Abstract
The use of minipigs as an alternative nonclinical species has increased in the last 20 years. The Society of Toxicologic Pathology (STP) has produced generic “best practice” recommendations for nervous system sampling in nonrodents during general toxicity studies (Toxicol Pathol 41[7]: 1028–1048, 2013), but their adaptation to the minipig has not been attempted. Here, we describe 2 trimming schemes suitable for evaluating the unique neuroanatomic features of the minipig brain in nonclinical toxicity studies. The first scheme is intended for general toxicity studies (Tier 1) to screen agents with unknown or no anticipated neurotoxic potential; this approach using 7 coronal hemisections accords with the published STP “best practice” recommendations. The second trimming scheme for neurotoxicity studies (Tier 2) uses 14 coronal hemisections and 2 full coronal sections to investigate toxicants where the nervous system is a suspected or known target organ. Collection of spinal cord, ganglia (somatic and autonomic), and nerves from minipigs during nonclinical studies should follow published STP “best practice” recommendations for sampling the central (CNS, Toxicol Pathol 41[7]: 1028–1048, 2013) and peripheral (PNS, Toxicol Pathol 46[4]: 372–402, 2018) nervous systems.
Keywords
Introduction
The use of minipigs (Sus scrofa domesticus) as an alternative nonrodent species for biomedical research has increased substantially over the last 20 years. 1 –3 The minipig is the second most common nonrodent species utilized in Europe and North America, after the Beagle dog. 4 Several dozen minipig breeds have been produced, but the most common for biomedical purposes are the Göttingen, Hanford, Sinclair, and Yucatan varieties 5 ; the Göttingen breed in particular features most often in translational medicine experiments and nonclinical safety studies of potential drugs (small molecules and biomolecules) and medical devices. 1 –3,6,7 The main advantage in using minipigs as nonrodent models for biomedical research is their closer anatomic and physiologic similarities to humans compared to other nonrodent species. An additional advantage of using minipigs is their relatively small size, which reduces the amounts of food, space, and test article needed to perform a study as well as facilitates animal handling.
Minipigs are employed to address a wide range of research questions. Minipig models have been used to explore cardiovascular, 8 –10 dermal, 11 –13 metabolic, 14 –16 neoplastic, 17 –20 neurologic, 21 –27 and reproductive 28 –31 diseases. The use of Göttingen minipigs in toxicologic research is supported by our rapidly expanding knowledge regarding species-specific drug disposition and metabolism, 32 responses to vehicles and excipients, 33 and spontaneous background findings. 34 –36 Ongoing efforts to effectively manage lineage variation in the Göttingen breed 37 have been instrumental in stabilizing the supply of experimental animals with a consistent genetic background. Minipigs appear to be better models for immunological evaluation compared to dogs, 38 and as such have become important models for examining the immunogenicity of novel biopharmaceuticals 39,40 and in preclinical safety evaluation of stem cells 41 and vaccines. 42,43 Finally, genetically engineered minipigs have been proposed as a potential source of cells, tissues, and organs for xenotransplantation into human patients. 44,45
For routine toxicity testing, current guidance by regulatory agencies 46,47 and international consortia 48,49 generally expects that organs of the central nervous system (CNS) and peripheral nervous system (PNS) should be subjected to macroscopic and microscopic evaluation in most studies. The Society of Toxicologic Pathology (STP) has produced generic “best practice” recommendations, consistent with such guidance, that cover sampling, trimming, and processing of CNS (Toxicol Pathol 41[7]: 1028–1048, 2013 68 ) and PNS (Toxicol Pathol 46[4]: 372–402, 2018 71 ) organs for rodents and nonrodents during general toxicity studies (performed to screen agents with unknown or no anticipated neurotoxic potential). However, unique neuroanatomic features typically require that these generic recommendations be adjusted to some degree for use in each species. Existing neuroanatomic studies of the minipig and pig nervous system 23,50 –53 typically do not give detailed minipig-specific descriptions for effective nervous system sampling during toxicity studies, while one excellent guide to tissue sampling for pig models 54 provides guidance for nervous system sampling that diverges to some degree from the STP recommendation for minimal CNS sampling during general toxicity studies. The objective of the current article is to provide trimming schemes conforming to the STP “best practice” recommendations that are suitable for sampling the minipig nervous system (especially the brain) during general toxicity studies (Tier 1) and neurotoxicity studies (Tier 2, performed to characterize toxicants where the nervous system is a suspected or known target organ).
Materials and Methods
Animals
All procedures were approved in advance by the Institutional Animal Care and Use Committee in accordance with relevant national regulations 55 and the Guide for the Care and Use of Laboratory Animals, 8th edition. 56 In keeping with current 3Rs (replace, reduce, refine) principles for animal-based research, production of this brain sampling atlas was conducted as a satellite investigation using extra tissue from animals used for another approved project.
Naive, young adult, male and female Göttingen minipigs (N = 20), aged 9 to 11 months and weighing 18 to 20 kg, were procured from Ellegard Göttingen Minipig and Marshall BioResources; for this study, brain samples were collected from Marshall minipigs while spinal cord and PNS tissues (ganglia and nerves) were obtained from Ellegard minipigs. Animals were housed individually in indoor pens (area 2.5 m2, height 3 m) equipped with sawdust bedding (which was changed weekly). All installations were cleaned daily and then disinfected with Desguard 20 (principal ingredient: 2% benzalkonium chloride). Minipigs received pelleted minipig chow (SMP (E) SQC; Special Diets Services) and municipal tap water (treated by reverse osmosis and ultraviolet radiation) ad libitum. Animals were kept in biologically clean rooms with high efficiency particulate air-filtered air and a 12-hour light/dark cycle. Room temperature and relative humidity were maintained at 19 ± 3 °C and approximately 50%, respectively. Bedding and food were autoclaved before use.
Tissue Acquisition and Processing
Animals were sedated with an intramuscular injection of ketamine (12 mg/kg) and then euthanized humanely using intravenous thiopental sodium anesthetic (1 ml/kg [trade name: Pentothal]) followed by an embutramide/mebenzonium iodide/tetracaine hydrochloride euthanasia cocktail (1 mL/kg [trade name: Tanax or T-61]). Brains were exposed by removing the dorsal calvaria and dura mater. Brain attachments (cranial nerves and spinal cord) were severed, and the brain was removed and fixed while intact by immersion for at least 48 to 72 hours at room temperature in 10 or more volumes of neutral buffered 10% formalin (pH 7.4) containing approximately 1% methanol as a stabilizing agent. In addition, cervical spinal cord was collected, removed from the meninges, and fixed by immersion with the brain.
Coronal trimming planes were positioned using prominent landmarks on the dorsal (Figure 1A), ventral (Figure 1B), and/or lateral (Figure 1C) surfaces of the brain. An initial cut to divide the fixed brain in half was made by placing the blade on the ventral brain surface between the optic chiasm rostrally and the infundibulum (pituitary stalk) caudally (Figure 2). An alternative for dividing the brain in half would be to place the blade on the dorsal brain surface in the middle of the suprasylvian gyri as they meet at the midline (ie, between the two 2 dashed rectangles in Figure 1A). Subsequently, a purpose-made rectangular brain matrix (developed in-house at Pfizer for obtaining coronal sections from brains of nonrodent species) was used to obtain 3.2-mm thick slices from both halves of the brain (Figure 3); construction details for this tool have been published previously. 57 Using this matrix, each brain may be parsed into 19 full-coronal slices having 2 flat surfaces as well as unleveled rostral and caudal ends that are not trimmed further (Figures 3 and 4). For most slices that were sampled, 1 half (ie, a coronal hemisection) was placed in its own tissue cassette; 2 small slices from the caudal hindbrain (Figure 4B, levels 18 and 19) were placed intact (ie, full coronal sections) in tissue cassettes. Tissues were processed routinely into paraffin. A 5-µm thick section was obtained for each block and stained with hematoxylin and eosin.
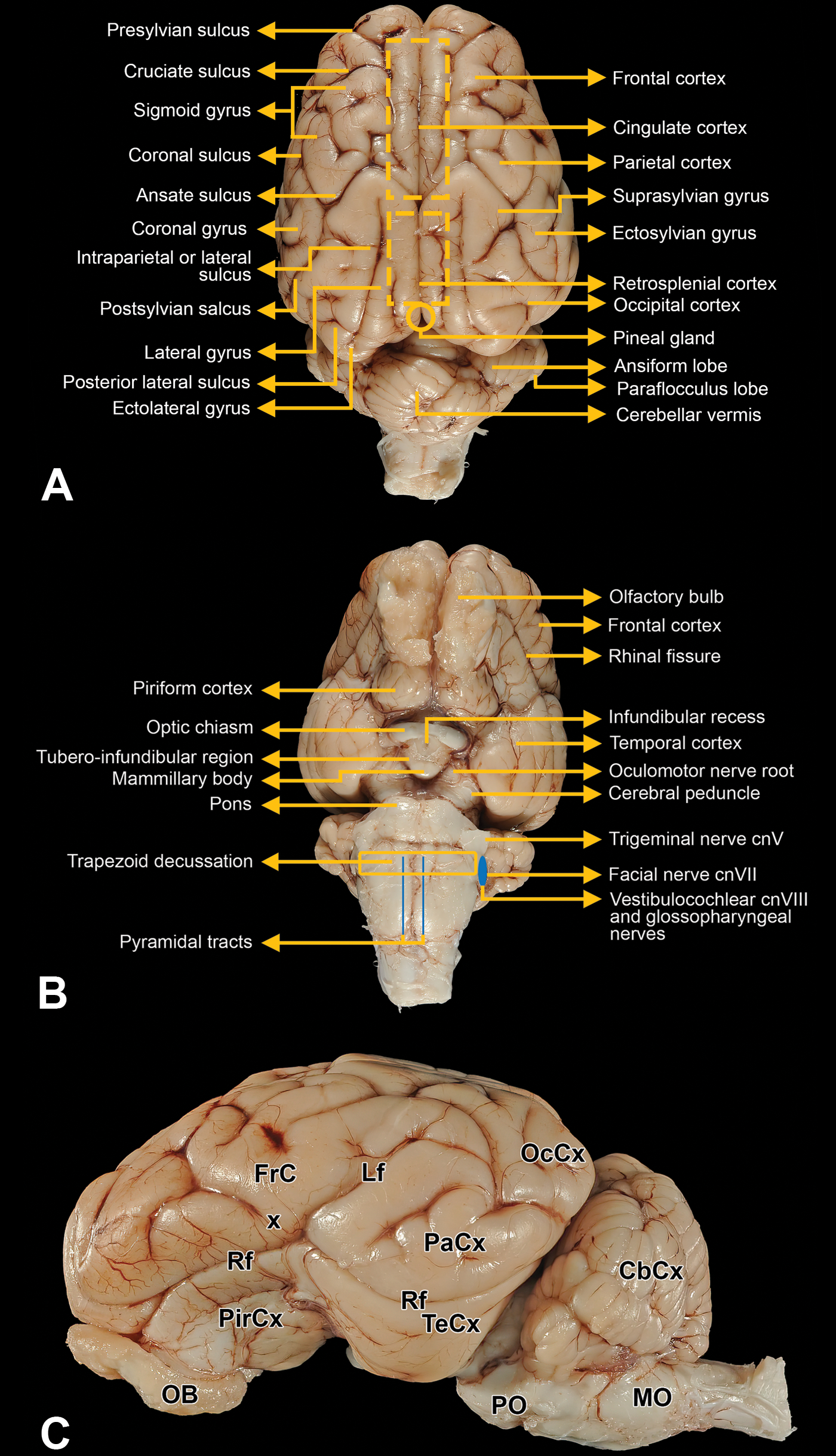
External landmarks on the dorsal (A), ventral (B), and lateral (C) surfaces of the minipig brain. These structures are useful for orienting blade placement when trimming the brain. In (C), CbCx indicates cerebellar cortex; FrCx, frontal cortex; Lf, lateral fissure; OB, olfactory bulb; MO, medulla oblongata; OcCx, occipital cortex; PaCx, parietal cortex; PirCx, piriform cortex; PO, pons; Rf, rhinal fissure; TeCx, temporal cortex.
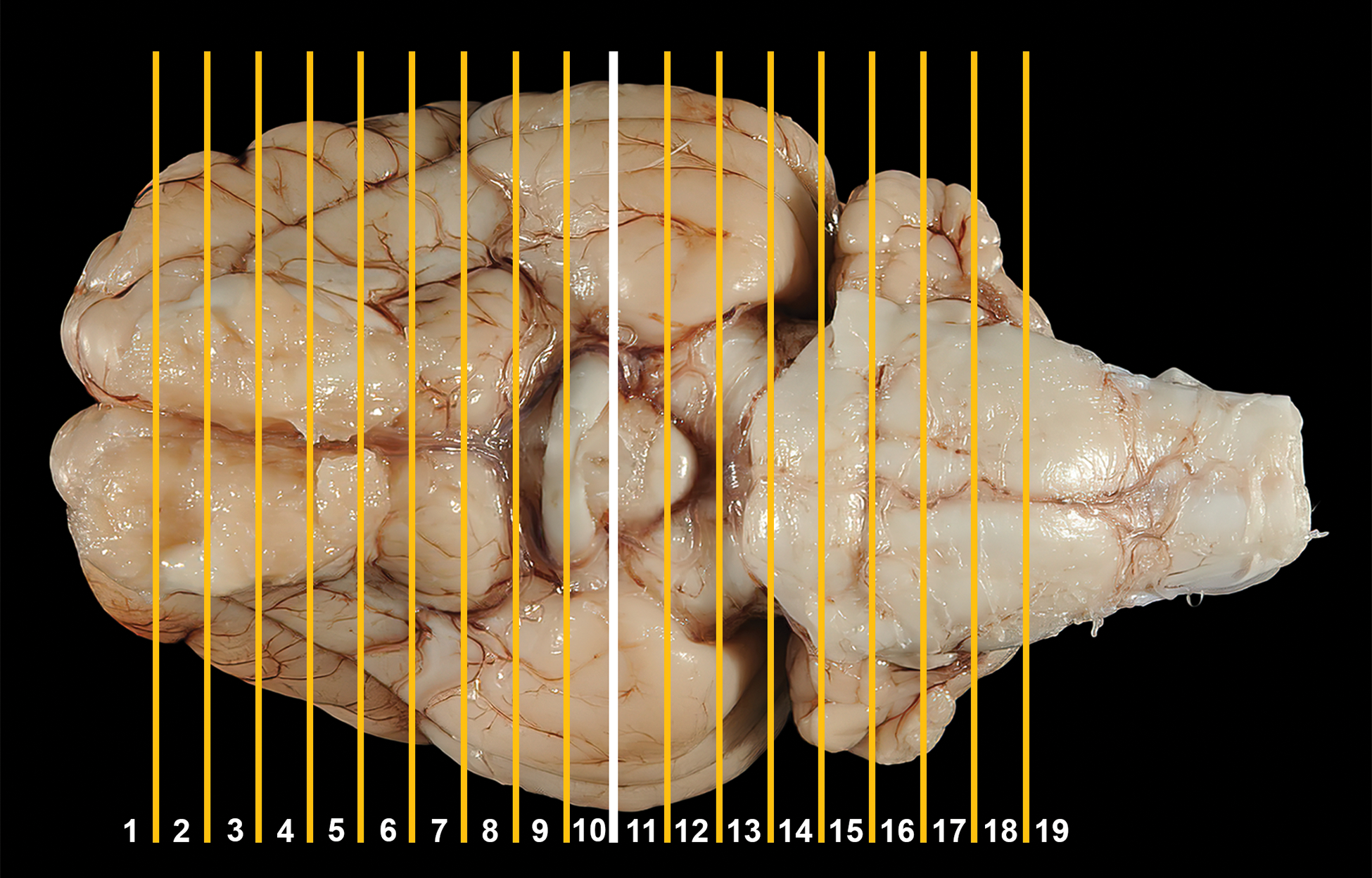
Location of trimming planes for minipig brain sectioning, ventral view. An initial cut (white line) to divide the fixed brain in half is made using the caudal aspect of the optic chiasm and the infundibulum as landmarks, after which additional parallel coronal cuts (yellow lines) are made rostrally and caudally to obtain a total of 19 3.2-mm thick slices. The tips of the rostral and caudal brain slices are not included in the sampling protocol.
