Abstract
The simple tripartite classification of sensory neurons as A-beta, A-delta, and C fibers fails to convey the complexity of the neurons that encode stimuli as diverse as the texture of a surface, the location of a pinprick, or the direction of hair movement as a breeze moves across the skin. It has also proven to be inadequate when investigating the molecular mechanisms underlying pain, which can encompass any combination of chemical, tactile, and thermal modalities. Beginning with a brief overview of visceral and sensory neuroanatomy, this review expands upon sensory innervation of the skin as a prime example of the heterogeneity and complexity of the somatosensory nervous system. Neuroscientists have characterized defining features of over 15 subtypes of sensory neurons that innervate the skin of the mouse. This has enabled the study of cell-specific mechanisms of pain, which suggests that diverse sensory neuron subtypes may have distinct susceptibilities to toxic injury and different roles in pathologic mechanisms underlying altered sensation. Leveraging this growing body of knowledge for preclinical trials and models of neurotoxicity can vastly improve our understanding of peripheral nervous system dysfunction, advancing the fields of toxicologic pathology and neuropathology alike.
Keywords
Introduction
A simple characterization of the sensory system assigns distinct functions to A-beta, A-delta, and C-fibers based on fiber size, cell body size, and level of myelination. 1,2 However, this tripartite classification of sensory neurons fails to convey the complexity of the neurons that encode stimuli as diverse as the temperature of the environment, texture of a surface, the direction of a breeze, or the precise location of a pinprick of the skin. Basic and translational neuroscientists have harnessed cutting-edge technology paired with rodent models of disease to examine cell-specific mechanisms of sensory physiology. The emerging understanding of sensory biology is that different types of sensation are driven by neuronal subtypes that are distinct in terms of their gene expression pattern, pattern of innervation of the end organ, specialized axon terminal morphology, characteristic electrophysiological properties, intercellular interactions within the dorsal root ganglion (DRG), and pattern of innervation of the spinal cord dorsal horn and brain stem. 3 -5 Not only do these distinguishing features confer selective responsivity to specific types of stimuli, these features may confer distinct patterns of plasticity after traumatic injury, toxic insult, or disease conditions, as described below. The study of skin afferents has generated the leading edge in our cellular and molecular understanding of sensory neuron biology, yielding key insight into sensory neuropathology. An update of the neural underpinnings of skin sensation can therefore provide a valuable perspective for any field of study that relies upon the histomorphologic or functional assessment of the sensory nervous system. As an aid for specialists engaged in several facets of preclinical trials and toxicology studies, this review of functional somatosensory anatomy includes (1) an overview of neuroanatomy of the peripheral sensory system, (2) a description of skin innervation by a wide array of nociceptor and mechanoreceptor subtypes, (3) examples of skin afferent pathology that can be identified by histology and immunohistochemistry, and (4) a synopsis of behavioral assays that can elucidate sensory pathology in rodent studies.
An Overview of Peripheral Sensory Neuroanatomy
Sensory neurons, also termed primary afferents, are derived from the neural crest. 2 Some neural crest-derived progenitors cells are bipotent, generating both sensory neurons and the satellite glia that envelope and support those neurons within sensory ganglia where they are housed. 6 Some satellite glia cells are thought to serve as neural progenitor cells in adult animals, giving rise to new sensory neurons with age, as seen in some rat strains, 7 and after certain types of nerve injury or cell death. 8 -10 These findings support the interesting prospect of adult neurogenesis in sensory ganglia. Electrical coupling between groups of satellite glia, mediated by gap junctions, facilitates coordination of their function. 11,12 The sheath formed by satellite glia surrounds the sensory neuron cell body, but is not continuous. Rather, it contains interspaced gaps that allow the penetration of dye tracers, neurotransmitters, and other macromolecules. 13 Similarly, sensory ganglion blood vessels are lined by a mixture of continuous and fenestrated endothelium 14 that also allows extravasation of dye tracers and macromolecules. As a consequence, unlike central nervous system (CNS) neurons that sit within the blood–brain barrier, sensory neuron cell bodies lack a barrier to the circulation. These structural features make it likely that sensory neurons are readily exposed to inflammatory mediators, systemic drugs, or toxins present in the systemic circulation, perhaps more so than many other organs in the body.
Sensory neurons are pseudounipolar neurons with a single axon that gives way to an axon bifurcation (T-shaped junction) with a peripheral and central branch. Sensory neurons transmit electrical signals from peripheral axon terminals in the end organ in a retrograde direction, through the axon bifurcation bypassing the cell body, directly into the central axon branch that terminates in the spinal cord or brain stem. All DRG subtypes express the synthetic enzymes required to make glutamate 15 and are capable of releasing glutamate at central axon terminals. 16 -18 As detailed below, at least some sensory neurons are also capable of releasing neurochemicals from their peripheral axon terminals, thereby affecting the functions of end-organ tissues, endothelium, vascular smooth muscle, and other neurons, as well as resident and infiltrating inflammatory cells. Interestingly, the cell soma of sensory neurons express synaptic vesicle markers and neurotransmitter receptors within the DRG and there is evidence of intraganglionic neurotransmitter release within the DRG. 19,20 The DRG neurons can also synthesize and release gamma-Aminobutyric acid (GABA), 21,22 while a subset of sensory neurons can release neuropeptides, as detailed below. Emerging evidence suggests GABAergic signaling within the DRG can act as a sort of “gate” wherein the cell soma can modulate signals at the axon bifurcation, as they are conducted from the peripheral axon to the central axon. 22 Communication within the DRG also includes glia–neuronal communication between satellite glia and sensory neurons mediated by adenosine triphosphate (ATP), purinergic receptors, and gap junctions. 23 There remains much to learn about local signaling within the DRG.
The sensory components of the peripheral nervous system (PNS) consist of both the visceral sensory system (Table 1) and the somatosensory system (Table 2). Although a detailed description of visceral sensory innervation is beyond the scope of this review, a summary is included as part of our overview of sensory neuroanatomy. The cell bodies of the visceral sensory system, also referred to as general visceral afferents (GVAs), are housed in bilateral ganglia including DRG as well as the geniculate ganglia of the facial nerve, distal glossopharyngeal ganglia (petrosal ganglia), and distal vagal ganglia (nodose ganglia) of cranial nerves (Cr.N.) VII, IX, and X, respectively (Table 1). The terminals of GVA of Cr.N. VII and IX innervate structures such as the tympanic membrane, middle ear, and carotid body, while visceral afferents of the vagus nerve (Cr.N. X) innervate the trachea, lungs, liver, pancreas, digestive tract, and bladder 1 (Table 1). The trachea, lungs, digestive tract, and urinary bladder also receive another set of sensory innervation, provided by DRG neurons located at corresponding levels of the spine. For organs with dual sensory innervation, the vagal sensory neurons are thought to mediate sensation of mechanical, chemical, and osmotic stimuli that induce reflex activity such as vomiting and increased gastrointestinal motility, while sensory neurons housed in the DRG are thought to be responsible for conscious perception of stretch and distention, as well as visceral pain. 1,24,25 Sensory axon terminals are found within the mucosa, submucosa, muscle, adventitia, and peritoneum (e.g. mesentery) of visceral organs and the pattern of visceral innervation may differ across neuronal subtypes that may serve distinct functions. 1,26 -29 Visceral sensory fibers are thought to be primarily C and A-delta fibers, most of which are mechanosensitive or chemosensitive. 27 -29 In addition, rapidly adapting A-beta mechanosensitive afferents have been identified in the trachea and airways where they are thought to contribute to cough reflexes and altered breathing patterns. 30,31 Although unresponsive to noxious stimuli under normal conditions, some visceral afferents become sensitized in the presence of irritants and inflammatory mediators through mechanisms that remain poorly understood. 26 The central axon terminals of visceral afferents of the Cr.N. VII, IX, and X are found in the nucleus of the solitary tract of the medulla. 1,32 However, some reports indicate the GVA axons of Cr.N. VII and IX terminate within the spinal trigeminal nuclei in at least some species, particularly for fibers that relay information about mechanosensation and pain. 1 Other reports suggest that a small number of GVA Cr.N. IX and X afferents synapse onto neurons of the dorsal vagal complex including the area postrema and dorsal motor nucleus of the vagus. 24,33,34 Visceral afferents arising from the DRG project to the deep layers of the spinal cord dorsal horn, with broad projections to many spinal levels near that particular DRG.
