Abstract
Recent “best practice” recommendations for peripheral nervous system sampling and processing provide guidance regarding nerve preparation for animal toxicity studies. This study explored the impact of delayed fixation, type of fixative, processing cycle times, starting ethanol concentration, and water bath temperature to improve nerve preservation in routinely prepared (paraffin-embedded, hematoxylin and eosin [H&E]-stained) sections. Sciatic nerves from adult Wistar rats (diameter, 1.04 ± 0.1 mm) and young domestic pigs (diameter 5.9 ± 1.2 mm) fixed at necropsy (“0” hours) or 3, 6, 12, or 24 hours after death were immersed in neutral-buffered 10% formalin containing 1.2% methanol (NBF) or methanol-free 4% formaldehyde (MFF) at room temperature. After fixation for 24 hours (rat) or 48 hours (pig), specimens were processed into paraffin, and ∼5-μm-thick sections were flattened on water baths set at 35°C, 40°C, or 45°C before H&E staining. Large-diameter nerves (pig) required longer processing cycles to ensure sufficient paraffin infiltration. For both small-diameter (rat) and large-diameter nerves, structural integrity was optimal if fixation by NBF or MFF occurred within 3 hours and the initial ethanol concentration for tissue processing was lowered to 50%. At all time points, structural preservation of nerve fibers was acceptable using NBF but was better with MFF. Use of a water bath at 35°C reduced processing-related nerve fiber separation within sections.
Introduction
Histopathological evaluation of the peripheral nervous system (PNS, comprised of nerves and ganglia) in conjunction with spinal cord (which carries central projections from the PNS) remains the “gold standard” for PNS hazard identification in animal toxicity studies for registering new biomedical products and chemicals. 1 –3 Recent “best practice” recommendations for Good Laboratory Practice (GLP)-compliant animal safety studies 3 are designed to address guidance provided by regulatory agencies around the world 4 while ensuring consistent, efficient, and effective sampling, processing, and evaluation of PNS tissues for the most common situations faced during both screening (general toxicity or “tier I”) and in-depth (dedicated neurotoxicity or “tier II”) studies. Screening is designed for routine pathologic assessment where the PNS is one system that needs to be evaluated and neurotoxicity is not anticipated. In this setting, organs and tissues from multiple systems are fixed by immersion (typically in commercial formulations of neutral-buffered 10% formalin [NBF] containing methanol as a stabilizer) and embedded in paraffin wax, after which sections stained with hematoxylin and eosin (H&E) are evaluated. More in-depth procedures are indicated when neurotoxicity affecting either the somatic (voluntary sensorimotor) and/or autonomic (involuntary homeostatic) PNS is suspected or likely to occur, and the nervous system is the single main focus of the study. For this latter scenario, animals are perfused with fixative (typically methanol-free aldehyde solutions) and embedded in paraffin or (for at least one nerve) plastic, after which serial sections are stained with H&E as well as special neurohistological stains to provide further information on cellular targets and/or neuropathologic mechanisms. 3,5 The choice of the methods to employ when evaluating the PNS for toxicity is made at the discretion of the sponsor. Where feasible, animal toxicity studies are conducted using the methods appropriate for general toxicity studies because immersion fixation and routine paraffin embedding are less laborious and also less costly.
The authors’ experience with current “best practice” recommendations for PNS sampling and processing 3 is that guidance regarding sciatic nerve evaluation during general toxicity studies does not address all factors that can impact nerve preservation. This study explored fixation and processing variables that might be adjusted to optimize nerve preservation for light microscopic evaluation of aldehyde-fixed, paraffin-embedded, H&E-stained sections for general toxicity studies in animals. Our data demonstrate that adjustments to several simple parameters in conventional nerve processing practices can substantially enhance structural integrity in conventional paraffin sections of sciatic nerve and that such adjustments vary with the species (or sample size).
Materials and Methods
Animals
Sciatic nerves were obtained as donations from dead adult Wistar rats (n = 35; total body weight, 225-340 g) and juvenile domestic pigs (n = 4; total body weight, approximately 30 kg) that had been assigned to other experimental protocols in which nerves were slated for disposal without use. All experiments for these animals had been approved in advance by the University of Missouri College of Veterinary Medicine and its Institutional Animal Care and Use Committee. All rats and pigs were control animals that had not been treated with any neurotoxic agent or subjected to any nerve-traumatizing procedure prior to necropsy.
Sciatic Nerve Harvesting
Sciatic nerves were collected bilaterally at necropsy (designated as time “0”), which was less than 15 minutes after euthanasia for rats or approximately 45 to 60 minutes after euthanasia for pigs. After reflection of the medial gluteal and biceps femoris muscles, the caliber (or diameter) of the exposed sciatic nerve was measured at mid-thigh with a fractional digital caliper (General Ultratech model 1433; Grainger Industrial Supply, Lake Forest, Illinois). For the rats, the proximal portion of the sciatic nerve covered by the medial aspect of the ilium was uncovered by removing the iliac crest with a small rongeur, after which the nerve was transected as close to the vertebral column as possible. In pigs, the sciatic nerve was severed just after the trunk coursed peripherally by passing through the greater sciatic foramen. With serrated forceps, nerves were carefully grasped at the proximal end and gently lifted, taking care to avoid stretching or compression. After elevation away from the underlying muscle, the nerves were cut at the popliteal fossa just above where the sciatic nerve branches into the tibial, fibular (or common peroneal), and sural nerves. The entire nerve was deposited on a wooden tongue depressor that had been wetted in advance with saline (pH 7.4), after which specimens were divided using a new scalpel blade to produce multiple segments of approximately 1 cm in length. Each segment was applied to a dry index card, after which samples were placed in fixative. For both species, segments from the proximal and distal portions of nerves were randomly distributed among the treatment groups.
