Abstract
Mer proto-oncogene tyrosine kinase (MerTK), expressed in the retinal pigment epithelium (RPE), regulates the phagocytosis of shed photoreceptor outer segments. To investigate the influence of dosing time on MerTK inhibitor UNC569-induced retinal toxicity, UNC569 at 100 mg/kg was orally administered to male mice at 2 different Zeitgeber times (ZT5.5 or ZT22) for 28 days. Electron microscopy was conducted at ZT2 after the final dosing. Additionally, the visual cycle components (11-cis-retinal, all-trans-retinal, all-trans-retinol, and 11-cis-retinol), which play an important role in maintaining retinal homeostasis, were quantified by liquid chromatography/mass spectrometry/mass spectrometry. Under electron microscopic examination, the number of phagosomes and phagolysosomes in the RPE increased in both the ZT5.5 and ZT22 administered groups, while endoplasmic reticulum dilatation in the RPE and chromatin aggregation of photoreceptor nuclei were observed only in the ZT22 administered group. No change was observed in any of the visual cycle components. These results suggest that the timing of the dosing in relation to the physiological MerTK phosphorylation affected the severity of changes in the RPE, leading to the apoptosis of the photoreceptor cells.
Keywords
Introduction
The circadian rhythm is a physiological phenomenon that fluctuates in a cycle of 24 hours. In general, it is formed endogenously and corrected by external stimuli, such as light, temperature, and food. In concern with this biological rhythm, the administration time of the certain drugs is known to affect the efficacy and toxicity, for example, tumor inhibition by anticancer drugs and hepatotoxicity by acetaminophen. 1,2
In the retina, photoreceptor cells and retinal pigment epithelium (RPE) are known to display a circadian rhythm. Photoreceptor cells lack the innate capacity to regenerate, whereas their photoreceptor outer segments (POSs) are continuously renewed. 3,4 The POS turnover starts with shedding their tips with the onset of light, followed by the phagocytosis of the shed POS by the RPE. Consequently, phagocytosis of the shed POS is also synchronized with circadian rhythms. 5
The phagocytosis of the shed POS consists of 3 phases: binding, internalizing, and digesting. The former 2 are receptor-mediated events, with the αvβ5 receptor and Mer proto-oncogene tyrosine kinase (MerTK) receptor mediating the binding and internalization of the shed POS, respectively. 6 -8 Mer proto-oncogene tyrosine kinase is a member of the TAM (Tyro 3, Axl, and Mer) transmembrane receptor tyrosine kinase subfamily and expressed in monocytes/macrophages, dendritic cells, natural killer cells, natural killer T cells, megakaryocytes, and platelets. In the retina, MerTK is expressed in the RPE and its phosphorylation was also shown to be synchronized with circadian rhythms in mice. 5 After internalization, similar to the general process of phagocytosis, the engulfed shed POSs are enclosed by cellular membranes forming phagosomes, followed by fusion with lysosomes forming phagolysosomes, and are finally degraded by lysosomal enzymes in the RPE. 9 In mice, POSs are entirely replaced by newly formed segments every 10 days. 3,4
The phagocytosis of shed POS is one of the most crucial roles of the RPE for the function and survival of photoreceptor cells, in addition to the transepithelial transport between photoreceptors and the choriocapillaris and the regeneration of 11-cis-retinal (11cRAL) in the visual cycle. 10 The failure of the phagocytic process in the RPE can evoke abnormalities in photoreceptor cells. The Royal College of Surgeons rat, a widely used model of retinal dystrophy, has a genomic deletion in the second exon of MerTK and demonstrates the loss of photoreceptor cells attributed to the failure of the RPE to phagocytize shed POS. 11,12
Recently, we reported that UNC569, a specific MerTK inhibitor, 13 induces ultrastructural changes both in the RPE and the photoreceptor cells of mice. 14 Since physiological MerTK phosphorylation and RPE phagocytosis are synchronized with circadian rhythms, we hypothesized that mice demonstrate diurnal variation in MerTK-induced retinal toxicity, that is, chronotoxicity. In the present study, ultrastructural changes in the retina were investigated in mice orally administered UNC569 at 2 different Zeitgeber times (ZT) to evaluate the effects of dosing time on the retinal toxicity caused by MerTK inhibition. Zeitgeber time is the time of the biological clock in the 12/12-h light–dark cycle, where the start time of the light and dark cycle is ZT0 and ZT12, respectively. Additionally, the visual cycle components, 11cRAL, all-trans-retinal (atRAL), all-trans-retinol (atROL), and 11-cis-retinol (11cROL), in the retina were measured to examine the relationship between the visual cycle and UNC569-induced lesions in photoreceptor cells.
Materials and Methods
Test Substance
UNC569 was synthesized at Daiichi Sankyo RD Novare Co, Ltd.
