Abstract
The microminipig has become an increasingly attractive animal model for various experimental practices because of its manageable size; however, studies of the histological features of the female reproductive organs in microminipigs are limited. The present study investigates the sexual development of the reproductive organs and the cyclical changes during the estrous cycle in female microminipigs. The ovaries, oviducts, uteri, and vaginal tissues from 33 animals aged 0 to 26 months were utilized in this study. By evaluating the large tertiary follicles, corpora lutea, and the regressing corpora lutea, we estimated that female microminipigs reached puberty at approximately 5 months of age and sexual maturity at 8 months of age. The appearance of the follicles and corpora lutea in the ovaries, as well as the epithelium in other reproductive organs, was synchronized with each phase of the estrous cycle and was identical to that in common domestic pigs. In addition, several spontaneous findings were observed, including mesonephric duct remnants adjacent to oviducts and mineralization in ovaries. Understanding the normal histology of the reproductive organs in microminipigs is crucial for advancing pathological evaluations for future toxicological studies.
Keywords
Minipigs have become particularly valuable nonrodent animal models for toxicological and pharmacological studies, replacing dogs and primates (Svendsen 2006; Bode et al. 2010). Among various strains of minipigs, microminipigs (Fuji Micra, Inc., Shizuoka, Japan) are becoming increasingly attractive alternatives for various experimental applications because of their manageable size of approximately 10 kg at 6 months of age (Kaneko et al. 2011). Background reference data on microminipigs have been collected and reported for several years. In the female reproductive system, the characteristics and the hormone profiles throughout the estrous cycle have been precisely described (Noguchi et al. 2015). However, there have been limited studies regarding the developmental and estrous cycle–dependent histological features of the female reproductive organs.
In general toxicology studies, the use of sexually mature animals is preferable, since the effects of tested drugs can differ between mature and immature animals. Moreover, the histological features of the immature reproductive organs can be misinterpreted as an effect of the test substance when using pubescent animals. In addition, in juvenile toxicity studies, toxicologic pathologists must be familiar with normal organ development in order to appropriately detect precocious or delayed development and differentiate this from abnormal development (Picut and Parker 2017). Moreover, animals must be of the optimal age for use in reproductive and developmental toxicity studies (Howroyd, Peter, and de Rijk 2016). Therefore, it is essential to understand organ development and sexual maturity in the animals utilized in these experiments. The age of sexual maturity in female minipigs has been well-documented and reviewed in several reports (Jørgensen 1998; Bode et al. 2010; Creasy 2012; Tortereau, Howroyd, and Lorentsen 2013; Peter et al. 2016; Howroyd, Peter, and de Rijk 2016). However, to the best to our knowledge, this information is lacking in microminipigs.
It is important for toxicologic pathologists to understand the underlying species-specific physiology for the proper interpretation of apparent differences in morphology. This is particularly important for the female reproductive tract, since the morphology is highly synchronized and extremely sensitive to hormonal perturbations (Halpern et al. 2016). The normal histology of female reproductive organs during the estrous cycle is well illustrated in Göttingen minipigs and is based on the state of the ovaries and on the changes in the appearance of the epithelial cells in the uterus. The cycle is classified into 5 phases: follicular/proliferation (F/P), ovulation, early-luteal (EL), mid-luteal (ML), and late-luteal (LL) phases (de Rijk et al. 2014). It remains uncertain whether the same criteria can be applied to microminipigs and whether minipig-specific features can be observed in microminipigs. Since any discrepancy in histological appearance may indicate an abnormality, a practical approach to the staging and understanding of the normal features of the estrous cycle in microminipigs is indispensable for future toxicology studies.
The objectives of the present study were to histologically evaluate the developmental changes observed in female reproductive organs of female microminipigs aged 0 to 26 months and to ascertain the morphological changes occurring during the estrous cycle in animals aged 8 to 26 months.
Material and Method
Animals
A total of 33 female microminipigs were used in this study. The animals, whose parental generation was provided by Fuji Micra, Inc. (Shizuoka, Japan), were bred and raised in a closed colony at the Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center (Shizuoka, Japan). The minipigs were housed in pairs or in a group (a maximum of 4 animals together) in pens of at least 0.54 m2 area/animal under conventional conditions with a 12-hr light and a 12-hr dark cycle. The animals were weaned at 4 weeks of age and then fed a commercial diet (Marubeni Nisshin Feed Co., Ltd., Tokyo, Japan), as instructed by Fuji Micra, Inc., at 2% to 3% of body weight/animal/day, with
Necropsy and Organ Weights
Before necropsy, all animals except for the stillborn ones were deeply anesthetized with intramascular injections of 0.1 ml/kg of Dormicum (5 mg/ml midazolam, Astellas Pharma, Inc., Tokyo, Japan) and 10 mg/kg of Ketamine (Fujita Pharmaceutical Co., Ltd., Tokyo, Japan). After a senior veterinarian confirmed the unconscious state of the animals, they were euthanized by exsanguination (Close et al. 1996). Body weight was measured at necropsy. Ovaries, oviducts, the uterus (including the cervix), and vagina were collected from each animal, and their weights were measured. Organ weights were expressed as absolute (in g) and relative (to terminal body weight) weights. In addition, the number of corpora lutea was counted macroscopically.
Histology Procedures
The collected tissues were fixed in 10% neutral buffered formalin. Medial sections were obtained for both ovaries. Oviducts were sectioned transversely in the middle of their lengths. Transverse sections of the uterus were made at the uterine horns, near their junction with the uterine body. In animals at 0 days and 1 week of age, whole ovaries, oviducts, and uteri were fixed and embedded in paraffin, since these tissues were too small to be trimmed. The sections corresponding to the abovementioned tissues were made and used for evaluation. The cervix and vagina were sectioned longitudinally in animals at 5 months of age and older. Transverse sections in the middle portion of the cervix and vagina, however, were made in animals at 0 days, 1 week, and 2.5 months of age, because these tissues were too small to cut longitudinally. Fixed samples were then dehydrated in a graded ethanol series and embedded in paraffin. Paraffin blocks were cut in 3-µm sections, and the serial sections were stained with hematoxylin and eosin and with periodic acid–Schiff (PAS) hematoxylin.
Histological Evaluation of Sexual Development
All 33 animals were used for this assessment. The histological development was evaluated using light microscopy by focusing on the changes in cellular appearance and microscopic structures. In the ovaries, the presence of the germ cell nests and the various follicular stages was recorded. The terminology used in the present study is as follows: germ cell nest, aggregation of oogonia derived from primordial germ cells; primordial follicle, oocyte surrounded by a simple squamous epithelium of flattened pregranulosa cells; primary follicle, oocyte surrounded by a simple epithelium of cuboidal granulosa cells; secondary follicle, oocyte surrounded by multiple layers of granulosa cells and a few theca cell layers; tertiary follicles, follicles possessing an antrum and surrounded by multiple layers of theca cells; and Graafian follicles, macroscopically visible preovulatory follicles (5 to 8 mm in diameter), possessing large fluid-filled cavities (Tortereau, Howroyd, and Lorentsen 2013; de Rijk et al. 2014; Dixon et al. 2014; Wear, McPike, and Watanabe 2016). Besides corpora lutea and regressing corpora lutea, atretic follicles were also recorded. In other organs, the histological development was evaluated based on the appearance of the epithelium. In addition, spontaneous findings in each organ were noted. The nomenclature used was as previously reported (Dixon et al. 2014).
Estrous Cycle–dependent Histological Morphology
Of all animals included in this study, a total of 12 animals (4 animals each at 8, 12, and 26 months of age) that had shown at least 1 regular estrous cycle (standing heat to standing heat) were used for this evaluation. To obtain histological material for all the phases of the estrous cycle, the time of necropsy was individually determined based on the signs of standing heat. Necropsy dates were referenced according to a previous report (de Rijk et al. 2014) and set as follows: the first day of standing heat (
Statistical Analysis
All data are represented as means and standard deviations. Paired
To determine whether there was a significant difference in the weights of reproductive organs between immature and pubertal to early mature females in microminipigs, 14 animals at 5 to 8 months of age were divided into two groups (the animals with no corpus luteum and the animals presenting a corpus luteum), and then the Student’s
Results
Biometric Data
Animal body weights (
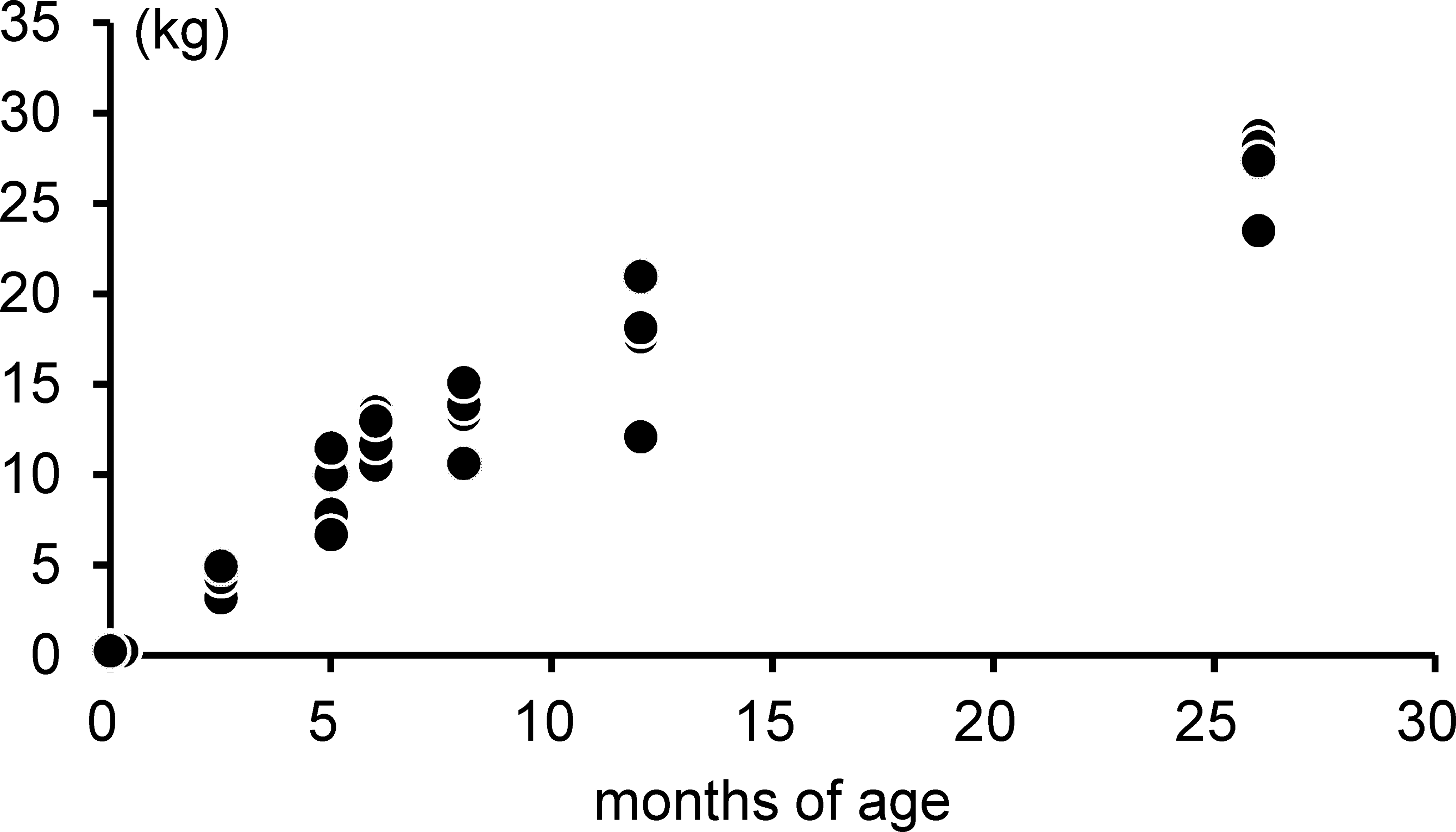
The development of the body weight in female microminipigs.
