Abstract
D-PLEX100 (D-PLEX) is a novel product candidate made of a polymer-lipid-based matrix (PLEX platform) which contains doxycycline that is being released at a constant rate for 30 days. D-PLEX was developed to prevent surgical site infections, which are a major global health challenge. Previous studies have shown its safety in adult humans, adult swine, and adult rabbits. The aim of this study was to assess the toxicity and safety of D-PLEX also in juvenile animals to support future clinical trials in pediatric patients. Yucatan miniature swine were selected as a model, primarily due to their relatively larger mass. D-PLEX or placebo (formulation without doxycycline) was administered locally to abdominal incisions, and the animal's safety parameters were followed for 9 months and compared to sham-control swine. There was no evidence of any systemic safety concern or local toxicity at the incision site in D-PLEX-treated animals. D-PLEX was detected after 1 month and was fully resorbed at the 3-month time point. The surgical incision sites were fully healed at the 6-month time point in all D-PLEX-treated animals. Toxicokinetic (TK) assessments revealed that doxycycline exhibited low Cmax and therefore minimal systemic exposure following a single dose of local administration. This study provides evidence for the safety of D-PLEX and PLEX-based formulation in juvenile miniature swine and supports its further testing in clinical pediatric population. In addition, it can be used as a reference for future preclinical studies aiming to evaluate the safety of other PLEX-based product candidates for the pediatric population.
Introduction
Surgical site infections (SSIs) are a major cause for mortality, increasing the risk by 11-fold, and represent a significant burden on the healthcare system, costing an estimated US$3.3 billion annually in the USA alone.1,2 SSIs can on average extend hospital stays by 9.7 days, resulting in an average of US$25,000 additional cost per case,1,3–5 and can delay postoperative treatment and worsen patient outcome. 6 To avoid the unwanted event of SSI, systemic antibiotic prophylaxis is often used. However, this method has limitations in its effectiveness since the local concentration of the given antibiotic at the surgical site may not reach the necessary levels, resulting in ineffective prophylaxis.7–9 The medical conditions known to be associated with lower local concentrations include peripheral vascular disease, obesity, and diabetes.9–12
To overcome this hurdle, various techniques for delivering antibiotics directly to the surgical site have been developed. Such a local drug delivery system is D-PLEX (PolyPid Ltd, Petah Tikva, Israel), which pairs an innovative Polymer-Lipid Encapsulation matriX (PLEX) technology platform with the broad-spectrum antibiotic, doxycycline. The PLEX matrix forms a protected drug reservoir and enables localized and continuous release of doxycycline for a period of 30 days. By that, D-PLEX can provide controlled antibacterial activity directly at the surgical site, while keeping systemic antibiotic exposure very low.9,13
The safety of D-PLEX in adult animals has been demonstrated previously in preclinical in vivo studies in a sternal surgical defect model in New Zealand white rabbits and in an abdominal surgical incision model in miniature swine. Full absorption of D-PLEX was observed after 9 and 6 months in both models, respectively.7,8 D-PLEX was also tested in adult patients, undergoing cardiac and colorectal surgeries, and was found to be safe in both types of surgery, and also effective in reducing superficial and deep SSIs in the colorectal surgery cohort.9,13 Nevertheless, the safety of D-PLEX in the pediatric population is still unknown. This information is particularly relevant because SSIs pose significant challenges in pediatric surgeries. Due to limited data, there is still uncertainty about the most effective prophylactic protocols.14,15 Even less is known about the use of local antibiotic prophylactic using biodegradable materials in preclinical models to evaluate their potential impact on pediatric development. It is for this reason that we performed this comprehensive study to assess the safety of D-PLEX in juvenile Yucatan miniature swine during 9 months of follow-up. The aim is to provide the groundwork for future clinical trials involving pediatric patients
Materials and Methods
Treatment Protocol
The study was conducted in Altasciences Preclinical Columbia, LLC, Missouri, USA, in compliance with the Food and Drug Administration (FDA) Good Laboratory Practice (GLP) regulations for Nonclinical Laboratory Studies (Title 21 of the Code of Federal Regulations, Part 58), as accepted by Regulatory Authorities throughout the European Union (OECD), Principles of Good Laboratory Practice, Japan (MHLW), and other countries that are signatories to the OECD Mutual Acceptance of Data Agreement. D-PLEX placebo (without Doxycycline) and D-PLEX were provided by PolyPid Ltd, Petah Tikva, Israel. D-PLEX is composed of β-tricalcium phosphate particles (50-100 μm) coated with a controlled release polymer-lipid-based matrix incorporating doxycycline hyclate.7,8
D-PLEX and Doxycycline Doses.
