Abstract
β-Secretase 1 (BACE1) represents an attractive target for the treatment of Alzheimer’s disease. In the course of development of a novel small molecule BACE1 inhibitor (AMG-8718), retinal thinning was observed in a 1-month toxicity study in the rat. To further understand the lesion, an investigational study was conducted whereby rats were treated daily with AMG-8718 for 1 month followed by a 2-month treatment-free phase. The earliest detectable change in the retina was an increase in autofluorescent granules in the retinal pigment epithelium (RPE) on day 5; however, there were no treatment-related light microscopic changes observed in the neuroretina and no changes observed by fundus autofluorescence or routine ophthalmoscopic examination after 28 days of dosing. Following 2 months of recovery, there was significant retinal thinning attributed to loss of photoreceptor nuclei from the outer nuclear layer. Electroretinographic changes were observed as early as day 14, before any microscopic evidence of photoreceptor loss. BACE1 knockout rats were generated and found to have normal retinal morphology indicating that the retinal toxicity induced by AMG-8718 was likely off-target. These results suggest that AMG-8718 impairs phagolysosomal function in the rat RPE, which leads to photoreceptor dysfunction and ultimately loss of photoreceptors.
Keywords
Introduction
Alzheimer’s disease (AD) is a fatal neurodegenerative disease characterized by accumulation of senile plaque deposits composed primarily of amyloid β (Aβ) peptides. Therapeutic strategies to reduce Aβ formation represent an attractive approach to prevent and treat AD. Aβ peptide is generated through sequential proteolytic processing initiated by the transmembrane aspartyl protease β-secretase 1, or β-site APP cleaving enzyme 1 (BACE1), from the amyloid β precursor protein (APP). BACE1 cleavage of APP is the rate-limiting step in Aβ production and therefore represents an attractive therapeutic target to reduce Aβ and plaque formation. Furthermore, a coding mutation in the APP gene that prevents BACE1 cleavage was recently found to be protective of AD and associated cognitive decline (Jonsson et al. 2012), which further supports the hypothesis that therapeutic inhibition of BACE1 may be protective of AD progression.
Development of small molecule inhibitors of BACE1 has been slow due to difficulties in engaging a large enzymatic active site while maintaining central nervous system penetration and a favorable pharmacokinetic and safety profile (Yuan et al. 2013). There are also concerns about potential on-target side effects of BACE1 inhibition due to the large number of BACE1 substrates that the enzyme is believed to cleave (Hemming et al. 2009; Kuhn et al. 2012). While numerous investigators have described BACE1 knockout (KO) mice as being viable, fertile, and with no gross or histological abnormalities (Luo et al. 2001, 2003; Roberds et al. 2001), others have reported effects on growth and mortality, adiposity, hypomyelination, schizophrenia-like behavior, seizures, and defects in cognition and motor coordination (Laird et al. 2005; Dominguez et al. 2005; Willem et al. 2006; Savonenko et al. 2008; Hu et al. 2010; Meakin et al. 2012; Cheret et al. 2013). The long-term effects of pharmacological inhibition of BACE1 in animals or humans have not been reported; however, late-stage clinical studies are underway.
Recently, an orally bioavailable small molecule inhibitor of BACE1 (LY2811376) was described as having robust Aβ lowering in humans (May et al. 2011), which further supports BACE1 as an attractive target to evaluate the role of Aβ in disease progression. However, development of LY2811376 was halted due to retinal toxicity in a 3-month rat toxicology study, which was characterized by an increase in autofluorescent granules (AFGs) in the retinal pigment epithelium (RPE) and photoreceptor degeneration (May et al. 2011). The retinal toxicity was considered an off-target effect not related to BACE 1 inhibition because similar retinal changes were observed in BACE1 KO mice treated with LY2811376, and the eyes of untreated BACE1 KO mice were normal. By contrast, Cai et al. (2012) have described increased AFG in the RPE, thinning of the neuroretina, reductions in phototopic electroretinography (ERG), and decreases in retinal capillary density in a different BACE1 KO mouse strain. The relevance of these nonclinical findings for patient safety is unclear.
In the course of drug development, we evaluated the small molecule BACE1 inhibitor AMG-8718 (Dineen et al. 2014) in a 1-month repeat-dose toxicity study in the Sprague-Dawley (SD) rat to support first-in-human trials. A dose-dependent increase in AFG in the RPE and hypertrophy of the RPE were observed after 1 month of treatment. Following a 2-month recovery period, the retinal change had progressed to retinal thinning, which was attributed to loss of photoreceptor nuclei in the outer nuclear layer (ONL). These observations prompted an investigational time-course study of the retinal findings in order to further characterize the morphological and functional effects on the rat retina and to inform appropriate clinical monitoring strategies. A BACE1 KO rat model was also developed and phenotyped in order to further evaluate the role of BACE1 in the retina.
Materials and Methods
Test Substance
AMG-8718 is a potent and orally efficacious BACE1 inhibitor (Dineen et al. 2014). In a biochemical enzyme assay, it inhibits BACE1 with a 50% inhibitory concentration (IC50) of 0.7 nM and lowers rat brain Aβ in vivo with an unbound plasma IC50 of 62 nM. AMG-8718 showed minimal selectivity over BACE2 (IC50 = 5.1 nM, 7×) and appreciable selectivity over cathepsin D (CatD; 2.3 μM, 3,285×).
Animals and Husbandry
Male SD rats (Rattus norvegicus, Crl:CD®(SD); 7–8 weeks of age) were cared for in accordance with the Guide for the Care and Use of Laboratory Animals, 8th edition. Animals were group-housed at an American Association for Accreditation of Laboratory Animal Care, international–accredited facility in nonsterile ventilated housing on nonaromatic bedding. All research protocols were approved by the Institutional Animal Care and Use Committee.
Animals had ad libitum access to pelleted feed (Lab Diet® Certified Rodent Diet #5002; PMI Nutrition International, Inc., Shoreview, MN) and water via an automatic watering system. Animals were maintained on a 12:12-hr light:dark cycle in rooms at 68°F to 79°F and 30% to 70% humidity range. To minimize exposure to fluorescent light, cages were placed on the bottom or middle rows of the cage rack during the study.
Study Design
Rats were acclimated for at least 5 days and randomized to treatment groups such that individual body weights were within 20% of the group mean body weight. Twenty rats were placed into 1 of the 3 cohorts of different study duration. Within each cohort, AMG-8718 was administered daily by oral gavage at 0, 10, 100, or 600 mg/kg (n = 5 rats/treatment group) in a solution of 2% hydroxypropyl methylcellulose, 1% Tween 80 in deionized water, pH 2.2 ± 0.2 adjusted with methanesulfonic acid and/or sodium hydroxide. Dose levels were selected based on retinal toxicity observed in a previous 1-month repeat dose rat toxicity study with a 2-month recovery and intended to provide sufficient exposure multiples above the rat BACE IC50 for brain Aβ lowering. Body weights were recorded prior to the first dose, once a week during the study, and on the day of necropsy (day 5, 29, or 85). Clinical observations were taken daily.
The first cohort was administered vehicle or AMG-8718 for 4 days and euthanized on day 5. Ophthalmic examinations were performed prior to the first dose and prior to necropsy (day 4 or 5). The second cohort was administered vehicle or AMG-8718 for 28 days and euthanized on day 29. Ophthalmic examinations were performed prior to the first dose, on day 13 or 14, and on day 26, 27, or 28 prior to necropsy on day 29. The third cohort was administered vehicle or AMG-8718 for 28 days followed by a 56-day treatment-free phase and euthanized on day 85. Ophthalmic examinations were performed prior to the first dose; prior to the end of treatment on day 26, 27, or 28; during the treatment-free phase on day 55, 56, or 57; and on day 82, 83, or 84 prior to necropsy on day 85. Ophthalmic examinations, as described subsequently, were staggered (e.g., days 26–28, 55–57, and 82–84) due to the need for multiple evaluations, overnight dark adaption, and accommodate the number of animals in the study.
In Vivo Ocular Assessment
A comprehensive morphometric and functional assessment of the retina was performed during the in-life portion of the study. This assessment included routine ophthalmic examination using a slit-lamp biomicroscope, fundus autofluorescence (FAF) imaging, optical coherence tomography (OCT), and ERG to evaluate retinal function. All animals had a comprehensive ophthalmic examination prior to the first dose to confirm the absence of preexisting anomalies that may confound data interpretation.
Ophthalmic examination using a slit-lamp biomicroscope was conducted on all animals prior to dosing. Animals designated for necropsy on day 28 were examined on day 26, and all animals designated for necropsy at the end of the treatment-free phase on day 85 were examined on day 82.
