Abstract
Differences were examined between male and female Sprague-Dawley rats in the response of 16 urinary biomarkers (measured using several assay platforms) to renal injury produced by gentamicin administered subcutaneously for 10 days at a dosage of 75 mg/kg. Urinary biomarkers expressed as fold difference from contemporaneous controls and renal histopathology were assessed after 3 and 10 doses. On day 4, minimal proximal tubular changes were observed microscopically in all males but no females; on day 11, more extensive and more severe injury was observed to a similar extent in all animals of both sexes. Modest increases (maximum 5-fold) in all urinary biomarkers (except epidermal growth factor [EGF], which was decreased) on day 4 and marked elevations (maximum 271-fold) on day 11 were seen consistently in both sexes. However, the magnitude of the increases differed between the sexes. On day 4, despite the lack of tubular injury, many biomarkers were more elevated in females than males but this rarely led to statistically significant sex differences; only 2 biomarkers (β2-microglobulin and total protein) showed a greater increase in males than females in line with the histopathology. On day 11, there were many more biomarkers that showed a statistically significant difference between the sexes in fold change with treatment; in line with the results on day 4, the majority of biomarkers were more increased in females than males. It remains unresolved if sex differences in the magnitude of biomarker response at injury threshold would lead to any difference in diagnostic interpretation between the sexes. These data highlight the need for publication of more studies using animals of both sexes to fully explore the influence of sex on the diagnostic performance of the novel biomarkers.
Keywords
Introduction
In order to minimize the occurrence of drug-associated nephrotoxicity, it is necessary either to prevent new nephrotoxic drugs from entering the market or to detect and manage nephrotoxicity efficiently when use of a drug with nephrotoxic liability is mandated by medical need (Bonventre et al. 2010). Serum indicators of whole kidney function, such as serum creatinine (sCr) and blood urea nitrogen (BUN), have been regarded for decades as the standard measurements for detection of kidney injury and are still widely used to monitor renal function and to detect drug-induced nephrotoxicity. The limitations of relying on elevated sCr and BUN to monitor impairment of renal function have been well documented (Bonventre et al. 2010; Frangogiannis 2012; Slocum, Heung, and Pennathur 2012). In addition to the delay in appearance of a rise in sCr and BUN due to the functional reserve of the kidney, interpretation can be complicated by the influence of extra-renal factors on their production and clearance. Thus, in view of the limitations of these two markers, novel renal safety biomarkers are sought to identify and monitor renal injury with high specificity and sensitivity (Vaidya et al. 2010).
In recent years, extensive effort has been directed toward the discovery and qualification of novel renal safety biomarkers that could be used to predict and monitor drug-associated nephrotoxicity. The Nephrotoxicity Working Group of the Critical Path Institute Predictive Safety Testing Consortium (PSTC) selected 23 exploratory renal biomarkers (Sistare et al. 2010) and systematically evaluated the utility of the most promising of these in multiple mechanistically distinct models of kidney injury in rats dosed with well-established nephrotoxicants (Dieterle et al. 2010; Vaidya et al. 2010; Yu et al. 2010). Seven of these urinary safety biomarkers were proposed to the regulatory authorities for qualification as kidney safety biomarkers for preclinical toxicology assessments. The Committee on Biomarkers of Nephrotoxicity of the Health and Environmental Sciences Institute (HESI) also evaluated a panel of renal safety biomarkers in the rat (Harpur et al. 2011). As a result of the efforts of these two consortia, 8 biomarkers-albumin, β2-microglobulin, total protein, cystatin C (CysC), kidney injury molecule-1 (Kim-1), clusterin, trefoil factor 3, and renal papillary antigen-1 (RPA-1) - were judged by regulatory authorities to be acceptable in specified contexts of nonclinical development for detection of acute drug-induced renal toxicity and to provide additional and complementary information to the currently available standard parameters (U.S. Food and Drug Administration [FDA] 2008, 2010; European Medicines Agency [EMA] 2009, 2010; Pharmaceuticals and Medical Devices Agency of Japan [PMDA] 2010). These newly qualified safety biomarkers are anticipated to facilitate early identification and elimination of drug candidates that are potentially nephrotoxic. It is also expected that, with better understanding of their diagnostic performance in humans, they will contribute to better safety monitoring and help prevent drug-induced kidney injury in clinical trials.
There are known sex differences in renal physiology, biochemistry, and toxicodynamics (reviewed in Sabolic et al. 2007; Trevisan et al. 2012) as well as in urinary excretion of protein biomarkers (Gautier et al. 2014). While there is evidence for these sex differences in several species, including dog and human, rats and mice have been more extensively studied. Despite this, almost all of the published data evaluated in the biomarker qualification decisions (Dieterle et al. 2010; Vaidya et al. 2010; Yu et al. 2010; Harpur et al. 2011) were generated only in male rats. Differences between the sexes in the magnitude of injury in response to renal trauma or differences in the constitutive presence of any chosen biomarker may not necessarily impair the diagnostic utility of that biomarker in both sexes. However, it would need to be shown that the response of the biomarker to an equivalent nature and severity of injury is comparable between sexes. Data demonstrating that renal biomarkers have equal utility in both sexes are sparse. Recently, Pinches et al. (2012) have begun to address this gap in our knowledge by studying the urinary biomarker response to cisplatin-induced renal injury in both male and female HAN Wistar rats. Reassuringly, their findings indicated that the urinary level of the biomarkers was related to the severity of the pathology and not overtly influenced by sex.
The study reported here was conducted under the auspices of the The International Life Sciences Institute (ILSI HESI) Committee on Biomarkers of Nephrotoxicity to examine differences between male and female Sprague-Dawley rats in the response of a wide range of urinary biomarkers to injury induced by gentamicin. An opportunity was also taken to evaluate the performance of several assay platforms. In an accompanying article, sex differences in urinary biomarkers in untreated male and female Sprague-Dawley rats are described (Gautier et al. 2014).
Method
Detailed methodology concerning the biomarker assays is presented in the companion article describing sex differences in urinary biomarkers in untreated male and female Sprague-Dawley rats (Gautier et al. 2014).
Animals and Husbandry
Sprague-Dawley rats [Crl:CD(SD)IGS BR] 7 weeks of age and weighing 130 to 230 g (females) and 250 to 350 (males) were acclimated for a minimum of 6 days before use. The rats were housed in pairs with a 12-hr light/12-hr dark cycle and had
Materials
Gentamicin sulfate (test article lot 047K07054) was purchased from Sigma-Aldrich Chimie (France).
