Abstract
This article summarizes a continuing education presentation on immunogenicity that was part of a continuing education course entitled, “Clinical Pathology of Biotherapeutics.” Immunogenicity of a biotherapeutic can have diverse impacts including altered systemic exposure and pharmacologic responses and, in a fraction of the cases, safety concerns including cross-reactive neutralization of endogenous proteins or sequela related to immune complex disease (ICD). In most cases, immune complexes are readily cleared from circulation; however, based on physiochemical properties, insoluble complexes form, activate complement, and deposit in tissues. Using published information and personal experience, a set of repeat-dose monkey toxicity studies with manifestations suggestive of ICD was reviewed to summarize the spectrum of clinical and pathology findings. The most common live-phase observation linked to ICD was an acute postdosing reaction following multiple dose administrations characterized by generalized collapse and attributed to acute complement activation. Less common live-phase observations were related to syndromes such as a consumptive coagulopathy or a protein losing nephropathy. The most common histologic change attributed to ICD was multi-organ vascular/perivascular inflammation followed by glomerulonephritis. The presentation concluded with a description of the challenges in assessing the relevance of immunogenicity-related reaction in monkey to human clinical use.
Introduction
This article summarizes one of the four presentations at a Continuing Education Session held in association with the Society of Toxicologic Pathology’s Thirty-seventh Annual Symposium. The Continuing Education Session focused on the clinical pathology evaluation of biotherapeutics in the context of nonclinical safety studies. The primary objective of this article was to review the potential impacts of immunogenicity on the nonclinical safety assessment of biotherapeutics. Following a brief introduction to the concepts of immunogenicity including mechanisms of formation of antidrug antibodies (ADAs), the remainder of the presentation focused on the various manifestation of immunogenicity in nonclinical safety studies. Particular focus was placed on the development of immune complex disease (ICD) in nonhuman primates administered monoclonal antibody-based constructs. The review concluded by addressing some of the challenges in understanding the relevance of the immunogenicity findings in animals to the safety of these agents in human clinical trials. This review did not provide a detailed analysis of mechanisms of immunogenicity or immune complex formation and did not review acute (type 1) hypersensitivity reactions. Several excellent reviews on nonclinical and clinical immunogenicity were highlighted for the audience (Leach et al. 2014; Ponce et al. 2009; Rojko et al. 2014; Mease, Kimzey, and Lansita 2017; De Groot and Scott 2007; Krishna and Nadler 2016) and served as an important basis for the presentation.
Immunogenicity: An Introduction
For the purposes of this review, immunogenicity was defined as the ability of a therapeutic agent to induce an immune response. Immunogenicity is most frequently a concern for protein-based therapeutics (hereafter referred to as biotherapeutics) and can occur in both animal and human settings (Krishna and Nadler 2016; Ponce et al. 2009). The antibody that is generated in response to a biotherapeutic has been variably referred to as an ADA, antitherapeutic antibody, primate antihuman antibody, or a human antihuman antibody, and the general response has been referred to as an anti-antibody response. The potential for immunogenicity to limit the use of biotherapeutics, particularly monoclonal antibodies, was an early concern for the field (Kuus-Reichel et al. 1994), and while immunogenicity remains an important topic, a wide array of monoclonal antibody and related protein constructs has proven to be highly effective therapeutics for a variety of disease states. In the clinical setting, immunogenicity can cause a variety of effects including loss of systemic exposure, loss of the desired therapeutic response, and less frequently safety concerns such as infusion reactions, cross-reactive neutralization of endogenous proteins that mediate critical biologic functions, and ICD. When immunogenicity issues arise late in the clinical development process, they are extremely costly to drug developers. As such, methods to predict and mitigate against clinical immunogenicity are active areas of investigation (Yin et al. 2015).
