Abstract
Microminipigs have become an attractive animal model for toxicology and pharmacology studies and for human disease models, owing to their manageable size. Although there are numerous reports of spontaneous age-related lesions in mice, rats, dogs, and monkeys, those in minipigs are scarce. In the present study, spontaneous age-related histopathological changes were investigated using 37 microminipigs (20 males and 17 females) that were 6 months to 10 years of age. Abnormal deposits of materials were evident in several animals from 6 years of age, and these deposits included amyloid in the renal medulla, thyroid gland, and adrenal gland, hyaline droplets in glomeruli, and fibrillar inclusions in neurons. Arterial sclerosing changes (intimal thickening, intimal proliferation, and medial mineralization) and proliferative lesions (hyperplasia of hepatocytes, follicular cells, Leydig cells, and uterine endometrial glands) were present at 4 years of age and beyond. Renal adenoma, uterine leiomyoma, and Leydig cell tumor were observed in several microminipigs. Moreover, glomerulosclerosis, renal interstitial fibrosis, thymic involution, and adrenocortical cell vacuolation were common in aging microminipigs. Since knowledge of age-related changes is helpful for pathologists, the basic information obtained in this study will be a useful reference for all future toxicity evaluations in microminipigs.
Introduction
Minipigs are one of the major animal species used in translational research because of their similarities with humans in anatomical and physiological characteristics. 1,2 Among several strains of minipigs, microminipigs (Fuji Micra Inc, Shizuoka, Japan) have become an increasingly attractive animal model and have been used in pharmacological and toxicological studies, 3 –7 human disease models, 8 –11 and studies of regenerative medicine. 12,13 In addition, microminipigs have considerable potential, particularly for long-term studies, because of their manageable size, reaching approximately 10 kg at sexual maturity and up to 20 to 30 kg at adulthood. 14 –16
A basic understanding of spontaneous age-related changes in the animal under investigation is essential for pathologists to recognize whether potential changes are induced by a study condition or spontaneously generated with age. There are numerous reports of spontaneous age-related lesions present in a number of laboratory animals, including mice, rats, dogs, and monkeys. 17 –24 In minipigs, a large number of literature regarding spontaneous histological changes in young to young adult minipigs exist, 25 –28 and there are several reports detailing the changes observed in female reproductive organs in aging individuals. 29 –31 Reports detailing systemic histological changes in aging minipigs, however, are scarce, particularly for microminipigs.
Various histological changes develop systemically with age in human and animals: abnormal deposits of materials in cells and tissues, arterial changes, proliferative changes, and thymic involution all occur regularly. 32 –34
The objective of this study is to clarify the incidence of spontaneous histological changes in aging microminipigs by focusing mainly on the changes mentioned above, through thorough histological and immunohistochemical evaluation.
Materials and Methods
Animals
A total of 37 microminipigs (20 males and 17 females) aged between 6 months and 10 years were utilized in this study. Ten microminipigs between the aged 6 and 12 months (7 males and 3 females) were used as control. Of the 27 aged animals (13 males and 14 females), 20 (7 males and 13 females) were retired breeders (Table 1). The animals were born and raised either in Fuji Micra Inc or Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center (Shizuoka, Japan). In both facilities, the animals were housed in a large group until age 3 months and then separated into male and female groups until age 6 months. After 6 months of age, the animals were housed individually in pens of at least 0.54 m2 area/animal, under conventional conditions with 12-hour light/dark cycles. The animals were weaned at 4 weeks of age and then fed a commercially available diet at 2% to 3% of body weight/animal/day with ad libitum access to water. The suppliers of the diet differed between the facilities: Marubeni Nisshin Feed Co, Ltd (Tokyo, Japan) or Chubushiryo Co, Ltd (Aichi, Japan) in Fuji Micra, Oriental Yeast Co, Ltd (Tokyo, Japan), or JA Higashinihon Kumiai Shiryou (Gunma, Japan) in Shizuoka Prefectural Research Institute. A number of pathogens were excluded or controlled for, and they were periodically monitored in both breeding facilities. Specifically, these pathogens included pseudorabies virus, porcine reproductive and respiratory syndrome virus, Actinobacillus pleuropneumoniae, Bordetella bronchiseptica, Brachyspira hyodysenteriae, Erysipelothrix rhusiopathiae, Haemophilus parasuis, Mycoplasma hyopneumoniae, Pasteurella multocida, Salmonella enterica, Coccidia (including Toxoplasma gondii), and internal parasites. During daily observations until necropsy, 7 (2 males and 5 females) of 27 aged animals showed signs of reproductive disorders, such as aspermia (clinically diagnosed by semen examination), low libido, subfertility, and silent heat, while the others (including the young control animals) were in good health. Before necropsy, all animals received intramuscular injections of 0.1 mL/kg midazolam (Dormicum 10 mg, Astellas Pharma Inc, Tokyo, Japan) and 10 mg/kg ketamine (Ketamine 5% [Fujita], Fujita Pharmaceutical Co, Ltd, Tokyo, Japan) as anesthesia. Animals were euthanized by exsanguination after a veterinarian confirmed depth of anesthesia by examining their state of immobility and insensibility to external stimuli: loss of both eyelid and corneal reflexes, and lack of response to skin pinching. 35 This study was conducted in accordance with the official regulations for animal care and use in Shizuoka prefectural Research Institute of Animal Industry, Swine and Poultry Research Center, which complies with the animal care and ethics described in the Guidelines for Proper Conduct of Animal Experiments issued by the Science Council of Japan (2006). 36
Clinical Manifestations and Macroscopic Findings in Aging Microminipigs.
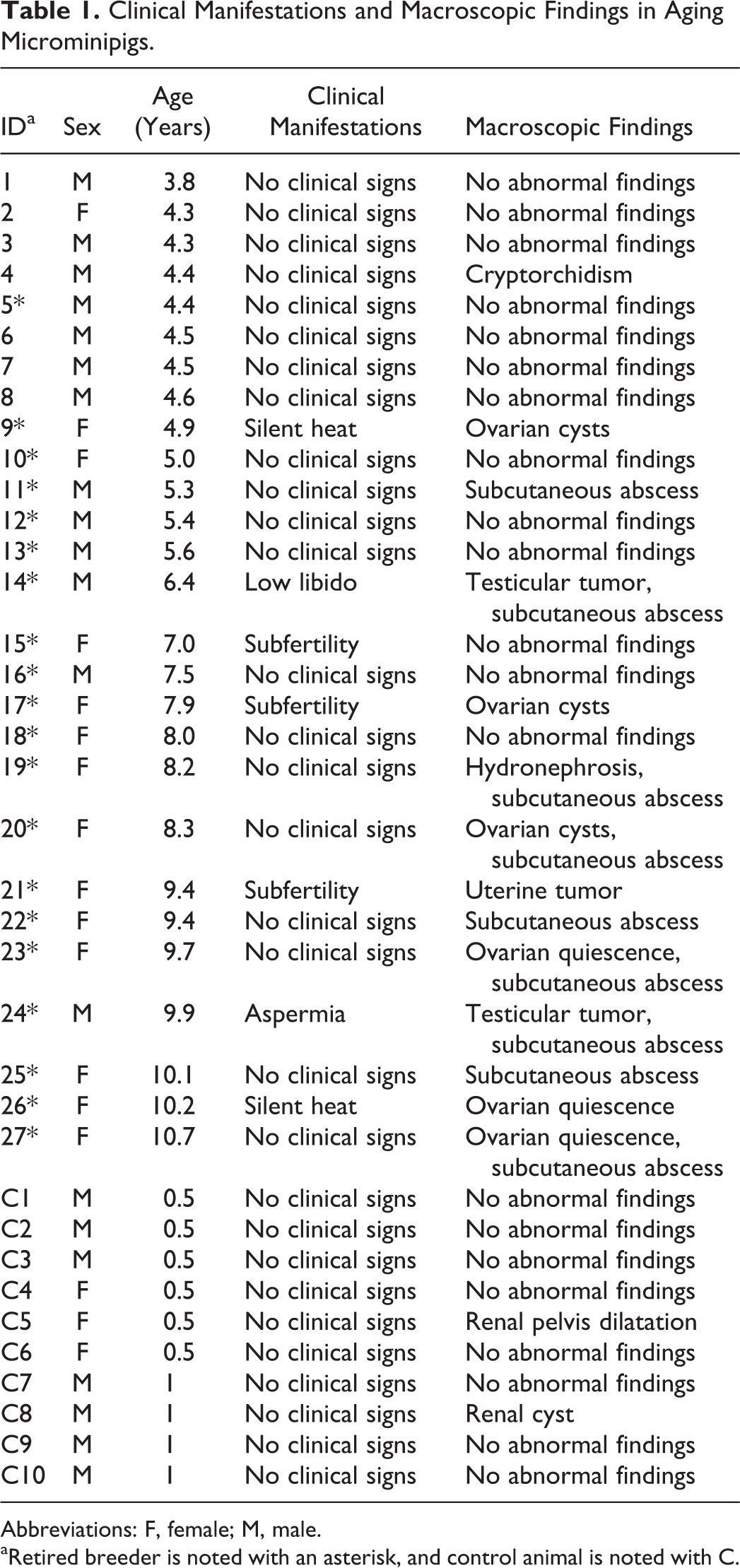
Abbreviations: F, female; M, male.
aRetired breeder is noted with an asterisk, and control animal is noted with C.
Histopathological Evaluation
Tissues were collected from the same area of each animal at necropsy and immersed into 10% neutral-buffered formalin, 37,38 and these tissues included the liver, gallbladder, spleen, kidney, urinary bladder, heart, aorta, lung, tongue, esophagus, stomach, duodenum, jejunum, ileum, cecum, colon, rectum, mandibular gland, parotid gland, pancreas, thymus (caudal part of the cervical portion), pituitary gland, thyroid gland, adrenal gland, epididymis, prostate gland, vesicular gland, bulbourethral gland, ovary, oviduct, uterus, uterine cervix, vagina, mammary gland, and brain. Testes and eyes were immersed into modified Davidson’s solution. 38,39 Fixed samples were routinely trimmed at the same cross section for each organ, dehydrated by ethanol gradient, and then embedded in paraffin. Paraffin blocks were cut in 3-µm sections and the sections were stained with hematoxylin and eosin (H&E) for routine histological examination. In this study, the findings were evaluated by focusing mainly on abnormal deposits of materials in cells and tissues, arterial changes, and proliferative changes. 32 –34 To better understand the incidence of age-related changes, the 27 aged animals were separated according to age into 3 groups: between 4 and 5 years (10 males and 3 females), between 6 and 8 years (2 males and 5 females), and between 9 and 10 years (1 male and 6 females).
Proliferating lesions of Leydig cells were classified as either Leydig cell hyperplasia or Leydig cell tumors. The criteria for distinguishing these 2 lesions were the degree of compression of adjacent testicular tissue, nuclear atypia, and diameter of foci (a focus with a diameter larger than 3 seminiferous tubules was interpreted to be a Leydig cell tumor). 40,41 To document the severity of a lesion, a 4-point grading system was used: 1, minimal; 2, slight; 3, moderate; and 4, severe. 42,43 For some lesions such as congenital anomalies, cyst(s), and neoplasms, the changes were recorded simply as present (P) rather than scoring a severity grade. 42
Special Stains and Immunohistochemistry
To better understand the changes observed, the following special staining analyses were performed: Congo red, Gallyas-Braak, Masson’s trichrome, periodic acid–Schiff hematoxylin (PAS-H), periodic acid–methenamine–silver with H&E (PAM), phosphotungstic acid–hematoxylin (PTAH), Schmorl’s method, silver impregnation method, Victoria blue with H&E, and von Kossa. Immunohistochemistry was performed using the ABC method (VECTASTAIN Universal Elite ABC kit, Vector Laboratories, Burlingame, California). Briefly, the antibody against α-smooth muscle actin (α-SMA, NBP2-33006, Novus Biochemicals, Centennial, Colorado) was utilized to locate smooth muscle cells in arteries and to determine the origin of the neoplastic lesion. Allograft inflammatory factor 1 (AIF-1/Iba-1, OAGA00004, Aviva Systems Biology Corp, San Diego, California) was used to detect the presence of macrophages in arteries. The sections were deparaffinized and, for antigen retrieval, heated 3 times at 98°C for 5 minutes each in a microwave oven for α-SMA, and treated for 30 minutes in boiled distilled water for AIF-1/Iba-1. The sections were then immersed in absolute methanol and 0.3% H2O2 for 30 minutes to quench endogenous peroxidase activity. After blocking nonspecific binding for 20 minutes, the slides were incubated with the primary antibodies overnight at 4°C. After being incubated with the biotinylated secondary antibody for 30 minutes at room temperature, the sections were incubated with VECTASTAIN ABC reagent for 30 minutes at room temperature and then with DAB (DAB peroxidase substrate kit, ImmPACT, Vector Laboratories) to detect immunoreactivity. The sections were washed twice in phosphate-buffered saline between each step. The counterstaining was performed with hematoxylin.
