Abstract
Serelaxin is a recombinant human relaxin-2 intended for cardiovascular indications. Inhalation was chosen as alternative route to intravenous to allow daily administration for chronic applications and home treatment. A total of 4 short-term studies were conducted in rats and cynomolgus monkeys with inhaled formulation of serelaxin at dose up to 10 mg/kg/d. All rats and cynomolgus macaques receiving serelaxin were exposed to the test item. One rat and approximately 50% of macaques developed immunogenicity, which did not appear to affect exposure. No adverse effect on respiratory function or systemic changes was noted. Both species developed similar microscopic lesions characterized by eosinophilic cell infiltration around bronchi; however, in the rat, this was more pronounced and extended to a perivascular location. In addition, in the rat, serelaxin showed eosinophilic crystalline material associated with macrophages in the alveoli and bronchioles. In macaques, serelaxin induced minimal macrophage infiltrates in alveoli and perivascular/peribronchiolar mononuclear cell infiltrations. The minimal airway eosinophilic/mononuclear inflammatory cell infiltrations were considered to be nonadverse in macaques due to the minimal severity and the lack of any other alterations in the lung parenchyma. In the rat, the presence of eosinophilic crystalline material and macrophage response, characterized as precipitated test article, was considered adverse.
Introduction
Serelaxin is a recombinant protein identical in structure to human relaxin-2 protein, a vasoactive peptide hormone, intended for cardiovascular indications (eg, acute heart failure).
1
A full preclinical safety package in rats and cynomolgus monkeys (
To allow chronic daily self-administration, inhalation was chosen as an alternative route of administration over the parenteral route. The large alveolar surface and high vascularization of the lung will allow good systemic absorption with ease of administration and also avoid hepatic first-pass metabolism. 2
In this article, we describe the nonclinical toxicological and toxicokinetic (TK) evaluation of serelaxin by the inhaled route in rats and cynomolgus macaques for 1 and 4 weeks.
Materials and Methods
Animals and Husbandry
Toxicology studies were conducted at Battelle Toxicology for the 1-week toxicity studies and at CRL for the GLP 4-week toxicity studies. Experimentally naive, purpose-bred, cynomolgus macaques (
All animals that underwent a comprehensive health evaluation were acclimated to routine handling procedures prior to study initiation and were not sedated for dose administration. Animals were assigned to one of the treatment groups by a stratified randomization scheme designed to achieve similar group mean body weights.
Study Designs
Rats and cynomolgus macaques were exposed by inhalation (nose-only in rat and head-only in cynomolgus macaques) to air (control) or serelaxin for 60 min/d for 7 or 28 consecutive days in the respective 1- and 4-week studies. In the 1-week rat exploratory study, male rats were assigned to 3 groups (5 animals/group and an additional 7 animals/group for TK) at the target doses of 0, 3, and 10 mg/kg/d. In the 4-week rat study, 4 groups (10 animals/sex/group) of rats were exposed at the target doses of 0, 1, 3, and 10 mg/kg/d. In the 1-week cynomolgus monkey exploratory study, male cynomolgus macaques (∼2.5-6.2 years old and 2.7-8.2 kg) were assigned to 3 groups (3 animals/group) at the target doses of 0, 0.6, and 6 mg/kg/d. In the 4-week cynomolgus monkey study, 4 groups (3 animals/sex/group) of cynomolgus macaques were exposed at the target doses of 0, 0.6, 2, and 6 mg/kg/d. In both 4-week studies, an additional 6 rats/sex or 2 cynomolgus macaques/sex in the control and high-dose group were kept for 8 weeks without any treatment, resulting in a total study duration for recovery animals of 12 weeks. The first day of dosing/exposure was set as Day 1 in the 4 performed toxicity studies.
Mortality checks (twice daily), clinical observations (cage-side observations 1-3 hours relative to the end of inhalation exposure), body weights (once prestudy and prior to dosing on days 1, 3 [rat only], and 7 in the 1-week and weekly in the 4-week and on the day of necropsy, fasted), and food consumption (once prestudy, then weekly) evaluations were performed.
Materials
Serelaxin is a recombinant human relaxin-2 protein formulated as a dry powder aerosol formulation (80:20 [wt/wt] serelaxin:vehicle).
Dosage to the animals was controlled by varying the aerosol concentrations of serelaxin within each exposure plenum and by exposure duration (outlined in Table 1). Aerosol concentrations were varied by using different sized test article reservoirs and varying the feed speed of a rotating brush generator supplied with a stream of predried compressed air. The resultant aerosol moved into a premixing chamber before feeding into the top of a positive flow delivery system/chamber. The target inhaled doses were estimated by using the minute volumes obtained from respiratory function measurement.
Target Inhaled Dose, Aerosol Concentration, and Achieved Inhaled Doses of Serelaxin in the 1- and 4-Week Rat and Cynomolgus Monkey Inhalation Studies.
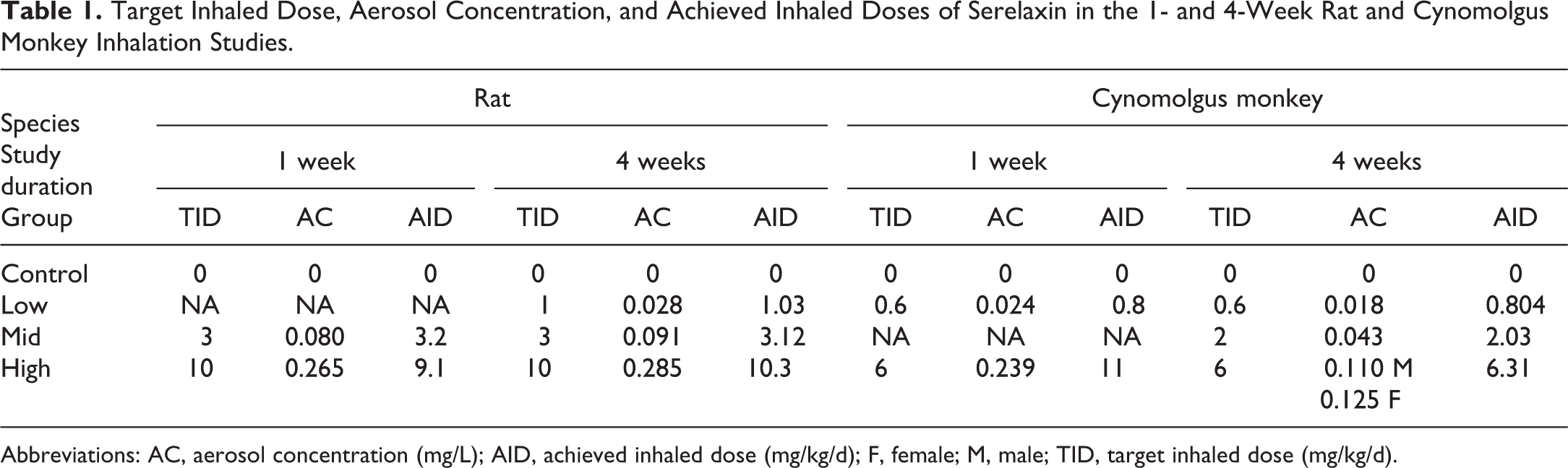
Abbreviations: AC, aerosol concentration (mg/L); AID, achieved inhaled dose (mg/kg/d); F, female; M, male; TID, target inhaled dose (mg/kg/d).

