Abstract
Idiopathic femoral head chondrolysis is a recognized condition in human adolescents and has previously been reported in two macaques at a biomedical research facility. A cluster of coxofemoral joint abnormalities consistent with this condition affecting seven cynomolgus macaques over a four-month period in 2016 and 2017 was observed at a single, nonclinical contract research facility, prompting enhanced physical examination screening efforts during animal receipt and pre-study evaluation to identify additional affected animals. This article summarizes the results of this investigation from November 2016 to March 2021, yielding 97 total cases for an overall incidence of 0.54% (97/17,898 macaques). Affected animals were presented with one or more of the following unilateral or bilateral findings on physical examination and/or diagnostic imaging: lameness, palpable coxofemoral joint abnormalities, femoral head atrophy with variable loss of articular cartilage and irregularity of the femoral head surface, enlarged joint space with effusion, and increased radiographic density of the femoral head. This condition prevented use of affected animals on study for 54% of the cases (52/97 animals). Recognition of this idiopathic condition is important in drug safety evaluation studies to distinguish it from test article–related effects.
Introduction
Musculoskeletal abnormalities in laboratory nonhuman primates may develop for a variety of reasons including traumatic injury, metabolic or neurologic disorders, xenobiotic/test article–related toxicity, or underlying genetic predispositions. 1 Identification and classification of the cause of these abnormalities are important in toxicologic pathology to distinguish test article–related effects from those that are independent of test article administration.
We identified an increased incidence of coxofemoral joint abnormalities that presented similarly to idiopathic femoral head chondrolysis in macaques over a 4-mo span in 2016 and 2017 at a nonclinical contract research facility. 2 Following this cluster of cases, enhanced musculoskeletal screening procedures were implemented in early 2017 for all primates upon receipt and prior to study placement. Our objectives were to investigate and report any patterns in animal source, signalment, physical examinations, radiography and other advanced imaging, necropsy, and histology for the affected animals with this condition.
Materials and Methods
A total of 17,898 macaques underwent enhanced physical examinations to identify coxofemoral joint abnormalities during intake and pre-test evaluations at a single, nonclinical contract research facility beginning in November 2016 and continuing over a period of 5 yr (2016-2021). As part of these initial physical exams, the following methods were implemented to increase detection of coxofemoral abnormalities: extension of the hind limbs to evaluate the upper legs for muscle mass symmetry, manipulation of the coxofemoral joints with abduction/rotation of the hind limbs for evaluation of the range of motion and flexion/extension, and palpation for abnormalities. Abnormal findings on physical examination were often subtle and included upper hind limb muscle mass asymmetry, coxofemoral joint crepitus, and limited range of motion and/or extension ability of the hind limb. Radiographic examination (ventrodorsal and frog-leg views of the coxofemoral joints) was performed on all animals with physical examination abnormalities. Initially, screening radiographs were required for all macaques prior to placement on studies of 3-mo duration or longer; however, screening radiographs are currently restricted to macaques intended for studies of at least 6-mo duration because of gained clinical experience and low risk of rapid deterioration in mild cases. During these screening radiographic exams, macaques with coxofemoral abnormalities are subjected to additional physical examination of the hind limbs under sedation and are evaluated for the potential need for pain management.
Selected macaques with a spectrum of coxofemoral joint abnormalities (unilateral and bilateral, mild to severe, and early to late detected disease) underwent additional diagnostic tests including magnetic resonance imaging (3 animals), fluoroscopic angiography (1 animal), joint fluid evaluation (9 animals), and/or necropsy with histopathology (30 animals). Tissues for histopathology were collected and fixed in 10% neutral buffered formalin, decalcified with hydrochloric or formic acid, and embedded in paraffin wax prior to sectioning to a 4- to 6-μm thickness and stained with H&E. All cytologic and histopathologic evaluations were performed by diplomats of the American College of Veterinary Pathologists (MM, SB, and/or DP).
