Abstract
The Society of Toxicologic Pathology charged a Nervous System Sampling Working Group with devising recommended practices to routinely screen the central nervous system (CNS) and peripheral nervous system (PNS) in Good Laboratory Practice–type nonclinical general toxicity studies. Brains should be weighed and trimmed similarly for all animals in a study. Certain structures should be sampled regularly: caudate/putamen, cerebellum, cerebral cortex, choroid plexus, eye (with optic nerve), hippocampus, hypothalamus, medulla oblongata, midbrain, nerve, olfactory bulb (rodents only), pons, spinal cord, and thalamus. Brain regions may be sampled bilaterally in rodents using 6 to 7 coronal sections, and unilaterally in nonrodents with 6 to 7 coronal hemisections. Spinal cord and nerves should be examined in transverse and longitudinal (or oblique) orientations. Most Working Group members considered immersion fixation in formalin (for CNS or PNS) or a solution containing acetic acid (for eye), paraffin embedding, and initial evaluation limited to hematoxylin and eosin (H&E)-stained sections to be acceptable for routine microscopic evaluation during general toxicity studies; other neurohistological methods may be undertaken if needed to better characterize H&E findings. Initial microscopic analyses should be qualitative and done with foreknowledge of treatments and doses (i.e., “unblinded”). The pathology report should clearly communicate structures that were assessed and methodological details. Since neuropathologic assessment is only one aspect of general toxicity studies, institutions should retain flexibility in customizing their sampling, processing, analytical, and reporting procedures as long as major neural targets are evaluated systematically.
Keywords
Background
The recognition that chemically induced neurological deficits are a major occupational health hazard (Connelly and Malkin 2007; Gobba 2003) has resulted in an extensive international effort to consider more cautiously the potential for neurological damage following xenobiotic exposure. One foundation of this effort is the neuropathology component of animal-based toxicity testing required to develop novel chemicals and small molecule pharmaceuticals. However, substantial variation exists across regulatory agencies (Bolon et al. 2011a) regarding preferred neuropathology practices for registering new compounds. The recommendations vary by species (non-rodents [e.g., dog, nonhuman primate vs. rodents; Krinke 1989; Morawietz 2004]); by age (e.g., developing rodents [U.S. Environmental Protection Agency {EPA} 1998b; Organisation for Economic Co-operation and Development {OECD} 2007; Garman et al. 2001; Bolon et al. 2006, 2011b] vs. adult rodents [Broxup 1991; EPA 1998a; OECD, 1997]); by the type of study (general toxicity screen vs. dedicated neurotoxicity bioassay; Bolon et al. 2011a); by the kind of industry (agrochemical firms vs. pharmaceutical companies, for which potential exposure levels and, therefore, risk-to-benefit assessments will vary); and according to whether the study was conducted by Good Laboratory Practices (GLP) standards to support product registration. Regulatory guidelines for conducting the neuropathology analysis of GLP-type general toxicity studies (i.e., screening or “Tier I” surveys) provide wide-ranging advice for designing experiments that will evaluate many disparate organs and systems. The multi-tissue analysis undertaken during general toxicity studies necessarily requires that the regulatory guidelines for such assays are less prescriptive than the directions for undertaking dedicated neurotoxicity studies (i.e., advanced or “Tier II” tests), which supply more detailed protocols designed to optimize the targeted assessment of nervous tissues (Bolon et al. 2011a).
The extent of the neuropathology assessment performed for general toxicity studies has the potential to affect the outcome of the analysis. The routine practice at many institutions for rodent brain sampling evaluates three or four brain levels (typically rostral forebrain [cerebral cortex and basal nuclei], caudal forebrain [cerebral cortex and hippocampus with either diencephalon or rostral midbrain], and hindbrain [usually cerebellum with pons and/or cerebellum with medulla oblongata]; Morawietz et al. 2004). In contrast, a recent discourse on comprehensive brain sampling (Switzer, Lowry-Franssen, and Benkovic 2011) reported that small structural lesions elicited by half of the 14 known chemical and pharmaceutical neurotoxicants reviewed might not (
Other factors must be considered when selecting among various brain sampling options. For example, the 3-level scheme may be sufficient when the test article is unable to cross the blood–brain barrier (e.g., large biomolecules). Similarly, an initial 3-level approach may be adjusted readily by sampling additional levels when the nervous system appears to be a target, as when nonpathology indices of neurological dysfunction (e.g., clinical neurological examinations, behavioral testing, quantification of test article concentrations in brain, known physical and target-binding properties of the test article, and/or a recognized mechanism of neuroactivity) are observed. This latter point emphasizes that the neuropathology portion of a general toxicity study is only one component for identifying potential neurotoxic liabilities. Indeed, this recognition is the rationale behind the routine use of 3-level brain sampling for rodents in general toxicity studies at many institutions.
An obvious challenge arising from the wide divergence between the 3- and 65-level approaches to brain sampling in general toxicity studies is to define a sampling strategy that effectively evaluates major sites for neurotoxic damage without unnecessarily complicating the neuropathology analysis. The number of brain levels that must be evaluated to ensure that major regions have been examined is a current subject of debate in the toxicologic pathology community (Hale et al. 2011). Accordingly, the Society of Toxicologic Pathology (STP) established a Working Group on Nervous System Sampling to assess current sampling practices as performed during standard nonclinical general toxicity studies and provide recommendations to guide the routine neuropathology portion of future general toxicity studies. The Working Group was given a charter with four specific aims. The first charge was to recommend what neural structures should be regularly sampled during GLP-type general toxicity studies (Tier I) performed in common mammalian test species (e.g., rodent, dog, and nonhuman primate). The second charge was to suggest tissue trimming schemes to facilitate sampling of these regions. The third charge was to define what conventional stains and special neurohistology procedures, if any, should be employed routinely in general toxicity studies. The fourth charge was to advocate what format should be utilized to most efficiently and effectively document microscopic findings of the neuropathology assessment in reports destined for review by regulatory bodies. The recommendations given below with respect to suggested procedures (Table 1) and particular neural structures to sample (Figure 1 and Table 2 [rodent] or Figure 2 and Table 3 [nonrodent species]) as well as means for documenting that they have been assessed (see example in Table 4) are based on experiences and opinions of the Working Group members 1 as well as selected input from the global toxicologic pathology community 2 received during a two-month public comment period in the third quarter of 2012. The rationale for recommending the proposed brain regions for sampling is given in the Appendix. Where consensus among Working Group members and/or STP members was clearly lacking on certain points, several mainstream options have been included and discussed with respect to their potential advantages and disadvantages.
Routine practice recommendations for microscopic evaluation of neural organs during nonclinical general toxicity studies.

Major landmarks for level orientation during brain sampling in GLP-type nonclinical general toxicity studies in adult rodents.

Major landmarks for level orientation during brain sampling in GLP-type nonclinical general toxicity studies in adults of nonrodent species.

Nervous system sampling—an explicit reporting example for use in pathology reports for nonclinical general toxicity studies.

aThis list is an example based on the recommendations given in this article and could be modified readily by an institution to fit its own research and development needs. All these brain structures are reliably present within the levels defined in Figures 1 and 2, and as such can be assured of evaluation without specific documentation.