Results
Macroscopic findings at necropsy are shown in Table 1. A unilateral testicular mass was observed in the boars with clinical signs of aspermia and low libido. Gross lesions in sows with subfertility and silent heat were either ovarian cysts, ovarian quiescence (absence of functional ovulatory follicles and corpora lutea), or a uterine mass. Hydronephrosis, cryptorchidism, ovarian cysts, or ovarian quiescence were found in 4 animals with no clinical signs. In addition, a subcutaneous abscess around the head or neck was observed in 9 animals older than 6 years. No clinical signs and no macroscopic findings, except for a small cyst and pelvis dilatation in the kidney of 2 individuals, were found in control animals. Histological changes observed were as follows.
Abnormal Deposits of Materials
In all animals older than 4 years, fine brown granules were deposited in centrilobular hepatocytes and in Leydig cells throughout the testis (Supplemental Figure S1A and B). The pigments accumulated in cardiac myocytes, follicular cells of the thyroid gland, and neurons and glial cells in the brain in all animals older than 6 years (Supplemental Figure S1C-E). The amount of deposition within the liver, heart, brain, thyroid gland, and testis apparently increased with age. In several animals, there were mild focal pigmentations in the epithelial cells of the renal proximal tubule (Supplemental Figure S1F) and in adrenal cortical cells. Note that pigments were slightly deposited in the liver and testis of several control animals. These fine brown pigments in H&E sections stained dark blue to dark greenish blue with the Schmorl’s method, purple-red with PAS reaction, and negative with Berlin blue, confirming that the pigments were lipofuscin (Table 2, Supplemental Figure S1, insets).
Incidence of Abnormal Deposits of Materials and Mineralizations in Aging Microminipigs.
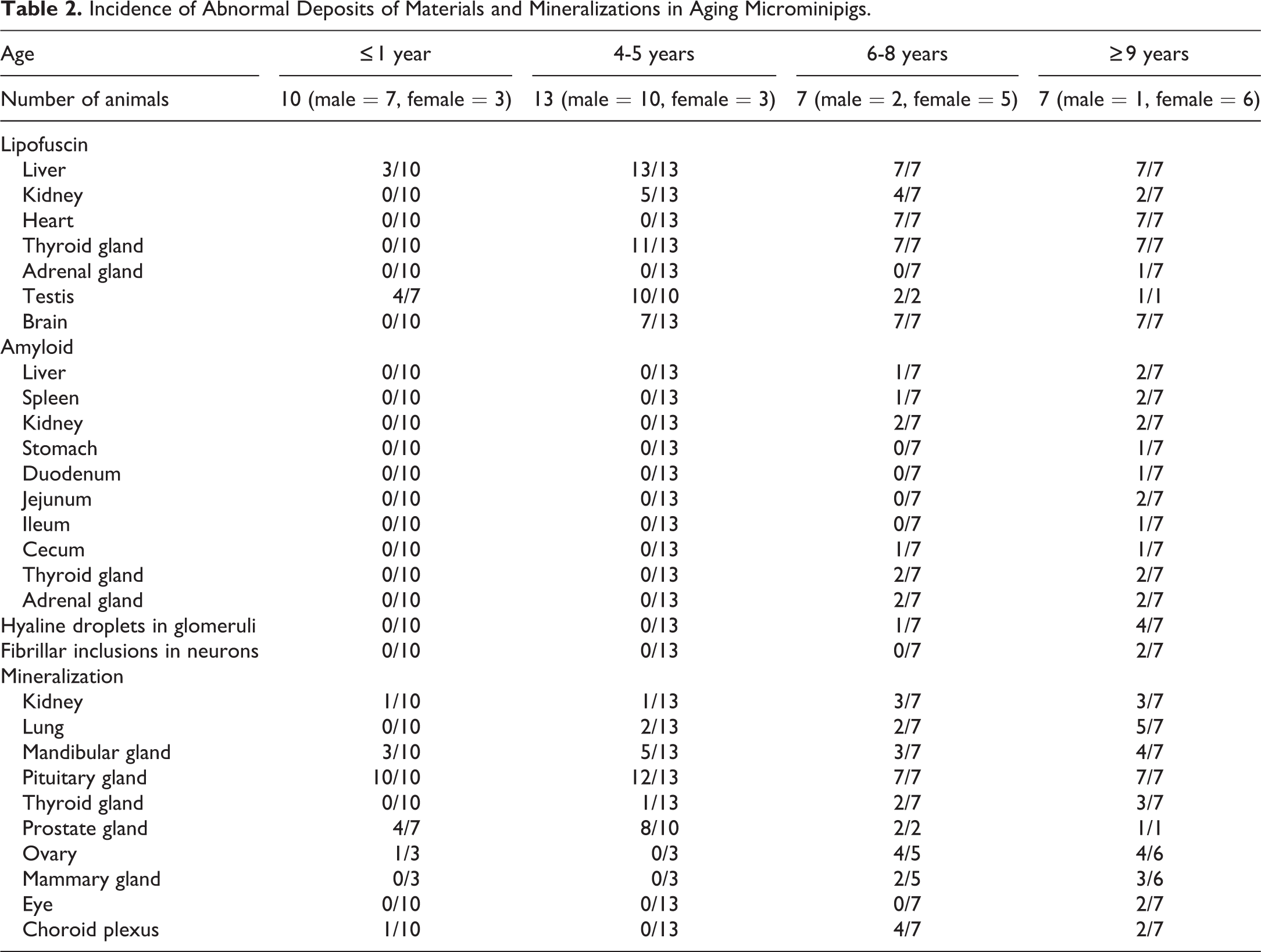
Materials that ranged from eosinophilic homogeneous to slightly fibrillar in composition were observed in certain organs of 4 animals older than 8 years (2 at 8 years of age, 1 at 9 years of age, and 1 at 10 years of age). The eosinophilic material was deposited around the tubules in the medullary interstitium of the kidney (Figure 1A), in the interstitial compartment between thyroid follicles (Figure 1D), in the walls of arterioles and extracellular space between cortical cells in the adrenal gland (Figure 1G), in the wall of central arteries and in splenic cords in the spleen, and in the walls of arterioles in the liver, stomach, duodenum, jejunum, ileum, and cecum. Kidney, thyroid gland, and adrenal gland were the specific organs where eosinophilic material was present in all 4 animals. The amount of deposition in these 3 organs at 8 years of age was apparently less than that found within the organs of animals at 9 and 10 years of age. Additionally, eosinophilic material in the liver was observed in animals at 9 and 10 years of age, while the material was present in 1 of 2 animals at 8 years of age. Deposition within the spleen and gastrointestinal tract was observed in animals only at 9 and/or 10 years of age. These eosinophilic extracellular substances stained orange to red color by Congo red staining (Figure 1B, E, and H) and produced apple-green birefringence viewed under cross-polarized light (Figure 1C, F, and I); therefore, the material was identified as amyloid.
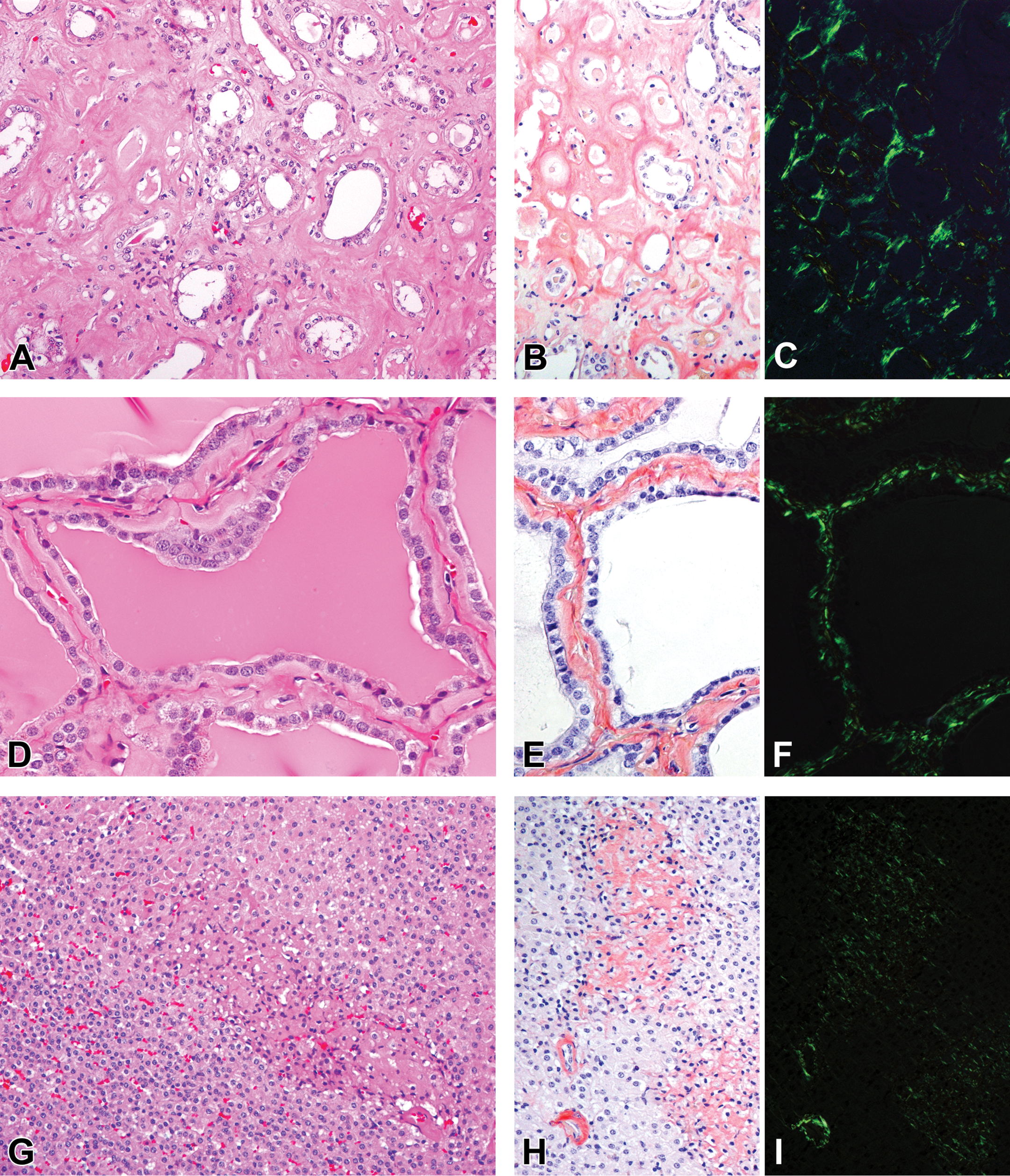
Amyloid deposition in aging microminipigs. Extracellular eosinophilic homogeneous material is deposited in the sections stained with H&E (A, D, and G) and Congo red (B, E, and H) and produces apple-green birefringence viewed under cross-polarized light with Congo red (C, F, and I). The deposits are observed: A–C in the medullary interstitium in the kidney of a microminipig at 10 years of age, D-F in the interstitium between follicles in the thyroid gland of a microminipig at 9 years of age, G–I in the extracellular space between cortical cells and the wall of arterioles in the adrenal gland of a microminipig at 10 years of age. H&E indicates hematoxylin and eosin.
In 5 animals older than 7 years, eosinophilic hyaline droplets of various sizes were observed in the mesangial areas in the renal glomeruli (Figure 2). The number of droplets varied between individuals and also differed between glomeruli in each individual. The droplets were focally to diffusely distributed throughout the kidney, but segmentally to globally deposited within glomeruli. Most of the mesangial cells in the lesion became plump and distorted by the droplets. Relatively small droplets were occasionally seen in the cytoplasm of mesangial cells. The droplets were stained purple red with the PAS reaction and deep blue with PTAH (Figure 2, insets). PAM–stained sections revealed that the basement membrane was thin and partially fragmented around the droplets (Figure 2B). The more severe the changes of the basement membrane became, the more prominent the hyaline droplet formed. No crescent formation in Bowman’s capsules or hyaline droplets in the epithelial cells of renal proximal tubules was observed.
