Abstract
Microminipigs are becoming increasingly attractive alternatives for various experimental applications, such as general toxicology studies, owing to their manageable size. However, there are limited studies on the male reproductive organs of microminipigs, particularly on the histological aspects of sexual maturity. To clarify the development of male reproductive organs, 35 male microminipigs, aged 0 to 12 months, were used in this study. Histological and histomorphological evaluation was performed based on spermatogenic development, measurement of tubular structure in testes and epididymides, and histological progress of accessory glands. In addition, spontaneous testicular changes were quantitatively assessed. Histologically, male microminipigs sexually matured around 4.5 months of age, when spermatogenesis in testes and structural development in genital organs were completed. Spontaneous testicular changes occurred in all the animals investigated. Multinucleated giant cell was most commonly observed, followed by hypospermatogenesis and tubular atrophy/hypoplasia. However, the number of affected tubules was less than 1% in testes after 4.5 months of age, suggesting that the influence of these changes on evaluation of toxicity studies may be minimal. It is preferable to use sexually mature animals in toxicology studies; therefore, the information obtained by the present study will be helpful for future toxicity evaluations in microminipigs.
Keywords
Minipigs have become particularly useful animal models for toxicology and pharmacology studies because of their well-accepted physiological and other similarities to humans (Svendsen 2006; Bode et al. 2010). Several strains of minipigs (Göttingen, Yucatan, Sinclair, Hanford, and others) are used as nonrodent toxicology models. Among minipigs, microminipigs (Fuji Micra Inc., Shizuoka, Japan) are becoming increasingly attractive alternatives for various experimental applications because of their manageable size (approximately 10 kg at 6 months of age) compared to other minipigs in general (15–25 kg at 6 months of age; Kaneko et al. 2011). In spite of their promising potential as laboratory animals, there have been limited studies on their male reproductive organs, particularly the histological aspects of their sexual maturity.
It is preferable to use sexually mature animals in toxicology studies; hence, it is essential to understand the sexual maturity age of the animals utilized in these experiments. In particular, the stage of maturity in males is an important consideration in the examination of the testes, because histopathological characteristics of immature and peripubertal testes may be indistinguishable from chemically induced toxic damages (Lanning et al. 2002). Severe drug-induced germ cell depletion, resulting in negligible germ cell number, can mimic the immature testis. In peripubertal testes, spermatogenesis can still be immature (lacking certain germ cell populations) and degenerating germ cells that slough into the tubular lumen can be a normal finding (Dreef, Van Esch, and De Rijk 2007).
During the evaluation of testicular toxicity, qualitative examination is initially performed to recognize subtle abnormalities in testicular morphology. In certain circumstances, it may be advantageous to quantify testicular changes, because several parameters have been found to be sensitive and reliable in detecting toxicity effects. Among various quantitative analyses, cell counting in seminiferous tubules is generally the most appropriate tool to detect abnormal cell population in affected testes (Berndtson 1977; Russell et al. 1990; Creasy 1997). In addition, spontaneous testicular changes have also been reported in sexually mature nonrodent experimental animals, such as minipigs, beagle dogs, rabbits, and monkeys (Dincer et al. 2007; Thuilliez et al. 2014; Jeppesen and Skydsgaard 2015; Helke et al. 2016a, 2016b; Rehm 2000; Goedken, Kerlin, and Morton 2008; Morton et al. 1986; Sato et al. 2012; Luetjens and Weinbauer 2012). Due to the complex nature of spermatogenesis, it is important to acknowledge spontaneous changes in the animals under investigation and to recognize these lesions as potentially incidental or not, when determining their significance in toxicity studies (Lanning et al. 2002). Thorough testicular examination by cell counts and spontaneous findings in seminiferous tubules have not been reported in microminipigs as yet; therefore, reference data in this context would be helpful for future toxicity studies.
The objectives of the present study were to determine the stage of sexual maturity by histological and morphometrical examinations and clarify the incidence and severity of spontaneous testicular findings in the microminipig.
Materials and Methods
Animals
A total of 35 male microminipigs were used for this study. The animals were bred and raised in a closed colony at the Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center (Shizuoka, Japan). Fuji Micra Inc. had provided the microminipigs for research purpose. The animals were housed in pairs or in a group until around 6 months of age and then housed individually in pens of at least 0.54 m2 area/animal, under conventional conditions with 12-hr light and 12-hr dark cycle. The animals were weaned at 4 weeks of age and then fed a commercial diet (Marubeni Nisshin Feed Co., Ltd., Tokyo, Japan) as instructed by Fuji Micra Inc., at 2% to 3% of body weight/animal/day, with
Necropsy and Organ Weights
Before necropsy, all animals, except stillborn animals, received intramuscular injections of 0.1-ml/kg Dormicum (5 mg/ml midazolam, Astellas Pharma Inc., Tokyo, Japan) and 10-mg/kg ketamine (Fujita Pharmaceutical Co., Ltd., Tokyo, Japan) as anesthesia. After a veterinarian confirmed the unconscious state of the animals, they were euthanized by exsanguination (Close et al. 1996). Body weight was measured at necropsy. Testes, epididymides, body of prostate, seminal vesicle, and bulbourethral glands were taken from each animal, and their weights were measured. For paired organs, individual weights were recorded. Organ weights were expressed as absolute (in grams) and relative (to terminal body weight) weights.
Histological Examination
Testes and epididymides were fixed in formalin–sucrose–acetic acid solution (24% v/v of a 37% solution of formaldehyde, 72% v/v of a 5% sucrose solution, and 4% v/v acetic acid) for 2 weeks or in modified Davidson’s solution (30% v/v of a 37% solution of formaldehyde, 15% v/v ethanol, 5% v/v glacial acetic acid, and 50% v/v distilled H2O) for 1 week (Maita et al. 1999; Latendresse et al. 2002). Accessory organs were fixed in 10% neutral-buffered formalin. Testes were trimmed transversely through the middle of the major axis. For animals with large testes, the testis was randomly trimmed to half or three quarters of the original size and used for examination. Epididymides were trimmed transversely in the middle of caput, corpus, and cauda. Fixed samples were then dehydrated in graded series of ethanol and embedded in paraffin. Paraffin blocks were sectioned at 3 µm and stained with hematoxylin and eosin (H&E). Histological development was evaluated using light microscopy by focusing on the changes in cellular appearance and microscopic structures.
Morphometry in Seminiferous Tubules and Ductus Epididymis
The mean diameter of seminiferous tubules was estimated from 30 randomly chosen round tubules in each testis, independent of their cycle stage. In animals that were developed enough for the tubular lumen to be recognizable, the height of the seminiferous epithelium was determined from 10 round tubules at stages VII and VIII, which were readily identified with H&E. Stage classification was based on cellular associations (Kangawa et al. 2016). For example, germ cells from stage VII tubules were composed of type A spermatogonia, preleptotene and pachytene spermatocytes, and round and elongated spermatids, whereas those from stage VIII tubules were composed of type A spermatogonia, leptotene and pachytene spermatocytes, and round spermatids. In the epididymis, the diameter and epithelial height were measured from 10 round ducts in the caput (proximal, distal), corpus, and cauda (proximal, distal). All morphometric analyses were performed using microscope imaging software NIS Elements BR3.0 (Nikon, Tokyo, Japan).
Cell Count in Seminiferous Tubules
The nuclei present in all gonocytes and immature Sertoli cells were manually counted in 10 randomly selected round tubules from immature animals. The nuclei in mature Sertoli cells were irregular in shape; therefore, all germ cell nuclei and Sertoli cell nucleoli, which became apparent and were spherical in shape in mature Sertoli cells, were counted manually in the animals after prepuberty (Berndtson 1977). The 10 round tubules used in the measurement of epithelial height at stages VII and VIII were used for this counting. Tubules at stages VII and VIII were chosen because stages around sperm release are commonly used for quantitative evaluation of toxicity in rodent animals, and the most sensitive parameters were found by counting leptotene and pachytene spermatocytes and round spermatids during these stages (Creasy 1997). These crude counts were then corrected for section thickness and nucleus or nucleolus diameter based on the method described by Abercrombie (1946). Abercrombie’s formula is as follows:

For this purpose, 10 nucleus and/or nucleolus diameters were measured (per animal) for each cell type analyzed, using microscope imaging software NIS Elements BR3.0. Based on the true count of germ cells and Sertoli cells, Sertoli cell efficiency and meiotic index were calculated. The former was estimated from the number of round spermatids per Sertoli cell. The latter was measured as the number of round spermatids produced per pachytene primary spermatocyte.
Quantitative Evaluation of Spontaneous Changes in Seminiferous Tubules
The total number of tubular cross-section profiles of each testis was recorded, and the absolute number of tubule cross sections with features of testicular changes was manually counted. For animals with large testes, the testis trimmed to half to three quarters of the original size for histological examination was used for quantitative evaluation. Histological terminology was derived from published nomenclature used in dogs (Rehm 2000; Lanning et al. 2002; Goedken, Kerlin, and Morton 2008). In brief, the definitions of terminologies used in the present study are as follows: hypospermatogenesis, abnormal absence or decrease in some or all germ cells within individual tubules; tubular atrophy/hypoplasia, Sertoli-cell-only or almost Sertoli-cell-only tubule; multinucleated giant cells, multinucleate aggregates of germ cells; swollen spermatocytes, spermatocytes with abnormally enlarged cytoplasm; and karyomegaly, a germ cell with abnormally enlarged nucleus.
Statistical Analysis
All data are represented in terms of mean and standard deviation (
Results
Biometric Data
Body weights (mean ±
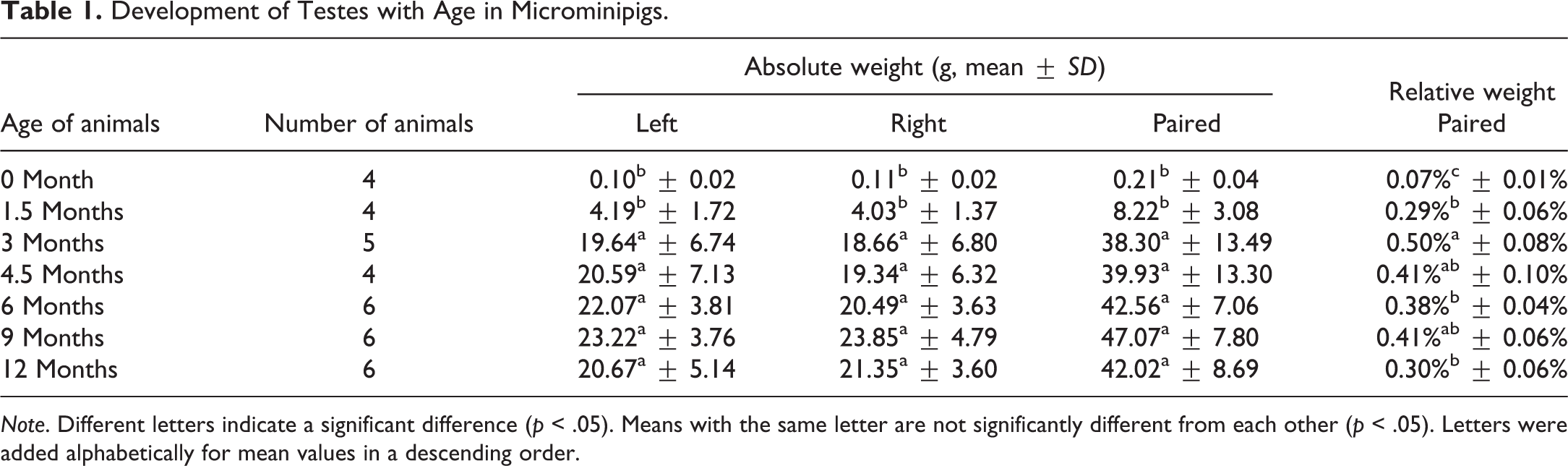
Development of Testes with Age in Microminipigs.
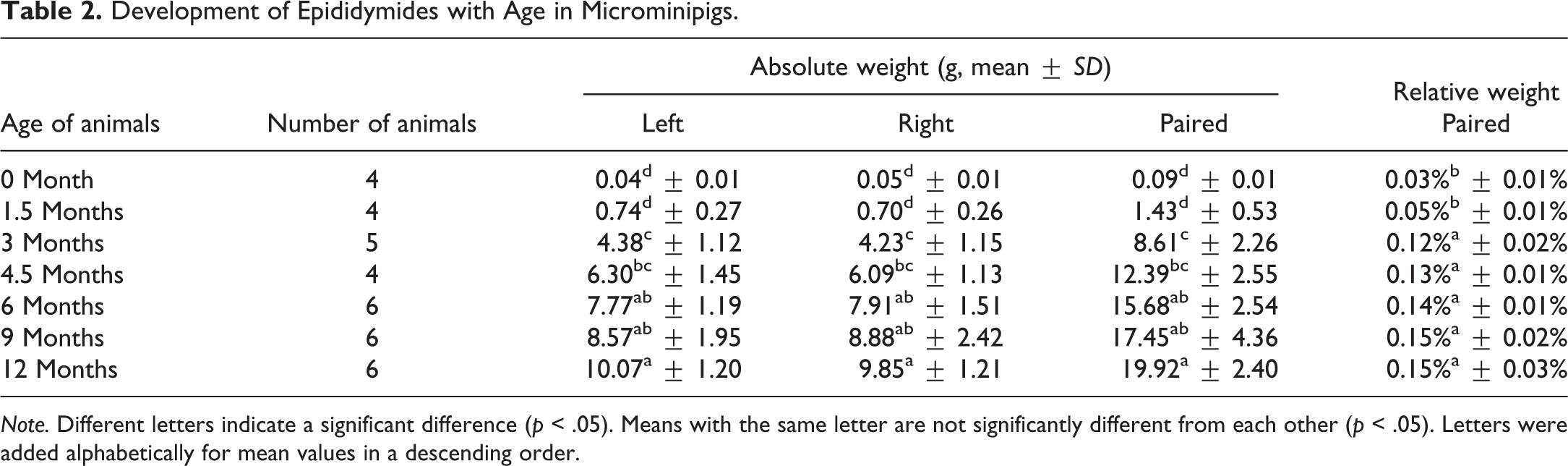
Development of Epididymides with Age in Microminipigs.