bThis structure is generally sampled only in general toxicology studies using rodents (removed with the brain in noninhalation studies, but sampled
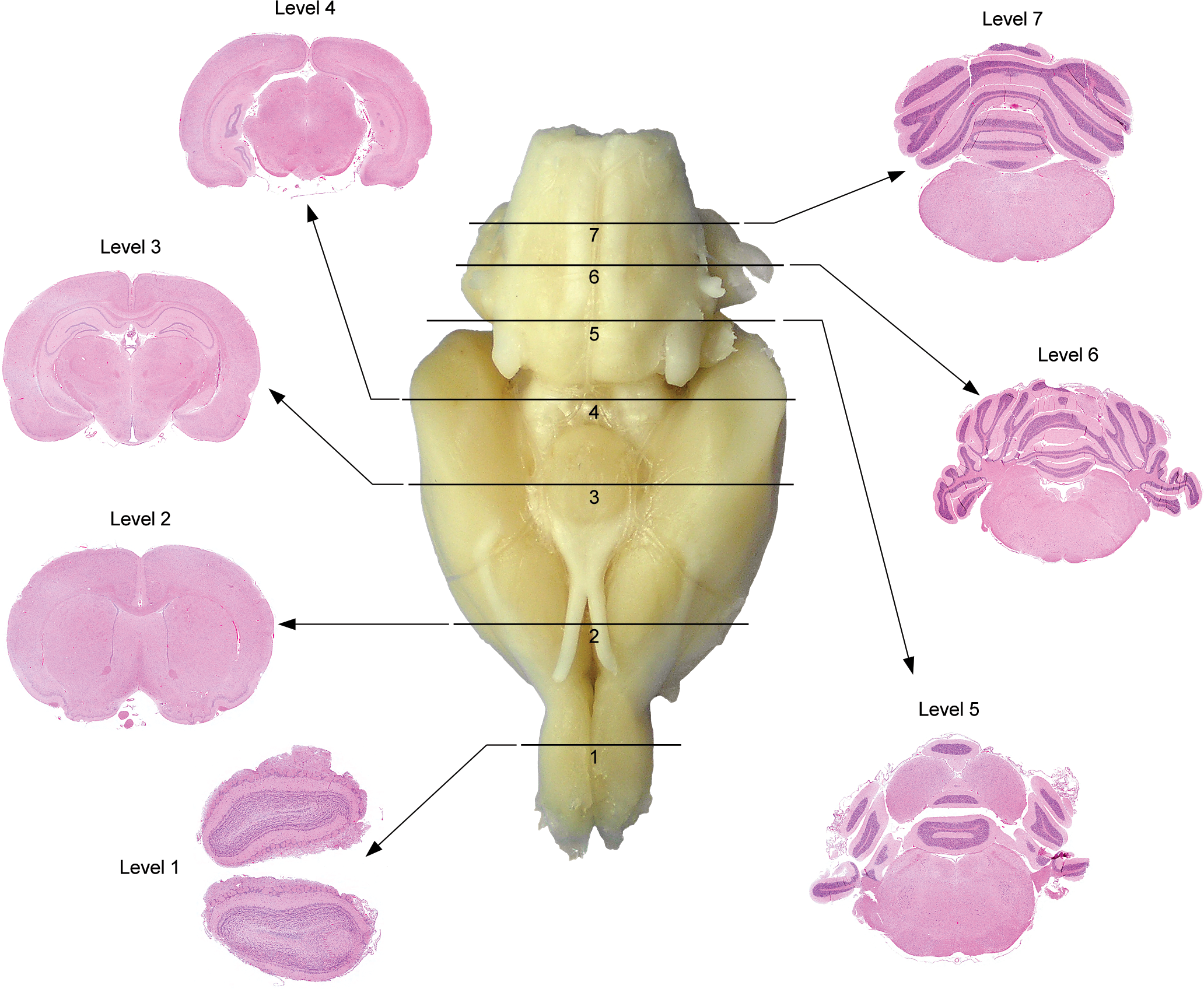
Trimming the adult rodent brain. Representation of external landmarks on the ventral surface of the adult rat brain (central whole-organ image) used to consistently attain coronal sections with generally similar internal structures (peripheral H&E-stained sections) for neuropathology assessment during general toxicity studies. The solid black lines show where to place transverse cuts, and the black arrows point to the sections produced by trimming the brain in this manner. (
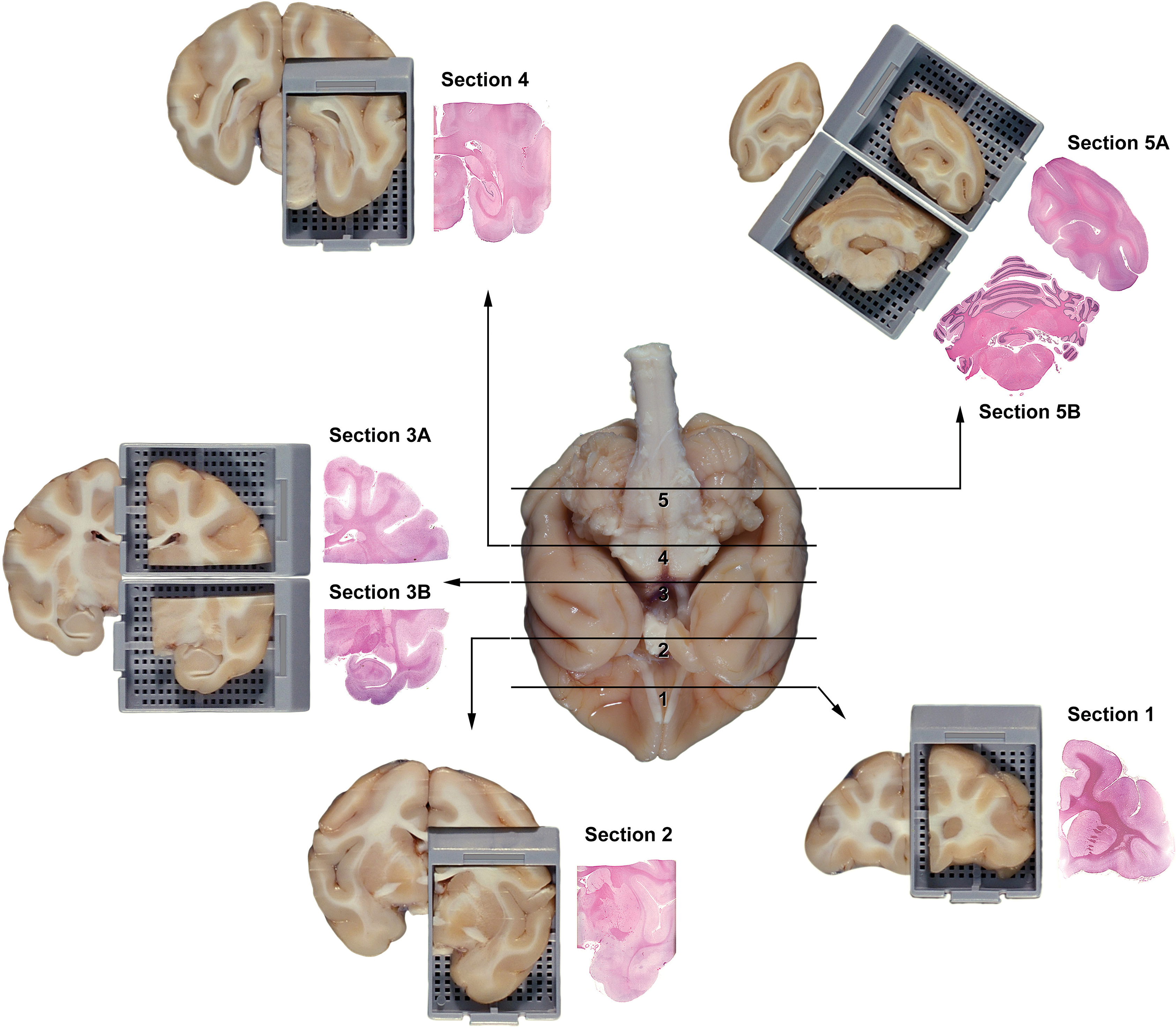
Trimming the adult nonrodent brain. Representation of external landmarks on the ventral surface of the adult nonhuman primate brain (cynomolgus monkey; central whole-organ image) used to consistently harvest coronal hemisections with generally comparable internal structures (peripheral H&E-stained sections) for neuropathology assessment during general toxicity studies (adapted from Pardo et al. 2012). The solid black lines show where to place transverse cuts, and the black arrows point to the sections produced by trimming the brain in this manner. The rostral face of each tissue slice (depicted in the tissue blocks) will be placed down in the cassette. (
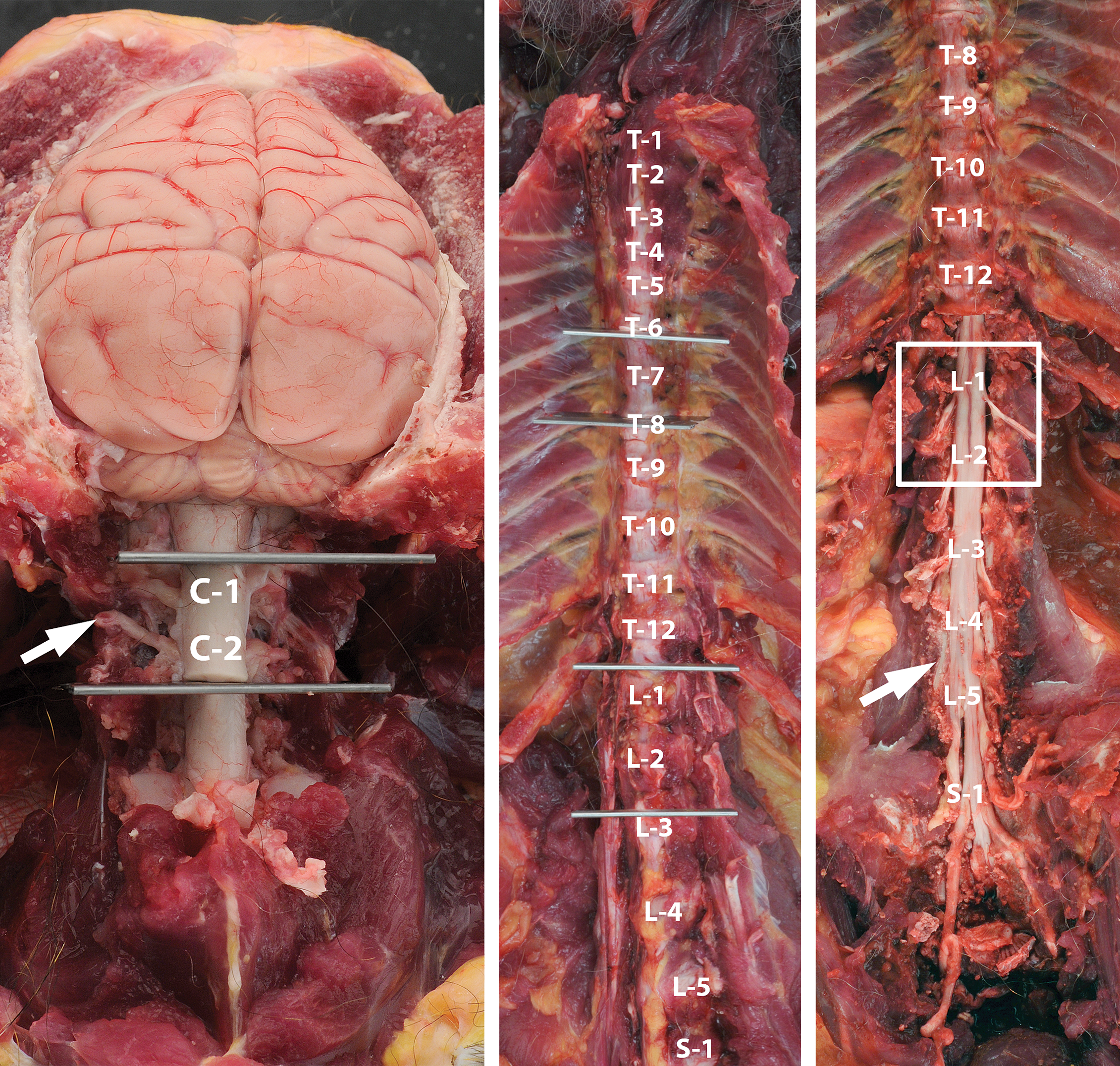
Harvesting recommended spinal cord segments. Sampling sites for the spinal cord of the adult nonrodent (cynomolgus monkey) to show the levels at which to obtain segments from the upper cervical (C1–C2), mid-thoracic (T6–T8), and lumbar intumescence (L4–L5) regions for neuropathology assessment during general toxicity studies. The regions to be harvested fall between the gray blades or within the white box; the white letter-number notations denote the identity of the associated vertebral bodies. (
Recommendations for Sampling, Processing, and Analysis of the CNS, PNS, and Eye during Nonclinical General Toxicity Studies
Tissue Collection
The manner in which the neural tissues are removed should be prescribed in either a facility standard operating procedure (SOP) or other reference document. This document should be developed in advance.
Collection of all nervous system samples should be performed to minimize structural artifacts produced by manipulation, compression, and traction of incompletely fixed tissue. The keys to keeping artifacts at a minimum are to limit pressure and stretching applied to neural tissues during the sampling process.
Brain Collection
Brain removal typically occurs at necropsy for general toxicity studies. Options for harvesting the brain vary to some degree. Important considerations in this regard are whether or not the brain is to be weighed (see below) and, for rodents, the exposure route (since olfactory bulbs often are processed
Any method of removing the skull cap to expose the brain is acceptable if it can be accomplished with no or minimal damage to superficial brain regions. Extraction usually is accomplished using rongeurs (for rodents, rabbits, and small nonhuman primates) or an oscillating saw (for dogs and larger nonhuman primates). Scissors should be available to remove the falx cerebri and tentorium cerebelli to prevent these tough membranes from shredding the delicate neural tissues that they border. Brains typically should be removed from the skull and immersed in fixative solution without any further trimming.
Recommended practice
The Working Group concludes that any careful brain collection practice may yield samples of suitable quality for microscopic assessment in general toxicity studies. The Working Group recommends that each facility follow a single standard approach for general toxicity studies in a given test species.
Spinal Cord Collection
Multiple methods for spinal cord collection may be used for general toxicity studies. The choice of method will depend on the test species and the purpose of the study.
The spinal cord of rodents may be fixed
Multiple options exist for obtaining spinal cord in rodents. The first is to complete the partial laminectomy so that the entire spinal cord may be removed. The second is to fix and process the spinal cord
The spinal cord of nonrodent species generally is removed at necropsy. The usual method is to isolate short sections (2–3 vertebrae in length) of the cervical, thoracic, and lumbar vertebral column by cutting through the appropriate intervertebral joints (Figure 3), using a thin metal instrument to cut the spinal nerve roots, and gently removing the spinal cord. An alternative is to undertake a partial laminectomy using an oscillating saw or pair of rongeurs, but this approach can significantly increase the length of the necropsy. After the cord has been isolated, the thick
Recommended practice