Animals
A total of 58 male BALB/c AnNCrlCrlj mice, aged 7 weeks, were obtained from Charles River Japan, Inc. The animals were individually housed in stainless steel mesh cages, in a room controlled at a temperature of 23 °C ± 2 °C and a relative humidity of 55% ± 15%. The animals were maintained under a 12-h light and 12-h dark cycle (11:00 on and 23:00 off) with exposure to 170 to 210 lux of fluorescent lighting during the light phase. Commercial pellets (CRF-1, Oriental Yeast Co, Ltd) and tap water were available ad libitum. All animal procedures were performed in accordance with the guidelines of the Animal Care and Use Committee of Daiichi Sankyo Co, Ltd (Tokyo, Japan).
Experimental Design
The experimental protocol for the dosing schedule, sampling schedule of the retinal tissue, and lighting conditions are shown in Figure 1 and were set based on the results of our previous study. The animals were divided into 2 groups, ZT5.5 and ZT22 groups. UNC569 at 100 mg/kg was orally administered once daily, for 14 or 28 days at ZT5.5 and ZT22. The animals were further subdivided into 2 groups for pathological examination (3 animals) and quantification of visual cycle components (5 animals) for each treatment period. Control animals were set to each group except for the pathological examination group of the 14-day treatment period and administered 0.5 wt/vol% methylcellulose in the same manner. The dose levels were set based on our previous results, in which ultrastructural changes in the RPE were observed without death or moribundity in animals during the 14-day administration period. 14 The first day of dosing was defined as day 1. The animals were euthanized by exsanguinations under isoflurane anesthesia and necropsied at ZT2 on day 15 or 29.
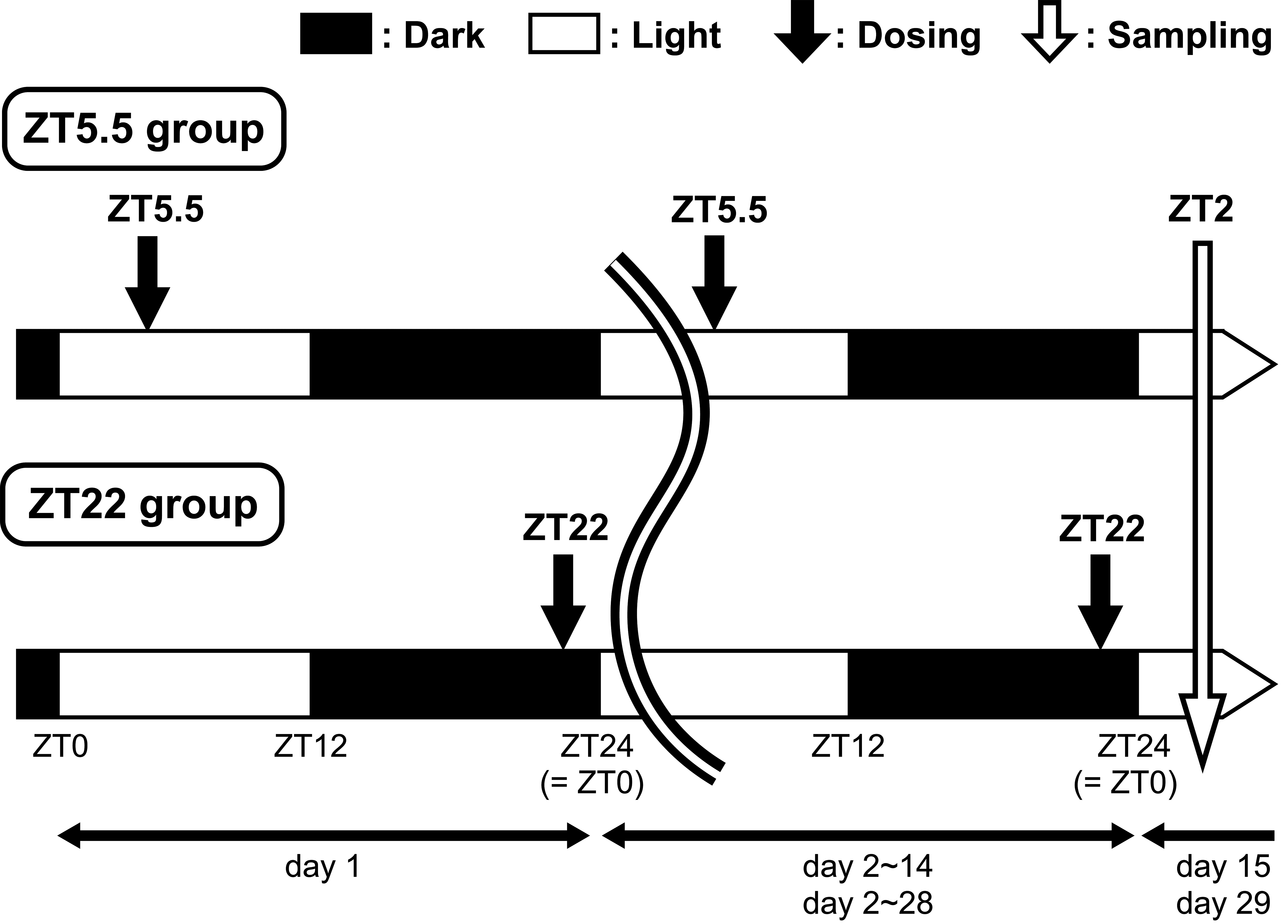
Time schedule for the dosing, sampling of the retinal tissue, and the lighting conditions.
