Abstract
Axonal dystrophy (AD) is a common age-related neurohistological finding in vertebrates that can be congenital or induced by xenobiotics, vitamin E deficiency, or trauma/compression. To understand the incidence and location of AD as a background finding in Beagle dogs used in routine toxicity studies, we examined central nervous system (CNS) and selected peripheral nervous system (PNS) tissues in twenty 18- to 24-month-old and ten 4- to 5-year-old control males and females. Both sexes were equally affected. The cuneate, gracile, and cochlear nuclei and the cerebellar white matter (rostral vermis) were the most common locations for AD. Incidence of AD increased with age in the cuneate nucleus, cerebellar white matter (rostral vermis), trigeminal nuclei/tracts, and lumbar spinal cord. Axonal dystrophy in the CNS was not accompanied by neuronal degeneration/necrosis, nerve fiber degeneration, and/or glial reaction. Axonal dystrophy was not observed in the PNS (sciatic nerve, vagus nerve branches, or gastrointestinal mural autonomic plexuses).
Keywords
Axonal dystrophy (AD) describes an accumulation of cytoskeletal elements and organelles that results in focal expansion of axons. 1- 4 Equivalent terms in the literature include “dystrophy, axonal” (the recommendation stated in the International Harmonisation of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice [InHAND] glossary 1 ), “neuroaxonal dystrophy,” “axonal swelling,” and “spheroid.” In vertebrates, AD can result from spontaneous, age-related changes affecting both sexes, as well as from genetic mutations, metabolic aberrations, or toxicant exposures. 1 -7
The present study evaluated spontaneously occurring AD in the central nervous system (CNS) and peripheral nervous system (PNS) of naïve Beagle dogs at 2 ages commonly encountered during conventional nonclinical toxicity testing. The objective of this work was to establish background incidence data for use in evaluating data sets from Beagle dogs in experimental studies.
The current study evaluated brain, spinal cord, and selected PNS tissues from twenty 18- to 24-month-old and ten 4- to 5-year-old, naïve Beagle dogs of both sexes (Marshall Farms, Marshall BioResources). This study of control animals was a satellite investigation to supplement our understanding of historical background incidence for a study that was conducted to support clinical development. All procedures were performed in accordance with established federal and state regulations and were reviewed and approved in advance by the Pfizer, Inc, Institutional Animal Care and Use Committee.
The dogs were euthanized humanely, the brain was harvested, and the corpus callosum was cut longitudinally at the midline to promote penetration of fixative. Brains were immersed in either a commercial formulation of neutral buffered 10% formalin (NBF, containing methanol as a stabilizing agent), pH 6.7 to 7.2 (Stat Lab Medical Products), or methanol-free 4% formaldehyde (often called “paraformaldehyde” [PFA]), pH 7.4 (Poly Scientific R&D Corp) as shown in Table 1 at a fixative to tissue volume ratio of approximately 20 to 1 for 3 (NBF) or 5 (PFA) days at room temperature (RT). The brain was trimmed according to Figure 1, after which tissues were retained in fixative for an additional 1 (NBF) or 2 (PFA) days at RT before being processed routinely into paraffin blocks. The forebrain and midbrain were evaluated unilaterally, while the hindbrain was evaluated bilaterally, an approach consistent with the recommended practices for brain trimming in nonclinical general toxicity studies. 8 Spinal cord (cervical, thoracic, and lumbar segments) and selected PNS tissues—sciatic nerve, vagus nerve branches (to the mesenteric lymph node and intestinal serosa), and autonomic plexuses in the gastrointestinal tract—also were removed, fixed in NBF, and processed using conventional methods. 8 -10
Demographics for Beagle Dogs.a

Abbreviations: F, female; M, male; NBF, neutral buffered 10% formalin, containing methanol; PFA, 4% paraformaldehyde (= methanol-free 4% formaldehyde).
a Spinal cord, sciatic nerves, branches of the vagus nerve within mesenteric lymph nodes, gastrointestinal tract, and pancreas (attached to duodenum) were fixed in NBF.