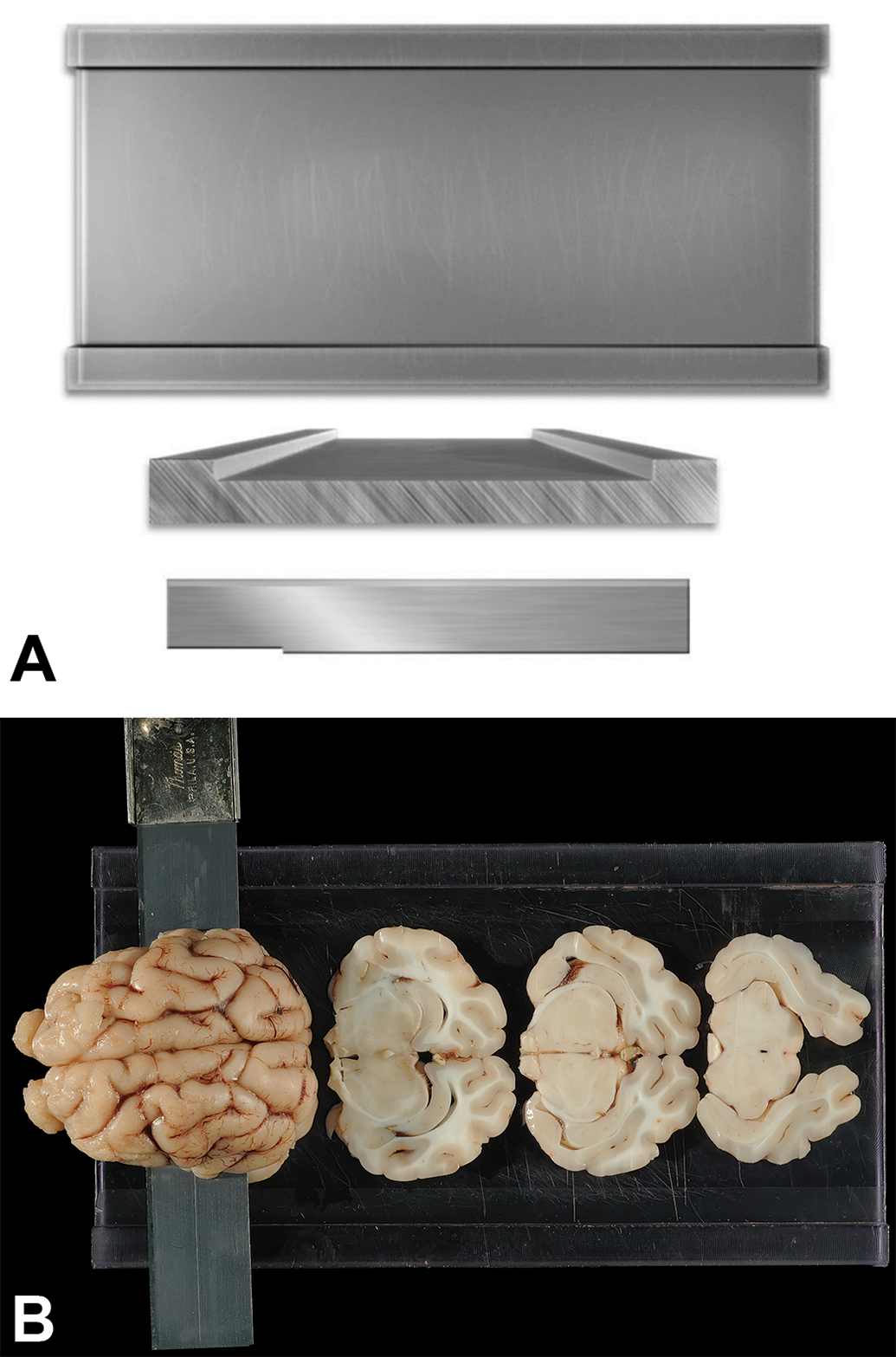
Rectangular fiberglass matrix to prepare coronal slices of nonrodent brains. A, The flat surface of the fixed brain half is placed “cut surface down” in the matrix between the 2 outer raised edges, which are 1 cm wide and 3.2 mm high. B, The tissue slicer blade resting on the raised edges is moved along the raised edges through the brain to produce 3.2-mm thick brain slices (of which 3 from the mid portion of the brain are shown here).
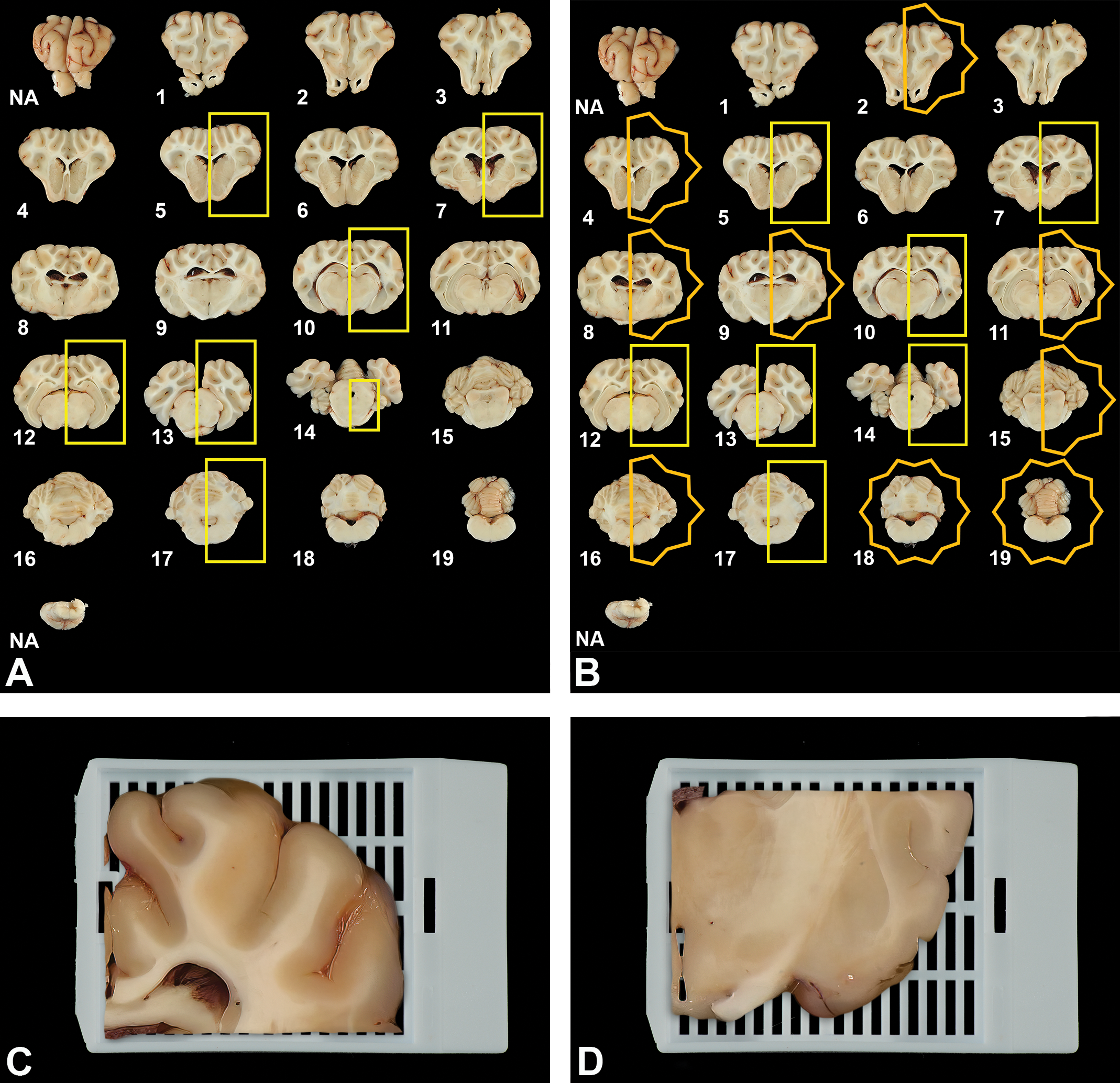
Brain trimming schemes for use in general toxicity (Tier 1, A) and dedicated neurotoxicity (Tier 2, B) studies. For both schemes, the fixed brain is sliced into 19 coronal sections. For general toxicity screening (A), a subset of 7 sections (yellow boxes) are selected at the levels of the frontal pole (No. 5); rostral (anterior) commissure (No. 7); mid-thalamus (No. 10); caudal thalamus (No. 12); occipital lobe and midbrain (No. 13); pons (No. 14); and cerebellum and brainstem (No. 17). For dedicated neurotoxicity studies (B), a subset of 16 (reached by excluding sections 1, 3, and 6) are evaluated. For both trimming schemes, the rostral face of each slice (shown here) is placed “down” in the cassette and paraffin boat to ensure that this plane is available for evaluation. Most of the brain sections are trimmed and processed as hemisections as they can typically be processed in standard tissue cassettes and mounted on standard (75 × 25 mm) glass slides. However, larger hemisections (eg, levels 8-12) may require bisection of the tissue and embedding of the 2 pieces in separate cassettes. This trimming adjustment is demonstrated using the dorsal (C) and ventral (D) aspects of the rostral thalamic section (level 9).
Neuroanatomic Annotation
The figures included in the current atlas are representative images for structures that might be found in sections made from the rostral faces of the 19 brain slices depicted in Figure 4A and B. Brain structures were annotated by comparison of minipig neuroanatomic features to those of comparable domains in other major nonrodent test species 58 –60 and humans 61 –64 as well as by reference to other neuroanatomic resources for the minipig and pig brain. 50,51,65,66 The neuroanatomic terminology conforms to the accepted Nomina Anatomica Veterinaria standard for quadruped animals. 67
Results
As in all species, nervous system sampling in the minipig for nonclinical safety testing should address both CNS and PNS structures. Harvesting the spinal cord (CNS) as well as ganglia (somatic and autonomic, as appropriate for the study objective) and nerves usually will conform to the published STP “best practice” recommendations for CNS (Toxicol Pathol 41[7]: 1028–1048, 2013 68 ) and PNS (Toxicol Pathol 46[4]: 372–402, 2018 71 ) sampling and processing that are applicable to all species.
Brain
Distinct trimming strategies were developed based on the different sampling needs for 2 classes of nonclinical safety studies. The first (or Tier 1) scheme is designed for general toxicity studies in which test articles with no or unknown potential for neurotoxicity are evaluated. This Tier 1 screen (Figure 4A) is consistent with the STP “best practice” recommendations for brain sampling as applied generally to nonrodent species. 68 The second (or Tier 2) trimming scheme (Figure 4B) is for dedicated neurotoxicity studies in which more extensive sampling is needed to evaluate effects on the nervous system. For both trimming schemes, the samples may be taken from the left hemisphere, right hemisphere, or a combination of the 2 since features evaluated during routine efficacy and toxicity studies are bilaterally comparable. In practice, institutions usually acquire all sections from a single hemisphere.
General toxicity studies
The Tier 1 scheme for trimming brains utilizes 7 coronal hemisections (outlined with yellow rectangles in Figure 4A) chosen to contain the predominant functional domains known to be targeted by proven neurotoxicants. Hemisections are used since they can typically be processed in standard tissue cassettes and mounted on standard (75 × 25 mm) glass slides; however, the largest sections may require bisection of the tissue and embedding into 2 cassettes (Figure 4C and D). The specified trimming planes are taken through the frontal cortex (level 5), rostral (anterior) commissure (level 7), mid thalamus (level 10), caudal thalamus and midbrain (level 12), occipital cortex and midbrain (level 13), pons (level 14), and cerebellum with medulla oblongata (level 17). On occasion, the study objectives may be served best by bilateral evaluation of features for a given brain region. In such instances, full coronal sections may be processed in over-sized cassettes, and the resulting sections mounted on nonstandard (eg, 75 × 50 mm) slides.
Dedicated neurotoxicity studies
The Tier 2 brain trimming scheme includes analysis of 14 coronal hemisections and 2 full coronal sections (Figure 4B). The increased number of sections is recommended since this approach is used in situations where a test article is suspected or known to produce neurotoxicity, thus warranting a more substantial evaluation. 62,68 –70 The 16 sections supplement the 7 levels (all coronal hemisections) utilized for Tier 1 screening (7 yellow rectangles in Figure 4A and B) with an additional 9 levels (7 coronal hemisections and 2 full coronal sections) that capture other major functional domains of the brain (7 yellow hemistars and 2 full stars in Figure 4B). Instances in which Tier 2 sampling may be warranted include when the test article has been reported to induce toxicity to any portion of the nervous system in published literature, when clinical observations from prior studies in other test species and/or from early human trials (rarely available) suggest that the test article may be neurotoxic, or when a quantitative structure–activity relationship exists between the test article and a similar agent with suspected or known neurotoxicity.
Possible adjustments to the trimming schemes
The 7-level (Tier 1) trimming scheme for general toxicity studies is the minimally acceptable approach to minipig brain sampling for nonclinical safety assessment to conform to the STP “best practice” recommendations for brain sampling in nonrodents during general toxicity studies. 68 This scheme is already reduced as far as possible as the microscopic evaluation is performed on coronal hemisections rather than the full sections that typically are assessed in rodents 68 and rabbits. 57 Accordingly, no further reduction to the Tier 1 scheme should be undertaken when trimming minipig brains.
Modest adjustments to the 16-level (Tier 2) trimming scheme for dedicated neurotoxicity studies might be selected at the discretion of the study design team and/or institution in certain cases, with appropriate scientific justification. For example, evaluation of fewer than 16 coronal hemisections might be elected by omitting one or more forebrain sections if the test article is suspected or known to induce effects only in the cerebellum, while one or more additional forebrain sections might be added if toxicity to the olfactory system is anticipated. In this context, the trimming levels to be omitted or added would be chosen by the study design team on a case-by-case basis.
Principal neuroanatomic features of the minipig brain
The mean brain weight is relatively stable in young adult Göttingen minipigs. Based on historical control data from 427 animals (Charles River Laboratories, Inc), the mean brain weight at 4 to 7 months of age is 56 g (range 47-65 g), while that at 6 to 8 months is 60 g (range 53-70 g). No significant difference exists between the brain weights of age-matched male and female Göttingen minipigs at these 2 time points.
The principal neuroanatomic features in all 19 brain hemisections from Göttingen minipigs are shown in Figures 5 –23. The key structures within each section are annotated. Due to spatial constraints, annotations are given only for the largest and most biologically and toxicologically significant structures (based on known regional function and known sensitivity to neurotoxicants [see Tables 1 and 2 as well as Supplemental Table 1]).
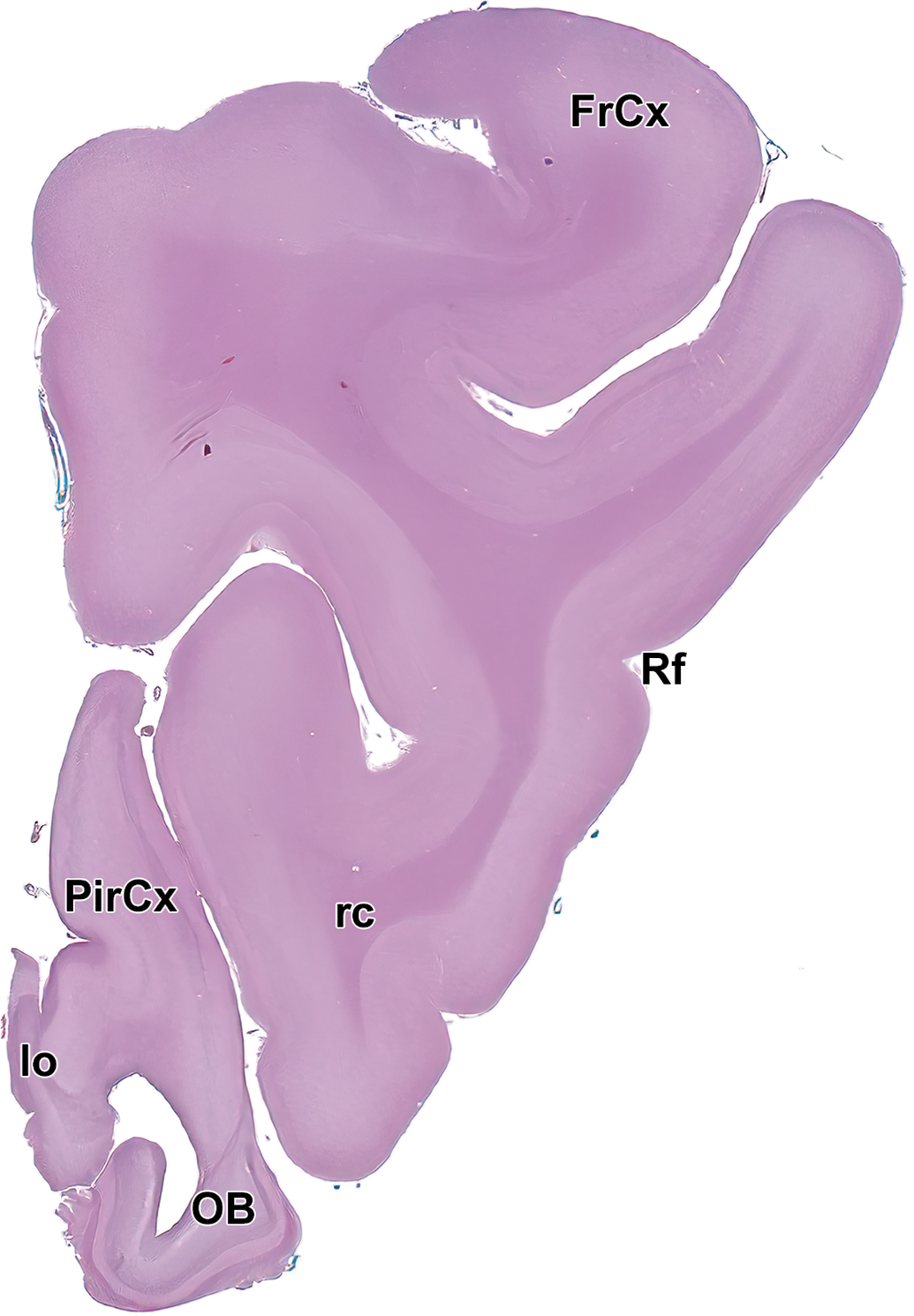
Level 1: Frontal pole and olfactory bulb I. FrCx indicates frontal cortex; lo, lateral olfactory tract; OB, olfactory lobe; PirCx, piriform cortex; rc, rostral commissure; and Rf, rhinal fissure.