Affected macaques were managed on a case-by-case basis by considering the severity of the radiographic changes, the degree of limb impairment when present, and the design and schedule of the intended study. Animals with mild disease were managed clinically with handling restrictions and assessment for need of pain management, while animals with moderate to severe disease were humanely euthanized or assigned to short-term studies.
This study was approved by the test facility’s Institutional Animal Care and Use Committee.
Results
There were 97 total cases with an overall incidence of 0.54% (97/17,898 macaques). Of the 97 affected animals, 54 were male (55.7%) and 43 were female (44.3%). Forty-eight animals were of Chinese origin (49.5%), 46 were Cambodian (47.4%), 2 were Rhesus (2.1%), and 1 was Mauritian (1%). During the initial cluster of cases and the following year, farm of origin for the affected cases was tracked and no correlation could be found that indicated a genetic or familial disposition. The range of age at detection was 17 to 138 mo (1.5-11.5 yr) with a mean age at detection of 38 mo (3.2 yr) and median age at detection of 34 mo (2.8 yr). The range of time from animal arrival at the facility to date of disease detection was 4 to 498 days with a mean time to detection of 83 days and median time to detection of 48 days. For 54% of the cases (52/97 animals), clinical signs were significant enough to warrant early termination and prevent use of these animals on study.
Of the 97 affected animals, 90 animals had unilateral disease (92.8%) and 7 animals had bilateral disease (7.2%). For unilaterally affected animals, the disease process was localized to the left limb in 57 animals (63.3%) and to the right limb in 33 animals (36.6%). Twenty-seven animals had visible limb impairment at the time of disease detection and 68 had subclinical disease that was identified upon further physical examination and clinical testing; for the remaining 2 animals, limb impairment status at the time of disease detection was not recorded for posterity. Affected animals were presented with one or more of the following findings: lameness, palpable coxofemoral joint abnormalities, femoral head atrophy with variable loss of articular cartilage and irregularity of the femoral head surface, enlarged joint space with inflammatory joint effusion, and increased radiographic density of the femoral head.
