skin, joints, and muscles of the limbs. The cervical nerves (C1–C7) emerge from the vertebral column rostral to the vertebra with the same number, whereas nerve C8 emerges rostral to the first thoracic vertebra. The remaining spinal nerves emerge caudal to the corresponding vertebrae.
The spinal cord in cynomolgus monkeys consists of central gray matter covered by white matter and surrounded by meninges (the dura mater, arachnoid, and pia mater). The center of the spinal cord holds the gray matter that contains symmetrical pairs of dorsal (posterior) and ventral (anterior) horns. The different levels of the spinal cord can be distinguished based on size, shape, and relative proportions of the white and gray matter.
Groups of myelinated axons that transmit sensory or motor information along the length of the spinal cord are located within three pairs of funiculi within the white matter. Within each funiculus, related groups of axons are bundled into tracts or fascicles that convey similar types of information. The posterior (dorsal) funiculi (medial to the dorsal root entry zones) contain fiber bundles that transmit somatosensory information to the brain. The lateral (between the dorsal root entry and the ventral root exit zones) and anterior (ventral; medial to the ventral root ext zones) funiculi have both ascending (sensory) and descending (motor) nerve tracts.
There are three types of fiber tracts: intersegmental, ascending, and descending. Intersegmental tracts project rostrally or caudally in the propriospinal tract that surrounds the outer edge of the central gray region, and these projections coordinate the different muscle groups that mediate spinal reflexes. The ascending tracts convey sensory information to the thalamus, cerebellum, or brainstem. Descending projections enable the cerebral cortex and brainstem to communicate with the spinal cord so that the brain can execute motor commands or modulate sensory transmission.
Cervical Levels
Cross-sections of the cervical cord have an oval shape. The funiculi in the cervical sections are large and are formed by the large number of ascending sensory tracts from the animal’s body and the descending motor tracts innervating groups of motor neurons. In addition, the anterior (ventral) horns are enlarged in sections C5–C8, because they contain additional motor neurons that innervate the upper limbs.
Thoracic Levels
Cross-sections of the thoracic cord are smaller than those of the cervical and lumbar cord, because the central gray region requires fewer neurons (which innervate the intercostal muscles of the trunk). Within the small lateral horns that protrude into the lateral funiculi, there is the intermediolateral cell column, which is composed of preganglionic sympathetic neurons of the autonomic system. All thoracic segments contain the dorsal nucleus of Clarke, which is located at the base of the dorsal horn and contains larger neurons. These nuclei are more prominent in the T10–T12 segments. In addition, the fasciculus cuneatus is present from T1–T6.
Lumbar Levels
Cross-sections of the lumbar cord have larger horns with a relatively smaller portion of white matter. The fasciculus gracilis is relatively narrowed compared to the thoracic cord. L1 and L2 have prominent lateral horns and dorsal nuclei of Clarke, but these structures are absent in L3–L5. The larger size of the anterior (ventral) horn is a result of the addition of motor neurons that innervate the muscles of the lower limbs.
Sacral and Coccygeal Levels
Sacral cord sections are characterized by small amounts of white matter and a relatively large proportion of gray matter. They have a thick gray commissure in the center of the cord. Caudal sections of the sacral cord become progressively thinner. The caudal segment is similar to sacral segments, but it is smaller in diameter.
Collection, Sampling, and Sectioning of the Cynomolgus Monkey Spinal Cord
Because some nuclei and fiber tracts are found only in certain levels of the spinal cord, at least one cross-section at the levels of the cervical region (near the brainstem), caudal thoracic region, and lumbar region should be sectioned and examined. The corresponding dorsal root ganglion and nerve roots should also be collected. The inclusion of a cervical cord section supports the evaluation of the distal portions of ascending tracts that pass through to the brainstem. Examination of lumbar sections provides for the analysis of the distal portions of descending tracts of the spinal cord. In a number of types of nerve fiber degeneration, the distal portions of the nerves may be the first to show morphologic changes detectable via light microscopic examination.
Some examples of specific nuclei and tracts occurring in cervical and lumbar cord are:
The intermediate septum is only present in the cervical and upper thoracic segments (C1–T6)
The fasciculus cuneatus is only present in the C1–T6 level sections.
The tectospinal tract is only present from C1–C4.
The sympathetic preganglionic neurons are located in the lateral horn of the spinal cord between segments T1 and L3. (Note that if parasympathetic alterations are suspected, sections of S2–S4 should be obtained.)