The Working Group judges that the spinal cord segments to be evaluated should be exposed or removed from the vertebral column at necropsy to ensure adequate fixation.
Nerve Collection
Various techniques may be used to acquire suitable samples for histology. The usual specimen is the sciatic nerve, which may be collected from one limb or, ideally, from both. The nerve is exposed by reflecting and/or removing the overlying skeletal muscle. In rodents, the whole hind limb may be fixed by immersion in NBF to avoid tension-induced structural artifacts. In nonrodent species, the unfixed nerve may be isolated and placed in a cassette, applied to an index card, or stapled to an acetate strip prior to fixation. If necessary to address specific questions, the location of the sample site (i.e., proximal vs. distal) can be identified by labeling one end of the specimen.
A unique consideration in nonhuman primates is the potential for damaging the sciatic nerve if an
In all species, acquisition of more distal hind limb nerves and glutaraldehyde fixation followed by postfixation in osmium tetroxide to better stabilize myelin lipids (Bolon et al. 2008) are common practices used during dedicated neurotoxicity studies (Tier II). These special sampling procedures are not currently a conventional component of general toxicity studies (Tier I), and the Working Group sees no evidence that they should become so.
In most general toxicity studies, the head, vertebral column, and carcass typically are discarded without isolating cranial nerve ganglia, DRG, parasympathetic ganglia (except those located within organs), and sympathetic chain ganglia unless there is sufficient reason (e.g., evidence of in-life neurological dysfunction) to retain these structures. The Working Group proposes no change to these practices during general toxicity studies.
Recommended practice
The Working Group deems that nerves may be exposed and then either fixed
Sensory Organ Collection
The eye with the optic nerve is the only special sense organ that commonly is evaluated during nonclinical general toxicity studies (Morawietz et al. 2004). The best preservation is achieved if the globe and attached optic nerve are gently removed from the orbit as soon as possible at necropsy, followed by removal of extraocular fat and skeletal muscle; glands associated with the eye (e.g., Harderian) may be left in place. Where possible, optic nerve specimens should be 0.5 cm in length to permit examination of nerve fibers over multiple internodes.
Specimens ideally should be identified specifically as being either the right or left globe, especially when the two eyes were subjected to different (i.e., local) treatments. Side-specific labeling is recommended but not essential after systemic administration since exposure to the test article would be predicted to be comparable in both globes.
Although globes of rodents may be fixed
Recommended practice
The Working Group believes that the eyes and attached optic nerves should be removed and cleaned of excess extraocular fat and muscle prior to fixation. Both globes should be collected. Individual labeling (i.e., left vs. right) is recommended.
Fixation
In our experience, immersion fixation affords an acceptable means of preserving the CNS and PNS of animals during general toxicity (Tier I) studies, while perfusion fixation is generally reserved for dedicated neurotoxicity (Tier II) studies or other specific objectives. The key to obtaining acceptable immersion fixation is standardizing the materials and techniques used in harvesting neural tissues.
Fixation of the CNS and PNS
Neutral buffered 10% formalin (NBF [∼3.7% formaldehyde]) is acceptable for routine fixation of the CNS and PNS in general toxicity studies. Ideally, brain and spinal cord should be fixed for at least 48 hr prior to trimming, particularly in the nonrodent species (which have comparatively large CNS organs). This practice will minimize the extent of cytoarchitectural artifacts, especially dark neurons (Garman 1990; Jortner 2006), that commonly arise when incompletely fixed neural tissue is handled.
Neural tissues typically are fixed together with other major organs and tissues during general toxicity studies. This practice reduces the cost and labor of conducting multi-animal necropsies but does not lessen overall preservation of CNS and PNS structures as long as adequate volumes of fixative are utilized. The ratio of fixative solution to tissue should be at least 10 volumes of fluid to one volume tissue. An advantage of NBF is that
Commercial formulations of NBF often contain methanol as a stabilizing agent (up to 15%). Methanol may induce structural artifacts in nervous tissue, such as cytoplasmic and organelle swelling, which may impact preservation at the ultrastructural level. This problem does not diminish the suitability of NBF as a routine fixative for preserving CNS and PNS tissues in general toxicity studies for light microscopic analysis.
