Abstract
Many preclinical investigations limit the evaluation of the peripheral nervous system (PNS) to paraffin-embedded sections/hematoxylin and eosin–stained sections of the sciatic nerve. This limitation ignores several key mechanisms of toxicity and anatomic differences that may interfere with an accurate assessment of test article effects on the neurons/neurites peripheral to the brain and spinal cord. Ganglion neurons may be exposed to higher concentrations of the test article as compared to neurons in the brain or spinal cord due to differences in capillary permeability. Many peripheral neuropathies are length-dependent, meaning distal nerves may show morphological changes before they are evident in the mid-sciatic nerve. Paraffin-embedded nerves are not optimal to assess myelin changes, notably those leading to demyelination. Differentiating between axonal or myelin degeneration may not be possible from the examination of paraffin-embedded sections. A sampling strategy more consistent with known mechanisms of toxicity, atraumatic harvest of tissues, optimized fixation, and the use of resin and paraffin-embedded sections will greatly enhance the pathologist’s ability to observe and characterize effects in the PNS.
This review article is a synopsis of information presented during a continuing education symposium (37th annual meeting of the Society of Toxicologic Pathologists, June 2018) on the peripheral nervous system (PNS). 1 As such, the information in this article primarily reflects those things learned (mostly the hard way) by the author over more than 25 years spent evaluating the nervous system. Topics to be covered include fixation, handling, harvest techniques, sample selection, and evaluation. Numerous, previously published sources cover much of this information, 2 –8 and the reader is referred to those sources as a supplement to this article. Each technique should be developed, practiced, and assessed to make sure mistakes and oversights happen before the necropsy of a regulatory study. As with nearly all things, the most important concept governing the harvest, handling, fixation, processing, and staining of nervous system tissues is to figure out what works best in your particular laboratory. There is more than one acceptable way to perform an intravascular perfusion, to fix tissues, to remove the spinal cord, and to embed and section ganglia and nerves. The work might be performed in-house or might be outsourced (in part or in entirety) to another laboratory which has already developed the techniques and/or has more experience in this specialized area. The best way is the way that works best—for each pathologist, the technicians working with that pathologist, and in general the testing facility/test site performing the work. Two quotations seemingly written to warn about the pitfalls regarding the PNS are: “Failing to plan is the same as planning to fail” (generally attributed to Benjamin Franklin) and “Good judgement comes from experience, but experience comes from bad judgement,” generally ascribed to just about everyone.
General Considerations
There is 1 nervous system, but the brain and spinal cord are often referred to as the central nervous system (CNS) and structures peripheral to the brain and spinal cord as the PNS. Pathologists should always think in terms of a single nervous system so some mention of the brain and spinal cord are included herein. The evaluation of the nervous system should provide study directors, regulatory personnel, clinicians, and eventually patients’ a degree of confidence that reasonable attempts were made to conduct a comprehensive assessment.
The brain and spinal cord have unique cell types, barriers, and anatomic arrangements (eg, they are encased in bone) because evolution dictated it was more important to protect these areas from injury than devote resources for efficient repair. Nervous system structures sitting outside bony enclosures are more susceptible to injury (both physical and partially, chemical), but they tend to repair more efficiently. It is important to note that “repair” in the nervous system essentially means repair (regeneration) of axons and/or myelin (white matter in the CNS) or the formation of new neuronal connections. 9 Neurogenesis may occur in some parts of the brain such as the hippocampus, 10 but for the purposes of repair in most morphological evaluations, loss of neurons is not going to resolve during the recovery period of a study. If a neuron dies, the loss includes the axons and dendrites and all the synapses/connections of those structures. Axons may span the CNS and PNS. Sensory neurons in dorsal ganglia have axons that may extend from the periphery, through the dorsal funiculi of the spinal cord, and onto the brain. In other words, there may be no such thing as a purely peripheral neuropathy. Apparent repair/recovery of damaged axons/myelin in the PNS during a dosing-free period may not, and probably does not, include repair of the central projections. 11 Axons from sensory neurons situated in dorsal root ganglia may appear to repair or recover or regenerate during a dosing-free period, but the central projection of that axon is unlikely to undergo anything resembling an effective repair.
Timing and Schedules
Processing and evaluating tissues from the PNS takes time, perhaps more time than sponsors would like for any given study. It is incumbent on the pathologist to make sure sufficient time is allowed for an optimal evaluation. Failure to do so threatens future patients and diminishes the scientific value of the profession of toxicological pathology. The pathology evaluation is a medical evaluation; it is not a technical exercise. Delicate dissections, handling, embedding, specialized staining, and evaluation of the nervous system tissues may take longer than typically would occur with the standard processing and evaluation of guideline-specified tissues. Quantitative investigations (intraepidermal nerve fiber density, neuron counts, other measures, etc) usually require that specimens across all groups be taken to stained slide, even if the evaluation will be performed in a stepwise fashion. Even small variations in fixation time, processing runs, laboratory conditions, and involved personnel can sufficiently alter the morphology of structures such that comparison of counts/measurements between groups is affected. Much of what is observed in the nervous system tissues that can easily be mistaken as an abnormality is, in fact, an artifact or a normal structure. Whatever can be done to mitigate the possible impact of the presence of artifacts in the various animals/groups should be done. This includes rigid attention to performing all activities, from necropsy to stained slide, across dose groups whenever possible.
Necropsy Room Versus the Histology Laboratory
The brain is typically removed at necropsy, although in studies where the animal is fixed using intravascular perfusion, it may be advantageous (especially to decrease dark neurons 12 ) to leave the brain in the skull (in fixative/refrigerated) for 24 hours before extraction. Removal of the spinal cord is a difficult, time-consuming process in any species, and a process that at many laboratories is best performed in the histology laboratory. It is the nature of the necropsy room to keep things moving, and unless dedicated personnel are present (especially for perfusion-fixed animals), it may be best to harvest the spinal column, then do the fine dissections under the typically more controlled environment of the histology laboratory.