Sciatic Nerve Fixation and Processing
Treatment groups (n = 4-6 nerve pieces/group) were defined by combinations of fixation and processing variables. “Fixation delay” as a factor comprised 5 time points: “0” (<15 minutes for rats, and 45-60 minutes for pigs = control), or fixation deferral for 3, 6, 12 or 24 hours after euthanasia. “Fixative solution” as a variable consisted of either a commercial formulation of NBF, consisting of approximately 4% formaldehyde containing 1.2% methanol as a stabilizer (Thermo Scientific, Cat No. 5705, Waltham, MA) or methanol-free 4% formaldehyde (MFF; Biotium, Inc, Cat No. 22023, Fremont, CA); MFF was included as an option since methanol is an organic solvent that has been posited to negatively impact lipid preservation in myelin sheaths. 3 For fixation, control nerve segments were immersed at time “0” in 3 mL of either NBF or MFF in 15-mL conical test tubes, while for other time points the nerve segments were deposited in empty (air-containing) 15-mL test tubes. All test tubes were sealed (to prevent fixative evaporation for controls or sample dehydration for deferred times 6 ) and held at room temperature (RT). After the end of the fixation delay, nerve segments in empty 15-mL test tubes were immersed in 1 mL of either NBF or MFF. Fixation was continued at RT for 24 hours for rats (small-diameter nerves) or 48 hours for pigs (large-diameter nerves).
Fixed nerves were trimmed in cross (transverse) and longitudinal orientations and processed into paraffin. “Starting ethanol concentration” as a factor was evaluated by comparing the initial concentration as used routinely in animal toxicity studies (70% 7 ) with a reduced starting concentration (50%, selected to permit a more gradual removal of water from lipid-rich nerve tissue). The ethanol used for histological processing in this study was a commercial preparation of reagent alcohol (ie, denatured ethanol mixed with small amounts of methanol and isopropyl alcohol). “Processing cycle lengths” as a variable consisted of adjusting the length of time spent incubating in each station on an automated tissue processor, adjusted based on the nerve diameter. In this study, a conventional processing method involved processing cycle lengths of 10 minutes or 40 to 60 minutes, which were applied on both small-diameter and large-diameter nerves. The optimal processing time was set at 20 minutes for small-diameter nerves and 45 minutes for large-diameter nerves. Following paraffin infiltration, cross and longitudinal segments of each specimen were embedded in a single-paraffin block. Approximately 5-μm-thick sections were cut from each block using either a semi-automated or a conventional manual microtome. “Water bath temperature” as a factor was evaluated by placing sections on water baths set at 35°C, 40°C, or 45°C, where the usual temperature used for flattening sections of non-neural tissues is between 40°C and 44°C. 7 Sections were captured on unfrosted glass or charged slides, stained with H&E using an automated histostainer, and cover-slipped with permanent mounting media (either Optic Mount Toluene; Mercedes Scientific, Lakewood Ranch, Florida or Tissue-Tek Glas medium; Sakura Finetek USA, Inc, Torrance, California).
Sciatic Nerve Evaluation
Sections were assessed by bright-field light microscopy for structural integrity and background findings. Serial examinations were performed independently by 2 American College of Veterinary Pathologists’ board-certified veterinary anatomic pathologists (J.S.F. followed by B.B.). The first parameter consisted of a qualitative evaluation of “distortion” (absence or presence of fissures). Two additional parameters were evaluated using tiered, semi-quantitative scoring schemes in both cross- and longitudinal sections: “preservation” (ie, how well axonal and myelin sheath integrities were maintained relative to the “0” time point) and “spread” (ie, how closely nerve fibers were packed within the section). The preservation score ranged from very good (ie, within normal limits), good, fair, down to poor. This score included the degree of an artifactual change termed “bubbling” indicative of clear and unstained vacuoles commonly observed diffusely in myelin sheaths of nerves following immersion fixation. 8 The spread score was assigned as none (ie, within normal limits), minimal, mild, moderate, or marked. The spread score is interdependent with the effect of water bath temperature (proportional relationship). These descriptive data were not subjected to statistical analysis. Representative images were obtained using a ScanScope XT at ×20 (Aperio Technologies, Inc, Vista, California).
Results
Independent assessments by 2 pathologists produced results indicating a similar interpretation for each of the factors defined above for both individuals (data not shown). No spontaneous background findings were observed in any of the sciatic nerve sections from either rats (n = 93 total pairs of sections [ie, cross and longitudinal orientations from a single nerve] from 35 animals) or pigs (n = 76 total pairs of sections from 4 animals).
Myelin Integrity Degrades If Fixation Is Delayed by 6 Hours or More
In this study, the diameter of adult rat sciatic nerves was 1.04 ± 0.1 mm (“small diameter”), while the diameter of juvenile pig sciatic nerves was 5.9 ± 1.2 mm (“large diameter”). For both small-diameter and large-diameter nerves, tissue and cytoarchitectural integrity was well preserved if fixation occurred within 3 hours by immersion in either NBF or MFF (Figures 1 and 2). At 6 hours, myelin sheaths exhibited “bubbling” (appearing as an irregular pattern of clear vacuoles covered by a thin mesh of intersecting eosinophilic strands), but the sheaths retained their regular diameter and remained intact. The degree of artifactual bubbling was accentuated if the interval of delayed fixation was longer than 6 hours and was especially pronounced at 12 hours (“overnight”) and 24 hours (Figures 1 and 2). For both small-diameter and large-diameter nerves maintained fresh after 6 hours, preservation was considered fair to poor following immersion fixation with NBF or MFF. A spread score of minimal to mild was attributed in each condition regarding the delay of fixation or type of fixative.
