Abstract
The authors describe a case in which well-circumscribed, expansile, and nonencapsulated nodular masses with missing digits were detected on the right and left feet in a 6-year-old female cynomolgus macaque from a routine toxicology study. Grossly, these masses were composed of variably sized and firm nodules containing white, chalklike material in the subcutaneous tissue on cross section. Microscopically, the nodules were composed of irregular lobules containing amorphous to granular, light to dark basophilic material that was surrounded by macrophages and multinucleated giant cells and separated by fibrous connective tissue. The nodule’s contents were von Kossa and Alizarin red S positive. Serum calcium and phosphorus levels of this monkey were within normal ranges. Based on the gross pathology, histopathology, serum chemistry, and histochemistry, a diagnosis of dystrophic calcinosis circumscripta was made. Dystrophic calcinosis circumscripta is an uncommon syndrome of mineralization that occurs following tissue damage, without abnormalities in calcium and phosphorus homeostasis, and it is characterized by deposition of calcium salts in soft tissues. To the best of the authors' knowledge, this is the first report of dystrophic calcinosis circumscripta in a cynomolgus macaque.
Calcinosis circumscripta is a form of ectopic mineralization characterized by single or multiple cutaneous nodules containing calcium salts (Wachtman et al. 2006). This syndrome has been reported in both humans and animals. In humans, calcinosis circumscripta is described clinically as firm, white dermal papules, plaques, or nodules that can occur in the skin and subcutaneous tissues in hands and knees and may be associated with pressure points, tendon sheaths, and terminal phalanges. Calcinosis circumscripta in humans is more common in females than males and may be associated with various connective tissue disorders such as Reynaud’s phenomenon, scleroderma, systemic lupus erythromatosus, telangiectasia, CREST syndrome, and dermatomyositis. In addition, trauma, insect bites, metabolic calcification (e.g., renal failure), and inherited disorders (e.g., pseudoxanthoma elasticum, Werner’s syndrome, and Ehlers-Danlos syndrome) may cause calcinosis circumscripta (Cohen 1980; Wong et al. 1986; Valdatta et al. 2003; Olsen and Chew 2006).
In animals, calcinosis circumscripta is clinically described as a raised, nodular, and firm to fluctuant mass and can occur in the tongue, margins of pinnae, spine, salivary gland, aorta, mammary glands, jejunum, and footpad (Berggren et al. 1965; Henderson and Giddens 1970; Anderson and King 1988; McEwan et al. 1992; Joffe 1996; Movassaghi 1999; Jeong et al. 2004). This syndrome has been reported in nonhuman primates, dogs, cats, horses, cows, buffalo, rabbits, turtles, and captive sitatunga (Dodd and Raker 1970; Ikede 1979; Stewart, Herling, and Dalinka 1983; Line, Ihrke, and Prahalada 1984; Anderson and King 1988; O’Brien and Wilkie 2001; Yanai et al. 2001; Yanai et al. 2002; Tafti, Hanna, and Bourque 2005).
In nonhuman primates, calcinosis circumscripta has been reported in two female rhesus macaques (Line, Ihrke, and Prahalada 1984). In addition, calcinosis circumscripta associated with a microchip implantation in a male common marmoset has been reported (Wachtman et al. 2006). Idiopathic calcinosis circumscripta has been described in an abstract in two cynomolgus monkeys (Anderson et al. 2007). However, no cases of dystrophic calcinosis circumscripta have been reported in cynomolgus monkeys, which are commonly used in toxicology studies. Here we report a case of dystrophic calcinosis circumscripta in a cynomolgus macaque.
