Abstract
Surgical site infections (SSIs) are a common surgical-related complication. To avoid these complications, a new biodegradable polymer–lipid encapsulation matrix that provides controlled release of doxycycline (doxycycline/polymer-lipid encapsulation matrix [D-PLEX]) has been developed. The aim of this comprehensive study was to evaluate the potential safety of D-PLEX100 in abdominal surgical site. D-PLEX100 was administered into incisions of abdominal surgical site in Yucatan miniature swine, which were followed for up to 6 months and compared to sham-control swine. The D-PLEX100 mass did not migrate from the incisional site, and there was no evidence for systemic toxicity or other safety concerns. Surgical incision sites, including the peritoneal surface, were fully healed at 6 months in all animals. Most of the D-PLEX100 mass was absorbed during the first 3 months, and by 6 months, D-PLEX100 was fully absorbed. Toxicokinetic evaluation revealed that doxycycline concentrations were evident at 30 minutes and persisted to 8 days (71 mg/kg) or at least 15 days (284 mg/kg) and were no longer present in plasma by day 29. This study supports the safety of D-PLEX100 and its favorable degradability profile. A clinical study is being performed to assess the safety and the efficacy of D-PLEX100 to prevent human abdominal SSIs.
Keywords
Introduction
Surgical site infection (SSI) is a very common complication, affecting 2% to 5% of all surgical patients in the Unites States, resulting in 300,000 to 500,000 affected patients annually. 1,2 It is the most common hospital-acquired infection, representing 31% of health care associated infections, 3 and in addition to morbidity and mortality, SSIs can result in increased length of hospital stay. 4 –6 Abdominal operations are especially susceptible for SSIs, and the incidence rates can reach more than 50% in some of the operations (eg, colostomies and colon cancer resection). 1,7,8 Therefore, there is a need for establishing proper perioperative methods to prevent SSIs in general and specifically during abdominal surgeries.
While systemic perioperative antibiotics are often administered as an SSI prophylaxis, this approach is limited due to insufficient antibiotic concentrations in the surgery site following damage to the local vasculature, adverse events resulting from systemic toxicity, and the formation of biofilm in the site of the surgery. 9,10 To avoid these limitations, methods for local administration of antibiotics have been established, with the ability of sustained delivery to allow the presence of a constant amount of antibiotic in the surgical defect for long durations. 11 Polymethylmethacrylate cement beads were the first polymeric carrier systems for local sustained antibiotic release. However, while providing longevity and structural support, they are nonabsorbable, used in osseous-related indications, and require an additional surgery for their removal. 12,13 In contrast, biodegradable vehicles do not require further surgery for their removal and provide prolonged action while avoiding the potential formation of a biofilm layer on their surface. 13 Nevertheless, many of the biodegradable vehicles, such as hydrogels and collagen, result in nonconstant release rate of the antibiotics, and some of them can lead to localized hypersensitivity reactions. 9,14,15
Recently, a new biodegradable polymer–lipid encapsulation matrix drug delivery platform (PLEX; PolyPid Ltd, Petah-Tikva, Israel) has been developed, which can be also utilized for local release of antibiotics. This matrix provides a continuous and prolonged release of antibiotics for 4 weeks, which is much longer than many other solutions such as hydrogels. The PLEX platform can be combined with nonheat-stable antibiotics, such as doxycycline (D-PLEX100), which is effective against a wide range of bacteria, including
Materials and Methods
Animal Husbandry and Maintenance
A total of 36 male and 36 female Yucatan miniature swine, 4- to 5-months old, weighing 16 to 25 kg, were supplied by Sinclair BioResources. The animals were housed in appropriate-sized cages, 1 animal per cage. The rooms were artificially lit for 12 hours each day. The study animals were acclimatized for at least 14 days before treatment initiation. The animals were supplied with water ad libitum and with certified miniswine diet (Teklad 7037C and/or 8753C; Envigo) once a day.
The study was conducted in Sinclair Research Center, LLC (Auxvasse, MO, USA), in accordance with all applicable sections of US Department of Agriculture (USDA) Policy 12. The study site is licensed by the USDA and accredited by the Association for Assessment and Accreditation of Laboratory Animal Care. Study procedures, including care of the animals, were reviewed and approved by the study site Institutional Animal Care and Use Committee prior to initiation of the exposure period.
Test Material
D-PLEX100, used in the present study, consists of tricalcium phosphate particles having a mean diameter of 50 to 100 µm and a polymer–lipid matrix encapsulating doxycycline. D-PLEX100 powder was supplied in a glass vial (5 g/vial). Prior to opening of the D-PLEX100 vials, each vial was shaken vigorously to achieve a homogenous powder. The appropriate amount of D-PLEX100 powder was hydrated with 0.9% sterile saline until a uniform wet powder appearance was achieved prior to administration to the incision site. D-PLEX100 was produced under Good manufacturing practices (GMP) conditions and tested for stability prior to administration.