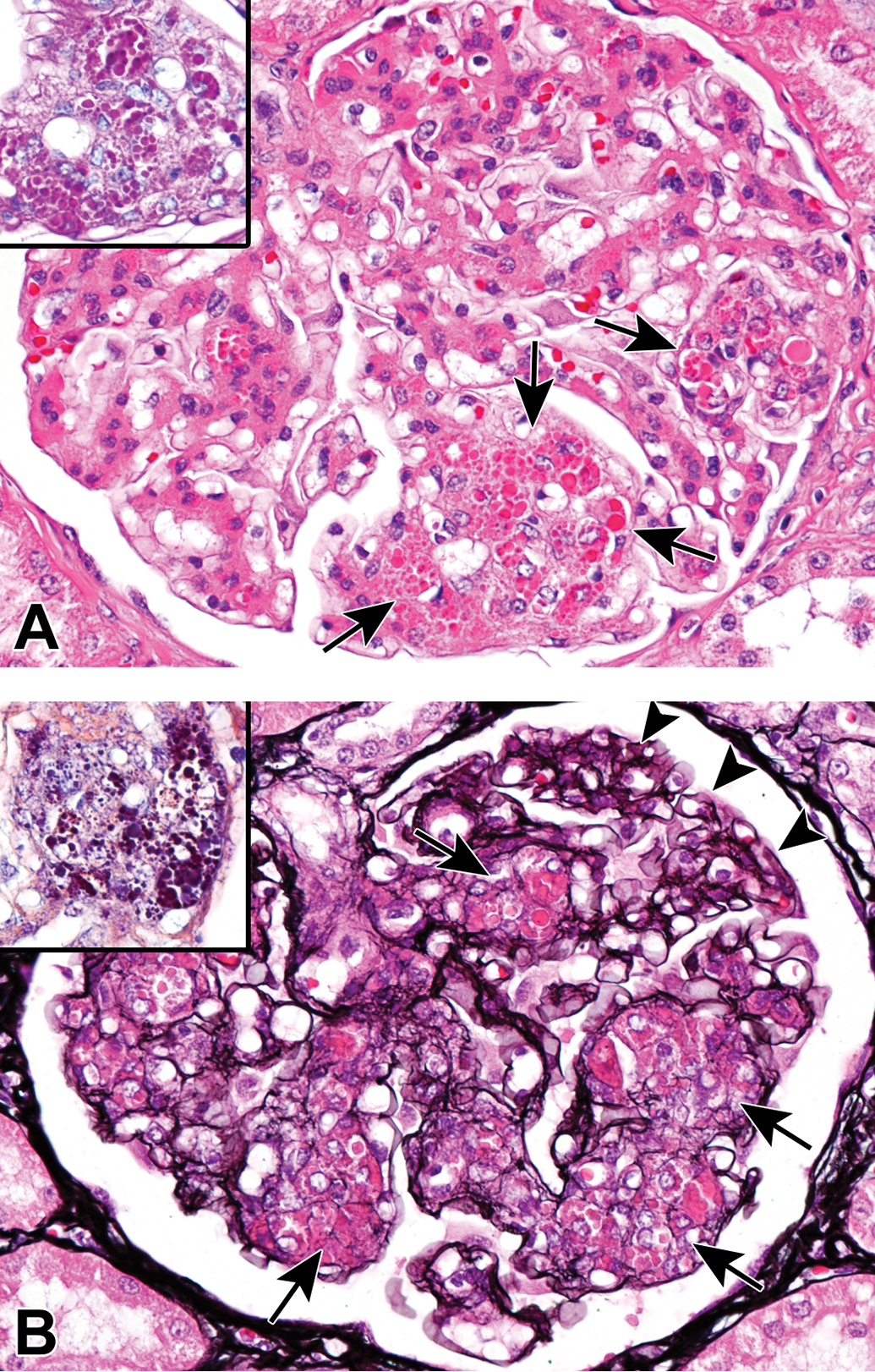
Hyaline droplet deposition in the kidney of an aging microminipig at 9 years of age. A, Eosinophilic hyaline droplets of various sizes are evident in the mesangial areas of glomerulus (arrows). H&E. Droplets are stained purple red with the PAS reaction (A, inset) and deep blue with PTAH (B, inset). B, The basement membrane is quite thin and partially fragmented around the droplets (arrows), compared to that of normal area (arrowheads). PAM staining. H&E indicates hematoxylin and eosin; PAM, periodic acid–methenamine; PAS, periodic acid–Schiff; PTAH, phosphotungstic acid–hematoxylin.
In 2 animals older than 9 years, an eosinophilic fibrillar intracytoplasmic inclusion was deposited in several neurons (Figure 3). The location of the inclusions was limited to the nucleus cuneatus of the medulla oblongata. The fibrillar materials were stained deep blue with PTAH (Figure 3, inset) but showed no reaction with PAS, Masson trichrome, PAM, or Gallyas-Braak staining.
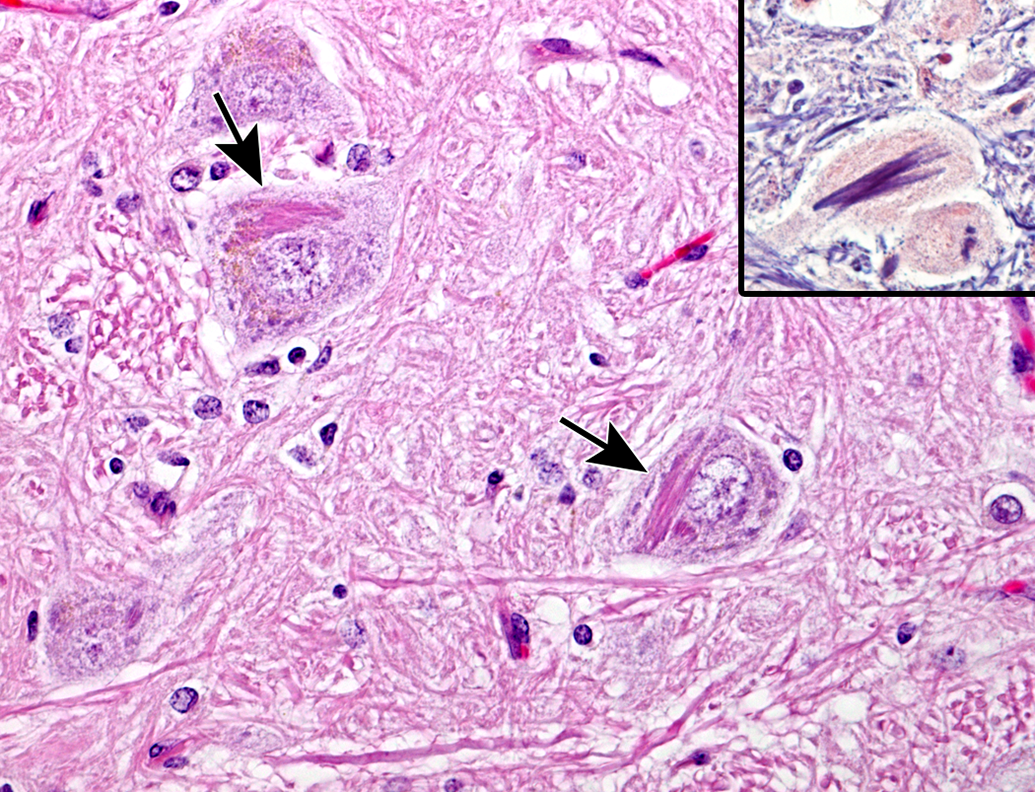
Eosinophilic fibrillar intracytoplasmic inclusion in the neurons at nucleus cuneatus in medulla oblongata of an aging microminipig at 9 years of age (arrows). H&E. The inclusions are stained deep blue with PTAH (inset). H&E indicates hematoxylin and eosin; PTAH, phosphotungstic acid–hematoxylin.
Slight-to-mild mineral deposits, stained dark brown to black by von Kossa staining and therefore confirmed as calcification, were present in the following locations: in the epithelial cells of collective tubules in the renal papilla, in the septum of the lung, in and around the ductal epithelial cells of the mandibular gland, within the colloid of the thyroid gland, within the colloid material in the pars intermedia of the pituitary gland (Supplemental Figure S2A), in the extracellular matrix of the choroid plexus, in the glandular epithelial cells and the glandular lumen of the prostate gland (Supplemental Figure S2B), and in and around the ductal epithelial cells of the mammary gland. Notably, minerals were deposited in the pituitary gland of almost all animals, including control animals.
Arterial Changes
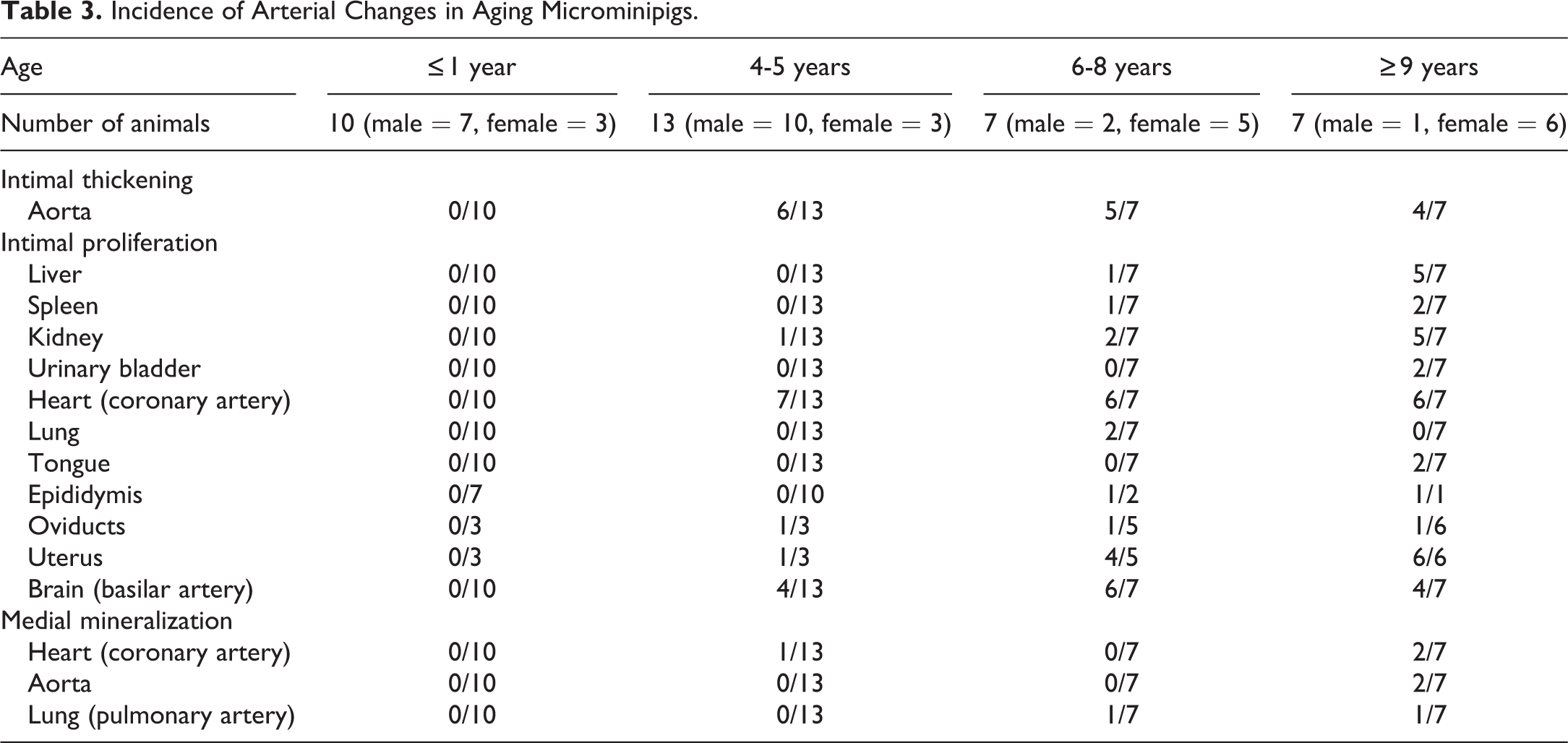
An increase in the matrix between the endothelium and the media, known as intimal thickening, was present partially to globally in the aorta at 4 years of age and thereafter (Table 3, Figure 4A). Special staining with Masson’s trichrome and Victoria blue and immunohistochemistry for α-SMA and AIF-1/Iba-1 revealed that the lesion consisted of loosely arranged collagenous tissue with a number of macrophages and a few smooth muscle cells (Figure 4B). In medium-to-large muscular arteries, intimal proliferation (the expansion of intima with cells) was observed at 4 years of age and thereafter. The coronary artery, the basilar artery, and arteries in the kidney and uterus were commonly affected (Figure 5A). Special staining with Masson’s trichrome and Victoria blue and immunohistochemistry for α-SMA and AIF-1/Iba-1 revealed that the intima was composed of a number of smooth muscle cells with few collagenous and elastic fibers, but macrophages were scarcely observed (Figure 5B).