Pathologists and technicians are trained to perform necropsies efficiently but also in a habitual manner, meaning any departure from those habits will require training and/or frequent reminders. Removal of the head from the C1 vertebrae is an example. A study where the cranial (superior) cervical ganglion (CCG) requires evaluation will necessitate leaving the head/neck arrangement intact, as removal of the head is very likely to either damage the ganglia or sufficiently distort the anatomy that finding the ganglion (intact) may be impossible.
Fixation
For most nonclinical studies, initial fixation of the nervous system tissues involves either immersion or intravascular perfusion using solutions of aldehyde-based fixatives. The most commonly used aldehyde fixatives are formaldehyde (HCHO) and glutaraldehyde. Ten percent neutral-buffered formalin (NBF) is a 10% dilution of 37% to 40% HCHO, and thus it is approximately 3.7% to 4% HCHO (concentrations vary somewhat). 13 Methanol is added to formalin to prevent the polymerization of HCHO into a solid form (paraformaldehyde). Methanol-free HCHO is usually prepared from paraformaldehyde powder. So although it is a commonly used terminology, there is no such thing as 4% paraformaldehyde: What is produced from paraformaldehyde powder is a solution in which the solute is 4% methanol-free HCHO. 14 Formaldehyde prepared from paraformaldehyde powder should be used within a few days, as the polymerization process starts somewhat rapidly. The use of HCHO prepared from paraformaldehyde powder is common in studies where nervous system tissues are being fixed. The reason for this is that the methanol in formalin is thought to be deleterious to myelin sheaths. This concern may be overrated, and each laboratory should come to their own conclusions. The use of 10% NBF instead of HCHO prepared from paraformaldehyde powder has the obvious advantages of being cheaper (as formalin is readily available in whatever quantities are needed) and/or less labor intensive to produce (since formalin is typically purchased in bulk). Formaldehyde (single aldehyde group) is a gas dissolved in solution. This small molecule in solution penetrates tissue rapidly, forming tissue bonds that result in excellent preservation of proteins but somewhat less suitable fixation of lipids. 15 Though penetration is rapid, reaction with proteins occurs somewhat slowly, 14 meaning it takes several days for immersion fixation to occur. Intravascular perfusion greatly reduces the distances fixatives have to penetrate to fix tissue so the process of protein stabilization can begin essentially immediately.
Glutaraldehyde (2 aldehyde groups) reacts quickly with protein but has several disadvantages. This fixative is slower (as compared to HCHO) to penetrate tissues, may interfere with paraffin penetration, and prolonged fixation at concentrations >2% may harden tissues such that suitable paraffin sections are nearly unattainable. Glutaraldehyde fixation may also interfere with immunohistochemical procedures and may impart some degree of autofluorescence to the tissues.
Osmium tetroxide (OT; usually used as a 1% or 2% solution) fixes proteins, but its main useful property is its superior (as compared to aldehyde fixatives) fixation of lipids including cell membranes/myelin. Osmication of tissues is necessary to provide optimal contrast when examining various tissues with transmission electron microscopy (TEM). As compared to paraffin embedded, formalin fixed specimens, the evaluation of OT fixed, resin embedded, toluidine blue-stained nerves will greatly enhance the evaluation of myelin. The use of OT is not compatible with glycol methacrylate as an embedding media, so use of a harder plastic/resin is necessary. Luxol fast blue may be a useful adjunct to paraffin-embedded, hematoxylin and eosin (PEHE)–stained sections, but it is not an optimal substitute for osmium fixation of nerve, brain, ganglia, or spinal cord. Osmium tetroxide is always limited to immersion fixation. Penetration is slow, and tissues immersed in osmium should be no larger than 2 mm in at least 1 direction, meaning specimens of larger nerves should be very gently cleared of surrounding soft tissue, and brain/spinal cord specimens should be no thicker than 2 mm. Osmium tetroxide is classified by the Environmental Protection Agency (EPA) as an acute hazardous waste, 16 meaning (in some states) generation of more than 1 L of OT per month classifies a laboratory as a large capacity generator, with all the regulations, permits, and stipulations that accompany that distinction.
In practical terms: Base your choice of fixatives/fixative methods on prior experience that provided optimal sections for your goals. If it is not prevented from other study end points (such as harvest of fresh tissue for various assays), use intravascular perfusion (generally with an initial saline ± heparin ± sodium nitrite [vasodilator] preflush) with an aldehyde-based fixative to enhance rapid fixation of tissues throughout the nervous system. If an end point does prevent intravascular fixation, consider perfusion with ice-cold buffered saline. Most studies with such end points involve some immediate sectioning of the brain, and this process will assist with making the brain easier (firmer) to slice fresh and at the same time, slow the process of autolysis providing the tissues for morphological evaluation are immersion fixed as quickly as possible. Use a 4% HCHO solution (10% NBF or 4% methanol-free HCHO) for intravascular perfusion or (if the inclusion of glutaraldehyde will not cause problems with later procedures) a combination of a HCHO/glutaraldehyde solution. Common combinations are 4% HCHO /1% glutaraldehyde
17
(McDowell-Trump’s solution) and 2% methanol-free HCHO/2.5% medical grade glutaraldehyde (half strength Karnosvky’s solution). The use of glutaraldehyde will make the perfused carcass rigid which may slow necropsy. Generally, for nonclinical studies, the use of HCHO alone as the perfusion fixative is sufficient. Use a 2.5% to 4% solution of glutaraldehyde for immersion fixation of smaller/thinner tissues (nerves, ganglia, small sections of brain/spinal cord) if producing resin sections or preparing the tissues for TEM. Glutaraldehyde solutions generally have lower osmotic concentrations than HCHO solutions, and this may assist with membrane preservation. Use a 1% to 2% solution of OT as a post-immersion fixative for nerves to be resin embedded.
One published regimen 18 for the fixation of sural nerve biopsies is:
Place straightened nerve on a wooden stick and immerse in cooled 2.5% glutaraldehyde in 0.025 M cacodylate buffer at pH 7.4 and osmolarity between 300 and 330 for 24 hours. Post-fix in 1% OT (pH 7.4) 3 to 5 hours (room temperature), then process through resin.
