Abstract
Although manuscripts for multiple species recommending nervous system sampling for histopathology evaluation in safety assessment have been published in the past 15 years, none have addressed the laboratory rabbit. Here, we describe 2 trimming schemes for evaluating the rabbit brain in nonclinical toxicity studies. In both schemes, the intact brain is cut in the coronal plane to permit bilateral assessment. The first scheme is recommended for general toxicity studies (tier 1) in screening agents where there is no anticipated neurotoxic potential; this 6-section approach is consistent with the Society of Toxicologic Pathology (STP) “best practice” recommendations for brain sampling in nonrodents (Toxicol Pathol 41: 1028-1048, 20131). The second trimming scheme is intended for dedicated neurotoxicity studies (tier 2) to characterize known or suspected neurotoxicants where the nervous system is a key target organ. This tier 2 strategy relies on coronal trimming of the whole brain into 3-mm-thick slices and then evaluating 12 sections. Collection of spinal cord, ganglia, and nerve specimens for rabbits during nonclinical studies should follow published STP “best practice” recommendations for sampling the central nervous system1 and peripheral nervous system (Toxicol Pathol 46: 372-402, 20182).
Keywords
Introduction
Rabbits (Oryctolagus cuniculus) are widely used as animal models for investigational basic research as well as in safety evaluation studies during the development of new therapeutic products (including small molecules, biologics/vaccines, and medical devices). Common roles for rabbits in biomedical research include production of antibodies, 3 pyrogen testing, 4 evaluation of novel test articles such as vaccines 5 and medical devices, 6 and as animal models for exploring cardiovascular, 7,8 infectious, 9,10 neurological, 3,11 and ocular 3 diseases. The White New Zealand (sometimes called the New Zealand White) rabbit is a generally accepted animal model for safety studies, principally for testing developmental toxicity, 12 reproductive toxicity, 13 local irritation, 14 and vaccine-induced reactivity. 5 Brains of rabbits and rodents share many neuroanatomic features—modest size (adult brain weights of 4 g and 10-12 g, respectively, for rats and rabbits); a lissencephalic (smooth) cerebrocortical surface; prominent olfactory bulbs; and a small forebrain-to-hindbrain-size ratio relative to other nonrodent test species. 15 Nonetheless, rabbits are lagomorphs, not rodents, and therefore can be utilized as a nonrodent species in nonclinical toxicity testing.
Multiple recent publications have given detailed recommendations for sampling (and often processing) the central and/or peripheral nervous systems (CNS and PNS, respectively) in rodent and nonrodent species to facilitate toxicologic pathology evaluation in nonclinical toxicity studies. 1,2,16 –24 In terms of nonrodents, these publications have emphasized sampling in dogs and nonhuman primates (NHPs), which are common test species for nonclinical safety assessment, and sometimes mention other species used to assess specific neurotoxicity questions (eg, chickens, pigs). Importantly, therefore, no existing publications specifically address optimal practices for sampling and processing the nervous system in rabbits for toxicologic pathology evaluation.
The main objective of the current article was to describe 2 trimming schemes for the rabbit brain—one for general toxicity studies (tier 1) that screen test articles without neurotoxic potential and the other for dedicated neurotoxicity studies (tier 2) that characterize suspected or known neurotoxic effects. The subsequent objective was to provide guidance on the relevance of neuroanatomical areas within the 2 trimming schemes to allow for meaningful histopathologic evaluation. Although information on sampling and processing additional CNS and PNS components of the rabbit nervous system is not the primary focus of this article, detailed information on collecting spinal cord, ganglia, and nerves is included and referenced as a resource for the reader. The information provided herein should be useful to toxicologists, pathologists, and biomedical research scientists involved in designing and conducting rabbit toxicology studies as well as histotechnicians and technical experts involved in precise tissue trimming and processing procedures.
Materials and Methods
Animals
All procedures were approved in advance by the Institutional Animal Care and Use Committee (IACUC) in accordance with US federal regulations 25 and the Guide for the Care and Use of Laboratory Animals. 26 In keeping with current 3Rs (replace, reduce, refine) principles for animal-based research, preparation of brains sampled for this article represented a satellite use of unneeded tissue from animals employed for another IACUC-approved project.
Naive young adult, female WNZ rabbits (N = 10) were procured from Envigo Global Services, Inc. Animals were housed individually in stainless steel cages (area = 4900 cm2, height = 46 cm). All cages held autoclaved corncob bedding (Andersons Lab Bedding Products). Animals received autoclaved pelleted rabbit chow (Certified Rabbit LabDiet 5322; PMI Nutrition International, LLC) and municipal chlorinated tap water (treated by reverse osmosis and ultraviolet radiation) ad libitum. Cages were kept in biologically clean rooms with HEPA (high-efficiency particulate air)-filtered air and a 12-hour light/dark cycle. Room temperature (RT) and relative humidity were maintained at 19 °C ± 3 °C and approximately 50%, respectively. Cages and bedding were autoclaved before weekly changes.
Tissue Acquisition and Processing
Animals were euthanized humanely using intravenous sodium pentobarbital. Brains were exposed by removing the dorsal calvarium and dura mater. Brain attachments (cranial nerves and spinal cord) were severed, and the brain was removed and fixed while intact by immersion in 10 to 20 volumes of neutral buffered 10% formalin (NBF, pH 7.4) at RT for at least 24 hours; this commercial NBF formulation contained approximately 1% methanol as a stabilizing agent. Specimens were fixed for approximately 5 days at RT before trimming.
Trimming planes were oriented using major landmarks on the dorsal (Figure 1A) and ventral (Figure 1B) surfaces of the brain. An initial cut to divide the fixed brain in half was made using the optic chiasm and infundibulum to position the blade (Figure 2). Subsequently, a purpose-made rectangular brain matrix developed in-house by Pfizer for acquiring coronal slices of NHP brains was adapted for use with rabbit brains. Using this tool (15-cm long, 8-cm wide, with 2 outer raised edges that are 1-cm wide and 3-mm high; see Figure 3A) and ventral external landmarks, a total of 15 full coronal slices were generated from each rabbit whole brain (Figures 2 and 3B, where Figure 3B shows 6 representative slices from the rostral half of the brain). The cutting blade used to make these slices was a Stadie-Riggs Tissue Slicer (Cat. No. 6727C18; Thomas Scientific). Each full coronal brain section was small enough (in size and thickness) to be placed intact in its own cassette. The rostral aspect of each brain slice was placed “down” in the cassettes. Brain slices then were processed routinely into paraffin. Five-micrometer-thick sections were obtained for each block and stained with hematoxylin and eosin (H&E).
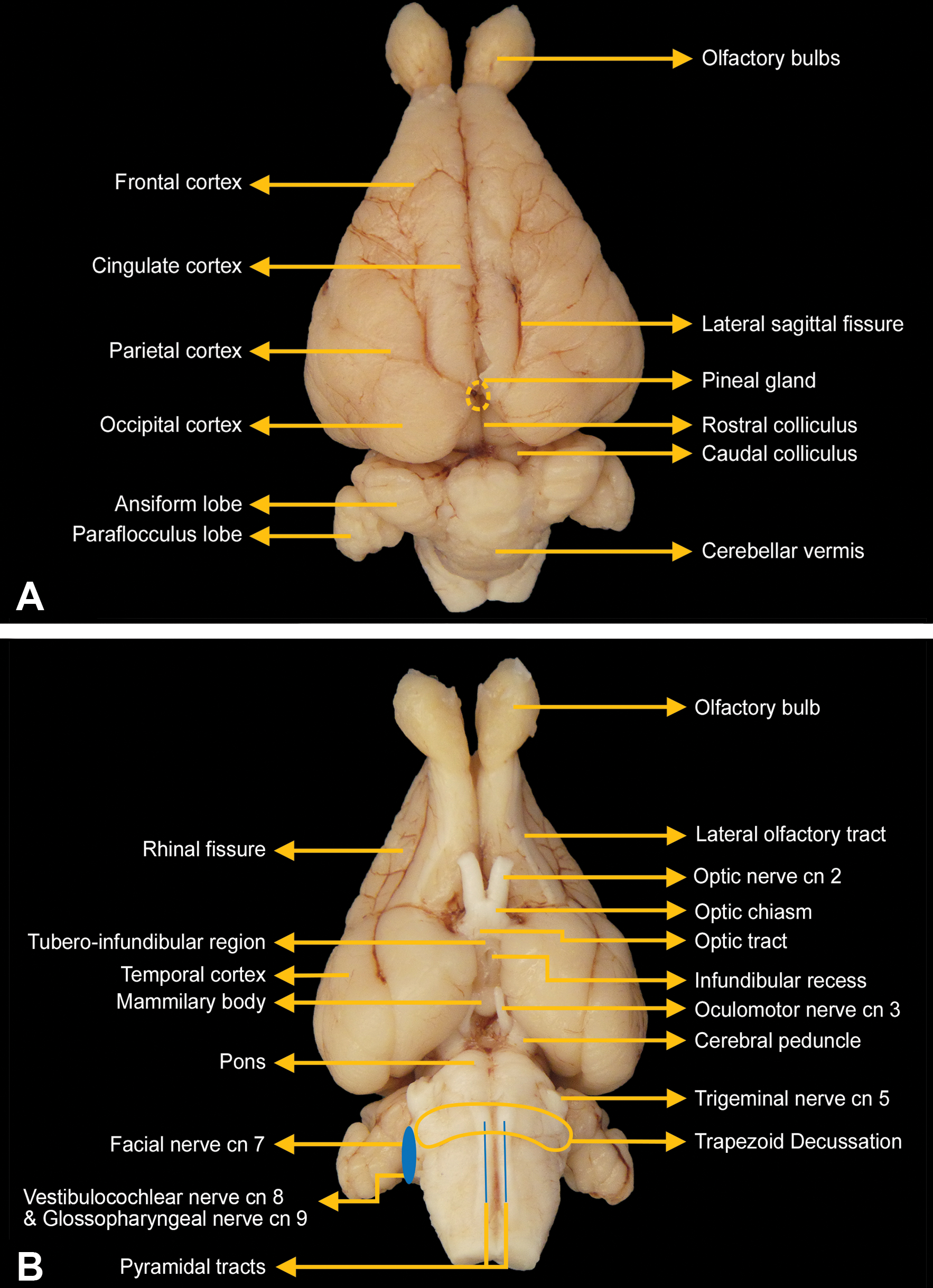
External landmarks on the dorsal (A) and ventral (B) surfaces of the rabbit brain. These structures are useful for orienting blade placement when trimming the brain.
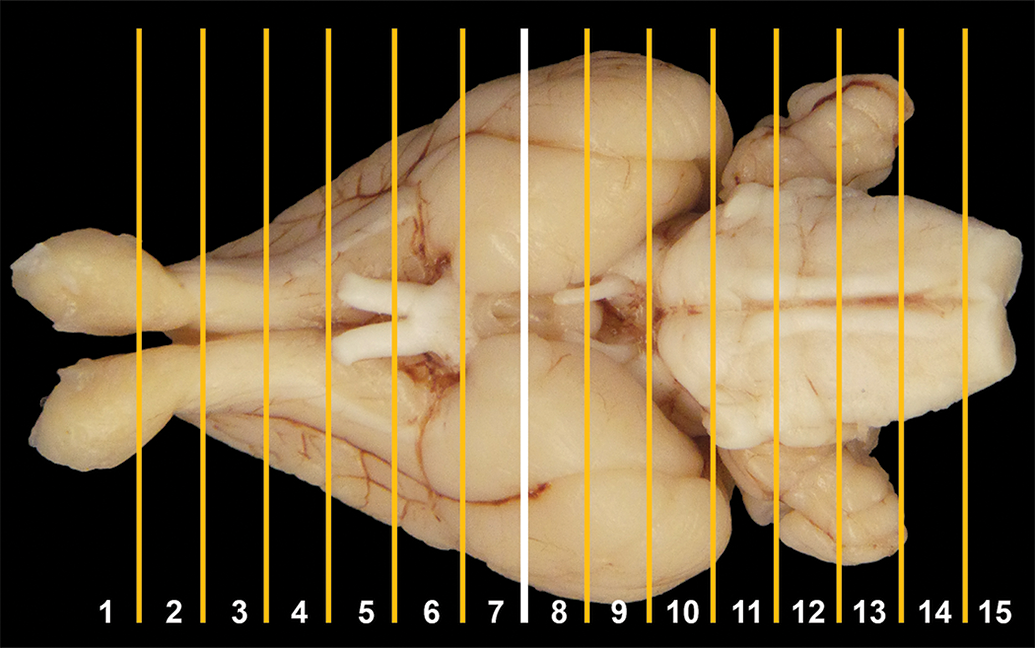
Location of trimming planes for rabbit brain sectioning, ventral view. An initial cut (white line) to divide the fixed brain in half is made using the caudal aspect of the optic chiasm and infundibulum, after which additional 3-mm-thick coronal cuts (yellow lines) are made rostrally and caudally to obtain a total of 15 slices.