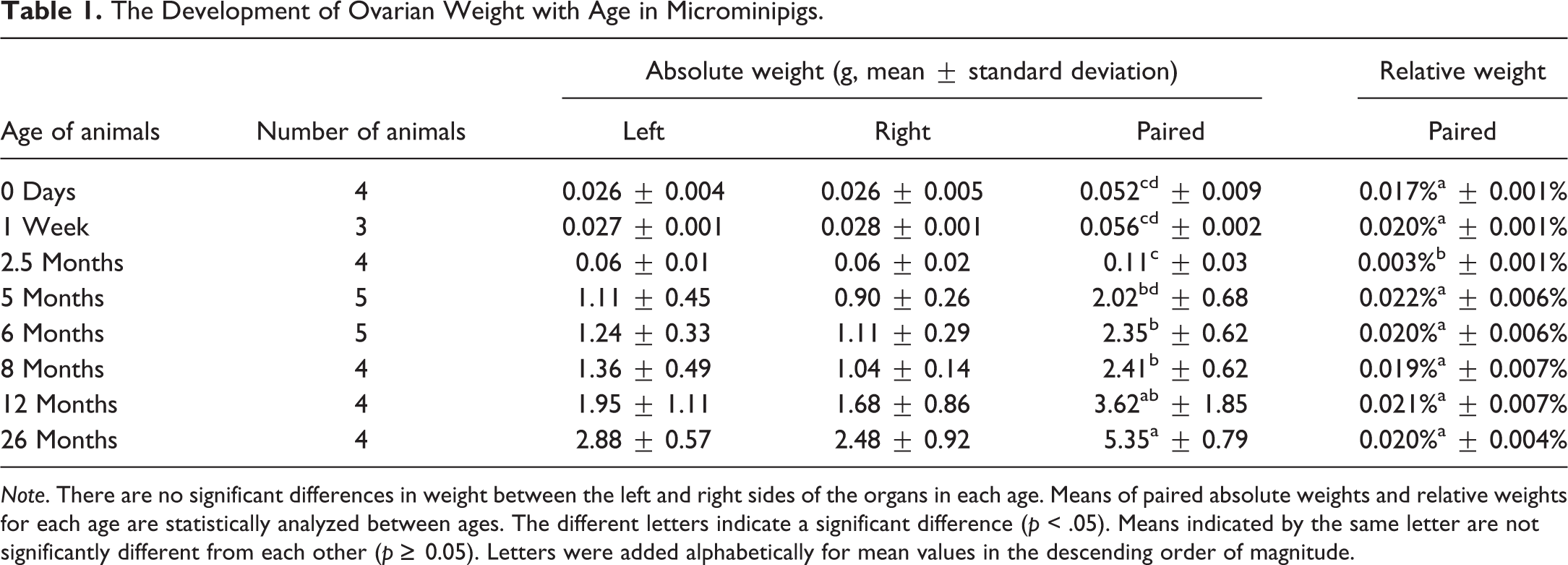
The Development of Ovarian Weight with Age in Microminipigs.
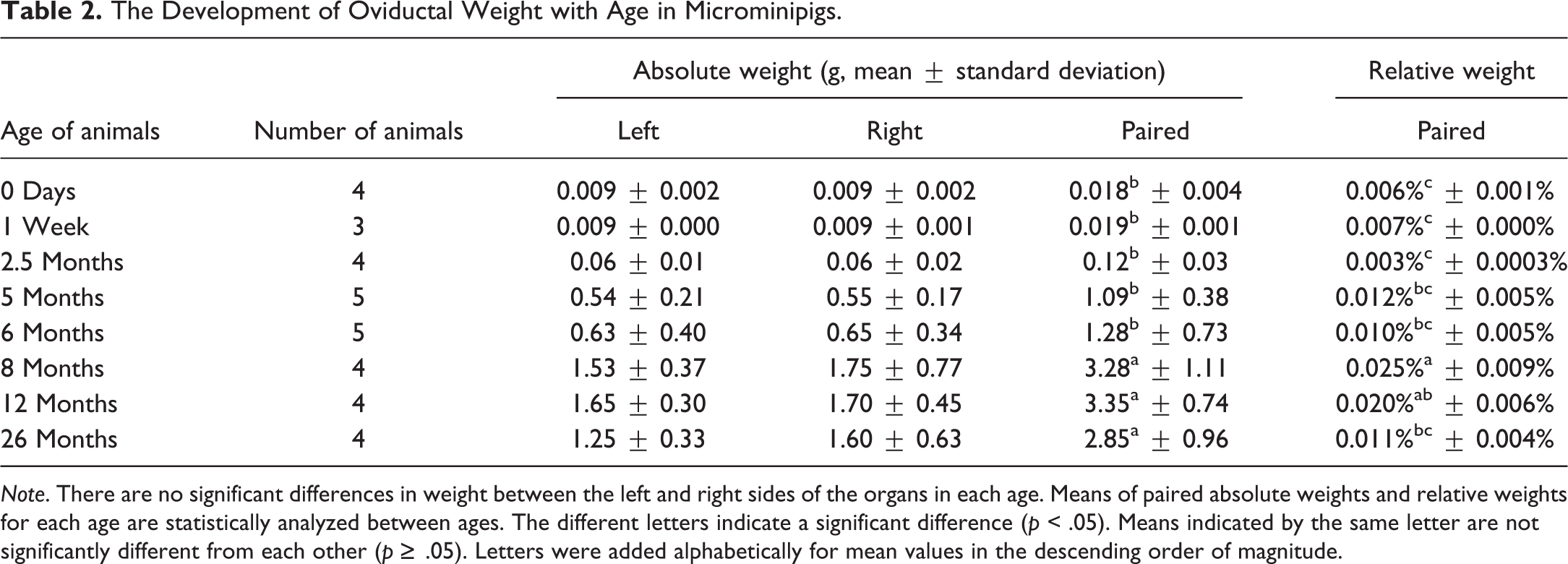
The Development of Oviductal Weight with Age in Microminipigs.
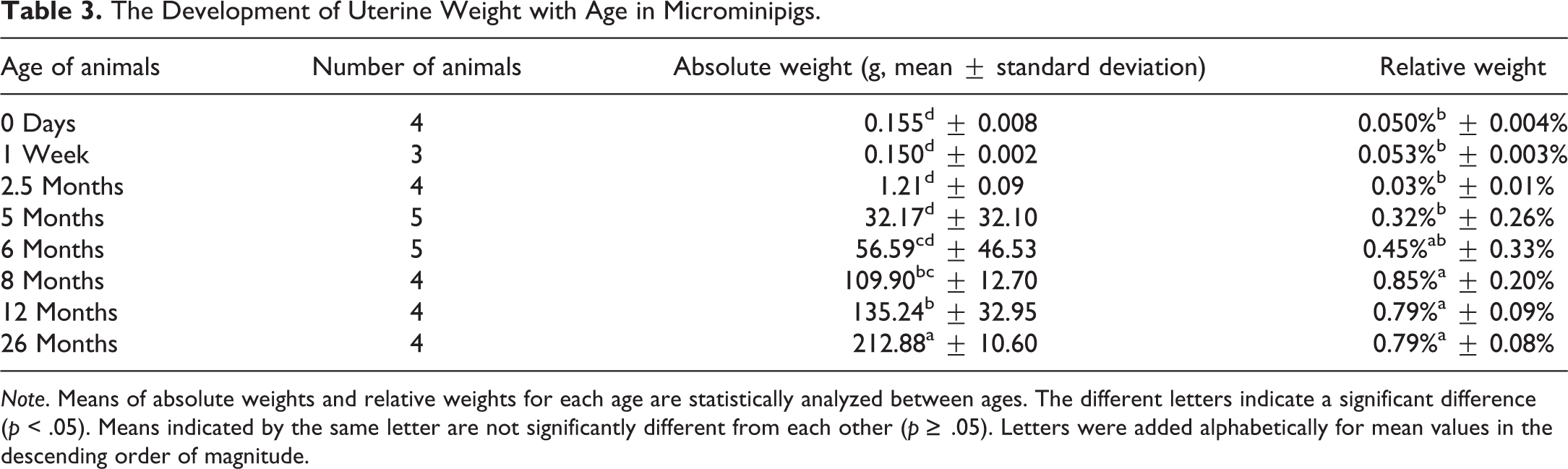
The Development of Uterine Weight with Age in Microminipigs.
The Development of Vaginal Weight with Age in Microminipigs.

The presence and number of corpora lutea identified by macroscopic observation are presented in Table 5. The total number of corpora lutea ranged from 4 to 9 in the animals investigated. Notably, several females at 8 and 12 months of age presented corpora lutea only on one side of the ovaries.
The Presence and Number of Macroscopic Corpora Lutea with Age in Microminipigs.

Histological Development
Ovaries
Figure 2 shows each stage of the follicular development observed in microminipigs. Histological features with age were as follows. At 0 days of age, the ovarian cortex consisted mainly of germ cell nests, and the internal areas of the nests were individualized to form primordial follicles (Figure 3A). Oogonial mitoses were occasionally observed. At 1 week of age, some germ cell nests were still present but only in a limited part of the subcapsular cortex. Most of the cortex was composed of primordial follicles, a few of which had developed to form primary follicles in the internal part of the cortex (Figure 3B). Secondary follicles first appeared at 2.5 months of age. Some of these contained small eosinophilic fluid-filled spaces, which were positive for PAS staining, within the layers of granulosa cells (Call–Exner bodies, Figure 3C). Germ cell nests were no longer observed at 2.5 months or in older animals. At 5 months of age, follicles at various stages of development, including tertiary and atretic follicles, were present in all the studied animals (Figure 3D). One of the 5 animals had corpora lutea but not regressing corpora lutea. This indicated that the female had started her first estrous cycle just before 5 months of age. Two of the 5 animals at 6 months of age presented corpora lutea. One of the animals presented well-developed stages of corpora lutea, while the other showed regressing corpora lutea, with growing large tertiary follicles (Figure 3E). However, hyalinized regressing corpora lutea, which represent the corpora lutea derived from the cycle preceding the previous cycle, were not yet present in any animals at 6 months of age. After 8 months of age, all animals presented hyalinized regressing corpora lutea, as well as corpora lutea, the appearance of which depended on the stage of the estrous cycle at necropsy (Figure 3F). Hence, all females had previously experienced several estrous cycles.
