Abstract
Aim: Nonsteroidal anti-inflammatory drugs (NSAIDs) are commonly used to manage the pain and inflammation. NSAIDs can cause serious side effects, including vision problems. However, the underlying mechanisms are still unclear. Therefore, we aimed to investigate the effect of meclofenamic acid (MFA) on retinal pigment epithelium (RPE). Materials and methods: In our study, we applied image analysis and whole-cell patch clamp recording to directly measure the effect of MFA on the gap junctional coupling between RPE cells. Results: Analysis of Lucifer yellow (LY) transfer revealed that the gap junction communication existed between RPE cells. Functional experiments using the whole-cell configuration of the patch clamp technique showed that a gap junction conductance also existed between this kind of cells. Importantly, MFA largely inhibited the gap junction conductance and induced the uncoupling of RPE cells.
Other NSAIDs, like aspirin and flufenamic acid (FFA), had the same effect. Conclusion: The gap junction functionally existed in RPE cells, which can be blocked by MFA. These findings may explain, at least partially, the vision problems with certain clinically used NSAIDs.
Introduction
Nonsteroidal anti-inflammatory drugs (NSAIDs) are widely used medications because they provide effective relief of chronic pain and inflammation via inhibition of cyclooxygenase (COX). However, visual side effects such as temporary blindness, visual field defect, 1 , 2 blurred vision, 3 , 4 scotomata, 4 , 5 color vision changes 4 , 6 have also been reported following short-term or long-term use. But, so far, the underlying mechanism is still unknown. Meclofenamic acid (MFA) is a common clinically used NSAID, which is a derivative of N-phenylanthranilic acid. Its well-known mechanism mediating their anti-inflammatory effects is inhibition of COX-1 and COX-2 that catalyze the biosynthesis of prostaglandins from arachidonic acid. However, accumulating data suggest that NSAIDs also act on other targets such as ion channels, via mechanisms different from the inhibition of the COX-prostaglandin pathway. Our previous studies showed that at high concentrations, MFA also inhibited ATP-sensitive K+ channels, 7 and ibuprofen can modulate cystic fibrosis transmembrance conductance regulator (CFTR), a cyclic adenosine monophosphate (cAMP)-gated Cl− channel. 8 CFTR is an ABC transporter-class ion channel that transports Cl− across epithelial cell membranes. Mutations of the CFTR gene affect the functioning of the Cl− channels in these cell membranes, leading to cystic fibrosis. NSAIDs such as diclofenac or flurbiprofen also inhibited acid-sensing ion channels on sensory nociceptor neurons. 9 Other fenamate compounds were found to potentiate large conductance Ca2+-activated K+ currents and gamma-butyric acid (GABAA) receptor Cl− channels. 10 , 11 MFA, although at high concentrations, displayed many desirable properties as a gap junction antagonist: high potency, water solubility, and relatively fast reversibility. 12 , 13 Recently studies identified that gap junction protein connexin43 (Cx43) existed in ARPE-19 cells (a human retinal pigment epithelial cell line), 14 , 15 but so far whether gap junction channels functional existed in ARPE-19 cells, and whether MFA also blocked this kind of channels, were still unknown.
Materials and methods
Chemical reagents
MFA, aspirin, and flufenamic acid (FFA) were purchased from Sigma (St. Louis, MO, USA), and lucifer yellow (LY) was from invitrogen (Carlsbad, CA, USA). Testing solutions were prepared shortly before usage. In accordance with manufacturer’s specific instructions, these compounds were first dissolved in double distilled water as stock and then diluted with extracellular perfusing solution at desired concentrations.
Cell culture
ARPE-19, obtained from American Tissue Culture Collection (ATCC, Manassas, VA, USA) was used in this study. Cells were cultured in Dulbecco's Modified Eagle's Medium/Nutrient Mixture F12 (DMEM/F12) with 15 mM 4-(2-hydroxyethyl)-1-piperazineethanesulfonic acid (HEPES) buffer, 10% (v/v) fetal bovine serum, 0.348% (w/v) additional sodium bicarbonate (NaHCO3), 2 mM
Dye injection
Using the patch clamp technique in the whole-cell configuration combined with intracellular dialysis of the fluorescent dye LY, gap junction between adjacent cells were analyzed by LY transfer. LY was added at a concentration of 1 mg/ml in the glass pipette.