Neuronal Subtypes of the Visceral Sensory System (General Visceral Afferents).
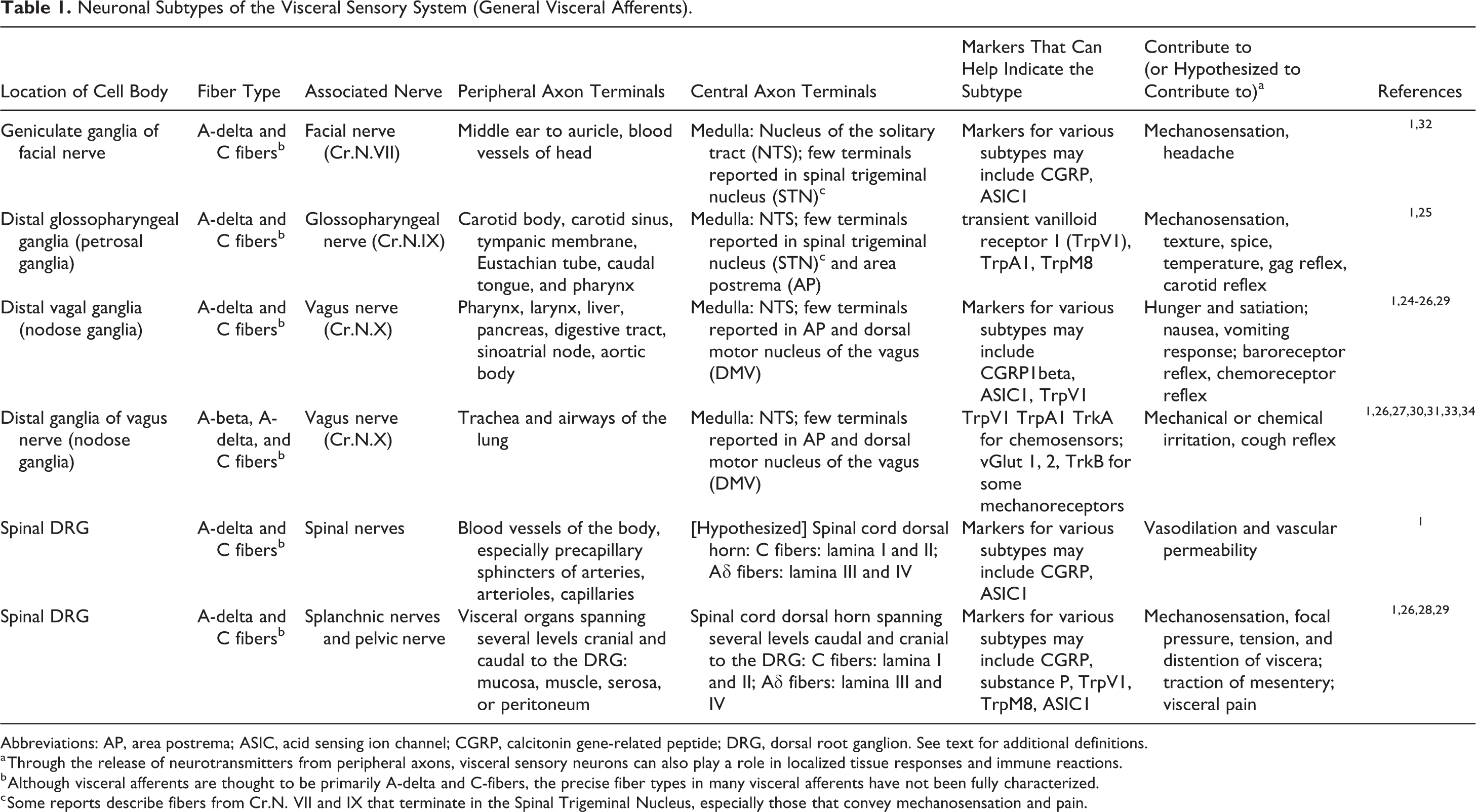
Abbreviations: AP, area postrema; ASIC, acid sensing ion channel; CGRP, calcitonin gene-related peptide; DRG, dorsal root ganglion. See text for additional definitions.
a Through the release of neurotransmitters from peripheral axons, visceral sensory neurons can also play a role in localized tissue responses and immune reactions.
b Although visceral afferents are thought to be primarily A-delta and C-fibers, the precise fiber types in many visceral afferents have not been fully characterized.
c Some reports describe fibers from Cr.N. VII and IX that terminate in the Spinal Trigeminal Nucleus, especially those that convey mechanosensation and pain.
Neuronal Subtypes of the Somatosensory System (General Somatic Afferents).