A, Schematic diagram of a rectangular fiber glass brain matrix to prepare coronal brain slices. This tool is 15-cm long, 8-cm wide, and has 2 outer raised edges that are 1-cm wide and 3-mm high. [This image is reproduced from “Technical guide for nervous system sampling of the cynomolgus monkey” [Pardo et al. 2012 20 ] with the permission of Sage Publications]. B, The flat surface of the fixed brain half (ie, a surface at the white line in Figure 2) is placed “cut surface down” in the matrix between the 2 outer raised edges. The Tissue Slicer Blade resting on the raised edges is used to produce fifteen 3-mm-thick brain slices (of which 5 from the rostral portion of the brain are shown here).
Neuroanatomic Annotation
The figures included in the present atlas are representative images for structures that might be observed in sections made from the rostral face of the 15 brain slices shown in Figures 4A and 4B. Brain structures were identified by comparison of rabbit features to those of similar regions in other species 27 –29 as well as by reference to recently published, detailed stereotaxic rabbit brain atlases generated via magnetic resonance imaging 30 –32 and out-of-print rabbit stereotaxic brain atlases 15,33 –37 available to the authors.
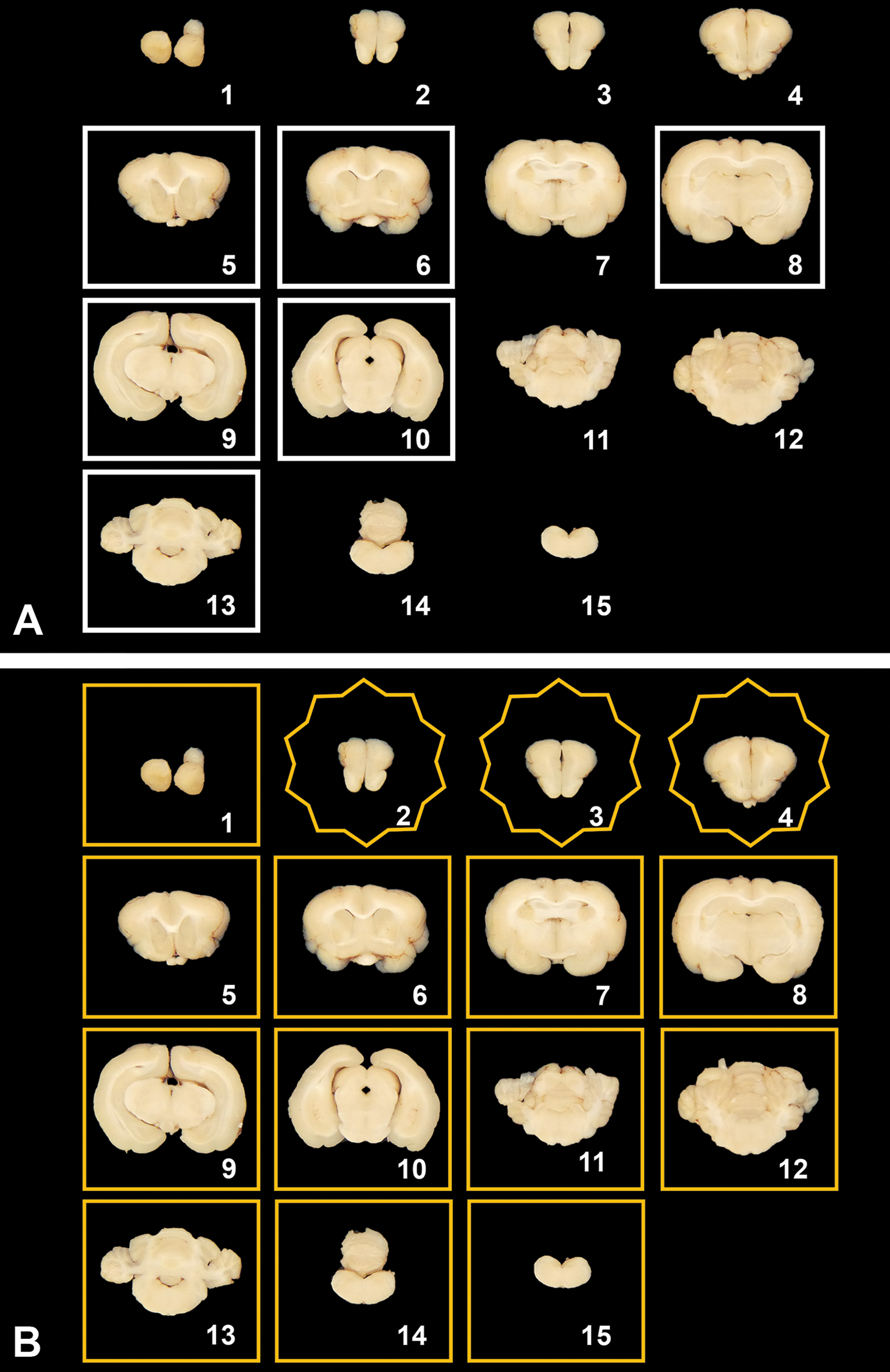
Brain trimming schemes for use in general toxicity (tier 1, A) and dedicated neurotoxicity (tier 2, B) studies. For both schemes, the fixed brain is sliced into 15 coronal sections. For general toxicity screening (A), a subset of 6 sections (framed by white boxes) are selected at the levels of the frontal pole (No. 5), rostral (anterior) commissure (No. 6), mid-thalamus (No. 8), caudal thalamus (No. 9), occipital lobe and midbrain (No. 10), and cerebellum and brainstem (No. 13). For dedicated neurotoxicity studies (B), all 15 sections or a subset of 12 (reached by excluding sections Nos. 2, 3, and 4 [10-pointed stars]) are evaluated. For both trimming schemes, the rostral face of each slice (shown here) is placed “down” in the cassette and paraffin boat to ensure that this plane is available for evaluation.
Neuroanatomic terminology conforms to the accepted Nomina Anatomica Veterinaria standard for quadruped animals. 38 Where considered to be helpful for medically trained researchers, the accepted medical nomenclature for certain structures as applied to humans is also included as parenthetical entries associated with the correct veterinary neuroanatomic term.
Results
As in all other species, nervous system sampling in the rabbit for nonclinical safety testing should address both CNS and PNS components. This procedure for rabbit brain sampling was adapted from published Society of Toxicologic Pathology (STP) “best practice” recommendations developed for nonrodents. 1 Harvesting the spinal cord (CNS) as well as ganglia and nerves in rabbits should follow the published STP “best practice” recommendations for CNS and PNS sampling and processing that are applicable to all species. 1,2
Brain
Two trimming schemes were devised based on the divergent sampling needs for 2 different categories of nonclinical safety studies. The first scheme applies to general toxicity studies used for screening test articles with no or unknown potential for neurotoxicity (tier 1) and closely follows the design for nonrodent test species as presented in the recently published STP “best practice” recommendations for brain sampling 1 (Figure 4A). The second scheme is for dedicated neurotoxicity studies to characterize test articles with anticipated or known neurotoxic capacity (tier 2), thus indicating that one or more components of the nervous system are target sites (Figure 4B). Principal neuroanatomic areas available for evaluation in each section are listed in Table 1 (for the general toxicity trimming scheme—tier 1) and Table 2 (for the dedicated neurotoxicity scheme—tier 2). Key functions and selected neural connections for the principal brain areas seen in the sections from these 15 brain slices are reviewed briefly in Online Appendix 1.
Major Landmarks Available for Evaluation in the 6-Section (Tier 1) Brain Trimming Schemea for General Toxicity Studies.
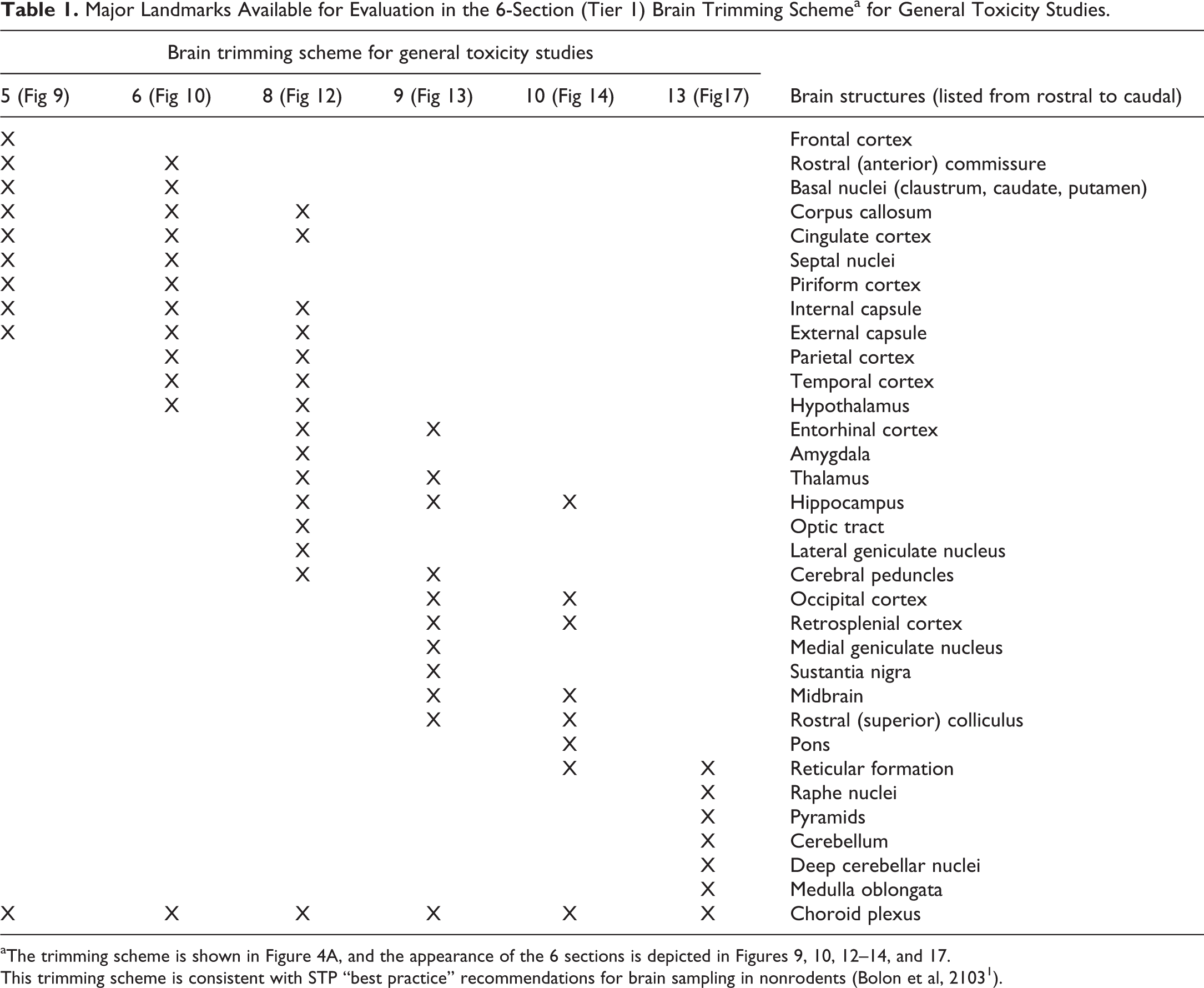
aThe trimming scheme is shown in Figure 4A, and the appearance of the 6 sections is depicted in Figures 9, 10, 12 –14, and 17.
This trimming scheme is consistent with STP “best practice” recommendations for brain sampling in nonrodents (Bolon et al, 2103 1 ).