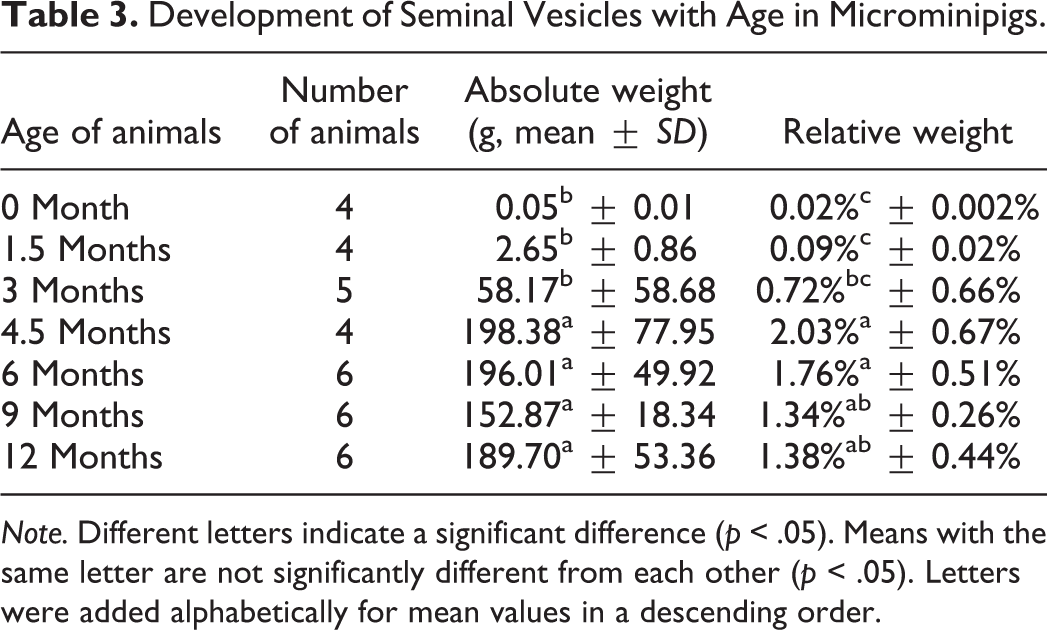
Development of Seminal Vesicles with Age in Microminipigs.
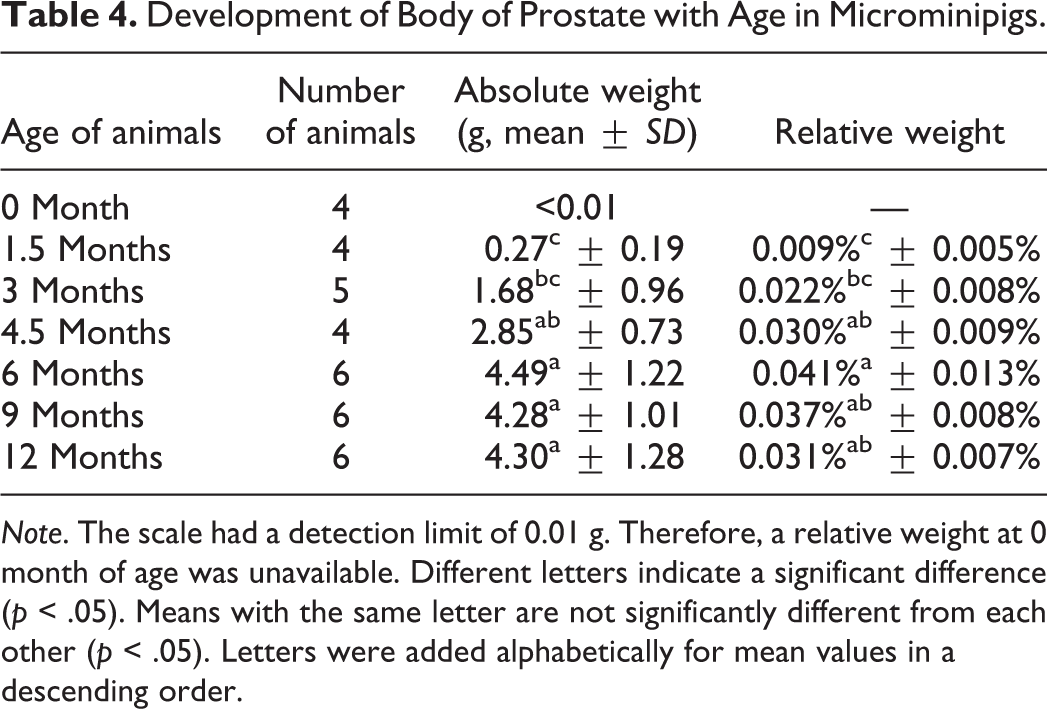
Development of Body of Prostate with Age in Microminipigs.
Development of Bulbourethral Glands with Age in Microminipigs.
Histological Findings
Testis
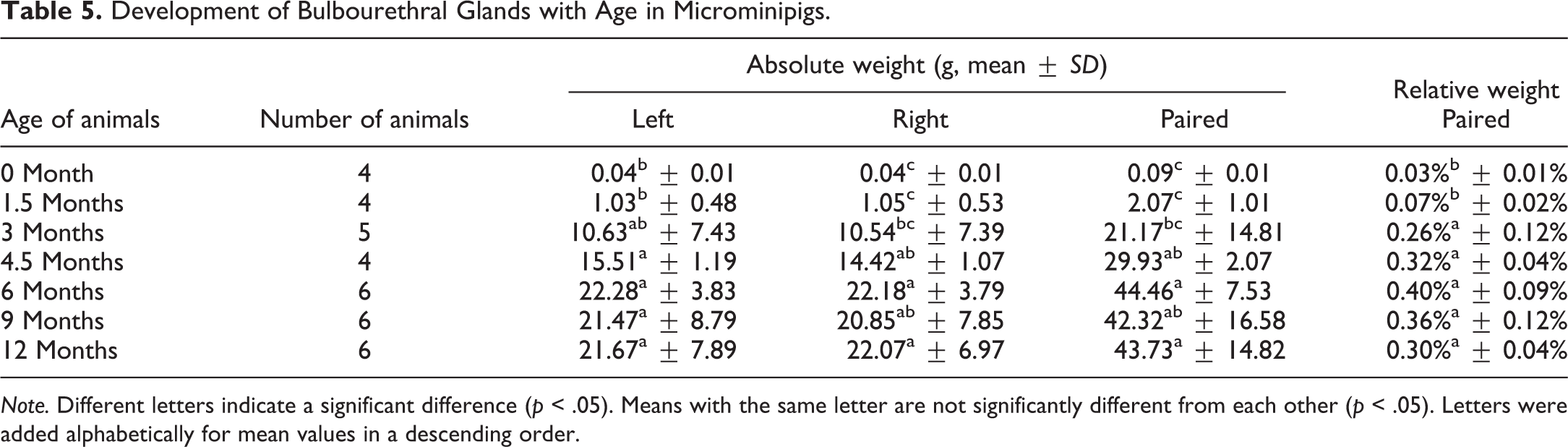
At 0 month of age, only Sertoli cells and gonocytes were observed in the seminiferous tubules (Figure 1A). Most of the cells located along the basement membrane had dark to crust-like-stained ovoid nuclei and were recognized as undifferentiated Sertoli cells. A few gonocytes, which had fine basophilic-stained round nuclei with a few chromatin granules and eosinophilic cytoplasm, were located along the basement membrane, and some were seen in the central region of the tubules. In addition, focal slight extramedullary hematopoiesis in the interstitium was observed in each animal at 0 month of age. Marked changes in the histological appearance of the testes were observed between ages 0 and 1.5 months. Most of the tubules at 1.5 months of age were composed of 2 to 4 layers of germ cells that were recognized as spermatogonia, spermatocytes, and round spermatids (Figure 1B). In a few tubules, there were a small number of elongated spermatids in the periphery of the lumen. Notably, the degree of tubular maturation differed depending on the location in the testis. Tubules situated in the internal part of the testis were composed of more layers (3–4) than those in the external part (2–3). As an incidental finding, focal mild hemorrhage with mononuclear cell infiltration was observed in 2 of the 4 animals at 1.5 months of age. Gonocytes were not seen at 1.5 months of age and thereafter. The number of each type of germ cells, including elongated spermatids, increased at age 3 months, and almost all seminiferous tubules throughout the testis showed active spermatogenesis (Figure 1C). At 4.5 months of age, all testes were composed of fully developed seminiferous tubules (Figure 1D). After 4.5 months, there were no apparent differences in the cell types and the density of seminiferous epithelium in the testis (Figure 1E and F). In contrast to seminiferous tubules, intertubular compartment, composed mainly of Leydig cells, showed few discernible differences with age, except that the area of Leydig cells at age 1.5 months appeared to be less prominent than those at other ages.
Histological development of testes in microminipigs. (A) At 0 month of age, seminiferous tubules were composed of immature Sertoli cells and gonocytes. Mitotic figures in gonocytes were occasionally observed (arrows). (B) At 1.5 months of age, seminiferous tubules were composed of spermatogonia, spermatocytes, spermatids, and Sertoli cells. Elongated spermatids were occasionally observed (arrowheads). (C) At 3 months of age, stage VII seminiferous tubules showed active spermatogenesis. At 4.5 months (D), 6 months (E), and 12 months (F) of age, stage VII seminiferous tubules showed fully developed spermatogenesis. Hematoxylin and eosin stain.