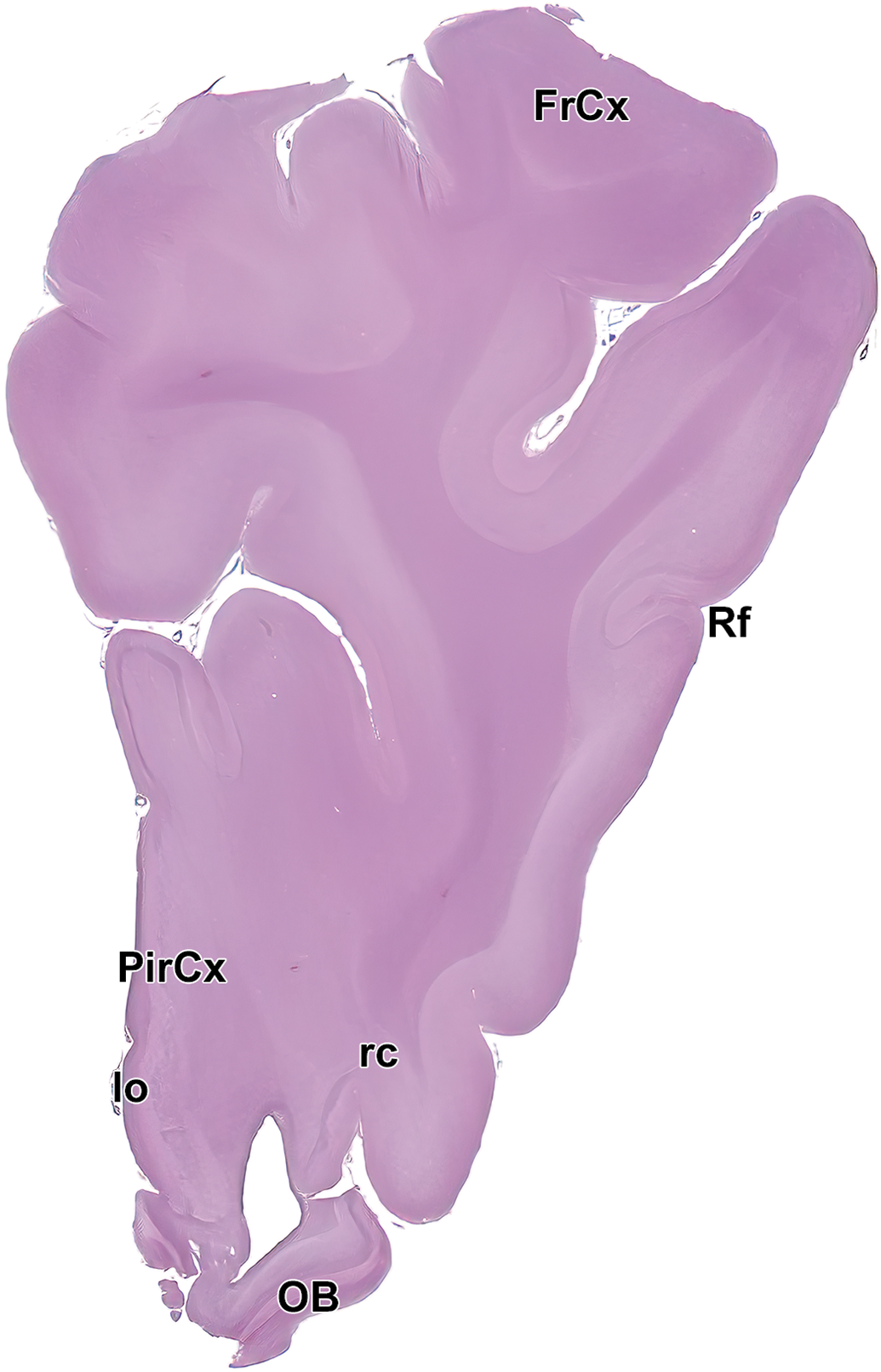
Level 2: Frontal pole and olfactory bulb II. FrCx indicates frontal cortex; lo, lateral olfactory tract; OB, olfactory lobe; PirCx, piriform cortex; rc, rostral commissure; and Rf, rhinal fissure.
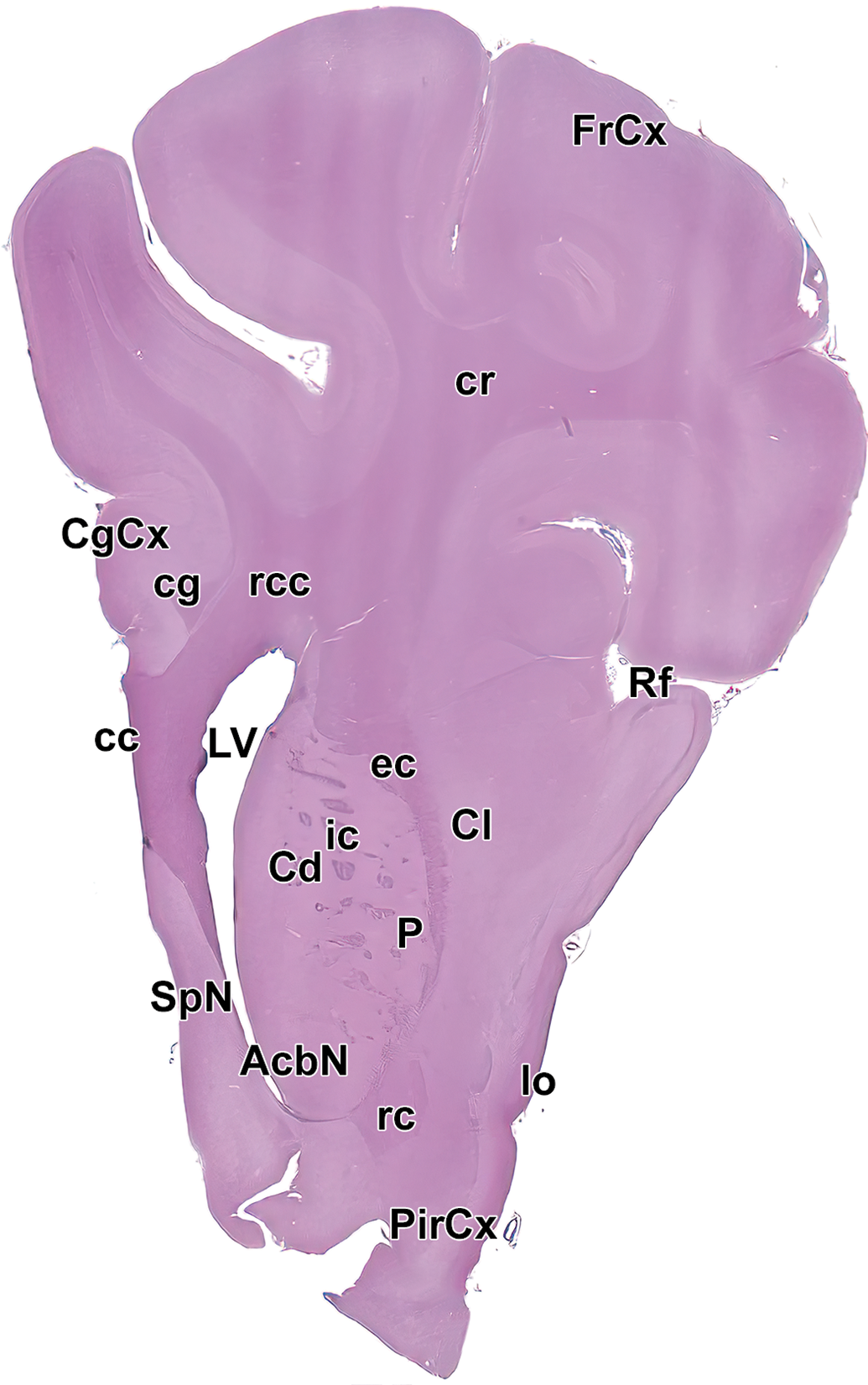
Level 3: Frontal pole, caudate nucleus, and septal nuclei I. AcbN indicates accumbens nucleus; cc, corpus callosum; Cd, caudate nucleus; CgCx, cingulate cortex; cg, cingulum; Cl, claustrum; cr, corona radiata; ec, external capsule; FrCx, frontal cortex; ic, internal capsule; lo, lateral olfactory tract; LV, left ventricle; P, putamen; PirCx, piriform cortex; rc, rostral commissure; rcc, radiation of the corpus callosum; Rf, rhinal fissure; and SpN, septal nuclei.
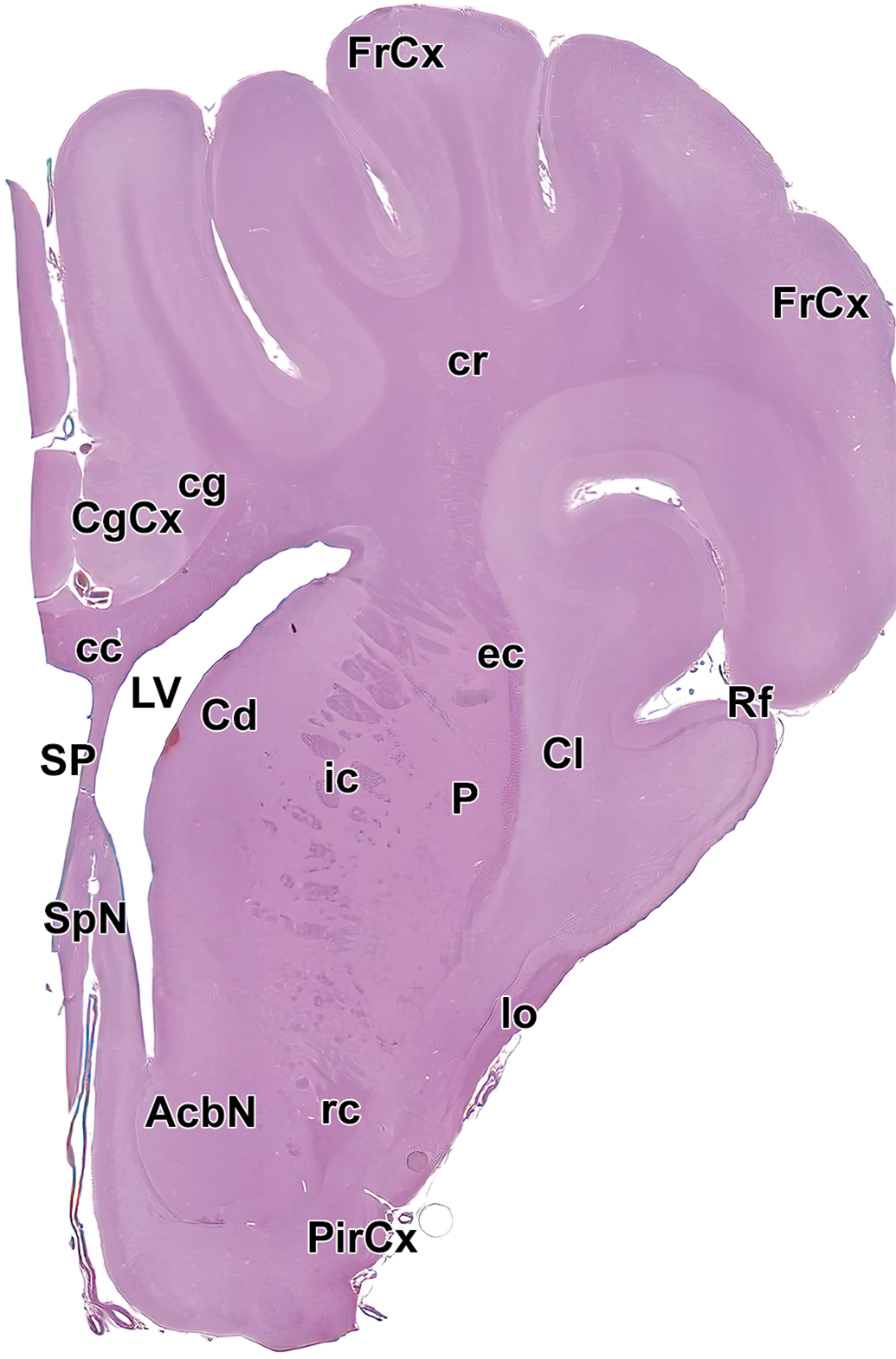
Level 4: Frontal pole, caudate nucleus, and septal nuclei II. AcbN indicates accumbens nucleus; cc, corpus callosum; Cd, caudate nucleus; cg, cingulum; CgCx, cingulate cortex; Cl, claustrum; cr, corona radiata; ec, external capsule; FrCx, frontal cortex; ic, internal capsule; lo, lateral olfactory tract; LV, left ventricle; P, putamen; PirCx, piriform cortex; rc, rostral commissure; rcc, radiation of the corpus callosum; Rf, rhinal fissure; SP, septum; and SpN, septal nuclei.
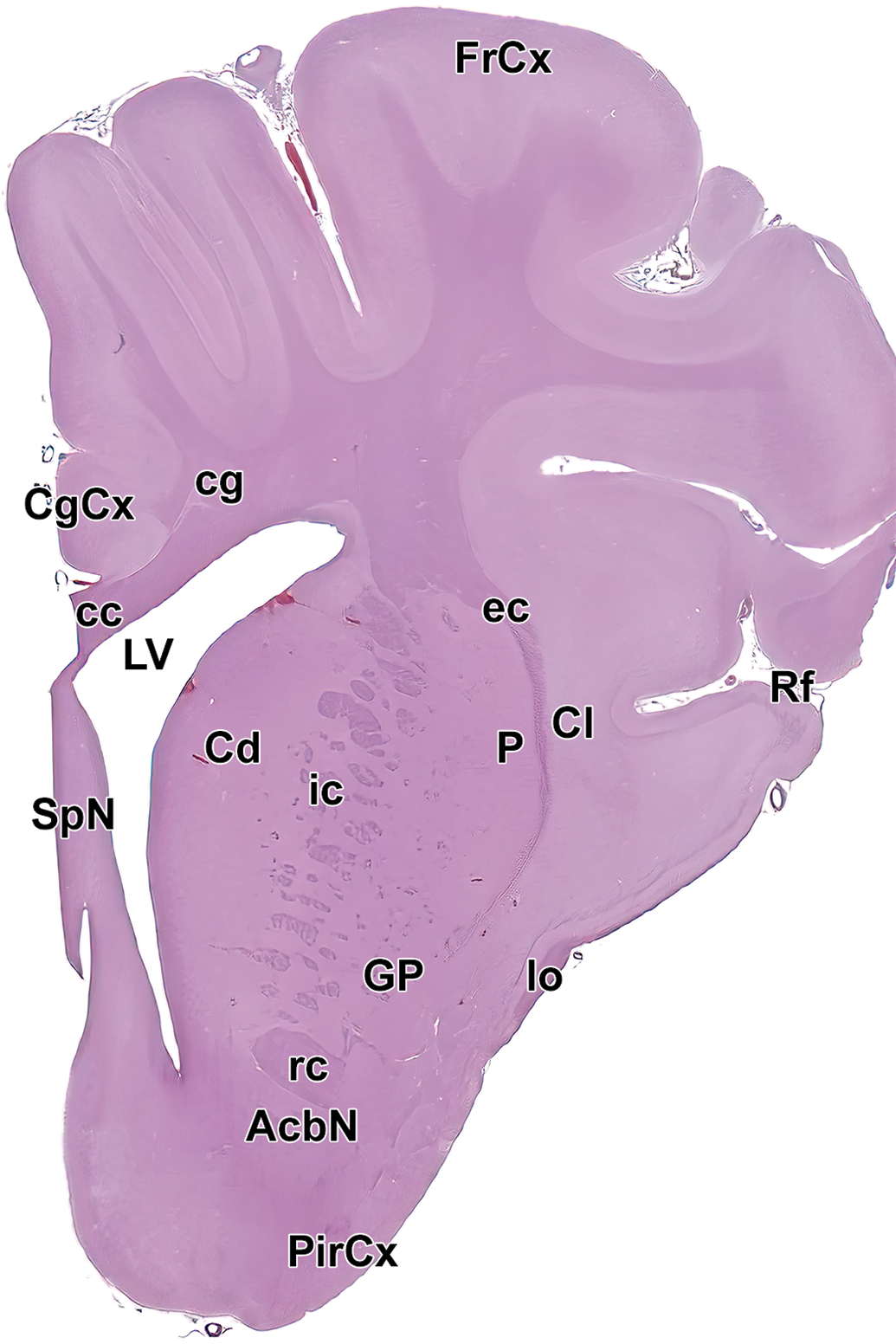
Level 5: Frontal pole, caudate nucleus, and septal nuclei III. AcbN indicates accumbens nucleus; cc, corpus callosum; Cd, caudate nucleus; cg, cingulum; CgCx, cingulate cortex; Cl, claustrum; ec, external capsule; FrCx, frontal cortex; GP, globus pallidus; ic, internal capsule; lo, lateral olfactory tract; LV, left ventricle; P, putamen; PirCx, piriform cortex; rc, rostral commissure; Rf, rhinal fissure; and SpN, septal nuclei.
Level 6: Frontal pole, caudate nucleus, and septal nuclei IV. AcbN indicates accumbens nucleus; cc, corpus callosum; Cd, caudate nucleus; cg, cingulum; CgCx, cingulate cortex; Cl, claustrum; ec, external capsule; FrCx, frontal cortex; GP, globus pallidus; ic, internal capsule; lo, lateral olfactory tract; LV, left ventricle; P, putamen; PirCx, piriform cortex; rc, rostral commissure; Rf, rhinal fissure; and SpN, septal nuclei.