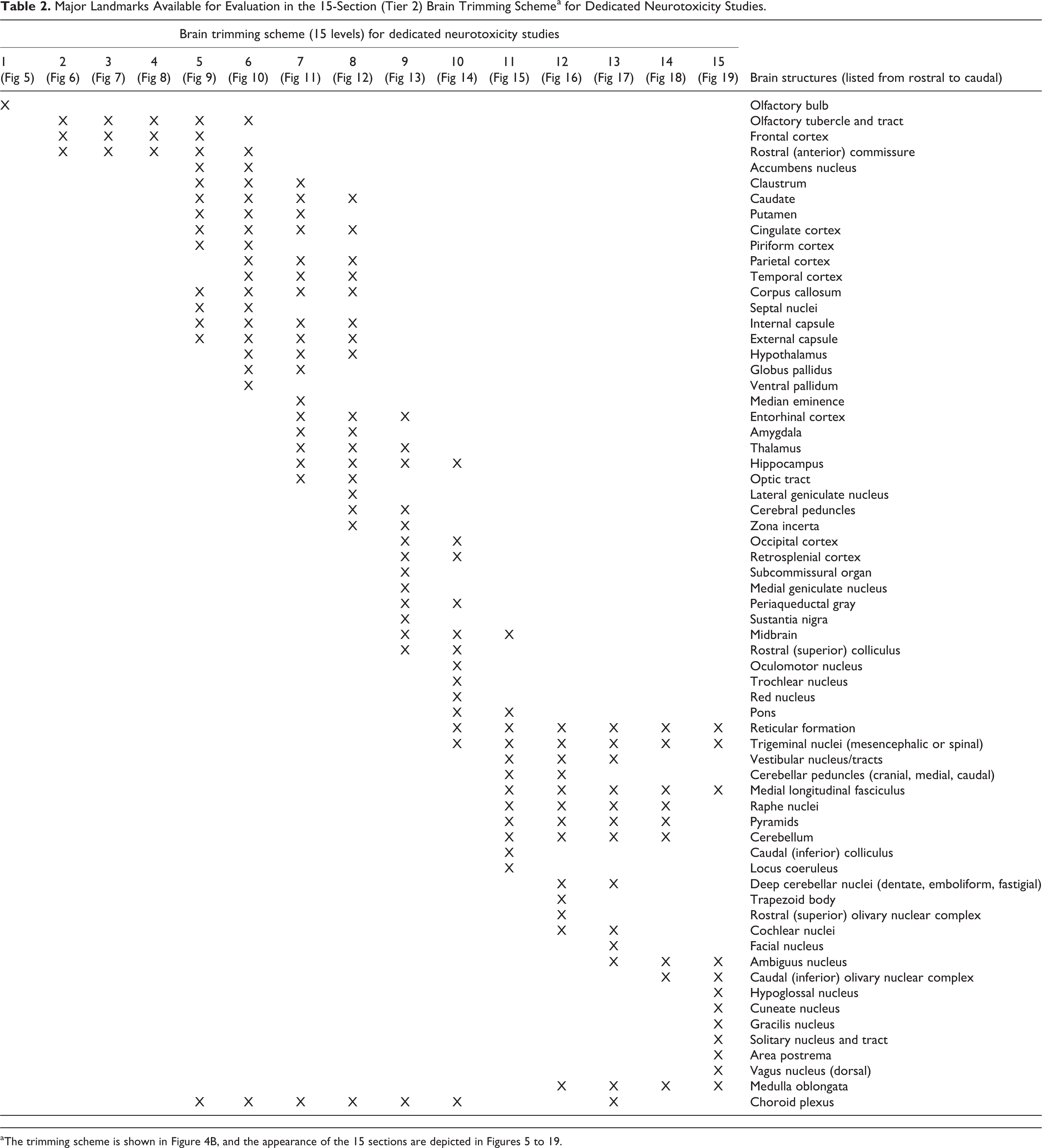
Major Landmarks Available for Evaluation in the 15-Section (Tier 2) Brain Trimming Schemea for Dedicated Neurotoxicity Studies.
General Toxicity Studies
The brain trimming scheme for screening studies encompasses 6 full coronal sections selected to show major areas known to be targeted by proven neurotoxicants. These sections represent a subset of the 15 coronal sections needed to fully trim the rabbit brain (Figure 4A). The specified trimming planes are taken through the frontal cortex (level 5), rostral (anterior) commissure (level 6), rostral thalamus (level 8), midbrain (caudal thalamus; level 9), pons and occipital cortex (level 10), and mid cerebellum overlying the medulla oblongata (level 13). Full coronal sections were chosen for screening (rather than the coronal hemisections necessary for larger nonrodent species) since (1) conventional tissue cassettes and glass slides readily accommodate complete cross sections of rabbit brain and (2) bilateral comparison of major structures facilitates analysis and interpretation of histopathologic changes in this organ.
Neurotoxicity Studies
The brain trimming scheme for characterizing known or suspected neurotoxicants targeting any component of the nervous system ideally should include an evaluation of all 12 full coronal sections (Figure 4B). This more detailed approach acknowledges that more brain sampling will be needed for adequate assessment when neurotoxicity is likely. 1,19,22,39 Such instances include when known or suspected target areas for potential neurotoxicity with the test article are available in published literature, when the target organ directly includes any specific component of the nervous system, when limited clinical observations from early acute studies in other animal species suggest neurotoxicity, when data in early human trials (rarely available) hint at potential neurotoxicity, or when extrapolation from other test articles with a putatively similar QSAR (quantitative structure–activity relationship) profile suggests a potential for neurotoxicity.
Possible Adjustments to Trimming Schemes
The 6-section trimming scheme for general toxicity studies represents the minimally acceptable sample for rabbit brain evaluation in nonclinical safety assessment. A permissible adaptation to this scheme would be to perform the histopathologic examination on 6 hemisections rather than 6 full sections, a strategy equivalent to the “best practice” recommendations applicable to other nonrodent species 1 as this ensures at least one full half of the brain at the recommended trimming planes will be evaluated. In general, this hemisection adaptation should be implemented for rabbits only if the study protocol has specified collection of unfixed tissue from the contralateral brain regions from the same animal to permit other analyses (eg, molecular events related to test article activity or measurements of test article distribution).
Adjustments to the 12-section trimming scheme for neurotoxicity studies might be performed at the discretion of the study design team and/or institution under certain situations. For example, for inhalation studies or when olfactory neurotoxicity is suspected, all 15 full coronal sections might be included in the evaluation since the olfactory brain is concentrated in the olfactory bulbs (level 1) and ventral forebrain (levels 2-4). In some instances, evaluation of fewer than 12 full coronal sections might be justified scientifically based on the choice to not review a particular subset of neuroanatomic structures where effects already are known to occur or where no unique neuroanatomic regions may be seen. In this context, the trimming levels to be omitted would be chosen on a case-by-case basis.
Principal Neuroanatomic Features of the Rabbit Brain
The appearance of the 12 to 15 (adding the extra 3 forebrain sections for inhalation studies) full coronal rabbit brain sections produced by the tier 2 (dedicated neurotoxicity) trimming scheme are depicted in Figures 5 to 19. The major neuroanatomic regions within each section are annotated. Due to spatial constraints, annotations are given only for the largest and/or most biologically and toxicologically significant structures (as judged based on known function and known sensitivity to neurotoxicants [see Online Appendix 1]).
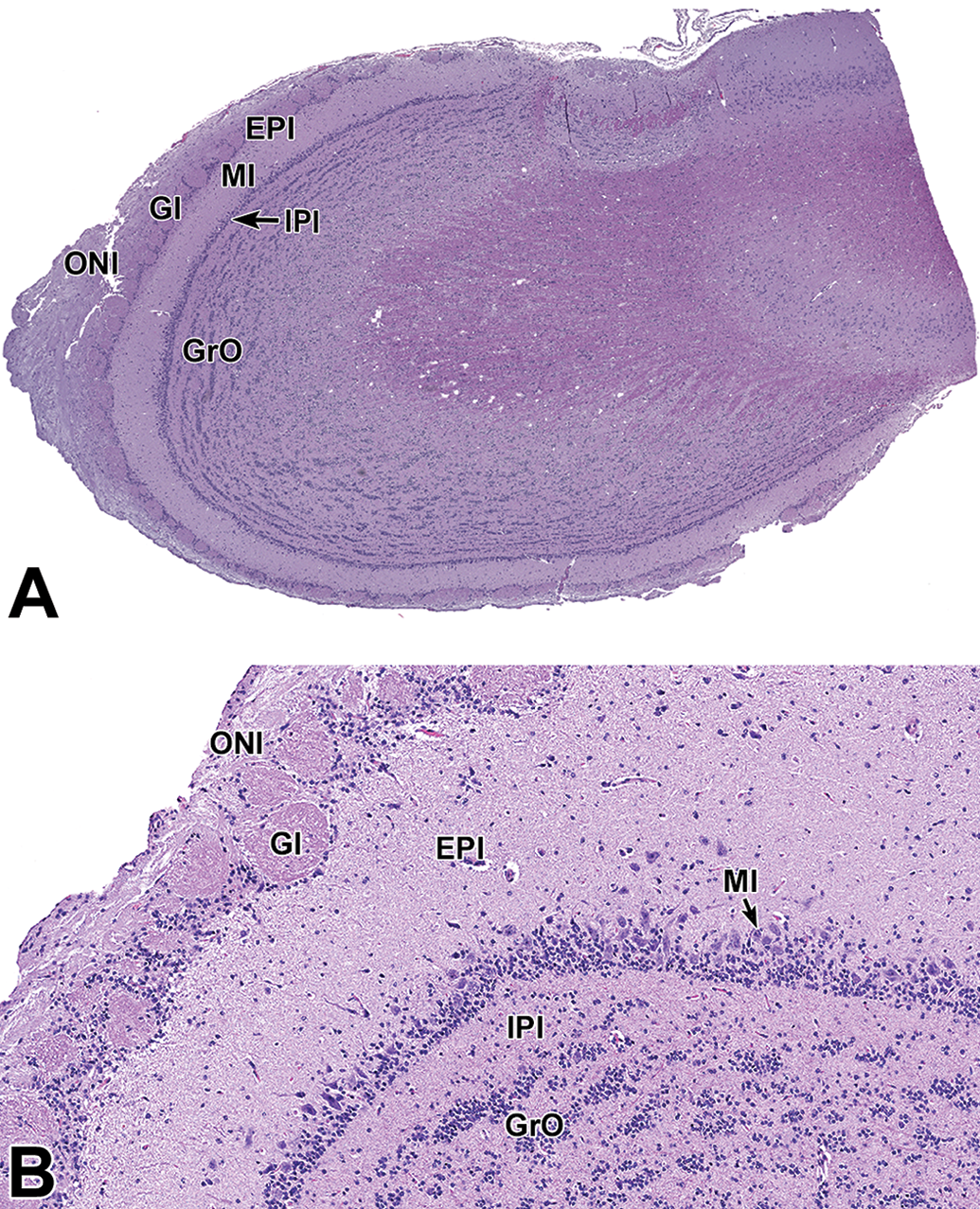
Level 1: Olfactory bulb (sagittal section). Low- (A) and high- (B) magnification views of a sagittal section depicting key histological areas. B, Higher magnification of figure A depicting a closer view of the olfactory lobe neuroanatomy. EPl indicates external plexiform layer; Gl, glomerular layer; GrO, granule cell layer; IPl, internal plexiform layer; MI, mitral cell layer; ONI, olfactory nerve layer.
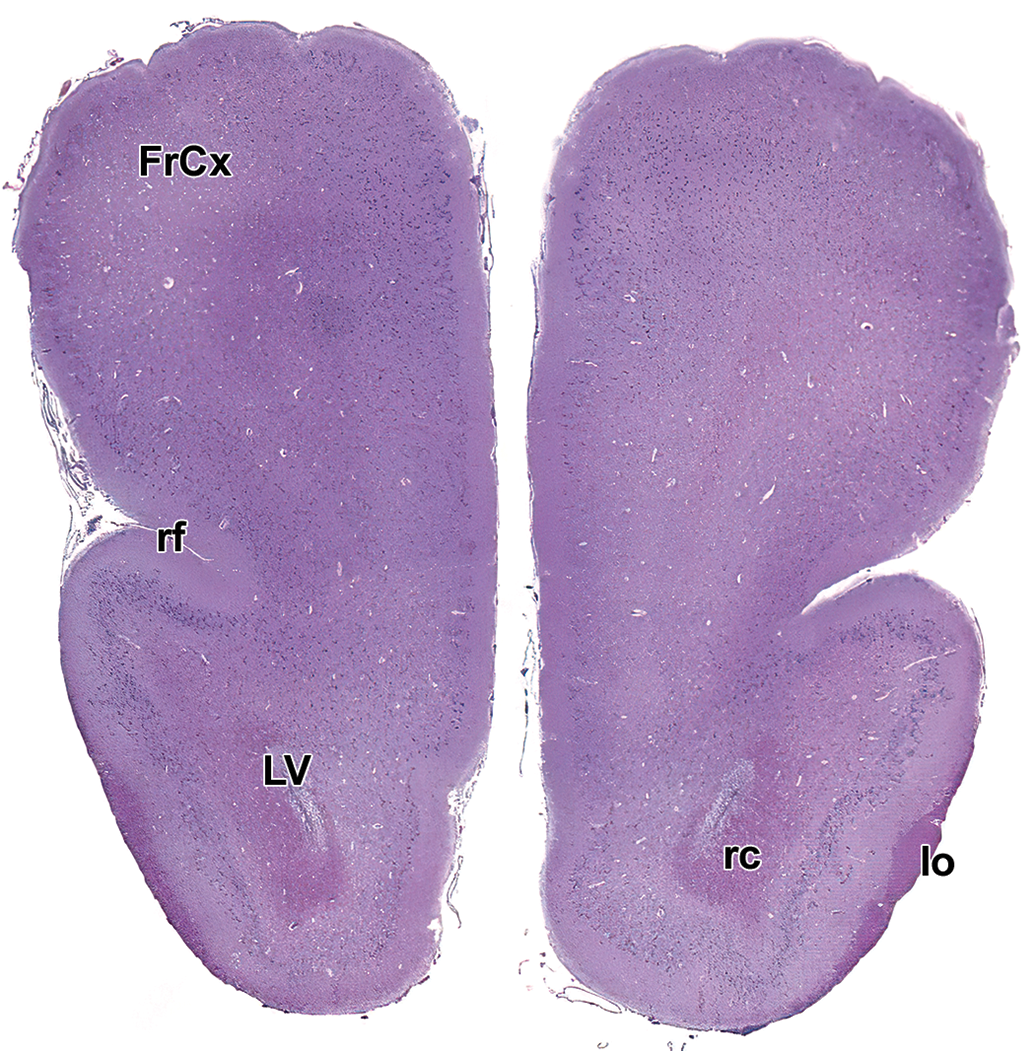
Level 2: Frontal pole and olfactory tract. FrCx indicates frontal cortex; lo, lateral olfactory tract; LV, lateral ventricle; rc, rostral (anterior) commissure; rf, rhinal fissure.