Ocular imaging was conducted using the Heidelberg Spectralis HRA + OCT (Heidelberg Engineering, Carlsbad, CA). Prior to imaging, pupils were dilated using topical tropicamide and phenylephrine, and the cornea was anesthetized with 0.5% proparacaine hydrochloride. Animals were sedated with inhaled isoflurane to effect, and the cornea was kept hydrated during imaging using ophthalmic saline eye wash.
With the goal of capturing an in vivo image of the granular autofluorescent deposits previously observed by fluorescence microscopy, FAF was used to evaluate the retina. Eyes were stimulated with blue light (465–490 nm) and imaged with a yellow-green (520–530 nm) band pass filter using a 55° wide angle noncontact lens with the optic nerve head as close to the center as possible (Luhmann et al. 2009; Secondi et al. 2012).
OCT was used to evaluate retinal thickness (Guo et al. 2010). Images were collected using a 30° lens at four quadrants approximately 15° from the optic nerve head from the right eye (at superior nasal, superior temporal, inferior nasal, and inferior temporal). Three points were placed on each OCT image of the retina (ImageJ; NIH Image): the vitreous/retina interface, the outer plexiform layer (OPL), and the apical layer of the RPE (Figure 1). Straight lines between 2 points and perpendicular to the retinal layers were measured and defined as the inner retinal thickness (vitreous to the OPL) and the outer retinal thickness (OPL to the apical layer of the RPE).
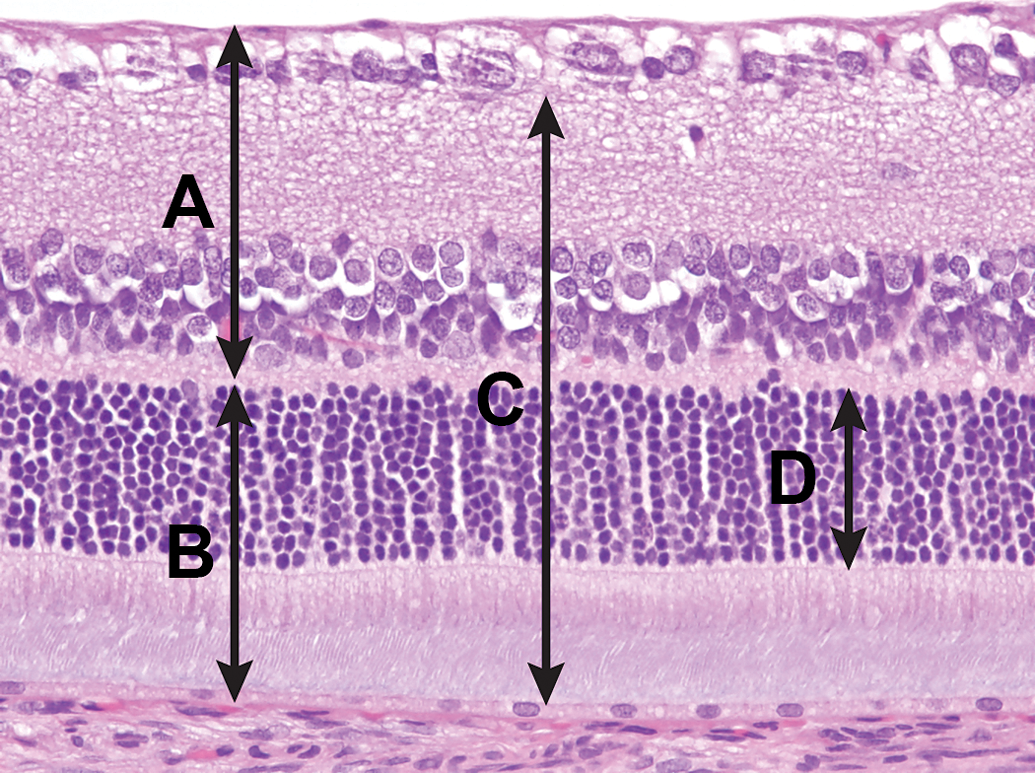
Illustration of thickness measurements and nuclei counts of the retina by OCT and light microscopy. Thickness of the inner (A) and outer retina (B) was obtained by OCT. Measurements of the neuroretina (C) and outer nuclear layer (D) were obtained from scanned light microscopic images. Nuclei counts conducted on 6 columns of the outer nuclear layer (D) were obtained using light microscopy. OCT = optical coherence tomography.
ERG was used to evaluate retinal function (Bayer et al. 2001). Animals were dark-adapted overnight, the eyes were dilated with topical 1% tropicamide and 10% phenylephrine, and the cornea was anesthetized with 0.5% proparacaine hydrochloride. Animals were sedated with a mixture of ketamine (60 mg/kg) and xylazine (5 mg/kg) prior to ERG evaluation. The active electrode was a gold loop embedded in a contact lens (Mayo Corp, Japan), the reference electrode was a stainless steel clip placed on the ear, and a ground electrode was a stainless steel needle placed in the nape of the neck. ERGs were recorded on both eyes using the Espion E2 recording system (Diagnosys, LLC, Lowell, MA). Under dark adaptation, A- and B-wave amplitudes were recorded over a 9 log range from 1 × 10−5 to 1,000 cd s/m2. After a 5-min light adaptation to a 30-cd/m2 rod suppressing white background light, phototopic (light adapted) A- and B-wave amplitudes were recorded at 10, 100, and 1,000 cd-s/m2. The A- and B-wave amplitudes are the principal measures of the ERG waveform and represent the electrical potential generated primarily by the photoreceptors (outer retina) and the bipolar and Müller cells (inner retina), respectively.
Toxicokinetics
Plasma (K2EDTA) was collected from the sublingual vein for drug concentration analysis from animals in cohort 1 on day 4 and from animals in cohort 2 on day 26 at predose, 0.5, 1.5, 5, 10, and 24 hr after dosing. Plasma samples were analyzed by liquid chromatography mass spectrometry using multiple reaction monitoring in positive ionization mode. The lower limit of quantitation in the assay was 5 ng/ml.
Postmortem Evaluation
Rats were euthanized with carbon dioxide following overnight fasting. Lung and mesenteric lymph nodes from all rats were preserved in 10% neutral-buffered formalin prior to dehydration and paraffin embedding. These tissues were collected due to a previous observation of AFG in these tissues in conjunction with AFG in the RPE. The eyes with optic nerve were marked with dye on the superior aspect of the globe for orientation and fixed using a solution of 3% glutaraldehyde in 0.1 M sodium phosphate (NaPO4) buffer at 20°C. After 40 to 60 min of fixation, the corneas were excised to allow greater penetration of the fixative. Following overnight fixation at 20°C, the lens was removed, and the eyes were retained in fixative for 48 hr prior to dehydration and paraffin embedding. After fixation, all tissues were embedded in paraffin, sectioned, stained with H&E, and examined by routine light microscopy and fluorescent microscopy. Histologic sections of eyes provided a superior/inferior plane of section. Fluorescent microscopy (Nikon Eclipse 90i, Nikon Corp., Tokyo, Japan) used a GFP HC HiSN Zero Shift Filter Set using a combination of excitation (450–490 nm), dichromatic mirror, and barrier filter (500–550 nm) for optimal visualization.
Retinal thickness in histologic sections (sectioned along the superior–inferior plane) was evaluated using linear measurements and by counting the number of photoreceptor nuclei in the ONL. For both methods, measurements were obtained at 2 locations approximately 750 μm from the center of the optic disk. The locations were free of processing artifact or blood vessels that would interfere with measurements. Retinal thickness was measured on both eyes from all rats using digital images scanned at 400× (Aperio Image Analysis System, Leica Microsystems, Buffalo Grove, IL). Linear measurements were perpendicular to the retina layers and included the ONL (between the outer and inner margins of the ONL) and the full retinal layer (defined here between the apical portion of the RPE and the outer margin of the ganglion cell layer; Figure 1). The outer margin of the ganglion cell layer was chosen in this study to avoid the inherent variability in the thickness and cellularity of this layer. Photoreceptor nuclear counts of both eyes from all rats on days 29 and 85 were obtained using light microscopy. Three noncontiguous columns of nuclei were counted at each location (n = 6 columns/eye). The mean thickness measurements for each treatment group were compared to the concurrent controls to determine the percentage change.
