Abstract
To study the effects of nicotine on retinal pigment epithelium (RPE) cells in vivo and in vitro, (Balb/c×C57Bl/6) F1 mice were given water containing 100 µg/mL nicotine for six months. Cultured fetal RPE cells were treated with nicotine or lipopolysaccharide for seventy-two hours. Expression of matrix metalloproteinase protein (MMP)2, MMP9, and VEGF was measured using Western blot. Expression of IL6 and IL8 was measured using real-time polymerase chain reaction or enzyme-linked immunosorbent assay. Electron microscopy was performed to observe the effects of nicotine on morphological changes of mice retina or cultured RPE cells, and filamentous actin in RPE cells was stained with phalloidin. Electron microscopy revealed that nicotine-treated mice showed thinner outer nuclear layers, fewer pigment granules in RPE cells, and a damaged photoreceptor–RPE interface when compared with age-control mice. When added to cultured RPE cells, nicotine induced accumulation of osmiophilic lamellated intracellular inclusions in cytoplasm, mitochondrion hypertrophy and vacuolar degeneration, and redistribution of actin in cells without affecting cell proliferation. Expression of MMP2 and MMP9 in nicotine-treated RPE cells was decreased. Nicotine-induced changes in RPE morphology and function provide insight into pathogenesis of smoking-related retinal diseases.
Introduction
Retinal pigment epithelial (RPE) cells form the outer layer of the blood–retinal barrier, which plays an important role in maintenance, metabolism, and function of photoreceptors. Retinal pigment epithelial cell dysfunction is involved in both hereditary and acquired ophthalmic diseases, such as age-related macular degeneration (AMD), in which the impairment of RPE cell functions is an early and crucial event in the molecular pathways leading to various macular degenerations (Nowak 2006; Rattner and Nathans 2006). Epidemiological studies consistently document that cigarette smoking is the primary environmental risk factor for all forms of AMD (Clemons et al. 2005; Christen et al. 1996; Evans 2001; Seddon et al. 1996). However, the pathogenic factors in cigarettes that account for this increased risk and the underlying molecular mechanisms are still not clear. A recent study showed that benzo(a)pyrene, a toxic element in cigarettes, may be involved, by altering cell biological processes in the RPE, in the formation of drusen in cigarette smokers (Wang et al. 2009). Although there are many chemical substances present in tobacco smoke, nicotine, one of the major toxic components, has been found to modulate oxidative accumulation and angiogenesis, which pathological processes that are shared by AMD and other disorders (Heeschen et al. 2001; Jain 2001). Exposure of mice to cigarette smoke also leads to sub-RPE deposit accumulation in several models (Espinosa-Heidmann et al. 2006; Fujihara et al. 2008; Ida et al. 2004), which indicates that smoke elements might interfere with function of RPE layers. However, direct evidence on how nicotine affects the biological status of RPE cells is lacking. In the present study, we demonstrated that feeding mice with nicotine or treating cultured RPE cells with nicotine causes morphologic changes in RPE cells or the RPE–photoreceptor interface, suggesting that nicotine might participate in the pathogenesis of RPE-related diseases by impairing the proper physiological function of RPE cells.
Materials and Methods
Animal Experiment Procedures
(Balb/c×C57Bl/6) F1 mice were obtained from the Laboratory Animal Center of Shandong Eye Institute (Shandong, China) and maintained in the animal care unit of this institute. Nicotine treatment of the mice started at the age of eight weeks by adding nicotine (Sigma, St. Louis, MO, USA) at 100 µg/mL to drinking water, whereas age-matched control mice received regular drinking water (Suner et al. 2004). All mice were fed standard laboratory animal food ad libitum and were maintained under a 12-hour light–dark cycle. Animal experiments were carried out following guidelines of the ARVO Statement for the Use of Animals in Ophthalmic and Vision Research. By the end of the nicotine treatment, the levels of nicotine in mice retina or blood were measured using high-performance liquid chromatography–tandem mass spectrometry (HPLC–tMS).
Transmission Electron Microscopy of Mouse Retina
After six months of nicotine feeding, the mice were sacrificed and the eyes were enucleated and fixed in 4% glutaraldehyde cacodylate buffer for ten minutes. The anterior sections of the eyes were removed along the corneoscleral limbal line, and fixation continued for one more hour. The posterior segments (retina, choroid, and sclera) were dissected into quarters in the ciliary body–perioptic nerve direction, followed by conventional protocols for preparing electron microscopy (EM) sections, and the samples were examined with a JEM-1200 transmission electron microscope or JSM-840 scanning electron microscope (JEOL, Tokyo, Japan).
