Abstract
Bacterial infections are a common complication after surgical procedures. Therefore, local delivery of antibiotics has been developed, including the use of biodegradable polymers. A newly developed product for prevention of surgical site infections is a polymer–lipid encapsulation matrix loaded with doxycycline, named D-PLEX100 (D-PLEX). We evaluated the toxicity and safety of D-PLEX using a sternal surgical defect model in rabbits. D-PLEX was tested with three different concentrations of doxycycline in comparison to sham-operated control after administration into the sternal surgical defect and on the ventral side of the sternum in New Zealand White (NZW) rabbits, following 15 months of exposure. No mortality or abnormal clinical findings were attributed to D-PLEX, and clinical pathology assays were normal. Histological examinations revealed no treatment-related adverse findings in any of the examined tissues, including the osseous and surrounding soft tissues. It has been shown that D-PLEX gradually degraded until complete disappearance after 9 months, and mainly during the first 3 months, in parallel to normal bone formation. In addition, the administration of D-PLEX did not affect sternal bone strength. This study adds to the growing data on preclinical safety studies utilizing biodegradable materials and provides information on the expected normal reaction to biodegradable materials in the sternum of NZW rabbits.
Surgical site infections (SSIs) are infections that involve the site of the surgical procedure and all the additional tissues that are involved in the surgery (ter Boo et al. 2015). They can be divided into superficial or deep incisional, or organ or deep-space infections (Pham et al. 2016). According to the U.S. Centers for Disease Control, SSIs were the second most common reason for hospital-acquired infections in 2002 and occurred in 2% of the surgical procedures (Klevens et al. 2007). The rate of SSIs and the incidence in which multidrug resistant pathogens are involved continue to increase, resulting in a major threat to the economy and the public health (Badia et al. 2017). In addition, SSIs result in a steep increase in morbidity and mortality following surgery (Kirkland et al. 1999) and can lead to delayed wound healing (ter Boo et al. 2015). Sternal wound infection is an example of an SSI with devastating complications, resulting in a 10.7% increase in 1-year mortality rate, longer hospitalization time, and higher costs (Sears et al. 2016). The incidence of such infections can reach more than 5% of sternal operations, which are being performed mainly for cardiac surgeries (Andreas et al. 2017; Calderwood et al. 2017; Reineke et al. 2018; Schimmer et al. 2017).
One of the key measures to prevent SSIs is perioperative administration of intravenous antimicrobial agents (Mangram et al. 1999). However, the ability of this approach to prevent SSI is compromised by damage during surgery to local vasculature resulting in insufficient local antibiotic concentrations (ter Boo et al. 2015). Increasing the dose of the administered antibiotics is not an appropriate approach due to systemic toxicity problems (ter Boo et al. 2015). Other factors that affect the efficacy of preventive perioperative systemic antibiotic administration include the formation of biofilms in the wound bed by the pathogens and the increasing rate of antibiotic resistance (Bayramov and Neff 2017; Harbarth and Samore 2005; May et al. 1998). In order to overcome some of these obstacles, ways for local administration of antibiotics have been developed. With such an approach, it is possible to better control the rate of antibiotic delivery and provide high drug concentrations in the surgical defect with low systemic exposure (Darouiche 2003; Richards et al. 2012). To provide a continuous supply of antibiotic coverage to the surgical defect site, sustained delivery systems have been developed. The first polymeric biomaterial to provide such bacterial defense was poly(methyl methacrylate) (PMMA) bone cement (Buchholz and Engelbrecht 1970). However, PMMA has problematic release patterns and it is not degradable, thus requiring a follow-up surgery to remove it from the body (Griffis et al. 2009). An optional method is to use antibiotic-loaded collagen, which is a degradable product, and is available commercially (Kilian et al. 2009). However, it presents nonmatching release times and degradation rates, in addition to rare biological responses that can result in localized hypersensitivity reactions (El-Husseiny et al. 2011; Friess 1998). Hydrogels, which are polymeric compounds with a high-water content, have been used for loading of antibiotics. Although they are easy to apply to the surgical defect, their use can result in nonconstant release of the antibiotics (ter Boo et al. 2015). Therefore, there is a need for development of new and improved systems for local antibiotic delivery, which are based on biodegradable polymers that can provide sustained release of the antibiotic without resulting in local side effects reactions (ter Boo et al. 2015).
A new biodegradable material that can be used for local antibiotic release is polymer–lipid encapsulation matrix (PLEX; PolyPid, Petah Tikva, Israel). This new technology allows a prolonged and continuous release of antibiotics for several weeks and even months, that is, much longer than known for hydrogels, and can be used together with non-heat-stable antibiotics, such as doxycycline (D-PLEX), which is known to be active against a broad spectrum of bacteria including staphylococci (Barger, Fragomen, and Rozbruch 2017). PLEX has been used before for controlled release of doxycycline and the antimicrobial peptide OP-145 (de Breij et al. 2016; Metsemakers et al. 2015). Nevertheless, there is no information on the safety of D-PLEX in preclinical animal models for long durations (Emanuel et al. 2012). Therefore, we performed this comprehensive study to assess the potential toxicity and safety of D-PLEX, specifically designed to continuously release doxycycline for a period of 3-4 weeks, administered into a sternal surgical defect (i.e., osseous and surrounding soft tissues) in New Zealand White (NZW) rabbits, following up to 15 months of exposure.
Materials and Methods
Animal Husbandry and Maintenance
A total of 80 male and 80 female rabbits (Oryctolagus cuniculus), 6- to 9-month-old, weighing 2.8 to 3.5 kg, were supplied by Covance (Denver, PA). The NZW rabbit was chosen for this study since it is a nonrodent animal model commonly accepted by the Food and Drug Administration (FDA) and other regulatory authorities for use in local tissue reaction studies, and is especially suitable for studies of bone infection/osteomyelitis (Patel et al. 2009), and has a large and strong enough sternum to create a suitable sternal surgical defect model that can withstand the surgical defect and the following animal activity throughout life (Wellisz et al. 2008). The animals were housed in appropriate-sized stainless steel cages with elevated wire mesh flooring, one animal per cage. The rooms were artificially lit for 12 hr each day. The study animals were acclimatized for at least 14 days before treatment initiation. The animals were supplied with water ad libitum and with Certified Lab Diet 5325 once a day at 125 g per animal. The study was conducted in accordance with the U.S. FDA GLP regulations, 21 CFR Part 58; USDA Policy #12 (March 25, 2011), U.S. Department of Agriculture; The American Veterinary Medical Association (AVMA) Guidelines for the Euthanasia of Animals: 2013 Edition, American Veterinary Medical Association; and Sinclair Research Center, LLC, Standard Operating Procedures Manual.