BACE1 KO Rats
BACE1 KO rats were created on the SD genetic background using CompoZr zinc finger nuclease (ZFN) technology by Sigma Advanced Genetic Engineering (SAGE, St Louis, MO). The ZFN pair was targeted to the first open reading frame of the rat BACE1 gene at the following binding positions, starting at genomic position 14825 (CTCTGACAGCTCAACATCctggtgGACACGGGCAGTAGTA; cleavage site in lowercase). The ZFN was microinjected into the pronucleus of 1-cell fertilized embryos from superovulated female SD rats and transferred to pseudopregnant foster mothers. A total of 35 pups from 4 litters were then screened for nonhomologous end joining-mediated mutations by designing polymerase chain reaction (PCR) primers that flank the ZFN targeting site, and the exact mutations were determined by sequencing. A founding heterozygous (F1) animal was identified that harbored a 49-bp deletion in exon 2 (14845–14893), which was predicted to generate a premature stop codon in exon 3 resulting in a truncated nonfunctional protein. This founding animal was subsequently backcrossed to wild type (WT) rats to ensure germline transmission. Heterozygous offspring were then bred to generate at least 4 to 5 rats/sex/genotype for phenotypic evaluation. Mutations were confirmed by PCR (forward primer tgacgatcaggtgacaggaa; reverse primer gtgtgaggtggggacaaaag). BACE1 protein expression in the brain was confirmed by Western blot using a rabbit monoclonal antibody #5606 to human BACE1 (Cell Signaling Technologies, Danvers, MA). Animals were evaluated for Aβ40 levels in the cerebral spinal fluid (CSF) and brain using a MesoScale Discovery immunoassay with a rodent N-terminal specific antibody for Aβ40 as the capture and a ruthenium-labeled antibody specific for the C-terminal region of Aβ40 as the detector (Dineen et al. 2014). Aβ40 concentrations were analyzed by one-way analysis of variance (ANOVA) followed by Dunnett’s multiple comparison analysis.
WT and BACE1 homozygous KO rats were evaluated for genotype-related changes in body weight and organ weights (liver, spleen, testes, heart, kidney, and brain) and routine clinical chemistry, hematology, and urinalysis parameters. Light microscopic evaluation of a full set of tissues was performed as described previously. Retinal thickness in histologic sections of both eyes from WT and KO animals was evaluated using linear measurements as described previously. Due to the unknown function of BACE1 in the retina, the photoreceptor layer, inner nuclear layer, and inner plexiform layer were measured in addition to the full neuroretina and ONL to provide a broader evaluation of retinal thickness.
Statistical Analysis
All data were analyzed using SAS v9.2 in the Windows VISTA Professional environment. Statistical significances were evaluated at p = .05 level.
Retinal thickness measurements from OCT were analyzed by repeated measure analysis of covariance (RMANCOVA) models (day −1 as the baseline covariate) assuming unstructured variance covariance matrix for repeated measures on an OCT image to assess the effect of treatment, the effect of OCT quadrants, their interactions and the time dependence of such effects. Electroretinogram parameters (maximum amplitudes) were analyzed by repeated measure analysis of variance (RMANOVA) models assuming unstructured variance–covariance matrix for repeated measures on an eye to assess the effect of treatment, the effect of eyes (right or left), their interactions, and the time dependence of such effects. If the model showed statistically significant interaction between time and treatment or between time and treatment-associated interactions, the RMANCOVA or RMANOVA was reduced to mixed effect analysis of variance–covariance models where the animal is considered as a random blocking variable to analyze the data at each time point.
Retinal morphometry parameters were analyzed by mixed effect ANOVA models at each necropsy time point (days 5, 29, and 85) to evaluate the treatment effect, location effect (left superior, left inferior, right superior, and right inferior), and the interaction between them. In each model, the animal is considered as a random blocking variable.
In all the analyses, Kenward and Roger’s method for calculation of degrees of freedom was used for the inferential test statistics. When there is a statistically significant treatment effect or a statistically significant interaction between treatment effect and other factors, pairwise comparisons among treatment groups were conducted at each level of the other factor.
Results
Daily dosing of AMG-8718 produced the expected increases in exposure in male rats, which were approximately 1, 7, and 21 times the rat BACE IC50 for Aβ lowering in the brain at 10, 100, and 600 mg/kg, respectively, based on exposures on day 26 (Table 1). There were no treatment-related effects observed by slit-lamp ophthalmoscopy or FAF during the treatment and recovery phases. There were also no effects on mortality, clinical signs, or body weight during the treatment and recovery phases.
Mean (standard deviation) toxicokinetic parameters in rats treated with AMG-8718 for 4 and 26 days.
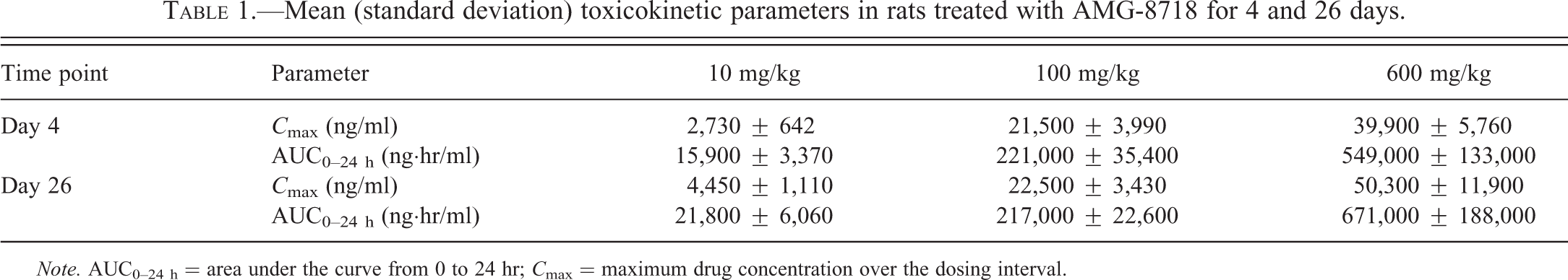
Note. AUC0–24 h = area under the curve from 0 to 24 hr; C max = maximum drug concentration over the dosing interval.
AMG-8718 Impairs Retinal Function
ERG was used to evaluate retinal function and characterize the relationship of ERG responses to morphologic changes in the retina. Electroretinograms were evaluated during the dosing phase (day 5 for cohort 1, days 14/15 for cohort 2, and days 27/28 for both cohorts 2 and 3) and during recovery phases (days 55/56 and days 83/84 for cohort 3). There were treatment-related decreases in dark-adapted A- and B-wave amplitude beginning on day 14/15 in the high-dose group only (Figure 2), which is consistent with a decrease in function of bipolar cells and rod photoreceptors. The effect persisted with a similar magnitude of change at the end of the dosing phase (day 27/28) and throughout the 2-month recovery phase indicating a lack of reversibility. Decreases in light-adapted B-wave amplitude, but not A-wave amplitude, were also observed (data not shown). There were no treatment-related changes in ERG parameters on day 5 or at the low- and mid-dose groups at any time point.
