Abstract
The use of miniature swine as a nonrodent species in safety assessment has continued to expand for over a decade, and they are becoming routinely used in toxicology and in pharmacology as well as a model for human diseases. Miniature swine models are regularly used for regulatory toxicity studies designed to assess safety of new therapeutic compounds given through different routes of exposure and are used as an alternative model to the canine or the nonhuman primate. Translational preclinical swine study data presented support the current finding that miniature swine are the animal model of choice for assessment of drug absorption, tolerance, and systemic toxicity following systemic exposures. Because research investigators need to be familiar with important anatomic and histopathologic features of the miniature swine in order to place toxicopathologic findings in their proper perspective, clinical and anatomic pathology data from a large number of Sinclair, Hanford, Yucatan, and Göttingen breeds from control groups from a wide variety of studies performed between 2004 and 2014 will be presented, compared, and partially illustrated.
Introduction
Miniature swine, also known as miniswine or minipigs, are becoming an established fixture in regulatory toxicity studies, providing an alternative in toxicology and safety studies to other nonrodent species such as canines and nonhuman primates (Ganderup et al. 2012). They are being used in safety assessments of new therapeutic compounds given through different routes of exposure as well as in models for human diseases such as diabetes, heart disease, and various skin conditions.
As new compounds are assessed in preclinical trials, it is imperative that normal background information be available in order to adequately determine the extent of product effects. Previous publications include studies examining and collecting data on specific features of individual breeds of miniature swine, including nasal passages structure (Kuper et al. 2012), respiratory system mechanics (Truchetti et al. 2014), intraocular features (Shafiee et al. 2008), and determination of female sexual maturity (Tortereau, Howroyd, and Lorentsen 2013). Additionally, there are reviews on the anatomy and physiology of swine as a species compared to humans and of miniature swine in general and their benefits in toxicology studies; these reviews also highlight deficiencies in background pathology or other information currently in the literature (Bode et al. 2010; Swindle et al. 2012). The increasing use of miniature swine in preclinical research draws attention to the need to be aware of differences among the individual miniature swine breeds as well as the need to be able to select the best model to meet specific trial needs. This publication is intended to provide a collection of important clinical, macroscopic, and histopathologic features of Sinclair, Hanford, Yucatan, and Göttingen miniature swine, the most common miniature swine breeds available in the United States (Swindle et al. 2012), to contribute to resources enabling investigators to place toxicopathologic findings of specific breeds of miniature swine in proper perspective. The translational preclinical swine study data presented will also support the growing finding that miniature swine are a reliable animal model of choice, as described by Bode et al. (2010), Helke and Swindle (2013), Ploemen et al. (2014), and Swindle et al. (2012), for assessment of drug absorption, tolerance, and systemic toxicity following systemic exposures.
Materials and Methods
Animals
Information was gathered from Sinclair, Hanford, Yucatan, and Göttingen miniature swine used as controls during toxicity testing or from in-house clinical investigations specifically designated for collecting baseline data. All information, with the exception of Göttingen anatomic pathology data, were collected at Sinclair Research Center (SRC), LLC. Sinclair, Hanford, and Yucatan miniature swine were between 3 and 12 months of age, and Göttingen miniature swine were between 3 and 14 months of age. The Göttingen anatomic pathology information is previously published data (Gad et al. 2015) that has been included for completeness. Sinclair, Hanford, and Yucatan miniature swine came from Sinclair BioResources, LLC (Figure 1), and the Göttingen miniature swine came from Marshall BioResources. All animal care and procedures performed at SRC were approved by the SRC Animal Care and Use Committee and conformed to the Guide for the Care and Use of Laboratory Animals published by the U.S. National Institutes of Health. Any animals that were euthanized were done so in accordance with the American Veterinary Medical Association (AVMA)’s published guidelines (AVMA 2013).