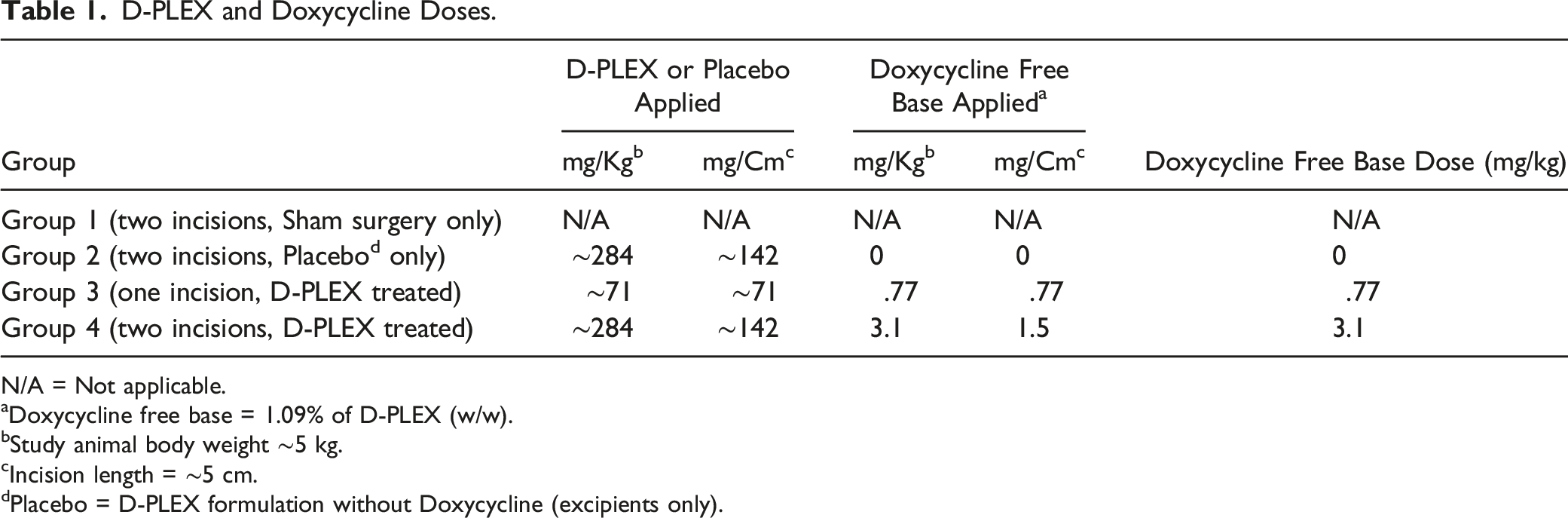
N/A = Not applicable.
aDoxycycline free base = 1.09% of D-PLEX (w/w).
bStudy animal body weight ∼5 kg.
cIncision length = ∼5 cm.
dPlacebo = D-PLEX formulation without Doxycycline (excipients only).
Surgical Procedures
Study Design.