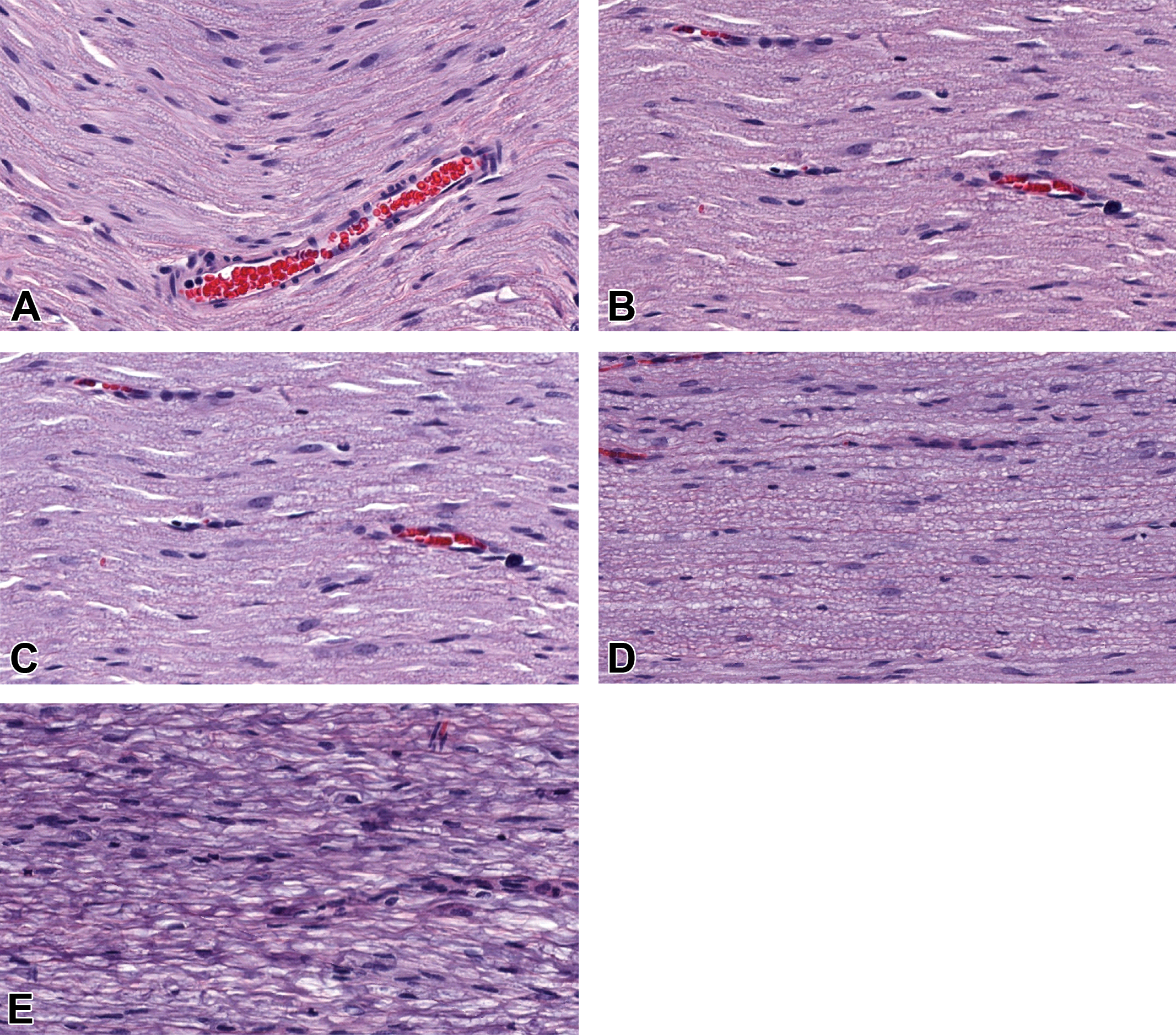
Morphological changes, chiefly “bubbling” (vacuolation) of myelin sheaths, in small-diameter rat sciatic nerves (longitudinal sections) were associated with delayed fixation using neutral-buffered 10% formalin (NBF). Times represent hours of fixation delay relative to necropsy (time “0”, equivalent to 15 minutes in rats). Such changes were minimal for NBF and if fixation was delayed up to 3 hours. Similar results were obtained with large-diameter pig sciatic nerves (data not shown). Delay of fixation: 0 hour (A), 3 hours (B), 6 hours (C), 12 hours (D), and 24 hours (E). Original objective magnification ×20. Additional processing: paraffin embedding, H&E staining.
Vacuolation of myelin sheaths in small-diameter rat sciatic nerves (longitudinal sections) was associated with delayed fixation using methanol-free 4% formaldehyde (MFF). Times represent hours of fixation delay relative to necropsy (time “0”, equivalent to 15 minutes in rats). Such changes were minimal for MFF if fixation was delayed up to 6 hours. Delay of fixation: 0 hours (A), 3 hours (B), 6 hours (C), 12 hours (D), and 24 hours (E). Original objective magnification ×20. Additional processing: paraffin embedding, H&E staining.
Preservation of Myelin Sheaths Is Improved by Fixation in MFF Rather Than NBF
Structural preservation of nerve fibers was acceptable using NBF but was better using MFF (Figures 1 and 2). This interpretation was based chiefly on the degree of bubbling seen in the myelin sheaths. When fixation occurred at necropsy (time “0”), preservation scores for MFF-fixed sections were “very good” for both small-diameter and large-diameter nerves, while those for NBF-fixed sections were “very good” for small-diameter nerves but only “good” for large-diameter nerves. After a fixation delay of 3 hours, preservation scores for small-diameter nerves were “good” or “very good” for both fixatives, while for large-diameter nerves, MFF yielded “good” scores and NBF typically produced “fair” scores. At later time points, both small-diameter and large-diameter nerves exhibited a comparable degree of myelin degradation. For a 6-hour delay, both MFF and NBF yielded “fair” or occasionally “good” scores, with MFF having slightly more “good” scores. At 12 hours, MFF-fixed sections received preservation scores of “fair” to sometimes “good,” while NBF-fixed sections were always “fair.” A “poor” preservation score was given to all NBF-fixed sections but only to a few MFF-fixed sections only after a fixation delay of 24 hours.
Nerve Section Integrity Depends on Diameter-Dependent Changes to Processing Conditions
The processing cycle conditions that proved most suitable for maintaining histological features of small-diameter versus large-diameter nerves are detailed in Table 1. A lower starting concentration of ethanol (50%) reduced the degree of distortion (fissures, irregular compression, and/or partial collapse) of nerve fascicle organization in contrast to the higher starting concentration of ethanol (70%) used in conventional processing encountered when all tissues (including nerves) from a given study are processed together, which is the typical practice for general toxicity studies in animals (Table 1 and Figure 3). This difference in optimizing myelin sheath integrity was especially notable for large-diameter nerves. Inclusion of a processing station with a lower starting concentration of ethanol to provide the best nerve preservation adds one more station of 20 minutes (for small-diameter rat nerves) or 45 minutes (for large-diameter pig nerves) to the processing protocol to optimally process sciatic nerves relative to non-neural tissues (Table 1).
Transverse sections of small-diameter (rat, A and B) and large-diameter (pig, C and D) sciatic nerves prepared after conventional processing parameters exhibited distortion (fissures, irregular compression, and/or partial collapse) of nerve fascicle organization (A and C) in contrast to sciatic nerves prepared with optimal conditions (B and D) as defined in Table 1. The optimal protocol for nerve processing includes a lower starting concentration of ethanol (50%) and longer processing cycles (20 minutes for small-diameter nerves and 45 minutes for large-diameter nerves), both of which preserve nerve structure by promoting more gradual tissue dehydration. Original objective magnifications: top row = ×10 (rat), bottom row = ×10 (pig). Additional processing: methanol-free 4% formaldehyde (MFF) fixation for 24 hours (rat) or 48 hours (pig), paraffin embedding, H&E staining.