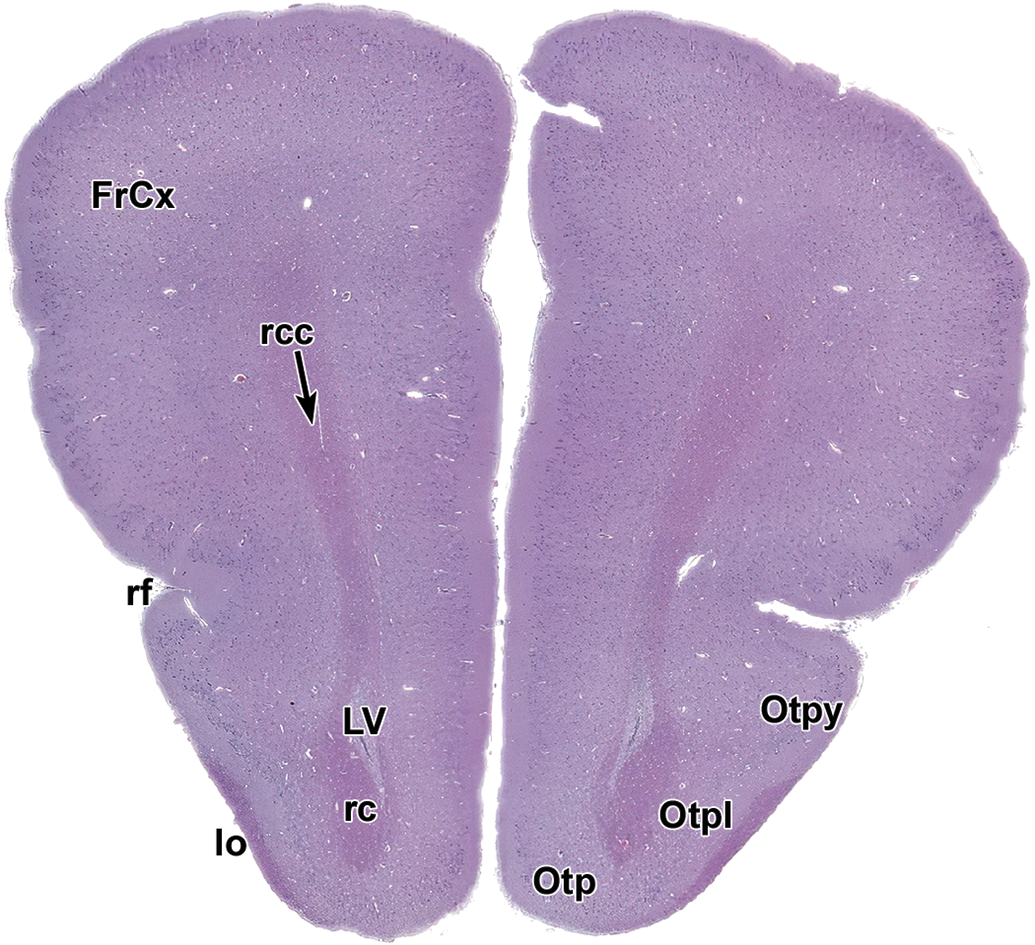
Level 3: Frontal pole and olfactory brain. FrCx indicates frontal cortex; lo, lateral olfactory tract; LV, lateral ventricle; Otp, olfactory tubercle, polymorphic layer; Otpl, olfactory tubercle, plexiform layer; Otpy, olfactory tubercle, pyramidal layer; rc, rostral (anterior) commissure; rcc, radiation of the corpus callosum; rf, rhinal fissure.
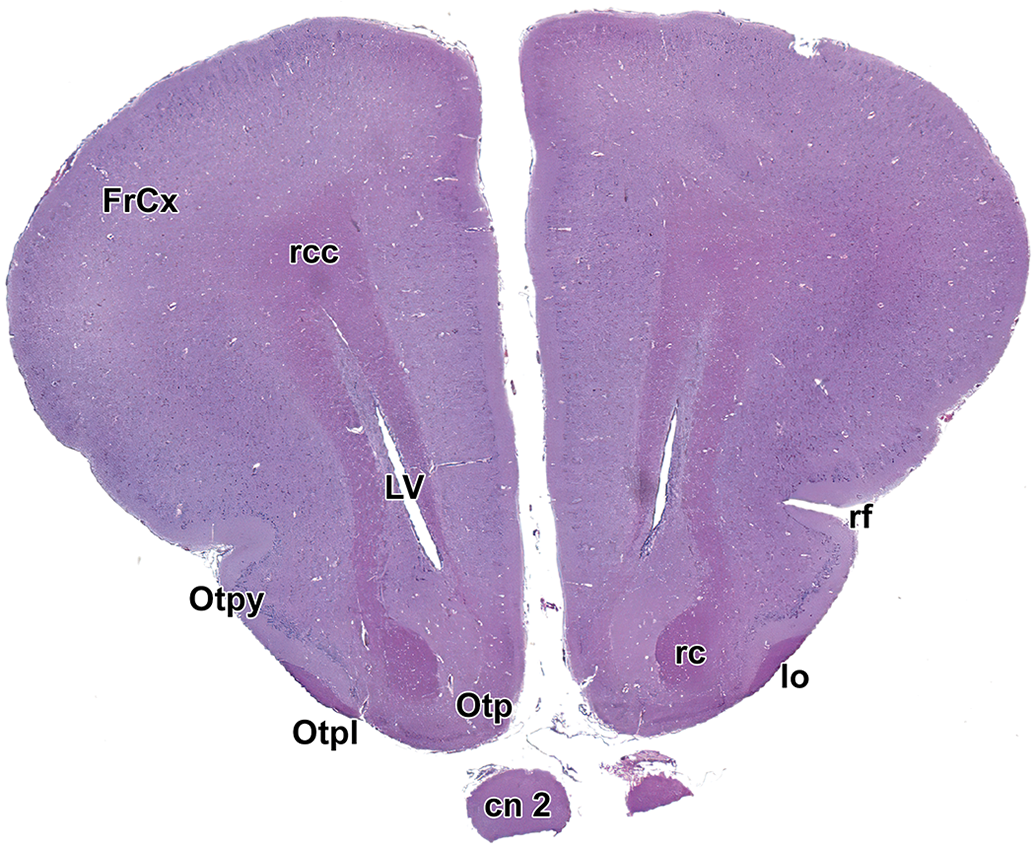
Level 4: Frontal cortex and olfactory brain. cn 2 indicates optic nerve; FrCx, frontal cortex; lo, lateral olfactory tract; LV, lateral ventricle; Otp, olfactory tubercle, polymorphic layer; Otpl, olfactory tubercle, plexiform layer; Otpy, olfactory tubercle, pyramidal layer; rc, rostral (anterior) commissure; rcc, radiation of the corpus callosum; rf, rhinal fissure.
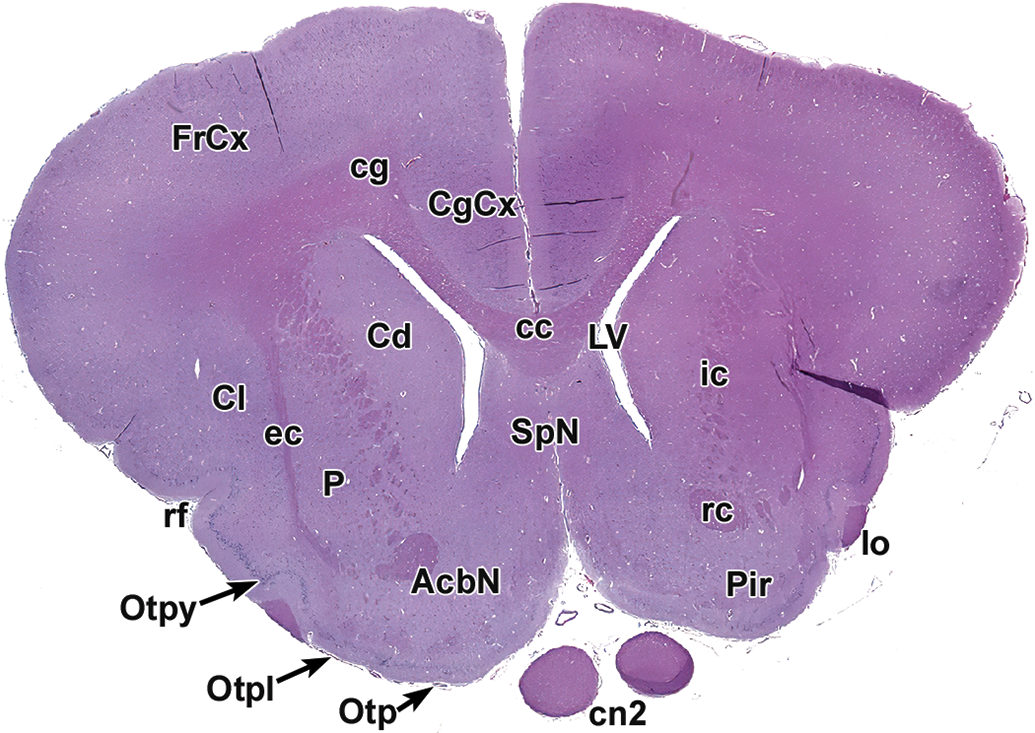
Level 5: Striatum and rostral (anterior) commissure. AcbN indicates accumbens nucleus; CgCx, cingulate cortex; cc, corpus callosum; cg, cingulum; Cd, caudate nucleus; Cl, claustrum; cn2, optic nerve; ec, external capsule; FrCx, frontal cortex; ic, internal capsule; lo, lateral olfactory tract; LV, lateral ventricle; Otp, olfactory tubercle, polymorphic layer; Otpl, olfactory tubercle, plexiform layer; Otpy, olfactory tubercle, pyramidal layer; P, putamen; Pir, piriform cortex; rc, rostral (anterior) commissure; rf, rhinal fissure; SpN, septal nuclei.
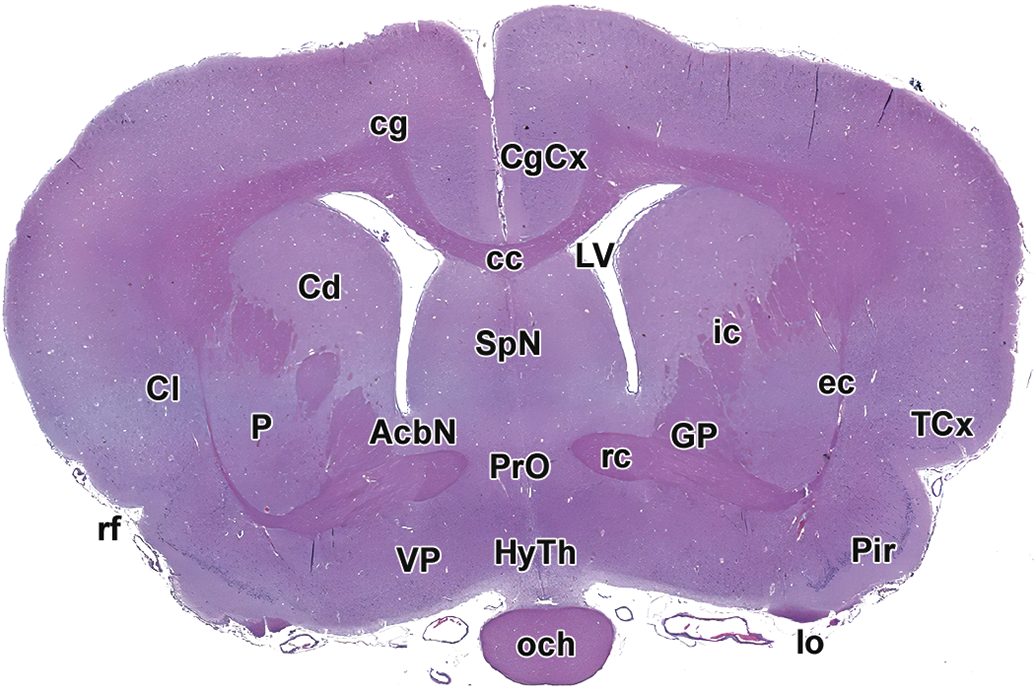
Level 6. Striatum and rostral (anterior) commissure. AcbN indicates accumbens nucleus; CgCx, cingulate cortex; cg, cingulum; cc, corpus callosum; Cd, caudate nucleus; Cl, claustrum; ec, external capsule; GP, globus pallidus; HyTh, hypothalamus; ic, internal capsule; lo, lateral olfactory tract; LV, lateral ventricle; och, optic chiasm; P, putamen; Pir, piriform cortex; PrO, preoptic hypothalamic nucleus/region; rc, rostral (anterior) commissure; rf, rhinal fissure; SpN, septal nuclei; TCx, temporal cortex; VP, ventral pallidum.
