Abstract
The inhalation route is a relatively novel drug delivery route for biotherapeutics and, as a result, there is a paucity of published data and experience within the toxicology/pathology community. In recent years, findings arising in toxicology studies with inhaled biologics have provoked concern and regulatory challenges due, in part, to the lack of understanding of the expected pathology, mechanisms, and adversity induced by this mode of delivery. In this manuscript, the authors describe 12 case studies, comprising 18 toxicology studies, using a range of inhaled biotherapeutics (monoclonal antibodies, fragment antigen-binding antibodies, domain antibodies, therapeutic proteins/peptides, and an oligonucleotide) in rodents, nonhuman primates (NHPs), and the rabbit in subacute (1 week) to chronic (26 weeks) toxicology studies. Analysis of the data revealed that many of these molecules were associated with a characteristic pattern of toxicity with high levels of immunogenicity. Microscopic changes in the airways consisted of a predominantly lymphoid perivascular/peribronchiolar (PV/PB) mononuclear inflammatory cell (MIC) infiltrate, whereas changes in the terminal airways/alveoli were characterized by simple (“uncomplicated”) increases in macrophages or inflammatory cell infiltrates ranging from mixed inflammatory cell infiltration to inflammation. The PV/PB MIC changes were considered most likely secondary to immunogenicity, whereas simple increases in alveolar macrophages were most likely secondary to clearance mechanisms. Alveolar inflammatory cell infiltrates and inflammation were likely induced by immune modulation or stimulation through pharmacologic effects on target biology or type III hypersensitivity (immune complex disease). Finally, a group of experts provide introductory thoughts regarding the adversity of inhaled biotherapeutics and the basis for reasonable differences of opinion that might arise between toxicologists, pathologists, and regulators.
Keywords
Introduction
In November 2017, the British Society of Toxicologic Pathology (BSTP) discussed the development challenges of inhaled biotherapeutics (biologically sourced drugs such as antibodies, proteins, or peptides), hereafter termed “biologics,” at its 32nd Annual Scientific Meeting (ASM) entitled “The Pathology and Toxicology of Biologics and Biotherapeutics.” This topic had recently also been reviewed by other societies and organizations, as the inhaled route was still a relatively novel method of biologic drug delivery, although the first inhaled proteins were investigated several decades ago. Findings from toxicity studies of inhaled biotherapeutics have raised several challenging toxicological questions and, not surprisingly, the subject sparked lively debate at the BSTP ASM. At the conclusion of the meeting, a decision was made to publish a white paper addressing the paucity of information and experience in the public domain and the absence of a consensus among the scientific community on the common or expected findings inherent to inhalation of any biologic regardless of target. A global survey was performed to collate and consolidate industry experience with the goal of presenting their findings in this special edition of Toxicologic Pathology.
This manuscript is a collection of 12 inhaled biologic case studies consisting of 18 toxicology studies from across the biopharmaceutical industry that reflect the interpretations of the toxicologists and pathologists who have experience with this route of drug delivery. The survey sought to gather industry-wide experience to increase understanding in 4 key areas: (1) microscopic findings associated with inhaled biologics, (2) plausible mechanism(s) for the changes induced by inhaled biologics, (3) relationship of the findings to immunogenicity associated with the inhaled route, and (4) a discussion of the factors determining adversity of the major pathology findings.
Our goal was to generate comparative data set for common and/or adverse findings associated with inhaled biologics, thereby providing guidance to the industry at large. The relatively broad selection of case examples presented, together with the experience and views of those who have worked in this field, provides substantial illumination on this complex area of toxicologic pathology. We hope that this data set and our interpretation of the pathology will act as a repository of information on which others might build. Ultimately, it is desired that the data set informs and facilitates discussions with global health authorities on the interpretation of lesions after the administration of inhaled biologics in regulatory toxicology studies.
Materials and Methods
A survey was conducted by the BSTP, European Society of Toxicologic Pathology, Society of Toxicologic Pathology, and the Association of Inhalation Toxicologists requesting case studies on inhaled biologics in 2018. Toxicologists and pathologists based in Europe and North America from a range of Pharmaceutical Companies, Contract Research Organizations, Consultancies, and the Medicines and Healthcare products Regulatory Agency formed a pathology working group to collect and analyze the data and provide insight into the pathology, immunogenicity, mechanistic considerations, and adversity induced by inhaled biologics. The case studies were anonymized to preserve test-article identity, and histopathology data (together with relevant ancillary data such as clinical pathology and clinical signs) were extracted to populate a template to generate a uniform data set across the series. The severity scores “mild” and “slight” (which are both normally scored as a “2” on a 4- or 5-point scoring system) were combined into the single score “mild” for consistency. Additional information (such as immunogenicity/anti-drug antibody [ADA] data) was requested as required but was not always available. Pathology data were merged into one of several root diagnoses using International Harmonization of Nomenclature and Diagnostic criteria (INHAND) terms, where possible, to generate consistent nomenclature across case studies. Unique terms were not converted. The original pathology scores were used to indicate lesion severity. All studies were conducted in accordance with each sponsor’s policy on the “Policy on the Care, Welfare and Treatment of Laboratory Animals” and reviewed by the Institutional Animal Care and Use Committee (IACUC). Furthermore, in Europe, there was an ethical review process at the institution where the work was performed and was conducted in an Organization for Economic Co-operation and Development (OECD) member country in accordance with OECD Test Guidelines. For North American based studies, the study plan and any amendment(s) or procedures involving the care and use of animals were reviewed and approved by the IACUC. During the study, the care and use of animals was conducted in accordance with the guidelines of the US National Research Council and the Canadian Council on Animal Care. All studies were, at a minimum, performed in accordance with the Principles of Good Laboratory Practice (GLP) if not formally designated as GLP compliant.
The discussion of adversity presented in this manuscript represents a consensus from the working group based on the data available and the judgment of internal experts from the working group (some of whom were the original pathologists for the case studies in question). Additional opinion from experts in inhalation pathology who were external to the group also contributed to the discussion of adversity and review of the manuscript.
A Note on Severity Scoring and Lesion Nomenclature
In order to understand how lesion severity is established and impacts adversity, and to communicate this to readers unfamiliar with this process, the authors considered it important to describe the standardized scoring system(s) employed by pathologists and the terms used to describe morphologic changes observed microscopically. Toxicologic pathologists use grading/nomenclature systems that vary according to study-specific details. Despite minor variations in approach and application, pathologists attempt to identify and interpret treatment-related changes and provide accurate and consistent data. 1,2
The severity grade (or score) of a microscopic observation is a semiquantitative assessment of the morphologic extent, complexity, and distribution of a finding. Most grading systems use either 4 or 5 grades (Table 2), and in general, the schemes align very well. Grades can be expressed as numbers (Numerical score, Table 1) correlating with words (Description, Table 1) that describe considerations (Broad definition, Table 1). Severity grade is normally established using 3 morphology-based criteria: extent of tissue/organ affected, complexity/features of the morphologic finding, and distribution/subanatomic topography (Table 1). Severity grades are most accurate when adjusted based on the spectrum of response observed in concurrent control animals, so, for example, when assessing dynamic lymphoid-related findings, such as increased bronchus-associated lymphoid tissue (BALT) cellularity in the lung, it is prudent to carefully examine all controls to determine the range of normal in order to identify treatment-related changes especially if they are subtle and low grade. This process ensures that background changes are not recorded as treatment-related effects.
Example 4- and 5-Point Grading Systems.

Key Determinants in the Assignment of Histopathology Severity Scores in Lung.
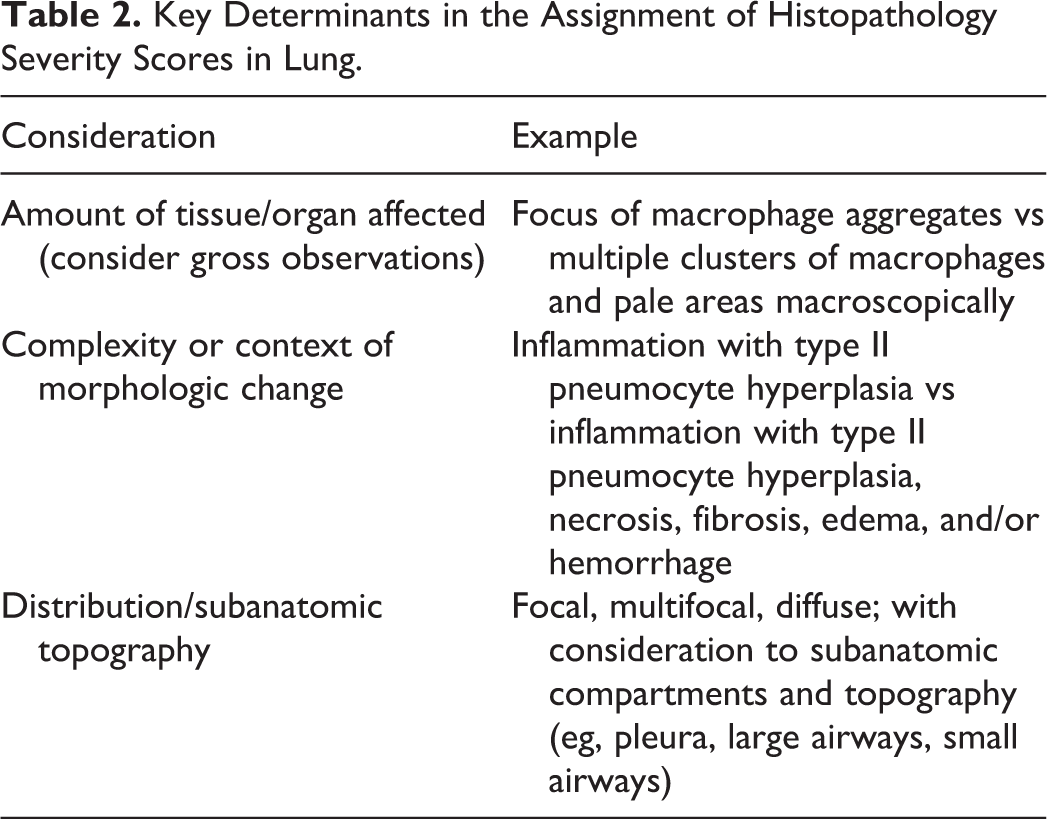
Organ-specific INHAND nomenclature serves to further standardize and characterize pathology changes, thereby providing helpful detail to subsequent reviewers who may not be pathologists. 1 In addition to standardized terminology, a clear description of each term and grade should be included in the narrative when describing adverse changes or when severity is a factor for adversity and/or critical dose effect level determinations (eg, no observed adverse effect level/lowest observed adverse effect level or the highest nonseverely toxic dose). 3 When assigning a severity grade, organ weights, clinical pathology, test article relationship, human relevance, biological impact, organ function, health status of the animal, reversibility, and adversity should not be considerations. 4
Results and Discussion
Part 1: Case Study Characteristics
Twelve inhaled biologic cases, each defined by a unique test article, consisting of 18 toxicology studies were donated to the inhaled biologics data set (Tables 3A and 3B) ranging in duration from a 7-day subacute study through to two 26-week chronic studies.
The majority of the studies consisted of peer-reviewed GLP toxicology studies intended to support first-in-human clinical trials. The most common toxicology nonclinical species was the nonhuman primate (NHP), most likely reflecting test article target cross-reactivity, but the data set also included 3 rodent studies (2 rat and 1 mouse) and 1 rabbit study. Test article modalities were generally equally distributed between antibodies (3 immunoglobulin single variable domains [ISVDs]/domain antibodies [Dabs], 2 fragment antigen-binding antibodies [Fabs], and 1 monoclonal antibody [Mab]) and protein/peptide therapeutics (5 in total). Additionally, 1 oligonucleotide therapeutic was also included. Dose levels of the inhaled test articles, despite the diversity of therapeutic products, were generally within the range of 1 to 20 mg/kg/d, but overall, the range was 9 µg/kg/d to 500 mg/kg/d.
General features
Treatment with all the inhaled biologics was well tolerated with either no or only minor changes in clinical observations, body weight, body weight gain, lung weight, food consumption, ophthalmology, and chest auscultation (Table 3A). No mortalities were attributed to direct effects of the test article but, in 2 case studies, single instances of bronchopneumonia were recorded in NHPs which were attributed to either the bronchoalveolar lavage (BAL) procedure (case study [CS] 5, 6-week NHP) or the inhalation exposure procedure (CS 11, 13-week NHP). In CS 12 (26-week NHP), a further mortality was attributed to immune complex disease (ICD).
Summary of 12 Case Study Preclinical Safety Data (Excluding Immunogenicity & Microscopic Data).
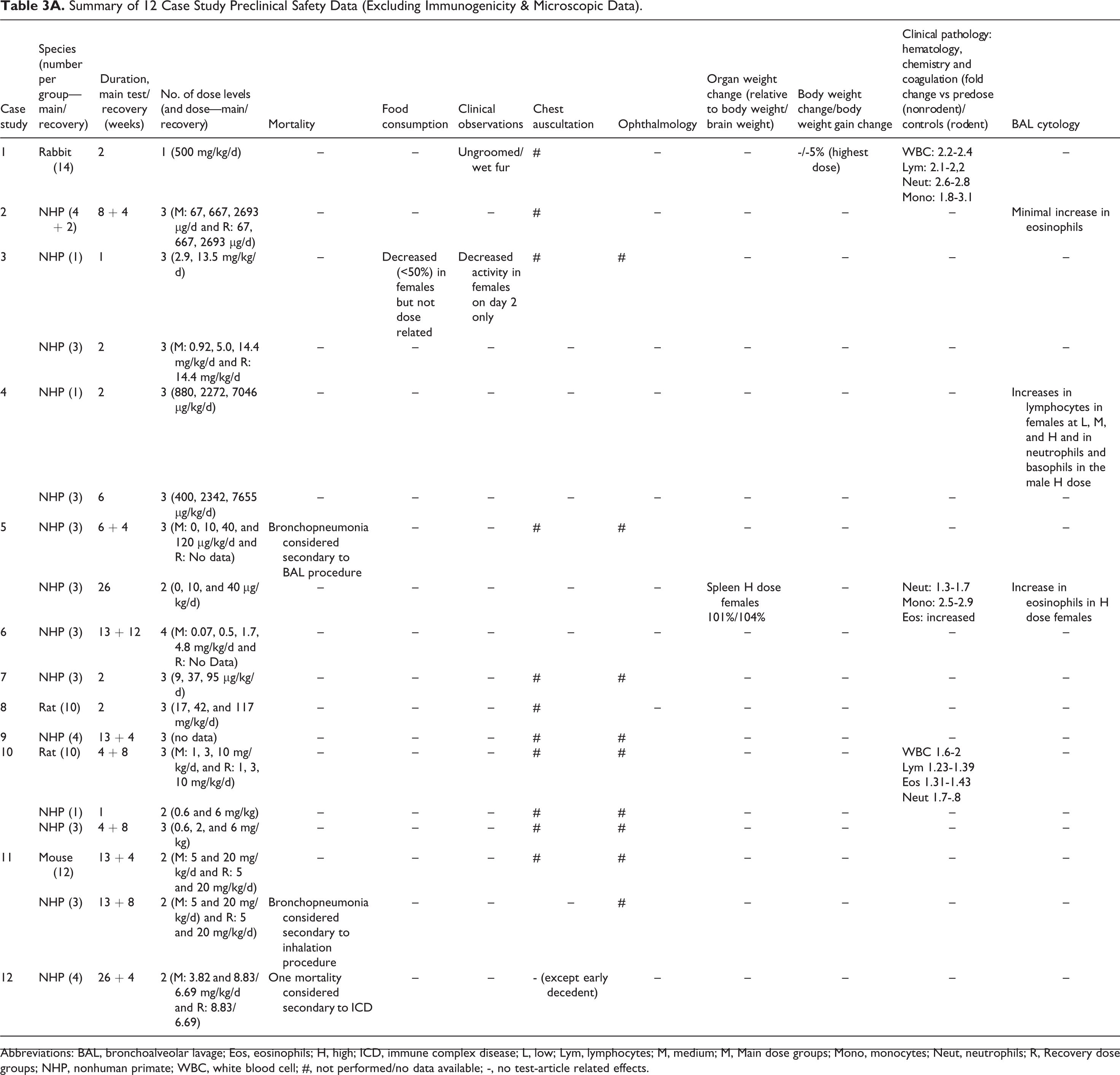
Abbreviations: BAL, bronchoalveolar lavage; Eos, eosinophils; H, high; ICD, immune complex disease; L, low; Lym, lymphocytes; M, medium; M, Main dose groups; Mono, monocytes; Neut, neutrophils; R, Recovery dose groups; NHP, nonhuman primate; WBC, white blood cell; #, not performed/no data available; -, no test-article related effects.