Specific Processing/Embedding Cycle Used for Small-Diameter (Rat) and Large-Diameter (Pig) Sciatic Nerves.a
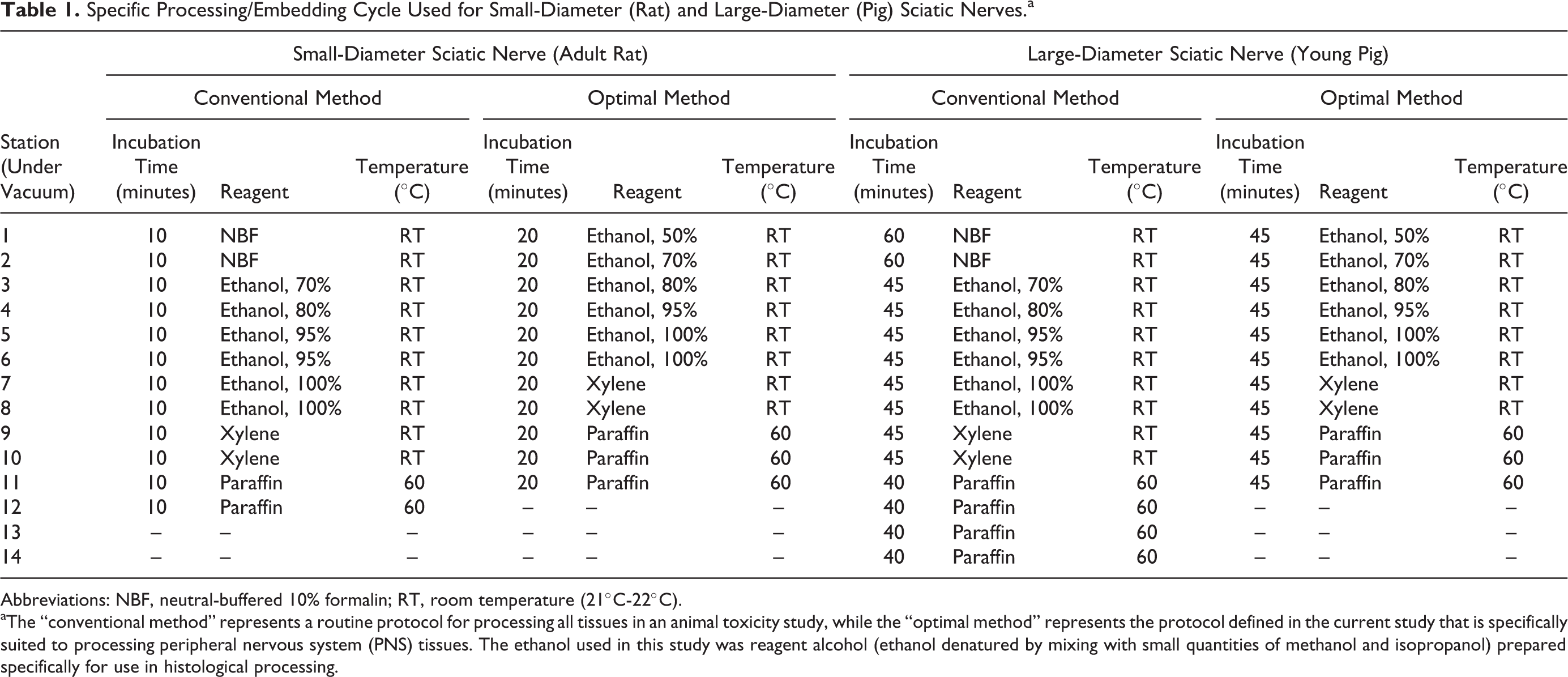
Abbreviations: NBF, neutral-buffered 10% formalin; RT, room temperature (21°C-22°C).
aThe “conventional method” represents a routine protocol for processing all tissues in an animal toxicity study, while the “optimal method” represents the protocol defined in the current study that is specifically suited to processing peripheral nervous system (PNS) tissues. The ethanol used in this study was reagent alcohol (ethanol denatured by mixing with small quantities of methanol and isopropanol) prepared specifically for use in histological processing.
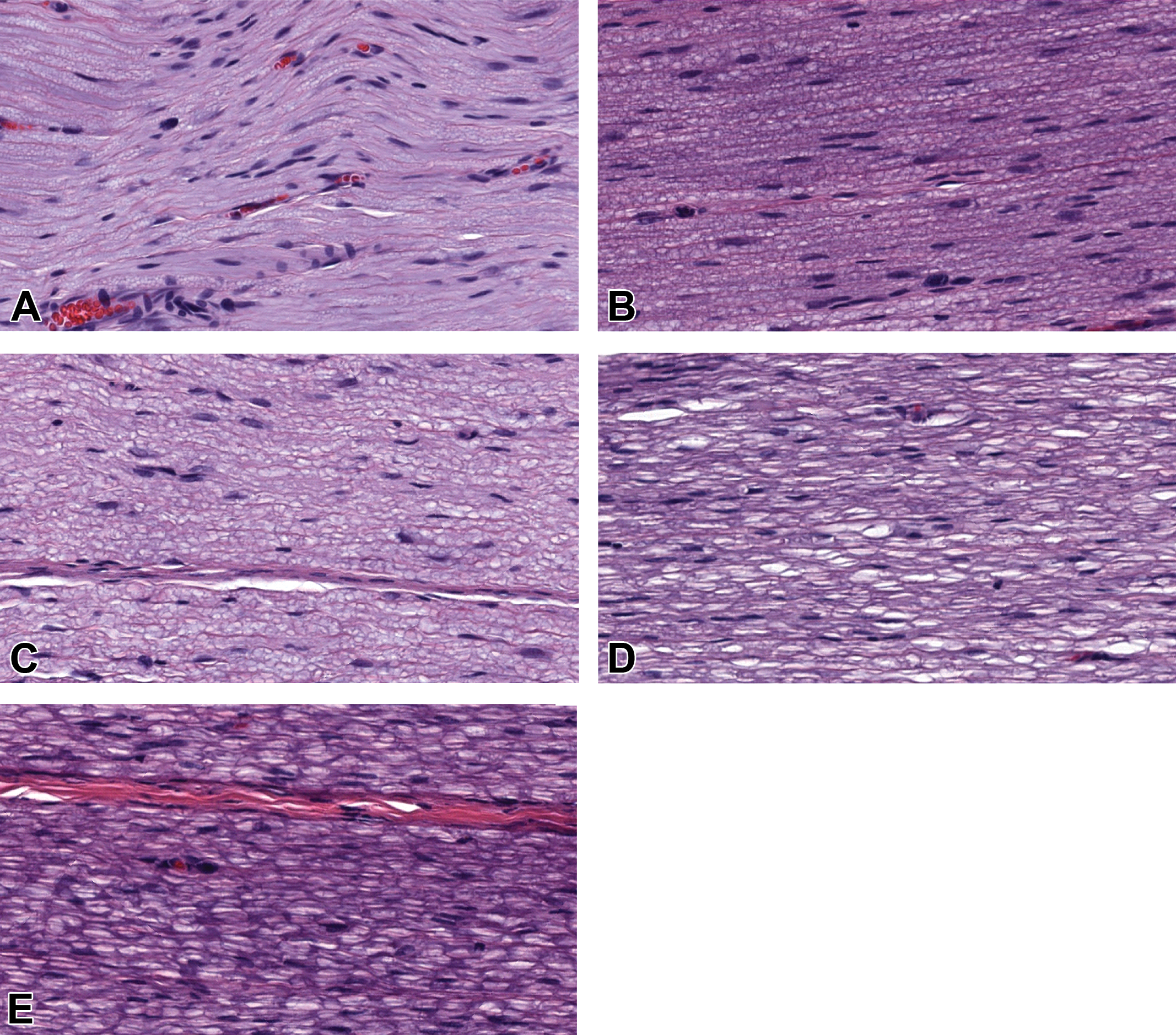
Large-Diameter Nerves Require Longer Processing Times
Large-diameter nerves (pig) required longer processing times per station (45 minutes) compared to small-diameter nerves (rat, 20 minutes per station) to retain myelin architecture. Tissue integrity was