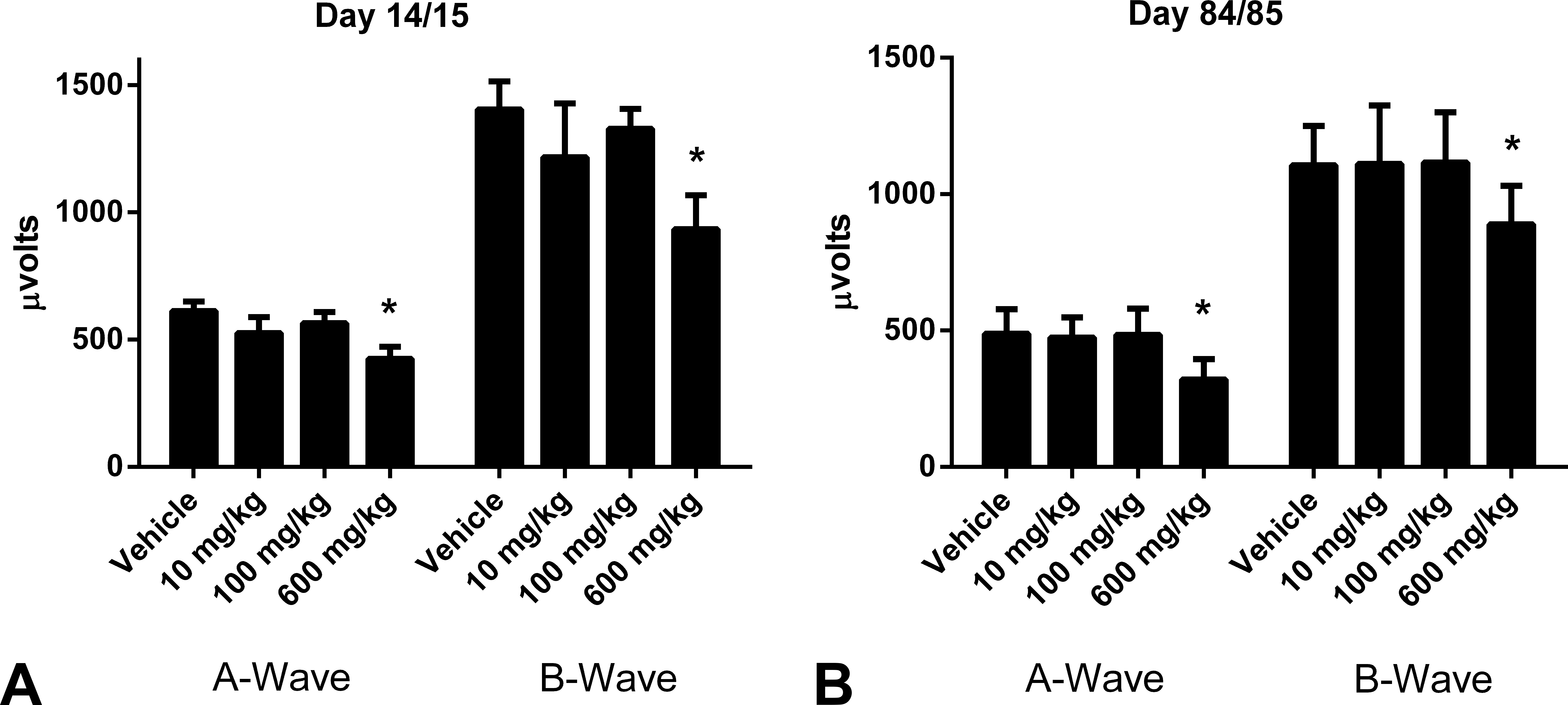
Evaluation of functional changes in the retina in rats treated with AMG-8718. Data expressed as mean ± standard deviation of dark-adapted maximum A- and B-wave amplitude on days 14/15 (A, n = 5/group) and 84/85 (B, n = 5/group). Similar results were observed on day 27/28 (not shown). Asterisk indicates statistically significant relative to time-matched vehicle control (p < .05).
AMG-8718 Induces Morphologic Changes in the RPE
The retinas were examined by light and fluorescence microscopy to detect and characterize morphologic changes in the RPE. When evaluated using fluorescence microscopy, there was a clear dose- and time-dependent increase in the amount of AFG in the RPE observed as early as day 5 in the mid- and high-dose group (Table 2; Figure 3). Although the AFG were best detected using the GFP filter set, the material was autofluorescent with multiple filters covering a broad spectrum. The increase in AFG was diffuse and uniform along the entire length of the retina with no evidence of regional differences. By light microscopy, there was a dose-dependent increase in the incidence of mild RPE hypertrophy on day 29. RPE hypertrophy correlated with a moderate increase in AFG and was attributed to the presence of the increased intracellular material. The RPE was otherwise normal, with no evidence of degeneration, necrosis, or vacuolation, and not associated with changes in any other layers of the retina. At the end of the recovery period, the severity of AFG in the RPE was reduced and RPE hypertrophy was not observed, indicating partial recovery of the morphological changes in the RPE.
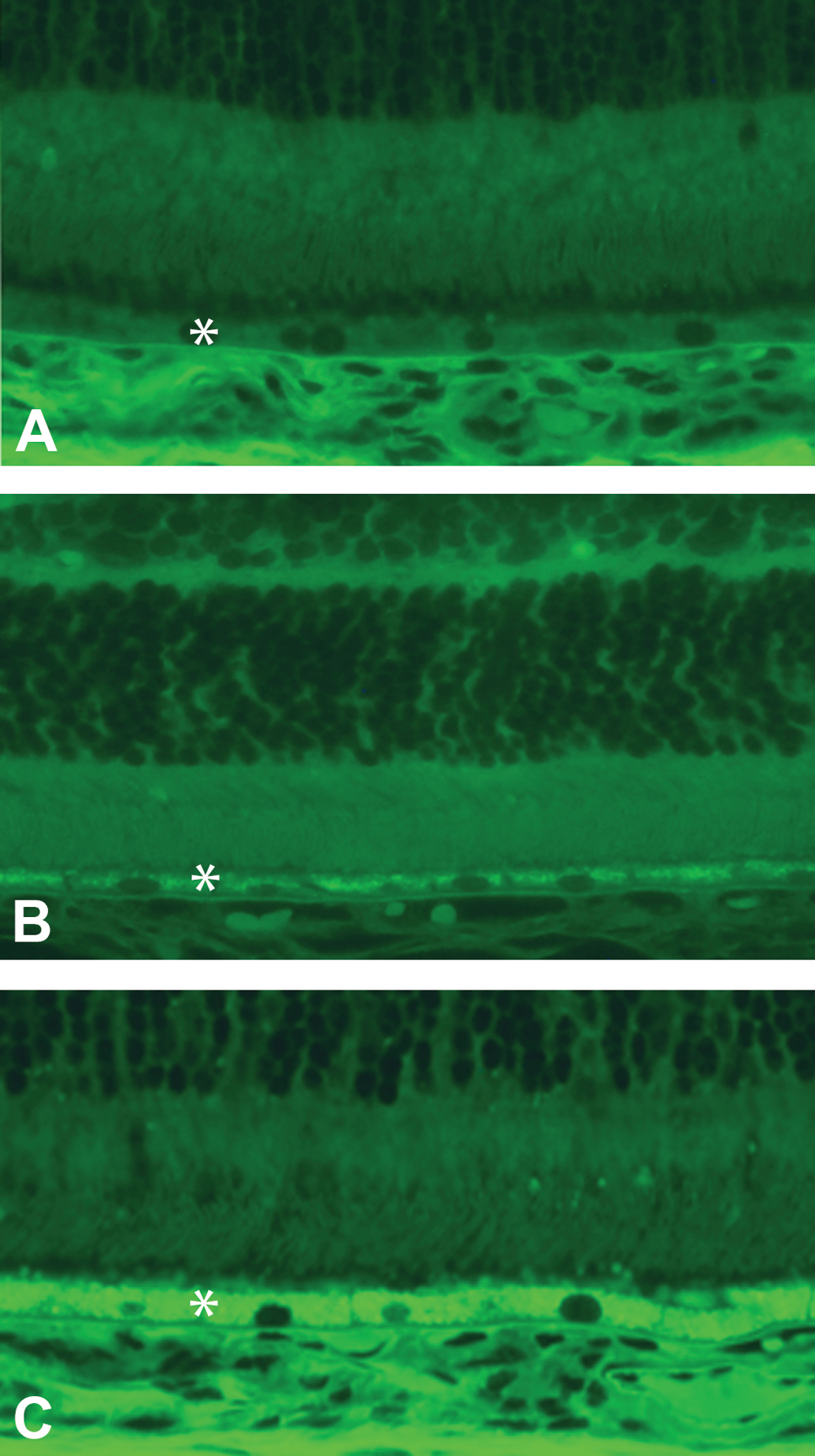
Fluorescence microscopy evaluation of the retina of rats treated with AMG-8718. Representative images from rats treated with vehicle (A) or 600 mg/kg of AMG-8718 (B and C). Asterisk indicates retinal pigment epithelial (RPE) layer. AMG-8718-treated rats have increased autofluorescent granules (AFGs) in the retinal pigment epithelium (mild in B; moderate in C). Moderate increase in AFG correlates with hypertrophy of RPE.
Light and fluorescence microscopy changes in the rat after 28 days of dosing of AMG-8718 followed by a 56-day treatment-free phase.