The monkey in this case was a 6-year-old female-cynomolgus macaque from a 39-week oral toxicity study. SNBL USA, Ltd. (hereafter SNBL USA) is accredited by the Association for Assessment and Accreditation of Laboratory Animal Care, has an Animal Welfare Assurance issued by the Office of Laboratory Animal Welfare, is registered with the United States Department of Agriculture, and has an Institutional Animal Care and Use Committee (IACUC) responsible for SNBL USA’s compliance with applicable laws and regulations concerning the humane care and use of laboratory animals. The SNBL IACUC reviewed and approved the animal use in this study. Animals in this study were housed in a temperature- and humidity-monitored environment. The targeted range of temperature and relative humidity was between 18°C and 29°C and 30% and 70%, respectively. An automatic lighting system provided a 12-hour diurnal cycle. The animals were individually housed in stainless-steel cages that complied with the Animal Welfare Act and were fed daily with Purina Certified Hi-Fiber Primate Diet (PMI Nutritional International Inc., St. Louis, MO). The diet was routinely analyzed for contaminants and found to be within the manufacturer’s specifications. No contaminants were present at levels that interfered with the outcome of the study. Fresh drinking water was provided ad libitum to all animals. The water was routinely analyzed for contaminants. No contaminants were expected to be present at levels that interfered with the outcome of the study. Clinical and physical examinations, body weight assessment, clinical hematology/biochemistry, and urinalysis were performed periodically throughout the conduct of the study. At study termination, the animal was euthanatized by exanguination after intravenous barbiturate-based anesthesia solution administration and necropsied. Multiple nodular, raised, and variably sized (0.5 × 0.5 × 0.5 cm and 1.5 × 1 × 1 cm) masses were noted on the left and right feet. In addition, the number 3 and 4 right fingers were missing. No gross pathology changes were seen in any of the internal organs. Multiple organ tissue samples, including the feet masses, were collected, fixed in 10% neutral buffered formalin, routinely processed to hematoxylin and eosin slides, and evaluated by light microscopy. The feet nodular masses contained white gritty material suggestive of calcinosis circumscripta. To further characterize the feet nodular lesions, histochemical (trichrome, von Kossa and Alizarin red S) and immunohistochemical (anti-CD68) stains were applied to tissue sections of the masses.
All of the parameters assessed during the in-life portion of the study were within normal limits. Serum calcium and inorganic phosphorus levels of this monkey were within normal ranges. Apart from the feet lesions and missing digits, there were no other gross pathology observations. The feet masses were composed of variably sized and firm nodules containing white chalklike material in the subcutaneous tissue on cross section (Figure 1A). Microscopically, the nodules were composed of irregular lobules containing amorphous to granular, light to dark basophilic material (mineral) that was surrounded by macrophages and multinucleated giant cells and separated by fibrous connective tissue (Figure 1B). The complete necropsy showed no further evidence of mineralization at necropsy or by histopathologic evaluation in multiple organs. By histochemistry, the nodules had trichrome positive fibrous connective tissue surrounding the mineral, and the contents were von Kossa and Alizarin red S positive (Figures 1C-E-5). Immunohistochemistry demonstrated that macrophages and multinucleated giant cells have immunoreactivity for CD68 (Figure 1F).
