Abstract
The delivery of biotherapeutic molecules (antibodies, proteins, peptides) and nucleic acids via the respiratory route has presented challenges for regulatory approval, due in part to a lack of understanding of the expected pathology, mechanisms of toxicity, and immunogenicity induced by the inhalation route. Although the first inhaled biotherapeutic was approved some time ago (Dornase Alfa, Pulmozyme; Genetech, 1993), no other inhaled biotherapeutics have been marketed for the treatment of human disease other than the inhaled insulins (Exubera; Pfizer, 2006 and Afrezza; Mannkind Corporation, 2014). As a result, scientific knowledge within the toxicologic pathology community is fragmented with precious little publicly available data. Therefore, one of the aims of this special edition was to generate a collection of manuscripts that pathologists and toxicologists could refer in order to understand the pathology, mechanisms of toxicity, immunogenicity, and challenges associated with the development of inhaled biotherapeutics.
A number of recent scientific symposia on inhaled biotherapeutics (British Society of Toxicologic Pathology [BSTP], Association of Inhalation Toxicologists [AIT] Joint Meeting in 2016, Envigo Annual Biologics Meeting 2017, CRL Biotech Symposium 2017, and the BSTP AGM 2018) highlighted the trials and tribulations experienced by several sponsors during the development of inhaled biopharmaceuticals. The lack of citable data, degree of regulatory challenge regarding the toxicity findings, and the fragmented nature of the pathology and toxicology experience within the community precipitated the call for a “white paper” to generate a pooled resource of knowledge to aid the development of inhaled biotherapeutics. As a result, a survey was performed by the BSTP, Society of Toxicologic Pathology (STP), European STP, and AIT to identify toxicologists and pathologists from all sectors of the industry (pharmaceutical, biotech, and contract research organization facilities [CROs]) who were willing to collaborate, share data, and ultimately publish manuscripts that would significantly expand knowledge in this area of toxicologic pathology. The resulting working group comprised over 35 individuals representing many of the pharmaceutical and CRO companies active in this area of research and culminated in 12 case studies that became the nucleus for the first 2 manuscripts in this special edition.
The first manuscript (Hall et al [this issue]) is a pathology-centric review of all 12 case studies that examines the pathologic changes induced by inhaled biotherapeutics. The authors describe common patterns of pathology and propose mechanistic rationales. For example, the changes in the airways appear to be morphologically and temporally similar to experimental models of lung immunization, and in support of this, immunogenicity and other data are presented (Figure 1). Similarly, changes in the alveoli are also discussed and categorized into 2 primary morphologically distinct patterns of change. The authors examine the available data and suggest that these changes may be due to either physiologic responses related to clearance of test article or pharmacologically mediated immunomodulation.
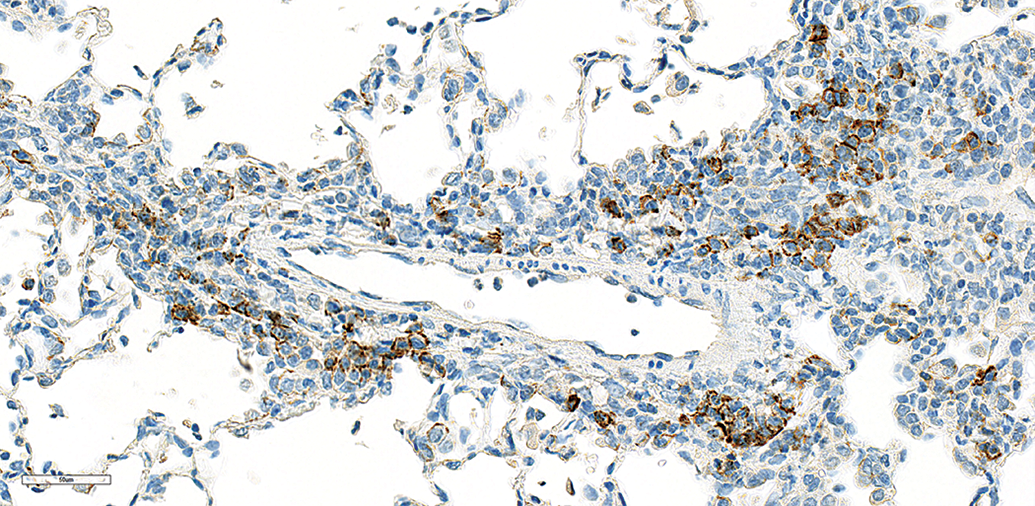
High power view of the lung from an NHP treated with an immunomodulatory inhaled biologic fragment antigen-binding (Fab) antibody that resulted in prominent perivascular/peribronchiolar (PV/PB) mononuclear inflammatory cell infiltrates (together with an alveolar mixed inflammatory cell infiltration). Immunohistochemical staining with the CD138 marker demonstrated strong granular membrane staining in a proportion of mononuclear cells indicating the presence of plasma cells.