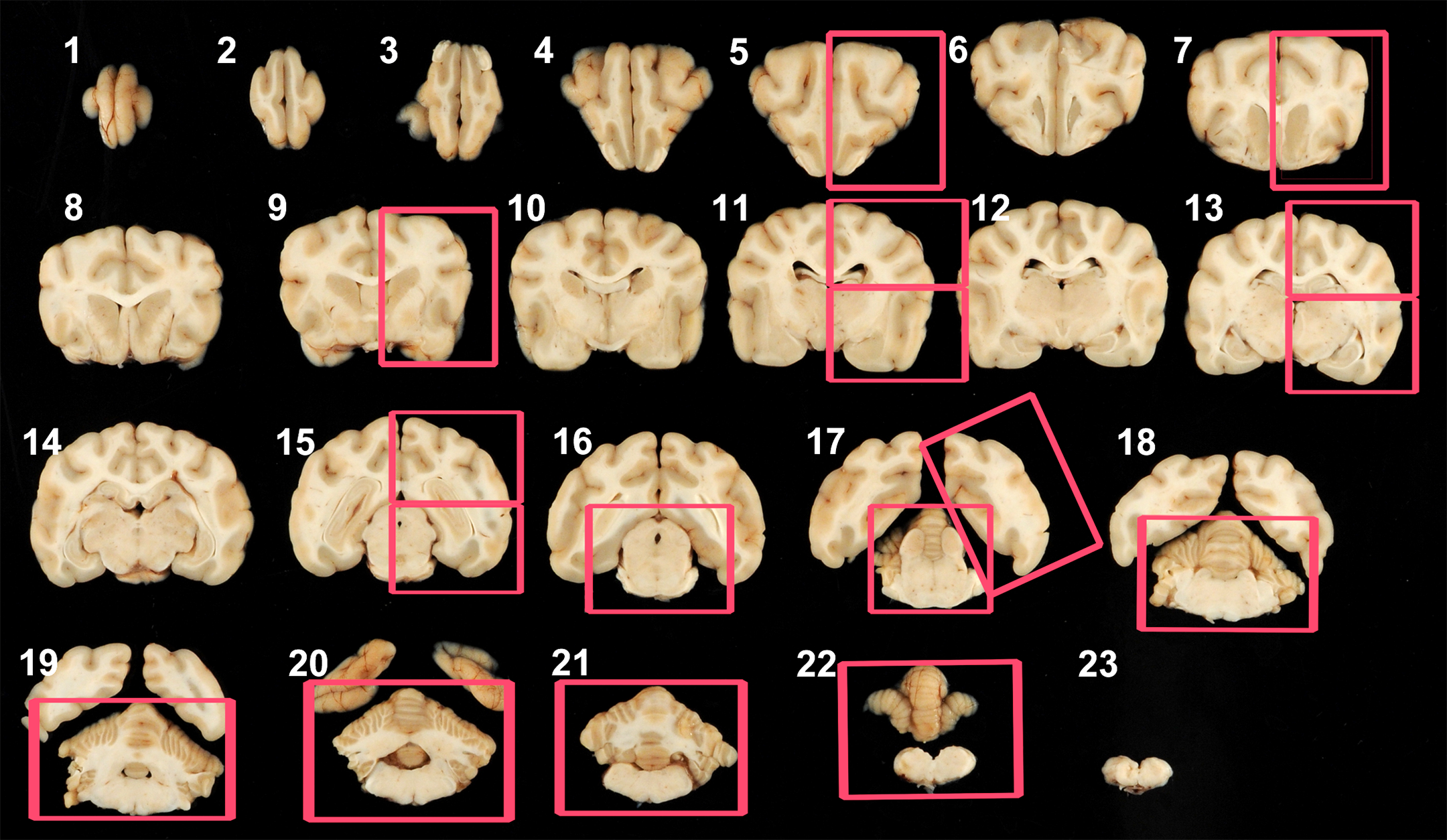
Brain sampling protocol used in this study. The fixed brain was sliced into 23 sections (3.2 mm in thickness). Red boxes for cross-sections (numbered from rostral to caudal) of levels 5 (unilateral section of the frontal pole); 7 (unilateral section containing the frontal cortex, caudate nuclei, and septum); 9 (unilateral section at the level of the rostral [anterior] commissure); 11 (unilateral dorsal and ventral sections at the level of the hypothalamus); 13 (unilateral dorsal and ventral sections at the level of hippocampus, substantia nigra, and thalamus); 15 (unilateral dorsal and ventral sections at the level of the midbrain and caudal hippocampus); 16 (whole section at the level of the pons); 17 (whole sections at the level of the occipital pole, rostral vermis, and rostral medulla oblongata); 18 (whole section of medulla oblongata); 19 (whole section of distal medulla oblongata and deep cerebellar nuclei); 20 (whole section of brainstem and deep cerebellar nuclei); 21 (whole section of mid brainstem); and 22 (whole section of caudal vermis and brainstem) were processed for light microscopic examination. Note that the rostral aspect of each slide was shown in this photo for demonstration purposes. The rostral aspect of selected sections were placed down in the embedding mold.
Five-micrometer-thick sections of all organs were stained with hematoxylin and eosin (H&E). Consistent with current toxicologic neuropathology practice, 1 section per block (ie, 1 H&E-stained section per region per animal) was prepared in this manner. 8 For brain, serial 5-µm-thick sections were cut from selected PFA-fixed brains of some 4- to 5-year-old dogs (animal nos. 21, 24, 25, 26, 28, 29, and 30) as initial evaluation of the related H&E-stained sections demonstrated sufficiently high AD numbers to permit their further characterization using special neurohistological methods. For histochemistry, brain sections containing medulla oblongata (and specifically the cochlear and cuneate nuclei) were processed with Giemsa, periodic acid–Schiff (PAS), Bielschowsky’s silver, Klüver-Barrera (Luxol fast blue [LFB] with cresyl violet [CV]), Lipofuscin, or Masson’s trichrome stains following conventional procedures. The Giemsa and PAS methods were performed on the DAKO Artisan Link Pro autostainer, using DAKO Artisan stain kits (Agilent Technologies, Inc). For immunohistochemistry (IHC), medulla oblongata sections were evaluated for axonal neurofilament content using a mouse monoclonal antineurofilament protein (NFP) 160 kDa immunoglobulin G1 antibody (clone NN18; Sigma-Aldrich) incubated at a dilution of 0.67 µg/mL for 20 minutes at RT on a Leica Bond Rx-automated stainer using the Bond Polymer Refine Detection kit (Leica Biosystems). The site specificity of AD was recorded during light microscopic examination using dog brain atlases by Palazzi 11 and Singer 12 to identify affected locations. The site-specific incidence of AD between younger and older dogs was compared statistically using an analysis of variance model in which the square root of the total AD count was analyzed using age, fixative type, and sex as factors for the statistical model. Calculations were performed by combining sexes in a site-specific manner since preliminary analyses showed no sex-related difference in AD incidence.