In addition, no test article-related changes were recorded in coagulation and clinical chemistry. Minor changes in white blood cell parameters were recorded for some case studies (CS 1, 2-week rabbit; CS 5, 26-week NHP; and CS 10, 4-week rat) consisting generally of 1- to 3-fold increases in various white blood cell subsets (lymphocytes, neutrophils, eosinophils, and monocytes; Table 3A). Bronchoalveolar lavage cytology (or data) was available in only 3 studies. Changes consisted of increases in eosinophils (CS 2, 8-week NHP and CS 5, 26-week NHP) or lymphocytes, neutrophils, and basophils (CS 4, 2-week NHP; Table 3A).
Inhaled biologics produce a characteristic pattern of toxicity
One study (CS 3, 1-week NHP) treated with an ISVD/Dab showed no microscopic effects (Table 3B). However, most studies, regardless of modality and duration, were associated with test article-related histologic changes (Table 3B), which occurred in 2 distinct but sometimes overlapping anatomic regions of the respiratory tract: centered on the airways of the lungs in a PV/PB location (and sometimes extending into neighboring alveoli) and in the alveoli and associated respiratory and terminal bronchiolar regions. Changes were also occasionally noted in the draining tracheobronchial lymph nodes and more rarely in the nasal cavity.
Summary of Case Study and Relevant Lung Pathology in the 12 Case Studies and 18 Toxicology Studies.
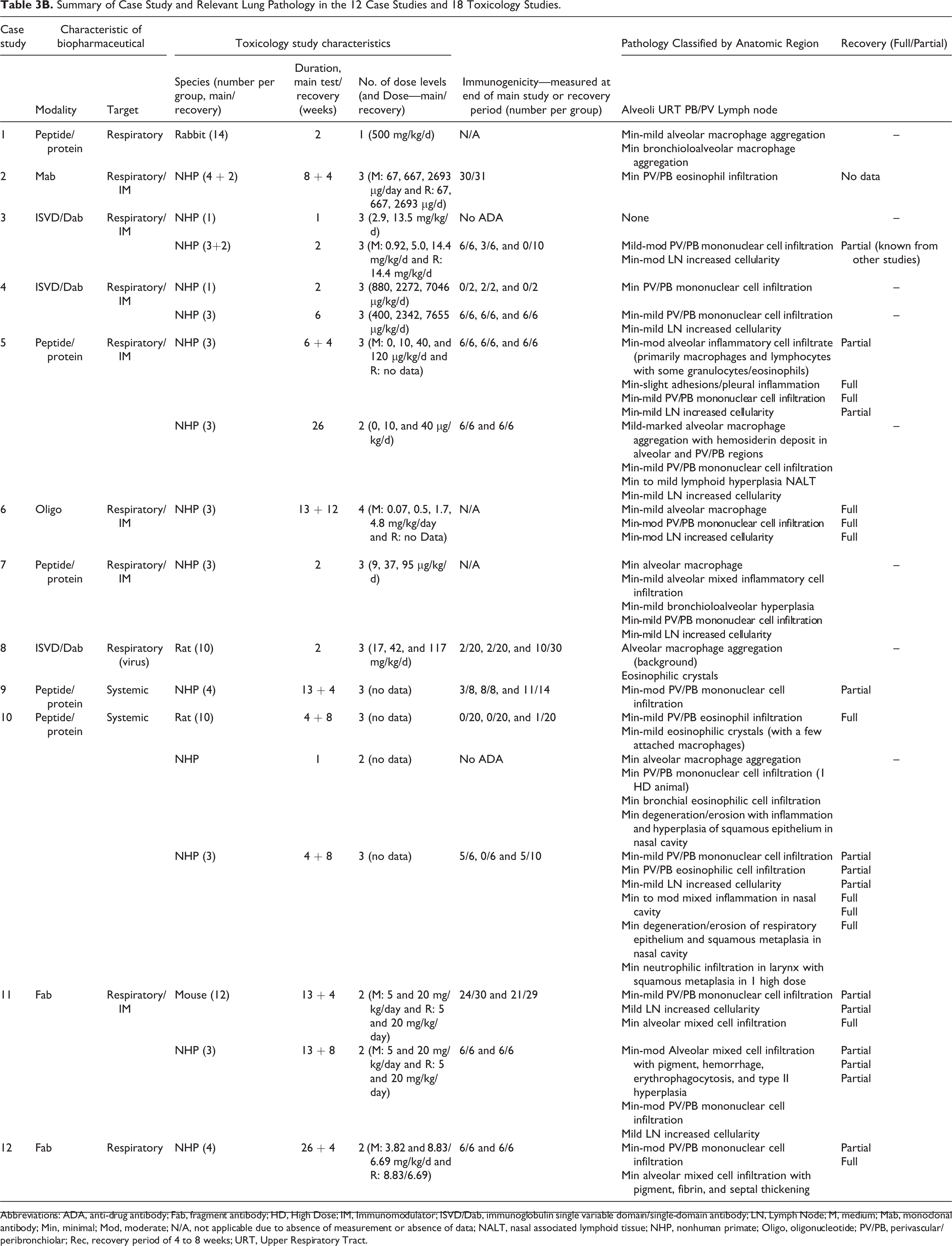
Abbreviations: ADA, anti-drug antibody; Fab, fragment antibody; HD, High Dose; IM, Immunomodulator; ISVD/Dab, immunoglobulin single variable domain/single-domain antibody; LN, Lymph Node; M, medium; Mab, monoclonal antibody; Min, minimal; Mod, moderate; N/A, not applicable due to absence of measurement or absence of data; NALT, nasal associated lymphoid tissue; NHP, nonhuman primate; Oligo, oligonucleotide; PV/PB, perivascular/peribronchiolar; Rec, recovery period of 4 to 8 weeks; URT, Upper Respiratory Tract.
Perivascular/peribronchiolar changes
The most frequent change noted in the perivascular/peribronchiolar (PV/PB) region of the lungs was an infiltration of mononuclear inflammatory cells (MIC; also called mononuclear cell infiltration/infiltrate) characterized principally by lymphocytes with fewer numbers of macrophages (Figures 1 and 3). These were orientated predominantly around airways and blood vessels and within the BALT (the latter sometimes referred to in study reports as BALT hyperplasia). In the most severely affected examples, the PV/PB MIC infiltrates occasionally extended into and distorted/thickened the terminal/respiratory bronchioles and neighboring alveoli. The PV/PB infiltrates were noted in 13 (72%) of 18 toxicology studies in this case series, ranging from 1 to 26 weeks in duration with minimal to moderate severity (Table 3B). Of the 5 studies where PV/PB MIC changes were not observed, 3 were of 2 weeks or shorter duration (CS 1, 2-week rabbit; CS 3, 1-week NHP; and CS 8, 2-week rat). This study length likely reflected the minimum period of time that was required for mononuclear cell infiltrates to infiltrate and accumulate to a level that could be detected by light microscopy given the kinetics of immune responses (ie, lymphocyte homing [approximately 10 days] to mucosal tissues after an immune response). 5
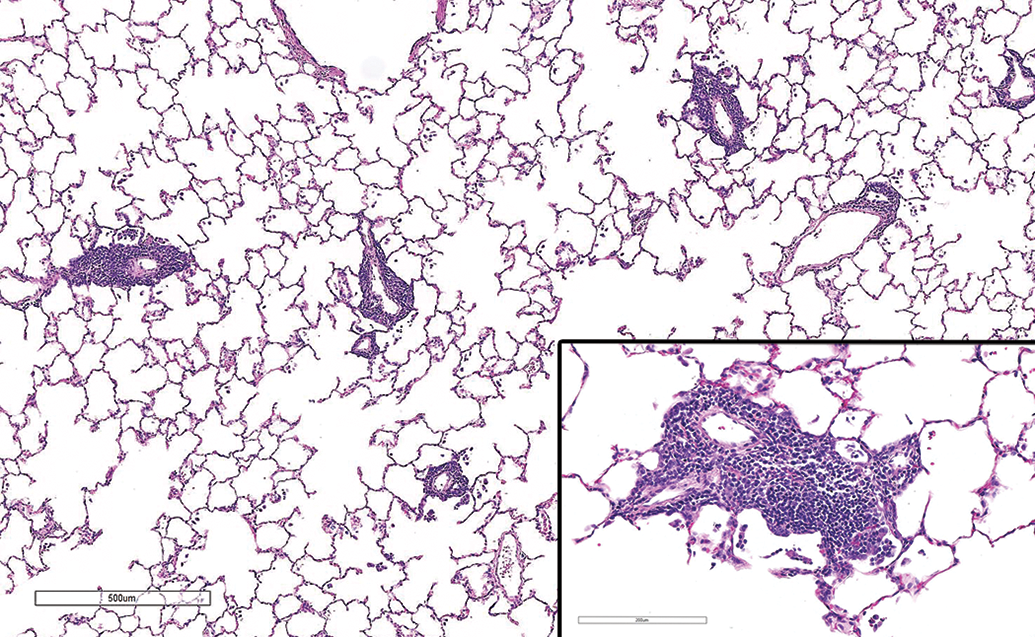
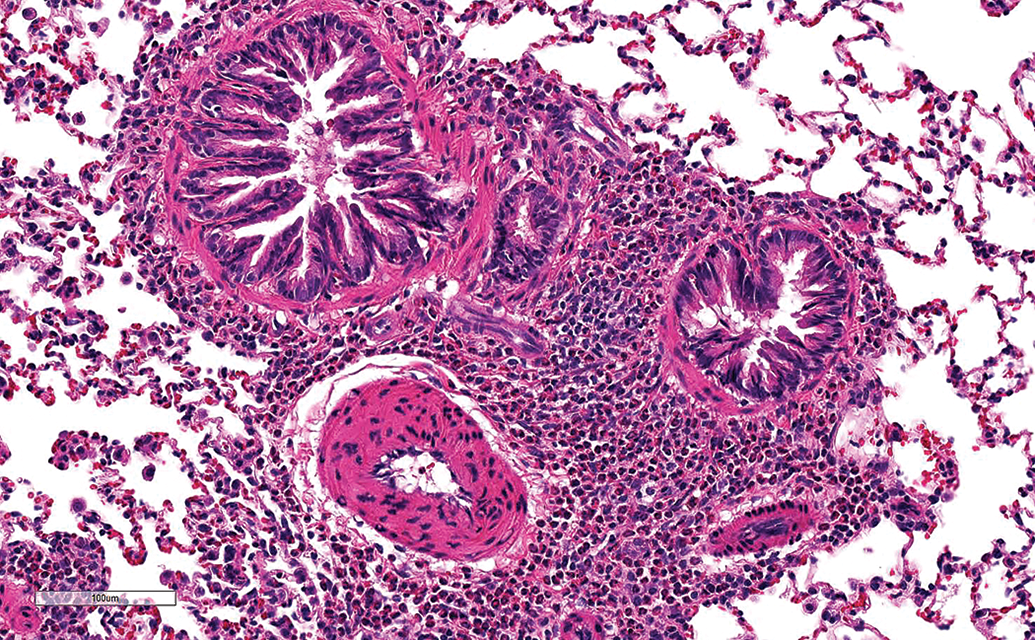
Low power H&E image of lung from a rat (case study 10) showing perivascular inflammatory cell infiltrate. Note arrangement of mononuclear inflammatory cells, predominantly lymphoid cells arranged around small vessels with scattered cells within alveoli. Inset—higher power view showing cellular details with perivascular location. H&E indicates hematoxylin and eosin.
High power H&E image of lung from a rat (case study 10) showing PV/PB inflammatory cell infiltrate composed mainly of eosinophils arranged around larger airways and arteries. H&E indicates hematoxylin and eosin; PV/PB, perivascular/peribronchiolar.
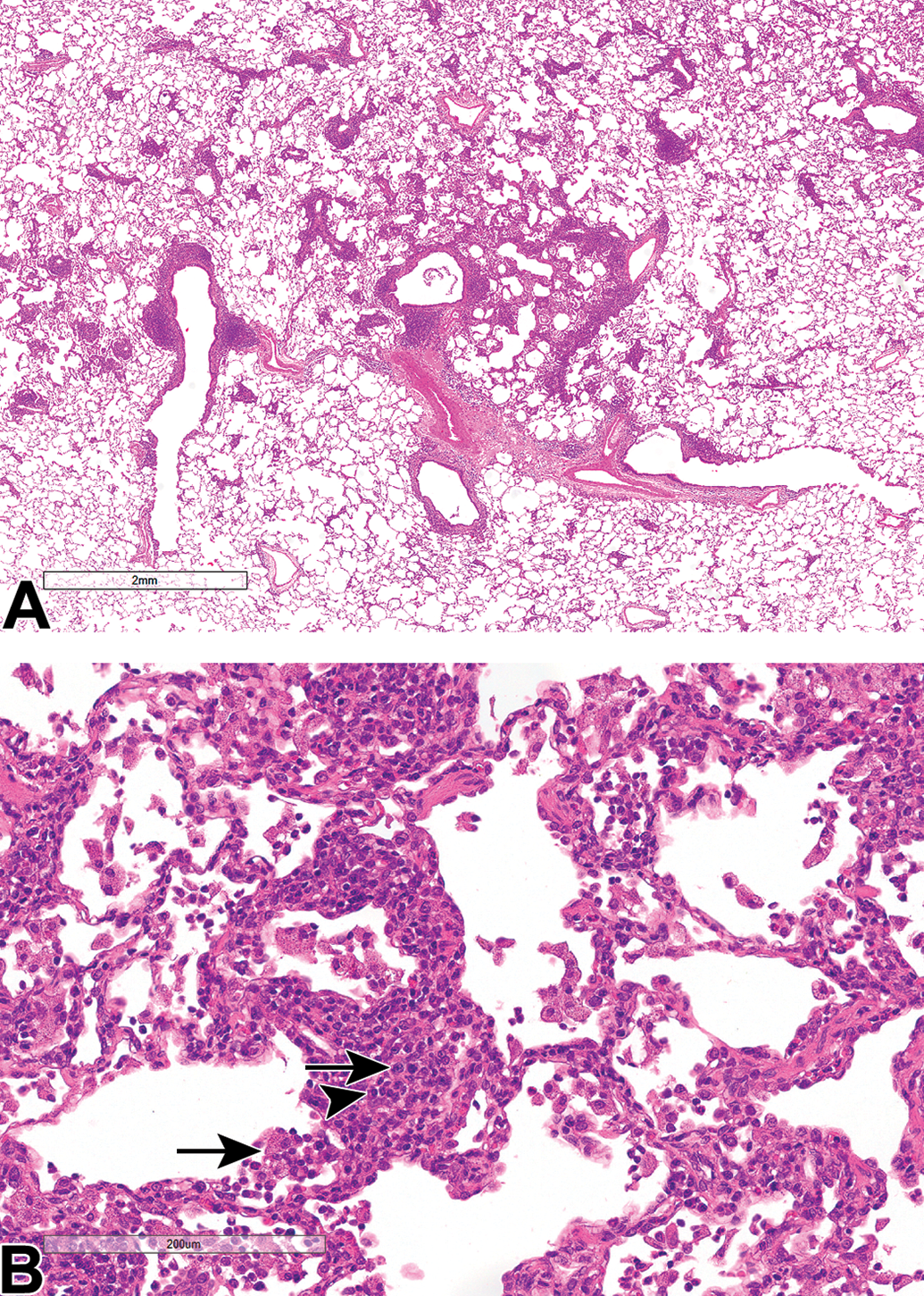
Low power H&E image of lung from an NHP (case study 11) showing (A) a more severe PV/PB and alveolar inflammatory cell infiltrate (compared to Figure 1), and (B) a higher power view showing details of PV/PB mononuclear cell inflammatory cell infiltrate (predominantly lymphocytes with fewer plasma cells [arrowheads] and macrophages [arrows]) and alveolar inflammatory cell infiltrate (consisting predominantly of macrophages with granular cytoplasm) extending into and distorting/thickening alveolar septa. H&E indicates hematoxylin and eosin; NHP, nonhuman primate; PV/PB, perivascular/peribronchiolar.
