Abstract
Microminipigs have become an attractive animal model for the toxicology- and pharmacology-related studies because of their manageable size. In this study, the development of the testicular interstitium and steroidogenesis in microminipigs, from 0 to 12 months of age, were investigated. Testicular interstitium was mostly composed of two types of Leydig cells (large and small Leydig cells) and a few macrophages and mast cells. Large Leydig cells were observed in the peritubular area throughout all the ages. Small Leydig cells were present in the interlobular and subcapsular areas at an early age and then gradually converted into large Leydig cells. Testicular composition of the Leydig cells began to increase after 3 months of age, when spermatogenesis was completed, and reached approximately 35% at 12 months. Steroidogenic enzymes in Leydig cells were detected by immunohistochemistry from 0 month of age. Serum testosterone levels increased substantially from 1.5 to 4.5 months of age, which coincided well with the age of sexual development previously reported in microminipigs. Because the interstitial space of the testis has dramatic variations between species, the basic information obtained in the present study will be a useful reference for all the future toxicity evaluations in microminipigs.
Keywords
The use of minipigs, as a nonrodent species, is considered an alternative to dogs and nonhuman primates for preclinical safety assessment of pharmaceuticals. They are now used routinely in toxicology- and pharmacology-related studies (Svendsen 2006; Nunoya et al. 2007; Bode et al. 2010; Colleton et al. 2016; Stricker-Krongrad et al. 2016). Among several different breeds of minipigs, microminipigs (Fuji Micra, Inc., Shizuoka, Japan) have become increasingly attractive as animal models for toxicology studies because of their manageable size: 9 kg to 13 kg at their sexual maturity (Kangawa et al. 2016, 2017). The age of sexual maturity in male microminipigs is known to be 4.5 months, according to the development of spermatogenesis and accessory glands (Kangawa et al. 2016). However, the morphological changes in the testicular interstitium and the characteristics of the steroidogenesis in the Leydig cells, during postnatal development of the testis in microminipigs, are unknown.
A basic understanding of the normal histology in the reproductive tract is essential for pathologists in their evaluations. A knowledge of the anatomical variations and development will facilitate an accurate interpretation of the changes observed in preclinical safety studies (Foley 2001). The interstitial space of the testis, all areas between the seminiferous tubules, has dramatic variations between species with regard to the number or amount of the Leydig cells, macrophages, mast cells, connective tissue, and lymphatics (Fawcett, Neaves, and Flores 1973; Nistal, Santamaria, and Paniagua 1984; Dirami, Poulter, and Cooke 1991; Anton et al. 1998; Foley 2001). Pigs especially have a larger amount of Leydig cells compared to other species, and the size of these cells differs depending on the location in the testis (Van Straaten and Wensing 1978; França, Avelar, and Almeida 2005). Furthermore, the function, the size, and the amount of Leydig cells and macrophages may be altered by the administration of certain drugs and hormones (Duckett et al. 1997a, 1997b).
The major reproductive function of the Leydig cells is the production and secretion of androgens and estrogens, which are necessary for the maintenance of reproductive and nonreproductive tissues (Sharpe 1988; O’Donnell, Stanton, and de Kretser 2017; Carreau et al. 2007). Androgens, primarily testosterone, have characteristic roles during male sexual differentiation, development and maintenance of male reproductive organs, and initiation and maintenance of spermatogenesis (George and Wilson 1994; McEwan and Brinkmann 2016). Estrogens, primarily estradiol, are also responsible for some crucial physiological functions in male reproduction and act synergistically with testosterone (Joshi and Raeside 1973; Rochira et al. 2016). In particular, high levels of circulating estrogens were one of the remarkable features of the male boar, compared to the males of other species and the females of the same species (Claus and Hoffmann 1980; Setchell et al. 1983; Schwarzenberger et al. 1993; Clapper, Clark, and Rempel 2000; Kanematsu et al. 2006). As the main function of the Leydig cell is steroidogenesis, any substance that interferes with this pathway will produce functional disturbances in the hormone balance (Creasy 2001). Therefore, it is important to understand the characteristics of steroidogenesis in Leydig cells, for a precise evaluation of male reproductive toxicity in microminipigs.
The objectives of the present study were to clarify the histological features of the testis interstitium and the characteristics of the steroidogenesis, in the Leydig cells during postnatal development in microminipigs.
Materials and Methods
Animals
In this study, 36 microminipigs, 6 each at 0, 1.5, 3, 4.5, 6, and 12 months of age, were utilized. From the 36 animals under study, 23 were previously used for the evaluation of sexual maturity: four individuals from each group of ages 0 (including three stillborns); 1.5, 3, 4.5, and 6 months; and three 12 months old individuals (Kangawa et al. 2016). The animals were bred in a closed colony, at the Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center (Shizuoka, Japan). Fuji Micra Inc. provided the parental generation of these animals for research purpose. Both male and female microminipigs were housed in a group from birth to 3 months of age, and then the male microminipigs were housed individually or in pairs, in pens of at least 0.54 m2 area/animal, under conventional conditions, with 12-hr light and 12-hr dark cycle. The animals were weaned at 4 weeks of age and then fed a commercial diet (Marubeni Nisshin Feed Co., Ltd., Tokyo, Japan), as instructed by Fuji Micra Inc., at 2% to 3% of body weight/animal/day, with
All experiments and procedures were approved by the Animal Care and Use Committee of Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center, which complies with the Guidelines for Proper Conduct of Animal Experiments (2006), provided by the Science Council of Japan.
Histological Examination
Testes were fixed in modified Davidson’s solution (30% v/v of a 37% solution of formaldehyde, 15% v/v ethanol, 5% v/v glacial acetic acid, and 50% v/v distilled H2O) for 1 week (Latendresse et al. 2002). They were trimmed transversely through the middle of the major axis because the composition of the median part of the testis in boars was considered to be representative for the whole testis (Rohloff and Hildebrand 1971). The fixed samples were then dehydrated in a series of graded ethanol and embedded in paraffin. Serial sections, 3 µm thick each, were obtained from the embedded testes and used for general and special staining and for immunohistochemistry analysis. Sections were stained with hematoxylin and eosin for general histological examination, with the periodic acid-Schiff (PAS) reaction, the Schmorl’s method, and the Berlin blue stain to confirm lipofuscin deposition, and with Masson’s trichrome for histomorphometrical analysis (Nandy 1971; Pearse 1980).
Morphometry of the Testicular Composition and the Leydig Cell Size
The testicular composition, within the cross section of the testis, was obtained by using the sections stained with Masson trichrome and microscope imaging software NIS Elements BR3.0 (Nikon, Tokyo, Japan). Measurements were performed based on previous reports on pigs, with minor modifications (Van Straaten and Wensing 1977; Almeida, Leal, and França 2006; Costa et al. 2010). Briefly, five randomly selected microscope fields of view, at a 100× magnification, were photographed from each testis. The areas occupied by the seminiferous tubules, Leydig cells, and interstitial tissue were measured. Then, the percentages of occupancy of each component were calculated. The tunica albuginea and the rete testis were not included in this analysis. The Leydig cell size was obtained by measuring the length of the cells using NIS Elements BR3.0. As the Leydig cells show different morphology depending on the location within the testis during the postnatal development in pigs (Van Straaten and Wensing 1978), 30 evenly square to polygonal Leydig cells from peritubular (the area surround the seminiferous tubule), interlobular (the area between the individual seminiferous tubules), and subcapsular (the area underneath the tunica albuginea) compartments of each animal were selected for measuring.
Immunohistochemistry for Leydig Cells and Macrophages
Immunohistochemistry was performed using the ABC method (VECTASTAIN Universal Elite ABC Kit, Vector Laboratories, Burlingame, CA, USA). Characteristics, dilutions, and antigen retrieval methods are shown in Table 1. Briefly, antibodies against the cytochrome P450 side chain cleavage enzyme (P450scc), 17-β hydroxysteroid dehydrogenase 3 (HSD17B3), and aromatase cytochrome P450 (P450arom) were utilized to detect the presence of the steroidogenic enzymes in the Leydig cells. The proliferating cell nuclear antigen (PCNA) was used to evaluate the proliferative activity of the Leydig cells, and the allograft inflammatory factor 1 (AIF-1/Iba-1) was used to locate the macrophages in the testis. The sections were deparaffinized and treated for antigen retrieval. They were immersed in absolute methanol and 0.3% H2O2 for 30 min to quench the endogenous peroxidase activity. After blocking nonspecific binding for 20 min, the slides were incubated with the primary antibodies for overnight at 4°C and then with the biotinylated secondary antibody for 30 min at room temperature. Then, the sections were incubated with VECTASTAIN® ABC Reagent for 30 min at room temperature followed by DAB (DAB Peroxidase Substrate Kit, ImmPACT, Vector Laboratories) to detect the immunoreactivity. The sections were washed twice in phosphate-buffered saline between each step. Counterstaining was performed with hematoxylin for steroidogenic enzymes and PCNA and with toluidine blue for AIF-1. Macrophages were confirmed by immunopositivity for AIF-1, and mast cells were by metachromasia for toluidine blue.
Characteristics, Dilutions, and Antigen Retrieval Methods in Immunohistochemistry.