Axonal dystrophy was identified in H&E-stained sections of brain and spinal cord but not in sciatic nerve, vagus nerve branches (mesenteric lymph node and serosa of the gastrointestinal tract), or autonomic plexuses in the walls of the gastrointestinal tract and within the pancreas (attached to the duodenum). Comparison of younger to older dogs indicated an age-related increase in the incidence of AD (based on the visible number of AD profiles in a given section) in some brain regions: cuneate nucleus; cerebellar white matter (rostral vermis); and trigeminal nuclei and tracts. Isolated (single) AD fibers were seen in numerous brain regions, especially in older animals, whereas in the most affected brain sites multiple axons exhibited AD. In the spinal cord, AD occurred in the gray matter and white matter of the cervical, thoracic, and lumbar divisions (Figure 2E and F), with a slightly increased incidence of AD in the lumbar segment in older compared to young dogs. For both ages and sexes, the formation of AD appeared to be independent in different brain domains as high numbers of AD fibers in 1 region did not suggest a heightened predisposition to develop AD in other regions of the same animal.
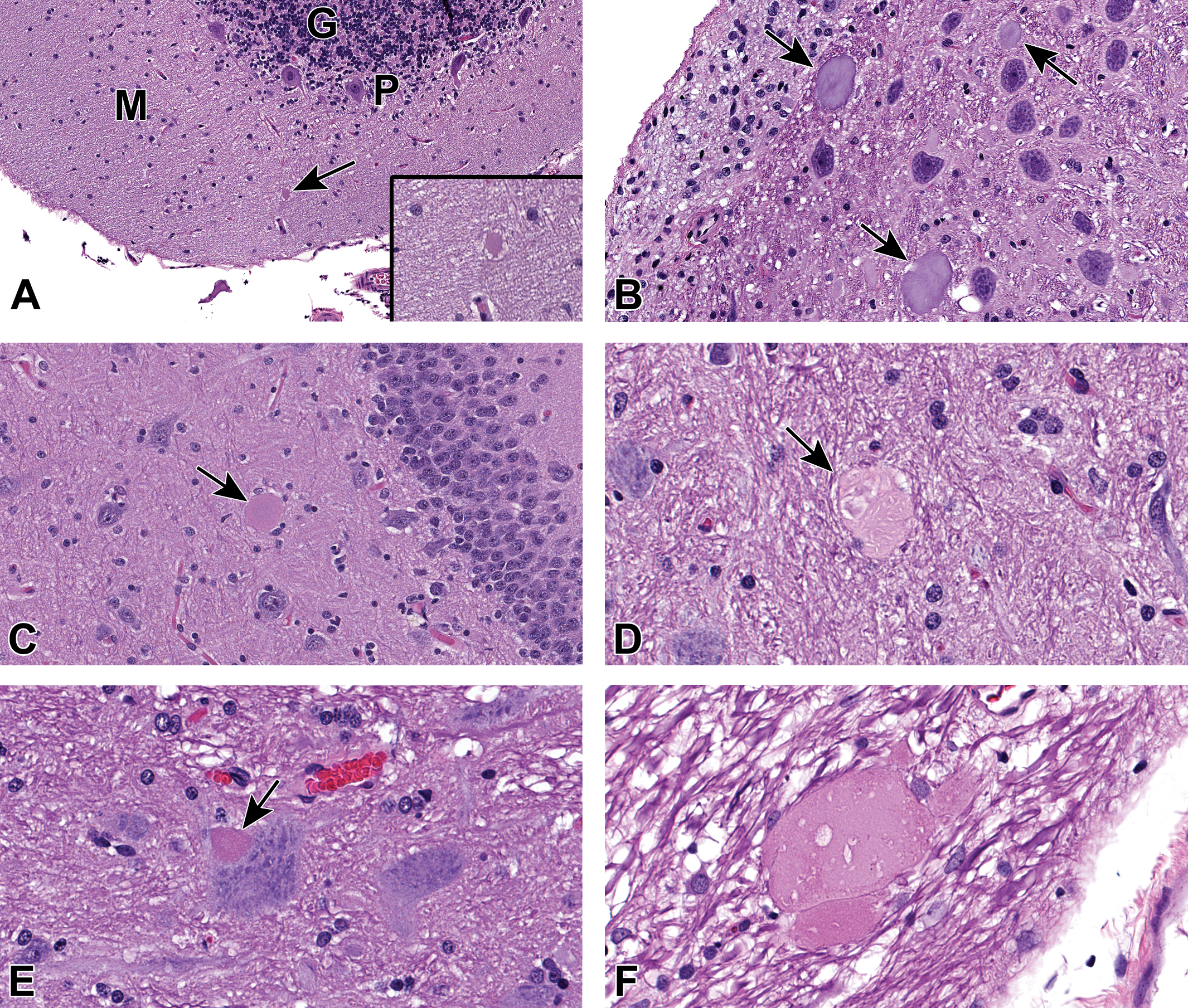
Variations in size, shape, and staining of nerve fibers with axonal dystrophy (AD) in different areas of the brain and spinal cord. A, AD in the molecular layer of the cerebellum of female dog no. 24 (arrow). M: molecular layer; P: Purkinje cells; G: granular layer. Large magnification of AD (inset). AD appears as an eosinophilic, rounded fiber. B, AD in the cochlear nucleus of male dog no. 7 (arrows). ADs are light basophilic, oval structures. C, Single AD in the hippocampus of male dog no. 21 (arrow). D, AD in the cuneate nucleus of female dog no. 26. Note the light eosinophilic staining and round- to oval-shaped structure with some vacuolated material inside (arrow). E, AD in the gray matter of the lumbar section of the spinal cord of male dog no. 21. The AD (arrow) is located near a large neuron. F, AD in the white matter of the lumbar section of the spinal cord of female dog no. 30. Note the variable pale to darker eosinophilic staining with some vacuoles.