Pathology
At necropsy, the eyes were removed from all animals. Right eyes were fixed in PAXgene Tissue (PreAnalytiX GmbH, Hombrechtikon) and embedded in paraffin, and histopathological specimens stained with hematoxylin and eosin (HE) were prepared for light microscopy. Left eyes were fixed in 2.5% glutaraldehyde, postfixed in 2% osmium tetroxide, and embedded in Quetol 812 (Nisshin EM) resin. Semithin sections were stained with toluidine blue for light microscopy, and ultra-thin sections were stained with uranyl acetate and Sato’s lead 15 for electron microscopy.
Quantification of Retinoids
At necropsy, the eyes were removed from all animals, and the cornea, lens, and vitreous were removed from the eyeballs. The obtained right and left retinal tissue samples, consisting of the retina, optic disc, choroid, and sclera, were homogenized with 500 µL of water/acetonitrile (1/1, vol/vol) and extracted by centrifugation at 800 × g at 4 °C for 5 minutes. The 50 µL of lysates were mixed with 10 µL of internal standard (all-trans Retinal-d5, Santa Cruz Biotechnology, Inc) and 140 µL of acetonitrile, and centrifuged at 13300 × g at 4 °C for 3 minutes. Measurement samples were obtained by adding 40 µL of purified water to 100 µL of lysates, and the concentration of 11cRAL, atRAL, atROL, and 11cROL were measured by liquid chromatography/mass spectrometry/mass spectrometry. All procedures, from anesthesia to measurements, were performed under red light as the handling of samples under fluorescent light would result in the isomerization and degradation of retinoids. 16
Statistical Analysis
The concentrations of visual cycle components are expressed as the mean ± standard deviation. For statistical analysis between the UNC569-treated ZT5.5 or ZT22 groups and each control group on days 15 or 29, the parameters were statistically analyzed by F test, and the t test and Welch test were performed for equal and unequal variances, respectively. These statistical analyses were performed using the SAS System Release 9.2 (SAS Institute, Inc). A P value less than 5% was considered statistically significant.
Results
There were no changes in the clinical signs or body weights of all animals.
Pathology
Microscopically, no histopathological changes were observed in the HE sections of all groups. Ultrastructural findings in electron microscopy are summarized in Table 1. On day 29, membrane-bound multilamellar bodies resembling POS were observed in the perinuclear zone of the RPE in the ZT5.5 and ZT22 vehicle control groups (Figure 2A and D). No differences were observed between these 2 control groups. On day 15, in the ZT5.5 and ZT22 UNC569 groups, membrane-bound multilamellar bodies were increased compared to those observed in the control, whereas no differences were observed between the ZT5.5 and ZT22 UNC569 groups (Figure 2B and E). Increased multilamellar bodies were mainly distributed from the apical cell surface to the perinuclear zone of the RPE. The lamellar elements were surrounded by a single membrane and showed various appearances. Some closely resembled the POS, whereas others showed thinner lamellae than those of the POS. Some lamellar elements were disorganized, forming electron-lucent areas. Additionally, multilamellar bodies with closely packed lamellar elements forming electron-dense cores were observed (Figure 2B and E; insets). On day 29, similar changes were observed in the ZT5.5 and ZT22 UNC569 groups (Figure 2C and F). Interestingly, the endoplasmic reticulum (ER) in the RPE was dilated only in the ZT22 UNC569 group on day 29 (Figure 2F). Furthermore, chromatin-condensed nuclei in the outer nuclear layer were observed only in the ZT22 UNC569 group on day 29 (Figure 3). No changes were observed in the organelles including the mitochondria of photoreceptor cells in all UNC569 groups.
