Abstract
Glomerular filtration rate is the gold-standard method for assessment of renal function but is rarely performed in routine toxicity studies. Standard serum biomarkers of renal function are insensitive and become elevated only with significant loss of organ function. Symmetric dimethylarginine (SDMA) is a ubiquitous analyte that is freely filtered by the glomerulus and can be detected in serum. It has shown utility for the detection of renal injury in dogs and cats in clinical veterinary practice, but the potential utility of SDMA to detect renal injury in preclinical species or toxicity studies has not been thoroughly investigated. We utilized a well-characterized glomerular toxicant, puromycin aminonucleoside, to induce podocyte injury and subsequent proteinuria in young male Sprague-Dawley rats. At the end of 1 or 2 weeks, blood, urine, and kidney tissue were collected for analysis. One week following a single 50 mg/kg dose, urea nitrogen, creatinine, and albumin mean values were within historical control ranges, while SDMA was increased. Glomerular changes in these animals included periodic acid–Schiff positive globules within podocytes, podocyte hypertrophy by light microscopy, and podocyte degeneration with effacement of foot processes by electron microscopy (EM). Taken together, our data indicate that SDMA may be a useful biomarker for early detection of glomerular toxicities in rats.
Introduction
The kidney is a common target of drug-induced injury. 1 Measurement of glomerular filtration rate (GFR) is the gold-standard method for assessment of kidney excretory function, however direct measurement of GFR is uncommonly performed in routine toxicologic assessments. 2 Instead, surrogate markers of GFR including serum creatinine (sCr) and blood urea nitrogen (BUN) are most commonly utilized. These analytes lack sensitivity, detecting only significant decreases in renal function (30% or 50% loss of glomerular filtration 3 ), and therefore have limited value in short-term toxicology studies. 4 To this end, urinary and serum biomarkers of kidney injury have been intensely investigated in recent years, and several novel, segment-specific candidates such as kidney injury marker 1 and neutrophil gelatinase-associated lipocalin (also known as lipocalin 2) have been qualified. 5 -7 An ideal biomarker of renal function would be translationally relevant, more sensitive, readily available, and stable in serum and/or plasma, and relatively unaffected by nonrenal factors such as nutritional status, body muscle mass, or preanalytical variables.
Symmetric dimethylarginine (SDMA) is a modified amino acid originating from intracellular proteins that undergo posttranslational modification and methylation followed by proteolysis. 8 The small size and positive charge of SDMA allow free filtration by the glomerulus in the kidney, with renal excretion being at least 90%. 8,9 Symmetric dimethylarginine is highly stable in serum and plasma, and its measurement is unaffected by lipid, hemoglobin, or bilirubin levels in the sample. 10 Furthermore, SDMA levels are not affected by age, diet, or muscle mass. 11 -13 Given these characteristics of SDMA as a ubiquitous analyte freely filtered by the glomerulus, it is a potentially valuable biomarker of glomerular filtration and overall renal function. Indeed, measurement of SDMA has been added as a recommended analyte for kidney disease staging in dogs and cats. 2
Puromycin aminonucleoside (PAN) is an antibiotic that is well-known to cause glomerular injury and has been studied as a model of minimal change disease in humans. 14 -16 Puromycin aminonucleoside causes nephrotic syndrome in rats, characterized by severe proteinuria originating from the glomerulus. 17,18 As early as 24 hours following a single dose of PAN in rats, ultrastructural injury to glomerular epithelial cells (podocytes) is characterized by blebbing, increased pinocytotic activity, accumulation of cytoplasmic vacuoles, loss and effacement of foot processes, and microvillous transformation, which correlate to the degree of albuminuria. 19 -23 Via light microscopy, the morphologic alterations are subtle and may feature minimally thickened glomerular basement membrane (GBM), swelling of podocytes, or scattered synechiae. 21 These morphologic changes correspond to damage and alteration of the glomerular size barrier and to GBM matrix proteins. 21,24 PAN-mediated damage to the glomerular filtration apparatus results in leakage of protein into the urine within 1 to 2 weeks of a single administration. 21 Secondary to protein leakage through the glomerular filtration barrier, tubular injury to proximal tubules is characterized by protein resorption droplets, loss of brush border, luminal dilation, accumulation of protein casts, and even epithelial cell swelling and rupture. 21,22 In order to investigate the potential utility of SDMA as a biomarker in the setting of primary glomerular injury in an acute toxicity model, male Sprague-Dawley rats were dosed with PAN once weekly for one or two weeks. Similar to previous descriptions of the toxicologic findings with PAN, we noted severe proteinuria with modest glomerular changes characterized by minimal to mild podocyte hypertrophy and degeneration with foot process effacement and microvillus transformation. Interestingly, these morphologic changes were accompanied by small increases in SDMA, while mean sCr and BUN concentrations remained within historical control data (HCD) ranges. These data suggest that SDMA could be a useful and sensitive tool in detecting certain acute glomerular toxicities in rats.
Materials and Methods
Animals
All procedures were approved by AbbVie’s Institutional Animal Care and Use Committee and carried out in American Association for Accreditation of Laboratory Animal Care-accredited facilities. Twenty male Sprague-Dawley (Crl: CD(SD)) rats between the ages of 56 and 58 days were sourced from Charles River Laboratories. Animals were housed two or three per cage for at least two days following receipt for acclimation and then randomly assigned to four dosing groups. During the study, animals were group housed two to three per cage in plastic, solid bottom hanging cages with bedding, equipped with feeders and automatic watering systems. Tecniplast metabolic cages were used for urine collections. Ad libitum certified Rodent Chow was provided and ad libitum reverse-osmosis chlorinated water (nonacidified) was provided. At the end of study, animals were euthanized via exsanguination under isoflurane anesthesia and necropsied.
