Abstract
Many pharmaceutical companies have recently elected to stop maintaining good laboratory practices (GLP) status of their R&D sites. Similar discussions have also been engaged in the (agro)chemical industry. This opinion paper examines the pros and cons of maintaining facility GLP status for the purposes of performing the pathology interpretation or peer reviews of GLP studies internally. The toxicologic pathologist provides gross and histomorphologic evaluation and interpretation of nonclinical exploratory and regulatory studies during drug and (agro)chemical development. This assessment significantly contributes to human risk assessment by characterizing the toxicological profile and discussing the human relevance of the findings. The toxicologic pathologist is a key contributor to compound development decisions (advancement or termination) and in the development of de-risking strategies for backup compounds, thus playing a critical role in helping to reduce the late attrition of drugs and chemicals. Maintaining GLP compliance is often perceived as a costly and cumbersome process; a common and short-term strategy to reduce the costs is to outsource regulatory toxicity studies. However, there are significant advantages in maintaining the GLP status for toxicologic pathology activities in-house including the sustainable retention of internal pathology expertise that has maintained the necessary training needed to manage GLP studies.
This is an opinion article submitted to the Toxicologic Pathology Forum. It represents the views of the authors. It does not constitute an official position of the Society of Toxicologic Pathology, British Society of Toxicological Pathology, or European Society of Toxicologic Pathology, and the views expressed might not reflect the best practices recommended by these Societies. This article should not be construed to represent the policies, positions, or opinions of their respective organizations, employers, or regulatory agencies.
Introduction
Several pharmaceutical companies have recently chosen to stop maintaining the good laboratory practices (GLP) status of their R&D sites and now outsource all GLP nonclinical safety studies to contract research organizations (CROs). Similar approaches have also been considered within the (agro)chemical industry. This article reflects the concerns of the authors, who are experienced pathologists, about this trend; it offers perspectives regarding current business practices and highlights the advantages of maintaining or establishing in-house GLP status from a pathologist’s perspective.
Regulatory authorities across the globe require that a series of nonclinical in vivo studies of new drugs, medical devices, food additives, and chemicals be conducted to characterize potential safety liabilities prior to testing in humans or addressing human risks. Given that the data and decisions made from these nonclinical studies are critical factors to ensure human safety, there is both an expectation and regulatory requirement that they are conducted using solid scientific principles and with rigorous attention to quality control and quality assurance (QA). The GLP framework, today a global standard, was introduced by the United States Food and Drug Administration decades ago, implementing regulations that focused on sound scientific methodology, data integrity, and documentation to establish consistent, reliable global standards of nonclinical safety testing.
The maintenance of GLP compliance is sometimes perceived to be a cumbersome process associated with a certain level of bureaucracy. For example, the degree of documentation required for GLP studies necessitates dedicated administrative structures that require more time, budget, and resources. This can be particularly the case when establishing a GLP-compliant facility for the first time. Some organizations lacking dedicated support units may not achieve the critical mass to adequately address GLP compliance. Nevertheless, ensuring solid scientific principles, including experimental repeatability, data accuracy, and documentation already requires resources, and ongoing maintenance of GLP status can often be less resource-intensive.
Current Situation in Pharmaceutical and (Agro)Chemical Companies
The outsourcing strategy of nonclinical programs differs from one company to another, but the authors identified 4 general business models that have emerged over time. The schematic classification and an overview of the GLP scope in these 4 models are given in Table 1. 1
In-House GLP Status in the 4 Models of Toxicology and Pathology Outsourcing Strategy.