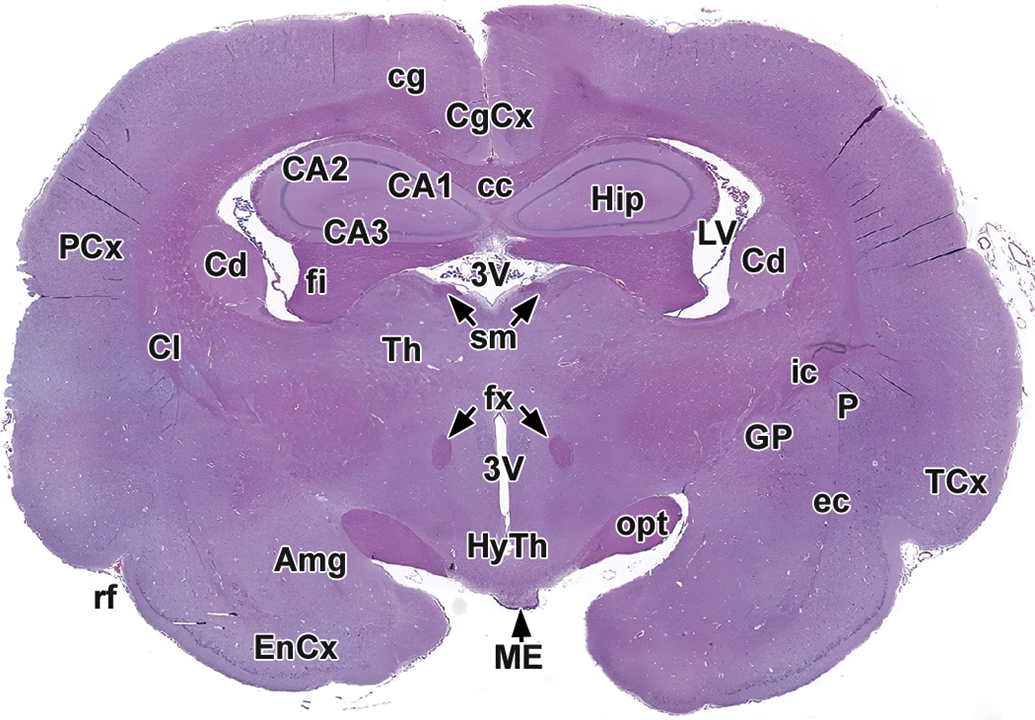
Level 7: Rostral thalamus, hypothalamus, and hippocampus. Amg indicates amygdala; CA1-3, cornu Ammonis fields 1-3; cc, corpus callosum; Cd, caudate nucleus; CgCx, cingulate cortex; cg, cingulum; Cl, claustrum; EnCx, entorhinal cortex; ec, external capsule; fi, fimbria; fx, fornix; GP, globus pallidus; Hip, hippocampus; HyTh, hypothalamus; ic, internal capsule; LV, lateral ventricle; ME, median eminence; opt, optic tract; P, putamen; PCx, parietal cortex; rf, rhinal fissure; sm, stria medullaris of thalamus; TCx, temporal cortex; Th, thalamus; 3V, third ventricle.
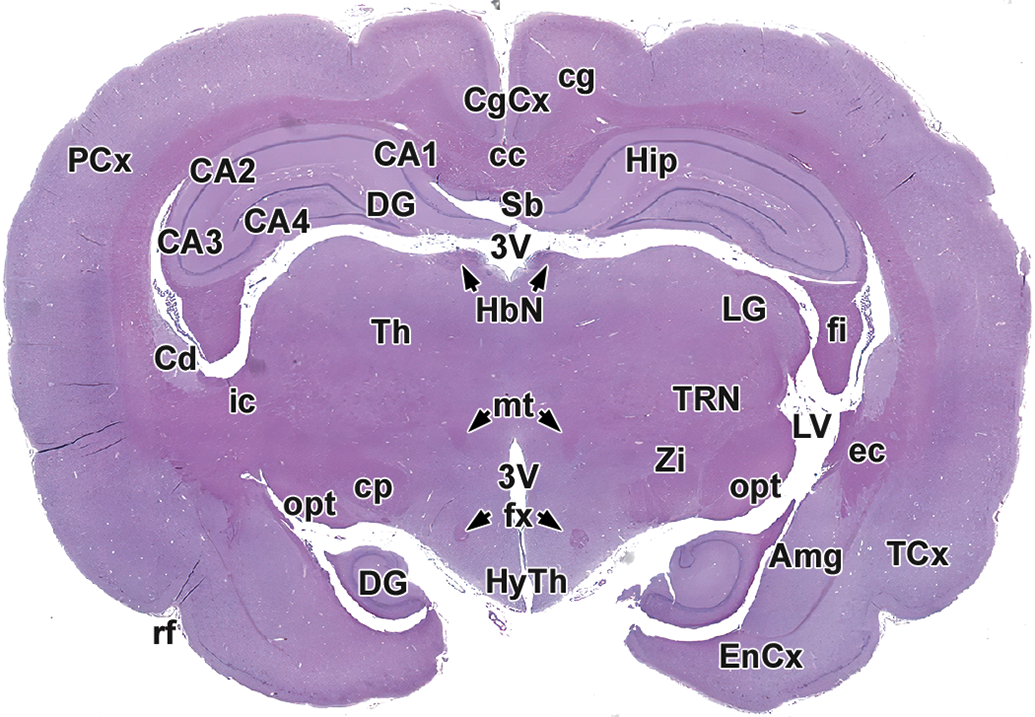
Level 8: Mid to caudal thalamus, hypothalamus, and hippocampus. Amg indicates amygdala; CA1-4, cornu Ammonis fields 1-4; cc, corpus callosum; Cd, caudate nucleus; cg, cingulum; cp, cerebral peduncle; DG, dentate gyrus of hippocampus; ec, external capsule; EnCx, entorhinal cortex; fi, fimbria; fx, fornix; HbN, habenular nuclei; Hip, hippocampus; HyTh, hypothalamus; ic, internal capsule; LG, lateral geniculate nucleus; LV, lateral ventricle; mt, mammillothalamic tract; opt, optic tract; PCx, parietal cortex; rf, rhinal fissure; Sb, subiculum; TCx, temporal cortex; T, thalamus; TRN, thalamic reticular nucleus; Zi, zona incerta; 3V, third ventricle.
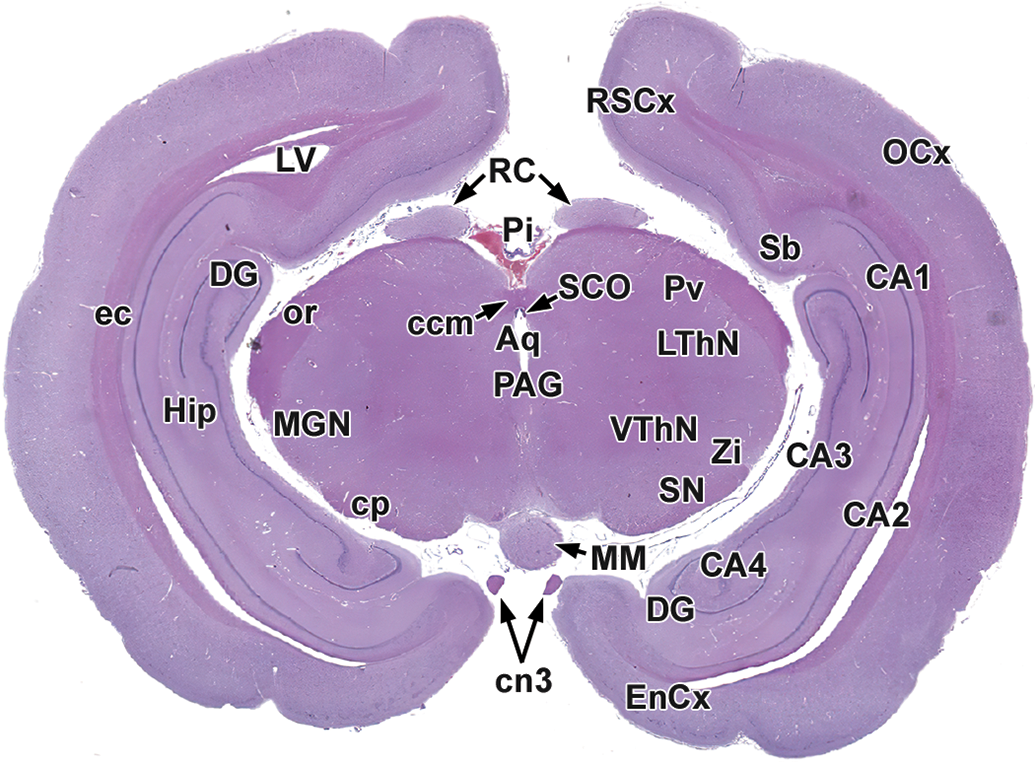
Level 9: Midbrain, substantia nigra, and occipital pole. Aq indicates mesencephalic aqueduct (of Sylvius); CA1-4, cornu Ammonis fields 1-4; cn3, oculomotor nerve; ccm, caudal (posterior) commissure; cp, cerebral peduncle; DG, dentate gyrus; EnCx, entorhinal cortex; ec, external capsule; Hip, hippocampus; LV, lateral ventricle; LThN, lateral thalamic nucleus; MGN, medial geniculate nucleus; MM, mammillary body; OCx, occipital cortex; or, optic radiation; PAG, periaqueductal gray; Pi, pineal gland; Pv, pulvinar nucleus; RSCx, retrospenial cortex; RC, rostral (superior) colliculus; Sb, subiculum; SN, substantia nigra; SCO, subcommissural organ; VThN, ventral thalamic nucleus; Zi, zona incerta.
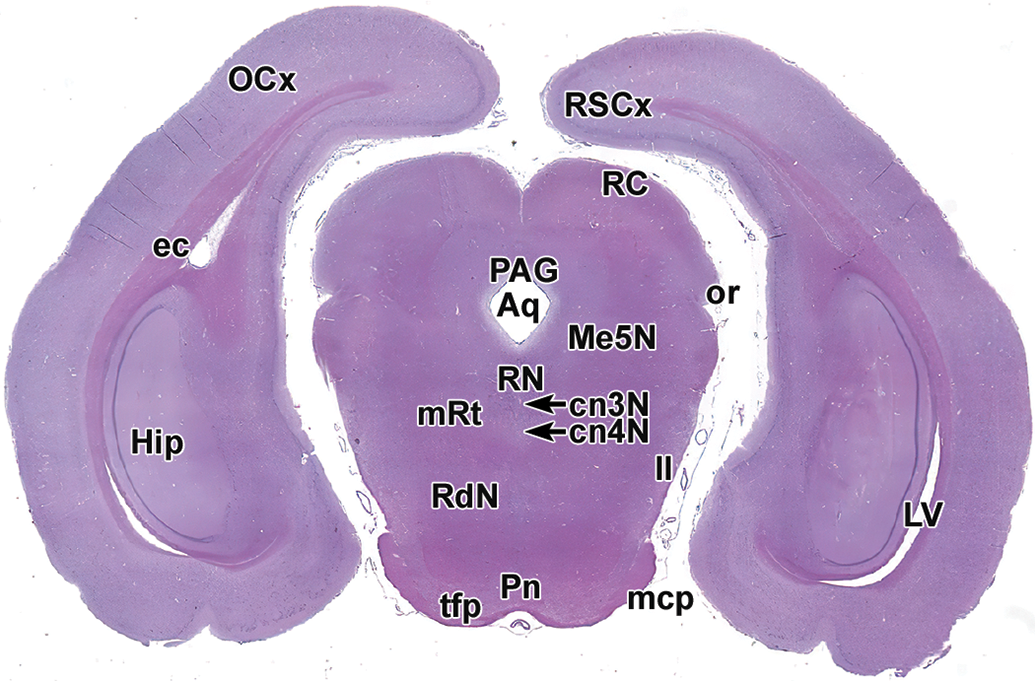
Level 10: Pons, hippocampus, and occipital pole. Aq indicates mesencephalic aqueduct (of Sylvius); cn3 N, oculomotor nucleus; cn4 N, trochlear nucleus; ec, external capsule; Hip, hippocampus; ll, lateral lemniscus; LV, lateral ventricle; mcp, middle cerebellar peduncle; Me5N, mesencephalic trigeminal nucleus; OCx, occipital cortex; or, optic radiation; PAG, periaqueductal gray; Pn, pontine nuclei; RdN, red nucleus; RSCx, retrosplenial cortex; RC, rostral (superior) colliculus; RN, raphe nuclei; mRt, reticular formation; tfp, transverse fibers of the pons.