Active concentration is the aerosol concentration of active ingredient as determined by chemical analysis.
The test-item atmosphere concentrations were continually monitored by a real-time Microdust Pro aerosol monitoring system to provide instantaneous qualitative feedback on temporal atmosphere concentration stability. In addition, chamber aerosol concentrations were measured at least 3 times during each exposure occasion from a reference sampling port using a gravimetric method. Filters were submitted for chemical analysis of active ingredient. Aerosol particle size distribution (PSD) was also measured at least once weekly for treated group, except control group 1 due to the lack of respirable mass.
Respiratory Measurements
Respiratory function measurements (tidal volume, respiratory rate, and derived minute volume) were obtained from a subset of animals: 3 rats/group and all cynomolgus macaques once pretreatment, days 1 and 7 in the 1-week studies; and 5 rats/sex/group and all cynomolgus macaques once pretreatment, during study weeks 1, 4, and 12 (end of recovery period) in the 4-week studies.
Rats were placed in “head-out” plethysmographs on the inhalation exposure system and allowed to acclimate to environmental conditions for approximately 15 minutes prior to each data collection period. Ventilatory parameters were obtained using a pneumotach connected to a pressure transducer for an approximate 15 minutes on predose, continuously during dosing, and out to 15 minutes after end of inhalation, and for an approximate 15-minute period at 24 ± 1 hours after end of inhalation.
The cynomolgus macaques were placed in restraint chairs and had the head only (1 week) or the oronasal mask (4 weeks) fitted that was attached to pneumotach and pressure transducer. Animals were given at least 5 minutes to acclimate to the experimental environment and then were monitored for 5-minute period predose, postdose (within 15 minutes of the end of inhalation exposure), and at 24 ± 1 hours after end of inhalation.
The data were analyzed and the parameters calculated using a microcomputer-based physiologic data acquisition system (Buxco or Ponemah Physiology Platform P3). The respiratory parameters were recorded as 30- or 5-second means, respectively, in 1- and 4-week rat studies, and reported as 15-minute means. The cynomolgus macaque respiratory parameters were reprocessed breath by breath and expressed as 5-minute means for the pretreatment, predose, and postdose time points.
Toxicokinetic Assessment and IG Analysis
Blood samples for TK and IG were collected into serum separator tubes from the sublingual vein in rats and from the cephalic or femoral veins in cynomolgus macaques. Serum samples for TK were obtained in the 1-week rat and cynomolgus monkey studies from TK animals on days 1 and 7 at predose, 5, 15, and 30 minutes, and 1, 2, 4, 8, and 24 hours post-inhalation exposure; and a single sample prior to termination on day 8. During the 4-week rat and cynomolgus monkey studies, serum samples were collected on days 1 and 28 at predose, 5 and 30 minutes, and 1, 3, 8, and 23 hours post-inhalation exposure; days 8, 15, and 22 at predose and 5 minutes postdose; day 29 (main phase terminal necropsy); and in recovery on day 30, study weeks 8 and 12 (recovery terminal necropsy). Serum serelaxin was determined by a validated sandwich ELISA (Quantikine ELISA kit, R&D Systems) in which serelaxin was captured by a monoclonal mouse anti-serelaxin antibody precoated onto wells and detected by a goat polyclonal anti-serelaxin HPR-conjugated secondary antibody. The relaxin kit standards from the commercially available Quantikine ELISA kit were used for the preparation of the calibration standards and quality control samples in pooled rat or cynomolgus macaque serum.
For systemic IG analysis, serum samples were obtained for all animals predose on days 1 and 8 in the 1-week rat and cynomolgus monkey studies. In the 4-week rat and cynomolgus monkey studies, serum samples were collected from all animals on day 7 (prestudy) and predose; on days 9, 15, 22, and 29 (main phase terminal necropsy); and in recovery on day 30 and study weeks 8 and 12 (recovery terminal necropsy). The method to measure anti-serelaxin antibodies (ADAs) was an electrochemiluminescence assay in which serelaxin was coated onto a plate and, positive control samples and study samples were added and incubated on the plate. Bound anti-serelaxin antibodies were detected by the addition of ruthenium-labeled serelaxin. The co-reactant tripropylamine was used to enhance electrochemiluminescence. The positive control used in this assay was a goat anti-serelaxin antibody, and the negative control was pooled rat or cynomolgus macaque serum. Specimens were defined as IG/screening positive when at or above the screening cut point.
In addition, in the 4-week rat and cynomolgus monkey studies at scheduled termination, a lobe (right caudal) of the lung was removed and bronchoalveolar lavage fluid (BALF) collection was performed for TK and IG sample analysis.
Clinical Pathology
In rats, blood samples were collected for clinical pathologic evaluations (hematology, serum chemistry, and/or coagulation) from the abdominal aorta on day 8 in the 1-week study and from jugular vein on day 26 and in study week 12 in the 4-week study in fasted rats. In cynomolgus macaques, blood samples were collected from the cephalic or femoral veins on study week 1 (prestudy), and day 8 in the 1-week study and during study week 4 prior to dosing and during the last week of recovery phase (study week 12) in the 4-week study. Standard hematology evaluation of whole blood in EDTA was performed using an ADVIA 120 automated hematology analyzer (Sysmex). Standard clinical chemistry evaluation of serum samples (see Supplement Table 2) was analyzed using a Roche Cobas 501 analyzer (Roche) or a Modular Analytics analyzer (Beckman-Coulter Instruments). For assessment of coagulation parameters, plasma from whole blood collected into tubes containing trisodium citrate anticoagulant was analyzed using an ACL 8000 coagulation analyzer or START4 Compact Stago analyzer (TCOAG Inc).
Urine for urinalysis (see Supplement Table 3) was collected overnight from metabolic cage on day 8 from fasted animals in the 1-week rat and cynomolgus monkey studies, on day 26 and study week 12 in the 4-week rat study. Animals were provided water during urine collection and fasted to prevent any food contamination of the urine specimen. Standard urinalysis was performed on a Clinitek 500 (ATLAS) or Urisys 1800 analyzer (Roche).
Anatomic Pathology
A complete necropsy was performed on all control and treated animals in all studies. Routine organ weights, with paired organs weighed together, were taken at necropsy. Tissues were fixed in 10% neutral-buffered formalin (NBF), embedded in paraffin, sectioned at a nominal thickness of 5 μm, and stained with hematoxylin and eosin. Histopathological examination was performed on a full panel of tissues (following Committee for Proprietary Medicinal Products, Food and Drug Administration, and Environmental Protection Agency guidelines). Special evaluations for inhalation studies were applied based on recommended guidelines. 3 Lungs were fixed with 10% NBF infused via the trachea at 25 cm of water constant pressure using a Marriot bottle. The larynx was preserved and sectioned at 3 levels. Nasal turbinates were flushed with formalin at necropsy and sectioned at 4 levels. All lung lobes with bronchi and longitudinal section of trachea through the bifurcation (carina) were processed. In rats, all processed tissues were examined for control and high-dose groups only, while only gross lesions and potential target organs were examined in intermediate dose(s). In cynomolgus macaques, all processed tissues from all groups were examined. Lesions were graded by a pathologist on a severity scale of 1 to 5 (1 = minimal, 2 = mild, 3 = moderate, 4 = marked, and 5 = severe).