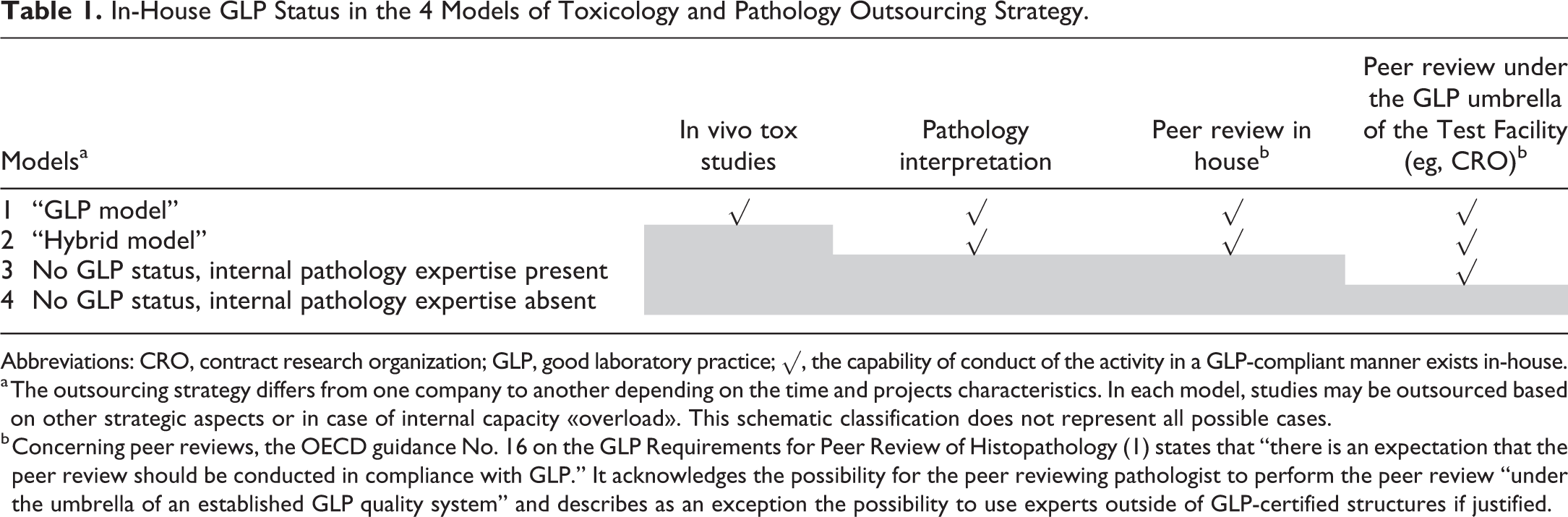
Abbreviations: CRO, contract research organization; GLP, good laboratory practice; √, the capability of conduct of the activity in a GLP-compliant manner exists in-house.
a The outsourcing strategy differs from one company to another depending on the time and projects characteristics. In each model, studies may be outsourced based on other strategic aspects or in case of internal capacity «overload». This schematic classification does not represent all possible cases.
b Concerning peer reviews, the OECD guidance No. 16 on the GLP Requirements for Peer Review of Histopathology (1) states that “there is an expectation that the peer review should be conducted in compliance with GLP.” It acknowledges the possibility for the peer reviewing pathologist to perform the peer review “under the umbrella of an established GLP quality system” and describes as an exception the possibility to use experts outside of GLP-certified structures if justified.
The Role of Toxicologic Pathologists in the Pharmaceutical and (Agro) Chemical Industry
The role of a toxicologic pathologist in drug or (agro)chemical development starts early on by supporting the establishment of suitable animal models, the planning and execution/evaluation of early pharmacological studies, and defining potential target liabilities. Toxicologic pathologists are major contributors to translational discussions providing input on differences and similarities between species. Pathological evaluation of exploratory and pivotal studies provides key information helping to characterize an overall toxicity profile including an in-depth understanding of pathophysiology and potential relevance to humans, all of which can be critical for the assessment of human risk. 2
Reducing the late attrition rate of drugs and chemicals is of major importance for both the pharmaceutical and (agro)chemical industries, and the unique expertise of toxicologic pathologists is essential to this objective as they may largely contribute to the early decision of project termination and help develop de-risking strategies for backup compounds.