Epididymis
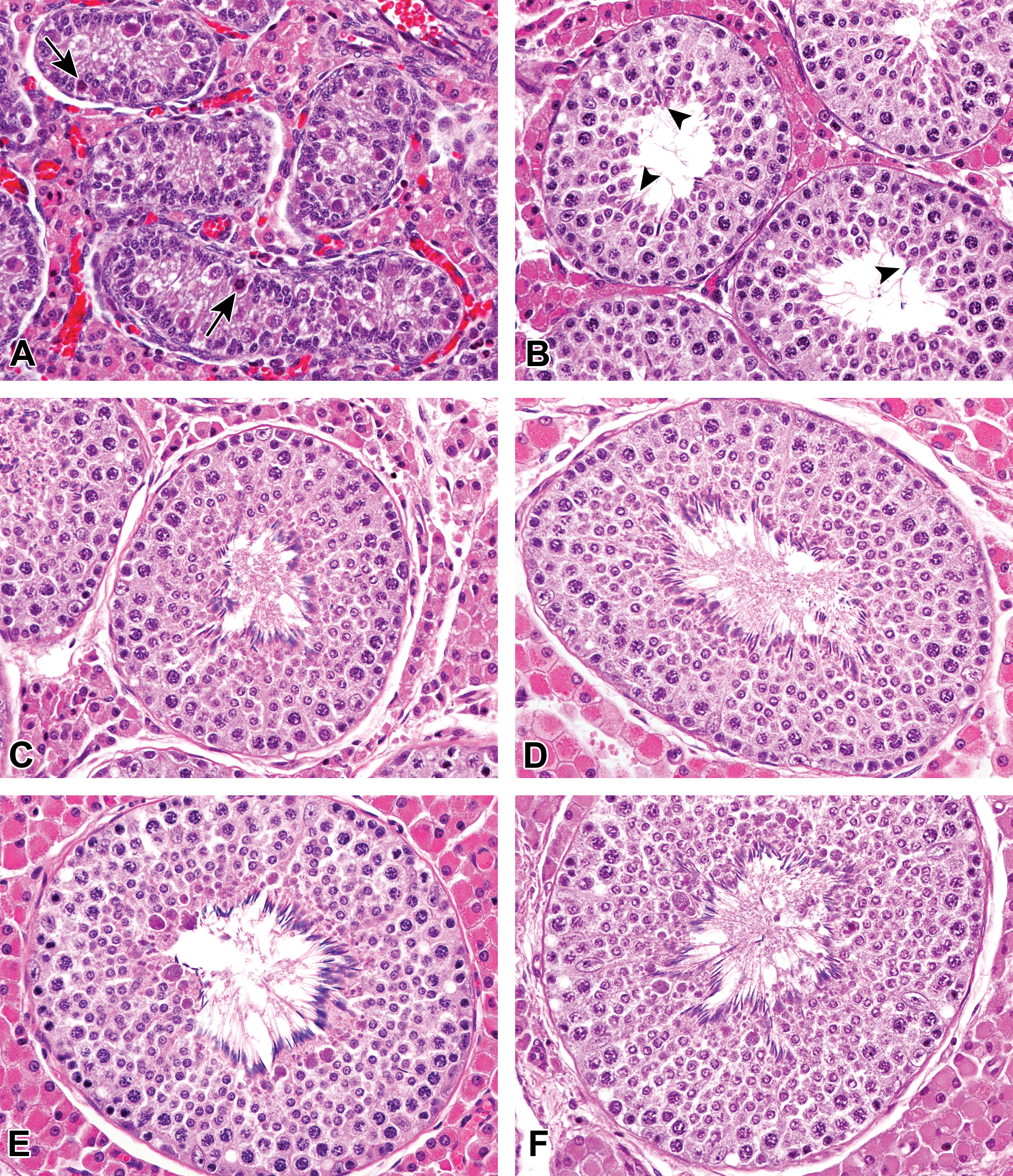
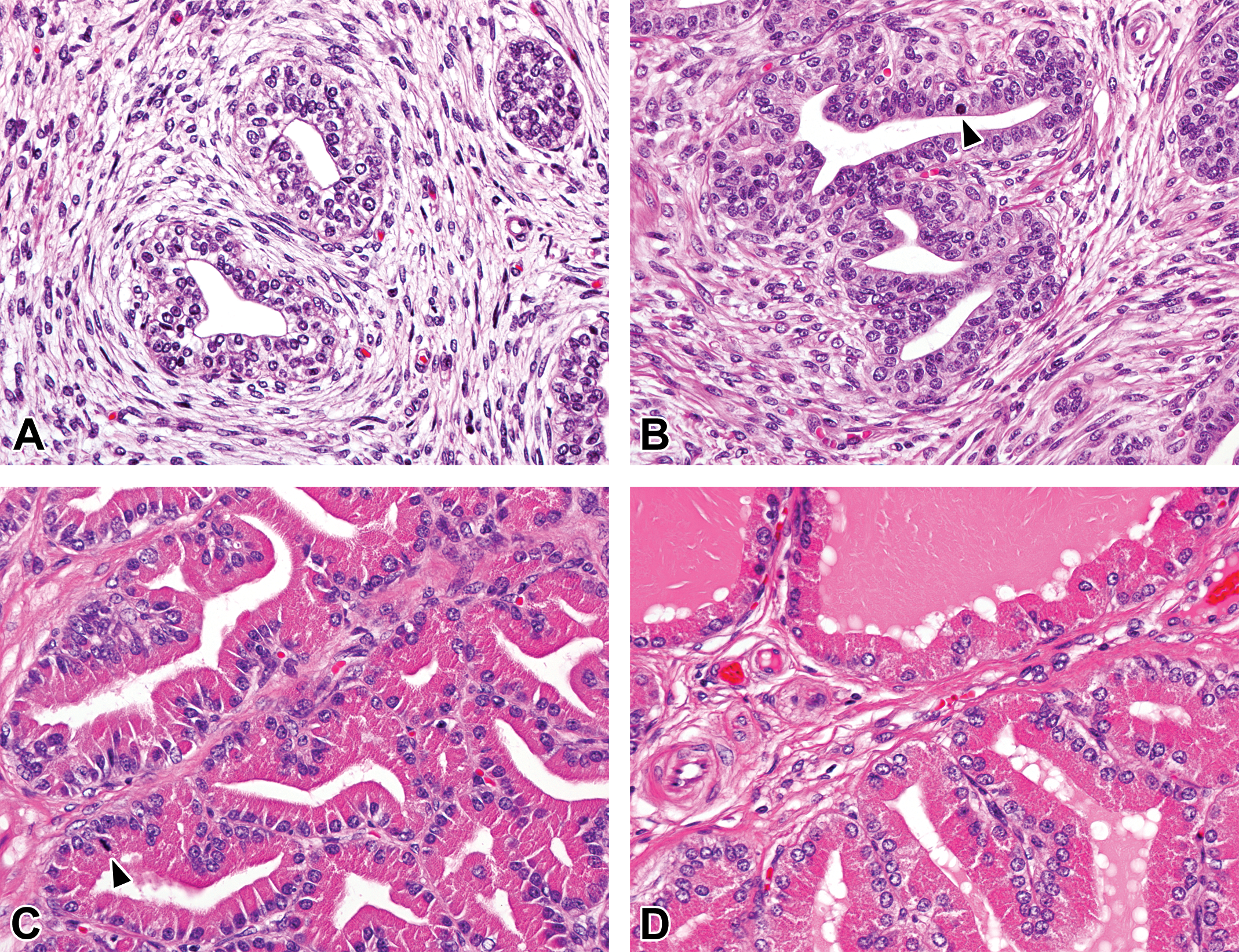
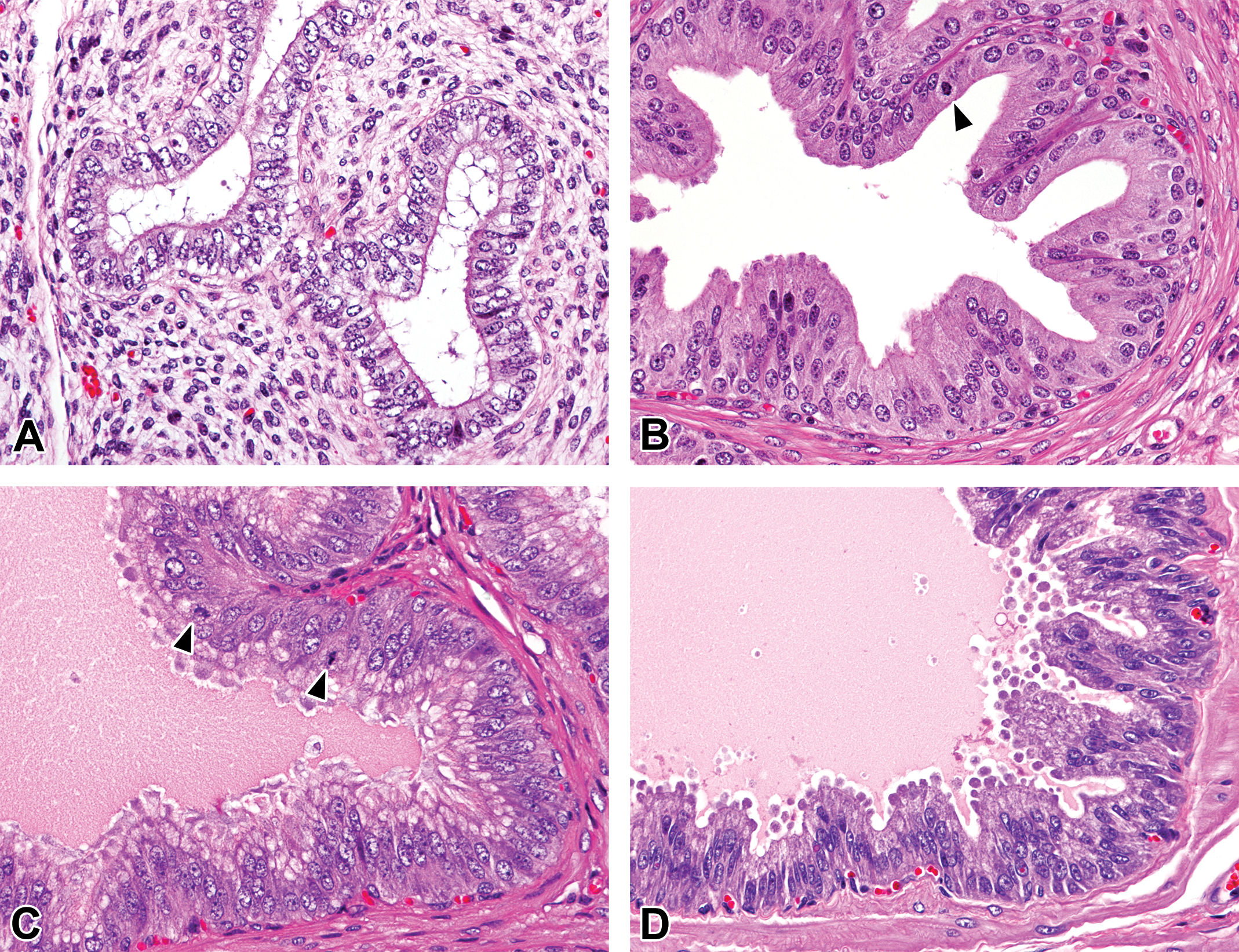
At 0 month of age, the ductus epithelium was composed of simple cuboidal to columnar epithelial cells. The epithelial cells had round to ovoid nucleus and possessed microvilli (stereocilia) in the caput, corpus, and cauda epididymis (Figures 2A, 3, and 4A). After 1.5 months of age, the ductus epididymis was lined with pseudostratified epithelium consisting of tall principal cells with long narrow nucleus and small basal cells with round to ovoid nucleus. The length of the microvilli increased at 1.5 months of age (Figures 2B, 3B, and 4B). Interestingly, nucleus of the principal cells in the caput epithelium gradually moved from the basal compartment at age 1.5 months to the central compartment at age 4.5 months (Figure 2). Spermatozoa first appeared in the ducts of the corpus and cauda epididymis at 3 months of age. Shed spermatocytes and spermatids were occasionally observed in the lumen of the ductus epididymis (Figure 4C). The height of the epithelium in the caput and corpus epididymis increased at 3 months and reached a maximum after 4.5 months of age. After 4.5 months of age, numerous spermatozoa with slight or without presence of shed germ cells were accumulated in the lumen of the caput (distal), corpus, and cauda epididymis (Figures 3D and 4D), and no apparent age-related histological changes were observed in the caput, corpus, and cauda epididymis.
Histological development of the proximal caput epididymis in microminipigs. (A) At 0 month of age, the ductus epithelium was composed of simple cuboidal to columnar epithelial cells with minute microvilli. (B) At 1.5 months of age, the ductus epididymis was lined with pseudostratified columnar epithelium with prominent microvilli. (C) At 3 months of age, the ductus epididymis was composed of highly columnar epithelium, showing almost mature appearance. (D) At 4.5 months of age, the fully developed epithelium consisted of ductus epididymis with triangular-shaped lumen. Hematoxylin and eosin stain.
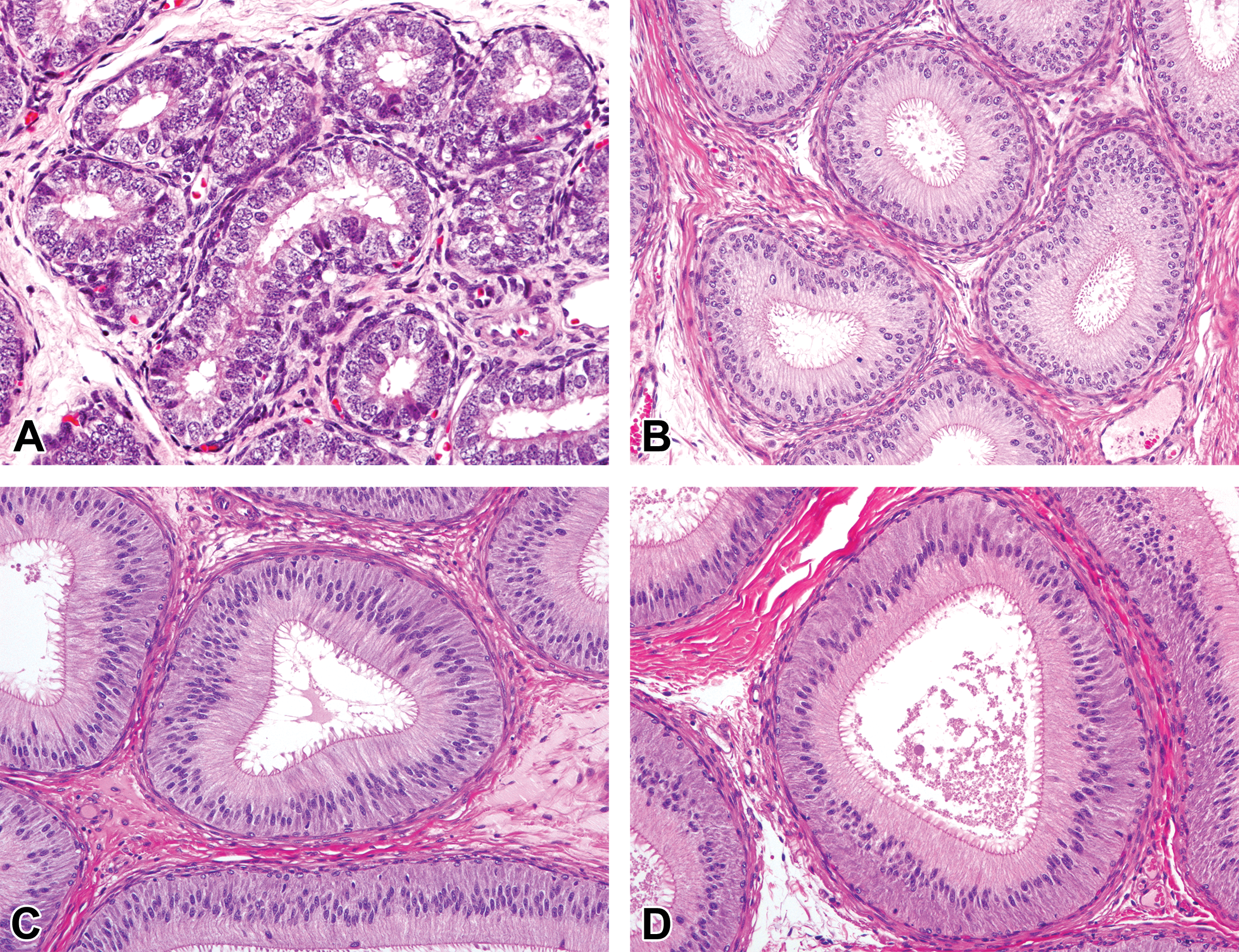
Histological development of the corpus epididymis in microminipigs. (A) At 0 month of age, the ductus epithelium was composed of simple cuboidal to columnar epithelial cells with minute microvilli. (B) At 1.5 months of age, the ductus epididymis was lined with pseudostratified columnar epithelium with prominent microvilli. (C) At 3 months of age, the ductus epididymis was composed of highly columnar epithelium with some mature sperms and cell debris in the lumen. (D) At 4.5 months of age, the fully developed epithelium consisted of ductus epididymis with abundant sperms in the lumen. Hematoxylin and eosin stain.