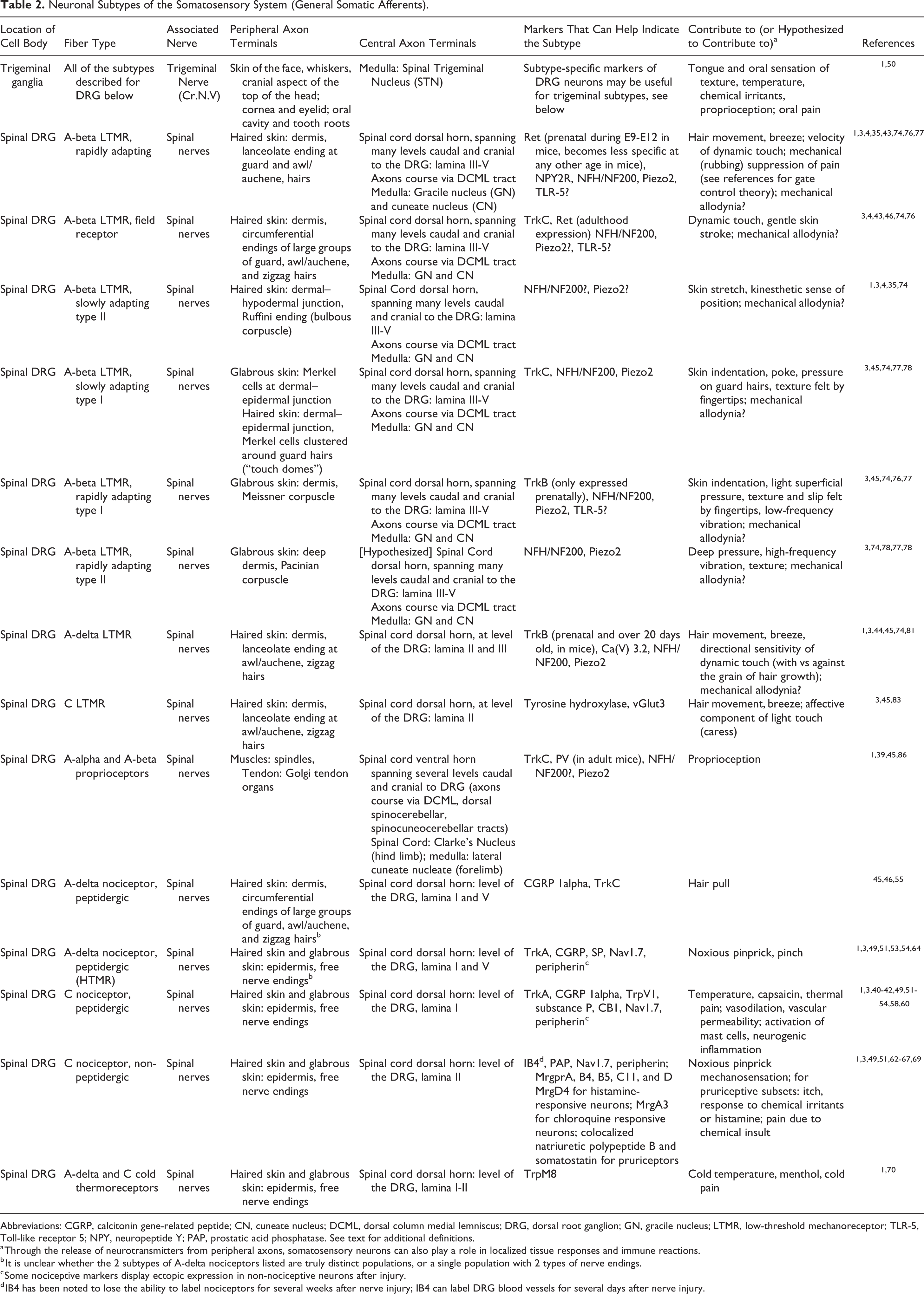
Abbreviations: CGRP, calcitonin gene-related peptide; CN, cuneate nucleus; DCML, dorsal column medial lemniscus; DRG, dorsal root ganglion; GN, gracile nucleus; LTMR, low-threshold mechanoreceptor; TLR-5, Toll-like receptor 5; NPY, neuropeptide Y; PAP, prostatic acid phosphatase. See text for additional definitions.
a Through the release of neurotransmitters from peripheral axons, somatosensory neurons can also play a role in localized tissue responses and immune reactions.
b It is unclear whether the 2 subtypes of A-delta nociceptors listed are truly distinct populations, or a single population with 2 types of nerve endings.
c Some nociceptive markers display ectopic expression in non-nociceptive neurons after injury.
d IB4 has been noted to lose the ability to label nociceptors for several weeks after nerve injury; IB4 can label DRG blood vessels for several days after nerve injury.
The somatosensory system, also referred to as a collection of general somatic afferents, conveys sensations of touch, pain, and position of the extremities, as summarized in Table 2. General somatic afferents of the head arise from cell bodies found primarily within the trigeminal ganglia, whose peripheral axons form the trigeminal nerve (Cr.N. V). Trigeminal neurons mediate touch, pain, and temperature sensation of the face and forehead as well as the oral and nasal mucosa (Table 2). The central axons of trigeminal neurons terminate in the spinal trigeminal nucleus. Somatic afferents of the caudal aspect of the head and the rest of the body consist of cell bodies found in spinal DRG, along with peripheral axons that innervate the end-organ and central axons that terminate in the spinal cord dorsal horn or medulla (Table 2). Classic descriptions of somatosensory neurons often simplistically ascribe C fibers and A-delta fibers to nociception and A-beta fibers to touch. There are also A-alpha and A-beta proprioceptors that innervate muscle and joint to encode the sensation of body position in space. Neuroscientists have identified key molecular markers and electrophysiological characteristics of various sensory neuron subtypes, enabling a more intricate understanding of the sensory neuron function, sensory deficits, and pain. Perhaps, this is exemplified best in mouse studies that have highlighted distinguishing features of over 15 different subtypes of sensory neurons that innervate the haired and glabrous skin, including at least 9 subtypes of touch neurons across C, A-delta, and A-beta fiber types. 4,35 Thus, the skin serves as an informative organ to understand the nuanced heterogeneity of the sensory system and to glimpse cell-specific mechanisms of dysregulation that occur after insult or injury to the nerve.