Pharmaceutical Companies
Pammolli et al 3 provided a comprehensive analysis of productivity of R&D organizations within the pharmaceutical industry comparing data from more than 45,000 projects between 1990 and 2017. These data demonstrated significant improvements over time in the overall efficiency of the selection process, with a more stringent screening of drug candidates and viable projects at early stages of the development. Improved validation of drug targets in terms of mechanism of action and of preclinical toxicity liabilities in early stages of drug discovery, including the use of in silico analyses, in vitro screens, and in vivo preliminary studies, was pivotal to ensuring the best drug candidates proceeded into development. However, the early stage toxicity studies may not be sufficient to predict every issue, and in vitro screening has limitations.
An internal survey conducted by Pfizer, covering 2012 to 2019, showed that approximately 80% of the reasons for attrition due to nonclinical observations were related to pathology issues. Although this survey illustrates the cases where attrition occurred and expertise of staff pathologists greatly contributed to the decision of project termination, toxicologic pathology expertise can also prove instrumental to establish the pathogenesis, relevance, and adversity of test item-related findings, de-risk toxicity issues and thus prevent the attrition of beneficial therapies or products. In addition, histopathological characterization of toxicologic findings can also provide useful information to develop the de-risking strategy for backup compounds. Internal pathologists also assist in defining the benefit/risk ratio of a project internally and in the context of summary regulatory documents.
(Agro)Chemical Companies
Toxicologic pathologists in the (agro)chemical industry have similar roles and contribute to similar types of studies when compared with their colleagues in the biopharmaceutical industry. The regulatory requirements for agrochemical compounds are extremely stringent, which can be explained by the different intended targets compared to pharmaceutical compounds. Agrochemical active ingredients are often intended to act as powerful pesticides; however, they also need to have an acceptable adverse profile for both environmental and human toxicity concerns as exemplified by the cutoff criteria applied for endocrine properties. 4 As a result, compound attrition rates in today’s agrochemical industry are generally comparable to that of pharmaceuticals. Adverse outcomes and morphological observations in toxicity studies still frequently lead to attrition despite robust in silico and in vitro early toxicity assessments. Missed or subtle adverse outcome pathways (unknown or unpredicted) are fortunately often detected early by the toxicologic pathologist. It is unfortunate that those data leading to the halt of agrochemical products during the research and development process are rarely published. 5
Advantages in Keeping In-House Expertise in Toxicologic Pathology
The strategic advantages of having the internal capability and knowledge to perform histopathology of all study types (including GLP-compliant subchronic and chronic) in-house studies are (1) preservation of relevant internal expertise/experience, (2) increased flexibility with regard to timelines, processes, and follow-up activities in case of critical findings or acceleration of projects, (3) taking maximum advantage of internal expertise with the histomorphologic evaluations conducted by pathologists with an understanding of the program and potential target tissues, and (4) attracting and training early career pathologists and developing/maintaining in house toxicologic pathology expertise. In-house pathologists often gain a deep understanding of the target, the mode of action and the associated biology, and therapeutic modality as they are often involved from the very early phases of a project. Furthermore, the pathologist may have prior experience with other similar compounds/targets or members of a drug class, which creates invaluable historical knowledge. This knowledge can facilitate consistency in diagnostic terminology, interpretation across different studies with the same test item or with backup molecules (directly on target-related, off-target, secondary, background findings), the evolution of such findings over time, the pathogenesis, and the safety assessment for regulatory documents. In-house pathology is a means to ensure a more flexible approach to project timelines and unforeseen additional investigative work. The direct communication and interaction with project management teams can speed up the decision-making and allow better alignment on project priorities and provide an avenue to quickly address specific questions. For example, fit-for-purpose approaches can be leveraged, such as expedited processing of potential target tissues and unscheduled animals and prioritizing that evaluation in advance of the remaining protocol tissues. Recuts, special stains, or additional samples are easily accessible in-house on short notice. Critical findings can be quickly discussed internally within the group of experienced pathologists and toxicologists, leading to continuous scientific exchange and strategic decisions. This can even provide the opportunity to amend ongoing studies to address concerns as they arise in real time. It is common practice for companies that have maintained GLP status to keep accelerated programs in-house for this purpose and obtain a competitive advantage. In addition, such programs generate the strongest internal engagement. Having both regulatory and experimental toxicologic pathology in-house allows fast and efficient communication, throughout the drug development process, including harmonization of nomenclature in reports over time. Furthermore, for studies under extraordinarily high timeline or strategic pressures (eg, Covid vaccine), direct communication lines between study pathologist/pathology management/portfolio and R&D management are pivotal to guide strategies in real time (eg, trigger backup decision or specific analyses). Decision can be made more quickly and efficiently saving time, resources, and money. The ability to provide a large spectrum of pathology opportunities can be an essential factor helping to attract early career pathologists to work for the pharmaceutical or (agro)chemical industry.