distorted, folded, and cracked, especially in longitudinal sections, if large-diameter nerves were prepared using the short incubation times that proved suitable for small-diameter nerves (Figure 4).
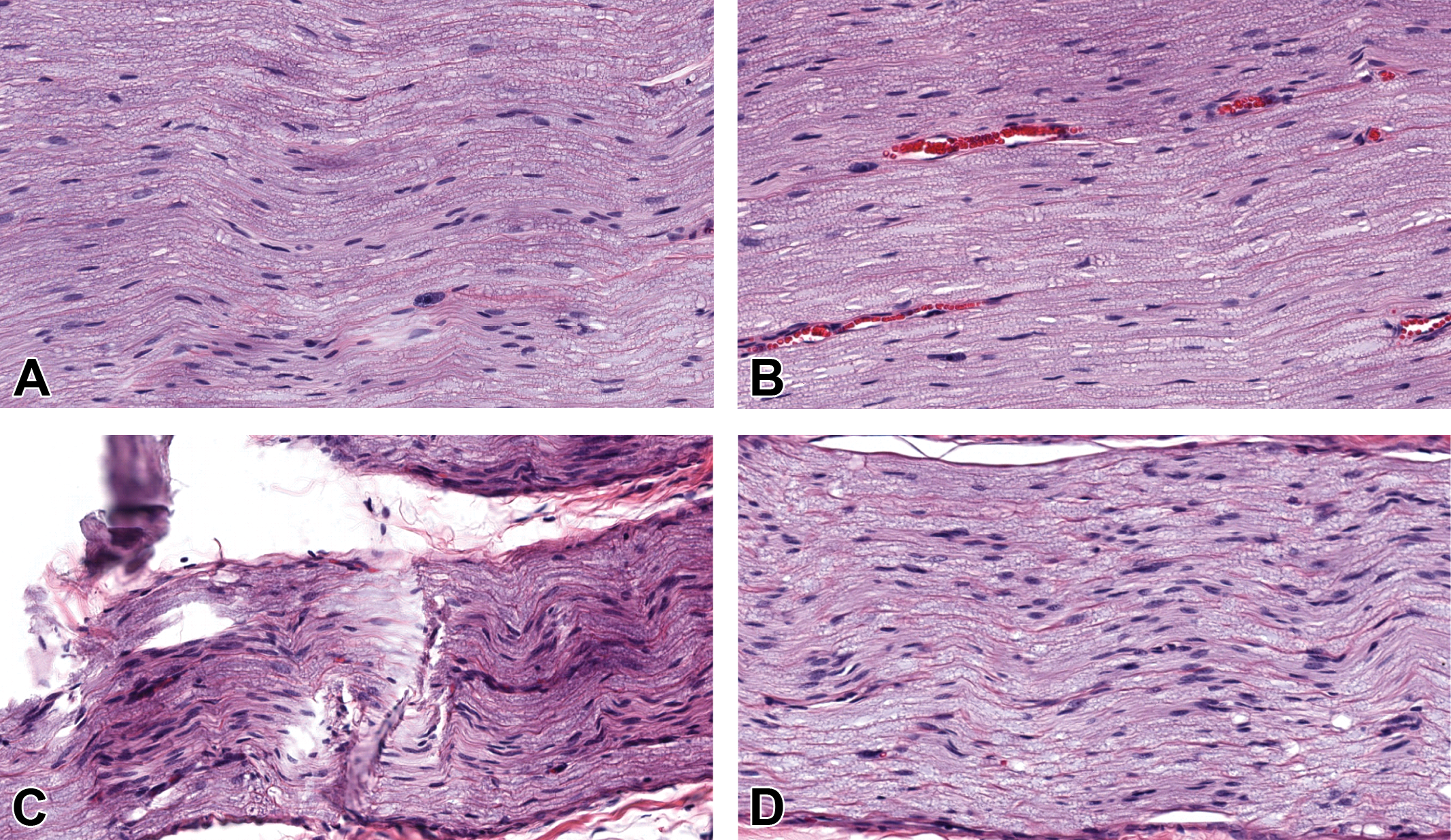
Different processing times per tissue processor station were required for optimal preservation of nerve profiles in small-diameter (rat, A and B) and large-diameter (pig, C and D) sciatic nerves (longitudinal sections). Tissue quality for small-diameter nerves was comparable for both short cycles (10 minutes per station, A) and long cycles (45 minutes per station, B). Large-diameter nerves were distorted and fragmented when processed using short cycles (10 minutes per station, C), and only long cycles yielded intact sections with well-preserved architecture (45 minutes per station, D). Original objective magnification: ×20. Additional processing: neutral-buffered 10% formalin (NBF) fixation for 24 hours (rat) or 48 hours (pig), paraffin embedding, H&E staining.