Study Design and Procedures
On arrival in the test facility, rats were randomly assigned to study groups as shown in Table 1. Rats were given the dose of 75 mg/kg/day of gentamicin (expressed in terms of free base) dissolved in 0.9% physiological saline or the same volume of saline (control animals) subcutaneously once daily for 3 or 10 days of treatment.
Dosage regimen, number of animals, and timing of key procedures.

aDoses are expressed in terms of gentamicin base, and dose volumes were based on the most recent body weight.
bDoses were administered subcutaneously once daily for 10 days from days 1 to 10.
c0.9% saline.
During the dosing period, animals were examined clinically at least twice daily and weighed every other day. On the days specified in Table 1, animals were placed in individual metabolism cages overnight (approximately, 16 hr) and urine samples were collected into ice-chilled containers. During the urine collections, animals were fasted but had free access to water. At the end of the collection period, urines were examined for color, appearance, and measurement of volume and specific gravity. After low speed centrifugation (400 g) at 4°C for 5 min, multiple aliquots of the supernatant urine were prepared and stored at −80°C until analyzed for creatinine, total protein, and novel biomarkers as described subsequently.
On days 4 and 11, 10 animals of each sex from both the control and gentamicin-treated groups were euthanized by exsanguination from the abdominal aorta under Aerrane/isoflurane anesthesia. A sample of abdominal aorta blood was reserved for clinical pathology analyses.
Histopathological Examination
Liver and kidneys were examined at necropsy for macroscopic abnormalities, weighed, and placed in 10% neutral phosphate-buffered formalin. Liver injury was not anticipated to result from this dosage regimen of gentamicin; however, liver was examined microscopically to confirm this, ensuring that no biomarkers of hepatic origin would confound the interpretation of the urinary biomarkers arising from the kidney. Sections of liver for microscopic evaluation consisted of 2 transverse sections from the left lateral and right lobes and 1 cross section of the median lobe. For the kidney, sections including the cortex, the medulla, and the papilla consisted of 1 transverse section through the middle third of 1 kidney and 1 longitudinal section through the middle third of the other kidney. The fixed tissues were processed, embedded in paraffin wax, cut (4 µm thickness), and stained with hematoxylin and eosin for microscopic examination.
For histopathological evaluation of tissue, a senior veterinary pathologist performed an initial assessment with full knowledge of the different groups (unblinded). After this initial assessment, the same pathologist performed a targeted, masked (blinded) evaluation of all groups to reevaluate findings as needed to resolve gentamicin-related microscopic findings from normal variation and to ensure the consistency of diagnosis and grading. At all stages of the histopathological evaluation, the pathologist did not have any knowledge of the biomarker data to avoid any bias in the analysis. The slides were reviewed by and discussed with a second board-certified veterinary pathologist. The terminology and the grading were in accordance with the agreed upon standardized HESI/PSTC histopathology lexicon (Sistare et al. 2010; Harpur et al. 2011).
Immunohistochemistry
Immunohistochemistry was performed on kidney sections from 3 males from the control group and 3 males from the gentamicin-treated group euthanized on day 11 to confirm the site of nephron injury. These sections were co-labeled with α-glutathione-S-transferase (α-GST) and calbindin-D28 for visualization of proximal (Rozell et al. 1993) and distal (Taylor, McIntosh, and Bordeaum 1982; Vekaria et al. 2006) tubules, respectively. An additional co-labeling was performed with aquaporin-2 for visualization of collecting ducts (Reilly and Ellison 2000).
Immunostaining was performed on tissue sections serially cut at 3-µm intervals. Briefly, sections were deparaffinized and rehydrated. Antigen retrieval was performed by immersion in a pH6 citrate buffer at 98°C for 20 min. Nonspecific staining was blocked with 3% hydrogen peroxide solution in methanol for 10 min and 10% goat serum in phosphate-buffered saline (PBS) for 30 min. Serial sections were incubated with either polyclonal rabbit anti-α-GST (Argutus Medical, Dublin, Ireland) at a concentration of 9.2 µg/ml, mouse monoclonal anti-calbindin-D-28K (Sigma-Aldrich, St Louis, MO) at a concentration of 7.4 µg/ml or polyclonal rabbit anti-aquaporin-2 (Sigma-Aldrich, St Louis, MO) at a concentration of 0.06 µg/ml for 30 min at room temperature. Primary antibodies were labeled with goat anti-mouse /anti-rabbit IgG coupled to horseradish peroxidase for 30 min, followed by 3′3-diaminobenzidine for 5 min (EnVisionTM Dual Link kit from Dako, Trappes, France). Sections were counterstained with Mayer’s hematoxylin, dehydrated, cleared, and coverslipped.
Traditional Clinical Pathology Parameters and Novel Biomarkers
The methods used to assay the traditional biomarkers in blood (sCr and BUN) and in urine (total protein, albumin, and NAG) as well as the novel urinary biomarkers are described in the companion article (Gautier et al. 2014). A description of the analytical validation and acceptance criteria can also be found in Gautier et al. (2014). A summary of the assay platforms used to assay the urinary biomarkers is shown in Table 2.
Summary of which biomarker assays were conducted on the different platforms.

aIndividual assay formats are described in the companion article (Gautier et al. 2014).
bThese biomarkers have been qualified for use in specified contexts of nonclinical development for detection of acute drug-induced renal toxicity in rat studies (European Medicines Agency [EMA] 2009, 2010; U.S. Food and Drug Administration [FDA] 2008, 2010). The others are still regarded as exploratory biomarkers.
cAssays on these platforms were conducted for these 3 biomarkers, but either all control samples were below the lower limit of quantification (LLOQ; cystatin C and osteopontin) or most control samples were below the LLOQ or, when quantifiable, were highly variable (GST-Yb1). Therefore, only data from the RBM platform are presented for these biomarkers.
Expression of Urinary Biomarker Data
Urinary biomarker values were normalized for urinary creatinine content by dividing the urinary concentration of the biomarker by the urinary concentration of creatinine. The rationale for this is presented in the companion article (Gautier et al. 2014).