Fixation of the Eye and Optic Nerve
The eye and optic nerve (i.e., cranial nerve II) are considered with the neuropathology assessment because these neural structures are CNS derivatives. The retina requires special consideration when selecting the most appropriate fixative due to the relative impermeability of the dense, fibrous sclera.
For nonclinical general toxicity studies, the recommended practice for preserving eyes is immersion in fixatives containing acetic acid, like Bouin’s solution, Davidson’s solution, or modified Davidson’s solution (Latendresse et al. 2002; Somps et al. 2009). Globes are fixed for 18 to 48 hr (depending on their size) and then transferred to 70% ethanol until trimming. Fixation in NBF has been reported to be acceptable for ocular preservation in long-term rodent studies (Morawietz et al. 2004), but the experience of Working Group members is that NBF is less suited for ocular fixation both because its relatively poor penetration power permits autolysis of the retina and tissues (especially the lens) often are so brittle that acquisition of intact sections is difficult.
Rat eyes typically are fixed while intact, but nonrodent eyes (rabbits, dogs, and nonhuman primates) usually fare better when a small aperture is made in the sclera so that fixative enters the vitreous chamber more easily (Somps et al. 2009). Institutional practices for creating apertures include placing a short slit (3–4 mm) or cutting a small window (3–4 mm2) in the nasal or temporal sclera just caudal to the equator; globes may be fixed by immersion for a brief period (5–30 min) to harden the sclera prior to making such cuts. An alternative means for retinal fixation is to introduce a 1:1 mixture of NBF and 4% glutaraldehyde in phosphate buffer by intravitreal injection. The usual injection volume required to render the globe firm to the touch is 0.1 to 0.5 ml.
Globes fixed in any solution may be transferred to NBF for storage until processing. This choice permits residual ocular specimens to be retained in a common receptacle with other organs. The use of 70% ethanol for storing Bouin’s-fixed eyes tends to encourage excessive hardening of the lens over time, thus making alcohol a less desirable method for long-term (i.e., greater than 7 days) archiving of ocular tissue. These approaches generally avoid potential artifacts of overfixation (e.g., spaces in the corneal stroma, vacuolation of corneal endothelial cells, fluid accumulation and excessive swelling/separation/fragmentation of lens fibers, and/or reduced detail of photoreceptor segments in the retina). The best strategy for obtaining high-quality eye sections while minimizing artifacts is to have experienced technical staff and validated sampling procedures.
Recommended practice
The Working Group believes that immersion fixation is an acceptable method for preserving the eye (retina) and optic nerve in general toxicity studies, although a small opening in the sclera should be considered when fixing large globes (especially in nonrodent species). The retina typically will be preserved suitably if immersed in Bouin’s solution, Davidson’s solution, or modified Davidson’s solution. Glutaraldehyde or mixtures of NBF and glutaraldehyde are acceptable if the fixative is introduced directly into the vitreous chamber.
Organ Weights
Brain weight, which is less prone to fluctuation in adults than is total body weight (Michael et al. 2007), should be considered in general toxicity studies as a means of calculating relative organ weights (i.e., organ-to-brain weight ratios) for non-neural viscera. Weights need not be collected for spinal cord, nerve, and eye.
Brain weights generally should be determined prior to further sectioning. Weights may be acquired either prior to fixation (i.e., in fresh, unfixed brain) or after fixation. However, brain weights should always be taken from the same type of preparation for all subjects in a given study. Practices (including fixation times) should be standardized for all animals in a study, and this information should be tracked in the historical database. Differences in fixation may alter brain weights, often serving to increase them (Bauchot 1967; Leibnitz 1967; Douglas et al. 1981).
Whenever brain weights are obtained, it is essential that the brains be removed and trimmed in a similar fashion. For example, a potential variable in rodent toxicity studies is whether or not the olfactory bulbs are removed from the skull. The large size of the olfactory bulbs in rodents (typically 6–7% of total brain weight) has the potential to substantially impact brain weight measurements. The decision regarding whether to include olfactory bulb may be dictated by the experimental design, as olfactory bulbs typically are left in the skull for inhalation toxicity studies. A common variable with nonhuman primate brains is the extent to which the rostral (frontal) cortex is penetrated by the oscillating saw during brain collection; impacts on brain weight may be minimized by employing trained prosectors who carefully select the location of the rostral cut needed to free the skull using reliable gross landmarks (e.g., the rostrodorsal margin of the orbital rim). Similarly, the level at which the brainstem is transected before weighing the brain must be standardized in all individuals of a given species. The brainstem should be transected at a standard distance caudal to the obex (i.e., the most readily identified landmark for reproducible trimming of this region). Some laboratories standardize brain size by cutting the brainstem with a scalpel immediately caudal to the foramen magnum, as the head is being detached from the body at necropsy.
Recommended practice
The Working Group holds that brain is the only neural organ that should be considered for routine weighing in general toxicity studies. Brain weights can be assessed before or after fixation. It is essential that brains from all study animals are removed and trimmed in a standard fashion before they are weighed. The Working Group counsels that olfactory bulbs be included when weighing the rodent brain if the study design does not involve the inhalation route of administration.
Tissue Trimming
The key balance to strike in nervous system sampling during nonclinical general toxicity studies is to inspect major targets of neurotoxic agents while not unnecessarily magnifying the cost, labor, and time required to complete the study. This balance is dictated by the nature of general toxicity studies, which serve as screens for major effects in multiple organ systems rather than comprehensive assessments of any single system. As such, the expectation is that major areas of the CNS and PNS (Table 4) will be surveyed in general toxicity studies (Tier I) and that more detailed evaluations (if warranted) will be left to subsequent dedicated neurotoxicity studies (Tier II) or added to general toxicity studies on a case-by-case basis using knowledge of the target or specific biologic/toxicologic effects.
Routine procedures for trimming neural tissues of common species should be detailed in either a facility SOP or similar reference document. Standardization of brain trimming planes in this fashion will promote consistency in brain sampling among animals for a given study. Establishing such conventions also may facilitate comparison of current data sets with historical control data.
In accordance with standard practice, grossly visible lesions in neural organs should be trimmed even if they are located in a region that would not normally be sampled in the typical trimming levels. Similarly, more extensive nervous system sampling may be warranted during general toxicity studies if routine sampling does not adequately address safety concerns raised by the presence of treatment-induced clinical abnormalities (not previously identified and characterized) and/or prior recognition of a specific anatomic area of potential toxicity that would not be consistently included in the routine survey samples. The decision whether to increase the extent of nervous system sampling in the course of a general toxicity study should be left to the professional judgments of the study pathologist and study director.
Care must be taken when trimming neural specimens to avoid introducing artifacts. Thick samples (e.g., brain and spinal cord) may be sliced at necropsy to permit more ready penetration of the fixative solution, although the Working Group recommends avoiding such slicing to avoid the generation of artifacts. If fresh organs are trimmed before they have been completely fixed, the cut edges should not be harvested and processed for microscopic examination.
Brain Trimming
General Principles
Several options are available for trimming the brain. The most common approach for small-sized brains (i.e., rodents) is to generate multiple full-face coronal (transverse) slices (Figure 1). For large-sized brains (i.e., nonrodents), typical preparations also produce tissue slices in coronal orientation; these are not full-face sections (unless 2 × 3-inch slides are used) but rather coronal hemisections (full or partial) from one side of the brain (Figure 2). An alternative sometimes employed for small-sized brains is to divide the organ longitudinally along the midline; this approach typically is utilized for non-GLP exploratory studies (Jordan et al. 2011). Following this latter approach, multiple coronal hemisections (usually 3–5) from one hemisphere as well as the intact opposite hemisphere (oriented with the mid-axial surface down in the cassette so that the connectivity of the white matter tracts may be viewed) are embedded in a single block. The preconditions for unilateral sampling are that bilateral or contralateral clinical signs are not observed
In our experience, the easiest means of procuring consistent sections in any species is to use definitive gross anatomic landmarks, which are best seen externally on the ventral aspect of the brain (Figure 1 [rodent] and Figure 2 [nonhuman primate]). The cutting blade may be oriented by free-hand trimming or using a brain matrix (designed to deliver slices of standard thickness [2–3 mm] with a consistent vertical orientation) or other positioning apparatus (e.g., a walled trough; Pardo et al. 2012) fitted for the appropriate species. The key to an effective brain survey during general toxicity studies will be to confirm that sections are well matched among subjects using internal neuroanatomic landmarks. Regulatory scientists who reviewed the Working Group’s recommendations preferred that rodent brains be trimmed with a brain matrix as this method standardizes the orientation and position of the trimming plane among animals. The Working Group recognizes that such reproducibility is desirable but notes that variations in brain size, especially in very young and very old rodents, preclude the unthinking adoption in all cases of a matrix device for brain trimming.