Culture of Human RPE Cells
Eyes of human abortuses were used as the source of primary RPE cells with approval by the Shandong Eye Institute Medical Ethics Committee and in compliance with the Declaration of Helsinki. Sheets of RPE were dissected from the choroids of fetal eye cups and cultured in low-calcium (0.05 mM Ca++) culture medium, which contained calcium-free minimum essential medium (MEM Eagle) with Earle’s salts (Sigma, St. Louis, MO, USA) and proposed supplements (all obtained from Sigma) plus bovine retina extract (Hu and Bok 2001). After primary RPE cells reached confluence, floating RPE cells were collected for cryopreservation or passage. Passaging was performed with medium containing supplements plus bovine retina extract (Sigma), but with normal calcium concentration (1.8 mM Ca++).
To study the effect of nicotine treatment on RPE proliferation, morphology, and function in an in vitro system, seeded fetal RPE cells were allowed to adhere to the culture surface before nicotine or lipopolysaccharide (LPS) were added to the culture medium. Lipopolysaccharide was used to detect a possible response of the RPE to TLR ligands and to reflect the responsiveness to foreign stimulators. The treatment lasted for three days. At the end of the incubation, the conventional MTT assay was used to quantify the cells in culture.
Electron Microscopy of Cultured RPE Cells
Fetal RPE cells were seeded on glass coverslips placed in twenty-four-well plates (for scanning EM study) or seeded in regular flasks (for transmission EM) and treated with 1 µM nicotine for seventy-two hours. After removal of the supernatant, the cells were rinsed with PBS and cells grown on coverslips were used for scanning EM following conventional procedures. The cells grown on flask surfaces were harvested by cautiously scraping using a cell scraper and collected by slow spinning, and the pellets were used for the transmission EM procedure as above.
Staining of F-actin with Phalloidin
Retinal pigment epithelium cells on coverslips (as above) were fixed with 4% neutralized polyformaldehyde for ten minutes, permealized with 0.2% Triton X-100 for ten minutes, blocked with goat serum for thirty minutes, stained with FITC-conjugated phalloidin (Alexis, San Diego, CA, USA) at 4°C overnight, and counterstained with DAPI. Phalloidin binds specifically at the interface between F-actin subunits (Cooper 1987). The samples were properly rinsed with phosphate-buffered saline between each step. A confocal microscope (C1-plus, Nikon, Japan) was used to capture fluorescence signals.
Enzyme-Linked Immunosorbent Assay
Retinal pigment epithelium cells were treated with 0.25, 0.5, 1, and 2 µM nicotine for seventy-two hours, and the supernatant was harvested for enzyme-linked immunosorbent assay (ELISA). IL-8 has been proposed to be one of the cytokines produced by RPE cells upon foreign insult (Elner et al. 1990; Yoshida et al. 2001). The concentration of IL8 in cultured RPE supernatant was performed using a BD human OptEIA IL8 set (BD Biosciences, San Diego, CA, USA) following the procedure provided by the supplier.
Real-Time Polymerase Chain Reaction
Total RNA was isolated from RPE cells using an RNeasy Mini Kit (Qiagen GmbH, Hilden, Germany) and reverse-transcribed to cDNA using an AMV first-strand cDNA synthesis kit (BBI, Toronto, Canada). Real-time polymerase chain reaction (PCR) was performed for evaluating expression of IL6 and IL8 using Taqman Universal PCR Master Mix in combination with specific primers and probes (Table 1) on an ABI Prism 7500 system (Applied Biosystems, Foster City, CA, USA), as recommended by the manufacturer. Each sample was assayed in duplicate. Cycling conditions were as follows: 10 minutes at 95°C followed by forty two-step cycles (15 seconds at 95°C and 1 minute at 60°C). The quantification data were analyzed with the SDS System Software, version 1.3.1 (Applied Biosystems, Singapore. Release date: October, 2005) using glyceraldehyde-3-phosphate dehydrogenase as reference.
Sequences of primers and probes used for real-time polymerase chain reaction.
Western Blot Analysis
Retinal pigment epithelium cells treated with or without nicotine were harvested into SDS-PAGE buffer. The samples were sonicated briefly, then separated by SDS-PAGE and immunoblotted using primary antibodies against matrix metalloproteinase protein (MMP) 2, MMP9, and VEGF (Boster, Wuhan, China). Horseradish-peroxidase-conjugated secondary antibodies and the ECL detection kit (Chemicon, Billerica, MA, USA) were used for the detection of specific proteins. The results were analyzed using Image J software, version 1.4 (National Institutes of Health, Bethesda, MD, USA).