In 2 case studies (CS 2, 8-week NHP; CS 10, 4-week rat; and CS 10, 1- and 4-week NHP), minimal to mild PV/PB eosinophil infiltration (Figure 2) was noted (Table 3B). In CS 2, this was seen in NHPs treated with a Mab for 60 days and correlated with the minimal increase in eosinophils noted by BAL cytology. In CS 10, this change was seen in both the rat and NHP studies treated with a protein/peptide for up to 4 weeks.
Alveolar changes
Alveolar changes (Figures 3
–8) featuring increased alveolar macrophages (occasionally extending into terminal/respiratory bronchioles) were noted in 9 toxicology studies and were associated with 4 patterns of pathology characterized by (1) simple infiltrates of minimal to mild numbers of macrophages without any other changes (most often recorded as macrophage aggregation or infiltration; corresponding to INHAND term “alveolar macrophages, increased”); (2) alveolar mixed inflammatory cell infiltrates characterized by minimal to moderate infiltrates of macrophages associated with lymphocytes and fewer granulocytes but no signs of inflammation or tissue damage; (3) alveolar inflammation characterized by more severe (up to marked) infiltrates of macrophages, lymphocytes, and neutrophils which was associated with bronchioloalveolar hyperplasia/type II pneumocyte hyperplasia and/or pigment, hemorrhage, fibrin, erythrophagocytosis, and (alveolar) septal thickening, (ie, morphologic indicators or inflammation); and (4) alveolar macrophages associated with minimal to mild eosinophilic crystalline deposits (Table 3B). These changes were noted in studies ranging from 1 to 26 weeks in duration. Minimal alveolar macrophage accumulation/aggregation was noted in 3 case studies (CS 1, 2-week rabbit; CS 6, 13-week NHP; and CS 10, 1-week NHP) with no other accompanying alveolar changes. In CS 1, relatively large amounts of test article were inhaled (500 mg/kg/d); whereas in CS 6, the dose level was more typical of other inhalation studies in this series (high dose level—4.8 mg/kg/d). For CS 10 (1-week NHP, n = 3), a minimal alveolar macrophage aggregate was noted but this was not seen in the more highly powered 4-week toxicology study (n = 6). In 2 case studies (CS 8, 2-week rat and CS 10, rat 4-week), minimal multifocal eosinophilic crystal deposits (which likely represented protein) were reported and were closely associated with macrophage infiltration (Figure 8). Infiltration was characterized by scattered foci of macrophages engulfing and surrounding crystalline material without signs of inflammation or damage to surrounding alveoli or terminal airways. In CS 8, this change (ie, eosinophilic crystal deposits) was considered a background finding. In 2 other case studies (CS 5, 6-week NHP and CS 11, 13-week mouse), mixed alveolar inflammatory cell infiltration was present characterized by minimal to moderate alveolar macrophages together with lymphocytes and fewer granulocytes. In addition, in CS 5 (6-week NHP), eosinophils were seen in the alveolar inflammatory cell infiltrate which correlated with the increase in eosinophils seen by BAL cytology in the 26-week NHP study. No changes indicative of alveolar tissue damage (such as hemorrhage or bronchioloalveolar/type II pneumocyte hyperplasia) were present although reversible pleural inflammation and adhesions were seen in CS 5. In 4 case studies (CS 5, 26-week NHP; CS 7, 2-week NHP; CS 11, 13-week NHP; and CS 12, 26-week NHP), alveolar inflammation was present characterized by minimal to marked alveolar macrophage infiltrate together with smaller numbers of neutrophils and lymphocytes (Figure 3) and accompanied variably by evidence of morphologic indicators of inflammation (alveolar/endothelial damage [bronchioloalveolar/type II pneumocyte hyperplasia, hemorrhage, fibrin, erythrophagocytosis, alveolar septal thickening (by infiltrating leukocytes] and/or hemosiderin pigment).
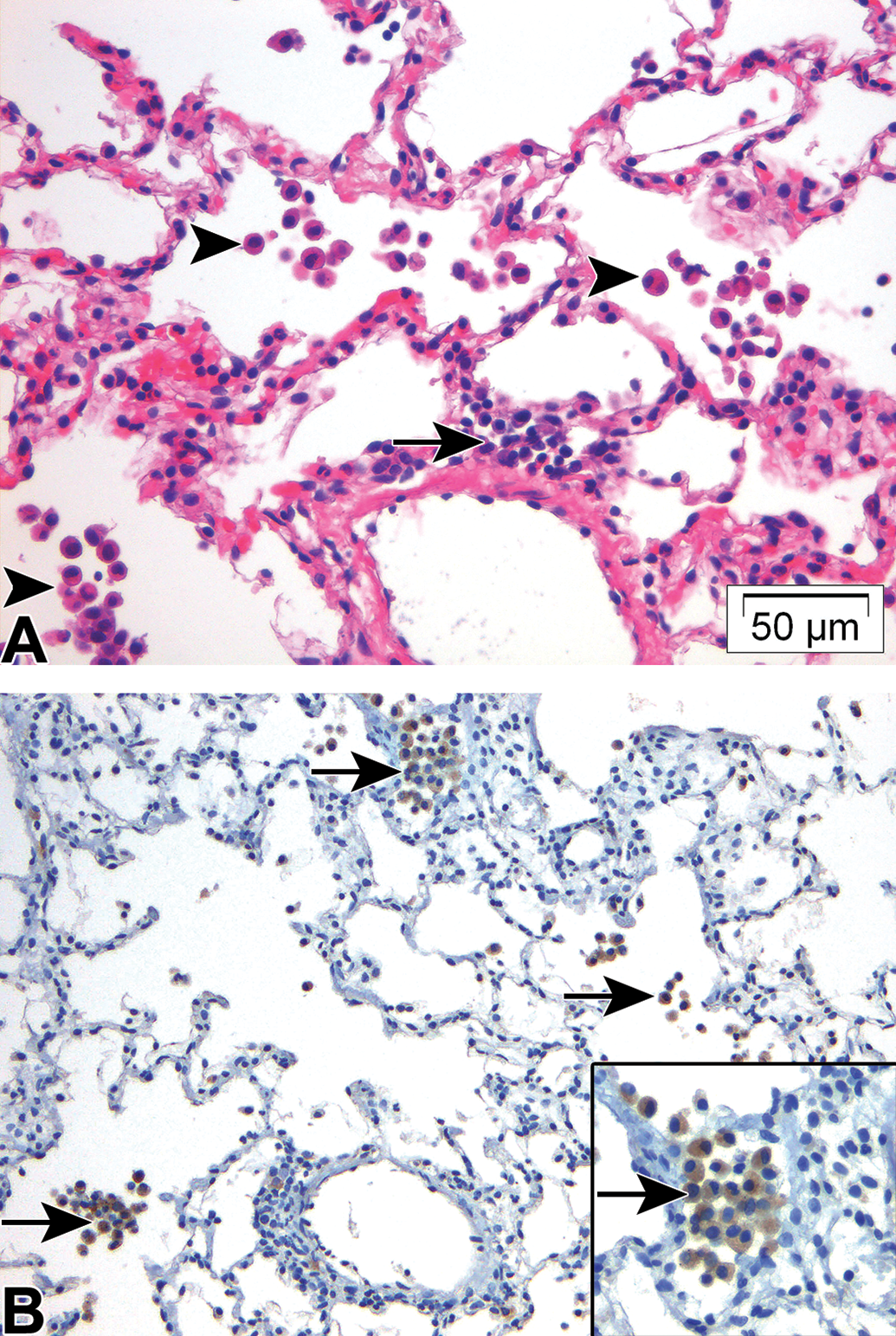
A, Low power H&E image of lung from NHP (case study 5) showing increased alveolar macrophages with smaller numbers of perivascular mononuclear cells (arrows) and (B) high power IHC image from the same study showing CD68 positively stained macrophages (arrows). Inset shows high power details of alveolar macrophages. Note relatively few cells in perivascular infiltrates stain positively for CD68. H&E indicates hematoxylin and eosin; IHC, immunohistochemistry; NHP, nonhuman primate.
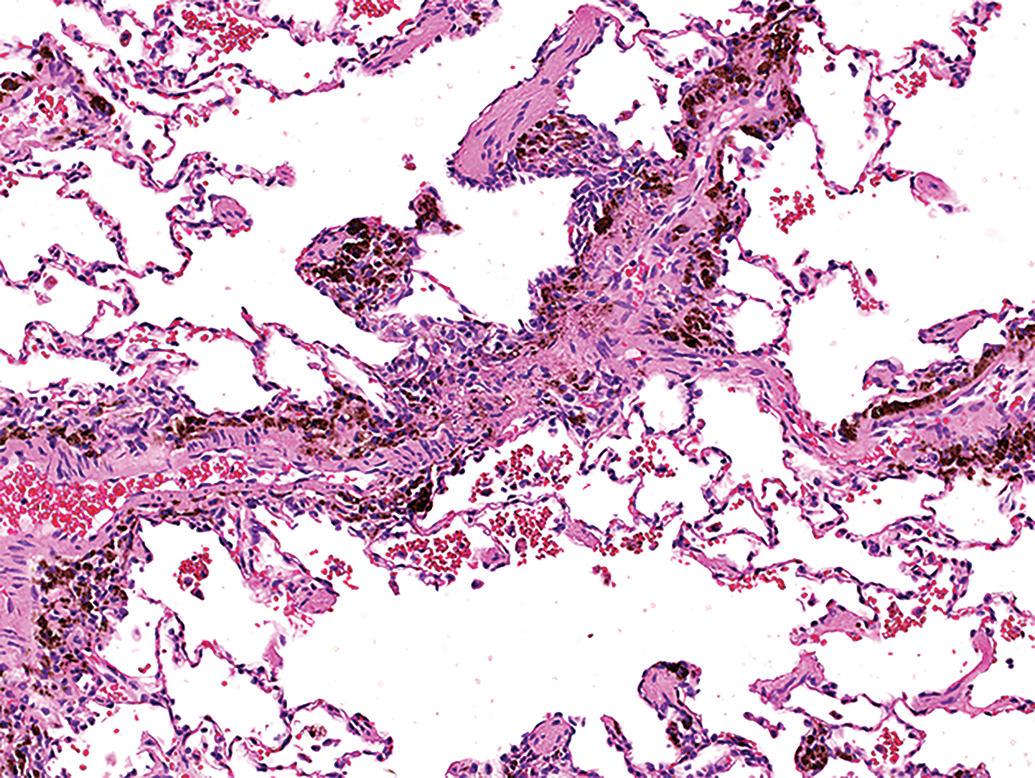
Low power H&E image of lung from an NHP (case study 12) diagnosed with immune complex disease showing hemosiderin pigment in a perivascular location together with alveolar hemorrhage. H&E indicates hematoxylin and eosin; NHP, nonhuman primate.
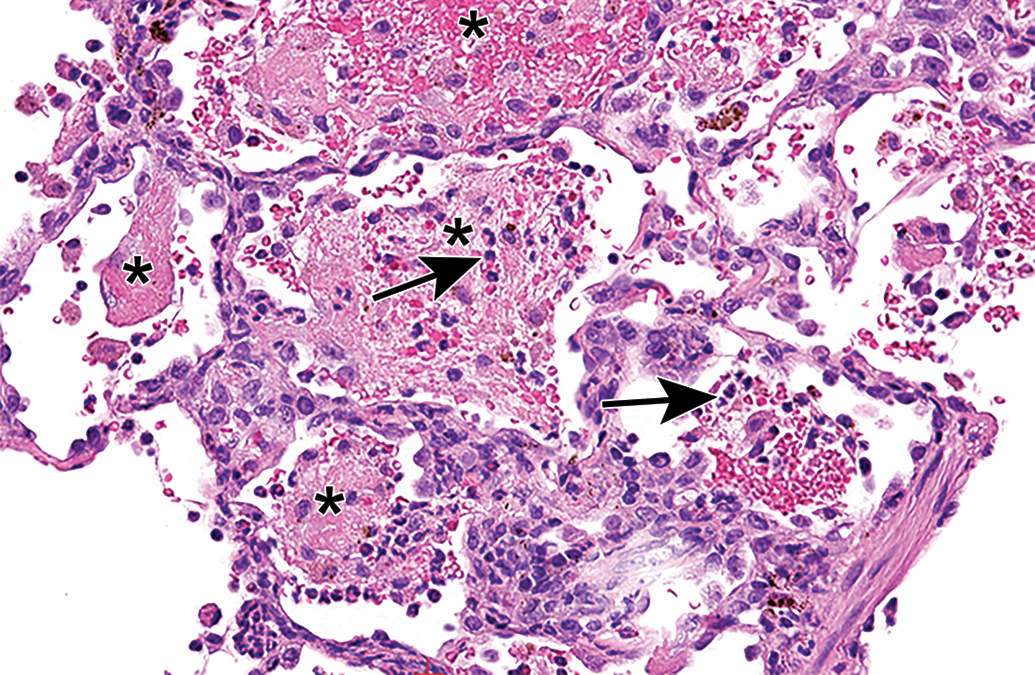
High power H&E image of lung from an NHP (case study 12) diagnosed with immune complex disease showing alveolar mixed inflammatory cell infiltrate (arrows) with hemorrhage, pigment, and fibrin (*). H&E indicates hematoxylin and eosin; NHP, nonhuman primate.
