Abstract
Peripheral nervous system (PNS) toxicity is a frequent adverse effect encountered in patients treated with certain therapeutics (e.g., antiretroviral drugs, cancer chemotherapeutics), in occupational workers exposed to industrial chemicals (e.g., solvents), or during accidental exposures to household chemicals and/or environmental agents (e.g., pesticides). However, the literature and expertise needed for the effective design, conduct, analysis, and reporting of safety studies to identify and define PNS toxicity are hard to find. This half-day course familiarized participants with basic PNS biology; causes and mechanisms of PNS pathology; classic methods and current best practice recommendations for PNS sampling, preparation, and evaluation; and examples of commonly observed lesions and artifacts. Three concluding case presentations synthesized information from the prior technical lectures by presenting real-world examples of lesions caused by drugs and chemicals to demonstrate how PNS toxicity may be addressed in evaluating product safety during nonclinical studies. Topics emphasized comparative and correlative data among animal species used in toxicity studies and clinical evaluation in humans in order to facilitate the translation of animal data into human risk assessment with respect to PNS toxicologic pathology.
Keywords
The peripheral nervous system (PNS) is a uniquely positioned network by which the central nervous system (CNS) communicates with all other organ systems throughout the body. Nerves of the PNS not only carry sensory information regarding internal and external environmental conditions but also motor signals to control the activities of cells, visceral organs, and musculoskeletal system. The essential nature of PNS functions requires that relevant elements of this system be assessed in suitable fashion when seeking to evaluate the adverse effect(s) of potential chemicals or during the development of new therapeutic agents (e.g., biologics, drugs) and chemicals (Bolon et al. 2018). However, information regarding practical means for conducting well-designed, comprehensive PNS toxicity studies and risk assessments is often hard to find in the scientific literature.
To address this lack, a half-day continuing education course on PNS neurotoxicity was conducted at the 2018 annual symposium of the Society of Toxicologic Pathology (STP). This article describes key points made by each of the eight speakers and is included here as a well-referenced basic introduction to examining the PNS during toxicity studies needed to evaluate new molecular entities.
The first presentation by Dr. Brad Bolon provided a brief review of PNS biology. He noted that nerves in both the somatic and autonomic divisions of the PNS are bundled nerve fibers where each fiber consists of an axon (efferent neuron process) insulated by variable quantities of myelin and encompassed in envelopes of fibrous connective tissue (Figure 1). The term “nerve fiber” is the generic designation for an axon and its myelin sheath. Additional cells observed in nerves include Schwann cells (the source of PNS myelin), endothelial cells, fibroblasts, and occasionally macrophages and mast cells.
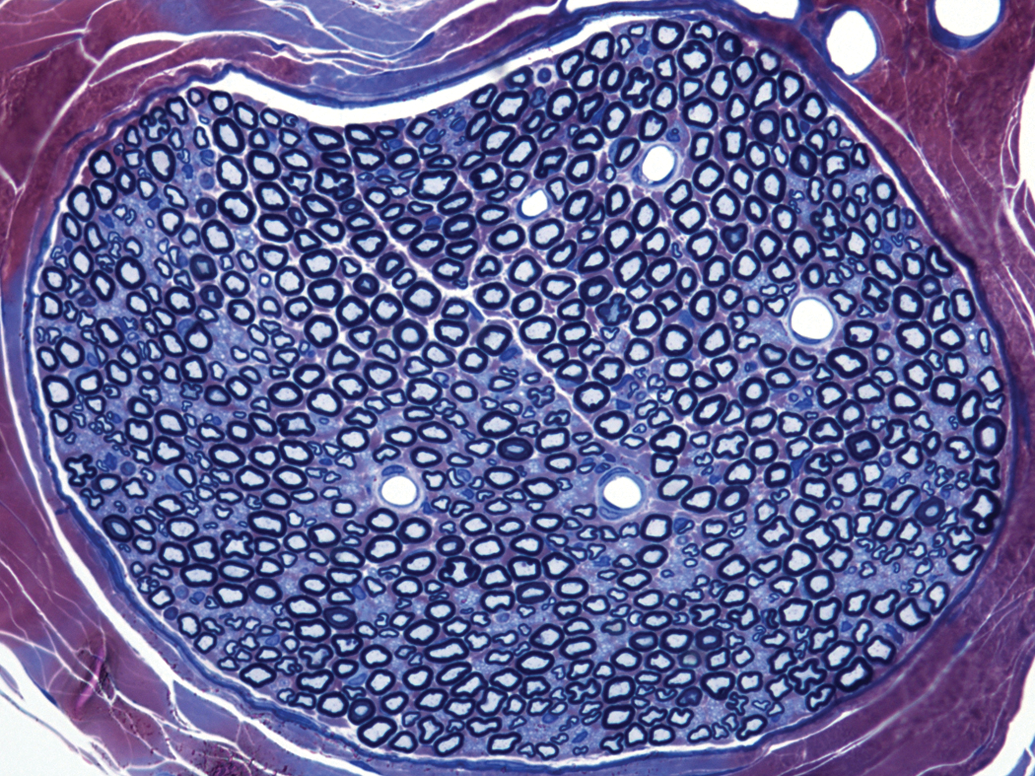
Nerve cross-sectional anatomy showing intermingled large-diameter nerve fibers (comprised of big, pale blue, usually elliptical axons surrounded by thick, dark myelin sheaths) and small-diameter nerve fibers (evident as clusters of small, pale blue, often irregular axons with very thin, dark myelin sheaths). The white spaces represent dilated capillaries, which sometimes include one or two crescent-shaped endothelial cell nuclei along their rims. The excellent resolution results from the special preparation methods used to highlight fine architectural details in nerves: perfusion fixation using 5% glutaraldehyde followed by immersion postfixation in 1% osmium tetroxide for 2 hr, hard plastic resin (epon) embedding, semithin sectioning, and staining with toluidine blue and safranin. Sciatic nerve, control rat. Figure reproduced from Jortner, B.S. (2011). Preparation and analysis of the peripheral nervous system. Toxicol. Pathol
Nerves contain both myelinated and unmyelinated nerve fibers. Myelinated fibers range in diameter from 2 to 20 μm. Organelles found in the axon cytoplasm (“axoplasm”) include mitochondria, smooth endoplasmic reticulum, and cytoskeletal elements (microtubules and neurofilaments); these organelles are dispersed evenly in normal axons but often form tangled clusters in toxicant-induced peripheral neuropathies. Unmyelinated axons generally are smaller in caliber (0.2–3.0 μm in diameter) than their myelinated counterparts and tend to form small bundles within nerve trunks (Figure 1). Thickly myelinated nerve fibers conduct impulses more rapidly and thus typically are dedicated to carrying high-speed motor and sensory signals in somatic nerves. Thinly myelinated nerves transmit slower impulses related to maintaining homeostatic equilibrium and thus are quite common in autonomic nerves.