Results
Morphological Changes of Retina in Nicotine-Fed Mice
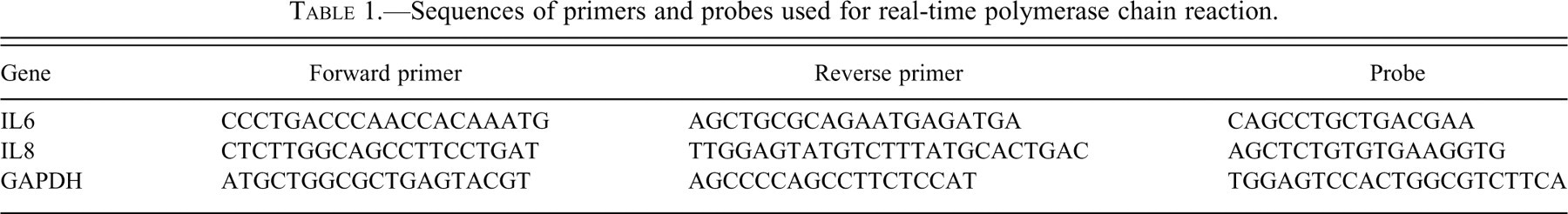
The HPLC-tMS method was validated first for measuring nicotine concentration in this assay. After drinking water containing 100 µg/mL nicotine for six months, the mean nicotine concentrations were 0.35 ng/mg (n = 4) and 42.2 ng/mL (n = 4), respectively, in the retina and blood of mice. Morphological alterations of the retina were observed by EM in nicotine-fed mice. Retinal pigment epithelium cells in normal mice contained many large, dense pigment granules indicating significant pigmentation, long RPE processes, and a clear photoreceptor–RPE interface (Figure 1A, 1C). In nicotine-fed mice, the pigment granules were significantly poor and sparse in RPE cells, processes were shortened, and the photoreceptor–RPE interface was damaged (Figure 1B, 1D). In normal mice, the outer nuclear layer contained more than ten cell layers and the cell arrangement was regular, whereas the outer nuclear layer was significantly decreased (only four or five layers) and more irregular in nicotine-treated mice (Figure 1E, 1F).
Morphology of retina in nicotine-fed mice (B, D, F) or age-controls (A, C, E). Compared to normal mice, nicotine-fed mice showed less pigment granules in RPE cells (B), damaged photoreceptor–RPE interface (D), and much thinner and damaged external granular layers (F). Original magnification: (A, B), ×2500, (C, D), ×4000, (E, F) ×1000. Four mice in each group were observed and given were representative illustrations. RPE, retinal pigment epithelium; ROS, rod outer segment; PG, pigment granule; ONL, outer nuclear layer.
Morphological Change in Nicotine-Treated Human RPE Cells
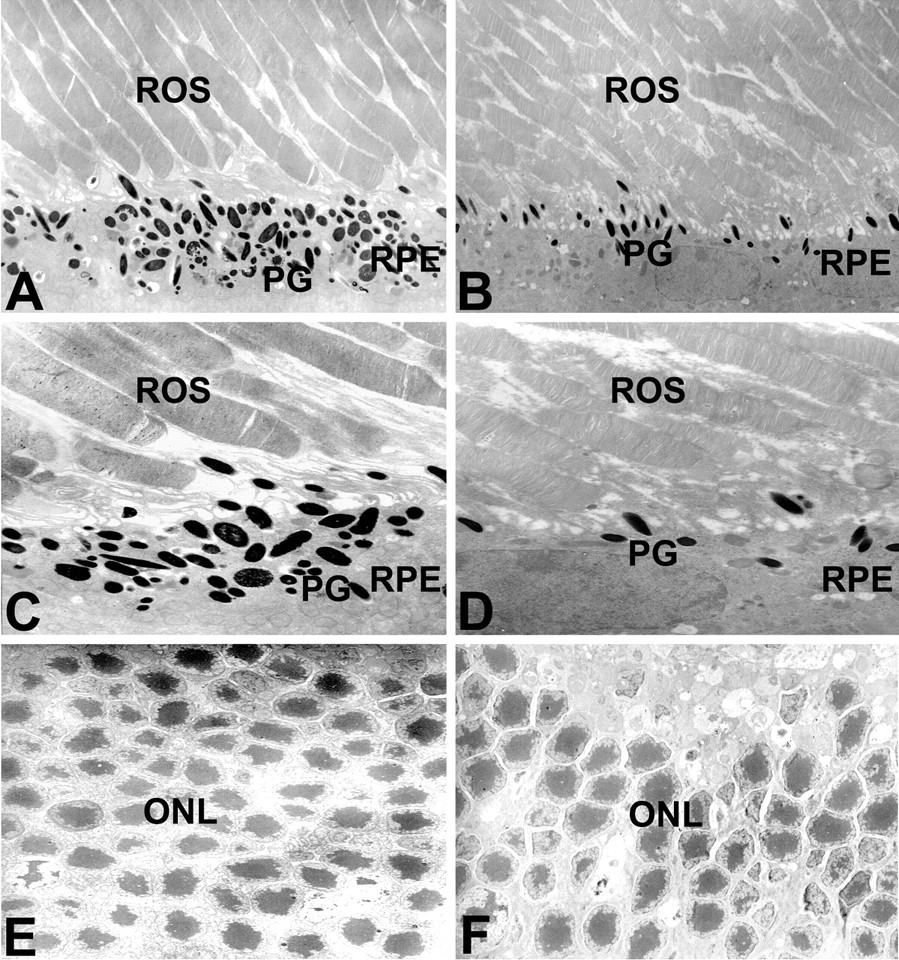
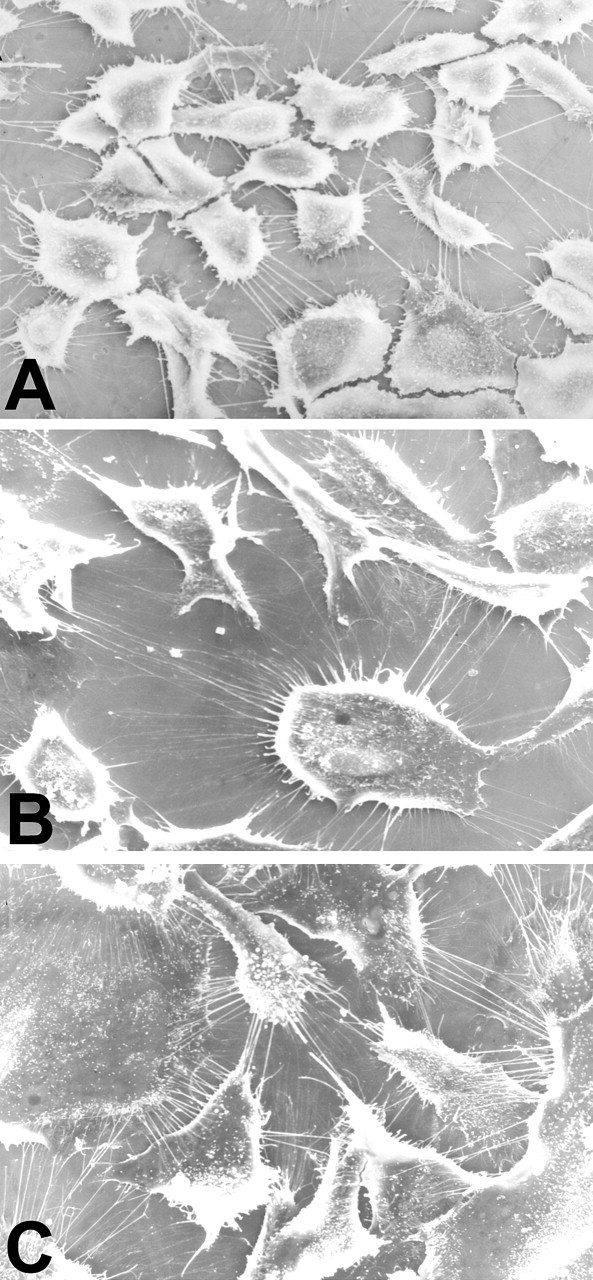