The second manuscript (Wolfreys et al [this issue]) is a toxicology-centric review focusing on the technical challenges often encountered in GLP toxicology studies when delivering biotherapeutics via the inhalation route. The authors describe how these challenges influence study design, efficacy and exposure, and the limitations of PK/PD (pharmacokinetic/pharmacodynamic) end points given the uncertainties of measuring drug in lung tissue or bronchoalveolar lavage fluid (BALF), especially with high levels of antidrug antibodies typically encountered in these types of studies.
Accompanying the main pathology review paper by Hall et al (this issue) are 3 case studies that describe in more detail the pathology-induced 2 inhaled single-domain antibodies (Haworth et al, this issue), an anti-TGFb1 humanized Fab (antigen-binding fragment) antibody (Hall et al 1 ) and a recombinant human protein (Flandre et al 2 ). A fourth case study describes the consequences of long-term inhalation of a novel insulin “Technosphere” inhalation powder (Afrezza). 3 The authors go on to present immunohistochemistry data suggesting a lack of carcinogenic potential. Finally, a fifth paper 4 provides a perspective on respiratory tract lesions induced by small molecule chemical entities (NCEs). As one of the largest comparison papers to date, using pooled data from 81 inhalation studies and 133 non-inhalation studies, evaluated at 3 geographically distinct CROs, the authors present background pathology incidences of common changes affecting the upper and lower respiratory tract of rodents and non-human primates (NHPs) as well as changes induced by inhaled NCEs. The data provide a useful and contemporary comparator to the inhaled biologics data provided in the pathology review paper by Hall et al (this issue).
Understanding the changes induced by inhaled biotherapeutics is very much dependent upon an accurate measurement of inhaled dose, as the severity and location of the pathology in the respiratory tract are determined by the deposited dose at the region in question. In a perspective on lung dose with inhaled biologics, Wolff 5 discusses the challenges of determining total delivered dose, the parameters that affect drug distribution, and the need to measure alveolar delivered dose as possibly the most important determinant of toxicity. The pathology review paper (Hall et al [this issue]) identified that alveolar toxicity may be associated with an inflammatory cell infiltration, and while histopathology provides the current gold standard by which to assess these changes, techniques such as analysis of bronchoalveolar lavage (BAL) fluid provide additional data that allow the quantitative assessment of inflammatory and epithelial cell content in a relatively noninvasive manner. To address this point, Poitout-Belissent et al 6 review the methods for BALF collection, the parameters that affect variability, and the factors to consider in the use of BAL for the assessment of safety of inhaled drugs, pollutants, and chemicals.
Hall et al (this issue) discuss 2 instances of bronchopneumonia due to opportunistic infections in their review of inhaled biotherapeutics. When opportunistic infections are diagnosed, it is incumbent upon the study pathologist to determine whether the lung changes were due to the inhaled test article or incidental in nature. While many procedural factors predispose to lung infection such as aspiration (during gavage studies) and BAL collection, test article pharmacology should always be considered, especially if the drug is an immunomodulator, as it may reduce immunological function through immunosuppression. To help readers in this regard, Flandre et al 7 review the most common opportunistic infections observed in the respiratory tract of NHPs and discuss how they might impact the interpretation of pathology findings.
Determining the adversity of a test article–related effect or observation is a subjective and often difficult endeavor. This is especially true in the case of histologic lesions that occur in studies of inhaled biologics, and in this edition, 2 papers provide important perspectives and guidance. In their pathology review, Hall et al discuss the factors contributing to lung adversity, why there might be areas of uncertainty, and what factors might contribute to a reasonable difference of opinion. Engelhardt et al 8 go on to discuss the assignment of adverse effect levels both in the general practice of toxicologic pathology but also with specific reference to changes in inhalation toxicology studies. This commentary presents 2 points of view which the authors term either focused (determining adversity at the level of a specific toxicity study) or broader (adversity considered in the context of the entire program with a focus on the clinical investigator) approach. While these views differ from some of the guidance provided in STP papers on adversity, they provide interesting concepts for pathologists and toxicologists to consider.
Finally, Macri et al 9 shine a spotlight on a well-known observation that is frequently recorded with inhaled NCEs and, more rarely, in inhaled biotherapeutic toxicology studies—namely laryngeal squamous metaplasia. These lesions develop in response to chronic irritation often induced by the more insoluble particle-like properties of NCEs and occasionally by biotherapeutics. 3 However, Macri et al 9 demonstrate a new finding that appears upon treatment cessation, where the laryngeal epithelium passes through a hitherto unrecorded transitional state, before full recovery.
Concluding Remarks
The purpose of this special edition was to generate a collection of citable data that could contribute to the relatively limited pathology and toxicology knowledge in the inhaled biotherapeutic space. We hope that these manuscripts encourage further publications to broaden the available literature, act as a guide to help pathologists interpret their data, and ultimately ease the regulatory path.
Footnotes
Author Contribution
Hall, Vahle, and Colman contributed equally.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