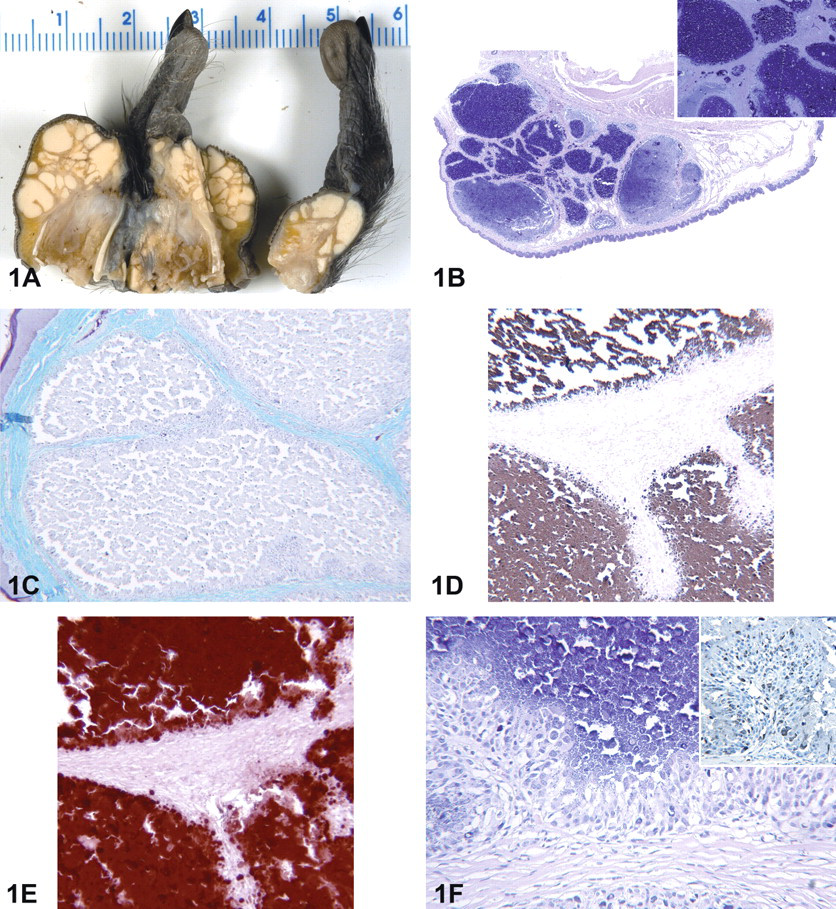
Female cynomolgus macaque (Macaca fascicularis). (A) Gross pathology of the right and left feet nodular masses. Nodular subcutaneous masses are composed of variably sized and firm nodules containing white chalklike material. (B) Hematoxylin and eosin histopathology of feet nodular masses. Variably sized nodules contained basophilic materials. Inset: Nodule contents are amorphous to granular, light to dark basophilic material. (C) Trichrome stain of the t nodular mass section from foot. Nodules had irregular structures surrounded by macrophages and multinucleated giant cells and were separated by trichrome positive fibrous connective tissue. (D) Von Kossa stain of the foot nodular mass section. Nodule contents are von Kossa positive. (E) Alizarin red S stain of the foot nodular mass section. Nodules contents are Alizarin red S positive. (F) Histopathology showing macrophages, multinucleated giant cells, and fibrous connective tissue surrounding the granular contents. Inset: Immunohistochemistry for CD-68 of the foot nodular mass section. Nodule CD-68 positive surrounding macrophages and multinucleated giant cells.
Abnormal deposition of calcium salts (calcium phosphate, calcium carbonate, calcium hydroxyapatite) is commonly referred to as pathologic calcification. Pathologic calcification of tissues can be divided into 3 categories: dystrophic, metastatic, or idiopathic calcification (Mendoza, Lavery, and Adam 1990). In dystrophic calcification, calcium salts are deposited in dying, damaged, or degenerate tissues despite normal serum levels of calcium and phosphate. Tissue damage in dystrophic calcification can be caused by necrosis, inflammation, trauma, infections, hemorrhage, foreign material, parasite migration, connective tissue pathology, or neoplasia. The pathogenesis of dystrophic calcification involves the formation of crystalline calcium phosphate mineral in the form of an apatite similar to hydroxyapatite of bone (Cotran, Kumar, and Collins 1999). Disorders that may be associated with dystrophic calcification are Ehlers-Danlos syndrome, pseudoxanthoma elasticum, arteriosclerosis obliterans, venous calcifications, crystal deposition disorders, and calcification resulting from neurologic disorders (Black and Kanat 1985). A recently described autosomal recessive disorder in humans is called familial tumoral calcinosis (FTC), particularly a form of the syndrome that is characterized by an absence of metabolic abnormalities, termed normophosphatemic (NFTC; Chefetz et al. 2008; Sprecher 2009). NFTC is associated with an absence of functional SAMD9, a putative tumor suppressor and anti-inflammatory protein (Chafetz et al. 2008). NFTC often manifests with nonspecific vasculitis-like erythematous rash and oral mucosal inflammation during the first year of life, and such eruptions end with painful calcified ulcerated nodules (Metzker et al. 1988). NFTC is thought to replicate many aspects of dystrophic calcinosis (Chefetz et al. 2008). The histopathologic features of NFTC are similar to those of dystrophic calcification. However, the female monkey in this case, unlike NFTC in humans, had no foot lesions early in her life and did not show involvement of the mucous membranes, and no erythematous rashes were noted before the development of the calcified nodules.