Statistical Analyses
For platform and day (day 4 and day 11) separately, log-transformed biomarker values (normalized for urinary creatinine content) were analyzed by analysis of covariance (ANCOVA) with treatment group and sex as fixed effects and log-transformed normalized baseline (day-2) biomarker values (normalized for urinary creatinine content) as a covariate (where available). Sex-specific treatment effects, differences in sex-specific effects, and corresponding 2-sided 95% confidence intervals were calculated and then converted to the ratio scale via the antilog transformation. Organ weight changes (absolute and relative to body weight) were analyzed by Student’s
Results
Gentamicin administration for 10 days was well tolerated (no clinical signs recorded), although a 10.3% and 13.1% decrease in body weight gain between day 1 and day 10 was observed in treated males and females rats, respectively, as compared to controls (data not shown). There were no macroscopic, microscopic, or organ weight changes in the liver resulting from gentamicin administration. These results are not presented or discussed further.
Kidney Weight Changes
Compared to mean values in controls, there were no kidney weight changes on day 4. Significant increases in mean absolute kidney weights (42% in males and 46% in females,
Macroscopic Observations in Kidney
On day 4, macroscopic findings in kidneys were limited to bilateral pale discoloration in 5/10 males and 3/10 females. Despite the absence of weight or microscopic correlation, this finding was considered related to gentamicin treatment.
On day 11, macroscopic findings in kidneys consisted of bilateral pale discoloration in all males and females and renal enlargement in 10/10 males and 7/10 females. These findings were correlated with organ weight increase and tubular epithelial cell degeneration/necrosis and tubular epithelial cell regeneration/basophilia in the cortex and the medulla.
Histopathology of Kidney
The histopathology results are summarized in Table 3. On day 4, minimal tubular epithelial degeneration of the S1 and S2 segments of the proximal convoluted tubule, characterized by the presence of intracytoplasmic hyaline droplets, was observed in 10/10 males and 0/10 females.
Incidence of histopathological findings in the kidneys.
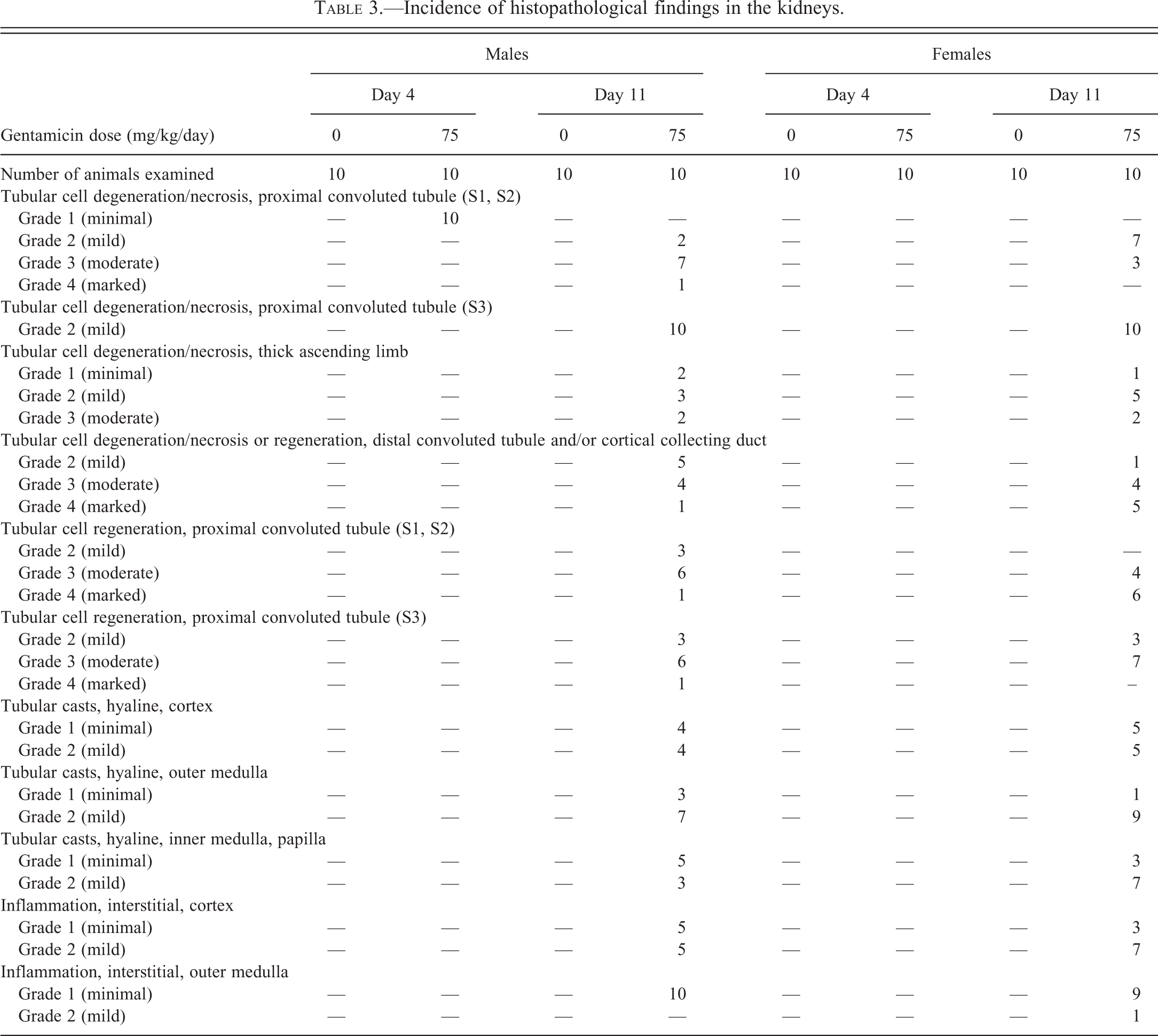
On day 11, microscopic kidney findings were more widespread and more severe than on day 4 and were noted in all animals. In the cortex, microscopic findings consisted of mild to marked tubular cell degeneration/necrosis (hyaline droplets, cell necrosis, cell sloughing, tubular dilatation with cell loss) and/or tubular regeneration (increased cytoplasmic basophilia, cell mitosis, cell anisokaryosis) of the S1 and S2 segments of the proximal convoluted tubule, the distal convoluted tubule, and/or the cortical collecting duct. Immunohistochemical staining with antibodies against α-GST confirmed the localization of these microscopic findings in the proximal tubules. Microscopic findings in the distal convoluted tubules and cortical collecting ducts were merged because it was difficult to differentiate them despite the use of immunohistochemical staining with antibodies against aquaporin-2 (for collecting duct) and calbindin-D28 (for distal tubules). In addition, minimal to mild intratubular hyaline casts were observed in the proximal convoluted tubules, distal convoluted tubules, and/or cortical collecting ducts in 8/10 males and 10/10 females. Minimal to mild foci of interstitial mixed inflammatory cell infiltrates were noted in all males and females. In the outer medulla, microscopic findings consisted of mild tubular cell degeneration/necrosis (cell necrosis, cell sloughing) and mild to marked tubular cell regeneration/basophilia (increased cytoplasmic basophilia, cell mitosis, cell anisokaryosis) of the S3 segment of the proximal tubule (pars recta) in 10/10 males and 10/10 females. Minimal to moderate tubular cell degeneration/necrosis of the thick ascending limb of Henle loop (reduced cytoplasmic eosinophilia) was observed in 7/10 males and 8/10 females. Minimal to mild intratubular hyaline casts in the thick ascending limb of Henle loop or outer medulla collecting ducts, and minimal to mild foci of interstitial mixed inflammatory cell infiltrates were noted in all males and females. In the inner medulla and papilla, microscopic findings consisted of minimal to mild intratubular hyaline casts in the collecting ducts in 8/10 males and 10/10 females.