We next determined whether nicotine added to the culture medium affects morphology or proliferation of cultured human RPE cells. Scanning electron microscopy demonstrated that RPE cells cultured with nicotine were flattened and thinner than control cells, the normal polygonal morphology was lost, the nuclei were less recognizable, and the variability of cell shapes was more significant compared with control groups. The extent and frequency of morphological changes of RPE cells depended on the dose of nicotine (Figure 2). Staining with phalloidin for F-actin in cultured RPE cells showed that F-actin was more likely to form spherical structures in control RPE cells but was more diffuse in nicotine-treated RPE cells (Figure 3). The transmission EM study revealed that a large number of osmiophilic, lamellated, intracellular inclusions (myelin figures) existed in nicotine-treated RPE cells (Figure 4B, 4C). Moreover, substantial mitochondrion hypertrophy and vacuolar degeneration was found in nicotine-treated RPE cells (Figure 4B, 4D). Even with these changes, no significant alteration of proliferation was observed when the RPE cells were treated with nicotine for 3 days (data not shown).
Morphological changes in nicotine-treated retinal pigment epithelium (RPE) cells. The cells were grown on coverslips and treated with agents for seventy-two hours. (A) RPE cells grown in medium; (B) RPE cells grown in 0.5 μM nicotine; (C) RPE cells grown in 1.0 μM nicotine. Original magnification: ×600. The experiment was repeated twice with similar results.
Redistribution of F-actin in retinal pigment epithelium (RPE) cells. (A, C) RPE cells grown in medium; (B, D) RPE cells grown in 1.0 μM nicotine. Original magnification: A, B, ×100; C, D (inlet of A, B, respectively), ×500. The experiment was repeated three times with similar results.
