Abstract
Peripheral nervous system (PNS) toxicity is surveyed inconsistently in nonclinical general toxicity studies. These Society of Toxicologic Pathology “best practice” recommendations are designed to ensure consistent, efficient, and effective sampling, processing, and evaluation of PNS tissues for four different situations encountered during nonclinical general toxicity (screening) and dedicated neurotoxicity studies. For toxicity studies where neurotoxicity is unknown or not anticipated (situation 1), PNS evaluation may be limited to one sensorimotor spinal nerve. If somatic PNS neurotoxicity is suspected (situation 2), analysis minimally should include three spinal nerves, multiple dorsal root ganglia, and a trigeminal ganglion. If autonomic PNS neuropathy is suspected (situation 3), parasympathetic and sympathetic ganglia should be assessed. For dedicated neurotoxicity studies where a neurotoxic effect is expected (situation 4), PNS sampling follows the strategy for situations 2 and/or 3, as dictated by functional or other compound/target-specific data. For all situations, bilateral sampling with unilateral processing is acceptable. For situations 1–3, PNS is processed conventionally (immersion in buffered formalin, paraffin embedding, and hematoxylin and eosin staining). For situation 4 (and situations 2 and 3 if resources and timing permit), perfusion fixation with methanol-free fixative is recommended. Where PNS neurotoxicity is suspected or likely, at least one (situations 2 and 3) or two (situation 4) nerve cross sections should be postfixed with glutaraldehyde and osmium before hard plastic resin embedding; soft plastic embedding is not a suitable substitute for hard plastic. Special methods may be used if warranted to further characterize PNS findings. Initial PNS analysis should be informed, not masked (“blinded”). Institutions may adapt these recommendations to fit their specific programmatic requirements but may need to explain in project documentation the rationale for their chosen PNS sampling, processing, and evaluation strategy.
Keywords
Background
Neurological deficits due to toxicant-induced peripheral neuropathy are a recognized consequence of accidental occupational or environmental exposures and some therapeutic treatments. Therefore, the neuropathology component of toxicity studies is a critical means for identifying potential hazards and assessing risks posed to humans by contact with new biomolecular or chemical entities.
Different regulatory agencies offer independent guidance 1 based on their distinct mandates, variable scientific levels of concern, and diverse uses of the agents they oversee regarding the specimens and procedures to be used in evaluating the integrity of the peripheral nervous system (PNS) when seeking to register new products (Bolon et al. 2011; Salvo and Butt 2011). The guidelines vary by the kind of industry (agrochemical vs. chemical vs. pharmaceutical vs. biopharmaceutical), differences in potential exposure levels, and ages of the test subjects (e.g., developing animals: Organisation for Economic Co-operation and Development [OECD] 2007; US Environmental Protection Agency [US EPA] 1998b vs. adults: OECD 1997; US Environmental Protection Agency 1998a). Guidelines also differ based on the aim of the study (hazard identification vs. safety assessment). For example, regulatory guidelines for performing the neuropathology analysis of good laboratory practice (GLP)-type general toxicity studies (i.e., screening or “Tier I” surveys) are fairly general since such studies assess the PNS as just one system among many organs and systems to be surveyed. In contrast, guidelines for GLP-type dedicated neurotoxicity studies (i.e., advanced or “Tier II” studies) are fairly detailed since assessment of the nervous system is the primary focus of the study (Bolon et al. 2011; Salvo and Butt 2011). However, substantial differences exist in the kinds of PNS toxicity that might be encountered (Table 1), and current guidelines do not address variations in approach that might be necessary to adequately investigate these divergent scenarios. Recent compilations reviewing published regulatory guidance in this area (Bolon et al. 2011; Salvo and Butt 2011) and/or individual regulatory guidelines should be consulted because guidance is reviewed and revised over time—as is presently occurring for the Toxic Substances Control Act (administered by the US EPA) and the “Redbook” guidance on food and color additives (overseen by the US Food and Drug Administration).
Different GLP-type Nonclinical Toxicity Studies in Which PNS Neurotoxicity Is Evaluated.
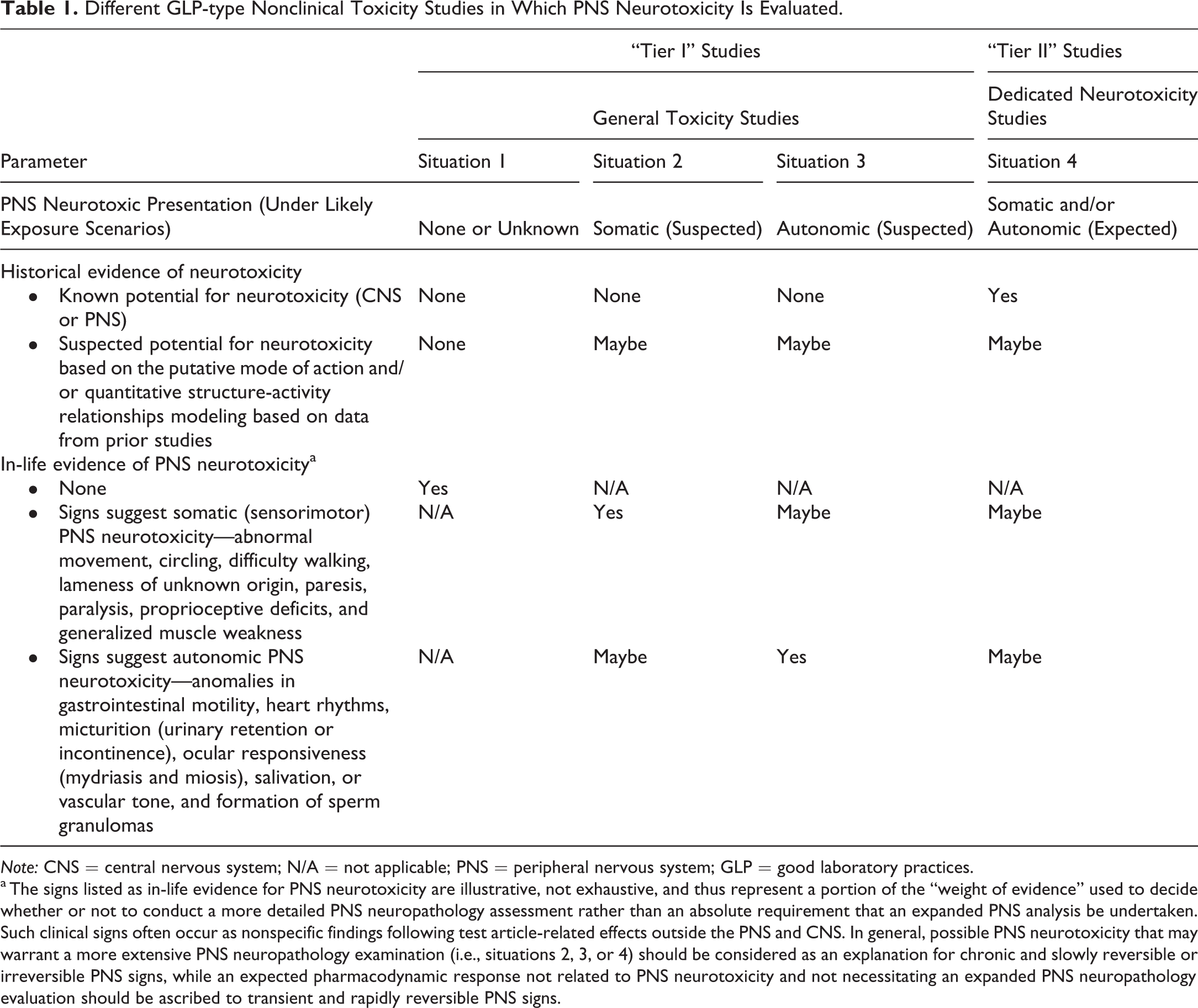
a The signs listed as in-life evidence for PNS neurotoxicity are illustrative, not exhaustive, and thus represent a portion of the “weight of evidence” used to decide whether or not to conduct a more detailed PNS neuropathology assessment rather than an absolute requirement that an expanded PNS analysis be undertaken. Such clinical signs often occur as nonspecific findings following test article-related effects outside the PNS and CNS. In general, possible PNS neurotoxicity that may warrant a more extensive PNS neuropathology examination (i.e., situations 2, 3, or 4) should be considered as an explanation for chronic and slowly reversible or irreversible PNS signs, while an expected pharmacodynamic response not related to PNS neurotoxicity and not necessitating an expanded PNS neuropathology evaluation should be ascribed to transient and rapidly reversible PNS signs.
When sampling the PNS, considerable care must be given to selecting the appropriate methodology (sampling scheme, fixatives, tissue orientation, embedding media, special stains, etc.) to ensure that tissue morphology is optimally preserved. Basic PNS sampling and processing methods were promulgated recently by a working group of the Society of Toxicologic Pathology (STP) tasked with establishing “best practice” recommendations for sampling and processing the central nervous system (CNS) for nonclinical general toxicity studies (Bolon, Garman, et al. 2013). Given the CNS focus, however, coverage of the PNS in this STP document was brief and did not specifically include recommendations encompassing different divisions of the PNS—somatic (sensorimotor) versus autonomic (parasympathetic and sympathetic)—or effectors controlled by the PNS (e.g., glands, skeletal muscle, or viscera). Accordingly, the STP established a new working group on PNS sampling, processing, and analysis to provide more specific recommendations appropriate to distinct varieties of peripheral neuropathies that might be encountered during the course of GLP-type nonclinical toxicity studies.
The working group was given a charter with multiple specific aims. The first charge was to recommend what PNS structures should be regularly sampled during GLP-type toxicity studies (Tier I and Tier II) performed in common vertebrate test species. The second charge was to suggest tissue processing procedures and trimming schemes to facilitate analysis of these regions. The third charge was to define what stains and special neurohistological procedures, if any, should be used routinely in PNS evaluations. The fourth charge was to consider when other special morphological techniques should be undertaken to provide a more complete assessment of PNS lesions. The fifth charge was to define appropriate means for assessing whether or not PNS recovery has taken place. The sixth charge was to propose what format should be used to most efficiently communicate histopathologic data for PNS tissues in reports destined for review by regulatory bodies. The recommendations given below with respect to particular neural structures to collect and evaluate or collect and retain (Table 2) as well as suggested sampling and processing procedures (Table 3), and also the means for documenting that they have been assessed, are based on the collective experiences and opinions of the working group members; 2 input from the global toxicologic pathology community 3 received during a 1-month-long public comment period in the fourth quarter of 2017 was also considered in defining the final recommendations. Where consensus among working group members and/or STP members was lacking on certain points, several options have been included below in a “Commentary” section, where the recommendations are discussed with respect to their advantages and disadvantages.
Recommended Sampling and Evaluation for Peripheral Nervous System (PNS) Tissues During Nonclinical Toxicity Studies.a
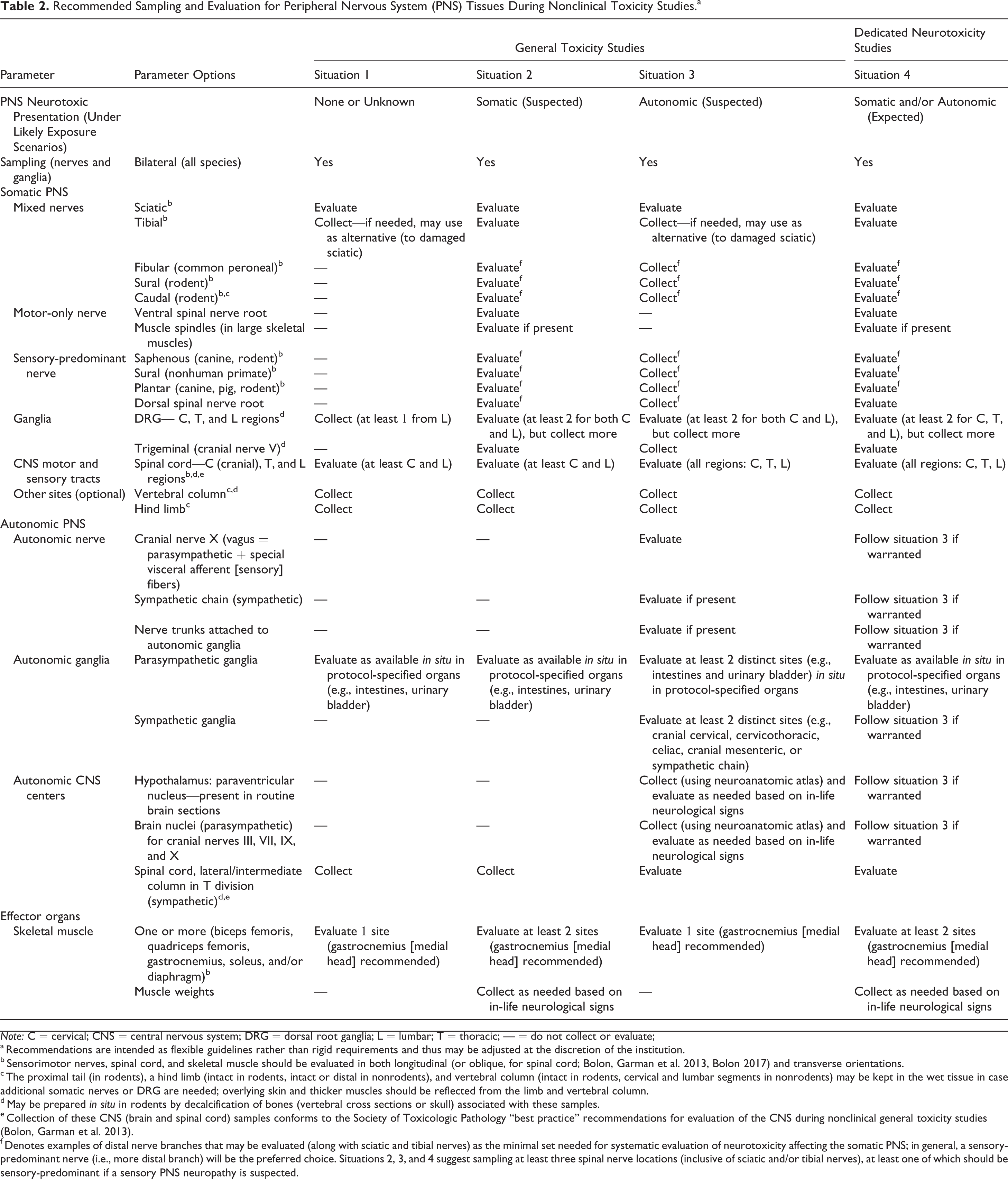
a Recommendations are intended as flexible guidelines rather than rigid requirements and thus may be adjusted at the discretion of the institution.
b Sensorimotor nerves, spinal cord, and skeletal muscle should be evaluated in both longitudinal (or oblique, for spinal cord; Bolon, Garman et al. 2013, Bolon 2017) and transverse orientations.
c The proximal tail (in rodents), a hind limb (intact in rodents, intact or distal in nonrodents), and vertebral column (intact in rodents, cervical and lumbar segments in nonrodents) may be kept in the wet tissue in case additional somatic nerves or DRG are needed; overlying skin and thicker muscles should be reflected from the limb and vertebral column.
d May be prepared
e Collection of these CNS (brain and spinal cord) samples conforms to the Society of Toxicologic Pathology “best practice” recommendations for evaluation of the CNS during nonclinical general toxicity studies (Bolon, Garman et al. 2013).
f Denotes examples of distal nerve branches that may be evaluated (along with sciatic and tibial nerves) as the minimal set needed for systematic evaluation of neurotoxicity affecting the somatic PNS; in general, a sensory-predominant nerve (i.e., more distal branch) will be the preferred choice. Situations 2, 3, and 4 suggest sampling at least three spinal nerve locations (inclusive of sciatic and/or tibial nerves), at least one of which should be sensory-predominant if a sensory PNS neuropathy is suspected.