Cytology of coxofemoral joint fluid was performed for 9 cases representing a variety of signalments and disease severities. For all cases, there were increased numbers of osteoblasts and osteoclasts and predominately histiocytic inflammation, which was considered to be consistent with reactive bone proliferation and tissue remodeling.
Radiographic findings included increased density of the small irregular femoral head and narrowed femoral neck with increased density, irregularity of the acetabulum, and enlarged joint space. Magnetic resonance imaging was additionally performed on 3 cases and T2 STIR (short tau inversion recovery) images showed a hyperintense signal within the coxofemoral joint, indicating excessive fluid and joint effusion (Figure 1).
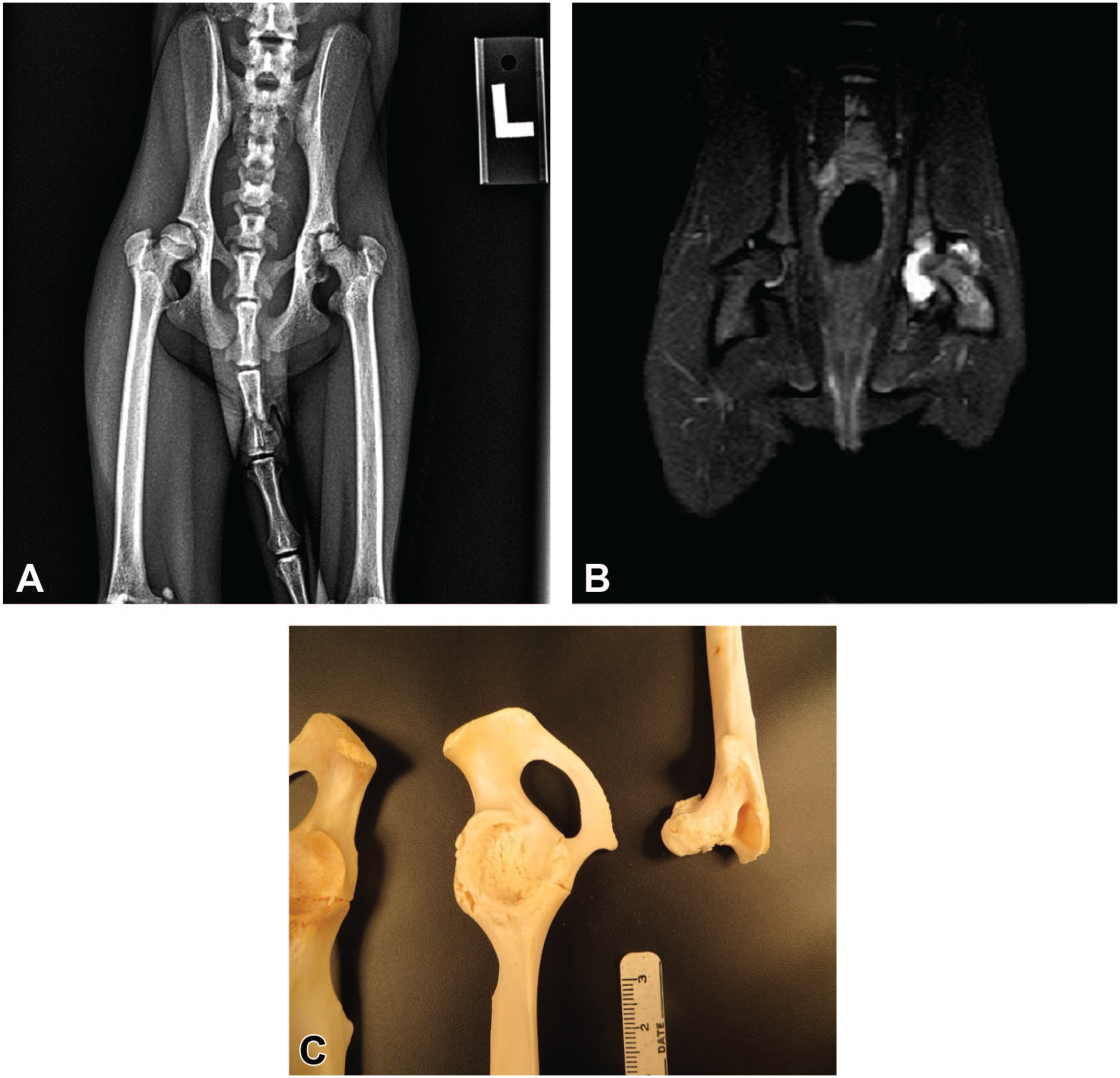
Radiographic, MRI, and macroscopic findings: (A) Ventrodorsal radiographs show increased radiodensity, narrowed femoral neck, irregular femoral head and acetabulum, and enlarged joint space of the left coxofemoral joint, (B) MRI T2 STIR images show a hyperintense signal within the left coxofemoral joint indicative of joint effusion, and (C) macroscopic findings include femoral head atrophy and irregularity of the femoral head and acetabulum. MRI indicates magnetic resonance imaging.
Gross necropsy and histopathology were performed for 30 cases; macroscopic and microscopic findings were generally similar between affected animals but varied in severity depending on the duration of disease.
Macroscopic findings included one or more of the following changes: femoral head atrophy (>15% loss by volume), irregular surface of the femoral head and/or acetabulum with segmental to complete loss of articular cartilage and variable ulceration of the exposed femoral head and acetabular bone, thickening of the coxofemoral joint capsule, excess fluid within the coxofemoral joint space, and atrophy of the hind limb muscles of the affected limb (Figure 1).
Microscopic findings included one or more of the following changes: irregular femoral and/or acetabular joint surfaces with segmental to complete loss of articular cartilage, eburnation of the subchondral bone with exposure of the medullary cavity, growth of fibrovascular connective tissue (pannus) across affected joint surfaces, synoviocyte hyperplasia, locally extensive bone remodeling with thickened trabeculae (osteosclerosis) and concurrent regions of osteolysis and/or osteonecrosis adjacent to areas of pannus, thinning of the femoral epiphyseal physis, and formation of enthesiophytes (bony proliferations within the insertion point of ligaments/tendons) along the capsule of the coxofemoral joint (Figure 2).