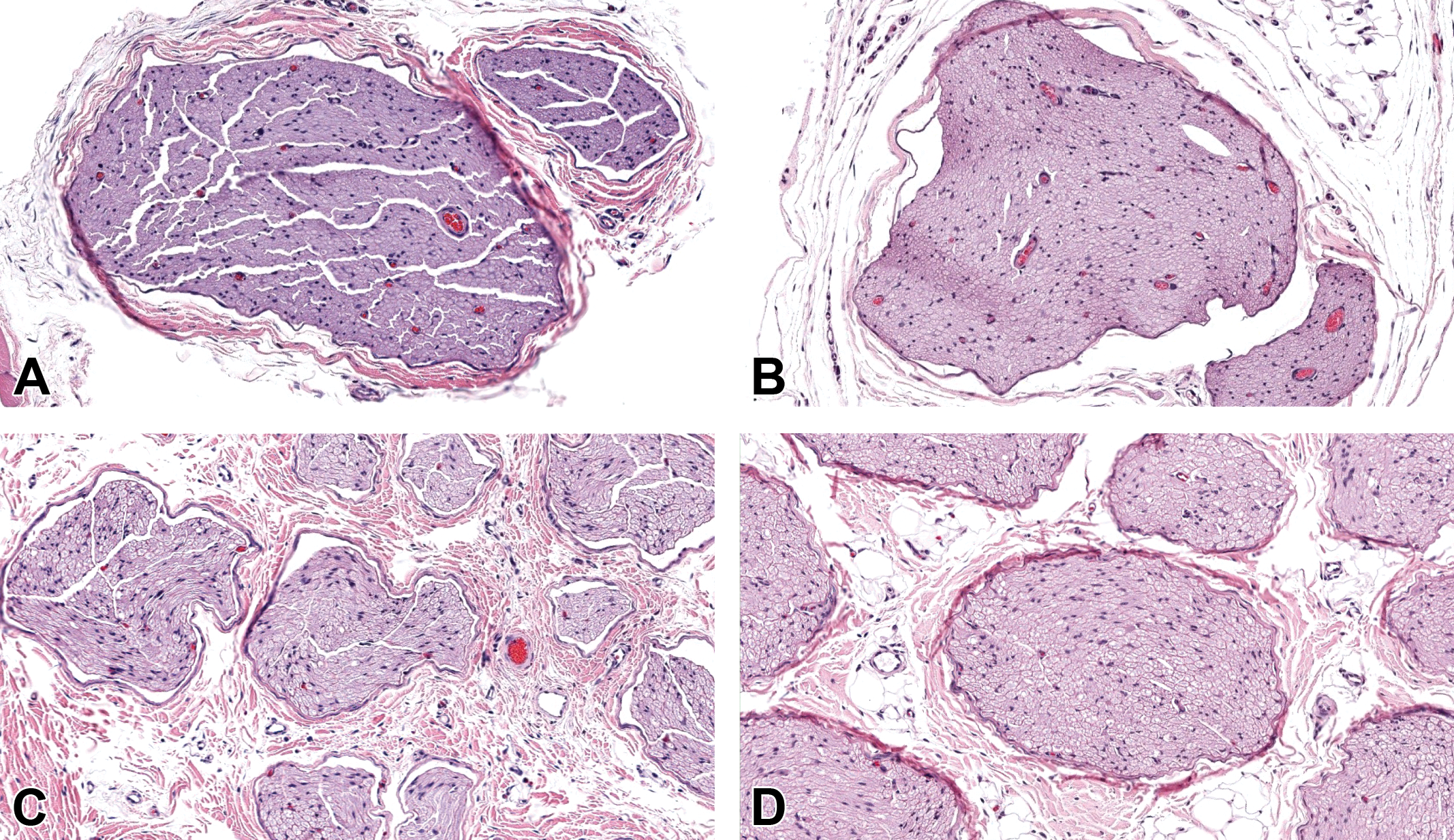
Lower Water Bath Temperatures Reduce Processing-Related Nerve Fascicle Separation
For both small-diameter (rat) and large-diameter (pig) sciatic nerves, use of a water bath at 35°C reduced processing-related section dissolution (ie, nerve fascicle separation) better than temperatures of ≥40°C (Figure 5). Nerve fibers, especially myelin sheaths, retained optimal cytoarchitectural integrity if sections were floated on 35°C water baths compared to higher temperatures.