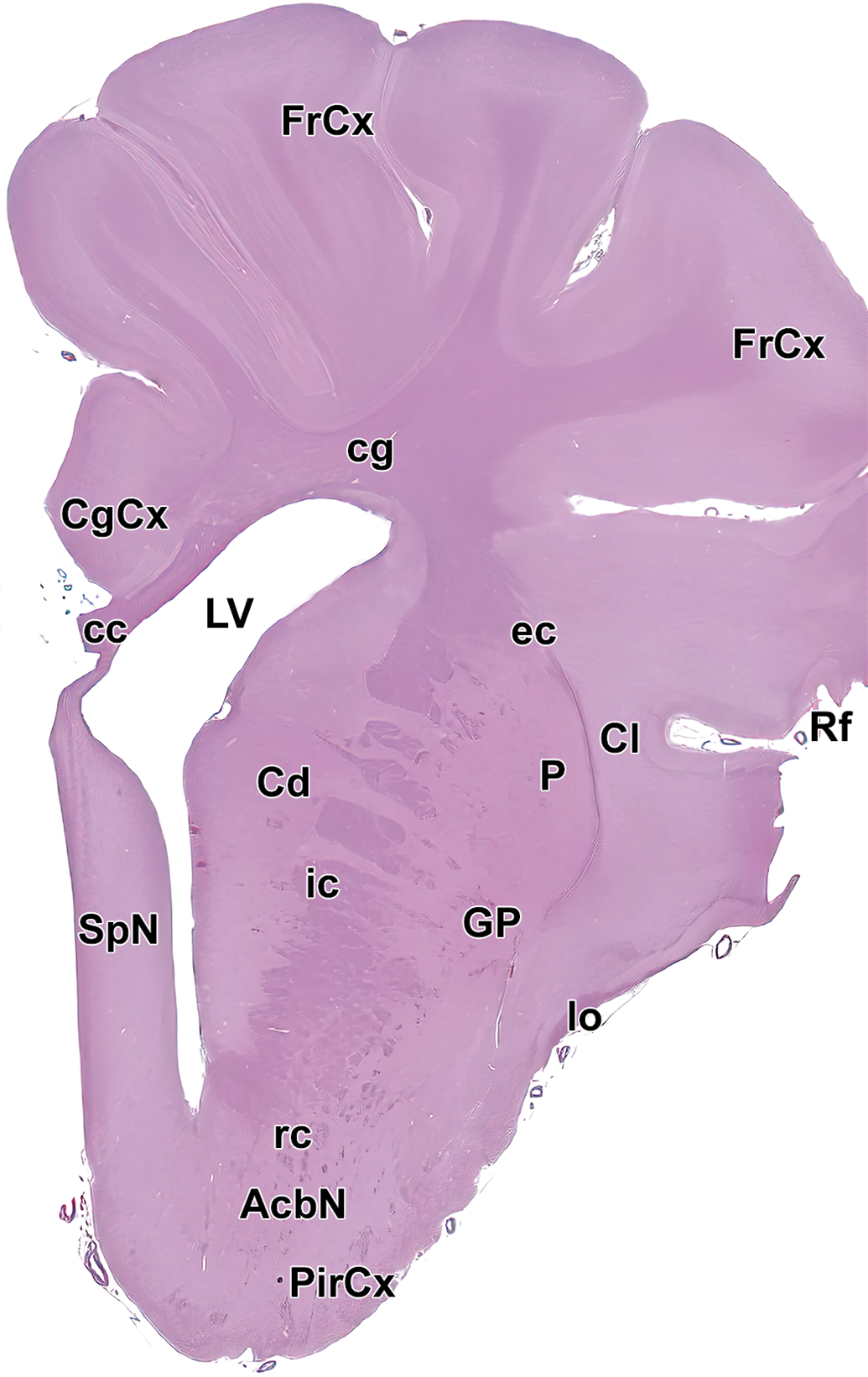
Level 7: Frontal, parietal, and temporal cortices; rostral commissure; and fornix. cc indicates corpus callosum; Cd, caudate nucleus; cg, cingulum; CgCx, Cingulate cortex; Cl, claustrum; CP, choroid plexus; ec, external capsule; FrCx, frontal cortex; fx, fornix; GP, globus pallidus; ic, internal capsule; IG, indusium griseum; Lf, lateral fissure; Ls, lateral sulcus; LV, left ventricle; P, putamen; PaCx, parietal cortex; PirCx, piriform cortex; PrO, preoptic region; rc, rostral commissure; Rf, rhinal fissure; st, stria terminalis; TeCx, temporal cortex; VP, ventral pallidum; and 3V, third ventricle.
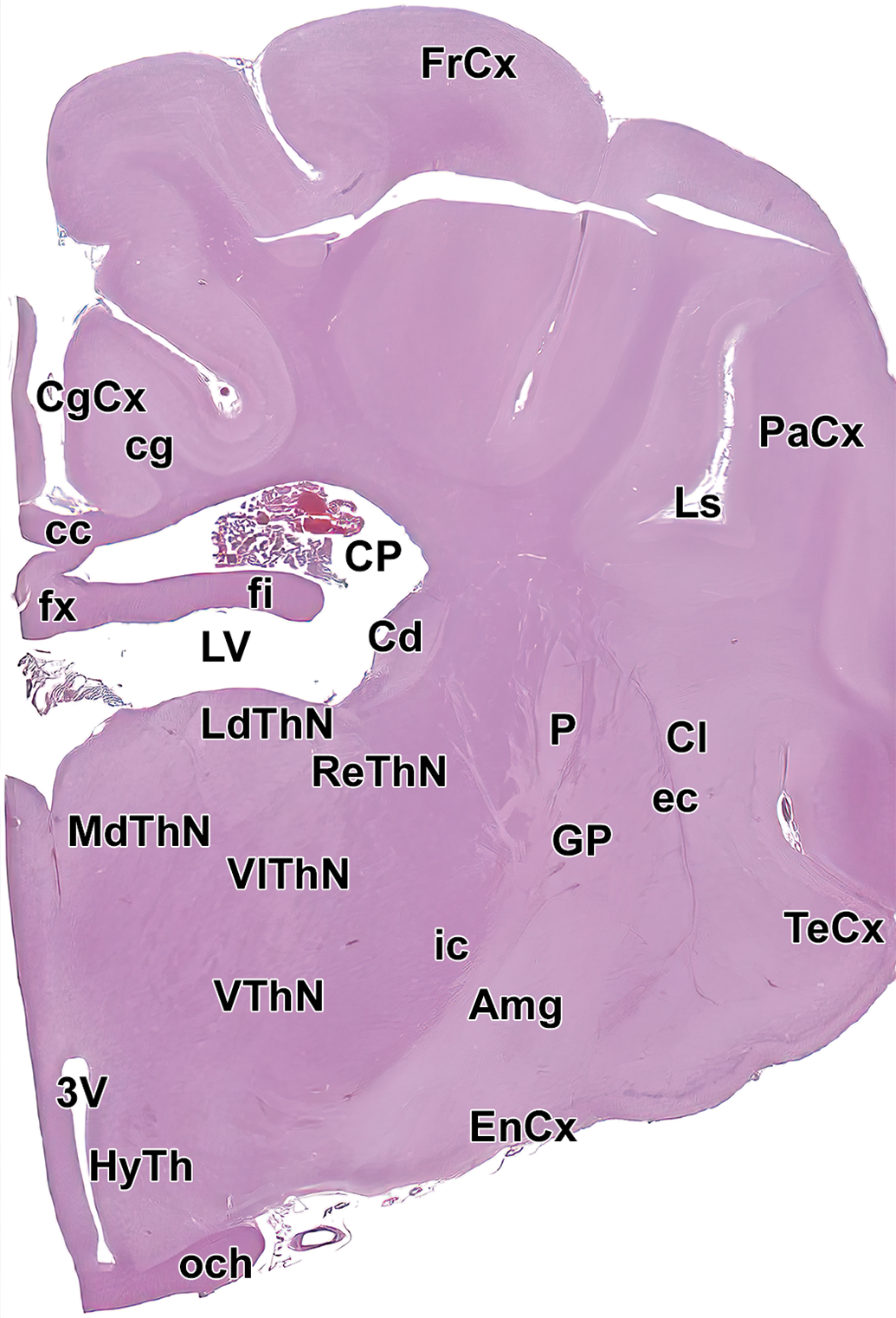
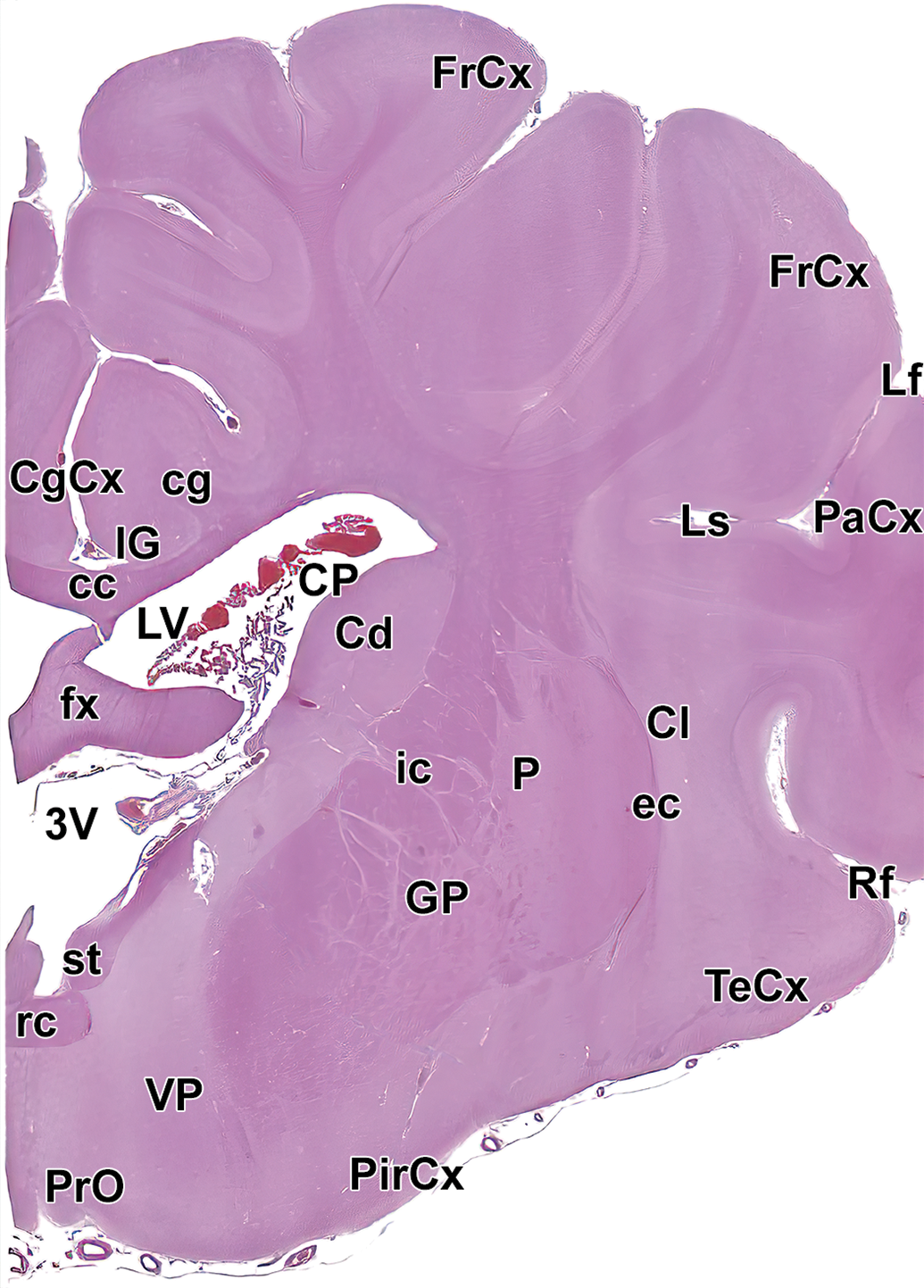
Level 8: Hypothalamus and rostral thalamus. Amg indicates amygdala; cc, corpus callosum; Cd, caudate nucleus; cg, cingulum; CgCx, cingulate cortex; Cl, claustrum; CP, choroid plexus; EnCx, entorhinal cortex; ec, external capsule; fi, fimbria; FrCx, frontal cortex; fx, fornix; GP, globus pallidus; HyTh, hypothalamus; ic, internal capsule; LdThN, lateral dorsal thalamic nucleus; Ls, lateral sulcus; LV, left ventricle; MdThN, medial dorsal thalamic nucleus; och, optic chiasm; P, putamen; PaCx, parietal cortex; ReThN, reticular thalamic nucleus; TeCx, temporal cortex; VlThN, ventral lateral thalamic nucleus; VThN, ventral thalamic nucleus; and 3V, third ventricle.
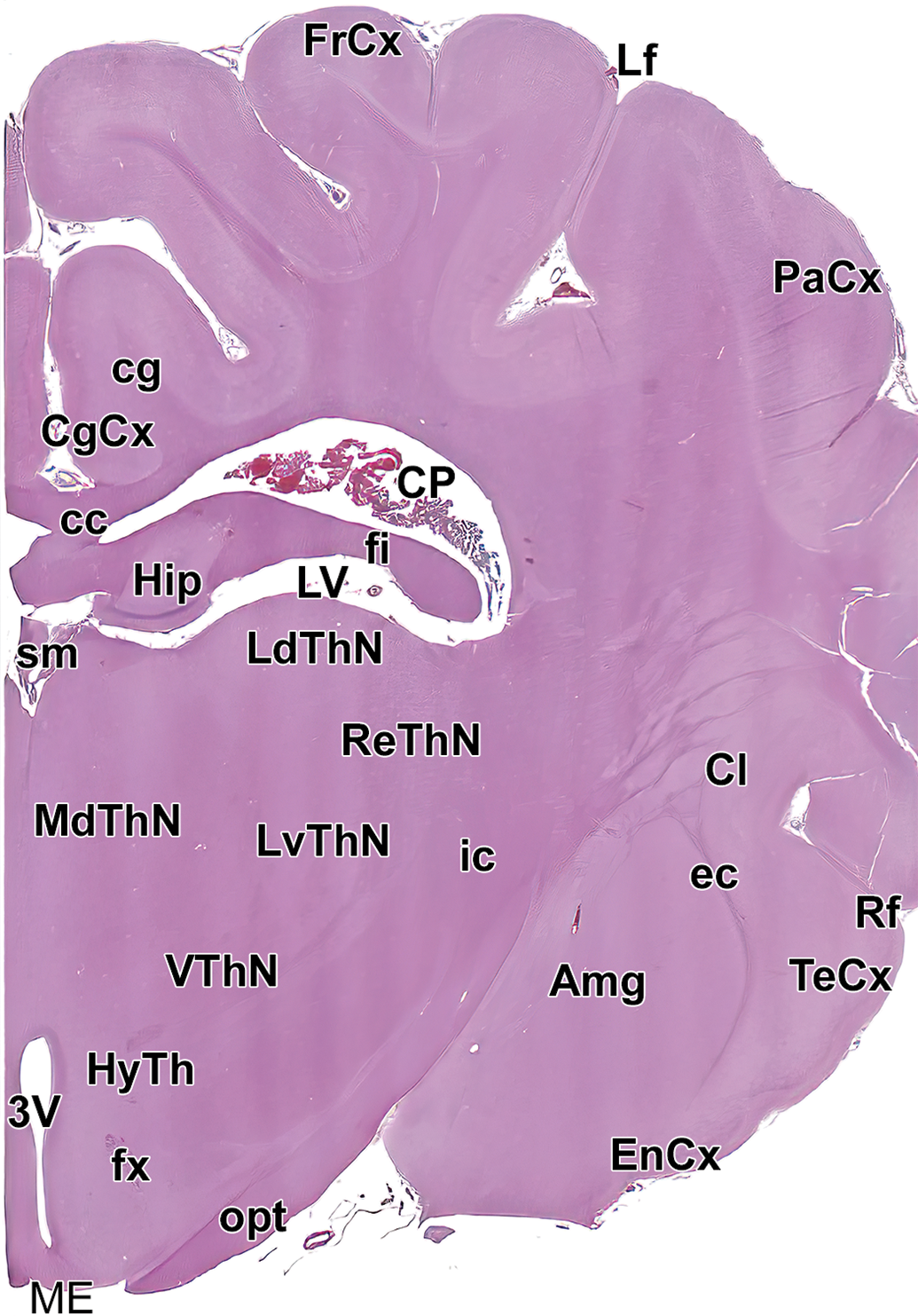
Level 9: Mid thalamus. Amg indicates amygdala; cc, corpus callosum; cg, cingulum; CgCx, cingulate cortex; Cl, claustrum; CP, choroid plexus; EnCx, entorhinal cortex; ec, external capsule; fi, fimbria; FrCx, frontal cortex; fx, fornix; Hip, hippocampus; HyTh, hypothalamus; ic, internal capsule; LdThN, lateral dorsal thalamic nucleus; Lf, lateral fissure; LV, left ventricle; LvThN, lateral ventral thalamic nucleus; MdThN, medial dorsal thalamic nucleus; ME, median eminence; opt, optic tract; PaCx, parietal cortex; ReThN, reticular thalamic nucleus; Rf, rhinal fissure; sm, stria medullaris of the thalamus; TeCx, temporal cortex; VThN, ventral thalamic nucleus; and 3V, third ventricle.