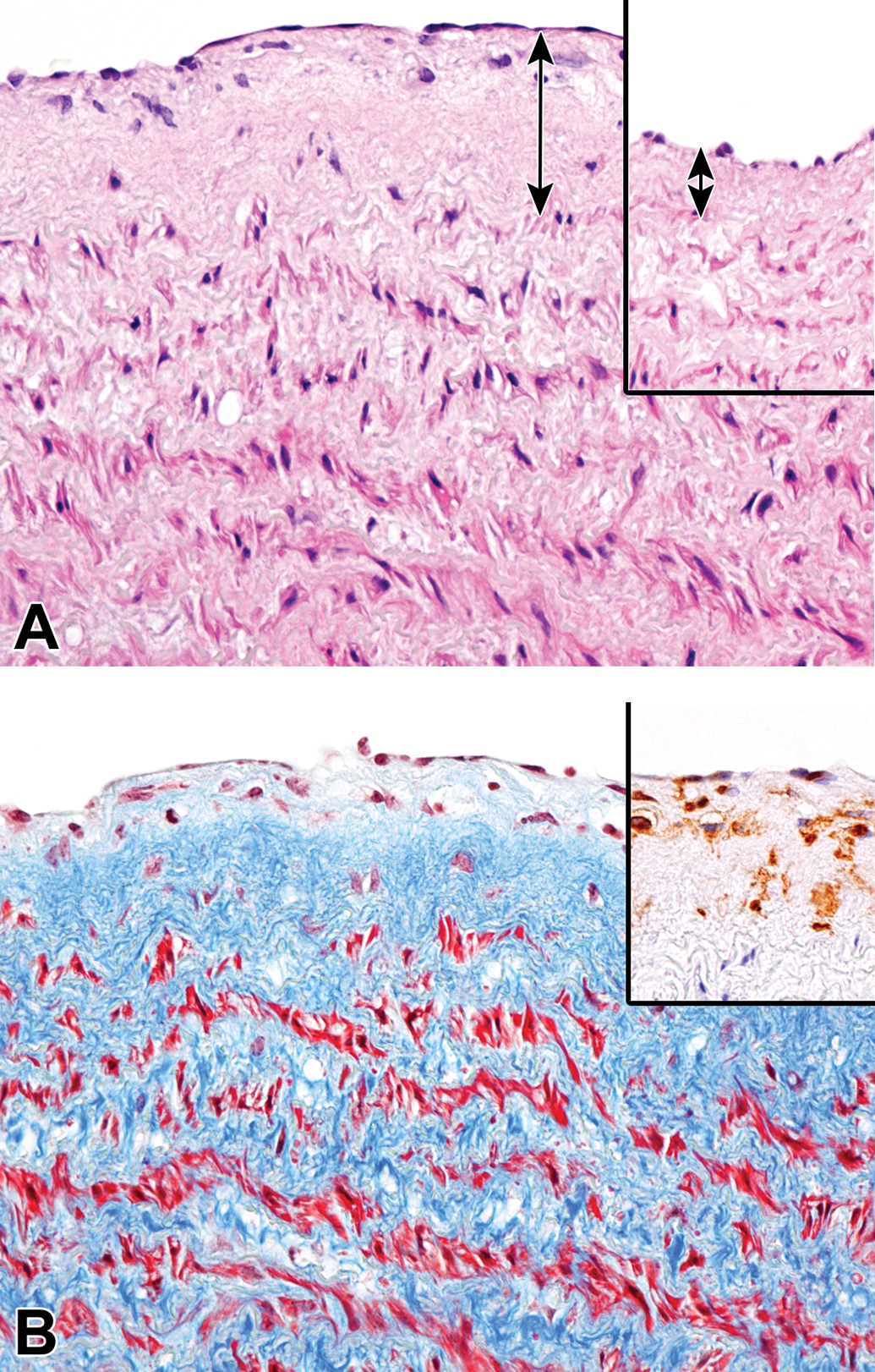
Intimal thickening in the aorta of an aging microminipig at 8 years of age. A, Extracellular matrix between the endothelium and the media increase compared to the normal part of the intima in the same animal (inset). Double-headed arrows indicate the thickness of the intima. H&E. B, The intima is composed of collagenous tissue and a number of cells. Masson’s trichrome staining. Inset: Most of the cells present in the intima are confirmed as macrophages; immunohistochemistry for AIF-1/Iba-1, counterstained with hematoxylin. AIF-1/Iba-1 indicates allograft inflammatory factor 1; H&E, hematoxylin and eosin.
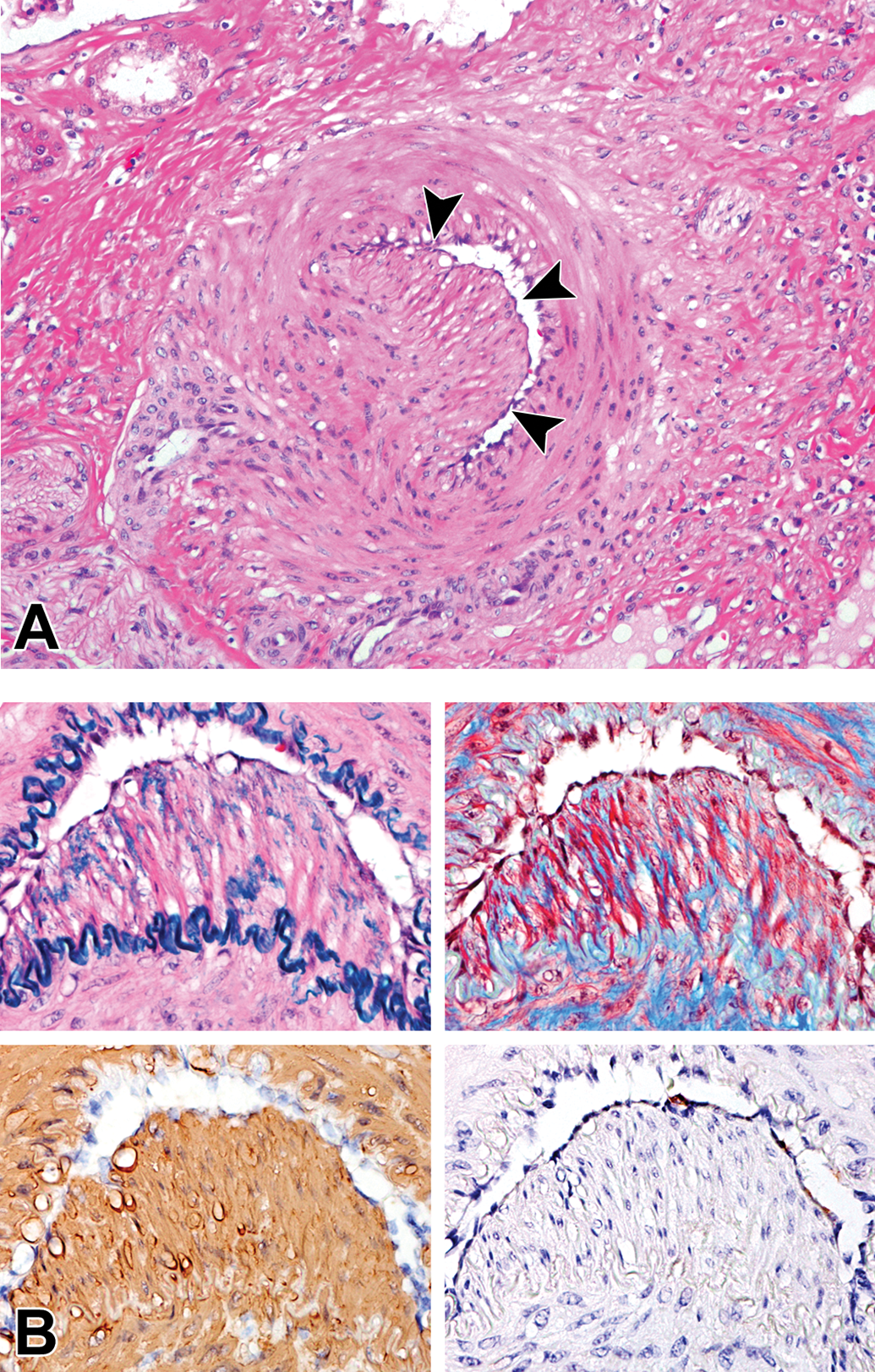
Intimal proliferation in the arcuate artery in the kidney of an aging microminipig at 10 years of age. A, The intima is expanded with cells and the lumen is compressed (arrowheads). H&E. B, The intima consists of a small amount of elastic and collagenous fibers and a number of smooth muscle cells, but macrophages are scarce. Serial sections stained with Victoria blue with H&E (upper left), Masson’s trichrome (upper right), and immunohistochemistry for α-SMA (lower left) and AIF-1/Iba-1 (lower right) counterstained with hematoxylin. AIF-1/Iba-1 indicates allograft inflammatory factor 1; H&E, hematoxylin and eosin; α-SMA, α-smooth muscle actin.
Incidence of Arterial Changes in Aging Microminipigs.
In 3 animals older than 7 years, foci of basophilic granular mineral deposits were present in the tunica media of elastic arteries and large muscular arteries (Figure 6). The deposits were stained dark brown to black by von Kossa and therefore confirmed as calcification. This medial mineralization was observed in the aorta, the coronary artery, and the pulmonary artery.
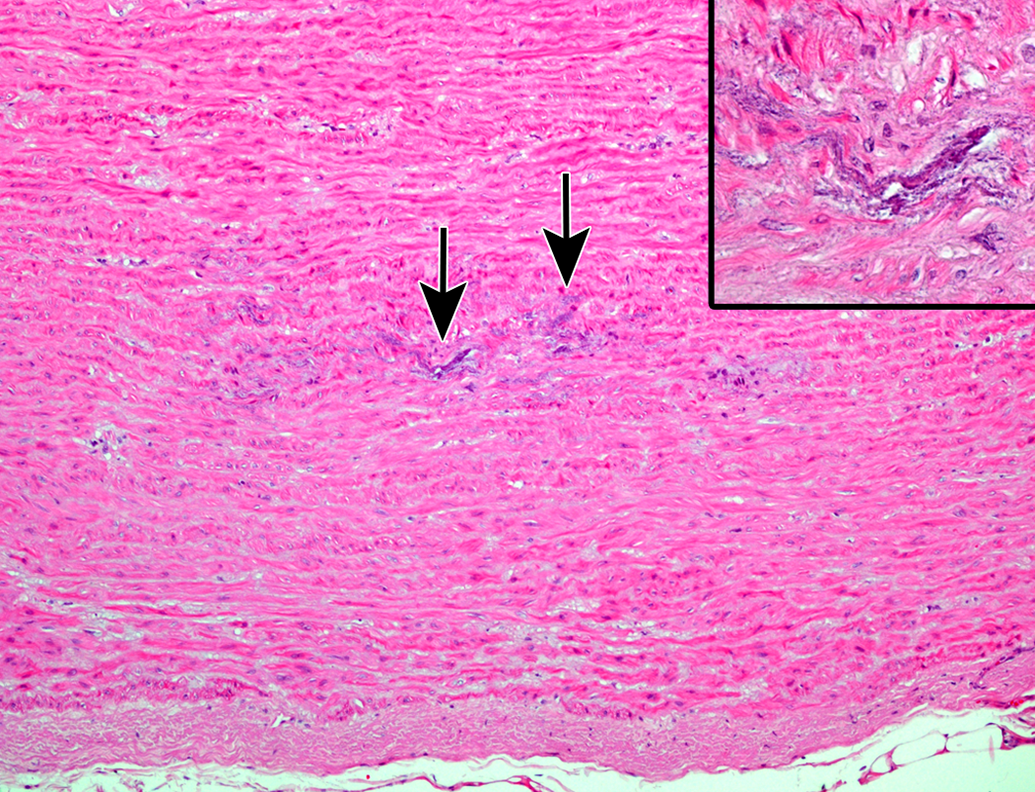
Medial mineralization in the aorta of an aging microminipig at 9 years of age. The arrows indicate mineral deposits. Inset: Higher magnification of the lesion where basophilic granular to crystalline materials are present, and lamellar arrangement of elastic fibers is distorted. H&E. H&E indicates hematoxylin and eosin.