Test Material
D-PLEX consists of tricalcium phosphate particles (50–100 µm) with polymer–lipid and various concentrations of doxycycline formulation. D-PLEX powder was supplied in a glass vial (5 g/vial) by PolyPid Ltd., Petah Tikva, Israel.
Treatment Protocol
The NZW rabbits were randomized into five separate cohorts (1-, 3-, 6-, 9-, and 15-month cohorts), each containing a sham-operated control group and three treatment groups per cohort (Table 1). On day 1 of the surgical procedure, all animals were induced under general anesthesia, and a longitudinal incision was made over the sternum, exposing the bone. A median sternal surgical defect (∼30–40 mm long, ∼1.0–1.5 mm wide, and approximately ∼1.5 mm deep partial thickness defect) was made along the midline using a surgical bur. A target amount of 214 mg of D-PLEX (equivalent on a per body weight basis to human use of one vial [5 g] of D-PLEX in a 70 kg human subject) with various concentrations of doxycycline in the various dose groups was immediately administered into the defect sites of groups 2 to 4 animals, following defect creation. An additional amount of 107 mg of D-PLEX was administered on the top of the sternum, yielding a nominal dose amount of 321 mg of D-PLEX per 3 kg in each animal in groups 2 to 4. Following administration of the test material, the sternal muscles were surgically closed using sutures, followed by the skin and tissues overlying the dose site. Each animal was monitored until it completely recovered from anesthesia and was then returned to standard housing for further follow-up and examinations until scheduled termination according to the assigned cohort.
Experimental Design.
Note: D-PLEX = polymer–lipid encapsulation matrix loaded with doxycycline.
Clinical Observations and Mortality
General, in-cage observations for mortality/moribundity were made at least twice daily (morning and evening). Detailed clinical observations were made at least once prior to surgical procedures and dosing on day 1, daily for the first week following surgery (days 2–8), and then weekly thereafter through termination. The animals were observed for any clinical signs of illness or reaction to treatment. Body weights were measured at least once prior to day’s 1 surgical and dosing procedures and then weekly thereafter through termination. Animal food consumption was determined at least a week prior to surgery/dose administration then weekly through termination. All animals were offered a daily maintenance amount of feed appropriate for the size of the animal.
All animals from each cohort were given ophthalmology examinations once prior to dosing and then once prior to termination. Animals were examined using an indirect ophthalmoscope and slit lamp biomicroscope.
Clinical Pathology and Toxicokinetics
Blood samples (at least 1 ml/animal) for clinical pathology analysis were collected via appropriate vessel from each study animal once during acclimation and prior to termination. The vessels that were used for venipuncture include the central ear artery, marginal ear vein, jugular, and cephalic veins. The blood was analyzed for hematological, coagulation, and biochemical parameters.
Blood samples (∼1–2 ml/time point) for toxicokinetics were obtained from groups 2 to 4 from the 15-month cohort predose and 0.5 hr; 1 hr; 2 and 4 hr; 10 and 12 hr; and 16, 24, 48, 72, and 96 hr after dosing and on day 8, day 15, day 22, day 29, and day 91.
Postmortem Procedures
A complete necropsy was conducted on all animals and organ weights were measured. Tissues were preserved in 10% neutral-buffered formalin (NBF) except for the eyes and testes that were fixed in Davidson’s and Modified Davidson’s, respectively, for 24 to 48 hr and then stored in 10% NBF.
Following euthanasia, the sternum was collected en bloc with a border of the attached ribs. Using the landmark measurement determined during day 1 surgery procedures, the sternal defect was located. The approximate center of the defect was determined, and a 20 mm section of the central defect sternum was collected (with musculature removed as much as possible), stored under refrigerated conditions, and submitted for mechanical testing evaluation.
For the 15-month cohort, additional tissues from the parietal pleura, including, but not limited to, the diaphragm and inner aspect of the chest wall in the region of the rib articulation with the sternum, adjacent to the sternotomy site were collected and stored in 10% NBF, and submitted for histology evaluation. The remaining cranial and caudal aspects of each sternum (with musculature remaining intact) were collected, stored in 10% NBF, and submitted for histology evaluation.
For histopathology of the sternum (all study animals, all cohorts), following decalcification of the sternal dose site (cranial and caudal aspects), the sternal samples were trimmed to include the surgical defect and any surrounding soft tissues. Histological sections through these tissues were cut at 4- to 5-μm thick and stained with hematoxylin and eosin (H&E). The focus of the assessment was on the presence of new bone deposition and bone remodeling (i.e., bone healing) within the sternum, as well as the presence or absence of any residual test article within the sternum defect and surrounding soft tissues. The cellular infiltration in the surgical defect site was scored using a semiquantitative grading of five grades (0 = no change, 1 = minimal changes, 2 = mild changes, 3 = moderate changes, 4 = severe changes), taking into consideration the severity of the changes (Schafer et al. 2018). All other collected tissues (groups 1 and 4) and any gross lesions (groups 1–4) were embedded in paraffin, sectioned, stained with H&E, and examined microscopically.
Mechanical Testing of Bone Strength
For mechanical testing of bone strength, the 20 mm central sternal sample with section of defect included a border of the attached ribs. These specimens were geometrically similar in dimensions (length and cross-sectional area) which should eliminate the influence of the cranial and caudal aspects which might contain native bone and/or cartilage. Tensile testing was performed on the sternum samples by Exponent, Inc. (Philadelphia, PA). The sternal samples were tested under Good Laboratory Practices GLP conditions, using a three-point bending test, with the load at bone rupture/failure reported as the primary measure of bone strength.
Statistical Analysis
The statistical analysis was performed using JMP® 8.0.2.2 (SAS Institute Inc., Cary, NC). Data for each sex were analyzed separately. For each end point, treatment groups were compared to the sham control (group 1) using group pairwise comparisons. For each specified end point and for all collection intervals, Dunnett’s test was used to compare the sham control (group 1) with each treatment group (groups 2–4) to assess events related to the test article alone. Results of all pairwise comparisons were evaluated at the 0.05 significance level. All end points were analyzed using two-tailed tests unless otherwise indicated. Historical data indicate that leukocyte counts (total and differential) are not normally distributed; therefore, a log transformation was performed on these data prior to group pairwise comparisons.