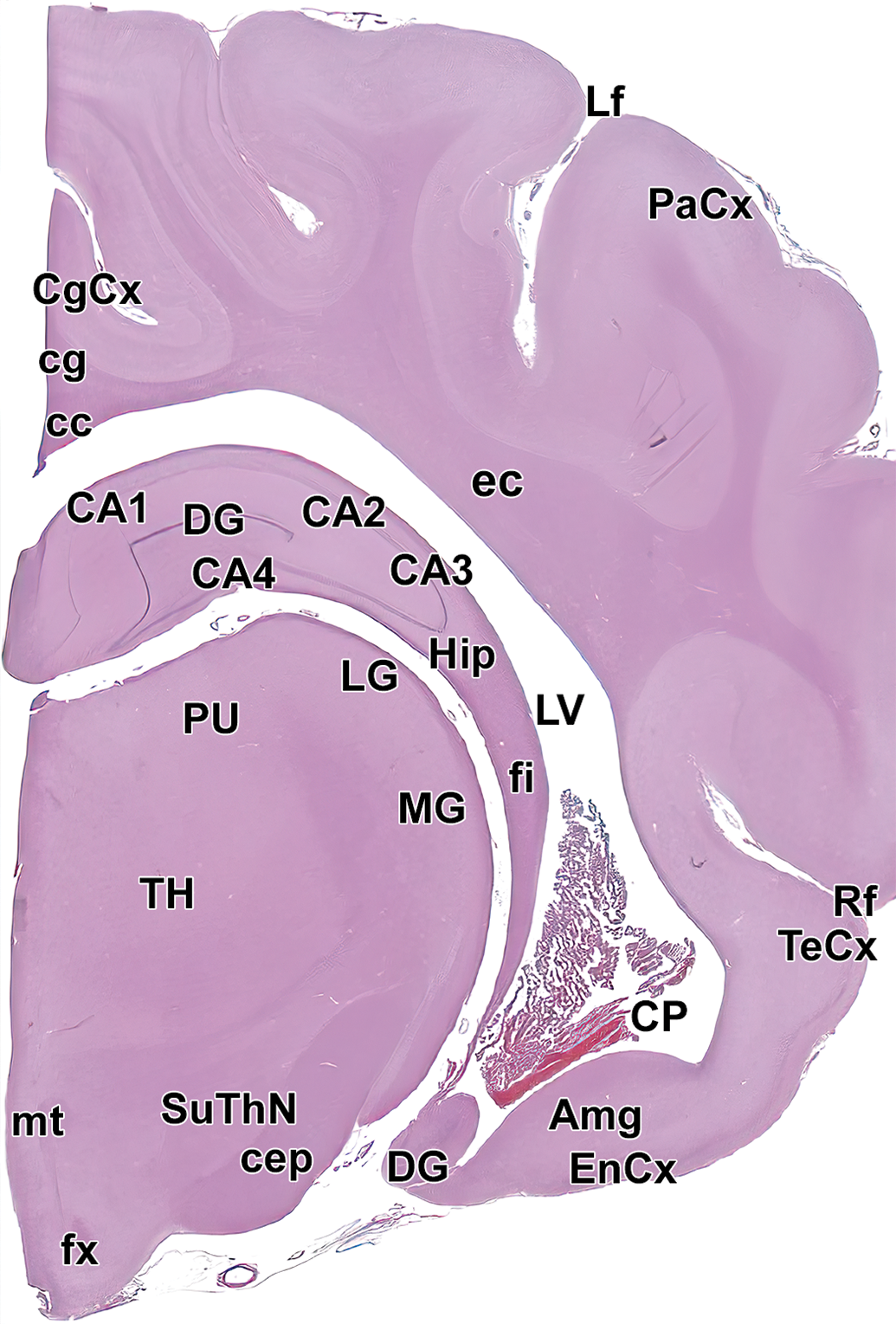
Level 10: Hippocampus and caudal thalamus I. Amg indicates amygdala; CA1-4, cornu Ammonis fields 1-4 of the hippocampus; cc, corpus callosum; cep, cerebral peduncle; cg, cingulum; CgCx, cingulate cortex; CP, choroid plexus; DG, dentate gyrus of hippocampus; ec, external capsule; EnCx, entorhinal cortex; fi, fimbria; fx, fornix; Hip, hippocampus; Lf, lateral fissure; LG, lateral geniculate nucleus; LV, lateral ventricle; MG, medial geniculate nucleus; mt, mammillothalamic tract; opt, optic tract; PaCx, parietal cortex; PU, pulvinar nucleus of the thalamus; Rf, rhinal fissure; SuThN, subthalamic nucleus; TeCx, temporal cortex; and TH, thalamus.
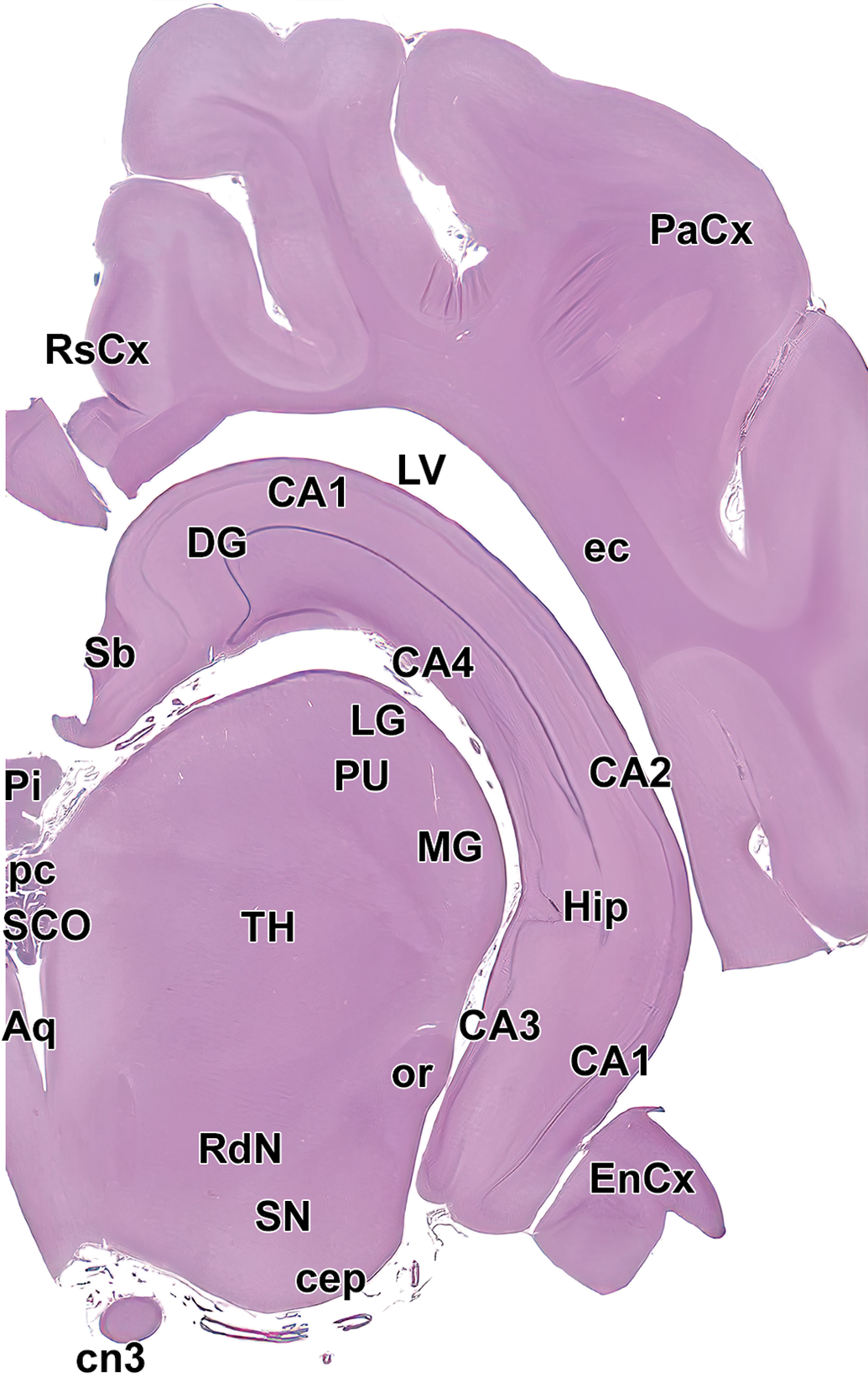
Level 11: Hippocampus and caudal thalamus II. Aq indicates mesencephalic aqueduct (of Sylvius); CA1-4, cornu Ammonis fields 1-4 of the hippocampus; cep, cerebral peduncle; cn3, oculomotor nerve (cranial nerve 3); DG, dentate gyrus of hippocampus; ec, external capsule; EnCx, entorhinal cortex; Hip, hippocampus; LG, lateral geniculate nucleus; LV, lateral ventricle; MG, medial geniculate nucleus; or, optic radiation; PaCx, parietal cortex; pc, posterior commissure; Pi, pineal gland; PU, pulvinar nucleus of the thalamus; RdN, red nucleus; RsCx, retrosplenial cortex; SCO, subcommissural organ; Sb, subiculum; SN, substantia nigra; and TH, thalamus.
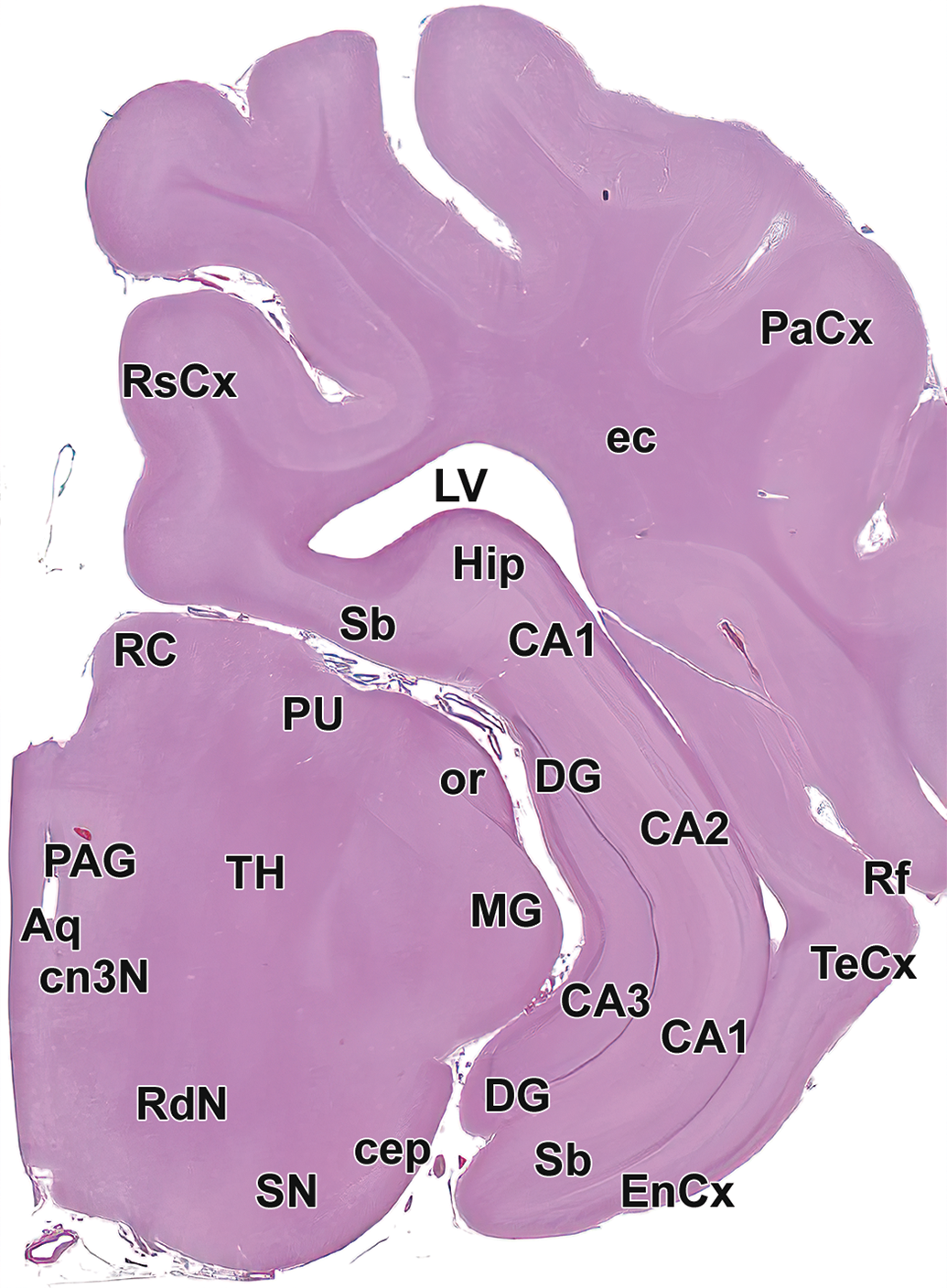
Level 12: Hippocampus and caudal thalamus III with rostral midbrain: Aq indicates mesencephalic aqueduct (of Sylvius); CA1-4, cornu Ammonis fields 1-4 of the hippocampus; cep, cerebral peduncle; cn3N, oculomotor nucleus; DG, dentate gyrus of hippocampus; ec, external capsule; EnCx, entorhinal cortex; Hip, hippocampus; LV, lateral ventricle; MG, medial geniculate nucleus; or, optic radiation; PaCx, parietal cortex; PAG, periaqueductal gray; PU, pulvinar nucleus of the thalamus; RC, rostral colliculus; RdN, red nucleus; Rf, rhinal fissure; RsCx, retrosplenial cortex; Sb, subiculum; SN, substantia nigra; TeCx, temporal cortex; and TH, thalamus.
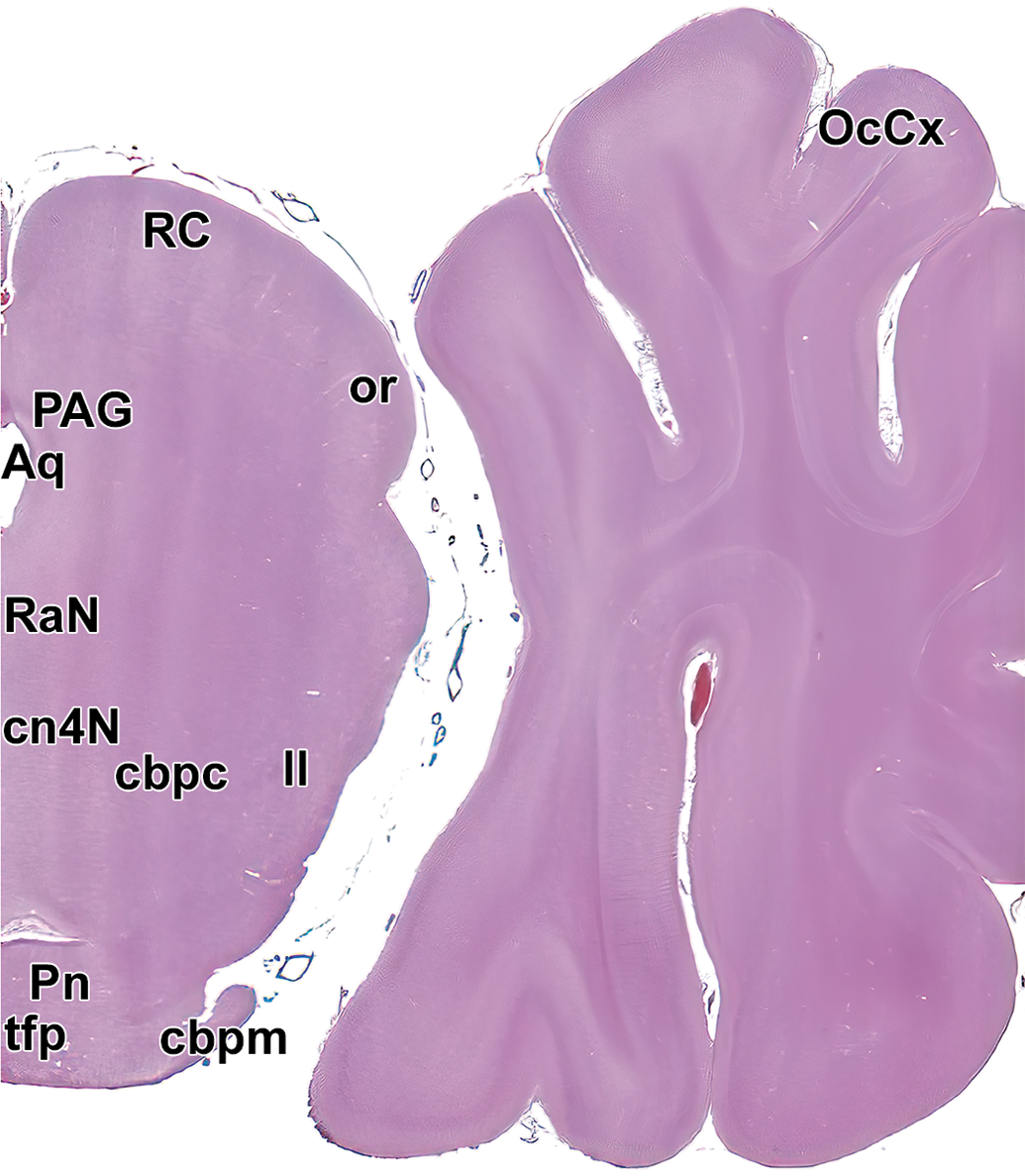
Level 13: Midbrain and occipital cortex I. Aq indicates mesencephalic aqueduct (of Sylvius); cbpc, cranial cerebellar peduncle; cn4N, trochlear nucleus; ll, lateral lemniscus; cbpm, middle cerebellar peduncle; OcCx, occipital cortex; or, optic radiation; PAG, periaqueductal gray; Pn, pontine nucleus; RaN, Raphe nuclei; RC, rostral colliculus; and tfp, transverse fibers of the pons.