Treatment Protocol
A total of 54 Yucatan miniature swine (n = 27/sex) were randomized into 3 cohorts with 3 treatment groups/cohort (groups 1-3; Table 1). On day 1 of the study, all animals were induced under general anesthesia. Each incision for groups 1, 2, and 3 was made to a depth that went through the peritoneum. The peritoneum was surgically closed using appropriate sutures. Group 1 animals received 2 incisions (sham control). Group 2 animals received a single lateral flank incision (∼10 cm) that was administered a single dose of D-PLEX100 at a dosage of ∼71 mg/kg (equivalent to the amount of one vial [5 g] of D-PLEX100 in 70 kg human). Group 3 animals received 2 lateral flank incisions (∼10 cm each), which were each administered a double dose (∼142 mg/kg) of D-PLEX100, resulting in a total dosage to the group 3 animals of ∼284 mg/kg, roughly the equivalent of 4 vials (20 g) of D-PLEX100 in a 70-kg human. D-PLEX100 was administered into the surgical site incision. The administration sites included the fascia suture line and the soft tissues of the abdominal wall along the whole length of the surgical wound. The soft tissues included the muscle, fat, and dermis but not the skin (suture line). The incisions were then closed without the use of a bandage. Each animal was monitored until it completely recovered from anesthesia and was then returned to standard housing for further follow-up and examinations until scheduled termination according to the assigned cohort. D-PLEX100 was supplied by PolyPid Ltd. Additional 18 animals (n = 9/sex) were used for the Toxicokinetics (TK) study. They were randomized to receive the same treatment as groups 2 and 3 (Table 1).
Experimental Design.

Abbreviations: D-PLEX100, doxycycline/polymer-lipid encapsulation matrix; NA, not applicable.
Clinical Observations and Mortality
In-cage observations for mortality/moribundity were made at least twice daily (morning and evening), and detailed clinical observations, including surgical treatment sites, were performed weekly. Physical examinations were made once prior to randomization and 3 days prior to scheduled termination. Physical examinations included general physical condition, skin, mucus membranes, eyes, ears, nose, and auscultation. Body weights were collected prior to randomization, once weekly, during the dosing phase, within 3 days of designated scheduled termination, and at designated scheduled termination per cohort. The brain, adrenal gland, pituitary gland, thyroid gland, heart, kidneys, liver/gallbladder, ovaries/oviducts, spleen, testes, thymus, and uterus were weighed for all animals at the scheduled necropsy. Paired organs were weighed together. Organ-to-terminal body weight and organ-to-brain weight percentages were also calculated. Food consumption was determined daily (beginning at least by day 1 dosing phase) through designated scheduled termination. Clinical observations for any clinical signs of illness or reaction to treatment were performed weekly, starting prior to randomization through designated scheduled termination. Ophthalmology examinations using an indirect ophthalmoscope and slit lamp biomicroscope were performed once prior to randomization and at designated scheduled termination (within 3 days of scheduled termination/cohort).
Clinical Pathology and Toxicokinetics
Blood samples for clinical pathology analysis were collected once prior to randomization and at designated scheduled termination (within 3 days of scheduled termination/cohort) via direct venipuncture of the jugular vein. The blood was analyzed for hematological, coagulation, and biochemical parameters. Urine samples for urinalysis were collected from all animals at overnight room temperature via collection trays under each cage tray.
Blood samples (3-6 mL/time point) for TK were obtained from the TK study cohorts pre-dose 0.5, 1, 2, 4, 10, 12, 16, 24, 48, 72, and 96 hours after dosing, and on days 8, 15, 22, and 29. Blood samples for doxycycline analysis were not taken from the sham group. Plasma samples were assayed for doxycycline by Wuxi Apptec/XenoBiotic Laboratories, Inc, using a validated bioanalytical method. Pharmacokinetic analysis was performed using a validated copy of Phoenix WinNonlin 8.1 (Certara, L.P.).