Serum Testosterone and Estradiol Measurements
Blood samples from each animal, except for the 3 stillborn animals at 0 month of age, were collected between 9 a.m. and 10 a.m. from the external jugular vein before necropsy. Serum was obtained by centrifugation, for 15 min at 3,000 rpm, and stored at −30°C. Serum testosterone levels were measured using DetectX Testosterone EIA Kit (Arbor Assays, Ann Arbor, MI) and the serum estradiol levels were measured using Estradiol ELISA Kit (Cayman Chemical, Ann Arbor, MI). For the steroid hormones determination, two 500 μl aliquots of each serum sample were extracted three times with 3 ml diethyl ether. The diethyl ether was evaporated using a centrifugal concentrator. The extracts were dissolved in 500 µl of EIA buffer and ELISA buffer. Each sample was then analyzed in duplicate by performing the assay in accordance with the manufacturer instructions. The sensitivity of these assays was 9.92 pg/ml for testosterone and 15 pg/ml for estradiol. The intra assay coefficients of variation were less than 15.8% for testosterone and 18.8% for estradiol. The cross reactivity of the testosterone EIA Kit was 56.8% for 5α-dihydrotestosterone, 0.27% for androstenedione, and 0.02% for estradiol, and that of the estradiol ELISA kit was 14.5% for estradiol-3-sulfate, 14% for estradiol-3-glucuronide, 12% for estrone, 10% for estradiol-17-glucuronide, and less than 0.01% for testosterone.
Results
Biometric Data
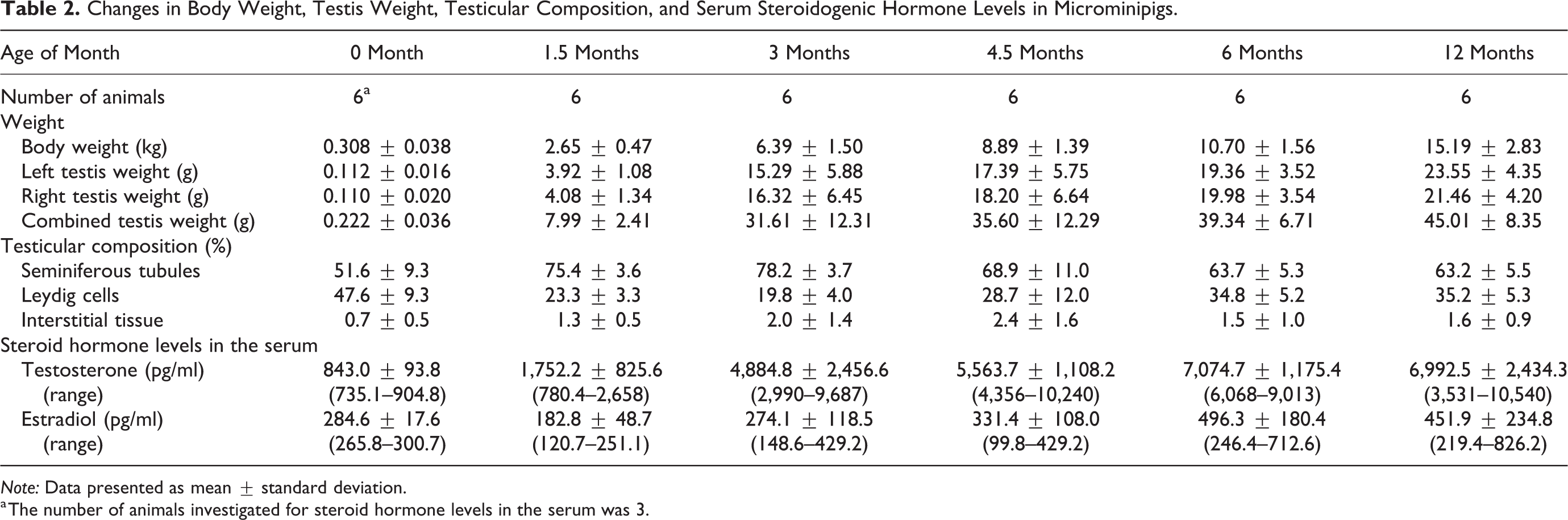
Body and testis weights (mean ± standard deviation) of the animals are shown in Table 2. Testis weight increased dramatically from 0 month to 3 months of age, after which the testis gained weight gradually, reaching maximum of the ages assessed at 12 months of age.
Changes in Body Weight, Testis Weight, Testicular Composition, and Serum Steroidogenic Hormone Levels in Microminipigs.
a The number of animals investigated for steroid hormone levels in the serum was 3.
Histological Findings
At 0 month of age, the Leydig cells were round to polygonal in shape with homogeneously eosinophilic cytoplasm and had a round or vesicular nucleus with one or two eccentrically located nucleoli (Figure 1a). There were hardly any morphological differences in the Leydig cells throughout the testis. The seminiferous tubules were composed of immature Sertoli cells and gonocytes.
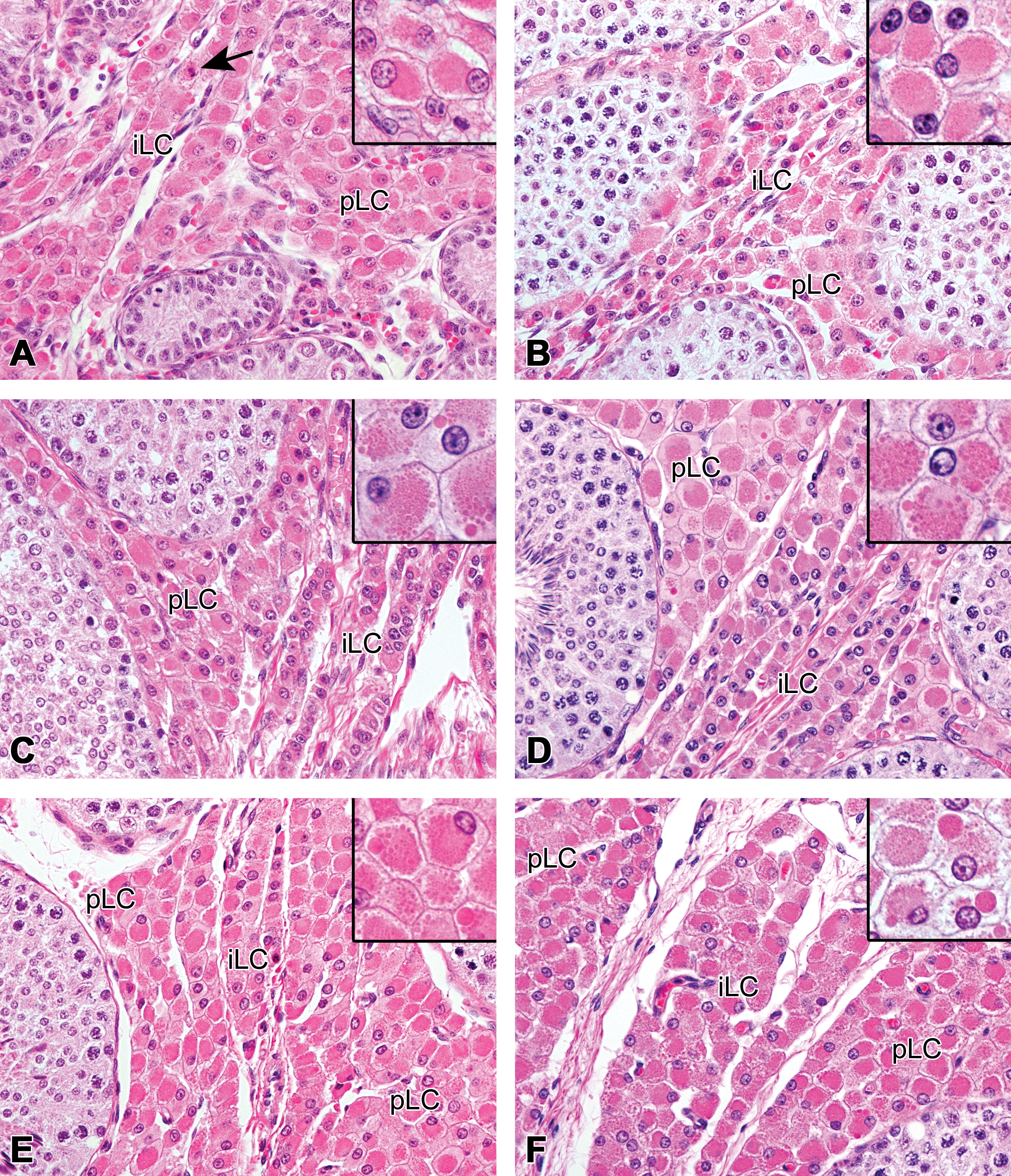
Development of the peritubular and interlobular Leydig cells in microminipigs. (a) At 0 month of age, both peritubular and interlobular Leydig cells had abundant eosinophilic cytoplasm. The arrow indicates a mitotic figure within the Leydig cell. Inset: Higher magnification of the peritubular Leydig cells. At 1.5 months (b), 3 months (c), and 4.5 months (d) of age, two types of the Leydig cells were easily recognized. Large peritubular Leydig cells surrounding the seminiferous tubule and small interlobular Leydig cells intermingled with the collagenous tissue. A seminiferous tubule showing active spermatogenesis at 4.5 months of age (d, left). (b)–(d) Insets: Higher magnification of the peritubular Leydig cells. Small- to middle-sized eosinophilic spheres started to appear in the cytoplasm from 3 months of age. (e) At 6 months of age, outer layers of interlobular Leydig cells became larger than those at 4.5 months of age. Inset: Higher magnification of the peritubular Leydig cells. Large eosinophilic spheres were present. (f) At 12 months of age, both peritubular and interlobular Leydig cells showed maximum development and were no longer distinguishable by their cellular appearance. Inset: Higher magnification of the peritubular Leydig cells. Large eosinophilic spheres became prominent. pLC = peritubular Leydig cell; iLC = interlobular Leydig cell. Hematoxylin and eosin staining.