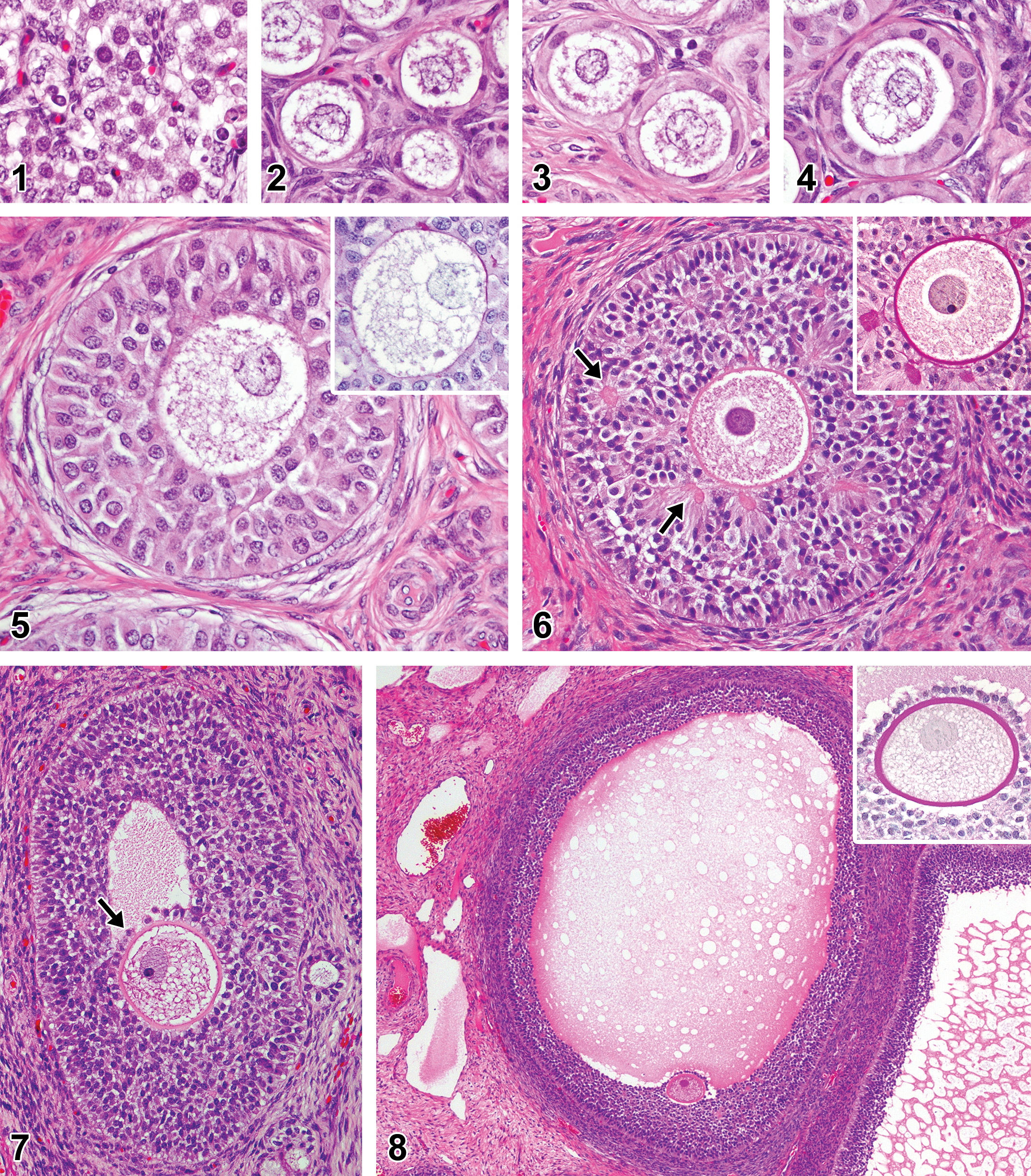
Follicular development in microminipigs. (1) Germ cell nest: cluster of germ cells with round nuclei containing various amount of chromatin. (2) Primordial follicles: oocytes surrounded by thin flattened pregranulosa cells. (3) Primordial follicles: oocytes surrounded by a single layer of squamous granulosa cells. (4) Primary follicles: oocyte surrounded by a single layer of cuboidal granulosa cells. (5) Secondary follicle: oocyte surrounded by multiple layers of granulosa cells. Inset: no periodic acid–Schiff (PAS)-positive material around the oocyte. (6) Secondary follicle (late stage): follicle contains some small fluid-filled spaces within the granulosa layers (arrows). Inset: small spaces are positive for PAS staining (Call–Exner bodies) and oocyte is surrounded by a thin PAS-positive layer (zona pellucida, PAS-hematoxylin stain). (7) Tertiary follicle: oocyte locates in the cumulus oophorus (arrow) and an antrum within granulosa layers is present. (8) Tertiary follicle (late stage): a large amount of faintly eosinophilic fluid accumulated within the antrum. Inset: oocyte surrounded by well-developed thick PAS-positive layer (zona pellucida) and a layer of granulosa cells (corona radiata, PAS-hematoxylin stain). If otherwise not specified, the staining represents hematoxylin and eosin staining.
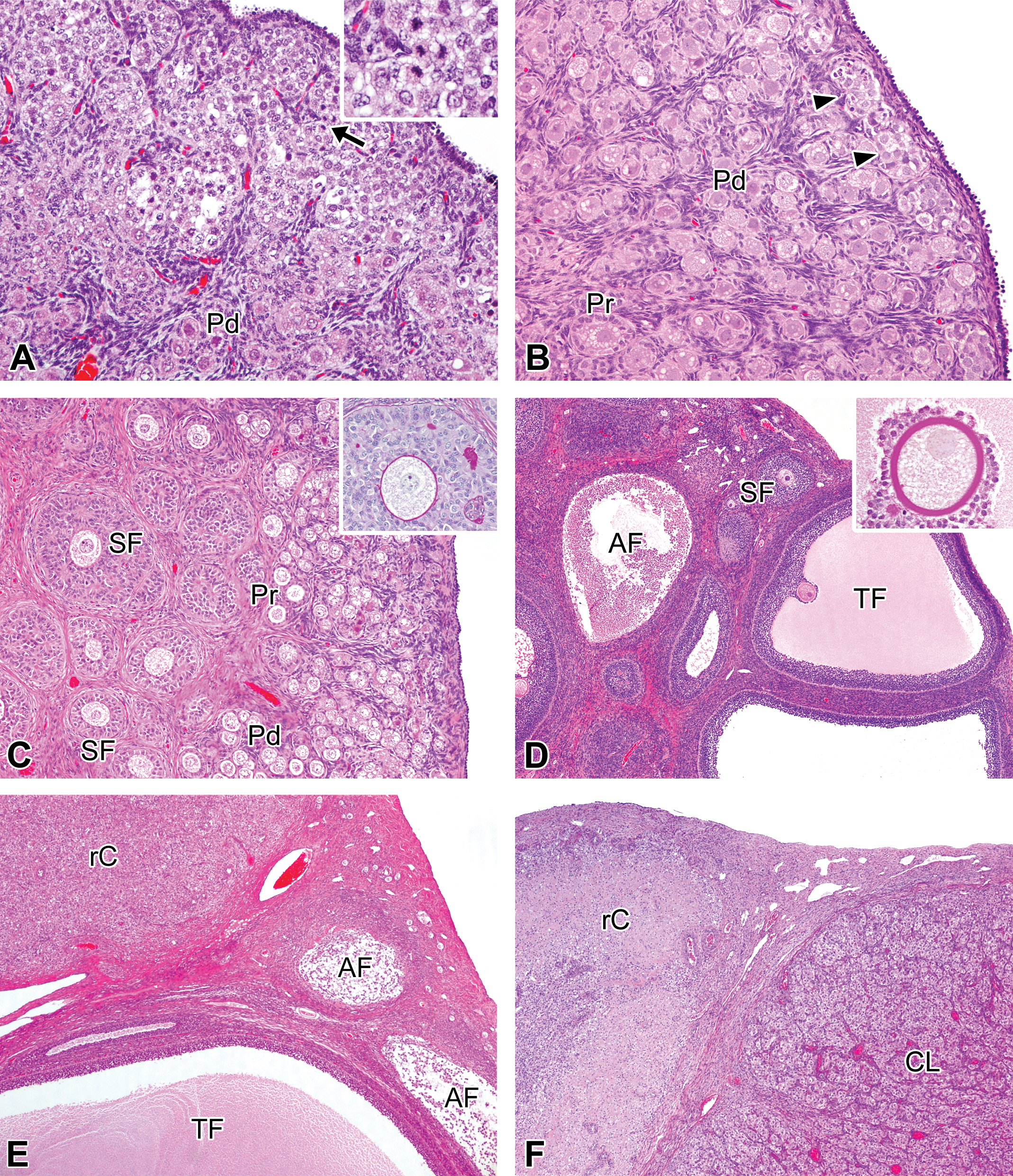
Histological features in the developmental ovaries in microminipigs. (A) At 0 days of age, ovarian cortex is composed mainly of germ cell nests. Primordial follicles are located adjacent to germ cell nests (lower left). The arrows indicate mitotic figures in oogonia (see also inset). (B) At 1 week of age, some germ cell nests are still present in subcapsular cortex (arrowheads). The cortex consists of a number of primordial follicles and a few primary follicles. (C) At 2.5 months of age, secondary follicles start to appear, containing periodic acid–Schiff (PAS)-positive materials within multiple layer of granulosa cells (Call–Exner body) and a thin undeveloped zona pellucida (inset, PAS-hematoxylin stain). A number of primordial and primary follicles are present in subcapsular cortex. (D) At 5 months of age, tertiary follicles with cumulous oophorus forming a stalk into the antral cavity and atretic follicles containing cellular debris are present. Inset: a well-developed zona pellucida showing strongly positive for PAS staining (PAS-hematoxylin stain). (E) At 6 months of age, a large tertiary follicle is located in the vicinity of a regressing corpus luteum and atretic follicles. (F) At 8 months of age, a well-developed corpus luteum is present adjacent to a regressing corpus luteum derived from the previous corpus luteum. In a female microminipig during the mid-luteal phase. If otherwise not specified, the staining represents hematoxylin and eosin staining. AF = atretic follicle; CL = corpus luteum; Pd = primordial follicle, Pr = primary follicle; rC = regressing corpus luteum; SF = secondary follicle; TF = tertiary follicle.
Oviducts
At 0 days and 1 week of age, the mucosal epithelia were lined with densely arranged columnar cells that were composed of secretory (nonciliated) and ciliated cells and formed mucosal folds of various heights, oriented toward the lumen. Most of the secretory cells located in the mucosal folds possessed microvilli on their luminal surface. Mitotic figures in epithelial cells were occasionally observed (Figure 4A). At 2.5 months of age, the mucosal folds became higher and more complex than those observed at younger ages. Secretory cells around the top of the mucosal folds displayed blebs on their luminal surface, whereas the cells in the basal compartment had relatively smooth luminal surfaces (Figure 4B). At 5 months of age, the blebs of the secretory cells became apparent throughout the mucosal folds, except for the basal compartment. The apoptotic figures started to appear at 5 months of age (Figure 4C). Their appearance at 6 months of age was quite similar to the one at 5 months of age. At 5 and 6 months of age, 3 females had well-developed or regressing corpora lutea in their ovaries. The structure of their mucosal epithelia was similar to the one in the ML or LL phase in mature females. At 8 months of age and thereafter, the mucosal folds had reached maximum development, and the appearance of the cells differed depending on the phase of the estrous cycle (Figure 4D). In contrast to the changes observed in secretory cells with age, the age-related morphological changes in ciliated cells were not obvious in any of the ages that we investigated.