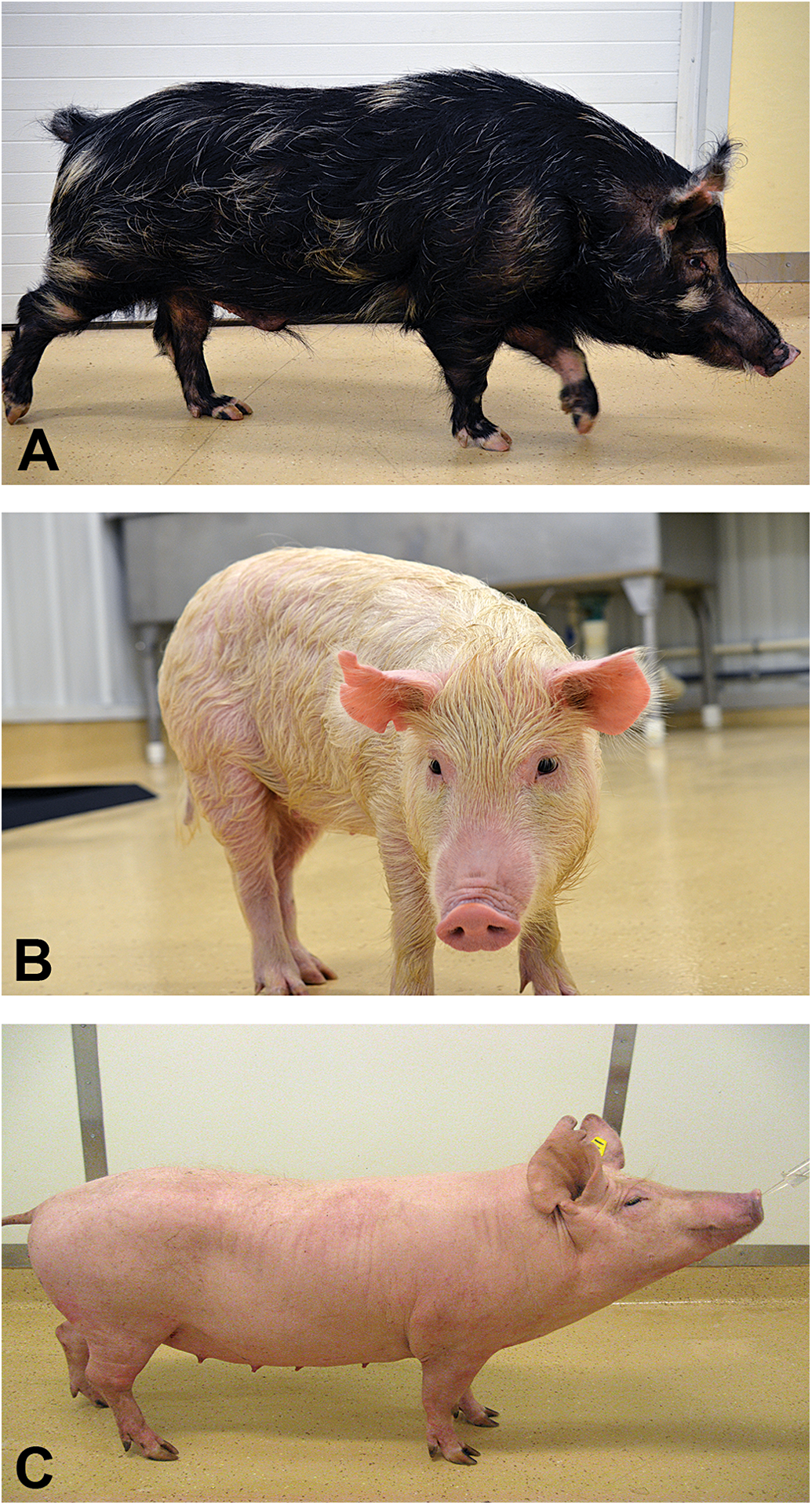
Photographs of three of the miniature swine breeds most commonly used in safety pharmacology and toxicology studies. A: Sinclair, B: Hanford, and C: Yucatan.
Housing
Environmental conditions included a 12-hr light/dark cycle with lights off at 6:00 p.m., an ambient temperature maintained at 15°C to 30°C (59°F–86°F), and humidity ranging from 12.8% to 100%. All miniature swine were individually housed except for four females who were housed in pairs. All pens had a minimum of 16.5 sq ft (3ft × 5.5ft) flooring made of raised polyvinyl chloride–coated self-spanned metal. The pens were made of stainless steel with either solid walls or solid lower walls with upper vertical bars, and all front gates were made of vertical bars. Environmental enrichment was provided via a metal chain suspended in each pen. A certified miniswine diet such as Harlan® Teklad #7037C or Purina® Sinclair S9 was fed at a maintenance amount once a day, and free access to deep well water was provided.
Complete Blood Count (CBC) and Serum Chemistry
CBC and chemistry testing were performed at Antech Diagnostic Laboratories (Antech) or in SRC’s quality-controlled laboratory; SRC uses Siemens Advia 120 for CBC analysis and an AU480 Chemistry Analyzer by Beckman Coulter. Hanford and Sinclair testing was performed at Antech, Yucatan testing was performed at either Antech or SRC, and Göttingen testing was performed at SRC.
Urinalysis
Hanford and Yucatan urinalysis samples were obtained via cystocentesis, and testing was performed at Antech. Sinclair urine samples were collected via metabolic cages and analyzed at either Antech or SRC. SRC urinalysis testing is comprised of Siemens Healthcare Diagnostic Urinalysis Strips read by a Clinitek Status Analyzer and microscopic sediment exams.
Anatomic Pathology
Organ weights were recorded at SRC at the time of necropsy. Gross and microscopic pathology were performed by board-certified anatomic pathologists. Göttingen histopathology was obtained from Gad et al. (2015) and included for comparison purposes. Histological findings were graded on a 4-grade system: minimal, slight, moderate, and marked.
Results
Basic Physiology and Comparative Anatomy
Organ weights
Absolute organ weights (Table S1), as would be expected, were smaller in younger populations. The results of the Hanford and Yucatan miniature swine were from animals more similar in age, and the absolute and relative organ weights of the two breeds were similar as well. The absolute organ weights of the Sinclair miniature swine were slightly smaller than the other two breeds, but the animals were 1 to 6 months younger than those of the other breeds at the time of data collection. The variation in relative organ weight, expressed as percentage of body weight, differed more widely across age and species than absolute weight. For example, the lungs with main stem bronchi, the pituitary gland, and the brain in the younger Sinclair population each comprised a notably higher percentage of body weight as compared to the other two breeds. Alternatively, the relative liver weight was a smaller percentage of body weight in the Hanford breed than the others, while the relative thyroid weight was a higher percentage of body weight in Yucatan than the other breeds. Conversely, the relative weights of the kidney, heart, and adrenal glands did not vary widely across the breeds.
