Abstract
Neuropathology of the peripheral nervous system (PNS) is an underappreciated area in toxicologic pathology. Toxicity to nerves and ganglia can result from toxic insults following exposure to environmental, occupational, and industrial chemicals; drugs and biologics; cosmetics and food additives; and even physical agents such as noise. The following introduction provides an overview of this special issue of
The peripheral nervous system (PNS) is the extension of the central nervous system (CNS), enabling the brain and spinal cord to extend their control throughout the body. However, this distinction between the central and the peripheral nervous systems is pedantic, since the integration of the two systems transcends neuroanatomical boundaries. That said, our understanding of neural function is enhanced by conceptually considering the structures and functions of the PNS and CNS separately. The initial step in comprehending PNS toxicologic pathology is to comprehend the primary components of the PNS (
Neuropathology in the PNS generally includes peripheral neuropathies and ganglionopathies. Peripheral neuropathy is the hallmark pathologic condition involving the peripheral nerves that can result in functional impairment of organ systems innervated by most cranial, spinal, and autonomic (sympathetic and parasympathetic) nerves. Ganglionopathy is a form of PNS pathology impacting neuron cell bodies in the dorsal root ganglia, cranial nerve ganglia, and/or autonomic ganglia. In general, PNS syndromes present as neuropathies associated with damage to either axons (“axonopathies”) or myelin (“myelinopathies”) rather than neuronopathies or ganglionopathies resulting from damage to nerve cell bodies or ganglion cell bodies, respectively. The pathogenesis of neuropathies and ganglionopathies may be localized (one affected structure) or multicentric (affecting many structures) and may present as an acute or chronic condition. Neuropathies may be reversible or permanent, given the regenerative capacity of axons and Schwann cells (the PNS glia that produce myelin sheaths in nerves), while ganglionopathies are generally considered to be permanent since severely damaged neurons lack the ability to regenerate and thus die.
Historically, peripheral neuropathy has been discovered, more often than not, in humans exposed to noxious chemical agents following accidental exposures, environmental disasters, or occupational tasks. Published literature on common contemporary conditions associated with peripheral neuropathy not only includes exposure to chemicals (alcohol, 4 numerous industrial chemicals [see Table 1]) and drugs (chemotherapeutics, 42,43 antiretroviral agents, 38,39 statins, 44 fluoroquinolone antibiotics, 45 immunosuppressant biologics) 45 but also includes classic medical conditions involving metabolic disruption (eg, diabetes) and/or nutrient imbalances (eg, vitamin B excess as well as alcohol-related vitamin deficiencies). 52 Most published references on peripheral neuropathy, including those incited by toxicants, lie within experimental and/or investigative pathology settings, where the pathogeneses of these lesions have been evaluated in animal models and humans. 53,54,55 In general, toxic and metabolic/nutritional damage in the PNS manifests as a polyneuropathy inducing more or less bilaterally symmetrical lesions affecting many nerves. Although damage to the PNS leading to peripheral neuropathy has held a definitive place in the evaluation of neurotoxicity, many challenges remain in the identification, characterization, and translation of peripheral neuropathy findings in animal toxicity studies. The focus of this special issue is primarily within the realm of regulatory toxicologic peripheral neuropathology in the context of safety assessment of regulated products.
Examples of Peripheral Nervous System Toxicants.a
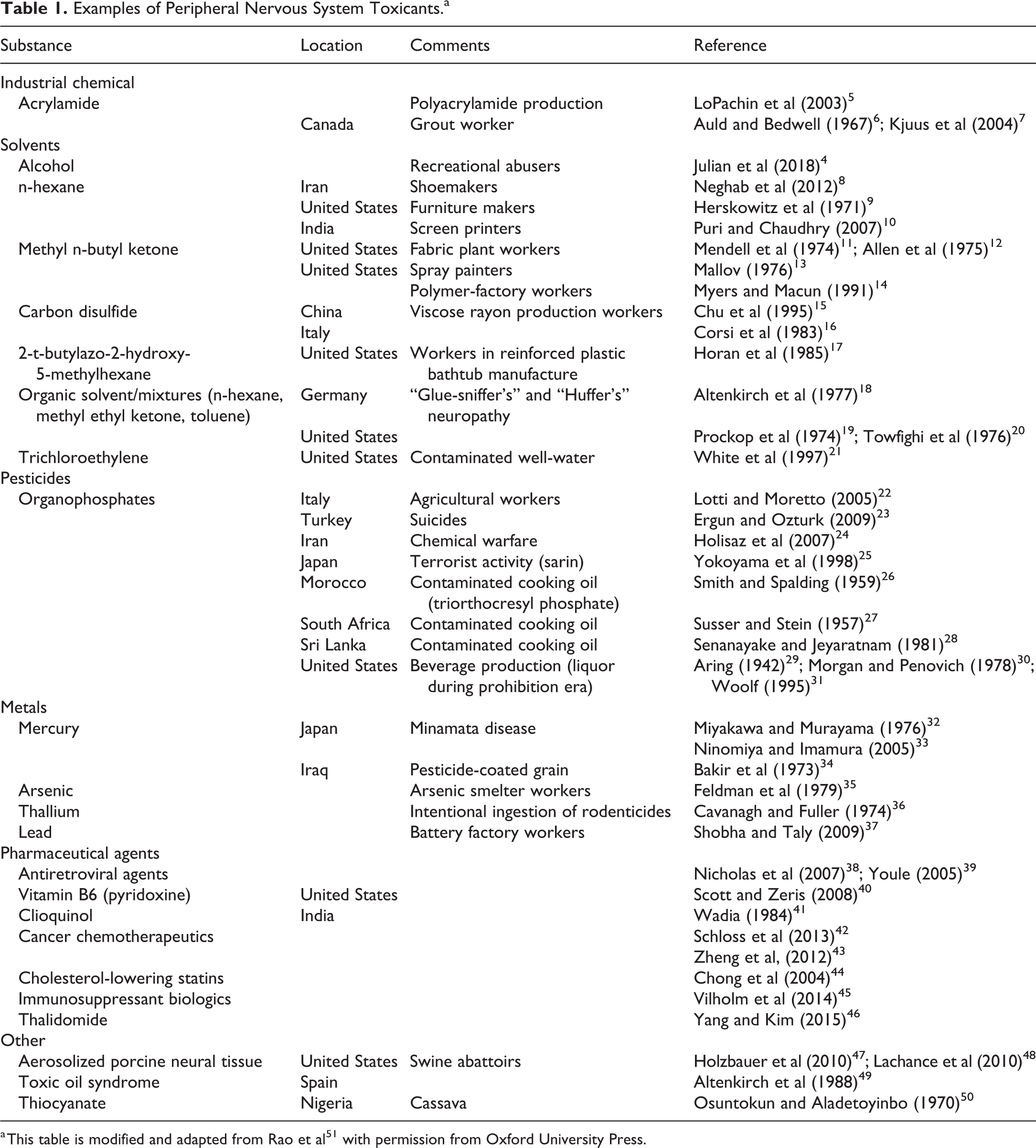
a This table is modified and adapted from Rao et al 51 with permission from Oxford University Press.