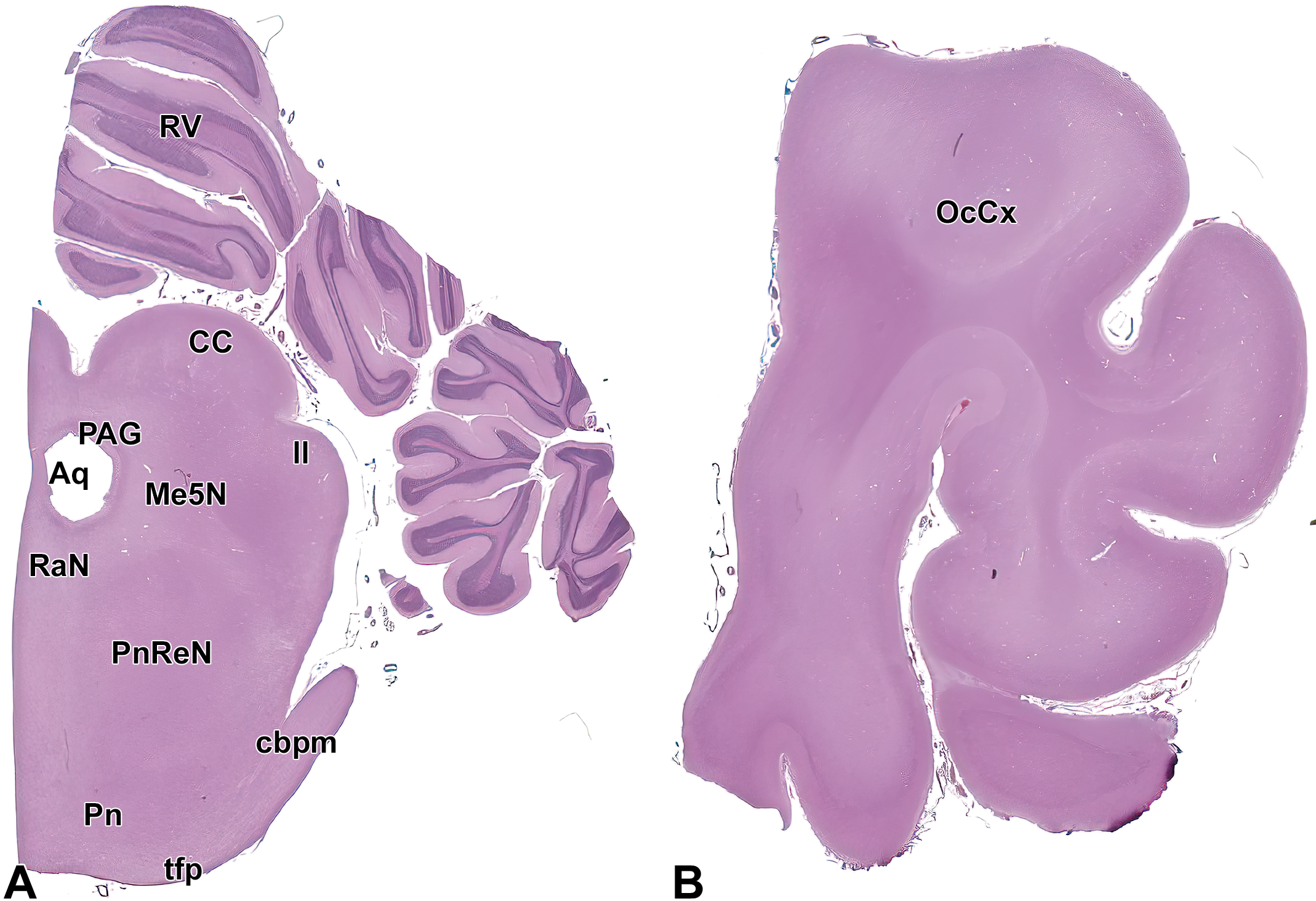
Level 14: Midbrain and occipital cortex II. (A) shows the caudal midbrain, pons, and cerebellar cortex while (B) demonstrates the occipital cortex. Aq indicates mesencephalic aqueduct (of Sylvius); CC, caudal colliculus; cbpm, middle cerebellar peduncle; ll, lateral lemniscus; Me5N, mesencephalic trigeminal nucleus; OcCx, occipital cortex; PAG, periaqueductal gray; Pn, pontine nucleus; PnReN, pontine reticular nucleus; RaN, raphe nuclei; RV, rostral vermis of cerebellum; and tfp, transverse fibers of the pons.
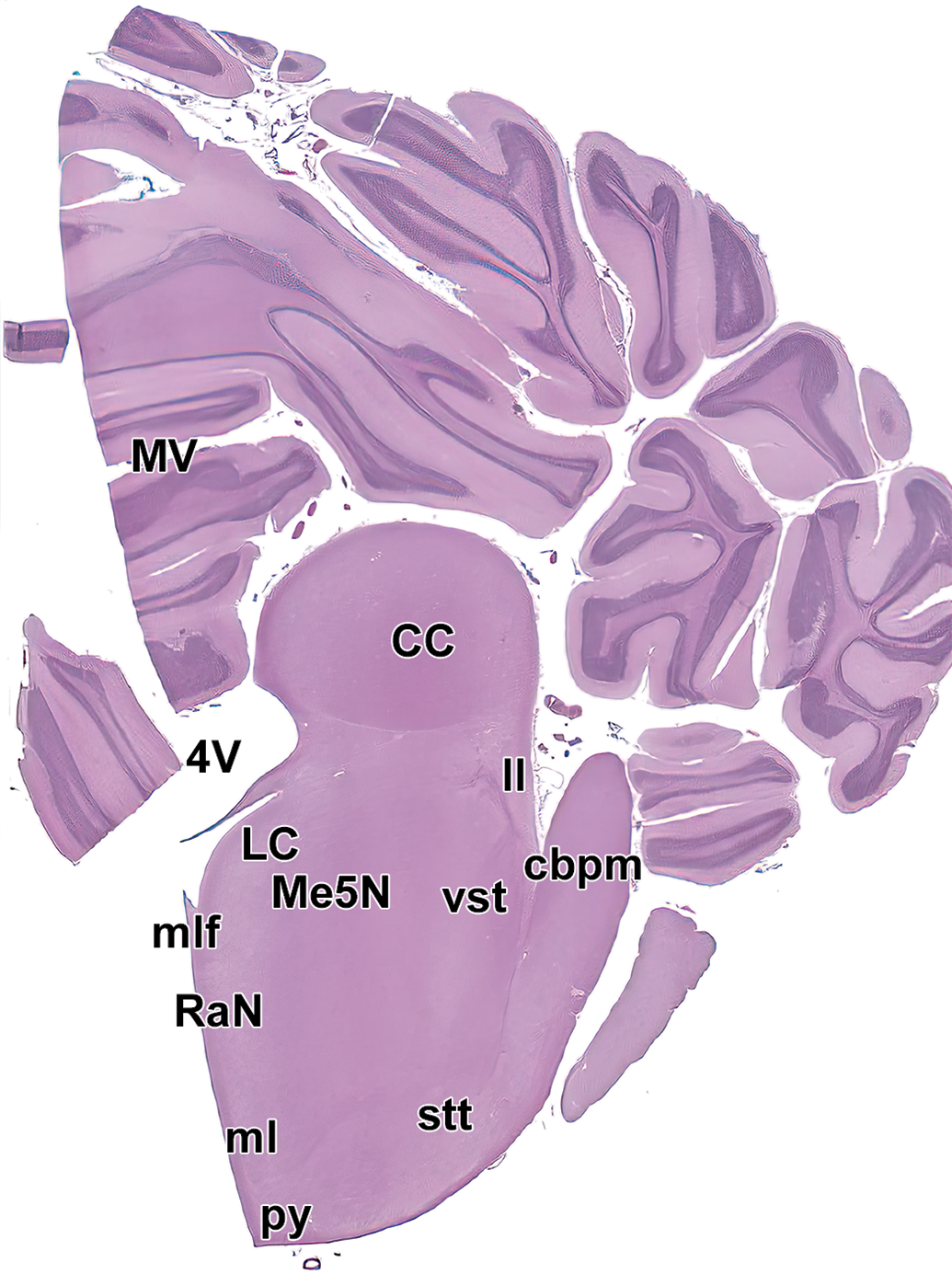
Level 15. Caudal colliculus (midbrain), midcerebellum, and pons. CC indicates caudal colliculus; cbpm, middle cerebellar peduncle; LC, locus coeruleus; ll, lateral lemniscus; Me5N, mesencephalic trigeminal nucleus; ml, medial lemniscus; mlf, medial longitudinal fasciculus; MV, middle vermis of cerebellum; py, pyramids; RaN, Raphe nuclei; stt, spinothalamic tract; vst, vestibulospinal tract; and 4V, fourth ventricle.
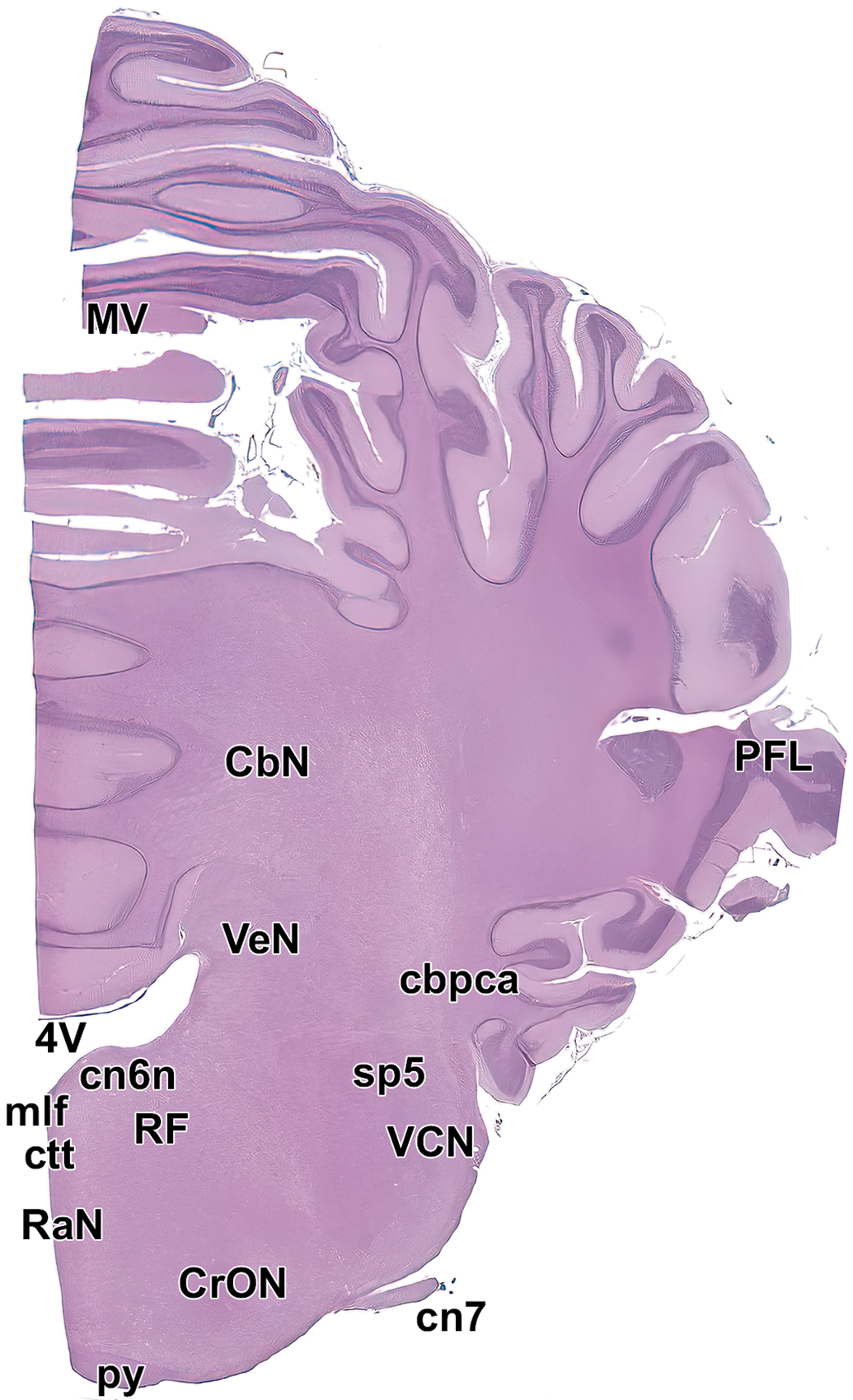
Level 16. Middle cerebellum and medulla oblongata. AbN indicates abducens nucleus; CbN, deep cerebellar nuclei; cbpca, cerebellar peduncle, caudal; cn7, facial nerve; CrON, rostral olivary nucleus; ctt, central tegmental tract; mlf, medial longitudinal fasciculus; MV, middle vermis; PFL, paraflocculus lobe of cerebellum; py, pyramids; RaN, raphe nuclei; RF, reticular formation; sp5, spinal trigeminal tract; VCN, ventral cochlear nucleus; VeN, vestibular nucleus; and 4V, fourth ventricle.
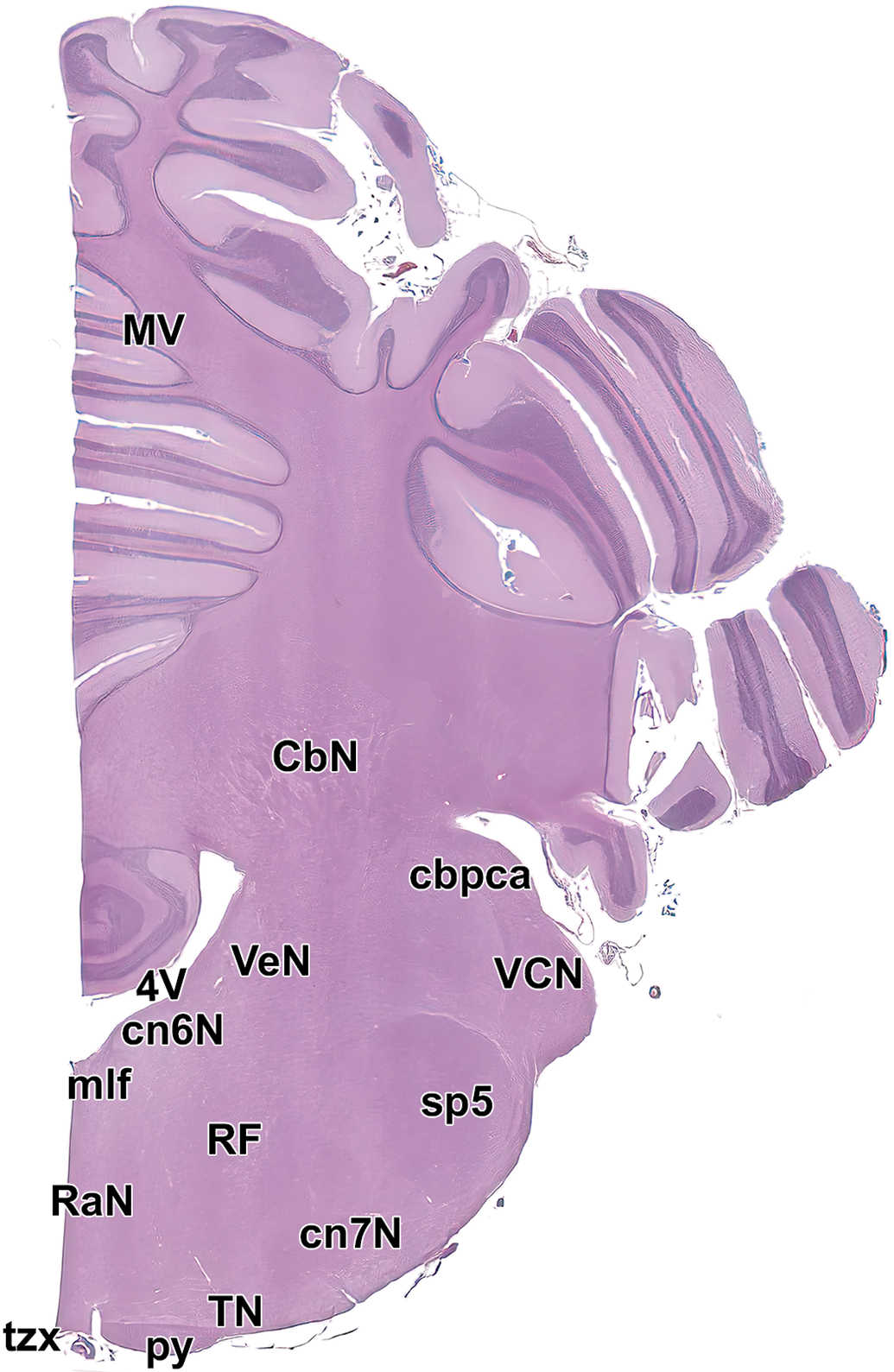
Level 17. Caudal cerebellum and medulla oblongata. CbN, deep cerebellar nuclei; cbpca, cerebellar peduncle, caudal; cn6N, abducens nucleus; cn7N, facial nucleus; mlf, medial longitudinal fasciculus; MV, middle vermis of cerebellum; py, pyramids; RaN, raphe nuclei; RF, reticular formation; sp5, spinal trigeminal tract; TN, trapezoid nucleus; tzx, decussation of the trapezoid body; VCN, ventral cochlear nucleus; VeN, vestibular nucleus; and 4V, fourth ventricle.