Toxicity Study
Puromycin aminonucleoside was administered subcutaneously to male Sprague-Dawley rats (5 rats per group) at 50 (groups 1 and 2) or 150 (groups 3 and 4) mg/kg/dose at a dose volume of 5 mL/kg. Groups 1 and 3 were administered a single dose on day 1 and necropsied on day 7. Groups 2 and 4 were administered two weekly doses on day 1 and day 8. Group 2 was necropsied on day 14. Due to clinical decline, group 4 was necropsied on days 9 and 10 (Table 1). Animals were monitored 2 to 4 hours postdose and once daily throughout the study period. Body weight was measured at baseline, on day 1, and on day 7 for all groups, and on day 14 for group 2. Urine was collected over a 24-hour period at baseline and on days 6 to 7 for all groups. Blood was collected by cardiac puncture at necropsy for clinical chemistry and SDMA analysis. At necropsy, kidney weights were recorded.
Puromycin Aminonucleoside Toxicity Study Design.
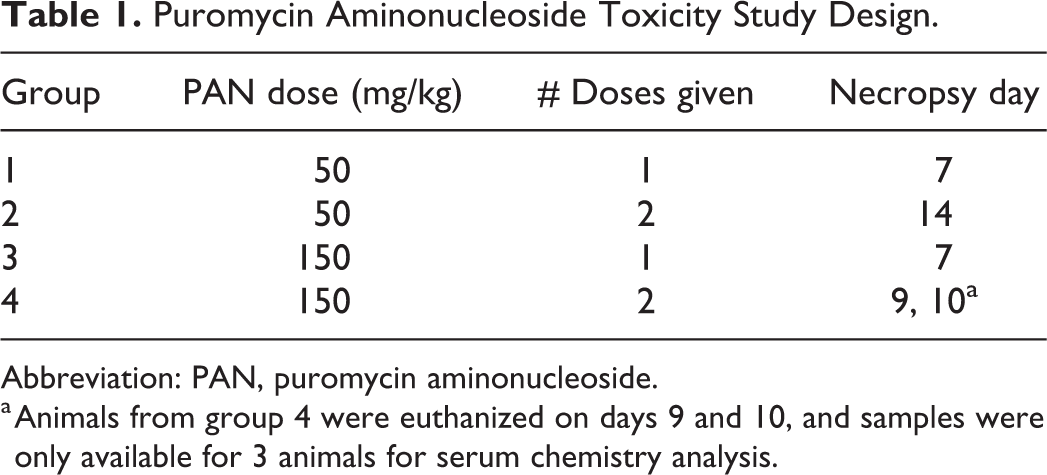
Abbreviation: PAN, puromycin aminonucleoside.
a Animals from group 4 were euthanized on days 9 and 10, and samples were only available for 3 animals for serum chemistry analysis.
Histologic Staining and Ultrastructural Analysis
Tissue sections of the right kidney, liver, heart, skeletal muscle, and pancreas were collected into 10% neutral buffered formalin, processed and paraffin-embedded in a routine manner, and stained with hematoxylin and eosin (H&E) for histologic evaluation. Kidney tissue from group 1 only was transferred to Karnovsky fixative and processed for transmission electron microscopy in a standard manner. 25
Sections of kidney from all groups were additionally stained with periodic acid–Schiff (PAS), and immunohistochemistry for nephrin was performed. The PAS staining was carried out without diastase digestion in the standard manner. Nephrin immunohistochemistry was carried out in a Bond Rx automated stainer (Leica Biosystems). Rabbit Anti-Nephrin monoclonal antibody (ab216341, Abcam) was used at 0.15 µg/mL with antigen retrieval using the citrate-based pH 6.0 BOND Epitope Retrieval Solution 1 (AR9961, Leica Biosystems) for 20 minutes. 3,3′-Diaminobenzidine (DAB) detection was done using Bond Polymer Refine detection (DS9800, Leica Biosystems).
Pathologic Evaluation
Sections of kidney were evaluated microscopically by board-certified veterinary anatomic pathologists (RK and LH). Comprehensive histologic evaluation of renal tissue resulted in application of qualitative 5-category scoring (minimal, mild, moderate, marked, severe).
For quantitative analysis of nephrin immunohistochemistry, slides were scanned at 40× to .mrxs (MIRAX-compatible virtual slide format) files on the Pannoramic 250 (3DHISTECH, Ltd). Analysis was conducted in QuPath 0.2.0-m2, an open-source, whole-slide image analysis software platform. 26 Preprocessing was performed to calibrate stain vectors. A grid was applied and three 4 mm 2 regions of interest (ROI) were delineated at the cranial pole cortex, antihilar cortex, and caudal pole cortex. Within each ROI, 10 glomeruli were manually annotated. The mean glomerular area did not differ between treatment groups (data not shown). The detect positive staining feature was applied to quantify positive pixels (representative of glomerular nephrin positivity) using a single DAB threshold (settings: downsample factor of 2.0, Gaussian sigma of 1 µm, hematoxylin threshold of 0.04 OD units, DAB threshold of 0.3 OD units). Mean glomerular positive percentage of stained pixels was calculated per animal (30 glomeruli/kidney) and per group. Data are represented as means ± 1 standard deviation, and statistically significant differences are indicated by asterisks.
Sample Collection and Analysis
Serum BUN, sCr, serum albumin, urine creatinine, urine protein, and urine microalbumin concentrations were measured using the Abbott Architect c16000. Serum SDMA concentration was measured using the IDEXX Catalyst One platform per manufacturer instructions. Historical control data (HCD) for BUN (7-15 mg/dL), sCr (0.3-0.7 mg/dL), and albumin (2.7-3.5 g/dL) were derived from an internal historical control database of male CRL: CD(SD) rats aged 8 to 10 weeks (N = 93-99) and are presented as mean ± 2 standard deviations. Historical control data for SDMA were derived from internal analysis of age/sex matched vehicle control male CRL: CD(SD) rats aged 8 to 9 weeks (N = 45) and are presented as mean ± 2 standard deviations (2-13 µg/dL, mean of 7.6 µg/dL; Supplemental Figure 1). These SDMA HCD data are consistent with external reference interval (RI) data on the Catalyst platform (5-11 µg/dL) published from IDEXX data. 27
Statistical Analysis
One-way analysis of variance was performed on nephrin immunohistochemical (IHC) data. Unpaired t test was performed on the SDMA HCD. A P value <.05 was considered statistically significant.