Myelin in the PNS is formed by Schwann cells, which are of two classes: myelinating and nonmyelinating. Each myelinating Schwann cell insulates one axon and spans a unit termed an internode (i.e., the length between two nodes of Ranvier, which are the nonmyelinated gaps between adjacent Schwann cells). The internodal length and thickness of the myelin sheath are proportional to the axon diameter, which in health remains relatively constant for a given nerve fiber. The bulk of Schwann cell cytoplasm is located near the nucleus (which tends to sit at the center of the internode) or in myelin (Schmidt–Lanterman) incisures (where cytoplasm squeezed out from between the myelin layers forms small lakes at regular intervals). Each nonmyelinating Schwann cell ensheathes multiple axons. Each Schwann cell and the axon it supports are surrounded by a basement membrane.
Nerve fibers within the PNS may arise in either the spinal cord or PNS ganglia. Dorsal root ganglia (DRGs) of the somatic PNS are located along the vertebral column and harbor sensory neurons with large nuclei and prominent nucleoli. The widely dispersed parasympathetic and sympathetic ganglia of the autonomic PNS are located throughout the body, with sympathetic ganglia typically positioned at a distance from innervated organs and parasympathetic ganglia located within the walls of innervated organs. For studies in which this is warranted, PNS evaluation should include not only assessment of nerves but also analysis of selected ganglia and the spinal cord (which carries centrally projecting axons from PNS neurons).
Dr. Mark Butt gave the second presentation that addressed sampling, handling, and fixation of tissues from all parts of the PNS. Selection of the correct approaches and procedures is critical in order to obtain appropriate samples and minimize processing-related artifacts. Recommended (“best”) practices for effective collection and processing of PNS specimens have been published recently by the STP (Bolon et al. 2018). Fixatives for PNS generally consist of 4% formaldehyde, either as methanol-containing commercial formulations of neutral buffered 10% formalin or methanol-free concoctions made from paraformaldehyde powder (to avoid methanol-related myelin distortion), medical-grade (monomer/low polymer) glutaraldehyde (usually at 2–4%), or a mixture of formaldehyde and glutaraldehyde (aka Karnovsky’s solution). Osmium tetroxide postfixation (typically at 1%) is useful for stabilizing lipid structures, including myelin (Bolon et al. 2018).
Unless another end point (e.g., quantitative polymerase chain reaction) prevents its use, intravascular perfusion fixation is generally preferred for PNS processing. However, in some cases, immersion fixation is the better practice. For example, immersion fixation without prior perfusion fixation is used if samples need to be harvested quickly at necropsy to assist with subsampling for molecular studies and/or to enhance the preservation of labile molecules (e.g., antigens in tissue blocks destined for immunohistochemical [IHC] analysis) and fragile structures (e.g., intraepidermal sensory nerve fiber endings in skin of the plantar surface of the foot) that are degraded by prolonged exposure to conventional fixative agents. Harvest and handling of delicate PNS tissues, which may require detailed dissection under a dissecting microscope, are often best performed following tissue fixation in the less hectic, more controlled environment of the histology laboratory since such dissections may require longer periods of time than are generally available in the necropsy room. Dissection of incompletely fixed PNS tissue, especially if done rapidly during a hectic necropsy session, may lead to processing artifacts related to postmortem crushing and/or stretching.
While most PNS evaluations will be performed on paraffin-embedded sections, other embedding media and staining methodologies may greatly enhance the examination. Frozen sections may be useful or even required for detailed IHC-dependent examinations (e.g., intraepidermal nerve fiber density) or when thicker sections (>10 μm) may be required (e.g., stereological evaluations of spinal cord and ganglia). Resin embedding of osmicated (i.e., osmium-fixed) nerves (Figure 1) will allow production of semi thin sections (<1 μm), which are ideally suited for detection of demyelinating disorders, analysis of neuropathies, estimation (or calculation) of the G-ratio (i.e., the ratio of axon diameter alone to the diameter of the axon plus its myelin sheath), and morphometrics (e.g., measuring myelinated fiber sizes).
Dr. Butt concluded his presentation by noting the importance of performing special and/or IHC stains in nonclinical studies of PNS neurotoxicity. Different stains are used depending on the objectives of the study. Some neurohistological stains used in PNS evaluation include Luxol fast blue (which may enhance visualization of myelin in paraffin-embedded sections), Bielschowsky’s silver (which labels neurofilament-packed structures like axons and may enhance the visualization of swollen axons), neuronal nuclear antigen (used to visualize neurons), glutamine synthetase (which labels satellite glial cells in ganglia), and toluidine blue (typically used to stain osmicated, resin-embedded sections). These special stains may provide information regarding cellular targets and/or disease mechanisms that are hard if not impossible to discern on routine hematoxylin and eosin–stained sections.
Dr. Bernie Jortner gave a third presentation that defined common structural lesions and artifacts in the PNS. He started with a brief review of PNS organization, emphasizing that the cell bodies of many neurons with peripherally directed axons lie within the CNS and some sensory neurons of the PNS have central projections. The PNS is also important in neurotoxicity investigations for its own sake due to its critical importance as the “real-time” system for transmitting signals to and from CNS control centers. Due to the regular distributions of nerve fibers (in nerve cross sections) and neurons (in DRG sections), the PNS is well adapted for morphometric study. Importantly, the PNS is the only portion of the nervous system that can exhibit effective repair of affected nerve fibers.