Special Investigations
Confocal Raman Microscopy was performed to determine the composition of the crystals observed microscopically in the lung of the 4-week rat study. For this purpose, sections (20 μM) of formalin-fixed lung from high-dose animals were submitted for identification of the crystalline material of interest by upright light microscopy (using a polarization filter). The crystalline material was then evaluated by molecular fingerprint using Raman single spectrum integration. The spectrum was compared to reference materials: vehicle (standard pharmaceutical excipient) and lyophilized purified serelaxin.
Results
General Observation
Inhalation exposure was well tolerated in all animals treated with serelaxin up to an estimated dose of 10.3 mg/kg/d in rats and 6.31 mg/kg/d in cynomolgus macaques for up to 28 consecutive days (outlined in Table 1). The average gravimetrically determined PSD analysis ranged from 1.9 to 3.0 µm mass median aerodynamic diameter across the 4 different studies and was considered ideal for inhalation exposures of aerosols to rats and cynomolgus macaques. The average exposure environmental temperatures ranged from 20 °C to 21 °C, and the average relative humidity was 26%.
There were no unscheduled deaths, no abnormal clinical observations, and no changes in body weights or food consumption attributable to serelaxin exposures in any of the rat or cynomolgus monkey toxicity studies.
Toxicokinetics and IG
In the rat and cynomolgus monkey toxicity studies, all animals receiving serelaxin were exposed to the test item. Serelaxin was not detected in either serum or BALF in control animals.
In the 1-week toxicity studies, the maximum exposure to serelaxin was observed between 0.083 and 1.1 hours postdose in rats and between 2 and 3 hours postdose in cynomolgus macaques. Exposure to serelaxin (
In the 4-week studies, dose-related systemic exposure to serelaxin was demonstrated in treated animals throughout the dosing phase. The overall exposure to serelaxin appeared to have increased between day 1 and day 28 following daily serelaxin administration. Following inhalation administration of serelaxin, the maximum exposure to serelaxin was from 1.5 to 4 hours on days 1 and 28 in rats, and between 2 and 4 hours postdose on day 1 and between 4 and 9 hours postdose on day 28 in cynomolgus macaques. In the rat, exposure was generally higher for females than males, while there was no gender difference in cynomolgus macaques. Exploratory analysis on BALF samples demonstrated airway exposure to serelaxin in all treated rats and cynomolgus macaques on day 29. Serelaxin concentrations and the total amount recovered in terminal BALF samples from main study animals showed interindividual variability within each group but generally increased in a dose-dependent manner across treated groups. In rats, serelaxin concentration in BALF was also higher in females compared with males in treated groups. There was no evidence of serelaxin in recovery animals.
Antidrug antibodies against serelaxin were detected in only 1 rat on day 50. No ADA was detected in the BALF samples, including the serum-positive ADA rat. In cynomolgus macaques, 10 of 22 animals treated with serelaxin (5/6 at 0.6 mg/kg, and 1/6 main and 4/4 recovery at 6 mg/kg) developed ADA against serelaxin as early as day 9 postdose. Only 1 high-dose animal tested positive in the BALF for ADA. There was no apparent impact of ADA on the serelaxin exposure in the rat and cynomolgus macaques positive for ADA in the inhalation studies.
Respiratory measurement
In the rat studies, following commencement of dosing, there was transient nonadverse change in a few respiratory parameters. In the 1-week study (see Table S4), the mean minute volume were decreased by 17% on day 1 and 11% on day 7 at 3 mg/kg/d and by 23% on day 1 and 9% on day 7 at 10 mg/kg/d, compared to the air control group. In the 4-week study (see Figures S1 and S2), a transient decrease in respiratory rate (11%-31%) occurred in animals, given 1 mg/kg/d during study weeks 1 (males and females) and 4 (males only). This decrease in respiratory rate was compensated by an increase in tidal volume (10%-26%) over the same period, and therefore, no change in overall minute volume was observed. Within 15 minutes of the completion of dosing, all respiratory parameters for treated animals were comparable to both baseline and control values. Given the transient nature, low magnitude of change in respiratory rate and tidal volume and the fact that overall minute volume was not affected, these changes were not considered adverse.
In the cynomolgus monkey studies, there were no changes in respiratory function attributable to serelaxin, when compared to air controls (see Table S5, Figures S3 and S4).
Clinical pathology
In both rat toxicity studies, serelaxin-related changes were minimal in nature and noted in hematology and clinical chemistry (see Table S6). There were no changes noted in hematology, clinical chemistry, coagulation, or urinalysis parameters in both cynomolgus monkey toxicity studies (see Table S7).
In the 4-week rat study, mild increases in eosinophil (Figure 1), neutrophil, and monocyte counts in males and females at ≥3 mg/kg/d, mild increases in lymphocytes in females at ≥1 mg/kg/d, and consequent increases in white blood cell counts in males at 3 mg/kg/d and females at ≥1 mg/kg/d were observed mainly in 2 or 3 individuals per group. These observations correlated with microscopic lung eosinophilic cell infiltration.