Results
Toxicologically Significant Findings Following Administration of PAN
Administration of PAN in rats for 14 days at 50 mg/kg resulted in the clinical observation of rough hair coat beginning on day 8. Animals dosed with 150 mg/kg had rough hair coats in both groups 3 and 4 beginning on day 2, while group 4 animals also had decreased activity, thin body appearance, and animals were in lateral recumbency with labored breathing beginning on day 9. Based on removal criteria for humane reasons, all animals in group 4 were euthanized on days 9 and 10 of study. Additionally, at 150 mg/kg PAN, animals had decreased body weight gain in groups 3 and 4 and decreased body weight in group 4 (Supplemental Figures 2 and 3). Poor clinical condition and weight loss in group 4 were attributed to compromised renal function. At necropsy, the kidneys in individual group 4 animals were pale and increased in size (Supplemental Figure 4).
Administration of PAN Results in Azotemia, Increased SDMA, and Proteinuria in Rats
Blood collected at necropsy was analyzed for BUN, sCr, albumin, and SDMA in order to evaluate renal excretory function (Table 2 and Figure 1). For group 1 animals, mean BUN (Figure 1A) and albumin (Figure 1B) were within the HCD, and four of five group 1 animals had sCr concentrations within the range of HCD values (Figure 1C). Group 1 serum SDMA concentration was at the high end or above the range of HCD or RI values for the three surviving animals (mean 13 ± 1.6 µg/dL; Figure 1D). For groups 3 and 4, mean sCr was elevated (mean 0.74 ± 0.06 and 2.7 ± 0.52 mg/dL, respectively). For groups 2 through 4, mean albumin was decreased (to 1.03 ± 0.05 g/dL) and both mean BUN and SDMA concentrations were elevated (to 312 ± 29.8 mg/dL and 80.7 ± 3.8 µg/dL, respectively). Collectively, the serum chemistry data suggest that higher doses of PAN, repeat dosing, and longer duration of dosing result in increasing renal injury. Within the context of the well-characterized PAN-induced kidney injury model, SDMA increased after one administration of 50 mg/kg PAN before consistent sCr increases and albumin decreases occurred. Further, SDMA performed at least as well as BUN, as concentrations from individual animals exceeded the range of historical control values (Supplemental Figure 1), while mean BUN was within the historical control range.
Mean Values of Serum Chemistry Parameters in Rats Dosed With PAN.
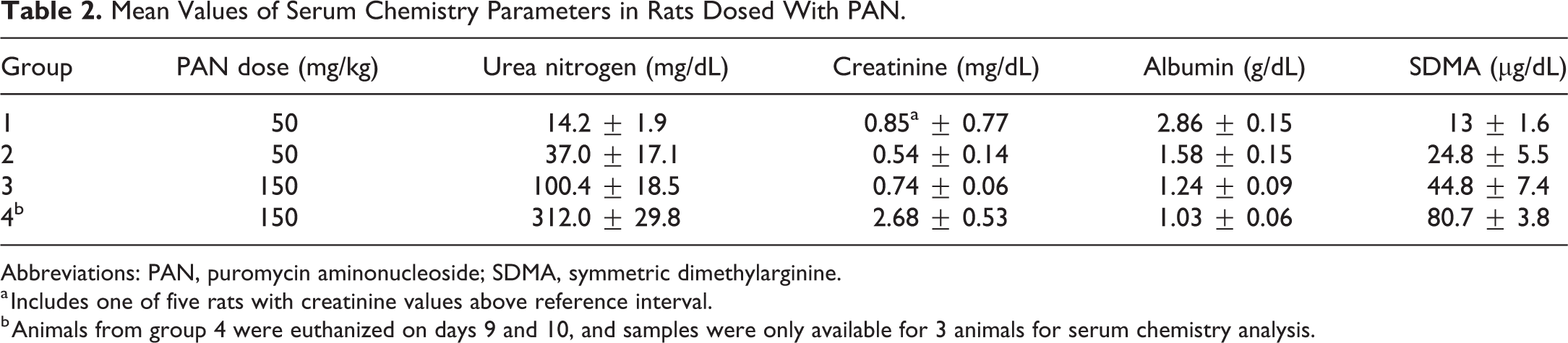
Abbreviations: PAN, puromycin aminonucleoside; SDMA, symmetric dimethylarginine.
a Includes one of five rats with creatinine values above reference interval.
b Animals from group 4 were euthanized on days 9 and 10, and samples were only available for 3 animals for serum chemistry analysis.
