Abstract
Treatment of nonhuman primates and mice with a humanized antigen-binding fragment (Fab) antibody (UCBFab) inhibiting transforming growth factor β via daily inhalation for up to 13 weeks resulted in low systemic exposure but high local exposure in the lung. Target engagement was demonstrated by reduced levels of signal transducers, phosphoSMAD and plasminogen activator inhibitor-1 in the bronchoalveolar lavage fluid (BALF). Treatment was associated with a high frequency and titer of antidrug antibodies, indicating high local immunogenicity, and local pathology within the lung and draining lymph nodes. Microscopic changes were characterized by perivascular (PV) and peribronchiolar (PB) mononuclear inflammatory cell (MIC) infiltrates that were principally lymphocytic in nature and mixed inflammatory cell infiltrates and/or inflammation within the alveoli. Immunohistochemical investigation revealed a predominantly CD68-positive macrophage and CD3- and CD8>CD4-positive T-cell response in the alveoli, whereas within the airways, there was a variable mixture of CD3-positive T cells, CD20-positive B cells, and CD68-positive macrophages. Increased cellularity of the draining lymph nodes was also noted, indicating the presence of an immune response to the inhaled test article. Morphologic changes did not progress over time, and all changes partially recovered. Increased leukocytes (principally macrophages) in BALF cytology correlated with the changes seen by histopathology.
Keywords
Introduction
Idiopathic pulmonary fibrosis (IPF) is a chronic progressive interstitial lung human disease of unknown etiology that occurs in adults and is characterized by progressive loss of parenchymal lung tissue and replacement with fibrous connective tissue. Histological appearance is similar to “interstitial pneumonia” and characterized by a patchwork of interstitial fibrosis and scarring with fibroblast foci but with only mild inflammation. Inflammation typically consists of foci of lymphocytes and plasma cells with isolated areas of normal lung tissue. 1 Idiopathic pulmonary fibrosis is typically progressive with a life expectancy of 2 to 5 years from time of diagnosis due to the lack of effective treatments.
Idiopathic pulmonary fibrosis commonly occurs at age 50 and beyond and is thought to arise after repeated injury to the alveolar epithelium leading to chronic inflammation and rounds of epithelial damage and regeneration. Ultimately, this results in stem cell exhaustion and abnormal reepithelialisation. 2 Transforming growth factor β (TGFβ) is thought to play a central role in this abnormal repair process by recruiting local and circulating/bone marrow–derived fibroblasts and promoting their transformation into activated myofibroblasts that migrate into areas of inflammation to deposit excessive extracellular matrix. 3,4 The principal source of TGFβ has long been thought to be the alveolar macrophage 5 ; although a more recent hypothesis proposes that upon injury, TGFβ originates from the activation of fibroblasts. 6 Inhibitors of TGFβ are efficacious in reversing changes in the bleomycin-induced model of lung fibrosis. 7 Therefore, inhibition of TGFβ seems an attractive therapeutic axis for IPF. However, TGFβ regulates a plethora of biological processes, including adult stem cell differentiation, immune regulation, wound healing, and inflammation, so alterations of specific components of the TGFβ signaling pathway may contribute to a broad range of pathologies. Previous experience with systemic inhibition of TGFβ has failed to demonstrate efficacy without adverse findings. For example, systemic administration of fresolimumab, a pan-TGFβ immunoglobulin G4 (IgG4) monoclonal antibody inhibitor, resulted in treatment-emergent keratoacanthomas and squamous cell carcinomas in humans. 8,9 In cynomolgus macaques, subchronic treatment with fresolimumab resulted in reversible epithelial hyperplasia of the bladder, nasal turbinate, and gingiva (with hemorrhage) that generally developed in areas of preexisting inflammation in the bladder. 10 In mice, subchronic treatment resulted in cystic epithelial hyperplasia and inflammation of the tongue, dental dysplasia, and epithelial hyperplasia and inflammation of both gingiva and esophagus. 11 Therefore, the local delivery of a drug-inhibiting TGFβ that would remain largely restricted to the lung was considered to be safer. Hence, the design of UCBFab, a humanized antigen-binding fragment (Fab) antibody, inhibits all 3 isoforms of TGFβ.
Inhaled biotherapeutics (biologics) are a relatively new platform of drug delivery relying on the respiratory system to distribute and deposit the drug product at the target location (usually the bronchioloalveolar region for particles with mass median aerodynamic diameter [MMAD] of <5 μM). 12 Although this strategy has been used for systemic indications due to rapid transport of some biologics into the blood, antibody constructs lacking the FcRn-binding sites are more restricted to the lung tissue. This approach has the advantage of achieving high local exposure while minimizing systemic toxicity, which is especially important for molecules that have tissue-damaging anti-oncogenic properties or neutralize essential functions.
While the inhalation route might be attractive from a systemic safety point of view, it is also a challenging drug delivery route due to the lack of publicly available literature describing class-specific effects of inhaled biologics. As a consequence, these effects are not well understood and are often considered adverse by experts in the field 13 and regulatory authorities. The lack of reliable and practical biomarkers to measure lung function or detect early damage and the relatively high level of lung disease in the human population coupled with the clinically silent early stages and irreversible nature of chronic interstitial lung disease (such as emphysema and IPF) heightens clinical and regulatory concern for lung-related toxicities. As a result, the algorithm used to set human clinical dosing based on the no-observed-adverse-effect-level identified in nonclinical studies is more conservative for lung delivery than for systemic dosing.
We have therefore generated case study data to describe the pathology, pharmacokinetics (PK), and immunogenicity associated with inhalation exposure of nonhuman primates (NHPs) and mice to a UCBFab that neutralizes TGFβ isoforms 1, 2, and 3 in toxicology studies of 2 to 13 weeks’ duration. The intention was to broaden the limited scientific body of literature in the public domain and describe the immunogenicity and potential mechanisms of toxicity of UCBFab. We also offer a weight of evidence-based explanation of the mechanism of the changes induced by inhalation of UCBFab.
Materials and Methods
Test Material
Subnanomolar concentrations of UCBFab were shown to inhibit TGFβ isoforms. The drug substance used across studies (termed UCBFab) was pure at more than 99.5% and was formulated in 50 mM histidine, 125 mM NaCl, and 0.005% (wt/vol) Polysorbate 20 and was diluted as required with the formulation buffer to achieve the required concentration for nebulization.
Animals and Treatment
Studies were conducted in both the United Kingdom and North America (Canada). For UK-based studies, all studies were performed in accordance with the standards of animal care and ethics described in “Guidance on the Operations of the Animals (Scientific Procedures) Act 1986” 14 issued by the UK Home Offices and conducted so that any clinical expression of toxicity remained within a moderate severity limit as described by the guidelines agreed with the UK Home Office Inspector. For North American–based studies, the study plan and any amendment(s) or procedures involving the care and use of animals were reviewed and approved by the Institutional Animal Care and Use Committee. During the study, the care and use of animals was conducted in accordance with the guidelines of the US National Research Council and the Canadian Council on Animal Care.
Animals were observed at least daily throughout the study for general health/mortality and morbidity and after dosing for potential reactions to UCBFab. A detailed clinical examination was performed daily, and body weight was recorded once or twice a week.
Cynomolgus Macaque
A number of studies were performed in cynomolgus monkeys (Table 1) originating from Mauritius or China. At the initiation of dosing, the animals were 2 to 4 years old and weighed between 2.2 and 4 kg. Animals were housed in groups by sex and by dose regimen and had access to enrichment.
Summary of Study Design for GLP and Non-GLP Toxicity Studies With UCBFab in NHPs and Mice.
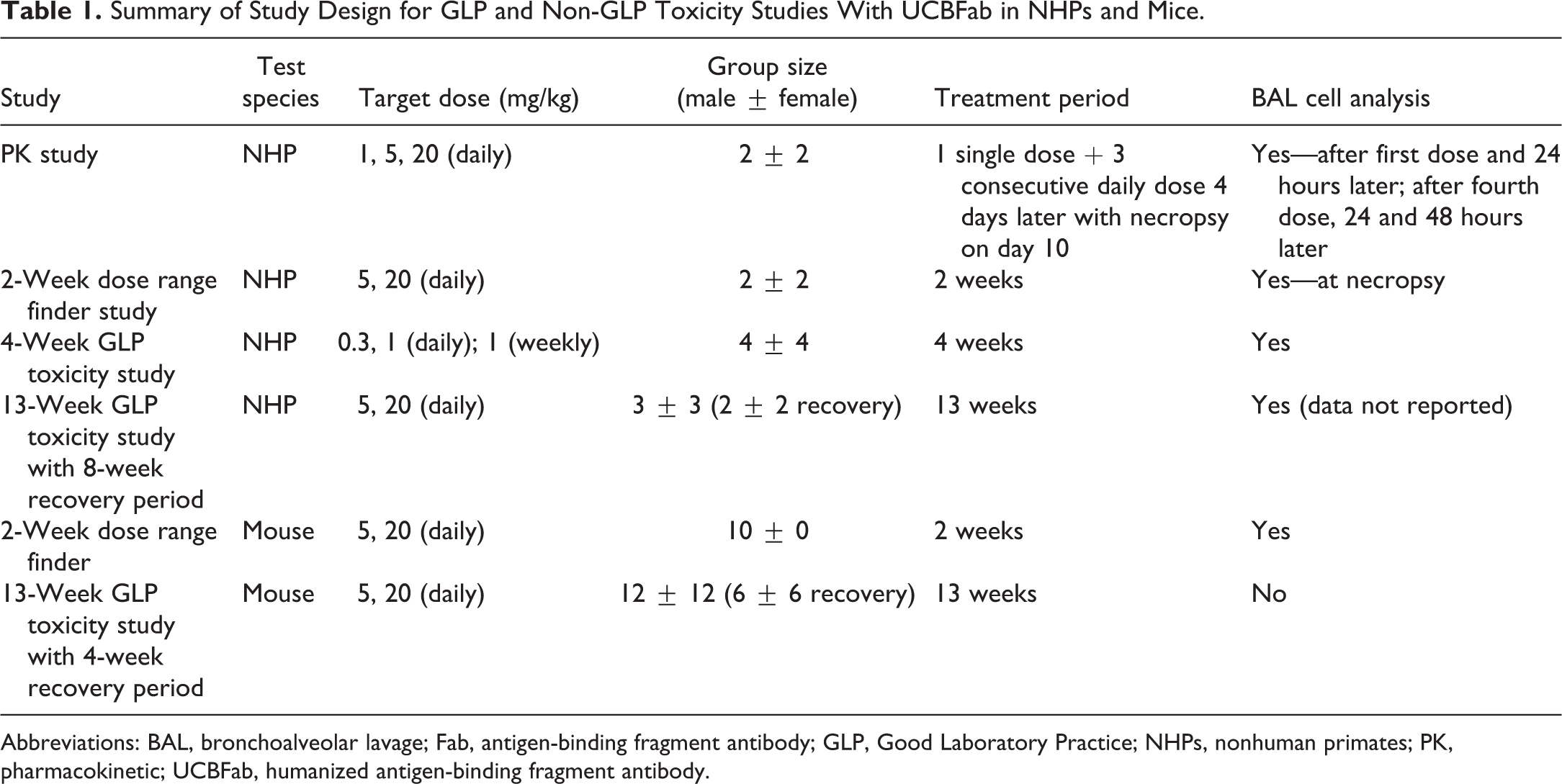
Abbreviations: BAL, bronchoalveolar lavage; Fab, antigen-binding fragment antibody; GLP, Good Laboratory Practice; NHPs, nonhuman primates; PK, pharmacokinetic; UCBFab, humanized antigen-binding fragment antibody.
For inhalation dosing, monkeys were seated in a purpose-designed restraint chair with a close-fitting face mask, connected via flexible plastic tubing to a plenum chamber or a linear manifold, depending on the experiment and required achieved dose. The mask operated as a flow past system with freshly generated aerosol being continuously delivered to and exhausted from the mask. The aerosol flow rate to each mask was controlled with a positive differential between input and exhaust. All air flow rates (delivered and extracted) were monitored visually using calibrated flow meters. Chamber air flow rates, temperature, and relative humidity levels were monitored and recorded at regular intervals during each daily exposure period.