Nerves being immersion fixed stick rather well to index cards or to a tongue depressor cut to fit standard size cassettes, and straight nerves are easier to clear of fat and trim for transverse sections. Perfusion-fixed nerves may remain straight without this extra step.
Regardless of the fixative used, the solution should be an appropriate pH (approximately physiologic; 6.8-7.4) and osmotic concentration (osmolarity; osmoles per liter of solution; an osmole is the number of moles of solute contributing to the osmotic pressure of a solution). For the diluted fixatives primarily used in pathology, osmolarity is essentially equal to osmolality (osmoles of solute per kilogram of solvent). The best means to determine osmolarity is to have the supplier provide the information, determine the osmolality using an osmometer (there are various methodologies) and consider this value to be essentially equal to the osmolarity or calculate the value. Because of the generally small numbers, molarity is typically expressed as milliosmoles (mOsm)/liter (L). Hypertonicity facilitates tissue penetration. Isotonic saline is approximately 250 to 350 mOsm/L. The osmolarity of 10% NBF varies but can be as high as 1300 mOsmol/L. 19 Because it is membranes that are most likely to be damaged by excessively hypertonic fixatives, use of fixatives that are only mildly hypertonic (400-600 mOsm) is best when tissues are to be examined using electron microscopy. 19,20
Tissue Harvest and Handling
Handling of the unfixed brain leads to an increase in the artifact known as the dark neurons. 12,21 For animals that are fixed via intravascular perfusion, delaying removal of the brain for 24 hours should decrease the dark neuron artifact. Extraction of the brain should be performed carefully and without loss of the olfactory bulbs. The hypothalamus region is often damaged, especially in monkeys, when the brain is removed and detaches from the pituitary gland. This separation should be performed using a sharp blade, and no prying of the brain out of the cranial cavity should take place.
Removal of the spinal cord, dorsal root ganglia, and spinal nerve roots is always time-consuming and tedious. Some laboratories prefer to remove the spinal cord with the spinal nerve roots and ganglia intact; others prefer to remove these separately. In the cervical and thoracic spinal cord, the vertebrae tend to correlate to the spinal cord segments: the spinal nerve roots directly exit the spinal canal through the immediately adjacent intervertebral foramen. While there is some interspecies variability, the lumbar and sacral spinal cord segments tend to be more cranial than their corresponding vertebrae, meaning the spinal nerve roots arising from these regions of the spinal cord traverse a variable distance caudally through the spinal canal before exiting. If maintaining this anatomic arrangement is important, the cord must be harvested with the spinal nerve roots/ganglia attached or careful dissection and labeling should ensure an accurate recreation of the in-life anatomic relationships. Necropsy and laboratory personnel must possess an understanding of this anatomy, as an epidural injection targeting, for example, the L4 dorsal spinal nerve root may be associated with spinal cord changes in the corresponding L4 spinal cord segment, which may be within the confines of lumbar vertebrae L1 to L2.
Removal of ganglia requires accurate dissection techniques and sufficient visualization of the ganglion to have confidence the entire structure has been removed. This is especially true for postsynaptic sympathetic ganglia, some of which are formed by the fusion of multiple ganglia. In any given animal, sympathetic ganglia may be round, oval, stellate, or hourglass-shaped depending on the manner and completeness of the fusion.
Harvest of nerves requires that no part of the nerve to be examined is damaged during harvest or removal of excess fat. If needed, the nerve should be harvested and labeled in a manner that allows a distinction between proximal and distal.
Tissue-Specific Considerations
Spinal Cord
The spinal cord should either be removed from the spinal column at necropsy (by a dedicated, non-time pressed, and trained technician) or in the histology laboratory. Considerable artifactual changes (mostly vacuolation) in the spinal cord may occur if the cord is left in the vertebral column while being fixed, although it is recognized this may be unavoidable due to practical constraints. Intravascular perfusion obviously makes this issue moot. Spinal cord evaluation should include at least 3 levels (cervical, thoracic, and lumbar) and perhaps more. For example, the cervical spinal cord might require 2 or more levels for optimal evaluation: through the cervical intumescence and a very cranial segment (C1/C2) to include the greatest extent of the white matter tracts coursing to/from the brain. Unless there is an implanted device and/or epidural/intrathecal delivery, each spinal cord level should be examined using (at minimum) a transverse and an oblique section. Oblique sections provide increased sensitivity (as compared to transverse sections) for observing subtle degeneration of axons and/or myelin sheaths. Longitudinal sections, unless the plan is to do multiple step sections through the thickness of the spinal cord, will miss some white tracts and neuronal areas. This completely defeats the reason for doing a section along the long axis of the spinal cord. Oblique sections are not perfect because they do not exactly parallel the course of most axons, but the benefit of having all gray columns and white matter tracts overrides this deficit. Figure 1 shows an example of an oblique section from a dog; note the inclusion of all needed anatomic regions. The pathologist may need to assist technical staff regarding how to orient/stabilize the spinal cord for trimming to produce optimal oblique sections.