Dystrophic calcification can be further divided into either localized (calcinosis circumscripta) or widespread (calcinosis universalis, calcinosis cutis). Apocrine sweat gland cysts, follicular cysts, and skin tumors have been associated with calcinosis circumscripta (Wachtman et al. 2006), while hyperadrenocorticism and diabetes mellitus have been associated with calcinosis universalis (Wilkinson et al. 1999). Metastatic calcification, on the other hand, may occur in normal undamaged tissues and is generally associated with abnormalities in calcium and phosphorous metabolism, and calcification can occur in several organs including the gastric mucosa, lung, kidneys, and arteries (Cotran, Kumar, and Collins 1999). The most common causes of metastatic calcification are vitamin D disorders and intoxication, parathyroid hormone hypesecretion, renal failure, and bone destruction due to neoplasia (Cotran, Kumar, and Collins 1999). The pathogenesis of metastatic calcification involves the loss of acid and increase in intracellular pH that predisposes tissues to calcification (Cotran, Kumar, and Collins 1999). Idiopathic calcification occurs in the absence of evident tissue or metabolic abnormalities.
The monkey in this case had clinical chemistry values that were within normal limits, and the lesion was restricted to the feet without involvement of any other organs. The detailed history of the feet lesion in this female monkey who was born in 2002 was as follows: multiple swellings (½-1 cm in size) were found on the dorsal surface of the right foot on July 30, 2007. On August 1, 2007, multiple raised, round, soft to firm nodules (3-5 mm in size) were noted on the right foot dorsal aspect. On August 24, 2007, the animal exhibited bearing weight on the affected foot, and surgical amputation was recommended by the supervising veterinarian. Surgical amputation of the third and fourth digit of the right foot was performed on September 6, 2007, and all small pockets of caseous lesions were curetted. The surgical site on the right foot appeared to have healed well by October 14, 2007. A small (3-mm), firm, raised mass was noted in the left foot between the third and fourth digits on September 12, 2007. At the time of necropsy on February 28, 2008, a mass (1.5 × 1 × 1 cm) was noted on the right footpad with missing number 3 and 4 digits. In addition, a mass (0.5 × 0.5 × 0.5 cm) was noted on the left foot. Therefore, the calcification in this monkey was characterized as dystrophic (i.e., related to feet trauma). Von Kossa and Alizarin red S stains have been used to detect calcium deposition (Marcos et al. 2006). The granular basophilic material had strong histochemical reactivity to both Von Kossa and Alizarin red S stains, which confirms the calcareous nature of the material. The location of these nodules in the monkey in this case and the microscopic, histochemical, and immunohistochemical features have similarities to calcinosis circumscripta reported in other species (dogs, cats, horses, cows, buffalo, rabbits, turtles, and captive sitatunga).
Calcinosis circumscripta treatment includes medical or surgical therapies (Mendoza, Lavery, and Adam 1990; Valdatta et al. 2003). Medical therapy may not be effective, and surgical excision has shown to be beneficial to relieve the symptoms in humans. Therapies in human medicine include intralesional corticosteroid injection, magnesium or aluminum hydroxide, etidronate disodium, and bisphosphonates (Valdatta et al. 2003). In veterinary medicine, complete resolution has been reported following surgical excision of the lesion (Davidson et al. 1998). However, recurrence of these lesions may occur (Valdatta et al. 2003).
Among laboratory animal species, no reports of dystrophic calcinosis circumscripta are available in cynomolgus macaques used in toxicology studies. Subcutaneous lesions in the digits of young cynomolgus monkeys are generally rare in drug safety studies. This case represents the first description of dystrophic calcinosis circumscripta in a cynomolgus monkey.
Footnotes
Acknowledgments
The authors thank Norbert Makori, Dale Morris, James Klaassen, Steven Meyer, and Ryoichi Nagata for the technical assistance. This work was sponsored and funded by Pfizer Inc.
Study was conducted at SNBL (Kenichi Sato) and sponsored and supported by Pfizer Inc (Zaher Radi).