At 1.5 months of age, two distinct groups of Leydig cells were easily recognized: the peritubular Leydig cells, which were large polygonal cells with abundant eosinophilic cytoplasm, and the interlobular and subcapsular Leydig cells, which were small cells with scant cytoplasm (Figures 1b and 2a). Both cells had a round to vesicular nucleus with one, rarely two, prominent nucleoli. The area adjacent to the tubules was closely packed with the peritubular Leydig cells. The interlobular and subcapsular Leydig cells were arranged in narrow rows, intermingled with small amount of connective tissue. The seminiferous tubules were still immature, but a few elongated spermatids were rarely observed on the surface of the seminiferous epithelium.
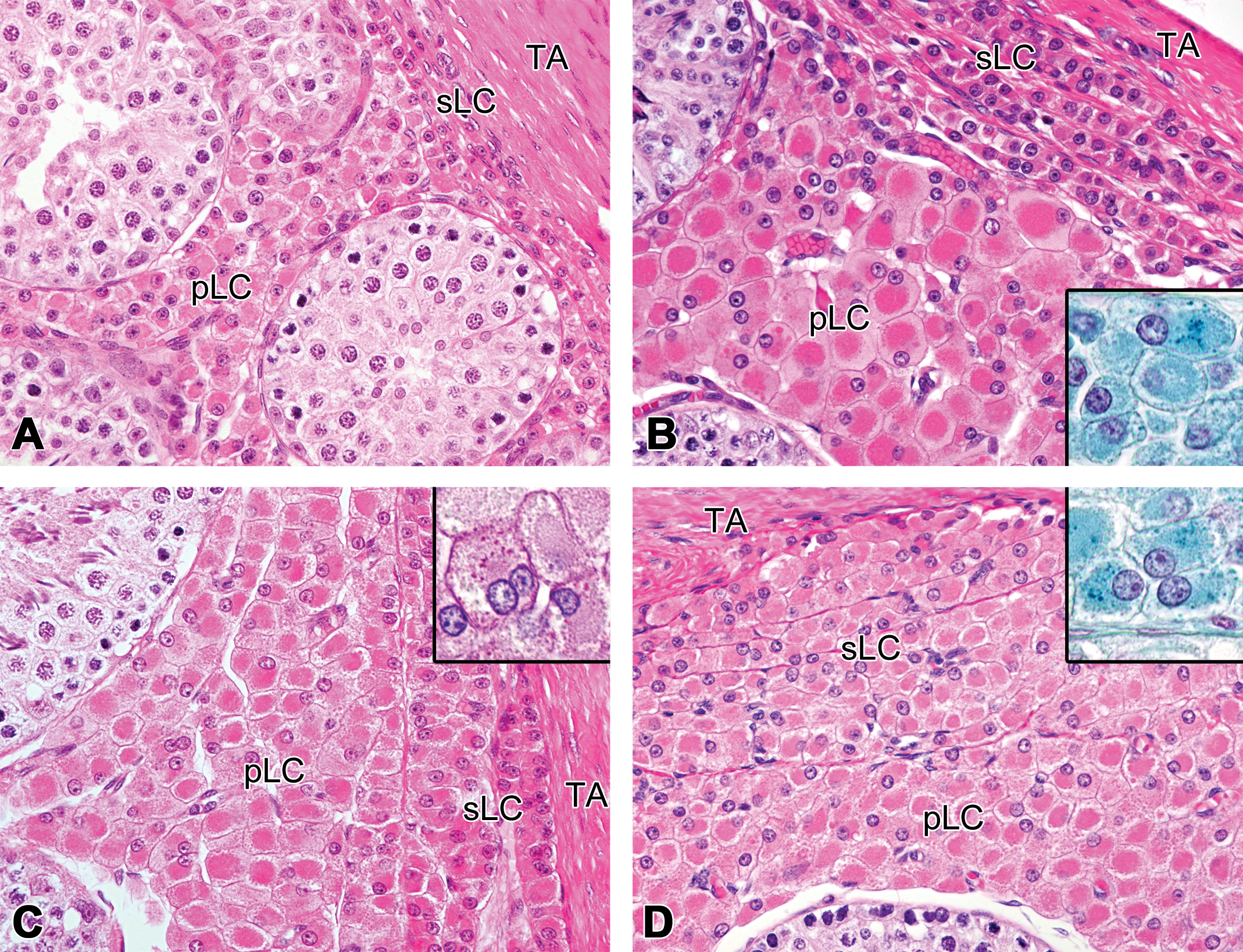
Development of the subcapsular and peritubular Leydig cells in microminipigs. At 1.5 months (a) and 4.5 months (b) of age, subcapsular Leydig cells were small and easily distinguishable from the peritubular Leydig cells. (b) Inset: Tiny lipofuscin granules within the cytoplasm of the peritubular Leydig cells were stained dark greenish blue by Schmorl method. (c) At 6 months of age, the inner layers of the subcapsular Leydig cells had more cytoplasm than those at 4.5 months of age. Inset: Lipofuscin granules in the binucleated peritubular Leydig cell (not shown in the lower magnification) were positive for periodic acid-Schiff reaction. (d) At 12 months of age, subcapsular Leydig cells showed maximum development and were no longer distinguishable from the peritubular Leydig cells by their cellular appearance. Inset: Lipofuscin granules in the peritubular Leydig cells stained by Schmorl method. pLC = peritubular Leydig cells; sLC = subcapsular Leydig cells; TA = tunica albuginea. Unless specified, the staining was performed with hematoxylin and eosin.
At 3 months of age, the peritubular Leydig cells increased in number, and a few cells started to have small eosinophilic spheres in their cytoplasm. There were no apparent morphological changes observed in the interlobular and subcapsular Leydig cells, compared with those of 1.5 months of age (Figure 1c). Active spermatogenesis within the seminiferous tubules was observed throughout the testis from 3 months of age.
At 4.5 months of age, the number of the peritubular Leydig cells increased even more, yielding several clusters of Leydig cells around the tubules. Several eosinophilic spheres of various sizes were occasionally observed within the cytoplasm of the peritubular Leydig cells. The outer layers of the interlobular Leydig cells and the inner most layer of the subcapsular Leydig cells were slightly larger, compared to those of 3 months of age (Figures 1d and 2b). Rarely, a few fine granular brown pigments started to appear in the cytoplasm of some Leydig cells. These pigments stained dark blue to dark greenish blue with Schmorl method, purple red with PAS reaction, and negative with Berlin blue, confirming that the pigments were lipofuscin (Figure 2b inset).
At 6 months of age, most of the interlobular Leydig cells and two to four inner layers of the subcapsular Leydig cells were larger, while one or two outer layers of the subcapsular Leydig cells remained small (Figures 1e and 2c). Lipofuscin pigments were occasionally observed in the Leydig cells throughout the testis (Figure 2c inset).
At 12 months of age, the interlobular and subcapsular Leydig cells had large amount of eosinophilic cytoplasm and were indistinguishable from the peritubular Leydig cells in their morphology (Figures 1f and 2d). The number of Leydig cells with lipofuscin, and the amount of lipofuscin granules within these cells, increased compare to those at 6 months of age (Figure 2d inset). Summary of the histological development of the Leydig cells is shown in Table 3.
Characteristics of the Leydig Cells, with Age, in Microminipigs.
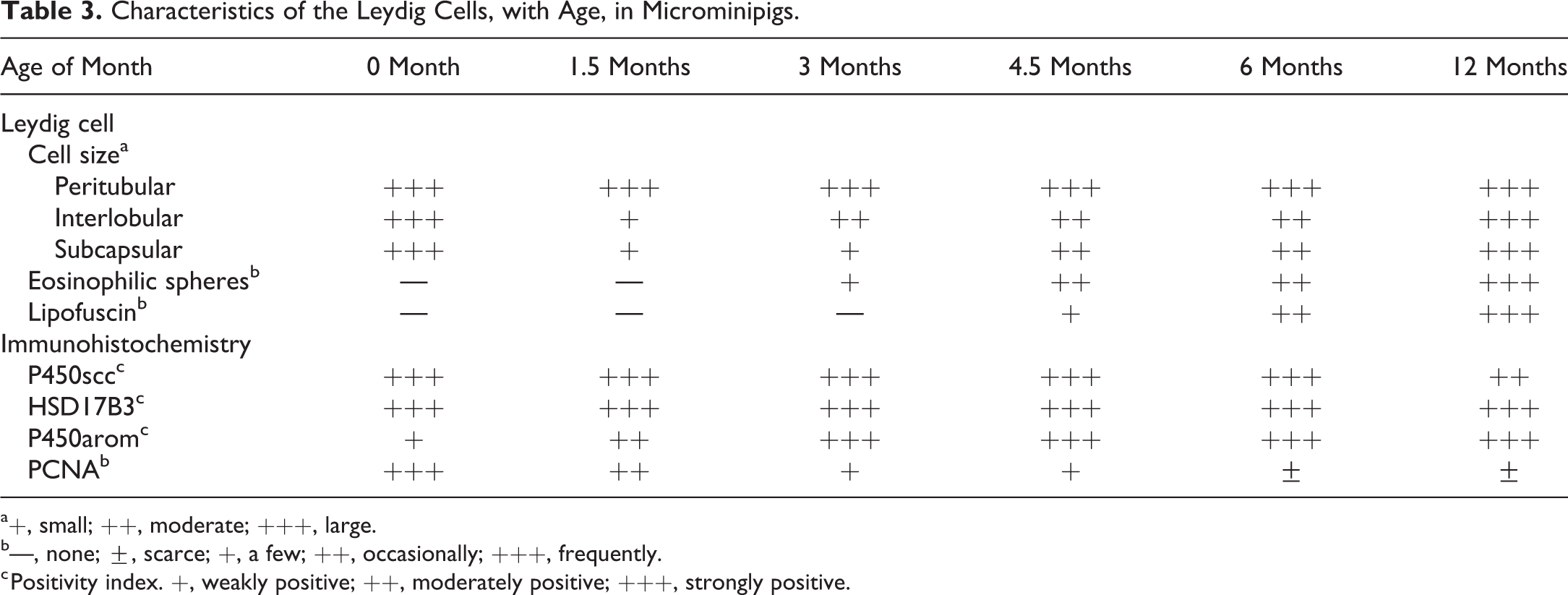
a+, small; ++, moderate; +++, large.