Axonal dystrophy exhibited a characteristic constellation of features in H&E-stained sections (Figure 2). Their sizes ranged from 10 to 200 µm in diameter. By H&E, AD fibers were pale to brightly eosinophilic or sometimes pale basophilic displaying granular basophilic material or variably sized vacuoles. Axonal dystrophy differed in shape from perfectly round to ovoid to elongate structures. Axonal dystrophy could be differentiated from similarly appearing neurosecretory (or Herring) bodies (NB, Figure 3), which contain antidiuretic and oxytocin hormones, based on their respective locations since AD occurred chiefly in the caudal brain while NB were commonly in the hypothalamus (including some optic nuclei) and median eminence. The distribution of AD in particular brain areas is demonstrated in Table 2. We found that the most common neuroanatomic areas with AD were the cuneate nucleus (Figure 4), gracile nucleus, cochlear nucleus, and cerebellar white matter (rostral vermis). Axonal dystrophy also occurred occasionally in the facial (cranial nerve VII) nuclei/tracts, molecular layer of the cerebellum (caudal vermis, Figure 2A), nuclei of the rostral (superior) and caudal (inferior) colliculi, hippocampus, and cerebral (temporal) cortex, among other sites (Table 2).
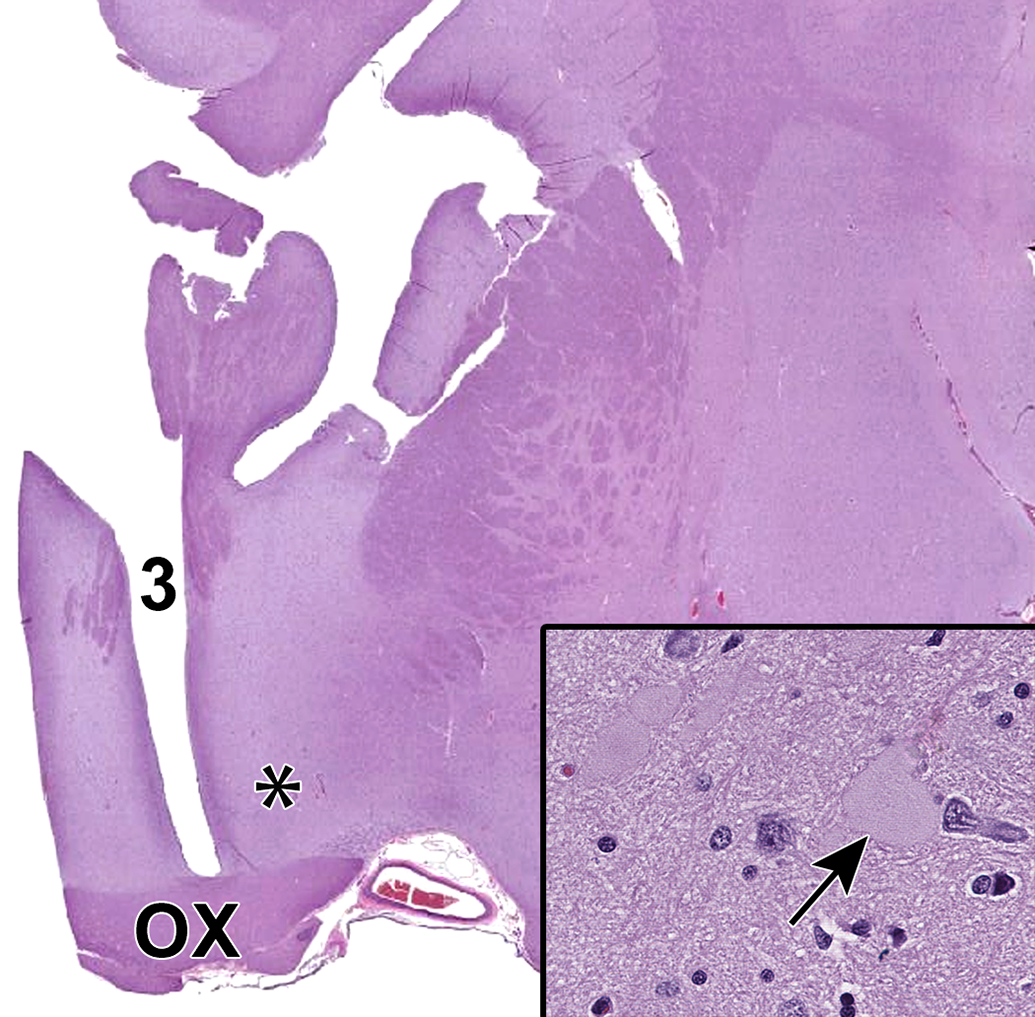
Neurosecretory (Herring) bodies (“physiologic AD”) in the ventral hypothalamus (asterisk) of female dog no. 16. For orientation, OX, optic chiasm; 3, third ventricle. Herring bodies have similar morphology to AD but can be differentiated based on their location in the ventral hypothalamus, usually in the median eminence and some optic nuclei. Inset: Herring body (arrow). AD indicates axonal dystrophy.