Other microscopic findings reported in kidneys were consistent with expected spontaneous change in Sprague-Dawley rats.
Traditional Clinical Pathology Results Including sCr and BUN
There were no findings on day 4 related to gentamicin administration. Findings on day 11 were associated with increases in mean BUN and sCr in both sexes as compared with sex-matched control animals (between 3.0- and 3.6-fold).
Biomarkers
Baseline (pretreatment) values
Urinary biomarker levels were measured in urine samples collected between day −3 to −2 but are not presented. However, the values were used as a covariate in the statistical assessment of gentamicin-induced changes. A comprehensive set of constitutive (baseline) values for all of the biomarkers measured in urine in this study can be found in the companion article (Gautier et al. 2014).
Biomarker responses to gentamicin-induced renal injury—Assay platform and sex as variables
Biomarker values are presented (Tables 4–6) using results from assays conducted on three platforms as shown in Table 2. The use of multiple assay platforms enabled 16 biomarkers to be measured. Of these 16, assay results were available for 9 biomarkers from 1 platform. For 7 biomarkers, assay results were available from 2 or 3 platforms allowing some limited platform comparisons to be made.
Comparison of treatment-induced changes in biomarker values in male and female rats measured on the various singleplex assays.
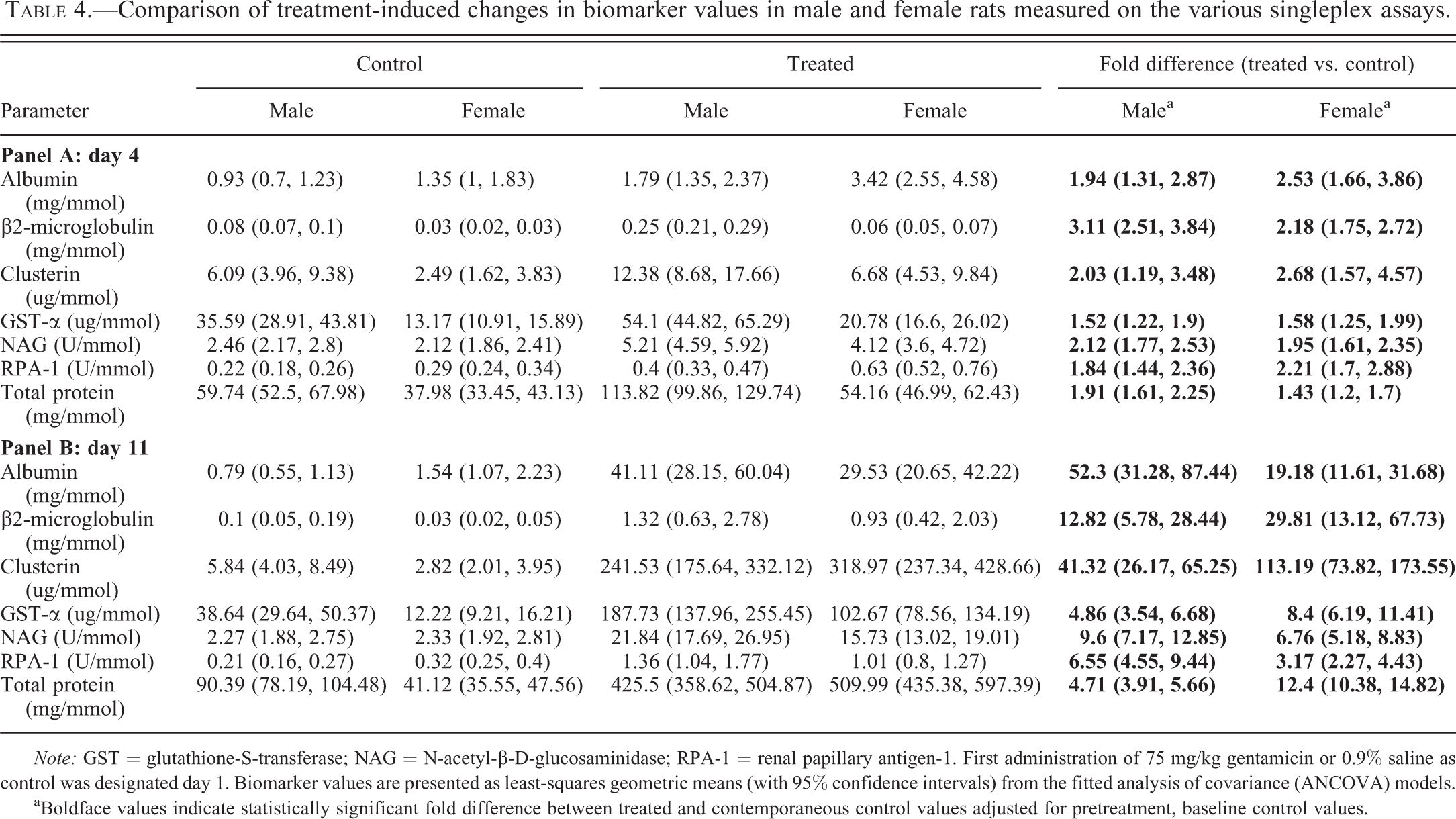
aBoldface values indicate statistically significant fold difference between treated and contemporaneous control values adjusted for pretreatment, baseline control values.
Comparison of treatment-induced changes in biomarker values in male and female rats measured on the RBM platform.