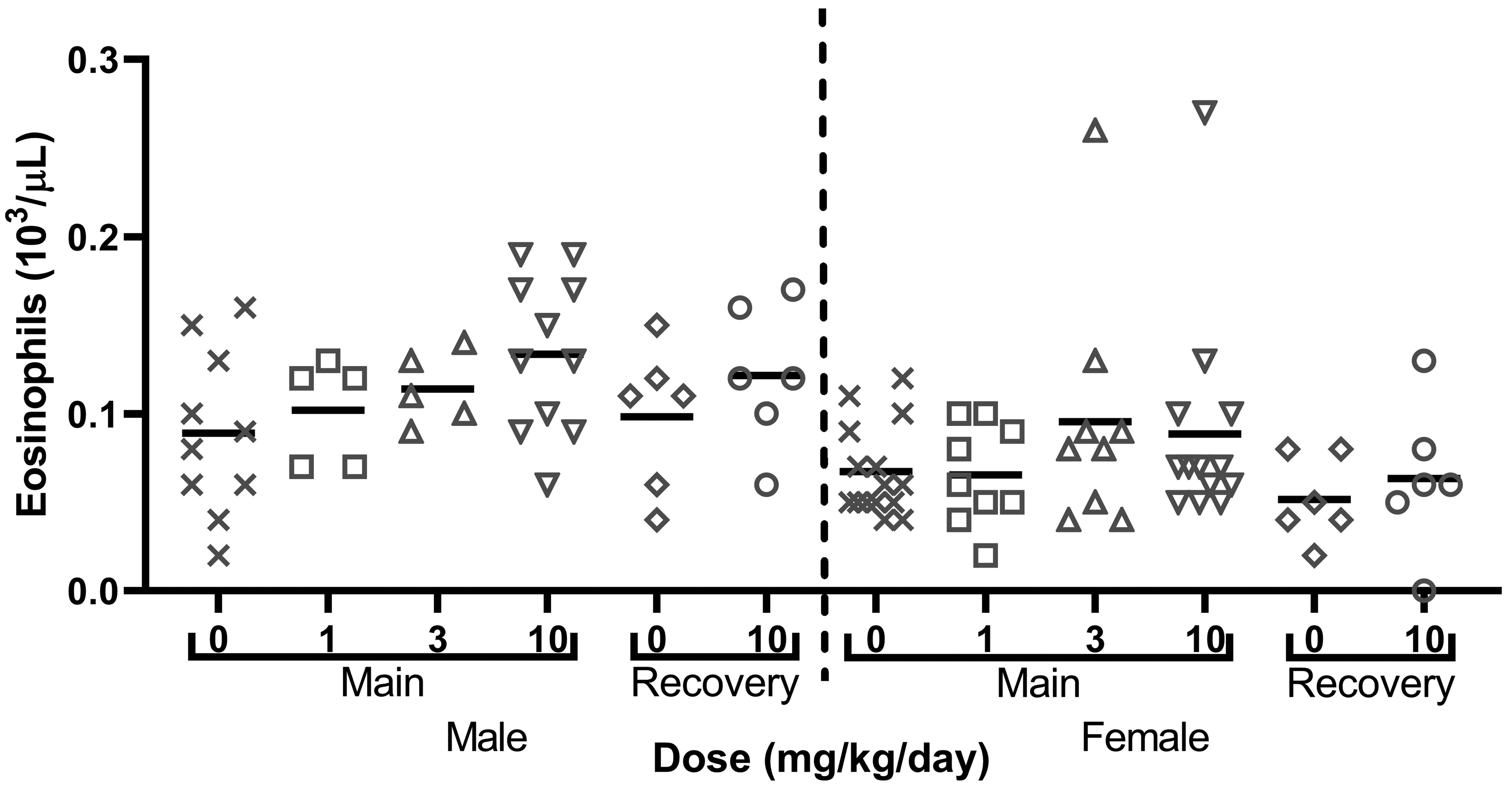
Dose-related nonstatistically significant increases in eosinophil counts at day 29 (main) with partial recovery at day 79 (recovery) in male and female rats in the 4-week study. Data are reported as individual values and mean. Clotted blood sampled were noted, especially in the 1 and 3 mg/kg/d male group resulting in low number (n = 5) of animals analyzed.
In both rat studies, there were minimal decreases in sodium and chloride concentration in males and females at all doses, which are likely related to the pharmacology of serelaxin. In addition, in the 4-week rat study, minimal increases in cholesterol concentration in males at ≥3 mg/kg/d were noted. Since these changes were not dose related and had no microscopic correlates, they were not considered adverse.
Anatomic pathology
There were no macroscopic findings or organ weight changes in both rat studies and in the 1-week cynomolgus monkey study. In the 4-week cynomolgus monkey study, enlarged tracheobronchial lymph nodes were observed in males at all doses and in females given 2 mg/kg/d of serelaxin. This correlated with microscopic findings of increased lymphoid cellularity (minimal to mild) within the lymph nodes. Following an 8-week recovery period, the tracheobronchial lymph node enlargement was still observed in one-fourth of animal that had been administered 6 mg/kg/d of serelaxin, suggesting partial reversibility of this finding.
Microscopic findings considered related to serelaxin administered by inhaled route were observed in lung in the 4-week rat study only and, together with nasal cavity observations, in both cynomolgus monkey studies (outlined in Table 2). In addition, in the 4-week rat study, microscopic findings in nonrespiratory tract tissues, consisting in uterus endometrial stromal hypertrophy and cervix stromal hypertrophy, were observed in female rats given ≥3 mg/kg/d. No serelaxin-related microscopic findings were observed in the 1-week rat study.
Summary of Relevant Histopathological Changes in the Lungs and Tracheobronchial Lymph Nodes in the 4-Week Rat Study and in the 1- and 4-Week Cynomolgus Monkey Studies.a
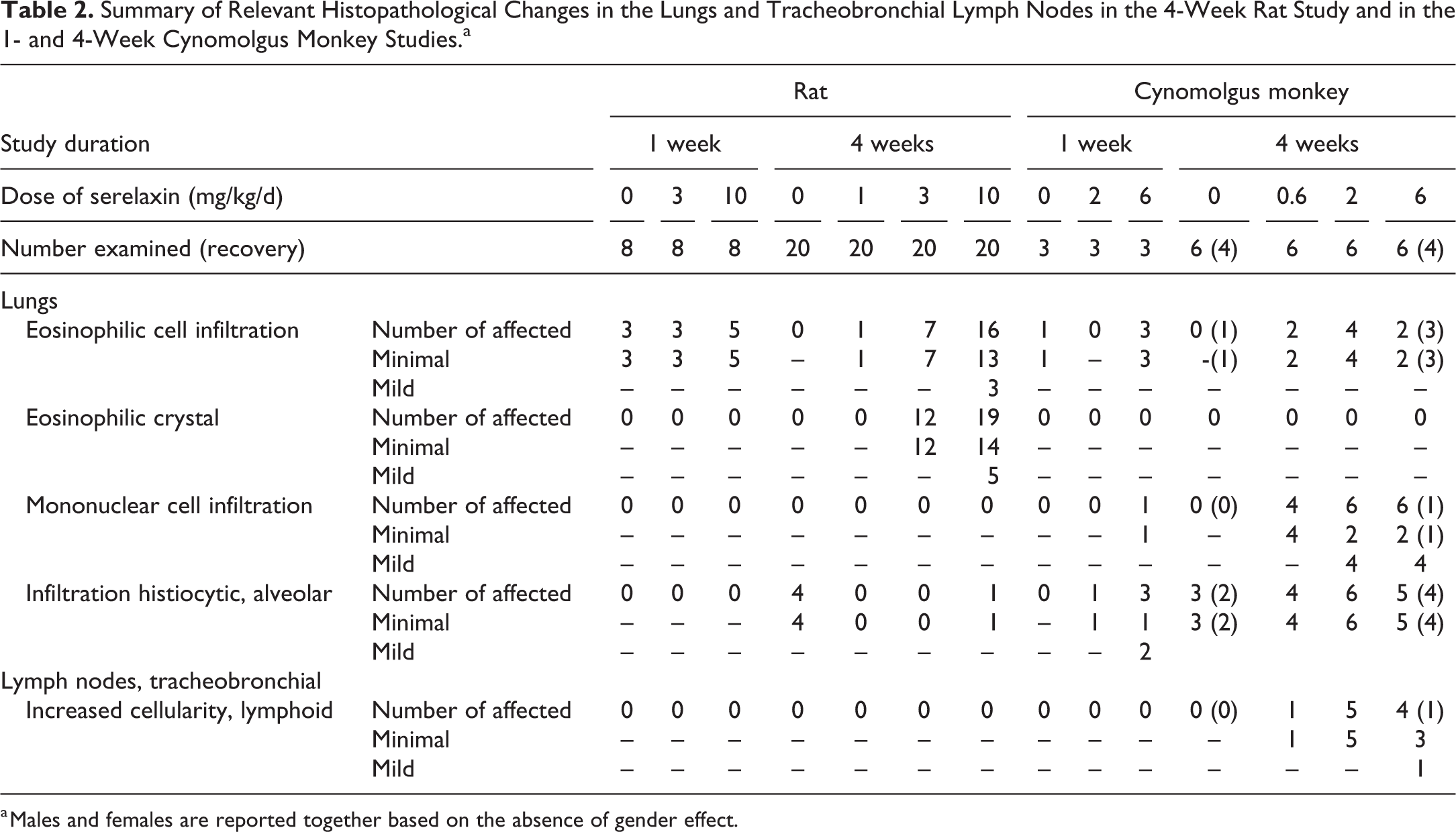
a Males and females are reported together based on the absence of gender effect.