Recommended Baseline Processing Strategies for Peripheral Nervous System (PNS) During Nonclinical Toxicity Studies.a
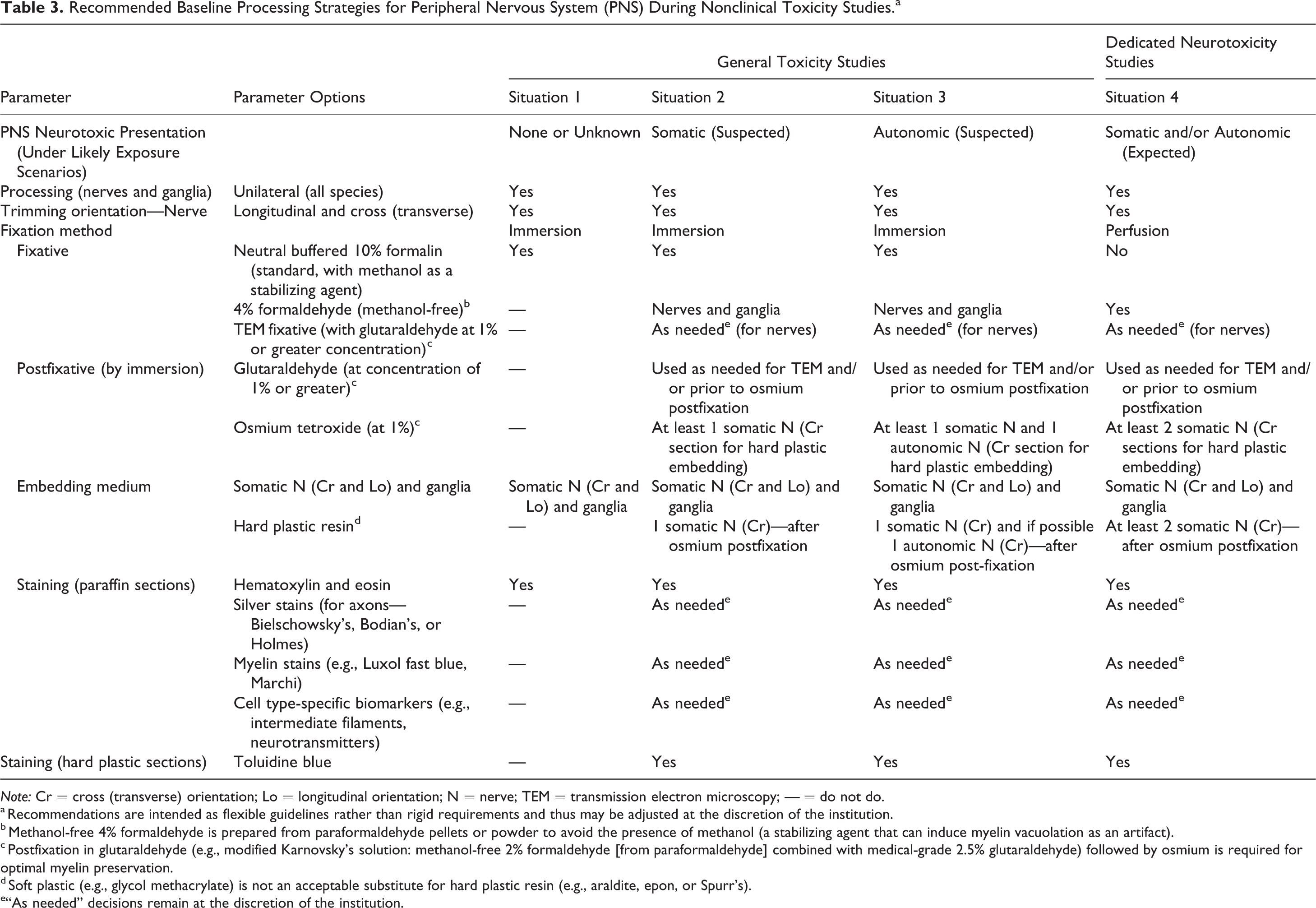
a Recommendations are intended as flexible guidelines rather than rigid requirements and thus may be adjusted at the discretion of the institution.
b Methanol-free 4% formaldehyde is prepared from paraformaldehyde pellets or powder to avoid the presence of methanol (a stabilizing agent that can induce myelin vacuolation as an artifact).
c Postfixation in glutaraldehyde (e.g., modified Karnovsky’s solution: methanol-free 2% formaldehyde [from paraformaldehyde] combined with medical-grade 2.5% glutaraldehyde) followed by osmium is required for optimal myelin preservation.
d Soft plastic (e.g., glycol methacrylate) is not an acceptable substitute for hard plastic resin (e.g., araldite, epon, or Spurr’s).
e“As needed” decisions remain at the discretion of the institution.
Situation-specific Recommendations for Sampling, Processing, and Analysis of the PNS during Toxicity Studies
Basic Philosophy
The working group concluded that a rigid “one-size-fits-all” approach to sampling, processing, and evaluating PNS tissues is inappropriate due to the variety of situations, modes of action (MOAs), molecular-initiating events, and potential target sites that might be encountered. Instead, the working group is of the unanimous opinion that the appropriate and achievable objective is to delineate a strategy for evaluating key PNS structures to differentiate common classes of neurotoxic lesions, but let the experiences and needs of individual institutions 4 drive selection of the specific battery of sampling, processing, and analytical methods undertaken to provide a suitable survey of the PNS. The methods used for implementing such institutional decisions should be articulated clearly in the study protocol and/or study report or alternatively documented in advance in an institutional standard operating procedure (SOP) or study-specific procedure. Such institutional decisions should be made using a “weight of evidence” (WOE) approach, where expanded sampling and evaluation of the PNS is considered only when evidence of PNS neurotoxicity is substantial enough to be an important factor in the final risk assessment. In general, WOE decisions incorporate such factors as the degree of PNS neurotoxicity versus toxicity to other target tissues (i.e., how sensitive is the PNS to the test article relative to other systems), the extent of PNS neurotoxicity that develops at relevant levels of exposure, and the variable sensitivities of different species, including humans, to the PNS neurotoxic effects of a given test article or compound class.
Scenarios for PNS Neurotoxicity
Four general situations during which PNS tissues may be sampled in the course of toxicity studies were considered (Table 1). For each, the recommended sampling strategy varies slightly based on the different locations in which the PNS is affected and the types of studies that are conducted. The first three situations involve general (Tier I or “screening”) toxicity studies, while the last scenario relates specifically to dedicated neurotoxicity (Tier II or “advanced”) assessments.
Situation 1 is a general toxicity study in which (1) no potential for PNS neurotoxicity was detected in data obtained during prior studies (
A side-by-side comparison of PNS specimens to collect and evaluate and baseline tissue sampling and processing recommendations for the four situations are given in Tables 2 and 3, respectively. The working group recommends that this information be used to define institutional SOPs or study-specific procedures that describe collection and processing for PNS tissues. These documents should be detailed but sufficiently flexible, so that the study director may adjust the study protocol as necessary to address the issues encountered during a particular situation.
The four situations noted above typically arise in nonclinical toxicity studies for adult animals, where PNS effects related to test article exposure occur with some frequency. No impediment exists with respect to applying these best practice recommendations to the PNS of developing animals. However, the working group notes that current practice in developmental neurotoxicity studies typically handles PNS sampling, processing, and analysis using the situation 1 recommendations for both the early (juvenile) and late (adult) time points. If other evidence suggests that a more wide-ranging examination is required to fully characterize the PNS findings, the more extensive PNS neuropathology examination typically is concentrated on the adult time point (Garman et al. 2016).
Best Practice Recommendations for All Four Situations
The PNS sampling strategy should be guided by observed in-life neurological signs or other information for the compound/target. The choice of which PNS samples to collect and whether or not special histology processing and/or investigative techniques need to be applied should be defined at the discretion of the institution for a given toxicity study using a WOE approach. For all situations, PNS structures (nerves, ganglia, and effector organs) typically should be collected bilaterally but may be processed and evaluated unilaterally. This strategy is acceptable since toxic lesions in the PNS are generally bilateral whereas background alterations secondary to nerve trauma (induced either
Where plastic embedding is undertaken, hard plastic resin is the recommended medium. Soft plastic (e.g., glycol methacrylate [GMA] or methyl methacrylate [MMA]) embedding offers no substantive improvement in resolution over paraffin embedding with respect to preserving myelin architecture, so soft plastic is not an acceptable substitute for hard plastic resin.
The recommended best practice for light microscopic evaluation is to undertake a tiered, semiquantitative analysis with foreknowledge of the study design. A subsequent masked (“blinded” or “coded”) analysis of PNS tissues with findings of concern may be conducted at the discretion of the study pathologist (or peer review pathologist) but usually is done only to aid in defining the dose response and/or establishing a “no observed adverse effect level” (NOAEL).
Best Practice Recommendations for Situation 1
For general toxicity studies with no known concern for PNS neurotoxicity (situation 1), the majority of the working group concurs that one large, mixed (i.e., sensorimotor) somatic nerve such as the sciatic nerve (or tibial nerve if the sciatic trunk has been traumatized), the autonomic ganglia within the walls of protocol-specified organs, the spinal cord (white matter tracts of the cervical and lumbar divisions), and one effector organ (e.g., the medial head of the gastrocnemius muscle) represent a suitable baseline PNS survey. At least one dorsal root ganglion (DRG
5
) from the lumbar division either isolated or (in rodents only) processed
Best Practice Recommendations for Situation 2
For general toxicity studies where somatic PNS neurotoxicity is a concern under likely exposure scenarios (situation 2), three spinal nerves are evaluated. Typically, the sciatic nerve and two or more of the following nerves (most of which are distal branches of the sciatic nerve) are sampled: tibial, fibular (i.e., common peroneal), plantar, saphenous, sural, or (in rodents) caudal nerves. The sciatic, tibial, and fibular nerves in all species and the sural and caudal nerves in rodents are mixed sensorimotor structures; the saphenous, plantar, and (in nonhuman primate) sural nerves are sensory-predominant branches (sometimes referred to as “sensory-only” nerves). Nerve selection generally should be based on in-life findings. Institutions may consider including sensory-predominant nerves to facilitate data translation among species since these distal branches can be biopsied more easily for clinical monitoring in humans. The spinal cord (at minimum the cervical and lumbar divisions), at least four DRG (two or more associated with both the cervical and lumbar intumescences, which supply nerve fibers to the brachial and lumbosacral plexuses, respectively), the associated spinal nerve roots, and one trigeminal (cranial nerve [CN] V [Gasserian]) ganglion 7 with associated nerve fibers are recommended for collection and evaluation.
Conventional processing conditions (immersion fixation in buffered formalin, paraffin embedding, and H&E staining) are suitable for PNS tissues, with three exceptions. First, methanol-free formaldehyde (MFF 8 ) or medical-grade glutaraldehyde (MGG, typically 2.5%) rather than NBF ideally should be employed to minimize processing artifacts. Generally, MFF is used for routine histologic preparations, while MGG is employed for samples in which postfixation with osmium is used to highlight myelin organization. The working group recognizes that this first adjustment may not be feasible on short notice, especially if the in-life PNS-related signs develop late in the course of a large general toxicity study. Second, if further characterization of PNS changes observed in H&E-stained sections is necessary, acquisition of serial sections for at least one mixed nerve may be considered for special neurohistological staining to highlight axonal morphology (silver stain) and explore myelin integrity (myelin stain). Third, at least one nerve cross section (usually a mixed-function distal trunk like the tibial or fibular nerve or a sensory-predominant branch like the sural or plantar nerve) should be postfixed by immersion in MGG followed by osmium, processed into hard plastic resin, and then stained with toluidine blue for light microscopic evaluation; this specialized technique better stabilizes myelin during processing than does fixation with MFF or NBF alone. The last two adjustments should be feasible regardless of whether MFF or NBF is utilized as the initial fixative at necropsy.
Best Practice Recommendations for Situation 3
For general toxicity studies where autonomic PNS neurotoxicity is a concern at relevant levels of exposure (situation 3), elements of the parasympathetic, sympathetic, and enteric 9 PNS should be evaluated, including multiple autonomic ganglia and, at the discretion of the institution, selected autonomic nerves (e.g., vagus for parasympathetic division and sympathetic chain for sympathetic division). Common postganglionic parasympathetic ganglia that may be assessed are found in the walls of protocol-specified hollow organs, most commonly the intestines (i.e., the submucosal [Meissner’s] and myenteric [Auerbach’s] ganglia) and urinary bladder, but ideally at sites related to in-life findings. Typical sympathetic sites to consider include the cranial cervical, cervicothoracic, celiac, cranial mesenteric, and/or sympathetic chain ganglia and their attached nerve roots, as well as the sinoatrial and atrioventricular nodes of the heart; in practice, working group members have found that the sympathetic structures that can be isolated most reliably by trained technicians are the cranial cervical ganglia in small animals and cervicothoracic ganglia in larger species, while the next easiest site in all species is the cranial mesenteric ganglion. In addition to autonomic PNS nerves and ganglia, somatic PNS nerves and ganglia should be collected as described in situation 2, and the sciatic nerve and at least four DRG (two each for the cervical and lumbar divisions) should be examined as a baseline somatic PNS screen. All spinal cord divisions (cervical, thoracic, and lumbar) should be collected and evaluated. Routine processing (immersion fixation in NBF or ideally MFF, paraffin embedding, and H&E staining) is suitable for most autonomic PNS samples. Postfixation with MGG and osmium followed by hard plastic embedding may be useful for small-caliber autonomic trunks despite the lower degree of myelination in most autonomic nerves.
Best Practice Recommendations for Situation 4
For dedicated neurotoxicity studies where PNS neurotoxicity is likely or certain (situation 4), expanded sampling and evaluation includes at least three spinal nerves (sciatic, tibial, and fibular, saphenous, sural, plantar, or caudal), white matter tracts of all three spinal cord divisions, multiple DRG and their associated spinal nerve roots, and a trigeminal ganglion. At least six DRG should be examined (two or more DRG for each of the cervical, thoracic, and lumbar spinal cord divisions). In general, DRG should be removed from the vertebral column rather than processed and evaluated
Adjustments to Recommended PNS Sampling, Processing, and Evaluation Practices
The working group recommendations for PNS sampling in situations 1–4 are designed to be applicable to cases where test articles have been delivered systemically (i.e., where all PNS tissues are liable to some degree of test article exposure) and thus may need to be modified for selected scenarios and/or unusual test articles. For example, even in situation 4 (where neurotoxicity is expected), less extensive PNS evaluation may be permissible if the pattern and severity of PNS lesions for the doses and/or the dosing regimen used in a study have been well defined in one or more previous studies; however, the working group recommends that all PNS tissues described in situation 4 still be collected and archived as wet tissue. Retained tissues that were fixed in MFF should be held in MFF until the PNS neuropathology is complete to avoid the potential for methanol-associated myelin artifacts, after which PNS samples may be transferred per institutional SOP to NBF for long-term archiving. Similarly, additional PNS samples (e.g., forelimb nerves) may have to be evaluated if clinical signs suggest that PNS damage has occurred at these sites. Finally, local delivery of a minimally diffusible test article 10 generally warrants increased collection and prioritized analysis of nerves near the administration site, while more distal PNS elements may be collected but retained as wet tissue without processing. Such modifications in sampling and evaluation may be made at the discretion of the institution. The rationale for such adjustments typically should be given in the study report.
Rationale for Recommended PNS Sampling, Processing, and Analysis Practices
Regulatory guidelines are fairly generic with respect to prescribing the PNS sampling strategy (Bolon et al. 2011; Salvo and Butt 2011), so common sense is an essential attribute when selecting the PNS tissues to collect and evaluate. Selection of PNS sites to sample and evaluate depends on the situation (Table 2). Reasonable flexibility is possible in the choice of PNS tissues, depending on institutional preference. A “WOE” approach should be employed in deciding whether or not expanded PNS evaluation will provide data relevant to the risk assessment. Situations in which PNS toxicity is judged to represent a modest hazard relative to more substantial test article–related findings observed in more sensitive systems and/or in which PNS toxicity occurs at exposure levels with substantial safety margins relative to predicted exposure levels may preclude the need for a substantial expansion, or permit only a modest expansion concentrated on the most sensitive target sites, in PNS sampling and examination.
Situation-specific PNS Sampling Strategies
Basic considerations
For screening in the absence of known or anticipated PNS neurotoxicity (situation 1), evaluation of one large mixed (sensorimotor) nerve is a suitable survey for PNS involvement. If PNS neurotoxicity is a concern (situations 2–4), PNS evaluation is expanded to include additional nerves and ganglia, with the choice depending on the nature of the in-life signs or other factors suggesting that one or more PNS sites might be sensitive to a test article or related molecules. Therefore, study protocols and institutional SOPs should facilitate collection of any PNS tissues that might be needed to explain the constellation of PNS-related clinical signs seen during the in-life portion of the study.