Skin Innervation as a Model of Sensory Biology
The neural underpinnings of skin sensation offer a perfect opportunity to explore the intricacies of sensory biology. Skin afferents demonstrate distinguishing features at each component of the neuron, including peripheral axon terminals, cell body, and axon terminals within the CNS. Sensory neurons have been functionally classified according to their neurophysiologic properties assessed using either ex vivo skin nerve preparations or in vivo electrophysiology in anesthetized rodents 36 -38 and by correlating these results with neurophysiological, behavioral, and psychophysical studies carried out in the skin of rodents, cats, nonhuman primates, and humans. Nociceptors are defined by the ability to respond to painful stimuli in a dose-dependent manner with no responsiveness to low-intensity mechanical stimuli. Pruriceptors encode the presence and intensity of itch-producing chemical and mechanical stimuli. The multiple populations of low-threshold mechanoreceptors (LTMRs) respond to low-intensity mechanical stimuli, encoding several modalities of light touch. As described in greater detail below and in Table 2, the identification of molecular markers enriched in particular subsets of sensory neurons has led to the further definitive identification and characterization of these sensory subtypes. Studies of knockout mice and human patients with loss-of-function mutations have likewise helped to elucidate functional roles of several molecular markers. 39 -42 This has been complemented by the more recent advent of publically available, large-scale gene expression atlases, single-cell transcriptomics, and the development of transgenic mouse lines with fluorescently labeled neuronal subtypes that aid in characterizing those subtypes during neurophysiology assays and anatomic studies (Figure 1). 43 -46
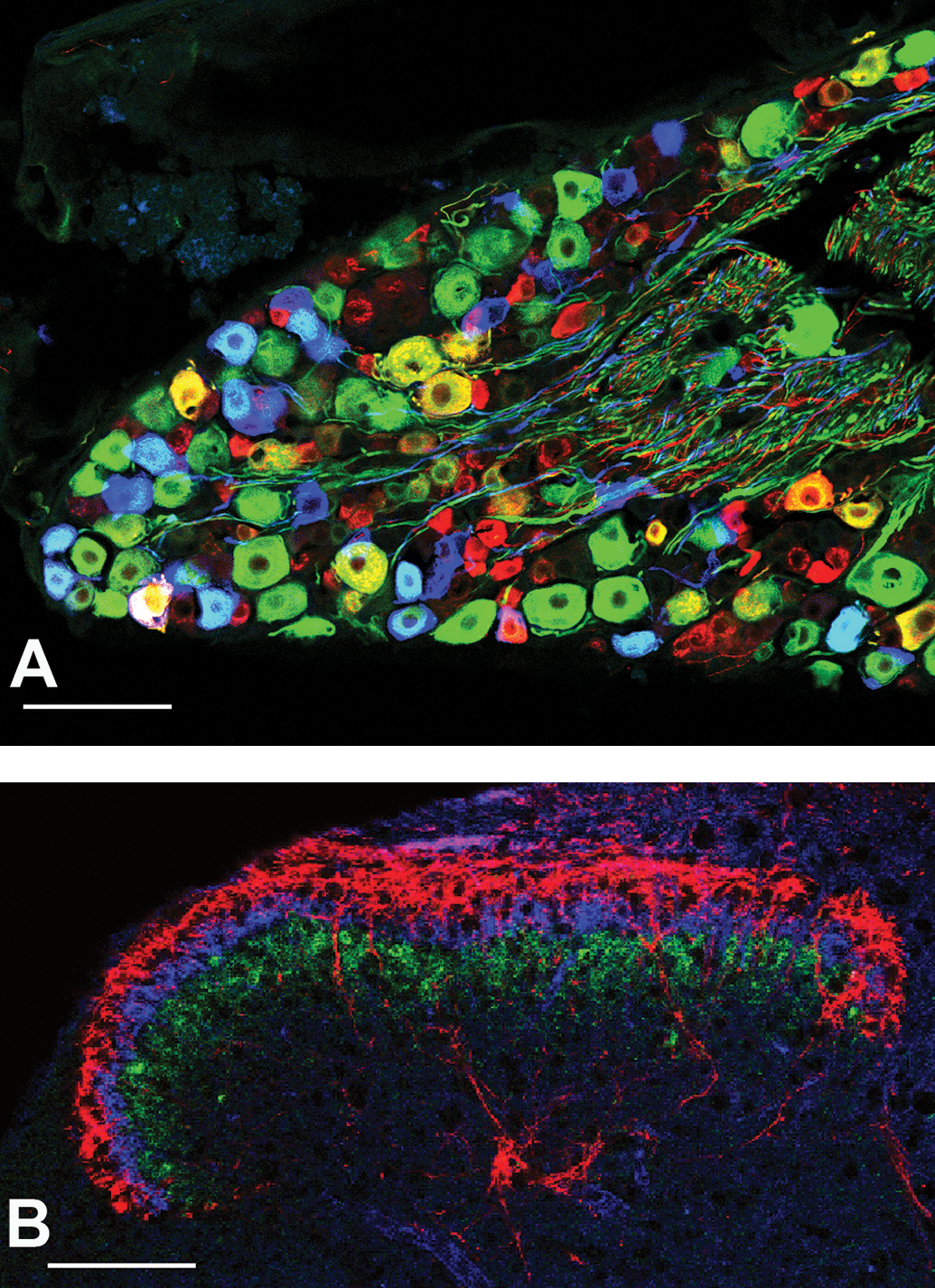
A, Confocal photomicrograph demonstrating the diversity of mouse DRG neurons. Immunofluorescent staining indicates the cytoplasmic expression of NF200 (green) and CGRP (red). Genetic labeling of A-delta LTMRs (blue, pseudocolor) was achieved through use of TrkB-specific expression of the tdTomato fluorescent reporter, induced on P22. These cytoplasmic markers stain the cell soma as well as axons. Neurons that appear yellow are colabeled with NF200 and CGRP. Neurons that appear teal are A-delta LTMRs colabeled with NF200. Neurons that appear white are A-delta LTMRs that are colabeled with NF200 and CGRP. Scale bar is 100 µm. B, Confocal photomicrograph of sensory neuron axon terminals in the spinal cord dorsal horn. The characteristic laminar organization of the spinal cord dorsal horn is exemplified by the central axon terminals of sensory neurons. CGRP (red) is expressed by peptidergic nociceptors, while IB4 (blue) labels non-peptidergic nociceptors, both found in the superficial lamina of the dorsal horn. Protein kinase C gamma (green) is a marker of spinal cord interneurons that serve as a useful landmark, dividing the superficial lamina from the deep lamina where A-beta and A-delta LTMR nerve terminals are found (not shown). Scale bar is 100 µm. CGRP, calcitonin gene-related peptide; DRG, dorsal root ganglion; IB4, Griffonia Simplicifolia Lectin I isolectin B4; LTMR, low-threshold mechanoreceptors, NF200, Neurofilament 200kD isoform.
As in other mammalian species, the cell bodies of rodent skin afferents consist of small-, medium-, and large-diameter neurons. Small-diameter neurons typically have unmyelinated axons and slow conduction velocities; these include peptidergic nociceptors, nonpeptidergic nociceptors, C-LTMRs, and pruriceptors. Medium- to large-diameter neurons of the A-delta subgroup have lightly myelinated axons and moderate conduction velocities; these include A-delta nociceptors and A-delta LTMRs. Other large diameter neurons of the DRG have large, heavily myelinated axons and fast conduction velocities, including several A-beta LTMR subtypes. Some studies have also identified A-beta nociceptors. 47,48 Many molecular markers that help identify these subtypes are delineated below and in Table 2. However, it should be noted that some of these markers, particularly those of nociceptors, may be less specific after nerve injury (see section “Features of Sensory Pathology Detected in the DRG”).
Peptidergic nociceptors include C fibers that are responsible for thermal sensation and pain in response to noxious heat or capsaicin. 49 Molecular markers include neuropeptides such as calcitonin gene-related peptide (CGRP) or substance P, as well as ion channels such as transient vanilloid receptor 1 (TrpV1) and endocannabinoid receptors like CB1. 50 –53 Peptidergic nociceptors also express tropomyosin receptor kinase A (TrkA) in adulthood, though TrkA is expressed across a broader range of neuronal subtypes during prenatal development. 53,54 The sodium channel Nav1.7 also mediates pain and is expressed by a subset of peptidergic nociceptors, along with a smaller subset of nonpeptidergic nociceptors, in a similar pattern as the marker peripherin. 41,42 The axon terminals of most peptidergic nociceptors are free nerve endings within the epidermis of haired and glabrous skin where they are well suited to detect breaks in the epidermal barrier to signal epidermal injury. There are also CGRP-positive A-delta nociceptors whose axon terminals form circumferential endings in the dermis, around hair follicles. 55 These are thought to signal pain associated with hair pull. Peptidergic nociceptors can release neuropeptides and other factors that influence inflammatory processes in the skin, a phenomenon termed neurogenic inflammation. For example, the release of CGRP, substance P, and neurokinin A from free nerve endings of the skin (and from sensory nerve endings of skin blood vessels) causes vasodilation and increased vascular permeability. 56 -58 Substance P can also prime mast cells and cause mast cell degranulation 59 through its activity at the Mas-related G-coupled protein receptor (Mrgpr or Mrg) ×2 receptor (Mrgpr×2 58,60,61 ). Some peptidergic nociceptors also express receptors for histamine, thereby encoding histamine-related itch. 62 Thus, peptidergic nociceptors can mediate bidirectional feedback, wherein histamine stimulates the release of neuropeptides that further increase histamine release. It is likely that sensory neurons also interface with other parts of wound healing and the immune response in ways we are just beginning to understand.