In the 4-week rat study, increased incidences of minimal to mild perivascular/peribronchiolar (PV/PB) eosinophilic cell infiltrates (Figure 2A) were noted in lungs of males at all doses and females given ≥3 mg/kg/d. Minimal focal PV/PB eosinophilic cell infiltration was also seen in the 1-week rat study; however, since this finding was observed with no significant incidence difference between control and treated, they were interpreted to be incidental in this study. In addition, minimal crystalline eosinophilic material (Figure 2) was observed within alveoli and/or bronchioles in males and females given ≥3 mg/kg/d. This material had a rectangular crystalline appearance with pale eosinophilic to a faint light blue, with a thin bright eosinophilic margin around the well-defined edges. Generally, there were a few macrophages associated with or attached to the edges of the material. There was no evidence of hemorrhage or damage to alveolar/bronchiolar epithelium in the vicinity of the material. Under polarized lights, no birefringence was observed. The Turnbull blue stain for iron/hemosiderin applied to lung sections was negative and indicated crystal was not composed of hemoglobin. Following the 8-week recovery period, there was complete recovery of the microscopic findings.
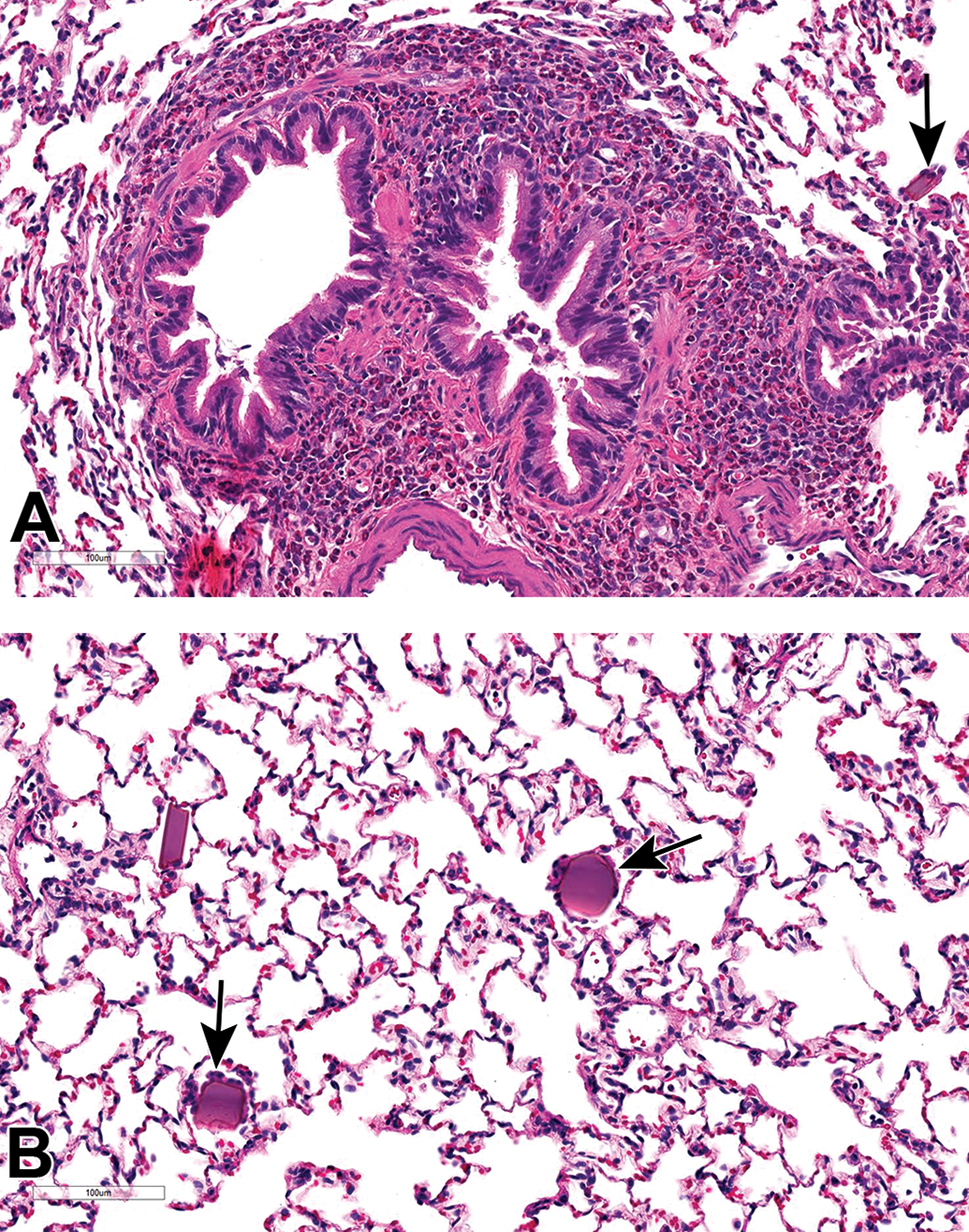
Histopathological changes in lungs of rats treated for 4 weeks with serelaxin. All sections stained with hematoxylin and eosin. Perivascular/peribronchiolar eosinophilic cell infiltrates (A) and crystalline eosinophilic material in alveoli surrounded by macrophages (arrows; A and B).
In cynomolgus macaques, minimal multifocal eosinophilic cell infiltrates (Figure 3A) were noted in both inhalation studies in all animals given 6 mg/kg in the 1-week study, and in 1 male administered 0.6 or 2 mg/kg/d and all females at all doses in the 4-week study. The eosinophilic cell infiltration was mostly PB following 1 week of administration while also infiltrating the bronchial epithelium after 4 weeks of treatment. In the 1-week cynomolgus monkey study, multifocal alveolar histiocytic infiltrates were observed in 1 cynomolgus macaque at 0.6 mg/kg/d and all cynomolgus macaques at 6 mg/kg/d. These infiltrates were characterized by discrete accumulations of large histiocytes within contiguous alveoli, occasionally expanding within the associated alveolar septa and sometimes associated with eosinophilic proteinaceous material. Minimal PV/PB mononuclear cell infiltration, primarily lymphocytic in nature, was seen in 1 male given 6 mg/kg/d in the 1-week study. Following 4 weeks of dosing, minimal to mild PV/PB mononuclear cell infiltration was seen in most animals at all doses and was associated with BALT lymphoid hyperplasia (follicular). Following an 8-week recovery period, there was partial recovery of most findings in the lung. One male still had PV/PB mononuclear cell infiltration and 2 female cynomolgus macaques still had bronchial eosinophilic infiltration at the end of the 8-week recovery period.