Collection of PNS samples (nerves, ganglia, and effector organs) for all four situations usually should be done bilaterally unless such an approach would impact another endpoint (e.g., collection of unfixed tissue for biochemical or molecular analysis). The rationale for this recommendation is that PNS neurotoxicity typically induces bilaterally symmetrical effects. Bilateral sampling can be done quickly by skilled technicians, and the retention of such specimens may permit additional characterization of unexpected findings or salvaging of a protocol-specified tissue where the sample that was processed initially was damaged in-life or during handling and processing; again, the choice of bilateral versus unilateral PNS collection should remain with the institution. Sample acquisition should be undertaken in a fashion that minimizes structural artifacts produced by manipulation, compression, and traction of incompletely fixed PNS tissue. The keys to curtailing artifacts are to limit handling (pressure and stretching applied to neural tissues during sampling), to promptly and carefully place tissues into properly prepared fixative and buffer solutions, and to maintain tissues at an appropriate temperature (generally room temperature [RT] for GLP-type toxicity studies) until additional processing may be undertaken.
In general, PNS samples should be individually identified. Sample identity may be assured by either placing each specimen in its own tissue cassette, applying it to a labeled index card (to which it will adhere due to the inherent stickiness of epineurial connective tissue), or stapling it (through one end, not the middle) to an acetate strip prior to fixation to maintain it in an extended (but not “stretched”) orientation (Jortner 2000). Stapling is the least desirable method due to the likelihood for “crushing” the tissue. The orientation of the proximal and distal ends of nerves can be identified by labeling one end.
Situation 1
In general toxicity studies where no neurotoxic potential is known or anticipated (situation 1), the minimal list of PNS tissues to be evaluated in all species is a readily accessible, large, spinal-origin somatic nerve, the autonomic ganglia within the walls of major viscera, the cervical and lumbar divisions of the spinal cord, and a skeletal muscle (i.e., richly innervated effector organ). This PNS sampling strategy is identical to that proposed in the STP best practices document for CNS sampling in nonclinical general toxicity studies (Bolon, Garman, et al. 2013) and reflects the current practice for general toxicity studies.
Nerves
The usual PNS sample for situation 1 is sciatic nerve. The rationale for selecting this nerve is that it contains both sensory and motor nerve fibers, which permits analysis of major peripheral sensorimotor structures in a single sample. The sciatic nerve is exposed by reflecting and/or removing the overlying skeletal muscle (Figure 1). Sciatic nerve samples commonly are acquired at a distal location (i.e., just proximal to where the tibial and fibular nerves branch, which occurs near the femorotibial joint [knee]), although collection at a more proximal site (i.e., midway between the vertebral column and knee) is a frequent alternative. The choice of sciatic nerve site to be sampled (proximal vs. distal) is left to the discretion of the institution.
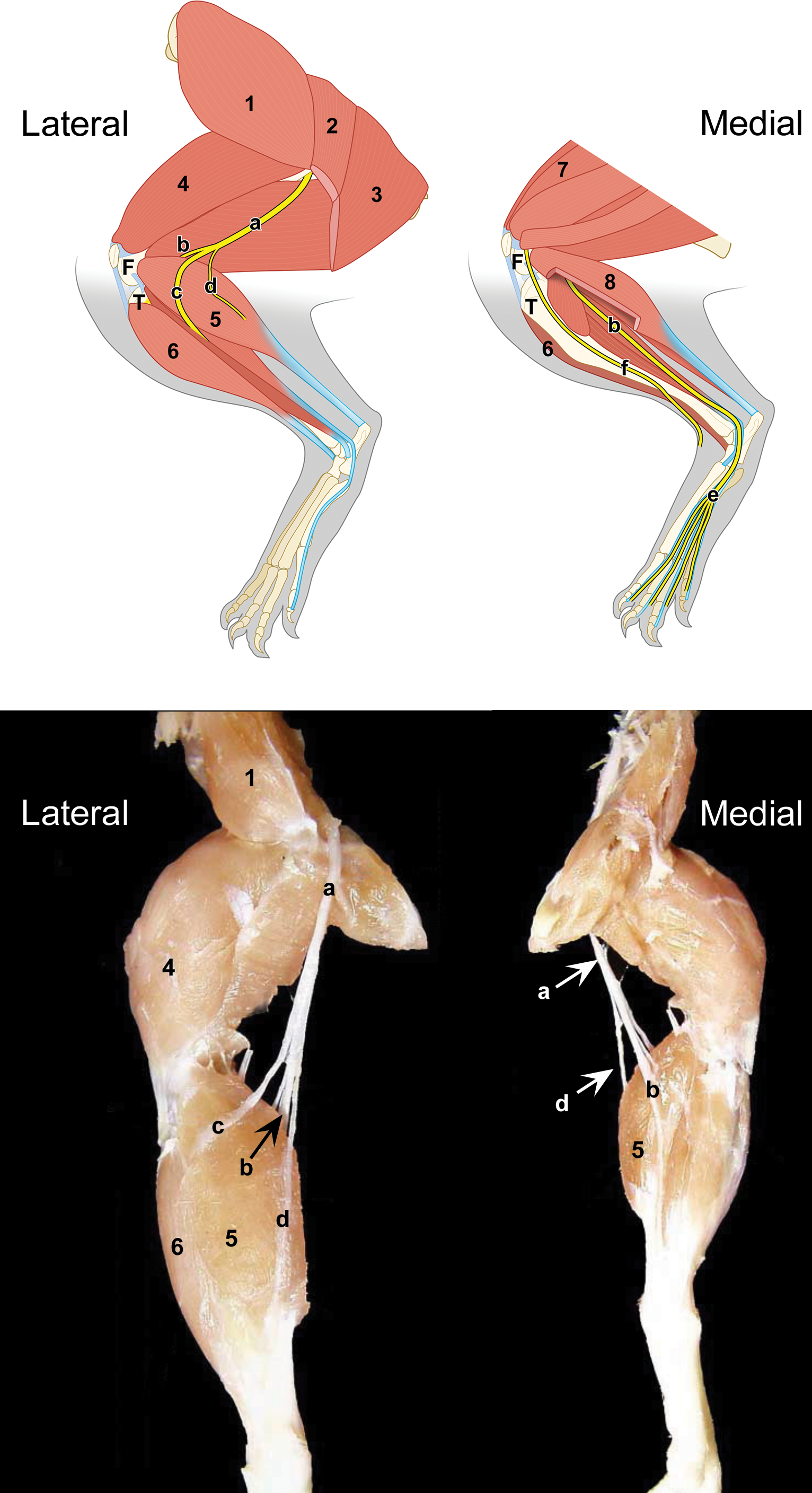
Locations for harvesting somatic nerves from the rodent hind limb. Nerves: a = sciatic; b = tibial; c = common peroneal (fibular); d = lateral sural; e = plantar, f = saphenous. Muscles: 1 = gluteus medius; 2 = biceps femoris; 3 = semitendinosus; 4 = quadriceps femoris; 5 = gastrocnemius lateralis; 6 = tibialis cranialis; 7 = rectus femoris; 8 = gastrocnemius medialis. Bones: F = femur (condyles); T = tibia. (Schematic diaphragm rendered by Mr. Tim Vojt, with confirmation of anatomic structures by Ms. Kate Anderson.)
A sciatic nerve branch, typically the tibial nerve (another trunk carrying both sensory and motor nerve fibers; Figure 1), may be evaluated instead of the sciatic nerve if likely artifactual changes might confound sciatic nerve analysis. A common scenario in which this substitution may be warranted is in nonhuman primates that have received intramuscular injections of ketamine in the region where the sciatic nerve is routinely collected. Chemical and mechanical trauma associated with such injections has been demonstrated to damage the nearby sciatic nerve trunk (Carrier and Donnelly 2014).
While sciatic nerve (or tibial nerve) commonly is the only PNS structure evaluated for situation 1, additional spinal-origin somatic nerves may be collected at necropsy at the discretion of the institution. Retaining other nerves in the archived wet tissues may prevent the need to repeat studies in the event that changes observed in the sciatic nerve necessitate evaluation of other portions of the PNS. A simple means for accomplishing this task in small species such as rodents is to retain an entire hind limb (after removing the skin) and the proximal tail (which contains the caudal nerves). In larger species, the distal nerve trunks should be removed at necropsy, although some institutions may prefer to save the distal limb (e.g., below the knee, with skin removed). Other nerves to consider for collection are listed below (under situations 2–4 and in Table 2). The choice of which additional nerves to harvest, if any, also should remain the decision of the institution.
Ganglia
A majority of working group members, with a few dissenters, recommend that DRG need not be evaluated routinely for situation 1 since no PNS neurotoxicity is known or anticipated. The working group does endorse collection and archiving of at least one lumbar DRG against the possibility that an explanation might need to be sought for lesions observed in the sciatic nerve. The rationale for this recommendation is that DRG, as well as the nerves they serve, lack effective neurovascular barriers (Olsson 1990; Abram et al. 2006; Sapunar et al. 2012) and thus may be exposed to test articles that are excluded from the CNS by the blood–brain barrier. Usually, the chosen DRG are associated with the spinal cord segments from which the sampled spinal nerve arises (i.e., the lumbar intumescence for the sciatic nerve and its branches; Table 4). A fast and simple means for retaining the DRG (and their associated spinal nerve roots) in the wet tissues is to harvest an extended portion (rodents) or region-specific segments (all species) of the vertebral column (after removing the overlying musculature and skin). The DRG may be processed and evaluated as isolated ganglia (all species) or
Expected Spinal Cord Origins of Principal Forelimb and Hind Limb Mixed Nerve Trunks for Common Vertebrate Species.
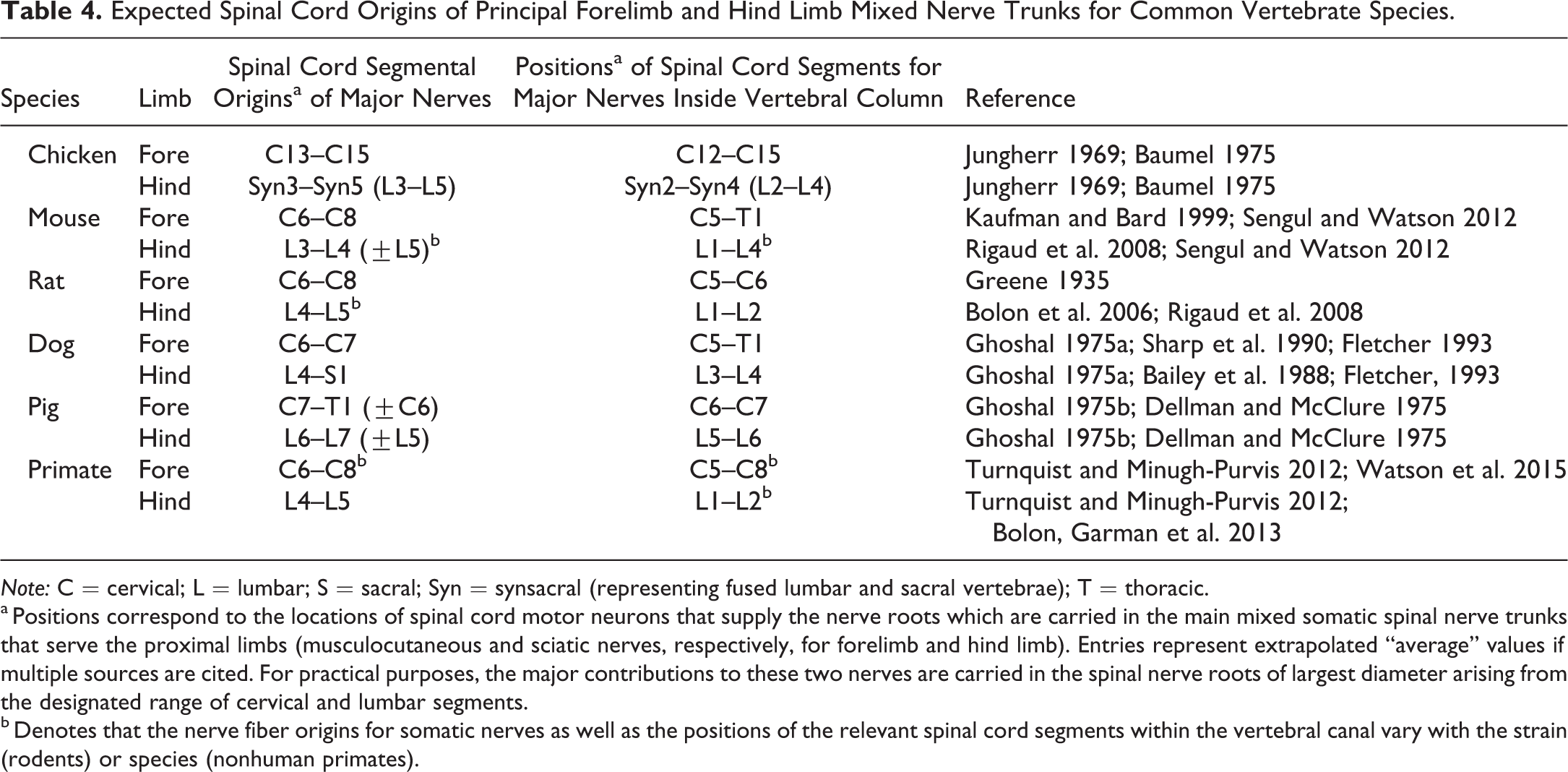
a Positions correspond to the locations of spinal cord motor neurons that supply the nerve roots which are carried in the main mixed somatic spinal nerve trunks that serve the proximal limbs (musculocutaneous and sciatic nerves, respectively, for forelimb and hind limb). Entries represent extrapolated “average” values if multiple sources are cited. For practical purposes, the major contributions to these two nerves are carried in the spinal nerve roots of largest diameter arising from the designated range of cervical and lumbar segments.
b Denotes that the nerve fiber origins for somatic nerves as well as the positions of the relevant spinal cord segments within the vertebral canal vary with the strain (rodents) or species (nonhuman primates).
Effector organs
In situation 1, skeletal muscle typically is examined as a protocol-specified tissue. Reductions in myofiber diameter may serve as indirect evidence of PNS damage due to nerve fiber (i.e., motor axon) degeneration if direct evidence of myopathic injury is not seen. Although tongue is a common choice for histologic evaluation of skeletal muscle (as a means of assessing many myofibers in several orientations in a single section), other skeletal muscle groups can be collected along with their innervating nerves. Muscles commonly selected for sampling are composed mainly of type I (“slow twitch,” fatigue-resistant) fibers (e.g., diaphragm and soleus) and/or type II (“fast twitch,” glycolytic) fibers (e.g., biceps femoris, quadriceps femoris, and gastrocnemius; Schiaffino and Reggiani 2011; Gropp 2017). Some investigators substitute biceps brachii (if the forelimb appears to be affected as a specimen comprised mainly of type II myofibers).
The working group recommends the gastrocnemius as the default effector organ sample since it has a mixed (but mainly type II fiber) composition (Armstrong and Phelps 1984), is a common site of neurogenic atrophy in both humans (Spencer and Schaumburg 1977) and animals with peripheral neuropathy, and the size of the muscle can be assessed qualitatively during life by palpation. The medial head of the gastrocnemius is larger, making it a logical choice for sampling this muscle. The biceps femoris is a suitable alternative sample as it is also a common location for detecting neurogenic atrophy. The exact choice of muscle(s) to sample and evaluate should be left to the discretion of the institution.
Situation 2
In general toxicity studies where in-life clinical signs or other data (e.g., MOA and QSAR similar to known PNS toxicants) suggest the potential for somatic (sensorimotor) PNS effects (situation 2), the number of PNS specimens subjected to light microscopic analysis should be expanded. Specific neurological evidence warranting additional sampling of the somatic PNS includes local or generalized signs of paresis, paralysis, proprioceptive defects, or muscle atrophy (Table 1). Nonspecific clinical observations related to possible somatic nervous system dysfunction (e.g., abnormal movement, circling, difficulty walking, lameness of unknown origin, and generalized skeletal muscle weakness) may also trigger collection of additional PNS samples, at the discretion of the institution. The clinical signs listed here as possible evidence for PNS neurotoxicity are illustrative, not inclusive, and generally would be considered to warrant a more detailed PNS neuropathology evaluation only if present as chronic and slowly reversible or irreversible adverse effects that clearly cannot be attributed to other systemic toxicities (anemia, dehydration, emaciation, emesis, neurotoxicity related to CNS targets, etc.).