It is important to recognize that many factors can affect immunogenicity. Product-specific factors that affect immunogenic potential often focus on the protein sequence of the construct; however, in many cases, the formulation, aggregates, impurities, and therapeutic target play important roles. Treatment-specific factors such as the dose, route, and frequency of administration also influence immunogenic potential. Patient-specific factors such as the disease state, concurrent illness or medications, and genetic background are also important and underscore how challenging it can be to predict immunogenic potential. While the production of ADAs can occur via both T-cell-dependent and T-cell-independent mechanisms (De Groot and Scott 2007), much of the focus regarding predicting immunogenicity is centered on identifying T-cell epitopes that drive T-cell help and antibody production.
Potential Impact of Immunogenicity on Animal Toxicity Studies
Similar to the clinical setting, immunogenicity can have a variety of impacts in animal models including the toxicity studies used to characterize the nonclinical safety profile of the biotherapeutic (Figure 1). Because the biotherapeutic is a human or humanized protein, the frequency and severity of the immunogenic response can be greater in the animal model than in the human clinical setting (Leach et al. 2014; Ponce et al. 2009). Despite the increased “foreignness” for the human protein in animals, in many cases, there may be no appreciable immunogenicity in the animal model. While robust data are not available on the frequency of immunogenicity in nonclinical toxicity studies, pathologists frequently encounter studies of biotherapeutics where there is essentially no impact of immunogenicity on the overall study outcome. In these cases, systemic exposures are maintained at adequate levels to assess toxicity, there is no loss of intended pharmacologic effects, and no sequela related to hypersensitivity or ICD.

The types of impacts that an immunogenic response to a biotherapeutic can have on a toxicity study. In some cases, very little impact is noted as there is not an appreciable impact on systemic exposure or pharmacologic or toxicologic effect. At the other end of the spectrum are those cases where immunogenicity can have substantial impact including morbidity or mortality, including but not limited to hypersensitivity reactions such as immune complex disease.
Antibody-mediated changes in the pharmacokinetic profile are one of the more common sequela of immunogenicity in toxicity studies. Antibodies that bind to the biotherapeutic and increase their clearance from systemic circulation are referred to as “clearing” antibodies and can markedly reduce systemic exposure and interfere with the ability to assess toxicity in either a single animal or entire dose groups. Various study designs can be considered in an effort to “dose through” immunogenicity including increasing administered dose or altering dose frequency or route of administrations (Ponce et al. 2009). While less common, there are clear examples where antibodies to the therapeutic protein have decreased systemic clearance resulting in increased systemic exposure (“sustaining antibodies”). In these cases, prolonged circulation time may increase pharmacologic activity and complicate assessments of toxicity. In addition to altering systemic exposure, the antibody to the biotherapeutic can bind at or near the target-binding epitope and inhibit the biotherapeutics ability to bind to the desired target. These antibodies are referred to as “neutralizing antibodies.” Both neutralizing and clearing antibodies, by reducing the pharmacologic effects of the biotherapeutic, can compromise the ability to assess the toxicity profile of the therapeutic. To aid in the overall interpretation of the toxicity study, it is important for the study pathologist to work with colleagues in toxicokinetics and immunology to assure an appropriate assay strategy is in place. These assays can include the measurement of the active circulating biotherapeutic, detection of circulating ADA levels, and measurement of pharmacodynamic end points in either the periphery or tissues.
The biologic significance of immunogenicity is heightened when the ADA neutralizes not only the administered biotherapeutic but also the biologic function of a critical endogenous protein (cross-reactive antibodies). For example, thrombocytopenia and pancytopenia have been reported in patients following the administration of recombinant human thrombopoietin (Basser et al. 2002; Li et al. 2001). Clearly, in both animal and human settings, the development of cross-reactive antibodies to endogenous proteins can have life-threatening consequences. Finally, as discussed in more detail below, the formation of immune complexes and sequela can have major impact on the interpretation of toxicity studies.