Histological development of the distal cauda epididymis in microminipigs. (A) At 0 month of age, the ductus epithelium was composed of simple columnar epithelial cells with immature microvilli. (B) At 1.5 months of age, the ductus epididymis was lined with pseudostratified epithelium with prominent microvilli. (C) At 3 months of age, mature sperms first appeared in the lumen. Cell debris and spermatocytes were also observed (arrows). (D) At 4.5 months of age, mature sperms accumulated in the lumen. Few cell debris were observed. Hematoxylin and eosin stain.
Prostate Gland
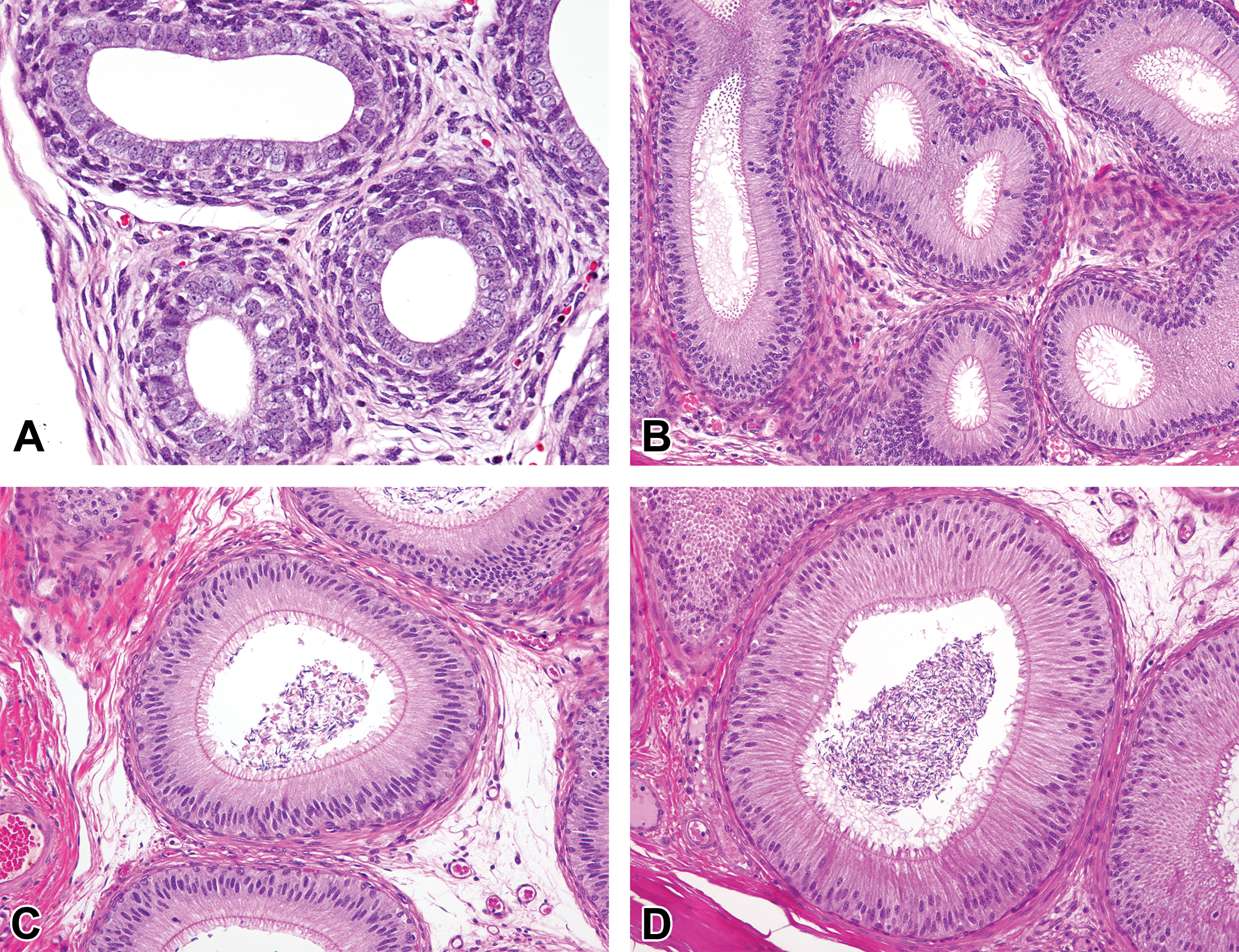
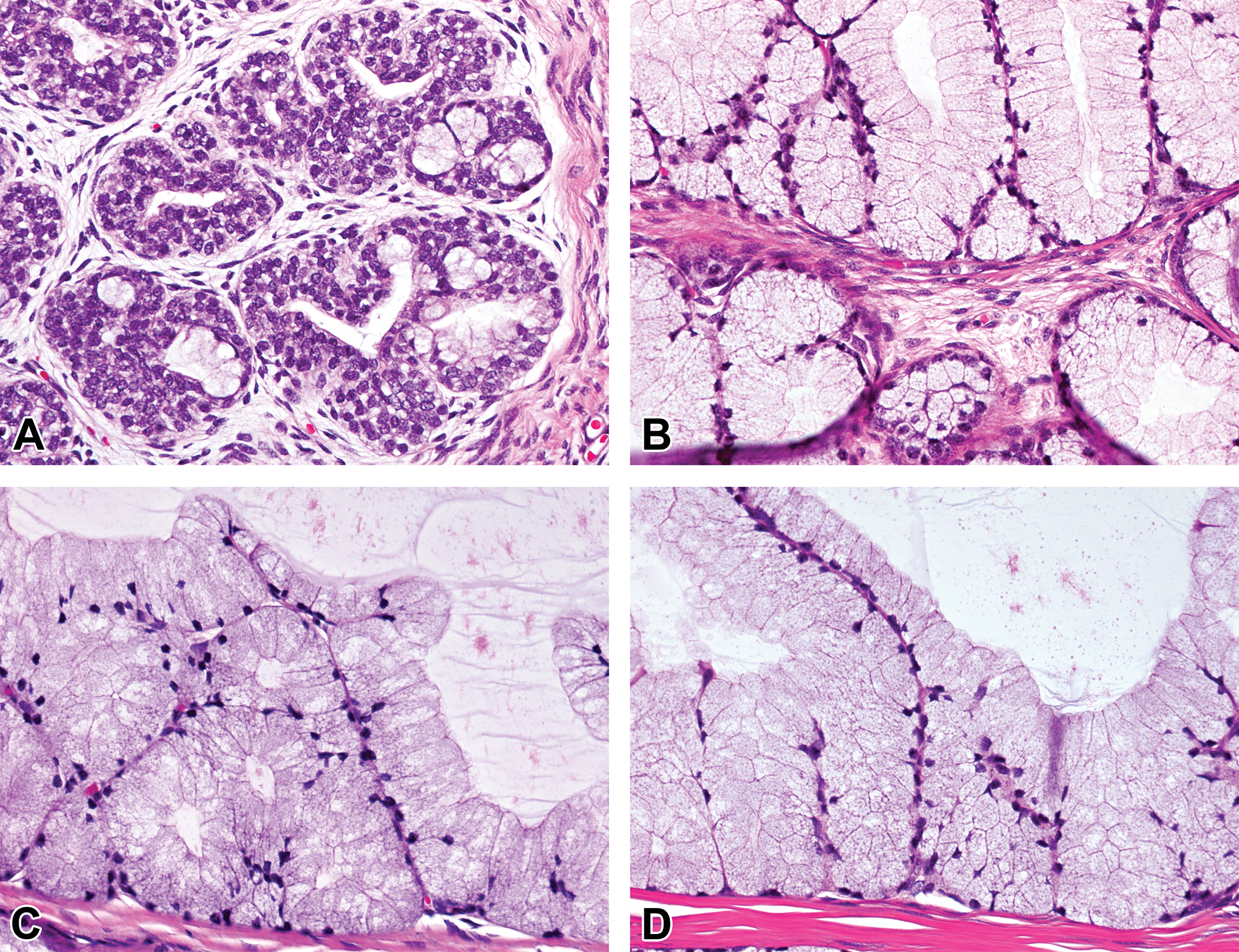
At 0 month of age, the prostatic tissue was composed mostly of connective tissue stroma with a few small glandular lobules surrounded by a number of spindle-shaped stromal cells. The glandular epithelium was lined by simple cuboidal to columnar cells with round nucleus and clear cytoplasm (Figure 5A). The lumen expanded and became irregularly shaped at age 1.5 months (Figure 5B). At 3 months of age, the number of the glands increased, and the cytoplasm of epithelial cells became highly eosinophilic with round nucleus at the basal side (Figure 5C). Mitotic figures were occasionally seen in the epithelial cells at ages 1.5 and 3 months. At 4.5 months of age, the size of the glands increased and eosinophilic secretion accumulated in most of the lumen (Figure 5D). After 4.5 months, there were no apparent differences in the histological appearance of the prostatic glands.
Histological development of prostate glands in microminipigs. (A) At 0 month of age, the prostatic tissue was mostly composed of connective tissue stroma with a few small glandular lobules. (B) At 1.5 months of age, the lumen was irregularly shaped with simple cuboidal to columnar epithelial cells. Mitotic figures were occasionally observed (arrowhead). (C) At 3 months of age, the number of glands increased and the epithelial cells showed highly eosinophilic cytoplasm. Mitotic figures were occasionally observed (arrowhead). (D) At 4.5 months of age, the size of glands increased and eosinophilic secretions accumulated in most of the lumen. Hematoxylin and eosin stain.
Seminal Vesicle
At 0 month of age, round to irregularly shaped small glands were surrounded by a large amount of connective tissue stroma. Pseudostratified columnar cells with minute microvilli lined the glandular lumen (Figure 6A). The size of the glands increased at 1.5 months of age. The epithelial cells varied in height with abundant cytoplasm, prominent microvilli, and ruffled luminal structures (Figure 6B). At 3 months of age, secretions from epithelial cells apparently became active because most of the lumen was filled with eosinophilic fluid (Figure 6C). Mitotic figures were occasionally observed in the epithelial cells at ages 1.5 and 3 months. After 4.5 months of age, almost all glands were filled with eosinophilic secretions (Figure 6D).
Histological development of seminal vesicles in microminipigs. (A) At 0 month of age, round to irregularly shaped small glands were surrounded by connective tissue stroma. (B) At 1.5 months of age, pseudostratified microvillous columnar cells lined the ruffled glandular lumen. Mitotic figures were occasionally observed (arrowhead). (C) At 3 months of age, the glands were lined by columnar epithelium with prominent microvilli, showing active secretion. Mitotic figures were occasionally observed (arrowheads). (D) At 4.5 months of age, the glands were filled with eosinophilic secretion. Hematoxylin and eosin stain.
Bulbourethral Gland
At 0 month of age, small glands were mainly composed of cuboidal cells with scant cytoplasm and surrounded by thin connective tissues. There were a few round to cuboidal cells with clear cytoplasm in some acini (Figure 7A). At 1.5 months of age, the glandular epithelial cells became highly columnar with abundant and slightly basophilic cytoplasm and thin nucleus on the basal edge of the cells (Figure 7B). The appearance of the cells did not seem to change after 1.5 months; however, the lumen of the glands expanded at 3 months of age and thereafter (Figure 7C and D).
Histological development of bulbourethral glands in microminipigs. (A) At 0 month of age, a few round to cuboidal cells with clear cytoplasm were observed in some glands. (B) At 1.5 months of age, the glandular epithelial cells were highly columnar with abundant and slightly basophilic cytoplasm. At 3 months (C) and 4.5 months (D) of age, the lumen of the glands expanded, with well-developed cellular composition. Hematoxylin and eosin stain.
Morphometry in Seminiferous Tubules and Ductus Epididymis
The parameters of the testis and epididymis morphometry are shown in Figures 8 and 9. The mean diameter of the seminiferous tubules increased markedly from 0 to 3 months of age and then reached a plateau at around 220 to 230 μm after 6 months (Figure 8). The lumen of the seminiferous tubules was hardly recognizable at 0 month but became apparent after 1.5 months of age (Figure 1). The epithelial height increased from 0 to 3 months and then reached a plateau at 70 to 75 μm in stage VII and 75 to 85 μm in stage VIII tubules. The mean diameter of the caput (proximal and distal) and corpus epididymides gradually increased until 4.5 months of age, with no substantial differences thereafter (Figure 9). The proximal cauda epididymis increased in diameter from 0 to 4.5 months and 9 to 12 months of age. The mean diameter of the distal part of cauda epididymis increased markedly from 0 to 4.5 months and steadily increased after 6 months of age to reach approximately 1,200 μm at 12 months. The mean height of the ductus epididymis epithelium, except distal part of the cauda, increased until 3 months of age, of which the proximal caput was almost twice as high as the others after 4.5 months. In contrast, there was little change with age in the mean height of the epithelium of the distal part of cauda epididymis.