The Working Group recommends that brain sampling for general toxicity studies include sections from throughout the brain of each animal. Certain brain structures should be sampled routinely: caudate/putamen, cerebellum, cerebral cortex, choroid plexus, hippocampus, hypothalamus, medulla oblongata, midbrain, olfactory bulb (for rodents), pons, and thalamus. Brain regions adjacent to these major structures also will be available for analysis, and the Working Group anticipates that pathologists will follow the routine practice of evaluating entire sections rather than limiting the examination to the main features. The Working Group recognizes that different institutions vary with respect to evaluation of the rodent olfactory bulb for general toxicity screens but judges that this structure merits consideration for routine examination due to its importance as a potential target for inhaled (Colin-Barenque et al. 1999) and ingested (Crews et al. 2000) small molecule toxicants as well as a rich source of neural stem cells (Lennington, Yang, and Conover 2003). That said, the Working Group acknowledges that significant functional and structural differences exist between macrosmatic species (i.e., olfactory-dependent, such as rodents) and microsmatic species (i.e., visually dependent, like humans), suggesting that routine sampling of the rodent olfactory bulb may have limited impact on hazard identification and risk assessment for humans. Accordingly, the Working Group recommends that institutions should retain the flexibility to decide for themselves whether or not to harvest olfactory bulbs for general toxicity studies in rodents, especially in assays where the test article is delivered by routes other than inhalation.
The brain trimming strategy to be used for rodent carcinogenicity bioassays also deserves further reflection. The current brain sampling practice for such studies in many institutions is to assess 3 to 4 coronal brain levels (generally rostral forebrain, caudal forebrain, and hindbrain; Morawietz et al. 2004). Proponents of maintaining this 3-level scheme hold that toxic (including preneoplastic) neural changes will be acceptably surveyed in multiple other GLP-type, repeat-dose, chronic (6- or 12-month), general toxicity studies that precede the carcinogenicity bioassay. Additional points raised in favor of the 3-level approach are that the prior removal of many subjects at interim necropsies spaced over long periods is ill-suited to evaluating typical end points of toxicity in a systematic manner and that expanding the number of brain levels to be evaluated will break with sampling standards for well-established repositories of historical data as the North American Control Animal Database (NACAD) and the Registry of Industrial Toxicology Animal-data (RITA). The contrasting position in support is that carcinogenicity studies may be viewed simply as a prolonged chronic study in which sampling more than 3 brain levels may offer a broader risk assessment for treatment-related effects (preneoplastic and small neoplastic lesions). More neoplasms have been reported in the brains of ethylene oxide-treated rats if the histopathological evaluation includes more than the three common sections (e.g., compare Garman, Snellings, and Maronpot 1985 to Garman, Snellings, and Maronpot 1986). Detection of the additional microscopic lesions did not alter the conclusion regarding the carcinogenic potential of this agent.
Practical Application
The Working Group concludes that the appropriate strategy for surveying the brain during nonclinical general toxicity studies is to focus on major structures that will be present in standard trimming levels (e.g., Figure 1 and Table 2 for rodent and Figure 2 and Table 3 for nonrodents) rather than debating the number, position, and/or orientation of specific brain levels that should be evaluated. In particular, the Working Group advocates that the brain be sampled to permit structural analysis of major centers engaged in cognition, homeostatic control, motor functions, and sensory modalities—with the specific choice of regions to be specified in the institution’s SOP or reference document. In other words, as long as the brain levels to be assessed contain key regions (e.g., those listed in the abstract) and any other areas needed to support institution-specific research initiatives, and as long as the same level exhibits relatively homologous features among all animals in the study, the choice of trimming strategy may be defined by the institution to support its own research and development focus.
Primary advantages of this structure-based approach are that the list of brain regions to be sampled routinely may be adapted to all species, and that flexibility is retained with respect to the orientation (coronal vs. parasagittal) in which the structures are viewed and the specific planes through which the brain is trimmed. In general, sufficient brain regions may be sampled bilaterally in rodents using 6 to 7 coronal levels (Figure 1) grouped on two standard-sized (25 × 75 mm) glass slides. Inclusion of an additional (8th) coronal level through the middle forebrain (i.e., midway between trimming planes 2 and 3 in Figure 1) would allow the entire rat brain to be embedded and available for analysis. In larger species (e.g., dog and nonhuman primate), screening of equivalent regions also can be accomplished using 6 to 7 partial coronal hemisections (Figure 2) grouped on 4 to 6 standard-sized slides. If hemisections of bigger brains need to be subdivided to fit on standard-sized slides, all pieces should be processed to ensure adequate sampling of both superficial and deep regions (i.e., cassettes 3A and 3B for trimming plane 3 in Figure 2). For systemically administered compounds, the Working Group deems that unilateral sampling in nonrodent species will be an adequate microscopic evaluation for general toxicity studies.
Recommended practice
The Working Group is of the opinion that brain sampling for general toxicity studies, including carcinogenicity studies, should focus on key structures that house the CNS centers for major cognitive, homeostatic, and somatosensory functions. In studies using rodents, the Working Group recommends that the olfactory bulb be considered among the key structures to be examined. The specific planes and orientations for trimming should be left to the discretion of the sponsoring institution as long as the resulting sections are fairly consistent among animals in the study. Where available, use of a brain matrix to select the trimming levels may increase the degree of structural homology in sections among animals from a given study and also among animals across different studies, but free-hand trimming is an acceptable practice in the hands of suitably trained technicians.
Spinal Cord Trimming
During general toxicity studies, the spinal cord typically is evaluated in the cervical, thoracic, and lumbar regions using transverse sections (Morawietz et al. 2004). The Working Group judges that a cranial cervical segment, a mid-thoracic segment, and lumbar intumescence segment should be used in the microscopic assessment for general toxicity studies. The cranial cervical region (approximately C1) is an important area to evaluate because this location is a sensitive site for evaluating damage to long ascending sensory tracts in the white matter (e.g., primary pathways: cuneate and gracile tracts and secondary pathway: spinocerebellar tract). A mid-thoracic segment (about T6–T8) permits analysis of the lateral horn (intermediolateral cell column), which holds preganglionic neurons for the sympathetic division of the autonomic nervous system. The lumbar intumescence (approximately L4–L5) is examined because its motor neurons supply the long axons that comprise the sciatic nerve, which is a typical site for PNS sampling; this part of the spinal cord also contains the distal portions of descending motor pathways (e.g., corticospinal tracts). Sampling of specific lumbar spinal cord segments and their CNS/PNS interface is not feasible as each segment exists in the vertebral canal at a level increasingly cranial to the corresponding vertebral body, so that trimming at vertebrae L4–L5 actually samples the sacral spinal cord and surrounding
While opinions differ regarding the appropriate method for trimming the spinal cord, the Working Group recommends that the routine spinal cord evaluation examine sections in both transverse and longitudinal (or oblique) orientation. Conventional transverse sections are warranted because they permit reliable analysis of gray matter and white matter, and the positions of somatotopically arranged fiber tracts and nuclear columns can be readily defined. The longitudinal/oblique plane is suggested for inclusion since, in the experiences of Working Group members, damage affecting only a few axons is far easier to see (especially in the thoracic segment) if extended lengths of white matter tracts encompassing multiple internodes are available for evaluation; by definition, such lengths cannot be observed in transverse sections. The longitudinal section may represent a true parasagittal plane taken lateral to the midline, or it may be an oblique section that angles across the midline; advantages of the oblique view are that it exposes a larger area of gray matter and central canal as well as all white matter tracts to examination. As with brain evaluation, consistent trimming is necessary to obtain relatively homologous spinal cord sections across all dose groups.
Recommended practice
The Working Group thinks that spinal cord sampling should include cranial cervical, mid-thoracic, and lumbar segments. Both transverse and either longitudinal or oblique orientations should be examined.
PNS Trimming
Sampling of the PNS during nonclinical general toxicity studies should include a large hind limb nerve (sciatic and/or tibial). Again, the Working Group recommends that the PNS analysis include nerve sections in both longitudinal and transverse orientations. The transverse plane often provides a readier means of assessing the density and numbers of myelinated and unmyelinated fibers. The longitudinal view is included because axonal damage is more readily detected if an extended length (multiple internodes) of the nerve fiber is available for examination. This representation may be achieved readily in nerve specimens approximately 1 cm in length.
In the experiences of Working Group members, autonomic ganglia and DRG need not be routinely examined during general toxicity studies except insofar as they occur within samples of other organs. If ganglia are to be evaluated specifically, the DRG may be examined
Recommended practice
The Working Group holds that the PNS may be screened sufficiently in general toxicity studies by examining a major somatosensory nerve (e.g., sciatic and/or tibial). The nerve should be sampled in both longitudinal and transverse orientations.
