Abstract
Samples of biologic specimens and their derivatives (eg, wet tissues, paraffin-embedded tissue blocks, histology slides, frozen tissues, whole blood, serum/plasma, and urine) are routinely collected during the course of nonclinical toxicity studies. Good Laboratory Practice regulations and/or guidance specify minimum requirements for specimen retention duration, with the caveat that retention of biologic specimens need not extend beyond the duration of sample stability. However, limited availability of published data regarding stability for various purposes following storage of each specimen type has resulted in confusion, uncertainty, and inconsistency as to the appropriate duration for storage of these specimens. To address these issues, a working group of the Society of Toxicologic Pathology Scientific and Regulatory Policy Committee was formed to review published information, regulations, and guidance pertinent to this topic and to summarize the current practices and rationales for retention duration through a survey-based approach. Information regarding experiences reaccessing biologic specimens and performing sample stability investigations was also collected. Based on this combined information, the working group developed several points to consider that may be referenced when developing or revising sample retention practices.
This Points to Consider article is a product of a Society of Toxicologic Pathology (STP) Working Group commissioned by the Scientific and Regulatory Policy Committee (SRPC) of the STP. It has been reviewed and approved by the SRPC and Executive Committee of the STP, the British Society of Toxicologic Pathologists (BSTP) and European Society of Toxicologic Pathology (ESTP) but it does not represent a formal Best Practice recommendation; rather, it is intended to provide key “points to consider” in designing nonclinical studies or interpreting data from toxicity and safety studies in support of regulatory submissions. The points expressed in this document are those of the authors and do not reflect views or policies of the employing institutions. Readers of Toxicologic Pathology are encouraged to send their thoughts on these articles or ideas for new topics to the Editor.
Keywords
Introduction
In the course of in vivo nonclinical toxicity studies, samples of a variety of biological specimens (tissues, blood, other body fluids) may be collected for analysis. The study protocol and institutional standard operating procedures (SOPs) define how these samples should be processed and evaluated, although in some cases the protocol may stipulate that samples be collected and held “for potential future analysis” or that residual material is archived after the completion of the primary analysis. In these instances, practical issues arise regarding the need to understand the appropriate limits of storage duration which will ensure that sample quality and stability are maintained to enable potential analysis or reanalysis. Several factors may influence specimen quality over time, including specimen type, processing procedures, and storage conditions (eg, room temperature vs frozen). For example, in typical toxicity studies, protocol-specified tissues are collected in 10% neutral buffered formalin (NBF), processed, and embedded into paraffin (referred to as formalin-fixed, paraffin-embedded or FFPE) blocks, from which tissue sections are prepared and mounted on glass slides. These sections are then stained routinely with hematoxylin and eosin (H&E) for histology and/or are potentially used for special staining, immunohistochemistry (IHC), or in situ hybridization (ISH) evaluations. 1 The FFPE blocks are considered optimal for long-term storage of tissue specimens, but there may also be residual or unprocessed tissues that are retained in sealed bags/jars with 10% NBF (“wet tissues”). Over time, tissues stored in 10% NBF may begin to accumulate artifacts due to excessive cross-linking of proteins, reduced formalin buffering capacity, tissue drying, or other effects. 2 At some point, these artifacts may impair histologic evaluation and render the specimens unsuitable for microscopic interpretation. Limited data are available regarding the period of time in storage required for these degradative processes to adversely impact specimen evaluation, and there is uncertainty on how to use this information to make informed decisions on retention and/or disposal of stored specimens. Although FFPE blocks and stained sections of tissues on glass slides (histology slides) are generally accepted to have superior stability compared to wet tissues, 3 blocks and slides may still have a practical life span after which the regulatory requirements, logistics, and/or costs of storage may outweigh the potential benefit of their continued retention. In addition, other specimen types such as those used for clinical pathology analysis (eg, whole blood [WB], serum/plasma, urine, blood and bone marrow smears) or other investigational samples (eg, frozen tissues) have variable stability limitations that are often poorly characterized, leading to similar uncertainty regarding appropriate retention/disposal of stored sample types. 4
Given the uncertainty in determining the appropriate length of time to retain biological specimens, some institutions may effectively archive specimens from Good Laboratory Practice (GLP) and/or non-GLP studies indefinitely in order to avoid noncompliance with even the most conservative interpretation of regulations and guidance. This approach may also be followed to (1) prevent accidental sample disposal in the event that novel safety liability issues or regulatory complexities arise that result in clinical hold and (2) leverage stored samples (by a protocol amendment or new study protocol) to avoid potentially having to repeat an in vivo study in the event additional information is needed. Although indefinite storage certainly ensures compliance with legal and regulatory responsibilities, this strategy also has disadvantages. For example, evaluation of degraded samples could confound interpretation and lead to erroneous conclusions. Furthermore, extended storage is associated with on- or off-site archival expenses and personnel resources to curate and monitor large banks of stored samples that may never be reaccessed (including equipment maintenance and ensuring sample containers remain intact).
Rationale for Sample Retention Working Group Formation and Manuscript Objectives
In order to aid the decision-making process regarding retention of biological samples from nonclinical toxicity studies, a working group comprised of anatomic and clinical pathologists representing industry (pharmaceutical, biotechnology, agrochemical, and medical device companies; hereafter collectively referred to as biotechnology/pharmaceutical companies), contract research organizations (CROs), academia, and government was formed under the auspices of the Society of Toxicologic Pathology (STP) Scientific and Regulatory Policy and Executive Committees. The primary goal of the working group was to collate available published data, regulations, and guidance from regulatory agencies with practical input collected from a survey on current sample retention practices of STP members working in biotechnology/pharmaceutical companies, CROs, government research laboratories, and academic institutions. The manuscript herein aims to provide points to consider in determining sample retention procedures for various biological sample types, with a particular focus on formalin-fixed wet tissues. A summary of information from selected regulations and guidance and published literature is presented in the context of the GLP framework for study conduct, followed by the results of the survey including the topics of (1) retention duration for various sample types, (2) the rationale for the retention duration, (3) investigative work that may have been performed to support sample stability decisions for retention duration, and (4) practical experience reaccessing stored samples. The information from these sources is integrated to provide support for proposed retention durations to consider for each major specimen type. These durations are not intended to be universal recommendations nor are they intended to supersede relevant regulations and/or guidance, but instead are to provide information that may be referenced as sample storage policies are being developed or revised.
History of GLP and Impact on Sample Retention Practices
The US Food and Drug Administration (FDA) requires that nonclinical studies of new drugs, devices, food additives, and chemicals be conducted in order to assess potential safety liabilities and efficacy in humans. 5,6 Human health and safety are dependent upon the conclusions made from these studies and the resultant risk assessments, and as such must be conducted according to sound scientific principles with strict attention to quality control and quality assurance. GLP is a quality control and integrity system of federal regulations that dictates the industry standards for organizational processes and the conditions under which nonclinical safety studies are planned, performed, monitored, recorded, reported, and archived. 7 The GLP framework was introduced in the 1970s by the FDA in response to a lack of quality control and scientific integrity reported in numerous nonclinical toxicity studies. This included cases of scientific misconduct, fraud and poor quality studies with inadequate or insufficient documentation, and animal health and welfare issues. 8 These new federal regulations outlined the minimum standards for conducting nonclinical laboratory studies that support or are intended to support applications for research or marketing permits for products regulated by the FDA. The objectives of GLP was to prevent undetected fraud, decrease public risk, reduce unnecessary use of animals, avoid acceptance of false or poor quality data, and ultimately to improve the quality and integrity of study data to ensure confidence in regulatory decisions.
One of the core tenets of GLP in the United States (Code of Federal Regulations [CFR], Title 21, Part 58) 9 is that in order to ensure consistent, reliable global standards of nonclinical safety testing, a third party must be able to reconstruct the study from what remains after completion; therefore, adequate data, documentation, and biologic samples must be available to support the study conduct and conclusions. Raw data, as pertaining to pathology, include the records of original observations such as the numerical data generated by automated blood or urine analyzers, necropsy records (verifying animal identity and any macroscopic lesions), body and organ weights, and the qualitative and semiquantitative histomorphologic observations and diagnostic interpretations made from microscopic evaluation of tissue sections by the toxicologic pathologist as presented in the signed and dated finalized pathology report. Biologic samples include blood, body fluids, wet tissues, and their derivatives such as FFPE tissue blocks or histology slides. More generally, biologic specimens (a term used interchangeably with “samples” in this manuscript) are defined by regulatory agencies as “any material derived from a test system for examination, analysis, [and/or] retention,” with some variability in the definition based on global region. 9 These data and specimens (samples) from GLP studies are retained during or after the finalization of the study at a GLP archive located either at the test facility or a commercial facility and should be protected against deterioration to the extent possible in order to be readily retrievable upon request. Test facility management and/or the sponsor of the study determine the length of retention for each specimen type and ensure appropriate SOPs are in place for archival and disposal.