ICD—Overview of Mechanisms
A thorough review of the mechanisms of immune complex formation, clearance, measurement, and pathophysiology was outside the scope of the presentation. The audience was referred to reviews of the topic by Rojko et al. (2014), an investigation of anti-infliximab immune complexes by Rojas et al. (2005), and methods to measure circulating immune complexes by Pierog et al. (2015). Key concepts discussed include the fact that in health, levels of circulating immune complexes are relatively low due to two primary clearance mechanisms. One mechanism consists of clearance via the FcγRIIa receptor on platelets, monocytes, and macrophages and is present across rodent and primates. The other mechanism, which is specific to primates, involves shuttling of immune complexes via the complement receptor 1on red blood cells. A variety of factors influence the physiochemical properties of immune complexes and dramatically effect clearance and deposition of immune complexes. Lattice size is one of the key factors, and the large lattices can form when there is antigen: Antibody equivalence is often insoluble and can lead to tissue deposition. Antigen and antibody ratios are not the sole determinant, as the concentration, valence, and charge of both antigen and antibody can influence immune complex formation and clearance. While these are useful concepts to understand, multiple factors can influence immune complex formation, clearance, and deposition, and it is not possible to predict the potential for a given biotherapeutic at a given dose and dose frequency to induce ICD in animals.
Immune complex formation leading to systemic and tissue effects is generally considered a type III hypersensitivity reaction in the classical Gel-Coombs classification scheme (Leach et al. 2014); however, the pathophysiology is much more complex than simple intravascular deposition of insoluble immune complexes. As summarized by Krishna and Nadler (2016), three key interrelated mechanisms contribute the reaction: (1) deposition of immune complexes in vasculature and sites of hyperfiltration leading to inflammation, (2) activation of complement by the immune complex, and (3) cross-linking of FcγR and complement receptor on the surface of a variety of immune cells leading the release of inflammatory mediators and chemotactive substances. Complement activation plays a key role in both the systemic and the tissue effects noted in ICD, and key mediators are the generation of the anaphylatoxins C3a, C4a, and C5a (Krishna and Nadler 2016).
Findings Attributed to ICD in Nonhuman Primate Toxicity Studies
To summarize the range of live-phase, laboratory, and morphologic findings that have been associated with ICD following the administration of biotherapeutics to nonhuman primates in toxicity studies, a group of 23 repeat-dose nonhuman primate studies was reviewed for this presentation. The level of detail available for each study varied greatly as the information was derived from publications, workshop presentations, personal communications, and personal experience. Key publications that either review a series of cases or present detailed results from a single-case experience include (Rojko et al. 2014), (Heyen et al. 2014), Husar et al. ( 2017), and Kronenberg et al. (2017). From the information available across the studies reviewed, key study design elements, live phase-findings, laboratory findings, and anatomic pathology findings were summarized.
Of the 23 studies examined, 22 used different biotherapeutics (two studies were reported for one of the biotherapeutics), and as expected, these were primarily monoclonal antibody-based biotherapeutics. In many cases, only general descriptions of the biotherapeutic were disclosed, so it was not possible to identify clear trends; however, ICD was reported in studies of both monospecific and bispecific antibodies and occurred across various isotypes of antibody (IgG1, IgG2, and IgG4). There were insufficient data to determine whether a certain type of antibody construct was more prone to developing ICD. In terms of methods of dose administration, manifestations of ICD were reported across both intravenous and subcutaneous administrations and in studies using weekly, twice-weekly, and once-monthly dose administrations. Although the administered dose on a milligram per kilogram basis was not reported for many studies, most studies reported the dose level (low, middle, and high) at which ICD was reported. Manifestations of ICD were observed across all dose levels and in various dose-level combinations. While it was difficult to assess the true dose response in all the studies due to limited individual animal reporting, ICD did not generally present in a dose-responsive manner. The incidence of ICD within a given study also varied widely, and in 36% of the studies, only a single animal was affected, whereas in 64% of the studies, multiple animals were affected.