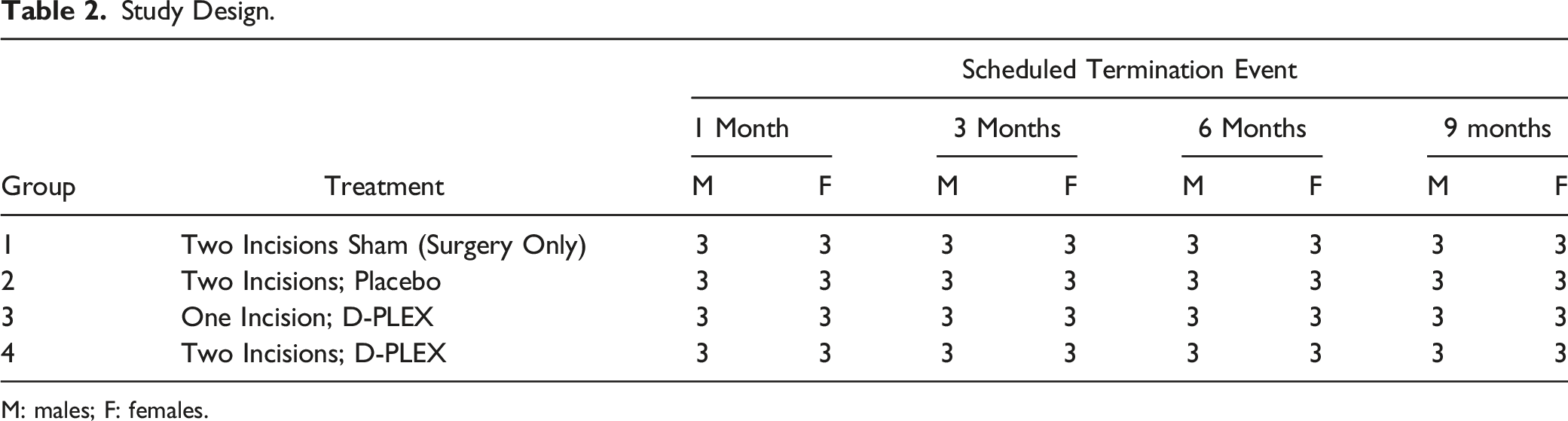
M: males; F: females.
Clinical Observations
In-cage observations were made at least twice daily, and detailed clinical observations were made weekly, including detailed evaluation of the incision site and dental assessment for teeth discoloration. Body weight measurements were performed at least weekly and food consumption was measured daily. Animals were tested for developmental landmarks (neurobehavioral testing) on Days 28 ± 1, 87 ± 3, 175 ± 4, and 265 ± 5 days. Ophthalmic examination was conducted once within four days of each respective scheduled termination.
Clinical Pathology and Toxicokinetics
Blood samples for clinical pathology (hematology, serum chemistry, and coagulation) were collected on Day 1 (prior to dose) then once within four days of each respective scheduled termination. Urine samples were collected once during acclimation then once within four days of each respective scheduled termination. Toxicokinetics samples were collected on Days 1 (Pre-dose, .5, 1, 2, 4, 10, 12, and 16 hours), 2 (24 ± 1 hours), 3 (48 ± 2 hours), 4 (72 ± 3 hours), 5 (96 ± 4 hours), 8, 15, 22, and 29.
Terminal Procedures
A complete gross necropsy was conducted on fasted animals and the following tissues were collected, preserved, and evaluated macroscopically and microscopically: aorta, bone marrow (femur and sternum), bone (femur and sternum), brain, cervix, incision site, epididymis, esophagus, eyes, and gallbladder. GALT (Gut-associated lymphoid tissue), adrenal gland, lacrimal gland, mammary gland, pituitary gland, prostate, mandibular salivary gland, thyroid gland with parathyroid, heart, kidneys, cecum, colon, rectum, larynx, liver, lung/bronchus, mandibular and mesenteric lymph nodes, quadriceps femoris, optic and sciatic nerves, ovaries/oviducts, pancreas, seminal vesicles, abdominal skin, duodenum, ileum, jejunum, cervical, lumbar and thoracic spinal cord, spleen, stomach, testes, thymus, tongue, trachea, urinary bladder, uterus, vagina, and vas deferens.