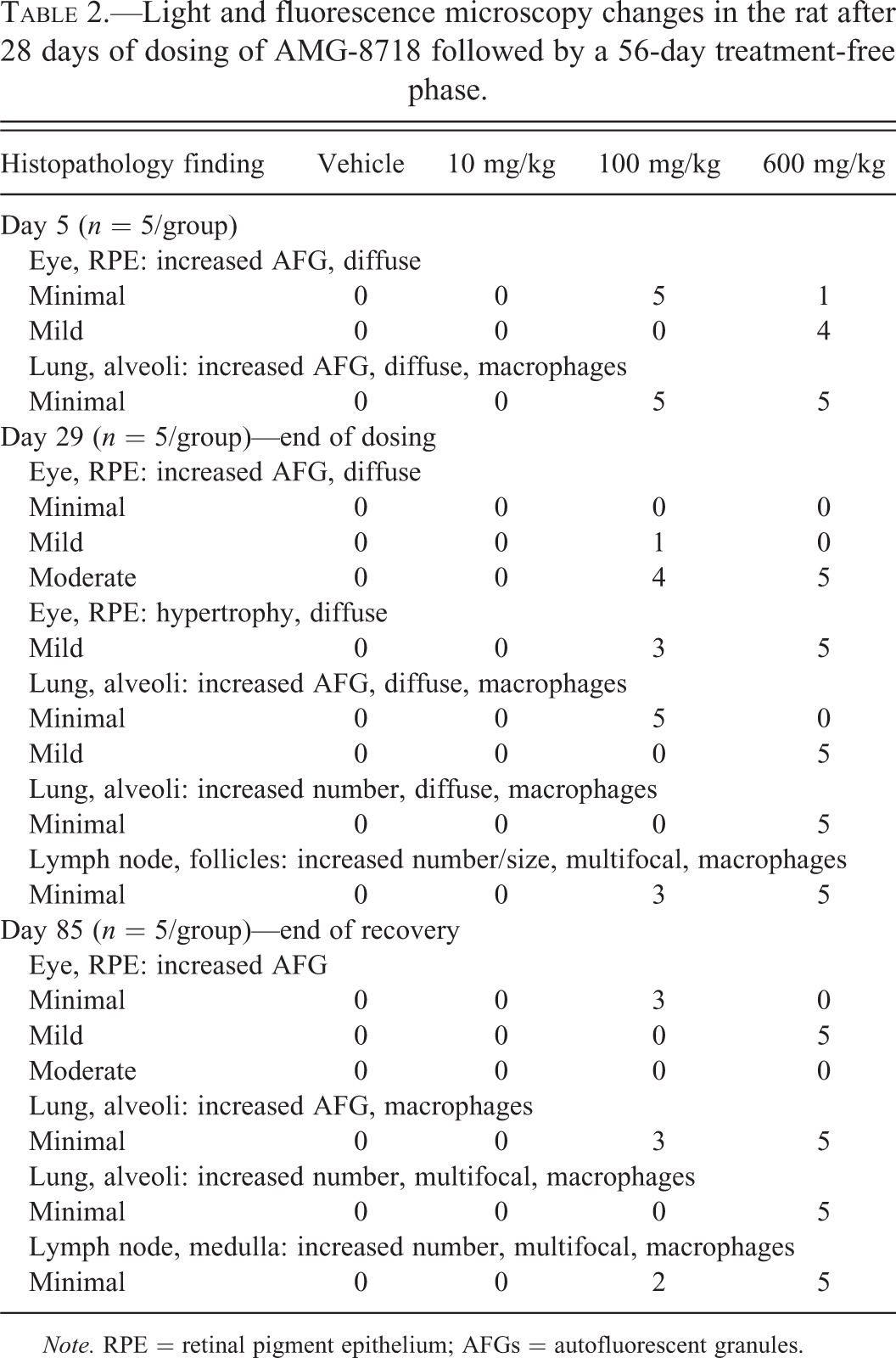
Note. RPE = retinal pigment epithelium; AFGs = autofluorescent granules.
AMG-8718 Induces Retinal Thinning Characterized by Loss of Photoreceptor Nuclei in the ONL
In the previous 1-month rat toxicology study, a diffuse and uniform decrease in retinal thickness in recovery-phase rats was observed by light microscopy. To determine whether this decrease could be detected noninvasively, OCT measurements of retinal thickness were obtained during the dosing (days 4, 13, and 26) and recovery phases (days 57 and 82) in this study. At the high dose, a treatment-related statistically significant decrease in outer retina thickness was observed on days 57 (−10%) and 82 (−13%; Table 3; Figure 4). A statistically significant decrease in outer retina thickness on day 29 in the high-dose group was of uncertain relationship to treatment because the magnitude of change was small and below the threshold of resolution for OCT measurement. A statistical comparison of retinal thickness measurements across the four quadrants sampled by OCT indicated no significant regional differences.
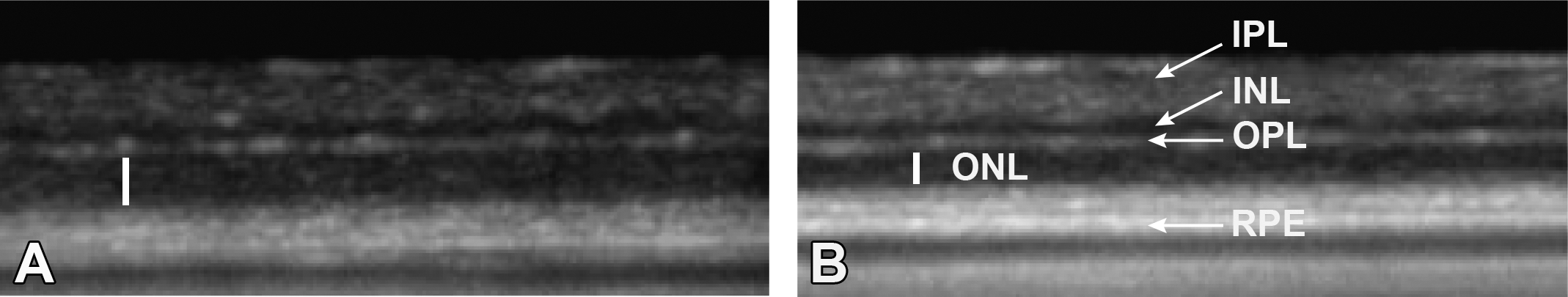
Optical coherence tomography evaluation of the retina of rats treated with AMG-8718. Representative images from rats treated with vehicle (A) or 600 mg/kg (B) of AMG-8718 for 28 days followed by a 56-day recovery phase. Vertical bar indicates outer nuclear layer (ONL), which is reduced in thickness in AMG-8718-treated rats. INL = inner nuclear layer; IPL = inner plexiform layer; OPL = outer plexiform layer; RPE = retinal pigment epithelium.
Mean (standard deviation) retinal thickness measurements and outer nuclear layer cell counts in the rat after 28 days of dosing of AMG-8718 followed by a 56-day treatment-free phase.
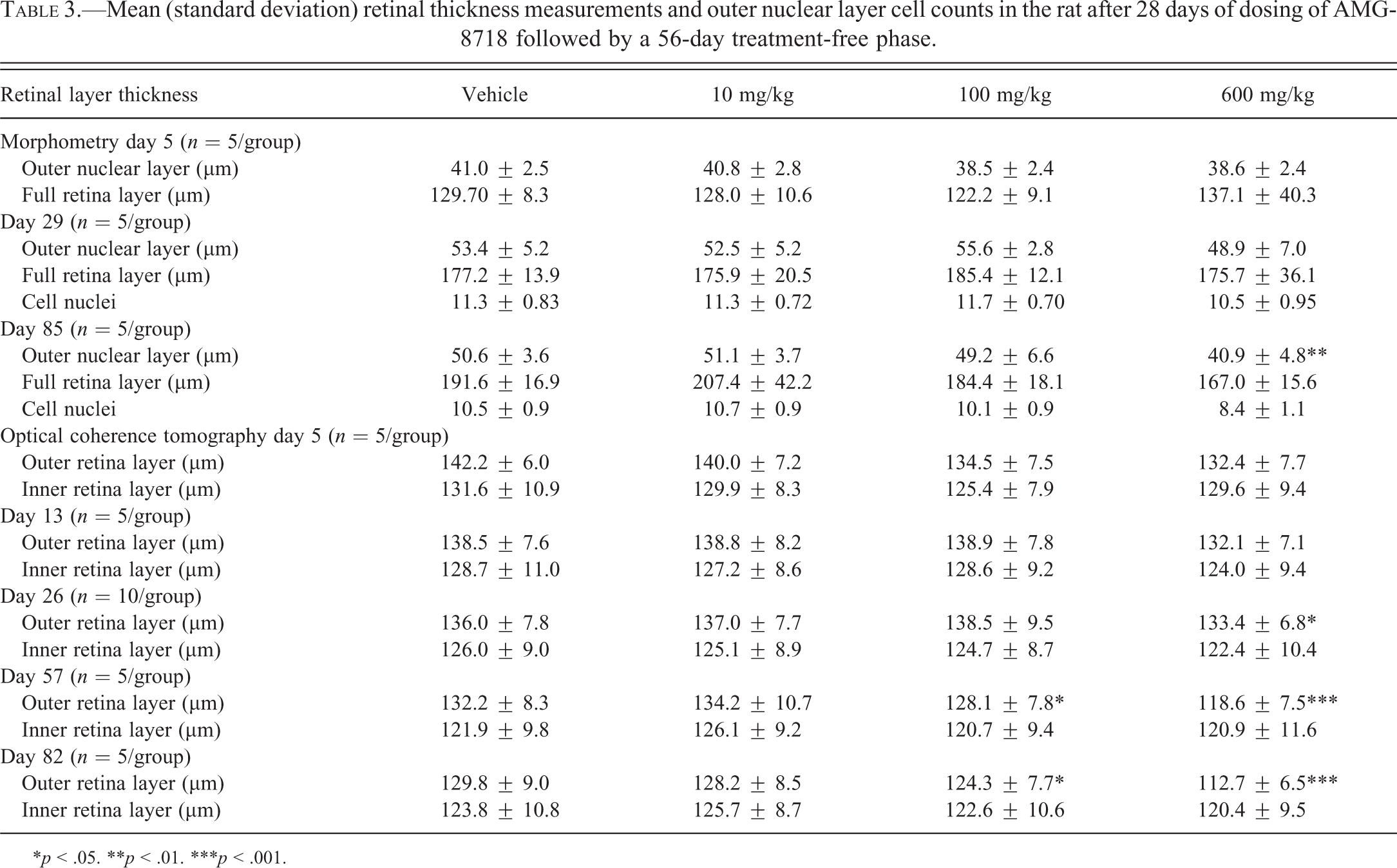
*p < .05. **p < .01. ***p < .001.