In 13 of the 23 studies (57%), findings attributable to ICD were reported during the live phase. While the clinical signs and laboratory findings reported were quite varied, for the purposes of this Continuing Education Session, live-phase findings were grouped into broad syndromes. The most common presentation or syndrome was designated an acute postdose reaction. These reactions occurred shortly after dose administration and most frequently occurred following the fourth or fifth dose administration. Clinical signs reported in nonhuman primates included hypoactivity, retching, emesis, excessive salivation, altered respiration, pallor, hypothermia, and pruritus. As discussed in Rojko et al. (2014), these may be a form of a generalized type III hypersensitivity reaction associated with acute complement activation. The clinical course for these animals varied widely with some showing spontaneous recovery, other requiring veterinary intervention with supportive case, and in other cases progressing to morbidity requiring early euthanasia. While the type and timing of laboratory data obtained in these animals was variable, the more common observations included transient decreases in neutrophil and platelet counts, increased fibrinogen and triglyceride concentrations, and decreases in albumin concentration. Complement data were reported in five of the 23 studies, and while the specific complement components analyzed varied, increases in C3a, C4a, C4d, and SC5b-9 were reported. Cytokine determinations were only reported in two of the 23 studies, and increased MCP1, Il-10, and IL6 were reported in one of these studies.
Although likely part of a continuum associated with acute complement activation, there were two additional acute live-phase syndromes identified in this case series. A subgroup of animals developed a consumptive coagulopathy. While many of these animals had clinical signs similar to those of the postdose reaction described above, additional live-phase observations included failure to clot at the phlebotomy site and/or petechial hemorrhage. While the laboratory data collected varied according to the specific study, laboratory findings reported in these animals included reductions in platelet counts, prolonged coagulation times, decreased fibrinogen concentration, and, in a single case, d-dimers were analyzed and reported as increased in concentration. Another syndrome that is likely associated with these acute postdose reactions included a subset of animals that had acute collapse postdosing, and the only histologic change identified was acute thrombosis. The level of detail reported in these studies did not allow a determination whether these animals also had laboratory data that would have been consistent with a consumptive coagulopathy and disseminated intravascular coagulation. The live-phase observations described above occurred shortly postdose. This is in contrast to the other important live-phase syndrome, nephrotic syndrome, in which there was a gradual onset of clinical signs and laboratory findings. These animals were often first detected based on live-phase observations of dependent edema, decreased body weight, and decreased food consumption and/or laboratory data of decreased serum albumins and increased urinary proteins. In each of these cases, histologic examination revealed glomerular disease attributed to immune complex deposition.
Rojka et al. (2014) provided a thorough review of the histologic findings associated with ICD as well as the immunohistochemical localization of various immune component with tissues. In the current series of studies, the general pattern of histologic changes was consistent with that reported by Rojko et al. Two of the 23 studies described clinical signs or laboratory findings that were attributed to ICD and acute complement activation but that did not have histologic lesions indicative of ICD. In the remaining 21 studies, in which histologic changes were attributed to ICD, perivascular and/or vascular inflammation was the most common histologic change occurring in 49% of the studies. Within a given study, the individual animal incidence of this change varied, and within an animal, the number of tissues affected was also highly variable ranging from a single vessel affected to widespread multi-organ involvement. Consistent with the sites of predilection for immune complex deposition, common sites were small to medium vessels in the serosa or mesentery, tortuous vessels in the urogenital tract, sites of hyperfiltration (choroid plexus, glomerulus), and the aortic root and coronary artery of the heart. In some cases, immunohistochemistry for immune components was used to confirm a diagnosis of immune complex vasculitis. Glomerulonephritis was less common than vasculitis in this series of cases and was reported in 16% of those studies in which histologic lesions attributable to immune complex occurred. The morphology of immune complex glomerulonephritis has been well described previously, and in this case series, key histologic findings reported were glomerular hypercellularity with hypersegmentation, increased mesangial matrix, thickened peripheral capillary loops, and, in more severe cases, synechia. Additional diagnostic methods were used in several of the studies and included localization of immune components within the lesion and electron microscopy to identify electron-dense deposits and further characterize the glomerular change. Other changes reported but not described in detail included fibrin thrombi in vessels not associated with vascular/perivascular inflammation and an increase in the size and/or number of Kupffer cells in the liver. In more advanced cases with severe vasculitis, a variety of secondary changes were reported including edema, hemorrhage, and necrosis of adjacent tissue.