Postmortem Procedures
Necropsy was performed on all animals, and organ weights were measured. The following tissues, when present, were collected from all animals: aorta, bone marrow (femur and sternum), bone (femur and sternum), brain, cervix, epididymis, esophagus, eyes, adrenal gland, lacrimal gland, mammary gland, pituitary gland, prostate, mandibular salivary gland, seminal vesicles, thyroid gland, gross lesions, heart, kidneys, cecum, colon, rectum, liver/gallbladder, lung/bronchus, mandibular and mesenteric lymph nodes, quadriceps femoris, optic and sciatic nerves, ovaries/oviducts, pancreas, abdominal skin, duodenum, ileum, jejunum, cervical, lumbar and thoracic spinal cord, spleen, stomach, testes, thymus, tongue, trachea, urinary bladder, uterus, and vagina. Tissues were preserved in 10% neutral-buffered formalin (NBF) except for the eyes and testes that were fixed in Davidson and Modified Davidson, respectively, for 24 to 48 hours and then stored in 10% NBF.
For incision/D-PLEX100 administration sites, full-thickness histopathology sections were prepared on 1 slide through the incision/D-PLEX100 site to verify whether there are any adhesions to parietal peritoneum beneath incision line. A semiquantitative evaluation of the amount of residual test article, local tissue effects, and the inflammatory response was performed. The amount of residual material was scored 0 to 4 based on abundance and frequency relative to the amount estimated to have been deposited at surgery (Table 2). All other parameters were graded semiquantitatively (Table 3). Parameters scored for assessment of the tissue response and healing included the following: fibrosis (along surgical planes, cutaneous, subcutaneous, and abdominal muscle), necrosis, neovascularization, peritoneal adhesions, suture-associated inflammation, surgical foreign microparticulate, and inflammatory cell types (neutrophils, eosinophils, lymphocytes, plasma cells, macrophages, giant cells, and phagocytic macrophages/giant cells specifically associated with residual test article). All parameters related to the surgical/implant site were summarized by group using means and averages. Statistical evaluation was not performed to the histology data (Supplementary Table 1, Histological scores for tissue responses and residual D-PLEX100 applied to abdominal incisions of miniature swine.).
Scoring of Residual D-PLEX in Tissue.

Abbreviation: D-PLEX, doxycycline/polymer-lipid encapsulation matrix.
Semiquantitative Grading of Pathological Findings.

Statistical Analysis
For statistical analysis, Bartlett test for homogeneity was performed with a significance level of 0.05. If the data were homogeneous (Bartlett test result
If the data were not homogeneous (Bartlett test result
Results
Mortality/Moribundity
On at least dosing phase days 127 through 132, one female animal from group 1 was observed with decreased food consumption, head tilt, and/or decreased activity. As a result of these abnormal findings, this animal was humanely euthanized on dosing phase day 132.
Periodically (dosing phase days 1 to 127), 1 male animal from group 2 was observed with decreased activity. Additionally, this animal was observed with decreased food consumption, head tilt, and continued decreased activity on dosing phase days 128 through 134. As a result of these abnormal findings, this animal was humanely euthanized on dosing phase day 134. In both cases, a specific cause of morbidity was not identified, but as both animals, a control and a test animal, died with similar clinical signs, the cause of death was considered unrelated to the surgical procedure or D-PLEX100 administration.
Clinical Observations
Except for the early deaths, there was no mortality or morbidity cases during the study, and no abnormal clinical signs were found. Body weights for male and female animals were similar for all study groups. In general, animal body weights were maintained or had a slight increase following surgery and then began to increase, as expected, through termination. There were no D-PLEX100-related effects on body weight or body weight change between treated and sham groups animals. There were no D-PLEX100-related findings with clinical or ophthalmologic observations.
Clinical Pathology
Following the initiation of treatment, there were no treatment-related differences for any hematology, clinical chemistry, coagulation, or urinalysis parameters between treated and control groups. Statistical analysis indicated a few isolated findings. These findings were minor and consistent with normal biologic variation and not considered to be related to test article exposure.
Toxicokinetics
After the administration of a single 71 mg/kg (single incision) or 284 mg/kg (142 mg/kg × 2 incisions) dose of D-PLEX100, plasma doxycycline concentrations were evident at 30 minutes and persisted to 8 days post dose at 71 mg/kg and to at least 15 days at 284 mg/kg. Plasma doxycycline exposure was similar between male and female swine. Doxycycline elimination half-life was approximately 50 hours in each group. There was a dose-related, but not dose proportional, increase in doxycycline mean Cmax (group 2: 101 ng/mL; group 3: 208 ng/mL). Variability in individual swine Cmax values ranged from approximately 25% to 40%. The median Tmax was 7.0- and 4.0 hours postdose for group 2 and group 3, respectively, (range: 4.0-16.0 hours). The mean elimination half-life was extended at approximately 50 hours in each group. The increase in doxycycline mean area under the curve (AUC)last was dose proportional at approximately 4 times greater in the group 3 swine than in group 2 (group 2: 2720 ± 487 hours × ng/mL; group 3: 12,200 ± 3400 hours × ng/mL) as well as the AUC0-∞ (group 2: 2900 ± 591 hours × ng/mL; group 3: 12,200 ± 3400 hours × ng/mL). Distribution volumes, based on the t1/2, and plasma clearances were similar between the 2 dose levels. This suggests that, across the dose range of 71 to 284 mg/kg, there is no saturation of doxycycline elimination/metabolic pathways. There was no evidence of a dose-dependent change in doxycycline Tmax Doxycycline was no longer present in plasma by day 29; therefore, post-day 29 TK blood samples were not assayed for doxycycline. Summary of the TK results are shown in Table 4.