Sexual Maturity Assessment
The sexual maturity assessment for males and females was based on morphologic evaluation. For males, this evaluation focused on spermatogenesis in the testes along with the morphology of the epididymides and accessory sex glands (seminal vesicles, prostate gland). For females, the assessment focused on the presence/absence of corpora lutea in the ovaries along with any evidence of repeated cycling in reproductive tissues. Animals were assigned to sexual maturity categories of immature, peripubertal, or mature based on the guidance published by Vidal et al. 16
Statistical Analysis
For statistical analysis, Bartlett’s test for homogeneity was performed with a significance level of .05. If the data was homogeneous (Bartlett’s test result
If the data was not homogeneous (Bartlett’s test result
Results
Mortality
There were a total of five unscheduled euthanasia/deaths—two in the sham control group (Group 1), two in the placebo control group (Group 2) and one in the D-PLEX high dose group (Group 4), between Days 5 and 41 following surgery. Mortality events were considered unrelated to the administration of D-PLEX, as four of the five animals were in the control groups, and based on the macroscopic and/or microscopic findings observed in these animals. The one D-PLEX high dose treated animal from Group 4 (284 mg/kg D-PLEX [3.1 mg/kg doxycycline]) was a female swine on Day 4 following surgery. There were no clinical observations noted prior to the animal’s death or macroscopic findings observed and the histopathological evaluation of systemic tissues did not reveal a cause of death for this animal. There was no evidence to suggest a relationship to D-PLEX administration. Microscopic findings of systemic tissues were limited to mild pulmonary hemorrhage and diffuse, mild congestion of the mandibular lymph nodes; these were agonal changes. Morphologic changes at the dosing surgical sites were characterized by neutrophil infiltrates (acute inflammation), diffuse edema, fibrin exudation and hemorrhage with necrotic skeletal muscle in all test groups, as would be expected at this early time point in this incisional surgical model.
Clinical Observations, Clinical Pathology and Body and Organ Weights
There were no D-PLEX placebo-or D-PLEX-related effects on clinical observations, body weight/body weight changes, food consumption, teeth coloration assessment, neurobehavioral and ophthalmic evaluations, hematology, coagulation, serum chemistry, and urinalysis parameters, or organ weights, throughout the time course of the study.
Toxicokinetics
Mean (SD) Doxycycline TK Parameters Following Surgical Incision Administration of D-PLEX in Combined Sex Animals.

Macroscopic Observations
There were no macroscopic observations related to the placebo control or test article in any of the tissues evaluated at the 1-, 3-, 6-, or 9-month time points.