Although in-house pathology expertise is considered as a critical resource for pharmaceutical and (agro)chemical company success and the need for this unique expertise is well-acknowledged within these industries, the way in which toxicologic pathologists are trained and the efforts required to develop and maintain their unique expertise is generally not fully appreciated outside the toxicologic pathology community.
Expertise in toxicologic pathology is not limited to histology slide evaluation, as the pathologist is often best qualified to critically put findings into perspective, integrate them with all the other toxicology study data and discuss them in light of the pathophysiology and mechanisms of action of the tested drug. To do so, pathologists are medically trained with special expertise/training in their discipline and often receive additional training as scientists (eg, PhDs, fellowships). Throughout their career, they learn from literature and attend continuing education and professional symposia. They expand and maintain their expertise mainly through conducting histomorphologic assessments and by interpreting, integrating, and communicating toxicology study data and playing an integral role in project teams developing drugs/chemicals. Professional education of early career colleagues relies on the continuous scientific exchange with more senior colleagues but, like a physician who needs to see a lot of patients to develop his diagnostic skills or a surgeon who needs to perform a lot of different surgeries, pathologists need to evaluate a lot of tissues, including nonremarkable tissues and experience many different cases both of which help to reinforce and expand their skills. A pathologist needs to be exposed directly to a wide variety of normal and abnormal tissues to build a real bench experience and mental library of normal inter-animal variation, age- and sex-related background findings, procedure-related pathology, and induced primary and secondary findings that can be observed in the wide range of tissues evaluated. Regular slide evaluations increase a pathologist’s acumen to detect subtle changes. Performing peer reviews also requires significant experience which can only be gained by repeated primary evaluation under the guidance of experienced pathologists. But it does not bring the same level of experience as only a portion of the slides are examined, and the level of responsibility is different (understanding/reconstructing vs deciding/designing).
The benefit of working under an in-house GLP umbrella is not, for a pathologist, related directly to the GLP status itself, but the consequences of this status exposing the pathologist to a wider diversity of pathology findings: • Good laboratory practice–compliant general toxicity studies are usually of longer duration: they may show more test item-related findings either off or on-target as some findings may develop only after a certain treatment duration, especially for small molecules. These studies also demonstrate more age-related or spontaneous findings. • Good laboratory practice–compliant general toxicity studies typically involve more animals, and the tissue sampling is extensive involving a long list of organs/tissues; this results in the pathologist having more slides and a larger tissue panel to examine as compared to non-GLP studies. By acting as study pathologist for in-house non-GLP studies, the pathologist can get early insight into many toxicology findings but the evaluation of more comprehensive GLP toxicity studies will generally provide more opportunity to see spontaneous as well as induced findings.
Overall, the perspective of having a balanced project team membership versus solely slide evaluation responsibilities contributes to the attractiveness of careers in pharmaceutical or (agro)chemical companies when competing for talent acquisition and to obtaining the most value from hired pathologists. This is also a key element for talent retention.