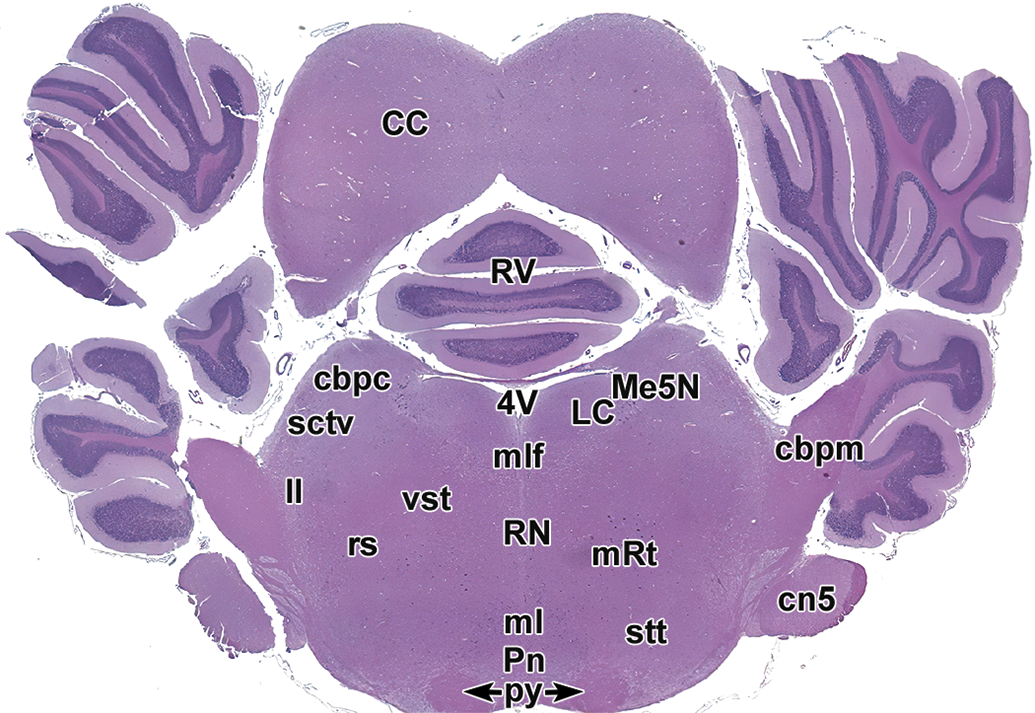
Level 11: Caudal colliculi, cerebellum, and brainstem. cbpc indicates cranial cerebellar peduncle; cbpm, middle cerebellar peduncle; CC, caudal (inferior) colliculus; cn5, trigeminal nerve; LC, locus coeruleus; ll, lateral lemniscus; ml, medial lemniscus; mlf, medial longitudinal fasciculus; Me5N, mesencephalic trigeminal nucleus; Pn, pontine nuclei; Py, pyramids; mRt, reticular formation; RN, raphe nuclei; rs, rubrospinal tract; RV, rostral vermis; sctv, ventral spinocerebellar tract; stt, spinothalamic tract; vst, vestibulospinal tract; 4V, fourth ventricle.
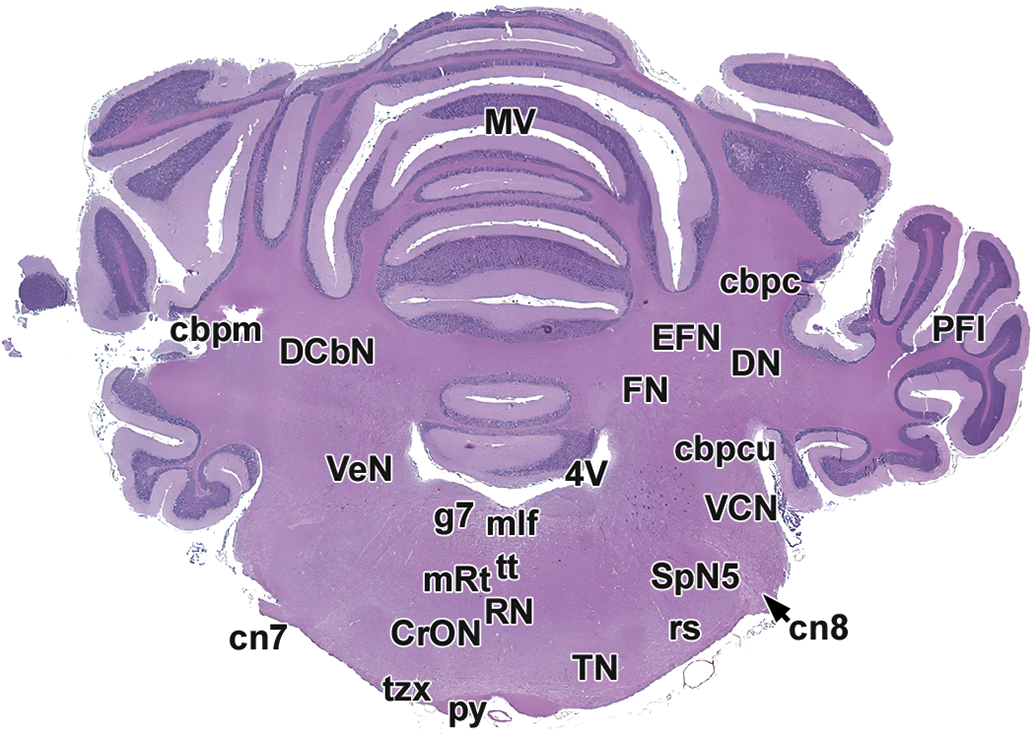
Level 12: Middle vermis, deep cerebellar nuclei, and rostral olivary nuclear complex. cbpc indicates cranial cerebellar peduncle; cbpcu, caudal cerebellar peduncle; cbpm, middle cerebellar peduncle; cn7, facial nerve; cn8, cochlear nerve (root); CrON, rostral olivary nuclei; DCbN, deep cerebellar nuclei (DN, EFN, FN); DN, dentate nucleus (part of DCbN); EFN, emboliform nucleus (part of DCbN); FN, fastigial nucleus (part of DCbN); g7, genu of the facial nerve; mlf, medial longitudinal fasciculus; MV, middle vermis; PFl, paraflocculus lobe; py, pyramids; RN, raphe nuclei; mRt, reticular formation; rs, rubrospinal tract; SpN5, spinal trigeminal nucleus; TN, trapezoid nucleus; tt, tectobulbar/tectospinal tract; tzx, decussation of the trapezoid body; VCN, ventral cochlear nucleus; VeN, vestibular nuclei; 4V, fourth ventricle.
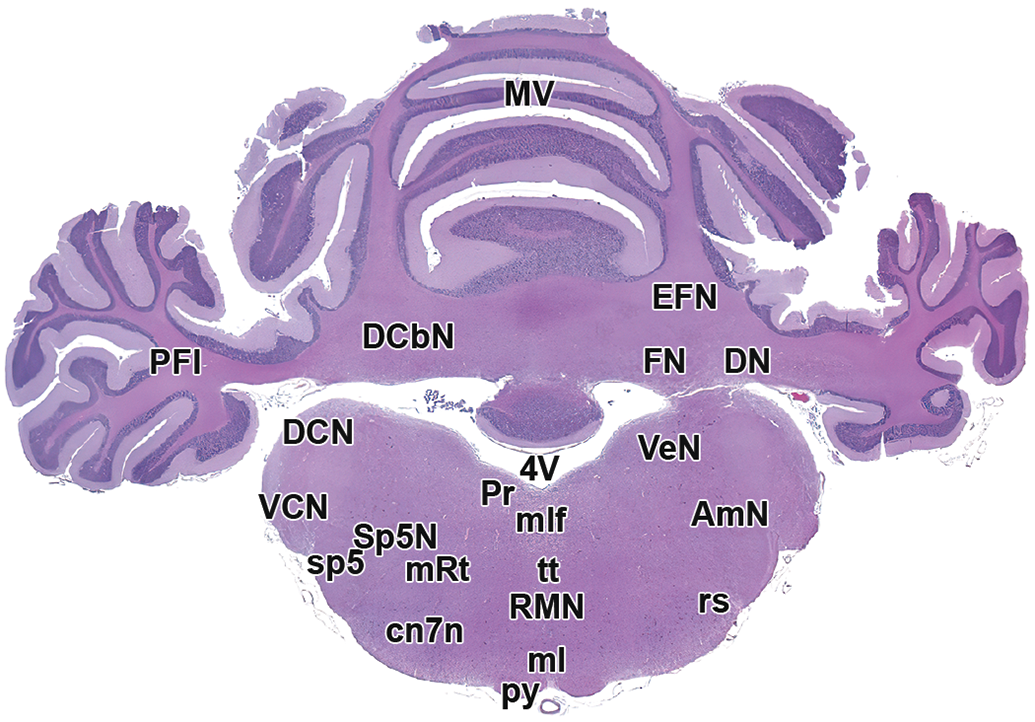
Level 13: Mid cerebellum and brainstem. AmN, ambiguus nucleus; cn7N, facial nerve nucleus; DCbN, deep cerebellar nuclei (DN, EFN, FN); DCN, dorsal cochlear nucleus; DN, dentate nucleus (DCbN); EFN, emboliform nucleus (DCbN); FN, fastigial nucleus (DCbN); ml, medial lemniscus; mlf, medial longitudinal fasciculus; MV, middle vermis; PFl; paraflocculus lobe; Pr, prepositus hypoglossal nucleus; Py, pyramids; RMN, raphe magnus nucleus; mRt, reticular formation; rs, rubrospinal tract; sp5, spinal trigeminal tract; Sp5N, spinal trigeminal nucleus; tt, tectobulbar/tectospinal tract; VCN, ventral cochlear nucleus; VeN, vestibular nucleus; 4V, fourth ventricle.
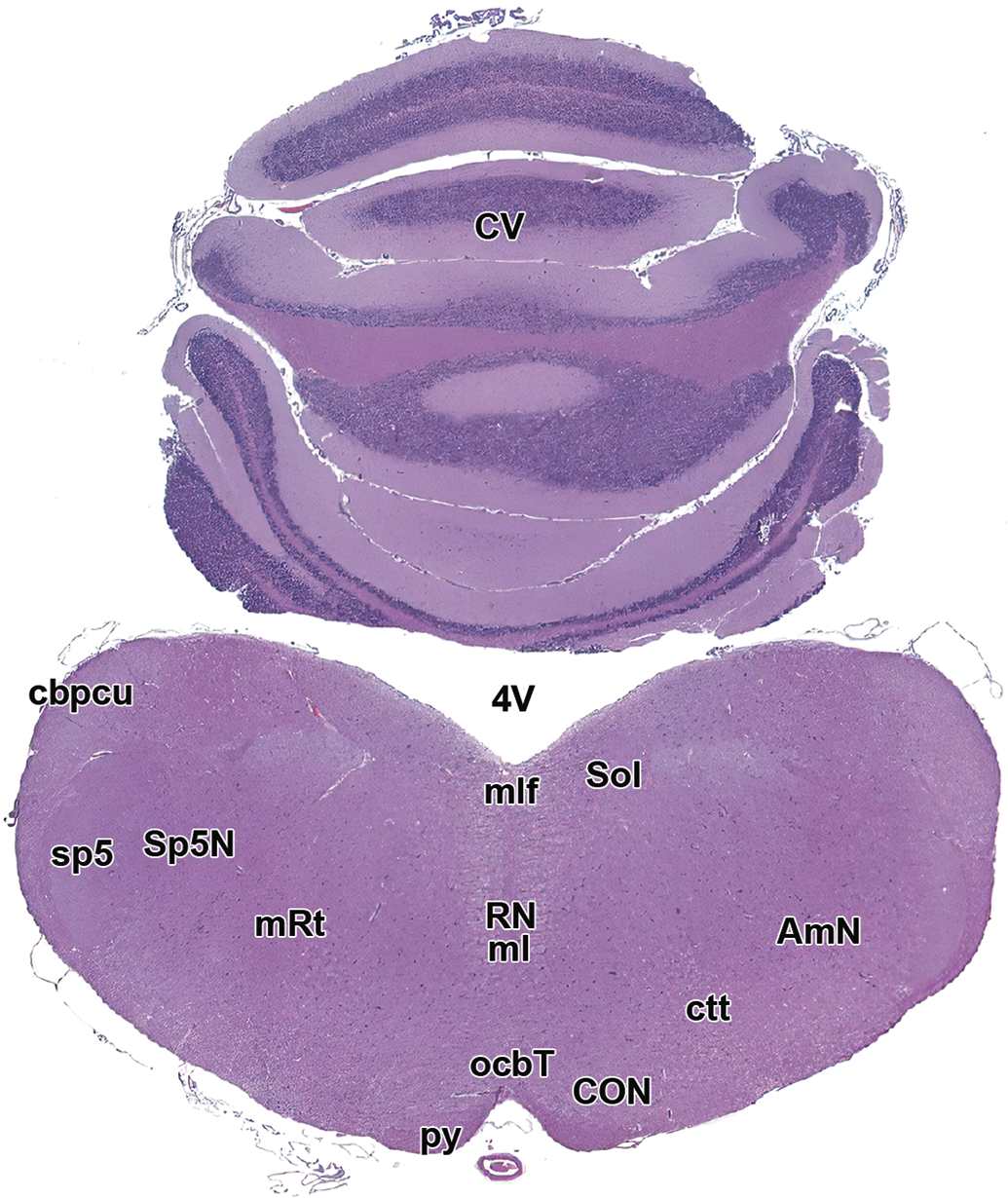
Level 14: Caudal vermis and brainstem. AmN indicates ambiguus nucleus; cbpcu, caudal cerebellar peduncle; CON, caudal olivary nucleus; ctt, central tegmental tract; CV, caudal vermis; ml, medial lemniscus; mlf, medial longitudinal fasciculus; ocbt, olivocerebellar tract; Py, pyramids; mRt, reticular formation; RN, raphe nuclei; Sol, solitary nucleus and tract; Sp5N, spinal trigeminal nucleus; sp5, spinal trigeminal tract; 4V, fourth ventricle.