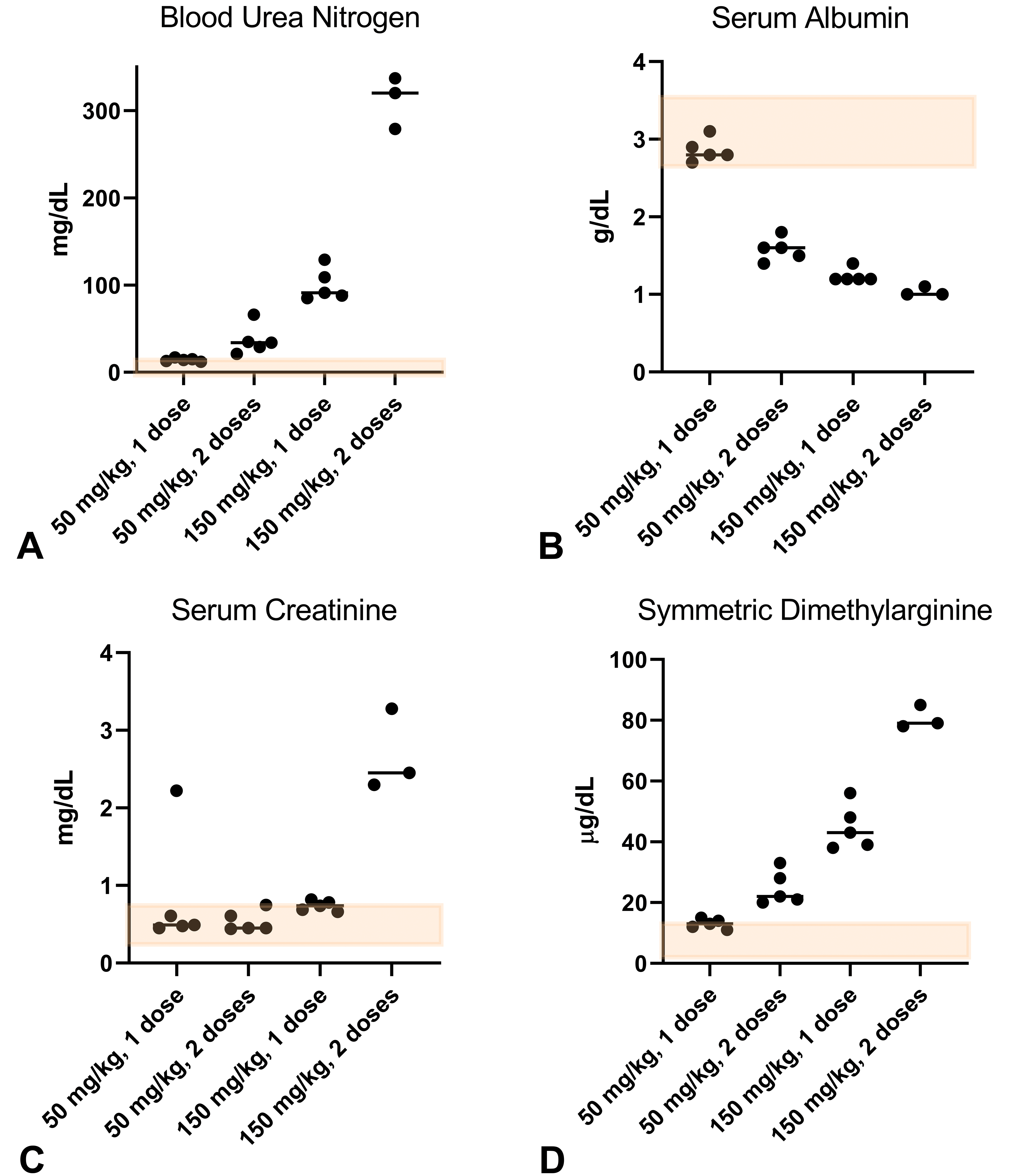
(A) Blood urea nitrogen increases with dose and duration of PAN dosing. (B) Serum albumin decreases with dose and duration. (C) Serum creatinine and (D) symmetric dimethylarginine increase with dose and duration. Bars represent mean values. Transparent orange boxes represent historical control data (BUN 7-15 mg/dL; sCr 0.3-0.7 mg/dL; Alb 2.7-3.5 g/dL; SDMA 2-13 µg/dL). Alb indicates albumin; BUN, blood urea nitrogen; sCr, serum creatinine; SDMA, symmetric dimethylarginine.
In urine samples collected on days 6/7 (Table 3), animals from groups 3 and 4 had urine protein concentrations exceeding the upper limit of quantification (>2000 mg/dL) and urine protein: creatinine (UPC) ratios of ≥9.57, which exceeded the normal published range. 28 On days 6/7, urine microalbumin was notably higher in animals administered 150 mg/kg PAN as compared to animals administered 50 mg/kg PAN. By day 14, urine microalbumin for animals administered two doses of 50 mg/kg PAN was notably higher than day 7 values, suggesting that the proteinuria worsens over time (data not shown).
Day 6/7 Mean Values of Urine Chemistry Parameters in Rats Dosed With PAN.
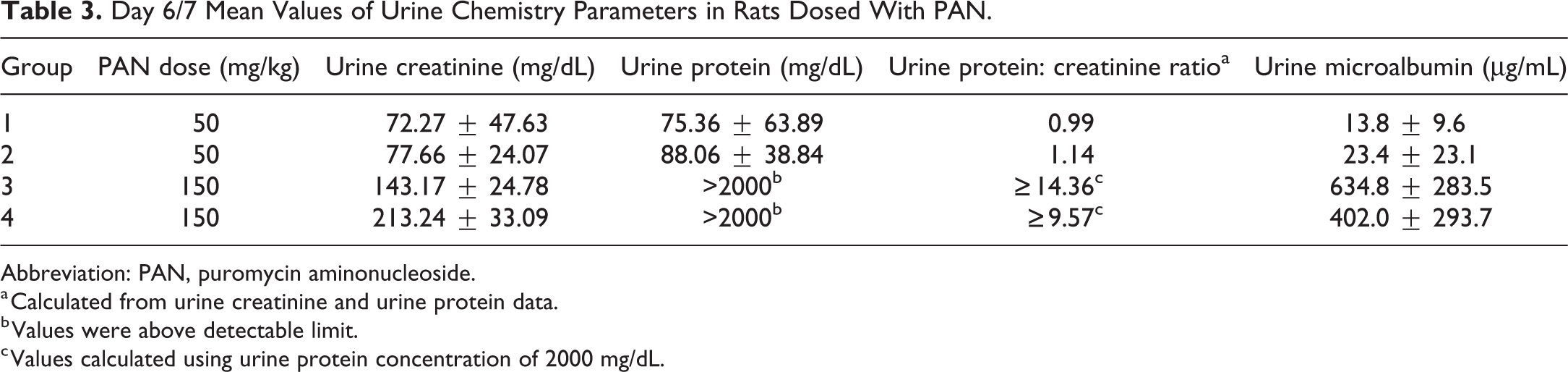
Abbreviation: PAN, puromycin aminonucleoside.
a Calculated from urine creatinine and urine protein data.
b Values were above detectable limit.
c Values calculated using urine protein concentration of 2000 mg/dL.
Administration of PAN Results in Tubular Protein and Glomerular Changes in Rats
Evaluation of kidney tissue by light microscopy resulted in podocyte hypertrophy, proteinaceous fluid in tubular lumena, proximal tubular epithelial degeneration/regeneration, and proximal tubular dilation (Figure 2). These findings were each scored on a 1 to 5 scale of minimal, mild, moderate, marked, and severe for every animal (Table 4). Podocyte hypertrophy was observed to a minimal degree in groups 1 through 3 and mild in group 4 animals. Tubular luminal protein was mild in groups 2 and 3 and moderate in group 4 animals. Group 4 animals also had moderate tubular dilation. Tubular degeneration and regeneration was of mild severity in group 2 animals and was minimal in groups 3 and 4.