Mean Doxycycline Toxicokinetic Parameters.a
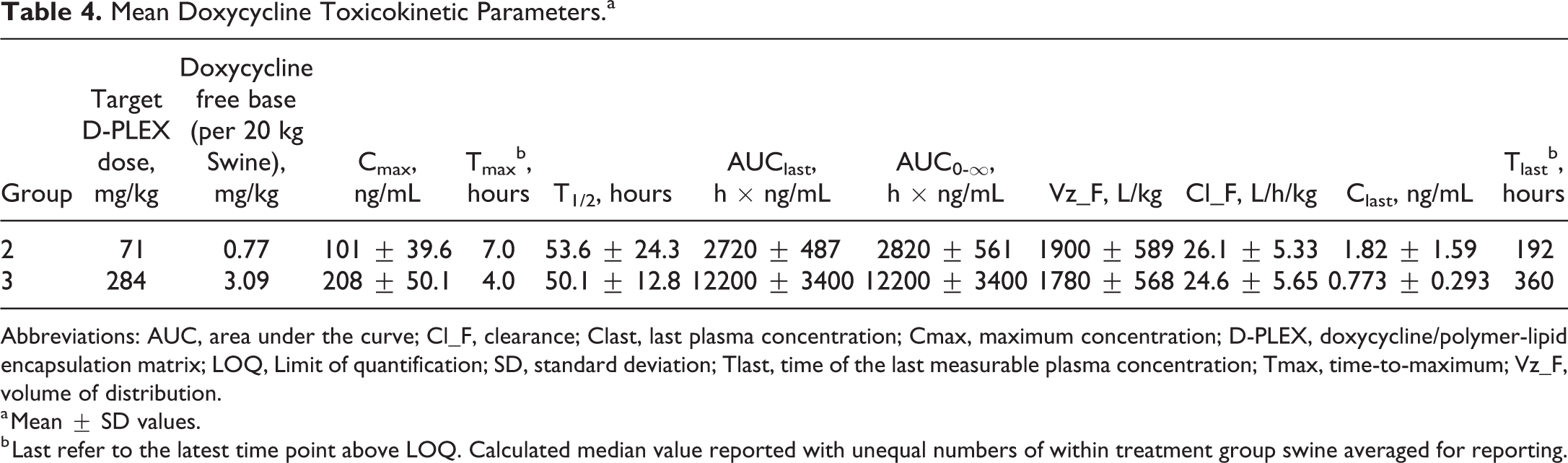
Abbreviations: AUC, area under the curve; Cl_F, clearance; Clast, last plasma concentration; Cmax, maximum concentration; D-PLEX, doxycycline/polymer-lipid encapsulation matrix; LOQ, Limit of quantification; SD, standard deviation; Tlast, time of the last measurable plasma concentration; Tmax, time-to-maximum; Vz_F, volume of distribution.
a Mean ± SD values.
b Last refer to the latest time point above LOQ. Calculated median value reported with unequal numbers of within treatment group swine averaged for reporting.
Organ Weights
There were no major differences between treated and control animals of either gender for any organ weights.
Macroscopic Observations
Abnormal consistency of the lungs was observed in a single group 1 male (3-month cohort), a single group 2 male (1-month cohort), and 2 group 3 females (1-month and 3-month cohorts). In the male animals, these observations correlated with minimal to mild, acute to chronic, and multifocal inflammation. This change is pathologically benign and coincidental. In the females, there was no microscopic correlation. Adhesions were noted grossly in a single group 1 female (3-month cohort) and a single group 3 female (1-month cohort). There were no D-PLEX100-related observations to gross pathology findings.