Dr. Jortner provided definitions and showed examples of several major toxicant-induced lesions of the PNS. “Neuropathy” is a broad term applicable to any disease affecting the PNS, including but not limited to those resulting from toxicant exposure. “Neuronopathy” (e.g., pyridoxine toxicity) is a condition in which the neuron cell body represents the chief target. Neuronopathies may be limited to somatic sensory neurons (i.e., “ganglionopathy”), somatic motor neurons, or autonomic neurons or it may affect two or all three components; the affected PNS neuron populations can be predicted based on the constellation of in-life clinical signs. “Axonopathy” (e.g., organophosphate-induced delayed neurotoxicity) results from damage that is primarily centered on the axon (Figure 2). Axonal damage may impact fibers of any diameter, but variants linked to toxicant exposure often affect large-diameter fibers preferentially and tend to develop first in the distal axon. Such distal portions are located at the greatest distance from the neuron cell body, which is where most key proteins needed to maintain axon integrity must be produced. “Myelinopathy” (e.g., riboflavin deficiency) arises from damage to the Schwann cell and/or myelin sheath. Accordingly, myelinopathy typically involves large-diameter nerve fibers of the somatic PNS. The distinct patterns of these polyneuropathies (i.e., affecting multiple nerves at once) are depicted in Figure 3. Because of reciprocal trophic influences, primary injury to axons leads to secondary myelin damage, while primary myelin abnormalities foster secondary axon disintegration. Therefore, since both axons and myelin are affected in end-stage lesions, efforts to distinguish the initial target typically need to be performed in earlier stages of neurotoxic conditions.
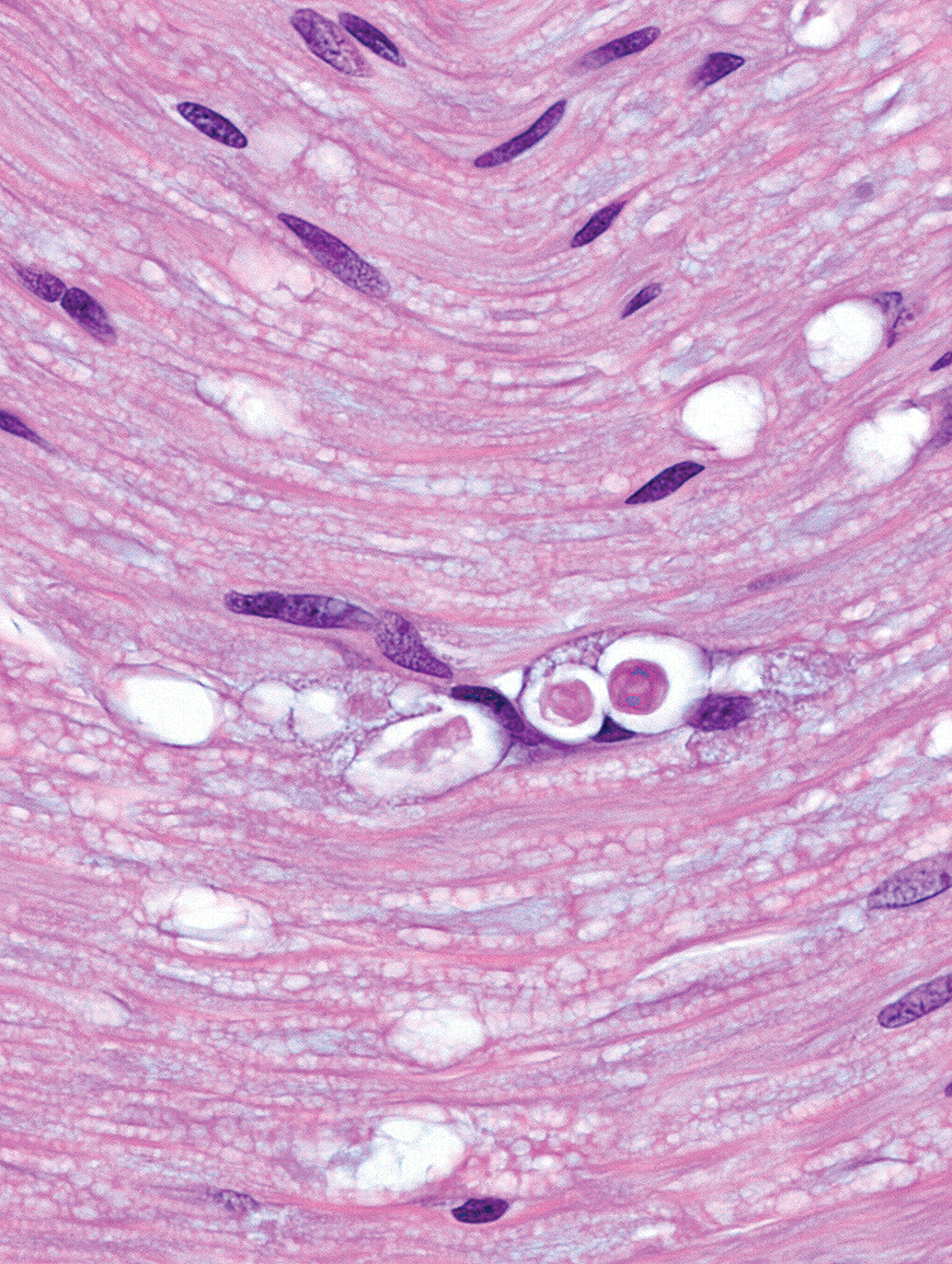
Nerve fiber degeneration (in this case due to axonal degeneration) characterized by vacuoles containing myelin and/or axonal debris (“digestion chambers”) and/or markedly distinct axonal swelling (“spheroids”). Sciatic nerve (longitudinal section), two-year-old control F344/N rat. Tissue preparation: immersion fixation in neutral buffered 10% formalin, paraffin embedding, hematoxylin and eosin staining.