Aerosols were generated using PARI e-flow vibrating mesh nebulizers (PARI Pharma GmbH). Each nebulizer was fitted with a reservoir and primed with vehicle or dose formulation as necessary, prior to aerosol generation/animal exposure. The amount of vehicle or dose formulation used over the period of aerosol generation was determined by weight difference.
Cynomolgus monkeys were treated with a nebulized aerosol formulation of UCBFab via a close-fitting face mask for 30 minutes at dose ranging between 0.3 and 20 mg/kg on a daily basis or intermittently in 4 separate studies consisting of a short-term repeat phase 3-day PK study, a 2-week non–good laboratory practice (GLP) dose range finding study and 2 GLP toxicity studies of 4 and 13 weeks’ duration, the latter having an 8-week recovery period (Table 1).
In the NHP PK study, animals (n = 2/sex/group) were exposed to a nebulized aerosol of UCBFab at a single dose of 1 or 20 mg/kg for 30 minutes on the first day, followed by a 4-day off-dosing period, then by a 3-day consecutive daily dosing period, and finally, by a 2-day off-dosing period with necropsy on day 10 of study, 48 hours after the last dose.
Mice
Approximately 6- to 8-week-old Crl: CD1(ICR) mice were exposed by flow-through snout only (Table 1); they were placed in restraint tubes connected to a plenum chamber where the aerosol generated by the PARI e-flow vibrating mesh nebulizer (PARI Pharma GmbH). Mice were treated daily for a duration of 30 minutes with a nebulized aerosol formulation of UCBFab at 5 to 20 mg/kg/d in a 2-week non-GLP dose range finding study and a 13-week GLP toxicity study with a 4-week recovery period (Table 1).
Delivered Dose
The estimated delivered doses in both species were derived based on measured aerosol concentration of UCBFab, calculated respiratory minute volume, exposure duration, and animal body weight, using the following criteria:
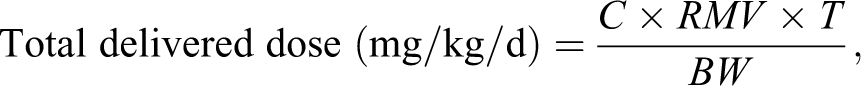
where C = aerosol concentration (mg/L of test item analytical base)
RMV = respiratory minute volume (L/min) *
T = duration of dosing (minutes)
BW = animal body weight (kg)
* RMV was calculated based on AIT equation: 0.608 × body weight (kg)0.852. 15
The determination of delivered concentrations was performed on each dosing day or at least twice weekly in long-term studies. Aerosols were sampled using a liquid impinger system comprising a liquid trap, vacuum pump, flow meter, and gas meter from a reference sampling port representative of the animal exposure face mask and test aerosol sampled for an appropriate duration at an appropriate flow rate. Analyses of aerosol samples were performed by ultra-performance liquid chromatography and high-performance liquid chromatography using validated analytical procedures. Stability of samples following nebulization was verified using size exclusion chromatography and verification of TGFβ1 activity using a gene reporter assay to confirm potency of the Fab after nebulization on a single occasion.
The particle size distribution of the test aerosols was determined by gravimetry using a Marple Model 298 Cascade Impactor for the monkey studies or a Marple 290 cascade impactor for mouse studies. Sampling was done on a regular basis over the course of the study from a reference sampling port representative of the animal’s exposure face mask (monkeys) or exposure tube (mice). The target particle size range for the MMAD was 2 to 4 µm for cynomolgus monkeys and 1 to 3 µm for mice.
Clinical Pathology and Bronchoalveolar Lavage Fluid Immunophenotyping
For NHPs, hematology and clinical chemistry were monitored at least once during pretest and at the end of the treatment phase (twice for the NHP 13-week study). In mice, hematology and clinical chemistry were monitored terminally. Blood samples were collected and analyzed for standard red and white blood cell (WBC) indices as well as plasma chemistry.
For NHPs, blood immunophenotyping was evaluated on at least 2 instances during pretest and at least 1 instance during the treatment phase to determine total T cells, T helper, T regulatory, cytotoxic lymphocyte natural killer (NK) cells, total B cells, naive B cells, memory B cells, mature B cells, monocytes, and neutrophils. Bronchoalveolar lavage fluid (BALF) immunophenotyping was performed with the same panel, plus activated T cells.
Necropsy and Histology
Animals from each study were subjected to a complete necropsy examination. Selected tissues were collected, weighed when appropriate, and representative samples were collected and preserved in 10% neutral-buffered formalin. Regarding the respiratory tract, the following tissues were collected: regional lymph nodes, trachea (proximal and distal), nasal cavity (proximal and distal), pharynx, larynx, carina, and lung. In the PK study, only lungs were collected, whereas in the NHP 4- and 13-week studies, the left cranial, middle, caudal, and right cranial lobe were processed for histological examination; the right middle lobe was isolated for BALF collection, the right caudal lobe snap frozen, and the right accessory lobe fixed for no more than 48 hours in formalin. Tissues for histological examination were embedded in paraffin, sectioned, mounted on glass slides, and stained with hematoxylin and eosin (H&E).
Serum Collection
Serum samples for toxicokinetics (TKs) and antidrug antibody (ADA) analysis were collected from main study animals at time points as appropriate for each study, after the first and last dose over 24 hours and on several occasions before nebulization (trough sample). In mice, samples were collected from dedicated animals.
Bronchoalveolar Lavage Fluid Collection
Bronchoalveolar lavage fluid was collected at necropsy from the 4- and 13-week NHP studies, but only reported for the 4-week study due to poor sample volume and quality in the 13-week study. The lungs were perfused with 0.9% saline via the pulmonary artery to remove congestion. The right middle lobe was tied-off lobe and then removed and washed with sterile saline at room temperature (3× 5 mL), and the aspirated liquid was collected into sterile plastic tubes. In the PK study, BALF was collected twice in anesthetized animals. An endotracheal tube was inserted into the respiratory tract until point of resistance, and BALF was collected by gentle flushing and aspirating 3× 3 mL/kg warm sterile saline. In mice, BALF samples were collected from dedicated animals at appropriate time points by dosing via intratracheal instillation of 3× 0.4 mL of 0.9% saline. One sample was used for automated total nucleated cell counts (ADVIA hematology analyzer) and manual differential WBC counts (cytospin preparation) of bronchial lavage fluid. A Halt protease and phosphatase inhibitor cocktail was added to the remainder BALF that was centrifuged 10 minutes at 500g in refrigerated conditions; supernatants were stored at −80 °C for UCBFab determination and cell pellets were snap frozen and stored at −80 °C.
Bioanalysis, Biomarkers, and TKs
Bioanalysis on serum and lung samples was performed by ligand-binding assay with a sandwich method by Mesoscale Delivery (MSD), an immobilized anti-UCBFab idiotypic antibody and MSD RuSulfo-TAG antihuman Kappa Light Chain goat Ab conjugate. Bioanalysis on BALF was performed by liquid chromatography mass spectrometry.
Sample analysis for immunogenicity was conducted on selected samples using a homogeneous bridging assay using biotinylated UCBFab and RuSulfo-tagged UCBFab and developed on the MSD platform. SMAD2/3 and phosphoSMAD2/3 (ser 465/467) levels were determined on lysed BALF cell pellets from NHP using specific Path Scan sandwich immunoassays reference 12000 and reference 7348, respectively, from Cell Signaling Technology. The TGFβ1 was quantified in mouse BALF supernatant using a sandwich immunoassay from R&D Systems (reference MB100B).
Plasminogen activator inhibitor-1 (PAI-1) was quantified in mouse BALF using a sandwich immunoassay from Abcam (reference ab157529). Toxicokinetic samples were determined using Phoenix WinNonlin V6.4 (Pharsight Corp) and a noncompartmental approach consistent with the inhalation route of administration. Standard deviations for Figure 1A, B, and D were determined using Excel.
Immunohistochemistry
Immunohistochemistry (IHC) was performed on formalin-fixed, paraffin-embedded (FFPE) lung sections from selected animals of the 13-week NHP toxicity study to evaluate the distribution of CD3 (T cells), CD20 (B cells), CD68 (macrophages), human IgG (indicative of UCBFab), endogenous monkey immunoglobulins (IgG, IgM, and IgA), and complement components (C3 and sC5b-9 [C9]) at the end of the treatment phase or the recovery phase. Additionally, the distribution and characterization of inflammatory cells positive for CD4, CD8, CD25, and CD206 was evaluated.
Positive and negative controls were used in every staining run. The positive controls for CD3 (Dako; catalog no. A0452), CD68 (Dako; catalog no. IR609), and CD20 (Dako; catalog no. M0755) were FFPE cynomolgus monkey mesenteric lymph node and spleen, and negative control was FFPE cynomolgus monkey skeletal muscle. The positive/negative controls for human IgG (confidential info.), monkey IgG (Bethyl Laboratories; catalog no. A140-202P), IgM (Bethyl Laboratories; catalog no. A80-100P), IgA (Bethyl Laboratories; catalog no. A80-102P), C3 (Quidel; catalog no. A304), and C9 (sC5b-9; Dako; catalog no. M0777) detection reagents were FFPE human placenta (positive and negative human IgG, IgA, IgM, C3, and sC5b-9 control tissue elements and monkey IgG-negative control tissue elements), monkey liver (human IgG-negative control tissue elements and monkey IgG, IgA, IgM, and sC5b-9-positive and sC5b-9-negative control tissue elements), and monkey lung (human IgG-negative control tissue, monkey IgG, IgA, IgM, and sC5b-9 positive and sC5b-9-negative tissue control elements). The positive controls for CD4, CD8, CD25, and CD206 were FFPE cynomolgus monkey spleen and thymus, and the negative controls were FFPE cynomolgus monkey lung and liver.
Rabbit anti-chicken IgY was used as the negative control antibody for the CD3 detection reagent, mouse anti-rabies virus was used as the negative control antibody for the CD20 detection reagent, whereas mouse anti-chicken IgY was used as the negative control antibody for the CD68 detection reagent. UV-resin spot slides with UCBFab, human IgG, and 2 irrelevant proteins were also used as control materials. Goat anti-rabbit IgG was used as the negative control antibody for the human IgG, monkey IgG, IgM, and IgA detection reagents, while goat anti-chicken IgY was used as the negative control antibody for the C3 detection reagent and mouse anti-rabies virus was used as the negative control antibody for sC5b-9.
The staining was performed according to internal standard operational procedures at CRL Frederick. Immunoperoxidase staining was used for demonstration of potential immune complex-related granular deposits. Additionally, one slide from each tissue was stained using H&E to act as an adjunctive slide for comparison only in the IHC evaluation. Suitable tissues from the Test Site paraffin tissue bank as well as commercially available reagents were used as the positive and negative control materials for IHC staining analyses, including lymph node, spleen, thymus, skeletal muscle, liver, and lung from cynomolgus monkeys and/or human placenta; UV-resin spot slides with UCBFab, human IgG, and 2 irrelevant proteins (recombinant human Oncostatin M and human hypercalcemia of malignancy peptide, amino acid residues 1-34) were also used as control materials. The average number of immunopositive cells across all sections was estimated by eye.
Statistical Analysis
For mouse studies, the following sequence of statistical tests was used for body weight, food consumption, clinical pathology, and organ weight data: A parametric analysis was performed if Bartlett test for variance homogeneity was not significant at the 1% level and a nonparametric analysis was performed if Bartlett test was still significant at the 1% level following both logarithmic and square-root transformations. For organ weight data, analysis of covariance was performed using terminal body weight as covariate unless nonparametric methods were applied. For clinical pathology data, if 75% of the data (across all groups) were the same value, Fisher exact tests were performed (see Appendix 1).