Electrophysiological recording and data acquisition
ARPE-19 cells were cultured to 50% confluence after 3 days of cell culture and then used for recordings. A single patch clamp method was used for studying gap junctions as described. 16 The extracellular perfusing solution contained (in mM) 140 NaCl, 1.3 CaCl2, 5.4 KCl, 2.0 MgCl2, 25 HEPES, pH 7.4 adjusted using NaOH. Whole-cell patch clamp recordings were performed by means of an Axopatch-1D amplifier (Axonal Instrument Co.). Glass patch pipettes had typical resistances of 3–5 MΩ and filled with the following solution (in mM): 150 K-gluconate, 1 CaCl2, 4 MgCl2, 11 EGTA, 10 HEPES, 4 K-ATP, pH 7.3 adjusted using KOH. All electrophysiological recordings were obtained at room temperature (21–22°C). For voltage clamp recordings, cells were constantly held at −60 mV. Transmembrane currents were elicited by depolarizing pulses stepped from potential of −80 to 60 mV (or −100 mV to100 mV) with 10 mV increment. Sustained outward current was taken as the mean current during the last 90 ms of the depolarizing pulse. For establishment of the current–voltage (I-V) relation, alternatively, voltage ramp protocols (−80 to 60 mV, 5s) were applied to the cell. For pharmacological studies, control or compound-containing solutions were focally applied onto the cell using a multi-barreled perfusion system (SF-77B, Warner Instruments). Data were collected online by means of Axon Digidata 1322A system with pClamp software (Axonal Instruments) and analyzed off-line with Clampfit (Axonal instruments) and SigmaPlot (SPSS).
Data analyses
Data are presented as mean ± SE. Statistical analysis was performed using Paired Student's t test. Statistical significance was set at p < 0.05.
Results
The gap junction coupling functional existed in ARPE-19 cells
Analysis of LY transfer revealed that the gap junction communication between ARPE-19 cells existed (Figure 1(a) top lane). To further study whether the gap junction communication functional existed between ARPE-19 cells, we made whole-cell patch voltage clamp recordings in ARPE-19 cells. The average liquid junction potential (LJP) was 12 ±2.8 mV. The average capacitance was 23.3 ± 4.35 pF (n = 10). The amplitudes of currents evoked by each voltage step were increased proportionally in the ARPE-19 cells, implying the development of a nonvoltage-dependent conductance (NVDC) which had a linear I-V relationship (Figure 2 (a1) and (a3)). This result showed that gap junction channels functional existed in ARPE-19 cells.

The effect of meclofenamic acid (MFA) on dye coupling in ARPE-19 cells. A control experiment shows the electrode impaling a single cell after 5 min of dye injection with 1 mg/ml Lucifer yellow (LY) (green). LY rapidly spread to the neighborhood cells. Black arrow shows the location of cell recorded (Lane a and b).The same protocol in the presence of 30 µM MFA produces a dramatic reduction in dye coupling. After a 15-min wash, LY still had not spread to other cells (Lane c). This indicates that coupling was irreversible in the presence of high concentration MFA.

Meclofenamic acid (MFA) inhibits a nonvoltage-dependent gap junctional conductance in ARPE-19 cells. Typical traces of transmembrane currents evoked by changing membrane potential (VM ) with voltage steps before and after MFA. The inset in (left lane) illustrates the VM -changing protocol (a1). MFA (30 µM) largely inhibits a nonvoltage-dependent gap junctional conductance in ARPE-19 cells (a2). Example I-V curves evoked by changing the VM using a voltage ramp protocol are shown (a3). This nonvoltage-dependent gap junctional conductance displays a linear I-V relationship. b shows the histograms of change in the gap junctional conductance. The currents are measured at −60 mV. The amplitudes of currents are normalized to the control (n = 11, **p < 0.01).