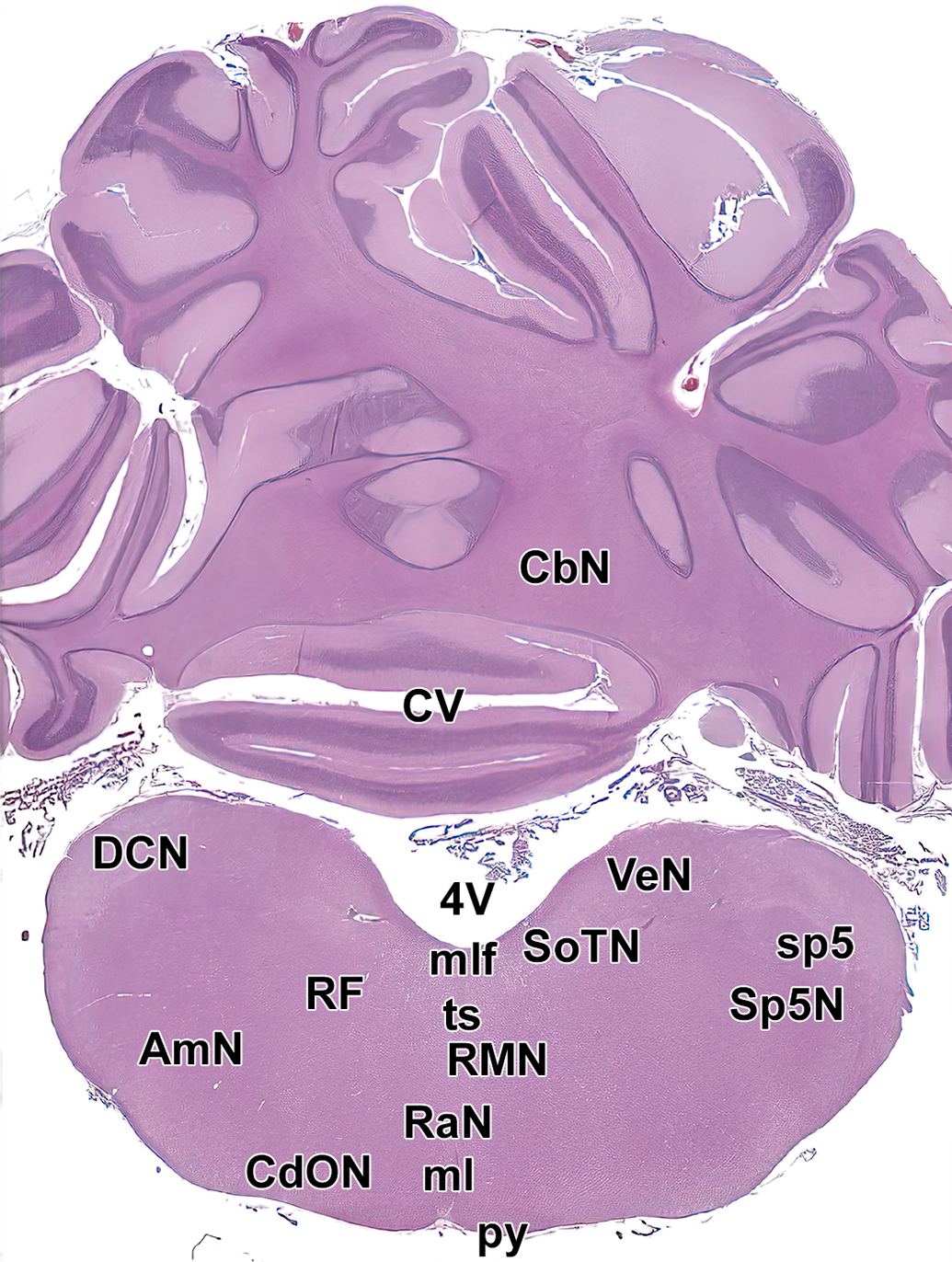
Level 18. Caudal cerebellum and medulla oblongata. AmN indicates ambiguous nucleus; CbN, deep cerebellar nuclei; CdON, caudal olivary nucleus; CV, caudal vermis of cerebellum; DCN, dorsal cochlear nucleus; ml, medial lemniscus; mlf, medial longitudinal fasciculus; py, pyramids; RaN, raphe nuclei; RF, reticular formation; RMN, raphe magnocellular nuclei; SoTN, solitary nucleus; sp5, spinal trigeminal tract; Sp5N, spinal trigeminal nucleus; ts, tectospinal tract; VeN, vestibular nucleus; and 4V, fourth ventricle.
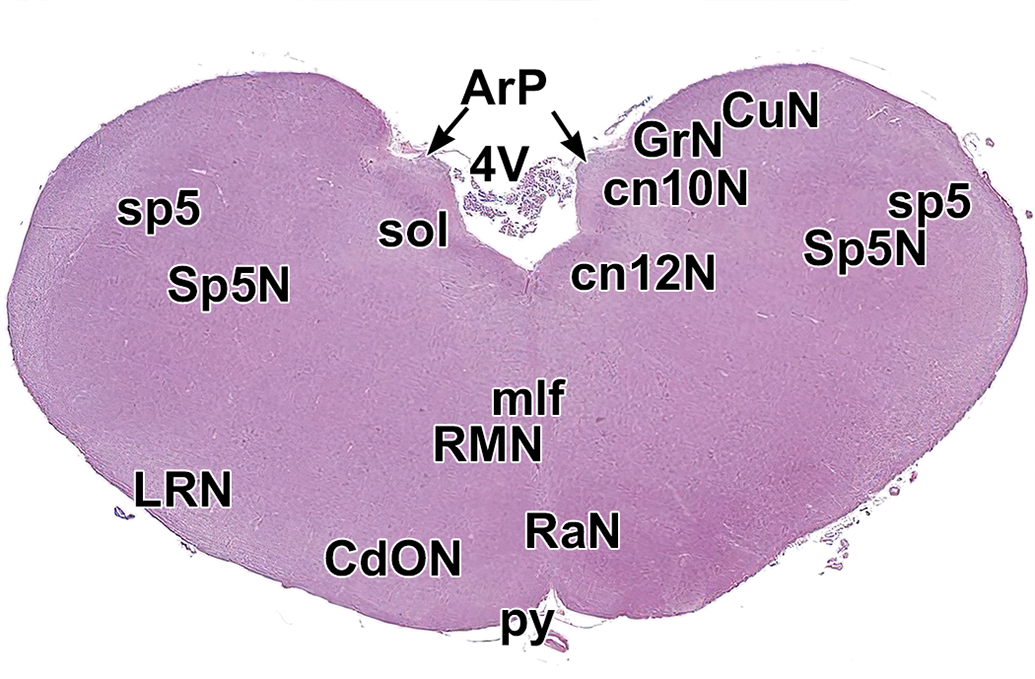
Level 19. Medulla oblongata. ArP indicates area postrema; CdON, caudal olivary nucleus; cn10N, vagus nerve nucleus (dorsal); cn12N, hypoglossal nucleus; CuN, cuneate nucleus; GrN, gracile nucleus; LRN, lateral reticular nucleus; mlf, medial longitudinal fasciculus; py, pyramids; RaN, raphe nucleus; RMN, raphe magnocellular nuclei; sol, solitary nucleus and tract; sp5, spinal trigeminal tract; Sp5N, spinal trigeminal nucleus; and 4V, fourth ventricle. (Irregular edges of the caudal vermis of the cerebellum for this level [present in Figure 4] were not included in this image due to tissue damage during processing.)
Major Landmarks Available for Evaluation in the 7-Section (Tier 1) Brain Trimming Scheme for General Toxicity Studies.
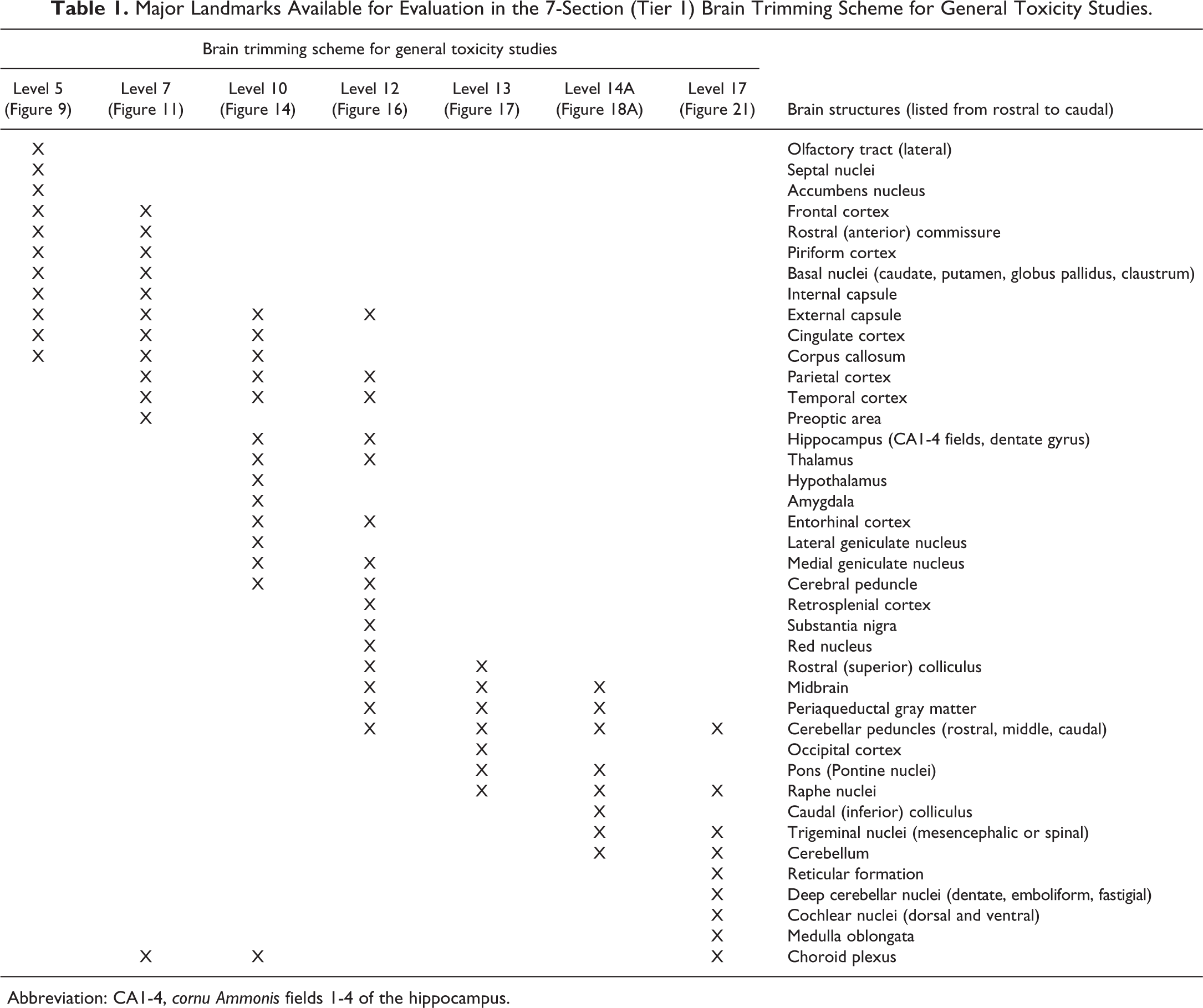
Abbreviation: CA1-4, cornu Ammonis fields 1-4 of the hippocampus.
Major Landmarks Available for Evaluation in the 16-Section (Tier 2) Brain Trimming Scheme** for Dedicated Neurotoxicity Studies.

Other Neural Organs: Spinal Cord, Ganglia, and Nerves
The STP “best practice” recommendations for collecting other nervous system tissues vary depending on the situation for spinal cord 68,71 and the PNS. 71 In general, neural structures outside the brain should be collected broadly and bilaterally. The reason for this approach is that a necropsy can only be performed once, so any tissues not obtained will be lost permanently.
Spinal cord
Three spinal cord divisions (cervical, thoracic, and lumbar) should be collected for both general toxicity and neurotoxicity studies (Figure 24). To promote adequate fixation, the spinal cord usually is harvested at necropsy by using medium-sized rongeurs to remove the very prominent dorsal and transverse vertebral processes and the vertebral arch, after which the exposed dura mater is grasped with forceps to elevate and remove the cord without touching the fragile neural tissue. Some pathologists may choose to extract short segments of vertebral column (spanning 1-2 vertebral bodies) and then immerse the entire block in fixative, but this approach may lead to delayed fixation of the bone-encased spinal cord at the center of the segment. Compared to humans, minipigs have narrow intervertebral spaces, 3 which may complicate removal of short lengths of vertebral column without traumatizing the nearby spinal cord.
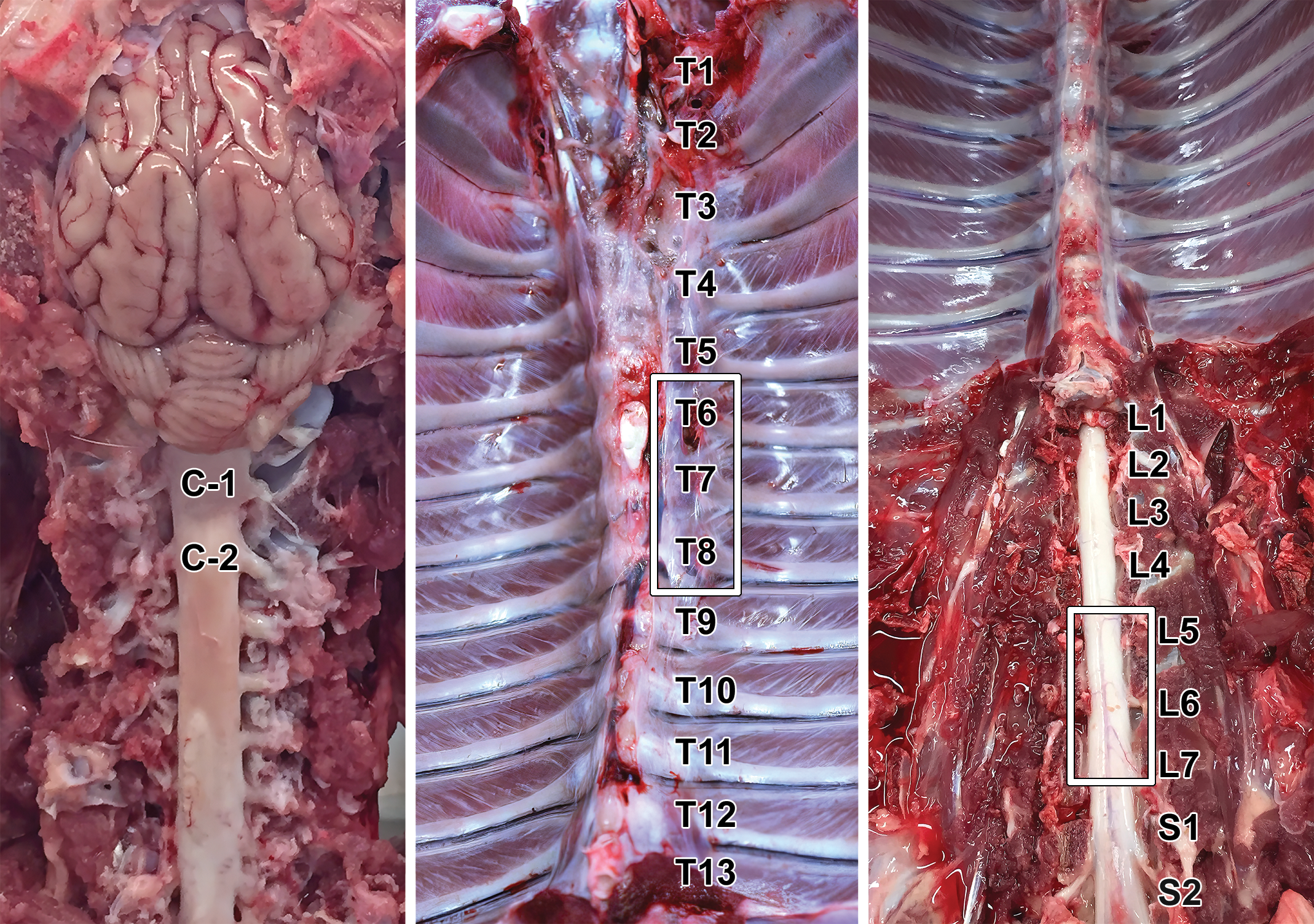
External landmarks used to identify spinal cord segments. Samples commonly are collected from the upper cervical (C1-C2), mid (T6-T8) to caudal (T9-T12) thoracic, and lumbar intumescence (L5-S1) regions for neuropathology assessment during general toxicity studies.
A major consideration for sampling the lumbar spinal cord in swine is that the numbered cord segments correspond approximately to the vertebrae with the same number. The lumbar intumescence, which resides in spinal cord segments lumbar L6 (and sometimes L5) to sacral S1, is located within vertebra L6 or sometimes L5 52,53 ; the cranial portion (L4-L6) of this spinal cord enlargement holds neurons that supply axons to the femoral nerve, while the caudal portion (S1-S3) houses neurons that travel in the sciatic nerve. 72 The cauda equina starts at the level of sacral vertebrae S2-S3. 2 These facts are important as the lumbar intumescence is less prominent in pigs than in other species, 52 and its location within the spinal canal is caudal to that of other principal nonclinical test species (inside vertebrae L1-2 in rodents and nonhuman primates or L3-4 in dogs 68 ) except rabbits (where the usual location is vertebrae L6-S1). 73
Spinal cord segments should be assessed both macroscopically and microscopically in gross (transverse) and longitudinal planes (Figure 25A). In general, samples from all 3 spinal cord divisions should be processed for histopathologic evaluation since some nuclei and fiber tracts are absent in some spinal cord divisions. The authors have found that specimens taken between cervical segments C1-C3 (Figure 25B), thoracic segments T9-T12, and lumbosacral segments L5-S1 provide acceptable views of key spinal cord domains; in general, the cross section should be taken through the rostral third of the spinal cord sample for each division, while the longitudinal plane should cross the caudal two-thirds of the sample (Figure 25A). The authors prefer the longitudinal oblique trimming plane for the microscopic evaluation because it permits histopathologic analysis of more spinal cord white matter and gray matter than is possible in some other longitudinal orientations, 68,74 which greatly facilitates detection of nerve fiber degeneration. Two other longitudinal orientations are possible, 74 but in the authors’ experience these trimming planes yield sections with more anatomic variation in both white matter and gray matter structures.