Average Incidences of Axonal Dystrophy for Common Brain and Spinal Cord Regions in Beagle Dogs, by Age.
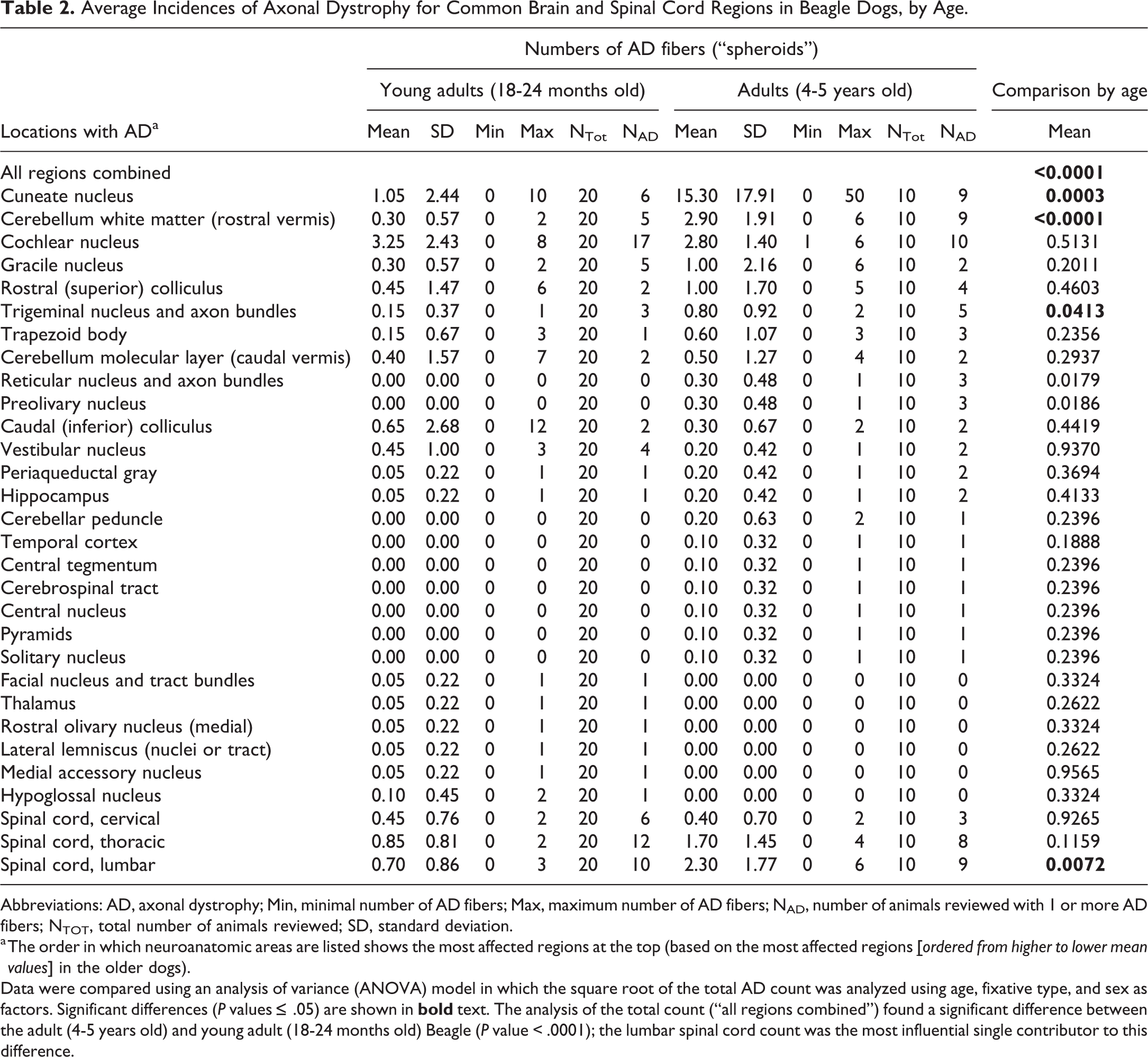
Abbreviations: AD, axonal dystrophy; Min, minimal number of AD fibers; Max, maximum number of AD fibers; NAD, number of animals reviewed with 1 or more AD fibers; NTOT, total number of animals reviewed; SD, standard deviation.