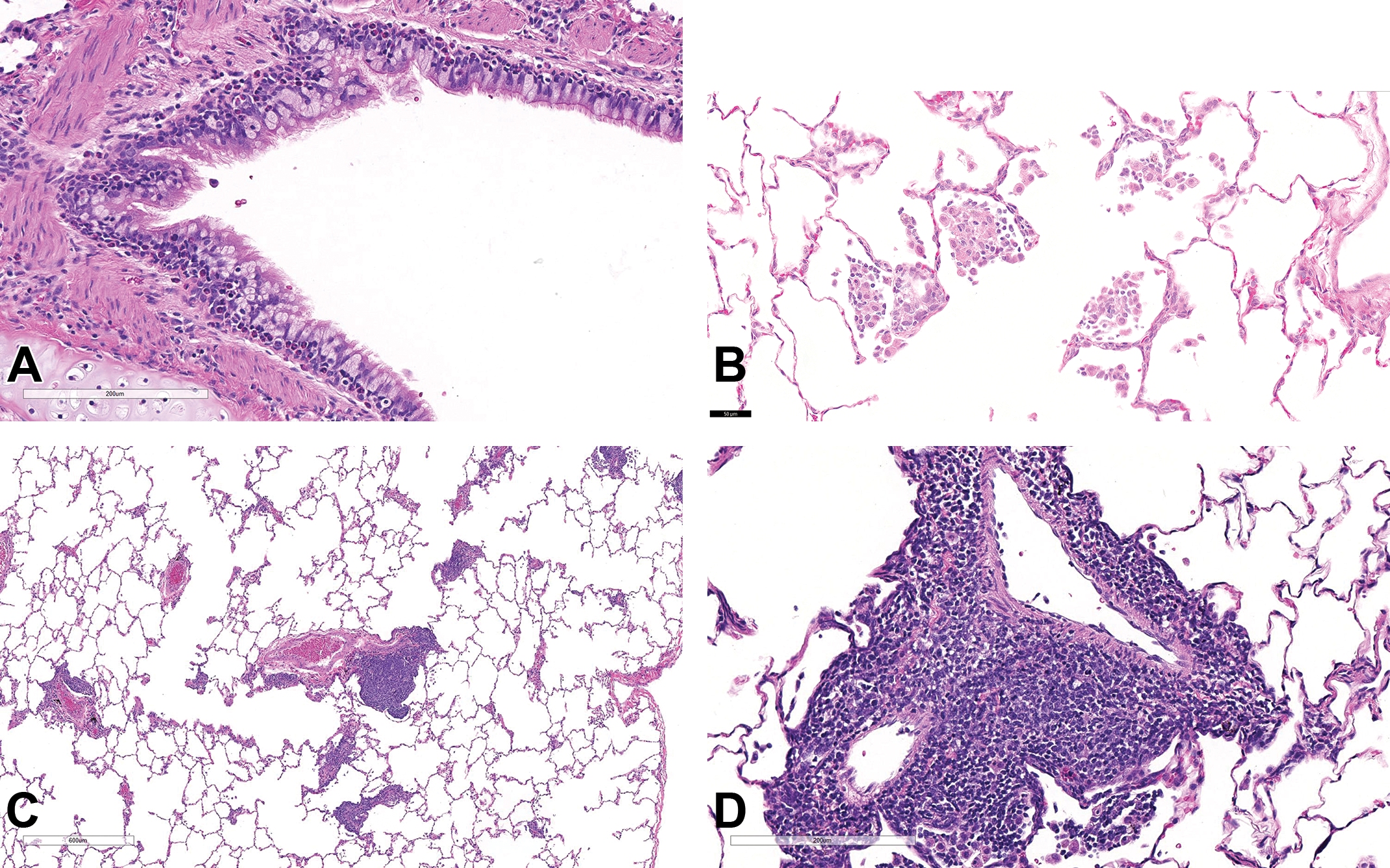
Histopathological changes in the lungs of cynomolgus macaques treated for 4 weeks with serelaxin. All sections stained with hematoxylin and eosin. Peribronchiolar/bronchiolar eosinophilic cell infiltration (A), alveolar histiocytic infiltrates (B), and perivascular/peribronchiolar mononuclear cell infiltration (C and D).
In cynomolgus macaques, neutrophilic inflammation, vacuolation of olfactory epithelium, squamous metaplasia of respiratory epithelium, and/or squamous epithelium degeneration, hyperplasia, and/or erosion (Figure 4) were observed in the nasal cavity of a some animals exposed to serelaxin at all doses in both studies (see Table S8). These lesions were most prominent in the rostral section and along the septum and were not seen in control animals. The respiratory and squamous epithelial cells were rounded with pyknotic nuclei and hypereosinophilic cytoplasm. Occasionally, the squamous epithelium was denuded with associated inflammation transmigrating into the nasal cavity. Hyaline droplets were occasionally seen in the cytoplasm of respiratory and squamous epithelial cells. The inflammation expanded the submucosa underlying the squamous epithelium, which was sometimes thickened with 2 to 5 layers of overlying keratinized epithelium. In addition, in the 4-week cynomolgus monkey study, there was minimal neutrophilic cell infiltration in the larynx of males and females at ≥2 mg/kg/d of serelaxin and in the carina of one male at 6 mg/kg/d. Minimal squamous metaplasia in the larynx was also seen in 1 female administered 6 mg/kg/d of serelaxin.