Nonpeptidergic nociceptors are C fibers with free nerve endings in the epidermis. They are often identified by the binding of Griffonia Simplicifolia Lectin I isolectin B4 (IB4), immunostaining for prostatic acid phosphatase (PAP), or the expression of the Mrgpr D receptor. 63,64 It should be noted that PAP also stains a small number of peptidergic nociceptors, particularly in mice. 65 Nonpeptidergic nociceptors sense noxious mechanical pain, 49 though some may be polymodal with the ability to encode an itch response to chemical irritants. Members of the Mrg family of proteins are emerging as receptors for specific nonhistamine pruritogens like chloroquine (MrgA3 66 ), while a more general marker of pruriceptive “itch” neurons is the colocalized expression of both natriuretic polypeptide B and somatostatin. 67 These itch-related proteins are primarily expressed by nonpeptidergic nociceptors, although a few itch-related neuronal subtypes, or pruriceptors, do not encode pain and are dedicated only to the sense of itch. Gastrin-releasing peptide can also contribute to itch, but its expression in DRG neurons has been called into question. 68,69 Cold thermoreceptors are nonpeptidergic A-delta and C fibers that express transient receptor potential cation channel subfamily M member 8 (TrpM8) and mediate the sensation of cool temperatures and menthol. 70
Mechanoreceptors, in contrast to nociceptors, are less well understood due to the historical challenges of identifying LTMR subtypes in tissue sections. Over time, several molecular markers have been validated to distinguish broad subsets of the LTMR subtypes described below. Immunostaining for the 200 kD neurofilament “heavy” isoform (NFH or NF200) can distinguish nociceptors and C-LTMRs (both NFH immunonegative) from NFH immunopositive, heavily myelinated sensory subtypes including several A-beta LTMRs, A-delta LTMRs, A-delta nociceptors, and proprioceptors. 3,71 Maf-A is a more selective marker, restricted to LTMRs, and expressed among several if not all LTMR subtypes. 72,73 Mechanosensitive Piezo2 channels are expressed by LTMR subtypes and are critical for touch sensation in the skin. 74 However, Piezo2 is not specific for LTMRs, as it is also expressed in a small subset of nociceptors that also express cholinergic receptor nicotinic alpha 3 subunit and can respond to noxious mechanical stimulation under specific sensitized conditions. 75 An intriguing marker that seems selective for A-beta neurons is Toll-like receptor 5, 76 though it remains unclear which of the 6 different subtypes of A-beta LTMRs express it.
Identification of these molecular markers as well as others with more limited distribution in specific subtypes has aided in better characterization of the distinct functions of mechanoreceptor subtypes. A unique A-beta LTMR subtype innervates each of the specialized touch organs of the dermis, with axon terminals that are often intertwined with terminal Schwann cells or lamellar cells. 5 Rapidly adapting subtype I A-beta LTMRs innervate Meissner (tactile) corpuscles located in dermal papillae of glabrous skin of the finger tips, lips, prehensile tails, and mystacial pad, depending on the species. 3 Rapidly adapting A-beta neurons show activity at both the start and cessation of a continuous mechanical stimulus but are silent in the middle of that stimulus. The A-beta subtype that innervates Meissner corpuscles is thought to contribute to tactile acuity and sensation of fine texture. Rapidly adapting subtype II A-beta LTMRs innervate Pacinian corpuscles (lamellar corpuscles) with the deep dermis of glabrous and haired skin and contribute to deep pressure, vibration, and texture sensation. 3 Ruffini endings (bulbous corpuscles), located between the deep dermis and hypodermis, are innervated by slowly adapting type II A-beta LTMRs that encode stretch and tension of the skin and fascia. 3 However, there is some controversy because Ruffini endings have been difficult to demonstrate in the skin of some species, despite electrophysiologic evidence of slowly adapting type II A-beta LTMRs. 77,78 Merkel cells (Merkel discs) form synaptic contacts with slowly adapting type I A-beta LTMRs (SAI A-beta LTMRs). They are found scattered within the basal layer of the epidermis of glabrous skin while, in haired skin, they cluster around guard hairs in structures called “touch domes.” 3,45 With the assistance of associated Merkel cells, the SAI A-beta LTMRs show continuous, slowly adapting activity in response to skin indentation and help encode sustained pressure and 2-point discrimination. 4,45 Tropomyosin receptor kinase type C (TrkC) has been used to help identify SAI A-beta LTMRs, though it is also expressed by proprioceptors, peptidergic nociceptors, and A-beta field receptors (see below). 45,46,79
The hair follicle functions as a specialized touch organ that is innervated by an assortment of LTMRs, depending on the type of hair. The longitudinal lanceolate endings of LTMRs, interposed with similarly shaped processes of terminal Schwann cells, are aligned at the basal lamina of the outer root sheath of the hair follicle, such that movement of the follicle is tightly linked to mechanical movement of the axon terminal cell membrane. 80 This cell membrane movement activates mechanosensitive proteins such as Piezo2 channels. 74 Another type of axon terminal that innervates the hair follicle is the circumferential ending, which encircles hair follicles and longitudinal lanceolate axon terminals. Circumferential endings are not sensitive to hair deflection; rather, they help encode skin indentation and hair pull.
There are several different varieties of hairs in mammalian skin including guard hairs, awl/auchene hairs, and zigzag hairs; each exhibits a distinct complement of sensory innervation. The large-diameter guard hairs are innervated by 3 distinct types of A-beta LTMRs. In addition to SAI A-beta LTMRs that innervate touch domes as mentioned above, deeper in the dermis, the guard hair follicle is innervated by longitudinal lanceolate endings of rapidly adapting A-beta LTMRs (RA A-beta LTMRs), a subtype that is distinct from the A-beta neurons that innervate glabrous skin. In the haired skin, RA A-beta neurons encode the sensation of hair deflection, the velocity of dynamic touch, and air puff or breeze. 4,45 These neurons express Ret during E9-E12 of embryogenesis, after which time Ret becomes a less selective marker. 4,45 Rapidly adapting A-beta LTMRs also innervate awl/auchene hairs. The third type of A-beta neuron that innervates guard hairs are A-beta field receptors, aptly named because a single neuron of this subtype has a wide receptive field, innervating up to 180 hair follicles, compared to 5 to 40 follicles per RA A-beta neurons and 1 follicle per SAI A-beta neuron. 46 A-beta field receptors innervate every type of hair follicle: guard hairs, awl/auchene hairs, and zigzag hairs. A-beta field receptors form weakly mechanosensitive circumferential endings such that there is summation in response to a stimulus moving across the hairs of the receptive field, encoding stroking of the skin. Molecular markers that identify A-beta field receptors in mice include the combination of TrkC expression with Ret expression after postnatal day 5.