The medial longitudinal fasciculus is present only in sections of cervical cord.
An efficient method for sectioning the spinal cord for general toxicity studies in cynomolgus monkeys can be accomplished by performing a total or partial laminectomy. After removing the skin, spinal muscles, and adipose and connective tissues, the dorsal and transverse processes of the vertebrae can be removed using a bone saw or rongeur (World Precision Instruments, Sarasota, FL), being careful not to damage the cord. The entire cord (complete laminectomy) or segments at the levels of C1–C4, T10–T12, and L1–L4 (partial laminectomy) can be removed, along with the associated spinal nerve roots and corresponding dorsal root ganglion. The meninges can be used to stabilize and exert traction on the spinal cord during removal. Doing so will prevent mechanical destruction of the spinal cord parenchyma. Cross-sections of the spinal cord provide neuroanatomic orientation (Figure 10A). Oblique sections of the spinal cord (Figure 10B) provide an analysis of the funiculi in a semilongitudinal fashion, and lateral or longitudinal sections will improve optimal evaluation of nerve fiber degeneration. One single cross-section of each segment (C1–C4, T10–T12, and L1–L4) placed in one cassette, including the nerve roots with dorsal root ganglion (if possible), can provide an adequate analysis of most neural pathways of the spinal cord (Figure 10C). For general toxicity studies, staining of selected sections of cord can be limited to hematoxylin and eosin (H&E), although special chemical and immunohistochemical stains such as Luxol fast blue, glial fibrillary acidic protein, Iba1, calbindin, and silver techniques such as Bodian’s or Bielschowsky’s (to name just a few) can be performed to better characterize findings observed by routine light microscopic examination. Figures 11–13 depict neuroanatomic areas in the cervical, thoracic, and lumbar cord segments. Tables 8 and 9 briefly describe functions and neuroanatomy of the spinal cord segments shown in Figures 11–13.
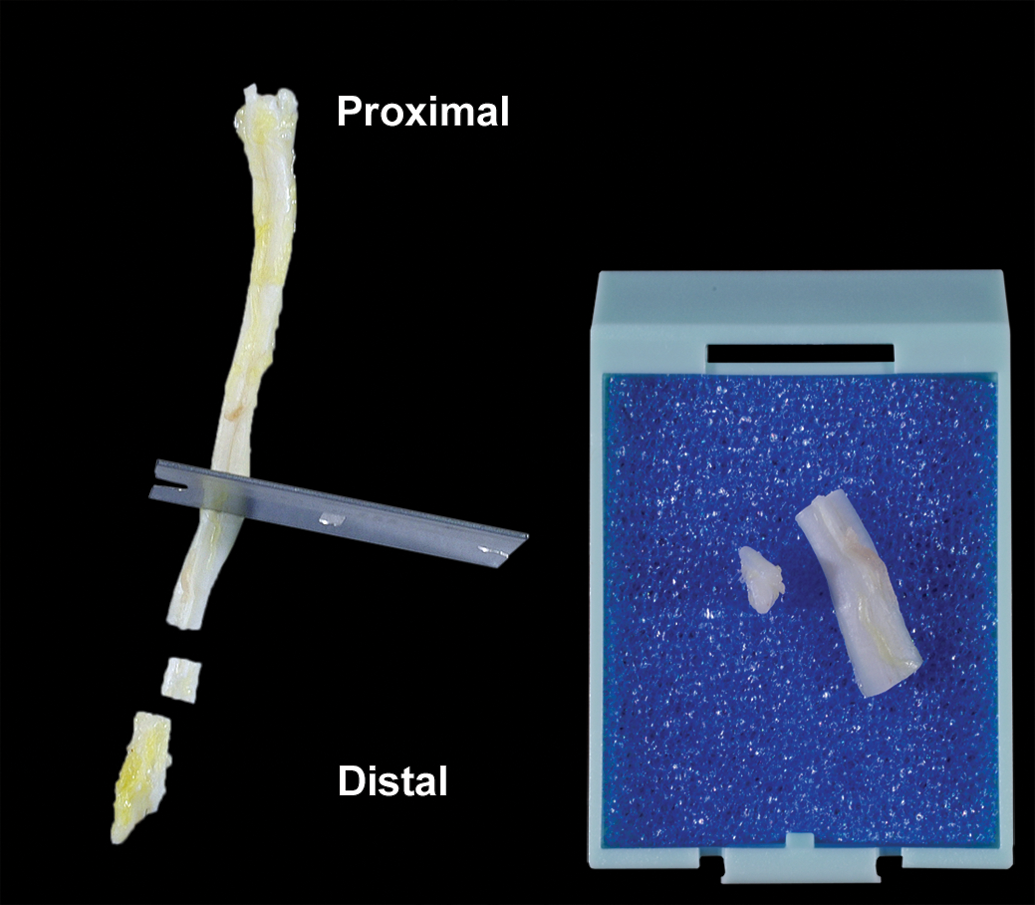
Longitudinal and cross-sections of the sciatic nerve placed in the cassette.