Nerves
Multiple mixed (sensorimotor) spinal nerves are sampled bilaterally during the initial tissue analysis (Figure 1; Spencer and Schaumburg 1977). As noted in situation 1, sciatic nerve samples usually are collected at a distal location (i.e., near the femorotibial joint) due to the increased sensitivity of distal sensory nerve fibers to neurotoxic agents (Martyn and Hughes 1997; Azhary et al. 2010). More proximal sampling may be elected by some institutions for characterizing myelin changes since the proximal sciatic nerve is populated by larger Schwann cells, covering longer axonal lengths, such that the loss of individual cells is more obvious relative to depletion of the smaller Schwann cells in more distal nerves (Friede and Bischhausen 1982; Krinke 2011). The choice of sciatic nerve site to be sampled (proximal or distal, or both) is left to the discretion of the institution.
In addition to the sciatic nerve, the choice of other nerves to collect may be dictated by the spectrum of neurological signs observed in-life or may conform to a predefined battery specified in an institutional SOP. Typically, distal nerve branches are preferred for evaluation since they usually contain a high proportion of sensory axons, and clinical cases of peripheral neuropathy often present as altered sensation (Martyn and Hughes 1997; Azhary et al. 2010). Furthermore, hind limb nerves rather than forelimb nerves usually are sampled in toxicity testing because the longer nerve fibers that serve the hind limb usually are affected first during peripheral neuropathies (Krinke 2011). That said, forelimb nerve branches also should be harvested if the in-life neurological signs suggest that forelimb function has been affected. Evaluation of nerves near the administration site may be prioritized in instances where a locally delivered test article has limited systemic bioavailability.
At least three spinal-origin nerves (usually sciatic nerve and two of its branches) are recommended for evaluation, but the decision regarding which nerves to assess should be guided by the clinical signs and left to the discretion of the institution. The tibial (all species), fibular (all species), and/or sural (rodents; Peyronnard et al. 1986) nerves are common choices as they are mixed sensorimotor tributaries of the sciatic nerve. In rodents, the caudal nerve (a mixed nerve that extends the entire length of the tail) may also be considered for evaluation as electrophysiological testing (e.g., nerve conduction velocity) combined with light microscopic examination of this nerve affords an opportunity to correlate structural and functional findings related to PNS neurotoxicity (Schaumburg et al. 2010). Some working group members have found that aldehyde fixation of the proximal to middle tail (via intravascular perfusion or immersion) allows for later harvest and analysis of caudal nerve. In general, nerves are evaluated unilaterally (in which case nerves that are to be examined for a given animal typically are harvested from the same side), but bilateral evaluation may be considered at the discretion of the institution or if necessitated when iatrogenic nerve damage is likely due to in-life trauma (e.g., intramuscular injection sites).
Collection of dedicated sensory-predominant or motor-only nerves generally is not necessary for safety assessment since the approach to microscopic evaluation is similar for both mixed- and single-modality nerves. However, if observed clinical signs are indicative of a sensory neuropathy (which is the most common presentation of peripheral polyneuropathy in humans and animals), the working group recommends that at least one PNS specimen be a sensory-predominant nerve. Readily accessible sites include the plantar (usually the lateral branch in dogs [Ghoshal 1975a], but the medial branch in rodents [Sant’Anna et al. 2016] and pigs [Ghoshal 1975b]), saphenous (dogs: Braund et al. 1980, and rodents: LaMotte, Kapadia, and Shapiro 1991), and sural (primates [including humans]; Butt et al. 2014) nerves. Evaluation typically may be undertaken using H&E supplemented if necessary by special neurohistological procedures (see below). However, effective examination of unmyelinated (Group C) sensory nerve fibers may necessitate transmission electron microscopy (TEM) because these elements have diameters less than 1 µm, which is near the effective resolution of conventional bright-field microscopes. The only motor-specific nerves in all species are the ventral spinal nerve roots, which may be assessed individually or in sections that also include the sensory-only dorsal spinal nerve root and its associated DRG. For this purpose, serial DRG sections may be necessary to ensure that the desired nerve root is examined as their morphologic features are identical. The choice regarding whether or not to sample sensory-predominant and/or motor-only nerves should be left to the institution.
CNs other than CN V typically are sampled and analyzed only if in-life neurological signs suggest that their function has been compromised (reviewed in Bolon and O’Brien 2011) or if they can be seen within other protocol-specified tissues. For instance, cross sections of CN I (olfactory nerve, an unmyelinated sensory nerve) and major branches of CN V (trigeminal nerve, a mixed somatic nerve) may be readily observed
Ganglia
If evidence of a somatic peripheral neuropathy is observed, at least two DRG should be evaluated for both the cervical and lumbar divisions of the spinal cord (i.e., at least four total DRG). The DRG need not be paired (i.e., represent bilateral samples from the same spinal cord segment). The inclusion of cervical DRG is recommended as evaluation of these samples represents the sole assessment of the forelimb PNS in standard nonclinical toxicity studies. The best practice for GLP-type nonclinical studies is to remove DRG from the vertebral column (Figure 2) when the spinal cord is extracted (Bolon, Garman, et al. 2013) to preclude the induction of handling artifacts associated with vertebral decalcification needed for
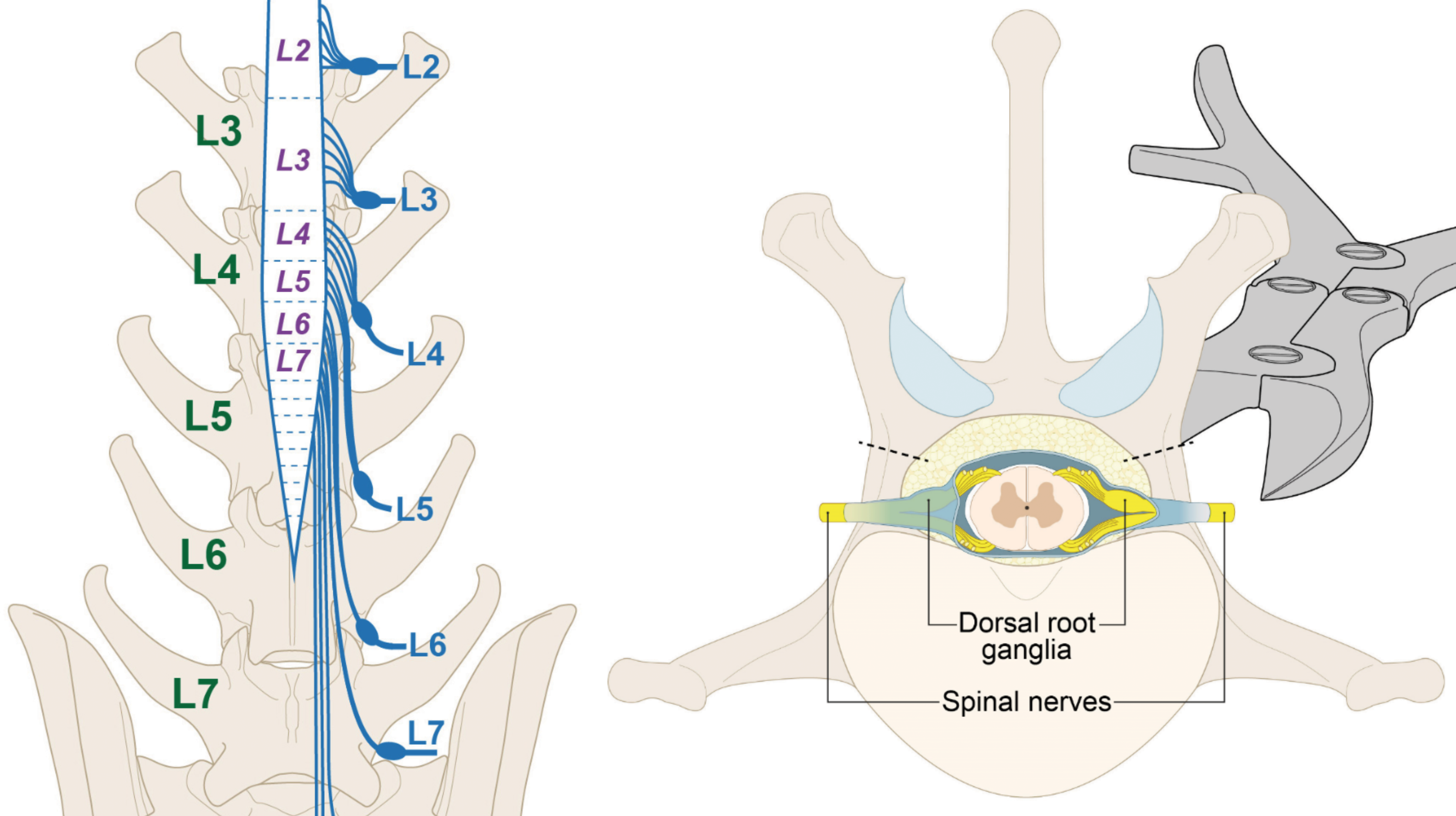
Diagrams show how to approach and localize dorsal root ganglia (DRG) associated with the origin of the sciatic nerve. Left panel: note that caudal segments of lumbar spinal cord (L4–L7) are displaced cranially relative to the DRG and vertebrae of the same number. Regional anatomy is based on the dog vertebral column. Right panel: DRG are best approached via removing the vertebral arches (at the location of the dotted lines at 2 o’clock and 10 o’clock) using bone-cutting rongeurs. (Images crafted by Mr. Tim Vojt.)
In addition to DRG, the trigeminal ganglion (i.e., the sensory ganglion of CN V) should be collected for evaluation. Ganglia of the autonomic PNS are assessed when seen
Effector organs
Skeletal muscle from sites other than the tongue should be examined from two or more distinct muscles. The specific sampling location(s) may be left to institutional preference and the parameters of the study design (e.g., muscle near sites of locally delivered test articles should also be sampled). The working group recommends that gastrocnemius serve as the default choice for one of the two muscle specimens.
Muscle weights acquired at necropsy may provide an indirect but quantitative means of discriminating peripheral neuropathic effects but should be employed at the discretion of the institution. Weights typically are acquired from isolated biceps brachii, biceps femoris, gastrocnemius, and/or quadriceps femoris, which can be easily identified and collected in a consistent fashion (Greene 1935; Vleggeert-Lankamp 2007; Magette 2012). When a muscle weight will be included as an end point, the working group recommends the gastrocnemius for weighing since peripheral neuropathies usually occur first in portions of longer axons (which in the hind limb are most distant from their supporting neuronal cell bodies; Krinke 2011). Care is required in interpreting the relevance of muscle weights as meaningful conclusions may be impossible in situations where animals were in moribund condition or impacted by other toxicities (e.g., if weights were gathered from samples taken near the site of local test article administration or from long-term recumbent animals) or where historical control data are limited or nonexistent. In such cases, muscle weight measurements have little impact on the risk assessment for PNS toxicity.
Where present within muscle sections, muscle spindles (i.e., sensory end organs) and intramuscular nerves should be assessed. The choice regarding how to record test article–related findings observed in these structures should be left to the institution.
Central (CNS) motor and sensory tracts
An adequate evaluation of somatic PNS neurotoxicity requires that the motor and sensory pathways in the spinal cord also be evaluated. At minimum, this analysis should incorporate examination of longitudinal (or oblique) and transverse sections through the cranial cervical division (particularly C1, which is near the termini of the cuneate and gracile fascicles, which are the key sensory tracts in the white matter of the dorsal funiculus) and lumbar divisions. The rationale for this recommendation is that many fiber tracts in the spinal cord white matter carry axons from PNS neurons and sensory receptors; because spinal cord has a minimal capacity to regenerate, damage to PNS-derived axons often is more apparent in the spinal cord. Spinal cord evidence indicative of PNS injury is especially prominent in the relatively large dorsal funiculus of the cranial cervical region, which harbors all the sensory axons arising directly from DRG serving the fore and hind limbs. The actual level at which samples are obtained is dictated by the test species (Bolon, Garman, et al. 2013; Watson et al. 2015) and in rodents may be influenced by the strain (Rigaud et al. 2008). The position of the lumbar intumescence within the vertebral canal varies modestly among rodent strains and nonhuman primate species. Therefore, a practical approach to correctly sampling the mouse and monkey lumbar spinal cord segments from which sciatic nerve-targeted axons arise is to harvest the spinal cord that is housed within the relevant lumbar vertebrae (Table 4).
Situation 3
In general toxicity studies where autonomic PNS neurotoxicity is suspected (situation 3), expanded sampling of autonomic PNS structures is necessary. Evidence warranting more extensive autonomic PNS sampling includes nonspecific signs of visceral dysfunction including abnormalities in gastrointestinal motility, heart rhythms, micturition (urinary retention or incontinence), ocular responsiveness (mydriasis and miosis), salivation, or vascular tone (Mathias 2003; Table 1). A WOE approach is especially important in deciding whether or not to engage in expanded sampling and analysis of the autonomic PNS. In general, isolated and/or transient signs of visceral distress (e.g., affecting one or two autonomic functions) usually reflect signs of toxicity to extraneural organs rather than to the autonomic PNS and thus would not serve as a trigger for increased autonomic PNS sampling. Instead, expanded autonomic PNS collection is recommended if a generalized autonomic dysregulation is suggested by multiple anomalous signs originating in the autonomic CNS or PNS, especially if adverse effects persist over a long period and are only slowly reversible or irreversible.
When autonomic PNS neurotoxicity is suspected, care should be taken to properly define the extent to which the histopathologic evaluation of the nervous system should be increased. Sometimes both autonomic divisions (i.e., parasympathetic and sympathetic) may be affected at once, which would warrant careful sampling to obtain representative specimens of both divisions. In addition, autonomic neuropathies may also be accompanied by somatic neuropathies, which may warrant expanded sampling of the somatic PNS (as defined for situation 2 above) as well. As noted above, the final PNS sampling strategy should be driven by the constellation of PNS-related in-life neurological signs.
Nerves
Although the number of autonomic nerves conducive for ready sampling is limited, multiple autonomic nerves may be considered for assessment during the initial analysis for situation 3. Autonomic PNS sampling may include parasympathetic (e.g., CN X [vagus]) and/or sympathetic (e.g., sympathetic chain branches) nerves (Figures 3 and 4). Somatic nerve sampling often mirrors that described above for situation 2 (Figure 1). The decision regarding which autonomic nerves (if any) and somatic nerves to examine remains with the institution.