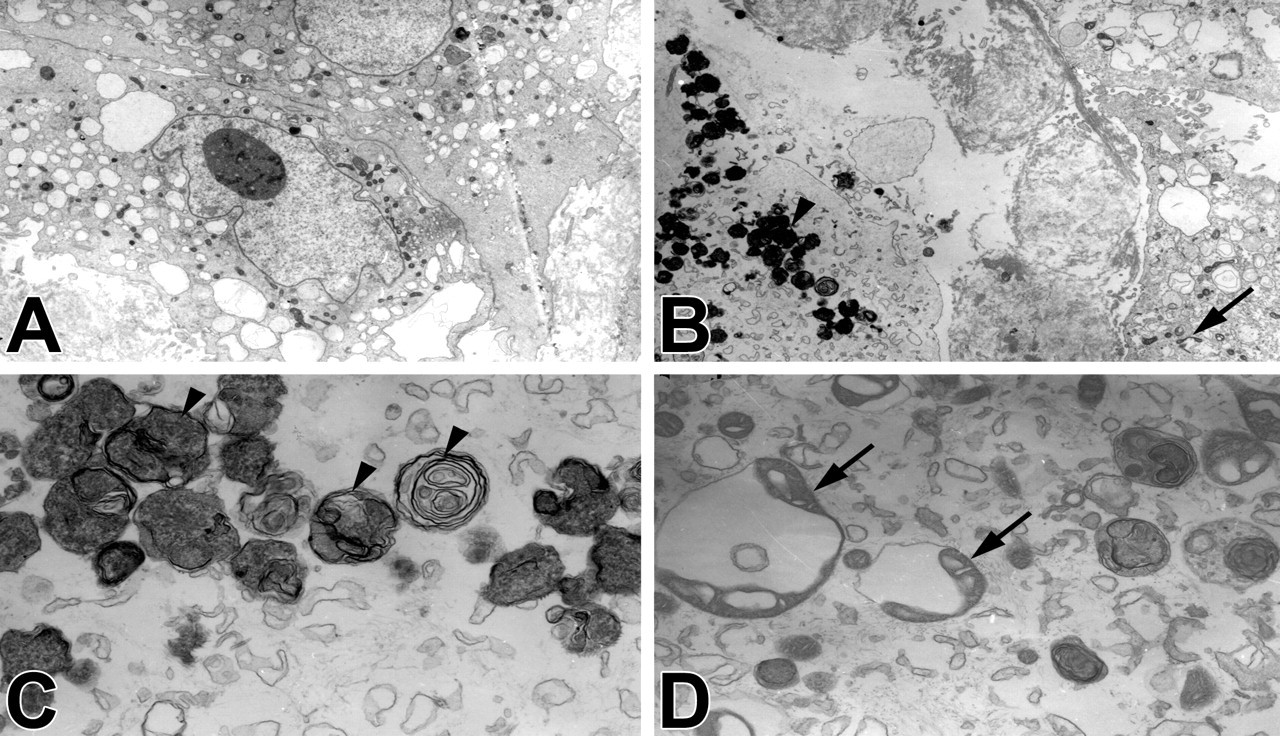
Ultrastructural changes of cultured retinal pigment epithelium cells in response to nicotine treatment. Cells were cultured on a flask surface and treated with medium (A), 1.0 μM nicotine for seventy-two hours (B–D). The triangle marks indicate detailed structures of osimiophilic lamella bodies, the arrows indicated the mitochondrion hypertrophy and vacuolar degeneration. Original magnification: (A, B) ×2000; (C, D) ×8000. The experiment was repeated three times with similar results.
Effects of Nicotine on Cytokine Production of Human RPE Cells
Real-time PCR results showed that expression of both IL6 and IL8 was markedly increased in LPS-treated RPE cells, but not in nicotine-treated cells (Figure 5A). Cultured RPE cells constitutively produced detectable IL8 in the medium, and this production could be greatly increased by LPS. However, up to 2 µM nicotine did not change IL8 levels in the culture medium (Figure 5B).