Microscopic Observations
1-Month Time Point
The morphologic appearances of the dosing sites in the Sham Control (Group 1), Placebo Control group (Group 2), and D-PLEX groups (Groups 3 and 4) were comparable. They were characterized by the presence of fibrosis and neovascularization (granulation tissue) of similar intensity (usually mild-to-moderate) in the region of the surgically created incision (Figure 1(A)-(D)). Similarly, the degree and incidence of inflammation was comparable across all groups. Specifically, in the Placebo control group (Group 2) and both D-PLEX groups (Groups 3 and 4), there were infiltrations of minimal-to-mild numbers of lymphocytes, macrophages, and multinucleated giant cells within the surgical tissue planes. This foreign body-type response was composed of phagocytic macrophages and giant cells in association with minimal-to-mild (Grade 1 to Grade 2) amounts of material (sometimes calcified), consistent with residual placebo (PLEX excipients as a biodegradable drug delivery platform) or D-PLEX (PLEX excipients plus doxycycline). For the D-PLEX groups (Groups 3 and 4), there were no apparent dose-related differences in the intensity of the inflammatory response. Other findings noted across groups included the presence of microgranulomas associated with surgical foreign material (gauze/cotton and/or hair-shaft fragments) or suture material-related inflammation. (A) Low magnification image of the left dosing site from a female swine treated with D-PLEX placebo (Group 2) at 1 month, demonstrating a grade 2 fibrosis and grade 3 neovascularization at the incision site (circles). Arrows indicate grade 2 PLEX residual material. High magnification image of the boxed area is provided in Figure 1(B). (B) High magnification image of the boxed area of Figure 1(A), demonstrating the residual PLEX material, some of it appears as calcified material (arrows), associated with macrophages and multinucleated giant cells (arrowheads). (C) Low magnification image of the right dosing site from a female swine treated with D-PLEX 3.1 mg/kg (Group 4) at 1 month, demonstrating a grade 2 fibrosis and grade 3 neovascularization (circles) at incision site. Asterisks indicate suture material. The boxed area and arrow indicate grade 1 residual D-PLEX material. High magnification image of the boxed area is provided in Figure 1(D). (D) High magnification image of the boxed area of Figure 1(C), demonstrating the residual D-PLEX material, some of it appears as calcified material (arrows), associated with macrophages and multinucleated giant cells (arrowheads). (E) Low magnification image of the left dosing site from a male swine treated with D-PLEX placebo (Group 2) at 9 months, demonstrating the healed incision site with well-organized fibrosis (circles). High magnification image of the boxed area is provided in