Results
Mortality/Moribundity
Three rabbits required euthanasia and one animal died following day 1 surgery/dose administration events. These unscheduled deaths were not attributed to D-PLEX exposure. Instead, these animals were euthanized/died due to complications following the surgical procedure. These animals were replaced by reserve (extra) animals that had been acclimated per cohort. On day 446, one female from group 3 15-month cohort was observed with difficulty in breathing and was pale in color during blood collection procedures. Veterinary interventions were unsuccessful, and the animal died. The cause of death was attributed to a large hematoma that was concluded as iatrogenic, occurring during blood collection procedures. There were no macroscopic or microscopic findings in any of the tissues obtained from this animal related to the administration of D-PLEX nor any findings related to morbidity. This animal constituted the only mortality over the entire postsurgery exposure period.
Clinical Observations
The most common abnormal clinical finding for 1-month cohort animals was varying degrees of erythema and edema (slight, moderate, or severe) at the incision site of males and females from all study groups. The findings were expected due to the type of surgical procedure, and generally subsided after approximately four-week postsurgery. Additionally, there were infrequent findings of alopecia observed in males and females from most groups within the cohort which were considered incidental and not related to D-PLEX exposure.
For 3-month cohort animals, most of erythema and edema findings, which were attributed to normal tissue response following surgery, resolved within two- to three-weeks postsurgery. Alopecia in males and females from all study groups was more frequently noted within this cohort (3 month) as well as 6-, 9-, and 15-month cohorts, as compared to animals in the 1-month cohort, and was generally slight to moderate, located on the chest area, at or near the incision site. The alopecia findings were generally attributed to incidental animal behavior and not related to D-PLEX exposure. Also, erythema and edema findings were observed in animals from the 6-, 9-, and 15-month cohort following surgery, like 1- and 3-month cohorts, most of which had resolved within two weeks postsurgery.
On day 27, one female from group 4 15-month cohort was observed with a ∼4 cm × 3 cm wound extending from midsternum to the right axillary region. The wound was full-thickness with necrotic edges and appeared to be self-inflicted trauma. The wound was treated with debridement and sutures. The animal had no other significant wound-related findings for the remainder of the study.
Overall, all animals consumed sufficient food to maintain an adequate level of nutrition and remained healthy during the study. Statistical analysis indicated a few isolated findings which were not considered to be related to D-PLEX exposure.
Body weights for male and female animals were similar for all study groups. In general, animal body weights decreased following surgery and then began to increase approximately two weeks postsurgery and continued to increase through termination. The observed decrease in body weight corresponded to a decrease in daily food consumption following surgery. Statistical analysis indicated a few isolated findings which were not considered to be related to D-PLEX exposure.
Overall, there were few ocular findings. The only noteworthy findings were observed for animals in the 1- and 15-month cohorts. Ocular discharge was the most common finding observed for animals in the 1-month cohort but was not assessed to be greater than grade +2 at any time point (Supplementary Table 21). None of the abnormal findings for animals in the 1- or 15-month cohorts were associated with D-PLEX exposure. No animals from the 3-, 6-, or 9-month cohorts had abnormal ocular findings at any examination time point.
Clinical Pathology
Following the initiation of treatment, there were no statistically significant differences for any hematology or coagulation parameters between treated and control group animals in the 1- or 3-month cohorts. Although intergroup differences for isolated hematology and chemistry parameters were noted at designated termination events for the 6-, 9-, and 15-month cohorts, the findings were consistent with normal biologic variation, and not considered to be related to D-PLEX exposure (Supplementary Tables 1–20).
Toxicokinetics
Doxycycline T max was reached at 2.5 to 3.8 hr. Doxycycline t 1/2 was dose dependent. D-PLEX with 4.8 mg/kg doxycycline free base was associated with the greatest systemic doxycycline exposure, the greatest persistence of plasma doxycycline concentrations, with plasma doxycycline exposure up to 29-day postdose administration. C max was not dose proportional while area under the curve (AUC) was proportional. Summary of the TK results is shown in Table 2. At all three dose levels, there were no obvious sex differences in doxycycline systemic exposure. Therefore, sex-combined results were used for further reporting.
Mean Doxycycline Toxicokinetic Parameters (Mean ± SD Values).
Exposure expressed as AUC0-last increased in a dose-proportional manner, but C max showed a less than dose-proportional increase.
Organ Weights
Kidney weight and kidney-to-body weight ratio results were statistically significantly increased for the 6-month cohort group 2 animals relative to corresponding group 1 controls. Thymus weights and thymus to body and brain weight ratios were statistically significantly decreased in 15-month cohort groups 2 and 3 male animals relative to corresponding group 1 controls. As these differences did not follow a dose–response pattern and were not significant for corresponding groups 2 and 3 female animals, the findings are consistent with normal biologic variation, and not attributed to D-PLEX exposure.
Histopathology
At the examinations made at the sternal administration sites at both the 1- and 3-month time points, D-PLEX was observed microscopically at all three dose levels. D-PLEX was found to be associated with a cellular infiltration predominated by macrophages, multinucleated giant cells, and lymphocytes in the surrounding soft tissue and the sternum bone defect. Additionally, evidence of new bone formation that incorporated D-PLEX within the surgically created sternal defects has been detected (Figures 1 and 2, Table 3). Nevertheless, a dramatic decrease in the amount of product granules was observed after 3 months compared to the 1-month cohort animals. Trabeculae of new bone formed at the sternum by one month were characterized by the presence of woven bone (Figure 2). By three months, there was a decreased amount of new bone formation with remodeling as characterized by maturation of the previously deposited new bone and reestablishment of the cortex consistent with evidence of bone healing at the sternal defect (Figure 3). By the 6-month time point, residual D-PLEX was no longer present in the surrounding soft tissue in any of the sternum sections from males while residual D-PLEX was only present in limited quantities in one group 2 female, two group 3 females, and one group 4 female only in the sternum bone defect (Figure 4 and Table 3). No residual D-PLEX was present at the 9- and 15-month examinations (Figures 5 and 6, Table 3). By the 6-, 9-, and 15-month time points, no new bone formation was noted as bone remodeling appeared complete and the surgical sites healed for the administration sites in a similar fashion for the D-PLEX-treated sternotomy sites as compared to the sham sites (Figures 5 and 6, Table 3).