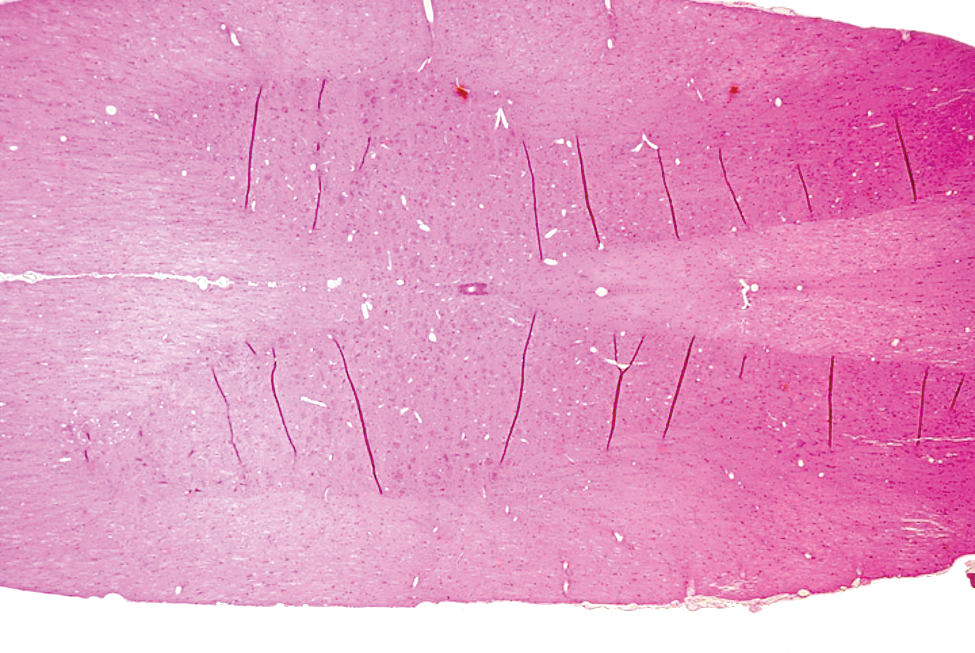
Intravascular perfusion fixation with 10% NBF, paraffin embedding, H&E stain, 5-µ section. Oblique section of the thoracic spinal cord from a canine. Cranial is to the right; caudal is to the left. The ventral (primarily descending), dorsal (primarily ascending), and lateral (ascending and descending) white matter tracts and dorsal and ventral gray matter are present bilaterally. Wrinkles (dark lines) in the gray matter sometimes occur because of the different consistencies of the gray and white matter. H&E indicates hematoxylin and eosin; NBF, neutral-buffered formalin.
If there is an implanted device and/or test article administration in, around, or near the spinal cord, then multiple transverse sections spanning the site of administration should be evaluated. Transverse sections are preferred in this situation because they allow the spinal cord to be examined with the dura (and epidural) intact. In rodents, transverse sections of the spinal cord can be very successfully prepared/examined by decalcifying a portion of the spinal column (Figure 2).
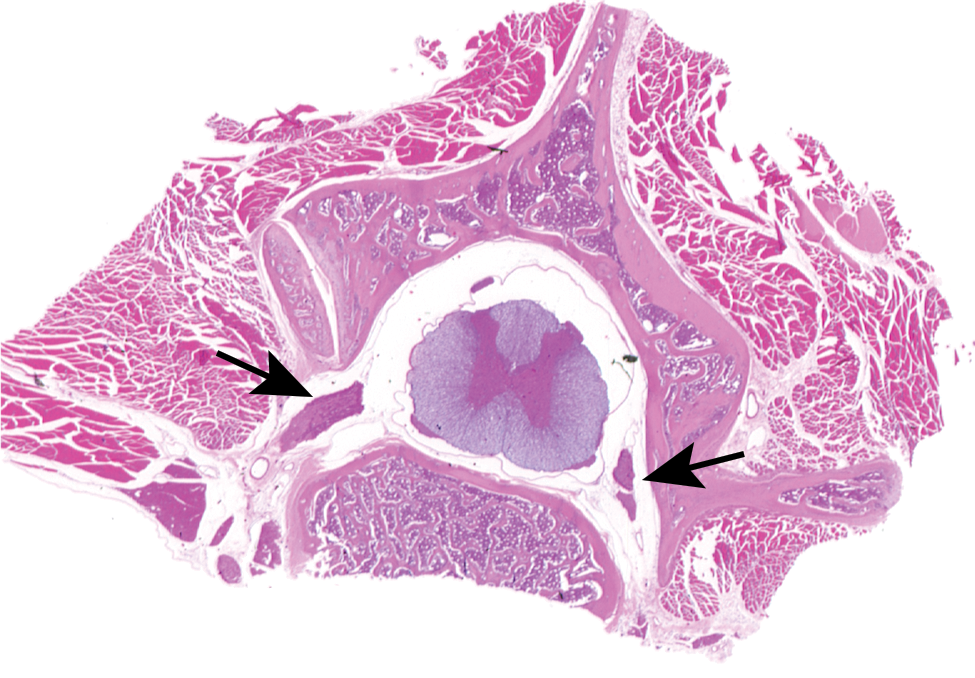
Intravascular perfusion fixation with 10% NBF, paraffin embedding, H&E stain, 5-µ section. Decalcified spinal column/spinal cord from a rat. The quality of the spinal cord sections is unaffected by the decalcification process. A dorsal root ganglion (left arrow) and spinal nerves (right arrow) are within intervertebral foramen. The dura, intrathecal space, and epidural region are readily examined in this type of preparation. H&E indicates hematoxylin and eosin; NBF, neutral-buffered formalin.
For most studies, the spinal cord will be examined using paraffin embedding, although thin slices of the spinal cord are easily post-fixed in osmium and examined as resin sections. When paraffin-embedded, all spinal cord sections should be examined using H&E (for general morphology) and immunohistochemical stains to detect astrocyte and microglial reactions. In particular, microglial reactions can be invisible on H&E staining. It is the opinion of the author that stains selective for neuronal necrosis (such as Fluoro-Jade B) do not increase the sensitivity of the evaluation of spinal cord neurons, as the neurons are arranged into discrete gray matter areas adequately assessed on H&E-stained sections. Other stains that may prove useful are nonselective silver stains (such as Bielschowsky’s) for axons (this stain may assist in identifying axonal degeneration, axonal swellings, or axon loss), Luxol Fast Blue (as a general myelin stain, but this stain is not a suitable substitute for osmicated resin sections), substance P (for detecting alterations in sensory innervation), and Neuronal Nuclear (NeuN) antigen (for identifying/counting neurons).
Spinal Nerve Roots and Dorsal Root Ganglia
Spinal nerve roots are evaluated on specifically harvested tissue (usually dorsal root ganglia with adjacent nerve roots) and also on spinal cord sections. Dorsal roots contain sensory/afferent axons sending information to neurons in the spinal cord and brain. Ventral roots contain efferent/motor axons sending information to muscles and in the thoracic/lumbar regions also contain sympathetic axons from presynaptic neurons. Differentiating dorsal from ventral roots may be important in determining whether or not a neuropathy is predominantly sensory or nonspecific. The pathologist should make an effort to site morphological changes as affecting dorsal and/or ventral nerve roots. The following features (see Figure 3) may assist in differentiating dorsal from ventral spinal nerve roots when the distinction is not obvious.