Review of Major Global Regulations, Guidelines, and Published Literature for Retention of Nonclinical Biologic Specimens
Consistent with the goals of GLP, global regulations and/or guidance for specimen and data retention from nonclinical studies are intended to allow for independent verification of study conclusions should issues arise during the drug development process, for example, in the case of unexpected findings from clinical trials or as part of regulatory inspections. In particular, sample retention is intended to allow pathology findings to be re-evaluated and confirmed by the sponsor or a third-party pathologist as needed. Some directions regarding minimum retention periods may be specified by regional or national regulations and/or guidance, after which time the materials may be discarded, however some gaps or opportunities for uncertainty may remain (Table 1). For example, current FDA regulation (REF 21 CFR 58.195 and 21 CFR 58.190[a]) 10 specifies that in the United States these nonclinical biological specimens should be retained for various periods dependent on the relationship to regulatory submission, for example, “2 years following the date on which an application for a research or marketing permit…is approved” or…”5 years following the date on which the results of the nonclinical laboratory study are submitted…”. However, the regulation also contains a key caveat related to tissue quality which may be a source of inconsistent interpretation: “Wet specimens (except those specimens obtained from mutagenicity tests and wet specimens of blood, urine, feces, and biological fluids)…which are relatively fragile and differ markedly in stability and quality during storage, shall be retained only as long as the quality of the preparation affords evaluation.” Similarly, the European Organization for Economic Co-operation and Development guideline ENV/JM/MONO (2007) 11 strongly recommends that sample retention schedules should be defined by the appropriate regulatory authorities according to the period during which GLP regulatory inspections may occur or at least “3 inspection cycles,” so that inspectors can evaluate the GLP compliance of the test facility and the respective studies. The guidance also refers to sample stability, indicating perishable samples should be “discarded when the quality of the material no longer permits evaluation” (assuming optimal storage conditions and adequate justification and documentation of the disposal) but does not refer to specific durations for either of these rationales or provide information related to quality assurance/quality control. The Japanese Ministry of Health, Labour and Welfare Ordinance 21 on GLP for Nonclinical Safety Studies of Drugs 12 notes that “the test facility management shall properly retain study-related materials in archives” but provides no further information related to the expected duration of retention.
Selected Health Authority Recommended Sample Retention Guidance in Relationship to Regulatory Submissions.
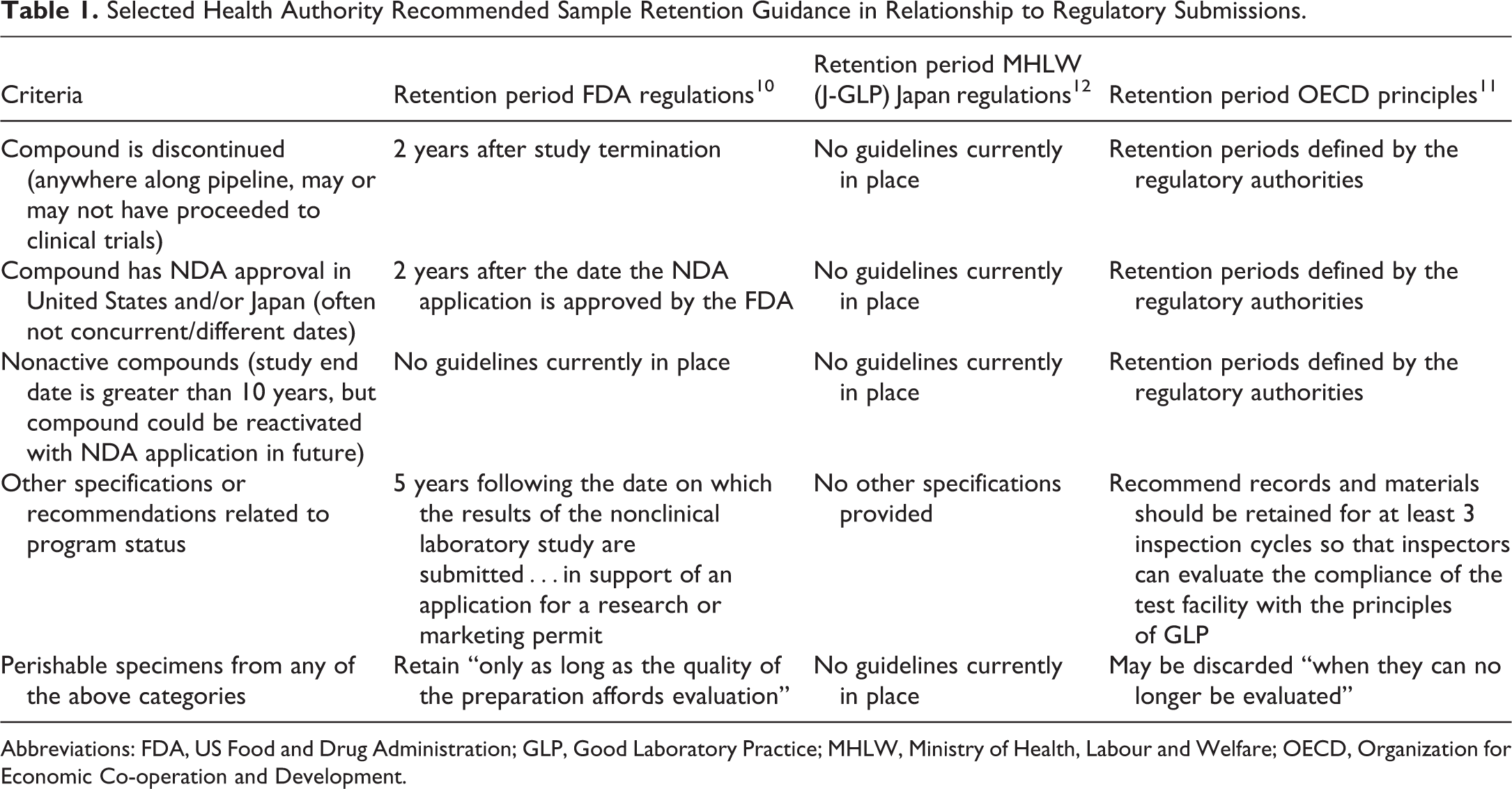
Abbreviations: FDA, US Food and Drug Administration; GLP, Good Laboratory Practice; MHLW, Ministry of Health, Labour and Welfare; OECD, Organization for Economic Co-operation and Development.
Other types of institutions such as academic or national animal health diagnostic laboratories also collect nonclinical biological samples but may not always be subject to the same restrictions that dictate retention of samples from studies intended to support clinical trials. Therefore, space considerations and other convenience factors may play a more important role in determining sample retention durations in some cases, and varying durations of retention based on individual preference may be more common. For example, in the American Association of Veterinary Laboratory Diagnosticians Requirements for an Accredited Veterinary Medical Diagnostic Laboratory, 13 recommended durations for retention of biologic specimens are not specified and briefly reference that laboratories should have “procedures for…the preservation of specimens.” Additional description stating the goals of GLP criteria is provided regarding retention of data generated from the initial assays. Specifically, institutions should “retain for a defined period of time original observations, derived data, calibration records, staff records, a copy of each test report issued and any other information necessary to recreate the activity.” However, a specific duration for retention of these materials is not specified. The Royal College of Pathologists Best Practice Recommendations for Storage and Retention of Animal Tissues and Records (G184) 14 details specific periods for retention of various specimen types for the purpose of allowing “correlation with results from previous and subsequent specimens, [and] responding to queries or challenge, audit and quality assurance,” with consideration for the “ethical and legal acceptability of continued storage,” but the scope of the guidance excludes “pathological studies carried out as part of GLP regulatory studies under the UK GLP Compliance Monitoring Programme.” Recommended retention periods in this document include retaining WB, plasma, serum, and other bodily fluids, aspirates and swabs for 24 to 48 hours after issuance of the final diagnostic report by the laboratory “unless sample deterioration precludes storage.” The recommendation also states that remaining serum or plasma should be stored for “as long as practicable, either frozen or lyophilized” and indefinitely where possible “to facilitate retrospective studies in the identification of emerging infections and vaccination program monitoring.” Wet tissues are recommended to be stored for “4 weeks after issue of final report” and stained histology slides for at least 10 years depending “on their nature and purpose.” For paraffin- or resin-embedded tissue blocks, “storage for at least 10 years is recommended, if facilities permit,” but it is also recommended that the need for archiving can be reviewed after 5 years. Similar durations for several specimen types are recommended in the multinational International Organization for Standardization ISO/IEC 17025 Accreditation Application appendix, 15 for example, most clinical pathology samples are only recommended to be stored for a week (with the exception of blood smears to be stored 2 months and wet [fixed but unblocked] tissues for a month). Recognizing their importance as a historical resource, glass histology slides for microscopic evaluation were recommended to be stored for 3 years and FFPE blocks for 10 years. Notably, none of these resources included the rationale for selection of these specific retention durations.