Neuroanatomic Areas Included in the Gray Matter of the Spinal Cord (Figures 11, 12, and 13)


Ascending and Descending Tracts in the White Matter of the Spinal Cord


Functional neuroanatomy of the brain areas included in Tables 2–7 (Figures 4–9) were obtained from Afifi and Bergman 2005; Alloway and Pritchard 2007; Crosby et al. 1962; Hendelman 2005; and Nolte 2009.
Functional neuroanatomy of the spinal cord included in Tables 7 and 8 (Figures 10–12) were obtained from Alloway and Pritchard 2007; Crosby et al. 1962; Hendelman 2005; and King 1999.
The Peripheral Nerve
General Descriptive Anatomy of the Cynomolgus Monkey Peripheral Nerve
Peripheral nerves are responsible for afferent and efferent communication between the body and the central nervous system. The cell bodies of afferent fibers are located in the dorsal root ganglia and are referred to as pseudounipolar neurons. The efferent fibers of the ventral nerve roots originate from cell bodies located in the lateral or ventral horns of the spinal cord. After leaving the spinal cord, these efferent fibers merge with distal processes of the afferent fibers to form a mixed spinal nerve; however, efferent fibers originating from motor neurons in lamina IX proceed to skeletal muscle and terminate in neuromuscular junctions.
The brachial and sciatic nerves are formed by the spinal nerves C5–T1 and L4–S3, respectively, although variation in the origin of the brachial nerve was observed in one monkey by the first author. The brachial nerve is responsible for cutaneous and muscular innervation of the entire upper limb, with the exceptions of the trapezius and intercostobrachial muscles, which are innervated by the spinal accessory nerve and intercostobranchial nerves, respectively. The brachial nerve gives origin to the axillary, radial, and ulnar nerves, as well as to other important nerves. The sciatic nerve innervates the skin of the legs, muscles of the back of the thigh, and leg and foot muscles such as the biceps femoris, semitendinosus, semimembranous, and adductor magnus. The sciatic nerve branches to form the tibial and common peroneal nerves.
Collection, Sampling, and Sectioning of the Cynomolgus Monkey Peripheral Nerve
For most general toxicity studies, the evaluation of longitudinal and cross-sections of the sciatic nerve from one of the legs is adequate to examine the peripheral nerve system (Figure 14). The section can be taken at the mid-thigh level, just prior to the point of branching of the sciatic nerve into the tibial and peroneal nerves. The longitudinal sample should be ≥ 1 cm in length and fit in a standard cassette. It is highly recommended to mark with ink the proximal versus the distal aspect of the nerve to keep the nerve orientation at sectioning. Examination of the sciatic nerve of both legs would be recommended if local injection site trauma to the nerve is suspected. Additional peripheral nerves such as the tibial, femoral, sural, and brachial nerves may be collected if clinical signs suggest peripheral nerve dysfunction.
Conclusions
There is general agreement among neuropathologists that a greater degree of brain sampling is necessary to adequately perform safety evaluations. The technique described in this paper provides a consistent way to sample and evaluate the nervous system of cynomolgus monkeys in general toxicity studies. This method is readily adaptable to species other than nonhuman primates, such as dogs and minipigs, and this method can readily be learned by prosectors and histotechnicians with relatively basic training in neuroanatomy.
Footnotes
Acknowledgments
The authors thanks the technical staff in DSRD Pfizer, Inc., Groton, CT composed of Mrs. Catherine A. Tabor, Ms. Angela Aleksa, Ms. Natalka Kopcyk, Mrs. Denise Pernal, Ms. Stacy Kwasniewski, Ms. Laura Kearney, and Mrs. Kim Kowsz for their excellent technical support with necropsies and possessing histopathologic slides used in this publication and CDROM. In addition, the authors want to thank Mr. David Sabio at EPL for his contribution in art and design of the CDROM of the Cynomolgus Monkey Nervous System Trimming Protocol.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