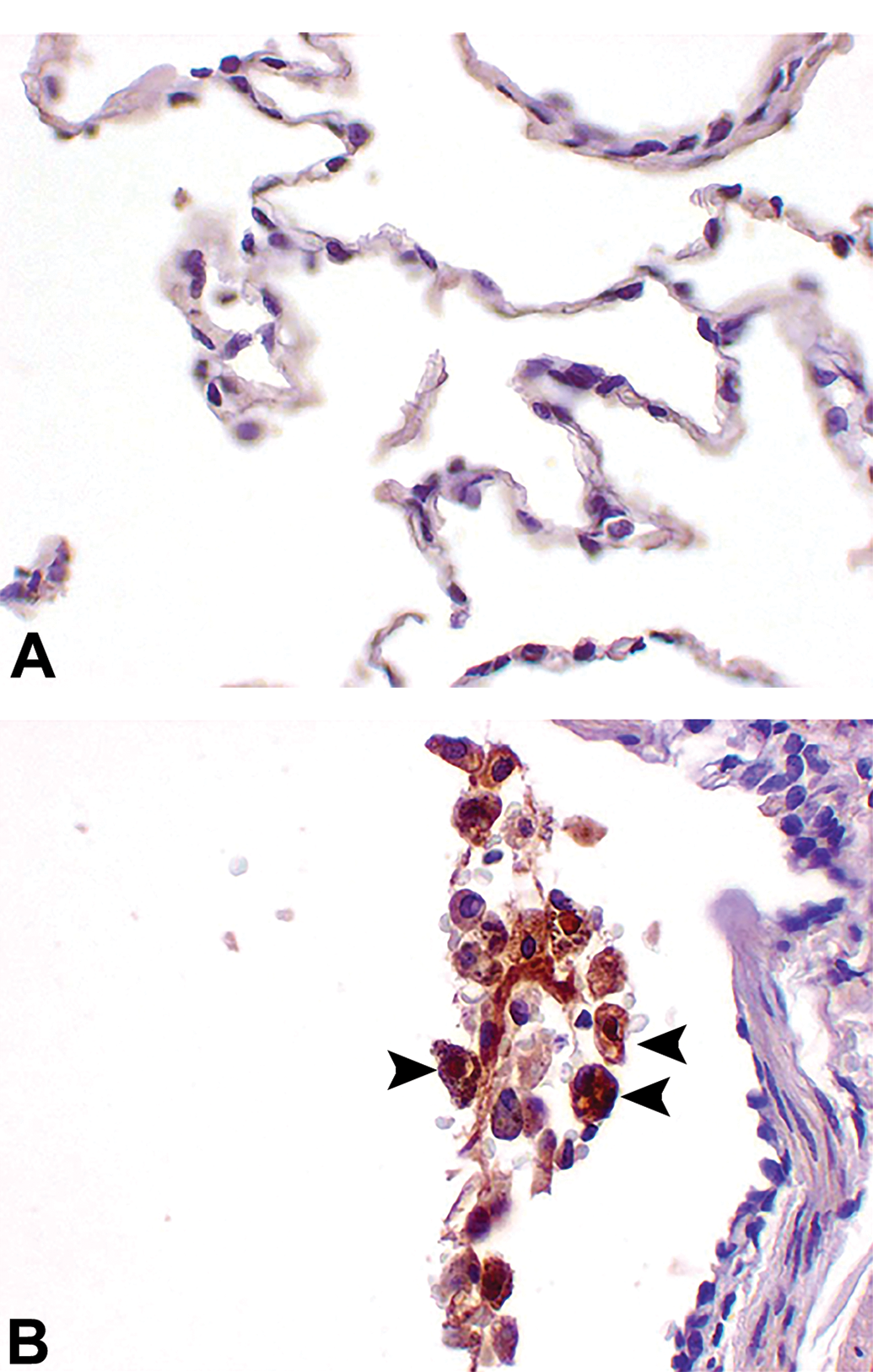
High power IHC stained image of lung from an NHP (case study 12) stained with an antihuman IgG to recognize the test article (humanized Fab) from (A) control animal and (B) an animal treated with the test article at 20 mg/kg/d. Note strong staining of alveolar macrophages in treated animal indicating endocytic update of test article (arrowheads). IgG indicates immunoglobulin G; IHC, immunohistochemistry; NHP, nonhuman primate.
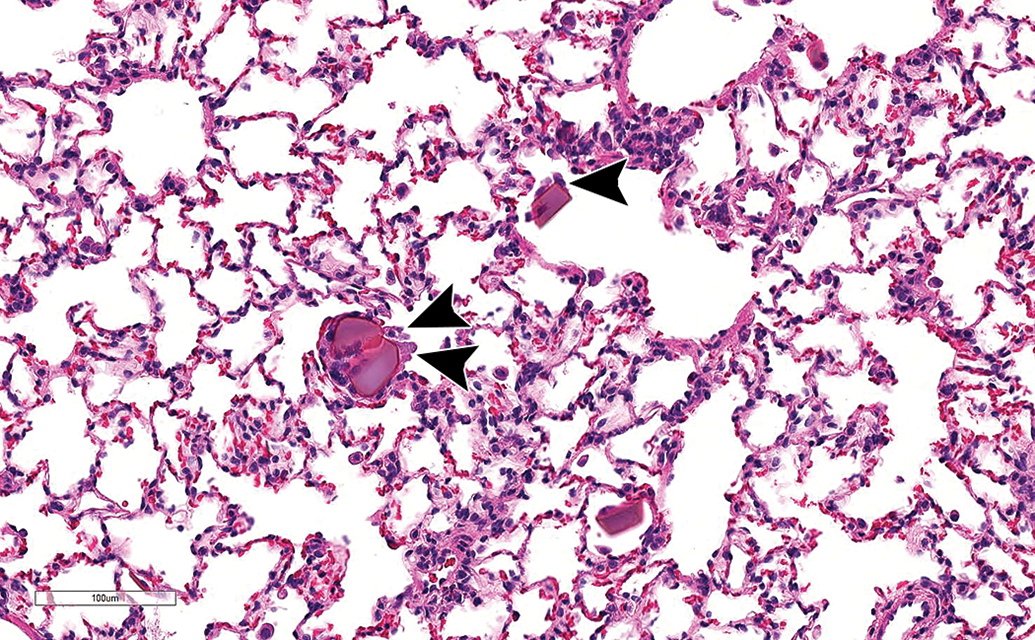
Medium power H&E image of lung from a rat (case study 10) showing alveolar eosinophilic crystals with small numbers of attached macrophages (arrowheads). H&E indicates hematoxylin and eosin.
Draining lymph nodes
Minimal to moderate increased cellularity (lymphoid hyperplasia) was noted in the draining (tracheobronchial/mediastinal) lymph nodes of 9 toxicology studies (Table 3B) characterized by increased lymphoid cellularity of the paracortex and/or germinal centers. These changes were apparent after 2 weeks of treatment and were present in studies up to 26 weeks in duration.
Nasal cavities
Changes in the nasal cavities were seen in 3 toxicology studies. In CS 5 (6-week NHP), minimal to mild hyperplasia of the nasal associated lymphoid tissue (NALT) was noted together with lymphoid hyperplasia of the BALT in the lung.
In CS 10 (1- and 4-week NHP), minimal to moderate inflammation within the nasal cavity was seen. This was accompanied by minimal degeneration/erosion and squamous metaplasia/hyperplasia of the respiratory epithelium together with neutrophilic infiltration and squamous metaplasia of the larynx (in 1 high dose animal).
Recovery
Partial recovery was noted in the majority of studies for those that included a recovery phase, with a tendency for the lymphoid changes (PV/PB MIC infiltrates in the lung and lymphoid hyperplasia in the draining lymph node) to persist as might be expected for an immune response with inherent memory effects. However, full recovery of increased alveolar macrophages, PV/PB MIC infiltrates, and increased cellularity of the draining lymph node was observed when given sufficient time (CS 6, 13-week NHP; Table 3B).
Differences between biologics and small molecules (new chemical entities)
Comparison of the pattern and frequency of changes induced by inhaled biologics (seen in these case studies) to those induced by inhaled small molecules (new chemical entities [NCEs]; seen in the companion review, Gregori et al, which featured 81 inhalation studies) highlighted similarities in the pathology induced in the lungs, but there were differences in the frequency and anatomic distribution of induced lung changes. Additionally, there were differences in the frequency of responses of other tissues to inhaled NCEs (particularly in the nasal cavity, trachea and larynx).
Similarities in pathology indicated common and limited patterns of response of the respiratory tract to inhaled molecules, whereas differences in frequency and anatomic distribution were considered to reflect differences in the mechanism of the induced change. For small molecules, “irritancy” and clearance of poorly soluble particles were significant contributing factors to toxicity, whereas with inhaled biologics, other mechanisms appeared to be important (see “Part 3: Mechanisms of Toxicity” section).
In the lungs, the most frequently induced change with inhaled NCEs (seen in approximately 20% to 50% of studies in rats and NHPs) was increased alveolar macrophages (Gregori et al, this issue), which most likely represented a clearance response to more poorly soluble particles. Similar uncomplicated increased alveolar macrophages were also seen with inhaled biologics and likely reflected macrophage clearance of test article. However, the frequency of PV/PB MIC infiltrates in both NHPs and rats induced by NCEs was relatively low (30% or 3/10 NHP studies and 3.4% of rat studies) compared to inhaled biologics where these changes were seen in 72% of toxicology studies. Of note, in 2 of the 3 NHP NCE studies, the PV/PB MIC changes were likely secondary to the immunomodulatory action of the drugs, that is, a pharmacologic response rather than an immune response per se.
In addition, in the NHP, induced changes in the nasal cavity (erosion/ulceration) and trachea (squamous metaplasia and inflammatory cell infiltrates) were relatively common (20% of studies) with inhaled NCEs, whereas in our series of case studies, degenerative changes in the upper respiratory tract were relatively rare (recorded in only 1 CS [8.3%]). In the rat, unlike in our case studies, changes associated with NCE inhalation in the larynx (squamous metaplasia and cartilage necrosis) and nasal cavity (goblet cell hyperplasia and squamous metaplasia) were relatively common (seen in up to 20%-30% of studies; Gregori et al, this issue) most likely reflecting an “irritant” effect of poorly soluble particles.
Part 2: Antidrug Antibodies
Overview
The production of ADA, due to a host immune response against foreign epitopes present in large molecule therapeutics, occurs frequently in both animal and human settings. 6,7 The frequency of occurrence and the magnitude of the response to biotherapeutics vary considerably due to a plethora of risk factors—biophysical (protein sequence, posttranslational modifications, formulation, aggregates, and impurities), biological (dose, route, frequency of administration, and therapeutic target), and patient-specific factors (such as the disease state, concurrent illness or medications, and genetic background) 8 as well as others (reviewed in Krishna and Nadler 6 ). These risk factors combine to influence the immunogenicity of the molecule. 6 All these factors ultimately converge on the adaptive immune system resulting in the expansion and selection of antigen-specific T and B cells and recruitment to sites of antigen exposure. B cells, in the form of plasma cells, secrete antibodies (including secretory immunoglobulin A [IgA] at mucosal sites) that bind to the drug target in an attempt to eliminate it. The consequences of this varies in terms of both pharmacokinetics/pharmacodynamics (PK/PD), that is, loss or increase in systemic exposure and consequently loss or enhanced therapeutic response. Less frequently, these changes result in safety implications. Safety concerns include infusion reactions and ICD. 9,10 Also, cross-reactive neutralization of endogenous proteins that mediate critical biologic functions 11 such as recombinant human factor VIIa, 12 thrombopoietin, 13 and erythropoietin 14,15 have been demonstrated.
Is the inhalation route likely to be more immunogenic than other routes?
The respiratory system has evolved a number of strategies to overcome microbial invasion such as physical barriers, secreted antimicrobial factors, and most importantly, a robust innate and adaptive immune system. 16,17 Given the variety of factors that influence immunogenicity and the priming of the respiratory system to respond to infection, it is not surprising that the route of delivery might be a critical factor influencing the overall immunogenicity of a molecule. However, while the inhalation route, together with the intradermal and subcutaneous routes, might be associated with increased immunogenicity compared to the intravenous and oral routes, 18,19 there is a paucity of published literature to confirm this and to describe the degree of immunogenicity that might be expected from this route.
Early studies demonstrated that the mucosal surface might act to induce immunological tolerance, at least in the context of sensitization to aeroallergens. 20 This contention was somewhat supported by the early literature in which the route of exposure was thought not to dramatically alter immunogenicity as it neither conferred 21 nor negated immunogenicity. 22 For example, daily exposure of mice for 3 weeks to human growth hormone (GH) resulted in the generation of ADAs and demonstrated that the inhalation route was less immunogenic than the parenteral route due to the development of lower serum titers, even after matching for total absorbed systemic exposure. 7
However, clinical data using inhaled dry powder human insulin 23,24 and inhaled nebulized Xolair (Omalizumab) showed the opposite effect. In 1 clinical trial, 1 to 10 mg of nebulized Xolair (Omalizumab) was administered for 8 weeks to 33 human patients with mild allergic asthma. This resulted in 1 patient treated at 10 mg (group size, n = 10) developing serum immunoglobulin A (IgA) and immunoglobulin G (IgG) antibodies from day 28, while 4 other patients at 10 mg had loss of exposure at day 56 but no serum antibodies could be detected. The authors speculated that the inhalation route might be more immunogenic than the parenteral route as significant ADA had never been detected following intravenous administration. 25 However, these results may have been influenced by biophysical factors such as aggregation that was not evaluated and may have contributed to the increased immunogenicity of the aerosolized product. 26,27
In another clinical trial, treatment with Exubera by both the subcutaneous and inhaled route demonstrated increased antibody binding to insulin (ADA) via the inhalation route that peaked after 6 to 12 months of exposure. The ADA production was greatest for patients with type I diabetes compared to those with type II diabetes. No increases in ADA were noted for patients with type II diabetes who had not previously used insulin, 28 indicating that prior exposure was required to see differences in ADA response between these 2 routes of exposure. Increased ADAs were maintained for the duration of the 2-year extension trial but were not associated with any safety, hypersensitivity, pulmonary function, or efficacy effects. 28
Nonclinical Case Studies Are Associated With High Levels of Immunogenicity
Analysis of the case studies showed that the inhalation route was indeed highly immunogenic and that immunogenicity (indicated by detection of ADA in BAL fluid [BALF] or serum) was associated with increased presence of lymphocytes in the lungs (recorded as PV/PB MIC infiltrates) and draining lymph nodes (increased cellularity).