A review of current literature identified publications that describe tissue quality in selected tissues stored in 10% NBF for durations from 1 to 18 years. 16- 18 These studies detected artifacts in H&E-stained tissue sections prepared from tissues stored in 10% NBF for 1 year or longer including vacuoles, crystals, diminished H&E staining capacity, formalin pigment (acid hematin), and tissue disruption. Despite these findings, the authors of these studies concluded that 14 years of storage in 10% NBF was the cutoff for evaluability of tissues; however, the criteria used for this determination were not defined. 16 For other techniques such as IHC, ISH, or RNA extraction, the sample stability time in 10% NBF may be much shorter and may differ on a case-by-case basis depending on IHC/ISH target or other factors. 2,19,20 For example, in one study performed by the Japanese Society of Pathology, RNA extracted from wet tissues contained time-related degradation and artifacts (cysteine/threonine substitution) after as short as 1 week of storage. 20 Of note, this manuscript also determined handling and storage parameters (in liquid nitrogen) for frozen samples or tissues processed in optimal cutting temperature that allowed good quality RNA extraction from samples up to 10 years old. 20
Published sample stability information is more readily available for several clinical pathology sample types, with stability generally being shorter for fluid specimens relative to fixed tissues, FFPE blocks, and histology slides. 21 Refrigerated (3-4 °C) or room temperature (20-25 °C) WB samples are generally regarded as having a short stability window (<24 hours), with samples for standard hematology evaluation ideally analyzed within 1 to 6 hours of collection. 22 The short duration of stability of WB samples is largely due to rapid lysis of cellular components in fluid as well as changes in cell volumes during storage. Although refrigeration may slightly prolong WB stability for hematology analysis, the ability to freeze these samples in long-term storage is generally limited. 23 Fresh plasma samples for coagulation testing are also generally analyzed within 6 hours of collection, although frozen long-term stability (−24 to −74 °C for at least 18 months) has been demonstrated for many coagulation proteins. 24,25 Stability of urine analytes measured in routine urinalysis panels is typically ≤24 hours under refrigeration and routine urinalysis is recommended to be completed within 6 hours of collection at room temperature. 26,27 Urine specimens intended for renal or other biomarker/protein analysis may be stored frozen (−20 to −70 °C) with variable analyte stability (days to years) depending on the specific biomarker assessed. 28- 30 Serum/plasma samples for routine clinical chemistry analysis, depending on the specific analyte, are also generally stable for 3 to 6 months of frozen storage; however, much longer stability (up to 10 years or longer) has been demonstrated for some biomarkers including some large proteins. 31 -33
Survey Results: Current Practices and Rationale for Retention of Samples From Nonclinical Toxicity Studies
Survey Format and Response Demographics
An electronic survey was conducted to collect data from respondents at multiple institution types (biotechnology/pharmaceutical companies, CROs, government research laboratories, and academic institutions) regarding current retention duration practices for various biologic samples including wet tissues, paraffin- or resin- embedded blocks, frozen tissue blocks, stained or immunolabeled histology slides, and several types of clinical pathology samples (Supplementary Figure 1). The rationale for specified retention periods was also requested (eg, regulations and/or guidance, sample stability determination, etc.). Additional questions addressed the frequency and purpose of sample re-evaluation/reanalysis after initial archival. Respondents submitted one response per institution (or per site, for multi-site institutions).
Sixty-five unique survey responses were received; however, not all respondents answered all questions. Of the 65 responses, 33 (50.8%) were from industry, specifically 29 pharmaceutical/biotechnology companies, 3 medical device companies, and 1 agrochemical company (Figure 1A). Respondent companies spanned all size ranges from <50 to >20,000 employees, with 501 to 5,000 total employees most commonly reported (18/33 [54.5%] respondents) and included an average of 6 employed toxicologic pathologists (excluding consultants; Figure 1B and C). Sixteen (24.6%) survey responses were received from CROs of varying size (6 individual responses represented different sites of 2 respective larger parent organizations), including small CROs with <50 employees (2/16 [12.5%]), medium CROs with 50 to 500 employees (8/16 [50%]), and large CROs with 501 to 20,000 employees (6/16 [37.5%]). Contract research organizations reported employing 2 to 28 anatomic and/or clinical pathologists per site, generally proportional in number to the size of the company. Seven (10.8%) responses were obtained from individuals representing private consulting practices (comprised of 1 toxicologic pathologist in 6 of 7 cases). Five (7.7%) responses were received from government research laboratories (4 located in the United States and 1 for which the location was not specified) and 4 (6.2%) were received from academic facilities (2 research laboratories and 2 research pathology core facilities).
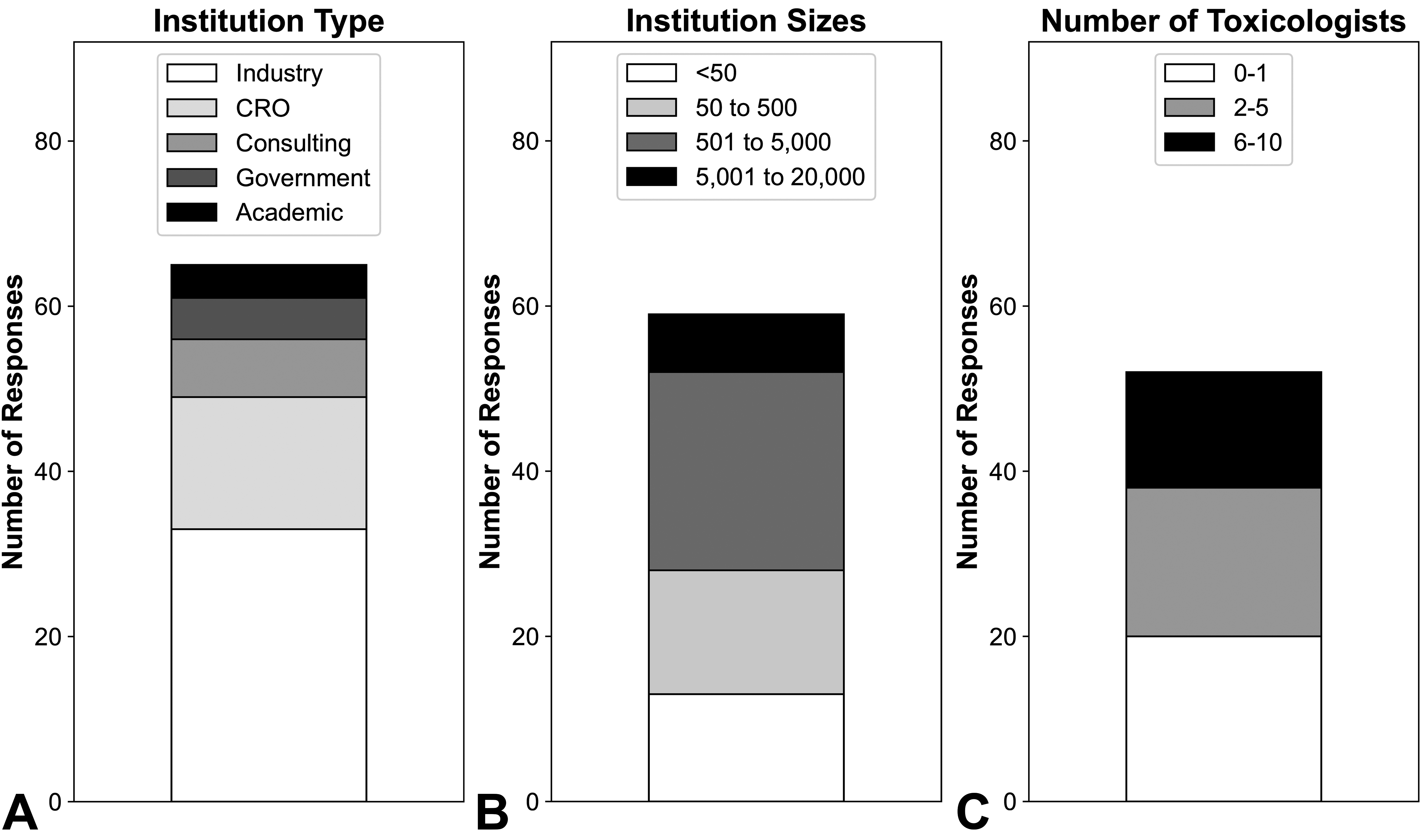
Survey demographics. Reported demographics of survey respondents with respect to (A) institution type, (B) overall institution size, and (C) number of employed toxicologists/toxicologic pathologists (excluding consultants). The majority of respondents were from biotechnology/pharmaceutical companies, with representation from institutions of varying size.