a The order in which neuroanatomic areas are listed shows the most affected regions at the top (based on the most affected regions
Data were compared using an analysis of variance (ANOVA) model in which the square root of the total AD count was analyzed using age, fixative type, and sex as factors. Significant differences (P values ≤ .05) are shown in
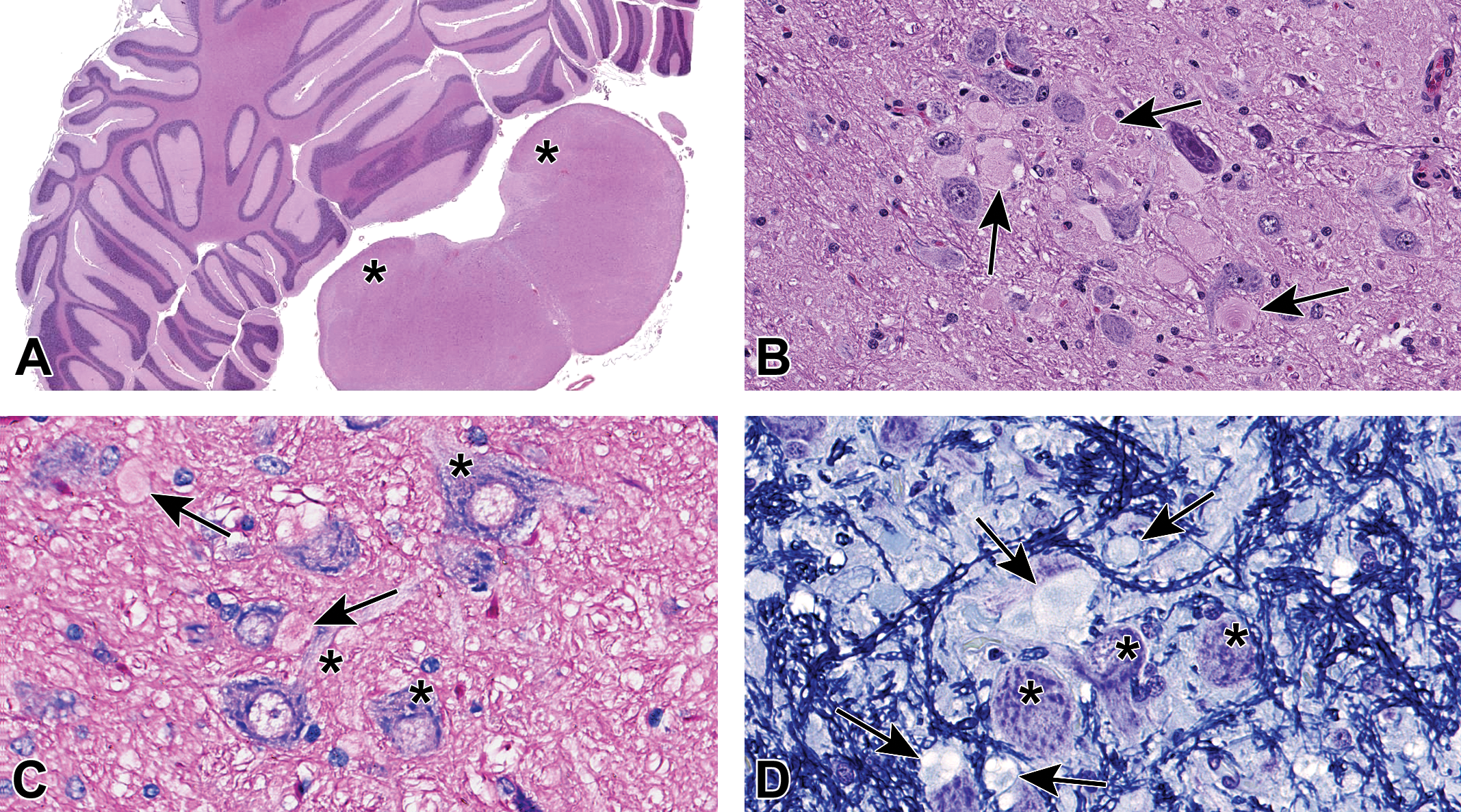
Axonal dystrophy (AD) in the cuneate nucleus of female dog no. 26. A, Lower magnification of brain section 21 containing the cuneate nuclei bilaterally (asterisks). B, Higher magnification of AD in the cuneate nuclei (arrows depict some AD). C, Giemsa stain of the cuneate nuclei. Axonal dystrophy stained pale pink (arrows), whereas neuronal cell bodies and axons exhibited strong basophilic staining (asterisks). D, Klüver-Barrera stain (Luxol fast blue/cresyl violet [LFB/CV]) in the cuneate nucleus. The AD (arrows) had a glassy, vacuolated appearance, and were either surrounded by a single, thin, irregularly disrupted LFB-positive membrane consistent with myelin or lacked a surrounding membrane (consistent with myelin loss [“naked axons”]). The cell bodies of neurons (asterisks) were CV-positive showing a clear demarcation between the cell and nearby affected axons.
Overall, analysis of AD distribution found a significant difference in incidence between younger and older dogs (P value < .0001), but no difference between males and females. The size of AD fibers and their histological appearance were comparable at both ages and in both sexes. No statistically significant difference was detected in the incidence of AD in different neuroanatomic sites of brain preserved with a fixative containing (NBF) or lacking (PFA) methanol, nor did the fixative impact AD appearance (axoplasm color, degree of vacuolation, etc).
Special histochemical and IHC procedures further delineated AD structure. A fine demarcation between pale pink AD and darkly stained neuronal cell bodies was evident with Giemsa (Figure 4C). Axonal dystrophy did not stain with Klüver-Barrera but instead had a glassy homogeneous appearance and were surrounded by a thin to irregular, disrupted, LFB-positive membrane consistent with myelin (Figure 4D). The cell bodies of neurons were CV-positive, while axons and AD were not (Figure 4D). Both Bielschowsky’s silver (Figure 5B) and anti-NFP stained AD (Figure 5D), unaffected axons, and neuronal cell bodies similarly since all these structures are rich in neurofilaments. Axonal dystrophy fibers irregularly were positive for the PAS and Lipofuscin stains but were negative for Masson’s trichrome.