b—, none; ±, scarce; +, a few; ++, occasionally; +++, frequently.
c Positivity index. +, weakly positive; ++, moderately positive; +++, strongly positive.
In addition, the following spontaneous changes were observed: foci of extramedullary hematopoiesis, composed of small round cells with intensely basophilic nuclei and scant cytoplasm, within the testicular interstitium of the all animals at 0 month of age (Figures 3a and 8a), abnormally large, binucleated Leydig cells on rare occasions at 1.5 months to 6 months of age (Figures 2c inset and 3b).
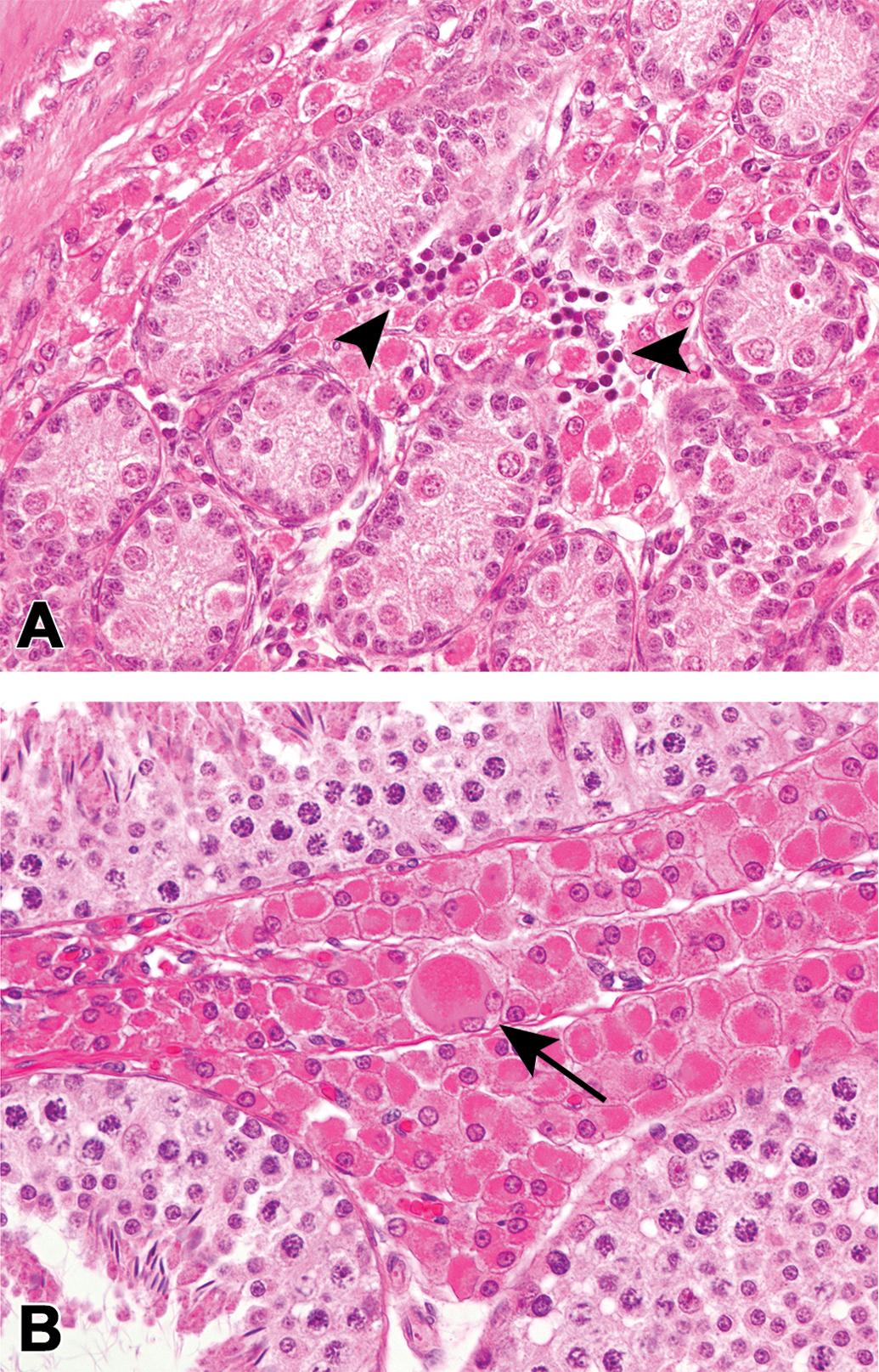
Spontaneous changes in the testicular interstitium of the microminipigs. (a) Extramedullary hematopoiesis, the aggregates of small round cells that have intensely basophilic nuclei with scant cytoplasm, at 0 month of age (arrowheads). (b) A plump binucleated Leydig cell, the cell that is much larger than the other Leydig cells seen in this figure, at 1.5 months of age (arrow). Hematoxylin and eosin staining.
Morphometry of the Testicular Composition and the Leydig Cell Size
Changes in the testicular composition, with age, are shown in Table 2. The Leydig cells occupied half of the cross section of the testis at 0 month of age, and then their ratio dropped to approximately 20% at 1.5 and 3 months of age. The area occupied by the Leydig cells, gradually increased from 4.5 months of age, reaching 35% at 6 and 12 months of age. The area of the seminiferous tubule showed changes contrary to that of the Leydig cells: increased up to three quarter of the testis from 0 to 3 months of age and then dropped to approximately 65% at 6 and 12 months of age. The interstitial tissue mainly consisted of connective tissue and blood vessels. The area of the interstitial tissue was approximately 3% of the testicular composition at any age investigated. Figure 4 represents the changes in the Leydig cells size with age. The peritubular, interlobular, and subcapsular Leydig cells were approximately 17 µm in length at 0 month of age. The interlobular and subcapsular Leydig cells decreased to 10 µm in length at 1.5 months of age, the smallest size observed among all the investigated ages, and then gradually increased, reaching 18 µm in length at 12 months of age. On the other hand, the peritubular Leydig cells showed almost no changes or a slight increase in size with age.