Contract Research Laboratories as Partners
Good laboratory practice–compliant toxicity studies may be perceived as essentially “routine” studies, in contrast to early investigative studies; therefore, some sponsors may choose to outsource their GLP studies in order to focus more in-house resources on the less routine investigative activities for strategic reasons. However, the authors agree that although the parameters to be evaluated in GLP toxicity studies are largely framed by international guidelines and are therefore, mainly standard, the toxicity findings cannot be fully anticipated and there is no such thing as a “routine” toxicity study. Any study result may become critical to identify issues of importance for the fate of the drug. Therefore, GLP toxicity studies of longer duration equally deserve the fullest attention including true partnership between the sponsor company and the CRO. In addition, some study types (eg, biodistribution studies for gene therapy) that are performed under GLP status can be quite complex with the inclusion of an extensive list of non-GLP investigative analyses (eg, vector and transgene detection in tissues) that result in increased administrative and logistic burden and cost to a point where these are often more efficiently managed at the sponsor’s site rather than as a multisite study.
In the authors’ experience, the main arguments used to justify a subcontracting strategy are often related to cost savings. However, a side-by-side comparison with internal costs is not always easy to perform. The resources spent on monitoring adequately outsourced studies, pathology peer reviews, specimen shipment and chain of custody management, report reviews, and follow-up meetings are often overlooked and not included in the side-by-side comparison.
Additionally, the currently observed tendency of consolidation of CROs by acquisitions leads to only a few internationally recognized providers in the market and reduced business competition among CROs. Although the administrative effort for the customer may be diminished for the placement of studies, the dependency on a certain preferred provider is much higher. Enhanced competition for time slots may result in less opportunity to negotiate and thus in increased costs, longer lead times prior to study start, or delays in completion of projects.
Excellent toxicologic pathology expertise is found at CROs, but CRO pathologists are naturally not completely integrated at a project level or as familiar with subsequent regulatory steps as would be the case for in-house pathologists. There is, therefore, a risk for sponsor companies to become fully dependent on CRO study interpretations as this could have an impact on the wider integration and interpretation of pathology findings at the project level making it more challenging to design proper and efficient risk assessment strategies. Performing in house studies also allows companies to build and capitalize on their own internal historical knowledge.
Future Perspective: Will Digital Pathology Change the Paradigm?
It is the conviction of the authors that the undergoing digitalization of the toxicologic pathology field does not impact the advantages of an in-house GLP status. In the future, the histopathological evaluation may be performed using glass or digital slides, and the prerequisites to consider the use of digital pathology for primary reading or peer review have been discussed. 6,7 As was the case in the field of radiology, digitalization of toxicologic pathology is likely to advance pathology expertise, allowing real time or much faster discussion of findings with peers and/or machine learning augmentation for diagnoses. Via the rapidity and ease of exchange, digital pathology capabilities can increase the flexibility and balance between in-house and external pathology, independent of the site of the test facility. 8 The implementation of digital pathology and the forthcoming progresses around artificial intelligence could provide flexibility, but these tools do not aim to replace the unique expertise of toxicologic pathologists. The basic need of pathologists is to be exposed to a variety of findings, the in-house benefits of integrating findings into a development project and the advantages of internal operational flexibility obtained from in-house GLP capacity will remain the same.
Conclusion
This article reflects the discussions and opinions of the authors, who are experienced pathologists from a broad range of pharma and (agro)chemical companies. It highlights the advantages of maintaining in-house GLP status from a pathologist’s perspective. The authors would like to encourage each company to stress test their current strategy.
The short-term vision of cost reduction, when abandoning GLP status, should be counter-balanced by a long-term vision of investment in a very specific scientific expertise that remains mandatory for successful product development. The cost of reestablishing GLP status in the future, once given up, may turn out to be prohibitive as the GLP expertise may be lost.
Considering the current and future shortage of pathologists on the market, attracting and maintaining an in-house toxicologic pathology expertise will be essential for pharmaceutical and (agro)chemical companies.
Overall, despite the cost, resources, and administrative investments, there are considerable advantages in maintaining the GLP status for toxicologic pathology activities in-house as it ensures internal expertise in both GLP compliance and pathology as well as potentially shorter timelines, flexibility, and more integrated scientific input and comprehensive oversight from early investigative studies to late-stage nonclinical studies.
Footnotes
Acknowledgments
The authors would like to thank Catherine Botteron, Richard Haworth, and Kuno Wuersch, who contributed actively to the first discussions on the topic.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