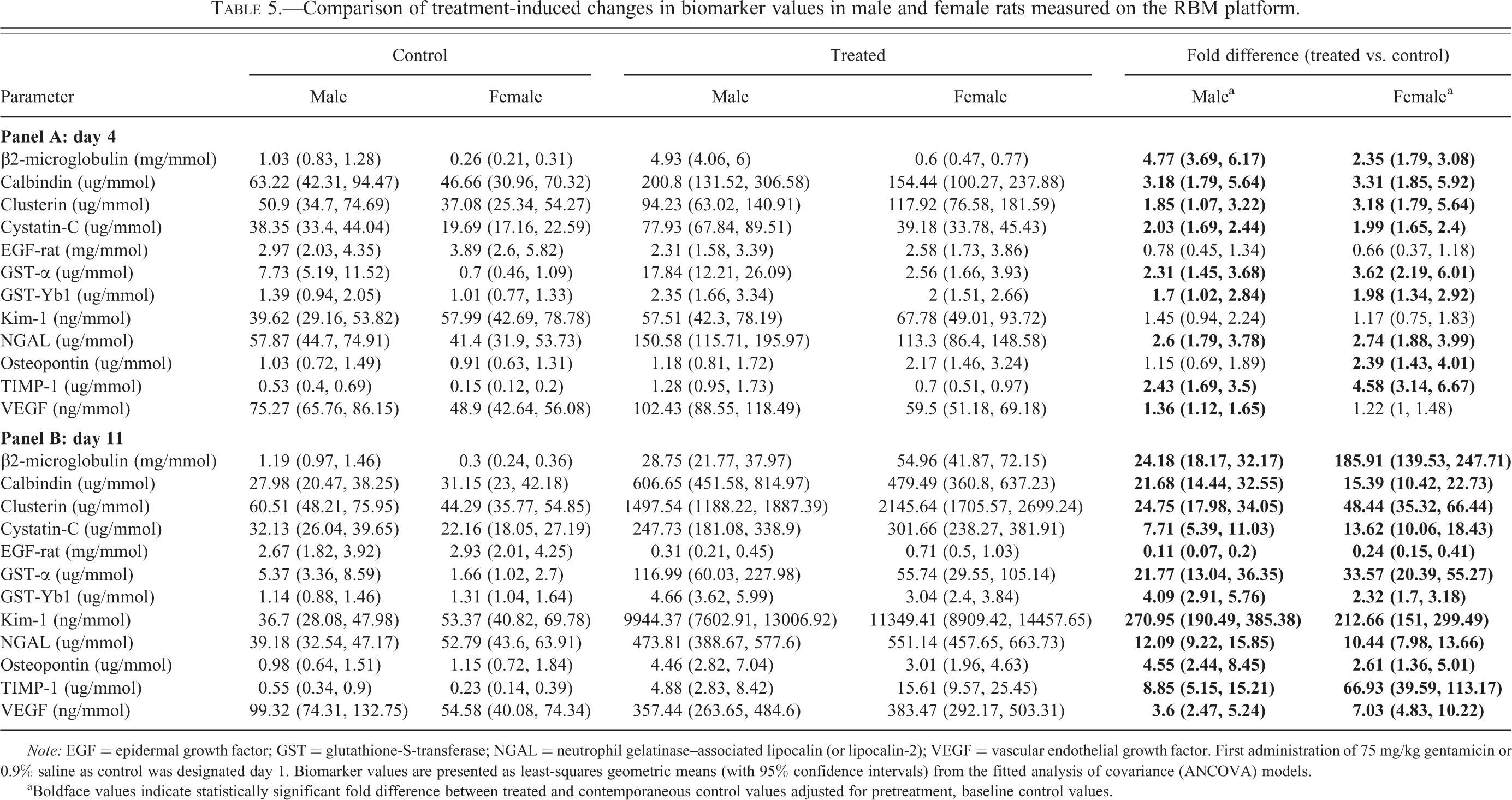
aBoldface values indicate statistically significant fold difference between treated and contemporaneous control values adjusted for pretreatment, baseline control values.
Comparison of treatment-induced changes in biomarker values in male and female rats measured on the MSD platform.
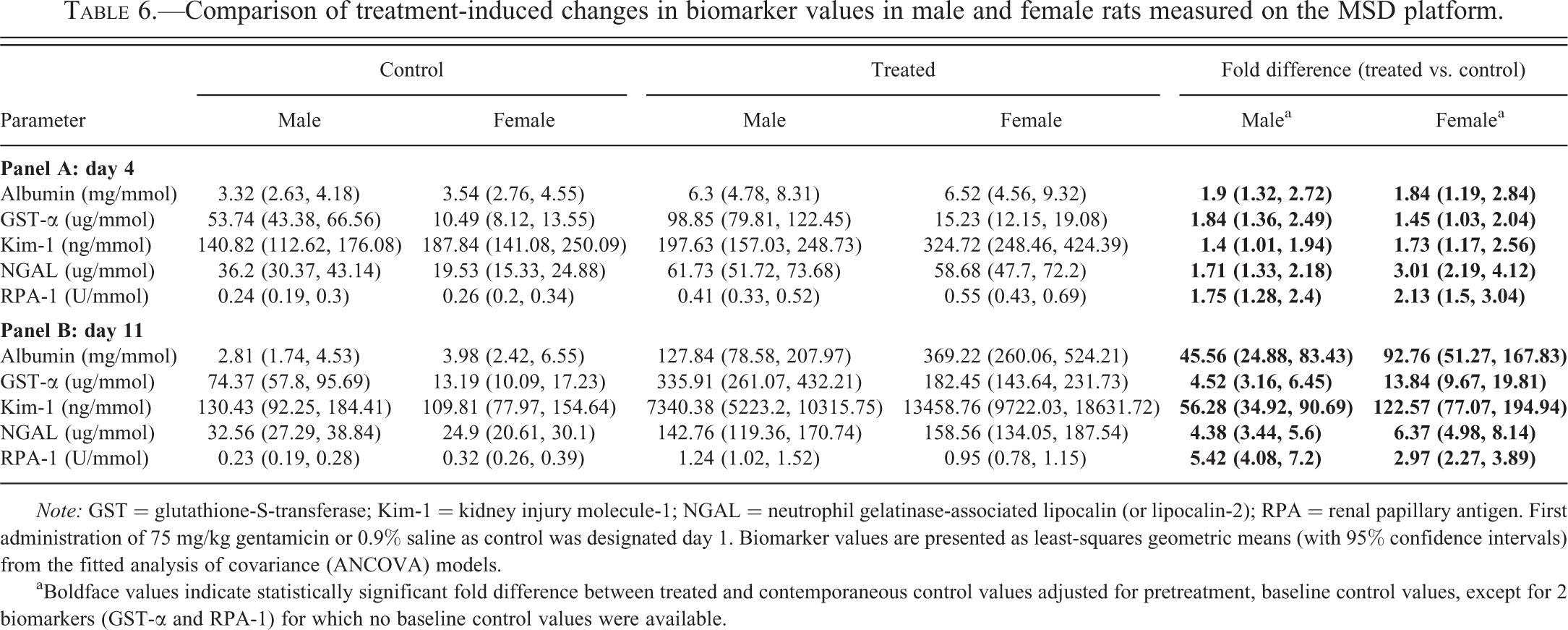
aBoldface values indicate statistically significant fold difference between treated and contemporaneous control values adjusted for pretreatment, baseline control values, except for 2 biomarkers (GST-α and RPA-1) for which no baseline control values were available.