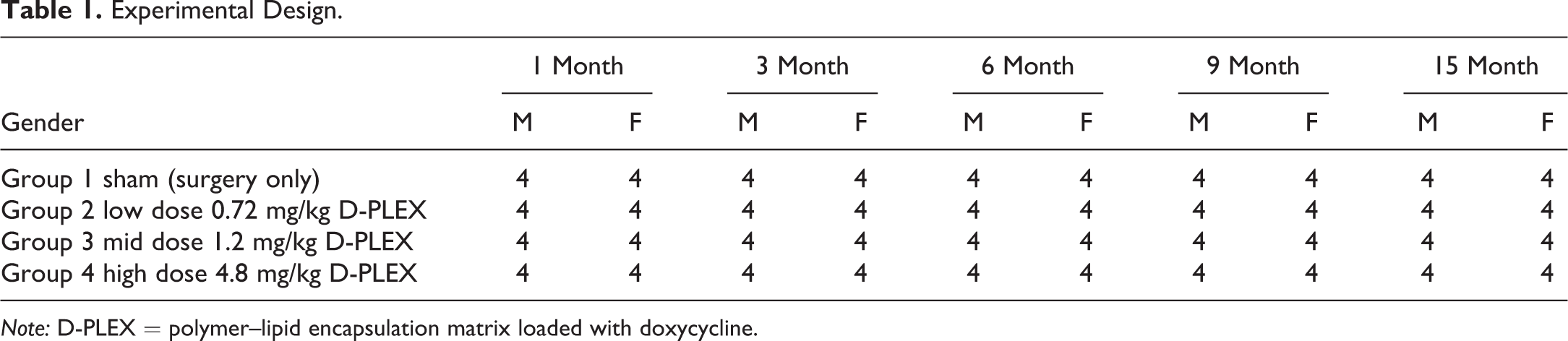
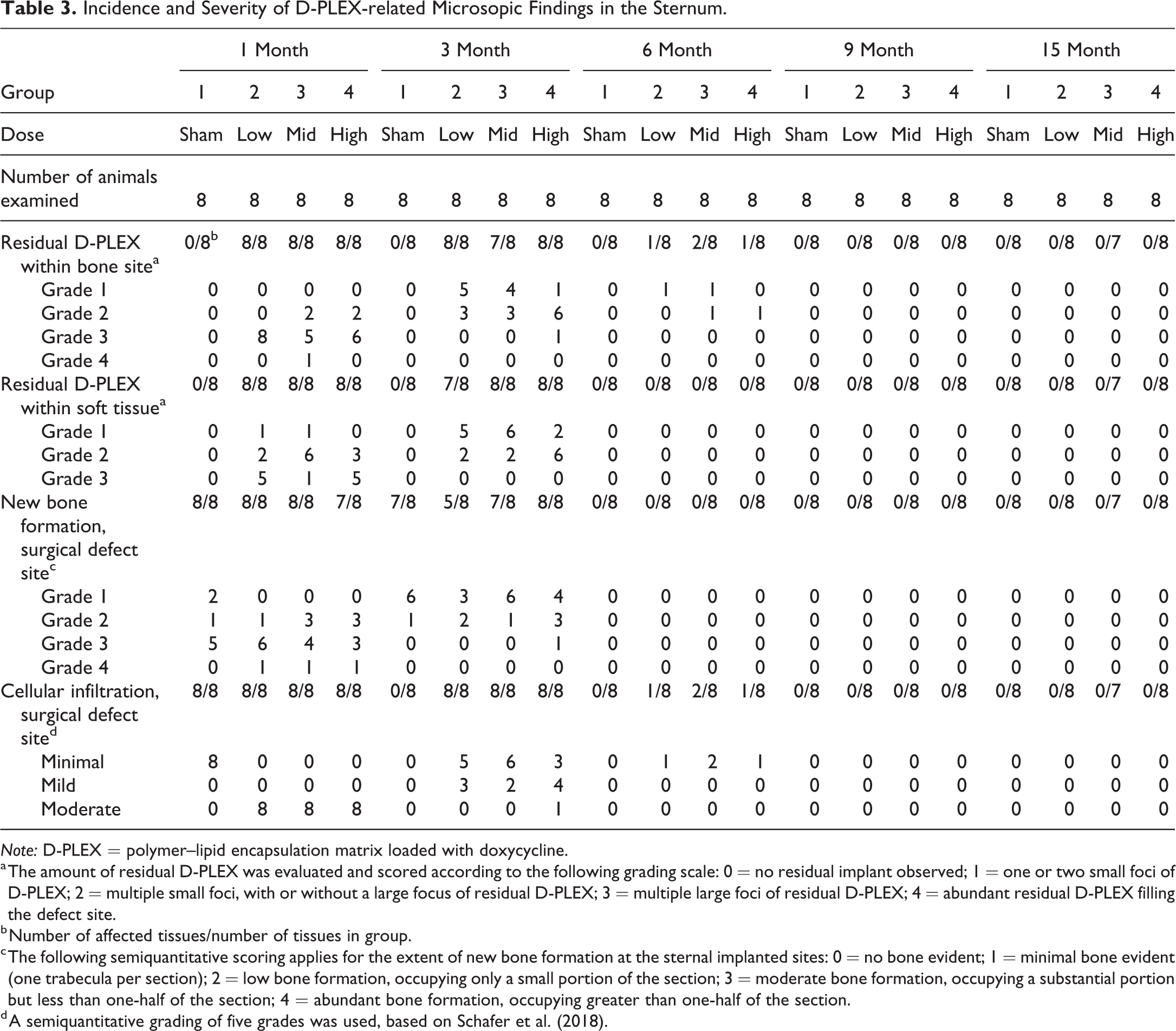
Section of the sternum from a female rabbit. Sham operated. 1-month postoperation. (A) The surgically operated defect area is indicated by double-headed arrow. Arrows indicate new bone formation. Asterisk indicates bone marrow. (B) A close-up image of the new bone trabeculae (asterisk) at the surgically created defect composed of woven bone lined by numerous osteoblasts (arrows).