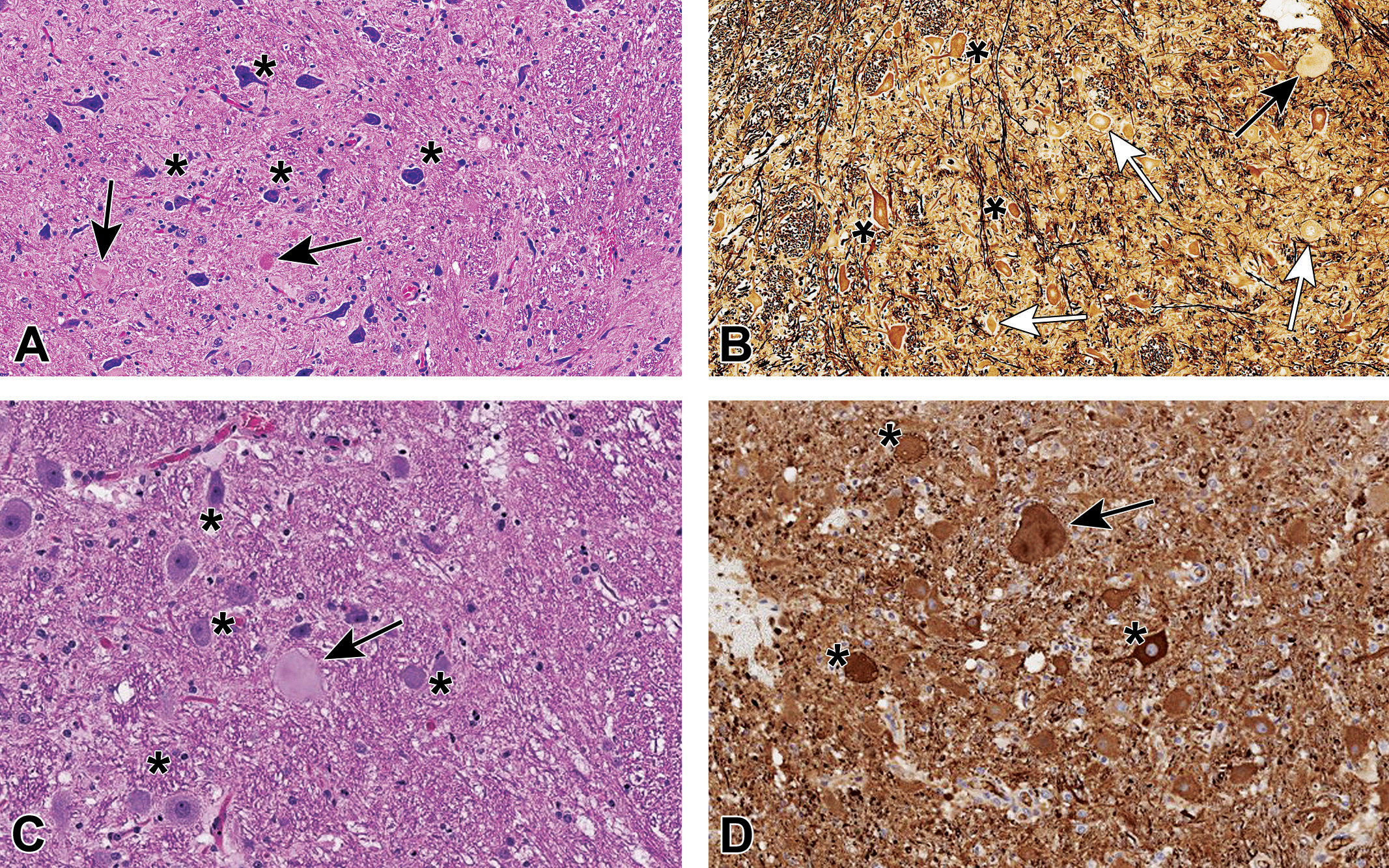
A-D, Axonal dystrophy (AD) in brainstem nuclei of Beagle dogs. A, Cuneate nucleus of female no. 29 showing AD fibers (black arrows) and neuronal soma (asterisks); H&E. B, Cuneate nucleus of female no. 29, stained with Bielschowsky’s silver, showing a large AD fiber (black arrow) and neurons with weak (white arrows) or more intense (asterisks) staining. Note that neuronal cell bodies, normal axons, and AD axons all are positive, making difficult the differentiation among them. C, Cochlear nucleus of an adult control Beagle dog from a different study showing a large AD fiber (black arrow) and multiple neurons (black asterisks); H&E. D, Cochlear nucleus of the same control Beagle dog from (C), labeled with anti-neurofilament protein (NFP), depicting the same AD fiber (black arrow) from (C) and many neurons (asterisks). Note that neuronal cell bodies, normal axons, and AD fibers all are NFP-positive, making difficult the differentiation between AD and normal neuronal components. H&E indicates hematoxylin and eosin.