Similar to the previous study, a decrease in retinal thickness in recovery-phase rats at 600 mg/kg was observed by light microscopy (Figure 5). The decrease in retinal thickness was diffuse and uniform across the full length of the retina, consistent with the changes observed by fluorescent microscopy and OCT. There were no other treatment-related light microscopic changes in the eyes at any dose. To further characterize the change, the thickness of the full retina and the ONL were obtained on histological sections (Table 3). Nuclei count of the ONL was also assessed to rule out the possibility of fixation artifacts. There was a treatment-related decrease in the mean full retina (−12.9%) and ONL thickness (−19.1%) in the high-dose, recovery-phase group only. The decrease in ONL thickness was consistent with a decrease in nuclei count (−20.0%) within the ONL. Treatment-related decreases in mean retinal thickness measurements were not observed at lower dose levels at the end of recovery or on day 5 or 29 at any dose level. There was a minor decrease in nuclei count in the ONL (−7.1%) in the high-dose group on day 29; however, the relationship to treatment was uncertain because the change was small and there was no correlative change in ONL thickness.
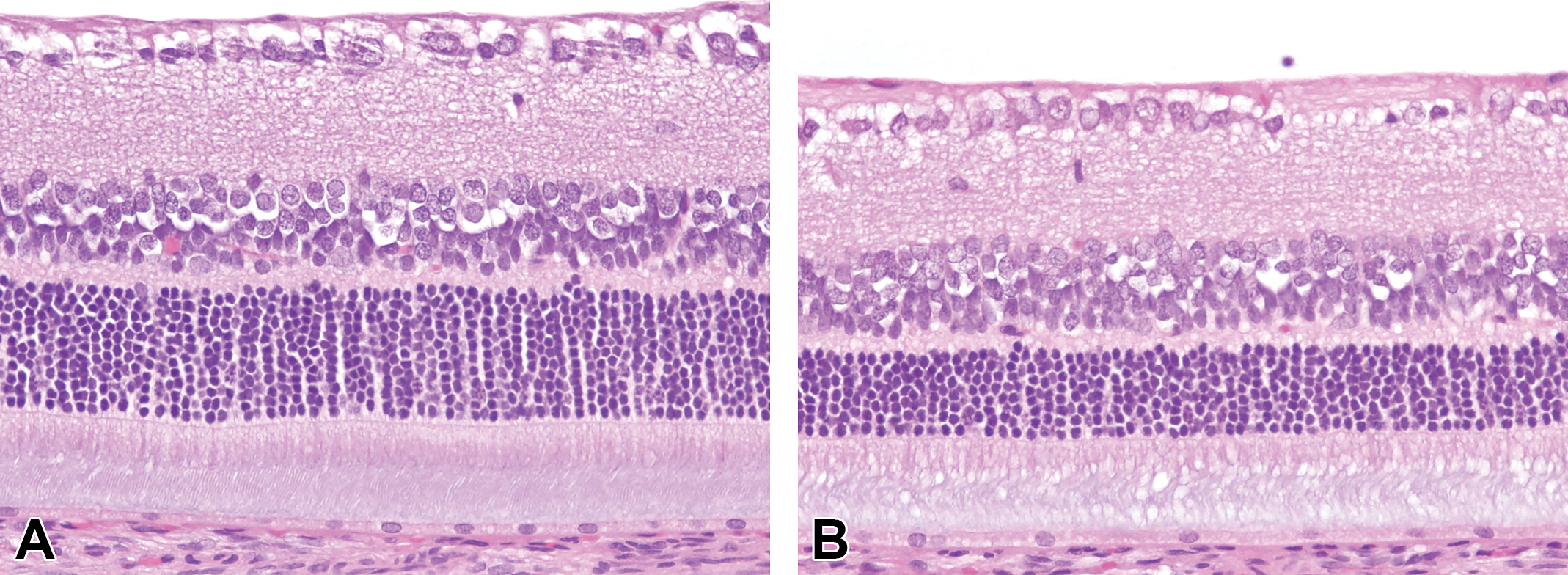
Light microscopic evaluation of the retina of rats treated with AMG-8718. Representative images from rats treated with vehicle (A) or 600 mg/kg (B) of AMG-8718 for 28 days followed by a 56-day recovery phase. AMG-8718-treated rats have reduced thickness of the outer nuclear layer, indicated by the arrow.
AMG-8718 Induces Morphologic Changes in the Lung and Lymph Node
Morphologic changes in macrophages of the lung and lymph node were similar to the changes noted in the RPE (Table 2). In the lung, morphologic changes included a diffuse increase in alveolar macrophages, many of which had an increase in intracytoplasmic AFG. Increased AFG in alveolar macrophages were observed at ≥100 mg/kg on days 5 and 29 of the dosing phase. At the high dose, the severity of this change was time dependent with an increase from days 5 to 29. The diffuse increase in alveolar macrophages was noted at 600 mg/kg on day 29 only. There were no other morphologic changes (e.g., vacuolation or pigment) in the alveolar macrophages. At the end of the recovery phase, the diffuse increase in alveolar macrophages observed in the dosing-phase animals was replaced by discreet aggregates of alveolar macrophages, while the increased AFG was reduced relative to day 29, suggesting partial recovery.
In the lymph node, there was a dose-dependent increase in the size and number of macrophages in lymphoid follicles at ≥100 mg/kg on day 29 only. This change was characterized by large macrophages filled with bright eosinophilic phagocytized debris. Although similar macrophages were occasionally noted in control animal lymph nodes, the affected treated animal lymph nodes had an increased number of them. These cells in both controls and treated animals were intensely autofluorescent, and there was no evidence of vacuolation. At the end of the recovery phase, there were multiple foci of macrophage aggregates in the medulla, immediately subjacent to the lymphoid follicles, which may suggest partial recovery.
BACE1 KO Rats Have Normal Retinal Morphology
BACE1 KO rats were constructed using ZFNs targeted to exon 2, which led to a founding heterozygote animal with a mutation predicted to generate a premature stop codon in exon 3 and a truncated nonfunctional protein. To confirm functional deletion of the BACE1 gene, Aβ levels were evaluated in the CSF and brain of WT, heterozygous, and homozygous KO rats. Consistent with a role of BACE1 in Aβ formation, levels of both CSF and brain Aβ were significantly decreased in homozygous KO animals (Figure 6). Western blots of brain samples also confirmed lack of detectable BACE1 protein in homozygous KO rats (data not shown). No significant difference in CSF and brain Aβ was observed in heterozygous animals compared to WT littermates.