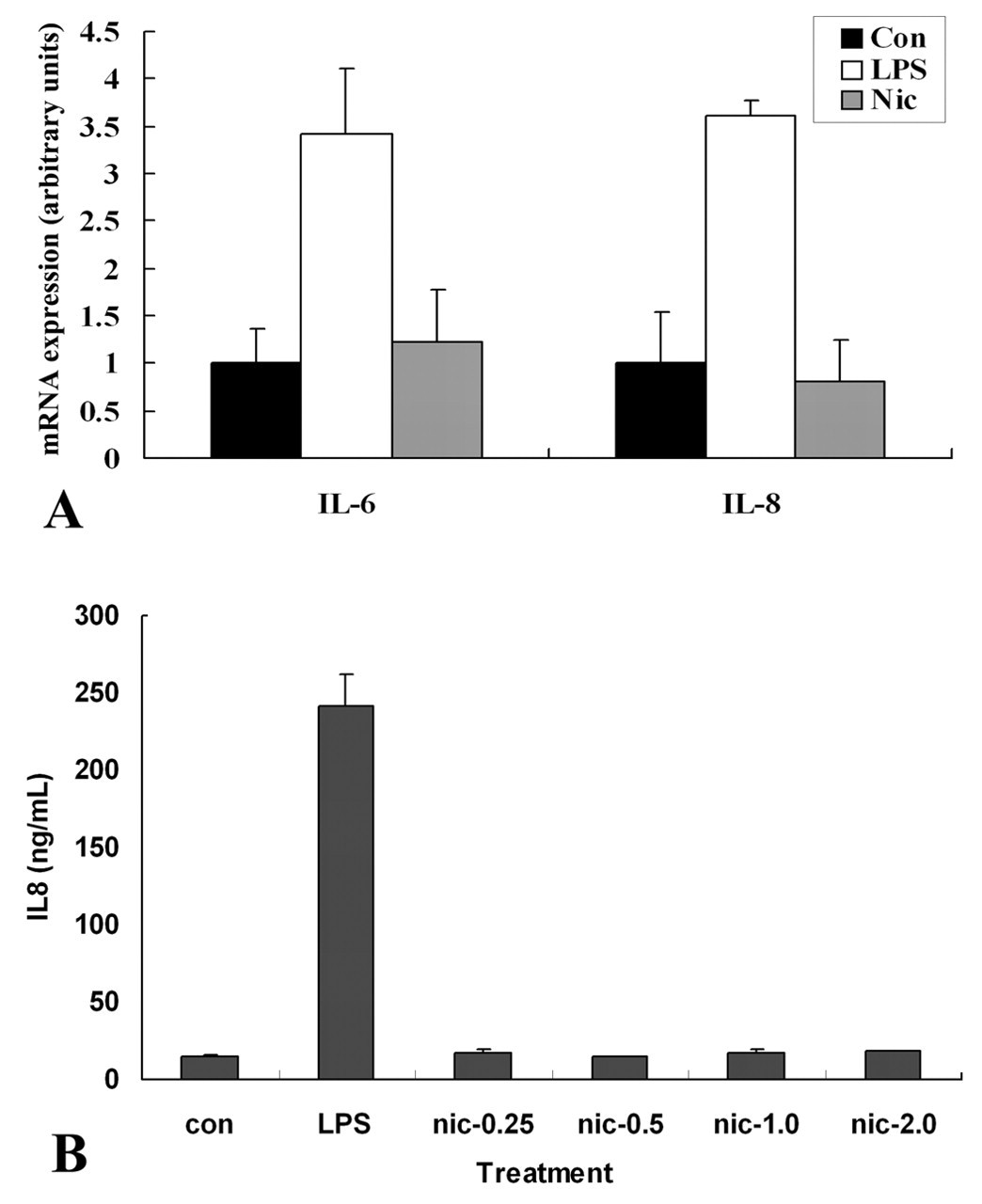
Real-time analysis of IL8 and IL6 in cultured retinal pigment epithelium (RPE) cells (A) and enzyme-linked immunosorbent assay analysis of IL8 in supernatants of RPE cells (B). Con, control group; LPS, treated with 1 µg/mL LPS; Nic-0.25, Nic-0.5, Nic-1.0, Nic-2.0, treated with 0.25, 0.5, 1.0, 2.0 µM nicotine. The experiments were done three times with similar results.
Effects of Nicotine on VEGF, MMP2, and MMP9 expression
Expression of VEGF, MMP2, and MMP9 was determined by Western blot. MMP2 was significantly decreased and MMP9 was slightly decreased in nicotine-treated RPE cells, whereas VEGF protein level remained stable in the presence or absence of nicotine (Figure 6).