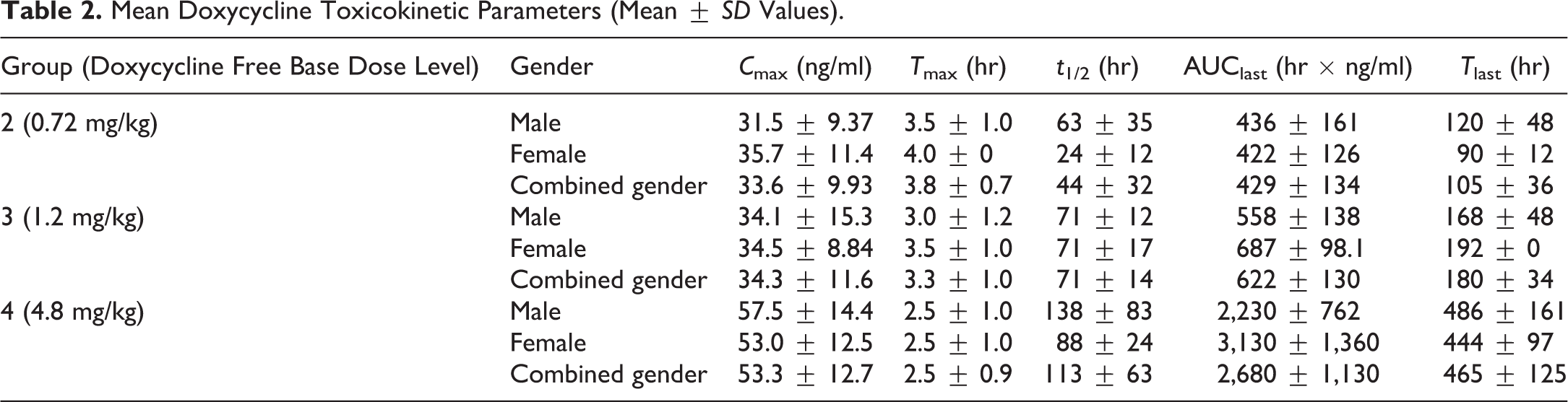
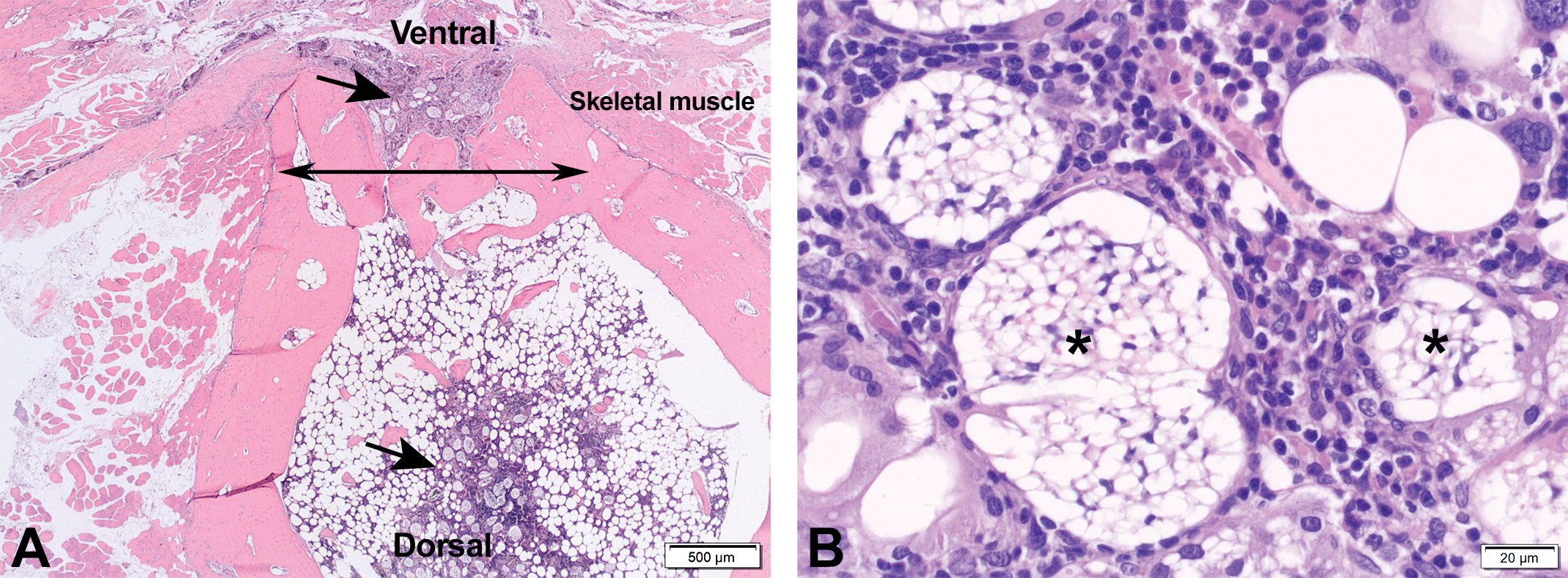
(A) Section of the sternum, implantation site, from a male rabbit from group 4 (high dose, 4.8 mg/kg D-PLEX), 1-month time point. Areas of residual D-PLEX (arrows), located at the region of surgically created sternal defect (i.e., across the double-headed arrow). (B) A close-up of the previous photo, showing the residual D-PLEX (arrow) associated with multinucleated giant cells, lymphocyte, and macrophage infiltrates in spaces surrounding the D-PLEX. D-PLEX = polymer–lipid encapsulation matrix loaded with doxycycline.
Incidence and Severity of D-PLEX-related Microsopic Findings in the Sternum.
Note: D-PLEX = polymer–lipid encapsulation matrix loaded with doxycycline.
a The amount of residual D-PLEX was evaluated and scored according to the following grading scale: 0 = no residual implant observed; 1 = one or two small foci of D-PLEX; 2 = multiple small foci, with or without a large focus of residual D-PLEX; 3 = multiple large foci of residual D-PLEX; 4 = abundant residual D-PLEX filling the defect site.
b Number of affected tissues/number of tissues in group.
c The following semiquantitative scoring applies for the extent of new bone formation at the sternal implanted sites: 0 = no bone evident; 1 = minimal bone evident (one trabecula per section); 2 = low bone formation, occupying only a small portion of the section; 3 = moderate bone formation, occupying a substantial portion but less than one-half of the section; 4 = abundant bone formation, occupying greater than one-half of the section.
d A semiquantitative grading of five grades was used, based on Schafer et al. (2018).