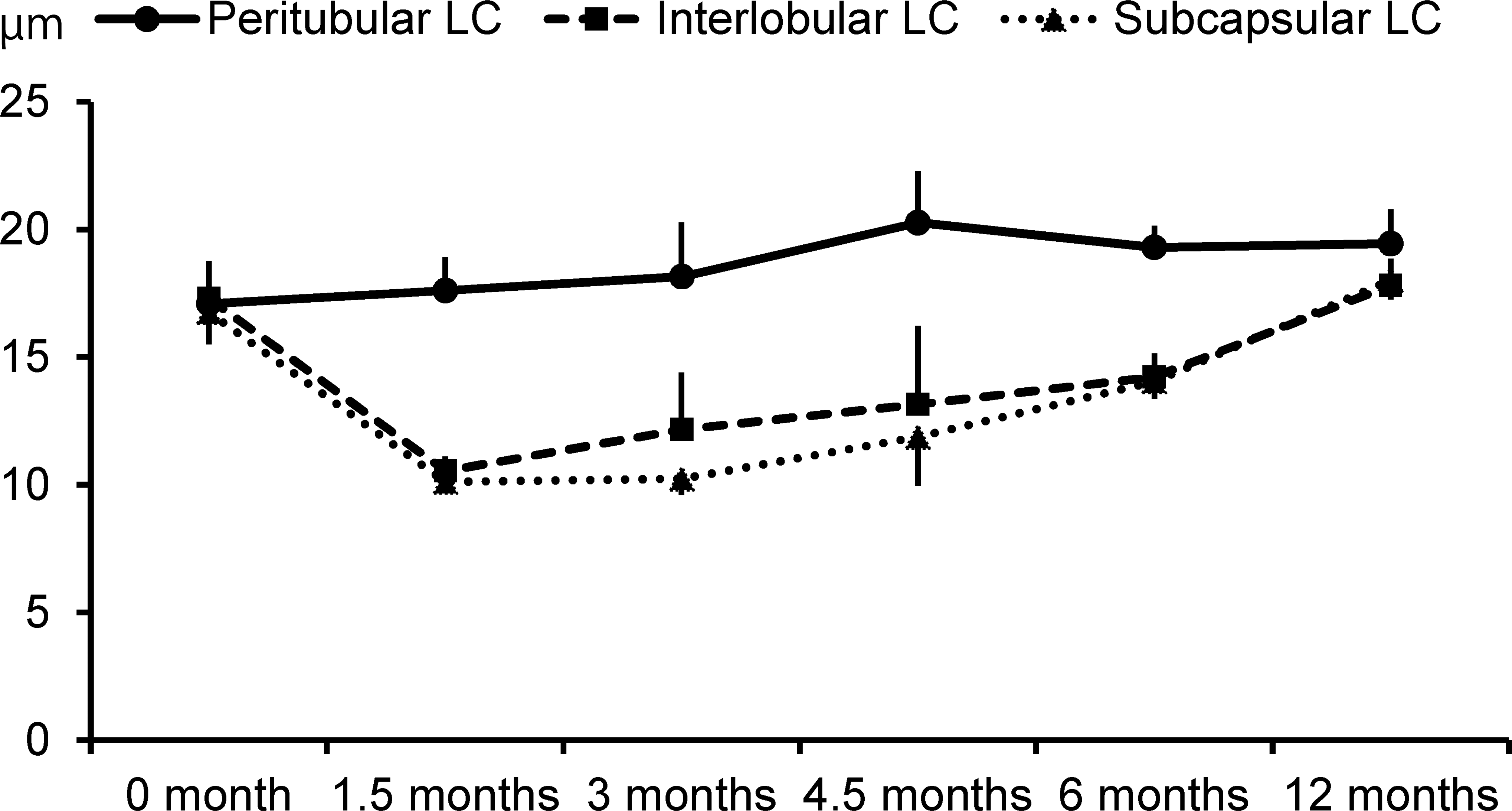
Changes in the length of the peritubular, interlobular, and subcapsular Leydig cells with age (mean ± standard deviation). LC = Leydig cell.
Immunohistochemistry
All Leydig cells, at any investigated age and any location in the testis, were immunopositive for P450scc, HSD17B3, and P450arom, although there were slight differences in intensity in various parts of the cytoplasm (Figures 5 –7 and Table 3). While the immunopositivity tended to be concentrated in the cell center, the peripheral cytoplasm was relatively free for HSD17B3 and P450arom, a positive reaction for P450scc was evident with various sized spheres observed from 3 months of age, as well as the granular cytoplasm at all ages (Insets in Figures 5b, 6b, and 7b). PCNA-positive nuclei were occasionally observed in the Leydig cells throughout the testis at 0 and 1.5 months of age (Figure 8a). At 3 months of age and thereafter, the PCNA positive cells in the peritubular area were hardly observed, while there were a few PCNA positive cells in the interlobular and subcapsular areas (Figure 8b). The PCNA-positive cells in the interlobular and subcapsular areas later decreased and were scarcely observed at 12 months of age. In the sections stained with PCNA, the nuclei of the gonocytes and a few Sertoli cells at 0 month of age, and those of spermatogonia and the early phase spermatocytes at 1.5 months of age and thereafter, showed strong immunopositivity, but the nuclei of the spermatids were completely negative. The hematopoietic cells observed at 0 month of age were positive for PCNA (Figure 8a), but negative for P450scc, HSD17B3, P450arom, and AIF-1/Iba-1. Macrophages and the mast cells observed within the testis are shown in Table 4. Macrophages were present mainly in the peritubular and interlobular areas, and the rete testis, but few were also present in the subcapsular area and the tunica albuginea, regardless of the age. Most of the macrophages were located along the basal membrane of the tubules and the interstitial collagenous tissue (Figure 9). Mast cells were observed only in tunica albuginea at 0 month of age. At 1.5 months of age and thereafter, mast cells were often present in the interlobular area, rete testis, and the tunica albuginea, and a few were present in the peritubular and subcapsular areas (Figure 9). In addition, several mast cells occasionally gathered around the arterioles located in the interlobular area and the tunica albuginea.
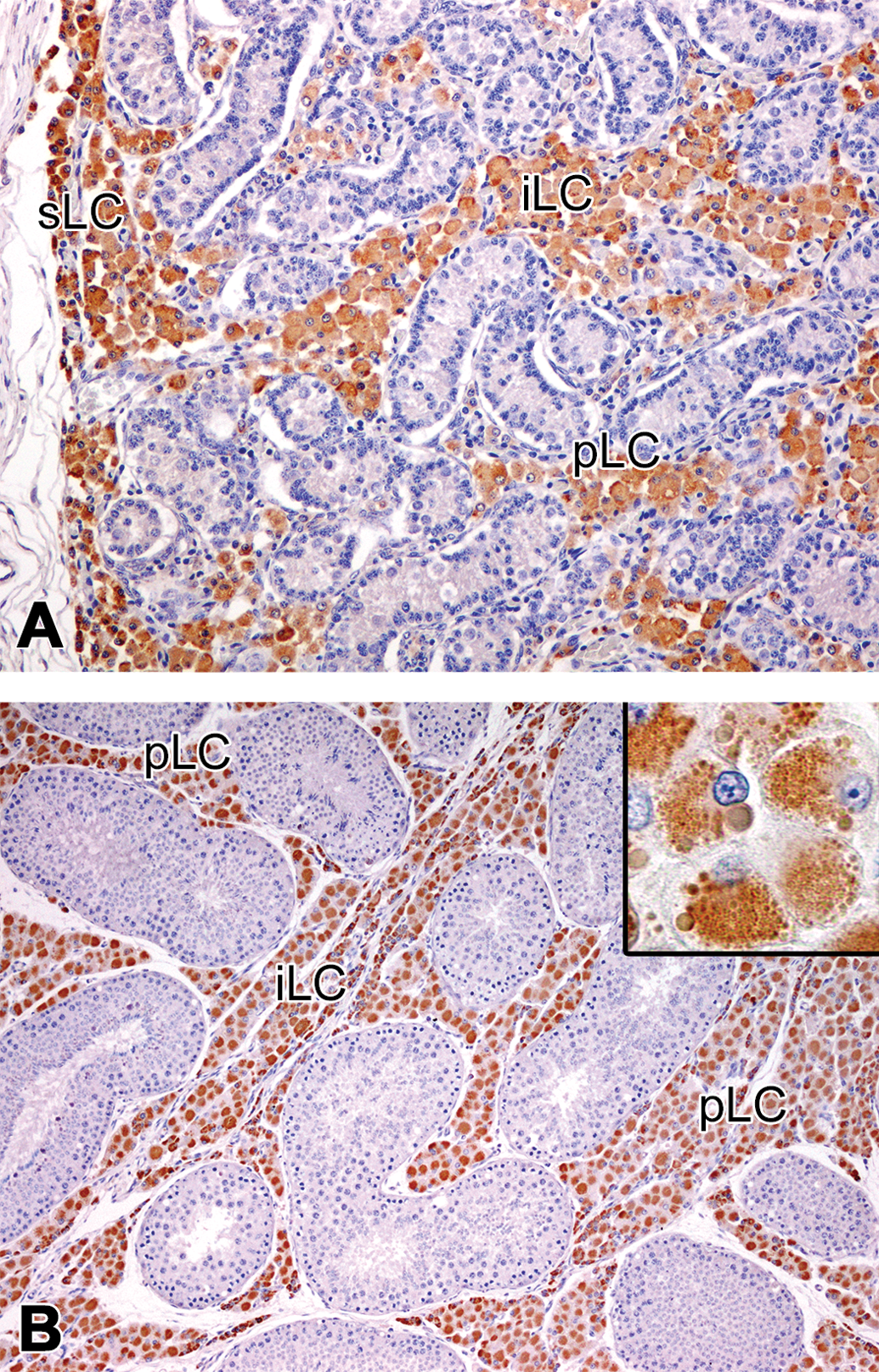
Immunolocalization of cytochrome P450 side chain cleavage (P450scc) in the testis of microminipigs. (a) At 0 month of age, Leydig cells in the peritubular, interlobular, and subcapsular areas were positive for P450scc. (b) At 4.5 months of age, the peritubular and interlobular Leydig cells were positive for P450scc. Inset: Higher magnification of the immunopositive peritubular Leydig cells. Various-sized spheres were P450scc-positive. pLC = peritubular Leydig cell; iLC = interlobular Leydig cell; sLC = subcapsular Leydig cells. Counterstained with hematoxylin.