Diagnostic Approach to ICD
The author provided a brief overview of the diagnostic tools that can be used to investigate findings that may be attributable to ICD (Figure 2). For many of the laboratory alterations that may be of interest such as detection of acute phase responses or acute complement activation, sampling timing is critical. As such, during the study design phase, the study team can consider a sample banking strategy where the appropriate matrix (serum and/or plasma) is banked both prestudy (to establish individual animal baseline values) and at various intervals both pre- and postdose during the study. In those cases where there are unexpected events and prospective samples were not obtained, residual samples from toxicokinetic or routine clinical pathology samples may be available. An advantage of the toxicokinetic samples, if available and in the appropriate matrix, is that they are typically obtained at multiple intervals postdose. In addition to laboratory data, having timely and consistent recording of live-phase observations is important in those situations where one or more animals develop clinical signs postdose administration. The live-phase observer should strive to use consistent terminology across the study to allow study personnel to determine whether different animals are having similar presentations.
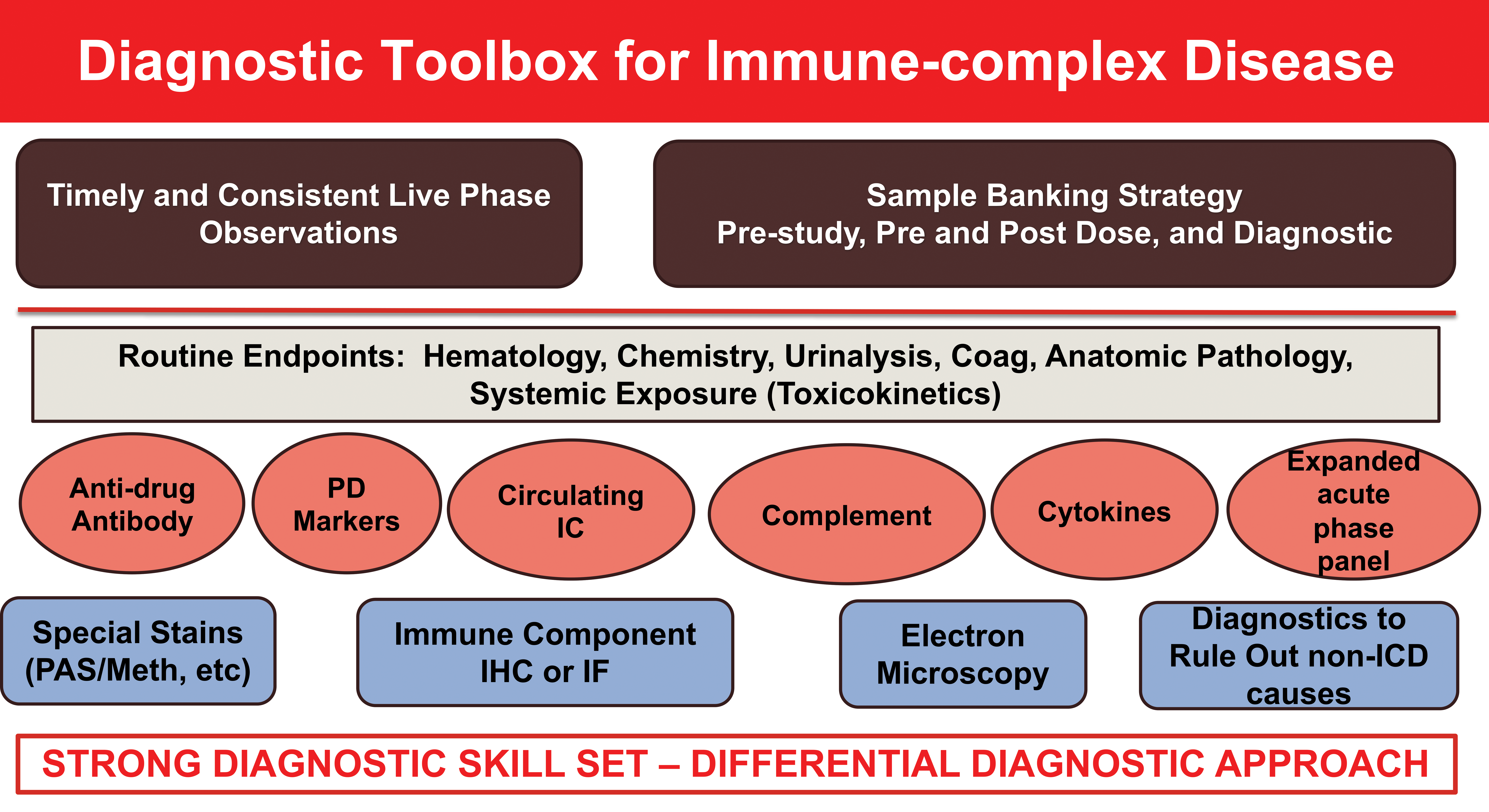
Various end points and approaches study teams can consider in developing a weight-of-evidence assessment regarding immune complex disease in animal toxicity studies. The author emphasized a case-by-case approach to determining which end points are most appropriate for a given animal or study.