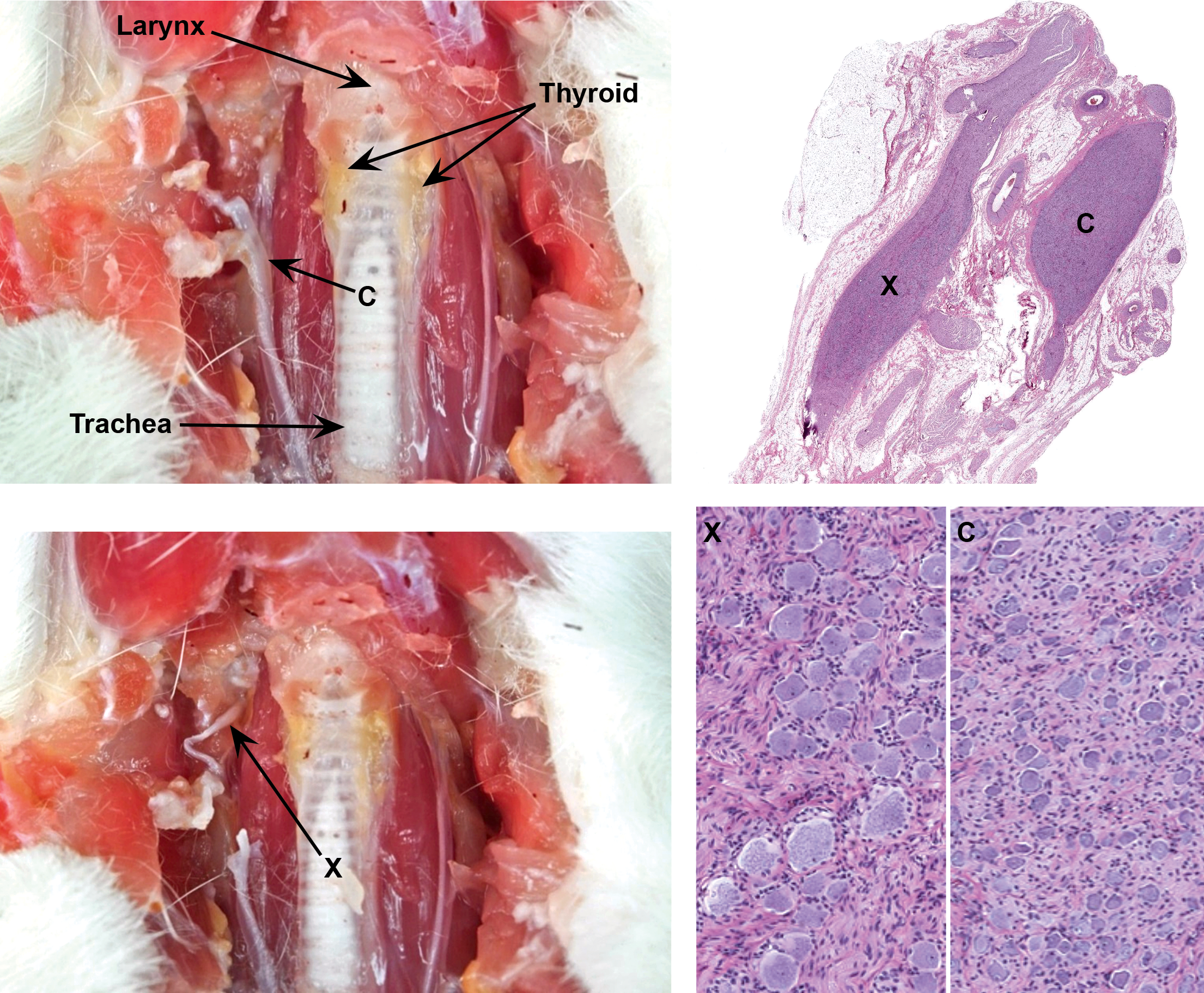
Location of key autonomic ganglia in the cervical region. Two cervical ganglia, the cranial (superior) cervical ganglion (C, a part of the sympathetic division) and the caudal vagal (nodose) ganglion (X, a visceral afferent [i.e., sensory] element), may be isolated adjacent to the trachea and thyroid glands in the vicinity of the bifurcation of the carotid artery. In histologic sections, the two ganglia can be distinguished readily because the sympathetic neurons in the cranial cervical ganglion are much smaller than the sensory neurons in the caudal vagal ganglion. Samples: left column = adult rat (provided courtesy of Dr. Magalie Boucher, by permission); right column = adult Beagle dog showing collection of both the cranial cervical ganglion (C) and caudal vagal ganglion (X) in the same histologic section and high-power depictions of these ganglia at the same magnification demonstrating their distinct features. Processing (right image): immersion fixation in neutral buffered 10% formalin, paraffin embedding, sectioning at 4 μm, hematoxylin and eosin staining.
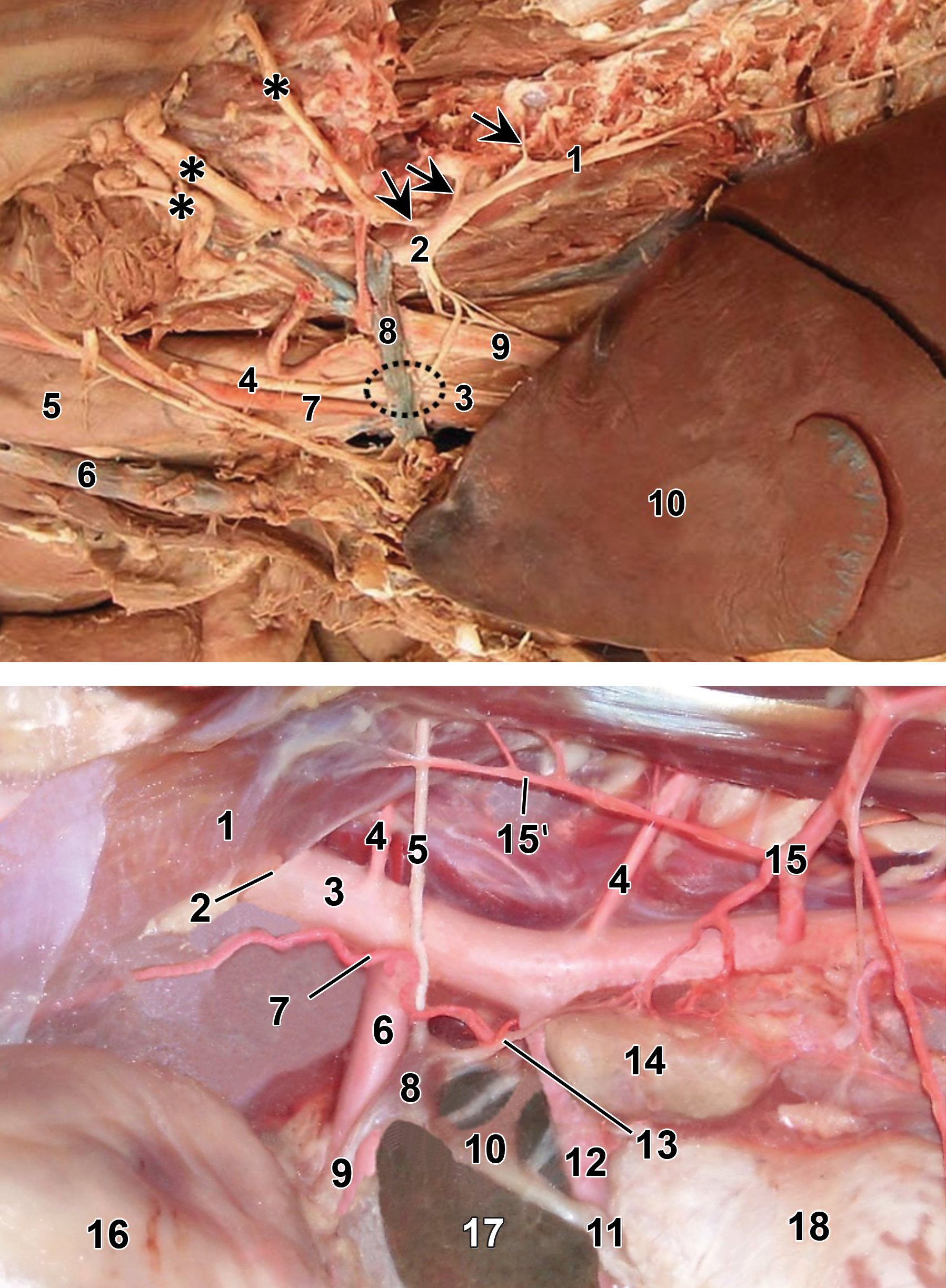
Location of key autonomic ganglia and nerves in the thoracic and abdominal regions. In the thorax (upper) of a cadaver dog, the major autonomic (sympathetic) structures are the sympathetic trunk (1), rami communicantes (arrows) extending from the spinal nerve roots, cervicothoracic ganglion (2), vagus nerve (3), and vagosympathetic trunk (4). Other labeled structures are the spinal nerve ventral branches that form the brachial plexus (asterisks), esophagus (5), external jugular vein (6), left common carotid artery (7), costocervical trunk (8), left subclavian artery (9), and left cranial lobe of the lung (10). The position of the middle cervical ganglion (another sympathetic structure) beneath the costocervical trunk is outlined by a dashed oval. (Image republished from Image 1 of the “Carnivore Dissection, Lab 12” portal, Clarkson et al. 2015, as informed by content in the “Canine Autonomic Nervous System” portal, Fletcher and Brown 2010, with permission of Dr. Thomas Fletcher.) In the abdomen (lower) of a cadaver cat, the primary autonomic (sympathetic) structures are the celiac ganglion (8) and cranial mesenteric ganglion (10). Other labeled structures include the diaphragm (1), aortic hiatus (2), aorta (3), lumbar artery (4), major splanchnic nerve (5), celiac artery (6), caudal phrenic artery (7), celiac plexus (9), cranial mesenteric plexus (11), cranial mesenteric artery (12), minor splanchnic nerve (13), adrenal gland (14), cranial abdominal artery (15) with its diaphragmatic branch (15’), stomach (16), liver (caudate lobe, 17), and kidney (18). (Image republished from https://en.wikipedia.org/wiki/Celiac_ganglia; last accessed March 1, 2018.) The working group has observed that autonomic ganglia in unfixed carcasses are paler (light tan or white) and rarely appear brown or yellow as they do here in fixed tissues.
Ganglia
Where feasible, ganglionic sampling should be based on in-life findings (i.e., visceral dysfunction). However, in most cases, intramural autonomic (parasympathetic) ganglia in protocol-specified hollow organs (e.g., intestines, urinary bladder) represent the most common autonomic ganglia available for evaluation in situation 3. If intramural ganglia are missing from initial sections, then preparation of additional tissue sections from protocol-specified viscera should be considered.
In addition, several sympathetic ganglia may be considered for analysis. The working group recommends that at least two sympathetic ganglia be evaluated if an autonomic PNS neuropathy is suspected. Frequently sampled sites include the cranial (superior) cervical ganglion (Figure 3), cervicothoracic ganglion (Figure 4), cranial (superior) mesenteric ganglion (Figure 4), and celiac ganglion (Figure 4); if present in heart sections, the sinoatrial and atrioventricular nodes may also be assessed. Working group members have found that the cranial cervical ganglia in small animals and cervicothoracic ganglia in larger species, and the cranial mesenteric ganglia in all species, are the most readily harvested sympathetic ganglia. The caudal vagal (nodose) ganglion—which is a sensory (visceral afferent) portion of CN X—is easily confused with the cranial cervical ganglion since both are located in proximity to the bifurcation of the carotid artery, although the two ganglia can be differentiated readily in tissue sections (Figure 3). Somatic sensory PNS ganglia, such as multiple DRG (cervical and lumbar) and a trigeminal (CN V) ganglion, should also be considered for sampling.
Effector organs
In most toxicity studies, the list of protocol-specified tissues will include multiple effector organs that are innervated by the autonomic PNS (e.g., glands, heart, and hollow organs with abundant smooth muscle like the digestive tract and urinary bladder).
Lesions of the autonomic PNS have been linked on occasion to structural changes in some effector organs. For example, systemic administration of ganglioplegic drugs (i.e., “ganglionic blockers,” which inhibit transmission between preganglionic and postganglionic autonomic neurons in both the parasympathetic and sympathetic systems) can induce sperm granulomas in the epididymis of rats (Bhathal, Gerkens, and Mashford 1974). However, sperm granulomas are a common incidental background finding in this species, so their presence should not be interpreted as confirmation that a test article produces autonomic dysfunction in the absence of additional evidence to support this conclusion.
Central (CNS) autonomic centers
Preganglionic neurons for autonomic nerves reside in various brain nuclei (parasympathetic role) and the lateral (intermediate) column of the thoracic ± rostral lumbar spinal cord (sympathetic role). The hypothalamus serves many significant autonomic tasks. The most important autonomic structure in this region is the paraventricular nucleus (PVN) of the hypothalamus, which contains neuroendocrine cells that innervate the median eminence and pituitary gland (Ulrich-Lai and Herman 2009). In rodent brains trimmed according to current STP best practices for CNS sampling (Bolon, Garman, et al. 2013), the PVN should be present in level 3. CNs III, VII, IX, and X carry both somatic motor and parasympathetic nerve fibers; the parasympathetic components innervate involuntary functions of multiple muscles and glands. Locations of these brainstem parasympathetic nuclei reside outside the seven levels recommended for assessment under current STP best practices for CNS sampling (Bolon, Garman, et al. 2013) and instead will need to be localized using a species-specific neuroanatomy atlas (Paxinos, Huang, and Toga 2000; Paxinos and Franklin 2001; Paxinos and Watson 2007; Palazzi 2011) if in-life signs warrant their assessment. The lateral column of the sacral spinal cord also contains preganglionic autonomic neurons. Dogma for the past century has classed these sacral neurons as parasympathetic, but recent functional and molecular data indicate that these neurons may actually regulate sympathetic functions in pelvic viscera (Espinosa-Medina et al. 2016). These CNS sites may be considered for sampling and evaluation at the discretion of the institution if the potential for an autonomic neuropathy is present.
Situation 4
In dedicated neurotoxicity studies where a CNS or PNS susceptibility is likely or known (situation 4), expanded sampling is required to more fully characterize neurotoxic hazards. Because the nervous system is the main focus of the study, more extensive sampling of the PNS (and CNS) typically is expected by regulatory agencies. This approach is applicable to both adult (Rao et al. 2011; Pardo et al. 2012; Bolon, Garman, et al. 2013) and developmental (Bolon et al. 2006; Garman et al. 2016) neurotoxicity studies in mammals and to organophosphate-induced delayed neurotoxicity in hens (Krinke et al. 1979, 1997). Sampling in situation 4 may be curtailed at the discretion of the institution if PNS lesions for a test article already have been well characterized.
Nerves
Multiple (three or more) spinal-origin nerves are sampled, as defined in situation 2 above. The precise choice of spinal-origin nerves is left to the discretion of the institution, although more distal locations and predominantly sensory nerves should be emphasized due to their early involvement in toxicant-induced peripheral neuropathies. Where nerve conduction velocity is tested (e.g., in dogs, the fibular nerve for motor fibers and the sural nerve for sensory fibers; in rats, the caudal nerve), the same nerves should be considered for microscopic examination to permit direct structure-to-function correlations; in the authors’ experience, electrophysiological testing does not induce structural changes in nerves that would confound assessment of test article–related changes. However, the working group recommends that the contralateral nerve also be collected, so that any structural changes observed in the tested nerve can be verified (at the discretion of the institution) in an untested trunk, thereby providing evidence that the anatomic findings are related to test article exposure rather than electrophysiological stimulation. Autonomic nerves typically are not collected unless in-life neurological signs suggest that lesions may exist in the autonomic PNS, in which case the autonomic nerves as defined in situation 3 may be considered for collection as well.
Ganglia
Multiple DRG (more than the four collected in situation 2) should be examined. At least two should be harvested and assessed for each spinal cord division (cervical, thoracic, and lumbar); the DRG do not need to be paired (i.e., ganglia from both sides at the same level). Some institutions collect a dozen or more, especially in studies that involve direct epidural or intrathecal delivery or in which in-life neurological signs show that the sensory PNS represents a sensitive target organ. In studies where the PNS findings seen at relevant exposure levels are likely to contribute to the risk assessment, the working group members concur that it is impossible to assess too many DRG since neurotoxic changes in these structures do not develop in a uniform manner in these organs. In rodents and other small animals, DRG may be evaluated
Sites for collecting cervical and lumbar DRG are the same ones recommended above for situation 2 (Table 4). The thoracic DRG typically are collected from the middle of that division. For DRG investigations, it is important to remember that while all DRG are located immediately adjacent to the vertebra of the same designation (i.e., DRG L5 is immediately caudal to vertebra L5), the spinal cord segment associated with a DRG frequently is present cranial to the vertebra bearing the same designation (i.e., spinal cord segment L5 is located in vertebra L1-2 in rodents; Bolon, Garman, et al. 2013; Table 4).
As with situation 2, the trigeminal ganglion (for CN V) and parasympathetic ganglia as available in other protocol-specified organs are recommended for analysis. If neurological signs suggest that autonomic dysfunction may be present, sampling of autonomic ganglia may be expanded to include some of the specimens listed for situation 3.
Effector organs
If the known potential for neurotoxicity suggests that neural lesions are localized to somatic nerves and/or ganglia, skeletal muscle should be examined for at least two distinct sites, as defined above for situation 2. Organ weights may be obtained after whole-body perfusion fixation for one or more isolated muscle bellies, at the discretion of the institution, and the isolated muscles may be employed thereafter for histopathologic analysis.
Situation-specific Fixation Options for PNS
Situation 1
For general toxicity studies in which PNS neurotoxicity is not known, suspected, or observed during life, the PNS is fixed using the same regimen applied to the nonneural tissues: immersion in NBF, commercial formulations of which contain 3.7–4% formaldehyde and approximately 1% (v/v) methanol (included as a stabilizer to extend the shelf life by slowing polymerization of formaldehyde monomers into paraformaldehyde [PFA] polymers; Kiernan 2000, 2008). Methanol is a solvent and therefore may induce morphologic artifacts in PNS, especially vacuoles and splitting of lipid-rich myelin sheaths (Garman 2011a). Nonetheless, due to cost and ready availability, NBF is still the preferred PNS fixative for general toxicity studies without a predefined need for a special assessment of the nervous system.