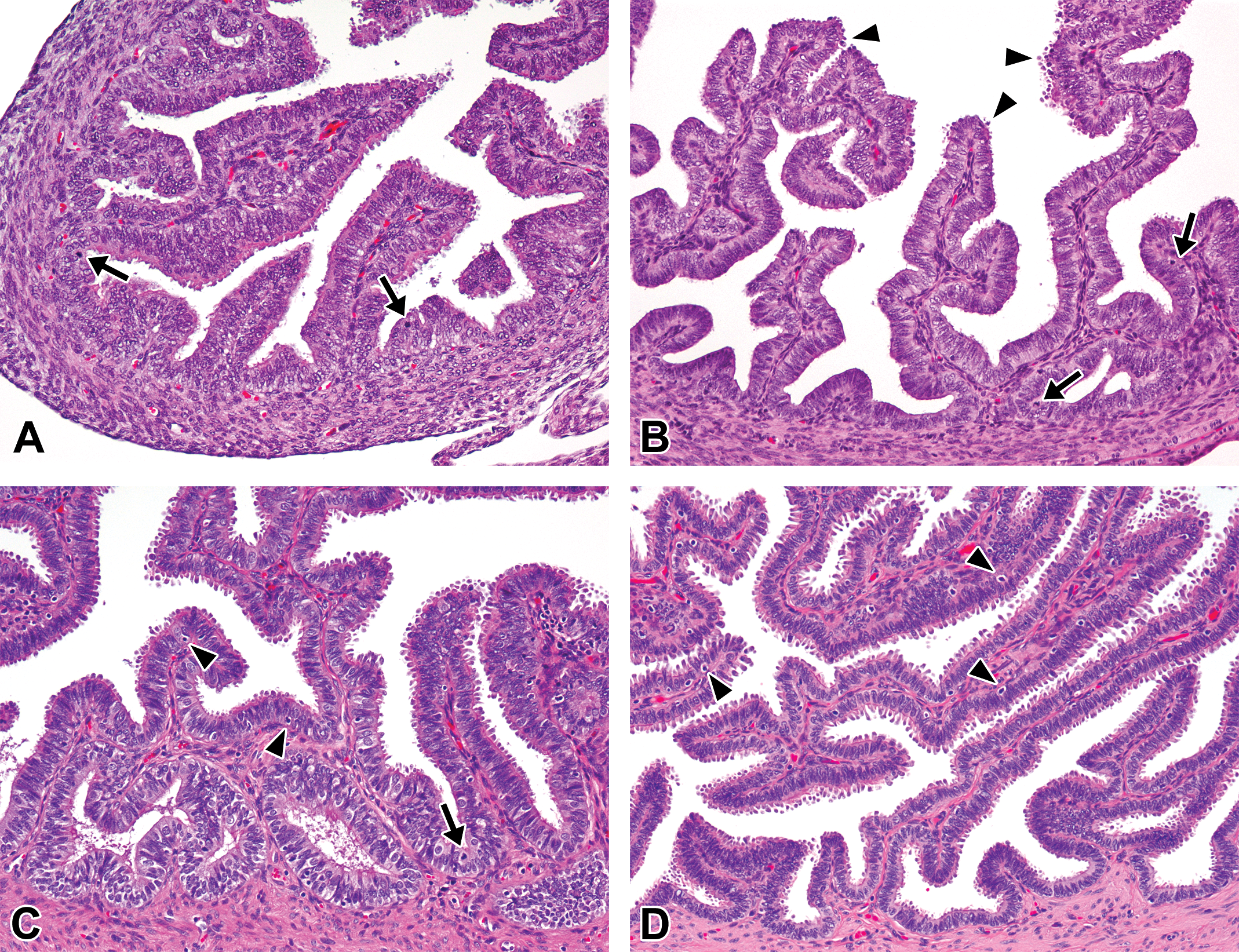
Histological development of the oviducts in microminipigs. (A) At 0 days of age, the mucosal epithelium is composed of densely arranged columnar secretory and ciliated cells, forming mucosal folds toward the lumen. The arrows indicate mitotic figures. (B) At 2.5 months of age, the taller and branched folds are present. Secretory cells around top of the mucosal folds possess small blebs on the surface (arrowheads). The arrows indicate mitotic figures. (C) At 5 months of age, the mucosal folds consist mainly of secretory cells with blebs on the surface. The basal part of the epithelium is highly columnar with clear to slightly eosinophilic cytoplasm. The arrows indicate mitoses and the arrowheads indicate apoptosis. (D) At 8 months of age, the fully developed mucosal folds, including basal parts, are composed of secretory cells possessing prominent blebs. The apoptotic figures are frequently present (arrowheads). In a female microminipig during the mid-luteal phase. Hematoxylin and eosin stain.
Uterus
At 0 days and 1 week of age, the mucosal surface was composed of pseudostratified columnar microvillous epithelial cells, and the lumen was round in shape. The uterine glands were not recognizable (Figure 5A). At 2.5 months of age, the uterine lumen became irregular in shape, and the uterine glands started to appear within the endometrium. The glandular epithelium was composed of cuboidal and columnar cells, but no secretory activity was observed. The glandular lumen became narrower as it went deeper in the endometrium (Figure 5B). At 5 to 6 months of age, there were frequent apoptotic bodies derived from mucosal epithelial cells, and a few eosinophilic granulocytes underneath the epithelium. The number of uterine glands increased in the endometrium. Within the uterine glands, the nuclei were located at the base of the cells, and occasional mitotic figures were observed (Figure 5C). In 3 females at 5 and 6 months of age, which possessed well-developed or regressing corpora lutea in their ovaries, the features of the endometrium were identical to those in the ML or LL phases in mature females. At 8 months of age and beyond, the luminal epithelium formed apparent mucosal folds, and the endometrium was maximally developed. The appearance of these tissues depended on the phase of the estrous cycle (Figure 5D).
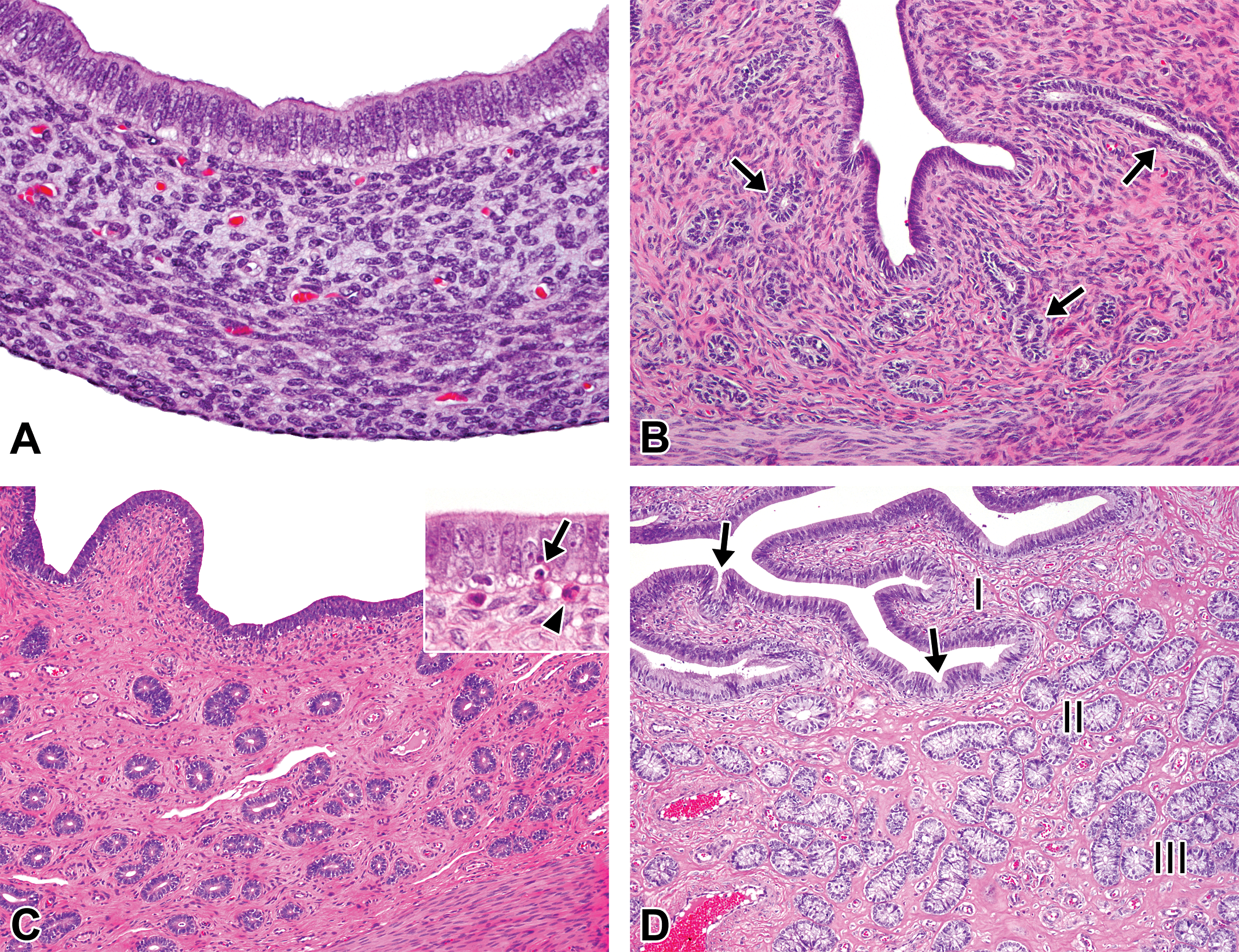
Histological development of the uterus in microminipigs. (A) At 0 days of age, the mucosal surface is lined by pseudostratified columnar microvillous epithelial cells. The stroma consists of densely arranged spindle cells. The uterine glands are not present. (B) At 2.5 months of age, the small immature uterine glands start to appear within collagenous tissue in endometrium (arrows). (C) At 5 months of age, the endometrium is composed of a number of uterine glands and a larger amount of collagenous component. Inset: the luminal epithelium contains frequent apoptotic bodies (arrow), and a few eosinophilic granulocytes (arrowhead) are present below the epithelium. (D) At 8 months of age, the fully developed luminal and glandular epithelium consist of highly columnar epithelial cells. The mucosal folds and a few epithelial invaginations (arrows) are present. The roman numbers indicate three functional zones in endometrium. In a female microminipig during the mid-luteal phase. I = zone I, II = zone II, and III = zone III. Hematoxylin and eosin stain.
Cervix
At 0 days and 1 week of age, the mucosal surface was composed of 2 types of epithelial cells. The luminal part of the surface comprised multiple layers of stratified squamous epithelial cells. In contrast, the invaginated part was composed of pseudostratified columnar microvillous epithelial cells (Figure 6A). At 2.5 months of age, the mucosal folds started to appear, while the cellular composition was similar to those of the younger ages (Figure 6B). At 5 to 6 months of age, the transitional area between the endocervix and the ectocervix was easily recognizable, and it contained nodular squamous cells beneath or within the epithelium (Figure 6C). The endocervix was composed of columnar microvillous epithelial cells, whereas the ectocervix was composed of stratified squamous cells. At 8 months of age and beyond, the epithelium formed well-developed mucosal folds. The appearance of these folds differed slightly between the different phases of the estrous cycle (Figure 6D).