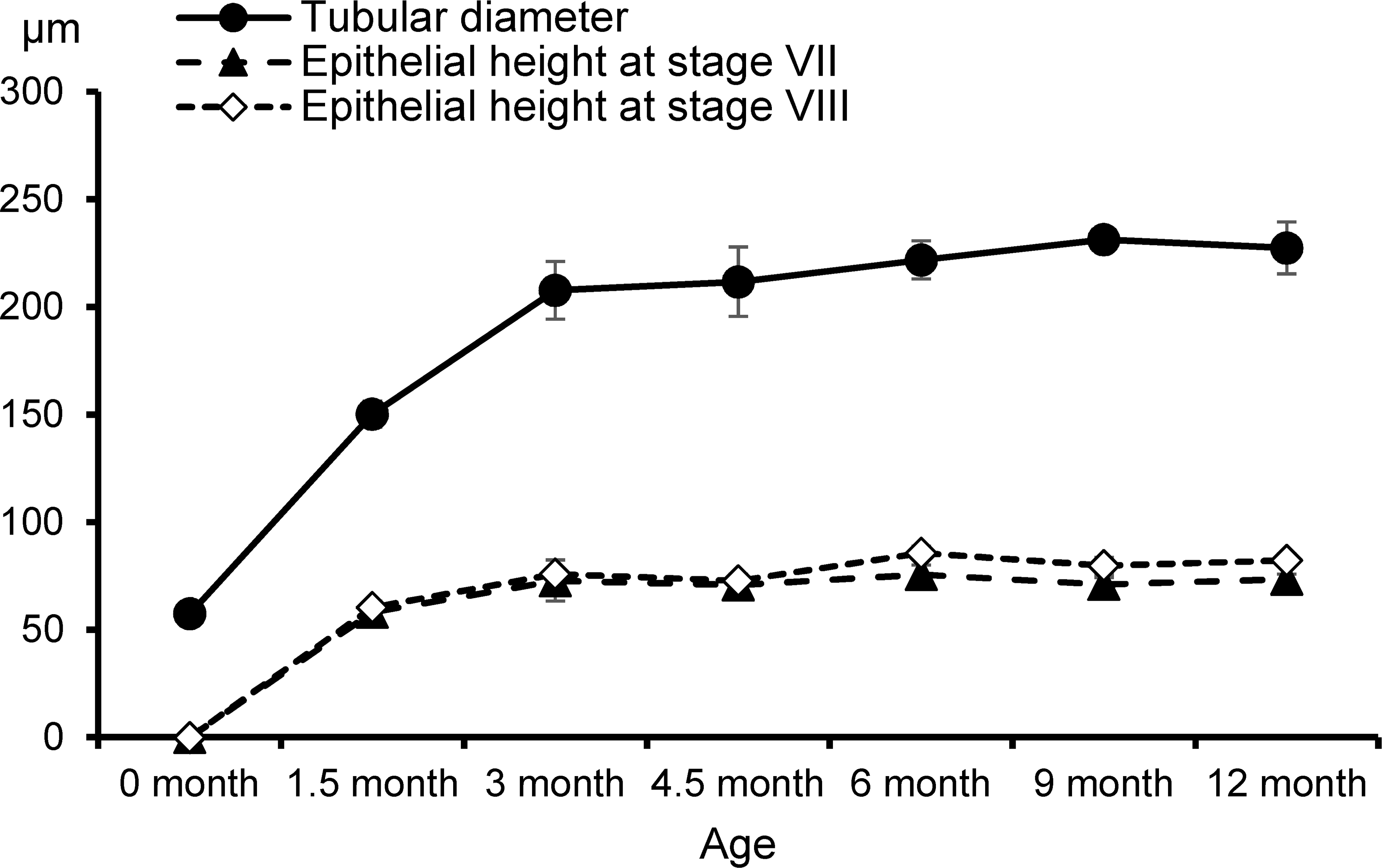
Development of seminiferous tubules indicated by tubular diameter and epithelial height at stages VII and VIII in microminipigs.
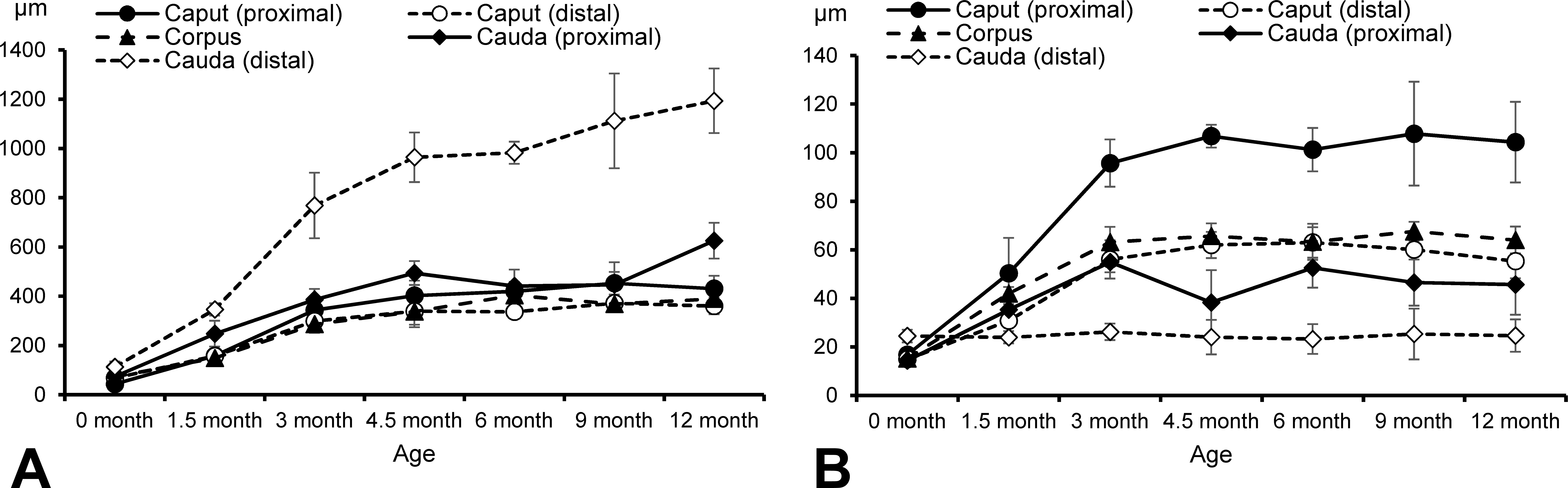
Development of epididymides in the caput, corpus, and cauda indicated by ductus diameter (A) and epithelial height (B) in microminipigs.
Cell Count in Seminiferous Tubules
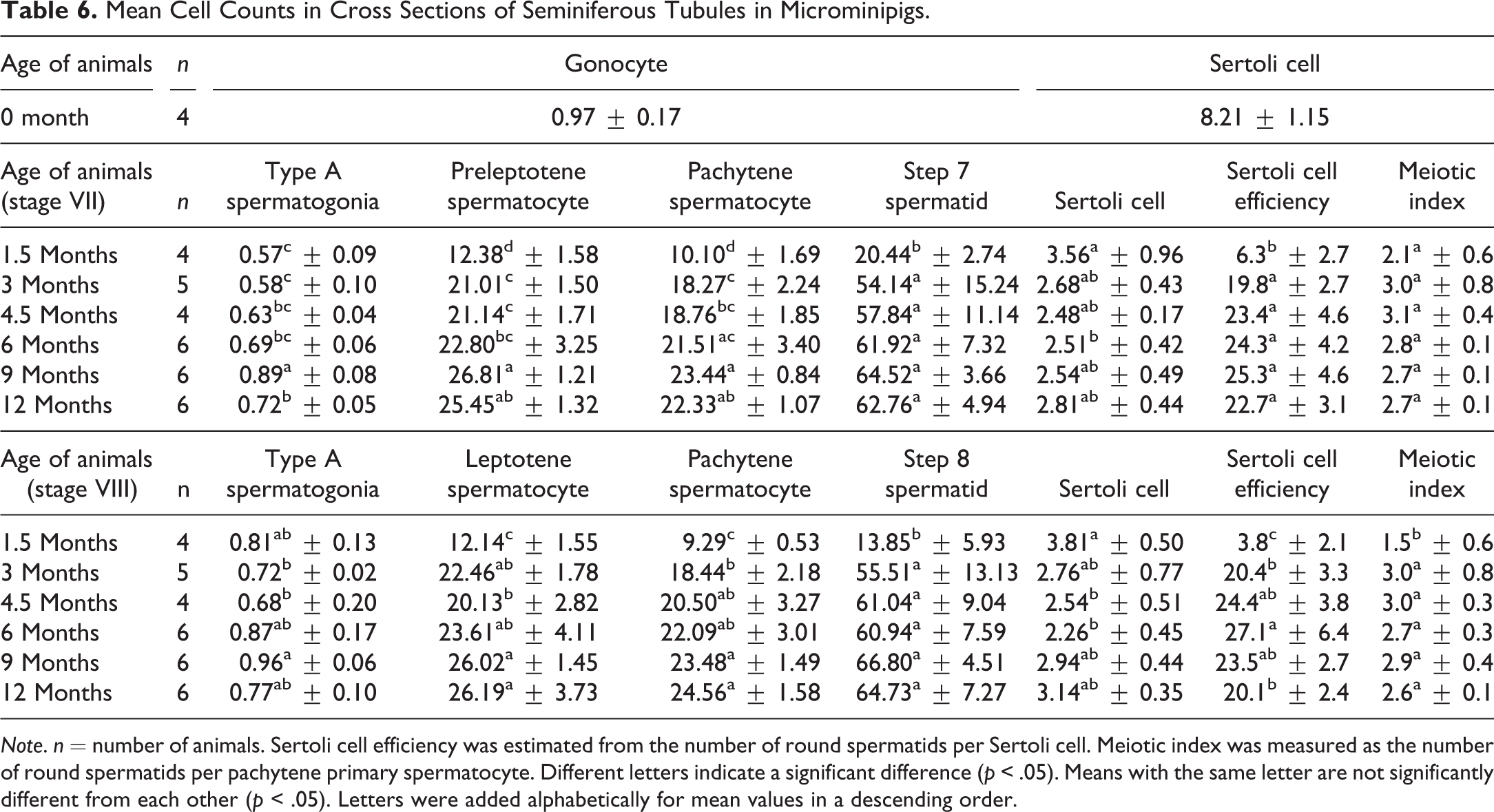
Cell counts of each type within the seminiferous epithelium at different ages in seminiferous tubules are shown in Table 6. At 0 month of age, nucleoli in Sertoli cells were inconspicuous due to immaturity, so that diameters and numbers of round nuclei were measured for a true cell count calculation. The number of gonocytes was only one eighth of that of Sertoli cells at 0 month. Staging of the seminiferous epithelium cycle was applicable in all animals at 1.5 months and over, even though fully active spermatogenesis throughout a testis occurred only from 4.5 months, as mentioned earlier in histological findings. Significant increases in the number of preleptotene, leptotene, and pachytene spermatocytes and spermatids in stages VII and VIII tubules were observed between 1.5 months and 3–12 months. Although each cell count was not fully statistically significant, the mean number of type A spermatogonia, spermatocytes, and spermatids appeared to increase until 9 months of age in both stages. Sertoli cell efficiency in stages VII and VIII tubules increased significantly between 1.5 months and 3 to 12 months, and the means were more than 20 in both stages beginning at 4.5 months. The meiotic index in stage VIII was significantly lower at 1.5 months than at 3–12 months; however, no significant difference was observed in stage VII.
Mean Cell Counts in Cross Sections of Seminiferous Tubules in Microminipigs.