Survey Results: Institutional Practices, Retention Duration, and Documentation of Various Sample Types From GLP Nonclinical Toxicity Studies
A total of 54 (83.1%) of 65 respondents indicated that their institutions had defined practices for retention of samples from GLP nonclinical studies intended for regulatory filing, including all CROs and government research laboratories. The 11 respondents whose institutions did not have standard practices were primarily from private consulting practices (4 of 7 respondents in this category, 57.1%) but also included a minority of respondents from biotechnology/pharmaceutical companies (6 of 33, 18.2%) or academic laboratories (1 of 4, 25%). No further information concerning sample retention practices was provided from these respondents.
The duration of sample retention varied by sample type, with wet tissues, tissue blocks, and slides generally retained for longer periods than fluid samples (serum/plasma, WB, and urine; Figure 2). In general, the small number of respondents from academia and government precluded meaningful comparisons based on type of institution. However, comments from individual respondents are included below where relevant.
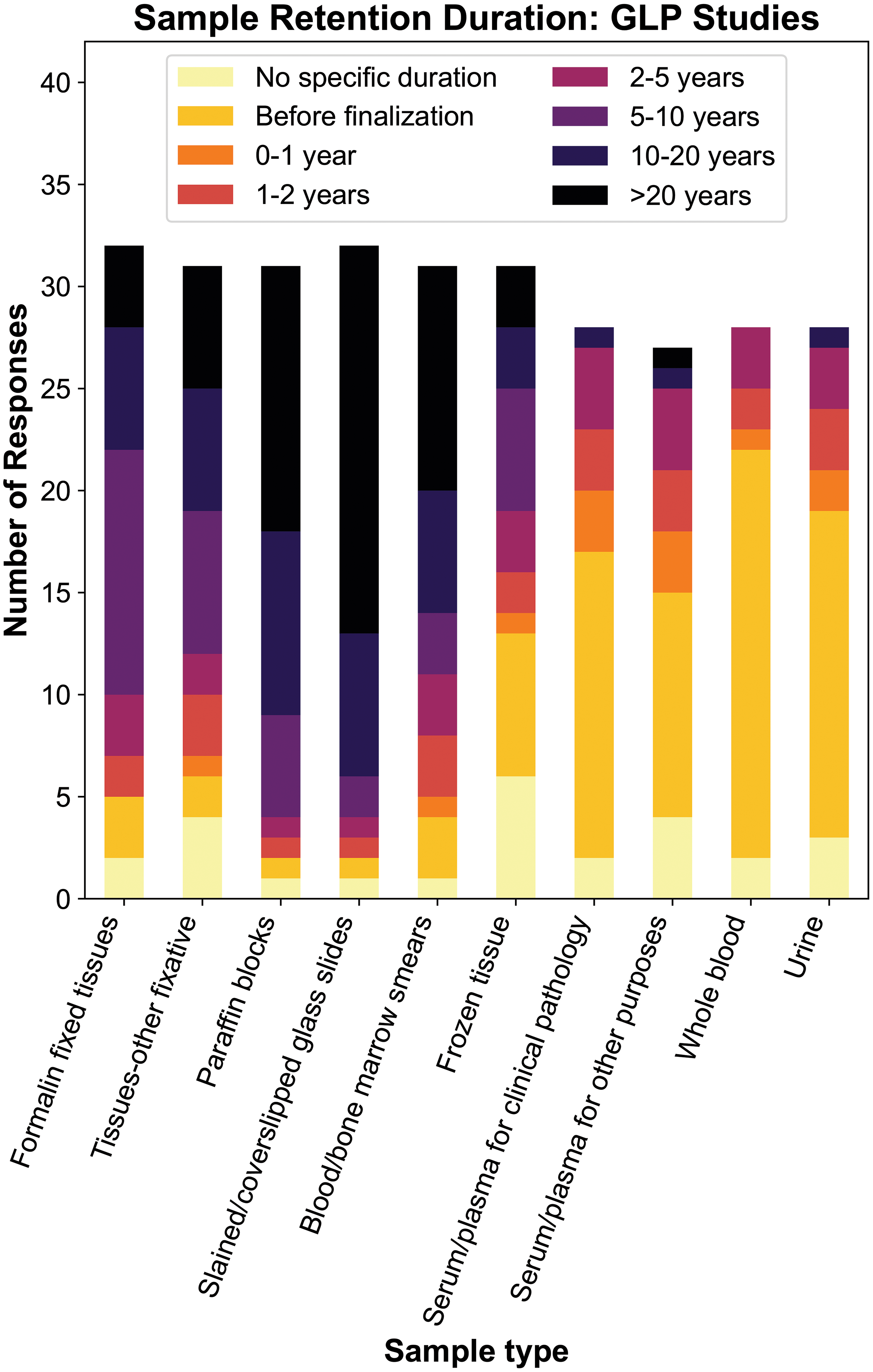
Reported retention duration for biologic specimens collected from GLP toxicity studies. Tissues, blocks, and histology slides are generally retained for longer durations than clinical pathology samples, whereas frozen tissue retention was highly variable. GLP indicates Good Laboratory Practice.
Formalin-fixed wet tissues from GLP studies were most commonly reported as being retained for 5 to 10 years before being discarded (12 of 32, 37.5%), although responses spanned all duration categories (ie, “discarded at/before study finalization” to “20+ years/indefinite”; Figure 2). Two additional respondents (2 of 32, 6.3%) indicated they had no designated retention time for wet tissues in 10% NBF. Retention duration for tissues from GLP studies stored in fixatives other than 10% NBF (eg, skeletons in glycerin) was generally as described above, with a few respondents indicating distinct retention periods (whether longer or shorter). Retention duration for frozen tissues was highly variable, with 6 (19.4%) of 31 respondents having no defined time for this sample type and other responses distributed across the range from “Discarded at study finalization” to “20+ years/indefinite” with no clear trends.
Paraffin blocks and stained/cover slipped/labeled glass histology slides from GLP studies were commonly retained longer than wet tissues, with “20+ years/indefinite” the most commonly selected response category for both sample types (13 of 31, 42% and 18 of 32, 59%, respectively). Most respondents indicated that the retention durations for blood or bone marrow smears were similar to those for the other types of histology slides; however, 8 (25.8%) of 31 respondents indicated shorter retention times. Of note, the survey did not attempt to differentiate retention durations for H&E-stained slides versus histochemical stained or immunolabeled slides. Additionally, although electron microscopy blocks were rarely specifically mentioned by survey respondents, one biotechnology/pharmaceutical company indicated that their retention time was the same as for paraffin blocks.
For clinical pathology fluid specimens from GLP studies, respondents most commonly indicated that these types of specimens are discarded at or before study finalization (serum/plasma [15 of 28, 53.6%], WB [20 of 28, 71.4%], and urine [16 of 28, 57.1%]). However, 37% of organizations retained one or more these specimen types for periods up to 5 years. It was uncommon for fluid specimens to be retained beyond 5 years (≤2 of 28 [7.1%] respondents).
A final survey question in this section referred to the procedures for documenting when samples were discarded, given that this documentation is included in the description of GLP regulation as a component required to reconstruct a study by supporting study conduct and may be requested as part of regulatory inspections. Respondents indicated sample disposition was documented in an electronic sample database (6 of 33, 18.2%), a memo/letter (16 of 33, 48.5%), or both (10 of 33, 30.3%).
Survey Results: Retention Duration for Samples From Non-GLP or Investigative Nonclinical Toxicity Studies
The majority of respondents across institution types (up to 26 of 31, 83.8%, depending on sample type) reported the same retention practices for GLP and non-GLP toxicity studies, especially for non-GLP studies intended for regulatory filing (survey data not shown). Of the 5 respondents who indicated distinct practices for non-GLP studies, 3 respondents indicated shorter retention duration for formalin-fixed wet tissues from non-GLP studies compared to GLP studies (eg, 1-2 years for non-GLP studies vs 2-5 years or 5-10 years for GLP studies) and 2 respondents had no specific retention period for non-GLP wet tissues. Depending on the sample type, the retention practices for investigative studies not intended for regulatory submission were generally different from those of GLP studies, with up to 13 (41.9%) of 31 respondents reporting shorter or undefined retention periods for samples collected from investigative studies, with samples often discarded at study finalization (Figure 3).