MFA inhibited the gap junctional coupling in the ARPE-19 cells
We examined the effects of MFA on the dye coupling between ARPE-19 cells. MFA produced a dramatic reduction in the coupling between the ARPE-19 cells (Figure (1a) bottom lane). Black arrow showed the location of cell recorded (Figure (1b)). After a 15-min wash, LY still had not spread to other cells, indicating that the uncoupling was irreversible in the presence of high concentration MFA (Figure (1c) bottom lane). The whole patch clamp recording revealed that application of MFA (30 µM) to the ARPE-19 cells significantly suppressed the gap junction conductance (Figure 2). Other NSAIDs, like aspirin (10 mM) and FFA (30µM), had the same effect (Figure 3).
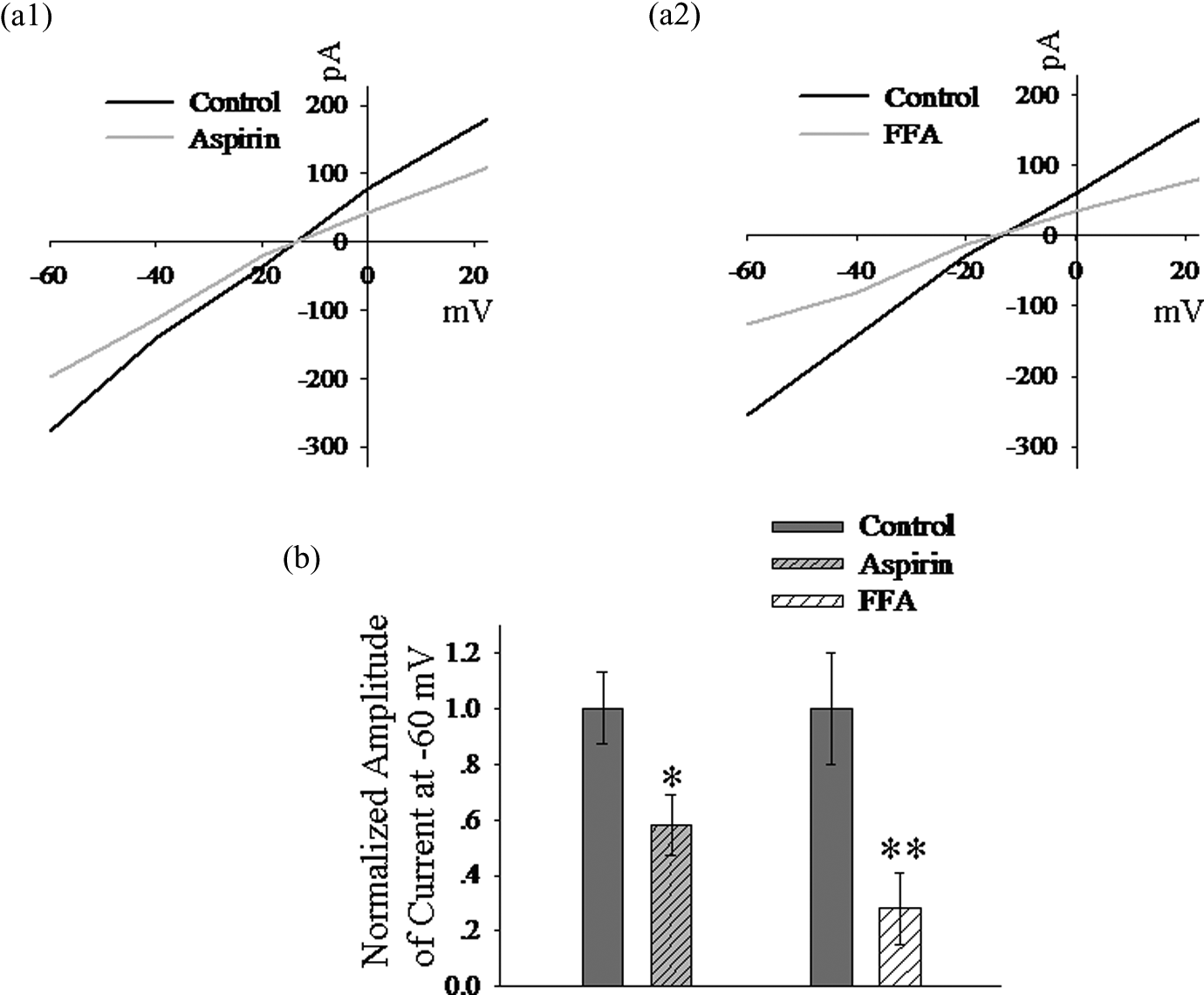
Aspirin and flufenamic acid (FFA) suppress the gap junctional conductance in ARPE-19 cells. a1 and a2 display the example I-V curves after aspirin (10 mM) and FFA(30 µM). b shows the histograms of change in the gap junctional conductance. The currents are measured at −60 mV. The amplitudes of currents are normalized to the control (n = 13, *p < 0.05, **p < 0.01).