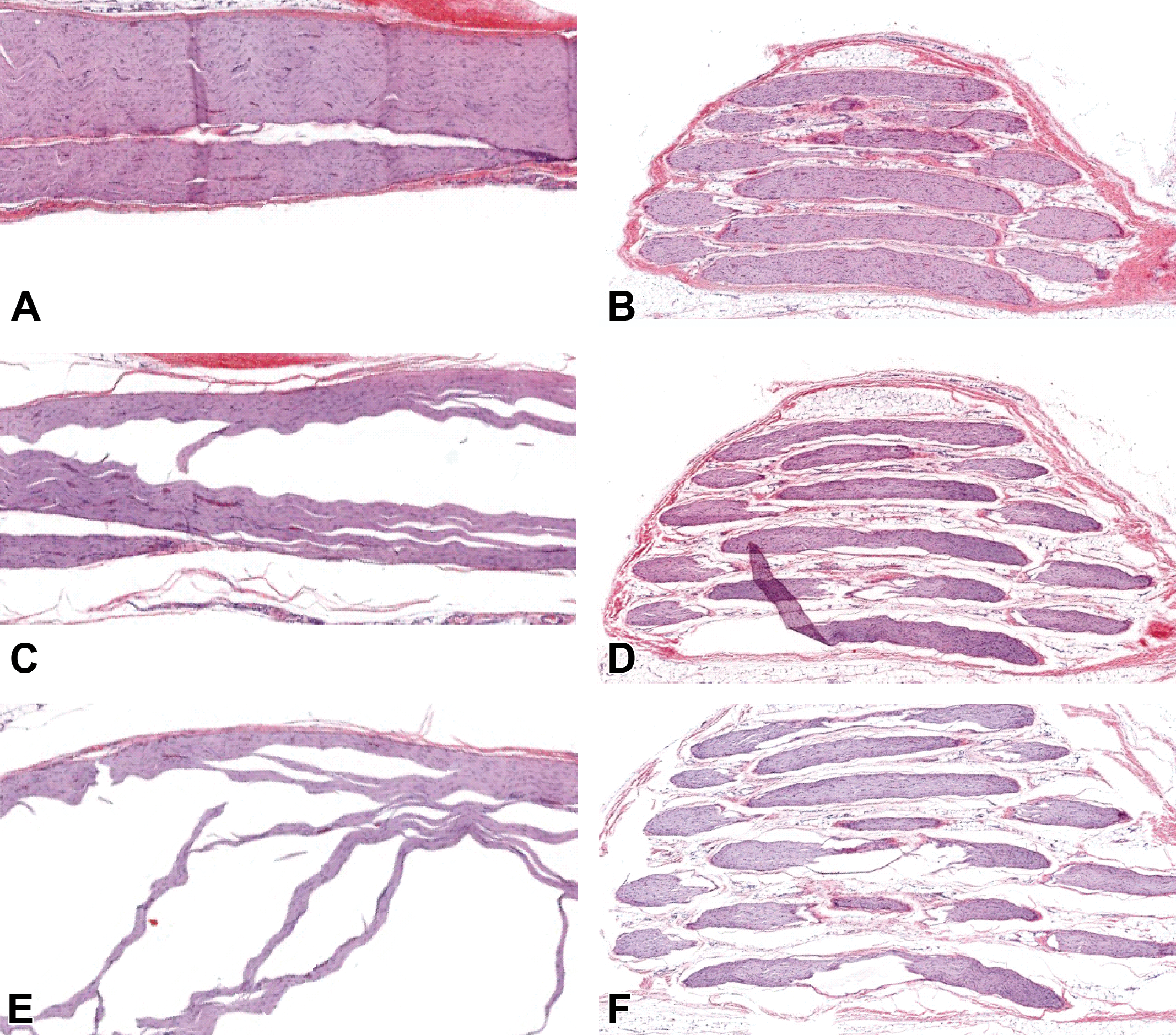
Longitudinal sections of small-diameter (rat, A, C, and E) and large-diameter (pig, B, D, and F) sciatic nerves were less expanded (ie, exhibited less artifactual loss [“shelling out”] of nerve fibers and separation of nerve fascicles) when floated on water baths with a lower temperature (35°C) relative to baths set at the higher temperatures typically used for other tissues (40°C or 45°C). Water bath temperature: 35°C (A and B), 40°C (C and D), 45°C (E and F). Original objective magnification: ×1. Additional processing: neutral-buffered 10% formalin (NBF) fixation for 24 hours (rat) or 48 hours (pig), paraffin embedding, H&E staining.
Discussion
Peripheral nerves are very susceptible to development of processing artifacts that can mimic pathological changes. 9 In this study, the impact of different parameters such as delayed fixation, type of fixative, processing times, starting ethanol concentration, and water bath temperature on sciatic nerve preservation was investigated to provide further information for optimizing PNS processing for animal studies. The paucity of information on these parameters applied to PNS fixation and processing for tissues from GLP-compliant safety studies was the rationale for the present optimization study.
A delay of fixation was the first variable tested since artifactual changes such as diffuse swelling of cells and loss of resolution and dissolution of membranes (particularly myelin sheaths of nerve fibers) are observed if fixation is delayed. 9 We explored the degree to which sciatic nerve fixation could be delayed and still yield interpretable features. Dissection of PNS samples in general toxicity studies in animals often occurs toward the end of the necropsy, which engenders a delay ranging up to 30 minutes in rodents and 1 hour in large non-rodents at scheduled terminal procedures. For unscheduled deaths, animals may be found an unspecified number of hours after death, while animals that die late in the day may be held overnight (typically in a cooler at 4°C-8°C) leading to longer delays in fixation that may have a significant impact on tissue preservation. In this study, fixation delays of up to 3 hours at RT still conserved cellular architecture in the axons and myelin of both small-diameter and large-diameter sciatic nerves, while lag times over 3 hours led to modest degradation of myelin for both small-diameter and large-diameter trunks. Even shorter lag times in fixation are necessary to retain molecular signatures in tissue for histochemistry and immunohistochemistry, with known limitations for embryonic and adult tissues being limited to 15 to 20 minutes and 45 to 50 minutes, respectively. 10,11 Fixation delays of up to 6 hours may be possible if the unfixed specimens are stored at 4°C since cold temperatures are known to retard autolytic changes. 11 This study did not explore the impact of refrigeration on nerve preservation, but this parameter was judged to be irrelevant given the routine practice in general toxicity studies to acquire and immediately fix PNS tissues at necropsy (ie, within 30 minutes for small-diameter nerves of rodents and 1 hour for large-diameter nerves of nonrodents).
The second variable, the choice of fixative for immersion fixation, evaluated conventional NBF (the usual choice for general toxicity studies 7,12 ) versus MFF. Use of NBF has 2 advantages, the ability to mingle PNS samples with other tissues in a single container for each animal and cheapness, while MFF has the advantage of having no methanol (which may damage myelin lipids 3 ) but is costlier and will require preparation (ie, labeling and filling) of a second container to hold PNS tissues for fixation. Both MFF and NBF provide suitable myelin preservation of small-diameter rat nerves, while MFF affords modestly enhanced myelin protection in large-diameter pig nerves relative to NBF. If nerves are fixed at necropsy, our data suggest that NBF is a suitable fixative in general toxicity studies for rodents (where no substantial myelin dissolution develops) and nonrodents (where any minimal degradation in large-diameter trunks would not be severe enough to confound the evaluation for subtle neurotoxic effects). Of course, optimal preservation for detailed evaluation of PNS tissues as recognized in current best practice recommendations requires initial fixation by perfusion rather than immersion and (for at least one nerve) postfixation in glutaraldehyde and osmium tetroxide (to better stabilize myelin) followed by embedding in hard plastic resin (to permit production of thinner sections with better resolution of cellular features, especially myelin sheaths). 3
The third variable evaluated was the starting concentration of ethanol used for the initial dehydration step in tissue processing. The ethanol concentration gradient has an impact on preservation of cellular features of lipid-rich tissues, including myelin sheaths of nerves. Conventional processing begins with an initial dehydration step of 70% alcohol, which is suitable for most tissues. 7 In this study, optimal preservation of nerve fiber integrity resulted from processing that employed a lower concentration of ethanol (50%) in the initial step. The more gradual concentration gradient provided by this additional ethanol step affords more complete dehydration. Expanding the concentration gradient by adding a lower starting point (30%-50%) provides for slower and less destructive removal of cell and tissue water and limits abrupt passive diffusion of fluid across membranes that may cause cellular distortion. 7 These data highlight the advisability of devising discrete processing programs for specific tissues based on their unique biochemical properties.