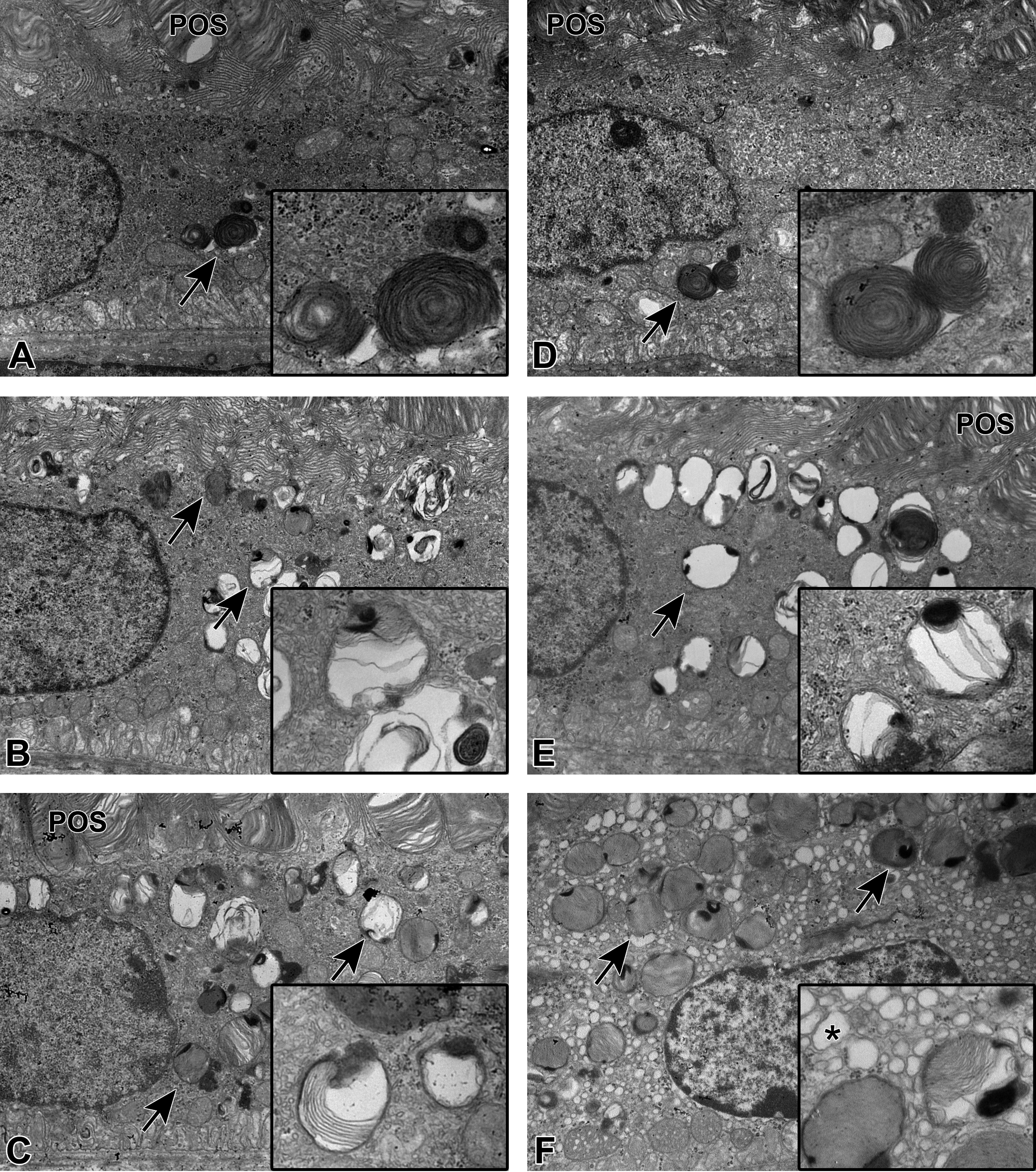
Electron microscopic appearance of the retinal pigment epithelium in mice treated with UNC569. A, Zeitgeber time (ZT) 5.5 vehicle control group and (D) ZT22 vehicle control group on day 29. Membrane-bound multilamellar bodies (arrows) resembling photoreceptor outer segments (POSs) were observed in the perinuclear zone of retinal pigment epithelium (RPE). Insets, Higher magnification of POS-resembled multilamellar bodies. B, ZT5.5 UNC569 group and (E) ZT22 UNC569 group on day 15. Increased membrane-bound multilamellar bodies (arrows) distributed from the apical surface to the perinuclear zone of the RPE. Some lamellar elements were disorganized, forming electron-lucent areas and electron-dense cores emphasized in insets. C, ZT5.5 UNC569 group and (F) ZT22 UNC569 group on day 29. In addition to the same changes observed on day 15 in both ZT5.5 and ZT22 groups, the dilated endoplasmic reticulum (ER) in the RPE was observed only in the ZT22 UNC569 group. Inset, Higher magnification clearly indicated the dilatation of ER (asterisk). Increased membrane-bound multilamellar bodies (arrows) were observed, as well as in ZT5.5 and ZT22 UNC569 groups on day 15. Original objective: ×10,000.