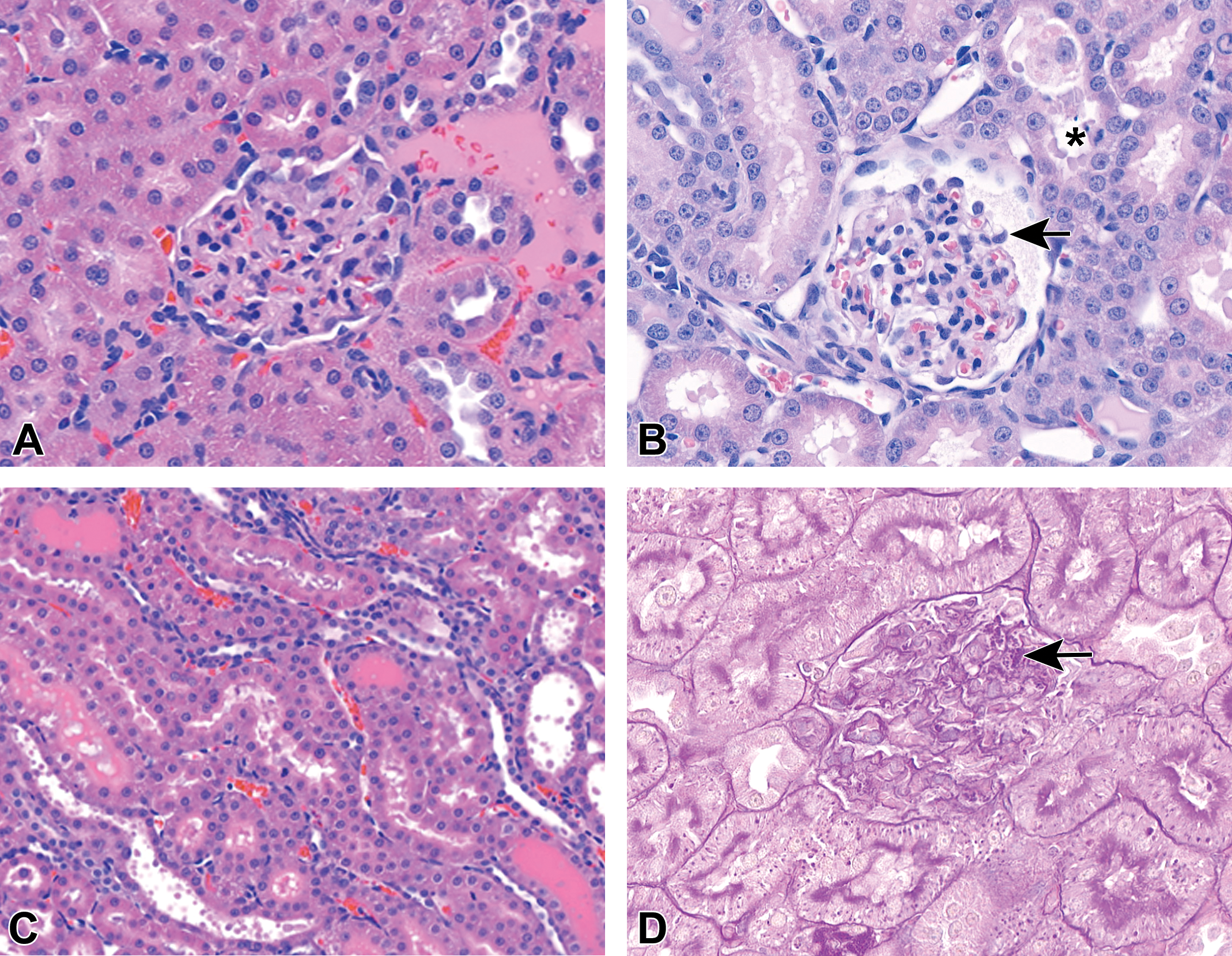
(A) Representative glomeruli from group 1 and (B) group 4. Note the increased size of podocytes with dose and duration as well as vacuolation of podocytes (arrow). Also note multifocal tubular epithelial degeneration (asterisk). (C) Tubular degeneration with loss of brush border, attenuated epithelium, and luminal protein in group 3. H&E. (D) Representative glomerulus from group 1 animal. Note protein resorption droplets in glomerular tuft (arrow). PAS. H&E indicates hematoxylin and eosin; PAS, periodic acid–Schiff.
Severity of Histologic Diagnoses in Kidney of Rats Administered PAN.
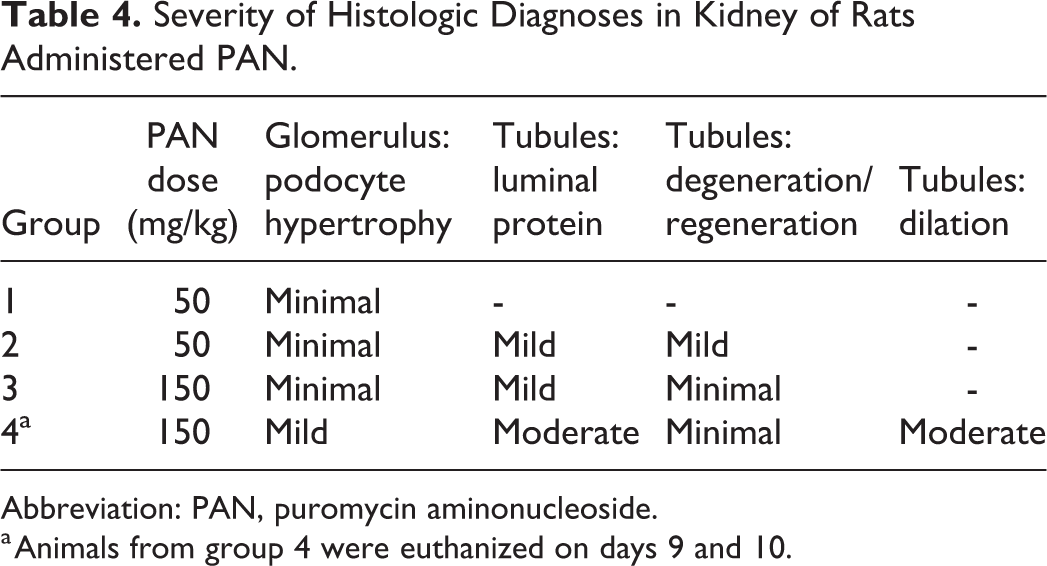
Abbreviation: PAN, puromycin aminonucleoside.
a Animals from group 4 were euthanized on days 9 and 10.