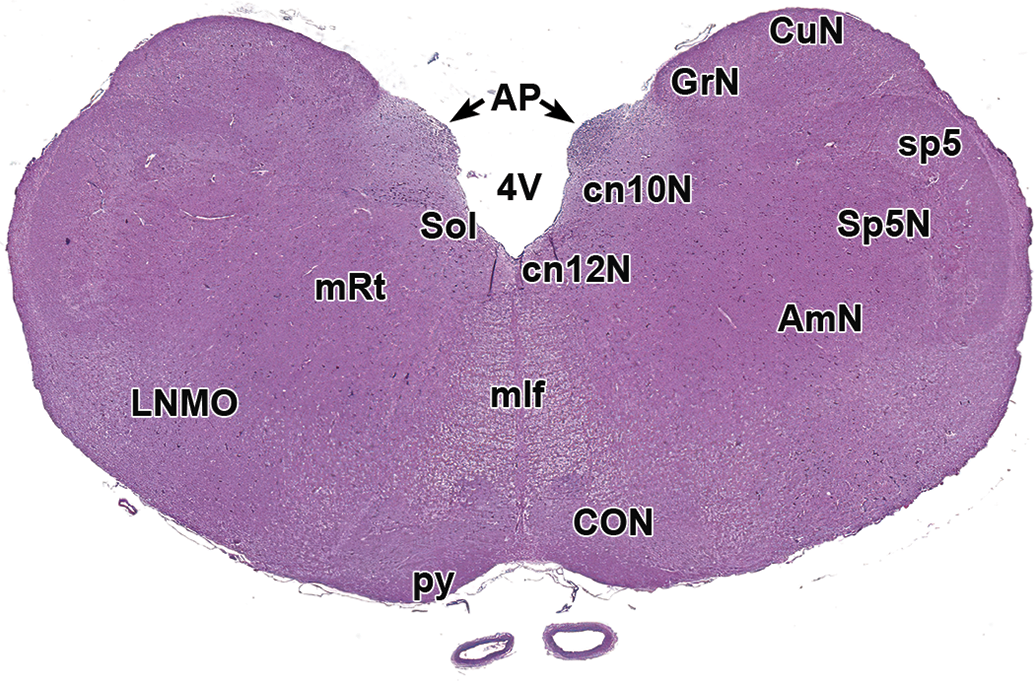
Level 15: Brainstem and caudal olivary nuclear complex. AmN indicates ambiguus nucleus; AP, area postrema; cN10N, vagus nerve nucleus (dorsal); cN12N, hypoglossal nerve nucleus; CON, caudal olivary nucleus; CuN, cuneate nucleus; GrN, gracile nucleus; LNMO, lateral nucleus of the medulla oblongata (lateral reticular nucleus); mlf, medial longitudinal fasciculus; py, pyramids; mRt, reticular formation; Sol, solitary tract nucleus and tract; Sp5N, spinal trigeminal nucleus; sp5, spinal trigeminal nucleus tract; 4V, fourth ventricle.
Other Neural Organs: Spinal Cord, Ganglia, and Nerves
The STP “best practice” recommendations for collecting other nervous system organs vary depending on the situation for spinal cord 1 and for PNS. 2 In general, neural structures should be sampled extensively and bilaterally. The rationale for this strategy is that the necropsy is conducted at a single point in time, and any tissues not harvested during this procedure will be lost forever.
Spinal Cord
Three spinal cord segments (cervical, thoracic, and lumbar) should be harvested for both general toxicity and dedicated neurotoxicity studies. These segments (typically as long as 2-3 vertebral bodies) may be left in situ for fixation or removed from the vertebral canal. For removal, spinal cord generally is harvested by utilizing medium rongeurs to remove the dorsal and transverse vertebral processes, after which forceps are used to grasp the dura mater and extract the spinal cord without touching the delicate neural tissue. Compared to humans, rabbits have larger epidural and smaller subarachnoid spaces, 40 which makes removal of the spinal cord relatively easy in rabbits. Spinal cord segments should be evaluated both macroscopically and microscopically in cross (transverse) and longitudinal planes (Figure 20). A longitudinal oblique orientation is quite useful for this purpose since it permits histopathologic analysis of more white matter than is feasible in some other longitudinal orientations. 1,18
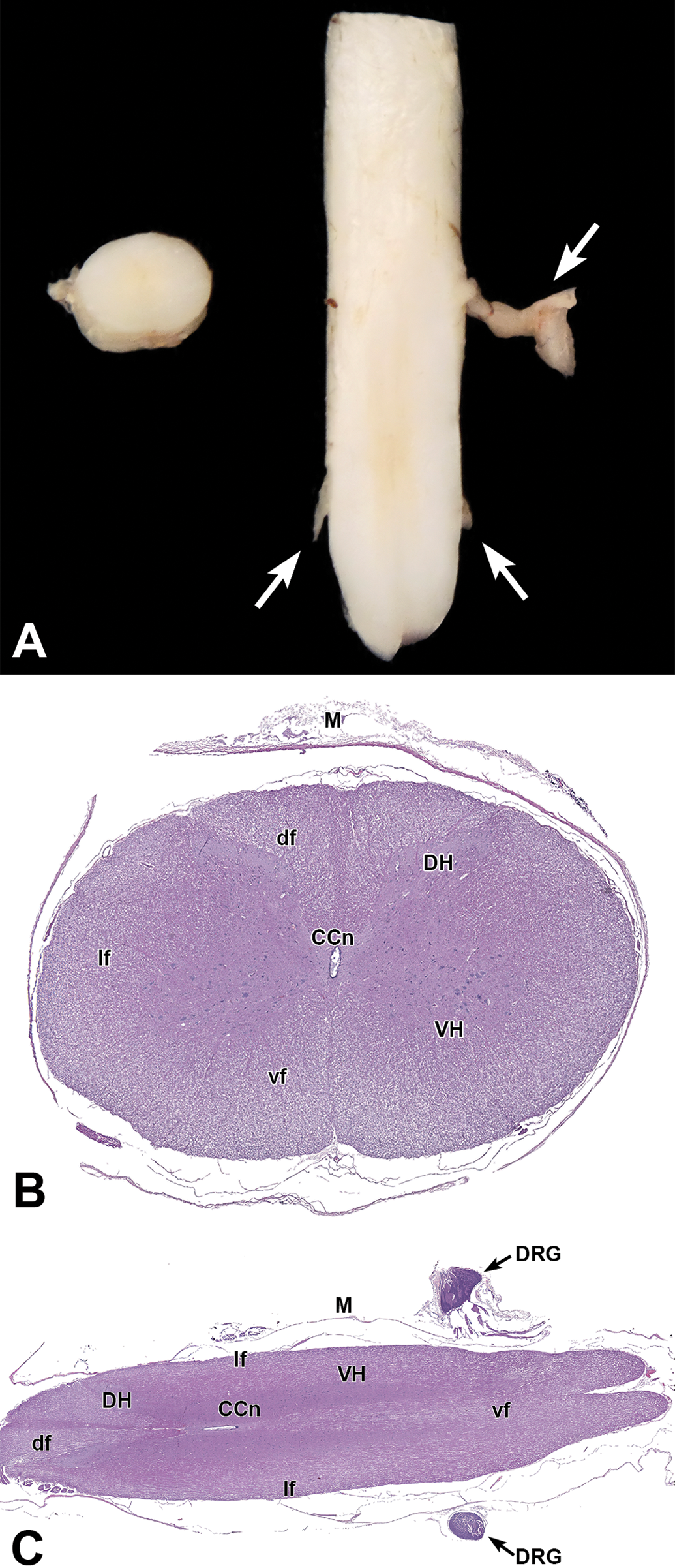
Spinal cord. (A) Macroscopic images of transverse (cross) and longitudinal oblique sections of cervical cord. Arrows denote dorsal root ganglia (DRG). (B) Transverse and (C) longitudinal oblique sections of cervical cord. Gray matter: DH, dorsal horn; VH, ventral horn. White matter: df, dorsal funiculi; lf, lateral funiculi; vf, ventral funiculi. Other features: CCn, central canal; M, meninge; Arrow in C = DRG.
Typically, the spinal cord is removed from the vertebral column for histological processing to avoid damage produced by acid-based bone-decalcifying reagents. However, some pathologists leave the spinal cord in the vertebral canal for histological processing to maintain the integrity of the spinal cord connections to the spinal nerve roots and dorsal root ganglia (DRG). In this case, the fixed vertebral column segments are decalcified by immersion in a solution of formic acid (Klaus Weber, personal communication, 2020). An alternative decalcification procedure might be immersion in a 1:4 mixture of formic acid and sodium formate, which removes mineral more slowly and thus is gentler toward the soft neural tissues.
An important consideration for sampling the lumbar spinal cord segment in rabbits is that, unlike other test species, the numbered cord segments correspond approximately to the vertebrae with the same number. The rabbit spinal cord has been reported to terminate within the second sacral vertebra (S2) in 79.3%, the first sacral vertebra (S1) in 19.0%, and within the third sacral vertebra (S3) in 1.7% of the 64 WNZ rabbits in the study. 41 In rabbits, the spinal cord segments that contribute to the sciatic nerve are L6-7 and S1-3, while spinal cord segments L4-6 (ie, those that innervate the sciatic nerve in other species) supply the femoral nerve in rabbits. 41 Thus, the best spinal cord segments for sampling the neurons providing axons to the sciatic nerves will be found in vertebrae L6 to S1 in most rabbits, which is caudal to the locations in other mammals (L1-2 in rodents and NHPs or L3-4 in dogs) used in nonclinical toxicity testing. 1
Ganglia
Sampling of PNS ganglia varies depending on the study purpose. For most studies, sampling is typically limited to somatic sensory ganglia such as the DRG and trigeminal ganglia (cranial nerve V). 2,42 These sensory ganglia usually are sampled when there is a possibility to be targeted by a test article. 43 –46 Autonomic ganglia generally are obtained for evaluation only as they occur within the walls of hollow nonneural organs sampled as part of the protocol-specified tissue list.
For general toxicity studies, at least 1 lumbar DRG should be harvested if no neurological signs are observed in life, while 2 DRG each for the cervical and lumbar segments should be collected if neurological signs are seen during life. The largest lumbar DRG in rabbits is L6, 41 which is consistent with this segment’s major contribution to the sciatic and femoral nerves.
For dedicated neurotoxicity studies, at least 2 DRG each from the cervical (typically C5-T1), thoracic (T6-8), and lumbar (L4-6 and/or L6-S1) segments as well as both trigeminal ganglia should be procured. Most study protocols recommend sampling DRG that are associated with the major nerves supplying the forelimbs (C5-T1) and hind limbs (in rabbits, L4-6 for the femoral nerve and L6-S1 for the sciatic nerve). The T6-8 DRG is chosen commonly since this location represents the midrange for this spinal cord segment. Autonomic ganglia (eg, cranial [superior] cervical, cervicothoracic, and cranial mesenteric) should be considered for collection and retention in wet tissue even if neurological signs are not noted during life. The location of these ganglia in rabbits could not be demonstrated for the current satellite study because the torso and its neural elements were dedicated to the objective of the primary study, but their position in rabbits 47,48 is similar to that of comparable ganglia in other species. 2
Ideally, the DRG and trigeminal ganglia are removed from their bony surroundings for histological processing to avoid acid-induced damage during decalcification. This harvesting may be done at necropsy, but more frequently DRG are fixed in situ and then teased out painstakingly in the laboratory using a pair of fine dissecting forceps (eg, Dumont No. 5) and a stereomicroscope. A labor-saving alternative for DRG employed by some pathologists, especially for general toxicity studies, is to process them along with the vertebrae after decalcification by immersion in 1:4 mixture of formic acid and sodium formate for 3 weeks, as described above (K. Weber, personal communication, 2020). Subsequently, the decalcified vertebral column is trimmed near the level of the intervertebral cartilage so that the DRG are near the trimming plane after embedding.