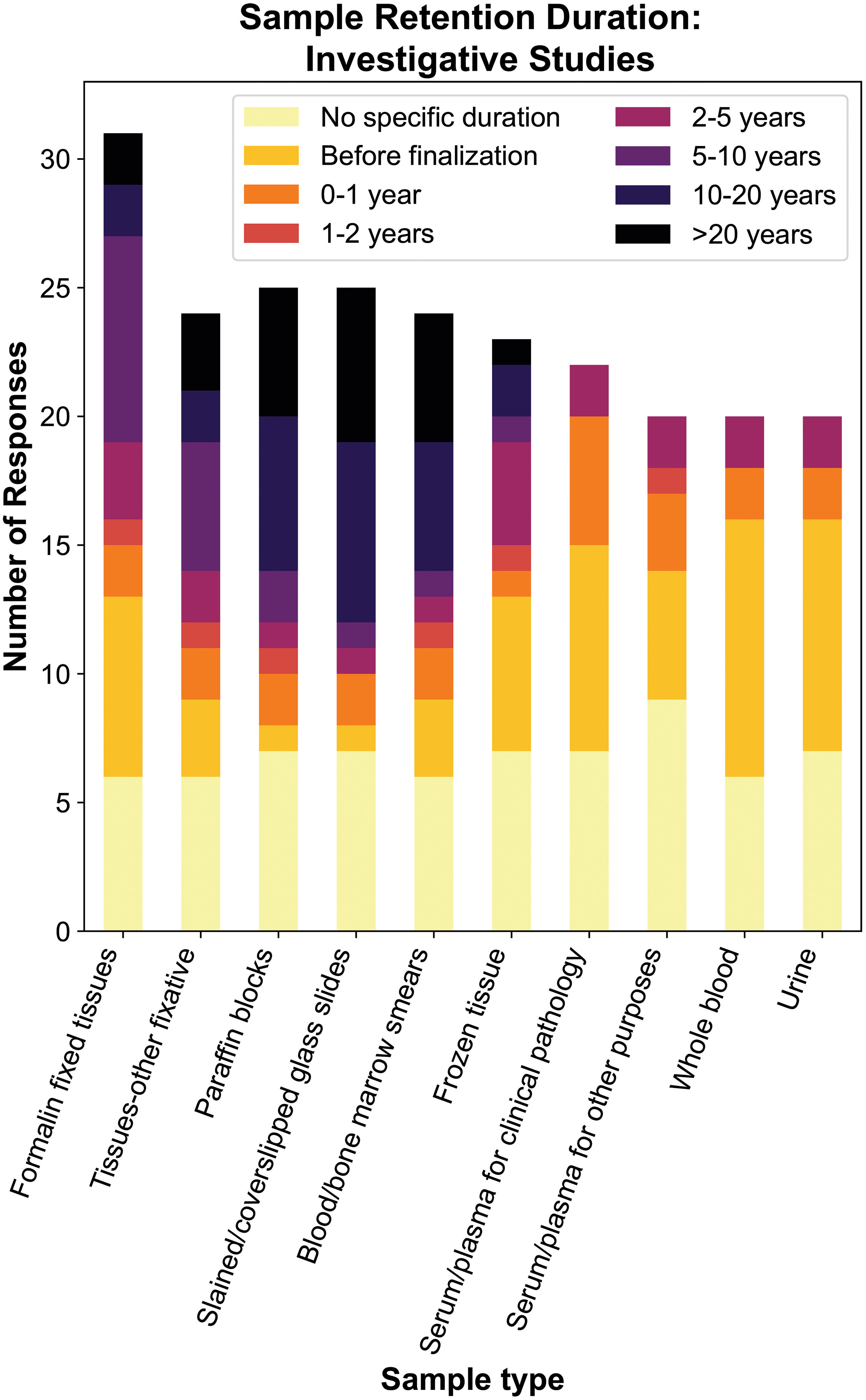
Reported retention duration for various sample types from investigative studies not intended for regulatory filing. Compared to GLP or non-GLP toxicity studies intended for regulatory filing, a higher percentage of respondents indicated they lacked specific retention durations or had shorter retention duration across sample types for investigative studies not intended for filing. GLP indicates Good Laboratory Practice.
The clinical stage of the program also affected sample retention duration for 10 (37.0%) of 27 biotechnology/pharmaceutical companies or CRO respondents that had standardized sample retention practices in place. Longer retention times were generally reported for compounds being tested in clinical trials and shorter retention times for discontinued and/or early discovery stage projects. Rarely, biotechnology/pharmaceutical respondents noted distinct retention policies for marketed products, and 2 CRO respondents noted deferral to sponsor requirements as rationale for differing practices based on program clinical stage.
Survey Results: Rationale for Retention Duration of Samples From GLP Nonclinical Toxicity Studies
Compliance with regulations and/or guidance(s) was the most common rationale cited for the retention period of wet tissues stored in 10% NBF or other fixatives, FFPE blocks, stained/labeled histology slides, and (to a lesser degree) blood/bone marrow smears (Figure 4). Additional information provided by some respondents recognized local and regional differences in regulation and/or guidance specifications (eg, one industrial respondent highlighted specific differences based on location, ie, 15 years retention for Germany, 5 years for Spain/United Kingdom, 10 years for all other countries). Another biotechnology/pharmaceutical company respondent highlighted the perceived likelihood of needing to answer potential regulatory queries over the course of development as part of the rationale for their internal retention practices. In general, respondents who cited regulations and guidance as the primary rationale for storage duration had a longer duration of retention than those with other responses; however, even among respondents citing similar rationale, there was variability in reported retention times. For example, the wet tissue retention duration for those basing their retention practices primarily on regulations and/or guidance varied from 5 to 10 years (n = 5) to 20+ years/indefinite (n = 4). This variation may represent regional differences in regulation specifications, variability in interpretation of the guidance, and/or application of other secondary considerations.
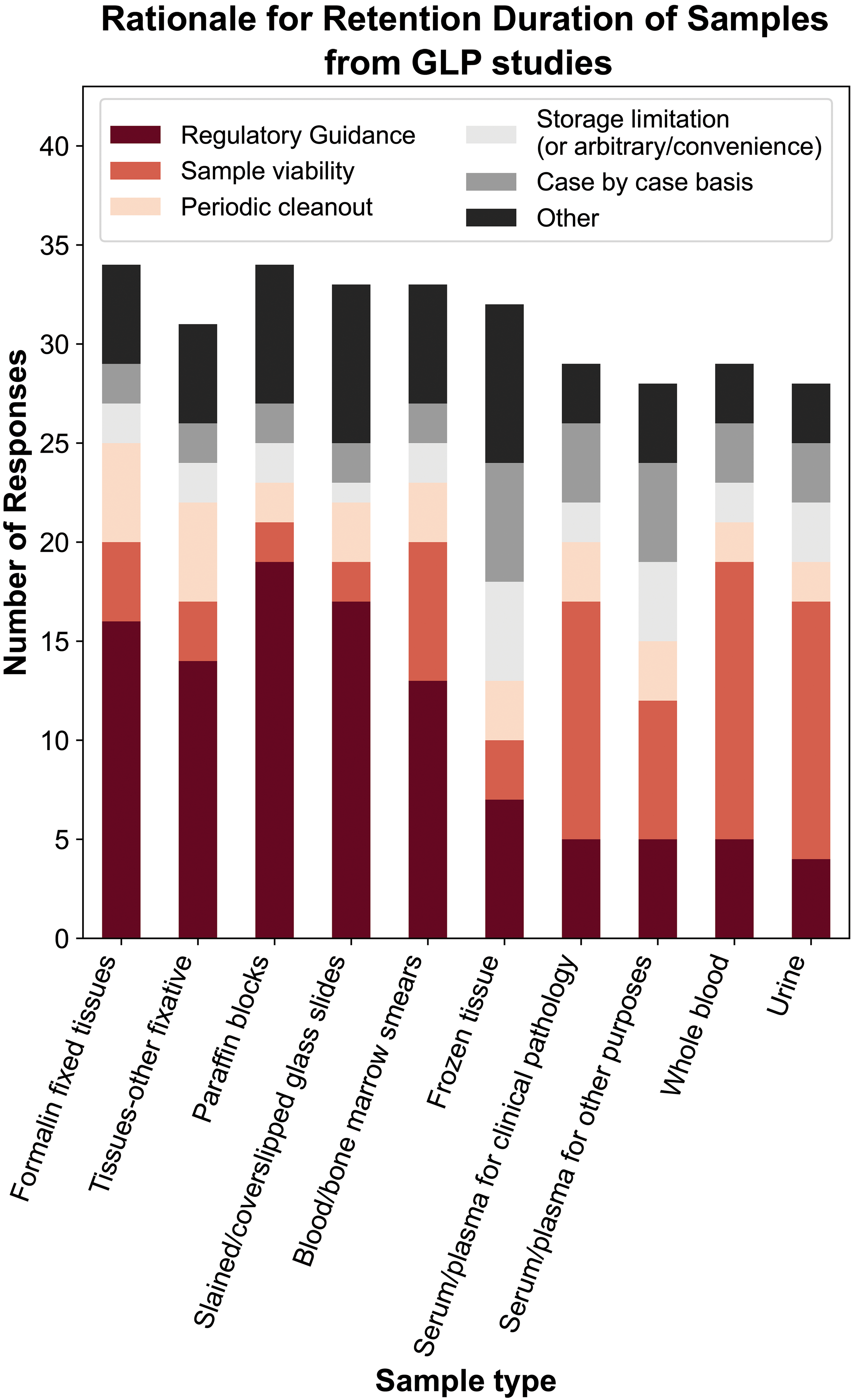
Rationale for retention duration of samples from nonclinical GLP toxicity studies. The most common reported rationale for retention duration of tissues, blocks, and histology slides was regulation and/or guidance, whereas sample stability was the most commonly cited for clinical pathology samples. For frozen tissues, there was variability in rationale, with site specific factors (eg, periodic cleanouts or storage limitations) identified more commonly. GLP indicate Good Laboratory Practice.