For NHP studies, the following statistical tests were used for body weight, food consumption, clinical pathology, and organ weight data: Parametric and nonparametric statistical tests were conducted at the 5% significance level. All pairwise comparisons were conducted using 2-sided tests and were reported at the 0.1%, 1%, and 5% levels. Levene test was used to assess the homogeneity of group variances and a 1-way analysis of variance F test if Levene test was not significant or the Kruskal-Wallis test if it was. Pairwise comparisons were conducted using Dunnett or Dunn test. A t test was performed if Levene test was not significant or Wilcoxon rank-sum test if it was.
Results
Studies and Dosing
Cynomolgus monkeys were treated with a nebulized aerosol formulation of UCBFab at 0.3 to 30 mg/kg/d, whereas CD-1 mice were treated with a nebulized aerosol formulation of UCBFab at 5 to 20 mg/kg/d (Table 1). Intended dose and measured dose were very similar for both species.
Nonhuman Primate
Treatment with UCBFab was well tolerated at all doses and dose regimens with no biologically significant effects on clinical signs, body weight, body weight gain, ophthalmoscopy, electrocardiology, auscultation, urine analysis, coagulation, clinical pathology, immunophenotyping, or respiratory function assessment. The particle size was in the expected range for deep lung deposition (MMAD 2-5 µM; geometric SD up to ±2.8 µM).
Exposure
In the PK study, serum bioanalysis demonstrated systemic exposure in all animals, whereas BALF bioanalysis demonstrated deposition of test item in the lungs of all animals. The delivered doses were within −15% to +12% of the target. UCBFab accumulation between the first and the fourth dose was observed in the 2 dose groups, with an accumulation index of 2.1 (Cmax) and 8.1 (area under the curve [AUC]) for 1 mg/kg and 3.2 (Cmax) and 17.6 (AUC) for 20 mg/kg. After the fourth repeat dose in BALF, accumulation was observed for all doses, with accumulation indexes based on AUC of 6.9 and 12.7 for 1 and 20 mg/kg, respectively. Interestingly, there was a good correlation between serum and BALF exposure (Figure 1A), before ADA interference developed. UCBFab concentrations observed in BALF were higher than those in the systemic circulation as can be expected for a Fab fragment, in agreement with the low permeability of large proteins and the absence of active transport via FcRn (Figure 1A).
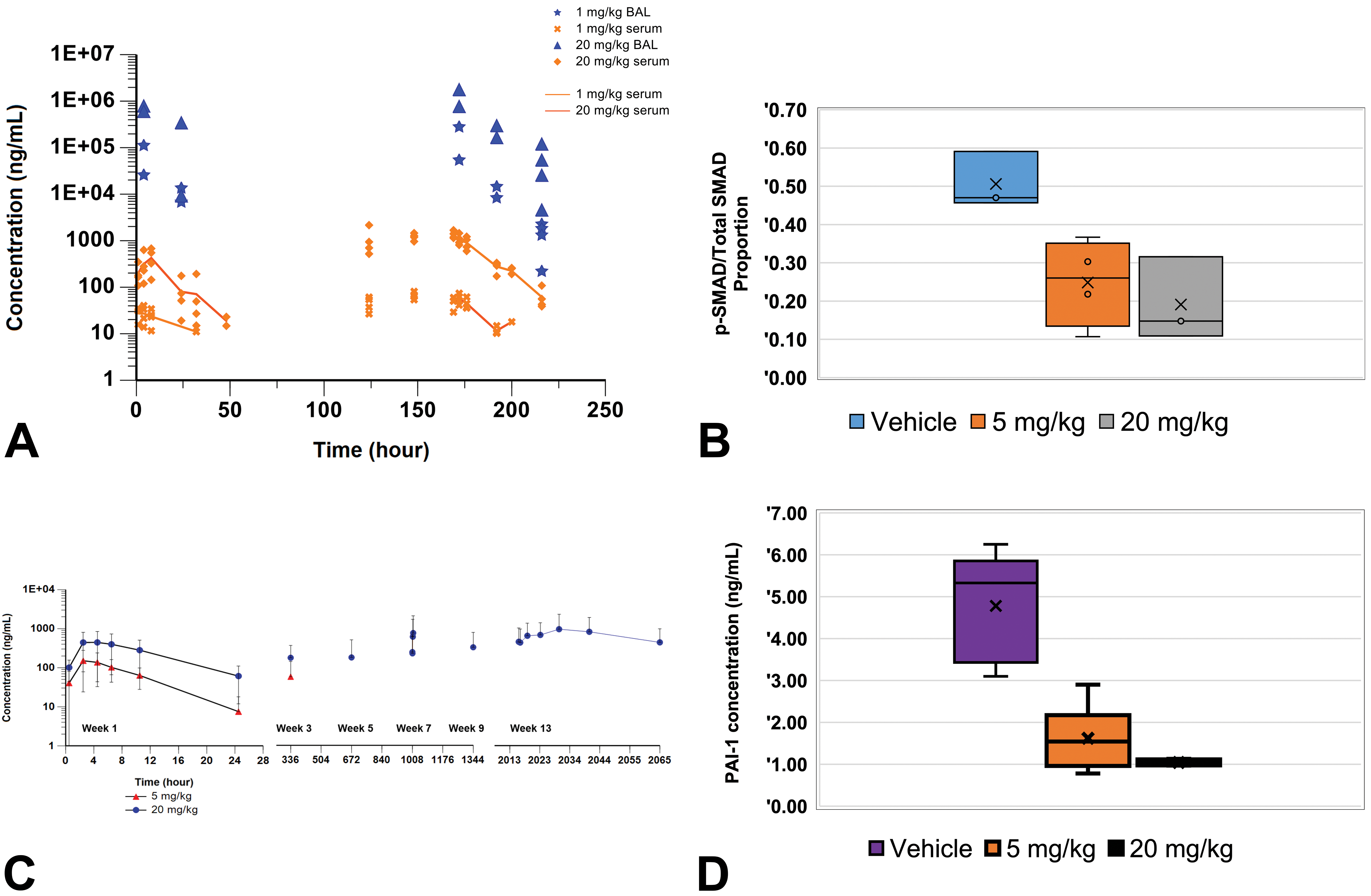
A, UCBFab exposure in serum of cynomolgus monkeys after a single daily dose, followed by 3 consecutive daily doses, 5 days later at 1, 5, and 20 mg/kg. Bronchoalveolar lavage (BAL) fluid was collected from monkeys in a sparse manner due to the invasiveness of the procedure at 4, 24, 32, and 48 hours after dosing as indicated with 2 samplings per time point, except at necropsy where all 4 animals were sampled. n = 4 per group. Due to the misdosing of the 5 mg/kg group, this group was excluded from the study. B, Levels of pSMAD2 versus total SMAD2/3 in the BAL fluid of cynomolgus monkeys. n = 3 to 4 as indicated. C, Mean serum concentrations after daily administration through inhalation of 5 or 20 mg/kg of UCBFab. D, Mouse plasminogen activator inhibitor-1 levels in BAL fluid taken from lungs of untreated and treated mice. UCBFab indicates humanized antigen-binding fragment antibody.
In the 2-week study, exposure over the 2-week period was maintained at both 5 and 20 mg/kg of UCBFab dose levels. Exposure was higher in some animals after the last dose and appeared supraproportional. The AUC accumulation index was estimated to be approximately 9 at 5 mg/kg and 7 for the 20 mg/kg dose. Exposure was also confirmed in lung homogenates and in BALF, which was collected 4 hours after the last dose. In this study, phosphorylated SMAD2/3 was shown to be downregulated in BALF cells, as illustrated by a decrease in the ratio between pSMAD2/3 and total SMAD2/3, demonstrating the expected pharmacological effect upon inhibition of TGFβ (Figure 1B).
In the 4-week study, animals test article, measured 24 hours after the last dose, was present at higher levels in BALF than in serum, indicating slow absorption into the circulation. Overall serum exposure was similar between daily and weekly dosing on day 1 (AUC0-t: 296 ng·hr/mL vs 264 ng·hr/mL). However, concentrations became undetectable in a number of animals given daily dosing over time (most likely due to ADA given the high levels detected in the 13-week study), whereas accumulation was noted in the weekly dosed animals (AUC0-t was 1090 ng·hr/mL on day 22).
In the 13-week study, animals’ systemic exposure on day 1 was comparable to that observed in the 2-week DRF study. All animals treated at 20 mg/kg/d showed systemic exposure throughout the study, whereas no quantifiable serum exposure was measured for any animals in the 5 mg/kg/d dose group from week 5 onward, presumably due to clearing and/or neutralizing ADA. After 13 weeks of dosing, inhalation of UCBFab at 20mg/kg/d led to accumulation indexes of 6.9 (Cmax) and 11.6 (AUC), respectively (similar to other studies; Figure 1C).
Immunogenicity
Antidrug antibodies were measured in the serum for the PK study and 4- and 13-week study. In the PK study, serum samples were collected predose and at days 1, 6, 7, and 8. All predose samples were negative. At the end of the study (day 8), all but 1 animal (treated at 1 mg/kg/d) remained negative for ADA. The positive animal had very low values, as could be expected based on the timing of sample collection after the first dose.
In the 4-week study on day 1, all animals were ADA negative. Animals dosed daily rapidly seroconverted, with only 3/8 animals at 0.3 mg/kg/d remaining negative on day 15 and all animals being positive by day 28. By contrast, in the group dosed at 1 mg/kg/wk, all animals remained negative on day 15 and 2 of 8 animals were still negative after 4 weeks.
In the 13-week study, a similar high incidence of ADA was noted. All animals tested negative for anti-UCBFab antibodies prior to treatment, but all treated animals seroconverted, resulting in 100% treatment-emergent ADA by week 3 and on all subsequent occasions. The titer increased from week 3 to week 7 and remained at high levels throughout the rest of the sampling time points up to the end of the dosing period in both dose groups. The titer, expressed as normalized signals, tended to be higher for animals treated at 5 mg/kg/d compared to those treated at 20 mg/kg/d. This was matched by the loss of exposure as demonstrated in Figure 1C.