In response to gentamicin-induced injury, all biomarker levels in urine were increased with the exception of EGF-rat, which was decreased (Tables 4–6). The magnitude of the changes, expressed as fold difference between treated and control, was small on day 4 (never exceeding 5-fold), when the injury, assessed microscopically, was minor in males and absent in females (Table 3). On day 11, when more marked injury was evident microscopically in both sexes (Table 3), the biomarker changes were much larger (up to 271-fold). For all biomarkers, the direction of the changes (increased in every case except for EGF, which was decreased) was absolutely consistent across platforms on both days and in both sexes although there were a few differences in the magnitude of change between data generated on different platforms and many differences in the magnitude of change between the sexes, particularly on day 11.
Although the fold differences (treated vs. control) on day 4 (Tables 4 [panel A], 5 [panel A], and 6 [panel A]) were small relative to the large differences on day 11 (Tables 4 [panel B], 5 [panel B], and 6 [panel B]), they were statistically significant across platforms and sexes, with very few exceptions. The exceptions included increases in osteopontin and vascular endothelial growth factor (VEGF) in treated animals on day 4 (data available for these 2 biomarkers only on the RBM platform; Table 5 [panel A]), which were statistically significant only in females or males, respectively. In the case of Kim-1, statistically significant increases on day 4 were seen only with data generated on the MSD platform (Table 6 [panel A]).
On day 4, as illustrated by the ratio of the fold changes induced by treatment in each sex (Table 7), there were statistically significant differences between the sexes for 5 biomarkers even if the magnitude of the sex difference ratios was never greater than 2-fold: β2-microglobulin (maximum 2-fold greater in males); osteopontin and TIMP-1 (approximately, 2-fold greater in females); protein (1.3-fold greater in males); NGAL (1.8-fold greater in females but only on the MSD platform).
Male to female ratios of fold changes in biomarker responses to gentamicin treatment.
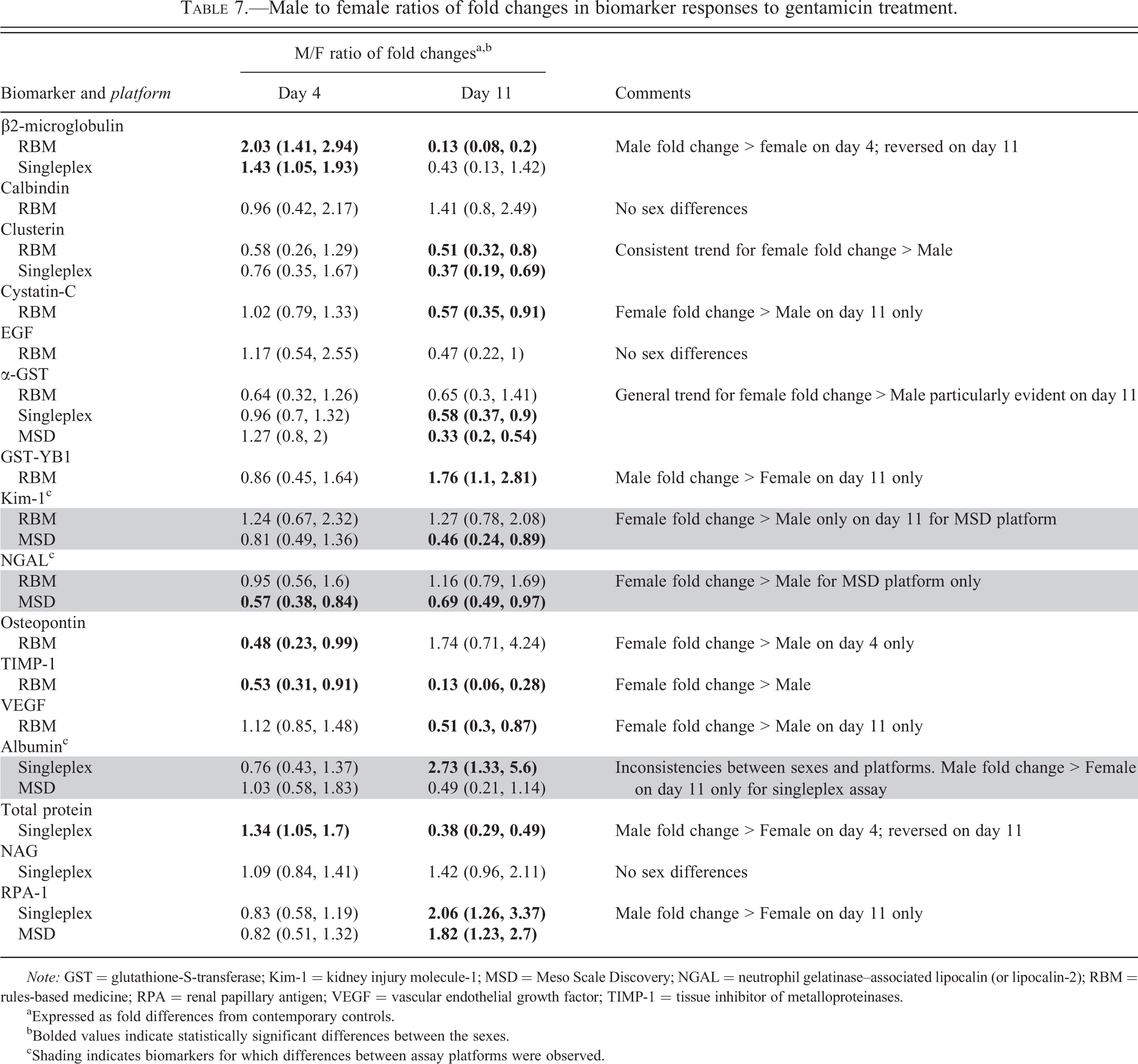
aExpressed as fold differences from contemporary controls.
bBolded values indicate statistically significant differences between the sexes.
cShading indicates biomarkers for which differences between assay platforms were observed.