In this current series of 12 case studies, immunogenicity was assessed either systemically and/or in BALF in 15 of 18 toxicology studies. Our data revealed a high frequency of ADA with a trend toward increasing frequency of ADA positive animals in studies of longer duration (Figure 9). For studies where ADA data were reported, no ADA was detectable after 1 week of treatment in 2 case studies. This rose to an intermediate frequency of ADA positive animals after 2 weeks of treatment in 2 further case studies and a high or very high frequency of ADA after 4 or more weeks of treatment in 4 case studies, with 100% frequency in 4 case studies of 6 to 26 weeks of duration (Table 3A and Figure 9). These data are consistent with the increased likelihood of breaking tolerance and induction of ADAs after repeated dosing or increased drug exposure 6 as well as the clinical picture seen in humans with inhaled Exubera where peak ADA production occurs after 6 to 12 months of treatment. 28
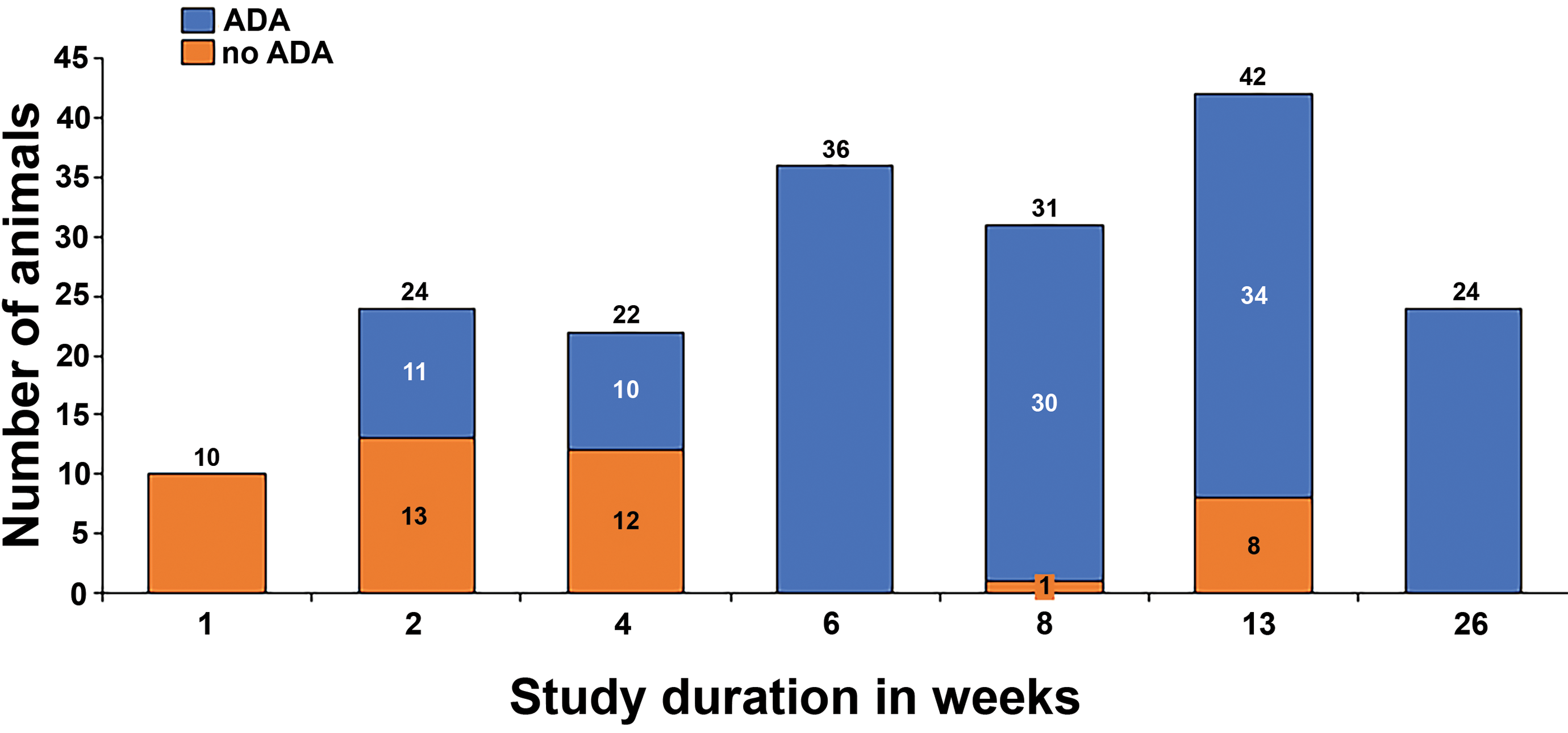
Incidence of immunogenicity showing the number of ADA positive (orange) and ADA negative (blue) NHPs versus study duration based on 8 case studies (12 toxicology studies). ADA indicates anti-drug antibody; NHPs, nonhuman primates.
The lack of immunogenicity (ADA) in the two 1-week studies is consistent with the expected kinetics of an immune response. Experimental intratracheal immunization of mice with sheep red blood cells required 5 days before expansion of the regional lymph nodes could be detected (via a plaque formation assay) 29 with at least 2 more days required to see the accumulation of lymphocytes within the respiratory tract. Lymphocyte infiltration was characterized by perivascular infiltrates of lymphocytes around small vessels (ie, postcapillary venules). 5 Recruitment is now known to be co-ordinated by specific adhesion receptors on high endothelial venules that ensure homing from the local draining lymph node back to the original source of antigen exposure. 30,31 In some cases, 2 immunizations are required, 29 resulting in local, lobe specific, mucosal immunity 30 initiated and maintained by the accumulation and proliferation of effector lymphocytes. 32 In CS 5, proliferation of local lymphocytes (BALT hyperplasia) was measured immunohistochemically (Figure 10) and was seen to occur in parallel to ADA synthesis throughout the duration of treatment (weeks 6 to 26—albeit at reduced levels at 26 weeks compared to 6 weeks) consistent with Kaltreider’s 5 initial observations suggesting that the PV/PB infiltrates represent a local immune response to the inhaled protein.
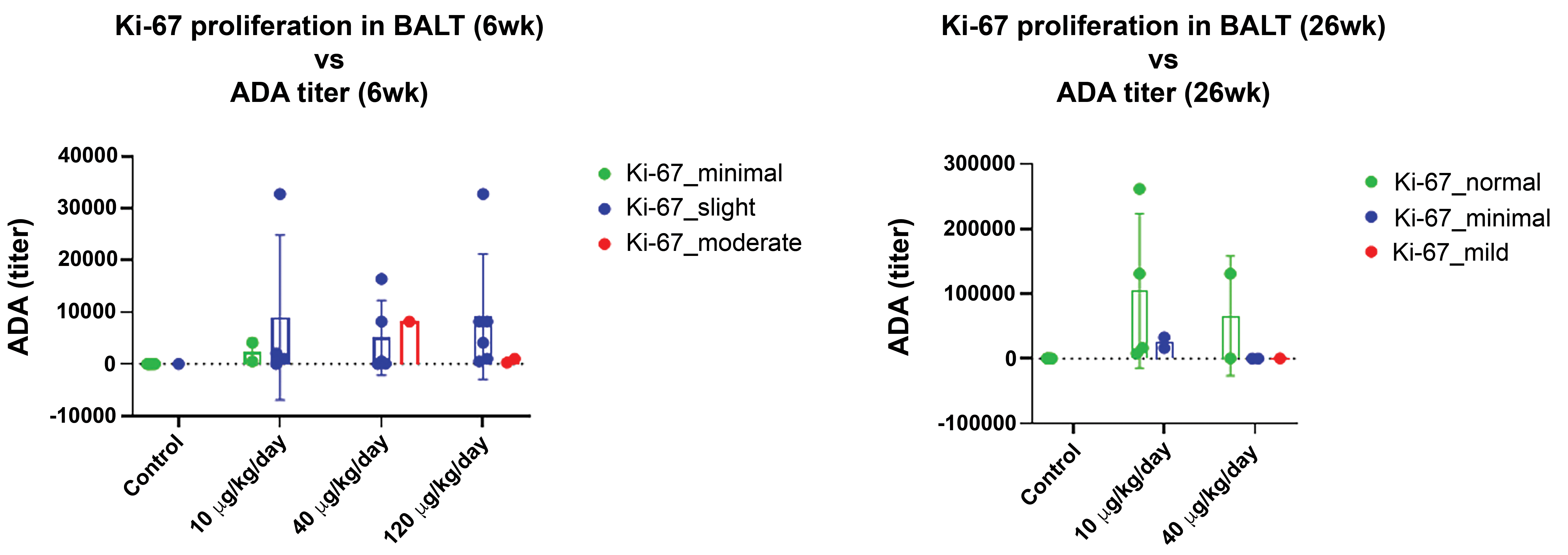
Graph illustrating Ki67 IHC proliferation marker versus ADA titer and dose after 6 week and 26 weeks of treatment. ADA indicates anti-drug antibody; IHC, immunohistochemistry.
Further analysis of the two, 1-week studies (CS 3, 1-week NHP and CS 10, 1-week NHP) also demonstrated that ADA production was closely correlated with the appearance of PV/PB lymphocytes as both the 1-week studies showed no pathology findings (CS 3) or very limited pathology (1 animal in the high dose group with minimal mononuclear infiltrates, CS 10). However, after 2 weeks of treatment (CS 3), ADA and PV/PB/alveolar lymphocytic infiltrates (with increased lymphoid cellularity of tracheobronchial lymph nodes) were seen which strongly suggested that PV/PB infiltrates are the source of ADA and that approximately 2 weeks are required to detect this change histologically. Finally, in the 4-week rat study (CS 10), there was again a general absence of ADA (detected in only 1 high dose rat) with no accompanying PV/PB MIC infiltrates.
For CS 9, ADA production was measured in 2 studies that compared the inhalation route to the subcutaneous dose route. The studies were matched for dosing duration, and exposure and ADA titers were measured using the same ADA detection format. A higher frequency of serum ADA (with 100% incidence in the mid-dose group) was seen with the inhalation route compared to the subcutaneous route, where only a minimal incidence of ADA was seen (Figure 11).
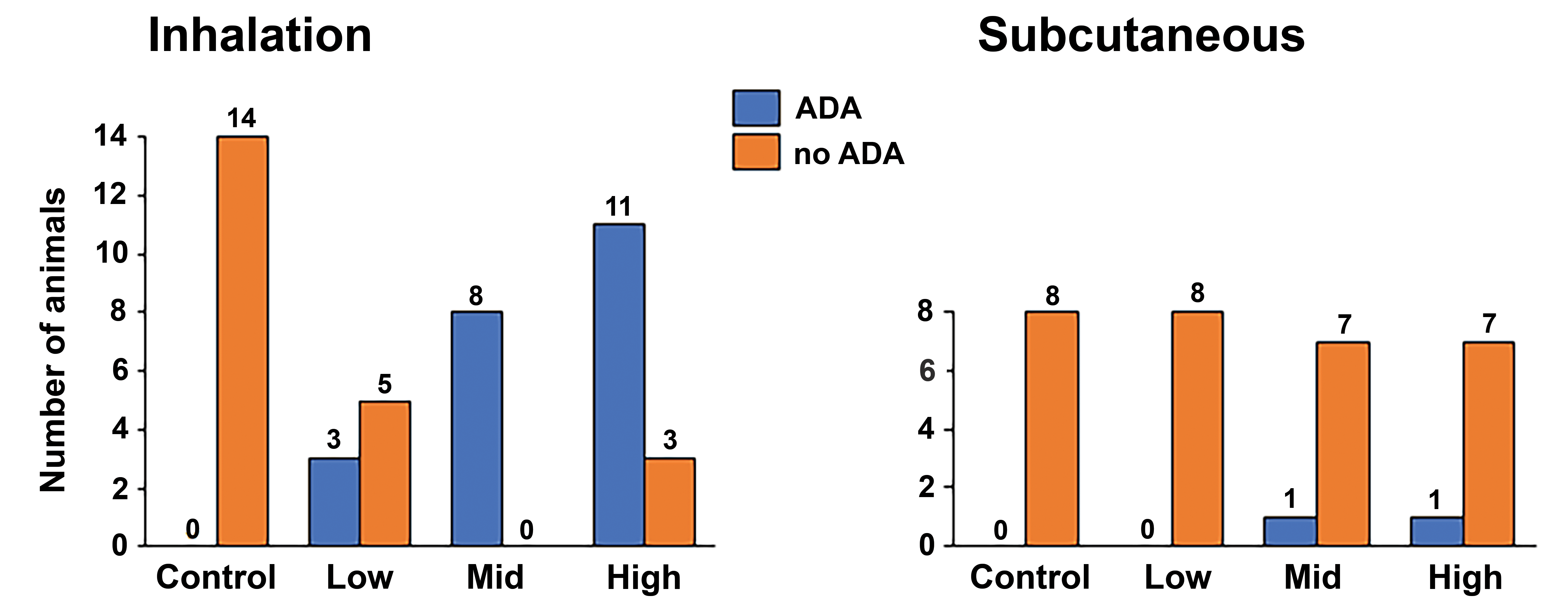
Case study 9 showing comparative incidence of ADA absence (blue) or presence (orange) following administration of a peptide/protein via the inhalation or subcutaneous route for 3 months. ADA indicates anti-drug antibody.
Finally, correlation of ADA data with microscopic findings using aggregated results from 138 NHPs split across 10 toxicology studies constituting 6 case studies (Figure 12) revealed that 103 NHPs developed PV/PB MIC of which 77 animals (75%) also developed ADA, indicating that immunogenicity was positively associated with these infiltrates. Furthermore, PV/PB MIC infiltration was positively associated with increased lymphoid cellularity of the tracheobronchial lymph nodes since 92% of NHPs with enlarged tracheobronchial lymph nodes also had PV/PB MIC infiltrates.
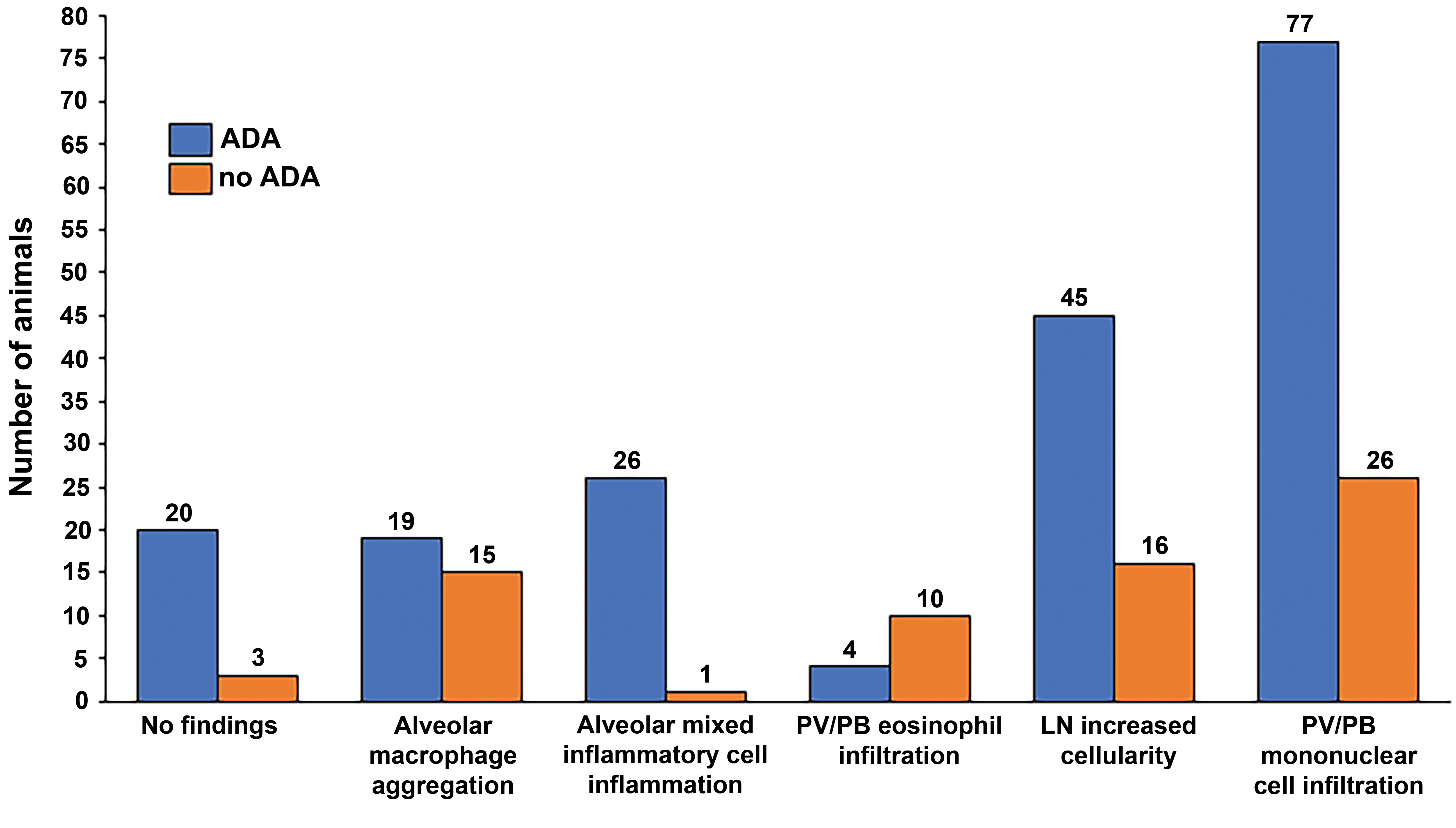
Incidence of immunogenicity (ADA positive [orange] and ADA negative [blue]) versus microscopic findings in 138 treated NHPs from 10 toxicology studies where raw data were available. ADA indicates anti-drug antibody; NHP, nonhuman primate.