Sample stability was the most commonly cited rationale for the retention of fluid samples in GLP studies, including serum/plasma samples intended for clinical pathology (12 of 29, 41.3%), serum/plasma for biomarkers or other purposes (7 of 28, 25%), WB (14 of 29, 48.3%), and urine (13 of 27, 48.1%), whereas regulations and/or guidance and other rationales were infrequently cited (Figure 4). For frozen tissues, there was high variability in the rationale for retention (as for the reported retention duration itself), with several common responses reported at similar frequencies: case-by-case basis (8 of 31, 25.8%), regulations and/or guidance (7 of 31, 22.6%), arbitrary/convenience (6 of 31, 19.4%), and sample stability determination (4 of 31, 12.9%). Almost half of the CRO respondents (3 of 7, 42.9%) indicated deferral to client directives for disposal of this sample type.
Respondents were also asked to comment on storage costs, which were highly variable depending upon storage conditions (refrigerated, frozen, electronic data), sample size, space (by cubic foot), and other variables. Additional details on pricing were not provided, although it was mentioned that the cost of storage can be a motivating factor for sample disposal.
Survey Results: Institutional Efforts to Investigate Wet Tissue Sample Stability Duration
Although sample stability determination was not commonly used as the primary rationale for disposal of wet tissues, 15 (33.3%) of 45 respondents on this topic indicated that investigative work to determine the stability of wet tissues stored in 10% NBF had been performed at their institutions for various purposes. Investigative work was most commonly directed at determining sample stability for IHC after storage durations up to 20 years and was reported by 4 biotechnology/pharmaceutical company respondents and 2 CROs. Investigative work to determine stability of wet tissues for standard H&E assessment following storage for durations up to 30 years was also performed by 3 biotechnology/pharmaceutical company respondents and 2 CROs. Two biotechnology/pharmaceutical company respondents provided additional information for this question, with 1 indicating that wet tissue quality was deemed adequate for H&E histologic evaluation after up to 12 years of storage in 10% NBF. Another respondent stated that sample stability durations for stained histology slides and FFPE tissue blocks were determined to be up to 25 years and indefinitely, respectively. Other investigational work mentioned by individual respondents included quality evaluation of tissues by electron microscopy after storage in formalin and evaluation of transcriptomic probes, although the conclusions of these investigations were not disclosed.
Although several responses/comments regarding clinical pathology specimens included consideration of internal validation and stability testing during establishment of sample storage and reassessment practices, the current survey did not specifically request information related to investigative work performed to determine sample stability of clinical pathology samples. Current standards in validation for clinical pathology and biomarker methods commonly include testing sample stability under a variety of conditions (eg, room temperature [20-25 °C], refrigerated [3 to 4 °C], frozen [−20 to −70 °C], after ≥1 freeze thaw cycle, etc). 34 -36 Clinical pathology laboratories may perform various elements of this stability testing in house during method validation, particularly for routine clinical pathology analyses, and/or may refer to the literature for information on expected stability duration.
Survey Results: Experience Reaccessing Samples From Nonclinical Toxicity Studies
Forty-four responses were obtained regarding how frequently additional tissue sections from archived formalin-fixed wet tissues or FFPE blocks had been reaccessed to supplement the original histopathology analysis (ie, after the initial round of processing according to the study protocol), and most respondents responded “never” or “rarely” (Figure 5A). The most commonly cited reasons for reaccessing wet tissues were to further investigate endpoints in the original study, for example, by obtaining additional tissue sections for H&E staining, or to perform histochemical stains, IHC, or ISH (Figure 5B). For other activities performed with reaccessed wet tissues, 2 CRO respondents gave the example of evaluating potential additional target tissues or equivocal findings specifically in low or intermediate dose/exposure groups, and one indicated that this reanalysis was most frequently performed within 1 to 2 years of the initial study. Less commonly cited reasons for reaccessing wet tissues and/or FFPE blocks included molecular or ultrastructural pathology evaluations (eg, DNA methylation patterns, gene expression [RNA] analysis by RNA sequencing, electron microscopy, etc). Only one biotechnology/pharmaceutical company respondent cited a regulatory request to reexamine the tissue due to issues related to recording findings during the primary evaluation. Limited information was provided regarding the perceived value of some of these analyses and quality of the samples upon reaccession. However, 1 academic respondent cited DNA of poor quality in wet tissues upon extraction after prolonged storage (of unspecified duration) in formalin due to a high level of DNA degradation that resulted in inconsistent molecular results. The 2 respondents who commented on the value of reaccessing wet tissues for generating H&E sections for standard histology evaluation did not cite any quality concerns, except 1 CRO respondent who identified artifactual vacuolation in the brain. In addition, 1 government respondent noted that the DNA quality when reaccessed from archived FFPE blocks was variable (permissive for polymerase chain reaction amplification and Sanger sequencing but challenging for next-generation sequencing) but emphasized that DNA quality was nonetheless perceived to be better than from archived wet tissues.
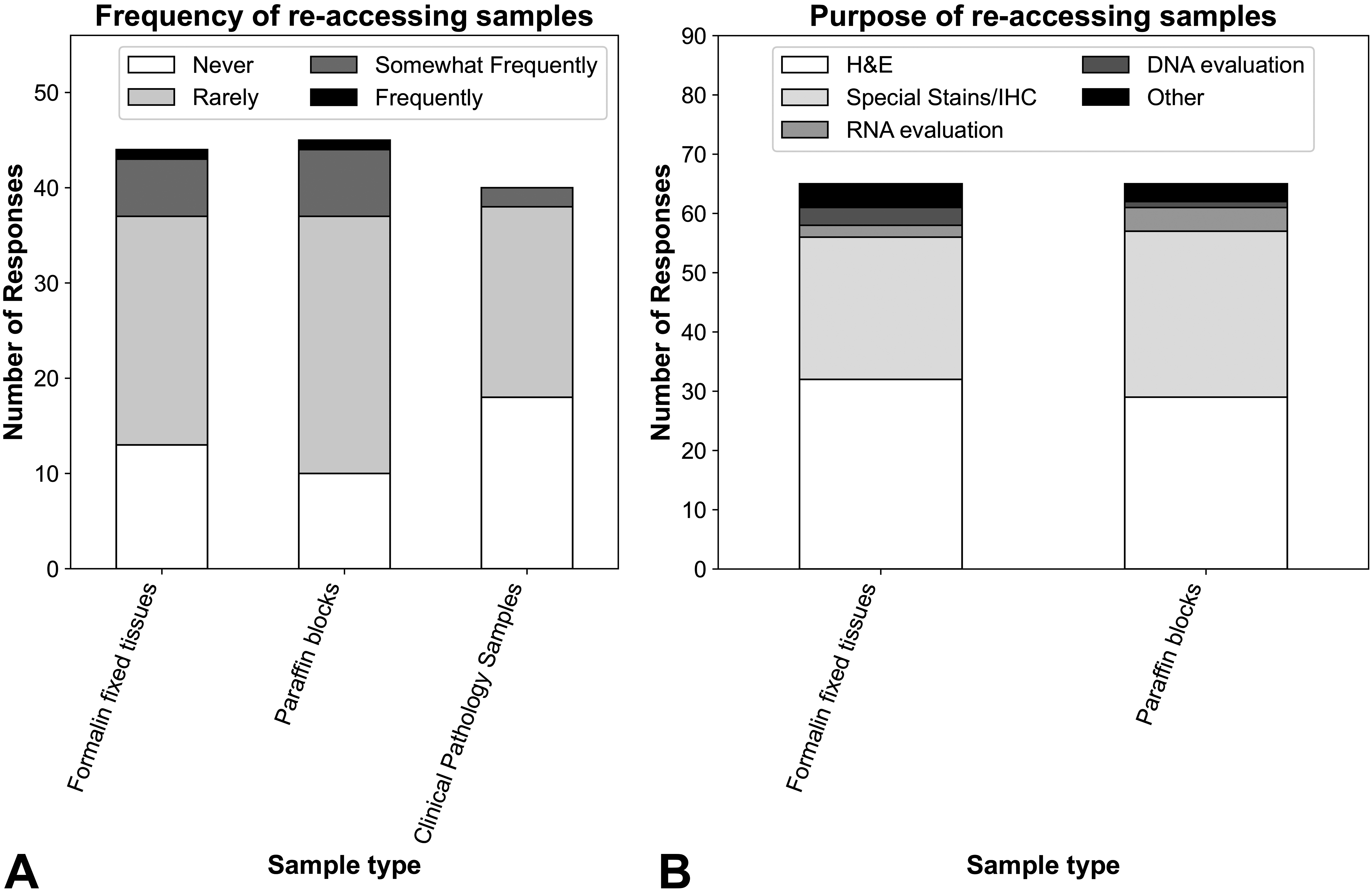
Reported experience reaccessing archived biologic samples from nonclinical toxicity studies. A, Across surveyed sample types, survey respondents most commonly indicated biologic samples were rarely or never reaccessed after initial analysis. B, When wet tissues or FFPE tissue blocks were reaccessed, the most common purpose was to obtain additional tissue sections for H&E staining to perform for special histochemical stains, IHC, or ISH. Molecular methods to evaluate RNA (eg, gene expression analysis by RNASeq, microarray, or RT-PCR) or DNA (eg, evaluation for mutations or methylation patterns) were also rarely performed. Clinical pathology samples were reaccessed too infrequently to identify patterns in the purpose for reaccessing them (data not shown). FFPE indicates formalin-fixed, paraffin-embedded; H&E, hematoxylin and eosin; IHC, immunohistochemistry; ISH, in situ hybridization; RT-PCR, reverse transcription polymerase chain reaction.