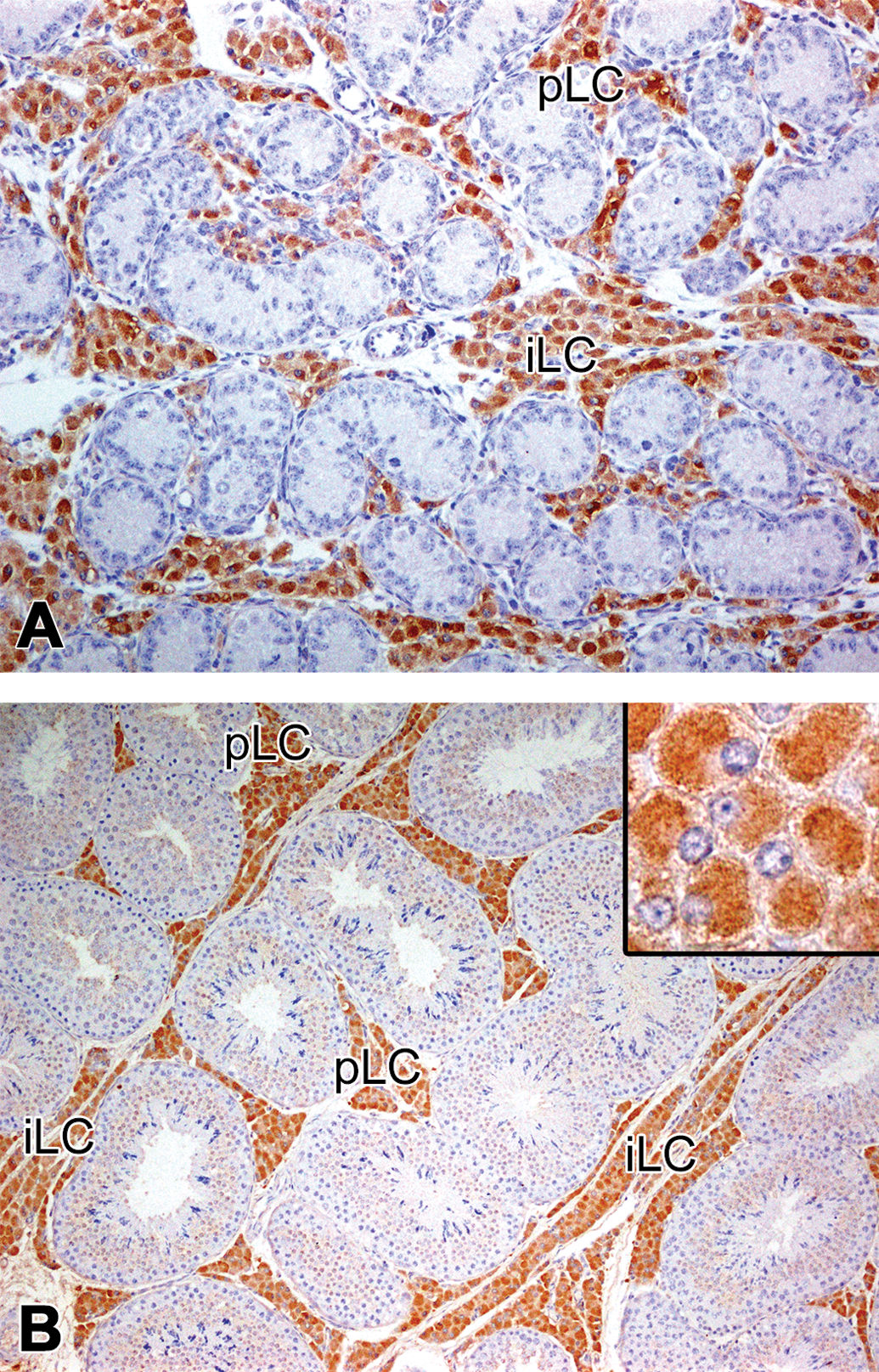
Immunolocalization of 17β-hydroxysteroid dehydrogenase 3 (HSD17B3) in the testis of microminipigs. At 0 month (a) and 4.5 months (b) of age, the peritubular and interlobular Leydig cells were strongly positive for HSD17B3. (b) Inset: Higher magnification of the positive Leydig cells. The central area in the cell was densely positive, while the peripheral cytoplasm was relatively free. pLC = peritubular Leydig cell; iLC = interlobular Leydig cell. Counterstained with hematoxylin.
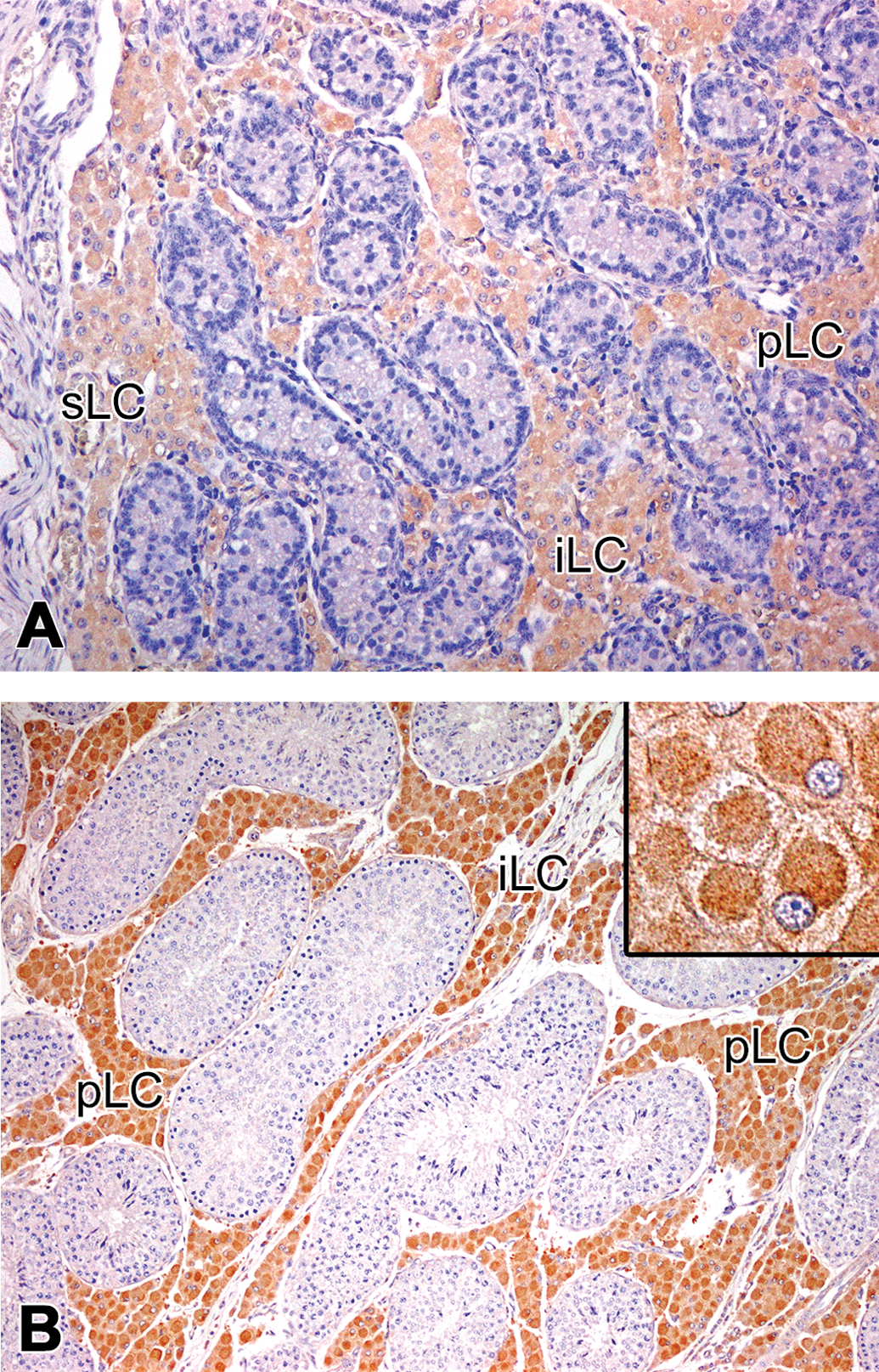
Immunolocalization of cytochrome P450 aromatase (P450arom) in the testis of microminipigs. At 0 month (a) and 4.5 months (b) of age, the peritubular, interlobular, and subcapsular Leydig cells were positive for P450arom. (b) Insets: Higher magnification of the positive Leydig cells. P450arom appeared to be concentrated in the cell center while the peripheral cytoplasm was almost free. pLC = peritubular Leydig cell; iLC = interlobular Leydig cell; sLC = subcapsular Leydig cell. Counterstained with hematoxylin.