Unscheduled Deaths
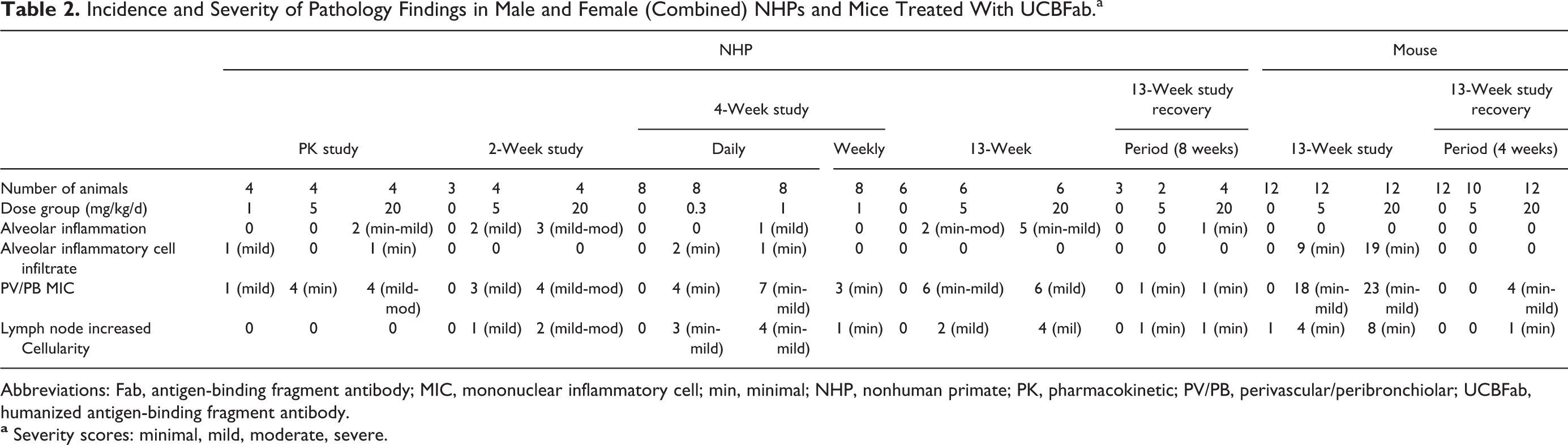
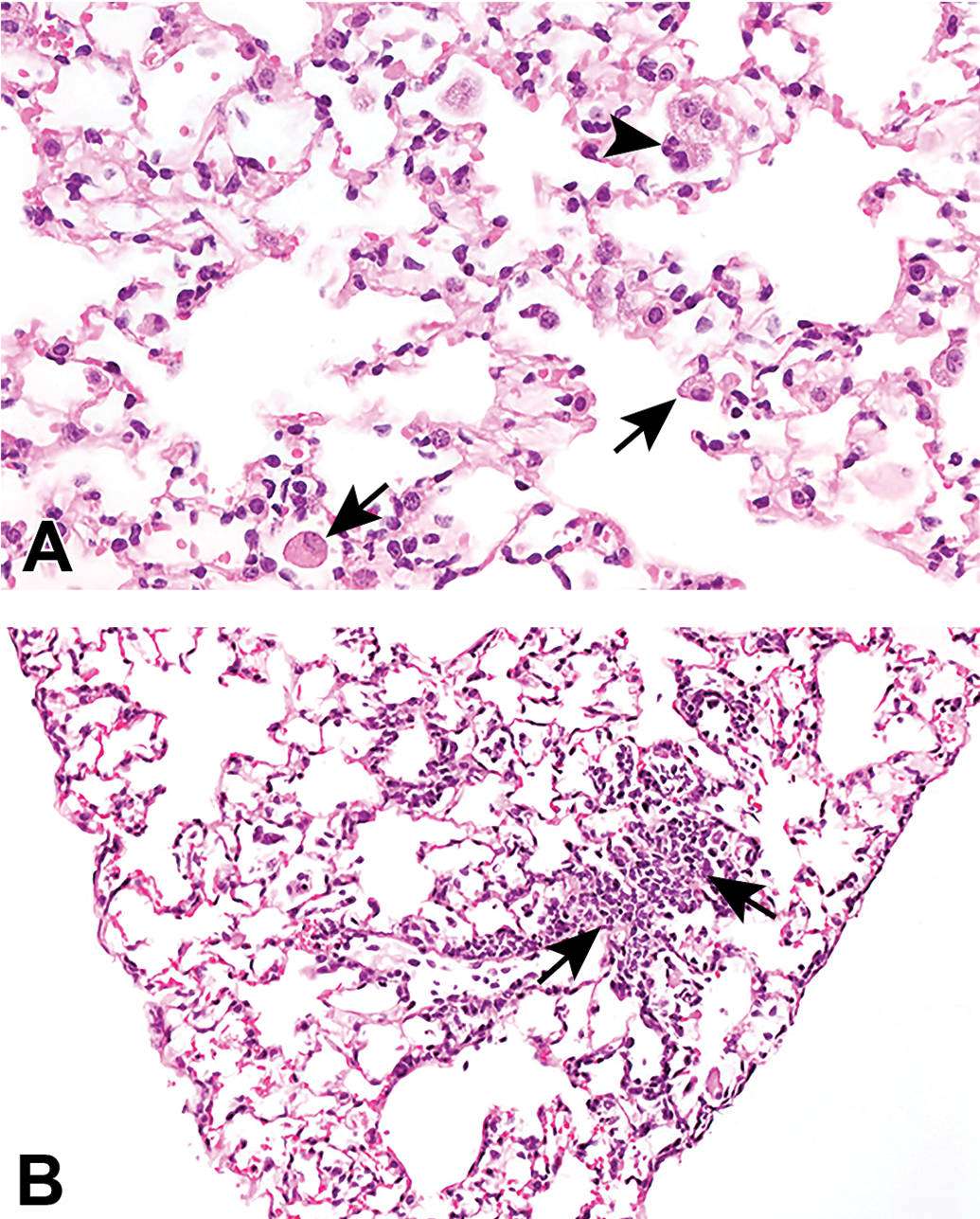
No mortalities were seen in any of the NHP studies that could be clearly ascribed to UCBFab. In the 13-week study, 1 animal treated with UCBFab at 5 mg/kg/d was euthanized on treatment day 37 after a brief period of illness lasting 2 days consisting of high rectal temperature (40.1 °C), poor condition, and abnormal lung sounds. Necropsy revealed a firm, mottled, and enlarged right lung lobe with adhesions between the right lung lobes, and enlarged bronchial lymph nodes. Histological examination of the right caudal lung lobe showed marked alveolar inflammation, macrophage aggregation, and exudation (correlating with firm, mottled, and enlarged noted at necropsy), with pleural inflammation extending to the adjacent lung lobes (correlating with adhesions; Figure 2A and B). Treatment-related findings of a minimal severity (which were similar to those observed in other treated animals) were seen diffusely throughout the other lung lobes. The cause of death was mixed bacterial pneumonia, which was culture positive for Streptococcus and Pseudomonas. The cause of the infection was considered secondary to the inhalation procedure as no other cases were recorded in a total of 5 NHP and 2 mouse studies and there was no indication of immunosuppression induced by UCBFab.
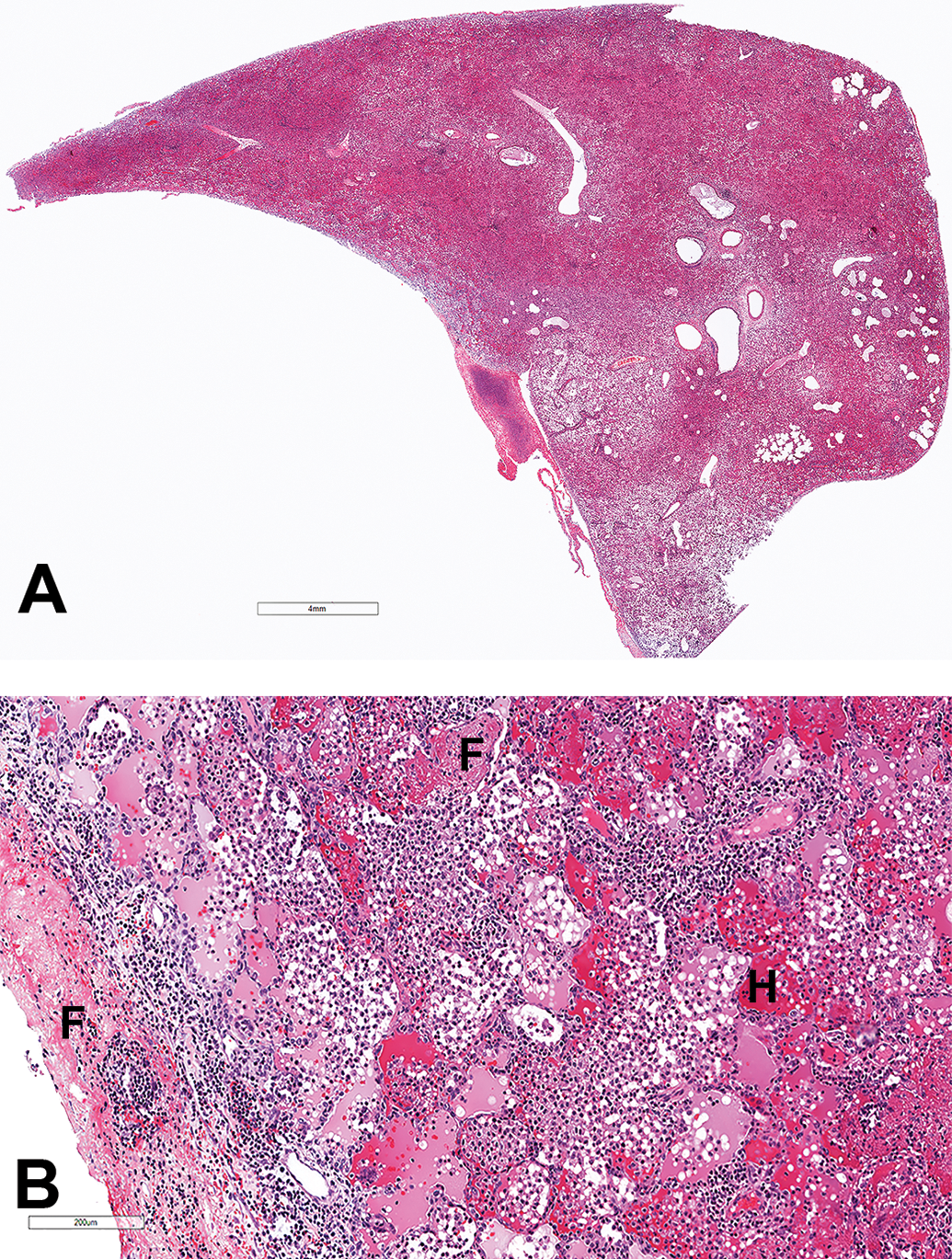
Low- and high-power hematoxylin and eosin stained images of NHP lung showing (A) consolidation of lung lobe and (B) intra-alveolar hemorrhage (H), fibrin exudation (F), and mononuclear inflammatory cells (predominantly lymphocytes and macrophages). Fibrinous exudate and inflammatory cells extend to the pleural surface. NHP indicates nonhuman primate.
Necropsy
In the PK study, abnormal consistency (“spongy”) of the lungs was noted in the majority of UCBFab-treated animals with discoloration (mottled appearance) in some animals which correlated occasionally with agonal congestion. This finding correlated with pink stained lavage fluid noted after the inhalation procedure and most likely reflected iatrogenic damage with hemorrhage due to the insertion of the cannula tube. However, BALF was collected under anesthesia in 2 occasions over a short period of time.
Enlargement of the draining tracheobronchial lymph node was observed in the 2- and 4-week NHP studies in animals treated at ≥1 mg/kg/d (females) and at ≥0.3 mg/kg/d (males). This macroscopic change generally correlated with increased lymphoid cellularity of the lymph node. These macroscopic changes were not reported after 13 weeks of treatment. No effects were noted on organ weights.
Histopathology
Treatment with UCBFab resulted in microscopic changes that were multifocal and generally minimal-to-mild severity. Changes were only noted in the lungs and draining tracheobronchial lymph nodes, due to local exposure to UCBFab, and were consistent across studies, regardless of the duration.
In the PK study, changes consisted of either (1) minimal-to-mild chronic active/mixed alveolar inflammation (hereafter termed alveolar inflammation) or, in some animals, minimal-to-mild inflammatory cell infiltration, and (2) mild-to-moderate PV/PB mononuclear cell infiltrate. Alveolar inflammation extended to the pleura in some animals and was characterized by macrophage and neutrophilic infiltration with erythrophagocytosis and type 2 pneumocyte hyperplasia. The PV/PB mononuclear cell infiltrate consisted principally of lymphocytes but with neutrophils, lymphocytes, and monocytes in 2 animals (Table 2).
Incidence and Severity of Pathology Findings in Male and Female (Combined) NHPs and Mice Treated With UCBFab.a
Abbreviations: Fab, antigen-binding fragment antibody; MIC, mononuclear inflammatory cell; min, minimal; NHP, nonhuman primate; PK, pharmacokinetic; PV/PB, perivascular/peribronchiolar; UCBFab, humanized antigen-binding fragment antibody.
In the 2-week study, changes in the lungs consisted of minimal-to-moderate alveolar inflammation characterized by increased alveolar macrophage aggregation and multinucleate giant cells together with type 2 pneumocyte hyperplasia and scattered polymorphonuclear cells in the bronchioloalveolar and PV areas. In the airways, mild-to-moderate PV/PB mononuclear infiltrate was present composed principally of lymphocytes that extended into the respiratory bronchioles in some animals. Both these changes were present in a subset of animals treated at 5 and 20 mg/kg/d with no obvious sex bias. Diffuse mild increased lymphoid cellularity of the tracheobronchial lymph node cortex was also present in 1 female at 5 mg/kg/d and one female at 20 mg/kg/d (Table 2).