Immersion fixation in NBF is conducted at RT for at least 24 hr. The ratio of fixative solution to tissue should be at least 10 volumes of fluid to one volume of tissue. The quality of PNS preservation using methanol-containing NBF is acceptable provided that tissues are harvested quickly and not handled excessively (to avoid crush and stretch artifacts). If desired, MFF may be utilized for selected specimens at the discretion of the institution to preserve methanol-sensitive antigens for later IHC detection, but this practice is not done for entire studies for situation 1.
Situations 2 and 3
For general toxicity studies in which a concern for somatic (situation 2) or autonomic (situation 3) PNS neurotoxicity is projected by in-life neurological signs, PNS fixation typically is identical to that employed in situation 1: immersion in NBF (3.7% formaldehyde with 1% methanol). Where feasible (e.g., where in-life neurological signs develop early enough in the course of a study to allow bulk acquisition of specialty reagents), institutions may elect at their discretion to perform immersion fixation in MFF (e.g., methanol-free 4% formaldehyde) as the absence of methanol improves myelin integrity.
Some institutions may prefer to employ whole-body perfusion fixation if PNS neurotoxicity is suggested by in-life neurological signs (Table 3) and providing that additional study end points do not preclude this manner of fixation. Perfusion fixation may alter certain parameters commonly included in the data sets of GLP-type toxicity studies, particularly organ weights and the microscopic integrity of highly vascular organs (e.g., lung and spleen). Except for the lungs and possibly the spleen and heart, comparison of organ weights among groups should be possible for perfusion-fixed tissues from animals in the same study, if the laboratory has an established track record of successfully performing the perfusion procedure. Comparison of organ weights from perfusion-fixed animals with historical control data from immersion-fixed animals is not recommended. Technical details for whole-body perfusion fixation are given below under situation 4.
Situation 4
For dedicated neurotoxicity studies in which an impact on the nervous system (PNS or CNS or both) is likely or certain (situation 4), whole-body perfusion using MFF or another methanol-free fixative (e.g., 2.5% MGG) is recommended. Because perfusion fixation can impact the ability to assess other protocol-specified organs, collection of PNS (and CNS) samples from perfused animals is commonly done on a satellite group specifically designated for neuropathology evaluation.
For intravascular perfusion, fixative is introduced into either the left cardiac ventricle or aorta of a deeply anesthetized animal through a blunt metal needle or plastic cannula at a pressure of 120 to 150 mm Hg (approximately equal to vertebrate systolic blood pressure) by perfusion pump or a gravity drip system (Fix and Garman 2000). Species-appropriate needle sizes are 21 to 25 gauge in mice and young rats, 19 to 21 gauge in adult rats, and 14 to 18 gauge (or even greater) in nonrodents (Hancock et al. 2005; Bolon and Butt 2014). A preflush of physiological saline may be given to prevent thrombi from forming in small blood vessels as the fixative contacts blood cells and plasma proteins. Inclusion of a vasodilator (e.g., sodium nitrite, 1 mg/ml) and/or anticoagulant (e.g., sodium heparin, 1,000 IU/L of solution) in the preflush maximizes vessel patency. The choice of using a preflush (with or without anticoagulants and vasodilators) should be left to the institution’s discretion. The volumes of preflush and fixative to infuse usually are determined by the need to adequately preserve the brain and spinal cord and vary by the species. Each laboratory should develop their own protocols for intravascular perfusion especially concerning the duration, volume, and rate of perfusion. Fifty to 100 mL in adult mice, 500 to 1,000 mL in adult rats, and 3 to 5 L (or more) in larger species are suggested as starting points for the amount of fixative solution to instill; the amount of preflush typically is between 30% and 50% of these volumes. Both preflush and fixative solutions may be perfused at either RT or 4°, but RT solutions may produce fewer artifacts in neural tissues (Hancock et al. 2005; Bolon and Butt 2014).
The consensus recommendation of the working group is that MFF is a perfusion fixative of choice for preserving PNS (and CNS) tissues for routine light microscopic analysis. If TEM is also to be undertaken, inclusion of MGG is recommended as another component of the perfusate to better preserve cytoarchitectural details and reduce artifactual changes in myelin. These two aldehydes may be applied sequentially (usually using MFF to begin) or in combination. Two common mixtures are modified Karnovsky’s solution (2% MFF and 2.5% MGG) and McDowell/Trump solution (4% MFF and 1.0% MGG); in the working group’s experience, the most common choice is modified Karnovsky’s solution. Fixatives for TEM often are made in 0.1 M cacodylate or phosphate buffer (pH 7.4). Cacodylate solutions have a longer shelf life but contain arsenic and thus require extra care during use and disposal. For combination fixatives, intact ganglia or nerves are postfixed by immersion in fresh fixative at 4°C for 2 to 24 hr, after which tissue is transferred to fresh, ice-cold buffer. The reason for reduced fixation length with glutaraldehyde is that this agent renders tissues hard and brittle through its ability to more effectively cross-link molecules (Kiernan 2000). Extended storage in glutaraldehyde-containing fixatives results in excessive tissue hardening that may lead to fragmentation of the samples during sectioning.
Postfixation
For scenarios in which PNS neurotoxicity is suspected (situations 2 and 3) or likely (situation 4), or where regulatory guidelines require plastic embedding of nerve (EPA 1998a), selected nerve samples require additional fixation to adequately stabilize lipids in myelin. For this purpose, one (situations 2 and 3) or at least two (situation 4) nerves—usually spinal-origin somatic trunks rather than autonomic branches—are postfixed in MGG and then osmium tetroxide 12 (Bolon et al. 2008; Raimondo et al. 2009). Osmium must be used with glutaraldehyde to best maintain cellular structures (Penttila, Kalimo, and Trump 1974).
Isolated PNS samples (typically nerve cross sections) first are immersed in MGG for at least two hours (Dyck 2005; Bilbao and Schmidt 2015). A common composition is 2.5% MGG in 0.025 M cacodylate buffer, pH 7.4, at an osmolarity of 300 to 330 mOsm. Fixation may be done at RT or 4°C (usually overnight to 24 hr), after which fixed tissue may be stored in buffer. Postfixation in MGG is utilized for tissues fixed in NBF or MFF but is not needed for samples in which MGG was part of the perfusate. Subsequently, samples are immersed in 1% osmium tetroxide in 0.1 M phosphate buffer, pH 7.4 at RT for 1 to 4 hr, after which tissues are shifted to buffer. Osmium penetrates poorly (approximately 1 mm total; Dykstra 1992), so prior to osmication, PNS samples must be cleaned of surrounding adipose and connective tissue—without injuring the neural elements. Large samples (e.g., sciatic nerves of nonrodents) may need to be trimmed into thin slices to facilitate osmium permeation into the nerve center.
Strategies for Trimming PNS Samples
Great care should be exercised when handling nerves and ganglia (even when fixed) as even subtle manipulation may cause artifactual changes. Tissue trimming of the PNS includes one or more nerve trunks and skeletal muscle (an effector organ) in all four situations as well as DRG (including spinal nerve roots) and/or autonomic ganglia for situations 2–4. Nerves and skeletal muscle should be trimmed to permit analysis of fibers in both cross (transverse) and longitudinal orientations. Particular attention should be given to evaluating skeletal muscle in cross sections because the morphological features of myofibers affected by PNS lesions (e.g., “fiber group atrophy” from denervation) are assessed most readily in this orientation. Myofibers in the diaphragm and tongue are arranged in crisscrossing patterns that preclude most fibers from being viewed in truly longitudinal and cross orientations, thereby adding to the challenge of detecting “fiber group atrophy.”
A properly prepared nerve cross section (Figure 5) allows for an assessment of the density and numbers of myelinated axons and to a lesser extent those of unmyelinated axons (Raimondo et al. 2009). The cross section also allows for an evaluation of myelin integrity (including discrimination between demyelination and remyelination) and may reveal Schwann cell changes not readily seen in longitudinal sections. The longitudinal section provides a means for demonstrating axonal or myelin damage spanning several internodes (Figure 5) and may, due to the length of nerve examined, allow for a better assessment of associated changes such as inflammatory reactions. Longitudinal nerve samples should be approximately 1 cm long if feasible (Bolon, Garman, et al. 2013) to ensure that sufficient numbers of nerve fibers will be visible over extended lengths. Spinal nerve roots may be isolated if necessary for evaluation (after embedding in hard plastic resin) but generally are embedded along with their associated DRG, typically in longitudinal orientation (Figure 6).
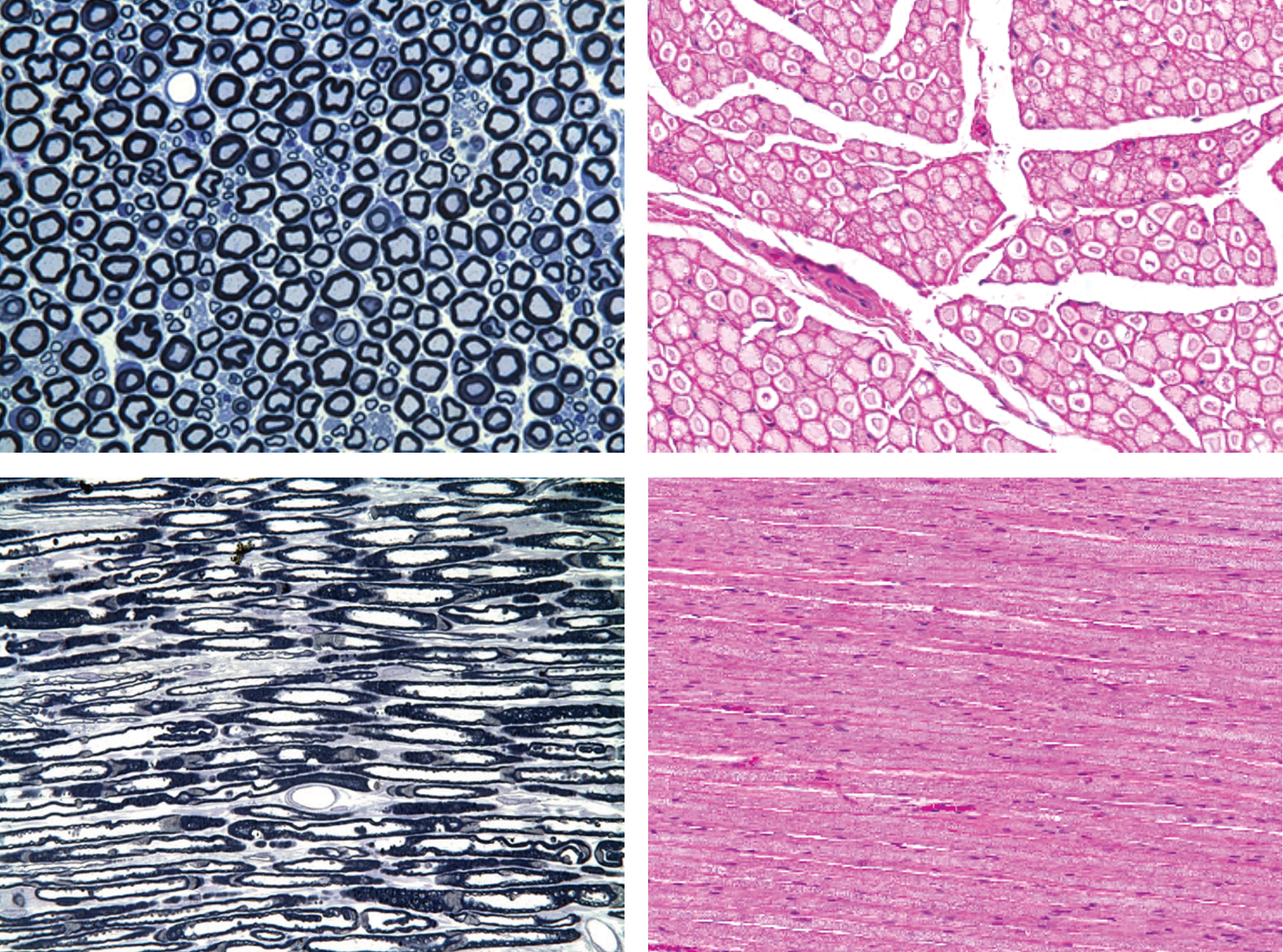
Nerves should be available for histopathologic analysis in both cross (top row) and longitudinal (bottom row) orientations. The cross (transverse) view allows comparison of the numbers and densities of myelinated nerve fibers (large-caliber, pale blue axons bounded by thick, dark myelin sheaths) and possibly unmyelinated fibers (small-caliber axons with minimal myelin [often found in small clusters]), although such fine discrimination is only possible in specially prepared nerves exhibiting high contrast between axons (pale) and myelin sheaths (dark; upper left panel) and not in routinely processed sections (upper right panel) where contrast is modest and extensive clear space exists between nerve fiber bundles as a very common processing artifact. The longitudinal plane permits axonal and myelin integrity to be assessed over extended distances. Samples: sciatic nerve from normal (i.e., control) adult rat. Processing: left column = whole-body perfusion fixation with 4% glutaraldehyde, postfixation in 1% osmium tetroxide, hard plastic resin (epon) embedding, sectioning at 1 μm, toluidine blue staining; right column = immersion fixation in neutral buffered 10% formalin, no glutaraldehyde or osmium postfixation, paraffin embedding, sectioning at 4 μm, hematoxylin and eosin staining. (Images of hard plastic-embedded nerves [left column] were provided courtesy of Dr. William Valentine, by permission.)
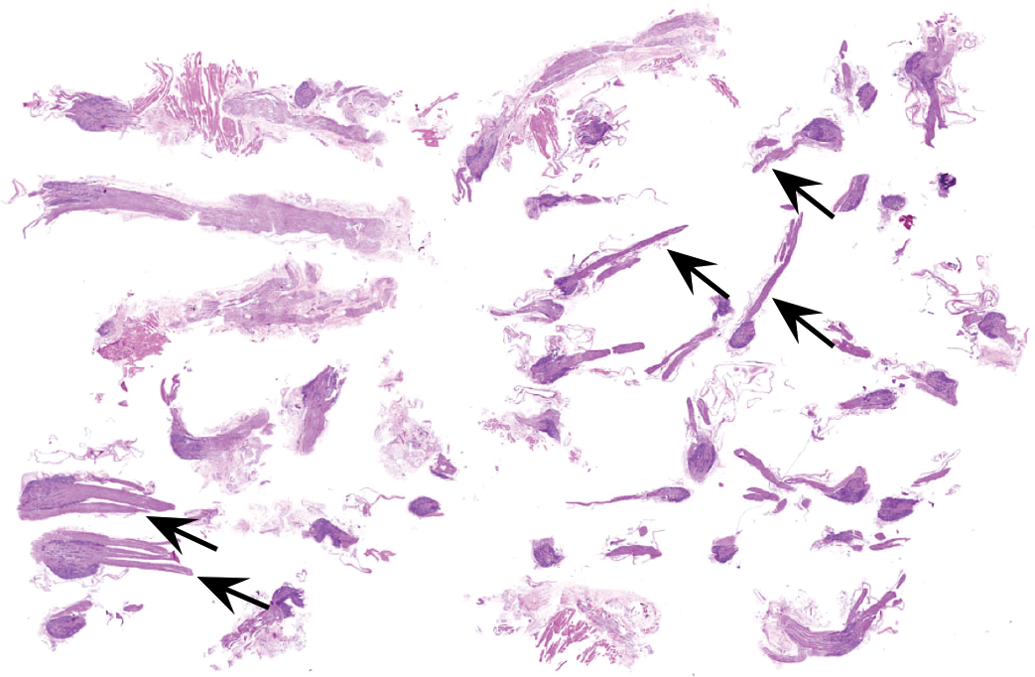
Placement of multiple ganglia in a single cassette ensures that sufficient tissue is available for histopathologic evaluation. The grouping may be ganglia from all spinal cord divisions (as shown here) or alternatively grouping as a single spinal cord division (e.g., cervical, thoracic, or lumbar). Sample: dorsal root ganglia and spinal nerve roots (arrows) from adult control rat. Processing: immersion fixation in neutral buffered 10% formalin, paraffin embedding, sectioning at 4 μm, hematoxylin and eosin staining.