Figure 1(F). (F) High magnification image of the boxed area of Figure 1(E), demonstrating birefringent microparticulate consistent with minute amount of inert residual PLEX with associated macrophages (arrow). Arrowheads indicate minimal focus of adjacent infiltrating lymphocytes. (G) Low magnification image of the right dosing site from a male swine treated with D-PLEX 3.1 mg/kg (Group 4) at 9 months, demonstrating the healed incision site with well-organized fibrosis (between thick lines). High magnification image of the boxed area is provided in Figure 1(H). (H) High magnification image of the boxed area of Figure 1(G), demonstrating residual suture material (asterisk) with associated inflammatory cell infiltrates surrounded by fibrosis.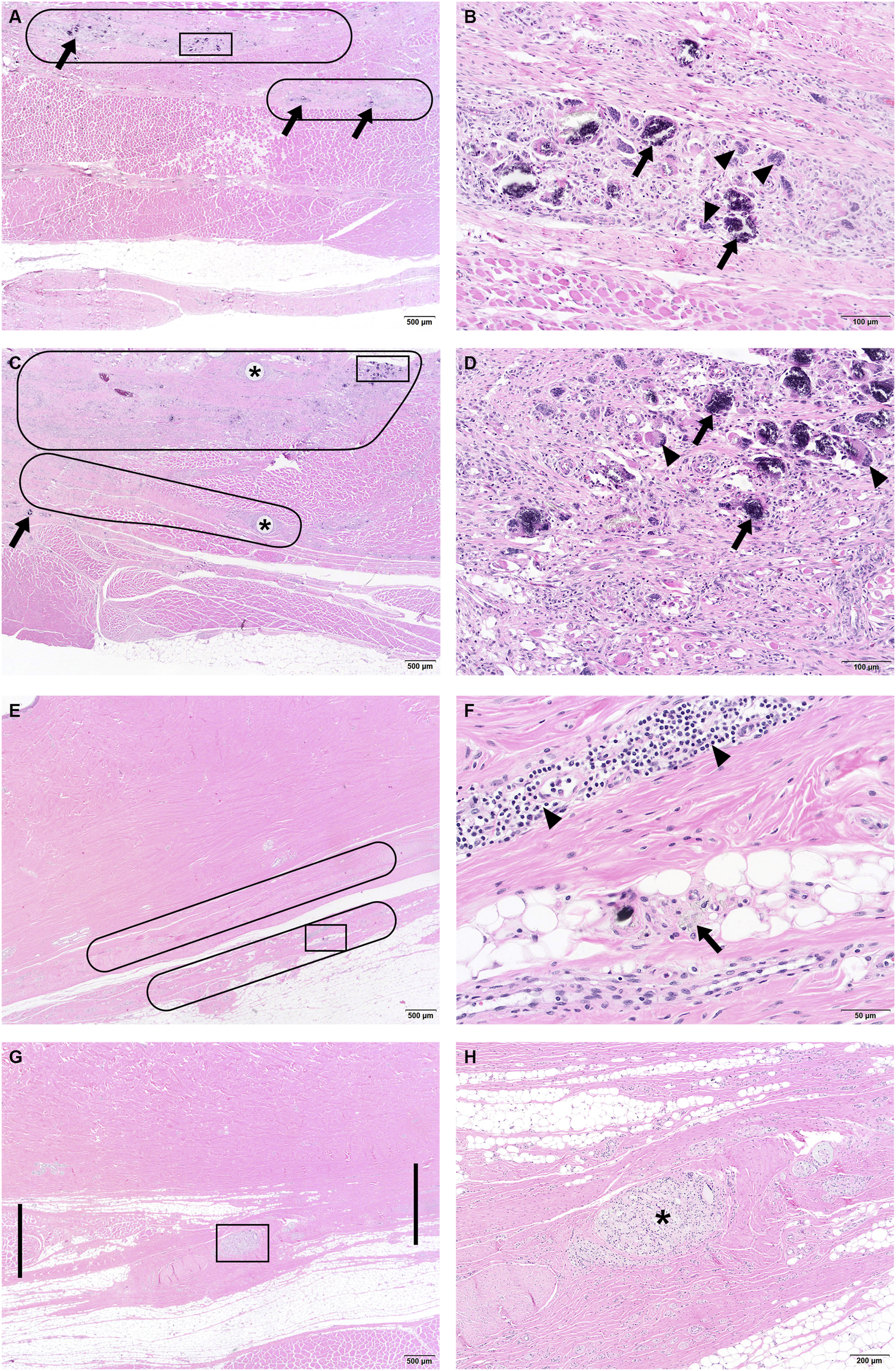
3-Month Time Point
The dosing sites across all groups continued to have a comparable microscopic appearance with the presence of more organized, healing collagen matrix (fibrosis) with slightly reduced neovascularization as the wound sites continued to mature. Inflammatory cell infiltration (lymphocytes, macrophages, and/or giant cells) decreased in intensity in all groups when compared to the 1-month time point (minimal severity degree). In the D-PLEX placebo and D-PLEX groups (Group 2 and Groups 3-4, respectively), no residual excipient (PLEX) was present, consistent with complete biodegradation of the biomaterial. Only trivial amounts (sometimes calcified) were present as trace quantities in association with infrequent phagocytic macrophages and/or giant cells. The residual amounts of these microparticulates were observed in quantities comparable to surgical foreign microparticulates or other poorly characterized iatrogenic microparticulates (i.e., gauze/cotton fragments or suture fragments) typically associated within surgical sites or wound sites that completely heal in pre-clinical surgical studies in laboratory swine. Additionally, there were no deleterious tissue alterations at the dosing sites in association with the trace microparticulates in all groups.
6-Month and 9-Month Time Points
The dosing/wound sites continued to heal without complication for all treatment groups and were regarded to be completely healed. The degree of extracellular collagen matrix was stable in quantity but increased in quality as collagen was more organized and the connective tissue more clearly bridged the incision line across bundles of skeletal muscle of the abdominal wall. Inflammation was of decreased incidence at the 6-month time point and was essentially negligible across all groups by the 9-month time point. By the 6-month time point only trace microparticulates were observed. By the 9-month time point, evidence of these trace microparticulates was notably reduced in all groups, consistent with continued biodegradation (Figure 1(E)-(H)).