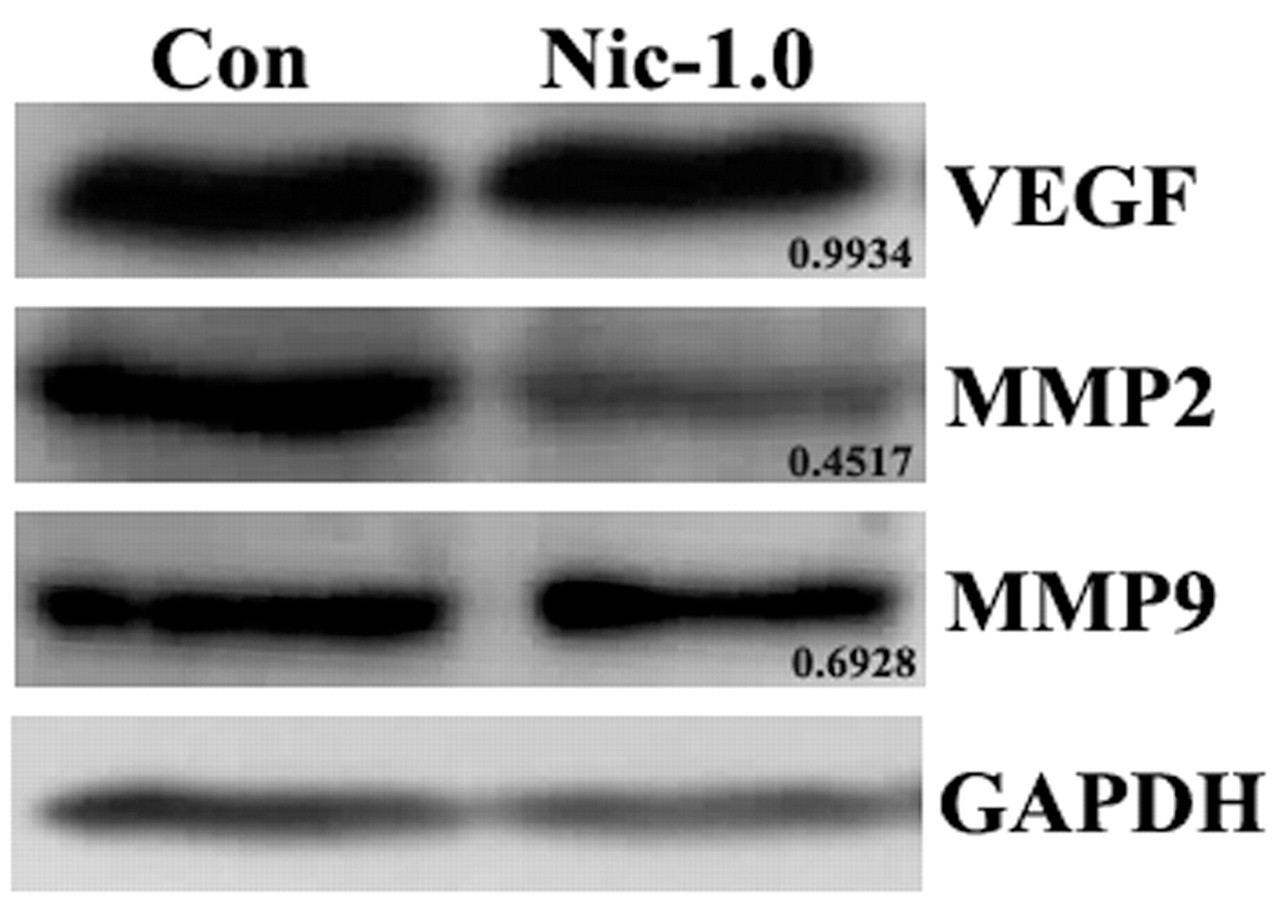
Western blot analysis of matrix metalloproteinase protein (MMP)2, MMP9, and VEGF of retinal pigment epithelium cells. Con, control group; Nic-1.0, treated with 1.0 µM nicotine. The pixel of each band was obtained with ImageJ software, and the relative density of each gene to GAPDH was calculated. The relative densities in control group were arbitrarily set at 1.0, and the ratios of relative densities in nicotine group were obtained by comparing to those in control group.
Discussion
Cigarette smoking is an environmental risk factor for many diseases, including AMD (Evans 2001). It has been proven that giving mice water containing nicotine (usually at 100 μg/mL) mimics the situation of chronic, moderate smokers (Heeschen et al. 2001). This model is thus widely used to study the effect of chronic nicotine toxicity on choroidal neovascularization (Suner et al. 2004). Here, we were able to show that in the present experimental situation, the local concentrations of nicotine were 0.35 ng/mL in retina and 42.2 ng/mL in blood, which was about the same level as in the plasma of smokers (5–44 ng/mL) (Leikin and Paloucek 2007; McCusker et al. 1982; Russell et al. 1975), depending on the brand of cigarette and time of measurement after smoking.
Previous studies have shown that degeneration of RPE cells is one of the initial events in AMD (Kopitz et al. 2004; Nowak 2006). Since epidemiology studies show that cigarette smoking is the greatest environmental risk factor of AMD, investigating the effects of nicotine on RPE cells may lead to clues of pathological mechanisms leading to AMD development in cigarette smokers. Patton first showed that the cigarette constituent benzo[a]pyrene, may crosslink with cellular DNA and delay RPE growth (Patton et al. 2002), and Sharma showed that another toxic element of cigarette smoke, benzo(e)pyrene, leads to apoptosis of human RPE cells in vitro (Sharma et al. 2008). Notably, both the Cousins and Handa groups have further shown that mice treated with nicotine for six months had a damaged retina–choroid complex, that is, higher apoptotic RPE incidence and increased deposition onto Bruch’s membrane (Espinosa-Heidmann et al. 2006; Fujihara et al. 2008).