In the 4-week study with daily dosing, similar but slightly less severe changes (compared to the PK and 2-week studies) were seen in the airways characterized by a dose-dependent minimal-to-mild PV/PB MIC infiltrate, which was sometimes associated with prominent bronchial-associated lymphoid tissue. In the alveoli, 1 male treated at 1 mg/kg/d and 1 female treated at 0.3 mg/kg/d showed minimal-to-mild alveolar inflammation. These changes were focal in distribution and characterized by an accumulation of macrophages with fewer neutrophils in the alveolar spaces and interstitial tissue and were associated with type 2 pneumocyte hyperplasia, hemorrhage with erythrophagocytosis, and/or cell debris in alveoli. In other animals, alveolar changes were less severe and were characterized by minimal alveolar mixed cell infiltration consisting of a scant accumulation of macrophages and neutrophils occupying alveolar spaces. Minimal-to-mild increased lymphoid cellularity was seen in the tracheobronchial lymph nodes in all male and 1 female animals (treated at 1 mg/kg/d). Less severe changes were seen in animals treated weekly despite similar overall exposures, consisting of only minimal PV/PB MIC infiltrate (without alveolar inflammation or inflammatory cell infiltrates) and 1 animal with minimal increased lymphoid cellularity of the tracheobronchial lymph nodes (Table 2).
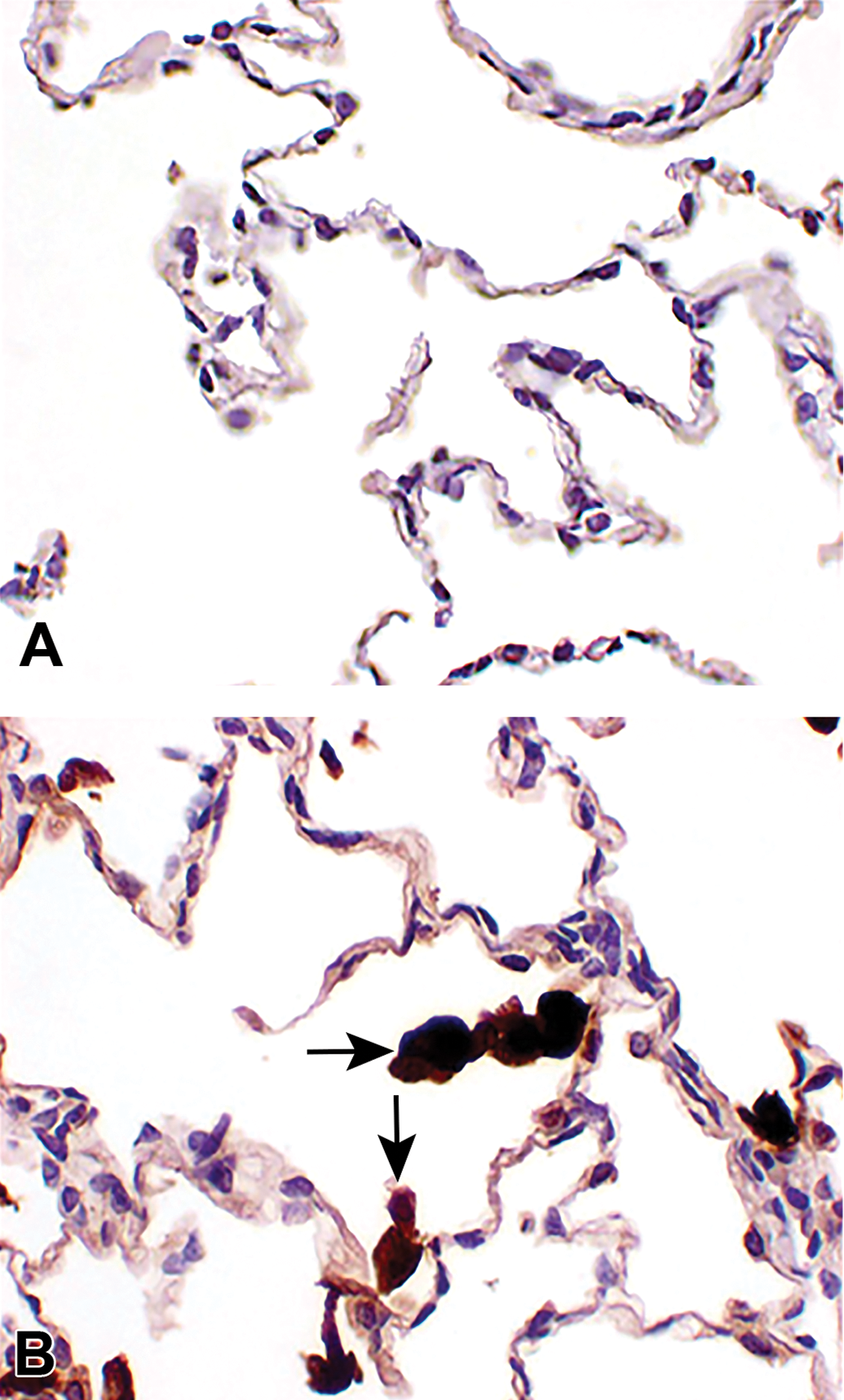
In the 13-week study (Table 2), similar changes were seen to those in the 2-week study, characterized by minimal-to-mild PV/PB MIC (Figure 3A and B) and minimal-to-moderate alveolar inflammation with macrophages, mononuclear cells (Figure 3B), and occasional neutrophils present within alveolar septa and airspaces, and mild increased cellularity of the tracheobronchial lymph nodes (Table 2). In the most severe cases, alveolar inflammatory cell and PV/PB MIC infiltrates became confluent, leading to distension and thickening of terminal/respiratory bronchioles and adjacent alveoli (Figure 3C).
Low- and high-power hematoxylin and eosin–stained images of NHP lung showing (A) PV/PB mononuclear cell infiltrates (arrowheads) arranged as focal aggregates of cells in a predominately PV and PB pattern, and (B) PV/PB aggregate (arrows) arranged around smaller airways and vessels with mononuclear cells (principally macrophages and lymphocytes) free within alveoli. C, A more severe case of alveolar inflammatory cell infiltrate and PV/PB infiltrate showing confluence and distension of terminal/respiratory bronchioles and alveoli by an MIC infiltrate. Alveolar macrophages show a conspicuous granular cytoplasm (arrows). MIC indicates mononuclear inflammatory cell PV/PB, perivascular/peribronchiolar. NHP indicates nonhuman primate.
Recovery
An 8-week recovery arm was included in the 13-week study. At the end of the treatment-free period, PV/PB MIC and alveolar inflammation were present with reduced incidence and severity, indicating partial recovery (Table 2).
Immunohistochemistry Phenotyping of Inflammatory Cell Infiltrates
Immunohistochemistry was performed on the lung to determine whether the changes seen during histological examination might be due to immune complex disease (ICD). Immune complex disease presents classically as a multi-organ vasculitis and/or glomerulonephritis for systemically administered immunogenic biotherapeutics and is thought to be due to deposition of granular deposits composed of test article, host antibody (IgG, IgM, or IgA), and/or complement. 16
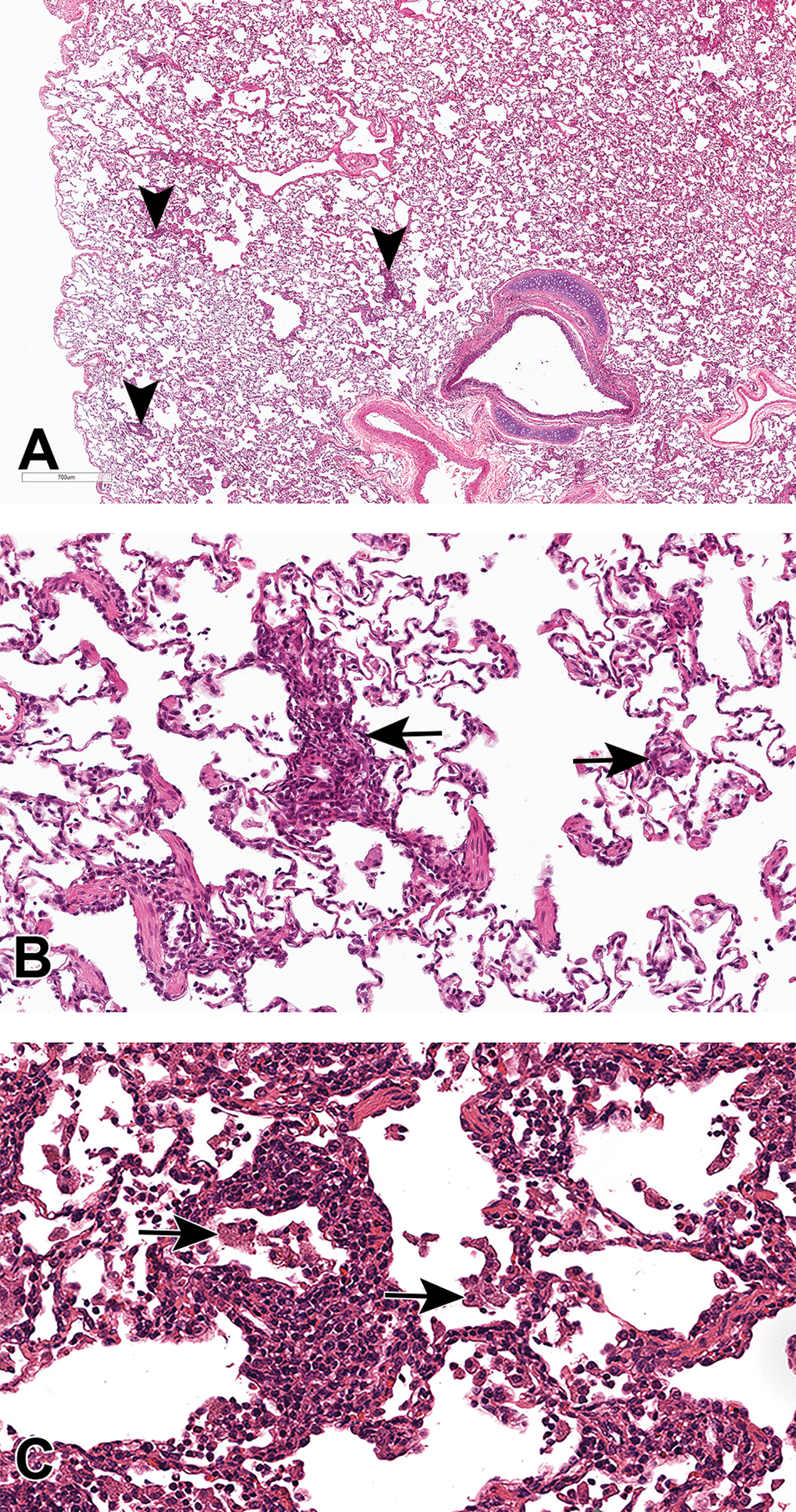
Histopathological evaluation, however, revealed no evidence of granular deposits, indicating that ICD was unlikely to be the cause. Test article was detected in alveolar macrophages (Figure 4A and B) and pneumocytes lining alveoli in the lungs of most UCBFab-dosed main study animals, as well as intravascular and/or extravascular in the lungs of select UCBFab-dosed main study animals as expected. The test item was not detected in the lungs of UCBFab-dosed recovery study animals 8 weeks after treatment cessation.
Immunohistochemical stained images showing the presence of test article (antihuman immunoglobulin G primary antibody) in (A) control and (B) treated animals with strong staining within macrophages (arrows).