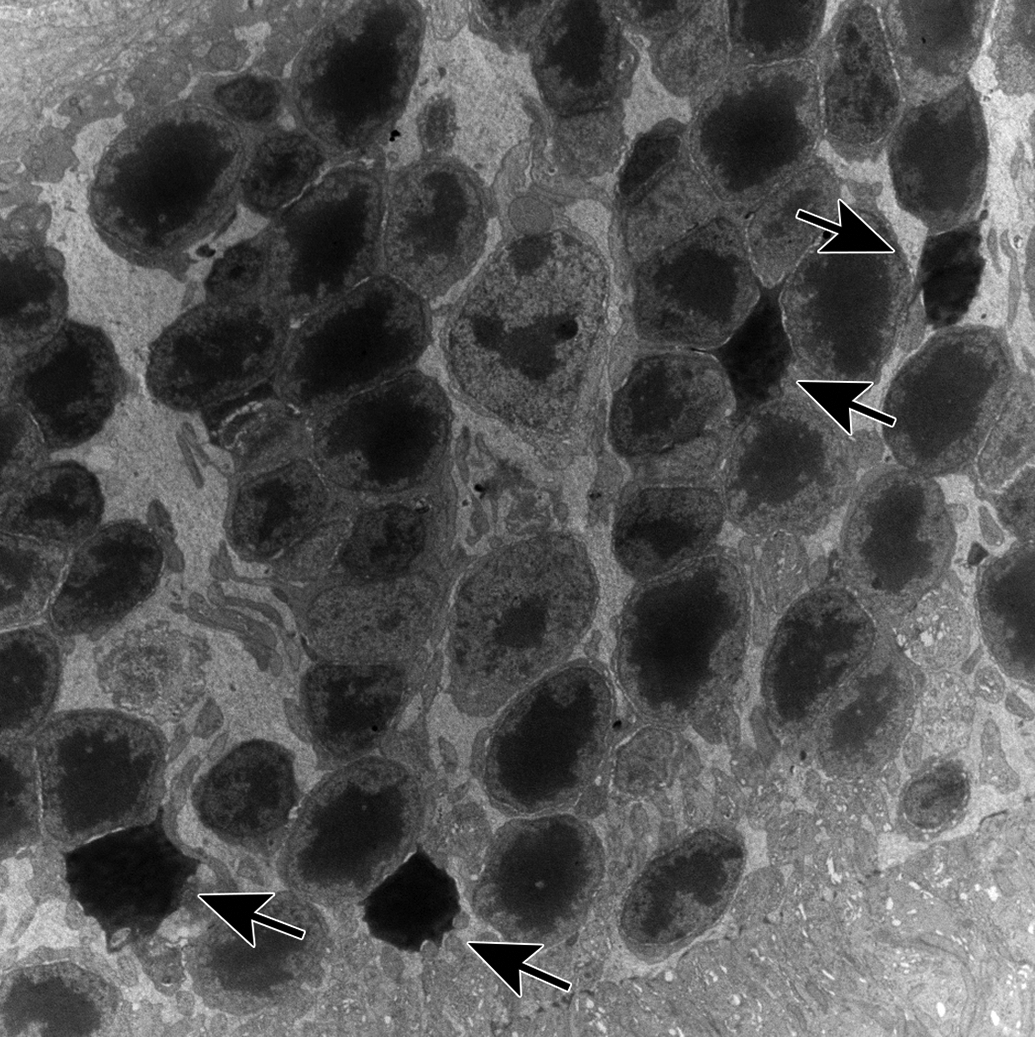
Electron microscopic appearance of photoreceptor cells in mice in the ZT22 UNC569 group on day 29. Chromatin-condensed nuclei increased in the outer nuclear layer (arrows). Original objective: ×3000. ZT indicates Zeitgeber time.
Summary of Ultrastructural Findings in the Retina of Mice Treated With UNC569.

Abbreviations: NE, not existed; RPE, retinal pigment epithelium; ZT, Zeitgeber time; -, no change.
a Increased multilamellar bodies in the RPE.
b Dilatation of the endoplasmic reticulum in the RPE.
c Chromatin-condensed nuclei in the outer nuclear layer.
Quantification of Retinoids
The concentrations of 11cRAL, atRAL, atROL, and 11cROL demonstrated no statistically significant alterations between the ZT5.5 and ZT22 UNC569 groups and each vehicle control group on day 29 (Figure 4). There was no statistically significant change on day 15, either (data not shown).