Similar analysis of alveolar mixed inflammatory cell infiltration also showed a very high correlation with ADA status. Alveolar mixed inflammatory cell infiltration was less frequently observed with an incidence of 20% (27 animals) in the reviewed toxicology studies. However, all but 1 animal that developed alveolar mixed inflammatory cell infiltration was ADA positive (96%; Figure 12), indicating a very high association between ADA positivity and alveolar mixed inflammatory cell infiltration, consistent with immune stimulation as a factor in this change (discussed in Part 3: Mechanisms of Toxicity section). However, this latter data set was relatively limited in size, so further work is required to corroborate the significance of this mechanism.
Inhaled biologics: the translation of ADA formation and immune-mediated pathology observed in animals to risk in humans
Current thinking states that immune-mediated responses in the form of ADAs or immunopathology observed in animals following administration of a biologic are not necessarily predictive of the same reaction occurring in humans. 9 However, whenever ADAs are detected, their impact on the interpretation of the study results should be assessed (ICH, S6). 33 This statement is likely as applicable to the inhalation route of administration as for other routes (ie, subcutaneous, intradermal and intravenous), since the same basic immunological mechanisms are common to all routes. As such, a fully humanized biologic, identical to the endogenous native human protein, may be recognized as “foreign or non-self” and elicit an immune reaction in test animals, but it is more likely to be recognized as “self” and nonimmunogenic in humans. The risk of idiosyncratic immune reactions in humans cannot currently be predicted by testing in animals. 34 Similarly, the immunogenicity response in humans is also not entirely predictable since treatment of type I and type II diabetic patients with Exubera resulted in immunogenicity and anti-insulin antibodies in some clinical trials but not in others. 35
The unpredictable nature of ADA and lack of correlation between clinical and nonclinical data are further illustrated by Pulmozyme (dornase alfa; recombinant human deoxyribonuclease [rhDNase]), an inhaled mucolytic used primarily in pediatric patients with cystic fibrosis. In 4-week and 26-week studies, rats and NHPs showed an increased incidence or severity of pulmonary lesions with elevated antibody titers. 36 However, clinical trials in pediatric patients up to 2 years in duration showed that in humans, Pulmozyme was well tolerated and adverse events were rare (<1/1000). Significantly, less than 5% of patients treated with Pulmozyme developed antibodies to the drug and none developed immunoglobulin E antibodies. 36 The lack of significant immunogenicity in patients is likely due to the use of a fully human recombinant product.
In general, animal studies measuring immunogenicity in response to humanized biotherapeutics have limited predictive power for human patients. However, these studies may not be entirely without merit as the data generated from our case studies indicate that the inhalation route is highly immunogenic in animals. Although limited immunogenicity in the case of inhaled Exubera and Pulmozyme did not result in adverse events in humans or changes in pulmonary function, 28,37 it would be prudent to ensure careful and robust monitoring of ADA during clinical trials of inhaled biotherapeutics. Should immunogenicity be detected in human patients, it is possible that similar types of pathology to that encountered in animal studies would occur. The Food and Drug Administration (FDA) guideline states: “The extent of information required to perform a risk-benefit assessment will vary among individual products, depending on product origin and features, the immune responses of concern, the target disease indication, and the proposed patient population” (FDA, Guidance to Industry, 2014). 38 One should therefore be aware of a potentially increased risk of immunogenicity via the respiratory route during the design of new candidate molecules, especially if a high frequency of immunogenicity was encountered in the animal studies. As is typical for safety assessment, a case-by-case approach should be adopted with careful assessment of the potential impact and translation of ADA from the nonclinical to clinical stage.
Part 3: Mechanisms of Toxicity
Structure and function of BALT
The lung has an enormous surface area which provides opportunity for contamination with particles and microbes. Although the upper airways are colonized by commensal bacteria, viruses, phages, and fungi (the lung microbiome) mostly from inhaled particles, in the healthy individual, the lower airways are sterile. The microbiome remains in check by a very efficient system of host defenses (cough, epithelial barrier, mucociliary escalator, innate immunity, acquired immunity, and secreted host factors). 39 Anatomically, the BALT is located in the mucosa beneath the epithelium and in the lamina propria around the arteries and airways. It is present in all mammalian species developing in some constitutively as a secondary lymphoid organ or, as in humans and in the mouse, as a tertiary lymphoid organ after induction by inflammation or infection. 40 In these latter species, BALT is more loosely arranged throughout the lung, often adjacent to the pulmonary artery. 40 Bronchus-associated lymphoid tissue is not present in the fetus but develops postnatally, resulting in increased numbers of lymphocytes within the first 2 weeks of life with discrete B and T cell areas developing by weeks 4 to 12. 41
Pivotal to the acquired immune response is the BALT, which is composed of a zone of mononuclear cells (B cells, T cells, macrophages, and dendritic cells) that form loose aggregates around high endothelial venules. 41 Its function is to sample and respond to antigen, acquired locally either through microbial invasion, the lymphatic system, or via specialized microfold (M) cells located in dome epithelium. It is therefore primed to respond to foreign particles (pathogens, foreign proteins, etc) and does so by increasing its size and prominence (physiological hyperplasia). 41 –43 As a result, BALT differs in appearance between conventional and germ-free housed animals. 41
Immunogenicity as a Mechanism for the PV/PB MIC Infiltrates
As previously discussed, the inhaled platform appears to be a highly immunogenic route of delivery resulting in a high frequency of ADA positive animals and a good correlation between ADA positive status and presence of PV/PB lymphoid infiltrates. The histological pattern of PV/PB lymphoid infiltration, together with enlarged regional lymph nodes and NALT hyperplasia in CS 5, plus the kinetics of immune reactions in the lung strongly indicate that the PV/PB lymphoid infiltrates represent a local immune response to inhaled foreign proteins or nucleic acids that are apparent histologically after about 2 weeks of treatment. However, it should be noted that BALT reactions are not exclusively initiated or maintained by an immune response since inflammatory reactions can also give rise to BALT hyperplasia. Immune stimulation and inflammation serve to enhance the function of the other; inflammation increasing the exposure of the immune system to antigens, whereas injured cells release proinflammatory molecules and danger signals that further stimulate inflammation and activate dendritic cells to induce an immune response. 44 Once induced, inducible BALT may persist for months and participate in immune responses. 45
Airway damage/inflammation was not a consistent driver for PV/PB MIC infiltrates
In our series of case studies, 5 case studies (3, 4, 6, 9, and 10) consisting of 9 toxicology studies in total did not show any changes indicative of “damage” (ie, degeneration, necrosis, or inflammation in the airways or alveoli), even after 26 weeks of treatment. In a further 2 case studies (CS 5, 6-week NHP and CS 11, 13-week mouse), PV/PB MIC infiltrates were seen together with alveolar mixed inflammatory cell infiltrates (consisting of macrophages with lymphocytes and neutrophils) without morphologic changes indicative of damage/inflammation indicating that alveolar damage per se was not responsible for the PV/PB changes. It is therefore unlikely that in these case studies, damage/inflammation was a driver for the observed PV/PB changes. However, 3 case studies, comprised of 3 toxicology studies, showed PV/PB changes accompanied by alveolar mixed inflammatory cell infiltration with bronchioloalveolar hyperplasia (CS 7) and/or alveolar hemorrhage/pigment/type II pneumocyte hyperplasia (CS 11, 13-week NHP) and/or alveolar septal thickening (CS 12). In these cases, it was not possible to distinguish whether inflammation or immunogenicity was the main driver for the PV/PB changes.
Immunogenicity related to foreignness induces PV/PB MIC infiltrates
Repeated inhalation of nebulized protein (eg, ovalbumin or human GH) results in robust anatomic changes characterized by perivascular lymphoid cuffing in the adventitia of small and medium sized pulmonary veins. 7,46 These changes are very similar to those seen in experimental immunization of mice, 29 dogs, and NHPs, 30 and morphologically and temporally similar to the PV/PB MIC infiltrates seen in our case studies. Not surprisingly, inhalation of the marketed drug Pulmozyme (rhDNase), which shows low (80%) sequence homology in the rat, was associated with both ADA production and PV/PB MIC infiltrates and alveolitis in the rat and NHP, 47 whereas inhalation of insulin which has much higher sequence homology (98.1% in the NHP) resulted in no adverse pathology or significant ADA production (reviewed in Appendix A). 23,24,48 –50 As might be expected, intravenous administration of rhDNase to rats and NHPs resulted in no treatment-related histological changes in the lungs 47 consistent with lack of local immunogenicity. Similarly, no adverse effects have been observed in patients treated for up to 2 years with rhDNase/Pulmozyme (presumably due to lack of significant immunogenicity) even in patients in which ADAs have been detected. 50 However, it should be noted that mild inflammatory changes may be difficult to detect in the intended patient population (cystic fibrosis) but, despite this, no significant adverse changes are likely given that lung function improved over the clinical trial period. 37 A similar pattern of changes was seen in experimentally administered inhaled bovine GH to rats, which produced no PV/PB lung changes, whereas treatment with inhaled human GH, which shows only 69% sequence homology to the rat, 51 resulted in PV/PB lymphoid (mononuclear cell) cuffing. 7,37
Inhaled NCEs induce a lower frequency of PV/PB MIC infiltrates compared to biologics
Finally, analysis of the lung changes induced by NCEs in a large multicenter study consisting of 81 inhalation studies (made up of 44 unique NCE test articles) showed that PV/PB MIC changes in the lung were a relatively infrequent finding in the rat (0% and 6.9% study incidence in Sprague-Dawley and Han Wistar rats, respectively). This difference in incidence likely reflects an absence of immunogenicity associated with small molecules. In NHPs, the incidence was 30% (3 of 10 studies; Gregori et al, this issue). In this latter case, further analysis showed that the PV/PB MIC infiltrates were accompanied by lymphoid hyperplasia of the draining lymph nodes and were associated in 2 cases with pharmacologic modulation of the target biology, as 2 of the 3 NCEs were immunomodulators.
Clearance as a Mechanism of Alveolar Macrophages Accumulation
Particle deposition
Particles deposit in the lung according to defined parameters determined by the patient (breathing pattern, lung disease/lung geometry) and the nature of the inhaled particles (size, size distribution, shape, charge, density, and hygroscopicity). 52 Deposition occurs primarily through 3 well-defined mechanisms, namely inertial impaction, sedimentation, and diffusion which are largely determined by particle size, 52 although other parameters such as particle shape and electrostatic charge influence these mechanics. 53 These properties are often bioengineered into medicinal products in order to target specific regions of the lung with greater accuracy. In our series of 12 case studies, all but 1 CS (16 of 18 toxicology studies) reported a mass median aerodynamic diameters (MMADs) <5 µm, which would result in a greater proportion of particles deposited within the terminal airways and alveoli. Particles with MMADs between 1 and 5 µm tend to deposit by sedimentation in this region of the lung due to the lower flow velocities encountered there. 53
Clearance of particles from the lower respiratory tract occurs through several mechanisms. In the airways, the mucociliary escalator efficiently transports particles up to the oropharynx, whereas in the alveoli, resident macrophages patrol the surface epithelium and phagocytose insoluble or poorly soluble particles as well as endocytose more soluble particles. Macrophages are considered “professional” phagocytic cells because they have considerable capacity to ingest a range of small and large (up to 30 µm diameter) particles 54 that can result in these cells containing large quantities of particles 55 that might eventually result in particle overload. 56 In this respect, they outclass other phagocytic cells such as epithelial cells and fibroblasts. 57 Macrophages are then cleared into the systemic circulation by transepithelial migration or transported into the gastrointestinal tract via the muco-ciliary escalator. Lung particles may also be removed through the epithelium by active or passive transport into capillaries and lymphatics and then into the systemic circulation. Therapeutic proteins may be degraded by locally secreted and cell-associated proteases. 7
Response to particles
The response of the lung to inhaled, poorly soluble particles has been well characterized in animals. Chronic exposure at high concentrations to crystals (silica, cholesterol, and asbestos), nanoparticles, and inert, poorly soluble particles such as carbon black or TiO2 results in lung tissue accumulation that may result in lung injury. In rats exposed to high concentrations of insoluble particulates, “particle overload” of macrophages may ensue. The threshold for this effect occurs when the volume of ingested particles exceeds 6% of the normal macrophage volume, which results in reduced clearance and macrophage motility. Virtual cessation of particle clearance occurs when ingested particulates reach 60% of macrophage volume. 56 Lesions associated with high concentrations of inhaled insoluble particles are characterized by chronic active inflammation with neutrophil and macrophage infiltration that resembles a form of pathology originally termed “frustrated phagocytosis.” Macrophages are recruited and activated by pattern recognition and scavenger receptors, 58 appearing enlarged with vacuolated or granular cytoplasm (“foamy macrophages”) in histological sections. Chronic inflammation may ensue leading to secondary changes such as fibrosis, emphysema, and even neoplasia. 59 –62
Exposure to lower levels of material usually results in much more benign outcomes. Occupational and environmental nuisance dusts such as ragweed pollen, ambient environmental particulates, and occupational dusts typically constitute a daily inhaled dose ranging from 0.7 to 50 mg. 37 Exposure at this level is usually associated with very little or no toxicity due to rapid clearance by resident alveolar macrophages. 61,63 Similarly, low dose exposure to TiO2 (5 mg/m3) and carbon black (1 mg/m3) for up to 13 weeks of duration in rats does not result in inflammation but merely in a reversible increase in macrophages. 64 These increases in macrophages are considered nonadverse adaptive/physiological responses intended to clear particulate matter. 63 They are frequently encountered in the lungs of control animals as a normal background finding that is often described as an accumulation of intra-alveolar (foamy) macrophages, or (foamy) macrophage aggregates (Gregori et al, this issue) often seen, especially in rat studies, at the bronchioloalveolar junction. 60
Clearance mechanisms stimulate alveolar macrophage infiltrations
In our series of 12 case studies, 8 case studies were associated with alveolar inflammatory cell infiltrates that involved macrophages (Table 4) of which 4 case studies (CS 1, 2-week rabbit; CS 6, 13-week NHP; CS 8, 2-week rat; and CS 10, 4-week rat and 1-week NHP) consisted of pure (uncomplicated) populations of macrophages without accompanying signs of damage or inflammation. The principal driver for increased alveolar macrophages was considered likely to be phagocytosis (clearance) of inhaled drug material.
Summary of Alveolar Pathology and Proposed Mechanisms of Toxicity.