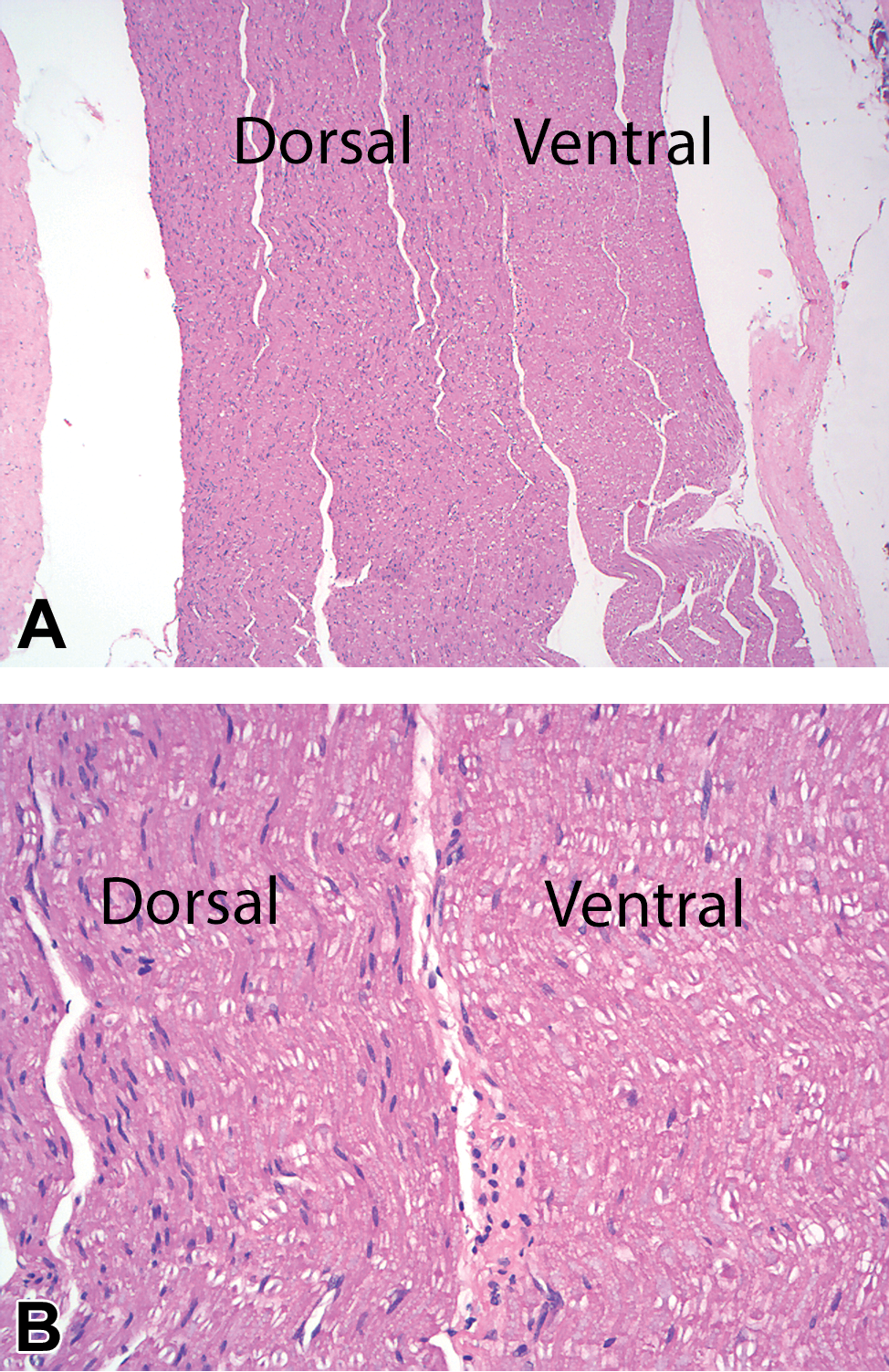
A, Intravascular perfusion with chilled saline followed by immersion fixation with 10% NBF, paraffin embedding, H&E stain, 5-µ section. Spinal nerve roots (lumbar L4) from a cynomolgus monkey. The dorsal root is larger and generally more cellular than the adjacent ventral root. B, Intravascular perfusion with chilled saline followed by immersion fixation with 10% NBF, paraffin embedding, H&E stain, 5-µ section. Spinal nerve roots (lumbar L4) from a cynomolgus monkey. A, Higher power image of the spinal nerve roots. Axons (palely basophilic linear/curvy structures coursing through the nerve) in the ventral root have a more uniform diameter as compared to the dorsal root. In the dorsal root, cellularity is increased due to more Schwann cells supporting more numerous, smaller diameter axons. H&E indicates hematoxylin and eosin; NBF, neutral-buffered formalin.
Size: In the regions of the cervical and lumbar intumescence, dorsal roots from a particular spinal cord segment tend to be larger than ventral roots. In other regions (especially the thoracic), the roots from a particular spinal cord segment are usually similarly sized.
Dorsal roots tend to be slightly more cellular due to the greater percentage of smaller diameter axons and associated Schwann cells.
Ventral roots tend to have a more consistent axon size.
The preparations most useful for evaluating spinal nerve roots and dorsal root ganglia are paraffin-embedded, H&E (PEHE)-stained longitudinal sections that include both roots and ganglia and osmicated, resin-embedded, toluidine blue-stained transverse sections through the ganglion (which will include axons of the dorsal nerve root) and ventral root. Sensory ganglia are seldom listed as required tissues in regulatory guidelines. 22,23 For routes of administration that do not involve direct delivery to the CNS, test article exposure is likely to be higher in PNS neurons as compared to CNS neurons. This increased exposure is mostly due to increased permeability of the capillary endothelium 24,25 in ganglia as compared to the brain/spinal cord. Most if not all studies should include at least 1 dorsal root ganglion in the tissues to be examined list in the protocol.