Microscopic Observations
The histological scores for all parameters were similar in males and females, and the scores were combined for the sexes (Supplemental Table 1) for comparison and interpretation of the mean group scores for the tissue responses over time. In Group 2 (G2) and Group 3 (G3) residual test article group, mean scores were 1.5 and 1.8 at 1 month, 0.8 and 1 at 3 months, and 0 at 6 months, respectively. Calcified material attributed to residual D-PLEX100 paralleled the residual test article scores in G2 and G3 at 1 month (1.5 and 1.7, respectively) and 3 months (0.8 and 0.3, respectively). Calcified material scored 0.2 for both G2 and G3 at 6 months when D-PLEX100 was scored as 0. By 3 months, fibrosis scores decreased to 0.8 and 1 for these respective groups, and the scores in both groups was 0 by 6 months. Neovascularization scores were similar (range 1 to 1.2) for sham and both dose groups at 1 month, but neovascularization was absent at the subsequent 3- and 6-month time points. Necrosis was rare with a score of 0.2 only for G2 at 3 months. At 1 month, lymphocytes, macrophages, giant cells, and phagocytic cells were the predominant inflammatory cells in G2 and G3, scoring 0.8 for lymphocytes and 1 for all other cell types. A few eosinophils (score = 0.2) were only found in G2. The sham group scored 0.5 for lymphocytes, 0.3 for macrophages, and 0.1 for giant cells with no phagocytic cells. This indicated that the surgical procedure itself was partially responsible for the inflammatory response. By 3 months, inflammatory scores in G2 were slightly increased compared to 1 month for lymphocytes and macrophages (1 and 1.7) and similar for giant cells and decreased to 0.2 for phagocytic cells. Additional inflammatory cells including neutrophils (0.5), eosinophils (0.7), and plasma cells (0.3) were present in G2. A similar increase in number of and variety of inflammatory cells also occurred in G3 between 1 and 3 months, including neutrophils (0.1), eosinophils (0.1), lymphocytes (0.7), plasma cells (0.1), and giant cells (1), except that no phagocytic cells were present at 3 months. By 6 months, the number of and variety of inflammatory cells had again shifted to predominately lymphocytes (0.3 for G2, 0.4 for G3), macrophages (0.3 for G2, 0.8 for G3), and giant cells (0.3 for G2, 0.8 for G3), but phagocytic cell scores were slightly increased to 0.3 and 0.8 for G2 and G3, respectively, by 6 months. A few eosinophils were in G3 (0.1) but not G2 at 6 months. Only a few inflammatory cells (0.1 for each of lymphocytes, macrophages, and phagocytic cells) were in the sham groups at 3 months, and by 6 months no inflammatory cells were present. Although the amount of residual test article steadily decreased over time, neovascularization was entirely resolved by 3 months, and fibrosis was similar at 1 and 3 months before finally decreasing by 6 months. The inflammatory cell response was as expected for a foreign body response with a peak in the number of, and an expanded variety of inflammatory cells at 3 months but only lymphocytes, macrophages, and cells predominating at 1 and 6 months. Conversely, phagocytic cells were highest at 1 and 6 months with the nadir at 3 months. The general increase in inflammatory cells at 3 months versus 1 and 6 months may have masked the ability to identify phagocytosed material in macrophages and giant cells. Foreign particulates, suture-related microgranulomas, and peritoneal adhesions from the surgical procedure occurred without a specific relationship to any groups or time points. Overall, the semiquantitative evaluation did not identify any substantive differences in the tissue responses and biodegradation of D-PLEX100 delivered at ∼71 or 284 mg/kg, and over time the peak inflammatory response was at 3 months post surgery. Additionally, there were no test article–related changes in any of the systemic tissues examined microscopically.
Overall, the healing response at the treatment site (i.e., healing surgical plane) was comparable in control and treated groups at all time periods, and the morphology was characterized by a progressive fibrocellular response that resulted in all groups in fibrous bridging of the incision plane (Figure 1). Tissue healing was accompanied by a low-severity chronic inflammatory response that was primarily associated with the test article at 30 days and decreased overtime concurrently with decreasing amounts of test article. These changes were indicative on normal healing. It was observed that D-PLEX100 remained in the incision site until its full elimination.