On day 11, compared with day 4, there were much greater elevations of the levels of all biomarkers in urine (except for EGF which was decreased to a greater extent than on day 4). The fold increases (treated vs. control) were statistically significant for all biomarkers in both sexes and on all three platforms. However, despite the fact that the microscopic evaluation indicated comparable injury between the sexes, there were statistically significant sex differences on day 11 for a total of 12 biomarkers, as shown by the ratios between the fold increases in males and females (Table 7). Eight of these 12 biomarkers showed a sex difference in response to gentamicin treatment only on day 11 (Table 7). For 9 biomarkers (β2-microglobulin, clusterin, cystatin-C, TIMP-1, VEGF, GST-α, Kim-1, NGAL, and total protein), the fold change on day 11 was significantly greater in females than in males. For 3 biomarkers (albumin, GST-Yb1, and RPA-1), the reverse was true, that is, the fold change was greater in males. Some of these sex differences were large, for example, the treatment-related increases in urinary biomarkers were up to 8-fold greater in females in the case of β2-microglobulin and TIMP-1. Where there were data on more than 1 platform, there was generally good agreement with the exception of albumin, NGAL, and Kim-1. In the case of these 3 biomarkers, the significant sex difference was seen on only 1 platform (singleplex in the case of albumin and MSD in the case of NGAL and Kim-1). On the other platforms (MSD for albumin and RBM for NGAL and Kim-1), the direction of the sex difference was reversed, but this did not achieve statistical significance.
Discussion and Conclusions
Gentamicin Dose Selection
The renal toxicity of gentamicin in the rat has been studied extensively over many years, and it has been used as a toxicological tool in studies conducted to qualify the novel urinary biomarkers (Dieterle et al. 2010; Harpur et al. 2011; Vaidya et al. 2010; Yu et al. 2010). In the vast majority of these studies, including all those biomarker qualification studies that used gentamicin as a toxicological tool, only male rats were used. Thus, the study reported here was conducted to determine whether the urinary biomarker response was comparable in female rats. The dose of gentamicin used in this study was chosen both on the basis of literature and from a dose-range-finding study. Ozer et al. (2010) reported moderate to severe renal tubular degeneration, and regeneration after 9 daily intraperitoneal doses of 120 mg/kg gentamicin to male Sprague-Dawley rats. In a previous study in our laboratory, gentamicin, administered subcutaneously at a daily dose of 75 mg/kg for 14 days to male Wistar rats, produced mild degeneration and regeneration in the proximal tubule (Hoffmann et al. 2010). In a dose range-finding toxicity study in male Sprague-Dawley rats, conducted at Pfizer on behalf of the HESI Committee on Biomarkers of Nephrotoxicity, the dose of 75 mg/kg/day of gentamicin given subcutaneously for 7 days was clinically well tolerated and did not induce any significant increases in BUN or sCr. Histopathological findings in the kidneys at this dose level consisted of minimal to moderate tubular degeneration/regeneration associated with interstititial mononuclear cell infiltrates in all animals. On this basis, 75mg/kg/day for 10 days was expected to produce a range of pathology in the kidney ranging from mild to moderate in severity.
Evidence for Lack of Sex Difference in the Nephrotoxic Response to Gentamicin in the Sprague-Dawley Rat
There is evidence for sex differences in the nephrotoxic response of the rat kidney to some chemicals such as hexachlorobutadiene, acetaminophen and cisplatin (for review, see Trevisan et al. 2012). These sex differences in toxicity are generally attributable to sex differences in metabolism and/or renal transporter function. However, gentamicin is not metabolized and there is no clear evidence for sex differences in its nephrotoxic potential in the rat (Trevisan et al. 2012). If a sex difference in gentamicin toxicity exists, it appears to be restricted to the F344 strain. Thus, gentamicin (and another aminoglycoside antibiotic, tobramycin) was reported to be more nephrotoxic in male versus female F344 rats, but this sex difference in toxicity was not evident in Sprague-Dawley rats (Bennett et al. 1982; Goodrich and Hottendorf 1995), the strain of rat used here.
Histopathology Findings in the Present Study (Summarized in Table 3)
On day 4, histological evidence of toxicity in the proximal convoluted tubule, graded minimal, was seen in all male but no female rats. On day 11, a variety of microscopic lesions located in several segments of the tubule, ranging from mild to marked in severity, were seen in all animals of both sexes. Broadly, the findings were similar in both sexes although there were some differences between the sexes in incidence at each severity grade. Thus, male animals exhibited a higher incidence of moderate and marked severity grades for degeneration and necrosis in the proximal convoluted tubule (S1, S2) while the incidence of moderate to marked regeneration in the same region of the tubule was higher in females. Taken together, these differences between the sexes suggest a difference in the time course of the lesion progression from injury to repair rather than an overt difference in sensitivity to gentamicin nephrotoxicity. However, there was also a shift to more marked severity grades in female animals compared with males for degeneration and regeneration (combined) in the distal convoluted tubule and cortical collecting duct. The extensive renal microscopic lesions seen in all animals on day 11 were accompanied by large fold increases in the appearance of all studied biomarkers in urine from animals of both sexes. To investigate whether these differences between the sexes in incidence of severity grades for the same lesions could explain the observed sex differences in urinary biomarker levels, we examined whether there was a correlation between urinary biomarker values and severity grade for the previously mentioned microscopic lesions in individual animals (data not shown). This evaluation revealed no individual correlation between the fold increase in urinary biomarker levels and histopathology grade in either sex. Furthermore, it was seen that urinary biomarker fold increases were mostly higher in females at the same (or even a lower) severity grade of injury to that observed in males. Therefore, it is concluded that the sex differences in biomarker response to gentamicin-induced injury cannot be explained by sex differences in the severity of injury.
Biomarker Assay Platforms
The biomarker assay platforms used in this study have been evaluated using urine samples collected from untreated rats (Gautier et al. 2014). All of the assays performed satisfactorily with the exception of 4, namely the singleplex assay for cystatin-C, the singleplex and MSD assays for GST-Yb1, and the MSD assay for osteopontin (Table 2). Therefore, data generated for these 3 biomarkers on these platforms are not presented in the results tables and were excluded from all analyses. Data for each of these 3 biomarkers are available on the RBM platform.