Evaluation of PAS-stained kidney sections revealed PAS+ globules present in the cytoplasm of podocytes in one animal from group 1 (this animal is the one outlier for sCr level) and in all animals from groups 2 through 4. From animals in groups 2 through 4 having tubules containing luminal protein, there were also large PAS+ intracytoplasmic droplets in tubular epithelium. These were interpreted as protein-resorption droplets (Figure 2D).
Nephrin is an adhesion protein and component of the slit diaphragm of glomerular podocytes, the expression of which has been shown to decrease in proteinuric and glomerular diseases. 29,30 Nephrin staining highlighted increasing size of podocytes with increasing dose of PAN and duration of exposure. Additionally, subjectively decreased intensity of staining in groups 3 and 4 was interpreted to be consistent with podocyte foot process degeneration (Figure 3). Quantification of nephrin IHC staining demonstrated a dose-dependent decrease in glomerular nephrin, indicated by reduced DAB pixel area, which reached significance in group 4 compared to groups 1 (P =.0029) and 2 (P =.0098; Figure 3C).
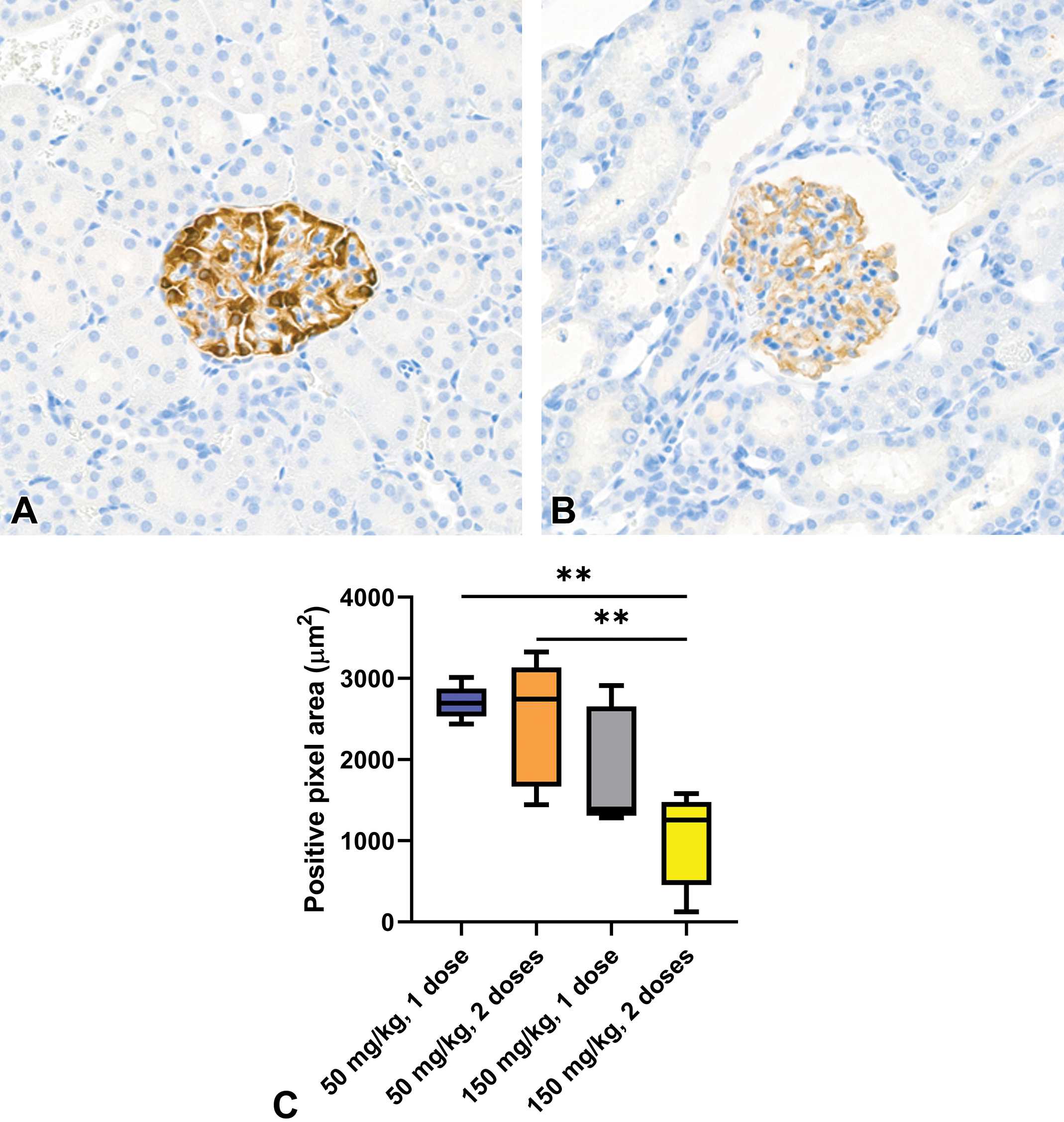
(A) Representative glomeruli from group 1 and (B) group 4. Note the decrease in intensity of DAB staining and irregularity of staining in group 4. Nephrin IHC. (C) Quantification of nephrin IHC. 50 mg/kg, 1 dose versus 150 mg/kg, 2 doses, P = .0029; 50 mg/kg, 2 doses versus 150 mg/kg, 2 doses, P = .0098. DAB indicates 3,3′-diaminobenzidine; IHC, immunohistochemical.