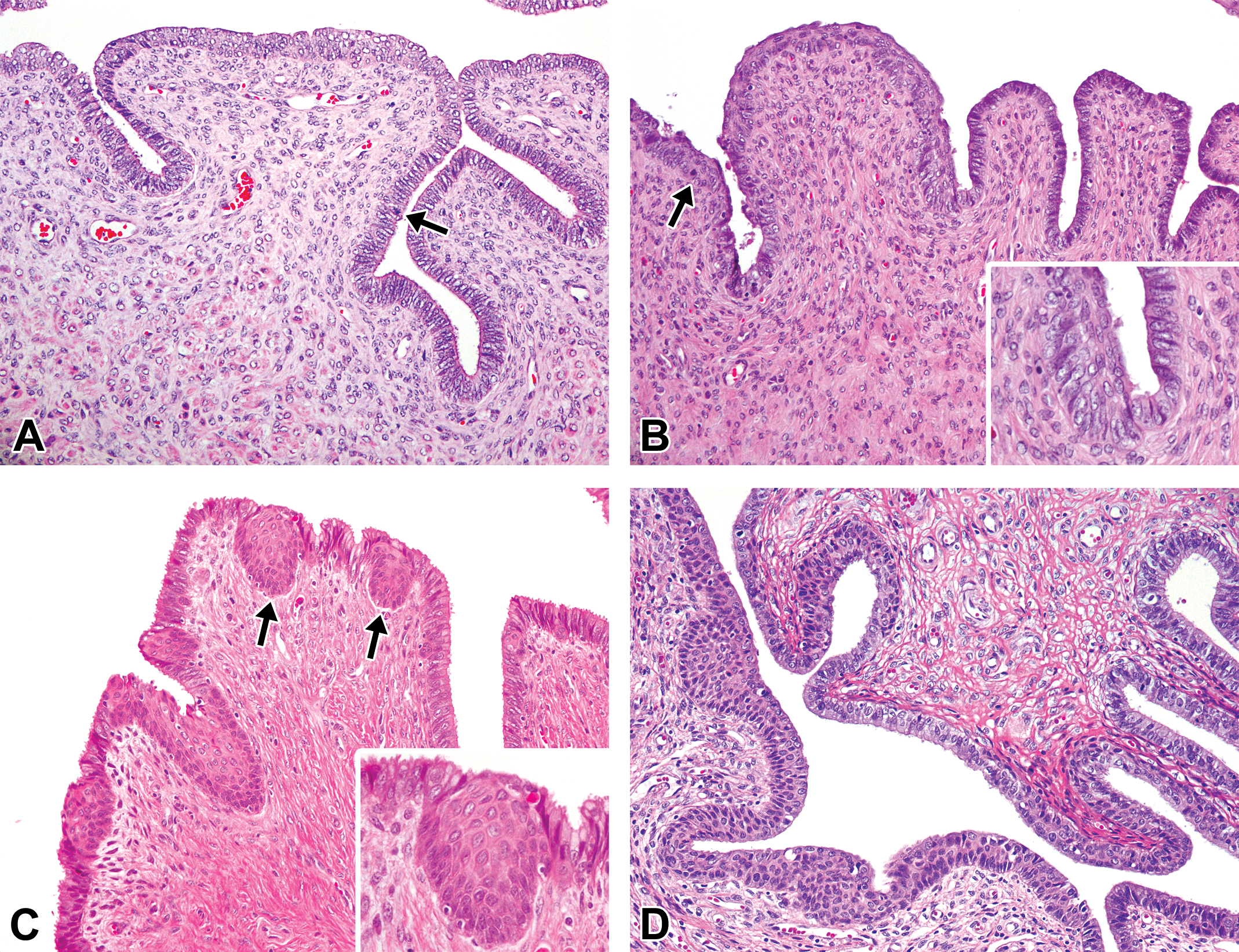
Histological development of the cervix in microminipigs. (A) Mucosal surface at 0 days of age composed of 3 to 4 layers of stratified squamous epithelial cells at the luminal part and pseudostratified columnar epithelial cells at the invaginated part. The arrow indicates a mitotic figure. (B) Mucosal folds starting to appear at 2.5 months of age. Inset: higher magnification of the transitional area of the epithelia. Multiple layers of the columnar and stratified cells are present. (C) The transitional area between the endocervix and ectocervix at 5 months of age. Nodular squamous cells beneath or within the mucosal epithelium (arrows). Inset: higher magnification of the squamous nodule. (D) The transitional area between the endocervix and the ectocervix at 8 months of age, with well-developed mucosal folds of various thicknesses. In a female microminipig during the early-luteal phase. Hematoxylin and eosin staining.
Vagina
At 0 days and 1 week of age, the mucosal surface was composed of 2 types of epithelia. The flattened part of the surface comprised 3 to 4 layers of stratified squamous epithelial cells, though these cells were somewhat irregularly arranged. In contrast, the invaginated part was composed of pseudostratified columnar microvillous epithelial cells (Figure 7A). At 2.5 months of age, the extent of the epithelial invagination became shallower, and the height of the columnar epithelial cells was reduced (Figure 7B). Moreover, a few eosinophilic granulocytes started to appear. At 5 to 6 months of age, the mucosal surface was composed of 3 to 4 layers of stratified squamous epithelial cells that were regularly arranged (Figure 7C). At 8 months of age and beyond, the epithelium became thicker and was composed of more than 5 layers of stratified squamous epithelial cells. However, there were differences in the epithelial height, the number and appearance of the epithelial layers, and the extent of cellular infiltrates depending on the phases of the estrous cycle (Figure 7D).
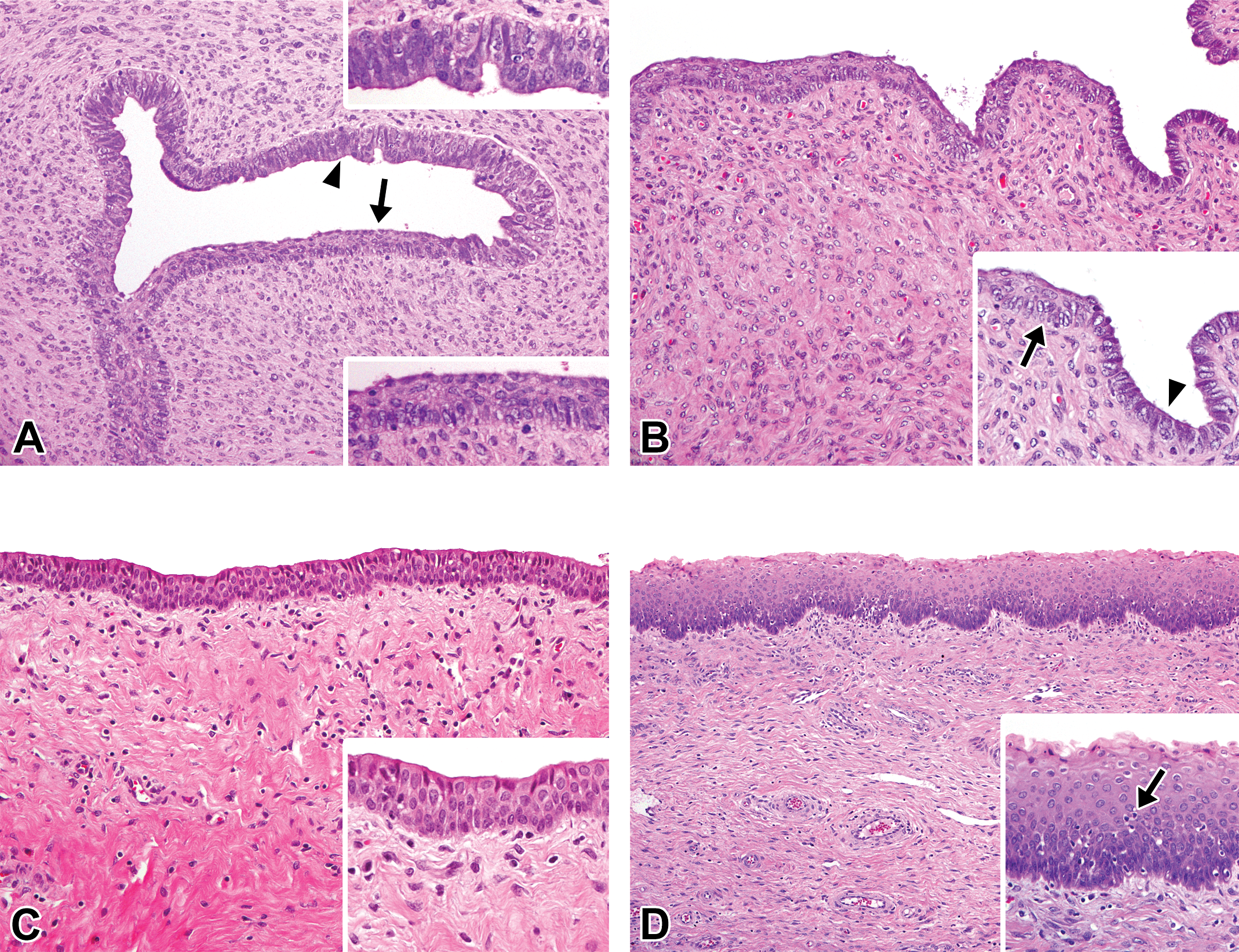
Histological development of the vagina in microminipigs. (A) At 0 days of age, the mucosal surface is composed of stratified squamous epithelium (arrow) and pseudostratified columnar microvillous epithelium (arrowhead). A number of stromal cells are present in the lamina propria. Inset (lower right): higher magnification of the area indicated by the arrow. Inset (upper right): higher magnification of the area indicated by the arrowhead. (B) At 2.5 months of age, the mucosal surface is still composed of stratified squamous epithelium (inset, arrow) and pseudostratified columnar microvillous epithelium (inset, arrowhead). The height of columnar epithelium became lower. (C) At 5 months of age, the mucosal surface consists of 3 to 4 layers of stratified squamous epithelial cells. The lamina propria is comprised of a large amount of collagenous component. Inset: higher magnification of the mucosal epithelium. (D) At 8 months of age, the mucosal epithelium is composed of 7 to 9 layers of stratified squamous epithelial cells. Inset: higher magnification of the mucosal epithelium. The apoptotic figure is present (arrow). In a female microminipig during the early-luteal phase. Hematoxylin and eosin stain.
Spontaneous Findings
The most frequently observed changes were the mesonephric duct remnants (epoophoron) near the oviduct and the mineralization that occurred in the ovary. A focal to multifocal nodular ductal structure was unilaterally or bilaterally observed adjacent to the oviduct in 13 of the 33 females investigated: 3 of the 4 animals at 0 days; 2 of 3 at 1 week of age; 3 of 4 at 2.5 months of age; 2 of 5 at 5 months of age; and 1 of 4 at 8, 12, and 26 months of age (Figure 8A and B). Mineralization was present in the ovaries in 6 of the 33 animals investigated: 1 of the 5 females at 5 months of age, 3 of 5 at 6 months of age, and 1 of 4 at both 8 and 12 months of age. All instances of mineralization were observed within atretic follicles and appeared to be deposited on remnants of oocytes (Figure 8C). Other findings are detailed below. Polyovular follicles were observed in 2 of the 4 animals at 2.5 months of age and in 1 of the 5 animals at both 5 and 6 months of age (Figure 8D). In the ovary, brown pigments were deposited within macrophages and/or interstitial cells in 1 of the 4 animals at 2.5 and 12 months of age (Figure 8E). In the uterus, a few brown pigments were also observed within the endometrium in 1 of the 4 females at 26 months of age. Although their incidence was low, solitary paraovarian cysts, which are known to originate from mesonephric duct remnants, were unilaterally present adjacent to the isthmus of the oviduct in 1 of the 5 animals at 5 months of age (Figure 8F).