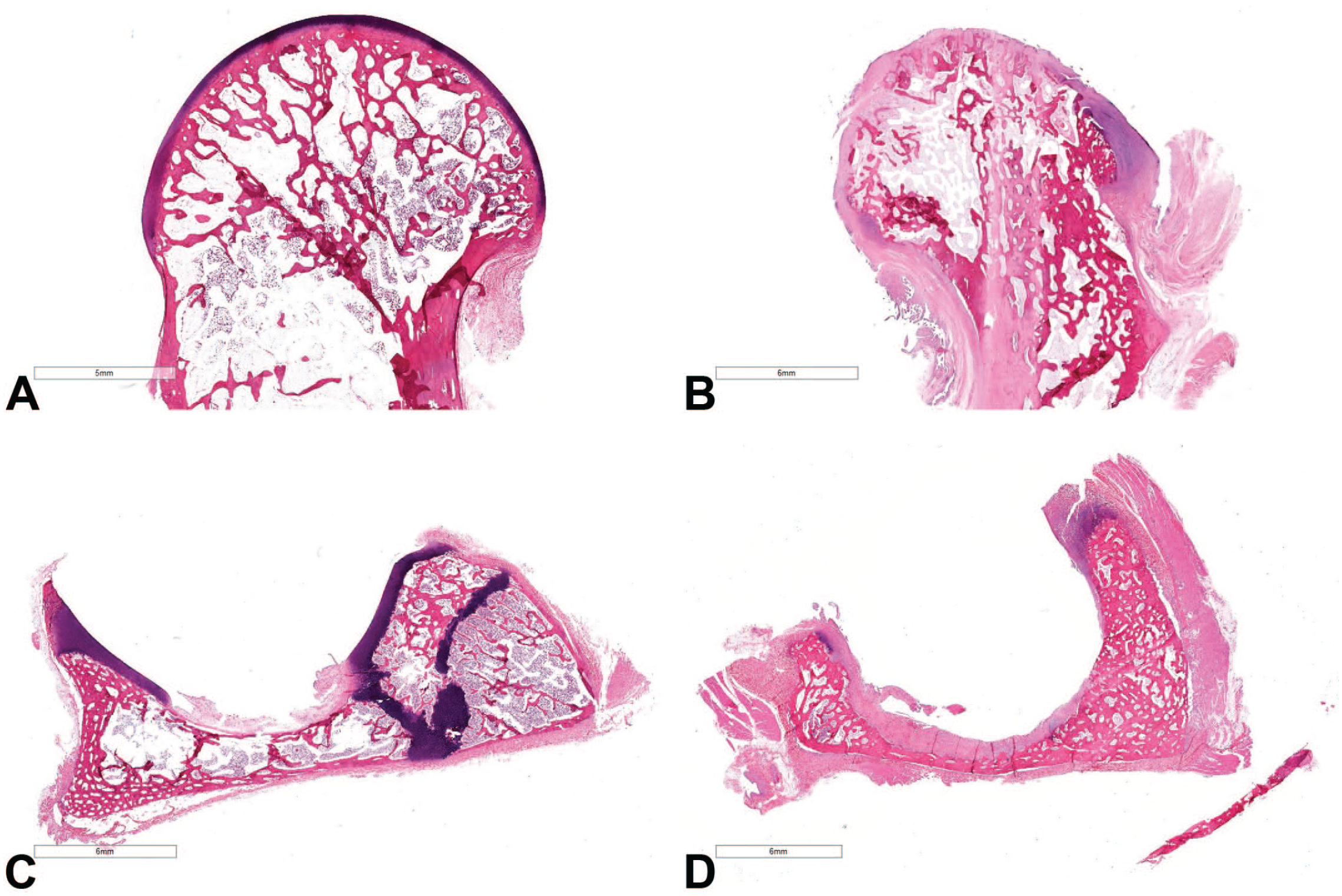
Histopathologic findings: (A) Normal femoral head, (B) affected femoral head with irregular, misshapen joint surfaces and loss of articular cartilage, (C) normal acetabulum, and (D) affected acetabulum with loss of articular cartilage and irregularity of the joint surfaces.