A-delta LTMRs (previously termed D-hair nerve fibers) have longitudinal lanceolate endings with a unique, polarized innervation of the caudal aspect of the hair follicle. In adult animals, markers such as TrkB 45 and the voltage-gated calcium channel 3.2 (CaV 3.2 81 ) can help identify A-delta-LTMRs. Interestingly, brain-derived neurotrophic factor produced on the caudal aspect of the developing follicle of awl/auchene and zigzag hairs binds to TrkB and directs the unique patterning of A-delta LTMRs. 44 This enables A-delta LTMRs to encode the directional sensitivity of hair follicle movement, showing the most sensitivity to deflection of hair in the cranial direction, against the direction of hair growth.
C-LTMRs innervate awl/auchene and zigzag hairs with longitudinal lanceolate endings. In mice older than postnatal day 13, C-LTMRs express tyrosine hydroxylase (TH), the synthetic enzyme for the molecule dihydroxyphenylalanine (DOPA). 45 This is noteworthy in light of the frequent use of TH immunostaining to identify sympathetic motor neurons, as TH-positive axon terminals may include C-LTMR skin afferents 4,45 or TH-positive visceral afferents. 82 C-LTMRs respond to hair deflection and thereby contribute to the sensation of breeze and the affective component of stroking the skin, for example, the pleasant sensation of caress or petting. 4,83
Nociceptor and mechanoreceptor projections to the CNS terminate predominantly in the spinal cord where axon terminals are distributed within particular layers, or laminae, of the dorsal horn. The dorsal horn also has a somatotopic organization with medially projecting axon terminals that innervate distal limb afferents and laterally projecting axon terminals that innervate more proximal structures. The superficial dorsal horn contains the central axon terminals of peptidergic nociceptors (lamina I and outer lamina II), nonpeptidergic nociceptors (lamina II), and C-LTMRs (lamina II; Figures 1B and 2). A-delta LTMRs terminate in the mid dorsal horn (lamina II-III), while A-beta LTMRs terminate in the deeper dorsal horn (lamina III through V). Protein kinase C gamma labels deep lamina II spinal cord interneurons, serving as a useful structural marker that divides superficial lamina from deep lamina of the dorsal horn (Figure 1B). Most somatosensory neurons enter the dorsal horn at the level of the spinal cord that corresponds to the DRG where their cell body is found (though one must keep in mind the relative caudal movement of both vertebrae and DRG as the spine lengthens during growth of the animal). Studies in transgenic mice with genetic labeling of nonpeptidergic nociceptors has revealed that about ∼25% of nociceptive afferents enter the dorsal horn 1 to 2 segments away from their DRG. 84,85 A-beta LTMR subtypes (and proprioceptors, 86 see Table 2) have a more expansive longitudinal distribution of central axon terminals. The central axons of A-beta LTMRs bifurcate before entering the dorsal horn, forming 2 branches that extend rostrally and caudally in the ipsilateral dorsal column medial lemniscus (DCML) white matter tracts. Collaterals sprout off of the rostral and caudal branches to enter the dorsal horn at many spinal levels, while the rostral branch continues to the ipsilateral brain stem. Lumbosacral and caudal thoracic A-beta LTMRs terminate in the gracile nucleus (GN), while cranial thoracic and cervical A-beta LTMRs terminate in the cuneate nucleus (CN) of the medulla. 45,46
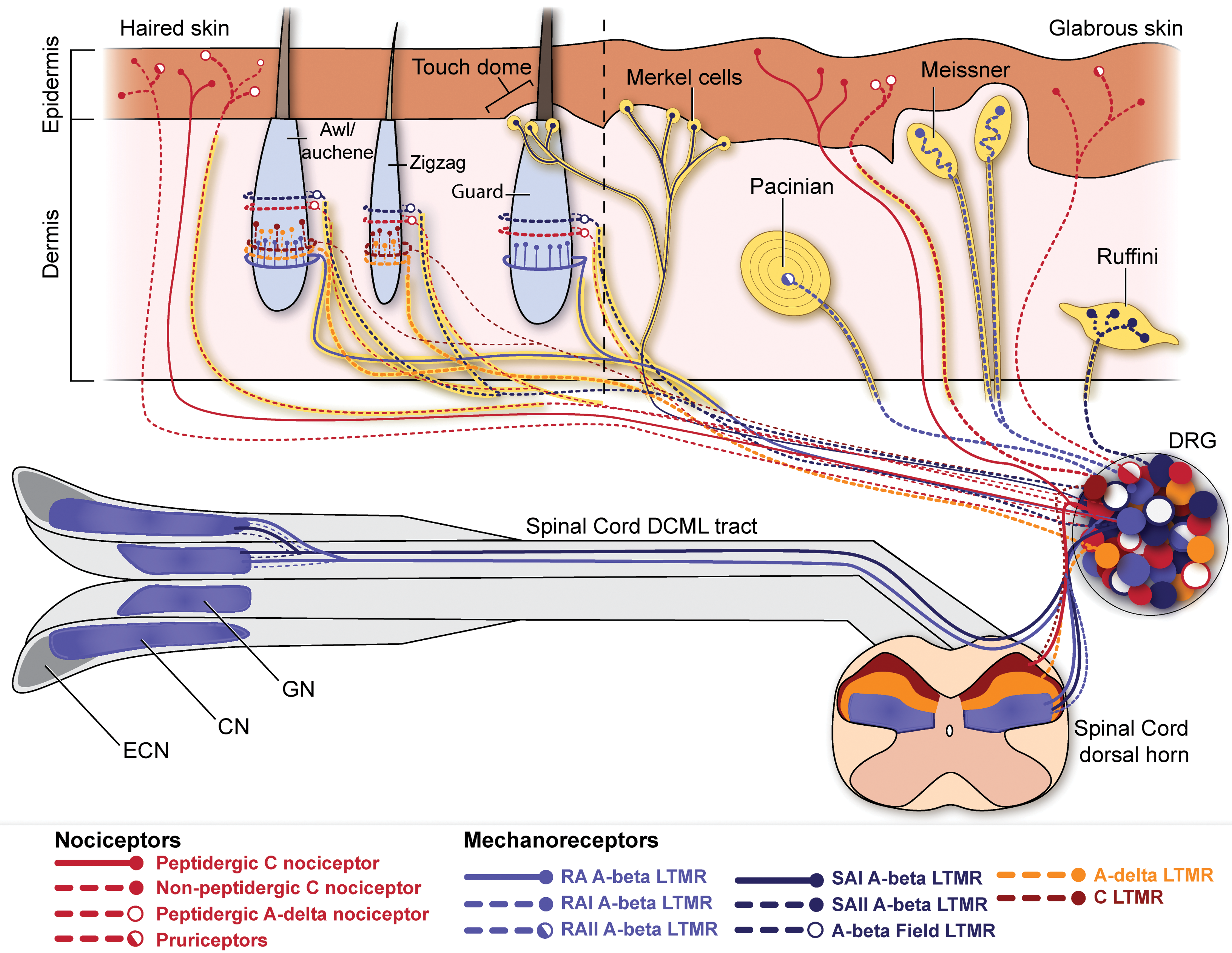
Sensory afferents of the haired and glabrous skin. The distinct patterns of skin innervation by sensory neuron subtypes demonstrate the nuanced heterogeneity of the somatosensory nervous system. The majority of peripheral axons of nociceptors and pruriceptors form free nerve endings in the epidermis, while low-threshold mechanoreceptors (LTMRs) innervate specialized touch organs or hair follicles within the dermis or dermal–epidermal junction. Two distinct subtypes of rapidly adapting A-beta LTMRs (RA A-beta) innervate Meissner corpuscles and Pacinian corpuscles while a third type forms longitudinal lanceolate endings around the hair follicle of large guard hairs and awl/auchene hairs. Two distinct types of slowly adapting A-beta neurons (SA A-beta LTMRs) innervate Ruffini endings and Merkel cells. Merkel cells are found in the basal layer of the epidermis, scattered individually in the glabrous skin and clustered in groups of 10 to 20 around guard hair follicles in haired skin. A-beta field receptors form circumferential endings around the follicles of every type of hair in the mouse skin. A-delta LTMRs and C-LTMRs form longitudinal lanceolate endings around the follicles of awl/auchene hairs and the other type of smaller hairs, zigzag hairs. The central axons of sensory neurons likewise exhibit subtype-specific patterns. All nociceptors and C-LTMRs terminate in the superficial laminae of the spinal cord dorsal horn. Adelta-LTMRs terminate slightly deeper in lamina III of the dorsal horn. All A-beta LTMRs have axon terminals in the deep laminae of the dorsal horn as well as a collateral branches that descend (not shown) and ascend the spinal cord to terminate in the dorsal horn rostral and caudal to the DRG. Ascending collaterals of A-beta neurons terminate in the gracile nucleus (GN) and cuneate nucleus (CN). The external cuneate nucleus (ECN) contains axon terminals of A-alpha and A-beta proprioceptors that innervate muscle (not shown). DRG indicates dorsal root ganglion.