In the field of toxicologic pathology, especially regulatory toxicologic pathology, additional improvements in the assessment of PNS structure and function are needed to better understand the relationship among lesions, altered nerve function, and adversity in the context of regulatory submissions and clinical settings. However, the parameters defining appropriate sampling and processing to capture potential lesions in animal toxicity studies historically have been inconsistent and generally have lacked translational results between animals and humans. For example, assessment of functional end points indicative of potential peripheral neurotoxicity in clinical trials for pharmaceutical products would be better assessed by a neurologist rather than an internist. Screening for peripheral neurotoxicity in regulatory animal studies has generally been limited to histopathologic evaluation of the sciatic nerve using only hematoxylin and eosin–stained sections. However, experience in humans has shown that peripheral neuropathy often presents initially as a “glove-and-stocking” pattern of sensory deprivation (ie, functional deficits) affecting more distal nerve trunks and nerve endings. In animal studies, when clinical signs (such as decreased grip strength in functional observational batteries), excessive grooming, skin lesions, automutilation of digits, or dogs walking on the dorsal surface of their paws are noted, they should be investigated further via nerve conduction velocity tests (
End points for sensory neuropathy evaluation in animal toxicity studies is often neglected due to the specialized nature and limited implementation of the available techniques needed for such evaluations. A review of regulatory guidance documents for animal toxicity studies defining PNS evaluation generally reveals meager recommendations for sampling and processing (
In general, peripheral neurotoxicity evaluation in routine toxicity screening studies with test articles of unknown neurotoxic potential has been limited to assessment of clinical signs for evidence of late-stage motor disturbances. The lexicon for capturing such clinical signs is generally variable among institutions and limited to alterations in gait and movement. Traditional morphologic evaluation of the PNS in toxicity testing for new products typically is limited to a cursory examination of somatic (voluntary motor and sensory) constituents by assessing one major nerve and, occasionally, one or a few of its distal nerve branches. In contrast, autonomic elements that mediate involuntary motor and sensory functions to maintain homeostasis are largely ignored. As a result, the editors of this Special Issue have endeavored to collect and collate the experiences of toxicologic neuropathologists in the STP’s Special Interest Group in Neuropathology, participants in a recent continuing education course on the “Toxicologic Pathology of the Peripheral Nervous System” (STP Annual Meeting 2018), and allied scientists who perform PNS functional testing to provide readers with a wide spectrum covering fundamental principles and practical techniques for assessing toxicologic neuropathology affecting the PNS. This Special Issue is organized into two sections of related articles.
Part 1 is entitled “Methods for PNS Neurotoxicity Evaluation and Interpretation.” This section highlights conventional and other special morphologic techniques for sampling, processing, and evaluating multiple PNS components. Nuggets of practical information drawn from Dr Mark Butt’s
59
extensive experience are included in his article on the sampling and processing of PNS components. Determination of optimal fixation variables (fixative, processing cycle times, and water temperature) for the routinely prepared paraffin-embedded sections used in evaluating sciatic nerves is investigated by
Part 2 on “Toxicologic Neuropathology of the PNS” covers peripheral neuropathy findings commonly encountered in paraffin-embedded sections (
The Special Issue editors hope that this compilation of articles on toxicant-induced PNS neuropathology reflecting peripheral neurotoxicity to nerves and ganglia will be useful to readers of
Footnotes
Authors’ Note
This editorial reflects the views of Deepa B. Rao and should not be construed as representing views or policies of her previous employer—the US Food and Drug Administration.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