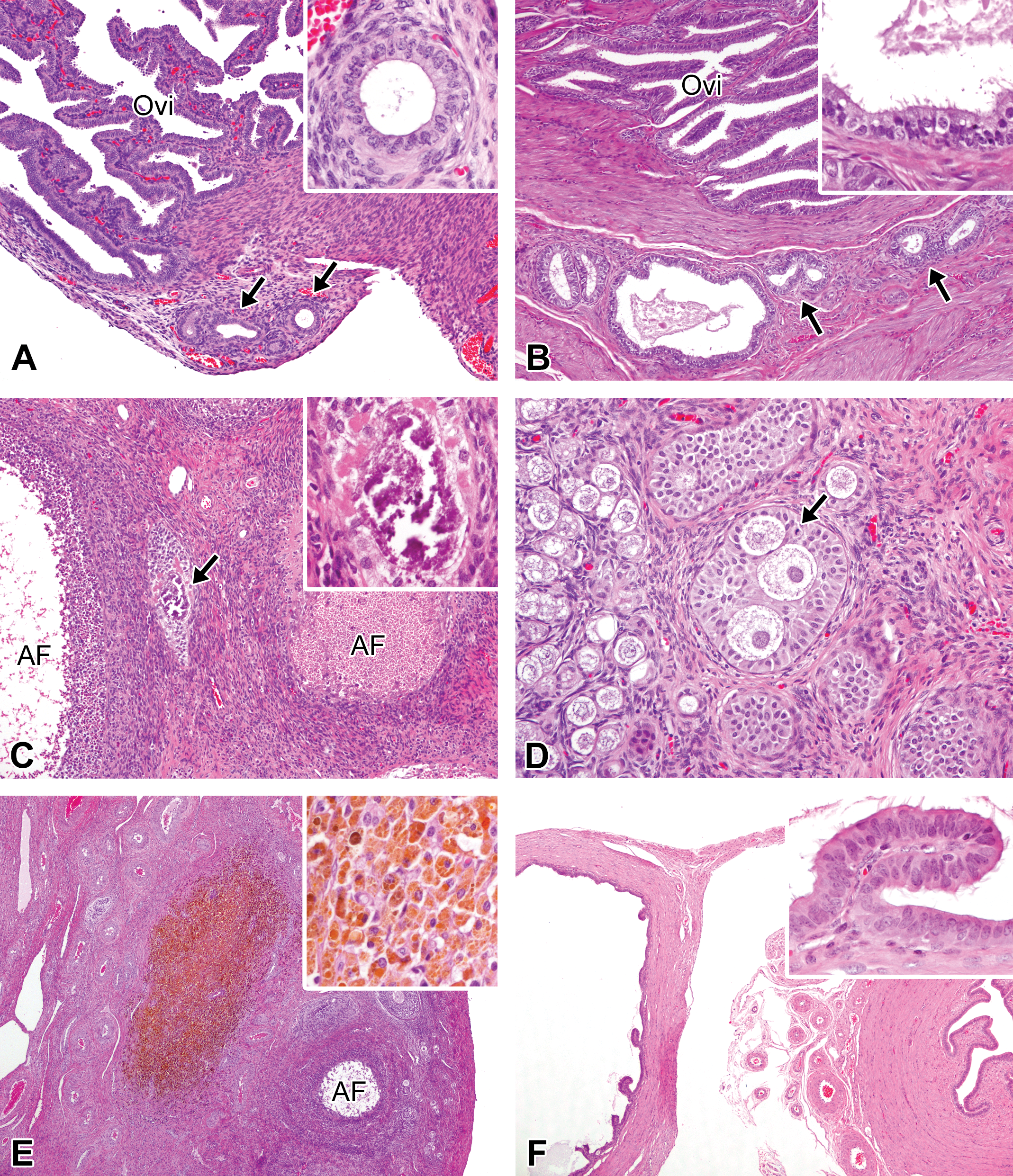
Spontaneous findings in female microminipigs. (A) Mesonephric duct remnants at 2.5 months of age. A few ductal structures are present adjacent to the oviduct. Inset: higher magnification of the duct. The lumen is lined by cuboidal to columnar epithelial cells. (B) Mesonephric duct remnants at 12 months of age. Multiple foci of ductal structure are located within the muscular layer of the oviduct (arrows). Inset: higher magnification of the duct that is comprised of pseudostratified ciliated and nonciliated columnar epithelial cells with some fluid in the lumen. (C) Mineralization in the ovary at 5 months of age. Mineral deposits are present between atretic follicles (arrow). Inset: higher magnification of the deposits. (D) Polyovular follicle in the ovary at 2.5 months of age. Three oocytes surrounded by multiple layers of granulosa cells are present. The arrow indicates mitosis in granulosa cell. Nests of normal primordial follicles are seen (left side). (E) Brown pigments in the ovary at 12 months of age. A focal area composed of brown pigments-laden macrophages is present. Inset: higher magnification of the ovarian macrophages containing brown pigments. (F) Paraovarian cysts at 5 months of age. A cyst (left) is present adjacent to the isthmus of the oviduct (right). Inset: higher magnification of the papillary projection of the cyst. The cyst is lined by pseudostratified ciliated and nonciliated columnar epithelial cells. Hematoxylin and eosin staining. AF = atretic follicle; Ovi = oviduct.
Estrous Cycle–dependent Histological Morphology
In the 12 animals that were investigated, the mean length of the estrous cycle was 20.6 ± 1.0 days, while the duration of standing heat was 1.6 ± 0.7 days. The cyclical features that were commonly observed at each phase of the cycle are presented in Figures 9 to 13. The histological changes observed in the ovaries and in the uterus were identical to those described in a previous report in Göttingen minipigs (de Rijk et al. 2014), and the oviducts and vagina were those described in the domestic pigs (Steinbach and Smidt 1970; Wu, Carlson, and First 1976; Zagórowski, Wrobel, and Schilling 1976; Walter and Bavdek 1997). The detailed histological features in these organs were described in the Online Supplemental Files. In contrast to the ovaries, oviducts, uterus, and vagina, the cervix displayed slight changes during the different phases of the estrous cycle. The height of the mucosal epithelium in the endocervix was higher in the F/P to EL phases than in the ML to LL phases. However, epithelial cells in all estrous cycle phases contained PAS-positive material, located on the luminal side of the cytoplasm (Figure 14). In the ectocervix, there were few discernible changes during the estrous cycle. One consistent feature was the presence of a few sloughed epithelial cells and of cellular debris along the surface of the mucosal epithelium during ovulation and the EL phases. These features were similar to those detected in the vaginal epithelium during ovulation and EL phases.
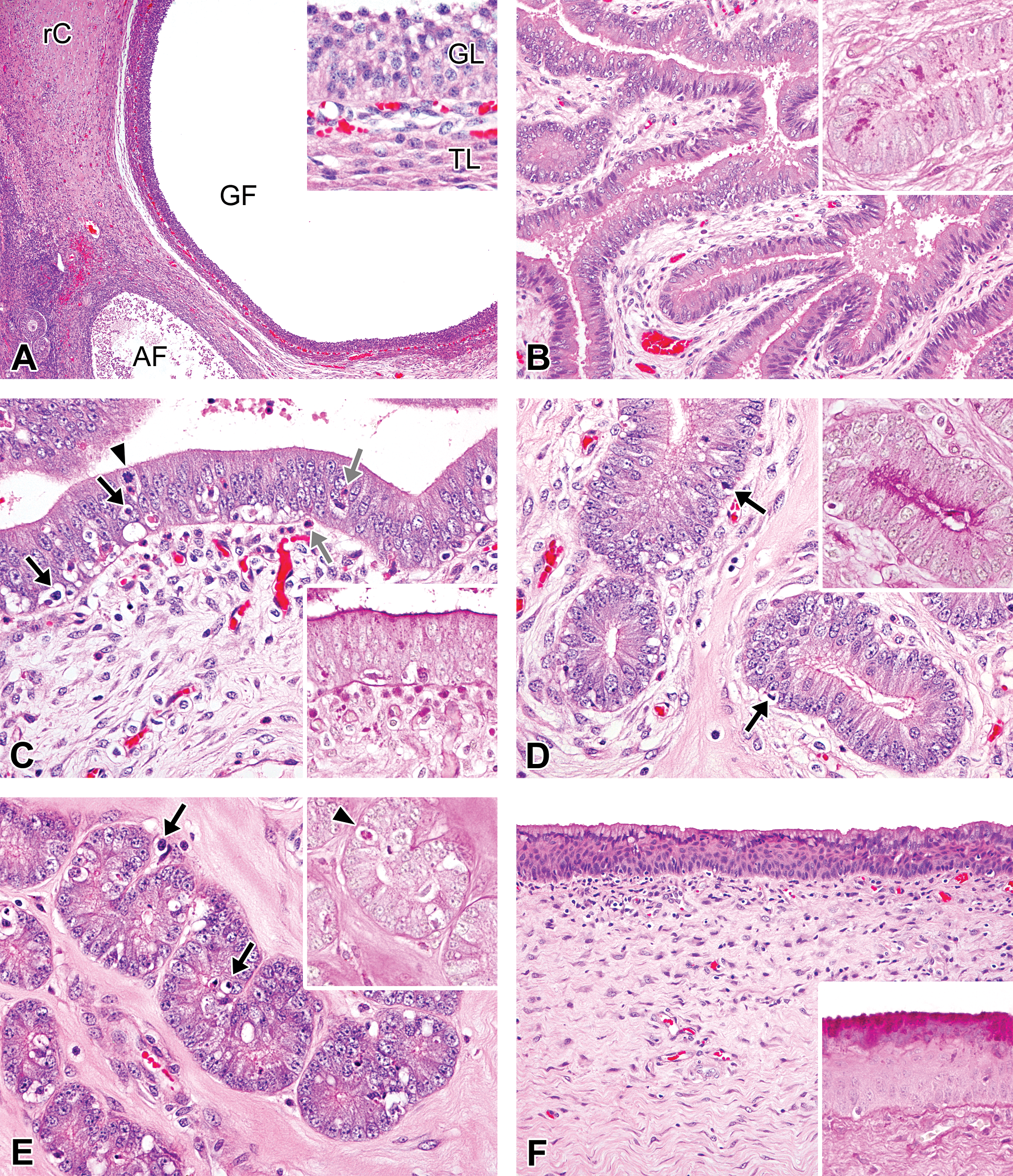
Photomicrographs of the ovaries, oviducts, uterus, and vagina in the follicular/proliferation phase in microminipigs. (A) Ovary: a Graafian follicle adjacent to a regressing corpus luteum and an atretic follicle is present. Inset: the follicle is lined by multiple layers of granulosa cells and surrounded by a theca cell layer. (B) Oviduct: the mucosal epithelium, supported by edematous stroma, is composed mainly of highly columnar pseudostratified secretory cells, and eosinophilic secretion is present in the lumen. Inset: periodic acid–Schiff (PAS)-positive materials in the epithelial cells. (C) Luminal epithelium of uterus: the pseudostratified epithelium contains a number of apoptotic bodies (black arrows) and a few mitotic cells (arrowhead). Eosinophilic granulocytes are also present within and underneath the epithelium (gray arrows). Inset: the surface of the luminal epithelium is positive for PAS staining (PAS-hematoxylin stain). (D) Uterine glands in zone II: the glands are lined by highly columnar pseudostratified cells with weakly eosinophilic cytoplasm. The arrows indicate apoptosis of the epithelial cells. Inset: the surface of the glandular epithelium is positive for PAS staining (PAS-hematoxylin stain). (E) Uterine glands in zone III: the glands are lined by columnar pseudostratified cells with eosinophilic cytoplasm. The arrows indicate apoptosis of the epithelial cells. Inset: PAS staining shows positive for an apoptotic cell (arrowhead) but not for glandular epithelium. (F) Vagina: stratum mucification is apparent in the superficial layer of the mucosal epithelium. Inset: the superficial mucoid cells are strongly positive for PAS staining (PAS-hematoxylin stain). If otherwise not specified, the staining represents hematoxylin and eosin staining. AF = atretic follicle; GF = Graafian follicle; GL = granulosa cell layer; rC = regressing corpus luteum; TL = theca cell layer.