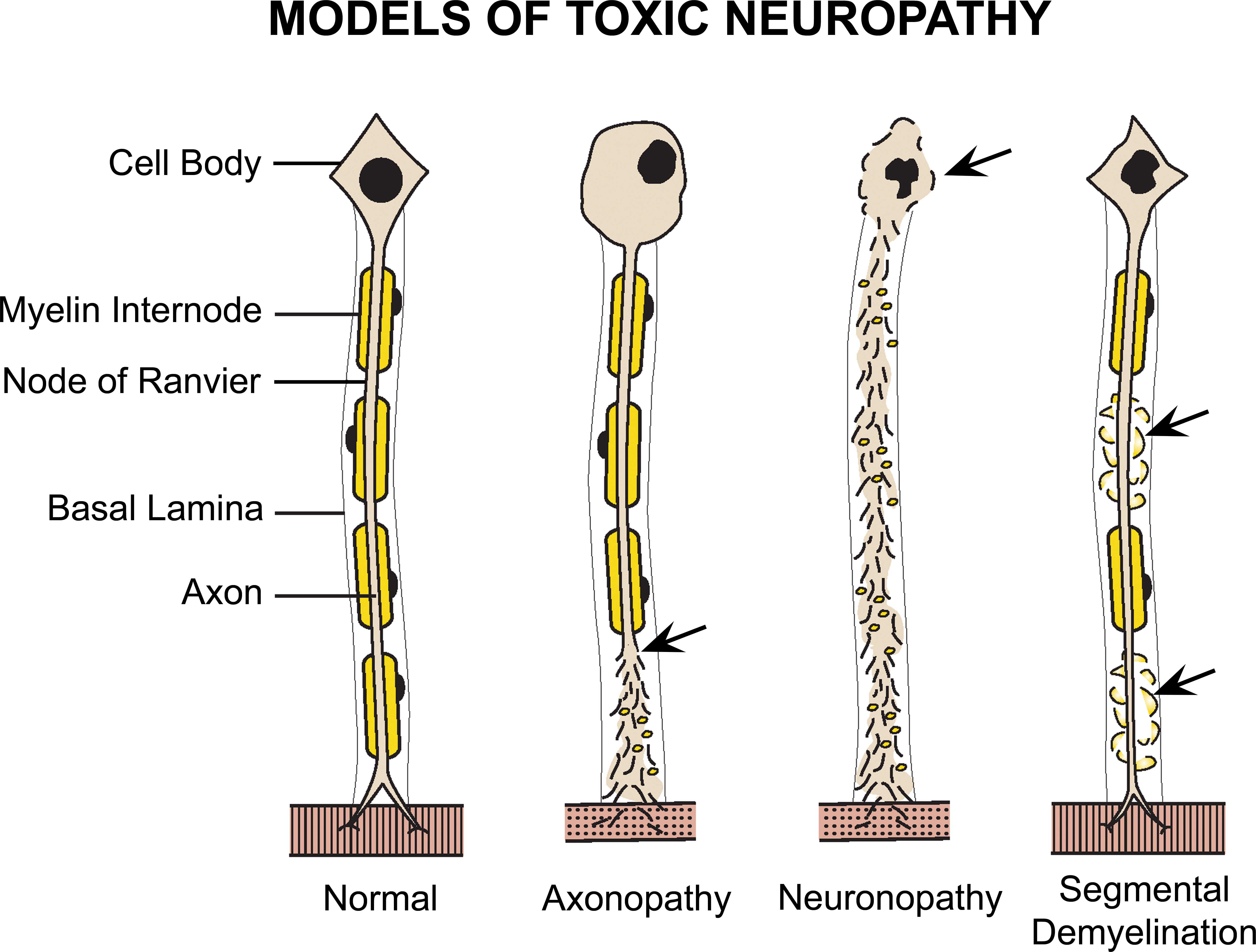
Compared to the normal condition (left), injury (shown by arrows) leads to different patterns of toxicant-induced injury to myelinated peripheral nerve fibers depending on the target. Primary damage to the axon (axonopathy) leads to loss of the distal axon and secondary disintegration of its myelin sheath with compensatory enlargement and chromatolysis of the neuron (i.e., the “axonal reaction”) to support axon regrowth. Primary injury to the cell body (neuronopathy) results in destruction of the neuron cell body and irreparable secondary loss of its axon and the corresponding myelin sheath. Primary damage to the myelin sheath or Schwann cell leads to segmental demyelination in association with retained axons early (as shown), although in later stage, the axon may degenerate due to the loss of trophic factors produced by Schwann cells.
A key distinction between the CNS and PNS is that repair by nerve fiber regeneration may occur in the PNS. The pattern of recovery in the PNS differs with the type of damage. Primary axonopathies are associated with Schwann cell proliferation in linear columns (Figure 4) followed by subsequent axonal regrowth through these columns, while primary myelinopathies appear as increased numbers of Schwann cell nuclei (because newly produced Schwann cells usually are smaller) surrounding intact axons. Axonal restoration takes time due to the slow rate at which axons regrow (approximately 0.5–1 mm/day; Morfini et al. 2012). The degree of axonal regeneration in the PNS may be underestimated by light microscopic evaluation (relative to transmission electron microscopic examination), especially small-diameter nerve fibers, due to the inability to fully assess subtle indications of nascent restoration (Ronchi et al. 2014). Remyelination as a regenerative event in myelinopathies may occur in both the PNS and CNS.