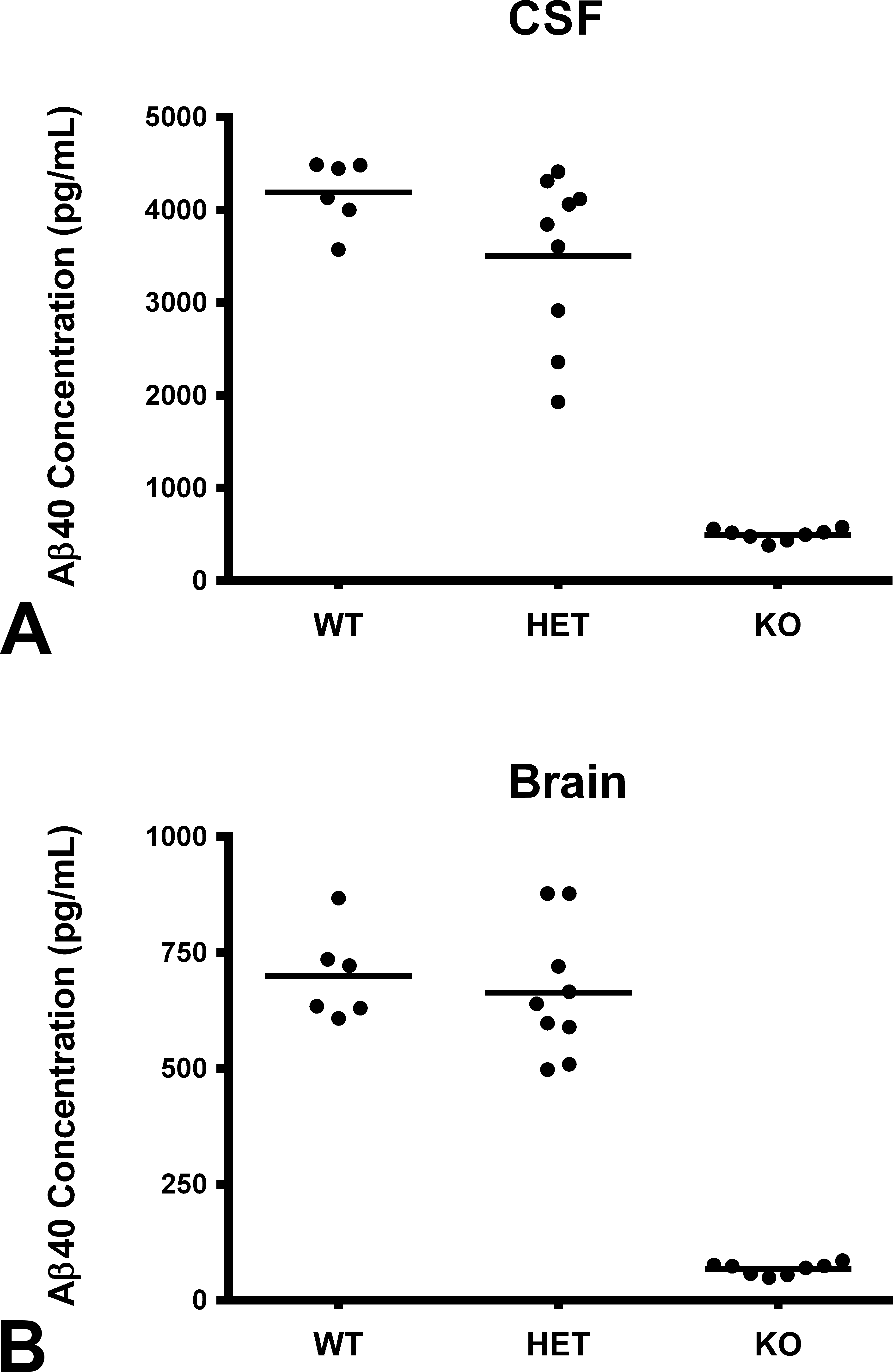
Aβ40 measurements in CSF and brain. Wild-type (WT, n = 6), heterozygous (HET, n = 9), and homozygous (KO, n = 8) knockout rats (aggregated across sex) were evaluated for Aβ40 levels in the CSF (A) and brain (B) as described in the Methods section. Levels of Aβ40 levels in the CSF and brain of KO rats were significantly (p < .0001) lower than WT and HET rats. Horizontal line represents mean value. CSF = cerebral spinal fluid; KO = knockout.
Body weights were 6% lower in male and female BACE1 KO rats; however, there were no other genotype-related changes in clinical chemistry, hematology, urine chemistry, organ weights, or histopathology (panel of 30 tissues). Retinal morphology in KO rats was within normal limits of age-matched controls (Figure 7). In addition, there were no statistically significant genotype-related differences observed in retinal thickness within each sex or combined, and the level of AFG in the RPE was also comparable across sexes and genotypes. There were also no genotype-related changes in the lung and lymph node.
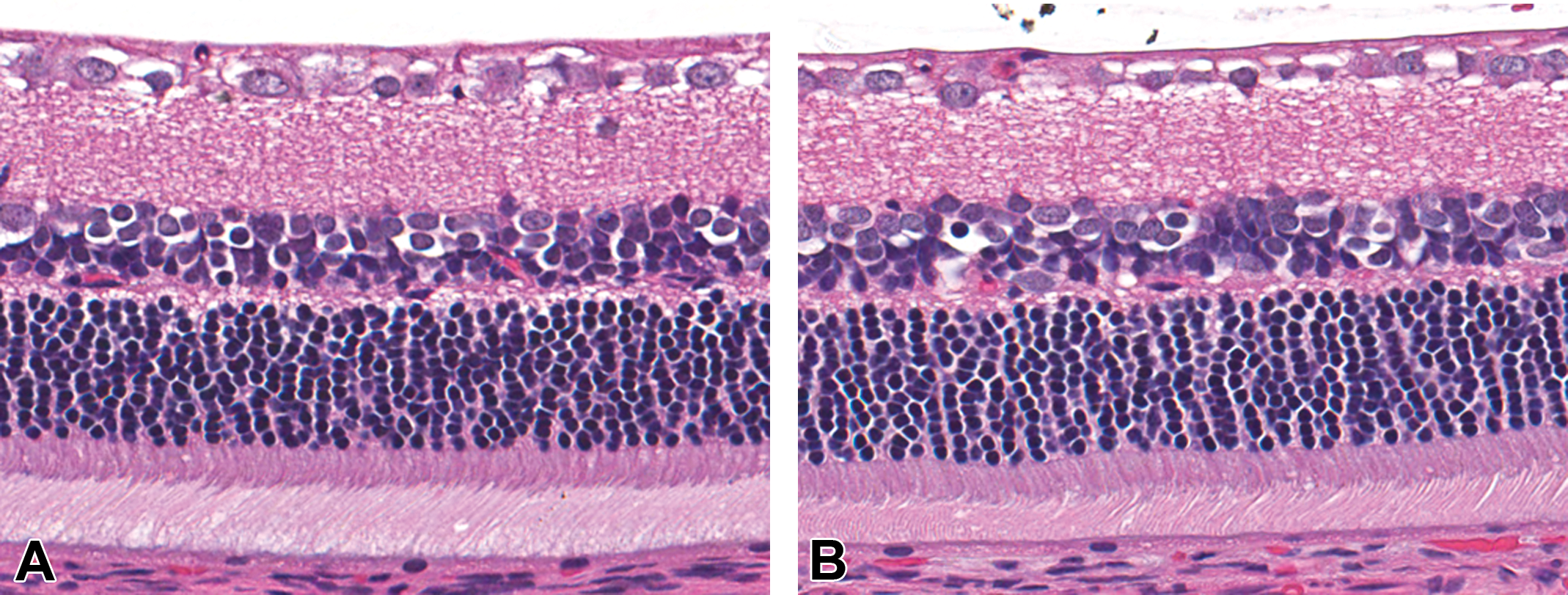
Light microscopic evaluation of the retina of wild-type and homozygous BACE1 knockout rats. Representative images from wild-type (A) and knockout rats (B). Retinal morphology in knockout rats was within normal limits of age-matched controls. BACE1 = β-site amyloid β precursor protein cleaving enzyme 1.
Discussion
Our studies further characterize the morphologic features and functional consequences of a reproducible retinal toxicity induced by a novel small molecule BACE inhibitor. After 1 month of daily dosing and a 2-month treatment-free period, a significant decrease in neuroretinal thickness was observed in the high-dose recovery-phase animals. The decrease in neuroretinal thickness was characterized as a loss of photoreceptor nuclei from the ONL. The time-course investigation revealed that the effects of AMG-8718 on the retina were first manifested by a dose-dependent increase in AFG in the RPE as early as day 5, which progressed with time (Table 2). Parallel changes were also observed in macrophages within the lung and lymph node follicles. Earlier detection of increased AFG in the RPE could not be reliably detected based on results of similar compounds tested in the rat (data not shown). Increases in AFG in the choroid plexus were noted in the original 1-month study with AMG-8718 but were not reevaluated here. In addition, macrophages were enlarged and more numerous in the lung, lymph node, gut-associated lymphoid tissue, and spleen when evaluated in the original 1-month study. These changes suggest that other phagocytically active cell types are targeted by AMG-8718, suggesting a disruption in the phagolysosomal process.