Clinical pathology
CBC findings are presented in Table S2. While all four breeds had a higher neutrophil count than any other white cell type, there was notable variation in distribution of the different types of white blood cells across species. Sinclair miniature swine had the highest mean lymphocyte count (11.62 ± 3.83 × 103/mm3), Yucatan miniature swine had the highest neutrophil count (4.34 ± 2.73 × 103/mm3), and Göttingen miniature swine had a markedly higher eosinophil count than the others (1,961 ± 1,222/mm3 vs. a range of 180–420/mm3 in the other breeds). The Göttingen breed had subjectively lower hematocrit than the other breeds (mean of 28.2% vs. 39.5–47.4% in the other breeds). Chemistry results, as shown in Table S3, also exhibited mild variation across breeds. The average glucose level of the Hanford breed was at least 0.5 mmol/L higher than the other breeds. Sinclair miniature swine had the lowest urea concentration of the four breeds, but they shared the highest creatinine content with the Hanford miniature swine. Total protein and albumin levels were quite consistent across the breeds. Sinclair and Göttingen had the highest triglyceride values of the four breeds, but only Göttingen miniature swine had higher total cholesterol concentrations (2.33 ± 0.57 mmol/L), while the other three breeds had average total cholesterol concentrations that ranged from 1.99 to 2.05 mmol/L. Göttingen results were among the lower values of bilirubin, alanine aminotransferase, and aspartate aminotransferase but were second only to Hanford in having the highest alkaline phosphatase concentration. Overall, electrolyte levels (Na+, K+, Cl−, Ca2+, and PO4 2−) were comparable across the breeds, with the exception that the Göttingen had a lower Na+, K+, and Cl− than the remaining breeds.
Table S4 shows some basic urinalysis results to take into consideration for comparison with toxicology and drug safety studies. Sinclair miniature swine had a lower average urine pH of 7.1 to 7.3 than did Hanford or Yucatan who had pH levels around 8.0. When male and female data are considered together, though, urine specific gravity was fairly consistent across breeds. The Hanford and Yucatan urine samples were collected by cystocentesis; both breeds had higher proteinuria and a glucosuria that was not present in the Sinclair breed, which had urine collected via metabolic cages.
Anatomic Pathology
Macroscopic findings
The macroscopic findings are displayed in Table S5. No gross pathologic findings were observed in the population of 39 Yucatan miniature swine. Among the remaining breeds, lung lesions of red foci, solidifications, and adhesions were the most common finding; this is largely attributed to pigs’ general tendency to inhale food particles while they eat. There was a larger prevalence of reddened thymus and thyroid in the Göttingen breed than the others; reddened thyroid glands can often be a result of venipuncture. The Göttingen miniature swine also had occurrences of reddened urinary bladder mucosa; this can be a finding secondary to cystocentesis.
Microscopic findings
There is a wide range of background microscopic findings possible in miniature swine, as seen in Table S6. The tissues with the most observed microscopic changes were lungs, kidneys, stomach, and liver; and the most commonly observed microscopic findings were cell infiltration and inflammation. Mononuclear cell infiltration was characterized by focal to multifocal infiltration of macrophages with fewer lymphocytes and occasional plasma cells, unaccompanied by degenerative or necrotic changes, and was predominantly seen in the renal cortex of kidneys of the three evaluated breeds: Sinclair, Hanford, and Yucatan (Figure 2). Other tissues in which mononuclear cell infiltration was commonly observed were brain, liver, salivary glands (Figure 3), skin, heart, urinary bladder, and lungs. The severity grade of mononuclear cell infiltration was predominantly minimal. Mixed cell infiltration was not uncommonly seen in the lungs, liver, and esophagus of Hanford miniature swine; this finding was not observed in any tissue from other breeds other than the lungs of Yucatan miniature swine. Mixed cell infiltration was characterized by focal or multifocal foci of neutrophils admixed with macrophages and lymphocytes unaccompanied by degenerative or necrotic changes. The severity grade of mixed cell infiltration was predominantly minimal.