In the current study, we observed greatly reduced numbers of pigment granules in RPE cells, shortened processes of RPE cells, as well as loss of overlying photoreceptors (Figure 1A–1D) and obviously decreased numbers of cells in the outer nuclear layer in nicotine-treated mice compared with control mice (Figure 1E, 1F). However, the time course and chronological sequence of these changes deserve further investigation. Although not examined, we also have good reason to postulate that the structural alteration of RPE cells and photoreceptors described here would result in dysfunction of RPE cells and photoreceptors, and might result in impaired vision of the animals.
In the in vitro study, nicotine treatment of RPE cells led to morphology alterations. Although control RPE cells were more likely to have the natural hexagonal shape and maintain a substantial thickness even when isolated from each other (Figure 2A), nicotine-treated RPE cells tended to spread like a thin sheet on the culture surface (Figure 2B, 2C). These changes might be correlated with redistribution of F-actin upon nicotine treatment (Figure 3), as has been noticed in cytokine-challenged RPE cells (Li et al. 2007; Zech et al. 1998). Transmission EM showed that nicotine-treated RPE cells were rich in osmiophilic lamellar structures, and mitochondrion hypertrophy and vacuolar degeneration were also present (Figure 4). We propose that the appearance of osmiophilic lamella bodies and deformed mitochondria reflect the changes in the functional status of the cells upon acute challenge with nicotine. However, we are unclear as to how these changes influence the biological functions of RPE cells, or whether these lamellar bodies are released into the extracellular space or remain within cells. It is also uncertain whether these apparently abnormal organelles have any relationship with the slowly formed sub-RPE deposits that have been observed in previous studies (Cousins et al. 2002; Espinosa-Heidmann et al. 2004; Rakoczy et al. 2002). Furthermore, studies are necessary to identify possible functional deficits in RPE cells as a result of nicotine administration. Surprisingly, and in contrast to earlier reports (Patton et al. 2002), we were not able to detect the effect of nicotine on RPE growth in the current model, even with obvious morphological changes present. Confirming our results, Patil et al. (2009) found that ARPE-19 cells showed no change in either cell viability or caspase-3/7 activity after nicotine treatment.
IL8 has been proposed to be one of the main cytokines secreted by RPE cells during their response to foreign challenge stresses like proinflammatory/cytokine treatment (Bian et al. 1996; Ebihara et al. 2007; Elner et al. 1990; Fukuoka et al. 2003) or injury (Yoshida et al. 2001). In some cases, nicotine was reported to inhibit IL8 production in various cells (Kalayciyan et al. 2007; Louvet et al. 1999; Sugano et al. 1998; Patton et al. 2006), but in other cases, nicotine was reported to increase IL8 production (Iho et al. 2003; Wendell and Stein 2001). In this study, although cultured RPE cells spontaneously secreted certain a amount of IL8 and LPS dramatically enhanced IL8 production, up to 2 µM of nicotine in RPE culture failed to affect IL8 production, either at the mRNA level (Figure 5A) or the protein level (Figure 5B). Similar results were obtained with IL6 (Figure 5A), implying that RPE may not respond the same way to nicotine as it does to proinflammatory stimuli.
Members of the MMPs are expressed in RPE cells to a varying extent and are involved in several RPE-related pathological processes, including choroidal neovascularization (Kvanta et al. 2000; Steen et al. 1998). It has also been shown that smoke components modulate VEGF and MMP expression in cells and in live animals (Seagrave et al. 2004; Wright et al. 2002). In our experiment, we found that nicotine treatment decreased the protein level of MMP2 and MMP9, but nicotine did not alter the protein expression of VEGF by RPE cells (Figure 6).
Taken together, the results of this study displayed that nicotine, as one of the major toxic elements of cigarette smoke, damaged RPE cell structure under both in vivo and in vitro conditions. This finding supports the clinical observation that smoking is involved in pathogenesis of RPE-related diseases like AMD. Extensive studies are necessary to further investigate the molecular basis for nicotine’s effect on RPE cells.
Footnotes
Acknowledgment
We thank Liming Lin for technical assistance with HPLC–tMS and Robert Mullins at University of Iowa for editing the manuscript. This work was supported by the Natural Science Foundation of China (30471645), a Shandong Provincial Healthcare Research Grant (2005), and the Taishan Scholar Program QDU-EYE, Shandong, China.
This work was supported by the Natural Science Foundation of China (30471645), a Shandong Provincial Healthcare Research Grant (2005), and the Taishan Scholar Program QDU-EYE, Shandong, China.