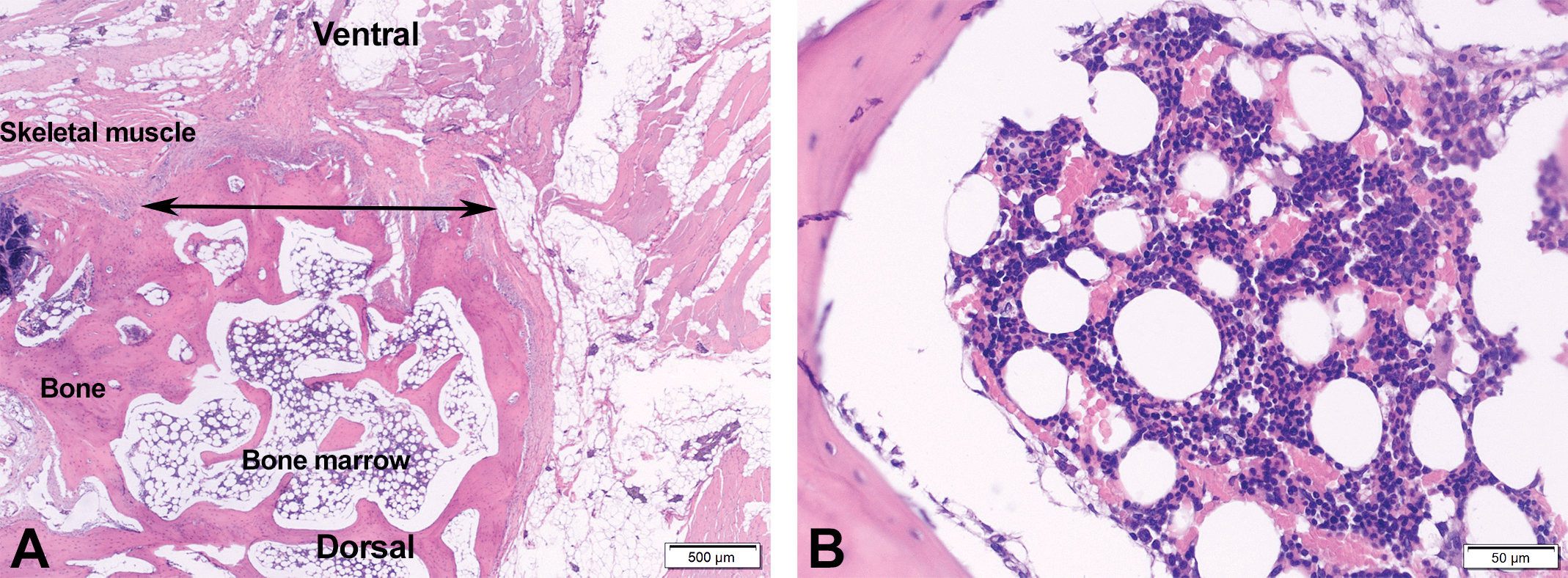
(A) Section of the sternum, implantation site, from a male rabbit from group 4 (high dose, 4.8 mg/kg D-PLEX), 3-month time point. Evidence of bone healing across the sternum surgical site (double-headed arrow) with foci of residual test article (arrows) within the marrow space and within the area of bone healing. (B) A close-up of the previous photo, showing the residual D-PLEX (asterisks) with associated cellular response. D-PLEX = polymer–lipid encapsulation matrix loaded with doxycycline.
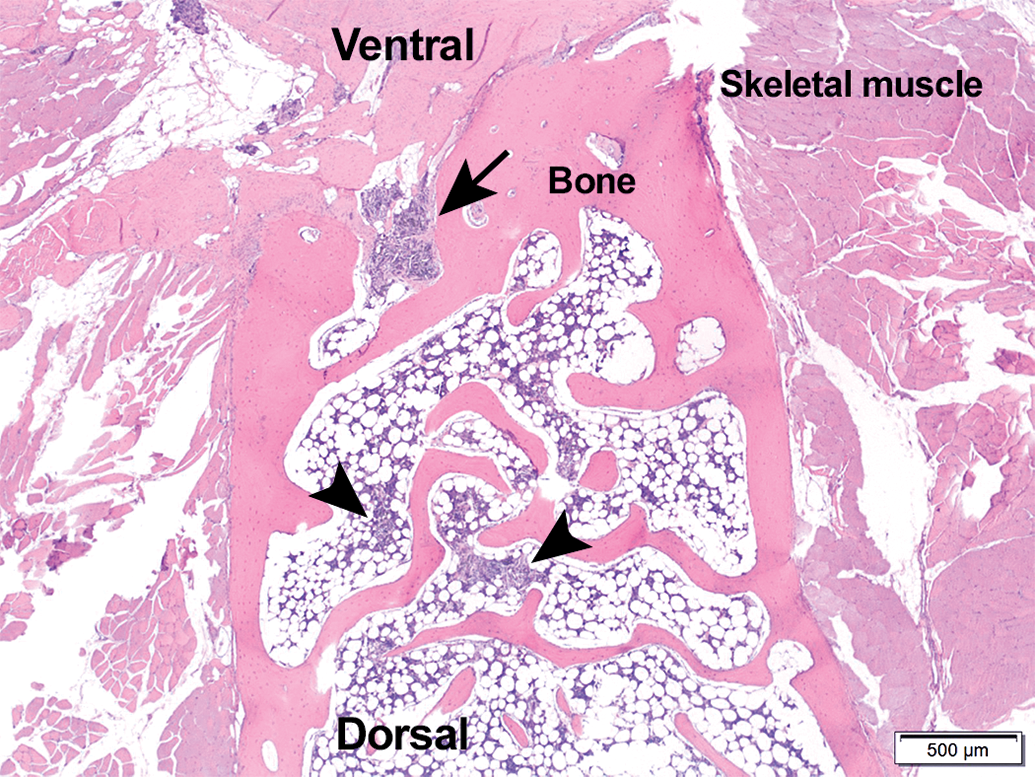
(A) Section of the sternum, implantation site, from a male rabbit from group 4 (high dose, 4.8 mg/kg D-PLEX), 6-month time point. Healed bone across the surgical site (double-headed arrow). No residual D-PLEX is apparent. (B) A close-up of the previous photo. Bone marrow—no residual D-PLEX is apparent. D-PLEX = polymer–lipid encapsulation matrix loaded with doxycycline.
Section of the sternum, implantation site, from a male rabbit from group 4 (high dose, 4.8 mg/kg D-PLEX), 9-month time point. Healed bone across surgical site. There is remaining foreign material (i.e., gauze/cotton fibers) associated with inflammation (arrow and arrowheads). The foreign material demonstrated birefringence using polarizing filters. D-PLEX = polymer–lipid encapsulation matrix loaded with doxycycline.