Structural and Neurochemical Responses to Injury in Sensory Neurons
Features of Sensory Pathology Detected in the CNS
While investigators have uncovered many molecular and cellular mechanisms for sensation under healthy conditions, relatively less is known about the pathological changes that occur in sensory subtypes after injury or disease. Much of the sensory pathology that has been unearthed in histopathology studies and in neuroscience research has focused on the CNS, as it is sampled more routinely. The mechanisms underlying subtype-selective pathology are poorly understood, but some insight can be gained from examination of primary afferents axon terminals in the spinal cord dorsal horn, DCML tract, and medulla. Immunolabeling in mouse models of traumatic nerve injury demonstrated unilateral regional loss of spinal cord axon terminals of nonpeptidergic nociceptors, but not peptidergic nociceptors. 87,88 After nerve injury, A-beta LTMRs acquire ectopic neuropeptide Y (NPY) expression that can be seen in the corresponding brain stem nuclei. 89 -91 In the brain stem, the GN, CN, and external cuneate nucleus (ECN, also termed accessory CN or lateral CN) contain axon terminals of A-beta LTMRs from the hind limb, A-beta LTMRs from the forelimb, and proprioceptors from the forelimb, respectively. In fact, there are reports of spheroids, axonal dystrophy, or axonal degeneration within these dorsal column nuclei in many species. These lesions are considered an incidental or age-related change when present in low numbers, 92,93 but can increase in disease conditions that target sensory neurons. 94 -96 The presence of spheroids in the dorsal column nuclei of the brain stem but not the dorsal horn suggests pathology that is selective for A-beta LTMRs and proprioceptors, but spares nociceptors and A-delta LTMRs. When samples are collected in accordance with Society for Toxicologic Pathology best practices, level 7 sections of the mouse brain are likely to include the CN and ECN and the rostral GN. 97,98 For other sectioning protocols or other larger species, coronal brain sections that include the mid cerebellum and vestibular nuclei may include the rostral aspect of the ECN, but may be too rostral for assessment of the GN and CN. 97,98 In those cases, specialized sections of the caudal medulla may be required to assess the GN and CN.
One example that demonstrates the utility of CNS assessment of sensory neuron pathology comes from the description of increased number of spheroids in the spinal cord white matter and brain stem as part of the neurodegenerative disease, equine degenerative myeloencephalopathy (EDM). 96 Spheroids within the spinal cord and brain stem dorsal column nuclei suggests degeneration of both proprioceptors and A-beta LTMRs, and one study has successfully linked the spheroids to sensory neurons originating from the DRGs. 96 This work suggests the intriguing idea that EDM is largely a sensory peripheral neuropathy. A corollary of this hypothesis is that these dorsal column spheroids could be examples of distal axonal degeneration given the relative length of the A-beta LTMRs: The peripheral branch of A-beta LTMR extends from the skin and is continuous with the central branch that reaches the medulla. An A-beta neuron that extends from the tip of the toe to the brain stem GN may have the longest axon in the body in most species.
Features of Sensory Pathology Detected in Peripheral Axons
The sciatic nerve provides another opportunity to evaluate sensory axons, though the large number of motor neurons may obfuscate changes limited to particular sensory subtypes. The sural nerve is primarily sensory and may thus better highlight changes that are restricted to sensory neuron subtypes. Unfortunately, neither the sciatic nor sural nerves are ideal for assessing neuropathies that are limited to the distal aspect of the longest axons, as are common among toxic neuropathies as well as clinical entities such as diabetic peripheral neuropathies, HIV-associated neuropathy, and chemotherapy-induced peripheral neuropathy. These distal polyneuropathies typically present with a “glove-and-stocking” distribution of sensory deficits or neuropathic pain beginning with the skin of the distal extremities and sternum. As such, peripheral axon terminals in the skin present an alluring window into the sensory nervous system. In fact, the evaluation of sensory axon terminals in skin biopsies from the ankle has become a validated component of the diagnosis of pain associated with human diabetic peripheral neuropathy and other peripheral sensory neuropathies such as familial amyloid polyneuropathy 99 and HIV sensory neuropathy. 100 Using immunolabeling of the pan-neuronal marker protein gene product 9.5 (PGP 9.5), these biopsies are evaluated for a loss of intraepidermal nerve fibers (IENF; ie, free nerve endings of nociceptors) that has been linked to neuropathic pain. Skin innervation patterns remain relatively unexplored in veterinary diagnostics but is gaining ground in translational pain research. For example, in a model of HIV-associated neuropathy, a loss of IENF in glabrous skin has been reported in SIV-infected macaques, 101,102 along with a loss of trigeminal sensory innervation of the cornea. 103 However, the relationship between skin innervation patterns and pain must be interpreted with some knowledge of the underlying biological process. There is likewise a loss of IENF with deafferentation following axotomy or toxic ablation of peptidergic nerve fibers with resiniferatoxin, 104,105 procedures that lead to sensory deficits rather than pain or hypersensitivity. In addition, studies of nerve regeneration following injury have linked restored nociception to increased nerve sprouting from uninjured afferents, resulting in increased IENF density. 106 Unfortunately, LTMR nerve terminals are found primarily in the dermis, rather than the epidermis, and thus are not assessed in IENF studies. As a consequence, there is a gap in our knowledge of mechanoreceptor pathology and the potential role of LTMRs in sensory deficits or pain. Albeit relatively unexplored, the pathology of haired skin innervation is particularly intriguing given that mechanical allodynia is often described in human patients as pain resulting from a light, moving stroke of the haired skin. Further validation of histologic changes, perhaps in combination with ancillary studies, is needed to interpret altered sensory nerve terminals in the skin and other end organs.