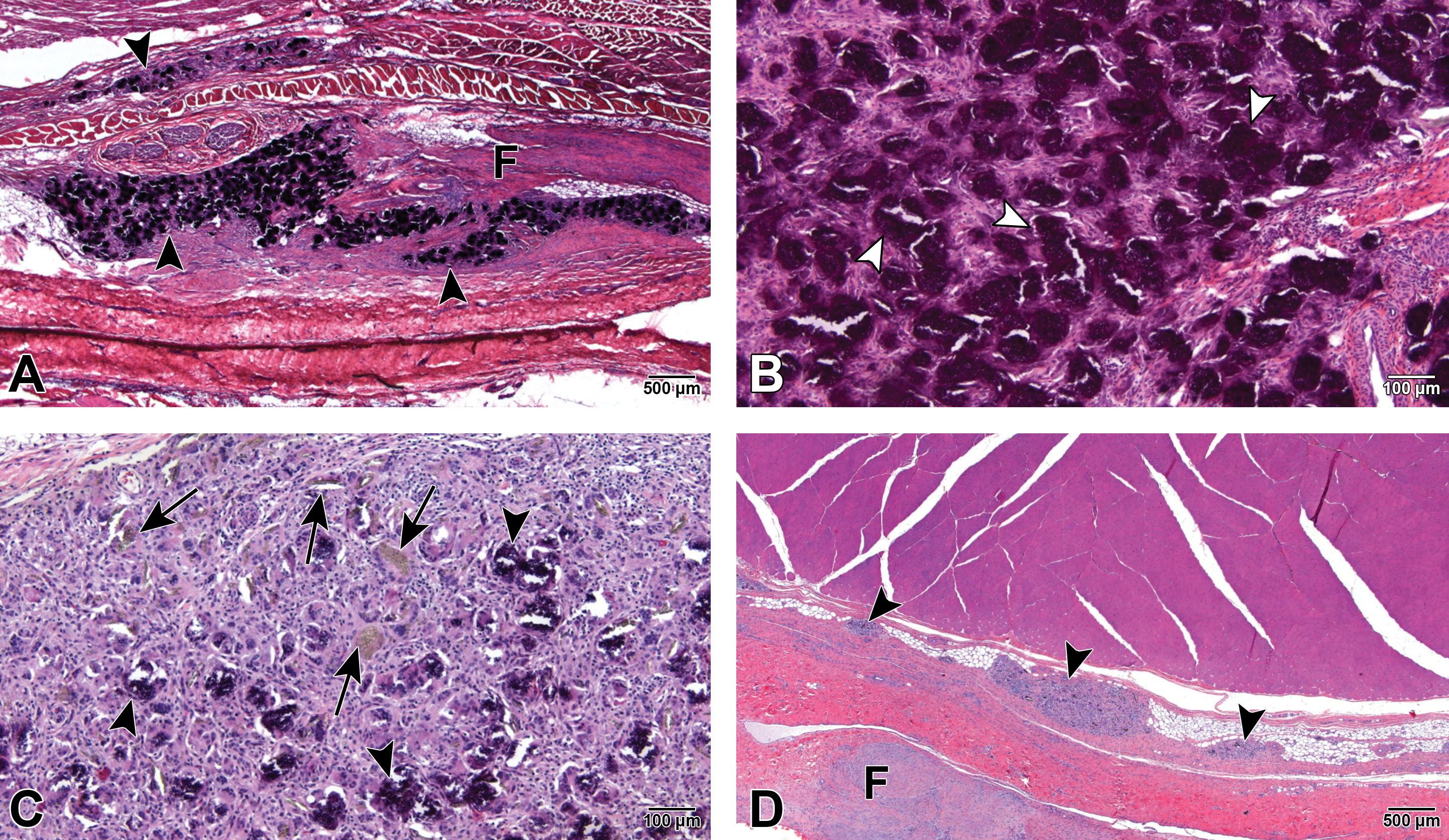
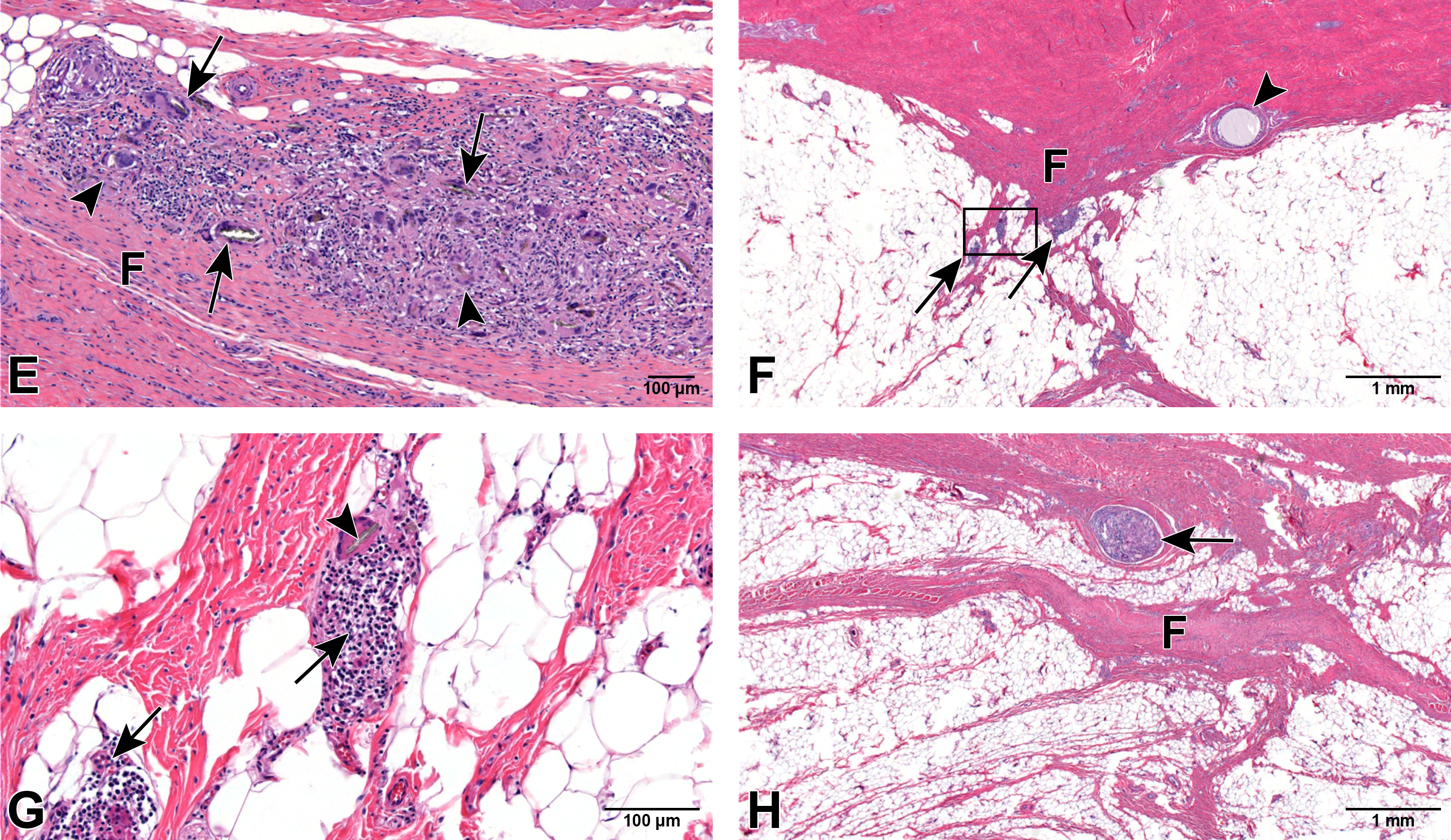
A, Section from the left incision site from a male group 2 animal (D-PLEX [∼71 mg/kg]), 1-month termination group. Arrowheads = foci of chronic inflammation associated with moderate residual test article that is frequently calcified (dark blue); F = mild localized fibrosis in the subcutaneous tissue. Stained with H&E. B, Higher magnification of the left incision site from a male group 2 animal (D-PLEX [∼71 mg/kg]), 1-month termination group. Arrowheads = calcified residual D-PLEX. Stained with H&E. C, section from the left incision site from a female group 2 animal (D-PLEX [∼71 mg/kg]), 1-month termination group. Arrows = clusters of birefringent granular material, consistent with mild residual D-PLEX, associated with slight chronic foreign body inflammatory response; arrowheads = calcified residual D-PLEX. Stained with H&E. D, Section from the left incision site from a male group 2 animal (D-PLEX [∼71 mg/kg]), 3-month termination group. Arrowheads = rare clusters of birefringent granular material, consistent with minimal residual D-PLEX, associated with slight chronic foreign body inflammatory response; F = mild fibrosis within the subcutaneous tissue. Stained with H&E. E, Section from the left incision site from a male group 2 animal (D-PLEX [∼71 mg/kg]), 3-month termination group. Arrows = clusters of birefringent granular material, consistent with minimal residual D-PLEX, associated with slight chronic foreign body inflammatory response (arrowheads); F = mild fibrosis within the subcutaneous tissue. Stained with H&E. F, Section from the left incision site from a female group 2 animal (D-PLEX [∼71 mg/kg]), 6-month termination group. Arrowhead = minimal microgranulomatous inflammatory response associated with degrading suture material; F = slight fibrous tissue scar along healed incision plane; arrows = minimal residual chronic inflammatory cell aggregates along the surgical plane; black rectangle = area detailed in next figure. Stained with H&E. G, higher magnification of the black rectangle in F. Arrowhead = elongated birefringent microparticulate consistent with minute amount of residual D-PLEX encased within a foreign body giant cell; arrows = slight chronic lymphohistiocytic inflammatory response. H, Section from the left incision site from a male Group 1 animal (sham control), 6-month termination group. Arrow = minimal microgranulomatous inflammatory response associated with degrading suture material. F = slight fibrous tissue scar along healed incision plane. Stained with H&E. D-PLEX indicates doxycycline/polymer-lipid encapsulation matrix; H&E, hematoxylin and eosin.