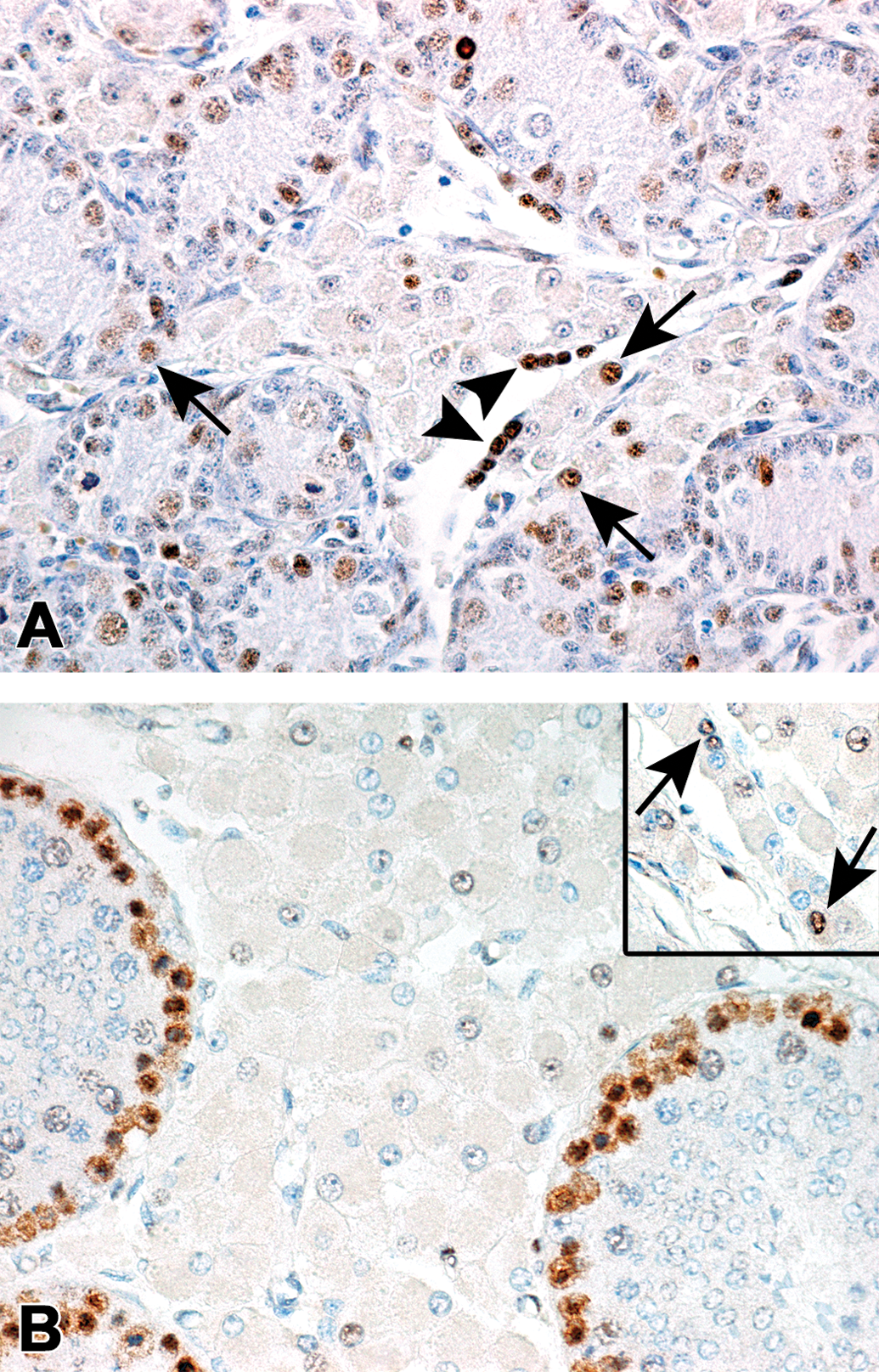
Immunoreactivity of the Leydig cells against the proliferating cell nuclear antigen (PCNA) in microminipigs. (a) At 0 month of age, nuclei of the Leydig cells were occasionally positive (arrows). The arrowheads indicated PCNA-positive nuclei in extramedullary hematopoiesis. (b) At 4.5 months of age, PCNA-positive cells were no longer observed in the peritubular Leydig cells. In contrast, spermatogonia and early phase spermatocytes in the seminiferous tubule were strongly positive for PCNA. Inset: PCNA-positive interlobular Leydig cells (arrows). Counterstained with hematoxylin.
The Extent of Macrophages and Mast Cells Present in the Testis with Age in Microminipigs.
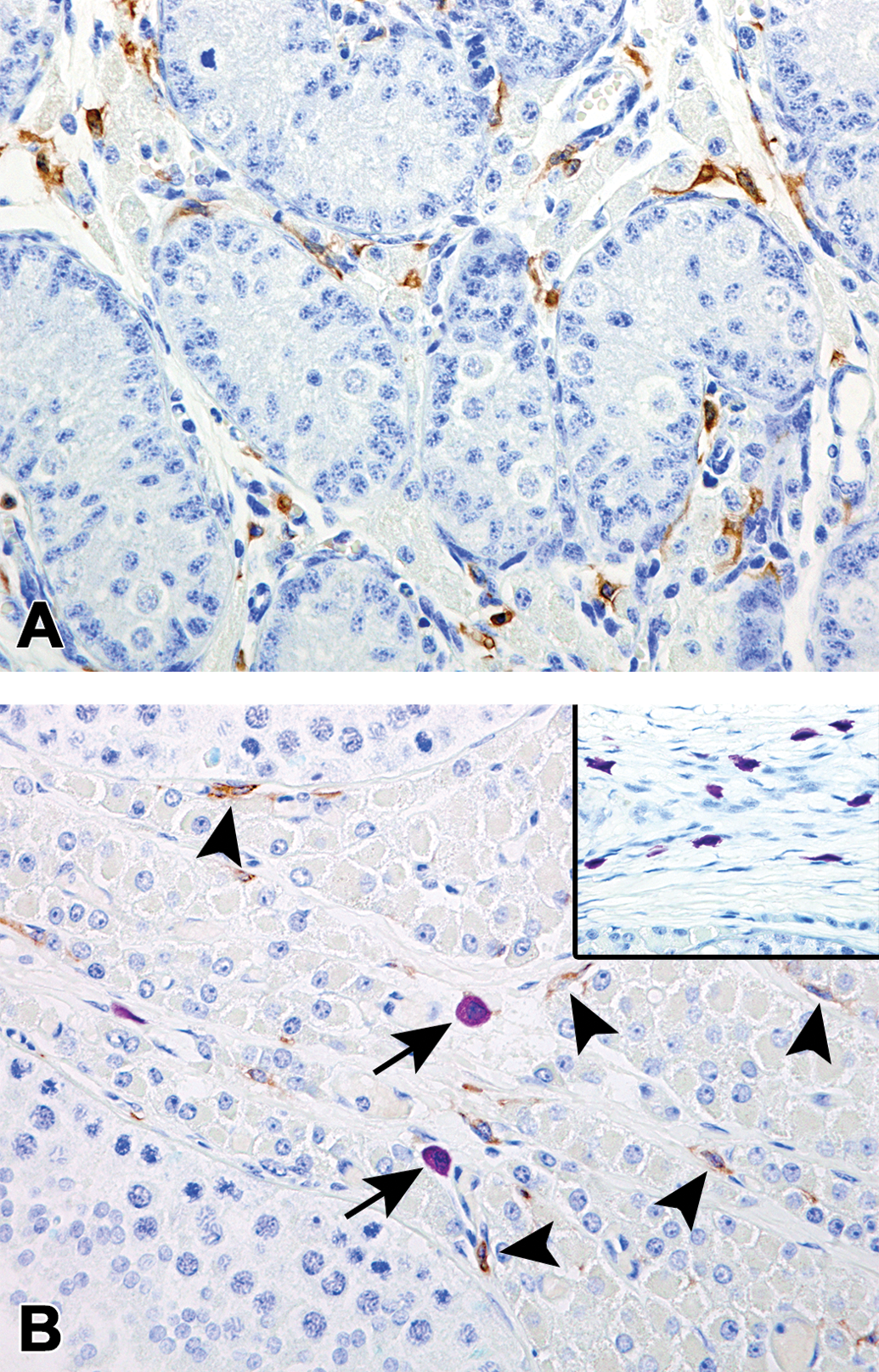
Photomicrographs of the macrophages and mast cells located in the testis of microminipigs. (a) At 0 month of age, macrophages were frequently observed but no mast cells were seen in the interstitium. (b) At 12 months of age, macrophages with scant cytoplasm were located along the basal membrane of the seminiferous tubules and the interstitial collagenous tissue (arrowheads). A few mast cells were also seen in the interlobular area (arrows). Inset: Numerous mast cells within the tunica albuginea. Immunohistochemistry against allograft inflammatory factor 1 (AIF-1/Iba-1) counterstained with toluidine blue.
Serum Testosterone and Estradiol Levels
The serum testosterone and estradiol concentrations, with age, are shown in Table 2. The testosterone concentration tended to increase from 0 month to 6 months of age. The values differed considerably between individuals at each investigated age, except for 0 month of age. In contrast, the estradiol concentration did not increase as much as the testosterone, and there was substantial individual variability at 3 months of age and thereafter. Individual data obtained in the present study are shown in Supplemental Table.
Discussion
The development and characteristics of spermatogenesis in the various laboratory animals are well illustrated, but there have been limited studies on the interstitial tissue of the testis, particularly in minipigs. This report is the first to describe the development and characteristics of the testicular interstitium in microminipigs, based on the histological and hormonal investigations.
Compared to other mammalian species, the testis of a pig is remarkable, due to the abundance of its interstitial compartment, especially the Leydig cells (Bascom and Osterud 1927). In the present study, two distinct types of Leydig cells were recognized: one was a large polygonal cell with abundant eosinophilic cytoplasm and the other was a small cell with scant cytoplasm. The third type was a minor one: a transitional cell, the size of which was between the two types of the cell mentioned above. The small Leydig cells first appeared in the interlobular and subcapsular areas at 1.5 months of age, and afterward, the small cells were converted into large Leydig cells with age and disappeared completely at 12 months of age. Similar observations in the development of the Leydig cells were obtained in the domestic boar (Van Straaten and Wensing 1978; Choi et al. 2009). Thus, we can assume that the large Leydig cell was a well-matured cell, and the small Leydig cell was an immature or a reserve cell generated sometime between 0 and 1.5 months of age, even though the differences in the immunoreactivity against steroidogenic enzymes were not detected regardless of the Leydig cell size. Progenitor cells of the Leydig cell are derived from spindle-shaped mesenchymal cells, located close to either the seminiferous tubules or the blood vessels in the testicular interstitium (Mendis-Handagama and Ariyaratne 2001; Davidoff et al. 2004; Teerds et al. 2007). Although finding a clue to the origin of the Leydig cells is beyond the scope of this study, the small Leydig cell in microminipigs may arise from a mesenchymal precursor cell residing in the interlobular and/or subcapsular interstitial areas.