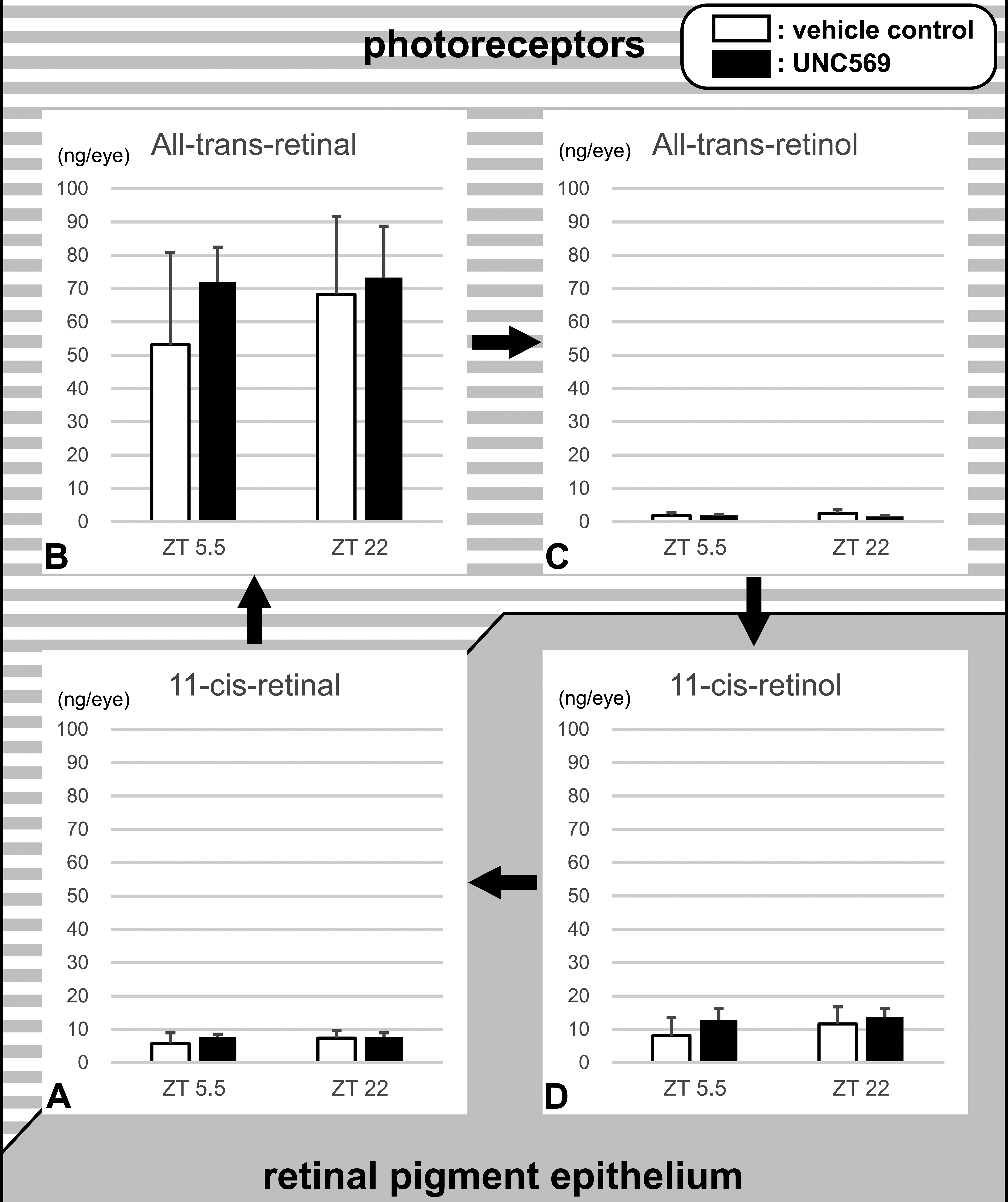
Retinoid concentrations in the retinal tissue samples of mice treated with UNC569 for 28 days. No statistically significant change was found in any retinoids. Background texture represents the localization of each retinoid. Data are expressed as mean ± SD (n = 5). SD indicates standard deviation.
Discussion
Our study revealed that the dosing time affected the severity of retinal changes induced by UNC569 in mice.
Ultrastructurally, membrane-bound multilamellar bodies in the RPE increased in both the ZT5.5 and ZT22 UNC569 groups after 14- and 28-day administration similar to our previous study. 14 These structures in the RPE were considered to be phagosomes and phagolysosomes, and a variety of appearances including electron-lucent areas and electron-dense cores were suggested to have reflected the different stages of the intracellular digestion process. Interestingly, dilatation of the ER in the RPE, indicative of the ER stress, and chromatin-condensed nuclei of the photoreceptor cells, and indicative of the early phase of apoptosis were observed only in the ZT22 UNC569 group after the 28-day administration. These differences between ZT22 and ZT5.5 could be related to the timing of UNC569-induced inhibition of physiological MerTK phosphorylation. The previous study demonstrated that the physiological MerTK phosphorylation continued from ZT2 to ZT5, and UNC569 inhibited the phosphorylation for 17 h after 100 mg/kg of dosing. 14 Because of this 17 h inhibition period, the inhibitory activity in the ZT22 UNC569 group was thought to have persisted up to ZT15 (ZT22 + 17 h = ZT39 = ZT15) and those in the ZT5.5 UNC569 groups was up to ZT22.5 (ZT5.5 + 17 h = ZT22.5). The former period included the duration from ZT2 to ZT5, the duration of physiological MerTK phosphorylation, whereas the latter period did not (Figure 5). In other words, more severe changes in the ZT22 UNC569 group as compared to the ZT5.5 UNC569 group could be due to the inhibitory activity from ZT2 to ZT5, the duration of physiological MerTK phosphorylation. The present study demonstrated that the dosing time affected the severity of UNC569-induced retinal toxicity.