Discussion
The use of biomaterials for infection prophylaxis has been studied extensively in recent years in an effort to find the compound that can provide the best local delivery of antibiotics with the best safety characteristics. The first polymeric material that has been used clinically for this purpose was poly(methylmethacrylate) (PMMA) that can be administered as beads to prevent infection of large soft tissue wounds, acute Osteomyelitis, and open fractures, in addition to traditional applications, such as persistent and acute prosthetic joint infections, nonunions, and chronic osteomyelitis. 24 –26 However, use of PMMA has several drawbacks, including the fact that the release of antibiotics is not complete, 27 and the fact that local levels of antibiotics are decreasing rapidly. 9 However, the main drawback of PMMA is that it is not degradable, thereby posing a risk for foreign body colonization by bacteria. Therefore, the use of PMMA requires a follow-up surgery to remove it from the tissue, a procedure that carries an additional risk for SSI 12 in addition to the risk of tissue toxicity from unreacted methacrylate. 28
An approach to eliminate the need for a follow-up surgery to remove the antibiotic-secreting biomaterial is to use biodegradable compounds. For example, collagen has been utilized as a biodegradable material; however, its use was accompanied by the nonmatching release times and degradation rates and can rarely result in local hypersensitivity reactions
15
and even was associated with elevated SSIs rate when used as gentamicin collagen sponge.
29
To avoid the adverse biological responses, synthetic biodegradable implants have been developed, such as poly(D,
Similar to the administration of D-PLEX100 in the rabbit sternum soft tissue, 20 administration of D-PLEX100 to the abdominal surgical incision sites of Yucatan miniature swine was evaluated for up to 6 months and produced no evidence of systemic toxicity and no safety concerns. Surgical incision sites, including the peritoneal surface, were fully healed at 6 months in the sham control (group 1) and D-PLEX100 groups (groups 2 and 3). By 6 months, D-PLEX100 was considered essentially fully absorbed (score 0) meaning per scoring definition that test article was not found or was rarely observed as isolated particles within giant cells (trace amounts), and most of the mass was absorbed already after 3 months (score 1). The trace amounts of materials detected at 6 months were no more prominent than iatrogenic microparticulate of surgical origin. Therefore, D-PLEX100 was considered essentially absorbed at 6 months.
Although inflammation was evident in the incision site of the animals from groups 2 and 3, the changes observed were not considered to be adverse, as they were of low severity (generally score 1, rarely 2), not dose dependent, and represented the expected phagocytic and foreign body response to D-PLEX100 as seen also for other biodegradable materials and in other species. 36 There was a fibrotic response that was consistent with healing from the surgical procedure. Calcification of the test article observed some what prominently at 1 month decreased gradually overtime concurrently with biomaterial absorption with no associated adverse effect on tissue healing. Furthermore, the response remained of low severity and showed no untoward effect on the integrity of the healed incision and was not considered to be clinically meaningful.
A moderate pyogranulomatous response was observed in one female from group 2 at 3 months. However, due to its isolated occurrence at the mid-dose only (group 2), this unusual response may represent an idiosyncratic allergic reaction or more likely could be due to a bacterial contamination of the surgical field during the administration procedure and is not likely to be toxicologically relevant. Similar granulomatous responses have been reported in association with polymer-coated stents and are not unusual in preclinical stent studies in the swine species and are generally regarded as a limitation of the model. 37 –39
The current study, together with the previous study performed in sternal surgical defect model in rabbits, further supports the safety of D-PLEX100 and its favorable degradability profile. Further research is warranted to evaluate the safety and degradability of D-PLEX100 also in humans.
Supplementary Materials
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320928902 - D-PLEX100 in an Abdominal Surgery Incision Model in Miniature Swine: Safety Study
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320928902 for D-PLEX100 in an Abdominal Surgery Incision Model in Miniature Swine: Safety Study by Yuval Ramot, Shlomo Nedvetzki, Sefi Rosenfeld, Serge D. Rousselle, Abraham Nyska and Noam Emanuel in Toxicologic Pathology
Footnotes
Authors’ Note
Yuval Ramot and Shlomo Nedvetzki contributed equally to this work.
Acknowledgments
The contribution of Mr Timothy J. Madsen, Sinclair Research Center, LLC (SRC), MO, USA, is deeply appreciated for the management and evaluation of the study. The authors would like to thank Dalit Hazan and Tzvia Selzer (Polypid Ltd) for manuscript review and revision.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Shlomo Nedvetzki, Sefi Rosenfeld, and Noam Emanuel are employees of PolyPid Ltd. Abraham Nyska is a consultant to Polypid.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by PolyPid Ltd, Petah-Tikva, Israel.
Supplementary Materials
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