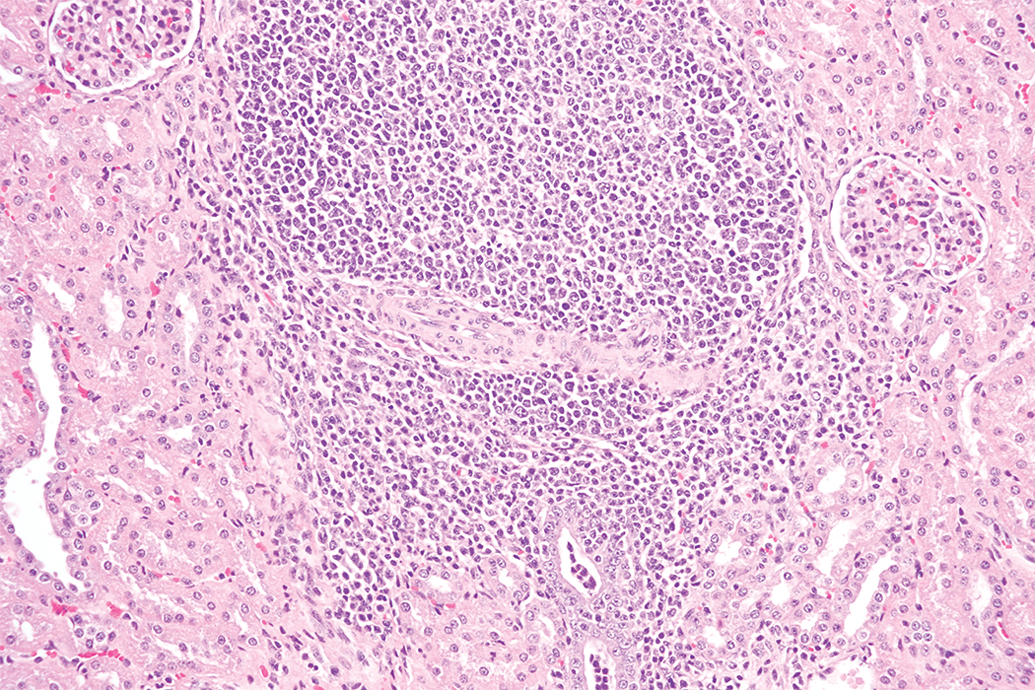
Kidney from a Sinclair miniature swine with mononuclear cell infiltration within the renal cortex. 40× objective; hematoxylin and eosin stain.
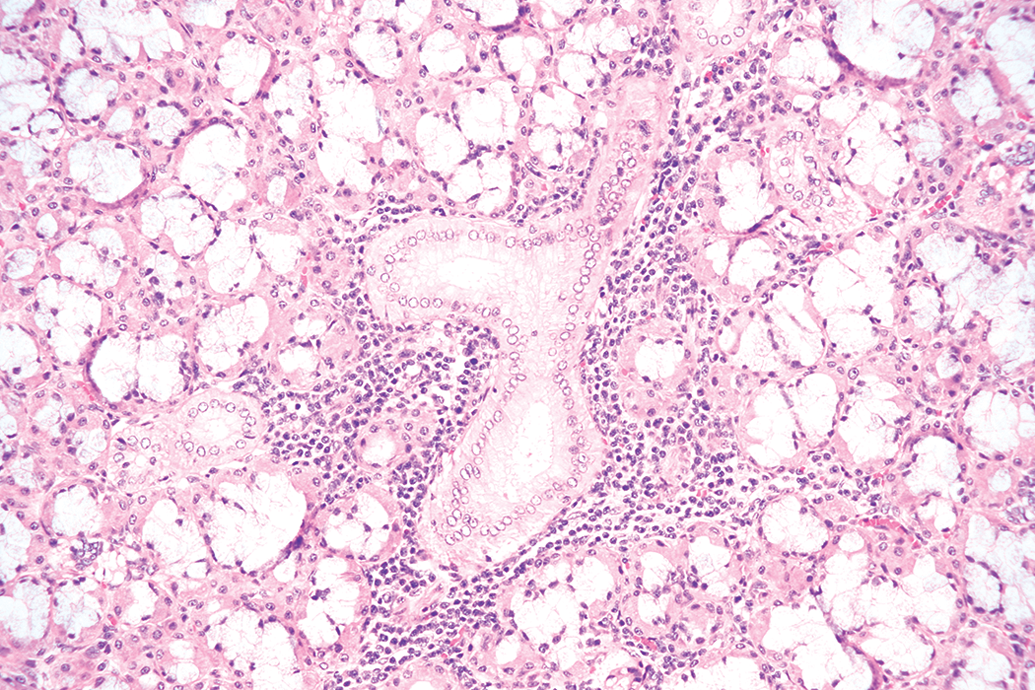
Salivary gland from a Hanford miniature swine with mononuclear cell infiltration within the glandular acini and duct. 20× objective; hematoxylin and eosin stain.
Inflammation was observed in several tissues from the same three breeds. The tissues most significantly affected by inflammation were the lungs where severity grade ranged from minimal to marked (Figures 4 and 5). Granulomatous inflammation (Table S6: granuloma) was observed in the lungs of the Hanford breed, and bronchopneumonia and pneumonia (Table S6: inflammation, lung) in the Yucatan miniature swine. Occasionally, some of the microscopically observed pulmonary inflammation correlated with macroscopic observations such as red discoloration. Inflammation, unlike mononuclear or mixed cell infiltration, was characterized by aggregates of inflammatory cells (neutrophils, macrophages, and lymphocytes) accompanied by degenerative or necrotic changes in the affected tissues. In one case, bacteria were present in the lungs of a Yucatan minipig with inflammation within the lungs. Other more commonly seen locations of inflammation were in the eyes (conjunctivitis in Yucatan), kidneys (arteritis/periarteritis and mixed inflammation in Hanford), heart (specifically arteritis/periarteritis in Hanford; epicarditis in Yucatan, Figure 6), trachea (Hanford), skin (perivascular inflammation in Hanford), and occasionally in the esophagus of all three evaluated breeds. Minimally severe inflammation has previously been frequently observed in the Göttingen breed as well, namely arteritis and periarteritis associated with fibrinoid necrosis, most commonly in the epididymides, heart, intestines, kidney, lung, spleen, stomach, and urinary bladder as well as inflammation of the glandular stomach (Gad et al. 2015).