For situation 3, isolated sympathetic ganglia should be processed in a fashion similar to other ganglia.
Situation-specific PNS Embedding Strategies
Embedding of PNS tissues is a critical factor in determining the data quality derived from the evaluation of PNS tissues. Paraffin allows detection of primary degenerative and infiltrative processes and therefore is a suitable embedding medium for PNS samples in general toxicity studies where PNS neurotoxicity is not a concern (situation 1). Paraffin is also used for most specimens in general toxicity studies where PNS neurotoxicity is suspected (situations 2 and 3) as well as in dedicated neurotoxicity studies where neurotoxicity (CNS and/or PNS) is likely or certain (situation 4) due to its low cost, ready availability, and suitability of such tissues for special pathology procedures (IHC, etc.). One neurotoxicity testing guideline states that “[p]lastic embedding is required for tissue samples from the peripheral nervous system” (US EPA 1998a, p. 6). The intent of this recommendation is to improve discrimination of fine cellular detail in myelinated and unmyelinated fibers. Use of plastic embedding media permits acquisition of thinner sections, thus providing improved resolution of cellular features (especially myelin sheath architecture).
Plastic embedding is expensive and labor-intensive. In studies where it is deemed that plastic embedding is warranted, the working group advises the following practical adaptation of regulatory guidance requiring plastic embedding for the PNS.
The working group recommends plastic embedding for at least one (situations 2 and 3) or two (situations 4) nerve cross sections (Figure 5), which are scenarios in which a concern exists that a test article may elicit PNS neurotoxicity. Indeed, for situation 3, nerve fibers (and especially the myelin sheaths) of autonomic nerves often are so small in diameter that plastic sections of osmicated nerves may be essential for light microscopic assessment. In such cases, PNS specimens slated for plastic embedding should have been postfixed in MGG and osmium. Cross sections of these nerve samples permit ready evaluation of the features and diameters for both axons and complete nerve fibers (i.e., axons plus myelin). Plastic embedding of longitudinal nerve sections is used less often as osmium deposition in myelin may obscure features in superimposed PNS nerve fibers due to overlap of the metal-impregnated myelin sheaths. However, plastic-embedded longitudinal nerve sections may be useful for evaluating nodes of Ranvier.
Several working group members suggest that laboratories and sponsoring institutions be encouraged to consider adjusting their PNS processing procedures for situation 1 to incorporate routine preparation of one osmicated, plastic-embedded nerve cross section as a means of attaining ideal morphologic preservation for PNS samples. However, the majority of the working group accepts that this modification may not be practical for the many general toxicity studies where no concern exists that the test article has induced PNS neurotoxicity and thus should be used in situation 1 at the discretion of the institution.
Plastic embedding for nerve samples usually employs one of two variants: “hard plastic” (hydrophobic) resins such as araldite, epon, or Spurr’s, or combinations thereof (e.g., epon-araldite); or “soft plastic” (hydrophilic) materials like GMA and MMA. Section thicknesses that are reproducibly attainable for PNS using hard plastic (<1 μm, Figure 5) and soft plastic (2 μm, Figure 7) are considerably reduced relative to that which is readily achievable for paraffin (4–6 μm, Figures 5 and 7). Soft plastics are more expensive than paraffin but are less costly and easier to process and section than are hard plastics. However, hard plastics can be used with osmicated PNS samples while soft plastics are not compatible with osmium; thus, myelin lamellae are only imperfectly conserved in soft plastic sections, which negates the original reason why plastic embedding of PNS tissues was required (EPA 1998a). The working group is of the unanimous opinion that soft plastic embedding media offer very little if any improvement in cytological resolution over paraffin embedding for nonosmicated nerve samples (Figure 7) and that soft plastic offers substantially inferior tissue preservation relative to hard plastic combined with osmication (Figure 5). Accordingly, the working group recommends hard plastic resin (of osmicated samples) as the best practice for plastic embedding of PNS and further advises that the use of soft plastic is not a suitable alternative for hard plastic embedding of the PNS. Methodological details for hard plastic embedding are found in the manufacturer’s instructions available with commercially available kits.
Soft plastic (left panel) is not a suitable medium to comply with regulatory guidelines that mandate plastic embedding of nerves, as soft plastic provides no significant improvement in resolution relative to conventional paraffin embedding (right panel). Samples: sciatic nerve from normal (i.e., control) adult rat. Processing: left column = whole-body perfusion fixation in neutral buffered 10% formalin, no osmication, soft plastic (glycol methacrylate) embedding, sectioning at 2 μm, hematoxylin and eosin (H&E) staining; right column = immersion fixation in neutral buffered 10% formalin, no osmication, paraffin embedding, sectioning at 4 μm, H&E staining.
Osmium-impregnated nerves may be embedded in paraffin (Bolon, Butt, et al. 2013) or frozen sections (Strich 1968). The preservation and visualization of myelin is enhanced in osmicated, paraffin-embedded, and frozen nerve sections in comparison to nonosmicated, paraffin-embedded, and frozen nerve sections, respectively, but remains inferior to osmicated, hard plastic resin-embedded sections. Therefore, the working group recommends that paraffin embedding of osmicated tissues be avoided as a substitute for hard plastic resin embedding.
For dedicated neurotoxicity studies (situation 4), the working group recommends that nerves and DRG should be embedded in individual blocks (with or without other tissues), so that lesions may be tracked to identifiable PNS sites. Alternatively, some institutions place many DRG from all spinal cord divisions in one cassette (Figure 6) or group DRG from specific spinal cord segments into separate cassettes. The working group recommends that sampling more DRG is preferable, even if the exact locations of each individual DRG are not tracked. Therefore, the choice of embedding scheme (individual vs. multiple samples in one block) is left to the discretion of the institution.
For both paraffin- and plastic-embedded specimens, the working group recommends that all PNS tissues from the treatment groups selected for initial evaluation (e.g., high-dose and control animals) should be processed in the same time frame to avoid any systematic variation (e.g., block effects) in such technical factors as the lengths of time spent in fixative or dehydrating solutions. If a quantitative end point has been built into the study design, tissues from all study groups for which the end point might need to be collected should be processed into blocks at the beginning of the study, even if sectioning of the blocks for intermediate dose groups will be delayed. This strategy will greatly reduce the likelihood that variations in processing will impact the quantitative data. A key means of standardizing the effects of fixation and processing across treatment groups is to include cassettes from animals in the different cohorts within each processing “run,” so that handling-related artifacts are balanced by mixing samples from all dose groups.
Situation-specific PNS Staining Strategies
Staining of PNS tissues employs H&E for paraffin sections and toluidine blue for hard plastic resin sections (Table 3). The working group recommends these two stains as a suitable initial screen for PNS specimens in toxicity studies. When preparing PNS tissues, an important consideration is that delayed processing of some treatment groups (as opposed to immediate processing of all groups into blocks) may result in altered tinctorial intensity in H&E-stained nerve sections, which might confound any quantitative or
When needed, the working group recommends stains for axons and myelin in paraffin sections as the most useful special methods for further evaluating PNS findings related to test article exposure. Silver stains (e.g., Bielschowsky’s [Figure 8], Bodian’s, or Holmes) or IHC methods to detect neurofilament proteins are helpful to highlight neurofilament-rich structures, including axons and cytoplasmic organelles in neuronal cell bodies, and for demonstrating damaged axons in axonopathy (e.g., fragmentation) or neuroaxonal dystrophy (e.g., axonal spheroids). Myelin-staining methods used in PNS tissues include Luxol fast blue (Figure 8) and Sudan black, which are especially beneficial for intact myelin, IHC to detect myelin basic protein, and the Marchi stain, which often is used to reveal demyelination (Strich 1968). The reasons for recommending these procedures are that axons and myelin are the two key components of PNS structures, and thus, many laboratories routinely perform these stains.
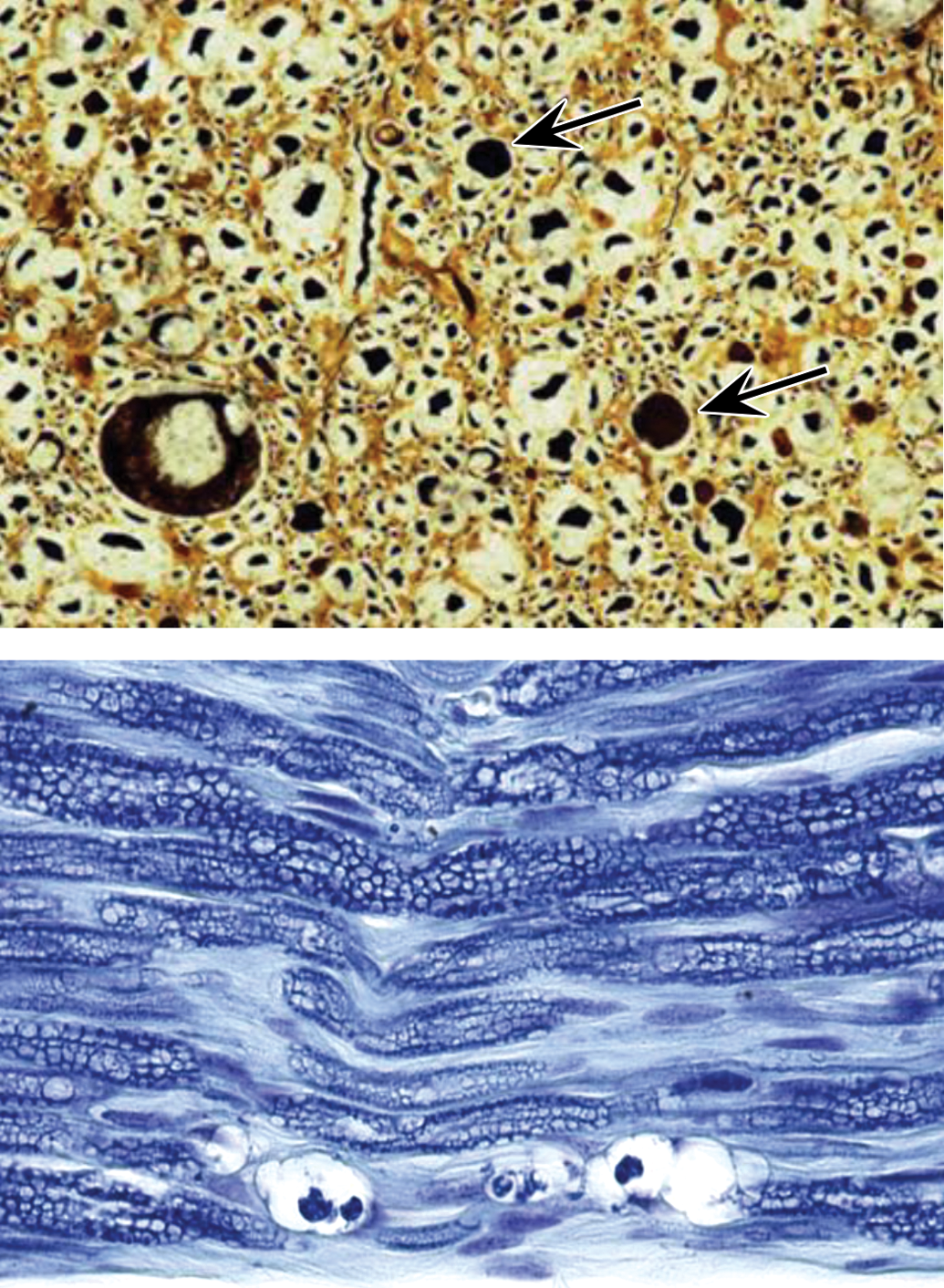
Special methods used to highlight nerve fibers include silver and myelin stains. Upper panel: Bielschowsky’s silver stain demonstrates axons and neuronal cytoplasm as dark profiles against a pale background. Arrows indicate swollen axons. Lower panel: Luxol fast blue stain reveals myelin as intact dark blue sheaths. A single degenerating axon is revealed at the bottom of the image as a series of vacuoles containing fragmented debris. The myriad tiny, clear vacuoles in the myelin sheaths represent a processing artifact. Processing: immersion fixation in neutral buffered 10% formalin, paraffin embedding, sectioning at 4 μm.
Special neurohistological methods used to showcase neurotoxic damage in the CNS typically are not utilized when evaluating PNS neurotoxicity. Routine IHC methods to demonstrate glial fibrillary acidic protein (GFAP, upregulated in reactive astrocytes and in some satellite glial cells) and ionized calcium-binding adaptor molecule 1 (Iba1, expressed by microglia and macrophages) may be used in DRG to detect satellite glial cells and activated macrophages, respectively (Patro, Nagayach, and Patro 2010; Ton et al. 2013). However, such techniques typically are deployed in the research setting rather than in toxicity testing. In ganglia, glutamine synthetase (a preferred marker for satellite glial cells; Miller, Richards, and Kriebel 2002; Schaeffer et al. 2010) and CD68 (a macrophage marker; Jimenez-Andrade et al. 2006) may be used with or in place of anti-GFAP and anti-Iba1 procedures; leukocyte biomarkers (e.g., anti-CD3 for T-lymphocytes) may be helpful in differentiating inflammation from increased satellite cell numbers. Fluoro-Jade, a fluorescent stain used to detect necrotic neuronal cell bodies in the CNS (Schmued and Hopkins 2000; Schmued et al. 2005), may be attempted in the PNS to detect degenerating neuronal cell bodies in ganglia (Marmiroli et al. 2009). In the experience of working group members, Fluoro-Jade has not been consistently helpful in discriminating dying ganglionic neuronal cell bodies in DRG and autonomic ganglia (Krinke et al. 2001). The “red dead” morphology observed in necrotic neurons in the brain (Garman 2011b; Bolon, Butt, et al. 2013) typically is not observed in ganglia. Where a marker besides H&E is required to confirm that ganglionic neuronal cell bodies are dying or dead, the working group recommends anti-caspase-3 IHC (Schmeichel, Schmelzer, and Low 2003), terminal deoxynucleotidyl transferase dUTP nick end labeling (TUNEL: Oliveira et al. 1997; Lee et al. 2017), or TEM.
While the use of soft plastic is not recommended for PNS tissues, archival samples embedded in this medium may be stained routinely with H&E. Other procedures that may be undertaken in soft plastic-embedded PNS tissues include silver stains for axons and histochemical stains (e.g., Sudan black; Cerri and Sasso-Cerri 2003) or IHC methods (Mueller et al. 2000) to reveal myelin lipids. The experience of working group members is that special techniques are applied to soft plastic-embedded PNS specimens mainly in the research setting.
Hard plastic-embedded nerve cross sections usually are osmicated during processing and then stained with toluidine blue (Figure 5). The concentration of toluidine blue used for this purpose varies among laboratories but typically is set at 1% (1:100; Hancock et al. 2005). Paraphenylenediamine may also be employed to highlight lipid-rich cell membranes in osmicated, hard plastic resin-embedded sections (Shirai et al. 2016) including myelin sheaths (Sadun, Smith, and Kenyon 1983).
Special histochemical procedures may be applied to differentiate various myofiber types in skeletal muscles (Armstrong and Phelps 1984; Kremzier 1984; Staron et al. 1999; Kammoun et al. 2014; Gropp 2017). These methods generally are not used to evaluate muscle samples in situations where PNS neurotoxicity is a concern as muscle lesions due to PNS damage (i.e., fiber group atrophy) may be seen easily by H&E.
Special Procedures for Evaluating PNS Neurotoxicity
If warranted, additional techniques may be undertaken to better characterize PNS lesions. Examples include TEM (Peters, Palay, and Webster 1991), morphometry (Diemer 1982; Kristiansen and Nyengaard 2012; Butt et al. 2014), stereology (Hyman, Gomez, and Irizarry 1998; Butt et al. 2014), teased fiber preparations (Krinke, Vidotto, and Weber 2000; Hancock et al. 2005), and quantification of intraepidermal nerve fiber ending density (IENFD; Lauria, Cornblath, et al. 2005; Lauria, Lombardi, et al. 2005; Myers and Peltier 2013; Mangus et al. 2016), motor end plates (Francis et al. 2011), and muscle spindles (Krinke et al. 1978; Muller et al. 2008). A detailed consideration of such special procedures is beyond the scope of this article. Decisions regarding whether or not to deploy these methods should be guided by data showing the neurotoxic potential of a test article to the PNS, usually the presence of in-life neurological signs or prior knowledge that the test article or a related molecule produces morphological effects in the PNS. Another important reason for quantifying IENFD, motor end plates, and/or muscle spindles may be to confirm the absence of neuropathy to provide evidence that nerve signaling is intact. The choice regarding whether or not to use these special procedures should be left to the discretion of the institution.