Two glomeruli for each group 1 animal were additionally examined by electron microscopy. Ultrastructural diagnoses of podocytes included accumulation of electron-dense cytoplasmic globules (interpreted as protein and correlating to PAS+ globules by light microscopy), cellular degeneration of podocytes characterized by accumulation of cytoplasmic vacuoles and foot process swelling and effacement with multifocal microvillus transformation (correlating to nephrin staining changes by light microscopy; Figure 4). Thus, our pathologic findings following PAN dosing in rats are consistent with published reports.
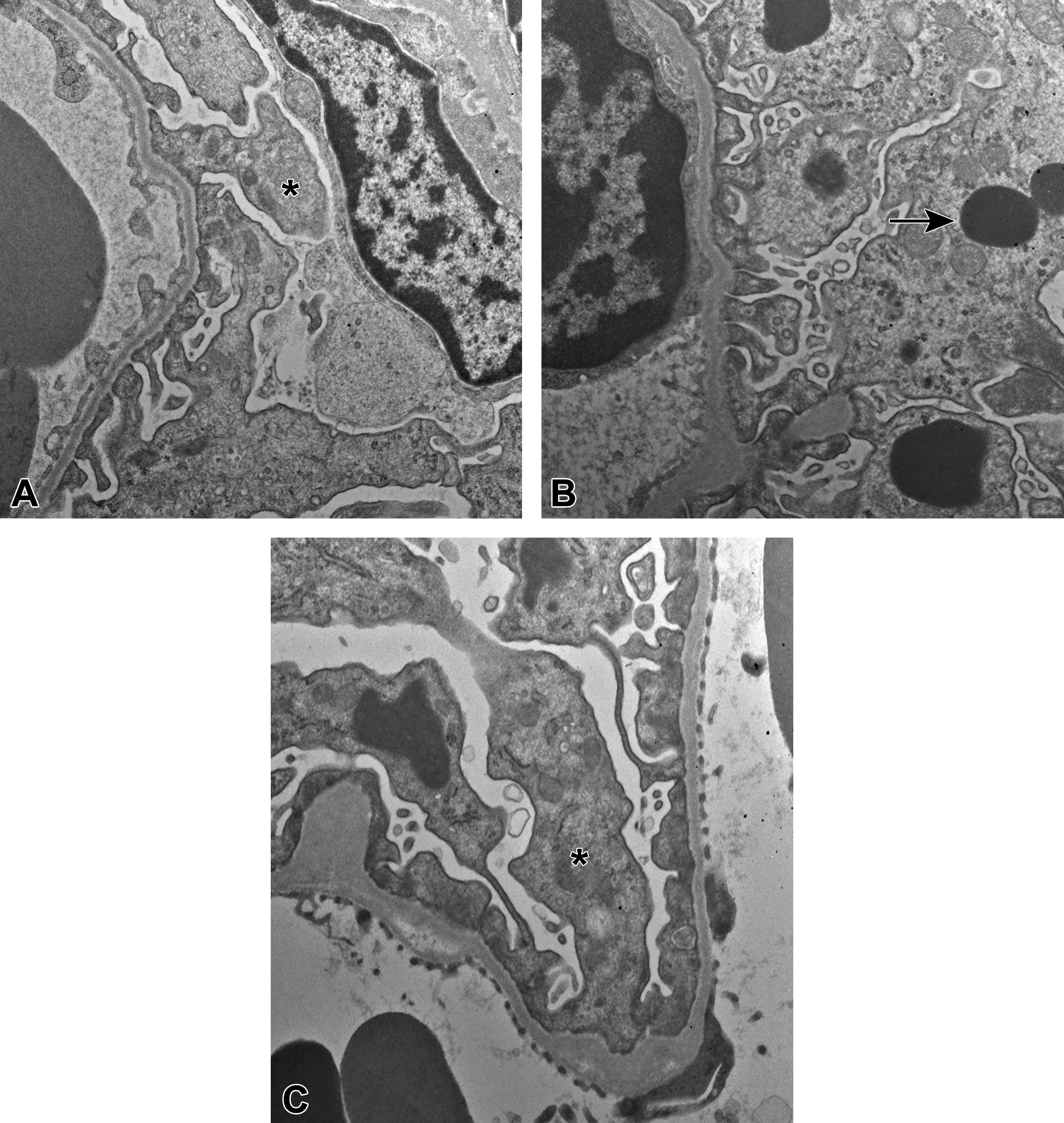
Representative electron microscopy images of glomerular basement membrane and podocyte foot processes from group 1 animals. Note effacement of foot processes (asterisks), podocyte cellular degeneration, and electron dense globules interpreted as protein resorption droplets (arrow).
Discussion
The goal of this pilot exploratory toxicology study was to determine the response of the novel biomarker SDMA as well as standard end points of renal injury following administration of a reference nephrotoxicant. This study used the PAN model to understand time- and dose-dependent changes in SDMA relative to alterations in traditional indicators of renal injury such as BUN, creatinine, and proteinuria. Results of this study would shed light on the ability of SDMA to be a valuable biomarker for glomerular damage-induced renal toxicity in rats by outperforming BUN and/or creatinine. Animals administered a single dose of 50 mg/kg PAN and terminated after 7 days had no significant clinical signs or gross necropsy findings. Levels of standard biomarkers of renal injury (BUN, sCr, serum albumin, and albuminuria) were generally within HCD ranges, while SDMA values were elevated with improved temporal sensitivity over creatinine. Histologically, there were no significant changes in glomeruli or in the tubulointerstitial compartment in H&E-stained sections; findings were limited to minimally subjectively increased size of podocytes in some glomeruli. Importantly, group 1 animals displayed ultrastructurally evident glomerular damage that correlated with the increases in SDMA, while there was no correlation between the electron microscopy findings and other parameters of kidney damage measured.
Across groups, when evaluating the kidney by PAS staining, protein resorption droplets were apparent within podocytes in many glomeruli and within affected tubules. The proposed pathogenesis is PAN-mediated damage to the glomerular filtration apparatus, allowing larger molecular weight proteins (such as albumin) to pass into the ultrafiltrate. The amount of albumin in the ultrafiltrate subsequently overwhelms the resorption capacity of the tubular epithelium, leading to aggregation of protein in protein resorption droplets which are detectable by PAS staining. There are recent studies to suggest that endocytosis by glomerular podocytes themselves also plays an important role in protein regulation, consistent with our finding of presumptive protein resorption droplets in the podocyte cytoplasm. 31,32 Protein-induced tubular damage likely contributed significantly to highly elevated levels of BUN, sCr, and SDMA in groups 2 through 4 in our study.