Sensory Organ Trimming
Intact eyes and attached optic nerves of adult rodents are placed in a cassette without further trimming for histological processing and embedding. More pristine preservation of retinal tissue in adult rats is reportedly obtained by creating a small scleral window to permit better entry of solutions into and escape of air bubbles out of the vitreous chamber during tissue processing. Both eyes may be processed in the same cassette if the sides do not need to be individually identified. Rodent eyes should be trimmed in the vertical plane to include the retina and a longitudinally oriented specimen of the optic nerve. Routine inclusion of an optic nerve cross-section when sampling the eye and optic nerve is recommended by some practitioners.
Eyes of larger animals typically are trimmed after fixation to produce a sagittal (mid-axial) representation through the entire globe (Render et al. 2008; Somps et al. 2009). Ideally, sections will sample the retina through the region of the optic disc (i.e., the site at which the ganglion cell axons exit the eye to form the optic nerve) in addition to other non-neural ocular structures. The Working Group considers this practice to be sufficient for general toxicity studies as it allows all intraocular and periocular tissues to be evaluated in a single block. The specimen is harvested using a sharp, fairly rigid blade (e.g., Tissue Slicer Blade; Thomas Scientific, Swedesboro, NJ) to penetrate the sclera (the site of the first cut), lens, and cornea in sequence. In dogs, a perpendicular (vertical) cut is achieved by right angle orientation to the ciliary groove, which is visible on the caudal surface of the globe; this orientation allows sampling of both tapetal (dorsal and central) and nontapetal (ventral and peripheral) portions of the retina. Rabbit eyes, though lacking a tapetum, usually are trimmed in the perpendicular plane as well. For nonhuman primates, the eye is trimmed along a horizontal plane in order to include the macula in the section (Render et al. 2008). Since any cuts after the initial slice may cause artifacts, the trimmed specimen should be as thick as possible. This is permitted by use of extra-deep cassettes (e.g., Tissue-TEK® Mega Cassette; Sakura Finetek USA, Inc., Torrance, CA). Alternatively, the caudal portion of the globe and retina may be isolated so that the wall of the vitreous chamber, including the attached retina, may be embedded without the hard lens. Much care is needed with this latter technique to avoid artifacts such as retinal detachment.
Recommended practice
The Working Group recommends that eyes be trimmed to obtain a sagittal section of the retina. The trimming plane will vary by species, but the orientation for a given species should be consistent among all animals of a given study. The optic nerve will be sampled (usually in longitudinal orientation) at its point of attachment to the globe. Inclusion of an optic nerve cross section should be considered.
Tissue Processing
For nonclinical general toxicity studies, the Working Group recommends that all neural specimens from the treatment groups selected for initial evaluation should be processed in the same time frame to avoid any systematic variation in such factors as the lengths of time spent in fixative or various dehydrating solutions. A good means of standardizing the effects of fixation and processing across treatment groups is to include cassettes from animals in different treatment cohorts within each processing “run” so that any handling-related artifacts are balanced across all dose groups. That said, each institution retains the flexibility to select which samples should be selected for immediate processing (e.g., control and high-dose animals only) and which should be obtained but archived with the wet tissues (e.g., low-dose and mid-dose animals, olfactory bulbs from noninhalation studies).
Neural tissues slated for evaluation in general toxicity studies typically are subjected to the same histological processing regimen (i.e., paraffin embedding) that is employed for samples from other organ systems. The Working Group supports continuation of this practice as it is compatible with the screening nature of general toxicity studies and is cost-effective (because no special processing is required for neural specimens). However, an important processing consideration for neural tissues (especially CNS) is that they not be held in alcohol baths of automated tissue processors for prolonged periods (e.g., over the weekend) as this practice may result in large, irregular holes in white matter tracts (especially the deep folia of the cerebellum).
Recommended practice
The Working Group reckons that the morphology of neural specimens (CNS, PNS, and eye/optic nerve) may be suitably preserved for general toxicity studies by routine embedding in paraffin. The neural tissues can be processed at the same time as tissues from other body systems. Samples from different treatment groups should be mixed randomly across processing runs.
Neurohistology Methods
The Working Group recommends that routine microscopic assessments for general toxicity studies be based on standard H&E-stained sections. Targeted neurohistological techniques need not be performed during general toxicity studies as a routine practice.
In instances where neurologic behavioral or morphologic changes discovered during the preliminary assessment warrant further review, additional sections from affected region(s) or additional domains may be cut later to permit special procedures to be performed. Examples of likely ancillary methods include conventional neurohistology stains, such as cresyl violet (to reveal fine neuronal cytoarchitecture) and Luxol fast blue (to view myelin integrity); various immunohistochemical biomarkers of cell origin, like glial fibrillary acidic protein (to detect reactive astrocytes at sites of subacute to chronic brain injury) and ionized calcium binding adaptor molecule 1 (Iba1, to reveal microglia); and techniques to detect neuronal degeneration, such as amino cupric silver, Fluoro-Jade B, or Fluoro-Jade C. The recent literature contains many superb articles that address the rationale and technical considerations for deploying these special procedures (Fix and Garman 2000; Fix et al. 1996; Barone et al. 2000; Switzer 2000; Krinke et al. 2001; Switzer and Butt 2011; Schmued et al. 2005).
Recommended practice
Most Working Group members conclude that the initial screen for toxicant-induced microscopic changes in the CNS and PNS during general toxicity studies may be done acceptably using H&E alone. Additional targeted neurohistology procedures may be performed
Approach to Neuropathology Assessment
The strategy used for evaluating the nervous system during general toxicity studies is a qualitative assessment equivalent to that for any other organ or tissue (Table 1). The initial analysis usually involves comparing samples from control and high-dose groups, with evaluation of animals in other dose groups being undertaken later if warranted by findings in the high-dose cohort. The Working Group thinks that quantitative methods (e.g., morphometry and stereology) need not be a routine practice in general toxicity studies.
The initial microscopic evaluation of neural tissues from general toxicity studies generally should be conducted in an uncoded (“unblinded” or “unmasked”) fashion. In other words, the study pathologist should be acquainted
Recommended practice
The Working Group judges that the routine microscopic evaluation of nervous tissues for general toxicity studies should be a qualitative analysis performed in an uncoded (“unblinded”) fashion.
Neuropathology Documentation in the General Toxicity Study Report
The final report for a general toxicity study, as it relates to evaluation of the nervous system, will contain all the parts of a conventional pathology report (e.g., a pathology narrative together with individual animal and summary data tables) while providing detailed descriptions of particular neuropathology methods used for the study. With this requirement in mind, the Working Group holds that the Methods section should be constructed in one of two formats. Regardless of the choice, the study pathologist must be free to use professional judgment in determining the most appropriate method to categorize lesions.
Implicit reporting option
In its simplest form, this approach reports that major regions of the brain and spinal cord as well as the nerve, eye, and any other protocol-specified neural tissues were sampled, processed, and examined in the appropriate dose groups. If this approach is taken, it would be helpful (but is not required) to define the trimming planes used for brain sampling in the Methods section, either by specifying external (e.g., optic chiasm) or internal (e.g., anterior commissure) anatomic landmarks used to position the cuts and/or by providing a species-specific reference for brain trimming (e.g., rodents: Rao et al. 2011; dogs: Garman 2003; nonhuman primates: Pardo et al. 2012). Alternatively, many institutions instead list such organ-specific details in a SOP or similar reference document rather than stating them for particular organs within every pathology report; this approach is an acceptable means of addressing the issue. Rodent brain trimming also may be defined by noting the approximate Bregma levels for trimming (as presented in published species-specific brain atlases 3 ). If appropriate, lesions in the individual animal data tables are referenced to regions that are affected under the specific organ (brain, spinal cord, eye, etc.) in the diagnosis, modifiers, or comment section (e.g., “brain, neuronal necrosis, frontal cortex” or “brain, neuronal necrosis [Comment: in frontal and occipital cortex]”). If a lesion is defined in a discrete region, the same region should be assessed in as many other animals as possible so that the true incidence of its occurrence in the various dose groups may be ascertained. However, when multiple regions of an organ contain a lesion that has no obvious regional distribution (e.g. “brain, perivascular infiltrate, mononuclear cell”), identification of the specific brain region affected may not be necessary or appropriate. Structures that have no changes are not listed in the data tables (an “implicit reporting” arrangement in which unmentioned structures are considered to have been “within normal limits”). If there are no lesions in an entire organ, the organ is marked as normal.
If implicit reporting is selected, the regulatory scientists who reviewed the final draft of these recommendations uniformly requested that the final pathology report include sufficient methodological detail to understand how the neuropathology analysis was performed. From their perspective, the most desirable mechanisms for transmitting this information when an implicit reporting format is used would be to either include the institutional SOPs/reference documents for nervous system sampling and processing as appendices or else insert literature citations on detailed brain trimming practices for the test species within the Methods section.