Proliferative Changes
Hepatocellular hyperplasia and uterine gland hyperplasia were 2 common proliferative findings in aging microminipigs (Table 4). Hepatocellular hyperplasia was present focally to multifocally and was randomly distributed within the liver (16 affected animals of 27 total examined animals). There were variations in the size of foci and in the cellular appearance (Figure 7). The size of foci ranged from less than 1 lobule to multiple lobules less than 1 cm in diameter. The small foci were localized in the area either adjacent to portal tracts in the peripheral zone or around the central vein in the central zone. Hepatocytes within the foci were of normal to enlarged size with eosinophilic and/or clear cytoplasm. Binucleate and polyploid cells were occasionally observed, while mitotic figures were uncommon. The foci were composed of tortuous hepatic cords in varying degrees and were well demarcated from normal parenchyma. However, the compression of the sinusoids within the focus and the surrounding liver tissue became prominent as the foci increased their size. In addition, portal areas and central veins were not present in small foci but were seen in large nodular foci. No fibrosis or inflammatory cell infiltration was evident in or around the foci. Hematopoietic cells were occasionally observed within the large nodular foci (Figure 7D, inset).
Incidence of Proliferative Changes in Aging Microminipigs.

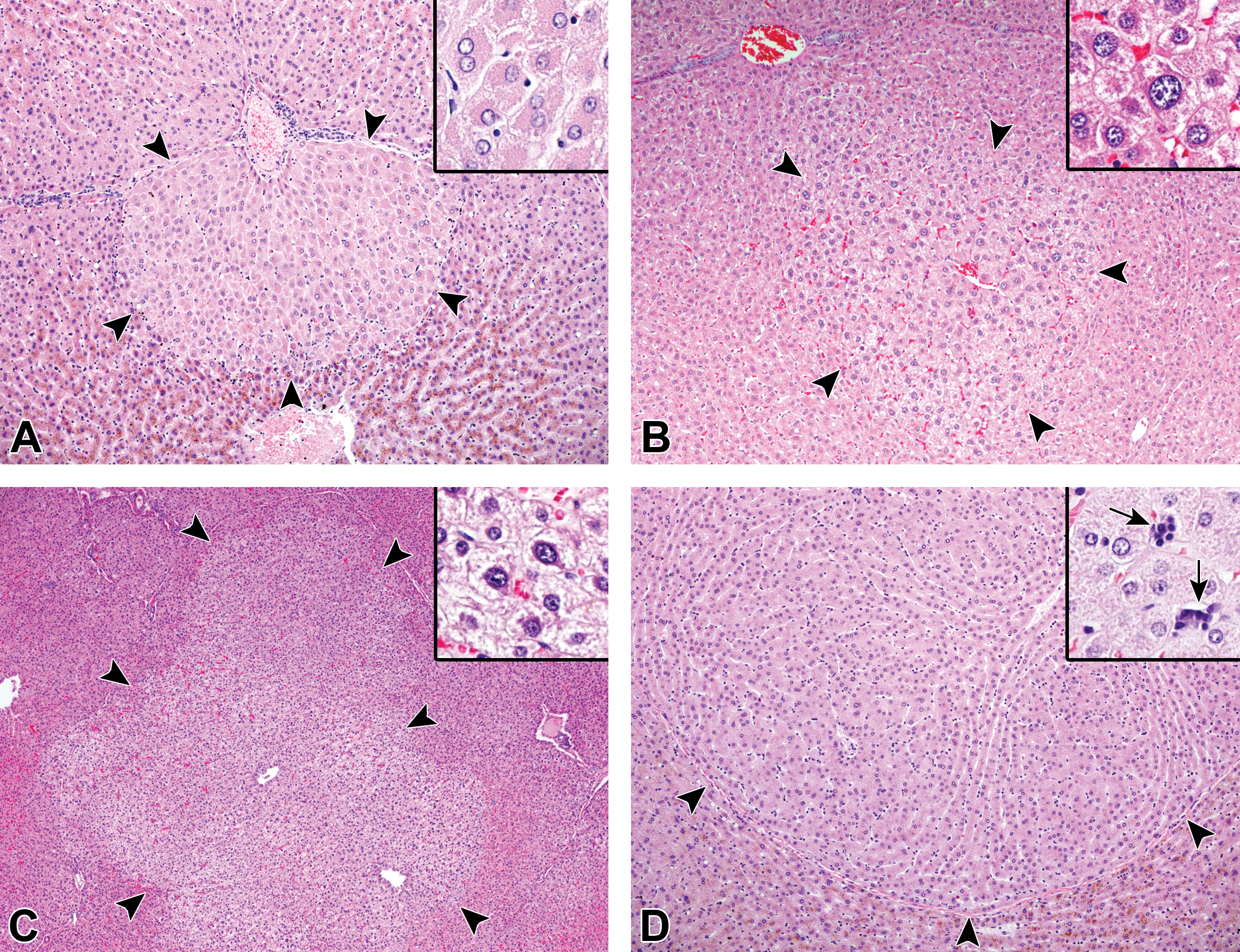
Hepatocellular hyperplasia in aging microminipigs. A, The focus is located in peripheral to intermediate zone and consists of polygonal hepatocytes with eosinophilic cytoplasm in a microminipig at 8 years of age (arrowheads). Inset: Higher magnification of the hyperplastic cells, which are slightly enlarged, and binucleated cells are occasionally observed. B, The focus is present in central to intermediate zone, slightly compressing the surrounding liver tissue in a microminipig at 8 years of age (arrowheads). Inset: Higher magnification of the hyperplastic cells; the enlarged cell with polyploidy is evident. C, The focus is about the size of several lobules and central veins are present in a microminipig at 10 years of age (arrowheads). Inset: Higher magnification of the hyperplastic cells, which are slightly enlarged and have clear cytoplasm with mild anisokaryosis. D, The large focus, composed of the cells with eosinophilic cytoplasm, is well demarcated from normal parenchyma in a microminipig at 9 years of age (arrowheads). Inset: Higher magnification of the hyperplastic cells; the arrows indicate hematopoietic cells (not shown in the lower magnification). H&E. H&E indicates hematoxylin and eosin.
Uterine gland hyperplasia was present focally and was clearly distinguishable from normal endometrium, occasionally accompanied by mild fibrosis between the hyperplastic glands (8 of 14, Supplemental Figure S3). Most of the glands consisted of compactly arranged cuboidal to columnar epithelial cells and lumens were frequently distorted.
In the thyroid of a few aging animals, follicular cell hyperplasia was observed focally and was well demarcated from normal parenchyma without forming a capsule (3 of 27, Figure 8). The foci were composed of columnar cells with eosinophilic cytoplasm and occasionally contained papillary infoldings of the epithelial cells, cellular debris, mineralized colloid, and cholesterol crystals in the colloid.
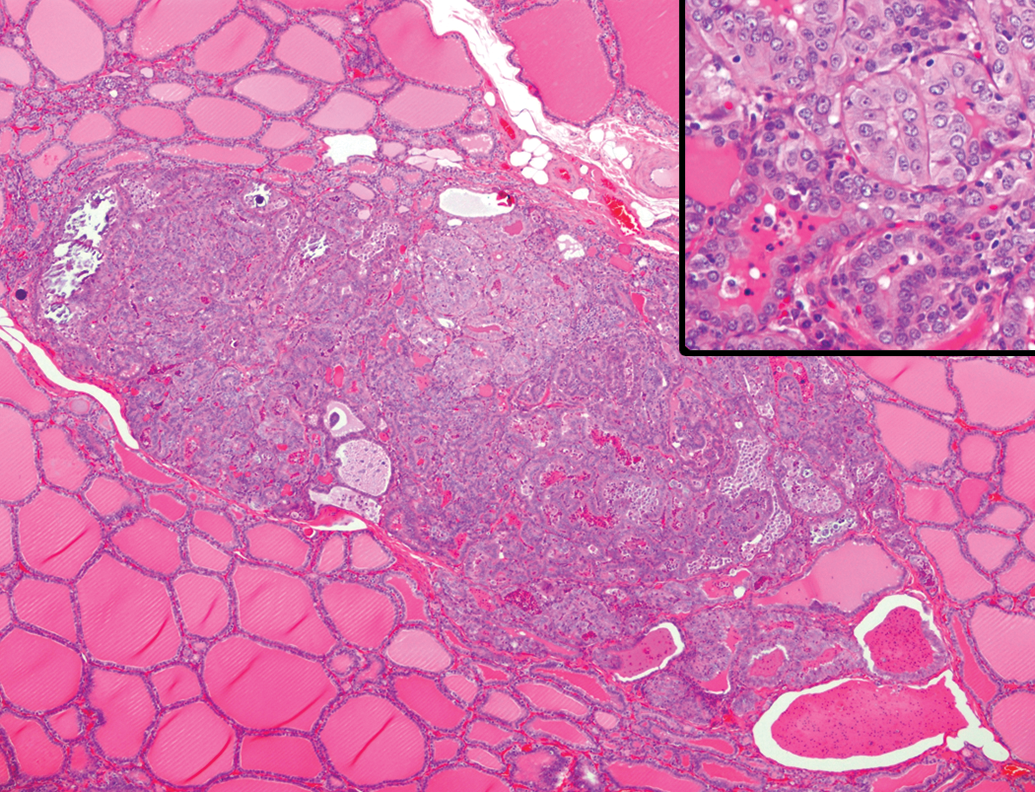
Follicular cell hyperplasia in the thyroid gland of an aging microminipig at 5 years of age. The focus is well demarcated from normal parenchyma. A capsule is not present between the focus and normal follicles. Inset: Higher magnification of the focus that is composed of cuboidal and columnar epithelial cells arranged in a follicular pattern. Colloids with cellular debris are observed. H&E. H&E indicates hematoxylin and eosin.
Hyperplasia and tumors of Leydig cells were present focally or multifocally in the testis of a few individuals (2 of 13). Both changes coexisted in the same testis and were well circumscribed from the normal parenchyma. The hyperplastic foci consisted of large polyhedral cells with abundant eosinophilic cytoplasm, arranged in a solid sheet (Figure 9). Notably, lipofuscin deposition in the proliferating Leydig cells was less severe than normal Leydig cells in the same testis. In Leydig cell tumors, anisokaryosis, intranuclear inclusions, and cellular polymorphism became more prominent as the lesions increased their size (Figure 10). The mass possessed cystic spaces of various sizes, which contained proteinaceous materials, and focal areas of necrosis with mineral deposition were occasionally observed. Mitotic figures were scarce.
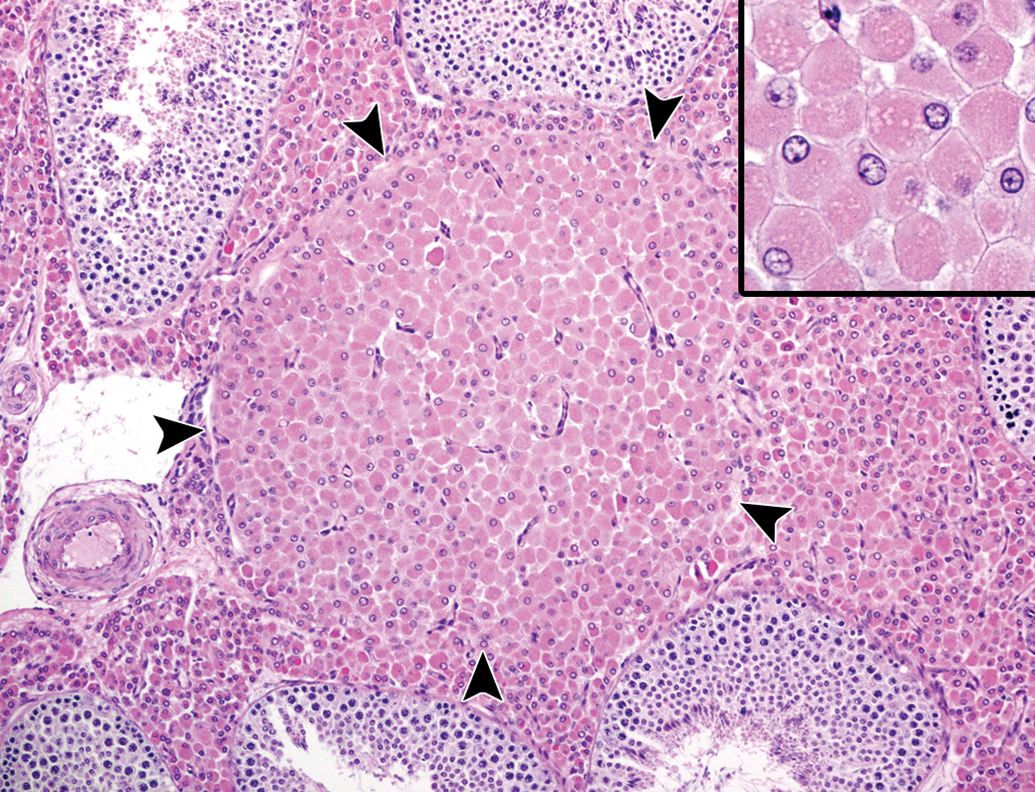
Leydig cell hyperplasia in the testis of an aging microminipig at 6 years of age. The solid aggregation of Leydig cells is present between seminiferous tubules (arrowheads). Inset: Higher magnification of the cells, which are enlarged with abundant eosinophilic cytoplasm. H&E. H&E indicates hematoxylin and eosin.