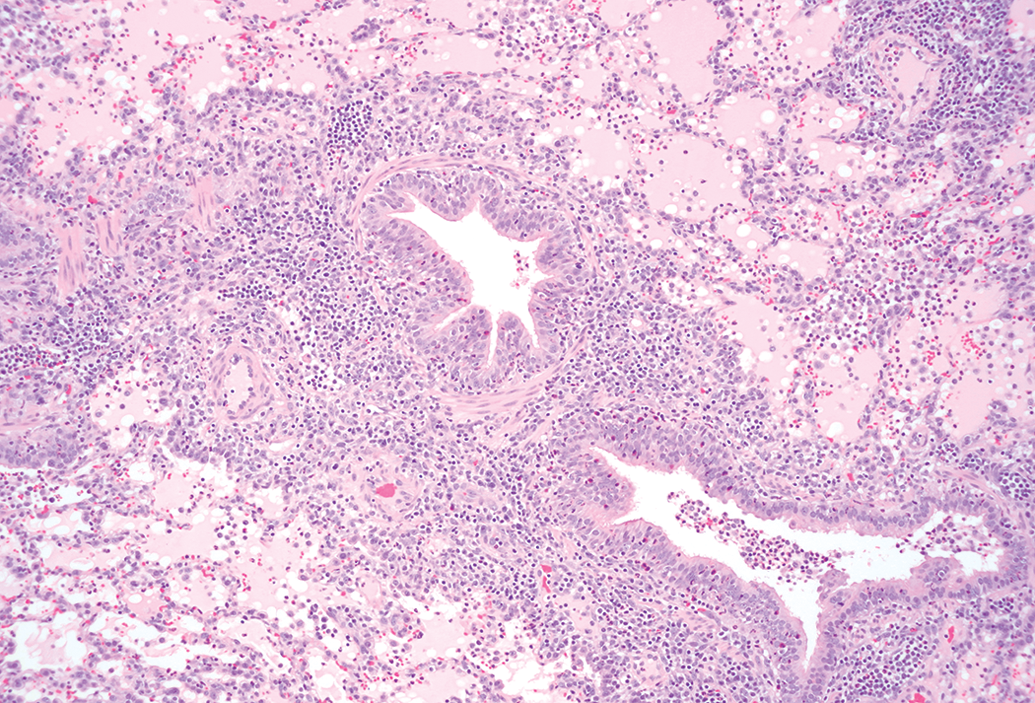
Lungs from a Hanford miniature swine with mixed inflammation of the bronchiolar wall and lumina, alveolar spaces and walls, and edema of the alveolar spaces (pneumonia) 10× objective; hematoxylin and eosin stain.
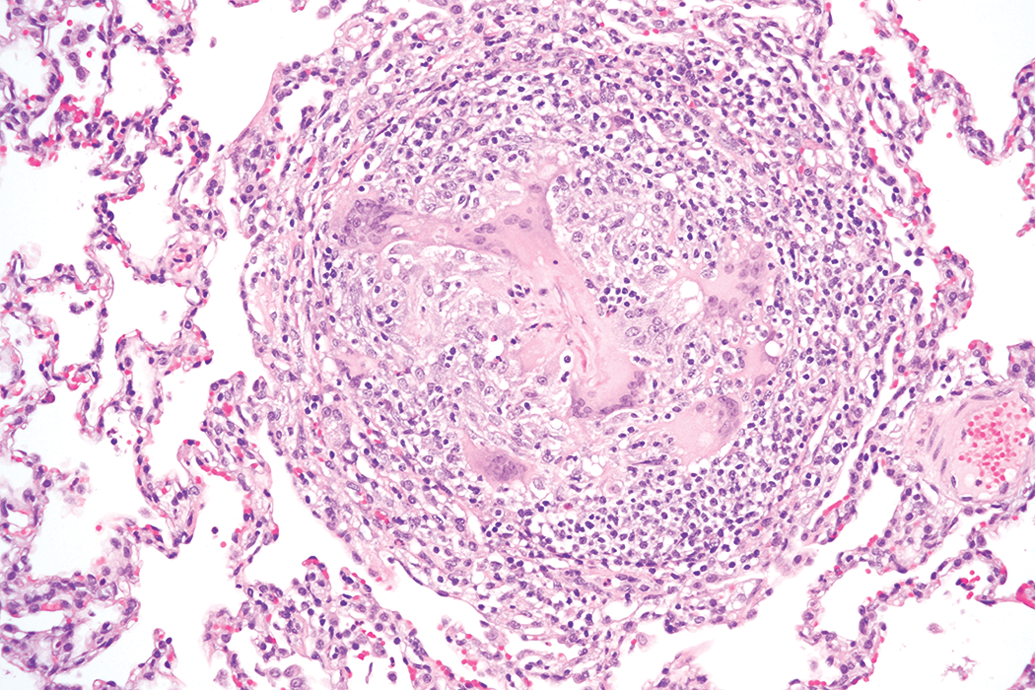
Lungs from a Hanford miniature swine with granulomatous inflammation and granuloma formation. 40× objective; hematoxylin and eosin stain.