Discussion
In this study, we identified that gap junction functionally existed in RPE, which can be blocked by MFA. These findings may explain, at least partially, the vision problems with certain clinically used NSAIDs.
One of the principal findings was that gap junction functionally existed in RPE. Gap junction channels allow direct exchange of ions, second messengers, metabolites, and other small molecules (up to ∼1200 kDa) between the cytoplasm of adjacent cells. This type of intercellular coupling is known to regulate cell proliferation and differentiation.17–19 Previous studies revealed that gap junction proteins were expressed in RPE cells. 14 , 15 Gap junction hemichannels from the pigment epithelium regulate neural retinal progenitor proliferation 20 and the differentiation of retinal pigment epithelial cells, 21 NSAIDs are widely used medications to relieve pain and inflammation in clinic. However, these drugs also cause a few common side effects including nausea, vomiting, diarrhea, and constipation. Some uncommon side effects also have been reported such as temporary blindness, visual field defect, blurred vision, scotomata, and color vision changes1–6 following short-term or long-term use. Evidence suggests that NSAIDs can effectively cross the blood–brain barrier. 22 The relatively high therapeutic plasma concentrations are achieved in the brain with 80 µM and 20 to 70 µM for mefenamic and niflumic acids, respectively,22–24 suggesting that appreciable brain levels might be achieved. MFA is a gap junction antagonist, 12 , 25 but it is unclear so far whether MFA blocks the gap junction communication between the RPE cells. Our electrophysiological results and image analysis identified that gap junction functionally existed between the RPE cells, which was irreversibly inhibited by MFA. RPE is a nonrenewable single cell layer situated in the posterior segment of the eye between the neural retina and the choroid. Its functions include maintenance of blood–retinal barrier properties, phagocytosis, and continuous digestion of growing photoreceptor outer segments and maintenance of the volume and composition of the sub-retinal fluid 26 , 27 and is indispensable for the survival and function of retinal photoreceptors. 28 Primary changes in the RPE cells and the associated secondary changes in the neural retina result in proliferative and degenerative diseases of retina, such as proliferative vitreoretinopathy, diabetic retinopathy, and age-related macula degeneration. 29 MFA also blocks the electrical synapse currents of horizontal cells, retinal AII amacrine, and on-cone bipolar cell. 12 , 25 Gap junction uncoupling of retinal cells induced by MFA may be one of the factors that causes the vision problems with certain clinically used NSAIDs. Vision may be damaged with high doses or long-term use of NSAIDs. As a result, doctor should be cautious about taking NSAIDs if patients have vision problems.
Footnotes
Authors’ Note
The authors Nannan Ning and Ying Wen contributed equally to this work.
Declaration of Conflicting Interests
The authors declared no conflicts of interest.
Funding
This work was funded by grants to Jingxin Li from National Natural Science Foundation of China (NSFC 81070503).