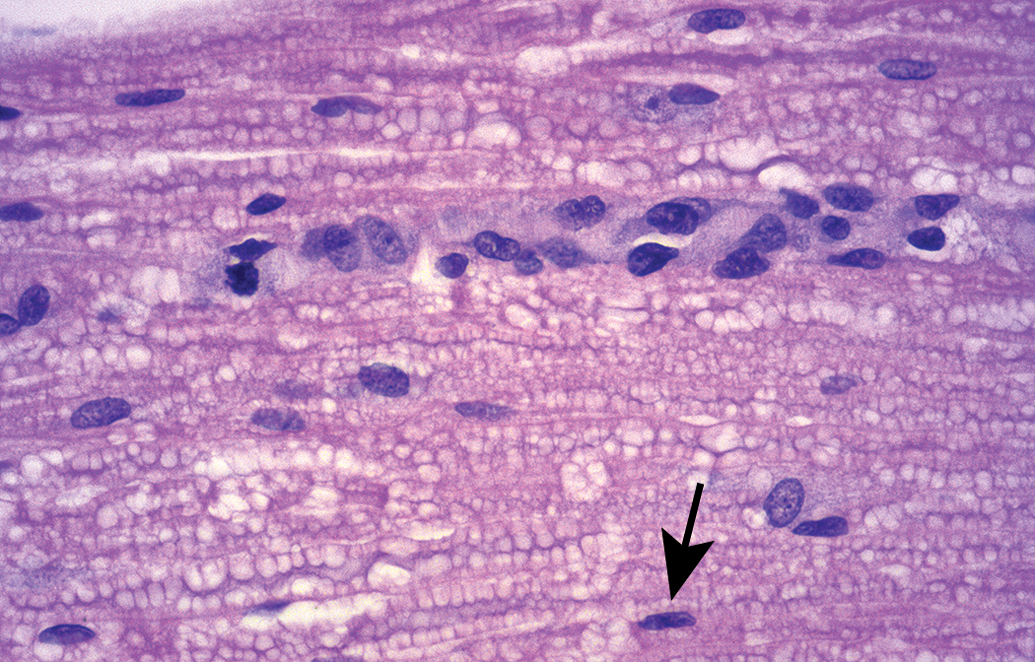
Increased Schwann cell proliferation (termed a “Büngner band”) is characterized by linear columns of plump, closely packed glial cells with abundant pale basophilic cytoplasm and large oval nuclei. These structures, which fill former “digestion chambers” once the debris left by degenerating axons (see Figure 2) has been removed, provide a permissive microenvironment to cradle regenerating axons. The arrow denotes the narrow, elongated nucleus of a “resting” Schwann cell that is providing the myelin sheath for an intact axon. Clear, colorless vacuoles (“bubbling”) within myelin sheaths throughout the section represent an artifact of immersion fixation with conventional (i.e., methanol-containing) formalin. Sciatic nerve (longitudinal section), control rat. Tissue preparation: immersion fixation in neutral buffered 10% formalin, paraffin embedding, hematoxylin and eosin staining.
Dr. Jortner also presented several common histological artifacts that may confound assessment of PNS lesion patterns. The most frequent processing-related artifacts include nerve fiber distortions associated with compression of unfixed tissue and/or immersion fixation using standard (methanol-containing) formalin (Figure 4), myelin splitting, and axon shrinkage. He concluded his talk by noting that osmium postfixation, epoxy resin embedding, semithin sectioning, and toluidine blue staining produce sections that provide excellent resolution by light microscopy (Figure 1), allowing critical evaluation of such samples. The fine detail evident in such preparations explains their selection as the standard methods employed for PNS analysis in the human diagnostic setting and in many PNS-focused basic experimental studies in animals.
The fourth presentation on classes and mechanisms of PNS neurotoxicants was delivered by Dr. Bill Valentine. He stated that humans have historically served the role of sentinel species for many neurotoxic agents with many documented neuropathies resulting from occupational and environmental exposures. Peripheral polyneuropathies have also been a frequent but unanticipated consequence of certain therapeutic interventions (e.g., antineoplastic chemotherapy). To rectify this situation and prevent the occurrence of human cases in the future, considerable effort is being directed at finding efficient and effective methods to identify potential PNS neurotoxicants during product development. There are many recognized PNS neurotoxicants, but information on their molecular mechanisms is scarce. Nevertheless, considerable benefit can be gained by mechanistic investigations (e.g., formulating structure–activity relationships), which then can be useful for predicting the neurotoxic effects of other agents that work through a similar mechanism, designing less neurotoxic analogues that retain desirable biological effects, developing sensitive noninvasive biomarkers, and designing relevant in vitro screening systems.
Critical to conducting mechanistic studies is an initial working hypothesis. An important component to developing such hypotheses is an accurate characterization of the neuropathy and selection of an appropriate model system; choosing among various models typically requires an accurate morphological evaluation. Knowledge of the primary lesion (e.g., neuronopathy, axonopathy, or myelinopathy) combined with an understanding of the unique structural and functional properties of the PNS can often provide initial insight into potential molecular targets. In some cases, agents with known mechanisms that produce similar PNS neurotoxic lesions may also provide guidance for initiating mechanistic studies. Evaluations of PNS neurotoxic mechanisms often combine experiments using in vitro systems, measurement of peripheral surrogate markers, and utilization of structural analogs that complement standard morphological methods. The general flow diagram for such integrated experiments is outlined in Figure 5.