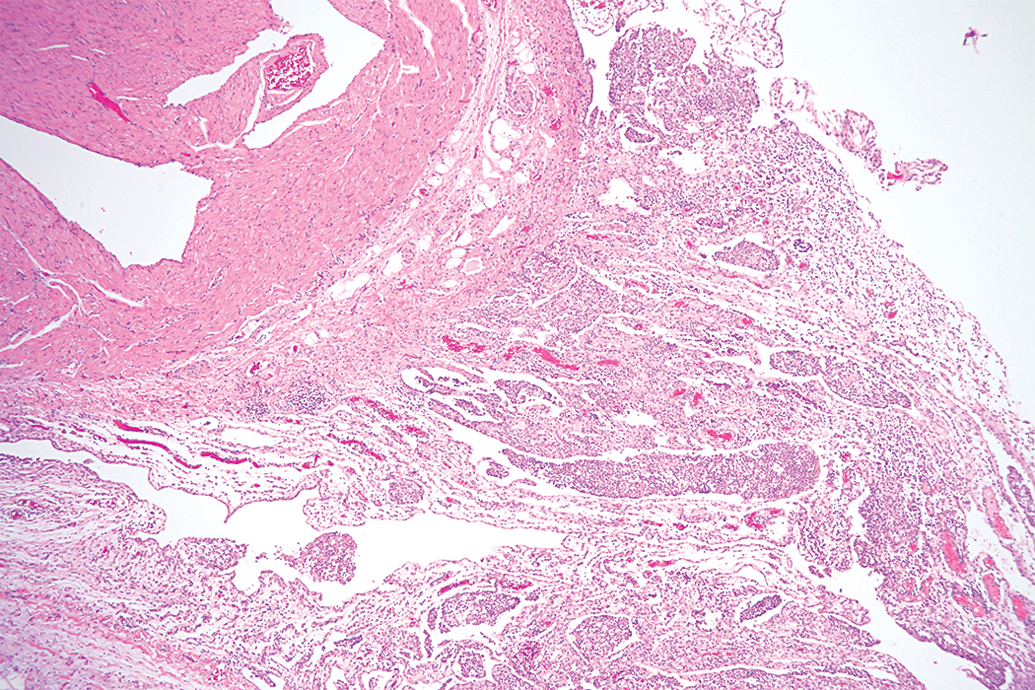
Atrium and epicardium from a Yucutan miniature swine with subacute inflammation infiltration the epicardium. 10× objective; hematoxylin and eosin stain.
Granulomas or microgranulomas were observed in lungs and cervical lymph nodes; the granulomas had higher severity grades than the microgranulomas. Lymphoid hyperplasia (Table S6: increased lymphoid nodules/tissue) was observed in the mandibular and mesenteric lymph nodes as well as the gastric mucosa and submucosa, and esophageal submucosa of Hanford miniature swine. Lymphoid hyperplasia was not observed in Sinclair or Yucatan miniature swine. Fibrosis was more frequently noted in Hanford kidneys and pleura than in any other breed or location (Figure 7). Unilateral or bilateral testicular tubular hypoplasia or atrophy of unknown etiology was a common background finding unique to Göttingen miniature swine. Hemorrhage was more frequently observed in the lungs, cervical, mandibular and mesenteric (Figure 8) lymph nodes, and stomach. Hemorrhage observed in the mandibular lymph nodes, thymus, and thyroid glands were mostly secondary to blood sampling procedures. Congestion was most often observed in the lungs and spleen. There was a higher incidence of gastric findings in Yucatan when compared to Sinclair and Hanford, particularly with increased occurrence of gastric hyperkeratosis in Yucatan miniature swine. Clear cell foci characterized by enlarged hepatocytes with clear cytoplasm in the perinuclear region was also seen very occasionally in the Göttingen breed (Gad et al. 2015).