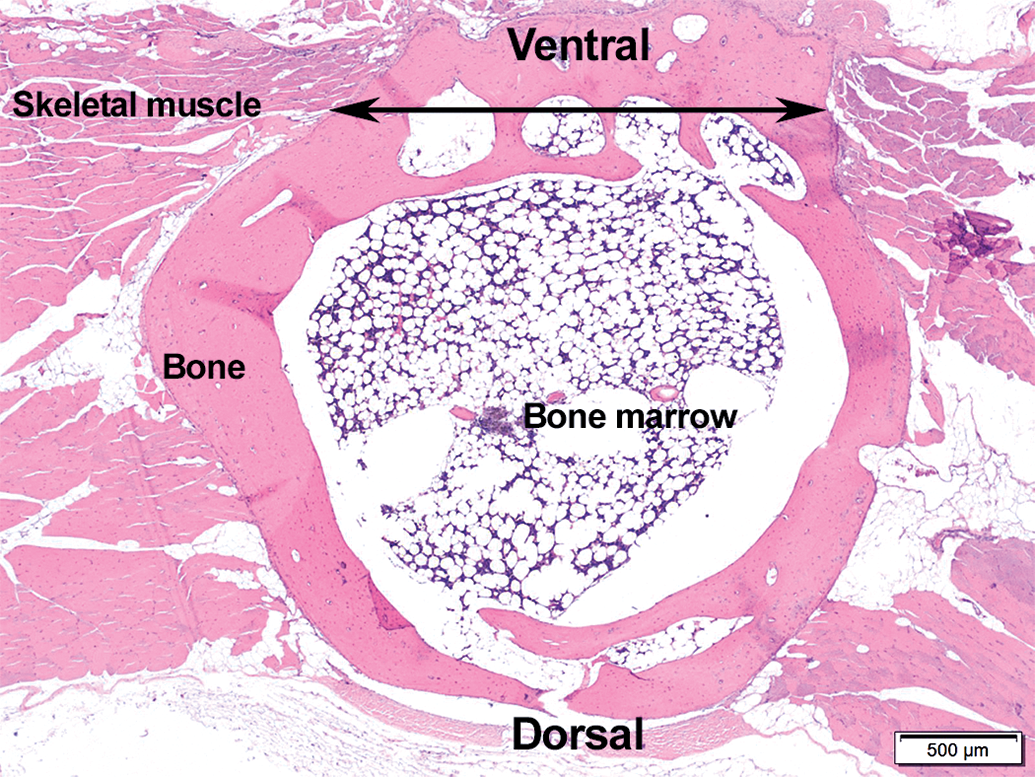
Section of the sternum, implantation site, from a male rabbit from group 4 (high dose, 4.8 mg/kg D-PLEX), 15-month time point. Healed bone across surgical site (double-headed arrow). No residual D-PLEX is apparent. D-PLEX = polymer–lipid encapsulation matrix loaded with doxycycline.
Occasionally, birefringent material was observed in some administration site specimens across all groups (sham control and D-PLEX-treated groups) and at all time points (Figures 5 and 7). This material was composed of small irregularly shaped debris, circular to elliptical to cylindrical structures, or particulates (often in areas of D-PLEX) that were most consistent with gauze/cotton fibers by bright field microscopy. This birefringent material induced a limited, focal foreign body-type response consisting of macrophages and/or multinucleated giant cells.
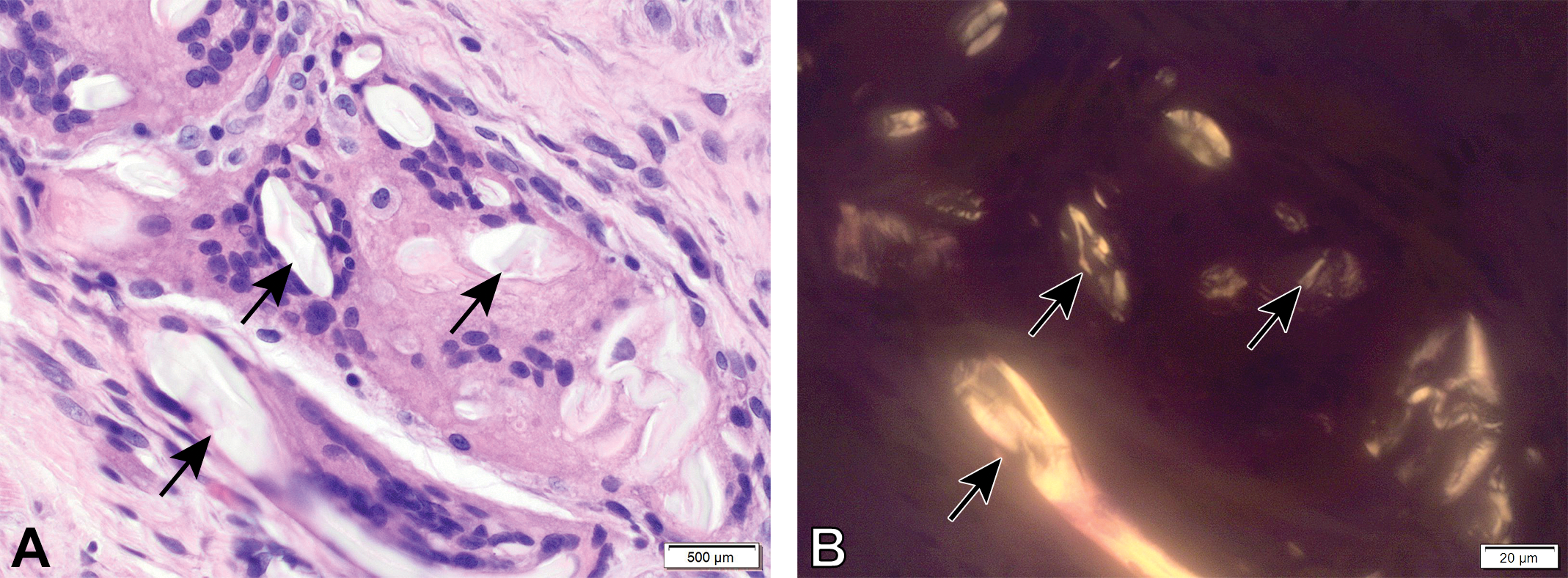
Section of the sternum from a male rabbit. Sham operated. 1-month postoperation. (A) Arrows indicate foreign material with foreign body response, located at the site of surgically operated defect. (B) Under polarization light, the foreign material is demonstrating birefringence, consistent with gauze/cotton fibers.
Mechanical Testing
Flexural strength values varied without a treatment-related trend and only occasional changes within the multiple comparisons made at the several cohorts attained statistical significance, but mostly in the direction of higher strength. Therefore, it was concluded that the administration of D-PLEX did not affect sternal bone strength after 15-month. Maximum flexural stress results are presented in Table 4.
Mean Maximum Flexural Stress.