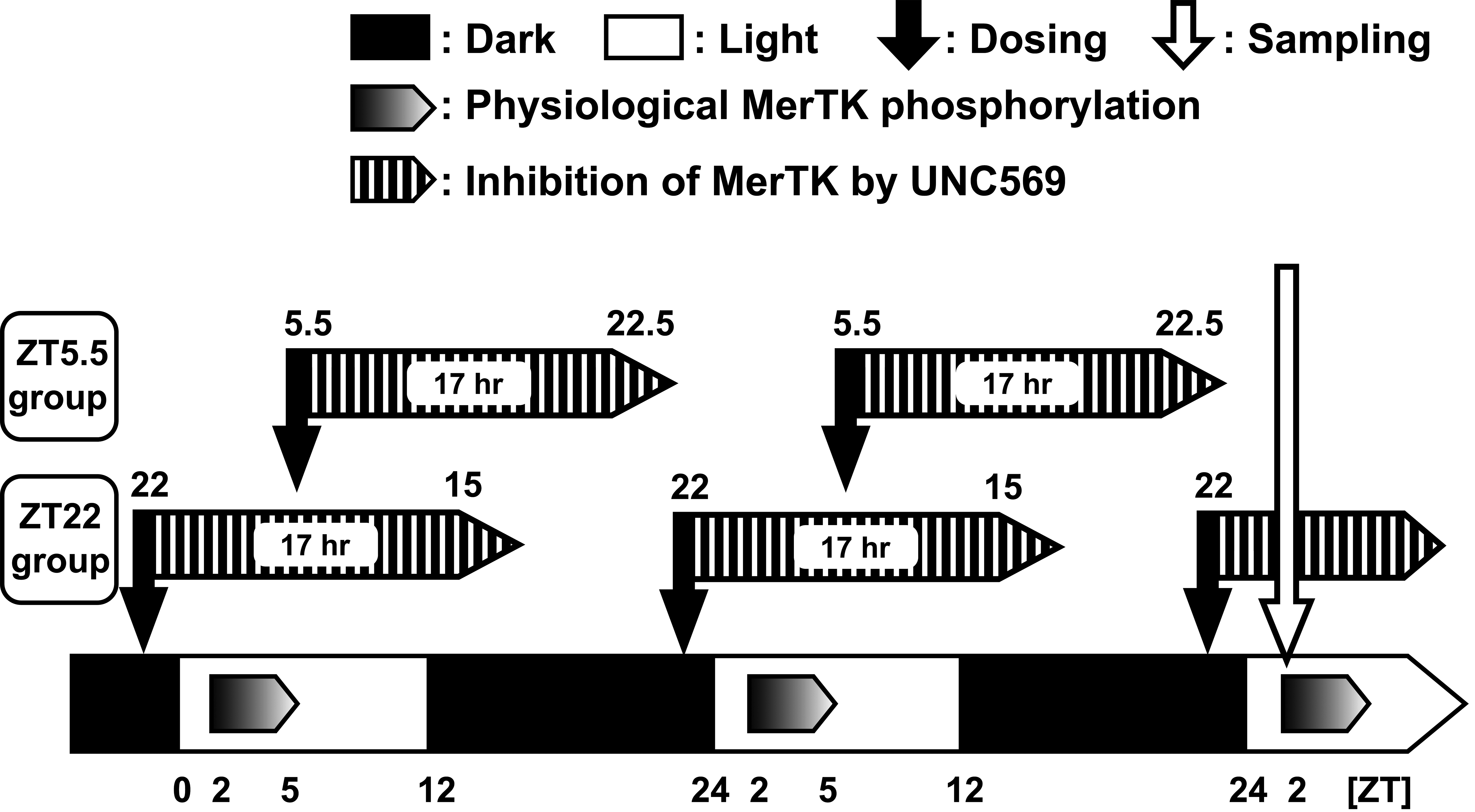
Timings between phosphorylation of MerTK under normal condition and inhibition of MerTK by UNC569 in the present study. MerTK indicates Mer proto-oncogene tyrosine kinase.