Quantitative Evaluation of Microscopic Changes in Seminiferous Tubules
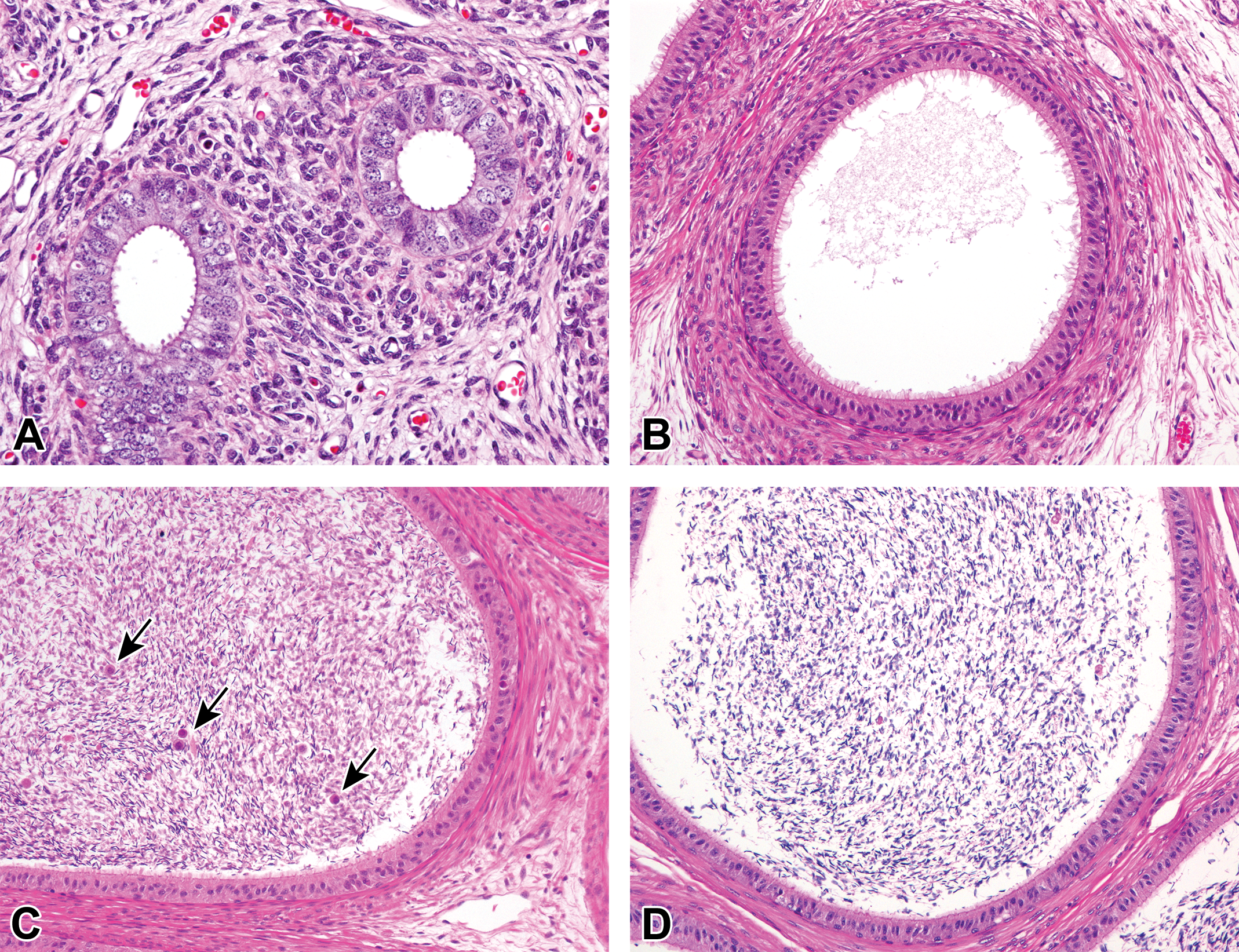
The animals from 1.5 months of age were used for this histopathological evaluation because testicular composition was incomplete and spontaneous changes were unrecognizable at 0 month. The number of tubular cross sections investigated per testis ranged from 1,756 to 5,650 with a mean value of 2,912 tubules per testis between 1.5 and 12 months. The results are shown in Table 7. Hypospermatogenesis was commonly observed from 3 months of age, occurring in 84% of the animals (26 of 31 pigs). Of these 26 animals, 23 (88%) had changes in bilateral sides of the testes. Absence of round and/or elongated spermatids was most commonly observed in testes with hypospermatogenesis (Figure 10). Obvious hypospermatogenesis was not seen at 1.5 months of age because spermatogenesis was incomplete in the testes and lack of spermatids in the tubule was commonly observed. The extent of hypospermatogenesis was maximum of 0.54% at 3 months and minimum of 0.14% at 9 months of age. Tubular atrophy/hypoplasia was seen in 48% of the animals (15 of 31 pigs) and occurred bilaterally in 5 of the 15 pigs (33%; Figure 11). At 1.5 months of age, atrophic/hypoplastic tubules were not observed in the testes examined. At ages 3 and 4.5 months, only 1 animal each showed tubular atrophy/hypoplasia. The incidence increased with age to 83% (5 of the 6 pigs) at ages 9 and 12 months. In addition, there was an age-dependent increase in the extent of tubular atrophy/hypoplasia, with maximum of 0.29% tubules affected at 12 months of age. Multinucleated giant cells occurred at any age in 94% of the animals (29 of the 31 pigs; Figures 12 and 13). Of these 29 pigs, 26 (90%) exhibited the changes bilaterally. Multinucleated giant cells sometimes existed in the tubules with hypospermatogenesis and tubular atrophy/hypoplasia. All animals from ages 1.5 to 9 months exhibited tubules containing multinuclear germ cells, whereas 4 of the 6 animals showed this change at 12 months of age. The extent peaked at 2.47% at 1.5 months and was minimum of 0.14% at 9 months of age. Other histological changes were less severe than those before mentioned. Swollen spermatocytes were observed in 19% of the animals (6 of the 31 pigs; Figure 13), mainly occurring in young animals aged 1.5 and 3 months. Tubules with karyomegaly were seen in 39% of the animals (12 of the 31 pigs; Figure 14). The incidence was higher in ages 1.5 and 3 months and gradually declined to 0% at 12 months. The extents of both swollen spermatocytes and karyomegaly were quite minimal and less than 0.1% in all ages examined.
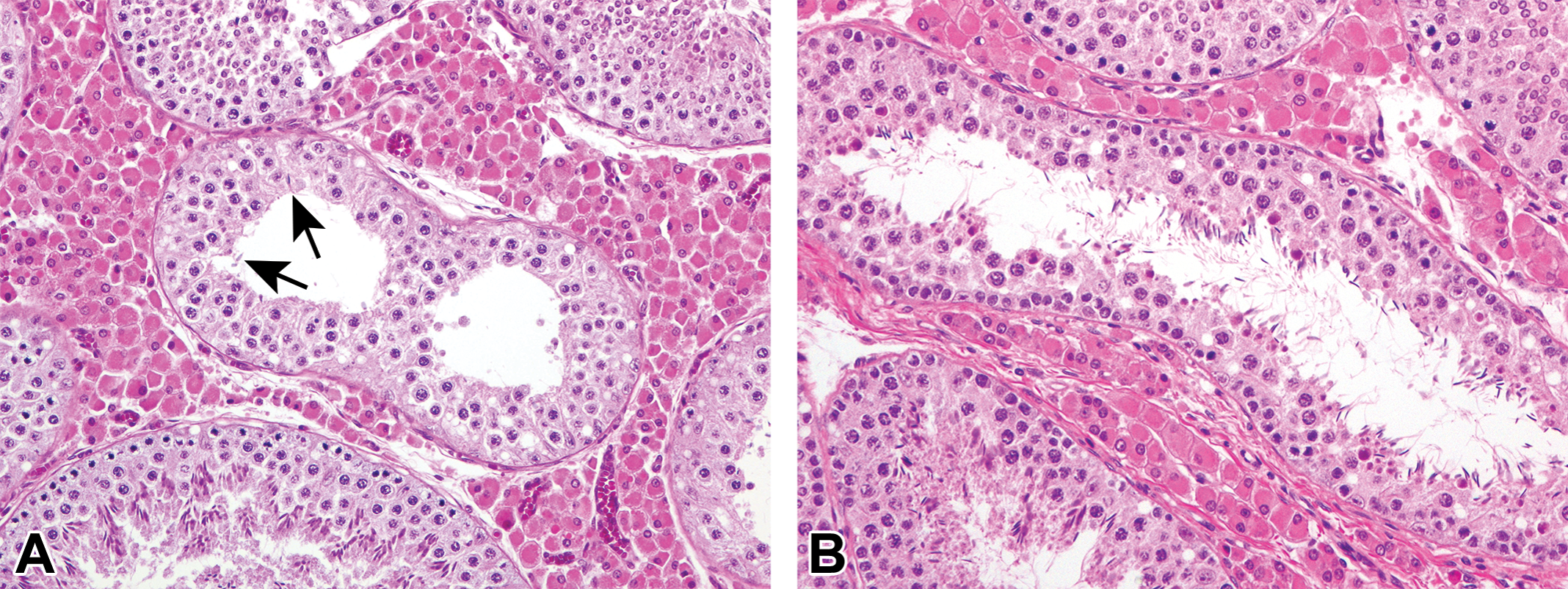
Testicular tubules with hypospermatogenesis that showed depletion or abnormally reduced number of germ cells. (A) Tubules showing reduced number of round spermatids and pachytene spermatocytes. A few elongated spermatids were seen in the luminal part of the tubules (arrows), at 9 months of age. (B) Tubules lacked round spermatids, although there were moderate number of elongated spermatids, residual bodies, and pachytene spermatocytes at 4.5 months of age. Hematoxylin and eosin stain.
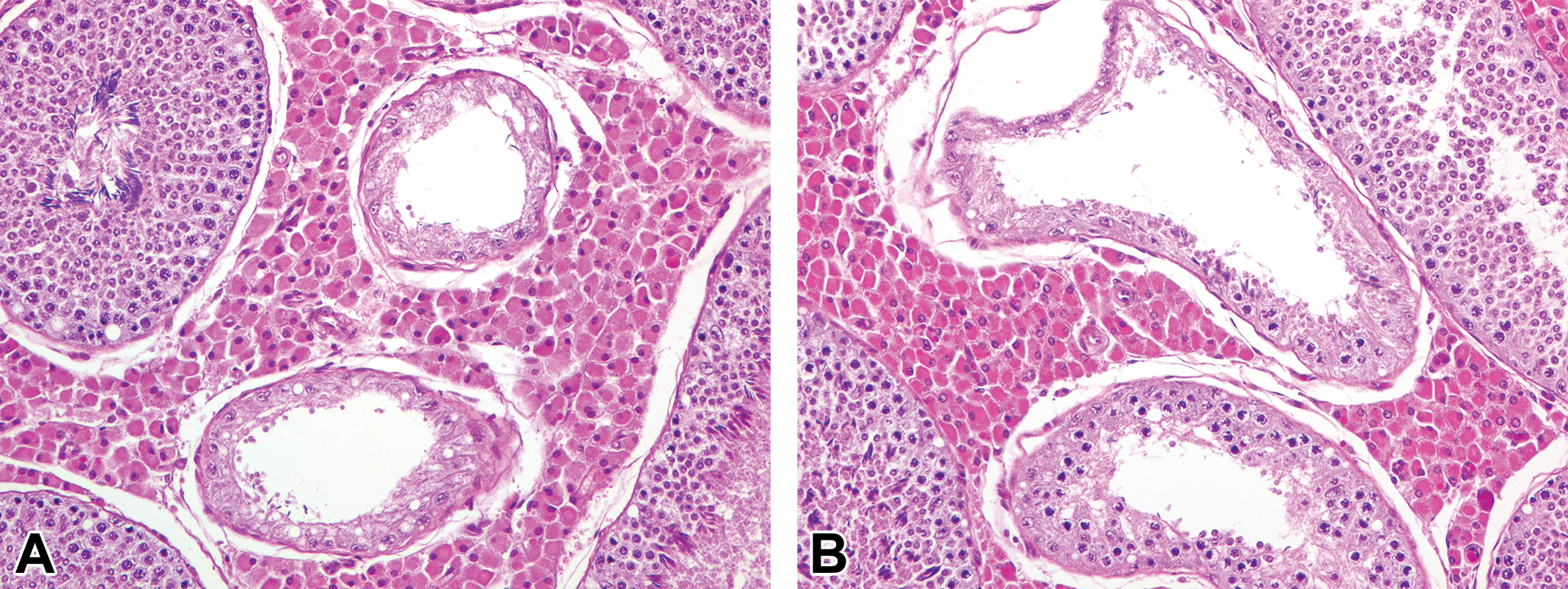
Tubular atrophy/hypoplasia with only Sertoli cells or almost only Sertoli cells. (A) Small tubules composed of only columnar Sertoli cells next to tubules with normal spermatogenesis at 3 months of age. (B) Irregularly shaped tubules containing almost only Sertoli cells (upper) in close association with tubules of hypospermatogenesis (bottom) at 12 months of age. Hematoxylin and eosin stain.
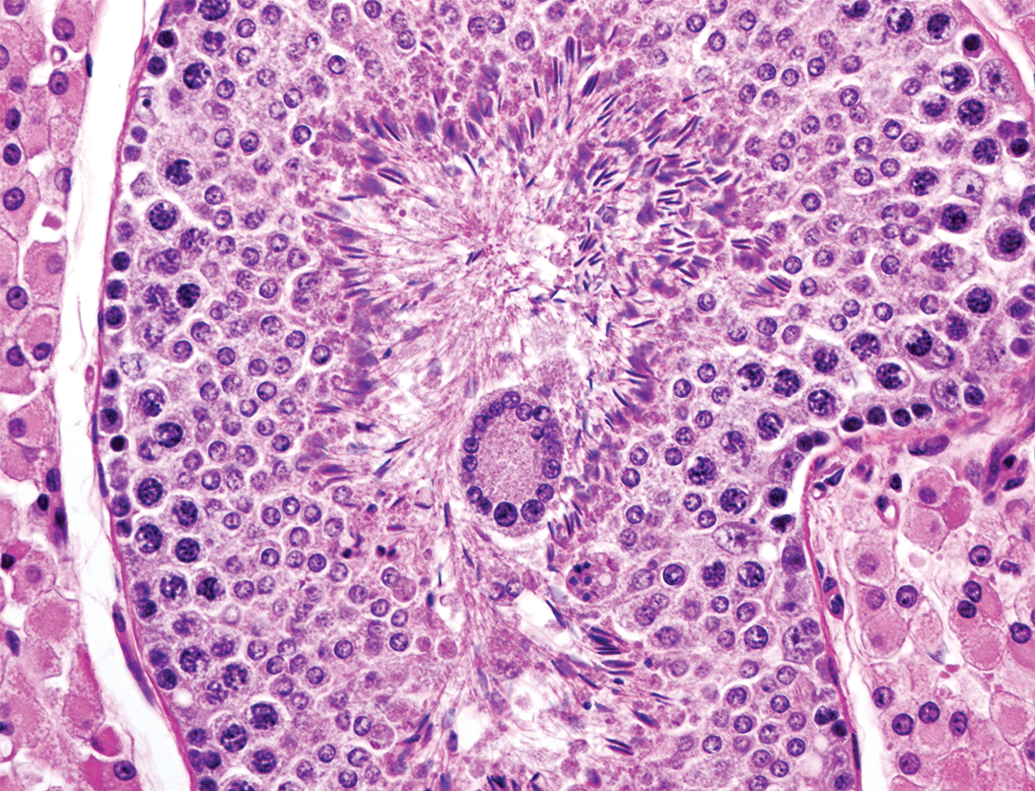
Multinucleated giant cell in the lumen of the tubule showing normal spermatogenesis at 6 months of age. Hematoxylin and eosin stain.