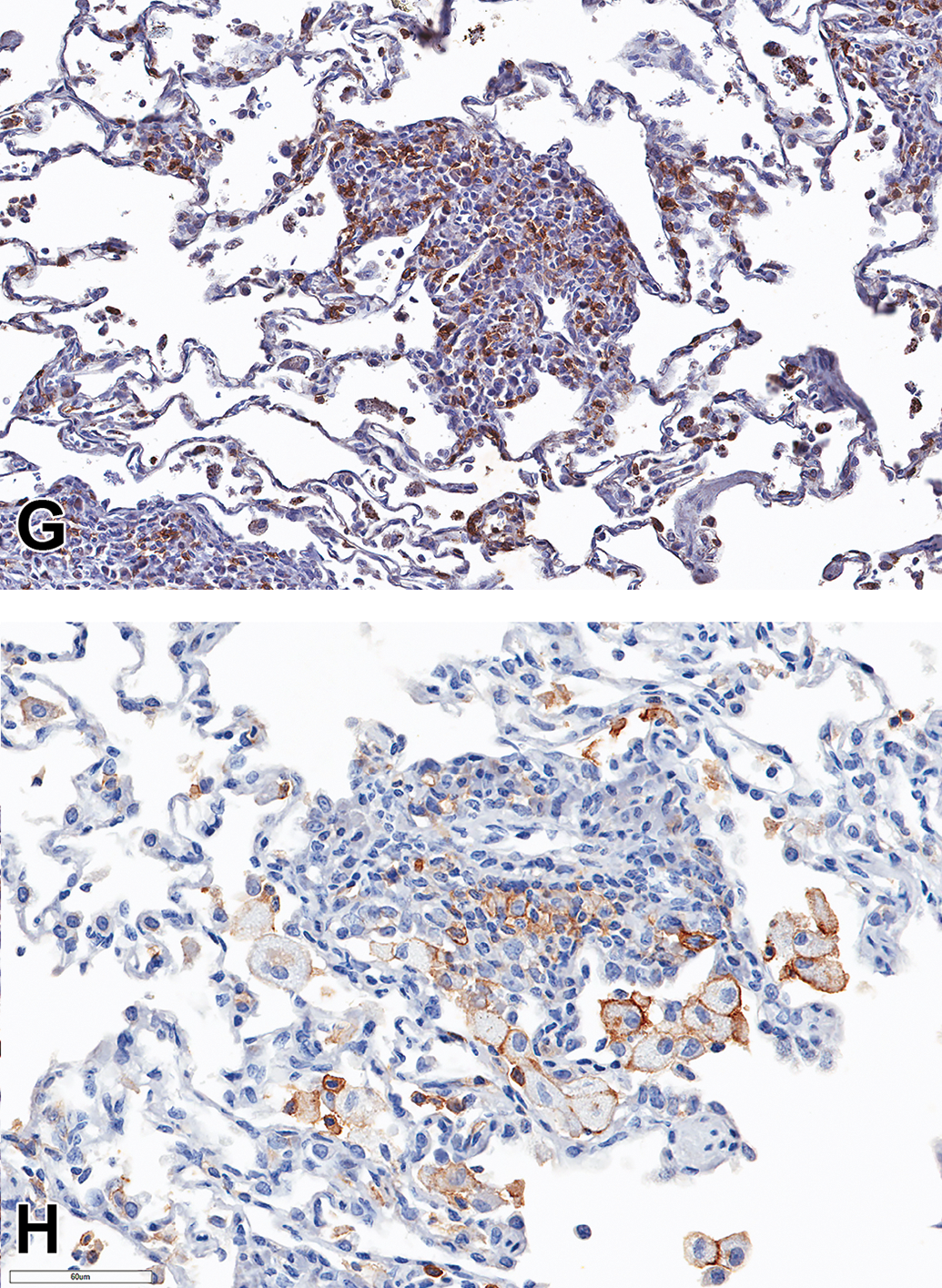
CD3 (Figure 5A), CD4 (Figure 5B), and CD68 (Figure 5C and D) IHC markers were performed on the inflammatory cell infiltrates seen in the airways and alveoli of the lung from animals treated with UCBFab at 5 and 20 mg/kg/d. In the alveoli, >75% of cells were CD68-positive macrophages, with the majority of the remaining cells staining for the T-cell marker CD3 (up to 25%; Figure 5A) and <1% staining for the B-cell maker CD20 (Figure 5E, arrow heads). Further characterization in 2 animals demonstrated that all alveolar macrophages displayed the M2 activation marker, CD206 (Figure 5F). The remaining mononuclear cells (predominantly lymphocytes) stained more frequently with CD8 (up to 60%-80%; Figure 5G) compared to the CD4 marker (c. 30%; Figure 5B), with approximately 20% to 50% of cells displaying the activation marker, CD25 (IL2Rα; Figure 5H and Table 3).
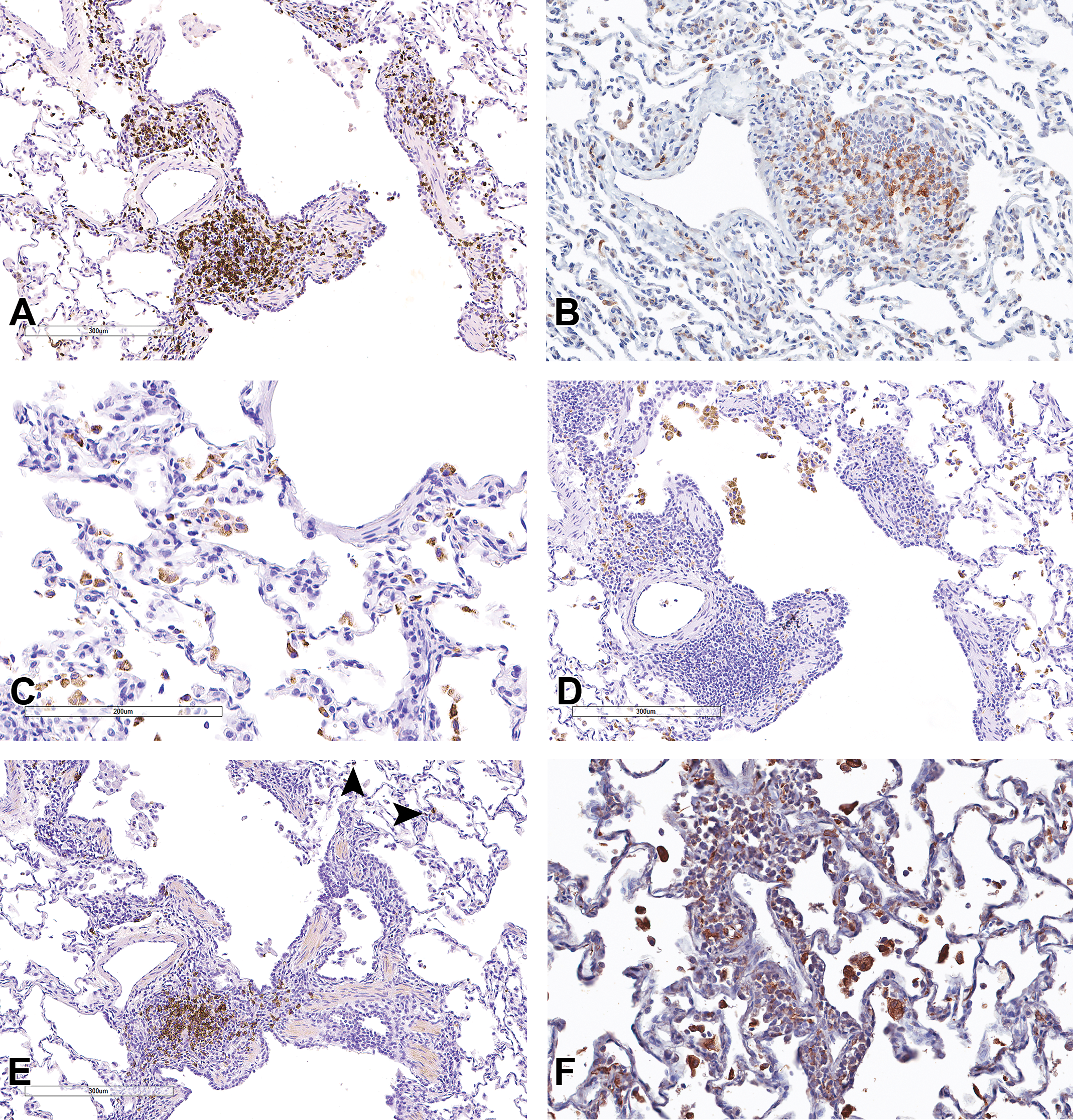
Immunohistochemical stained images (A-H) showing immunophenotyping of lymphocytes and macrophages in the airway perivascular/peribronchiolar infiltrates and alveoli. Immunostains used were (A) CD3, (B) CD4, (C and D) CD68, (E) CD20 (arrowheads indicate low frequency of CD20 cells in alveoli), (F) CD206, (G) CD8, and (H) CD25.
Percentage Frequency of Positively Stained Cells in the Inflammatory Infiltrates of NHPs Treated With UCBFab Split by Anatomical Region.
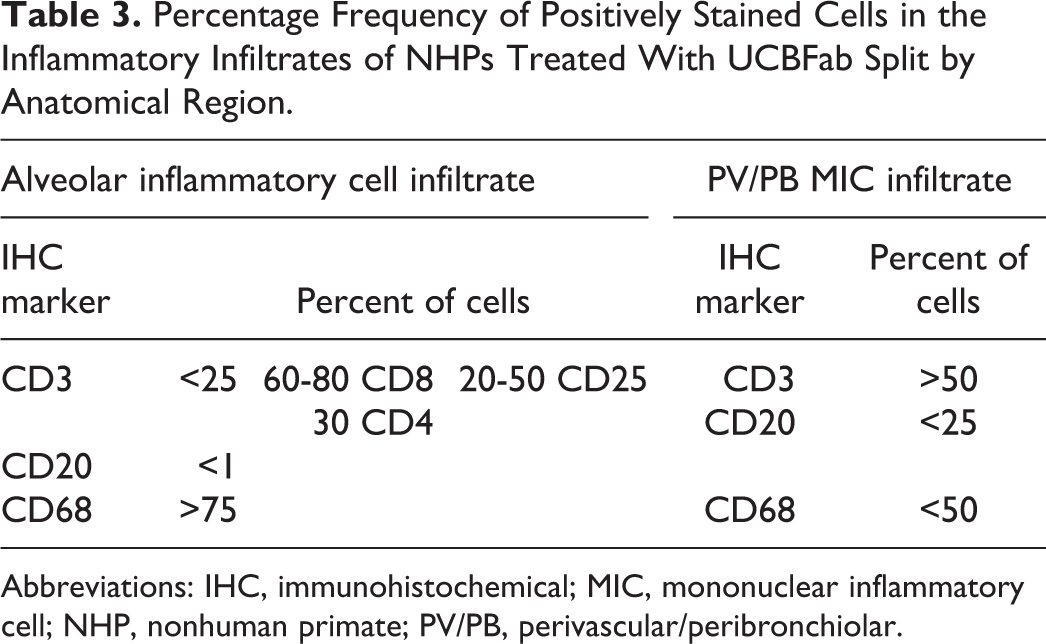
Abbreviations: IHC, immunohistochemical; MIC, mononuclear inflammatory cell; NHP, nonhuman primate; PV/PB, perivascular/peribronchiolar.
In the PV/PB MIC infiltrates, the composition of the infiltrates was quite variable, presumably reflecting the dynamics of an ongoing immune response. The majority of cells were CD3-positive T cells (>50%); Figure 5A), with the remaining cells being CD20-positive B cells (variable but overall <25%; Figure 5E) and with a variable infiltrate of CD68-positive macrophages (up to 50% in some foci; Figure 5D and Table 3).
Bronchoalveolar Lavage Fluid Changes
Bronchoalveolar lavage and analysis was performed in both the NHP PK and 4-week studies to assess changes in cell populations induced by treatment with UCBFab. In the PK study, BALF was performed twice each for all animals on week 4 and week 1 and then again on days 1 and 8 at either 4 or 24 hours postdose. On day 10, all animals were sampled approximately 48 hours after the last dose (day 8). No changes were seen in total cell counts. Similarly, BALF immunophenotyping showed no changes in the total T-cell population, T-cell subpopulations, NK cells, monocytes, B cell, or subpopulations of B cells.