Explicit reporting option
This strategy would include a specific list of key nervous system structures that were evaluated as a table in the Methods section of the pathology report (see Table 4 for one possible example). A list of this kind will distinctly document in a single place within the report that anatomic domains (defined by clear gross margins and/or unique cytoarchitectural features) supporting critical neural functions were chosen for screening. This approach presumes that all structures at the levels containing these key landmarks also will be assessed (which is standard practice for neuropathology evaluations during toxicity studies). Including a specific list of major structures is an efficient and effective means of verifying that a systematic survey has taken place, even when modest variability in orientation occurs among sections from different animals. An additional reason to consider explicit reporting of neuropathology data is that regulatory scientists who reviewed the final draft of these recommendations expressed an obvious preference for this format.
A necessary requirement for adoption of explicit reporting as a routine practice in GLP-type general toxicity studies is a practical mechanism for its deployment that does not require formal verification (“quality assurance”) that each site was evaluated in every animal. The Working Group judges that this mechanism will have two foundations. The first is a detailed SOP or similar reference document detailing the tissue trimming scheme; ideally, this guide will show low-magnification photographs of representative tissue sections resulting from the standard trimming planes used by that institution. The second foundation is a carefully selected set of major brain regions (e.g., the sample set listed in Table 4) that are assured of being included in the sections for all animals. The Working Group members believe that this latter standard is achievable using either free-hand trimming or brain matrices. The Working Group believes that this approach offers sufficient confirmation that the brain was screened in a suitable manner for general toxicity studies.
Neuroanatomical nomenclature
Regardless of the reporting method, unambiguous names (e.g., frontal cortex, medulla oblongata, lumbar intumescence, and sciatic nerve) are preferable to more generic terms (e.g., brainstem, cerebrum, hind brain, and nerve). These precise names should be reported in the data table as the sites for any neural lesions that occur in a specific region or pattern (examples: “brain, neuronal necrosis, frontal cortex” for the implicit option or “frontal cortex, neuronal necrosis” for the explicit option).
Nomenclature in all reports should conform to the most recent standards. Brain structures should be named using terms in appropriate brain atlases (e.g., dog: Palazzi 2011; mouse: Franklin and Paxinos 2007; nonhuman primate: Saleem and Logothetis 2007; Palazzi and Bordier 2008; Paxinos et al. 2008a, 2008b; rat: Paxinos and Watson 2007). Toxicant-induced neural lesions should be designated using accepted diagnostic nomenclature (Kaufmann et al. 2012).
Recommended practice
The Working Group deems that either the implicit option or the explicit option are acceptable means for reporting neuropathology data from general toxicity studies if the data tables and pathology narrative clearly identify regional patterns of lesion distribution within the nervous system. The key factor in selecting between these two reporting methods should be the ability to communicate sufficient detail regarding the nature and extent of the microscopic analysis to end users of the data set (e.g., institutional managers and regulators). In studies with neurotoxic lesions, explicit reporting is apt to more transparently communicate the extent of induced damage to specific neural structures. In studies where no neurotoxic lesions are found, implicit reporting certainly is sufficient.
Discussion
Basic Philosophy
The Nervous System Sampling Working Group was charged by the STP with: (1) considering the several current approaches to sampling neural organs during GLP-type nonclinical general toxicity studies and (2) then developing recommendations for a systematic approach to neural sampling in such screens. After considerable discussion, the Working Group members concluded that a rigid “one-size-fits-all” sampling approach is inappropriate. Instead, the Working Group is of the opinion that a more reasonable and attainable goal is to define a set of critical neural structures that should be examined and then let the experiences and needs of individual pathologists and institutions direct the exact choice of sampling, processing, and analytical procedures required to suitably screen the nervous system.
STP Recommendations for Nervous System Sampling in General Toxicity Studies
Accordingly, the Working Group proffers the following recommendations to guide nervous system sampling during GLP-type nonclinical general toxicity testing in the future (i.e., those studies providing pivotal information to guide human risk assessment and product development decisions). Examination of the brain in early non-GLP exploratory toxicity studies may not be necessary, so nervous system sampling in these studies should be more flexible.
For GLP-type general toxicity studies, both CNS and PNS tissues may be removed at necropsy and undergo the identical tissue processing procedures used for most non-neural organs (fixing by immersion in 10% formalin, embedding in paraffin, sectioning at 4–8 µm, mounting on standard-sized glass slides, and staining with H&E; Table 1). Major neuroanatomic regions serving critical functions (e.g., cognition, homeostatic control, motor activity and sensory functions) as well as common target sites of known neurotoxic agents (mentioned for selected structures in the right column of the Appendix) should be assessed specifically in a qualitative manner with full knowledge of the treatment groups and individual animal in-life, laboratory, and necropsy findings. Special neurohistological methods need not be undertaken routinely unless a change appreciated using H&E requires further characterization. The procedures for collecting and trimming nervous system tissues should be specified in an institutional SOP or reference document. The sites where microscopic findings with a recognizable regional distribution occur within the nervous system should be detailed in the final pathology report using appropriate nomenclature.
The Working Group’s “routine practice” recommendations are detailed enough to provide a more systematic analysis of the nervous system (especially the CNS) in general toxicity studies and yet flexible enough to permit their implementation via relatively modest revisions of current institutional practices. In particular, brain and spinal cord sampling schemes suitable for executing these recommendations exist already in the published literature. For instance, the revised sampling protocol for the rodent brain put forth by the U.S. National Toxicology Program (NTP) provides an instructive example of these nervous system sampling proposals as they might be applied to general toxicity studies in rodents (Rao et al. 2011). Two contract research organizations subsequently have tested this updated NTP approach on rodent general toxicity studies in their own facilities. Both organizations have concluded that the increased cost and time required to prepare one additional slide and to evaluate four more brain sections are warranted due to the increase in confidence that major nervous system targets have been systematically screened. Both groups now recommend this nervous system sampling strategy for all rodent general toxicity studies and carcinogenicity bioassays to be conducted at their institutions. This experience should help allay concerns expressed by many pathologists and pathology laboratory directors that more nervous system sampling during nonclinical general toxicity studies would be unduly burdensome. Similar sampling strategies have been defined for brains of nonrodent species (Garman 2003; Pardo et al. 2012). The Working Group predicts that CNS and PNS sampling of nonrodent species at many institutions already approaches or conforms to the recommendations set forth here, and thus should not represent a major departure from current practice.
Commentary on the Routine Practice Recommendations For Nervous System Sampling
The Working Group’s final draft of the routine practice recommendations included in this article were reviewed extensively by many scientists from numerous entities throughout the world, including academic, consulting, contract research, industrial, and regulatory institutions. Respondents were primarily toxicologic pathologists, but multiple remarks also were received from neurobiologists and regulatory scientists. Most reviewers stated their appreciation for the comprehensive nature of the recommendations; the balanced coverage of the arguments for and against various neural sampling schemes; the literature citations supporting the different views; and the flexibility offered to pathology practitioners and their institutions in choosing preferred approaches for evaluating the nervous system in nonclinical general toxicity studies. Nonetheless, several points of disagreement with the Working Group’s recommendations were raised by multiple reviewers, and are thus worthy of additional consideration here. These points are ordered below using the degree of feedback received on each theme, with the earlier paragraphs representing those for which a greater number of comments were received.
The first point of disputation was the paradigm for nervous system sampling to be employed for rodent carcinogenicity bioassays. Most toxicologic pathologists believe that the conventional sampling method used to populate historical databases (i.e., 3–4 coronal sections [Morawietz et al. 2004]) is appropriate for rodent carcinogenicity studies. Two main reasons were given for this belief: the value of rodent lifetime tests is overwhelmingly higher for defining the risk of neoplastic outcomes rather than toxic lesions, and agents shown to incite a significant neurocarcinogenic response have been identified reliably using the conventional 3-level scheme for histopathological evaluation. Furthermore, many reviewers stated that more brain sampling for rodent carcinogenicity bioassays already was undertaken when the weight of evidence from prior general toxicity studies warranted a more detailed neural examination. The Working Group holds that the flexibility of the sampling options offered in these recommendations already acknowledges these arguments, so institutions may establish their nervous system sampling scheme for carcinogenicity studies to best serve their own programmatic goals. The Working Group notes that utilization of a common sampling scheme for both general toxicity studies and carcinogenicity bioassays likely will reduce quality assurance issues that might arise in facilities where several brain trimming approaches are performed.