Common changes in the dorsal root ganglion of even control animals include neuronal autophagy in monkeys, 26 variations in cellularity (due to infiltrates of white blood cells and/or increased satellite glial cells and/or Schwann cells), and vacuolation. As with any neuronal population, vacuolation may indicate a test article-related change. Large, well defined, single (sometimes subdivided) vacuoles are commonly observed in ganglion neurons of control animals, and these likely represent artifactual change since the appearance is the same regardless of the time between dosing and necropsy (Figure 4). The occurrence of large, discrete vacuoles in ganglion neurons has been reported to be caused by long-term exposure to organophosphates in rats. 27 Neuronal necrosis may occur in sensory ganglia separated from or in association with inflammatory reactions. The administration of adeno-associated virus serotype 9 (AAV9) has been associated with necrosis of dorsal root ganglion neurons following intravenous and intrathecal delivery. 28 Neurons of caudal lumbar/sacral dorsal root ganglia may be preferentially affected (unpublished data from the author) in studies involving the administration of AAV. It is the experience of the author that Fluoro-Jade B and C do not label necrotic neurons in ganglia.
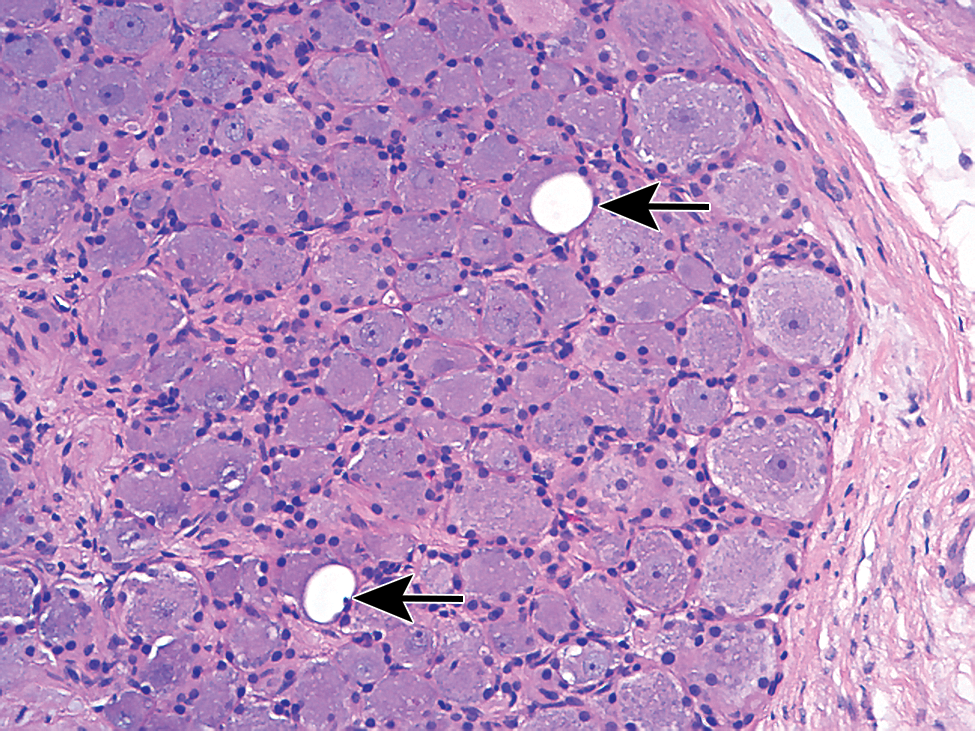
Intravascular perfusion with chilled saline followed by immersion fixation with 10% NBF, paraffin embedding, H&E stain, 5-µ section. Dorsal root ganglion from a cynomolgus monkey. Arrows indicate 2 discrete, large, clear vacuoles in neurons. This is a common artifactual change in dorsal root ganglia of multiple species. Note the orderly arrangement of the satellite glial cells around the periphery of the neurons. H&E indicates hematoxylin and eosin; NBF, neutral-buffered formalin.
Some test articles alter morphologic characteristics (cell size, nucleolar size, etc) of sensory neurons. 29,30 The use of cryoprotected, gelatin-embedded, thick (40-50 µ) frozen sections may be optimal for quantitative investigations including stereology to detect effects on cell numbers or cell/cell component (nucleus, nucleolus) size.
Other Sensory Ganglia
Some cranial nerves (notably the trigeminal nerve and facial nerve but also the vagus nerve) have associated sensory ganglia. Evaluation of these structures is similar to that described for dorsal root ganglia. The inferior sensory ganglion of the vagus nerve is often mistaken for the superior (cranial) CCG, so it is important to be able to differentiate sensory from sympathetic ganglia grossly and microscopically (see Figures 4 and 5).
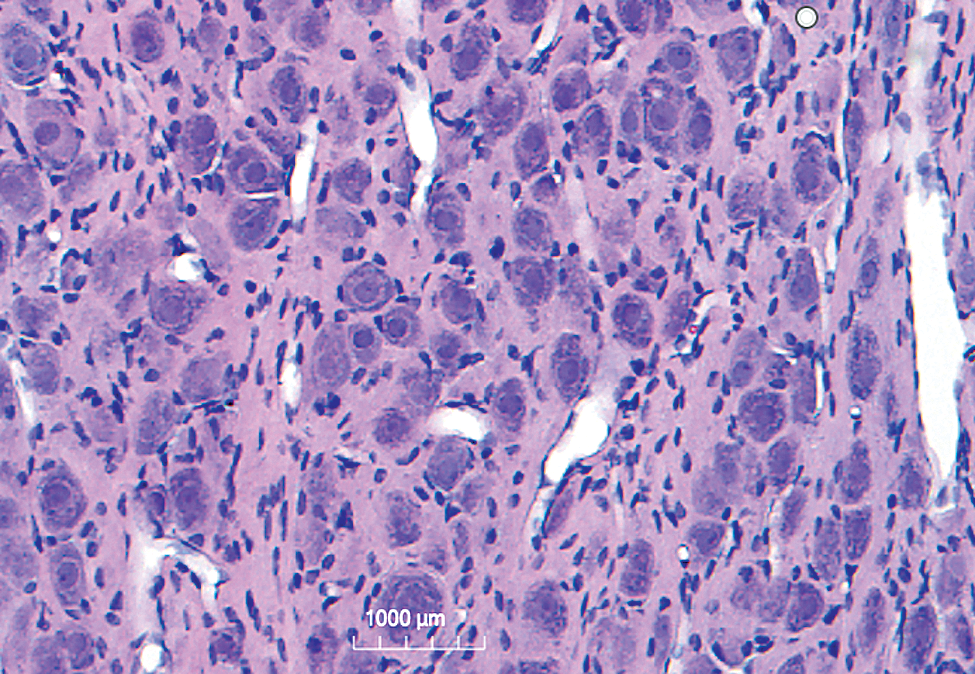
Intravascular perfusion fixation with 10% NBF, paraffin embedding, H&E stain, 5-µ section. Cervicothoracic ganglion from a cynomolgus monkey. The satellite glial cells associated with the neurons lack the more orderly arrangement present in sensory ganglia (see Figure 4). H&E indicates hematoxylin and eosin; NBF, neutral-buffered formalin.