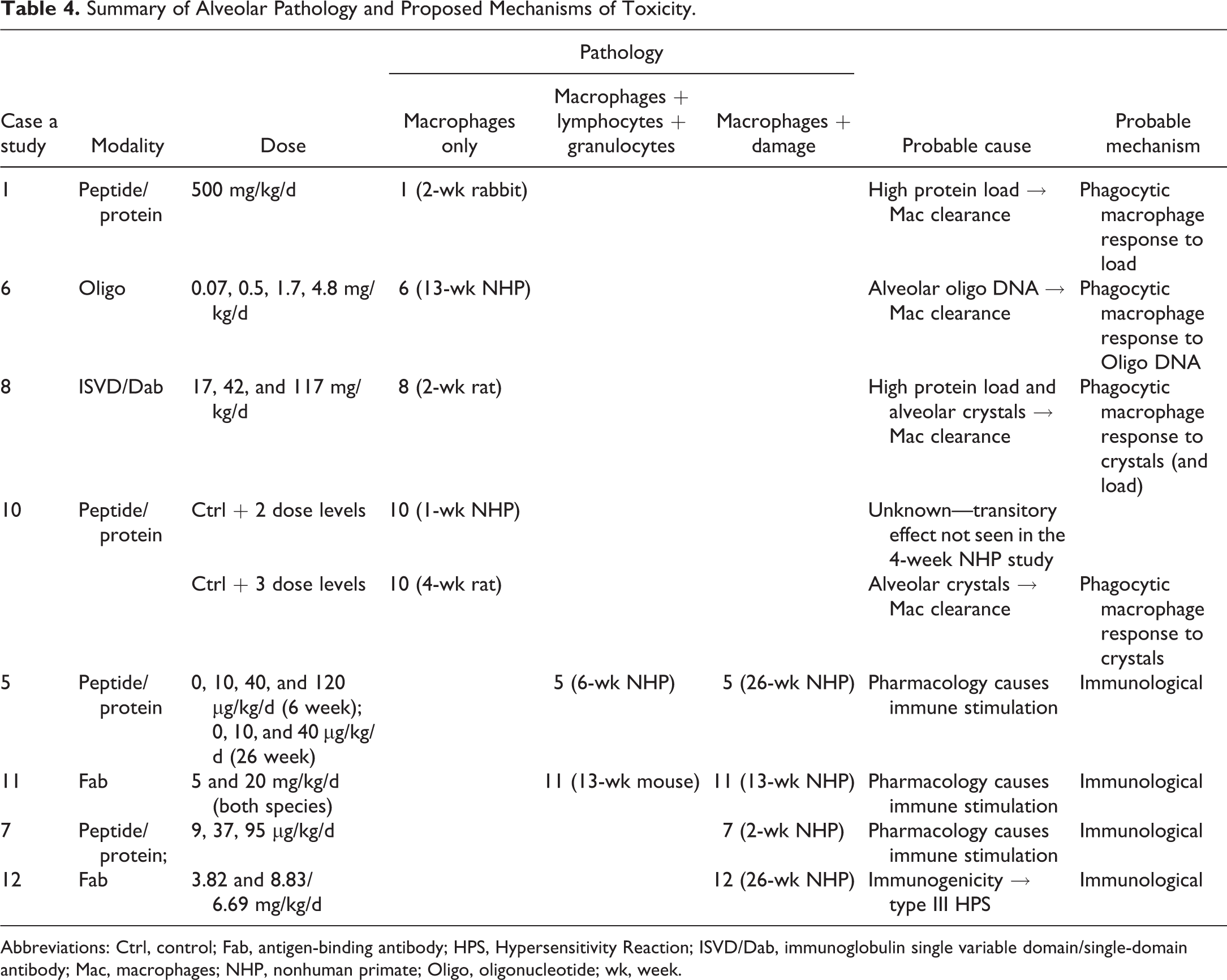
Abbreviations: Ctrl, control; Fab, antigen-binding antibody; HPS, Hypersensitivity Reaction; ISVD/Dab, immunoglobulin single variable domain/single-domain antibody; Mac, macrophages; NHP, nonhuman primate; Oligo, oligonucleotide; wk, week.
In CS 8 (2-week rat) and CS 10 (4-week rat study), eosinophilic crystals were identified and appeared to act as a focus for macrophage accumulation similar to that described for inhaled inert particles or hemoglobin crystals which stimulate macrophage accumulation and phagocytosis in the alveoli. 62,65 Morphologically, the lungs presented with multifocal intra-alveolar eosinophilic crystals surrounded by macrophages that appeared to be engulfing and attempting to phagocytose the crystals.
In CS 1, juvenile rabbits were exposed to a large dose (500 mg/kg/d) of inhaled protein/peptide test item for 13 days. This dose was significantly higher than the top dose employed in the other case studies, which typically tended to deliver micro- to modest (5-40) milligram dosages per day. In this CS, it is possible that the high protein load led to macrophage recruitment to clear excess protein within the alveolar space. However, despite recruitment of blood borne/interstitial macrophages to the alveolar compartment to augment the finite phagocytic capacity of alveolar macrophages to process material, phagocytosis declines beyond a limit. 57 It therefore seems reasonable to assume that at some level of inhaled protein load, protein clearance from the lungs saturates, overwhelming the resident alveolar macrophages, leading to recruitment and accumulation of macrophages from the circulating and interstitial pools in an attempt to maintain homeostasis. 66,67
Finally, in CS 6, NHPs were exposed for 13 weeks to an inhaled oligonucleotide at dosages up to 4.8 mg/kg/d, which resulted in a dose-dependent accumulation of foamy alveolar macrophages (together with foamy interstitial macrophages). Single- and double-stranded oligonucleotides are readily endocytosed into alveolar macrophages by receptor and nonreceptor mediated mechanisms. 68,69 This is an analogous mechanism to that seen in kidney tubules which appear basophilic in hematoxylin and eosin (H&E) sections or hepatic Kupffer cells after systemic administration of oligonucleotides. 70 Inhalation of an antisense oligonucleotide into the terminal airways and alveoli therefore likely induces endocytic clearance by the resident macrophage population in an attempt to remove foreign material, leading to activation, recruitment, and accumulation of further macrophages. Activation and possibly immunostimulation may be augmented by unmethylated cytosine-guanine motifs within oligonucleotides (especially phosphorothioate backbone oligonucleotides) as these activate toll-like 71 receptor 9 and induce chemotaxis. 72 However, in our CS, activation and immunostimulation were limited, as there was no indication of inflammation or damage even after 13 weeks of treatment. This finding is in line with other authors 73 –75 who have also observed simple accumulations of alveolar vacuolated (and sometimes hypertrophic/basophilic) macrophages without inflammation. Similar changes have also been observed in cultured RAW264.7 monocytes incubated with a single stranded phosphorothioate oligonucleotide where no effects were noted on cell viability, proinflammatory cytokines, or phagocytic potential. 74,76 However, it should be noted that these observations are based on a single example and further case studies are required to understand the responses to other inhaled oligonucleotides.
Alveolar inflammatory cell infiltrates with and without inflammation
Four case studies, comprising 6 toxicology studies, presented with alveolar mixed inflammatory cell infiltrates composed principally of macrophages with fewer numbers of lymphocytes and/or granulocytes. Two of these toxicology studies (CS 5, 6-week NHP and CS 11, 13-week mouse) were simple inflammatory cell infiltrates without any other changes. The other 4 toxicology studies (CS 5, 26-week NHP; CS 7, 2-week NHP; CS 11, 13-week NHP; and CS 12, 26-week NHP) were associated with inflammation, where mixed inflammatory cell infiltration was associated with histological evidence of tissue “damage” (hemorrhage/hemosiderin pigment and bronchioloalveolar/type II pneumocyte hyperplasia). The data from CS 5 and CS 11 demonstrated that simple alveolar inflammatory cell infiltration could progress to inflammation depending upon duration of exposure or species. For CS 5, progression was seen in the more chronic 26-week study, compared to the 6-week study (despite a lower top dose of 40 µg/kg/d vs 120 µg/kg/d). For CS 11, both toxicology studies were of equal duration (13 weeks) and exposure (5 to 20 mg/kg/d) but used different species (mouse vs NHP). Less severe alveolar pathology was observed in the mouse (infiltration) versus NHP (inflammation), possibly because the mouse received a lower exposure at the alveolar level due to increased impaction in the nose and airways compared to the NHP, 77,78 although differences in species response cannot be ruled out.
Immune stimulation as a mechanism for alveolar inflammation
In the 4 case studies that were associated with some form of damage or inflammation (CS 5, CS 7, CS 11, and CS 12; Table 4), 3 of the inhaled biotherapeutics (CS 5, CS 7, and CS 11) were immunomodulators whose target pharmacology would be expected to result in immunostimulation. Since the immune system is kept in check by a balance of pro- and anti-inflammatory mediators, it is not surprising that “removing the brakes” or “pressing the accelerator” resulted in inflammation. This is exemplified by a mouse knockout of transforming growth factor β1, a potent immunosuppressive cytokine, which leads to fibrotic and proliferative disease characterized by multifocal infiltrations of macrophages and lymphocytes into multiple organs. 79 It is therefore likely that in CS 5, CS 7, and CS 11, immune stimulation through modulation of target pharmacology resulted in alveolar inflammatory cell infiltration and progression to alveolar inflammation. This notion is consistent with the observation that alveolar mixed inflammatory cell infiltration was highly correlated with ADA positive status. In 1 CS however (CS 12), the target pharmacology was not associated with immune modulation. In this case, it is likely that immune stimulation resulted from a type III hypersensitivity reaction, as the pathology and dose response were consistent with ICD.
Characteristics of ICD
Immunogenicity and the resulting immune complexes that arise in conventional dosing studies can have significant effects on systemic exposure, pharmacology, and safety. Safety concerns from ICD are relatively infrequent but may result in histological changes characterized by multiorgan vasculitis and glomerulonephritis. 80 In the lung, this may present as thrombosis and vascular/perivascular inflammation. 81,82 Determination of ICD is not straight forward as it may present in any dose group with a tendency to show an inverse correlation with dose (ie, occur more frequently in the mid- and low dose groups) 9 and in a single animal (36% frequency of studies) or multiple animals (64% frequency of studies). 80 The clinicopathological response can vary significantly between animals even within the same dose group. Diagnosis is typically a weight of evidence approach based upon clinical signs, clinical pathology, immunohistochemistry (IHC), drug exposure, and identification of immunogenicity as no one test is reliably diagnostic since ADA titers may not correlate with any of the above. 9
Clinical signs, therefore, might be quite variable with no changes through to pruritus, somnolence, unconsciousness, and death. 9 Generally, clinical signs do not occur before 10 to 14 days of dosing (ie, the time required to elicit an immune response) unless there is preformed antibody. Other changes may include relatively minor signs such as decreased activity, skin reddening, and rapid breathing as well as changes in clinical pathology (decreased red blood cell mass, platelets, and decreased/increased white blood cells), alterations in PK/PD, drug exposure, and immunogenicity (presence of ADA). 9
Immunohistochemistry may be used to identify and colocalize immune complex (IC) deposits within the media or intima of the arteries in the tissues of interest. These are visualized as granular deposits, often in a subset of animals, consisting of complement factors, monkey immunoglobulin M (IgM) and IgG, and the human biotherapeutic. 10,82 Electron microscopy may also help demonstrate the presence of electron-dense deposits at the surface of endothelial cells or within the intima of arteries. 9 However, the concordance of ADA titers across all end points, that is, clinical signs, histology, and IHC is, at best, imprecise and, at worst, relatively rare. 80
In our series of case studies, CS 12 (26-wk NHP) showed alveolar changes that were reported to be secondary to ICD. No clinical signs were noted in the main study animals surviving to scheduled termination but, at histopathological examination, minimal alveolar hemorrhage, pigmented macrophages, and inflammation (characterized by macrophages and neutrophils with alveolar septal thickening) were seen in 1 male and 1 female NHP (group size, n = 4) treated with a Fab at 3.82 mg/kg/d (low dose) for 26 weeks. This was accompanied by high titers of ADA in all treated animals and loss of systemic exposure in the low dose group. In addition, 1 female animal, also from the low dose group treated at 3.82 mg/kg/d, was prematurely sacrificed for welfare reasons due to significant clinical signs attributed to ICD. No alveolar changes were seen in the high dose group (6.69 mg/kg/d), although mononuclear cell infiltrates (predominantly lymphocytes with occasional macrophages and plasma cells) were noted in a PV/PB location occasionally extending into the alveolar septa for most animals from all dose groups.
Further IHC work showed staining of test article in all treated animals together with a tendency for increased staining of monkey IgG, IgM, IgA, and complement in alveolar macrophages (and around inflamed blood vessels) in the animals with alveolar hemorrhage and inflammation. However, coprecipitation of granular IC/complement deposits could not be detected. Thus, while IHC failed to confirm ICD, the weight of evidence underpinned by the absence of changes in the high dose group, high levels of ADA, and loss of exposure suggests that ICD was the most likely cause of the alveolar changes in this CS. This supports the notion that immune stimulation (whether due to type III hypersensitivity as seen in CS 12 or pharmacologic modulation as seen in CS 5, CS 7, and CS 11) can induce alveolar inflammation.
Part 4: A Primer—Adversity of Inhaled Biologics
Hazard identification, adversity, and risk assessment
During the process of “slide reading,’ pathologists will normally identify and semiquantitatively score microscopic changes and attempt to ascribe them to treatment or background variation (spontaneous lesions) in a process called “hazard identification.” At the end of the study, pathologists together with relevant subject matter experts will discuss the changes together with relevant ancillary data and decide whether they are adverse or nonadverse—a process that is not always straightforward as ultimately it is an expert opinion based on objective and subjective data analysis. 83 This judgment is recorded in the report contributions (anatomic pathology report, clinical pathology report) and/or integrated toxicology study report depending upon client preference, with perhaps the latter report being the optimum, as the pathology reports may lack relevant details that contribute to this call. It is important to recognize that adversity can be a difficult assessment to make and that this determination is made on the totality of nonclinical data collected to date. The understanding of a histological lesion may therefore improve during the development of a molecule as data from more chronic toxicity studies inform upon the nature of the change. Once made, an adversity call has significant impact upon drug development as this is the major determinant in clinical dose setting and risk assessment. The risk assessment is an integrated assessment of all nonclinical data and occurs before regulatory acceptance and first in human clinical trials. Adversity defined in nonclinical studies is one of the principal components used in making a risk assessment. However, other important facets of the risk assessment include factors such as patient population, intended clinical indication, life expectancy, effectiveness of standard of care treatment, clinical monitorability, and ability to manage adverse effects. All these elements should be considered in the analysis of perceived risk versus benefit.
What is adversity?
Adversity is a fairly nebulous concept that is not always easy to apply based solely on morphologic changes seen in H&E-stained slides, especially in acute toxicology studies where ancillary data may be lacking and often progression of the lesion, with continued exposure, is unknown. Several definitions guide this process with perhaps studies of Keller et al
84
and Palazzi et al
85
providing the most useful advice: “…an adverse effect is a test item-related change…that likely results in an impairment of functional capacity to maintain homeostasis and/or an impairment of the capacity to respond to an additional challenge.” (Palazzi et al
85
) “Adverse Effect: A change…that results in an impairment of functional capacity, an impairment of the capacity to compensate for additional stress, or an increase in susceptibility to other influences.” (Keller et al
84
)
These publications focus on organ function and functional reserve capacity (ie, capacity to respond to additional stress) as the key measures of adversity since these measures are the underlying metric that determines overall health and reproductive function. Implicit in this idea is that organ function is inferred from the histological picture together with the totality of nonclinical data.
Another recent definition discusses adversity as a “harmful” effect which detrimentally affects overall health (Kerlin et al 86 ). This is a more abstract and subtly different definition to Keller and Palazzi; however, it does point toward the same end point, that is, the health status of the animal which is ultimately dependent upon organ function (the basis of Keller and Palazzi’s measure of adversity).