In general, increased phagosomes and phagolysosomes indicate 2 possible mechanisms: increased phagocytosis and suppressed degradation. In the previous study, we hypothesized that the increase could be attributed to an increase in phagocytosis owing to the sampling time of 24 h after the final dosing at ZT3.5, the apical localization as normal, and the absence of lipofuscin accumulation, which is a hallmark of long-term inhibition of POS digestion. However, in the present study, increased phagosomes and phagolysosomes were equally observed both in the ZT5.5 and ZT22 UNC569 groups necropsied at ZT2. Since ZT2 in the present study corresponded to 20.5 or 4 h after the final dosing in the ZT5.5 or ZT22 UNC569 groups, respectively, MerTK phosphorylation was thought to be inhibited in the ZT22 UNC569 group at sampling, whereas it was not in the ZT5.5 UNC569 group (Figure 5). Consequently, increased phagosomes and phagolysosomes, at least in the ZT22 UNC569 group, could be attributed to suppressed digestion of shed POS after engulfing and not to an increase in phagocytosis. On the contrary, we failed to explain the absence of an obvious difference in the amount of phagosomes and phagolysosomes or freshly engulfed shed POS between the ZT5.5 and ZT 22 UNC569 groups. This was inconsistent with the theory described above that MerTK phosphorylation at ZT2 could be restarted in the ZT5.5 UNC569 group. The duration of the inhibitory activity of UNC569 could be prolonged by the accumulation of UNC569 in plasma, owing to repeated administrations. Furthermore, the precise quantification of engulfed shed POS in the whole retina could be effective in comparing the amount of shed POS between the 2 timings. Further investigations are essential to verify the reasons for similar changes observed in the RPE in the ZT5.5 and ZT22 UNC569 groups.
To address the mechanism underlying UNC 569-induced injury in photoreceptor cells, which has no MerTK expression, we measured the amount of 11cRAL, atRAL, atROL, and 11cROL. The first step in vision transmission is the absorption of light by visual pigments (rhodopsin and cone opsin), the complex of the opsin protein, and the light-sensitive chromophore 11cRAL (Figure 4A). The capture of a photon results in the photoisomerization of 11cRAL to atRAL, which changes the conformation of the opsin protein, leading to the closure of cyclic GMP-gated cation channels and hyperpolarization of the photoreceptor cells (Figure 4B). Following photoisomerization and release from the opsin protein, atRAL is reduced to atROL (Figure 4C), transferred to the RPE, converted to 11cRAL via 11cROL (Figure 4D and A), transferred back to photoreceptor cells, and conjugated with an opsin protein to form a new and functional visual pigment. This metabolic pathway, involved in reactions in both the photoreceptor cells and the RPE, is called the visual cycle or the retinoid cycle and contributes to the maintenance of vision. 17,18 In addition, the delay in the reduction of atRAL, reactive vitamin A aldehyde, due to abnormalities in the visual cycle can damage photoreceptor cells. 19,20 In the present study, the amounts of atRAL and other components in visual cycle were not altered in the ZT22 UNC569 group compared to the control group. These results suggest that the UNC569-induced lesion of photoreceptor cells was not caused by abnormalities in the visual cycle.
In addition to the phagocytosis of shed POS and the maintenance of the visual cycle, another crucial role of the RPE is transepithelial transport for the survival of the photoreceptor cells. The RPE transports nutrients, ions, metabolites, and water between photoreceptors and choriocapillaris, and reduced transport can cause retinal degeneration. 10 The UNC569-induced lesion in photoreceptor cells might be caused by the disruption of transepithelial transport of the RPE, not by abnormalities in the visual cycle. This hypothesis is supported by the absence of any change in the measured visual cycle components as well as ER dilatation. Meanwhile, it is interesting that the supply of 11cRAL was not affected in the ZT22 UNC569 group despite apparent ultrastructural changes in the RPE, implying the significance of 11cRAL supply.
In conclusion, the timing of dosing influenced the severity of retinal toxicity induced by UNC569. UNC569 administered at ZT22 induced ER stress in the RPE and apoptosis of photoreceptor cells, in addition to suppressed digestion of shed POS in the RPE, whereas UNC569 administered at ZT5.5 only suppressed digestion. The present study suggests that the MerTK inhibitory activity of UNC569 during the physiological MerTK phosphorylation affected the severity of changes in the RPE, leading to the apoptosis of the photoreceptor cells, which lack the innate capacity to regenerate. Additionally, no changes in the visual cycle components and ER dilatation in the ZT22 group suggested that the disruption of transepithelial transport of the RPE caused the lesion of the photoreceptor cells. Our findings emphasize the importance of carefully planning experimental designs including the appropriate timing of dosing time for the investigators to evaluate drug-induced retinal toxicity potential and its clinical implications when candidates exhibit MerTK inhibitory activity.
Footnotes
Acknowledgments
We thank Yasuhiro Kinpara and Satoko Yahagi for performing the animal experiments, Yoshiko Ohshima for preparing the test substance, and Kunio Wada for synthesizing the test substance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