Autonomic Nervous System
The autonomic (involuntary) nervous system is part PNS (postsynaptic neurons) and a part CNS (presynaptic neurons). The system is composed of neurons that form the general and special visceral efferent (innervate smooth muscle) systems and the general and special afferent system, receiving information from viscera and sensory areas for taste and olfaction. 31
Presynaptic neurons of the sympathetic portion lie within the thoracic, lumbar, and sacral 32 spinal cord (intermediate gray column). Presynaptic neurons of the parasympathetic and enteric portions lie within brain stem nuclei (primarily nuclei of the vagus nerve).
The main sympathetic ganglia studied in preclinical investigations are the cranial (superior in monkeys) CCG, cervicothoracic ganglion (CTG; also known as stellate ganglion), and cranial (superior) mesenteric ganglion (CMG). The CCG is located by following the vagosympathetic trunk into the cranial neck region; the ganglion lies near the bifurcation of the carotid artery. 33,34 This ganglion innervates structures in the head. The CTG is formed by the fusion of the most distal cervical ganglion and the most cranial thoracic ganglion plus, in some animals, the second most cranial thoracic ganglion. It may be found by isolating the sympathetic trunk and following that trunk to the level of the first rib/vertebra C7. The CMG is located adjacent to the cranial mesenteric artery (CMA), which is the second branch (after the celiac artery) of the abdominal aorta. This ganglion is best harvested immediately upon opening the abdominal cavity, as any disruption to the local anatomy greatly increases the difficulty of locating this structure. The ganglion is usually not specifically located at necropsy, so a piece of soft tissue containing aorta and the proximal portions of the celiac/CMA from the diaphragm to the level of the left adrenal gland is harvested. The ganglion (near the origin of the CMA of the aorta) is removed in the histology laboratory at the time of trimming or the harvested soft tissue is step sectioned to locate the ganglion.
Sympathetic nerves containing axons from presynaptic neurons in the spinal cord leave the cord in the ventral nerve roots but quickly separate and course toward their target ganglia in the sympathetic chain running through the neck/thorax/abdomen. These sympathetic nerves are sometimes incorrectly diagnosed as having Schwann cell hyperplasia because they are more cellular as compared to the dorsal and ventral spinal nerve roots.
Performing stereology on sympathetic ganglion neurons usually requires the preparation of thick (≥40 µ) frozen sections, unless techniques that can utilize thin sections (physical disectors) are used. Cutting such sections requires specialized equipment (eg, sliding/freezing microtome) not readily available in most histology laboratories.
Unless specialized quantitative investigations such as stereology are being performed, sympathetic ganglia are typically evaluated in PEHE-stained sections. As compared to sensory ganglia, neurons in sympathetic ganglia are generally smaller and satellite glial cells tend to be less well organized (Figure 5). It is the author’s experience that some antibodies to NeuN are not reliable for performing total counts on neurons in sympathetic ganglia, so more traditional Nissl stains should be considered.
Peripheral Nerves
Most guidance documents and most preclinical studies limit the evaluation of peripheral nerves to the mid sciatic. This is probably not the best choice, given the typical drug-induced peripheral neuropathies begin with a “glove and stocking” distribution. 35 Many neuropathies are length-dependent, with the portions of the axon most distal from the neuronal cell body (for sensory neuropathies, neurons in sensory ganglia) affected initially and most severely. Evaluating the mid-sciatic nerve may well miss a neuropathy with axonal damage at more distal sites, such as the distal tibial nerve, the sural nerve, the plantar nerves, or in rodents, the caudal nerve in the tail. 36 Since tissues to be examined in any preclinical investigation should include those likely to be the most sensitive to test article effects, a nerve more distal than the sciatic nerve is likely to be most predictive of developing and/or subtle neuropathies.
Nerves should be examined as PEHE-stained longitudinal and transverse sections and also as an osmicated, resin embedded, toluidine blue–stained transverse section. The evaluation of the structure of myelin, using the light microscope, benefits greatly by the use of osmium-fixed specimens. On longitudinal and/or transverse PEHE-stained nerve sections, there may be obvious axon loss, and nonselective silver stains may assist in determining whether or not axons are present or absent. For subtle changes, the distinction between axonal degeneration and myelin degeneration may not be apparent. This is especially true for subtle myelinopathies, which may, on PEHE-stained sections, look identical to subtle axonopathies. The diagnostic value of osmicated/resin sections is quite apparent in numerous reviews on the PNS 4,6 and also apparent from the following statement quoted from Bilbao and Schmidt: 18
“In the assessment of a nerve specimen, examination of toluidine blue-stained plastic-embedded sections is critical.”
For any study, evaluation of PEHE-stained, longitudinal, and transverse sections of at least 1 distal nerve (eg, distal tibial nerve near the tibiotarsal joint and/or distal sural nerve; caudal nerve in rats) and an osmicated/resin-embedded, toluidine blue–stained transverse section of a distal nerve is recommended. The mid-sciatic nerve should be included to satisfy the guideline. In studies where there is any inclination of an effect in the PNS (situations 2 and 4 as defined in Bolon et al 7 ), multiple nerves, including distal nerves that include a branch of a predominantly sensory nerve (eg sural nerve), should be evaluated in paraffin and resin sections.