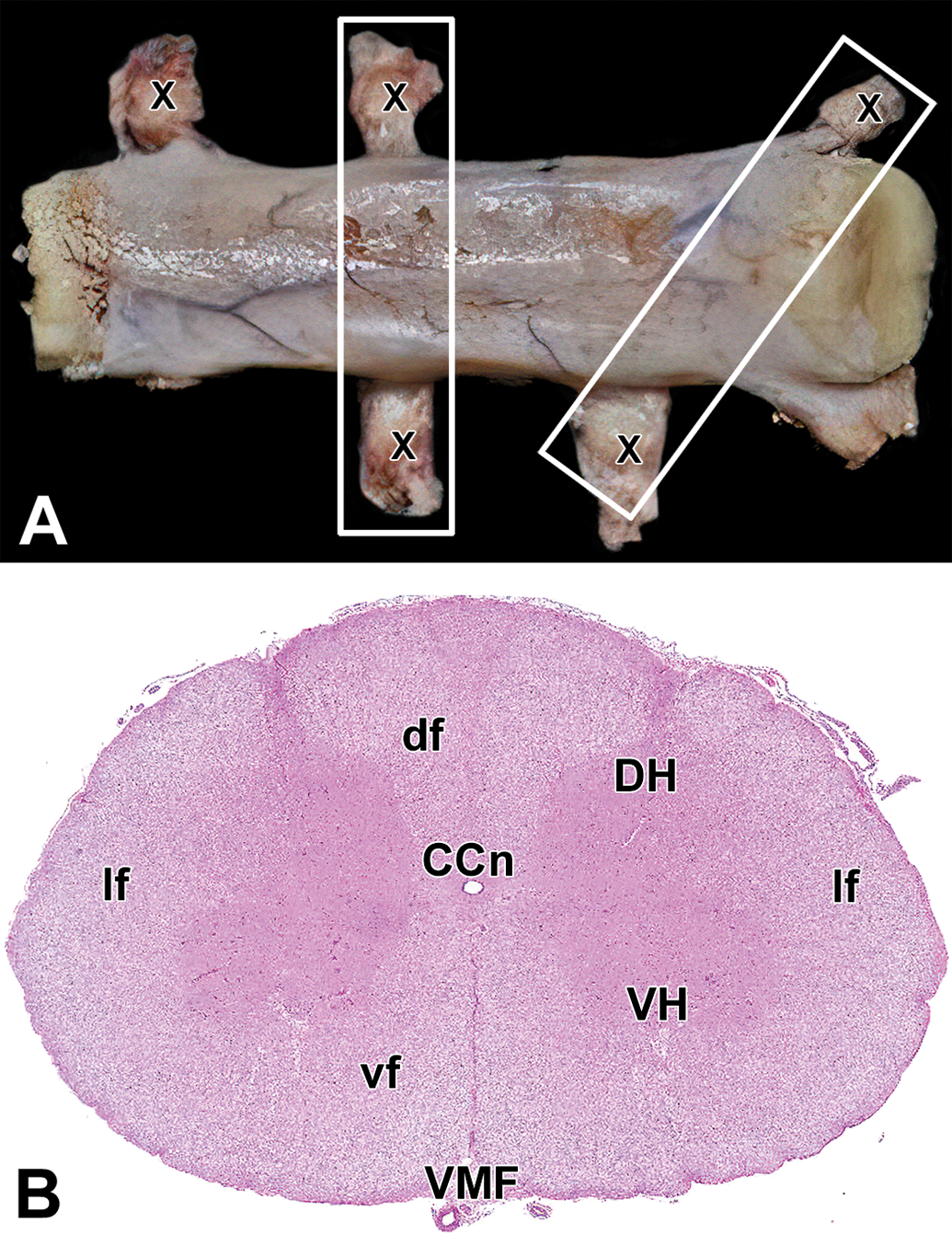
Spinal cord sampling and key internal landmarks. A, Slices of spinal cord should be harvested in both transverse (vertical box) and longitudinal oblique (tilted box) orientations. The Xs depict dorsal root ganglia (DRG). B, Cervical spinal cord (transverse section). Gray matter: DH indicates dorsal horn; VH, ventral horn. White matter: df, dorsal funiculi; lf, lateral funiculi; vf, ventral funiculi. Other features: CCn, central canal; VMF, ventral median fissure.
Ganglia
Sampling of ganglia in the PNS depends on the study objective. For most toxicity studies, only somatic ganglia are obtained purposely, while autonomic ganglia are assessed if they are present within or near protocol-specified non-neural tissues (eg, the walls of hollow organs). In this regard, typical somatic ganglia collected at necropsy are the dorsal root ganglia (DRG) and sometimes the trigeminal ganglia (cranial nerve V). 71,75 These somatic sensory ganglia usually are evaluated if there is a possibility that a test article might target their neurons. 76,77 Autonomic ganglia (usually for the sympathetic division) typically are collected only if the test article or a related material has been shown to produce effects to autonomic function. 76 –79
For general toxicity studies, at least 1 lumbar DRG should be collected and preserved in fixative to be processed later (if needed) for light microscopic examination. 71 Common collection sites for DRG in minipigs include those associated with the main nerve supplies to the forelimbs (C6-T1) and hind limbs (L5-S2) 71,72 ; in practical terms DRG sampling often occurs at locations geographically near the processed spinal cord segments (C1-C3; T9-T12; L5-S1). These DRGs should be procured, even if no neurological signs are seen during life. In the absence of neurological signs, DRG may be left in situ (ie, in their intervertebral locations) for fixation and archiving, and then removed and processed only if a decision is made to evaluate their microscopic structure. If neurological signs are observed during life, a minimum of 2 DRGs each should be harvested for the cervical, thoracic, and lumbar divisions. Collection of additional DRGs (at least 1 from each spinal cord division) is recommended for toxicity studies if the test article is a member of a therapeutic class (eg, antineoplastic agents, 80 some viral gene therapy vectors 81 ) known to target DRG neurons or if the test article is delivered directly into the neuroaxis (eg, via intracerebroventricular, intracisternal, or intrathecal injection or infusion).
For dedicated neurotoxicity studies, 2 (or ideally more) DRGs per spinal cord division as well as both trigeminal ganglia should be sampled, and autonomic ganglia should be considered for collection. 71 The DRGs are collected as noted above. Trigeminal ganglia and autonomic ganglia (eg, cranial [superior] cervical, cervicothoracic, and cranial mesenteric) may be gathered using published procedures. 75,82 Locations of major autonomic ganglia in minipigs could not be illustrated for the present satellite study because the torso was used to address the objective of the primary study, but the ganglionic positions in minipigs 83,84 are similar generally to those of equivalent ganglia in other nonrodent species. 71
Nerves
Collection of nerves from minipigs also depends on the study objective. For most toxicity studies, only somatic nerves are harvested unless autonomic (parasympathetic or sympathetic) signs are anticipated or seen, in which situations autonomic nerves may be obtained as well. Multiple large somatic nerve trunks offering ample tissue for microscopic evaluation may be accessed readily in minipig limbs (Figure 26).
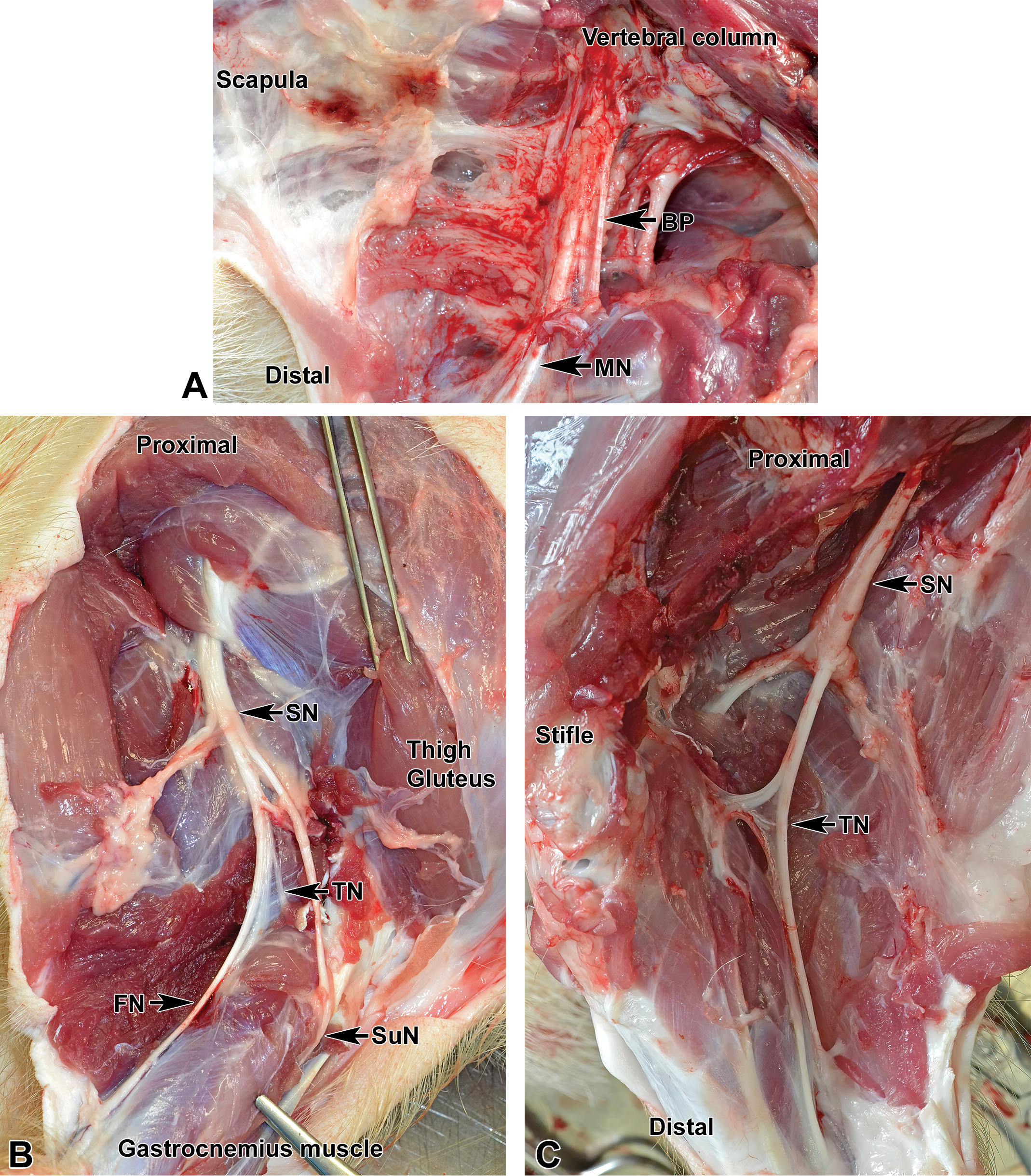
Locations of major somatic nerves. A, Medial aspect of the proximal foreleg. BP indicates brachial plexus; MN, median nerve. B and C, Lateral aspect of the proximal hind leg. FN indicates femoral nerve; SN, sciatic nerve; SuN, sural nerve; TN, tibial nerve; FN, fibular (peroneal) nerve.
For general toxicity studies, many institutions sample the sciatic nerve and one or more of its branches (eg, tibial and fibular nerves) but process only sciatic nerve for microscopic evaluation if no in-life neurological signs were observed. In general, nerves should be collected bilaterally to provide a reserve sample in the archived tissue but are evaluated unilaterally as neurotoxic test articles typically do not induce unilateral effects. 71
For neurotoxicity studies, several mixed sensorimotor nerves (generally the sciatic, tibial, and fibular at minimum) are sampled, processed, and evaluated. 71 In such studies, many institutions also collect at least 1 mixed sensorimotor nerve that serves the forelimb (eg, median [Figure 26A], radial, and/or ulnar) and/or a sensory-predominant nerve (eg, plantar 71 ).
Discussion
Our present article provides practical trimming schemes to ensure systematic sampling of the minipig nervous system during nonclinical toxicity testing that will be consistent with existing regulatory guidance, 46,47 globally harmonized testing expectations, 48,49 and industrial “best practice” recommendations 68,71 for evaluation of the CNS and PNS. For general toxicity studies, our 7-level (Tier 1) brain trimming protocol (7 coronal hemisections on 6-7 slides [Figure 4A]) follows STP “best practice” recommendations for brain trimming in nonrodent species. 68 For dedicated neurotoxicity studies, a more extensive 16-level (Tier 2) brain trimming protocol (16 coronal sections on 16 slides [Figure 4B]) allows processing of the entire brain as 14 hemisections rostrally and 2 full sections (for the last 2 levels containing caudal cerebellum and medulla oblongata). Acquisition of spinal cord, ganglia (somatic and autonomic), and nerves should follow published species-agnostic recommendations for these organs. 68,71 The CNS and PNS sampling schemes for Tier 1 and Tier 2 described in the current article are equivalent to but modestly expanded relative to the type 1 and type 2 schemes defined in an exquisitely detailed necropsy guide for pig biomedical models 54 ; this expansion is designed to provide further assurance to regulatory authorities that functionally and structurally complex neural domains have been sampled and evaluated adequately during nonclinical safety studies. The current annotated atlas also covers more brain levels (19 planes) compared to the annotated diagrams in the necropsy guide (10 planes).
The Tier 1 brain sampling strategy for general toxicity studies using nonrodents generally should not be reduced. The reason to avoid adjustments is that major neuroanatomic structures (delineated in Table 1 and Supplemental Table 1) need to be evaluated at least unilaterally, and our 7-level Tier 1 protocol provides the minimum means for performing this assessment in minipigs. The more extensive 16-level Tier 2 brain trimming protocol for neurotoxicity studies may be decreased to some extent if prior knowledge has demonstrated to the institution’s satisfaction that omission of one or a few levels will not impact hazard identification and characterization for a novel test article. Brain atlases for swine may be used to develop more focused trimming schemes to access particular brain regions for neurotoxicity studies. 23,50,51
Evaluation of the minipig brain for nonclinical safety testing and data translation with respect to other species requires some consideration. First, extra care must be taken in removing the pig brain to avoid traumatizing the delicate brain parenchyma with dissecting instruments. This care is necessary since the skull is quite thick (relative to the animal’s size) and certain structures are buried in various recesses within the skull. For example, the large olfactory bulbs are embedded in the rostroventral skull caudal to the cribriform plate; when desired, they can be collected with reasonable ease by fracturing the frontal and dorsal portions of the ethmoid bones. Similarly, the pituitary gland is enclosed within the sella turcica at the base of the skull and will remain in its original position in the skull after removal of the rest of the brain. Second, given the large size of the olfactory bulbs in swine, an important aspect of the study design is to determine whether or not these structures will be included when collecting brain weights. Olfactory bulbs account for 3% to 4% of brain mass in pigs, 85 so variable inclusion of olfactory bulbs likely will skew group mean brain weights given the small numbers of animals per group in routine nonrodent toxicity studies. Finally, the brain of minipigs is relatively large (relative to the animal’s size) and possesses prominent gyri and sulci (gyrencephalic) similar to other nonrodent test species (dogs, nonhuman primates) and humans. These neuroanatomic features coupled with the natural intelligence and the growing availability of neurological testing methods 86 –88 for this species have made the minipig an attractive animal model for studying human neurologic diseases. 22
Conclusion
The 2 trimming protocols detailed in this article describe systematic approaches for reliable and reproducible sampling of the minipig brain during nonclinical toxicity studies. The 7-level trimming scheme for general toxicity studies follows the STP “best practice” recommendations for brain sampling in nonrodent species during general toxicity studies. 68 Since this 7-level protocol is based on evaluation of hemisections rather than full coronal sections, it generally should not be reduced further. The 16-level trimming scheme for neurotoxicity studies acknowledges that increased brain sampling usually will be needed in cases where a test article is predicted or known to be neurotoxic. 60,68 –70 The authors believe that the descriptions and images provided in this article will allow toxicologic pathologists and toxicologists, technical personnel engaged in tissue sampling and processing, and scientists using minipigs in research to design more efficient and effective experiments with respect to macroscopic and microscopic evaluation of the nervous system.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211019941 - Nervous System Sampling for General Toxicity and Neurotoxicity Studies in the Laboratory Minipig With Emphasis on the Göttingen Minipig
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211019941 for Nervous System Sampling for General Toxicity and Neurotoxicity Studies in the Laboratory Minipig With Emphasis on the Göttingen Minipig by Ingrid D. Pardo, Rosa A. Manno, Raffaella Capobianco, Aaron M. Sargeant, James P. Morrison, Brad Bolon and Robert H. Garman in Toxicologic Pathology
Footnotes
Acknowledgments
Dr Ingrid D. Pardo would like to thank Mrs Hayley Newton Ritenour, Mrs Casey Ritenour, and Mrs Samantha Lee Levinson in the histopathology laboratory at Pfizer, Inc in Groton, Connecticut, for their help processing the tissues for light microscopic examination. Ms Andrea A. Koch of the Charles River Laboratories facility in Mattawan, Michigan, is also acknowledged for assistance with images. Dr Rosa A. Manno would like to thank Dr Andrea Grassetti (pathologist) and Enzo Insmenghi, Lucrezia Fabriani, and Alessia Pacelli, technicians in the Pathology Laboratories at ERBC, Pomezia, Italy, for their help in the neuroanatomical sampling and histological process. The authors also commend the efforts of Mr Walt Bobrowski and Ms Beth Mahler in optimizing the images.
Declaration of Conflicting Interests
The author(s) declared no potential, real or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