There were no differences in microscopic observations between the placebo- or D-PLEX-treated groups and the sham control group in any of the tested tissues.
Bone Growth and Skeletal Maturation Assessment
Mean Group Femur Length Measurements.
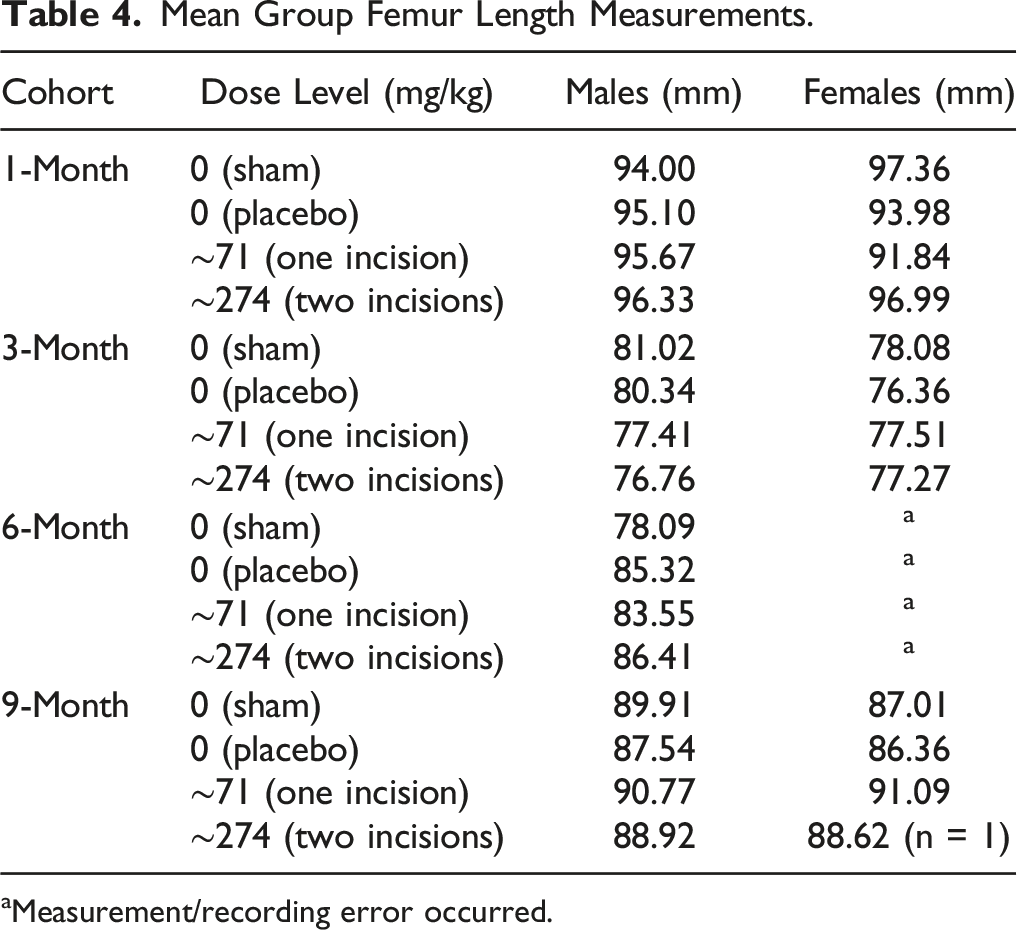
aMeasurement/recording error occurred.
Sexual Maturity Assessment
Sexual Maturity Assessment.

Discussion
Here we present the results of a GLP-compliant 9-months follow-up safety and TK study of D-PLEX in an abdominal surgical incision model in juvenile miniature swine. The animals were exposed to a single application of D-PLEX into abdominal surgical incision sites of juvenile Yucatan miniature swine. There were no test item-related effects in all the parameters evaluated in this study. Administration of D-PLEX placebo (PLEX platform without doxycycline) or D-PLEX in both tested doses was not associated with any local toxicity at the incision sites or with any systemic effects including sexual or skeletal maturation, suggesting good safety profile for both D-PLEX formulations and for the PLEX platform.