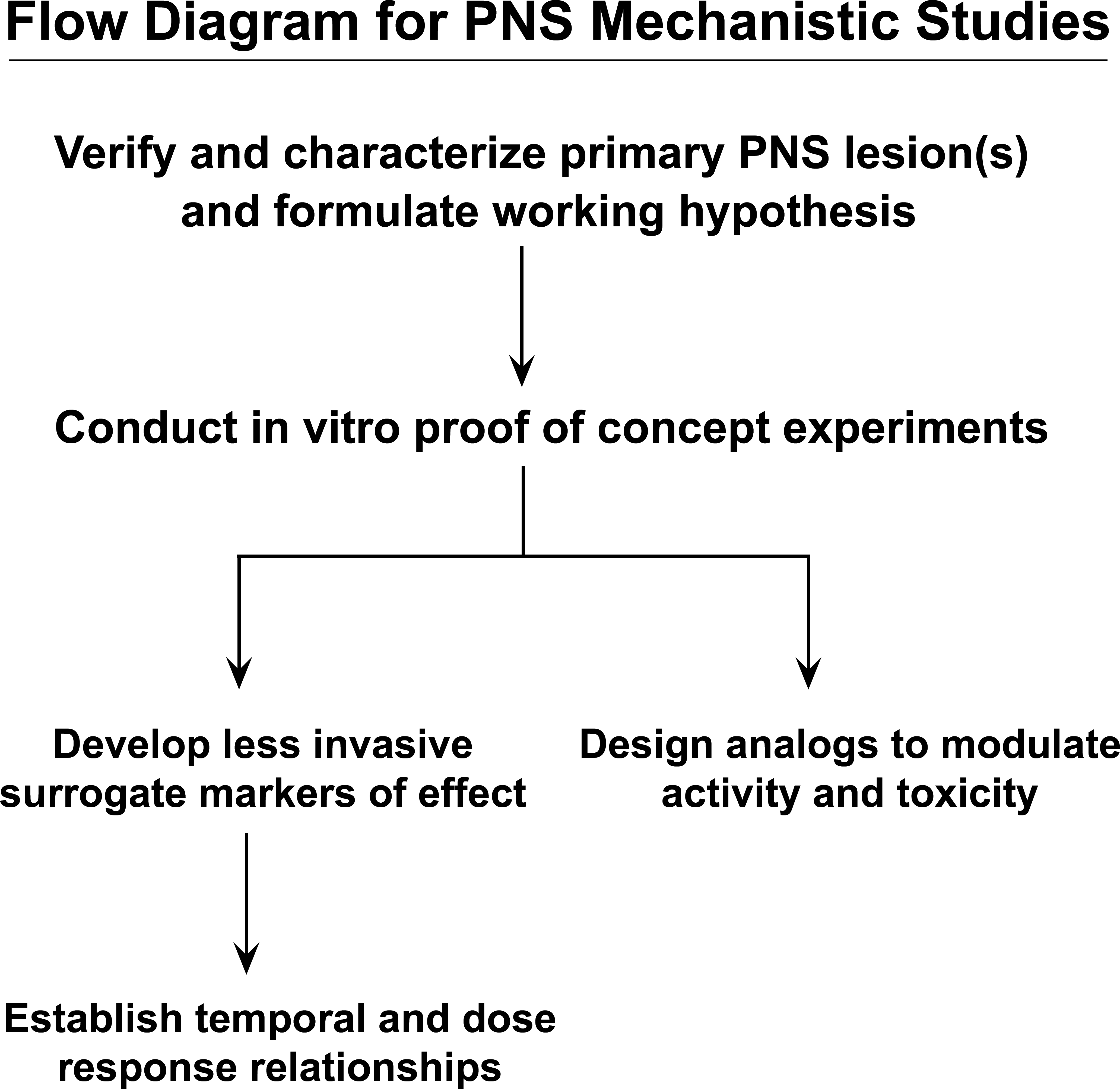
The general flow diagram showing integrated experiments for evaluating potential peripheral nervous system (PNS) neurotoxic conditions and mechanisms. These experiments may combine in vitro systems, attempt to measure peripheral (less invasive) surrogate markers to reduce the need for tissue acquisition and direct morphologic evaluation, and characterization of structural analogs to the neurotoxic agent to explore the full range of anatomic, functional, and molecular disturbances leading to PNS neurotoxicity.
Dr. Valentine concluded his presentation by reviewing work performed in his laboratory on the mechanisms and structure–activity relationships for several classical neurotoxicants that induce PNS axonopathies or myelinopathies. Investigations of carbon disulfide (CS2), dialkyldithiocarbamates, dithiocarbamate disulfides, and hexane have demonstrated the application of the integrated approach (Figure 5) and provided real-world examples of how knowledge of molecular mechanisms may be used to rationally design safer analogues and develop biomarkers of effect. While some agents (e.g., CS2) are directly neurotoxic, others (e.g., dithiocarbamates and hexane) must be metabolized to produce the neurotoxic molecule (Graham et al. 1995; Valentine et al. 1995). Other proposed mechanisms of PNS neurotoxicity include DNA damage (Englander 2013), enzymatic activation (Hidmark, Nawroth, and Fleming 2017), and mitochondrial disruption (Zhuo, Gorgun, and Englander 2018) in DRG neurons, which emphasizes the importance of evaluating the DRG in concert with their associated nerves when evaluating the PNS during nonclinical safety assessment.
Dr. Joe Arezzo delivered the last technical presentation, which introduced nonmorphological methods for evaluating the PNS. He emphasized that the nervous system is remarkably complex as axons that vary in length, speed, origin, and degree of myelination are charged with the continuous transmission and regeneration of electrical signals, often over considerable distances. Two of the most compelling issues that alter peripheral nerve function are exposure to medicines like chemotherapeutic agents and loss of the protective attributes of myelin. The loss of sensation produced by such insults is often progressive and manifests as diminished strength and balance as well as loss of speed and coordination.
One of the most challenging PNS insults is a length-dependent distal axonopathy with deficits that initially manifest at the distal segments of the affected nerves. While it is clear that histopathology provides the “gold standard” for the evaluation of neuropathies, there are valuable corollary measures that afford unique approaches for examining the onset and progression of such conditions. Most notably, measures that document changes in the speed, amplitude, and conduction of nerve impulses using a series of noninvasive surface electrodes can provide accurate quantitative measurements of nerve damage. Further, these noninvasive procedures allow for monitoring of changes in nerve function over time in the same subjects, and they often can identify functional deficits on the order of 5% to 8% compared to age-matched controls. The most striking value of nerve conduction velocity (NCV) may be the possibility of tracing functional recovery in individual subjects following changes in the underlying insult (i.e., removal of the toxicant, alteration of the dose). The ability to document changes in PNS function using noninvasive measures opens the door for the continuous monitoring of deficits and/or improvements in functionality.