The second point of contention raised by multiple reviewers was the means for reporting neuropathology data and specifically microscopic findings. The consensus preference of numerous toxicologic pathologists (and their institutions) is for an implicit reporting approach, for two reasons. First, the respondents felt that the general toxicity study series is a cumulative screen for risk which historically has achieved great success, and as such does not warrant major adjustment at this time. Reviewers emphasized that evidence of neurotoxicity in general toxicity studies already serves as a trigger for the study pathologist to provide more detailed topographic documentation of brain lesions through a judicious use of diagnostic nomenclature and an expanded narrative. The second reason given by respondents to support implicit reporting is the opinion that working groups with expertise in a specific organ/system always will argue for more documentation to fully capture the scope of toxicity in their tissue of interest. The Nervous System Sampling Working Group acknowledges this bias, but nonetheless considers that the complex three-dimensional anatomic, functional, and neurochemical complexities of the many hundred unique brain substructures offer a solid rationale for considering an explicit reporting format for this organ. Explicit reporting is the arrangement preferred by regulatory scientists for studies in which neurotoxic lesions have been detected as the details of the microscopic analysis and the implications of the microscopic data set for specific neural structures and functions may be communicated more clearly.
The third source of dissension voiced by many respondents was to question the Working Group recommendation that the olfactory bulb be sampled routinely in general toxicity studies performed with rodents. The choice of most toxicologic pathologists (and their institutions) is to harvest olfactory bulb only for studies in which the test article is administered by the inhalation route. Four arguments were raised to support this position: (1) most pharmaceutical agents are given by noninhalational routes, (2) blood-borne agents seldom result in functional or structural evidence of olfactory dysfunction, (3) humans depend on olfaction as a sensory modality to a minimal degree relative to rodents, and (4) the olfactory brain is not sampled routinely during general toxicity studies in nonrodent species. In other words, the ability to translate any lesions observed in the rodent olfactory bulbs to assess the potential risk of neurotoxicity in humans is likely to be negligible in most cases. The Working Group recognizes the foundation for these claims but maintains that routine inclusion of the olfactory bulb is appropriate for rodent studies due to the functional significance of this brain region in these species. That said, the flexibility of the Working Group recommendations affords institutions the latitude to decide for themselves whether or not to incorporate olfactory bulb into the list of protocol tissues for microscopic analysis during general toxicity studies in rodents.
A fourth point of disagreement with the Working Group’s recommendation was the contention that spinal cord sampling should be performed at multiple segments in two orientations (transverse and either longitudinal or oblique planes). Some toxicologic pathologists held that harvesting only the cervical spinal cord was acceptable for general toxicity studies, for three reasons. First, removal of the spinal cord is laborious in nonrodents due to the thick vertebral bodies, and requires considerable technical skill and time. Second, sections of cervical spinal cord contain contributions from all ascending (sensory) and descending (motor) white matter tracts, and thus represent an acceptable sample of these structures. Finally, additional spinal cord sampling beyond the cervical section routinely is done for general toxicity studies in which in-life neurological signs demonstrate the need for such an expansion. Other toxicologic pathologists sample 2 or 3 spinal cord segments but prefer to evaluate only transverse sections. The Working Group retains its recommendation that spinal cord should be sampled in the cervical, thoracic, and lumbar divisions so that region-specific structures will be available for microscopic evaluation. This view conforms to published recommendations based on negotiations among various industrial consortia (Morawietz et al. 2004), and a sampling strategy based on isolating short vertebral column regions (Figure 3) rather than partial laminectomy should minimize the labor involved in achieving this goal. The Working Group likewise maintains its position that spinal cord ideally should be examined in both transverse and longitudinal (or oblique) sections. The reason for this latter stance is Working Group members have found that subtle lesions in white matter often are more readily identified if axons and their myelin sheaths can be evaluated for relatively long distances rather than in cross-section.
A fifth point of strife regarded the suitability of immersion fixation as a routine practice for preserving nervous system samples from general toxicity studies. Comments on this subject were split unevenly between two diametrically opposed viewpoints. The majority of respondents were toxicologic pathologists who concurred with the Working Group’s recommendation that immersion fixation is an acceptable option for fixing neural organs harvested during general toxicity studies. Several reasons were given for holding this position. First, such “Tier I” surveys are designed to rapidly screen many organs from multiple systems, in which the various CNS and PNS structures represent only a subset of the important tissues. In addition, perfusion fixation may alter the morphology and weights of many non-neural organs (e.g., spleen) in ways that might preclude an effective examination of non-neural target organs. Finally, potential neurotoxic sequelae identified during general toxicity studies commonly are evaluated further in dedicated (“Tier II”) neurotoxicity studies where the focus on assessing neuropathologic end points requires perfusion fixation. The contrasting view, voiced by neurobiologists working in the research or regulatory settings, is that perfusion fixation is essential when seeking to preserve neural tissue for microscopic analysis. Adjustments to the Working Group’s recommendations that were proposed by neurobiology respondents to improve the outcome of immersion fixation are to block large specimens (e.g., nonrodent brains) and create openings into protected sites (e.g., eyes) at or shortly after (30 min) completion of the necropsy, as well as to increase the fixative-to-tissue ratio from the standard 10:1 used for non-neural specimens to at least 25:1 for brain and spinal cord. While adoption of these adjustments might provide marginal improvements in neural tissue preservation, the Working Group reasons that current immersion fixation practices for neural organs represent an acceptable trade-off between efficient harvesting of multiple tissues versus optimal preservation of neural organs for routine use in general toxicity studies.
A sixth point of debate centered on the appropriate battery of neurohistological stains required for effective routine microscopic analysis of nervous tissue sections during general toxicity studies. Again, remarks on this subject were divided unevenly between two opposite positions. Almost all respondents were toxicologic pathologists who believed that CNS and PNS tissues from general toxicity studies may be surveyed effectively using standard H&E-stained sections, followed if necessary by additional serial sections prepared to demonstrate particular lesions or cell markers. In contrast, neurobiologists indicated a preference for automatically producing a set of 4 to 6 brain sections for each animal processed to highlight general architecture, axonal alterations, neuronal integrity, myelin sufficiency, and reactive glia, as is currently undertaken in dedicated (Tier II) neurotoxicity studies. One neurobiologist further stated that brain sections should be standardized at a thickness of at least 25 µm to allow visualization of entire neuronal nuclei and dendritic arborizations, which can span dozens of micrometer. The Working Group concurs that a more systematic neuropathology evaluation would be provided by these modifications, but recognizes that their inclusion as a standard practice in a general toxicity study would defeat the purpose of such Tier I studies as rapid screens for effects in multiple organ systems. Therefore, the Working Group reckons that the decision if and when additional neurohistological methods might be needed in the course of general studies to more fully delineate neural findings should be left to the discretion of the study pathologist and institution.
A few respondents, representing mainly neurobiologists but also including a few toxicologic pathologists, suggested that the recommendation for the nerve to be submitted for microscopic evaluation should be amended from the automatic choice of the sciatic or tibial nerve to the purposeful selection of the most relevant nerve tissue (as supported by a scientific rationale [e.g., functional homology to humans] or the presence of clinical signs during life). The basis for this proposal is that different nerves carry distinct sets of information and utilize divergent molecular pathways. The Working Group judges that the collection of sciatic and/or tibial nerve should remain the recommended practice for general toxicity studies as the sciatic nerve is known to contain both motor and sensory fibers. In-life evidence of neurological dysfunction that can be linked to a specific nerve branch already will lead to sampling of additional nerves at necropsy.
Several respondents requested that recommendations be provided for sampling, processing, and evaluation of the pituitary gland. The basis for this request is that the pituitary gland resides in the
Concluding Remarks
Current opinions regarding how to design and report a suitable neuropathology evaluation for nonclinical general toxicity studies differ to some degree among pathologists and across institutions. The Nervous System Sampling Working Group has spent nearly 4 years attempting to balance these views while negotiating a proposed set of comprehensive yet flexible recommendations by which nervous system sampling, processing, and analysis across venues may become more standardized and systematic over time. The STP believes that the current recommended practices will provide a useful platform for increasing the harmony and quality of nervous system sampling among institutions and across geographic regions in the near term and will help to mold communal perspectives and guide debate on this topic in the future.
Footnotes
Appendix
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Acknowledgments
The Working Group thanks Dr. Robert C. Switzer III (NeuroScience Associates, Knoxville, TN) for providing theoretical and practical insights on neuroanatomical and neurohistological topics during the course of this project. The authors also thank Dr. Deepa Rao of Integrated Laboratory Systems, Inc. (ILS; Research Triangle Park [RTP], NC) and the National Toxicology Program (NTP), U.S. National Institute of Environmental Health Sciences (NIEHS; RTP, NC), as well as Mr. David Sabio of Experimental Pathology Laboratories, Inc. (EPL; RTP, NC) for assistance in assembling (Figure 1). Finally, the Group extends their appreciation to Mr. Walter Bobrowski of Pfizer, Inc. (Groton, CT) for assistance in compiling (Figures 2 and ![]() ).
).
Authors’ Note
This document has been reviewed in accordance with U.S. Environmental Protection Agency (EPA) policy and approved for publication. The views expressed do not reflect official EPA policy, and mention of trade names or commercial products does not constitute EPA endorsement or recommendation for use.