The TK parameters demonstrated dose proportional increases in doxycycline plasma concentrations with increased local administration dose. Mean Cmax values were 112 and 321 ng/mL (in the low and high D-PLEX doses, respectively) while reported systemic administration of doxycycline in swine resulted in a mean Cmax value of ∼1 μg/mL. 17 This further demonstrates the low systemic exposure of doxycycline and the safety of D-PLEX in juvenile animals.
We have also evaluated the safety and the incision sites healing in two previous preclinical studies. In one study, D-PLEX was evaluated in adult New Zealand White rabbits after its administration into a sternal surgical defect and on the ventral side of the sternum. 7 The rabbits were followed for 15 months, and no safety issues were found with the tested material. As expected, D-PLEX gradually degraded until it was fully resorbed by 9 months. Sternal bone strength was not affected by D-PLEX.
In the second study, the safety of D-PLEX in adult Yucatan miniature swine was tested using the same type of incision as in the present study. 8 Therefore, the current study allows us to compare the wound healing process in between adults and juvenile swine of the same strain as the previous study. In the previous abdominal model study, the swine were 4- to 5-month-old at study initiation (compared to 1-month-old at the current study). As in the current study, there was no evidence for systemic toxicity or other safety concerns in adult swine, and all D-PLEX material was fully absorbed by 6 months whereas most of the material was absorbed during the first 3 months. 8 In the current study, peritoneal surfaces in all groups were fully healed and there were no notable increases in abdominal adhesions to the incision site for D-PLEX-treated sites compared to the sham control sites. However, in juvenile animals, D-PLEX was fully resolved at the 3-month time point, which is quicker than the 6-month absorption rate observed in the older swine. 8 The difference in the absorption rate can be explained by an age-related delay in wound healing and less robust constructive remodeling in adult animals.18,19 Another possible explanation can be related to the notable faster drug release in juvenile vs adult animals as reflected in our studies by decreased Tmax and t1/2 in both tested doses (in mg/kg) supporting the accelerated metabolism in juveniles. 8 However, mean Cmax and AUC values were dose related (in mg/kg) and similar between juvenile and adult swine for both tested doses suggesting similar overall exposure but faster kinetics in the juvenile swine compared with the adult swine.
The similar absorption rate in the placebo group can further support the safety of the formulation, as well as the prolonged exposure to doxycycline also in juvenile swine. This information is encouraging for the future development of additional PLEX-based products to the pediatric population.
During the histopathological evaluation, we have noticed the presence of microgranulomas associated with surgical foreign material (gauze/cotton and/or hair-shaft fragments) or suture material-related inflammation (see, for example, Figure 1(C)). Such observations are normal findings and are a result of the surgical procedure or the routine wound care.7,20,21 It is however important to correctly interpret these findings, since biomaterials such as the ones used in the current study can also elicit a foreign-body reaction.
In summary, this study, along with the previous in vivo preclinical studies in adult rabbits and adult swine, demonstrates the safety of D-PLEX in abdominal and sternal surgical site models and provides evidence to support further testing of D-PLEX in the pediatric population. In addition, since it provides excellent documentation of the healing process of abdominal incision sites in juvenile minipigs, it can be used as a reference for future preclinical studies aiming to evaluate the safety of different PLEX-based treatments for the pediatric population.
Footnotes
Acknowledgments
The authors thank Tzvia Selzer from
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.D., S.N., S.R., and N.E. are all current or former employees of PolyPid, Ltd Or PolyPid, Inc. and own stock/stock options. A.N. is a consultant for PolyPid.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by PolyPid Ltd, Petah Tikva, Israel.