Outstanding work in conventional histopathologic analysis of autopsy and biopsy specimens has identified the causes, nature, and progression of nerve damage. This structure-oriented method has greatly increased our understanding of the subtypes of neuropathy, but unfortunately there has been little success in translating new knowledge into more efficient approaches to identifying and treating neuropathy. Electrophysiology, in which direct measurement of NCV provides an opportunity to assess functional integrity in thickly insulated, large-diameter nerve fibers using surface or deep electrodes, has allowed us to track the onset, progression, and in some cases the recovery of neuropathic damage. New procedures are now emerging (e.g., intraepidermal fiber density, diffusion tensor imaging, NCV measurement in small-diameter fibers) that offer the promise of fine-tuning PNS diagnoses in the future, perhaps even including simultaneous in situ morphologic and functional analyses (Lauria et al. 2005; Arezzo, Litwak, and Zotova 2011; Zotova and Arezzo 2013; Brienza et al. 2014; Ohana et al. 2014; Mangus et al. 2016).
The course concluded with three case presentations designed to encourage discussion among all speakers and the audience. The first case presentation concerning biomarkers for detecting autonomic dysfunction was presented by Dr. Ingrid Pardo. She discussed the need for noninvasive, fluid-based biomarkers that can help predict neurotoxicity or aid the understanding of neurological disease in nonclinical species and humans. Such biomarkers of neurological damage are uncommon for both the CNS and PNS (Vernau, Vernau, and Bolon 2011; Apostolidis et al. 2017), but many scientists are collaborating to develop and test biomarkers for neurotoxicity applications. Recent progress includes the Health Education Systems Incorporated Subcommittee on Translational Biomarkers of Neurotoxicity (Roberts et al. 2015), the European Union’s Innovative Medicines Initiative Translational Safety Biomarker Pipeline (IMI-2 TransBioLine) effort to identify and validate new biomarkers of CNS injury (http://ec.europa.eu/research/participants/portal/desktop/en/opportunities/h2020/topics/imi2-2017-13-11.html), and the recent U.S. Food and Drug Administration authorization of the first blood test to aid in the evaluation of traumatic brain injury (TBI) in humans (Takala et al. 2016). This blood test, which monitors serum glial fibrillary acidic protein and ubiquitin C-terminal hydrolase L1, will help physicians determine the need for a computed tomography scan in patients suspected of having mild TBI and thus prevent unnecessary neuroimaging and associated radiation exposure to patients (Takala et al. 2016). For the PNS, Dr. Pardo and her Pfizer colleagues validated (using control ranges derived from 50 Wistar rats) the norepinephrine metabolite 3-methoxy-4-hydroxyphenylethylene glycol (MHPG; Eisenhofer, Kopin, and Goldstein 2004) as a potential non-invasive biomarker for autonomic sympathetic dysfunction. This biomarker was measured in urine at different time points during and following treatment with high doses (50 and 100 mg/kg/day) of guanethidine, an antihypertensive drug that causes selective sympathetic autonomic neuron necrosis (Burnstock et al. 1971). Light microscopic and stereological evaluations were performed in the cranial (superior) cervical ganglion (SCG) immediately following dosing and after a 3- to 4-week recovery period. Guanethidine reduced neuron counts and led to a size increase in the remaining neurons in SCGs following the recovery period—changes that were not detected immediately following dosing—while producing a decrease in urinary MHPG levels both during treatment and following recovery compared to control values (Zahner et al. 2017). Thus, MHPG levels appear to be a sensitive biomarker of sympathetic PNS dysfunction.
The second case presentation delivered by Dr. Deepa Rao discussed degenerative effects in the sciatic nerve as a common age-related incidental finding in rats and the potential value of morphometric approaches to distinguish between age-related and chemically-induced findings. Such degenerative changes in nerves typically include vacuoles containing myelin and/or axonal debris (“digestion chambers”; Figure 2) and/or markedly distinct axonal swelling (“spheroids”). In chronic toxicity and carcinogenicity studies, such lesions often pose a challenge when a potential test article–related effect needs to be differentiated from an age-related spontaneous finding since end-stage changes in nerve fibers overlap and are seldom characteristic of a distinctive process (i.e., normal physiologic attrition vs. agent-related neurotoxicity), at least when assessed by light microscopy. In the present case from a 2-year rat carcinogenicity study, the features and incidences of sciatic nerve axonal degeneration in male and female control and high-dose Fischer 344/N rats (n = 50 per group per sex) were first graded by a routine informed (i.e., “unblinded”) histopathological evaluation by the study and two peer review pathologists. Although both peer reviewers suggested the potential for exacerbation by the test article, the potential that the finding represented age-related background changes could not be ruled out. A third peer review was requested, and the appointed pathologist (Dr. Rao) chose to employ a different method to address the question: a masked (“blinded”) morphometric analysis. For this purpose, the slides for both male and female, control and high-dose rats were randomized and the number of foci of axonal degeneration (defined as a cluster of vacuoles containing axonal and/or myelin debris, and/or axonal swelling) were counted in two longitudinal sections of sciatic nerve (∼1 cm long for each section) per rat using the 20× objective (to ensure that small lesions were seen). Morphometric data were tabulated for each section and slide, and a statistical analysis (one-sided Mann–Whitney test) of the unmasked data was conducted. To account for possible litter effects (passed on to the offspring through either the dam or the sire) as well as possible effects of body weight and age, an analysis of covariance (ANCOVA) was used to test for differences between the high-dose and control groups; by this test, the dam or sire was the random effect, dose was the fixed effect, and body weight and age at necropsy were covariates. These analyses were conducted on log-transformed standardized foci frequencies. Any differences in section area among animals were considered to be minimal, given that nerve sections for each of 50 rats per sex per group were ∼1 cm in length, consistently collected from the same location, and prepared using standard procedures for tissue processing across all dose groups. In studies with smaller group sizes (e.g., 10 rats per sex per group) or with tissue sections from larger organs, variations in section area should be accounted for in conducting the morphometric analysis. Results of the blinded quantitative peer review showed a statistically significant increase for high-dose males and females compared to controls, with females showing a greater difference compared to males. Therefore, these data suggested that an exacerbation of an age-related finding was present in high-dose animals.