In the 4-week study, BALF samples were collected at the end of the study. The lungs were perfused with 0.9% saline to flush out intravascular blood and then removed and weighed before obtaining a BALF sample from the left caudal lobe. In control animals, nucleated cell counts (range: 320-790/µL BALF) and leukocyte counts (range: 3-9 neutrophils, 13-119 lymphocytes, and 0-10 eosinophils/µL BALF) were very low (Table 4). Treatment with UCBFab at 0.3 and 1 mg/kg/d resulted in a dose-responsive mild-to-moderate increase in total nucleated, neutrophil, lymphocyte, and eosinophil cell counts (Table 4). Similar mild-to-moderate changes were noted for animals treated on a weekly schedule but with slightly fewer lymphocytes present in the BALF (which possibly correlated with the decreased ADA noted in this treatment group). For both dosing schedules, the presence of inflammatory cells in the BALF correlated with the presence of inflammatory cells noted in the lungs at histopathology (mixed inflammatory cell infiltration at 0.3 mg/kg/d and infiltration/inflammation at 1 mg/kg/d). In addition, BALF analysis indicated a significant increase in total nucleated cell counts, which was likely reflected the significant increase in alveolar macrophages seen at histopathology.
NHP BAL Fluid Analysis Showing Number of Control Animals Versus Treated Animals (Sex Combined) Together With Severitya of Change and Total Cell Count (in Bold).

Abbreviations: BAL, bronchoalveolar lavage; NHP, nonhuman primate; –, No change in measured parameter.
a Severity based on clinical pathologist judgement.
Mouse
Treatment with UCBFab at 5 and 20 mg/kg/d was well tolerated, with no drug-related mortality or significant effects on clinical signs, food consumption, body weight, ophthalmology, clinical chemistry, or immunophenotyping of the spleen.
Exposure
In the 2-week study, mice were dosed with 5 and 20 mg/kg/d. Although only 2 doses were compared, the PK was nonlinear between the 2 doses. Both Cmax/D and AUC/D increased between the first and last dose, demonstrating accumulation of UCBFab. The AUC accumulation was observed to be 33 and 46, respectively, for the low and high dose. Mean t1/2 estimates of 7.2 and 6.1 hours at the low and high dose, respectively, were as expected for a Fab. UCBFab content was also evaluated in BALF samples and lung tissue collected 2 or 6 hours after the last dose. UCBFab in BALF samples and lung homogenates from UCBFab-treated animals showed approximate dose proportionality between the 2 dose groups. The PAI-1 measurement in BALF showed a dose-dependent decrease in concentration with increased dose of UCBFab, whereas free TGFβ showed a simultaneous dose-response increase, thus showing a strong pharmacological effect (data not shown).
In the 13-week study, animals were dosed at 5 and 20 mg/kg/d of UCBFab. Inhalation of UCBFab at 5 and 20 mg/kg/d led to low peripheral exposure (AUC0-24 h day 1 below level of quantification and 346 ng·h/mL, respectively, for doses of 5 and 20 mg/kg, and accumulation over time with AUC0-24 h of 2000 and 12,000 ng·h/mL, respectively, on day 92), but high local exposure 2 to 6 hours after last dose (approximately 100 and 300 µg/mL BALF, or 45 and 90 µg/g lung tissue for the doses of 5 and 20 mg/kg, respectively), with an approximately 35-fold accumulation in BALF compared to first dose administration. Analysis of BALF showed increases in TGFβ (2.2× and 4.9× control values for both sexes combined) and decreases in PAI-1 (0.4× and 0.2× control values for both sexes combined) consistent with the expected pharmacologic action of UCBFab and similar to those observed in the 2-week study.
Immunogenicity
The presence of ADA was not evaluated in the 2-week study, and TKs did not indicate a potential interference. In the 13-week study, a low titer (within 2× screening cut point) of possibly preexisting antibodies to UCBFab was found on day 1 in all control mice and in 4 f 30 mice given 5 mg/kg/d and 2 of 30 mice given 20 mg/kg/d. As was noted in the primate studies, treatment with UCBFab resulted in a high incidence of treatment-emergent ADA, with most mice strongly positive (titers 100- to 1000-fold above the cut point) in both dose groups (24/30 mice treated at 5 mg/kg/d and 21/29 mice treated at 20 mg/kg/d).
Necropsy
No test item–related macroscopic abnormalities were noted at necropsy in either the 2- or 13-week studies. Statistically significant slight increases in female relative kidney organ weight (1.15× and 1.10× adjusted to body weight for low- and high-dose vs control group) and female lung weights (1.10× and 1.22× adjusted to body weight for low- and high-dose vs control group) were noted at necropsy in the 13-week study. At the end of the 4-week recovery period, kidney and lung organ weights were still slightly increased, but not statistically significant. No histological correlate could be associated with the increased organ weights noted in female animals, although it is possible that in the lungs, the inflammatory cell infiltrates contributed to this increase.
Histopathology
No test item–related changes were noted in any of the tissues examined in the 2-week study. In the 13-week study, the only changes observed were in the lungs and draining tracheobronchial lymph nodes. Similar but less severe changes than those noted in the 13-week NHP study were seen in mice treated with UCBFab at 5 and 20 mg/kg/d, characterized by minimal-to-mild dose-responsive PV/PB MIC in the airways of the lungs, minimal multifocal alveolar inflammatory cell infiltration (Figure 6A and B), and minimal increased lymphoid cellularity of the tracheobronchial lymph nodes (Table 2). Alveolar inflammatory cell infiltrates consisted of foamy macrophages and occasional lymphocytes (with neutrophils present in a few animals). Finally, in the draining tracheobronchial lymph nodes, minimal increased diffuse lymphoid cellularity of the cortex was noted.
Medium power hematoxylin and eosin–stained images of mouse lung showing (A) minimal mixed alveolar inflammatory cell infiltrate characterized predominantly by macrophages (arrows) with occasionally acute inflammatory cells (arrowhead) and (B) an area of predominantly perivascular/peribronchiolar lymphocytic infiltrate (arrows) into the terminal airways.
Recovery
At the end of the 4-week recovery period, a reduced incidence of PB/PV MIC and increased tracheobronchial lymph node cellularity were seen in animals previously treated with UCBFab at 20 mg/kg/d, indicating partial recovery. No alveolar changes were seen indicating full recovery in this compartment of the lung (Table 2).
Clinical Pathology
Hematology
No significant changes were noted in the 2-week study. The only changes in the 13-week study were slight increases in lymphocytes above the concurrent control range (1.12× and 1.41×) in male animals treated at 5 or 20 mg/kg/d, respectively. Female data were not available due to sample clotting.
Bronchoalveolar Lavage Fluid Changes
Bronchoalveolar lavage fluid analysis showed approximately dose proportional increase of UCBFab in the treated groups with approximately equal spread around the mean, whereas analysis of TGFβ and PAI-1 showed a dose-responsive accumulation of TGFβ and a dose-dependent decrease in concentration of PAI-1.
Discussion
Treatment with a nebulized aerosol formulation of UCBFab in cynomolgus macaques and CD-1 mice via the inhalation route at dosages between 0.3 and 20 mg/kg/d in a range of GLP and non-GLP toxicity studies resulted in very low systemic exposure as compared to lung or BALF achieved concentrations and was generally well tolerated with no significant effects on clinical signs, body weight, body weight gain, ophthalmoscopy, electrocardiology, auscultation, urine analysis, coagulation, clinical pathology, immunophenotyping, or respiratory function assessment. Two notable features were observed: (1) immunogenicity with a very high frequency of seroconversion in both species and decreases in systemic exposure and (2) pathological changes restricted to the lungs and draining lymph nodes, but no systemic effects. These changes were similar for both NHP and mouse and showed no significant morphologic progression over time. These results were in contrast to those normally seen with parenteral administration, where the incidence of immunogenicity is generally much lower and additional target organs beyond the respiratory tract are normally seen due to increased systemic exposure.
No mortalities could be clearly ascribed to treatment with UCBFab; however, one monkey treated at 5 mg/kg/d was euthanized after 37 days of treatment due to mixed Streptococcus and Pseudomonas pneumonia. These bacteria are normal residents within the oral cavity but are known to form aspiratable biofilms that can induce community-acquired and nosocomial (ventilator-associated) pneumonia in humans. 17,18 Since BALF was collected at week 20 prior to the start of the study and again at necropsy, it was unlikely that this was the cause of infection and therefore derived from other sources such as contamination of the rebreathing apparatus or possibly aspiration of orally derived, bacterially laden fomites, followed by penetration and colonization of the deep lung parenchyma. However, pneumonia is not generally regarded as a complication in inhalation studies.
Immunogenicity, ADA, and Exposure
Inhalation of UCBFab resulted in high local but low systemic exposure, indicating limited absorption into the circulation (Figure 1A) due partly to the inability to cycle Fab antibodies into the systemic circulation via the FcRn pathway. 19 A high incidence of systemic immunogenicity was observed in all monkey studies, except the PK study, and in the 13-week mouse study. The appearance of treatment-emergent ADA is consistent with the likelihood of breaking immune tolerance, which increases with increased exposure. 20 However, although most biotherapeutics have the potential to induce ADA in preclinical studies 21 because they are complex proteins designed to minimize immunogenicity in humans only, the frequency of ADA for biotherapeutics delivered systemically (ie, via a non-inhalation route) is generally much lower. In one retrospective evaluation involving 27 monoclonal antibodies, the frequency of ADA-positive NHPs was relatively low, with 41% of cases having an ADA-positive frequency of <6%. 21 The high level of ADA in this study is therefore in contrast to parenteral routes of delivery, but not unexpected, as the respiratory mucosa is generally believed to be a more immunogenic site 22,23 and consistent with that observed with other inhaled biotherapeutics (Hall et al, this issue). In addition, pharmacologic inhibition of the target, TGFβ, may also have contributed to increased immunogenicity.
Antidrug antibodies were not detected in the NHP PK study, which was terminated on day 10, consistent with the kinetics of an immune response, but were present in 4- and 13-week studies. An immune response typically takes approximately 5 to 8 days for expansion of antigen-specific B and T cells in draining lymph nodes before homing to the site of antigen exposure followed by antibody synthesis. 24,25 Not unexpectedly, generation of ADA was accompanied by a tendency toward loss of systemic exposure in NHP studies (Figure 1C), which was likely due to the formation of clearing antibodies. Humanized biotherapeutics tend to contain foreign epitopes for the test species and as a result often stimulate a polyclonal immune response, which undergoes affinity maturation over time. 26 Clearing and neutralizing antibodies have the capacity to cause loss of systemic exposure and pharmacodynamic effect in preclinical studies, which may manifest in human patients as loss of efficacy. One of the principal mechanisms of ADA-mediated clearance is due to the formation and binding of antigen–antibody complexes to FcRn with subsequent removal from the circulation and lysosomal degradation in the reticuloendothelial system. 27 However, the sustained exposure data in the 13-week mouse study combined with the presence of high ADA may be due to increased retention of UCBFab immune complexes mediated by non-neutralizing antibodies (that do not interfere with the free PK assay measurement) that promote recirculation via FcRn. Both TGFβ and PAI-1 determined at the end of the study indicated active drug after 13-week daily treatment for both doses despite the presence of high ADA.
Gross and Microscopic Pathology
Inhalation of UCBFab resulted in treatment-related changes in the lungs and draining lymph nodes in both the NHP and mouse. No systemic changes were identified consistent with high local but limited systemic exposure. Treatment-related changes were multifocal and were not associated with any clinical signs or with any changes in clinical pathology. They were less pronounced in mice and only present in the 13-week study, whereas they were already present in the NHP PK study that was terminated on day 10 of the study (Appendix 1) and did not progress with longer treatment (between 2- and 13-week daily dosing).
Nonhuman Primate
In the NHP PK study, gross effects (“mottling”) were confined to the lungs, which correlated occasionally with congestion. The only other gross finding of note was enlarged tracheobronchial drainage lymph nodes that correlated with the microscopic finding of increased cellularity consistent with lymphoid hyperplasia secondary to an immune response.