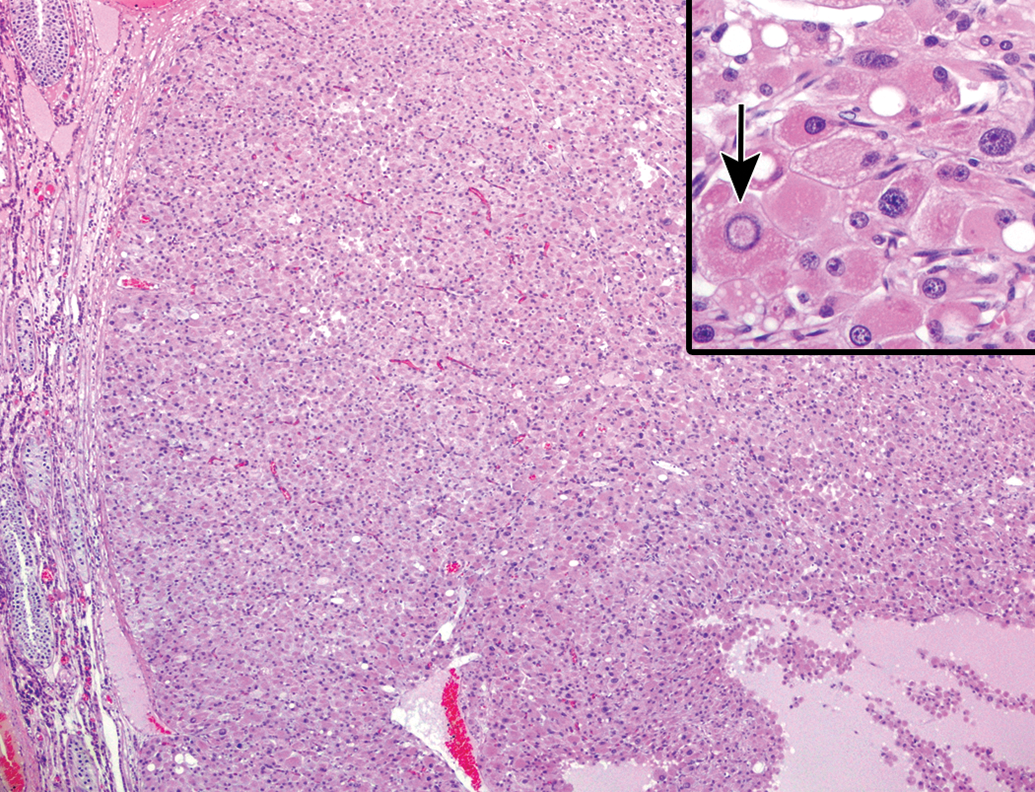
Leydig cell tumor in the testis of an aging microminipig at 6 years of age. The mass is well demarcated from the adjacent testicular tissue, but not encapsulated by fibrous connective tissues. Proteinaceous material is accumulated in the extracellular space. Inset: Higher magnification of the cells, which are of various sizes due to anisokaryosis, intranuclear inclusions (arrow), and cellular polymorphism. H&E. H&E indicates hematoxylin and eosin.
A single case of renal adenoma and uterine leiomyoma was present. Renal adenoma was not encapsulated but well circumscribed from the adjacent renal cortex (Figure 11). Multiple foci separated by thin strands of collagenous tissue comprised the lesion. Each focus was composed of a single layer of well-differentiated cuboidal epithelial cells with eosinophilic cytoplasm, and the cells lined papillary projections into the lumen. Mitotic figures were not observed. Uterine leiomyoma was located underneath the endometrium, replacing the normal myometrium in the lesion. The mass was well circumscribed and consisted of densely arranged interlacing bundles of spindle cells intermingled with a low level of vascularity, while collagenous tissues were scarce (Figure 12). The section stained with the silver impregnation method showed that each tumor cell was surrounded by thin bundles of reticular fibers, the so-called boxing image, and the tumor cells demonstrated strong immunostaining for α-SMA (Figure 12, insets). Nuclear pleomorphism was minimal and mitotic figures were rare.
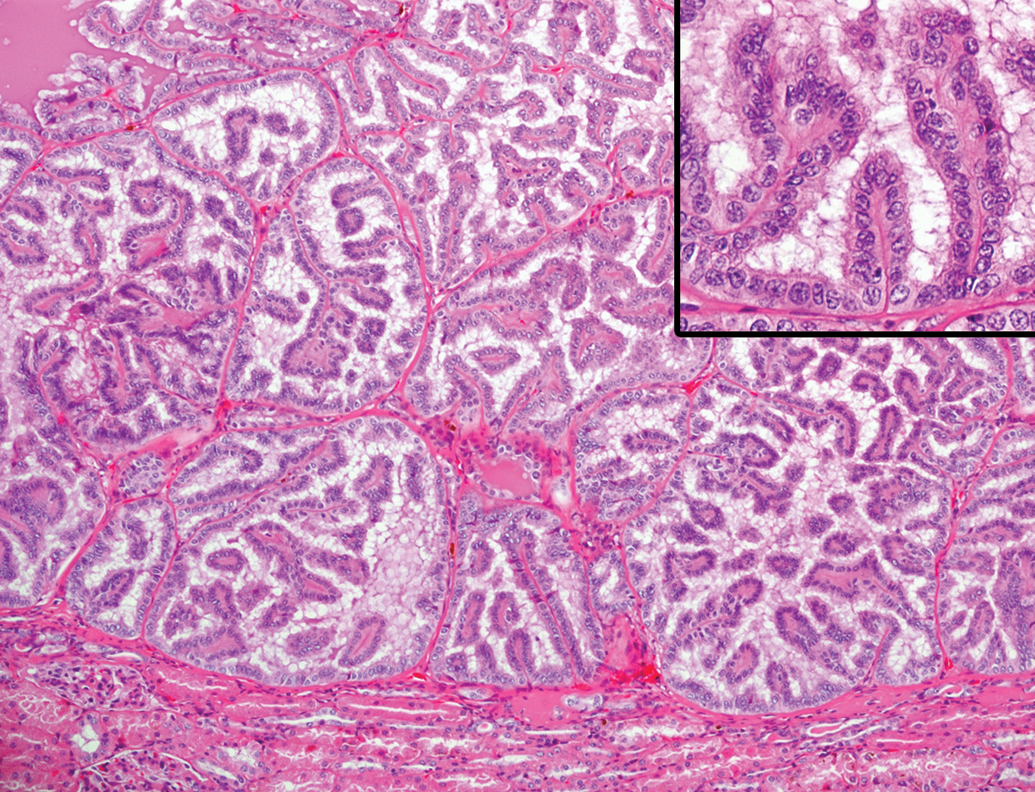
Renal adenoma in an aging microminipig at 7 years of age. The mass is composed of multiple circumscribed foci. Inset: Higher magnification of the foci, where the cells are well differentiated, showing papillary growth pattern. H&E. H&E indicates hematoxylin and eosin.
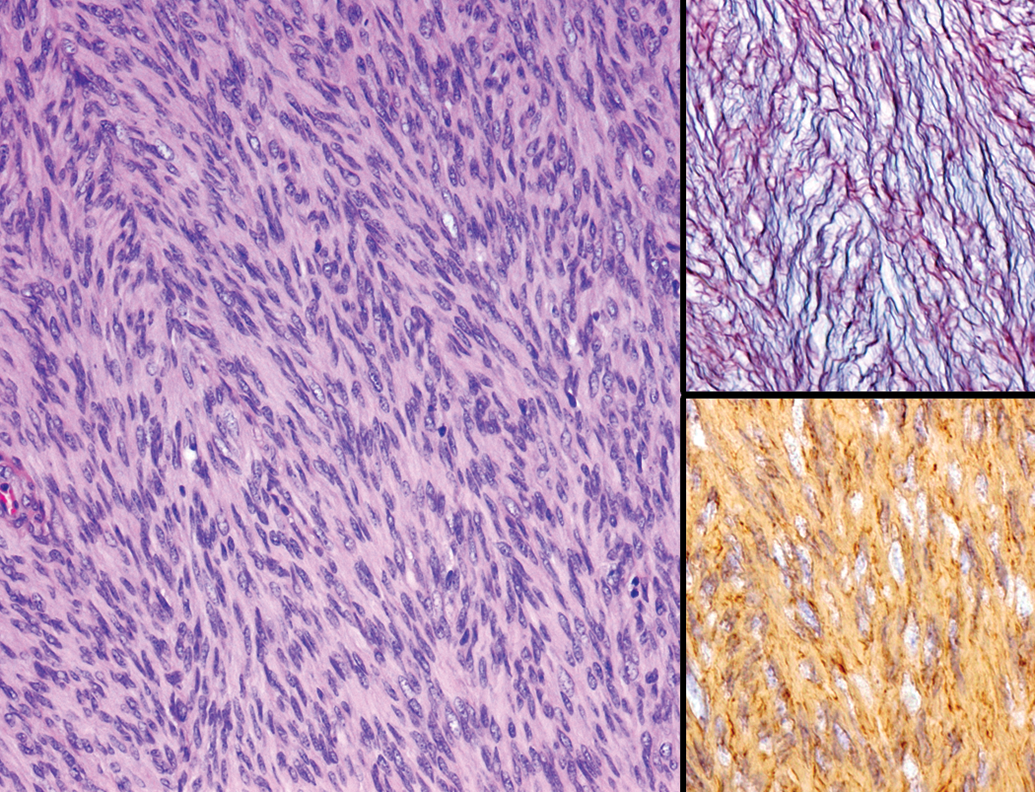
Leiomyoma in the uterus of an aging microminipig at 9 years of age. The mass is composed of densely arranged interlacing bundles of spindle cells intermingled with small amounts of vascularity. H&E. Inset (top): Each spindle cell is surrounded by thin bundles of reticular fiber, the so-called boxing image. Silver impregnation staining. Inset (bottom): The cells are positive for α-SMA; immunohistochemistry counterstained with hematoxylin. H&E indicates hematoxylin and eosin; α-SMA, α-smooth muscle actin.
Miscellaneous Changes
In the kidney, interstitial fibrosis (18 affected animals of 27 total examined animals) and glomerulosclerosis (21 of 27) were commonly observed in aging animals (Table 5, Supplemental Figure S4). Interstitial fibrosis stained bright blue with Masson’s trichrome and was present focally or multifocally in the cortex and focally to diffusely in the medulla, occasionally accompanied by mild mononuclear cell infiltration. The severity of the lesion varied between individuals. Segmental to global glomerulosclerosis was found multifocally, in a few to approximately half of glomeruli per cross section and was stained bright purple with PAS-H (Supplemental Figure S4, inset).
Incidence of Miscellaneous Changes in Aging Microminipigs.