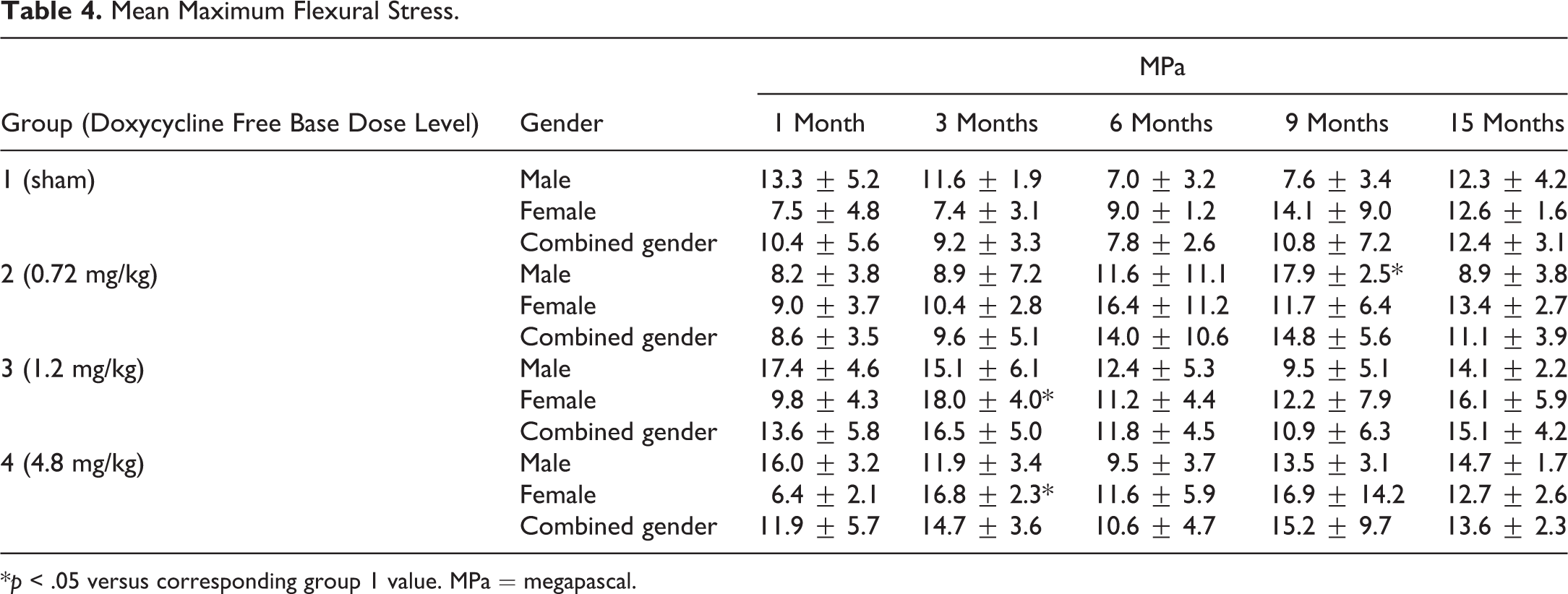
*p < .05 versus corresponding group 1 value. MPa = megapascal.
Discussion
Biodegradable drug delivery systems are gaining much popularity in different medical fields, and the number of such compounds that are in development is continually increasing. As a result, there is also an increase in the number of preclinical safety studies performed with such materials, developed for different administration and in a variety of animal models (Ramot, Nyska, et al. 2015 ; Ramot, Touitou, et al. 2015 ; Ramot, Haim-Zada, et al. 2016; Ramot, Rotkopf, et al. 2016; Stander et al. 2018; Barbosa Saliba et al. 2016; Fattal et al. 2014).
The study reported here shows the histopathological changes expected during the biodegradation of biocompatible matrix, specifically a novel PLEX, when administrated to sternal surgical defects. Here, we evaluated doxycycline-loaded PLEX intended for the prevention of SSI. Preclinical safety trials of antibiotic-releasing materials present a greater challenge than other materials, since a distinction should be made between possible adverse effects of the loaded antibiotics and the normal changes expected during the degradation process of the biodegradable matrix itself.
During the histopathological evaluation, gauze/cotton fibers were identified based on the typical appearance of the foreign material together with being birefringent. These fibers were introduced into the surgical defect at the time of surgery or subsequently during routine wound care (Ramot et al. 2008; Sari et al. 2006; Yeh et al. 1997). Making this diagnosis is of utmost importance during preclinical histopathological assessment, especially when evaluating biomaterials, which can by themselves result in a foreign-body-like reaction. Therefore, knowledge of the histopathological appearance of these materials, and the use of a birefringent microscope or in special cases even dedicated stains (e.g., in the case of suspected hair fibers) is crucial for the proper evaluation of the study.
Doxycycline mean T max was similar among all dose levels and ranged from 2.5 to 3.8 hr. Elimination half-life increased in a dose-dependent manner indicating nonlinear kinetics. However, the increase in elimination half-life with dose may be a bias related to the higher levels at the high dose which enable the detection of a slower elimination phase at the higher dose but an inability to detect this phase at the lower doses. It is worth noting that the terminal elimination phase is not a pure elimination phase, as it is masked by the long absorption (release) of doxycycline from the extended-release D-PLEX formulation into the central compartment (i.e., sternum surgical defect). It is therefore not surprising to see a dose-dependent increase in the terminal half-life under such conditions. This is in contrast to the expected elimination of doxycycline when administered as capsules in humans (Gschwend et al. 2007).
In conclusion, in this study, no deleterious effects on bone and soft tissue healing of the sternal surgical site and no systemic effects related to D-PLEX administration were observed. Based on these results, D-PLEX with 0.72, 1.2 and 4.8 mg/kg doxycycline free base when administered as a single dose into a sternal defect in NZW rabbits was determined to have no toxicity to both osseous and surrounding soft tissues for up to 15 months of follow-up. The safety of this product, however, still needs to be assessed in suitable clinical trials in humans before being approved for use.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623319837887 - Toxicity and Safety Study of D-PLEX100 in a Sternal Surgical Defect in New Zealand White Rabbits
Supplemental Material, DS1_TPX_10.1177_0192623319837887 for Toxicity and Safety Study of D-PLEX100 in a Sternal Surgical Defect in New Zealand White Rabbits by Yuval Ramot, Shlomo Nedvetzki, Sefi Rosenfeld, Noam Emanuel and Abraham Nyska in Toxicologic Pathology
Footnotes
Acknowledgments
The contribution of Mr. Timothy J. Madsen, Sinclair Research Center, LLC (SRC), MO, USA, and Dr. David S. Garlick, DVM, DACVP from HSRL, Mount Jackson, VA, USA, is deeply appreciated for the management and evaluation of the study. We would like to thanks Dalit Hazan and Tzvia Selzer (Polypid Ltd) for manuscript review and revision.
Author Contributions
Authors contributed to conception or design (SN, SR, NE, AN), data acquisition, analysis, or interpretation (YR, SN, SR, AN); drafting the manuscript (YR, SN, SR, NE, AN); and critically revising the manuscript (YR, SN, SR, NE, AN). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Shlomo Nedvetzki, Sefi Rosenfeld, and Noam Emanuel are employees of PolyPid Ltd. Abraham Nyska is a consultant to PolyPid.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by PolyPid Ltd., Petah Tikva, Israel.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