Pathologists make an adversity call based on the totality of all data types pertaining to the histopathological lesion, with the objective of assessing whether what is seen down the microscope would meaningfully and detrimentally affect organ function and hence the health/reproductive function of the animal. How an individual pathologist makes an adversity call can, on occasion, not be clear to toxicologists or regulators or even sometimes to other pathologists without adequate communication. This process is imperative in order to integrate the various data sets and scientific views from other areas of expertise in order to arrive at consensus. Nevertheless, lack of clarity and justification for the call has on occasion led to confusion, or reasonable differences of opinion that at least for the study and reviewing pathologists can usually be resolved at peer review.
In general, pathologists make a call of adversity based on: Nature and severity of the microscopic finding together with any macroscopic observations, effects on health (in-life findings/clinical signs/body weight etc), effects on clinical pathology, functional end points (if available), and injury/functional biomarkers (if available).
It should be emphasized that paradoxically, pathologists do not consider patient health when making a call of adversity in a nonclinical safety study, although it is patient health that the call is intended to protect. We instead consider the effect of the treatment on animal health, made by an objective assessment as possible of all available safety data, informed by experience, and aided by any specialist knowledge of the study pathologist, peer reviewing pathologist, consultant, toxicologist, or any other expert.
Do We Operate a “Zero Tolerance” Approach to Adversity?
A general maxim that many pathologists follow is that minimal or mild/slight lesions are more likely to be nonadverse compared to moderate, marked, or severe lesions, 85 although there are many exceptions to this rule. This makes sense if loss of function is the metric of adversity as changes in a limited number of cells are unlikely to affect the overall function of an organ in any meaningful way. The net outcome of this process is that pathologists ultimately assess organ function when they look at microscopic lesions because it is presumed that the structural finding(s) impacts organ function and therefore the health and/or reproductive function of the animal. What is implicit in this assessment and accepted by many pathologists/toxicologists is that limited damage is not necessarily considered adverse—however this varies from tissue to tissue, with much less tolerance in nonregenerating organs such as the heart and central nervous system. So, for example, minimal to slight squamous metaplasia of the larynx in rodents is regarded as nonadverse 87 but clearly some damage (epithelial necrosis and/or loss) has occurred to induce stem cell proliferation and differentiation. Similarly, minimal to mild alveolar macrophage infiltrates increase the diffusion barrier within the alveolar bed but are not considered adverse at minimal to mild levels as these do not meaningfully affect organ function. 63 Finally, minimal to mild nasal mucous cell hyperplasia/metaplasia is commonly considered adaptive (ie, nonadverse), 88 but as for laryngeal metaplasia, must arise from irritation and damage, leading to loss of epithelial cells and replacement by reactive hyperplasia.
What is not clear is what degree of “harm” or damage, if any, is permissible in organs like the lung—an organ with considerable capacity for self-renewal and with considerable functional reserve, especially in healthy animals typically encountered in nonclinical toxicology studies. Differing tolerance to damage, differing definitions of adversity in the literature, and the practical difficulties in translating this definition into a view of how a morphologic lesion might impact the health status of an animal through effects on organ function leads to inconsistencies of opinion between pathologists, toxicologists, and regulators, and even between regulatory agencies. Pertinent examples exist in the literature (Opinion Pieces: Toxicologic Pathology Forum 89,90 ) and are frequently encountered when submitting drugs for regulatory approval. Clearly, there is no universal standard to assess adversity. Differing opinions exist around whether minimal lung inflammation is regarded as adverse, since some “harm” has been done. As in the examples cited above in the context of the study in question, tolerance for limited damage is precedented if there has been no meaningful detrimental impact on organ function and no discernible detrimental effect on the animal’s health and well-being.
Similarly, differing opinion may exist on whether minimal to mild inflammatory cell infiltrates (ie, infiltrates of cells without histological evidence of inflammation or changes in lung structure) are adverse, even if again there is no meaningful detrimental effect on lung function or evidence of damage. In relation to this latter change, a number of questions are often asked by sponsors and regulators to understand the effects: Is it a response to occult damage? Are these quiescent or activated inflammatory cells?
The first question can be answered at the histological level—the current gold standard for safety assessment—by careful review of the slides to ensure that any changes seen in longer term studies are not accompanied by damage, or arising from damage, detected in more acute toxicology studies. For the second question—whether the cells represent activated cells or quiescent cells become a moot point in the absence of overt damage or meaningful detrimental effects on organ function/animal health. At the morphologic level, these types of changes are difficult to detect by light microscopy, and in the absence of any other changes, an alveolar mixed inflammatory cell infiltrate might be regarded as qualitatively similar to an alveolar macrophage infiltrate (which at the minimal or mild level is regarded by most pathologists as nonadverse 63 ).
Finally, differences of opinion may exist over whether PV/PB MIC infiltrates are adverse and whether they represent an adaptive immune response to foreign epitopes or an inflammatory response to occult damage. In the absence of mechanistic studies, one cannot definitively answer this question, but here, a weight of evidence approach can provide some insight as to whether an immune response is a plausible mechanism for the observed changes. Such data might include an assessment of both local and systemic ADA, a description of whether the cellular changes are immune cells centered around blood vessels (which represents the site of ingress of homing lymphocytes 7 ) and whether the pattern and kinetics of the changes match those of an immune response (2-week induction period and changes in regional lymph nodes indicating an on-going immune response). However, as for inflammatory cell infiltrates, these questions are somewhat academic in the absence of overt damage or meaningful detrimental effects on organ function/animal health when making assessments of adversity.
Differences of opinion therefore exist at all levels of the industry when assessing what is acceptable in nonclinical species. This is complicated by the clinical environment given the absence of monitorability and sensitive, noninvasive biomarkers of lung damage, absence (and disagreement in methods of measuring) of lung function, 91,92 the relatively high frequency of lung disease in the human population, the progressive nature of chronic lung disease, and the vulnerability of patient populations. A minimal or mild change in a healthy animal in a cage on study may be deemed nonadverse in the absence of meaningful detrimental effects on lung function as inferred from microscopic changes informed by all other safety data. However, this same change may be adverse in patients with significantly reduced lung function and comorbid pathology—for example—infiltrates of activated inflammatory cells within the lung of a healthy animal may be nonadverse in the nonclinical species but may be adverse in an inflamed lung of a patient through synergism with underlying pathophysiology and exacerbation of pre-existing inflammation. For regulators and clinicians responsible for patient health in clinical trials, it can be difficult to translate what is the human risk based on a minimal or mild nonadverse change identified in a healthy animal, especially one that has the capacity to progress to an adverse change with higher exposure. However, the former pertains to hazard identification and the adversity call is made in isolation of the clinical scenario, whereas risk and adversity in humans pertain to risk assessment and should integrate all the uncertainties associated with lesion progression and patient vulnerability. One should not confuse the 2 when making a call of adversity nor call something adverse in the nonclinical species based of an anticipated adverse effect when extrapolated to a vulnerable human population. Instead one needs to perform adequate risk assessment and ensure adequate safety margins to bridge areas of uncertainty in order to protect human health.
Regulatory Perspective
Nonclinical toxicology studies with inhaled biotherapeutics pose unique challenges to investigators and regulators alike. The relative paucity of background data and low numbers of regulatory submissions with such products mean that there is a clear lack of experience in interpreting the complex findings that are observed. This has led to some companies overinterpreting findings in individual study reports in an attempt to identify adverse and nonadverse findings when, in fact, there is a lack of understanding of the expected pathology and mechanism of actions of the investigational medicinal product within the respiratory system. In addition, an inconsistent approach from different pathologists and companies has led to different conclusions being made on similar findings. Therefore, a paper such as this, discussing case studies in an attempt to adopt a more consistent approach, especially with regard to grading of findings, is welcomed.
The goals of toxicology studies include the identification of potential target organs and the characterization of toxic effects with respect to those target organs. Dose–response relationships and the potential reversibility of findings are also important goals. Appropriate nonclinical studies are the basis of extrapolation to indicate possible risks to humans and the value of a toxicology study is only as good as its design and interpretation.
It is the ultimate responsibility of an applicant to ensure that toxicology study documents include a clear and cohesive interpretation of study findings. Essentially, findings in a study should be defined in relation to effects on the test species used and within the context of the given study. Test article-related effects should be described in isolation to other factors, and then decisions to consider them as adverse or nonadverse to the animals in the study should be justified based on a scientific rationale. Too often, investigators attempt to classify effects as nonadverse within a study to justify an “appropriate” safety margin for humans. However, it should be remembered that the investigational medicinal product is usually being administered to healthy animals in an unnatural situation with large multiples of the clinical dose and hence adverse effects in those animals should not be unexpected. The relevance of any such findings to humans is best discussed in the nonclinical overview regulatory documents rather than in individual study reports.
Limitations of Paper
This manuscript is a multicompany observational data set describing the pathology induced by 12 unique biotherapeutic molecules (therapeutic antibodies—Mabs, Fabs, ISVDs/Dabs, peptide/proteins, and 1 antisense oligonucleotide), split across 18 toxicology studies. Although this is a relatively large number of studies and gives some confidence to the robustness of the data, clearly knowledge in this relatively novel area of inhaled toxicology is limited, and further case studies are required to corroborate the conclusions. This is particularly relevant to inhaled oligonucleotides where only 1 example was represented. Although the observations pertaining to this molecule are accurate, it is possible or even probable that other oligonucleotides, with similar and different chemistries, will behave differently and may induce different pathology.
The proposed mechanisms of pathology, although scientifically plausible, are based on a weight of evidence approach. Since the main purpose of safety studies is hazard identification rather than mechanistic understanding of organ toxicity, further work is required to experimentally validate these mechanisms and demonstrate immunogenicity as the key driver underlying many of these changes. One such experiment might be to demonstrate ADA displayed on the surface of immune cells in the PV/PB MIC infiltrates. Detection of ADA positive immune cells would demonstrate the presence of an immune response in the respiratory tract which might indicate a physiological response to foreign epitopes rather than an inflammatory reaction to tissue damage. Hand in hand with mechanistic studies, sensitive biomarkers of lung injury are also required to more fully understand the adversity of changes seen in nonclinical species at the end of the treatment period.
Finally, toxicology studies are performed in healthy animals which generally have enormous functional reserve and limited functional demand on the respiratory system. Although these studies have proven to be an accurate predictor of toxicity in humans, there have been examples where this has not been the case, and nonclinical studies have not predicted serious adverse events in humans. 93,94 The underlying reasons for this are multifactorial and complex, but one obvious confounding factor is the lack of comorbid pathology in young animals which precludes the possibility of understanding whether a test molecule antagonizes or synergizes with an aberrant pathophysiological pathway. Hence the type, severity, and adversity of changes noted in animal studies may not accurately predict those in humans. Although this does not invalidate the toxicology studies or the accuracy of the adversity determination, regulators and sponsors need to be aware of the limitations of toxicologic assessment in nonclinical studies and not assume that there is a direct translation of these changes, or their interpretation, to a diseased burdened population.
Conclusions
Analysis of the 12 case studies, comprising 18 toxicology studies, revealed a characteristic pattern of toxicity associated with high levels of immunogenicity (ADA) and a pattern of microscopic changes characterized by PV/PB MIC (composed predominantly of lymphocytes), simple increases in macrophages, and alveolar inflammatory cell infiltrates ranging from mixed inflammatory cell infiltration through to inflammation. In addition, 1 CS was associated with severe alveolar inflammation and hemorrhage which was thought likely to be due to ICD. Three mechanisms were considered most likely to underlie these changes: immunogenicity due to exposure to foreign epitopes, impaired clearance of test article within the terminal airways and alveoli, and immune modulation or stimulation via pharmacologic effects on target biology or type III hypersensitivity (ICD).
Appendix A
Exubera, Recombinant Human Powdered Insulin, Pfizer
Rats and nonhuman primates (NHPs) treated with inhaled recombinant human powdered insulin at up to 6 mg/kg/d for 26 weeks showed no significant histopathological effects. Focal to multifocal alveolar inflammatory cells and focal to multifocal aggregates of alveolar histiocytes were noted in both the rat and NHP 6-month studies (including the excipient and air control groups) but most of these changes were attributed to the mechanical trauma of alveolar lavage or stress from the drug delivery system. Slight increases in antihuman antibodies were noted in several treated rats, but none were detected in the primate which shares similar sequence homology to the human form of insulin. 23,24
Afrezza (Technosphere-Insulin), Mannkind
Rats and dogs treated with inhaled recombinant human powdered insulin (delivered as Technosphere-insulin particles) at up to 2.18 mg/kg/d (1.96 mg Technosphere and 0.22 mg Insulin) for 39 weeks showed no significant treatment-related respiratory tract changes. 95 Eosinophilic globules and epithelial degeneration were noted in the nasal cavities of rats treated for 26 weeks and were thought to be due to irritation by the Technosphere particle component. 95 Alveolar and/or bronchial-interstitial neutrophilic infiltrates in the lungs of dogs treated for 39 weeks had an equivocal association with Afrezza exposure.
Human Insulin Inhaled Powder, Lilly
Treatment of dogs with inhaled powdered human insulin for 26 weeks at dose levels up to 701 µg/kg/d resulted in no effects on pulmonary assessments including histopathology and showed either low or no immunogenic potential. 49
Pulmozyme, Recombinant Human DNase
Treatment of rats and NHPs with inhaled recombinant human DNase at up to 2.2 mg/kg/d for 26 weeks resulted in perivascular (PV)/peribronchiolar (PB) mononuclear inflammatory cell infiltrates and alveolitis in both rats and NHPs. 47 Changes in the rats were characterized by mild to moderate alveolitis with inflammatory cell accumulation in alveolar spaces and a PV/interstitial inflammatory cell infiltrate and proliferation of type II pneumocytes. In the 26-week studies, the incidence was qualitatively similar to the 4-week studies but of reduced severity and incidence. In addition, an increased incidence of germinal centers was noted in the bronchus-associated lymphoid tissue (BALT) as well as in bronchial lymph nodes (the latter in female animals only). Similar changes were noted in the primates, characterized by increased PV lymphocytic cuffing, PB lymphoid hyperplasia, terminal airway bronchiolitis/alveolitis with increased eosinophil infiltrates, and pigment macrophages. 47 The titer of the antibody response in the 6-month primate study appeared to correlate with the severity of the pathology and was thought to be indicative of a type I hypersensitivity. 47
Inhaled Growth Hormone
Treatment of rats with inhaled human growth hormone at 0.27 mg/m3 for 0.3 hours/d for 28 days resulted in mild PV mononuclear cuffing of small and medium-sized pulmonary veins. No evidence of vessel wall destruction or karyorrhexis of leukocytes was observed. 7 Conversely, exposure of rats to bovine growth hormone for 7 days resulted in no lung abnormalities. 7 Although histological detection of small changes in BALT cellularity might be challenging after 1 week of exposure, this pattern of perivascular cuffing is consistent with a lack of significant immunogenicity to bovine growth hormone. 96 This was reflected in the much lower IgG titers elicited 37 due to much higher sequence homology of bovine growth hormone compared to human growth hormone (22/191 foreign amino acids compared to 67/191 foreign amino acids).
Footnotes
Authors’ Note
This review paper was endorsed by the British Society of Toxicologic Pathology (BSTP) Executive Council (EC). It has been reviewed and approved by the BSTP EC but does not represent a formal best practice recommendation. The views expressed in this article are those of the authors and do not necessarily represent the policies, positions, or opinions of their respective agencies and organizations.
Acknowledgments
The authors would like to thank the following people for their help in the preparation of this manuscript: John Vahle, Michael Dorato and Jeff Engelhardt for critically reviewing the manuscript. The authors would like to thank the pharmaceutical companies who donated case studies, and all the companies that donated time and resource to the preparation and review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