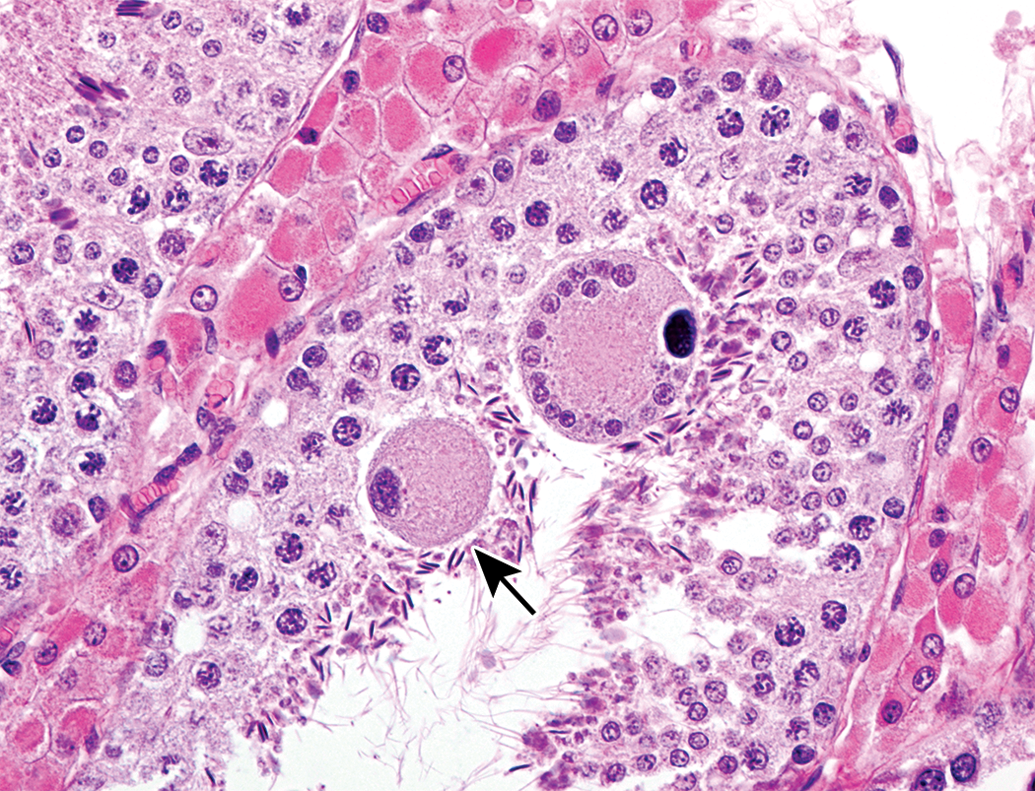
Swollen spermatocyte (arrow) and a multinucleated giant cell in the lumen of seminiferous tubule at 1.5 months of age. Hematoxylin and eosin stain.
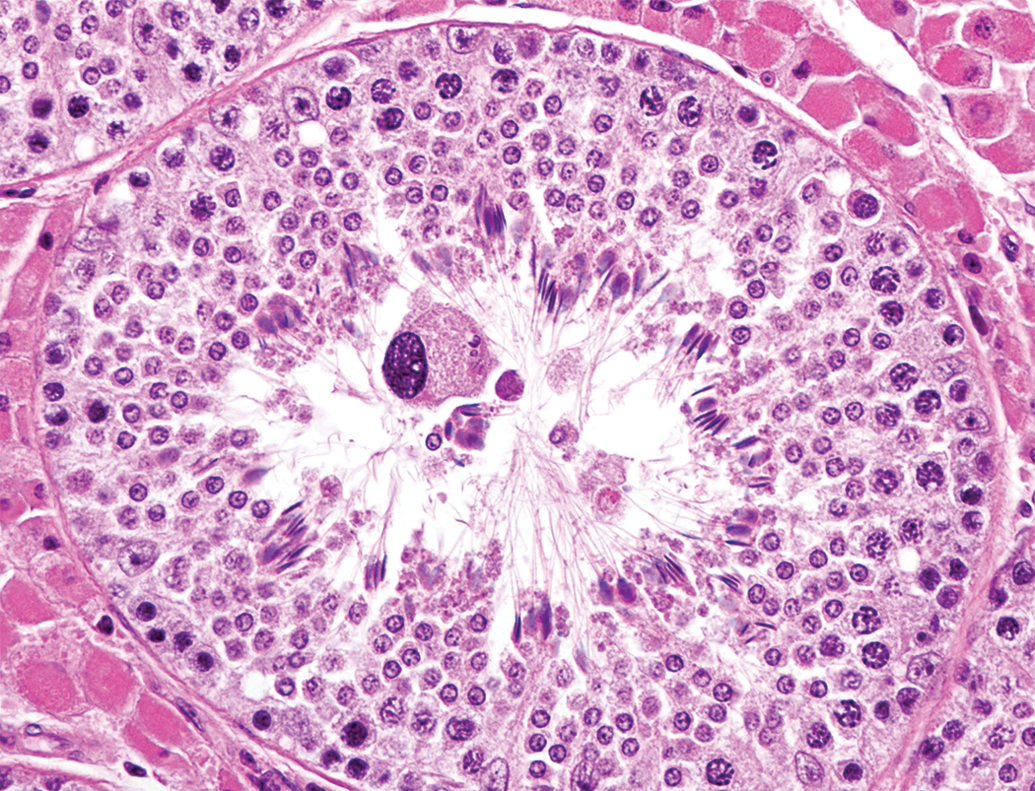
Karyomegaly derived from a spermatocyte in the lumen of a tubule at 3 months of age. Hematoxylin and eosin stain.
Spontaneous Testicular Changes in Microminipigs.
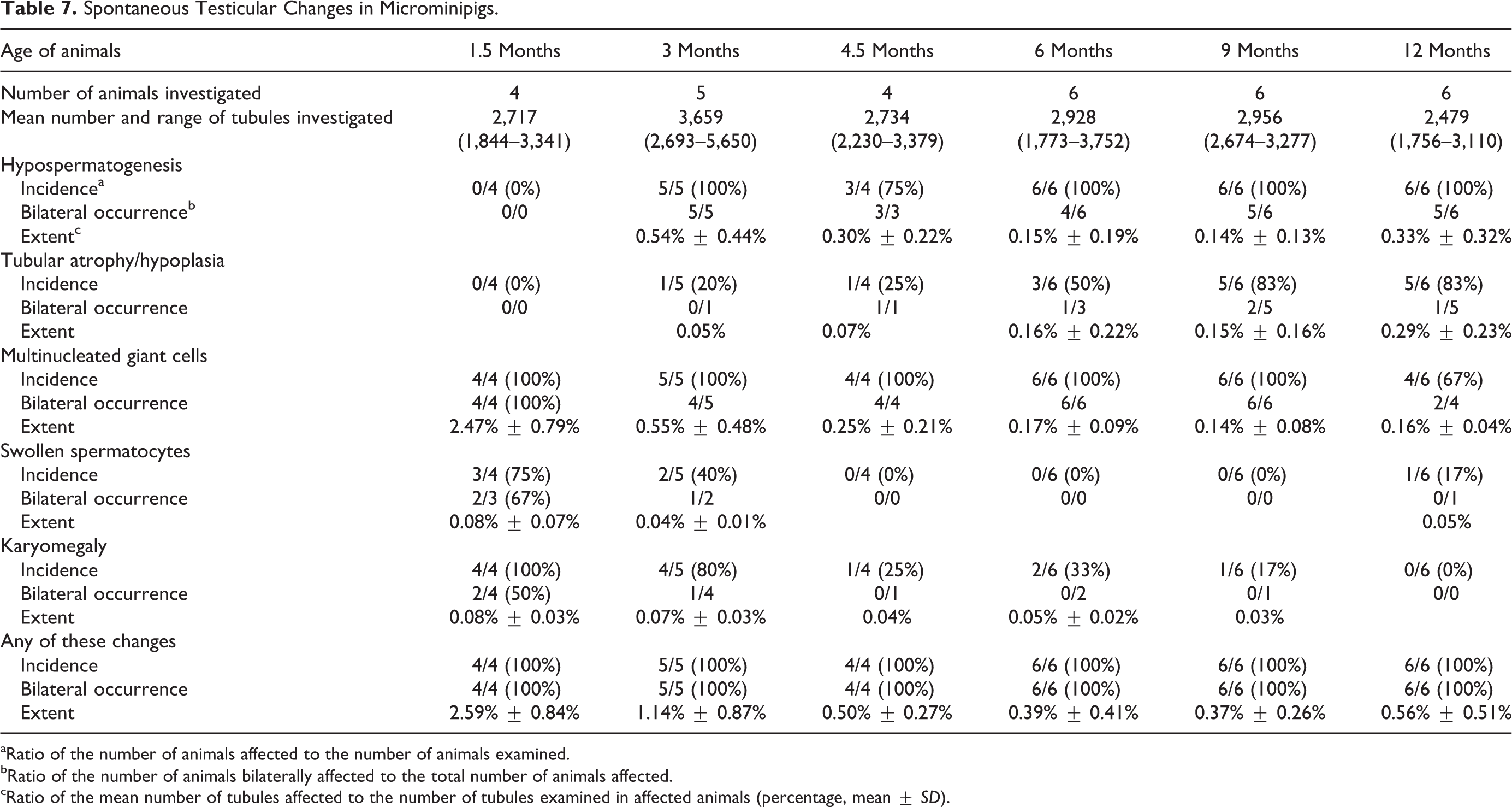
aRatio of the number of animals affected to the number of animals examined.
bRatio of the number of animals bilaterally affected to the total number of animals affected.
cRatio of the mean number of tubules affected to the number of tubules examined in affected animals (percentage, mean ±
Discussion
The present study illustrated the development of male reproductive organs and spontaneous occurrence of testicular changes in microminipigs aged between 0 and 12 months based on thorough histological and histomorphological investigations. This report is the first to describe the histological appearance and precise stage of sexual maturity in male microminipigs.
Summary of the results is shown in Table 8. Sperm production in testes and sperm storage in epididymides are generally considered as evidences of sexual maturity in males; however, completion of maturation occurs shortly thereafter. In addition to the analyses of the testis and epididymis, assessment of organ weights and histology of the accessory genital glands provides reliable information to decide the stage of maturity (Foley 2001). Based on the present assessment of reproductive organs, we deduced that male microminipigs are immature at 0 month, prepubertal at around 1.5 months, and pubertal at around 3 months of age followed by sexual maturity at around 4.5 months, when complete development of all reproductive organs was confirmed. In addition, we considered that pigs aged 4.5 to 6 months were young adults, because weight of accessory glands, such as prostate and bulbourethral glands, was still increasing. Moreover, adult life stage seemed to appear after 6 months of age, because accessory gland weights, except epididymides, reached a plateau.
Summary of the Development of Male Reproductive Organs in Microminipigs.