Testosterone plays an important role in the development and maintenance of spermatogenesis, male accessory glands, and external genitalia, as well as sexual behavior (Joshi and Raeside 1973; Colenbrander, Frankenhuis, and Wensing 1982; Waites, Speight, and Jenkins 1985; McEwan and Brinkmann 2016). During sexual development in male mammals, serum testosterone levels increase sharply at the prepubertal to pubertal phase (Colenbrander, Frankenhuis, and Wensing 1982; Waites, Speight, and Jenkins 1985; França et al. 2000). On the other hand, lipofuscin, often considered a hallmark of aging, accumulates progressively over time in the lysosomes of post mitotic cells (Terman and Brunk 1998; Brunk and Terman 2002). In general, lipofuscin pigments start to deposit in the Leydig cells around the age of sexual maturity and increased in prominence in the aged testis (L. Johnson and Neaves 1981; Almahbobi et al. 1988; Trainer 2012). In the present study, the serum testosterone levels increased substantially between 1.5 and 4.5 months of age, and lipofuscin pigments in the Leydig cells started to appear from 4.5 months of age. According to spermatogenesis and the development of accessory glands, the stage of sexual development in microminipigs has been reported as follows: prepubertal, 1.5 months of age; pubertal, 3 months of age; and sexually mature, 4.5 months of age (Kangawa et al. 2016). Considering these aspects, our data confirmed that the change in serum testosterone levels and the age of lipofuscin accumulation in the Leydig cells coincided well with the sexual development of in male microminipigs. In addition, eosinophilic spheres, which were positive for P450scc, started to appear in the Leydig cells at 3 months of age and thereafter. However, the significance of the spheres, in the pig testis, has not been elucidated so far. Similar age-related eosinophilic material, known as Reinke crystalloids, is present in human Leydig cells (Trainer 2012). The nature of these crystalloids is unknown, but they are presumed to represent a protein product of the cell (Nagano and Otsuki 1971). As P450scc is a membrane-bound protein associated with the mitochondrial membrane (Payne and Hales 2004), the spheres might represent some functional changes in the steroidogenesis with age.
A remarkable feature of the male domestic boar is high circulating estrogen concentrations, compared to the males of other species and females of the same species (Claus and Hoffmann 1980; Setchell et al. 1983; Schwarzenberger et al. 1993; Clapper, Clark, and Rempel 2000; Kanematsu et al. 2006). In domestic boars, serum estradiol levels gradually increase with age, and the estrogens act synergistically with testosterone, to stimulate the secretions from accessory glands and their sexual behavior (Joshi and Raeside 1973; Clapper, Clark, and Rempel 2000; Kanematsu et al. 2006). Estradiol concentrations in male microminipigs showed no obvious age-related changes in the present study, but the range of serum estradiol levels was similar to those reported in a previous study on the domestic boar (Clapper, Clark, and Rempel 2000; Kanematsu et al. 2006). Even though the role of estrogens in male reproductive system is still unclear, advances made using animal and human models suggest that the estrogens influence male sexual development, testis function, the hypothalamic–pituitary–testis axis, spermatogenesis, and ultimately male fertility (Rochira et al. 2016; K. J. Johnson, Heger, and Boekelheide 2012). As the regulation of male reproduction differs significantly between species, the biological significance of high rates of estrogen secretion in male microminipigs remains to be uncovered.
Comparative studies of the Leydig cells have provided evidence that the microminipigs are different from other species. Rats are the most commonly used experimental animals for reproduction and toxicology studies; therefore, the morphological features of their testis are well-documented (Russell et al. 1990). Rats have a small number of Leydig cells that are frequently within the lymphatic spaces, largely bathed in lymph fluid, and clustered around blood vessels. In addition, the resident tissue macrophages constitute about 20% of the interstitial cell population within the rat testis (Dirami, Poulter, and Cooke 1991; Hutson 1992). This is in contrast to the dog and monkey testis, where the interstitial areas have discrete lymphatic channels and the Leydig cells are embedded in the loose connective tissue (Fawcett, Neaves, and Flores 1973). In the present study, the interstitial compartment occupied approximately 30% to 35% of the cross section of the testis at 4.5 to 12 months of age, which is the age of sexual maturity in male microminipigs. The interstitium consisted mainly of closely packed Leydig cells with a few macrophages and mast cells, but lymphatics were scarce. According to published literature, the testicular compositions of the Leydig cells in sexually mature animals are reported as follows: rat, 2.8%; mouse, 4.3%; dog, 6.7%; domestic boar, 10%; monkey, 0.8%; and man, 6.4% (Fouquet, Meusy-Dessolle, and Dang 1984; Lowseth et al. 1990; França, Avelar, and Almeida 2005). Therefore, the large amount of the Leydig cells in microminipigs is one of the most prominent difference compared with other laboratory animals and domestic boar.
Extramedullary hematopoiesis is the formation and development of blood cells outside the medullary spaces of the bone marrow; it usually occurs postnatally as a pathological compensation due to a result of bone marrow failure (Johns and Christopher 2012; Chiu et al. 2015). In neonatal mice, humans, dogs, cats, cattle, and pigs, residual sites of fetal hematopoiesis persist mainly in spleen, but also in liver and lymph nodes, and then regress with growth and disappear prior to adulthood (Johns and Christopher 2012). In this study, extramedullary hematopoiesis was observed in the testes of all six animals investigated at 0 month of age. Since erythropoietin mRNA has been found in the testes of neonatal pigs, and the testes express significant amounts of the erythropoietin receptor, it has been suggested that a paracrine function of erythropoietin, inducing hematopoietic activity, exists in this organ (David et al. 2002, 2005). In addition, histological analyses showed that the testes of Yucatan minipigs, younger than 2 months old, had hematopoietic activity in 46.7% of the studied animals (Helke et al. 2016). Although it could be seen as an unusual finding, extramedullary hematopoiesis in the testis of microminipigs could also be interpreted as a normal physiological function.
In addition to phagocytosis of cellular debris from the testicular interstitium, testicular macrophages have a functional relationship with the Leydig cell development, in which they provide essential growth and differentiation factors to the Leydig cells (Khan, Teerds, and Dorrington 1992; Haschek, Rousseaux, and Wallig 2010). Mast cells are intricately involved in inflammation and fibrosis in a variety of organ systems. In particular, testicular mast cells increase in number at puberty, and their activation and migration are influenced by estrogens (Gaytan et al. 1989; Mechlin and Kogan 2012). In the present study, testicular macrophages were observed regardless of the age, while mast cells were present in the testis from 1.5 months of age, suggesting that the testicular macrophages and the mast cells in microminipigs have functions similar to those that have been described above. In the toxicity studies, macrophages and mast cells infiltrate in accordance with necrosis of the Leydig cells, induced by specific toxins (Molenaar et al. 1985; Bartlett, Kerr, and Sharpe 1986; Jackson et al. 1986). Moreover, increased numbers of mast cells have been reported in the testis of infertile men (Behrendt et al. 1981; Maseki et al. 1981). Additional investigation regarding the interaction between cytokines and bioactive substances in the testis will be needed to clarify the significance of the testicular macrophages and mast cells in microminipigs.
The present study revealed that the large amount of Leydig cells were typical of the testicular interstitium in microminipigs and that the development of the Leydig cells differed morphologically based on their location in the testis. Immunohistolocalization of the steroidogenic enzymes, and the presence of serum steroid hormones, suggested that the Leydig cells were capable of producing steroid hormones from 0 month of age. In addition, the age when serum testosterone levels increased coincided well with the previously reported age of sexual development (Kangawa et al. 2016). However, the significance of high serum estradiol levels in male microminipigs remains to be elucidated. As the histological manifestations of injury to the Leydig cell are generally identified as hyperplasia/hypertrophy, atrophy, and necrosis of the Leydig cells, together with macrophage and mast cell response (Haschek, Rousseaux, and Wallig 2010; Creasy et al. 2012), we expect the information obtained in the present study to be useful for future toxicity studies. Since there are species differences in endocrine feedback and in regulation of hormone receptor expression, therefore, further studies are required to comprehensively understand the function of the reproductive organs, and the morphological manifestations of the response to injury in microminipigs.
Supplemental Material
Supplemental Material, DS1_TPX_10.1177_0192623319827477 - Histological Changes of the Testicular Interstitium during Postnatal Development in Microminipigs
Supplemental Material, DS1_TPX_10.1177_0192623319827477 for Histological Changes of the Testicular Interstitium during Postnatal Development in Microminipigs by Akihisa Kangawa, Masayoshi Otake, Satoko Enya, Toshinori Yoshida and Masatoshi Shibata in Toxicologic Pathology
Footnotes
Acknowledgments
The authors would like to thank Prof. Motoki Sasaki (Obihiro University of Agriculture and Veterinary Medicine) for his advice during the early days of the study and Mr. Toshiaki Fukumura (Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center) for taking good care of the animals.
Author Contributions
Authors contributed to conception or design (AK); data acquisition, analysis, or interpretation (MO, SE, TY, MS); drafting the manuscript (AK); and critically revising the manuscript (MO, SE, TY, MS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