A critical function of the RPE is to phagocytose and digest photoreceptor outer segments (POSs) that are shed as part of the normal renewal of the light-sensitive component of the photoreceptors. Disruption of the phagolysosomal process is thought to lead to intracellular accumulation of debris originating from POS, typically observed as autofluorescent lipofuscin granules in the RPE of aged humans and animals. Disruption of phagolysosomal function in these studies is supported by the observation of increased AFG in multiple tissues. Moreover, ultrastructural characterization of the RPE from rats in a separate study with a structurally related compound showed an increase in phagolysosomes containing undigested POS material with the characteristic appearance of lipofuscin (data not shown). Similar results with a distinct BACE inhibitor LY2811376 were also reported by May et al. (2011); however, unlike LY2811376, AMG-8718 did not cause AFG in the neurons and glial cells of the brain or cause neuronal degeneration. This may be due to the shorter duration of exposure with AMG-8718 (1 vs. 3 months) or a unique selectivity profile. Hypertrophy of the RPE, which correlated with the severity of AFG accumulation, was attributed to the presence of increased intracellular material. Direct inhibition of phagocytosis is unlikely to be causal, given the normal appearance of the POS at these early time points when AFG were first observed. These effects collectively suggest disruption of the phagolysosomal pathway in the RPE, which leads to accumulation of undigested POS material in the RPE.
Given the role of the RPE in sustaining photoreceptor cells (Kevany and Palczewski 2010), it is hypothesized that inhibition of the phagolysosomal pathway and ensuing POS accumulation and incomplete degradation may negatively impact RPE function, and thus the ability of the RPE to sustain proper photoreceptor function and viability. Multiple lines of evidence support this (Sparrow and Boulton 2005). The decrease in retinal function observed by ERG after 14 days of treatment with AMG-8718, well before a detectable loss of photoreceptor nuclei from the ONL, is further evidence. The decrease in photoreceptor function appears to affect both rods and cones since both phototopic (light adapted) and scotopic (dark adapted) ERG signals were decreased. Given the relative paucity of cone relative to rod photoreceptors in rodents, light microscopy would not be expected to distinguish loss of cone from rod photoreceptors. While direct effects on the neuroretina cannot be discounted, there was no microscopic evidence to support this hypothesis. While the AFG accumulation showed evidence of partial reversibility, there was still considerable AFG in the RPE after the 2-month recovery period. This likely resulted in a continued decline in photoreceptor function and viability, which was evident by the sustained decrease in ERG amplitudes throughout the dosing and recovery periods, and the significant thinning of the retina observed at the end of the recovery period. It is unclear whether drug withdrawal at the earliest sign of increased AFG in the RPE would permit full reversibility and avoid impairment and/or degeneration to the photoreceptor layer. From this perspective, it was disappointing that fundus autofluorescent imaging was unable to detect a treatment-related increase in autofluorescence at any time point. Fundus imaging is routinely applied to the clinical monitoring of RPE toxicity for aminoquinolone antimalarials, although progression of the lesion upon cessation of therapy has been documented (Michaelides et al. 2011).
A previous report of retinal toxicity in the rat following chronic administration of a small molecule BACE inhibitor LY2811376, in addition to retinal abnormalities reported in a BACE1 KO mouse strain (Cai et al. 2012), has raised concerns that pharmacological inhibition of BACE1 may pose a risk of retinal toxicity in the clinic. The reports of numerous BACE1 proteolytic substrates (Hemming et al. 2009) and expression of BACE1 in the retina (Cai et al. 2012) have also increased uncertainty about the role of BACE1 in the retina. However, the results here and presented elsewhere collectively support that BACE1 deletion is not associated with retinal abnormalities in rodents and therefore the effects of AMG-8718 are likely off-target. To better compare the effects observed with BACE inhibitors in rat toxicology studies reported here and elsewhere (May et al. 2011) with genetic models of BACE1 deficiency, we developed and phenotyped a homozygous BACE1 KO rat on the SD genetic background. A significant decrease in brain and CSF Aβ levels in homozygous, but not WT or BACE1 heterozygous animals, confirmed functional deletion of BACE1 protein, consistent with effects in BACE1 KO mice (Luo et al. 2001; Roberds et al. 2001). The retina from BACE1 KO rats was microscopically normal and had a comparable level of AFG in the RPE relative to WT rats. A thorough morphometric evaluation of retinal thickness also demonstrated no significant differences between WT and KO rats. The lack of a detectable phenotype in the eyes of BACE1 KO rats is consistent with results of 2 independently generated BACE1 KO mouse models (Luo et al. 2001, 2003; May et al. 2011). May et al. (2011) also demonstrated that the retinal toxicity could be recapitulated in the BACE1 KO mice treated for 9 weeks with LY2811376, further arguing that BACE inhibitor–induced toxicity is not mediated by BACE1. By contrast, Cai et al. (2012) reported an increase in AFG in the RPE of BACE1 KO mice, consistent with BACE inhibitor–treated rats but also reported thinning of the outer and inner nuclear layers, atrophy of the RPE, neuronal apoptosis in the ganglion cell layer, a decrease in retinal vasculature, and a reduction in phototopic, but not scotopic, ERG. Without independent verification, it is unclear what is driving the phenotypic differences reported. The weight of evidence however indicates that the retinal toxicity induced in the rat by AMG-8718 is off-target.
The specific target mediating the retinal toxicity of AMG-8718 is currently unclear. The pathogenesis described herein suggests disruption of the phagolysosomal pathway, which could arise through various mechanisms including, but not limited to, inhibition of lysosomal fusion (Huynh et al. 2007), changes to lysosomal pH (Deguchi et al. 1994), inhibition of proteolysis via oxidative stress (Kaemmerer et al. 2007; Thampi et al. 2012), or direct inhibition of lysosomal proteases (Ivy et al. 1989). Similar observations of AFG in the RPE of rats treated with numerous structurally distinct small molecule BACE1 inhibitors suggest a unique metabolite is unlikely to contribute to the effects of AMG-8718. It is tempting to speculate that inhibition of the closely related aspartyl protease CatD may be causative based on the phenotypic similarities of neuronal lipofuscinosis and retinal abnormalities reported in some CatD-inactive or -deficient animal models (Koike et al. 2003; Zhang et al. 2005; Awano et al. 2006) and CatD-deficient humans (Steinfeld et al. 2006). However, AMG-8718-treated rats do not exhibit neuronal lipofuscinosis and degeneration; a defining phenotype of CatD-deficiency and neuronal ceroid lipofuscinoses in animals and man (Shacka and Roth 2007). This may be a consequence of the extent and/or duration of CatD inhibition, as neuronal lipofuscinosis and degeneration were reported in the brain of rats treated for 3 months with LY2811376 (May et al. 2011). Although biochemical selectivity against CatD may appear favorable for AMG-8718, such results may be misleading for basic compounds with the potential to distribute into acidic lysosomal compartments. In addition, effects on other aspartyl proteases, alone or in combination, cannot be completely ruled out.
In conclusion, we have characterized the pathogenesis of drug-induced retinal toxicity by a novel BACE1 inhibitor in the rat. This toxicity appears to manifest as a primary disruption of the phagolysosomal pathway in the RPE, which subsequently leads to irreversible retinal dysfunction and eventual loss of photoreceptors. A BACE1 KO rat model was developed and found to have normal retinal morphology, indicating that BACE1 deletion is not associated with retinal abnormalities in the rat. The putative off-target mediating the retinal toxicity of AMG-8718 is unknown, and therefore the relevance of these rodent findings for human safety is currently unclear. Given the conservation of RPE function across mammalian species, it is possible that humans could be susceptible to phagolysosomal disruption and retinal toxicity, as observed with aminoquinolone antimalarial drugs (Michaelides et al. 2011). Therefore, an appropriate clinical monitoring plan and an adequate safety margin would be prudent to provide additional assurance for patient safety.
Footnotes
Acknowledgments
We would like to thank Dr. SriniVas Sadda and Dr. Simon Peterson-Jones for consultation on the ophthalmic imaging and ERG evaluations, Paul Wen for the CSF and brain Aβ level determinations, and Danny Chui and Evelyn Prior for assistance with the rat knockout model development.
Author Contribution
M. Fielden contributed to the conception, design, analysis, and interpretation. J. Werner contributed to conception, design, analysis, and interpretation. J. Jamison contributed to acquisition, analysis, and interpretation. A. Coppi contributed to analysis and interpretation. D. Hickman contributed to analysis and interpretation. R. Dunn contributed to conception and design. E. Trueblood contributed to design, analysis, and interpretation. L. Zhou contributed to analysis and interpretation. C. Afshari contributed to conception, design, and interpretation. R. Lightfoot-Dunn contributed to conception and design and interpretation.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