Nerves
Sampling of nerves in rabbits also differs depending on the study purpose. For most studies, only somatic nerves are collected unless autonomic (parasympathetic or sympathetic) signs are expected or observed, in which case autonomic nerves may be harvested as well.
For general toxicity studies, many institutions collect and evaluate only the sciatic nerve if no neurological signs are seen in life (Figure 21). Multiple nerves (typically the mixed sensorimotor sciatic nerve trunk and its tibial and fibular branches) are collected bilaterally and evaluated if neurological signs were observed. Because it has been well established that neurotoxicants typically affect distal nerves first, reasoning for nonexamination of the tibial or fibular nerves should be included in studies with neurological signs where evaluation was limited to sciatic nerves.
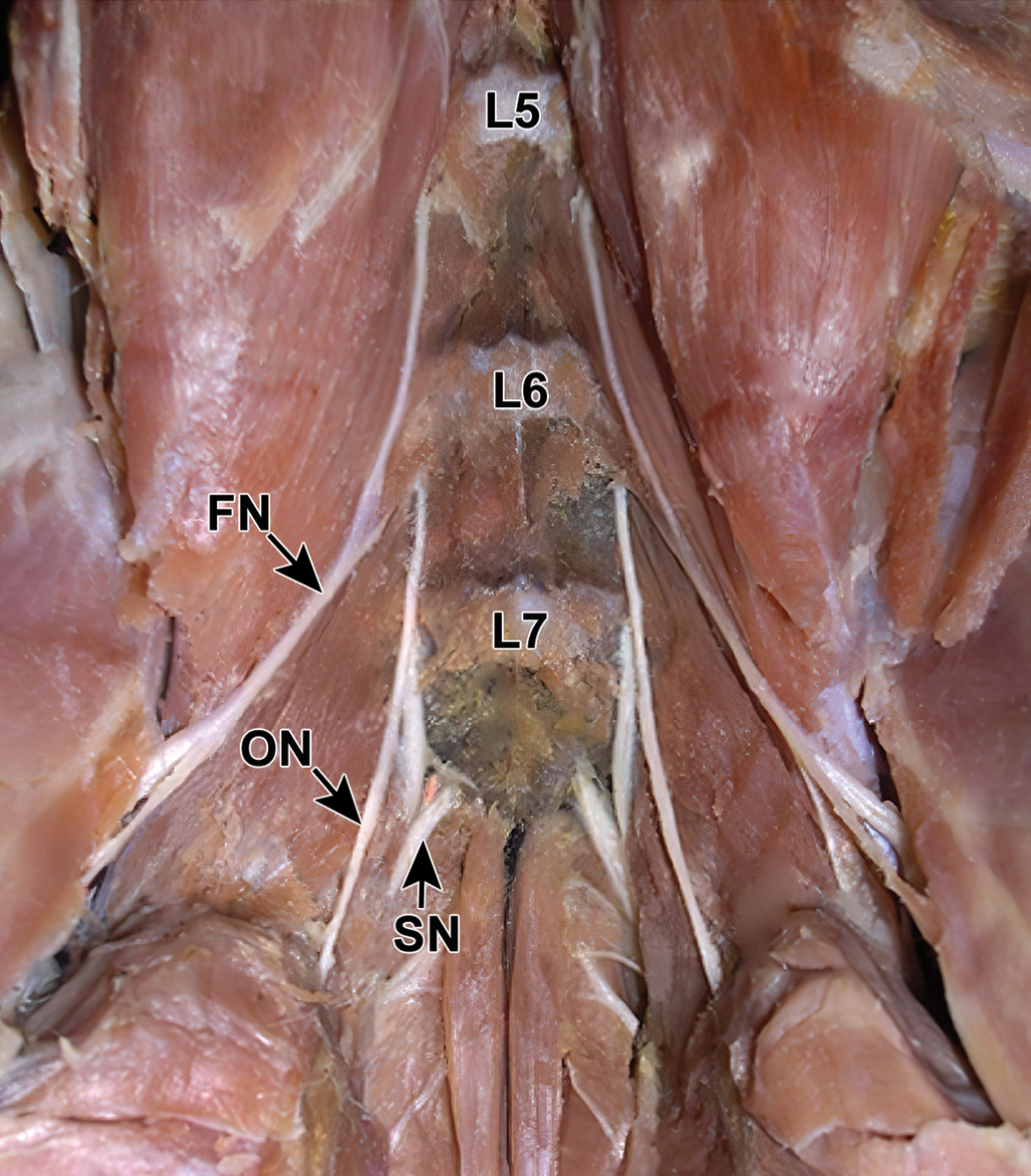
Location of the principal hind limb nerves in the rabbit. This of the sublumbar area of an adult White New Zealand (WNZ) rabbit shows the proximal portions of the femoral nerve (FN), obturator nerve (ON), and sciatic nerve (SN) with respect to the ventral vertebral bodies of the fifth (L5), sixth (L6), and seventh (L7) lumbar vertebrae. [This image was provided Dr. Marcelo Abidu-Figueiredo of the Department of Animal Biology, School of Veterinary Medicine, Universidade Federal Rural do Rio de Janeiro [UFRRJ], Brazil and is reproduced from “Origin and antimeric distribution of the obturator nerves in the New Zealand rabbits” [Medeiros do Nascimento et al., 2019 49 ] under a Creative Commons license.
For dedicated neurotoxicity studies in rabbits, the same 3 somatic nerves (sciatic, tibial, and fibular) form the foundation of the PNS assessment. However, where appropriate, other functionally specific trunks such as the motor-only obturator nerve 49 (Figure 21) or the sensory-predominant saphenous nerve 50 should be considered for collection as well.
Discussion
Our current article describes schemes for systematic sampling of the rabbit nervous system during animal toxicity testing in safety assessment. Specific instructions given here for both the CNS and PNS of rabbits are based on extrapolation of recently published STP “best practice” recommendations for nervous system sampling of other mammalian test species. 1,2 Particular emphasis is placed on demonstrating situation-dependent trimming options for the brain as well as defining what features may be seen and why they are important to evaluate in these standard rabbit brain sections.
Two strategies for brain trimming are described here, one for routine screening and a second for more detailed neurotoxicity assessment. For general toxicity studies (ie, a tier 1 screening approach), our 6-section protocol follows the STP “best practice” recommendations for brain trimming in nonrodent species, and thus permits the evaluation of all the key brain regions suggested for evaluation in nonclinical toxicity studies. 1 For dedicated neurotoxicity studies (ie, a tier 2 strategy for detailed neurotoxicity assessment), a more extensive 12-section trimming scheme allows the whole brain to be processed, thereby affording access to a greater number of key brain areas. The decision regarding whether further modifications of the sampling strategy might better suit an institution’s test article development plan may be undertaken using our detailed lists of key structures that are visible using each of the 2 trimming schemes (Tables 1 and 2) as well as the brief but thorough table regarding key functions served by major brain areas visible in these sections (Online Appendix 1). Collection of spinal cord, ganglia, and nerves should follow published species-agnostic recommendations for these organs. 1,2
Evaluation of the rabbit brain for nonclinical safety testing requires some special consideration relative to other test species, for 2 main reasons. The primary points relate to rabbit-specific brain dissection practices and neuroanatomy.
First, extra care must be taken when removing the rabbit brain to avoid damaging the brain parenchyma with dissecting tools. This concern is especially true for the cerebellum, which is in close contact with the overlying calvarium. The large, bulging olfactory bulbs are deeply entrenched in the rostral skull. When desired, they can be collected with reasonable ease by fracturing the frontal and dorsal portions of the ethmoid bones, but in most rabbit studies this region is not sampled specifically except in the uncommon instance where the test article is encountered via the inhalational route. For inhalation studies, the olfactory bulbs generally are evaluated while attached to the brain, and not left in situ. The pituitary gland is enclosed within the sella turcica at the base of the skull and will remain in its original position in the skull after removal of the rest of the brain.
Second, comparison of rabbit neuroanatomy to that of other nonrodent mammals requires some thoughtfulness in data translation among species. Rabbit brain is lissencephalic (ie, lacks gyri and sulci) and exhibits a greater degree of hindbrain compared to forebrain development relative to gyrencephalic species such as dogs, NHPs, and humans (where the brain surface features myriad folds [gyri] separated by crevices [sulci]). The lateral ventricles of rabbits are elongated and extend rostrally to the olfactory bulbs. 51 The mesencephalic aqueduct in rabbits is funnel-shaped and wide rostrally before attaining a narrower, round to oval configuration caudally. WNZ rabbits also exhibit some variation in the normal anatomy of the spinal cord and nerves. For example, in a survey of 7 rabbits, totals of 8 to 9 cervical, 12 thoracic, 6 to 7 lumbar, and 4 sacral spinal nerves were defined by corresponding 7 to 8 cervical, 12 thoracic, 6 to 7 lumbar, and 4 sacral vertebrae. 15 In another study, radiographic and necropsy analyses demonstrated thoracic (T) and lumbar (L) vertebrae counts of 12T/7L in 43.8%, 13T/6L in 32.8%, and 13T/7L in 23.4% of 64 WNZ rabbits. 41 Vertebral fractures are very common in rabbits under restraint and tend to occur at vertebra L7. This location is vulnerable due to the inability of the fragile skeleton to withstand the force applied by intense contractions of the hind limb muscles when kicking to escape restraint. Prosectors will need to be aware of this propensity since this L7 site is at the center of the favored vertebral column location (L6-S1) for sampling lumbar DRG that supply the rabbit sciatic nerve.
While not specifically part of the current study, one other aspect of rabbit neurobiology also deserves mention as it relates to nonclinical safety testing in this species. This point is historical control data with respect to normal brain weights in rabbits. Brain weights are comparable for adult male and female WNZ rabbits. Based on internal Pfizer data, weights of adult rabbit brain (N = 76 males and 76 females) taken without the olfactory bulbs (which remained in the skull) average 9.50 g (range, 7.30-10.40 g), with a mean brain/body weight ratio of 0.26 (range, 0.17-0.38). These values are consistent with previously reported rabbit brain weights in males (N = 8; mean = 10.25 g [range, 9.28-12.30 g]) and females (N = 6; mean = 10.12 g [range, 9.68-10.66 g]; Shek et al., 1986). The olfactory bulb of rabbits is relatively large, representing about 2.4% of the total brain weight, based on a very small pilot study performed at Pfizer. Accordingly, it is imperative to build the historical control data for rabbit brain weight by always including OR always excluding the olfactory bulbs when acquiring this measurement (note 1).
Conclusion
The 2 brain trimming schemes described in this article provide flexible yet systematic approaches for reliable and reproducible sampling of the rabbit brain during tier 1 nonclinical general toxicity as well as tier 2 dedicated neurotoxicity studies. The 6-section trimming scheme for general toxicity studies is consistent with the STP “best practice” recommendations for brain sampling in nonrodent species, 1 and generally should not be reduced further. The 12-section scheme for dedicated neurotoxicity studies conforms to the concept that suspected or known neurotoxicity requires more extensive sampling. 1,2,22,39 Sampling of the spinal cord, ganglia, and nerves is equivalent in rabbits to the protocols used in other nonclinical test species. 1,2 The descriptions and illustrations in this article will allow toxicologists, pathologists, histotechnicians, and basic research scientists using rabbits in regulatory and research projects to design more efficient and effective tissue sampling protocols.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320957637 - Nervous System Sampling for General Toxicity and Neurotoxicity Studies in Rabbits
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320957637 for Nervous System Sampling for General Toxicity and Neurotoxicity Studies in Rabbits by Ingrid D. Pardo, Deepa B. Rao, James P. Morrison, Colleen Huddleston, Alys E. Bradley, Brad Bolon and Robert H. Garman in Toxicologic Pathology
Footnotes
Note
Acknowledgments
Dr Ingrid D. Pardo would like to thank Mrs Hayley Newton Ritenour and Mr William Nowland in Pfizer Inc in Groton, CT for all of their help with brain collection and sampling. The authors want to thank Dr Lisa Lanigan for procuring brain and spinal cords in rabbits while, she was working for Charles River Laboratories in Ashland, OH, and Mr Walter Bobrowski, Lakeshore Digital Art, LLC (http://www.LakeshoreDigitalArt.com) for his excellent work in optimizing Figures 1 –21. In addition, the authors want to thank Dr Marcelo Abidu-Figueiredo of the Department of Animal Biology, School of Veterinary Medicine, UFRRJ, Brazil who gave us permission to reproduce Figure 21 (from Medeiros do Nascimento et al. [2019]49; Ciência Animal Brasileira, https://www.scielo.br/scielo.php?script=sci_arttext&pid=S1809-68912019000100315).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