The length of processing stations on automated tissue processors was the fourth parameter to be assessed. Protocols for processing PNS tissues vary among different histology laboratories, and it seems likely that some facilities will utilize a single common setup for most or all tissues without acknowledging that delicate PNS tissues deserve special consideration to produce the best structural preservation. Processing stations must be of sufficient length to ensure that tissue water has been fully replaced by infiltrating paraffin, and this requirement is especially true for lipid-rich PNS samples (Table 1). Utilization of short processing cycles applied to large-diameter sciatic nerves leads to extensive artifactual loss of architectural detail and clarity as well as cutting difficulties leading to section distortion or fragmentation due to inadequate paraffin infiltration. 13 The results of this study emphasize the importance of developing dedicated processing programs for specific tissues based on the divergent properties of samples from different species. 14
The impact of water bath temperature on section quality was the final variable evaluated in this study. According to our data, the delay of fixation and type of fixative do not have an impact on spread score. Water bath temperature can cause tissue expansion, and the extent of expansion depends on the tissue composition. Use of a water bath at 35°C reduced processing-related section artifacts—specifically dispersal of tissue elements—compared to either 40°C or 45°C. This fact may reflect basic nerve structure as hotter temperatures will promote more rapid melting of paraffin leading to release of nerve fascicles that lack the internal mesh of fibrous tissue that serves to stabilize organ integrity in non-neural sites. Low-temperature water baths will lessen paraffin melting and may permit floating sections to fold, which will obscure tissue architecture in the superimposed tissues. 15,16 Therefore, a fine line exists when selecting a water bath temperature for nerve processing in terms of which artifact is to be preferred: excessive spreading or folding; the authors noted this challenge particularly with respect to the large-diameter sciatic nerves. An implication arising from these observations is that histology laboratories tasked with processing multiple tissue types, as is typical of facilities that prepare tissues from animal toxicity studies, may need to develop PNS-specific sectioning protocols to permit procurement of the highest-quality nerve sections. Longer processing cycles in combination with lower water bath temperatures may be the best means of optimizing nerve section quality, especially for nonrodent PNS tissues.
Additional sources of PNS degradation deserve consideration but were not explored systematically in this study. The decision not to attempt optimization of these parameters was made based on existing literature that provides some guidance regarding their potential impact on PNS preservation. For example, the length of fixation can be estimated by the rate of aldehyde diffusion into tissue, which is estimated at 1 mm/h but may be slower for lipid-rich tissues. 7 In general, specimens should be fixed for 6 to 72 hours. 17,18 For specimens of 10 mm in thickness, fixation durations of 8 to 12 hours are generally indicated. 17 Prolonged fixation for more than 72 hours typically should be avoided because it may cause secondary shrinkage and hardening, nonspecific background staining, and loss of antigenicity. 19 Based on these facts, we calculated that fixation durations of 24 and 48 hours were suitable for small-diameter (rat, average diameter of ∼1.0 mm) and large-diameter (pig, average diameter of ∼6.0 mm) sciatic nerves, respectively. Our histopathological evaluation confirmed that PNS tissue integrity was preserved acceptably using these fixation times.
Similarly, most large-scale general toxicity studies (eg, n = 80 per study, or 10 animals/sex for each of 4 dose groups) utilize one of the 2 temperatures for the fixation step: RT (usually 21°C-22°C) or refrigeration (typically 4°C). Primary fixation at lower temperatures is known to slow autolysis but also will slow fixation. Even if immersed in ice-cold fixative, premature cooling of the unfixed tissue may lead to inadequate fixation of the specimen core due to slowed penetration by the fixative agent. 20 Fixation in warm buffered formaldehyde can increase the rate of fixation, 12 but also has the potential to increase the rate of autolysis and agglomeration of fixed molecules, impeding deeper penetration of the fixative agent. Histological and immunohistochemical features of biopsy samples from carcinomas (breast, colon pancreas, stomach) preserved in NBF have comparable features when fixed at low temperature and RT. 21 Taken together, these factors indicate that RT fixation, which is used most commonly in animal toxicity studies for convenience, is a suitable practice for PNS tissues.
In summary, existing regulatory guidelines 4 and best practice recommendations designed to address this regulatory guidance 3 provide direction for conducting the PNS assessment in animal toxicity studies but do not address all possible variables that can provide optimally preserved and processed PNS samples. The current experiment was designed to examine some key factors that have not been covered in these documents. Our current data have provided information regarding additional means of optimizing nerve preservation in general toxicity studies. First, nerve structure is best maintained when samples are fixed within 3 hours of death. Second, architectural definition in myelin sheaths of large-diameter (non-rodent) nerves is improved with MFF fixation, although for practical purposes NBF is suitable given the brief delay in fixation that characterizes typical necropsy practices for animal toxicity studies. Third, lipid-rich nerve tissue benefits from incorporating a more gradual dehydration process, by including an initial 50% ethanol bath as the first station on the automated tissue processor. Fourth, large-diameter nerves need longer processing cycles (45 minutes per station) relative to small-diameter nerves (20 minutes per station) to ensure optimal dehydration and paraffin infiltration. Finally, nerve sections retain optimal integrity if sections are floated on cooler water baths at 35°C compared to the higher temperatures typically used for other tissues. These new data should help institutions make rational decisions regarding whether or not, and how, to alter PNS fixation and processing practices to provide the best possible microscopic assessment of potential test article–related nerve lesions or in dedicated neurotoxicity study.
Footnotes
Acknowledgments
The authors would like to thank Dr Erin K. O’Connor, manager of Clinical Veterinary Services for the Office of Animal Resources at the University of Missouri, Columbia, MO, for her valuable assistance. The authors are grateful to the following University of Missouri principal investigators and laboratory members for sharing unwanted PNS tissues: Dr Frank W. Booth and Marie Elliot for the rat donations; and Dr Craig A. Emter, Dr Dylan T. Olver, and Jenna Edwards for the pig donations. The authors wish to acknowledge Roni Archuleta, Mark Butters, Susan Helming, and Candace Kassel for histologic technical support provided during the study.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