Using urine samples from untreated rats (Gautier et al. 2014), it has been shown that assay platforms for urinary biomarkers differ in the variability of the results produced but also, more importantly, in the absolute magnitude of the biomarker values obtained. The present study also permitted some limited but useful comparisons between assay platforms of the measured changes in urinary biomarkers induced by gentamicin administration. Although there was absolute consistency in the direction of the changes across platforms in both sexes on both day 4 and 11, there were some differences in the magnitude of change between data generated on different platforms. These differences translated into some disagreement between assay platforms in the assessment of sex differences in the changes in urinary biomarker values in response to gentamicin. The 3 observed occurrences of such disagreement (Kim-1, NGAL, and albumin) are highlighted in Table 7. In the case of all 3 biomarkers, there was a difference between platforms on day 11 in whether or not the male to female ratio of fold differences in response to gentamicin was significant. However, this has to be seen in context of the presence in both sexes of significant renal injury and universally large fold increases in biomarker values on day 11, irrespective of which assay was used (Tables 4–6). Thus, a statistically significant difference between assays in the ratio of the fold increase is unlikely to have any biological significance, that is, would be unlikely to influence the diagnostic utility of the biomarker. Only in the case of NGAL did the statistical significance of the male to female ratio of fold differences following gentamicin vary between platforms on day 4. Since the urinary biomarker increases on day 4 are occurring at renal injury levels close to the histologically detectable threshold, potentially this might have some diagnostic import. However, the range of fold increases across the sexes and assay platforms is small for NGAL on day 4 (ranging from a mean of 1.71 to 3.01) suggesting that this is a statistical quirk and unlikely to be biologically relevant.
While these 3 instances of lack of concordance between platforms are currently without explanation, it should be stressed that generally there was very good agreement between the different assays (Table 7). Some impact on the interpretation of results close to injury threshold cannot be completely ruled out. A more comprehensive comparative assessment of the performance of these assays at detecting small fold increases would be required to make recommendations as to which assays should be preferred.
Assessment of Sex Differences in Biomarker Response to Gentamicin Treatment
There were many significant changes in urinary biomarker levels in response to gentamicin treatment in both sexes on both days 4 and 11 (Tables 4–6) and the magnitude of these changes frequently differed between the sexes. There were just 3 of 16 biomarkers (calbindin, EGF, and NAG) that showed no statistically significant sex difference in biomarker response on either day (Table 7). For the remaining 13 biomarkers, where sex differences in biomarkers response were observed, in the majority (3 of the 5 on day 4 and 9 of the 12 on day 11) the response in females, as measured by fold difference from contemporary controls, was greater than in males.
On day 4, the 2 sex differences in the magnitude of change where male was greater than female (β2-microglobulin and total protein, Table 7) appear consistent with the microscopic findings in that the minor injury to the proximal tubule was reported only in males. However, for both β2-microglobulin and total protein, the sex difference in urinary biomarker levels on day 11 was reversed, that is, urinary increases were significantly greater in females than males. On day 4, in 3 instances (NGAL, osteopontin, and TIMP-1), the increases in urinary biomarker levels were significantly greater in females than males (Table 7), despite the lack of histopathological change in females. The sex difference in the case of osteopontin (2-fold greater in females) might reflect a true sex difference either in the nature of the underlying injury or in the osteopontin response to very minor injury. However, the fold increases in osteopontin levels on day 11, when more severe injury was present, were greater in males, but not significantly so. Two other biomarkers showed a significantly greater response in females than males on day 4 (TIMP-1 and NGAL). In the case of NGAL and TIMP-1, the increases in urinary levels were significantly greater in females than males on both day 4 and day 11 although in the case of NGAL, the sex difference was evident only with data generated on the MSD platform (Table 7). We have shown (Gautier et al. 2014) that there are sex differences in baseline values (mostly male greater than female) for several of the biomarkers studied here. Therefore, on day 11, the greater biomarker increases observed in urine of female rats as compared to male rats for some biomarkers like total protein, β2-microglobulin and TIMP-1 following gentamicin administration can be explained, at least in part, by lower baseline values in females as compared to males.
Given the very large increases in biomarker values observed consistently in both sexes on day 11, it is questionable whether the differences in magnitude of the changes would have led to any difference in diagnostic interpretation between the sexes at that time. What is evident is that the pattern of biomarker change was absolutely consistent in both sexes. The results on day 4 do indicate that the urinary levels of many of these biomarkers change in the presence of minor injury. The changes were seen in females as well as males although toxicity was observed microscopically only in males. It is not possible to say whether this is indicative of prodromal behavior in females. The existence of sex differences in urinary biomarker levels close to the threshold of renal injury has considerable implications for their diagnostic utility. Thus, it would be of great interest to confirm whether or not the sex differences in urinary biomarkers levels in the presence of minimal injury reported here are reproducible and, if confirmed, to elucidate the underlying mechanisms.
In addition to understanding better the exact nature of the response of these biomarkers to the kind of injury reported in this study, more extensive examination of the kidney using sensitive methods (including immunohistopathology) would be required to determine if there were underlying sex differences in the injury at locations other than the proximal tubule. The study had inadequate power (small number of animals with insufficient range of severity of microscopic lesions) to address adequately the key question of whether there would be any difference in the diagnostic utility of the biomarkers at injury threshold. Given the evident sex differences in constitutive values of many of these biomarkers in urine (Gautier et al. 2014) and the sex differences in response to injury reported here, this remains an important question to answer. Publication of preclinical development studies conducted using both male and female animals to investigate renal toxicity and incorporating measurement of novel renal biomarkers would be very helpful to resolve the remaining uncertainties.
Footnotes
Acknowledgment
We thank Pascal Le Priol from Sanofi for excellent technical assistance in this work.
Authors’ Note
The ILSI HESI Committee on Biomarkers of Nephrotoxicity is a consortium of pharmaceutical companies and government and academic scientists, whose mission is to advance development of biomarkers of renal toxicity that bridge from the preclinical to the clinical stages of drug development. HESI is an international, nonprofit organization that stimulates and supports scientific research that contributes to the collaborative identification and resolution of health and environmental issues of concern to the public, scientific community, government agencies, and industry.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication stems from work conducted as an in-kind contribution by a subgroup of members of the HESI Biomarkers of Nephrotoxicity Committee, which is coordinated and funded through ILSI HESI.