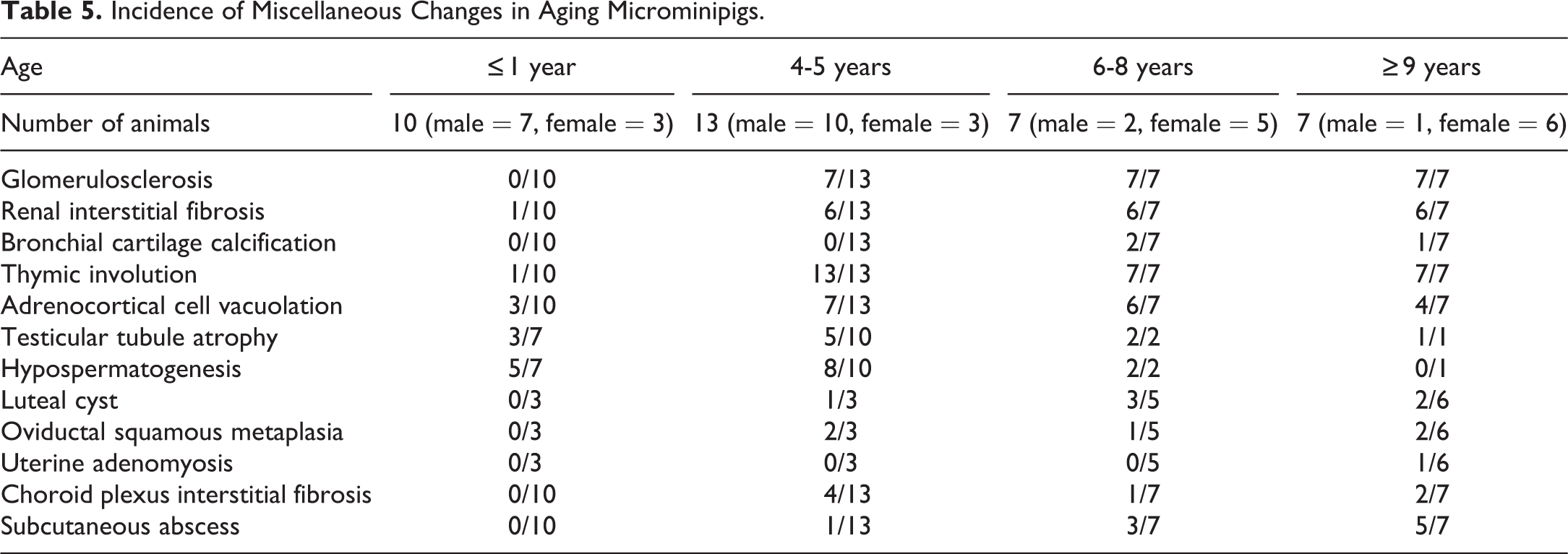
Thymic involution was macroscopically observed in both the cervical and thoracic portions of the thymus in all aging animals (27 of 27), while only 1 animal showed this change from the control group. In the involuting thymus at 4 to 5 years of age, there was a reduction in lymphoid cellularity, particularly in the cortex, and variable amounts of mature adipose tissue appeared between involuted lobules (Supplemental Figure S5). At 6 years of age and thereafter, the thymus parenchyma was replaced by mature adipose tissue containing scattered islands of small lymphocytes and small amounts of connective tissue.
In the adrenal gland, focal to multifocal cortical cell vacuolation was frequently observed in the internal part of the zona fasciculata (17 of 27, Supplemental Figure S6). The foci were well demarcated, and compression of adjacent cells was minimal. The cells were enlarged, containing numbers of small- to medium-sized clear vacuoles in the cytoplasm, and the nucleus occasionally became smaller, darker, and distorted in shape.
Tubular atrophy was occasionally present in the testes of all animals investigated, including control animals (11 of 20, Supplemental Figure S7). The number of affected tubules varied between individuals: a few tubules to approximately one-quarter of tubules per cross section, except for the testis with a Leydig cell tumor, in which almost all of the tubules were affected.
The ovarian cysts macroscopically observed in aging animals (6 of 14) were lined by several layers of polygonal luteal cells with abundant eosinophilic cytoplasm and small amounts of fibrous tissue; this was diagnosed as luteal cysts (Supplemental Figure S8).
Squamous metaplasia in the oviduct was focally or multifocally observed in several aging animals (5 of 14, Figure 13). The foci had a connection with normal oviductal columnar epithelium and were located in the lamina propria beneath the mucosa, with slight or mild mononuclear cell infiltration. Intercellular bridges and keratin filled lumens were occasionally evident within the foci.
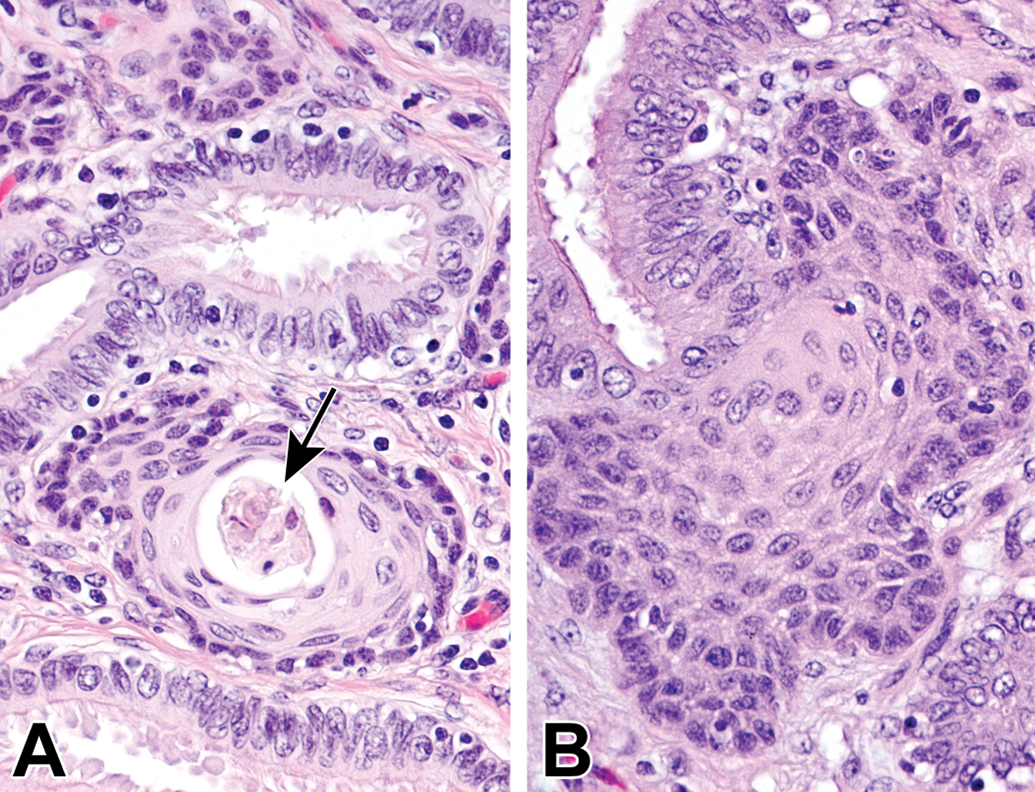
Squamous metaplasia in the oviduct of aging microminipigs. A, Squamous foci are present in the interstitium with mild mononuclear cell infiltration at 4 years of age. Keratinized cellular debris is accumulated in the lumen (arrow). B, Squamous focus has an obvious connection with the normal mucosal epithelium at 10 years of age. H&E. H&E indicates hematoxylin and eosin.
A single case of uterine adenomyosis was present in an animal at 9 years of age (Figure 14). The lesion was located within the myometrium and consisted of a number of uterine glands with highly columnar epithelium and a small amount of collagenous connective tissue. No evidence of atypia in glandular cells was observed.
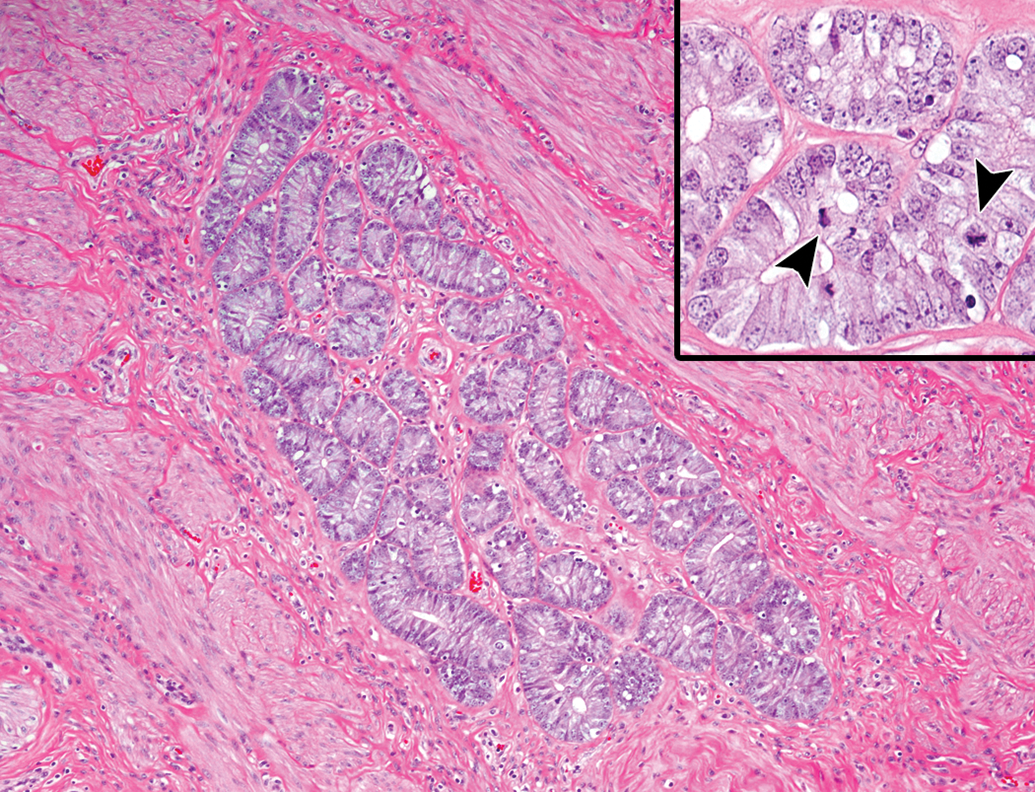
Adenomyosis in the uterus of an aging microminipig at 9 years of age. The clusters of uterine glands are present within the myometrium. Inset: Higher magnification of the glands. The glands consist of well-differentiated columnar cells with occasional mitotic figures. H&E. H&E indicates hematoxylin and eosin.
Interstitial fibrosis in the choroid plexus was occasionally observed in aging individuals (7 of 27, Supplemental Figure S9). The interstitial compartment was thickened with fine collagenous fibers and stained blue with Masson’s trichrome in varying degrees.
Individual data and the severity of the lesions, including other minor lesions, are presented in Supplemental Table 1.
Discussion
The present study illustrates the features and incidences of age-related histological changes observed in aging microminipigs, focusing on abnormal deposits of materials, arterial changes, and proliferative changes. This is the first study to describe the spontaneous age-related histological changes in microminipigs based on thorough histological investigations.
In minipigs, a number of instances detailing spontaneous histological changes in young to young adult minipigs have been reported, 25 –28 and some similarities and differences have been observed regarding the incidence of these changes compared with those observed in microminipigs. Mineralization is commonly observed not only within the prostate gland and the ovaries of Göttingen minipigs but also in the pituitaries of Göttingen minipigs and Yucatan minipigs. 25 –27 Vacuolation of the adrenal cortical cytoplasm is occasionally present in Göttingen minipigs, 25 –27 and tubular hypoplasia/atrophy is the most common incidental background finding in the testes of male Göttingen minipigs. 25 –27 Among all examined organs, the kidney exhibits the highest prevalence of inflammatory cell infiltration in Göttingen minipigs, Hanford minipigs, and Yucatan minipigs. 25 –28 Our present study suggests that these changes are also common in young and aging microminipigs. In Göttingen minipigs, only a single case or a very small number of cases of diffuse amyloidosis within the mandibular lymph node, Leydig cell hyperplasia and adenoma, and squamous metaplasia in the oviducts have been reported, 25 –27 and these were occasionally present in aging microminipigs. In contrast, necrotizing cholecystitis within the gallbladder is one of the most striking changes with fairly high incidence in Göttingen minipigs 25 ; however, this change was not observed in the present study.
Amyloidosis is a general term for a wide array of protein misfolding diseases caused by aggregation and deposition of abnormal fibrils in tissue, leading to organ dysfunction. 44 Pigs rarely develop amyloidosis, and there are only a few porcine cases reported so far. 45 –49 Those cases were identified as a consequence of reactive systemic amyloidosis, and underlying conditions were in association with bacterial infection, leukosis, and hyperimmunization. 45 –47,49 Most cases of amyloidosis in animals are of the reactive systemic type, and common sites of amyloid distribution are the kidney, liver, spleen, thyroid gland, adrenal gland, and small muscular arteries and arterioles in many organs. 50 –52 In this study, amyloid was deposited mainly in the renal medulla, thyroid gland, adrenal gland, and small arteries in the liver, spleen, and alimentary tract. Although no common underlying conditions were observed in the 4 animals with amyloidosis, the locations of amyloid deposition indicated that those microminipigs might have a reactive systemic type of amyloidosis. On the other hand, the localization of amyloid in the kidney is primarily glomerular in mouse and dogs, but medullary localization predominates often in cats and occasionally in cattle. 51,53 Glomerular amyloidosis is responsible for many cases of protein losing nephropathy in animals, causing notable proteinuria and uremia. In contrast, medullary amyloidosis is usually asymptomatic unless it results in papillary necrosis and is not grossly recognizable. 54 This observation was true of microminipigs exhibiting renal medullary amyloidosis, as none of the 4 animals showed signs of renal disorder. Further study will reveal the chemical and structural characteristics and species specificity of the amyloid deposited in microminipigs.