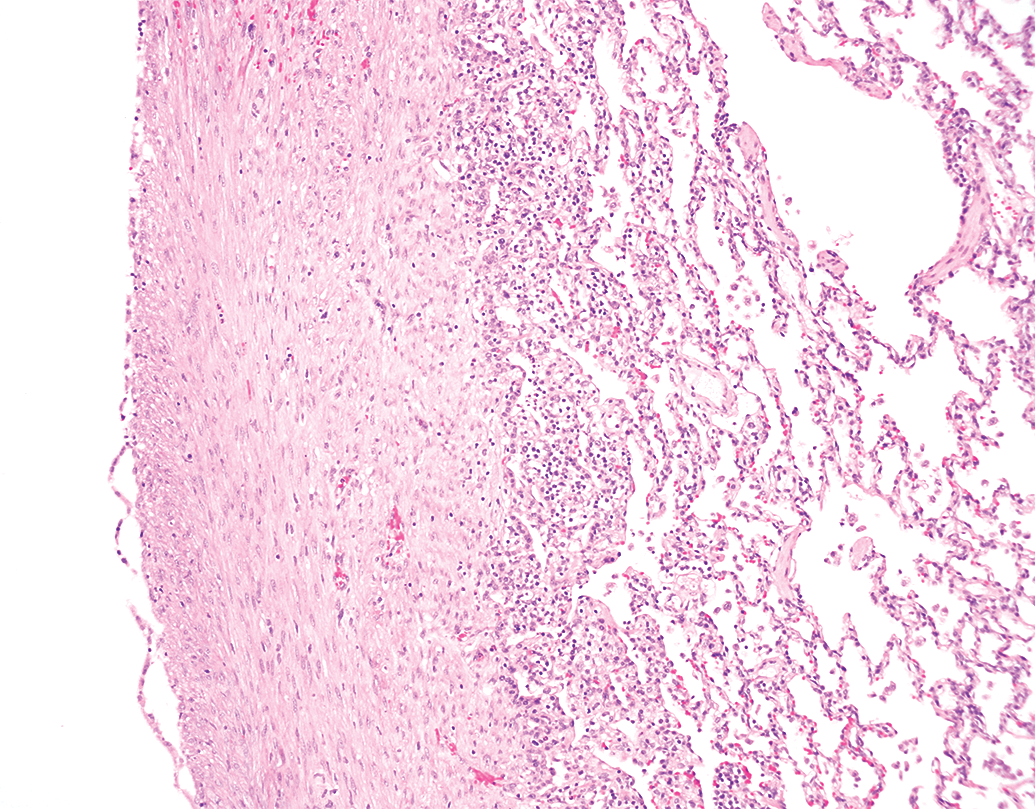
Lungs from a Yucutan miniature swine with pleural fibrosis. 40× objective; hematoxylin and eosin stain.
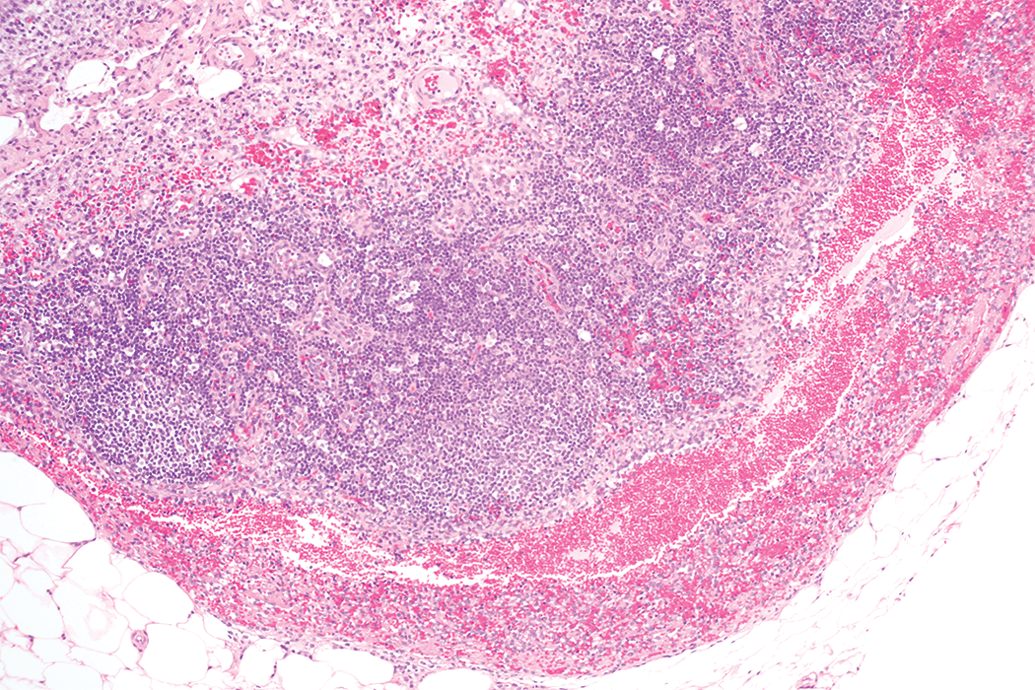
Mesenteric lymph nodes from a Hanford miniature swine with hemorrhage within the sinusoids. 20× objective; hematoxylin and eosin stain.
Discussion
The use of miniature swine (
Our assessment that normal clinical pathology values, as well as common background features and pathology, must be recognizable so as to keep the study findings in perspective has been expressed by others as well (Bode et al. 2010; Swindle et al. 2012; Vezzali et al. 2011). Toxicological findings must be correlated across all available study data for best interpretation of study findings. This outline of baseline and background results of testing common across toxicology studies achieves the objective of providing a resource detailing some variances that occur among these four breeds. Toxicology studies and drug safety assessments in miniature swine are generally performed at 3 to 12 months of age; at this age they are approaching sexual maturity, thus eliminating the difference in response that can be found in younger animals, and they are small and young enough to be financially and physically manageable. As shown in Table 1, miniature swine reach sexual maturity at 4 to 6 months of age, and body weight can vary greatly across the breeds at sexual maturity and beyond. At sexual maturity, the typical minipig weighs 20 to 40 kg as compared to 102 kg for the common pig and 8 to 15 kg for the dog. At this age, they begin to be comparable in size to humans without rapidly becoming too large to manage. Miniature swine are not fully grown until approximately 2 years of age, at which point the Göttingen miniature swine weigh 30 to 40 kg, Sinclairs weigh 55 to 70 kg, Hanfords weigh 80 to 95 kg, and Yucatans weigh 55 to 80 kg (Ganderup et al. 2012) or, in some cases, up to 83 kg (Kim et al. 2015).