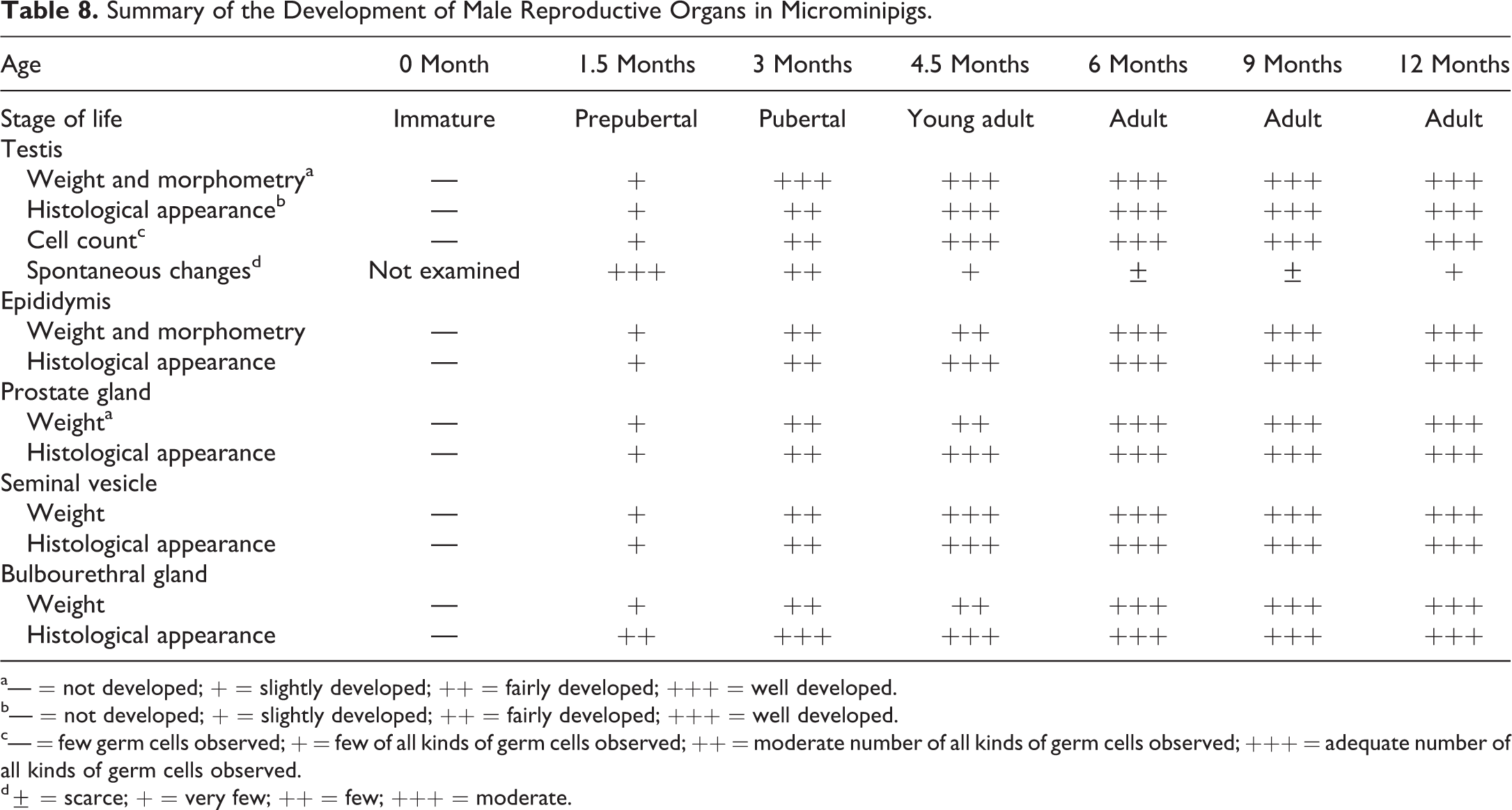
a— = not developed; + = slightly developed; ++ = fairly developed; +++ = well developed.
b— = not developed; + = slightly developed; ++ = fairly developed; +++ = well developed.
c— = few germ cells observed; + = few of all kinds of germ cells observed; ++ = moderate number of all kinds of germ cells observed; +++ = adequate number of all kinds of germ cells observed.
d± = scarce; + = very few; ++ = few; +++ = moderate.
Our findings revealed that male microminipigs might be showing precocious sexual maturity in comparison to other nonrodent animals because sexual maturity in dogs usually occurs at 9 to 10 months of age and in cynomolgus or macaque monkeys at approximately 4 to 5 years of age (Wolfe 1978; Kawakami, Tsutsui, and Ogasa 1991). The use of sexually mature animals is recommended for fine testicular morphological analysis when dealing with well-known testicular toxicants or unknown compounds in toxicology studies (Lanning et al. 2002). Microminipigs aged around 4.5 months and thereafter are suitable for the assessment of reproductive organs because they are at the young adult stage of life. For instance, during 4-week and 13-week toxicology studies, they would be sexually mature, and by the end of the studies, they would be approaching the adult stage. Sexual maturity in male minipigs is known to occur at around 6 months of age (Svendsen 2006; McAnulty et al. 2012). Although the definition of maturity may have been different in each study and consistent histological criteria may not have been applied, some strains of minipigs, such as Göttingen minipigs and Yucatan minipigs, have been reported to be precocious and sexually mature at 2 to 4 months of age and 4.4 months, respectively (Bode et al. 2010; Howroyd, Peter, and de Rijk 2016). The present study indicates that male microminipigs are one of the sexually precocious minipigs and potential candidates for studies that require sexually mature animals.
In toxicity studies, organ weights are used as a reliable parameter to identify treatment-related effects. Among these organs, testes are commonly weighed, because their weights are sensitive to toxicity due to perturbations in rapidly dividing spermatogenic cells. Epididymal and prostate gland weights are also considered sensitive indicators of toxic effect in specialized studies, such as reproduction studies (Michael et al. 2007). Weight changes of these reproductive organs may reflect edema, inflammation, or decreased sperm production (Sellers et al. 2007). Reproductive organ weights from immature animals must be evaluated with caution when their weights are lower than sexually mature counterparts, with interanimal variability (Foley 2001; Sellers et al. 2007). Furthermore, the use of relative (organ to body) weight is often helpful in evaluating treatment-related changes, particularly in nonrodents in which there are notable variations in either organ or body weight (Wooley 2003). Organ weight data collected in the present study would be helpful in evaluating toxicological assessment in studies using male microminipigs. In addition, testes weights could be used as an index to estimate the stage of spermatogenic development for a testis of around 20 g (in pairs, 40 g) and over showed active spermatogenesis in microminipigs.
Under the present study conditions, the body weight increased gradually during the whole period, whereas the relative weight of reproductive organs reached maximum at a certain age: for testes, 3 months; epididymides, 9 months; seminal vesicles, 4.5 months; and body of prostate and bulbourethral glands, 6 months. This result suggests that growth of reproductive organs is independent of body weight in microminipigs. This is consistent with a previous finding reported in boars (Allrich et al. 1983).
Size of seminiferous tubules is considered one of the most reliable markers for testicular maturation and damage, because an increase in number of spermatogenic cells and in production of seminiferous tubule fluid as the testis matures expands the size of seminiferous tubules and a decrease or loss of spermatogenic cells results in reduced size of seminiferous tubules (Russell et al. 1990; Creasy 1997). The mean diameter of the seminiferous tubule in which elongated spermatids first appeared was 150 μm at age 1.5 months. At 3 months of age and thereafter, the mean diameter reached more than 200 μm, where active spermatogenesis was confirmed by evaluating cell counts in the testis. The relation between seminiferous tubule diameter and spermatogenesis development was consistent with findings previously reported in domestic boars (Van Straaten and Wensing 1977; Harayama, Tsutsui, and Kato 1992; Itoh and Suzuki 1993). Despite the fact that microminipigs are much smaller in size than domestic pigs, microscopic features in their testis are similar to those in domestic boar testis. Histomorphological data from domestic boars and microminipigs may indicate that spermatogenic development is sustained genealogically.
Sertoli cell efficiencies and meiotic indexes were estimated by calculating the results of cell counts. Sertoli cells are the somatic cells residing among the germ cells in the seminiferous tubule, and they have important functions in spermatogenesis. One of these functions is to provide nutrition to the developing germ cells (Russell et al. 1990). The mean Sertoli cell efficiency in sexually mature microminipigs, which are older than 4.5 months, was 23.9 at stage VII and 23.7 at stage VIII. Sertoli cell efficiencies in various animal species are as follows: boar, 12.4; rabbit, 12.2; mouse, 10.9; rat, 8.0 to 10.3; and man, 3.0 (Franca and Russell 1998; Franca, Avelar, and Almeida 2005). Sertoli cell efficiency in microminipigs was more than twice as high as that in other animals. The mean meiotic index in sexually mature microminipigs was 2.8 at stage VII and stage VIII. Based on the fact that 1 primary spermatocyte divides into 4 spermatids by meiosis (maximum meiotic index is 4), the results indicated that almost one-third of the germ cells were lost during meiotic divisions in the microminipig seminiferous epithelium cycle, probably due to chromosomal damage and nondisjunction (Franca and Russell 1998; Franca, Avelar, and Almeida 2005). The meiotic indexes, which are almost same in various animals, except for humans, are as follows: boar, 3.2; rabbit, 3.3; mouse, 3.1; rat, 3.4; and man, 1.3 (Franca, Avelar, and Almeida 2005). Meiotic index in microminipigs was relatively low as compared to that in other animals. These results suggest that germ cell loss during meiosis might occur frequently, even though the supporting capacity of Sertoli cells for spermatids is higher in microminipigs than in other animals.
Spontaneous testicular changes have been precisely reported in rabbits and beagle dogs (Morton 1988; Rehm 2000; Goedken, Kerlin, and Morton 2008). Microminipigs have some differences in the incidence of these changes as compared to that in rabbits and beagle dogs. Hypospermatogenesis occurred in most of the microminipigs investigated, whereas the incidence was much less in rabbits (approximately 50%) and beagle dogs (30% according to Rehm 2000 and 20% according to Goedken, Kerlin, and Morton 2008). Tubular atrophy/hypoplasia in microminipigs occurred in 48% of animals (15 of 31); this value was lower in beagle dogs (30% according to Rehm 2000 and 26.3% according to Goedken, Kerlin, and Morton 2008). Tubular atrophy/hypoplasia in rabbits was not categorized by Morton, Weisbrode, Wyder, Maurer, and Capen (1986). Multinucleated giant cells were observed in almost all microminipigs, which were the same for rabbits (approximately 95%) and beagle dogs (98%). Swollen spermatocytes were not a common finding in microminipigs; however, they were present in 96% to 97% rabbits and beagle dogs. Differences among species, in the incidence of these changes, emphasize that pathologists should be aware of the intrinsic testicular characteristics of the animals under investigation.
Several previous studies have evaluated spontaneous findings in male reproductive organs in minipigs (Dincer et al. 2007; Thuilliez et al. 2014; Jeppesen and Skydsgaard 2015; Helke et al. 2016a, 2016b). Tubular atrophy/hypoplasia is the most common finding in the testes in Göttingen minipigs but not in Hanford or Yucatan minipigs (Helke et al. 2016a). Tubular hypoplasia/atrophy was found in 48% of all microminipigs (15 of 31) and the incidence increased with age, whereas the prevalence in Göttingen minipigs varied in different studies. However, a recent report by Helke et al. (2016b) reviewed previous articles and a large number of animals were evaluated. They found that 25% of Göttingen minipigs were affected by tubular hypoplasia/atrophy. Thus, the incidence may be higher in microminipigs than in Göttingen minipigs. Moderate to massive severity of tubular hypoplasia/atrophy was reported in some animals in Göttingen minipigs, in which a maximum of 50% to 75% of tubules were affected (Thuilliez et al. 2014; Jeppesen and Skydsgaard 2015; Helke et al. 2016a). The mean percentage of tubules affected was less than 0.3% in microminipigs of various ages, which may correspond to the minimal grade of alterations evaluated in previous reports. Although the number of animals investigated may not be sufficient compared to those in reports of Göttingen minipigs, the extent of tubular hypoplasia/atrophy was less severe in microminipigs than in Göttingen minipigs. The most common finding in microminipigs was multinucleated giant cells (94% of all animals, 29 of 31); however, very few studies have reported this and only minipigs aged 2 to 6 months were found to be affected in these studies. Multinucleated giant cells were observed in 1.3% of Göttingen minipigs, and tubular multinucleated cells were observed in 0.4% of Göttingen minipigs and 2.3% of Hanford minipigs (Helke et al. 2016a). The high incidence of multinucleated giant cells may be a histological feature of the testes in microminipigs. Extramedullary hematopoiesis was observed in all microminipigs at 0 months of age (4 of 4), but not in animals 1.5 months and older, which is consistent with the findings in young Yucatan minipigs: 100% of animals less than 2 months of age and 38% between 2 to 6 months of age (Helke et al. 2016a). Considering the nature of hematopoiesis in young animals, these findings may represent a normal state. In accessory organs of minipigs, spontaneous findings such as cysts, aspermia/oligospermia, luminal cell debris in the epididymis, mineralization in the prostate, and inflammatory cell infiltrate in the seminal vesicle and bulbourethral gland have been reported (Dincer et al. 2007; Jeppesen and Skydsgaard 2015; Helke et al. 2016a). In the present study, these changes were not observed. However, further studies are required to understand the occurrence of these events in microminipigs. In addition, hypospermatogenesis, swollen spermatocytes, and karyomegaly have not been characterized in the literature, and thus, the difference in the prevalence of these changes between microminipigs and other minipigs remains unclear.
The present study revealed that sexual maturity in male microminipigs was estimated to be around 4.5 months of age, based on the histomorphological appearance of reproductive organs, particularly in terms of active spermatogenesis and abundant sperm accumulation in the epididymis. In addition, spontaneous testicular changes at each stage of life were commonly observed in microminipigs, which are quite similar to those in other nonrodent animals. However, the severity of these changes was apparently mild; hence, their influence on the evaluation of testicular toxicity may be minimal. We expect the information obtained by the present study to be useful for future toxicity studies. A limited number of animals have been examined so far; therefore, additional studies on the physiological aspects, such as hormonal regulation of the male reproductive system, and morphological manifestations of response to injury are essential to comprehensively understand the function of the reproductive organs in microminipigs.
Footnotes
Acknowledgment
The authors thank Prof. Motoki Sasaki (Obihiro University of Agriculture and Veterinary Medicine) for his advice during the early days of the study and Mr. Toshiaki Fukumura (Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center) for taking good care of the animals.
Authors’ Contribution
Authors contributed to conception or design (AK); data acquisition, analysis, or interpretation (MO, SE, TY, MS); drafting the manuscript (AK); and critically revising the manuscript (MO, SE, TY, MS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