Discussion
Previous studies have identified a condition similar to the human entity of idiopathic femoral head chondrolysis in two macaques at a biomedical research laboratory. 2 In humans, idiopathic femoral head chondrolysis typically occurs in adolescents and is characterized by rapid loss of articular cartilage from the femoral head and acetabulum, resulting in progressive loss of function of the coxofemoral joint. Females and individuals of African American descent are affected at a higher rate, although the condition has been reported in males and multiple other ethnicities. 3 Reported clinical history typically includes chronic pain in the hip, groin, and/or upper leg that progresses to a loss of normal range of motion of the affected limb, disuse muscle atrophy, and fibrous ankylosis of the coxofemoral joint. 4 Radiographic findings include a symmetrically narrowed coxofemoral joint space with erosion of articular cartilage, abnormal femoral physis closure, periarticular osteoporosis, osteophyte formation along the femoral head and neck, and flattening of the femoral head. 5 It is a diagnosis of exclusion, and is made only in the absence of other inciting factors such as a history of trauma, clinical evidence of infection, or diagnostic findings supportive of other known disease entities. 6 Several causes have been speculated, including an underlying genetic predisposition, abnormal chondrocyte metabolism or chondrolytic enzyme release, and autoimmune-mediated destruction of the articular cartilage; however, none have been definitively confirmed. 4 There is no known treatment, and disease management is focused upon management of clinical symptoms with analgesics and physical therapy, or arthroplasty with soft tissue release in advanced cases that do not respond to conservative management.64
Other differential diagnoses for hip chondrolysis in primates include Legg-Calve-Perthes disease, pigmented villonodular synovitis, osteoarthritis, septic arthritis, rheumatoid arthritis, and traumatic injury. 5 In laboratory macaques used for nonclinical research, test article–related effects must also be considered. Legg-Calve-Perthes disease has been previously reported in a rhesus macaque at a research facility. 7 This condition is characterized by avascular necrosis of the femoral head; it occurs in adolescents (2-3 yr) and is thought to be a result of disruption of the blood supply to the proximal femoral epiphysis. 8 Key diagnostic features of Legg-Calve-Perthes disease that distinguish it from idiopathic femoral head chondrolysis include necrosis of the subchondral bone with initial preservation of the articular cartilage and a lack of premature femoral physeal fusion. 2 Pigmented villonodular synovitis of the hip is a type of tenosynovial giant cell tumor and is diagnosed based on evidence of villous or nodular synovial membrane proliferation. 9 It typically results in localized inflammation of the joint with destruction of articular cartilage but does not affect the underlying bone. 2 Osteoarthritis is reported in macaques between the ages of 5 and 15 yr and commonly affects weight-bearing joints such as the knee. 10 While features of severe osteoarthritis such as articular cartilage fibrillation and osteophyte formation are similar to what was observed in many of our cases, osteoarthritis in young animals confined to a single joint would be uncommon. 2 Other conditions such as septic arthritis, rheumatoid arthritis, and traumatic injury would have characteristic clinicopathologic findings that would distinguish them from idiopathic femoral head chondrolysis. Without clinicopathologic changes or a history of injury, other causes of hip degeneration such as septic arthritis, rheumatoid arthritis, and localized trauma were considered unlikely.
The collected data were considered to be most consistent with idiopathic femoral head chondrolysis. There were no obvious patterns in disease incidence related to sex, animal source, or species. While the incidence rate is low, the impact on study success and animal welfare can be significant if undetected, which is likely considering the frequent lack of clinical signs. An awareness of idiopathic femoral head chondrolysis in macaques is important in the proper separation of background findings and test article–related effects. In addition, early screening including careful physical examination focused on the hips and/or diagnostic imaging for proper study placement could help minimize study impacts and improve animal welfare.
Footnotes
Acknowledgements
The authors would like to thank the in-life, necropsy, and histology technicians for their help with animal care, clinical testing, and macroscopic/microscopic processing of the cases.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