The routine end points included in standard repeat-dose toxicity studies (live-phase observation, hematology, clinical chemistry, urinalysis, coagulation panels, anatomic pathology, and toxicokinetic data) provide a strong database and in some cases may be sufficient to provide a high index of suspicion that immunogenicity and ICD are present. Other assays that are often critical in the overall weight-of-evidence assessment include ADA analysis, assessment of pharmacodynamic markers to assess whether active drug is present, detection of circulating immune complex levels, complement and cytokine determinations, and an expanded panel of acute phase reactants.
With respect to tissue-based end points, immunohistochemical or immunofluorescent detection of immune components in the tissue of interest can be helpful and in some cases can provide a definitive diagnosis of ICD. Methods are available to detect not only various immunoglobulin and complement components from the monkey (e.g., monkey IgG, IgM, C3) but also to localize the therapeutic antibody within the tissue of interest. Other morphologic approaches that may be warranted include electron microscopy and special stains (e.g., Periodic Acid Schiff [PAS] on thin sections) to better characterize glomerular disease.
One of the most important aspects of the diagnostic or investigative approach is, for the study team and the study pathologists in particular, to bring a strong diagnostic skill set and differential diagnostic approach to the study. While it can be tempting to default to a diagnosis of ICD for any postdose reaction or vascular change that occurs in a monkey that has developed an immunogenic response, the study team needs to carefully rule out, as best as possible, that the changes are not either (a) directly attributable to the test article or (b) a spontaneous event unassociated with the test article. Another important challenge for the study team to deal with is the fact that in many cases, not all of the individual end points will correlate for a given animal. Because no single diagnostic test has perfect specificity and ICD manifests in a variety of ways, these cases often remain diagnostic and interpretive challenges. A relatively straightforward case might present with multiple episodes of acute postdose reactions, significant loss of systemic exposure, high titers of ADA, postdose complement activation, and histologic evidence of vasculitis and glomerulonephritis. In the studies summarized above, this type of “perfect concordance” was relatively rare. For example, at the high doses administered in some toxicity studies in an effort to overcome immunogenicity (“dosing through strategy” [Ponce et al. 2009]), despite considerable immunogenicity and accelerated clearance of the biotherapeutic, systemic exposure may not be dramatically impacted. In addition, assays for ADA may not be positive due to the design of the assay with respect to drug tolerance. Although not clearly reported in the case series examined, in the author’s experience, there is often not a direct correlation between ADA titer and the presence and/or severity of ICD. Some biotherapeutics can induce high ADA levels with marked reduction in systemic exposure and did not result in any evidence of ICD in the toxicity study.
Given these challenges, it can be difficult to determine the appropriate diagnostic approach for any given study. Laboratories and sponsors appear to vary widely in the intensity in which they investigate these findings. The author encouraged a case-by-case determination and suggested the study team consider a variety of factors in determining what level of evidence is warranted. Key questions for consideration are: What is the severity of the findings? Did the findings result in morbidity or mortality? What are the incidence and dose responsiveness of the findings across the study? What is the overall anatomic distribution of the findings (e.g., single vessel vs. widespread vasculitis)? What is the overall weight of evidence with the existing data? What additional data are more likely to be informative?