Histological changes consisted of PV/PB mononuclear cell infiltrates confined mainly to the airways and extending into the respiratory bronchioles in some animals and alveolar inflammatory cell infiltrates or, occasionally, inflammation. Immunohistochemical characterization in the 13-week study demonstrated that the PV/PB MIC infiltrates located within the airways consisted of a variable mixture of CD3-positive T cells (>50%), CD20-positive B cells (<25%), and CD68-positive macrophages (up to 50% in some foci), reflecting the normal populations of cells required to initiate and maintain an immune response. It is not clear whether the cell proportions merely reflect recruitment from the circulation during a normal immune response to the test article since the biological diversity of bronchial-associated lymphoid tissue (BALT) is very variable both within different regions of the lung and between animals 28 or had been additionally modified by pharmacologic inhibition of TGFβ as this molecule inhibits both B cell, T cell, and macrophage function. 29 The pattern of change was similar, regardless of treatment duration or dose level. The PV/PB mononuclear cell infiltrates were composed predominantly of lymphocytes located in a PV and PB location in airways but also extending into the respiratory bronchioles in some animals, accompanied by increased cellularity (lymphoid hyperplasia) of the drainage tracheobronchial lymph nodes in the 2-, 4-, and 13-week studies, consistent with an immune response to the inhaled Fab (humanized protein).
In the lung, the BALT is arranged in loose aggregates within the airway mucosa. Its function is to sample and respond to antigen, which it does by undergoing hyperplasia appearing as PV lymphoid cuffing around small- and medium-sized pulmonary veins. 30,31 This occurs typically over a 5- to 8-day period following antigen exposure in mice as a consequence of lymphoid hyperplasia and expansion within the draining lymph nodes followed by BALT hyperplasia and expansion within the lung at the site where the antigen was first encountered. 24,25 Given the similarity in the histological pattern and the kinetics of the PV/PB infiltrate to that seen in the reported literature, and the high level of immunogenicity with seroconversion encountered with the inhaled form of UCBFab, it is highly likely that this is an acquired immune response to the test article. Administration of fresolimumab, a pan-TGFβ IgG4 monoclonal antibody inhibitor, to mice and NHPs via the intravenous route at weekly doses of at least 10 mg/kg for 13 weeks produced a different spectrum of changes—chiefly epithelial hyperplasia of the bladder, nasal turbinate, and gingiva in primates and gingiva, tongue, and esophagus in mice—consistent with high sustained target engagement. It notably did not induce PV/PB mononuclear cell infiltrates in the lungs, 10 indicating that this particular change is unlikely to be pharmacologically mediated. This mechanism is also consistent with data obtained from other marketed and nonmarketed biotherapeutics where inhalation of proteins with immunogenic potential such as Pulmozyme (dornase α; human recombinant DNase) resulted in PV/PB changes similar to those that were reported, 32 whereas inhalation of nonimmunogenic insulins did not result in adverse pathology or ADA production. 33 –36 Finally, comparison to other case studies with inhaled biologics (Hall et al, this issue) indicates that PV/PB MIC infiltrates in the lungs appears to be a class effect of inhaled foreign biotherapeutics where it is frequently associated with high levels of ADA and lymphoid hyperplasia in the lungs and drainage lymph nodes (consistent with an immune response to the test article).
Within the alveolar bed, changes consisted variably of alveolar inflammatory cell infiltrates or alveolar inflammation. Alveolar inflammatory cell infiltrates were located within the alveoli and septa, but with no signs of inflammation or damage. This was in contrast to inflammatory changes that appeared as a progression of the inflammatory cell infiltrate and were associated with other changes indicative of damage such as erythrophagocytosis and type 2 pneumocyte hyperplasia.
Further investigation of the inflammatory cell infiltrates revealed that in the alveoli, most cells were CD68- and CD206-positive activated macrophages followed by reduced numbers of CD3-positive T cells and very few CD20-positive B cells. CD206 is a pattern recognition receptor (mannose receptor) expressed on the surface of macrophages and dendritic cells mediating the clearance of microbes and endogenous glycoproteins through recognition of high mannose N-linked glycans during endocytosis and phagocytosis (as well as other functions such as modulation of activation and antigen presentation). 37,38 Expression of CD206 by alveolar macrophages after treatment with UCBFab is consistent with a role in clearance of test article and correlates with the presence of test article within alveolar macrophages identified by IHC. Alveolar lymphocytes were also present in fewer number and consisted predominantly of CD8-positive T cells (with fewer CD4-positive cells) of which up to half of cells expressed the activation marker CD25 (IL2Ra). Activation of T cells within the alveolar compartment is consistent with the pro-inflammatory pharmacology of TGFβ inhibition, which normally maintains T cells in a quiescent and inactivated state. 39
Since IHC analysis revealed no evidence of ICD due to an absence of co-localized granular deposits of test article, host antibody (IgG, IgM, IgA), and/or complement, 16 ICD was unlikely to have accounted for the changes seen in the alveoli. In addition, it was not thought to be driven by the alveolar protein load since other Fab fragments from the same manufacturing platform given at comparable daily doses did not induce alveolar inflammation (personal observations). Moreover, experiments with weekly doses of UCBFab did not induce alveolar inflammatory changes, suggesting that the pharmacologic action of UCBFab is a contributing factor.
Other contributing factors might include immunogenicity since inhalation of “Pulmozyme”/rhDNase induced immunogenicity and similar, if not identical airway (PV/PB MIC) and alveolar inflammatory changes. 32 Similarly, inhalation of other immunogenic substances which lack pharmacologic action such as ovalbumin, pigeon serum, or grain dust has the capacity to induce alveolar inflammation, 30 sensitization, and even severe interstitial pneumonitis. 40,41
Probably, the main factor contributing to the alveolar changes was the pharmacologic action of TGFβ. Transforming growth factor β is a potent immunomodulator regulating cytokine production by immune cells, inhibiting T- and B-cell proliferation and function, downregulating NK activity, and regulating macrophage function. 29 In the lung, epithelial secretion of TGFβ maintains macrophages in an alternatively activated (M2c) resting state 42 and knockout of TGFβ1 in mice results in fatal histiocytic (macrophage)/lymphocytic inflammation in the lungs and heart. 29 Although the exact pathogenesis of the alveolar changes is not fully known, it is likely that TGFβ inhibition by UCBFab induced a pro-inflammatory environment in the alveoli that resulted in an inflammatory response. This hypothesis is consistent with the results of several other immunomodulators that also induced similar alveolar inflammation (Hall et al, this issue), indicating a common theme of exaggerated pharmacology, and consistent with data from this study showing suppressed phosphorylated SMAD2/3 in the BALF pellet. Reduced SMAD2/3 indicated target engagement and reduced signal transduction consistent with pharmacological inhibition of the target, TGFβ. However, whether the inflammatory response was a response to an autoantigen or driven in some part by an exaggerated immunological response to the foreign test article is unknown.
Mouse
In the mouse, exposure accumulation was noted at both doses and dose-related reduction of PAI-1 in BALF demonstrated efficacy for UCBFab. After 13 weeks of daily dosing, statistically significant slight increases in lung organ weight were noted in female animals, which correlated with the pathology changes. These changes were most likely significant in the mouse due to increased group size (n = 12 for the mouse vs n = 2-4 for the NHP). Histopathological examination of the lungs revealed similar but slightly less severe changes to those seen in the NHP. Since the mouse and the NHP 13-week studies were conducted with the same formulation of UCBFab and at the same dose, it is likely that milder changes were due to a lower deposited dose at the bronchioloalveolar region, 43,44 although differences in species response cannot be ruled out. Lymphocytic cell infiltrates into the alveoli and airways correlated (although with weak confidence due to loss of samples from clotting) with the hematology data where small increases in lymphocytes were identified during treatment. In addition, BALF cytology data from the NHP 4-week study where significant increases in leukocytes were seen also correlated with the alveolar inflammatory cell infiltrates. Finally, in the draining tracheobronchial lymph nodes, increased lymphoid cellularity was noted consistent with an immune response to the inhaled Fab protein.
Bronchoalveolar Lavage Fluid
Analysis of BALF in the PK study indicated that the data may have been influenced by procedural effects due to hemorrhage. Iatrogenic bleeding is a recognized, but relatively uncommon (0.26%-5% incidence) complication during flexible bronchoscopy in human clinical medicine. It can be related to a number of risk factors including procedural 45 –47 and may be more common in animal research that involves smaller species. 48 Other factors such as small group size, poor BALF mixing, and recovery may additionally contribute to poor BALF sample collection and quality in preclinical species. Nevertheless, BALF analysis in the NHP PK did not reveal any changes in cell counts or immunophenotype despite minimal-to-mild alveolar inflammation/inflammatory cell infiltrates. This result is in contrast to human clinical data where there is a high degree of correlation between bronchoalveolar lavage cytology and transbronchial biopsy noted clinically (sensitivity 93.44%; false negative 6.55%), 49 possibly as a result of more advanced disease in patients. Since this is a relatively common procedure in human clinical practice, this discrepancy might also reflect the increased technical expertise required to sample relatively small NHPs and the need to perform the technique routinely to maintain a high skill level.
Analysis of BALF during the NHP 4-week study, using a terminal technique in isolated lungs, correlated better with histopathology, possibly due to the procedure being performed at necropsy where the lung lobe could be removed, perfused, and isolated for aspiration. Control samples yielded low total cell number (320-790/μL, which represents total leukocytes and epithelial cells). Treated animals showed mild-to-moderate increases in total nucleated cell counts as well as neutrophils, lymphocytes, and eosinophils, which correlated with the histopathology changes. Since epithelial cells represent approximately 10% of total nucleated cell counts in normal animals, 48 macrophages were numerically the major cell type in lavage fluid in these animals, and most likely accounted for the significant increase in total cell count in treated animals since this was the predominant cell type noted at histopathology.
Conclusions
UCBFab was generally well tolerated in both the NHP and mouse. Target occupancy and signal transduction were confirmed by reduced levels of phosphorylated SMAD in BALF of NHP and a dose-dependent increase in TGFβ associated with a dose-dependent reduction in PAI-1 (proximal marker of target engagement). Inhalation of UCBFab resulted in high local exposure but low systemic exposure consistent with slow absorption into the systemic circulation due in part to the absence of active transport from the lung interstitium to vessels via the FcRn receptor in the absence of an Fc domain. Inhalation exposure resulted in high immunogenicity (both frequency and titer) and variable (and low) systemic exposure, resulting from the presence of clearing antibodies or, in some individuals, of non-neutralizing drug-sustaining antibodies. This, however, was not associated with ICD in the lungs or in peripheral tissues or adverse clinical signs. The PK study in NHPs demonstrated good correlation between serum and BALF concentrations of UCBFab before the development of immunogenicity that may interfere with systemic exposure (Figure 1A). Long-term dosing did not demonstrate accumulation of drug in the lung or in BALF. This is important since the initial serum concentration may help in the understanding local lung concentrations.
Inhalation of UCBFab resulted in 3 patterns of histological changes that were similar in both the NHP and mouse and were characterized by a PV/PB MIC infiltrate in the airways, an inflammatory cell infiltrate/inflammation in the alveoli, and increased lymphoid cellularity of the draining lymph nodes. In both species, findings showed no morphologic progression despite high local exposure and the presence of treatment-related changes for the duration of treatment.
The PV/PB MIC infiltrate was characterized by a predominantly lymphoid infiltrate (consisting of mainly T and B cells with some macrophages) that was thought to arise secondary to a local immune response in the lung. The alveolar inflammatory cell infiltrate and inflammation were both considered to be secondary to the sustained pharmacologic action of UCBFab upon daily dosing, although it is possible that some component of this change might have also be due to immunogenicity. Minimally increased lymphoid cellularity was noted in the draining tracheobronchial lymph nodes. All these changes showed partial reversal after a 4- or 8-week recovery period and would most likely have fully reversed given sufficient time. Alveolar changes were slightly less severe in mice compared to NHPs (inflammatory cell infiltrate vs inflammatory cell infiltrate with inflammation) most likely due to decreased exposure in the mouse, although it is possible that this could also be due in part to decreased sensitivity to the pharmacologic action of UCBFab. In the 4-week NHP study, BALF cytology correlated closely with the histopathology results.
Footnotes
Appendix A
Acknowledgments
The authors thank the following people for their help in the preparation and generation of data for this manuscript: Andrew Pilling who provided expert peer review, Florence Poitout, Nigel Young, Kieran Jones, and Jose Joby.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Peter Hall and Annick Cauvin are employed by UCB which is a pharmaceutical company that is involved in the development of medicines delivered by the respiratory route. These authors may also hold shares in UCB.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