Axonal degeneration in sciatic nerves is an expected age-related degenerative change (Cotard-Bartley et al. 1981; Mufson and Stein 1980; Mitsumori and Boorman 1990; McMartin et al. 1997) that occurs commonly in 2-year-old rats, including control animals. Often, these degenerative changes are attributed to weight-bearing trauma induced by housing rats in hanging wire-mesh cages, and thus typically the lesion incidence is higher in heavier males (Cotard-Bartley et al. 1981). However, in the current case, rats were not housed in hanging wire-mesh cages. The statistical comparisons between the high-dose and control groups, and between males and females, included statistical adjustments for increased survival in treated animals as well as body weight changes. As expected, both sexes had more degenerative foci with increasing age. On the other hand, body weight changes were significantly associated with the number of degenerative foci only in males. In fact, females exhibited more severe degeneration compared to males, which is consistent with chemically induced exacerbation of the age-related background degeneration. A detailed review of axonal degeneration as investigated in this study has been published elsewhere (Behl et al. 2013). Therefore, depending on the scientific question being investigated, in some instances, the extent and implications of a PNS change might be better assessed using a morphometric analysis rather than conventional histopathologic lesion grading. That said, with respect to the safety assessment setting, such morphometric analyses will represent a “second-tier” technique that may be deployed only as needed to clarify any discrepancies that remain after the routine histopathological evaluation has been completed.
The final case presentation by Dr. Alok Sharma described nerve fiber degeneration related to altered blood glucose concentrations from a toxicity study performed in euglycemic rats. The test article, which was being developed to treat type 2 diabetes, was administered daily via oral gavage for 6 months. Chronic hypoglycemia induced by test article treatment produced clinical signs of ataxia and limited hind limb use in males, and many animals had to be necropsied early due to severe debilitation. Microscopically, dose-dependent, minimal to moderate nerve fiber degeneration was present in the sciatic nerve and brachial plexus of mid- and high-dose males and high-dose females. Lesions were characterized by multifocal dilation/vacuolation of myelin sheaths, which were often empty and infrequently contained axonal and/or myelin debris (i.e., Wallerian-like degeneration). Glucodynamic data collected two weeks prior to moribund termination revealed dose-related decreases in serum glucose concentrations in males that were transient in low-dose (recovered by 8 hr postdose) and mid-dose (restored by 18 hr postdose) rats but were substantial and persistent in high-dose animals. The severity and incidence of nerve fiber degeneration were clearly correlated with the degree of profound hypoglycemia, and thus this change was interpreted to represent an exaggerated pharmacologic response rather than off-target PNS neurotoxicity.
Nerve fiber degeneration has been reported in multiple nonclinical species administered antidiabetic agents (such as glucokinase activators and long-acting basal insulin) due to persistent and profound hypoglycemia (Jensen, Bøgh, and Lykkesfeldt 2014; Pettersen et al. 2014; Tirmenstein et al. 2015; Jensen et al. 2016) as well as in hypoglycemic insulinoma patients (Heckmann et al. 2000; Striano et al. 2003). The pathogenesis of hypoglycemia-induced peripheral neuropathy remains unclear. Because the degenerative changes due to hypoglycemia occur both in the axon and myelin components of nerve fibers, and since few reports describe hypoglycemia-induced damage to ventral horn neurons, the proposed mechanism is that hypoglycemia causes a “dying back” peripheral neuropathy where both axonal and myelin degeneration occurs initially at distal ends of nerve fibers and then progresses to more proximal parts of nerves and ultimately to ventral horn neurons of the spinal cord (Mohseni 2014).
Peripheral neuropathy is a common complication seen in diabetic patients (World Health Organization 2016), who have long-standing hyperglycemia, as well as individuals treated with insulin, who develop hypoglycemia (Mohseni 2001). The peripheral neuropathy caused by these two blood glucose imbalances differ in their manifestations. Insulin-associated hypoglycemia produces lesions in large-diameter, thickly myelinated motor nerve fibers (Mohseni 2000; Jamali and Mohseni 2006); similar findings have been reported in animals with insulinomas (Van Ham et al. 1997; Sugimoto et al. 2004). In contrast, hyperglycemia mainly yields lesions in smaller-diameter sensory nerve fibers (Sima, Zhang, and Greene 1989; Jamali and Mohseni 2006). Nerve fiber degeneration and other hypoglycemia-induced microscopic findings such as neuronal degeneration, skeletal muscle atrophy, and vascular degeneration are not usually observed when hyperglycemic animal models of type 2 diabetes (e.g., Zucker diabetic fatty rats) are given antidiabetic molecules, which further supports exaggerated pharmacology as the cause of these findings observed in euglycemic rats (Tirmenstein et al. 2015).
The importance of the PNS as a potential target for adverse effects related to accidental or intentional exposure to chemicals and therapeutic agents warrants an increased emphasis on PNS evaluation as an important element in routine nonclinical safety assessment. This half-day continuing education session familiarized participants with multiple topics related to the basic structure, function, histological processing, and evaluation of the PNS in nonclinical species and human patients. In addition, typical PNS neurotoxic changes were compared and contrasted to common incidental findings and processing artifacts to aid audience members in performing more consistent PNS examinations. The course contents complemented and expanded on recently released “best practice” recommendations for PNS sampling, processing, and analysis applicable to nonclinical species (Bolon et al. 2018). Taken together, the contents of this short course and the best practices white paper are anticipated to help improve translation of PNS-related safety findings in nonclinical species to predict possible risk to humans.
Footnotes
Authors’ Note
This article reflects the views of Deepa B. Rao on research conducted during her tenure at the U.S. National Toxicology Program (NTP) and should not be construed as representing the views or policies of her current employer, the U.S. Food and Drug Administration.
Acknowledgments
The authors thank the staff and members of the Society of Toxicologic Pathology (STP) for selecting this continuing education course for presentation at the 2018 Annual STP Meeting and Ms. Beth Mahler for her aid in optimizing the resolution of the photomicrographs. Dr. Pardo acknowledges Dr. Georg Krinke for his scientific contribution in biomarkers for autonomic dysfunction. Dr. Rao acknowledges the contribution of Drs. Grace Kissling, David E. Malarkey, and Susan Elmore and support from the U.S. National Toxicology Program where the work for her case presentation was conducted.
Author Contributions
All authors (IP, DR, MB, BJ, WV, JA, AS, BB) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