Axonal dystrophy is a known age-related spontaneous finding in the nervous system, especially brain, and has been reported in many animal species and humans. 1 -4,13 -20 Increased AD numbers have been seen in several breeds of dogs due to autosomal mutations leading to progressive degeneration of central and/or peripheral neurons 21 -37 ; vitamin E deficiency, 4,38,39 presumably leading to increased oxidative stress 39 ; or exposure to toxicants that cause cytoskeletal cross-linking among other mechanisms of toxicity. 40 -49 Axonal dystrophy also has been described in dogs with chronic wasting diseases (eg, filariasis, tumor-related cachexia). 16 The Beagle dog is one of the most common nonrodent species used in nonclinical general toxicity studies, so it is important to understand the anticipated background level of AD in naïve dogs so that any apparent test article-related exacerbation in the incidence and/or severity of AD can be interpreted accurately.
In this study, we evaluated central and selected peripheral nervous tissues in naïve young adult (18-24 months) and adult (4-5 years), male and female Beagle dogs (where dogs used in general toxicity studies typically range from 6 to 24 months). The life span of the research Beagle dog is approximately 15 years with a median survival age of 12.5 years, but aging effects begin appearing in dogs kept in indoor pens as early as 5 years of age. 50 Our sampling strategy evaluated common CNS and PNS tissues included in nonclinical general toxicity studies as well as additional sampling for areas (eg, brainstem relay nuclei) where AD has been reported in the literature and a large autonomic nerve trunk (vagus, cranial nerve X) that carries visceral sensory and visceral motor (parasympathetic) information. Our data concur with prior studies indicating that AD fibers are concentrated in the caudal brain (brainstem relay nuclei and, to a lesser extent, midline white matter of the rostral vermis) and less frequently are scattered in the gray and white matter of the spinal cord; interestingly, we did not observe AD in either spinal nerve (sciatic) or autonomic nerves. Furthermore, the AD fibers in CNS regions were not linked to obvious neuronal lesions (eg, nerve fiber degeneration or neuronal necrosis) or inflammation (microglial or glia reactions) in any part of the CNS, likely because AD fibers do not typically rupture and invoke a tissue response. 1,2 The increased incidence of AD in the molecular layer of the caudal cerebellar vermis, which is located at the atlanto-occipital junction, suggests that head/neck extension leading to brain compression could be related to AD (Figure 2A). Importantly, the occurrence of more severe AD lesions was not associated with functional deficits. For instance, 2 older dogs (male no. 25 and female no. 26) had very prominent AD in the cuneate nuclei without any clinical signs indicative of proprioceptive dysfunction (alterations of positional and movement awareness of various body parts). Taken together, this incidental finding was judged to be nonadverse in these naïve (untreated control) Beagle dogs.
Because AD blended easily with the neuropil (axon-rich parenchyma) in H&E-stained brain and spinal cord sections, we tested various histochemical stain methods to try to facilitate the identification and characterization of AD. The most useful techniques to demonstrate AD in brainstem relay nuclei (ie, the most commonly affected sites in our dogs) were Giemsa and Klüver-Barrera (ie, LFB/CV). Both these techniques highlighted AD fibers in the cuneate nuclei as swollen, pale axons that were easily discriminated from more darkly stained neuronal cell bodies (Giemsa) or the adjacent myelin sheath (Klüver-Barrera). Bielschowsky’s silver and anti-NFP methods were not helpful in this regard since both neuronal cell bodies, normal axons, and AD all were positive and, by the main author’s experience, AD in the cochlear nuclei do not stain for Bielschowsky’s stain (Figure 5A-D). Similarly, PAS and Lipofuscin stains were unhelpful since they were variably negative to weakly (irregularly) positive. Masson’s trichrome was uniformly negative for both AD and surrounding normal tissue, and thus also lacked discriminatory power. A key finding during our study was that AD, which were concentrated in brainstem relay nuclei (Figure 2), can be segregated based on their site specificity and numbers from the somewhat similar-appearing neurosecretory (Herring) bodies that congregate in the ventral diencephalon (Figure 3).
Finally, our data indicated that AD could be seen equally well with methanol-containing (NBF) and methanol-free (PFA) fixatives. This finding is relevant since tissues collected during routine general toxicity studies are most frequently collected into NBF, whereas PFA may only be used for specialized neurotoxicity studies where alcohol-related white matter vacuolation (termed Buscaino bodies, mucocytes, or metachromatic bodies) 51,52 resulting from myelin extraction must be minimized.
In conclusion, spontaneous AD in naïve young adult (18-24 months) and adult (4-5 years) control Beagle dogs of both sexes occurred in the caudal brain and to a lesser extent the spinal cord but not in spinal (sciatic) or autonomic (vagus) nerves. Axonal dystrophy exhibited an age-related increase in the brain (especially cuneate nucleus, cerebellar white matter of the rostral vermis, and trigeminal nuclei and tracts) and spinal cord. Affected fibers are visible by light microscopy in conventionally prepared (NBF-fixed, paraffin-embedded) sections stained with H&E, although staining with Giemsa or Klüver-Barrera (ie, LFB/CV) can further highlight AD sites amid the neuropil. The authors recommend that AD be recorded when observed during animal toxicity studies, with comments regarding their neuroanatomic location, to build a historical control database that is better able to support the interpretation that this occasional finding is incidental.
Footnotes
Acknowledgments
The authors would like to thank Ms Denyse Laforge, Mr Casey Ritenour, Mr Dennis Pelletier, Mr Alan C. Opsahl. and Mr Thomas Kawabe for their support with different technical contributions related to the study and/or manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