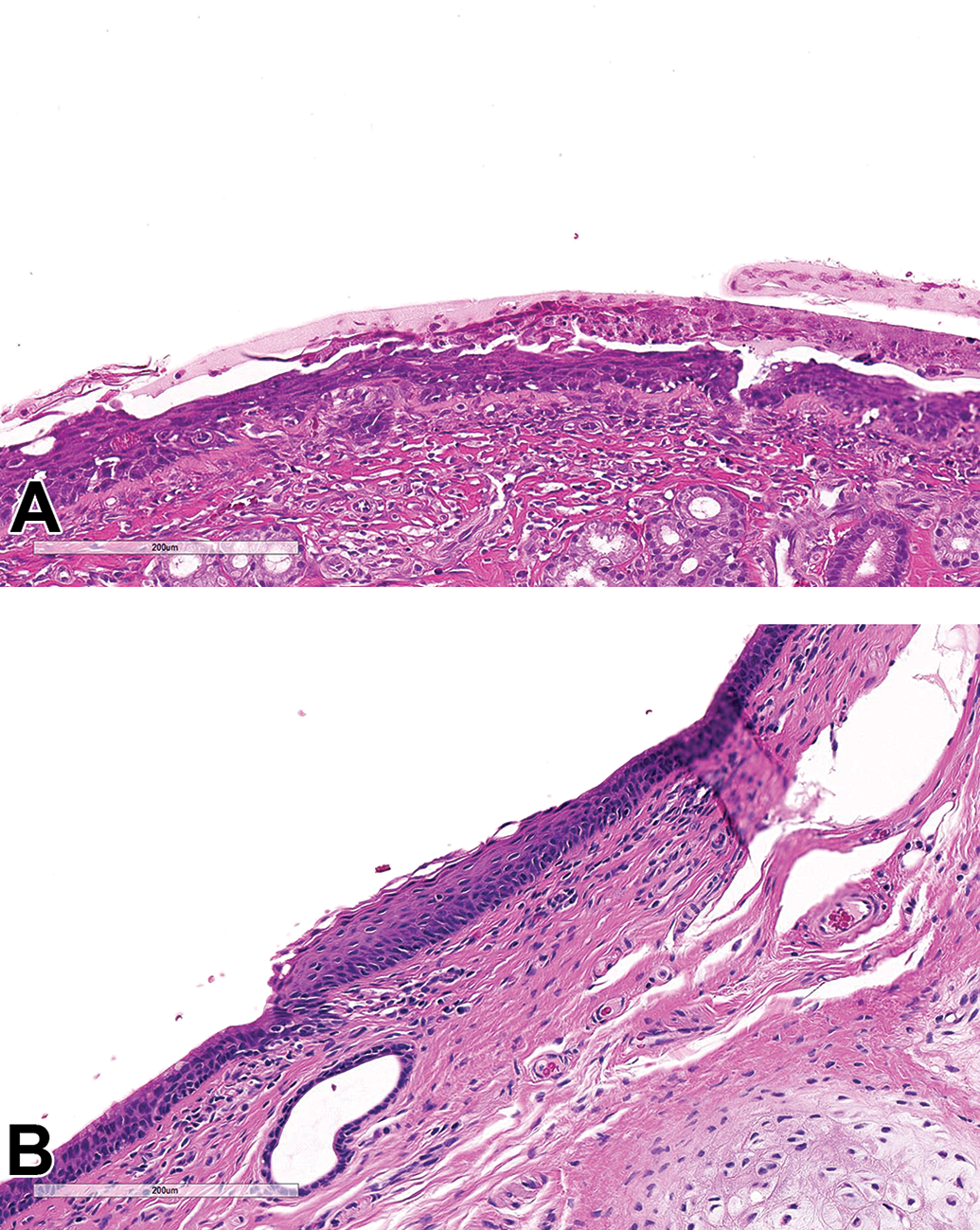
Nasal cavity in cynomolgus macaques treated for 4 weeks with serelaxin. All sections stained with hematoxylin and eosin. Squamous epithelium erosion (A) and squamous metaplasia of respiratory epithelium (B).
Determination of the composition of the eosinophilic crystals found
Determination of the composition of the eosinophilic crystals found in the lungs of rats treated for 4 weeks with serelaxin was performed on 6 such crystals/particles of material originating from 2 sections. All 6 crystalline materials evaluated showed matching and specific vibrational spectra indicating a single component. The spectral comparison of the Raman fingerprint of the crystals obtained from the histology sections with the extracted and averaged reference Raman spectrum obtained from lyophilized serelaxin showed a good correlation of spectral patterns in terms of peak position and, to a large extent, relative intensity (Figure 5). In conclusion, the crystalline particles evaluated in histology sections of treated animals were strongly related to serelaxin reference constituent and likely corresponded to the active molecule.
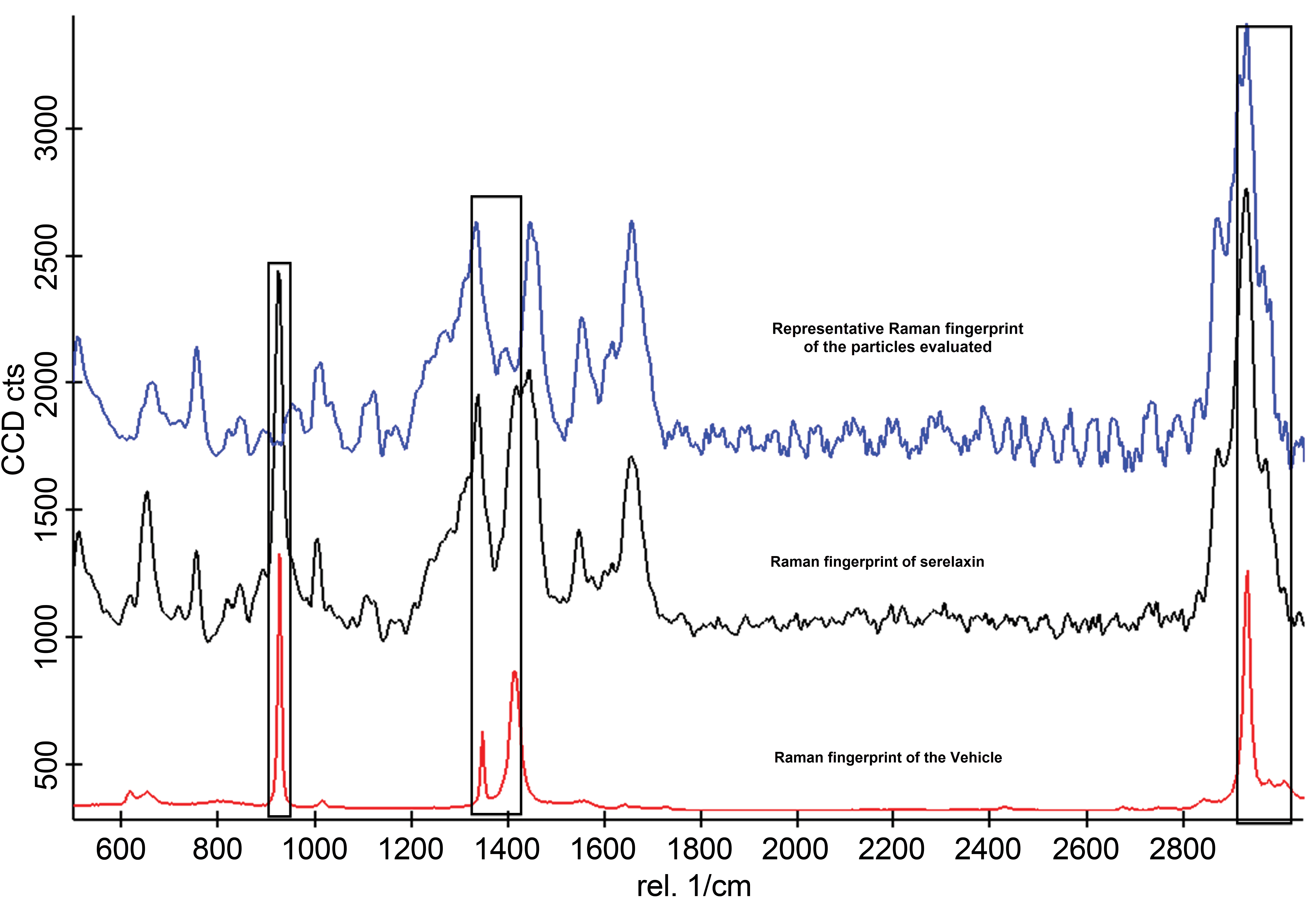
Detailed vibrational peak comparison showing correlation of spectrum associated with the particles of interest (blue spectrum) and lyophilized serelaxin (black spectrum) outside vehicle (red spectrum) interference (interference regions marked with black columns).