Intraepidermal Nerve Fiber Density
A very sensitive assessment of peripheral neuropathy, and 1 that provides quantitative data suitable for statistical analysis, is the determination of intraepidermal nerve fiber density. This assessment quantifies, via 1 of 2 methods (direct counts [most commonly used] or stereology [using an isotropic sphere virtual probe often referred to as “space balls”]), the sensory nerve fibers in the epidermis (plantar foot skin; rats, monkeys, dogs) or dermis (if evaluation of the epidermis is not feasible). The epidermal assessment basically counts nerve fibers crossing the dermal/epidermal junction (Figure 6) of an area where the epidermis is sufficiently thick (to provide a suitable background for counting) and free of hair (to allow for consistent sections to be cut) to allow for an accurate/repeatable analysis. There are numerous references describing the utility and nuances of this analysis. 37,38,39,40
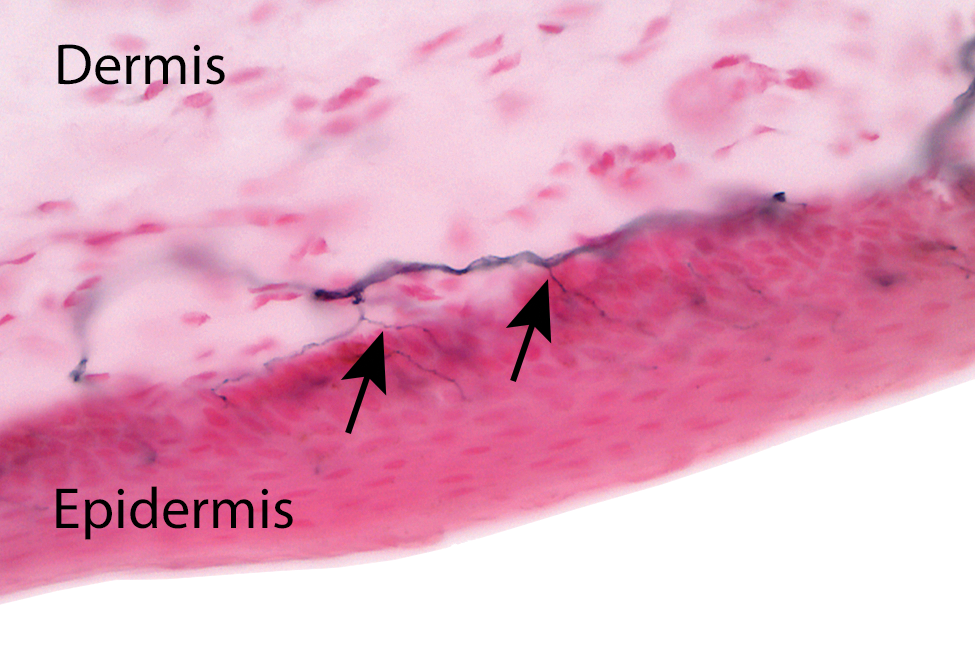
Immersion fixation in Zamboni’s fixative for 24 hours, frozen section, PGP 9.5 immunohistochemical stain for neurites with alizarin-red counter stain; 50-µ section. Plantar foot skin from a cynomolgus monkey. Sensory axons, originating from a dermal pedicle, cross over the dermal/epidermal junction to innervate the epidermis. The arrows indicate 2 axons as they cross from the dermis to the epidermis. To determine intraepidermal nerve fiber density, fibers are typically counted as they cross the junction, although stereologic methods to quantify total axon length in the epidermis are equally accurate (although a bit more time intensive).
The absolute key to this investigation lies in the harvest of high-quality, consistent specimens. Various procedures/reagents can be used, and each laboratory should develop the process that works best. One sample procedure used in rodents and monkeys is as follows: Using a new, 3-mm biopsy punch, obtain a sample of plantar skin/dermis from a site on the ball of the foot near the base of the digits. The same site (approximately) should be sampled for each animal. For transfer from animal to vial, grasp the dermal fat with forceps; the epidermis must remain untouched. Place the punch into Zamboni’s fixative (2% paraformaldehyde/picric acid) for 12 to 16 hours (being consistent within a 4-hour time window for all animals). Zamboni’s fixative is commercially available. The fixative must be kept cold, and the biopsy should remain chilled throughout the entire process of fixation/rinsing/shipment until microtomy. Rinse the biopsy 3 times in a 0.10 M chilled Sorensen’s phosphate buffer. Cryoprotect using a sucrose (20-30%) or glycerol (20%) solution (200 mL glycerol in 800 mL 0.1M Sorensen’s phosphate buffer). The sample should be processed as soon as possible but may be stored in the cryoprotectant for up to 1 week (or placed into a −20°C freezer for longer term storage).
The biopsies are sectioned using a sliding/freezing microtome and a 50-µ block advance, then stained (free floating) with a primary antibody to detect protein gene product 9.5, a pan-neuronal marker. At the author’s laboratory, a total of 24, 50-µ sections are cut from each biopsy, and 4 to 6 sections (selected in a systematic random manner) are mounted for staining. The sections are examined (blinded) for section quality, and 3 to 4 sections of the highest quality are used for nerve counts (also performed blinded).
Because this technique is so completely dependent on sample and staining consistency between animals, all biopsies that might have any possibility of being evaluated (ie, all dose groups including low and intermediate groups) should be taken to stained slide, even if the strategy is to initially evaluate the control and high-dose groups. There should be no variation between groups regarding how the samples are obtained, processed, or examined, and all samples (from each sacrifice interval) must be prepared in the same time frame. While these procedures can identify peripheral neuropathies inapparent in more proximal nerve sections, the process is time-consuming and fraught with potential problems at every step of specimen preparation. These techniques must be practiced and verified, and personnel must be specifically and thoroughly trained if there is any hope of success using these techniques. Considerable knowledge and tips for these techniques can be obtained by visiting the Johns Hopkins Medicine’s Cutaneous Nerve Lab’s website 41 and by reviewing recent articles on this subject.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