Features of Sensory Pathology Detected in the DRG
Because DRGs are not included in the routine autopsy of any species, there is a significant gap in our knowledge of the histopathologic and molecular changes that occur in sensory neurons in disease. Vacuoles within neuronal somata have been reported as a background lesion and may increase in some pathologic conditions in conjunction with axonal damage. 107 -109 The functional consequences of these vacuoles are poorly understood. Translational pain research has yielded a great deal of insight into sensory neuron pathology, as many cellular and molecular changes have been identified in the DRG in rodent models of nerve injury pain. Nerve injury induces the ectopic expression of several nociceptor-associated markers in non-nociceptive, large-diameter neurons, a phenomenon termed “phenotype switching.” Nociceptive markers including TrpV1, CGRP, and substance P have all demonstrated an increased expression in large-diameter, putative mechanoreceptors after injury. 110 -113 Another neuropeptide, NPY, is not expressed in the DRG under normal conditions but is highly expressed by injured A-beta mechanoreceptors after injury. 90,91 We are just beginning to understand the functional consequence of the ectopic expression of these nociceptive markers. It is not yet known, for example, what controls the release of neuropeptides that are ectopically expressed after injury. It has also not yet been established whether newly acquired expression of nociceptive markers renders those neurons susceptible to pharmacologic targeting of nociceptive pathways, as has been suggested by some studies of CGRP expression. 111
Other changes have been reported in DRG neurons after nerve injury and in other models of pain. Activating transcription factor 3 has emerged as a nuclear marker of injured, yet viable, sensory neurons. 114 -116 There is also an increase in the expression of gap junctions that leads to increased electrical coupling across satellite glia as well as increased neuron-glia and neuron–neuron coupling in rodent models of inflammatory pain, neuropathic pain, and visceral pain. 117 -120 A stimulus that excites a single neuron can thus lead to a spread of activity across surrounding satellite glia and adjacent neurons. Nerve injury also induces alterations in the pattern of IB4 binding including reduced IB4 staining of neurons and increased IB4 staining of blood vessels, 121 as well as the formation of ring structures around large-diameter neurons that include microglia/macrophage lineage cells, sympathetic axons that sprout from blood vessels, and IB4-labeled axons of putative nonpeptidergic nociceptors in addition to resident satellite glia. 121 -123 Cholera toxin B, often used as a neuronal tracer, typically demonstrates selective uptake by mechanoreceptors in the somatosensory system; however, it is less selective after nerve injury with uptake by nociceptors as well. 124 Although the mechanism for this altered pattern of uptake has not been identified, it suggests that injury may induce molecular changes in some cell types that alter their susceptibility to toxins or their binding by lectins like IB4.
Although many of these changes have been reported in injured neurons, it is important to note that uninjured, bystander neurons with axons or cell bodies adjacent to injured neurons can also exhibit pathologic changes. In cases of partial axotomy, where only a portion of skin innervation is lost, the uninjured nerves that still innervate the skin are responsible for transmission of stimulus-driven messages that ultimately evoke a pain response. Uninjured neurons can exhibit spontaneous electrical activity and phenotype switching and they and form over half of the population of mechanoreceptors that are encircled by rings of sprouting axons and microglia-like macrophages following injury. 123,125 -127
Behavioral Assays to Elucidate Sensory Pathology
Although it is tempting to draw conclusions about pain or sensory deficits based on molecular changes in sensory neurons, the functional significance of many injury-induced changes remains poorly understood. One way to validate potential histomorphologic and neurochemical alterations in primary afferents is to correlate those changes with behavioral readouts of sensory dysfunction. The standard battery of preclinical tests that may include IMPC, SHIRPA, and the Combined SHIRPA Dysmorphology protocols 128 -130 can offer limited sensory information if they include proprioceptive responses, ear twitch, whisker response, or palpebral reflex. More in-depth sensory information might be garnered from the use of behavioral tests that have been honed by pain research labs. The gold standards of functional assessment of allodynia and hyperalgesia include quantifying withdrawal responses to stimulation of the glabrous skin of the plantar hind paw, typically with the use of mechanical stimuli (eg, von Frey filament test) or thermal stimuli (eg, hot plate or radiant paw heating test). In addition, neuroscientists have continued to invent and optimize behavioral assays that detect modality-specific sensory deficits and hypersensitivities, including the challenging assessment of light touch sensation and sensation in haired skin. These assays quantitate behavioral responses to cold stimuli, responses to moving tactile stimuli (eg, dynamic brush assay), texture discrimination, vibratory sensation, responses to tactile stimuli applied to the facial region, and spontaneous pain behaviors (e.g. conditioned place preference test). The behavioral responses measured in each of these tests has a unique underlying biology that may be differentially affected by toxin, injury, or disease, as well as the cognitive and emotional state of the animal. Assays of sensory deficits are more difficult to develop than assays of sensory gain-of-function, but often utilize some of the same assays to quantitate decreased withdrawal responses in comparison to controls.
Conclusion
Standard diagnostic histopathology protocols and tier I screening protocols only scratch the surface of sensory pathology. The study of skin afferents has yielded insight into the neurobiology of the sensory system. The mechanisms underlying differential changes exhibited by subtypes of skin afferents are poorly understood. However, distinct patterns of pathology across different subtypes highlight the potential role that their defining neurophysiologic and anatomic features may play in selective susceptibility and pathogenesis. Notably, specific subtypes of touch neurons are emerging as early contributors to some types of chronic pain, placing new significance on nerve terminals within the dermis and the distinct innervation of haired skin. This enhanced understanding may help produce strategies such as selective blockade of neural activity through the direct molecular targeting of touch neurons. 76 A better understanding of interneuronal communication among sensory subtypes will shed light on the relative role of injured and uninjured bystander neurons in generating pathologic phenotypes. The neurobiology and plasticity of the DRG itself is of growing interest, particularly given that sensory neuron cell bodies within the DRG are readily exposed to the systemic circulation. This has implications not only for understanding toxicopathology and the effects of systemic disease but also for the development of novel therapeutics that need not cross the blood–brain barrier. An improved understanding of sensory neurobiology pathology can be leveraged to enhance a wide range of discovery and pre-clinical studies, advancing the fields of toxicologic pathology and translational pain research alike.
Footnotes
Acknowledgments
The authors would like to thank Catherine Stanley for her artistic input that helped to generate a diagram for this review article.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: M.J.C. is an inventor on a patent related to TRPV1 that is licensed through UCSF and Merck and he may be entitled to royalties on that patent. This conflict is being managed by the Johns Hopkins Office of Policy Coordination.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