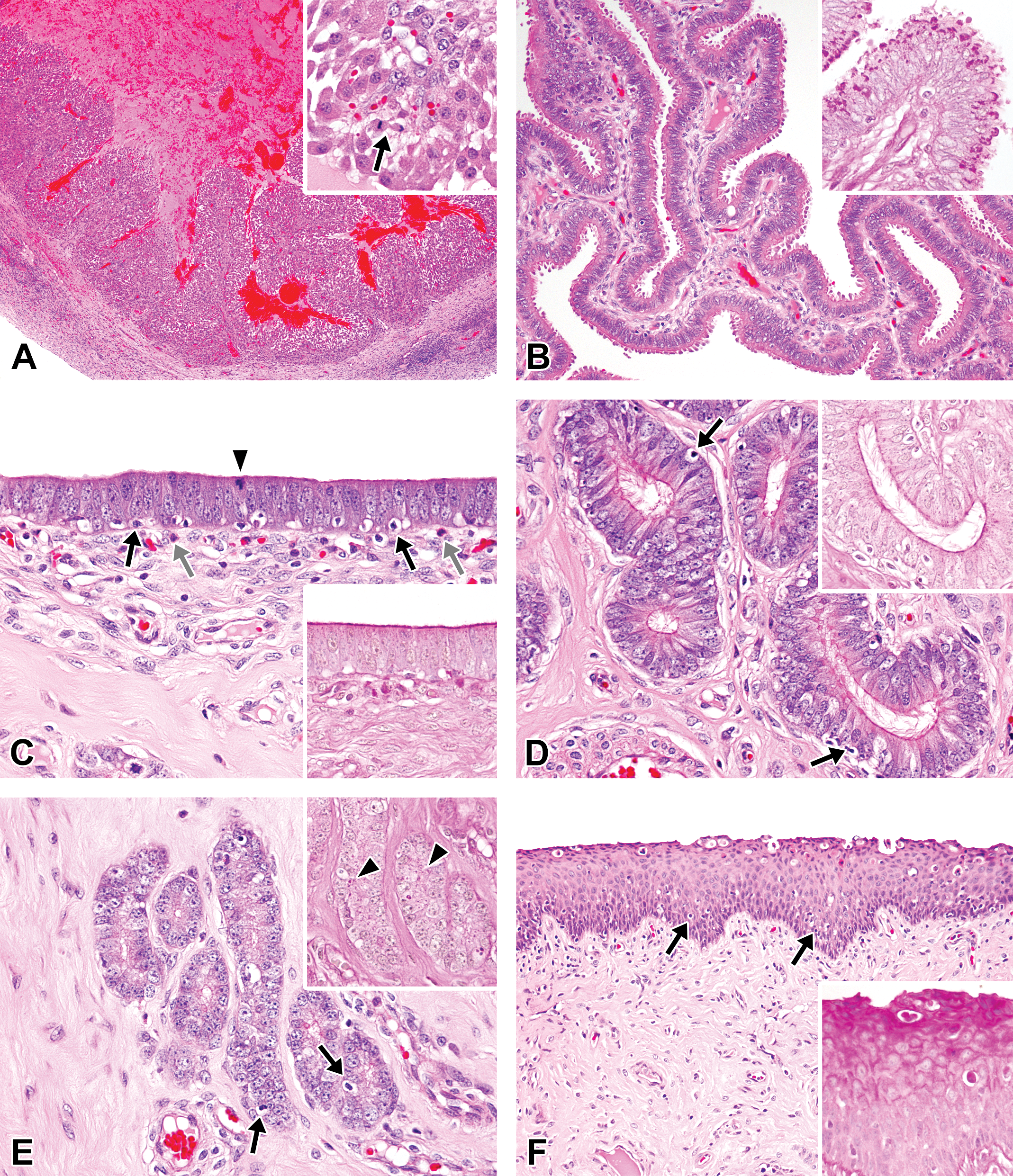
Photomicrographs of the ovaries, oviducts, uterus, and vagina in the ovulation phase in microminipigs. (A) Ovary: a newly forming corpus luteum contains a large amount of blood inside the lumen. Inset: higher magnification of the newly formed luteal cells, which are spindle to polygonal shape. The arrow indicates a mitotic figure. (B) Oviduct: the mucosal epithelium consists of ciliated epithelial cells and secretory cells with small blebs on the surface. Inset: the surface of the blebs on the epithelial cells is positive for periodic acid–Schiff (PAS) staining (PAS-hematoxylin stain). (C) Luminal epithelium of uterus: numerous apoptotic figures (black arrows) and occasional mitotic figures (arrowhead) are present within the pseudostratified epithelium. Eosinophilic granulocytes are also present underneath the luminal epithelium (gray arrows). Inset: the surface of the luminal epithelium is positive for PAS staining (PAS-hematoxylin stain). (D) Uterine glands in zone II: pseudostratified columnar epithelial cells contain weakly eosinophilic cytoplasm and apoptotic bodies are occasionally present (arrows). Inset: the surface of the luminal epithelium is weakly positive for PAS staining (PAS-hematoxylin stain). (E) Uterine glands in zone III: columnar epithelial cells possess eosinophilic fine granular cytoplasm. Apoptotic figures are present (arrows). Inset: a few tiny PAS-positive granules in the cytoplasm (arrowheads, PAS-hematoxylin stain). (F) Vagina: cellular debris is present along the surface of the epithelium. The arrows indicate apoptotic figures. Inset: the superficial layer is positive for PAS staining (PAS-hematoxylin stain). If otherwise not specified, the staining represents hematoxylin and eosin staining.
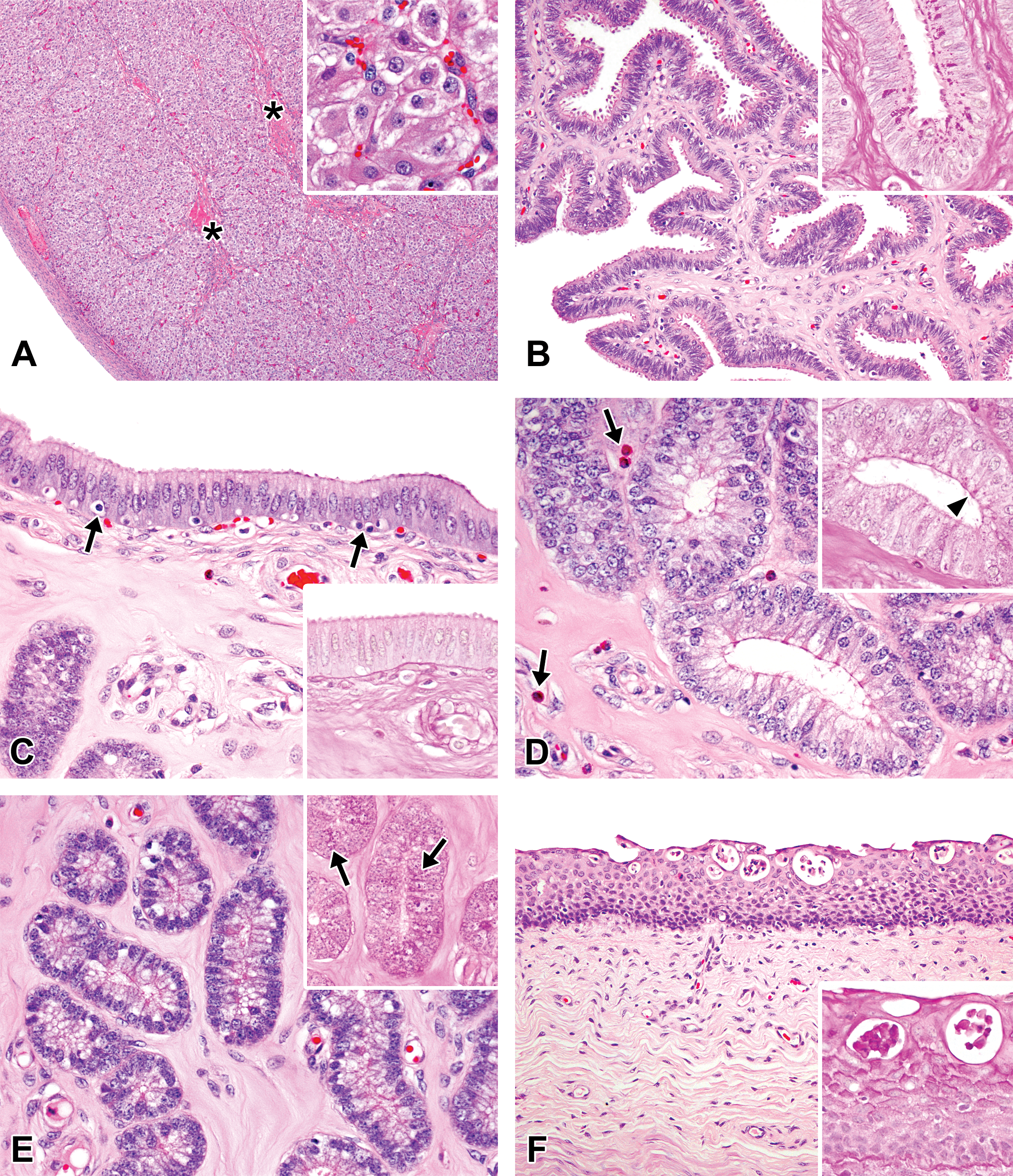
Photomicrographs of the ovaries, oviducts, uterus, and vagina in the early-luteal phase in microminipigs. (A) Ovary: corpus luteum consists of densely arranged proliferated luteal cells. A few small cavities are still present (asterisks). Inset: luteal cells are polygonal to round in shape, containing eosinophilic cytoplasm with peripheral foamy vacuoles. (B) Oviduct: the mucosal epithelium is composed mostly of secretory cells. Blebs on the surface become prominent (upper). Inset: periodic acid–Schiff (PAS)-positive granular materials within the epithelial cells at the bottom of mucosal folds (PAS-hematoxylin stain). (C) Luminal epithelium of uterus: the mucosal surface is lined by highly columnar epithelial cells with centrally arranged nuclei. Apoptotic figures are present (arrows). Inset: the luminal surface is no longer positive for PAS staining (PAS-hematoxylin stain). (D) Uterine glands in zone II: the glandular epithelium consists of highly columnar cells with clear to weakly eosinophilic cytoplasm. Some endometrial lymphocytes are present within the endometrial stroma (arrows). Inset: the luminal surface is no longer positive for PAS staining. A few PAS-positive granules within the cytoplasm become present, instead (arrowhead, PAS-hematoxylin stain). (E) Uterine glands in zone III: the glandular epithelium consists of columnar cells with clear cytoplasm. The nuclei are arranged in the basal portion of the cells. Inset: numerous PAS-positive granules in the cytoplasm (arrows, PAS-hematoxylin stain). (F) Vagina: a number of intraepithelial microabscesses are present near the surface. Inset: cellular debris is positive for PAS staining (PAS-hematoxylin stain). If otherwise not specified, the staining represents hematoxylin and eosin staining.