In group 1, by nephrin IHC, few podocytes were subjectively enlarged. This effect was more pronounced in groups 2 through 4. Additionally, DAB immunolabeling of nephrin was significantly reduced in group 4, as shown by quantification of glomerular DAB-stained area, suggesting decreased expression and/or altered distribution or morphology of the protein and/or potential loss of podocytes. In a study of nephrin expression in glomerulonephritis models, a patchy and less intense staining pattern was reported in PAN-treated rat glomeruli that was not apparent with other rat models of glomerular injury. 33 A downregulation of nephrin expression in PAN-treated rats was confirmed with reverse transcription polymerase chain reaction. 34,35 Despite minimal changes detected by light microscopy, there was evidence of podocyte injury ultrastructurally including foot process effacement. These findings are consistent with the reported ultrastructural lesions induced by PAN in rats and reinforces its use as a model for minimal change disease and the importance of electron microscopy for evaluation of glomerular injury. 19 -23,25
In our study, increases in SDMA generally mirrored increases in sCr and BUN. In humans with renal disease, SDMA level correlated highly with inulin clearance (a direct measure of GFR), creatinine clearance, as well as sCr. 36 The SDMA analysis by liquid chromatography-mass spectrometry has been validated by IDEXX in dogs, cats, horses, and rats. 27,37,38 In dogs and cats, SDMA levels increased earlier in the progression of spontaneous chronic kidney disease, when GFR was decreased as little as 40%. 2 In a colony of dogs with X-linked hereditary nephropathy, a genetic condition affecting type IV collagen in the glomerular basement membrane leading to robust proteinuria, SDMA increased during progression of preclinical disease to end-stage disease and strongly correlated with sCr and decreased GFR as measured by iohexol clearance. 10 In that study, SDMA identified a decrease in GFR of as little as 20%. As a result of the utility of SDMA to predict earlier phases of decreased renal function in dogs and cats, its detection was added to the International Renal Interest Society’s guidelines to stage kidney disease in dogs and cats. 2 In rats, partial or total nephrectomy has been shown to result in increased SDMA which correlated with creatinine clearance, sCr, and BUN. 39,40
The limitations of our investigative study include lack of concurrent control group, use of HCD for comparison of SDMA concentrations, and the use of a published reference interval for UPC ratio. Additionally, inclusion of end points such as specific gravity and urine sediment analysis would have provided additional context around interpretation of SDMA results. As a key goal of the study was to understand whether SDMA provided more sensitive detection of renal injury compared with standard end points for assessing renal injury such as sCr and BUN concentrations, a direct comparison among treatment groups was determined to be sufficient. Additionally, due to extensive historical control databases for Sprague-Dawley rats and in the spirit of the 3Rs (Replacement, Refinement, and Reduction), 41 an interpretation of elevation in these biomarkers could be made without concurrent controls. As for SDMA, IDEXX recently presented an extensive investigation into SDMA concentrations in normal Sprague-Dawley rats. These published values were comparable to our internal HCD, and the same analytical approach was used in this study as in the IDEXX investigation. 27
Our study demonstrates an acute glomerular toxicity that is detected by increases in SDMA but not BUN, sCr, or UPC. Furthermore, the PAN-induced toxicity at 50 mg/kg for 7 days was subtle such that special stains, IHC, and EM were needed to appropriately characterize the morphologic changes. Thus, SDMA may be a useful and sensitive biomarker for detecting some acute glomerular toxicities in rats.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211045341 - Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats
Supplemental Material, sj-docx-1-tpx-10.1177_01926233211045341 for Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats by Rebecca Kohnken, Lauren Himmel, Michael Logan, Richard Peterson, Sabyasachi Biswas, Christina Dunn and Bruce LeRoy in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-1-tpx-10.1177_01926233211045341 - Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats
Supplemental Material, sj-tif-1-tpx-10.1177_01926233211045341 for Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats by Rebecca Kohnken, Lauren Himmel, Michael Logan, Richard Peterson, Sabyasachi Biswas, Christina Dunn and Bruce LeRoy in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-2-tpx-10.1177_01926233211045341 - Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats
Supplemental Material, sj-tif-2-tpx-10.1177_01926233211045341 for Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats by Rebecca Kohnken, Lauren Himmel, Michael Logan, Richard Peterson, Sabyasachi Biswas, Christina Dunn and Bruce LeRoy in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-3-tpx-10.1177_01926233211045341 - Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats
Supplemental Material, sj-tif-3-tpx-10.1177_01926233211045341 for Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats by Rebecca Kohnken, Lauren Himmel, Michael Logan, Richard Peterson, Sabyasachi Biswas, Christina Dunn and Bruce LeRoy in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-4-tpx-10.1177_01926233211045341 - Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats
Supplemental Material, sj-tif-4-tpx-10.1177_01926233211045341 for Symmetric Dimethylarginine Is a Sensitive Biomarker of Glomerular Injury in Rats by Rebecca Kohnken, Lauren Himmel, Michael Logan, Richard Peterson, Sabyasachi Biswas, Christina Dunn and Bruce LeRoy in Toxicologic Pathology
Footnotes
Acknowledgments
The authors gratefully acknowledge the clinical pathology laboratory technicians, animal care staff, and investigative toxicology pathology in vivo team for their assistance with carrying out this study, in particular Jie Lai-Zhang at AbbVie.
Declaration of Conflicting Interests
The author(s) declared the following potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article: Rebecca Kohnken, Lauren Himmel, Michael Logan, Richard Peterson, Sabyasachi Biswas, Christina Dunn, and Bruce LeRoy are employees of AbbVie and may own AbbVie stock. AbbVie sponsored and funded the study; contributed to the design; participated in the collection, analysis, and interpretation of data, and in writing, reviewing, and approval of the final publication.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by AbbVie.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