For example, in a study where a single animal presents with a minimal to slight vascular lesion and the systemic exposure was markedly reduced, it may be warranted to indicate in the study report that the finding was potentially due to ICD rather than representing a direct test-article effect and not to pursue further evaluation. In contrast, a study with a high incidence of postdose reactions and vascular disease that resulted in morbidity and lacked a consistent correlation to altered systemic exposure might warrant additional analysis such as assessing complement activation or attempting to localize immune components in the tissue.
Challenges in Assessing Human Relevance
The presentation concluded with a brief discussion on how to assess the clinical relevance of immunogenicity-related findings in animal toxicity studies. To frame the discussion, three divergent approaches were reviewed. On one end of the spectrum was an approach that considers animal immunogenicity-related findings as not relevant to humans, since an immune response to a human or humanized protein is expected to be greater in animals than in humans. In this approach, the animal findings may not be used in the determination of a study no-observed-adverse-effect level and summary clinical regulatory, and clinical documents might not discuss the animal findings in detail. A downside of this approach is that it may be overly definitive in using language such as “not relevant” and may not provide the appropriate degree of transparency and interpretation of this complex phenomenon. At the other end of the spectrum is an approach that suggests since humans can develop immune responses to human or humanized protein therapeutics and since ICD can occur in humans, the animal findings are a key safety finding and should be used to determine human dose levels and drive clinical safety monitoring strategies. A downside of this approach is that it does not incorporate sufficient perspective regarding the generally poor predictive value animals have with respect to immunogenicity in general, and it may create greater clinical and/or regulatory concern for the findings than is warranted.
The author suggested an intermediate approach that indicates that while animal studies are often not predictive of immunogenicity and sequelae related to immunogenicity in humans, the animal findings should be carefully considered as part of the overall safety assessment. For example, if there is an on-target mechanistic basis for enhanced immunogenicity in animals, the findings might be considered more predictive for humans. In addition, for novel constructs that may be considered more “foreign” to both animals and human and for which there is limited clinical experience, additional caution might be warranted. One important point to keep in mind with this approach is that because immunogenicity does not generally occur in a dose-responsive manner, the animal findings should be considered more in terms of hazard identification, and it is likely not appropriate to set clinical dose ranges based on immunogenicity-related findings. In both the study and clinical regulatory documents describing immunogenicity-related findings, it is important to distinguish between immunogenicity-related finding, which have questionable translation for human, and direct on-target pharmacologic and toxicologic effects, which are more likely translatable to the human setting. In the author’s preferred approach, these direct on-target effects are most appropriate for setting the clinical dose range. Ultimately, the goal is to provide clinicians and regulators the transparency and perspective on the immunogenicity-related findings while providing clear descriptions on the nature and dose response of direct on-target effects.
The field currently lacks robust data to either definitively confirm or refute the statement that ICD in animals does not predict for similar effects in man. Certainly, there are examples in summary basis of approval documents where the findings in nonhuman primates did not predict for an increased immunogenicity or immune complex in humans. In an important paper discussing both animal and human data, Hussar et al. (2017) describe a high incidence and severity of ICD in monkeys administered an anti-CD20 monoclonal antibody; however, the accumulated clinical data have not identified an increased risk of events attributable to ICD.
The author closed the presentation by emphasizing to the audience that immunogenicity in nonclinical safety study is challenging to predict, and the sequelae associated with immunogenicity are diverse. Study teams mostly keep an open mind and use a weight-of-evidence approach when describing and interpreting findings that may be attributable to immunogenicity.
Footnotes
Acknowledgments
The author greatly benefited from collaborative discussions with pathology, toxicology and immunology colleagues over the years including Nancy Everds, William Siska, Charley Dean, Sven Kronenberg, Frank Brenan, Michael Leach, Jennifer Rojko, Rafael Ponce, Mark Evans, Heather Dale, Tara Arndt, Jamie Blackbourne, and Meredith Steeves. These discussions greatly aided the preparation of the continuing education presentation upon which this article was based.
Author Contributions
The author (JV) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. The author gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