Strategy for PNS Neuropathology Analysis
The approach used for initially evaluating the PNS during nonclinical toxicity studies in all four situations is a tiered, semiquantitative light microscopic examination (i.e., assigning lesion grades) equivalent in concept to that for any other organ or tissue (Schafer et al. 2018). Criteria used to set histopathologic grades for test article–related unusual or adverse findings should be defined using text descriptions (supplemented at the discretion of the pathologist and institution by illustrations). The analysis should identify the existence of morphological changes in PNS tissues and characterize the lesion pattern, so that the cell populations (e.g., neurons and/or Schwann cells) and structures (e.g., cell body vs. axon vs. myelin) targeted by the test article can be determined. The evaluation should consider that the effects on PNS elements are complicated by the interdependence of axons and myelin, such that primary injury to axons or myelin will elicit secondary responses from the adjacent cells.
Appropriate Use of Masked (“Blinded”) Analysis in Assessing PNS Neurotoxicity
The initial microscopic evaluation of PNS tissues from nonclinical toxicity studies for all four situations generally should be conducted in an informed (“unblinded” or “unmasked”) fashion. In other words, the study pathologist should receive
Once a PNS finding has been identified, a masked (blinded or coded)
Assessment of PNS Recovery in Nonclinical Studies
Unlike neurotoxic lesions in the brain and spinal cord, PNS lesions often can undergo repair, but such restoration (especially of damaged myelin) may be incomplete. If the study has been designed to include recovery groups, restoration of PNS elements may be examined during the light microscopic examination. Microscopic evidence of PNS recovery differs with the primary target site (i.e., axons or myelin sheaths). Primary axonopathies exhibit extensive Schwann cell proliferation, resulting in solid columns of Schwann cells (termed “bands of Büngner”) to support axonal regrowth. These bands produce less myelin than normal until the axon once again extends through the affected region. Recovery from primary myelinopathies is associated with increased nuclear numbers (which is required because new Schwann cells typically are smaller and therefore cover less axonal area than did original Schwann cells) and gradual restoration of myelin sheaths around intact axons. Teased fiber preparations demonstrate that myelin regeneration leads to decreased Schwann cell size and thus greater numbers of nodes of Ranvier. This enhanced cellularity is the structural basis for the reduced nerve conduction velocity available to individuals who have recovered from PNS damage, as signal propagation down the axon slows measurably at each node separating the smaller Schwann cells.
While PNS lesions may regress over time, the working group has no specific data to support selection of a particular recovery period for use in nonclinical toxicity studies. Complete restoration will require an extended period of time. For example, chickens treated with an organophosphorus ester exhibit partial but not complete recovery of nerve structure at 7 weeks postexposure, but nearly complete recovery by 9 weeks postexposure (Jortner et al. 1989). In rats where a traumatic (crush) injury has been applied to the median nerve, the number of nerve fibers was restored by 24 weeks, but axon diameters, nerve fiber diameters, and myelin thicknesses remained diminished (Muratori et al. 2012). The working group posits that visible evidence of PNS restoration might be seen but is not guaranteed to be evident during standard recovery periods in typical nonclinical studies (i.e., one-third to one-half of the time spent on treatment). The rationale for this supposition is that the rate of axonal regeneration is limited to the pace of slow anterograde axonal transport (i.e., approximately 0.5–1 mm/day; Morfini et al. 2012), which equates to axon regrowth of approximately 1.5 to 3 cm during a 4-week recovery period. If attempting to gauge the level of recovery, researchers should know that the degree of PNS regeneration may be underestimated by light microscopic evaluation (Ronchi et al. 2014). With these parameters in mind, which suggest that demonstrating PNS restoration may be difficult to detect and interpret (especially in larger species with their longer axons), the working group recommends that assessment of PNS recovery should be considered on a case-by-case basis when setting the length of recovery periods for nonclinical toxicity studies.
Neuropathology Documentation
With respect to communicating PNS neuropathology data, the final report for a toxicity study should contain all the parts of a conventional pathology report (e.g., a narrative together with individual animal and summary data tables) while providing detailed descriptions of the particular neuropathology techniques (e.g., fixative solutions and methods, embedding and staining procedures) used for the study. Where necessary to achieve clarity, institutions are encouraged to include the reasons why such choices were made within the study report.
In the experience of working group members, regulatory scientists have repeatedly expressed a preference that PNS lesions in the individual animal data tables be referenced to specific anatomical sites (e.g., “sciatic nerve” or “dorsal root ganglion”) rather than more generic terms (e.g., “nerve” or “peripheral nerve” or “ganglion”) and that the key PNS structures that were sampled are explicitly stated in the narrative. When assessing autonomic ganglia, institutions retain the discretion regarding whether or not findings in nonprotocol-specified neural structures (e.g., intramural autonomic ganglia in protocol-specified organs, muscle spindles) are to be recorded as a separate tissue (i.e., “enteric ganglia”) or as a notation under the tissue in which they reside (i.e., “jejunum” or “urinary bladder”). This choice may differ for situation 1 (general toxicity study with no in-life PNS signs) versus situations 2, 3, or 4 (studies for which PNS neurotoxicity is a concern). The working group concurs that the use of specific terms for protocol-specified PNS sites is the best practice for reporting PNS lesions. In addition, the working group recommends that apparent test article–related findings in any PNS elements (e.g., CN trunks, intramural autonomic ganglia) within protocol-specified nonneural tissues be entered as specific tissues (after protocol amendment) in histopathology data tables to provide documented proof that these structures were examined in systematic fashion. If full methodological details are not included specifically in the final report, they should be made available in an institutional reference document (e.g., SOP) detailing the PNS sampling and trimming scheme. The working group acknowledges that institutions retain flexibility in choosing for themselves whether or not to enact any, some, or none of these recommended best practices for reporting PNS neuropathology data in nonclinical studies.
Discussion
The working group unanimously holds that these best practice recommendations for PNS sampling, processing, and evaluation are detailed enough to provide for a systematic analysis of the PNS in GLP-type nonclinical toxicity studies for four distinct neurotoxicity scenarios and yet still sufficiently flexible to allow their implementation via relatively modest revisions of existing institutional practices. These same practices are also valid for sampling, processing, and evaluating the PNS during non-GLP investigative (i.e., pilot or exploratory) studies and likely would have utility when examining newer
The experiences of working group members suggest that PNS sampling at many institutions already approaches or conforms to the recommendations set forth here, especially for situations 1, 2, and 4—with the likely exception of the preferred plastic embedding medium (as discussed below). Therefore, adoption of these best practice recommendations for PNS structural assessment, as guided by functional evidence of possible PNS damage, should not represent a major departure from current practice for these three scenarios. The sampling recommendations where autonomic PNS neurotoxicity is a concern (situation 3) likely will require adjustments to existing institutional practices; given the extensive autonomic control of many physiologic processes, a discussion of what spectrum and chronicity of clinical signs might suggest a general effect on the autonomic nervous system warranting increased autonomic PNS sampling will also be in order. Common sense will need to be utilized during implementation of these recommendations as certain common clinical observations (e.g., emesis in dogs and nonhuman primates) occurring in isolation seldom will indicate the existence of autonomic PNS neurotoxicity and should not automatically be investigated as such. In short, the decision regarding which PNS tissues to sample and evaluate, and whether they may be assessed in an ongoing study versus designed into a future study, should be made at the discretion of the individual institution using a WOE approach where expanded PNS sampling and evaluation is done only in situations where the PNS is deemed to represent a target system that is likely to be an important factor in the risk assessment.
The principal adjustment that may be needed at many institutions to conform to the working group’s best practice recommendations likely will be to modify the plastic embedding protocol for PNS tissues. Current practice where PNS neurotoxicity is a concern (i.e., situations 2–4) often employs soft plastic media (e.g., GMA or MMA) for routinely fixed (i.e., NBF only) tissue. However, the working group unanimously agrees that optimal PNS preservation (especially of myelin) and evaluation requires initial fixation in MFF, postfixation in MGG followed by osmium, and embedding in hard plastic resin. The working group recognizes that many test facilities and contract histology laboratories may not be equipped at present with the specialized microtomy and hazardous waste reclamation equipment and procedures required to prepare hard plastic blocks and sections. Nonetheless, the working group unanimously judges that the data quality obtained using hard plastic-embedded cross sections of osmicated nerves offers the most effective means for meeting the intent of regulatory guidelines that require plastic embedding (EPA 1998a). Indeed, the working group consensus is that time spent evaluating one optimally processed nerve sample—a cross section postfixed in both MGG and osmium tetroxide, embedded in hard plastic resin, and cut at ≤1 µm—will be of greater value in understanding the nature and mechanism of toxicant-induced lesions in the PNS than will be the examination of multiple sections made using routine methods (i.e., NBF-fixed, nonosmicated, paraffin-embedded, 5 µm thick) or currently accepted specialty techniques (i.e., NBF-fixed, nonosmicated, soft plastic-embedded, 2 µm thick). A majority of the working group agrees that hard plastic embedding of nerve for general toxicity studies where PNS neurotoxicity is not expected (situation 1) is not feasible or necessary as a routine practice.
Commentary on the PNS Sampling and Processing Recommendations
The working group’s draft best practice recommendations included in this article were reviewed critically by numerous scientists from many entities around the globe including academic, consulting, contract research, industrial, and regulatory institutions. Respondents were primarily toxicologic pathologists, but multiple remarks were also received from toxicologists and regulatory scientists. Most reviewers articulated their admiration for the extensive nature of the recommendations, the impartial treatment of the viewpoints for and against various PNS sampling and processing strategies, the inclusion of literature sources addressing the various perspectives, and the flexibility afforded to institutions in selecting among different approaches for assessing the PNS in nonclinical toxicity studies. Nonetheless, two primary (and related) points of contention regarding the working group’s recommendations were raised by multiple reviewers. These two points as well as a third issue raised by a few pathologists are worthy of further explication to clarify the working group’s rationale for this proposition.
The foremost criticism was that the proposed best practice recommendations call for too much routine PNS sampling. Three main arguments were extended to support this opinion. The first was that revision of a study plan to accommodate additional PNS harvesting based on the development of in-life clinical signs near the terminal necropsy date is difficult if not impossible, especially for assays of short duration that have been outsourced to a contract research organization. The second reason is that many scientists and institutions question whether or not the cost in labor and time to collect more PNS samples adds sufficient value, given that such specimens typically are sent to the wet tissue archive and ultimately discarded without analysis. The third claim against additional PNS sampling is that this approach will extend the length of necropsies. In particular, multiple respondents questioned whether technicians should be expected to reliably dissect nonroutine autonomic PNS tissues (i.e., situations 3 ± 4) at relatively rapid speeds. The working group maintains that routine sampling and retention of more PNS sites (e.g., additional nerves besides the sciatic trunk, a few DRG associated with the origins of major nerve trunks, and nonroutine autonomic ganglia [where supported by in-life evidence of a potential autonomic peripheral neuropathy]) is a cost-effective strategy that should be considered for inclusion as a standard option in institutional SOPs or study-specific procedures. This approach is particularly important for settings in which PNS neurotoxicity is suspected (situations 2 and 3) or likely (situation 4) but may add value even in scenarios where no PNS neurotoxicity is known or anticipated (i.e., situation 1). The main support for this best practice recommendation is that additional sampling offers the opportunity to fully characterize a PNS finding in a single study, which may obviate the need for a follow-up dedicated neurotoxicity study unless a specialized PNS sampling and processing procedure is required for a particular endpoint (e.g., morphometric analysis of IENFD). The best practice recommendations for PNS sampling given in this article have been crafted to maximize the extent of PNS sampling while minimizing the additional time required for a properly trained and experienced prosector to complete a necropsy, which should limit any potential negative impact on tissue quality for other tissues and systems (i.e., an important focus for general toxicity studies that comprise situations 1–3). Nonetheless, the working group acknowledges that the institution remains the final arbiter of how many PNS tissues may need to be collected, processed, and evaluated for a given study when seeking to balance the benefits and risks of various analytical strategies in executing their product development program for a particular test article or compound class.
A second, corollary contention raised by multiple respondents was that bilateral sampling is not warranted, given that the proposed best practice recommendations support unilateral processing and evaluation as an acceptable approach to PNS analysis. The main rationale for this belief was that institutions do not perceive that evaluation of the contralateral PNS site will offer meaningful information for interpreting a subtle PNS finding. The working group notes that the desirability of bilateral sampling stems from the ability to discriminate potential iatrogenic lesions (e.g., sciatic nerve damage in nonhuman primates associated with chemical restraint injections into the muscle that covers the nerve trunk) from genuine PNS neurotoxic lesions (which typically exhibit bilateral symmetry). The working group does acknowledge that institutions retain the flexibility to address such issues in follow-up studies designed to specifically answer questions regarding PNS (and if necessary CNS) effects rather than by bilateral sampling during earlier studies. However, in the working group’s experience, follow-up studies to characterize possible PNS neurotoxic effects may need to include satellite groups with dosing at the exposure level for which the PNS finding was originally seen as PNS lesions may shift in character when a test article is administered for longer duration at a lower exposure level.
A minor source of dissension raised by a few respondents questioned the need to assess cervical DRG as a component of the PNS assessment for situations 2–4. The working group concedes that institutions have the flexibility to decide which DRG to evaluate as long as multiple DRG are evaluated, and at least some come from the lumbar division from which the sciatic nerve (i.e., the main nerve sample in nonclinical toxicity studies) originates. The working group wishes to reiterate its recommendation that at least some cervical DRG be evaluated since this sample typically provides the sole window for viewing potential neurotoxic effects in the forelimb PNS that may be processed in standard nonclinical studies.
Concluding Remarks
Current approaches to investigating PNS neurotoxicity during GLP-type toxicity studies and non-GLP investigative studies vary to some degree across institutions and appear to be distinguished more by application of a few time-tried methods rather than a reasoned exploration of the PNS as a potential target site for toxicity. The procedures for PNS collection, processing, analytical, and reporting practices should depend on the aims of the study and thus to a fair degree on institutional preference. However, a substantial improvement in the risk assessment for PNS neurotoxicity may be gained by improving the consistency of PNS sampling, processing, and evaluation. The STP believes that adoption of these best practice recommendations will provide a systematic yet malleable strategy for increasing the consistency, and thus the quality, of PNS sampling, processing, and analysis among institutions and across geographic regions over time.
Continuing advances in diverse fields like computational biology and noninvasive imaging (structural and functional) are transforming the modern practice of toxicologic neuropathology and human risk assessment. The STP believes that these best practice recommendations for PNS collection, processing, and evaluation may serve as a logical morphological “gold standard” against which emerging technologies and experimental neurotoxicity models may be measured.
Footnotes
Abbreviations
Ethical Practices
All procedures used to prepare macroscopic and microscopic images of animal specimens for this article were performed in accordance with regulations and established guidelines for humane treatment of research animals and were reviewed and approved in advance by an Institutional Animal Care and Use Committee.
Authors’ Disclaimer
This article has been reviewed in accordance with policies of the US Environmental Protection Agency (US EPA) and US Food and Drug Administration (US FDA) and approved for publication. The opinions expressed in this article solely represent those of the authors and should not be construed as representing the official views or policies of the US EPA or US FDA, nor should mention of trade names or commercial products constitute US EPA or US FDA endorsement or recommendation for use.
Acknowledgments
The authors would like to thank Dr. Magalie Boucher (Pfizer, Cambridge, MA), Dr. Thomas J. Fletcher (University of Minnesota, St. Paul, MN), and Dr. William Valentine (formerly of Vanderbilt University, Nashville, TN) for the generous provision of images used in this article, and would also like to thank Ms. Kate Anderson (Zalk Veterinary Medical Library, University of Missouri, Columbia, MO) for assistance in anatomical fact-checking for Figure 1, Mr. Tim Vojt (Columbus, OH) for superb craftsmanship of the schematic diagrams in Figures 1 and 2, and Ms. Beth Mahler for optimizing the images for publication.
Author Contribution
Authors contributed to conception or design (BB, GK, MB, DR, IP, BJ, RG, KJ, LA, JM, AS, MT); drafting the manuscript (BB); and critically revising the manuscript (BB, GK, MB, DR, IP, BJ, RG, KJ, LA, JM, AS, MT). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