Miniature Swine in Toxicity Testing.a

aUses: regulatory toxicity studies, dermal drug development, cardiovascular toxicity, and reproductive toxicology.
In acknowledgment of the probability that some of the information used in this study may be from younger miniature swine that have not completely reached histological sexual maturity, the occasional low incidence of findings of atrophy or underdevelopment of reproductive organs have not been a focus of analysis. One study detailed the need to be aware of the potential for histologic sexual immaturity in toxicology studies; they compared histological evidence of onset of puberty in Göttingen females with the previously mentioned widely accepted data stating that puberty onset of miniature swine is 4 to 6 months of age and found that only 50% of the females studied at approximately 6.5 months of age could be considered sexually mature based on histological findings (Tortereau, Howroyd, and Lorentsen 2013).
There were a limited number of repeated macroscopic findings observed and most were limited to the lungs. These pulmonary findings often correlated with inflammatory processes, hyperplasia of the bronchus-associated lymphoid tissue (BALT) or congestion and/or hemorrhage of the lungs. In spite of the varied incidence and severity grades of the inflammatory changes observed in the lungs, there were no signs of infectious agents. Therefore, most of the changes were considered to most likely be secondary to aspiration of food particles. Similar pulmonary changes have been observed in Göttingen miniature swine (Dincer et al. 2007; Jeppesen and Skydsgaard 2015). Most of the other microscopic findings observed in the tissues from Sinclair, Hanford, and Yucatan miniature swine were morphologically similar and comparable in incidence as described of Göttingen miniature swine (Jeppesen and Skydsgaard 2015), with the exception of hyperkeratosis of the stomach in Yucatan miniature swine. Characteristic spontaneous findings of Göttingen miniature swine such as cholescystitis and hypoplasia/aplasia of the gallbladder, myositis/myofiber necrosis of the skeletal muscle, and serous atrophy of the bone marrow (Gad et al. 2015) were not seen in any of the other three breeds compared here. The most frequent microscopic finding observed in Sinclair, Hanford, and Yucatan miniature swine was mononuclear cell infiltrate in several tissues; the kidneys were the tissue most affected with this change.
This collection of data would be more complete with some improvements, specifically if age of organ weights were more consistent across breeds, and if Göttingen data were available for all values as well. Another inconsistency is the fact that two breeds of miniature swine had urine samples collected via cystocentesis while the third breed was able to forego the stress of the collection procedure and had urine collected via metabolic cages. It is our experience that miniature swine can exhibit stress glycosuria in a manner similar to that of cats, thus the difference in collection methods affected the subsequent urinalysis results. In spite of these minor deficiencies, however, the information that is available thus far will provide many variables to enable guided selection for toxicology studies.
Conclusion
The use of miniature swine in research as a viable alternative to dogs or nonhuman primates has aroused increased interest during the past 10 years. Particular similarities to humans include their cardiovascular anatomy and physiology, integumentary system, gastrointestinal system and digestion, renal system, and immune system. Miniature swine are used in nonclinical pharmacology and standard preclinical studies such as repeat-dose, single-dose, teratology, fertility assessments; and absorption, distribution, metabolism, and excretion studies. They are also amenable to all methods of drug administration, including oral intubation, dietary, inhalation, dermal including administration to experimental wounds, multiple routes of injection, and continuous intravenous infusion.
Pathological evaluation of toxicity studies is concerned not only with the recognition of lesions caused by treatment directly but also with the identification of spontaneous lesions that may have increased in severity and/or frequency in treated animals. It is, therefore, vital to be aware of the background and incidentally occurring lesions as well as normal anatomic characteristics, such as organ weight, that can be observed during macroscopic and microscopic examinations of organs from laboratory animals in routine toxicity studies. In the present report, we focused on the breeds of miniature swine most commonly used in safety pharmacology and toxicology studies: the Sinclair, Hanford, Yucatan, and Göttingen. General characteristics of each breed were discussed as well as clinical pathology background and incidentally occurring lesions that can be observed during macroscopic and microscopic examinations of organs in routine toxicity studies. Additionally, individual variations were noted among breeds in baseline clinical pathology values, variations in organ weights across both age and breeds, and the most common postmortem lesions, such as inflammatory changes in the lungs and mononuclear cell infiltrates in multiple tissues, especially the kidneys. These findings also demonstrated the distinct absence of spontaneous findings characteristic to Göttingen miniature swine in the other strains of miniature swine. In conclusion, there is ample evidence supporting the need for background information in miniature swine altogether as well as individual findings specific to each breed. This information has been provided here in order to enable research investigators to place preclinical pharmaceutical and toxicologic study findings in their proper perspective.
Footnotes
Author Contribution
Authors contributed to conception or design (AS, CS); data acquisition, analysis, or interpretation (PM, SG, DS, GB); drafting the manuscript (AS, CS, PM); and critically revising the manuscript (AS, CS, PM, SG, DS, GB). All authors gave the final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