Reanalysis of archived clinical pathology samples to supplement the original study data also occurred “rarely to never” for the majority (38 of 40, 95%) of respondents. Individual responses detailing specific circumstances where reassessment had occurred were varied and included ancillary clinical chemistry tests, investigative/exploratory biomarker evaluation, hormone analysis, bone marrow/blood smear examination, and other analyses outside the scope of clinical pathology assessment (eg, receptor occupancy testing). Based on review of the relatively small number of supplemental written responses to this survey question, it is likely that some, if not many, of these analyses may have been pursued during the period prior to study finalization and not after sample archiving following completion of the study. However, information indicating the frequency of within-study reassessment (prior to study finalization) versus postfinalization analyses was not systematically captured by the survey.
Discussion
Summary of Sample Retention Information: Survey Results, Regulations and Guidance, Literature Data
The results of the survey indicate that most institutions have specific work practices in place to guide how long biologic specimens from nonclinical toxicity studies should be retained. However, in the current survey, there was high variability across respondents in the reported duration of those retention periods, which further varied by sample type. Regardless of the study type (GLP compliant or non-GLP; intended for regulatory filing or not) or relationship to product development stage, the reported retention durations often spanned the range of possible responses from “Discard at/before study finalization” to store “20+ years/indefinitely.” Although the majority of survey responses were received from biotechnology/pharmaceutical companies, trends in the reported retention duration generally appeared similar across institution types. Responses from academic and government research laboratories/core facilities suggested that conventions of the individual laboratory or region and/or space constraints rather than regulatory considerations determined the sample retention times at these types of institutions.
Most survey respondents indicated that the primary rationale guiding the retention period for wet tissues, FFPE tissue blocks, and histology slides was regulations and/or guidance. However, the variability in reported procedures from “Discard before/at study finalization” to store “20+years/indefinite” suggests uncertainty on how those regulations and guidances are interpreted and executed, a desire to be able to respond to global variability in guidelines, and/or a highly conservative response to the unlikely need to reaccess the sample. Practically speaking, most respondents indicated that they “rarely” or “never” reaccess these types of samples, and only 1 respondent indicated that a regulatory request prompted the review. For clinical pathology samples such as WB, serum/plasma, or urine, most survey respondents cited sample stability as the primary rationale for designating a specific retention period. Accordingly, most survey respondents reported discarding biological fluid samples at study finalization. Nevertheless, clinical pathology samples were reportedly stored for several years by some respondents, although these sample types were also “rarely” to “never” reaccessed.
Taken together, these results suggest that the generation of a universal set of standard sample retention practices would be beneficial to ensure biological samples are retained for as long as warranted without incurring unnecessary financial and resource management costs associated with indefinite storage. The standard should account for global regulations and guidance, sample stability, and the potential for reaccessing samples for each of the major sample and study types (wet tissues, FFPE blocks/glass histology slides, and clinical pathology samples from GLP, non-GLP, and investigative studies). Several global and regional regulations and guidance documents specify that the stability of wet tissues and other fragile samples should impact the duration of retention, but the criteria to determine adequate specimen quality are often undefined in these documents or leave room for differences in interpretation. Particularly for wet tissues, published literature on the stability after storage in 10% NBF to support retention decisions is limited. Tissue artifacts have been documented in some tissues (eg, brain) after as little as 1 year, whereas other tissues may remain useable for many years. 16- 18 However, establishing retention guidelines based on the documented appearance of artifacts in individual tissue types would not be logistically feasible even if detailed sample stability data were available to inform those criteria. Additionally, the extent to which pathologists can “evaluate through’’ these artifacts likely varies by pathologist experience, tissue type, and the histologic finding(s) being evaluated. Ideally, the duration of wet tissue retention should be uniform across all tissues and balanced between being short enough to ensure most tissues will still be of sufficient quality to allow microscopic evaluation but long enough to ensure that tissues are retained for any request for re-evaluation within a reasonable time frame. Conversely, FFPE tissue blocks and histology slides have been consistently determined to be stable for decades when stored under optimal conditions, so practical considerations in combination with regulations and guidance may be used to select a retention duration for these sample types. For tissue, blood, and bone marrow smear slides, decreased staining intensity is expected to occur over time and is likely to be variable depending on a number of factors; however, there is little available information regarding stability and rate of deterioration of these specimens as relates to standard histologic evaluation. For fluid clinical pathology samples, optimally each institution would establish stability under their own laboratory and storage conditions to supplement general guidance for retention duration (especially for common analytes), similar to the expectation that exists for analytical method validation studies.
Points to Consider for Retention Duration of Nonclinical Toxicity Study Samples
This manuscript integrates available data from current regulatory requirements, literature, and current practices and rationales provided by surveyed STP members to provide “points to consider” for storage duration of various sample types, particularly of wet tissues and clinical pathology samples that are more susceptible to degradation than tissues processed to FFPE tissue blocks or glass histology slides. These considerations are intended to meet regulatory expectations for sample retention and (in accordance with the “3Rs,” the principles of reducing, replacing, or refining the use of animals in testing) to prevent the need to run additional animal studies if samples are prematurely discarded, without necessitating indefinite storage of potentially degraded materials (Table 2). Although a single defined retention period for all sample types from all study types would be ideal, variability in applicable regulations and guidance and differences in sample characteristics dictate that each institution should review these points to consider and determine whether the retention durations presented for consideration in Table 2 meet their individual needs. However, in general, samples of test and reference items or specimens of all types may be discarded when the quality of the material no longer permits evaluation, with the expectation that storage conditions were optimal. It is good practice to refer to the storage devices’ history to determine equipment failures, power outages, moves, and so on that could possibly impact sample integrity. When samples of test and reference items or specimens are disposed of before the end of the required retention period, the reason for disposal should be justified and documented.
Proposed Retention Periods to Consider for Samples From Nonclinical Toxicity Studies.