One of the most noticeable changes observed in the present study was hyaline droplet deposition in glomeruli, occasionally accompanied by fragmentation of the basement membrane and phagocytosis of the mesangial cells. According to the literature, hyaline droplets were present in various degrees in the mesangium of the glomeruli with frequent mesangiolysis in domestic pigs, with or without causative gross lesions. 55 –58 Those hyaline droplets stained positive for PAS, deep blue with PTAH, and red with acid-fuchsin orange-G stain, indicating that the droplets were a proteinaceous substance. 56,57 A study at the ultrastructure level revealed that hyaline droplets were phagocytosed by mesangial cells or located in the paramesangial areas. 58 The features illustrated in the previous reports in domestic pigs were quite similar to those observed in the present study in microminipigs. Since the deposits are formed in animals with or without causative lesions, in addition lack of clinical signs, the pathogenesis and significance of hyaline droplets in glomeruli remain unclear so far. 51,56
In addition to lipofuscin, various age-related neuronal intracytoplasmic inclusions have been reported in mammals, such as neurofibrillary tangles, granulovacuolar degeneration, Hirano bodies, Lewy bodies, hyaline (colloid) inclusions, and Lafora bodies. 32,59 These inclusions are classified simply by conspicuous histological and ultrastructural characteristics demonstrated by light and electron microscopy. 60 The intraneural materials in the present study had a fibrillar appearance and were stained deep blue with PTAH but did not react with PAS, Masson’s trichrome, PAM, or Gallyas-Braak staining. As far as we know, none of the inclusions reported so far have similar characteristics to those seen in the present study. Neuronal inclusions can also be classified into 3 categories according to the histopathological significance: normal cellular components, nonspecific neuronal dysfunction, and evidence of specific diseases, and many of the neuronal inclusions are assigned to more than 1 category. 60 In addition, there are important differences in the topological distribution of neuronal inclusions; some are widely distributed throughout the gray matter and others have preferred areas or specific regions. 60 Since the microminipigs with neuronal fibrillary inclusions had no neurologic manifestation and the lesion was located specifically in the nucleus cuneatus, the neuronal intracytoplasmic fibrillary inclusions in aging microminipigs can be classified as inclusions with nonspecific neuronal dysfunction; therefore, the lesion might incidental.
Arteriosclerosis is defined as the thickening, hardening, and loss of elasticity of the walls of arteries and is recognized as an age-related disease that occurs frequently in many animal species but rarely causes clinical signs. 61 Arterial medial calcification also occurs frequently in the arteries of animals and may occur spontaneously in animals of advanced age. Both lesions are especially prominent in the aorta but can involve coronary arteries and other elastic and muscular arteries. 61 –63 The present study revealed that sclerotic changes such as intimal thickening and intimal proliferation occurred commonly in aging microminipigs and had a predilection for the aorta, coronary artery, and basilar artery. Compared with the incidence of intimal changes, medial calcification in aging microminipigs was present in a few animals and observed only in the aorta and the coronary and pulmonary arteries. Atherosclerosis, the vascular disease of greatest importance in humans, occurs infrequently in animals and rarely leads to clinical disease. 61 Pigs, however, are known to be atherosensitive and the lesions develop naturally in aged pigs. 62 In addition, microminipigs have been used as a suitable animal model for human atherosclerosis because the lesion is pathophysiologically similar with that in humans and can develop as early as 3 months of age with hyperlipidemia induced by a high-fat and high-cholesterol diet. 64,65 Microscopically, the lipid-laden foam cells that may be either altered macrophages or smooth muscle cells are characteristically observed in the atherosclerotic lesion. 62 In the present study, no obvious evidence of lipid deposition was detected in any arteries throughout the organs investigated, except for a few macrophages present within the thickening intima in the aorta. This may indicate that atherosclerosis does not occur spontaneously in aging microminipigs fed a normal diet, while sclerotic changes in the arteries are naturally developed with age.
Generally, 2 types of non-neoplastic, nonregenerative proliferative lesions of hepatocytes have been identified in animals: hepatocellular hyperplasia and focus of cellular alteration. 66 Nonregenerative hepatocellular hyperplasia in domestic animals is termed nodular hyperplasia and is a lesion of unknown pathogenesis that occurs frequently in older dogs but uncommonly in other domestic species. 67 Similar lesions have been observed in swine, with a reported incidence of 4 per 100,000 animals. 68 These data were derived only from a slaughterhouse study conducted in young pigs, so the biology of this lesion remains unclear. Hyperplastic nodules do not produce clinical signs or adverse effects on liver function and are found as an incidental lesion. 67 The nodules range from 2 mm to 3 cm in diameter, and there is no evidence of hepatic damage, encapsulation, or increase in fibrous tissue within the nodule. 67 Hepatocytes in the nodules of hyperplasia are, generally, enlarged with nuclei of normal appearance, but there may be binucleated hepatocytes and extramedullary hematopoiesis within the nodules. 69 On the other hand, the focus of cellular alteration is a localized proliferation of hepatocytes phenotypically different from the surrounding hepatocyte parenchyma; the lesion can occur spontaneously in aged rats and other rodents and can be induced by chemical treatment. 66 The foci are subclassified based on phenotypic and tinctorial features of the predominant cell: basophilic, eosinophilic, amphophilic, clear cell, and mixed cell type. 66 In the present study, some proliferative foci of hepatocytes showed features almost identical to nodular hyperplasia, but others consisted of eosinophilic or clear hepatocytes with polyploidy, which had some similarities with those of cellular alteration in rodents. Limited information has been reported on hepatocellular hyperplasia in pigs; therefore, additional studies are needed to understand the nature of this change.
Uterine leiomyoma is well documented in miniature pet pigs and pot-bellied pigs; the age range is between 5 and 19 years. 29 –31 Miniature pet pigs and pot-bellied pigs possess a strong predisposition for developing uterine leiomyomas, with a strong association between increased age and development of neoplastic lesions. 30,31 In addition, uterine smooth muscle tumors in pot-bellied pigs are suggested to be a valuable animal model for studying human fibroids. 30
It is important to recognize differences between species, especially in choosing a model for specific research purposes. In particular, understanding the age of sexual maturity, growth, and age-related changes is necessary for pathologists to accurately interpret the experimental results in the animal under investigation. 20,70,71 The age of sexual maturity in microminipigs is reported to be 4.5 months in the male and 8 months in the female. 15,72 The growth curve reaches a plateau around 18 months of age in females and 24 months of age in males. 14 The present study examining age-related changes in microminipigs revealed that among the abnormal materials studied, amyloid materials, hyaline droplets in glomeruli, and fibrillar inclusions in neurons were present at 7 years of age and beyond, while arterial changes and proliferative lesions were observed at 4 years of age and above. Based on previous studies on sexual maturity and growth 15,72 and the present results, the degree of maturity and the incidence of age-related changes in microminipigs are estimated in Figure 15. We hope that the basic information obtained by this study will be helpful for future studies in microminipigs.
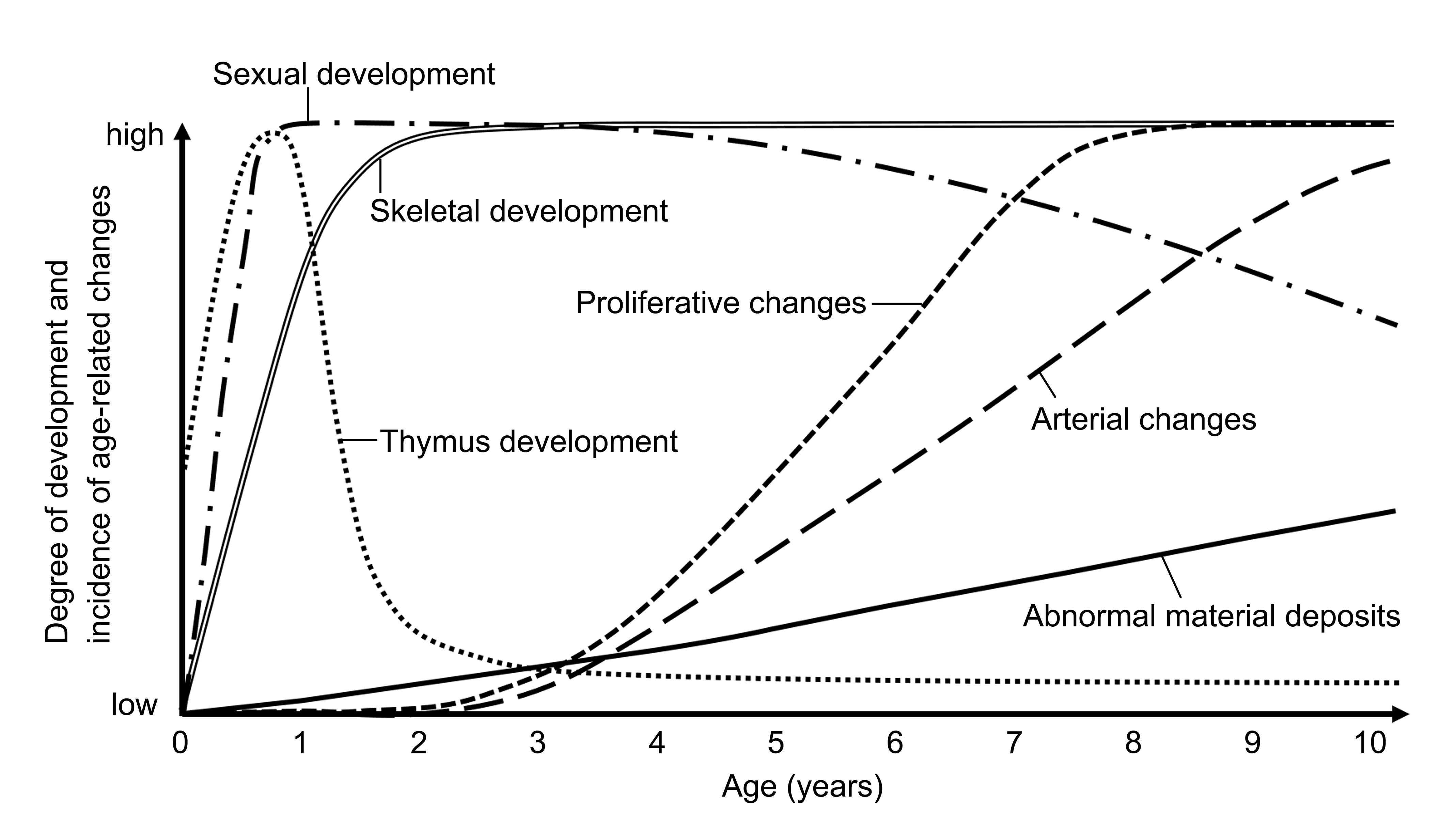
Degree of development and incidence of age-related changes observed in microminipigs. Horizontal axis represents age (in years), and vertical axis represents relative degree of development and incidence of age-related changes. Plotted curves correspond to different developmental features: thymus development (dotted line), sexual development (dash-dotted line), skeletal development (double line), incidence of abnormal material deposits (solid line), incidence of arterial changes (long-dashed line), and incidence of proliferative changes (short-dashed line).
In addition to the age-related changes observed in this study, various histopathological changes develop in senile man and animals, such as senile plaques and cerebral β-amyloid angiopathy in the brain, and atrophy in all of the body’s cells, tissues, and organs. 23,34 Further studies are necessary to fully understand the species-specific characteristics of aging microminipigs and to find whether microminipigs can be a useful animal model for human aging.
Supplemental Material
Supplemental Material, DS1-9_TPX_10.1177_0192623319861350 - Spontaneous Age-Related Histopathological Changes in Microminipigs
Supplemental Material, DS1-9_TPX_10.1177_0192623319861350 for Spontaneous Age-Related Histopathological Changes in Microminipigs by Akihisa Kangawa, Toshiaki Nishimura, Takashi Nishimura, Masayoshi Otake, Satoko Enya, Toshinori Yoshida and Masatoshi Shibata in Toxicologic Pathology
Footnotes
Acknowledgments
The authors thank Mr Toshiaki Fukumura (Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center) and breeding facility staff (Fuji Micra Inc) for taking good care of the animals.
Declaration of Conflicting Interests
The author(s) declared the following potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article: Toshiaki Nishimura and Takashi Nishimura are employees of Fuji Micra Inc.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