Discussion
Toxicological evaluation of serelaxin in rats and cynomolgus macaques revealed a good safety and tolerability profile following daily inhalation doses up to 10 mg/kg/d for up to 4 weeks. Animals were exposed systemically to serelaxin as expected. Pharmacology-related effects were observed in rat with decreases in sodium and chloride concentrations and microscopically in female reproductive tract tissues at ≥1mg/kg/d. The uterine and cervical stromal hypertrophy observed in female rats given ≥3 mg/kg/d was consistent with the expected pharmacological effects of relaxin in female rats. 4,5 Inhalation-specific toxicity findings were seen in both species following, mostly, 4 weeks of dosing.
A common microscopic finding in both species was PV/PB eosinophilic cell infiltration. It correlated with systemic eosinophilia in rats in which eosinophilic cell infiltrations were more pronounced compared to cynomolgus macaques. Minimal PV eosinophilic cell infiltration is an occasional background finding not only in rat lungs but also in cynomolgus macaque lungs. 6,7 In the 4-week rat study and 1- and 4-week cynomolgus monkey studies, the eosinophilic cell infiltrations were seen with higher incidence and severity compared to control groups and were considered treatment related. Etiology is not well known, but airway eosinophilia has been previously reported in the literature as a typical hypersensitivity response to postsensitization airway challenge to a variety of inhaled foreign protein/materials. 8 -12
In rats, the lungs had dose-dependent crystalline eosinophilic material present within alveoli and/or bronchioles (generally with associated or attached macrophages). Eosinophilic crystal can be occasionally observed in association with alveolar hemorrhage due to formation of hemoglobin crystals. 6,7,13 Other forms of crystal can also be found within macrophages associated with inflammatory reaction, often granulomatous, and correspond to inhalation of poorly soluble or inert particles (eg, crystalline silica), which could be located by their birefringence with polarized light. 7,14 In mice, eosinophilic crystals, composed of Ym1 protein, can be observed within macrophages along with an inflammatory reaction in lungs and within epithelial cells in few other tissues. 15 -17 In the present 4-week rat study, the negative Turnbull blue staining with the absence of hemorrhage and absence of birefringence or inflammatory reaction indicated that the eosinophilic material present in alveoli was not consistent with hemoglobin crystals or inert particles. Confocal Raman microscopy identified this material to be related to protein crystallization, most likely serelaxin. The crystalline material was no longer observed following the 8-week dose-free period, further confirming a likely relationship of this material to the inhaled material. In vivo protein crystallization in animal is a rare event but under certain condition (eg, pH, humidity, and temperature) can occur. 17 -19 In vitro protein crystallization is challenging, but Eigenbrot et al 20 were able to crystalize human relaxin to study its structure. In addition, further internal investigation demonstrated that serelaxin, either in vapor-phase hydration at high relative humidity or in a suspension of the powder in water, can crystalize under pH, humidity, and temperature conditions, representing the physiological lung environment in rat. Since these conditions are rodent-specific and crystalline material was not observed in cynomolgus macaques, it was considered unlikely to translate to human, although that eventuality could not be completely excluded.
In cynomolgus macaques, histopathological findings in the lung progressed from histiocytic infiltrates following 1 week of dosing to PV/PB mononuclear cell infiltration associated with increased lymphoid cellularity in the tracheobronchial lymph nodes following 4 weeks of dosing. The histiocytic infiltrate was considered to be a nonspecific response to inhaled foreign proteins as evidenced by the eosinophilic proteinaceous material. 21 -23 The lymphocytic response in the lungs and tracheobronchial lymph nodes was consistent with a nonspecific immunological reaction to inhaled foreign proteins, which was likely reflected by the high level of ADA. 24,25 These findings were, therefore, not considered to be serelaxin specific.
Nasal epithelial inflammation, hyperplasia, metaplasia, and/or erosions occurred in the rostral nasal cavity of cynomolgus macaques exposed to serelaxin. This finding, along with larynx (neutrophilic infiltration and squamous metaplasia) and carina (neutrophilic infiltration) changes, was consistent with the findings observed with inhaled powder vehicles (private communication from CRO). The cause of these observations may be direct exposure-induced epithelial cell damage or mechanical epithelial cell damage. 26
In summary, serelaxin was safe and well tolerated following inhalation for up to 4 weeks. Findings observed were considered consistent with a nonspecific immunological response to inhaled foreign protein; in this case, a recombinant human protein. In cynomolgus macaques, the no observed adverse effect level (NOAEL) was considered to be the maximal achieved dose of 6.31 mg/kg/d based on the absence of adverse effects, including the low severity, nonspecificity, and the partial recovery of the microscopic observations. In rat, the NOAEL was considered to be the achieved dose of 1.03 mg/kg/day due to the presence of intra-alveolar crystalline eosinophilic, material with a macrophage response, in the lungs at higher doses.
Supplemental Material
Supplemental Material, sj-tif-1-tpx-10.1177_0192623320943129 - Nonclinical Safety Assessment of an Inhaled Formulation of Serelaxin: A Recombinant Human Protein in Rats and Cynomolgus Monkeys (Macaca fascicularis )
Supplemental Material, sj-tif-1-tpx-10.1177_0192623320943129 for Nonclinical Safety Assessment of an Inhaled Formulation of Serelaxin: A Recombinant Human Protein in Rats and Cynomolgus Monkeys (
Supplemental Material
Supplemental Material, sj-tif-2-tpx-10.1177_0192623320943129 - Nonclinical Safety Assessment of an Inhaled Formulation of Serelaxin: A Recombinant Human Protein in Rats and Cynomolgus Monkeys (Macaca fascicularis )
Supplemental Material, sj-tif-2-tpx-10.1177_0192623320943129 for Nonclinical Safety Assessment of an Inhaled Formulation of Serelaxin: A Recombinant Human Protein in Rats and Cynomolgus Monkeys (
Supplemental Material
Supplemental Material, sj-tif-3-tpx-10.1177_0192623320943129 - Nonclinical Safety Assessment of an Inhaled Formulation of Serelaxin: A Recombinant Human Protein in Rats and Cynomolgus Monkeys (Macaca fascicularis )
Supplemental Material, sj-tif-3-tpx-10.1177_0192623320943129 for Nonclinical Safety Assessment of an Inhaled Formulation of Serelaxin: A Recombinant Human Protein in Rats and Cynomolgus Monkeys (
Supplemental Material
Supplemental Material, sj-tif-4-tpx-10.1177_0192623320943129 - Nonclinical Safety Assessment of an Inhaled Formulation of Serelaxin: A Recombinant Human Protein in Rats and Cynomolgus Monkeys (Macaca fascicularis )
Supplemental Material, sj-tif-4-tpx-10.1177_0192623320943129 for Nonclinical Safety Assessment of an Inhaled Formulation of Serelaxin: A Recombinant Human Protein in Rats and Cynomolgus Monkeys (
Footnotes
Acknowledgments
The authors thank Malak Bitar, Karyn Colman, Batelle Toxicology, and Charles River Laboratories Montreal for their expertise and support provided across these studies.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