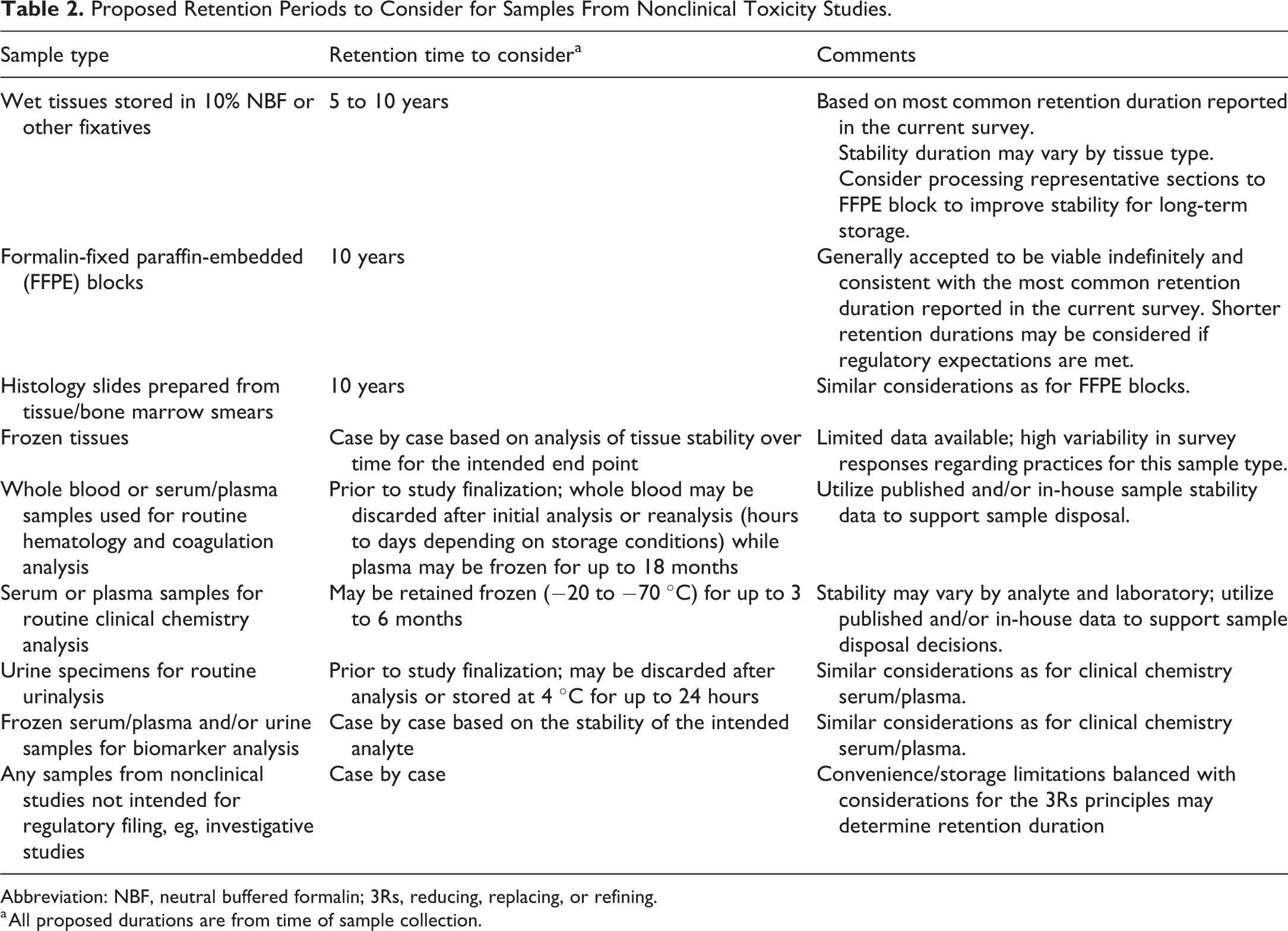
Abbreviation: NBF, neutral buffered formalin; 3Rs, reducing, replacing, or refining.
a All proposed durations are from time of sample collection.
Considerations for storage of formalin-fixed tissues, frozen tissues, FFPE blocks, and glass histology slides
A retention period of 5 to 10 years from the date of necropsy for tissues stored in 10% NBF may be considered to ensure the availability of high-quality tissue sections for evaluation and is consistent with current practices of the majority of survey respondents. Investigators should be aware that certain tissues (eg, brain, liver) may have reduced tissue quality after storage in 10% NBF for shorter durations than 5 years and may not be useable for all applications after 1 year, whereas others may remain stable for years to decades. 16 -18 Therefore, processing representative samples of all tissues at least to paraffin block as soon as feasible following the initial fixation period in 10% NBF may be desirable to support prolonged, temperature-controlled storage in the event reaccessing the sample is needed in the future. For frozen tissues, sample stability appears to be dependent on the specific analysis that the samples would undergo, and thus case-by-case analysis of tissue stability over time would be valuable to obtain objective data to guide retention practices.
Stained/cover slipped histology slides prepared from tissues and blood/bone marrow smears are also considered likely to remain stable for many years with proper storage, although staining intensity may decrease and/or issues with coverslips such as cracks or air bubbles may develop over time. Should additional tissue sections be needed (eg, for educational purposes, IHC, etc) or to replace original slides that have either been lost, broken, or have degraded in quality during storage, data from several studies indicate recuts from paraffin blocks provide material of acceptable quality even after decades when stored appropriately. 34 However, sample stability is not the only consideration for storage of FFPE tissue blocks and histology slides.Survey results indicated that the samples were rarely or never reaccessed, with only one instance of a regulatory request to revisit the histology findings due to incomplete data analysis in the initial study report cited by a respondent. Therefore, although most survey respondents retained these materials for >20 years/indefinitely, a shorter retention duration (eg, 10 years) may be considered. In particular, samples from non-GLP or investigative studies not intended for regulatory filing may be considered for earlier disposal, and storage space or cost limitations may play a larger role in determining when to discard these samples. Academic/government institutions may also be able to select shorter retention periods based on space considerations and perceptions regarding the likelihood of reaccessing the samples, particularly if studies are not intended to support regulatory filings. In the event that storage space and costs are not a consideration, institutions may prefer to retain paraffin blocks and slides from all studies for longer durations (ie, indefinitely) to ensure their availability if needed. Specifically, biologic samples from nonclinical studies supporting programs in active clinical development or with any potential regulatory issues may provide the highest likelihood of benefit from longer retention durations. Utilization of stored samples may prevent the need to repeat animal studies in some cases, in accordance with the 3Rs principle of reducing animal use. However, based on survey responses, this appears to be an infrequent occurrence.
Considerations for storage of clinical pathology fluid samples
For WB, serum/plasma, and urine, generating in-house sample stability data would provide the most objective data to determine appropriate retention times. Although not a replacement for collecting these data, some general considerations are as follows.
Fluid clinical pathology specimens are generally accepted to have a relatively short stability time frame. For example, WB specimens used for hematology analysis are often discarded after initial analysis within 24 hours of collection (or after any immediate reanalysis required for confirmation of results). 21,35 Although refrigeration (3-4 °C) may help to prolong the life span of WB samples not analyzed within a few hours of collection, storage-related artifacts may preclude meaningful evaluation for hematological investigations by 72 hours after collection, and freezing these samples is generally not advised. Similarly, prompt analysis of fresh plasma samples for routine coagulation testing and urine specimens for routine urinalysis (typically stored refrigerated and analyzed within 24 hours of collection) may aid in limiting storage-related artifacts; however, frozen long-term stability (−24 to −74 °C for >18 months) has been demonstrated for many coagulation proteins. 24,25 Serum or plasma may remain stable for analysis of many standard clinical chemistry analytes if frozen (−20 to −70 °C) for up to 3 to 6 months. 31 However, in the increasingly common scenario in which serum/plasma and/or urine are retained for potential future analysis (eg, of renal injury biomarkers or other nonstandard analytes), frozen samples can be potentially retained for years and still be useable for analysis of certain analytes. 33,35 -37 However, as analyte stability may vary substantially, the duration of retention for these purposes should take into consideration the types of biomarkers and the documented stability of those biomarkers based on in-house or published data.
Conclusions
The primary goal of this manuscript was to provide points to consider for retention time of formalin-fixed wet tissues and other biological sample types from nonclinical toxicology studies; these points were based on a survey of current practices and consideration for regulations, guidance, and available published data. Regulatory authorities provide general requirements for specimen retention in GLP-compliant studies, allowing for flexibility in retention time to ensure specimen quality and meaningful evaluation. However, in practice, there is wide variability in specimen retention times, and published data are limited to support retention decisions (for wet tissues in particular). To the working group’s knowledge, the survey-based data and points to consider reported herein provide the most current and comprehensive information on current sample retention practices for nonclinical toxicology samples and can be referenced during the development and/or review of institutional sample retention practices. Moving forward, institutions with internal data on sample stability are encouraged to publish their results in order to further support decision-making and refine sample retention guidelines based on objective information, particularly for wet and frozen tissues and serum/plasma biomarkers where the largest data gaps remain.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211049156 - Scientific and Regulatory Policy Committee Points to Consider: Biological Sample Retention From Nonclinical Toxicity Studies
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211049156 for Scientific and Regulatory Policy Committee Points to Consider: Biological Sample Retention From Nonclinical Toxicity Studies by Carole E. Harbison, Adam D. Aulbach, Bindu M. Bennet, Molly H. Boyle, Mary E. Carsillo, Torrie A. Crabbs, Natalie D. Keirstead, Krista M. D. La Perle, Arun R. Pandiri, Ahmed M. Shoieb and William D. Siska in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-pdf-1-tpx-10.1177_01926233211049156 - Scientific and Regulatory Policy Committee Points to Consider: Biological Sample Retention From Nonclinical Toxicity Studies
Supplemental Material, sj-pdf-1-tpx-10.1177_01926233211049156 for Scientific and Regulatory Policy Committee Points to Consider: Biological Sample Retention From Nonclinical Toxicity Studies by Carole E. Harbison, Adam D. Aulbach, Bindu M. Bennet, Molly H. Boyle, Mary E. Carsillo, Torrie A. Crabbs, Natalie D. Keirstead, Krista M. D. La Perle, Arun R. Pandiri, Ahmed M. Shoieb and William D. Siska in Toxicologic Pathology
Footnotes
Acknowledgment
The authors would like to thank Albert Swiston (Merck & Co., Inc., Kenilworth, NJ) for assistance with manuscript figure preparation.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