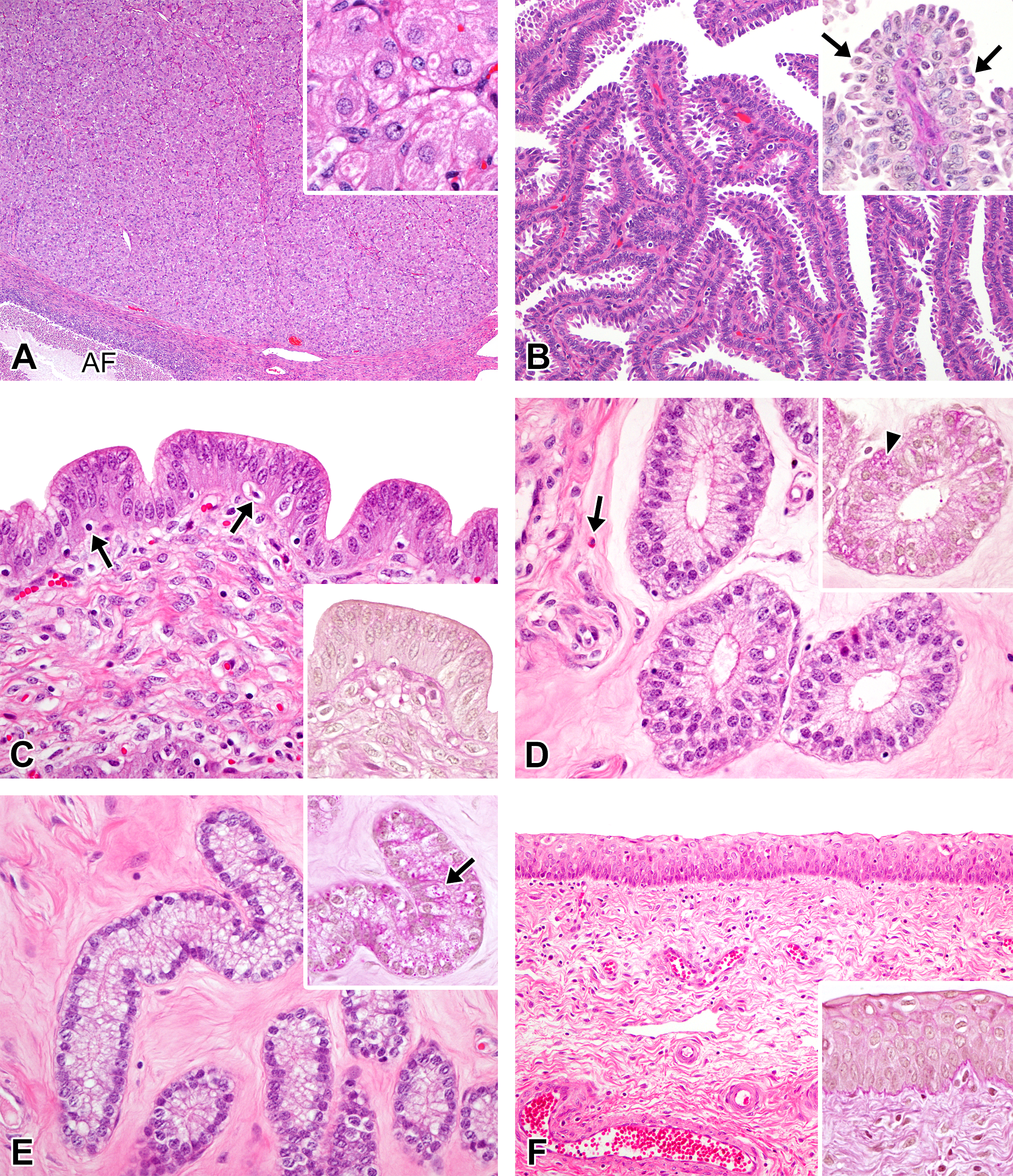
Photomicrographs of the ovaries, oviducts, uterus, and vagina in the mid-luteal phase in microminipigs. (A) Ovary: a fully mature corpus luteum is composed of densely arranged luteal cells. Inset: luteal cells contain abundant eosinophilic cytoplasm as well as a few vacuoles with eosinophilic core in center. (B) Oviduct: most of the mucosal epithelial cells have prominent blebs over the surface. Inset: higher magnification of the mucosal epithelium stained with periodic acid–Schiff (PAS). PAS-positive granules are no longer observed. The nuclei are often present within the blebs (arrows). (C) Luminal epithelium of uterus: the mucosal epithelium is lined by highly columnar epithelial cells and mucosal invagination is prominent. Nucleus are located in the center to apical of the cells. The arrows indicate apoptotic figures. Inset: the luminal surface is negative for PAS staining (PAS-hematoxylin stain). (D) Uterine glands in zone II: the glandular epithelium is in maximum height and composed of highly columnar cells with clear cytoplasm. The arrow indicates an endometrial lymphocyte. Inset: A number of PAS-positive fine granules are present in the glandular cells (arrowhead, PAS-hematoxylin stain). (E) Uterine glands in zone III: the glandular epithelium is in maximum height consisting of columnar cells with clear cytoplasm. Inset: a number of PAS-positive fine granules in the cytoplasm (arrow, PAS-hematoxylin stain). (F) Vagina: the mucosal epithelium is in regenerating state composed of 5 to 6 layers of epithelial cells. Inset: PAS positivity is no longer present (PAS-hematoxylin stain). If otherwise not specified, the staining represents hematoxylin and eosin staining. AF = atretic follicle.
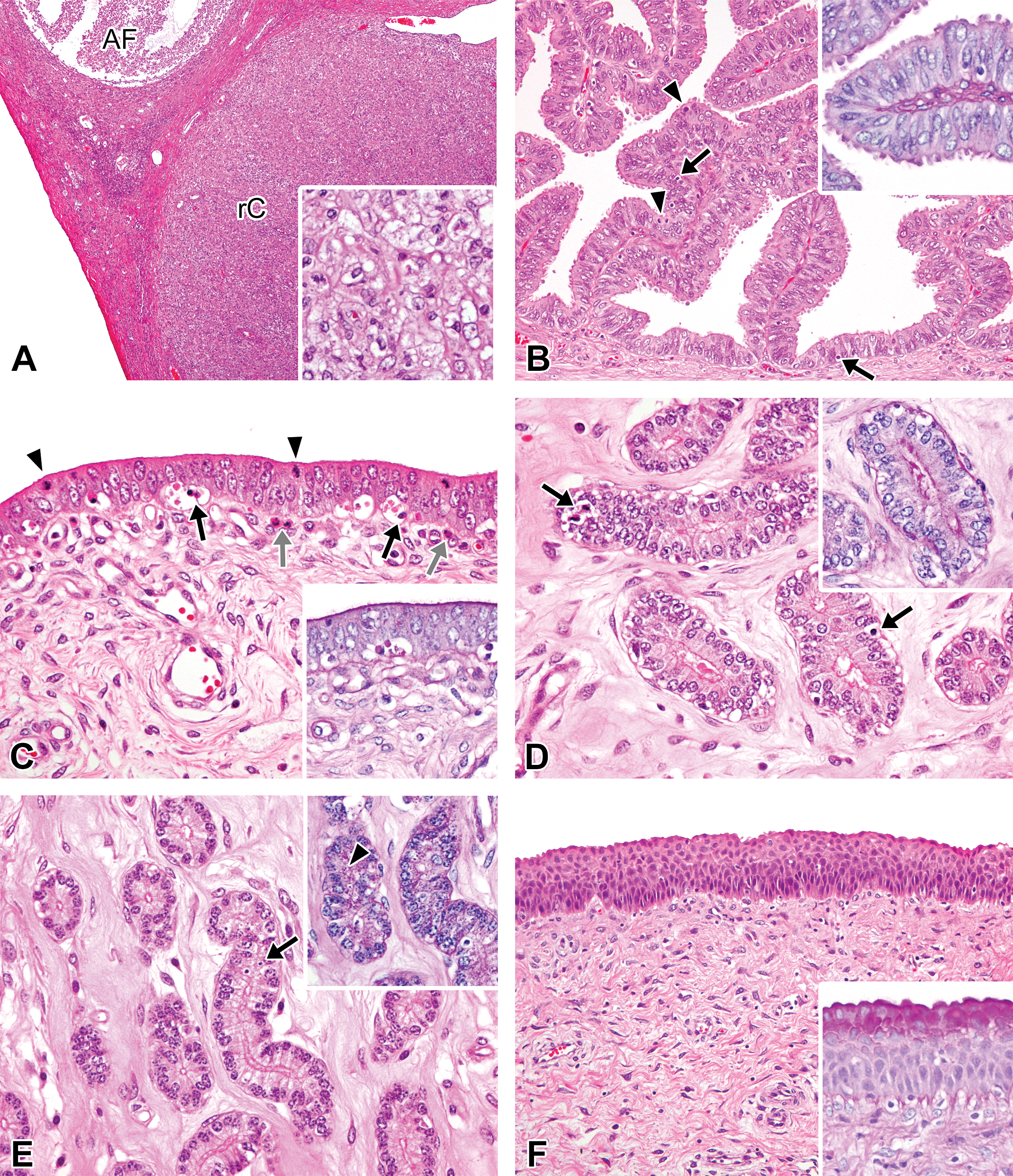
Photomicrographs of the ovaries, oviducts, uterus, and vagina in the late-luteal phase in microminipigs. (A) Ovary: regressing corpus lutea and an atretic follicle are present. Inset: the cytoplasm of the degenerating luteal cells becomes less eosinophilic with cytoplasmic vacuolations. (B) Oviduct: the mucosal epithelium consists of highly columnar epithelial cells. Blebs on the surface are much less prominent. Numerous apoptotic (arrows) and mitotic figures (arrowheads) are present. Inset: the luminal surface is faintly positive for periodic acid–Schiff (PAS) staining (PAS-hematoxylin stain). (C) Luminal epithelium of uterus: the pseudostratified epithelium contains numerous apoptotic bodies (black arrows) and mitotic figures (arrowheads). Eosinophilic granulocytes are present underneath the mucosal epithelium (gray arrows). Inset: the luminal surface is positive for PAS staining (PAS-hematoxylin stain). (D) Uterine glands in zone II: the glands are lined by columnar cells with weakly eosinophilic cytoplasm. The arrows indicate apoptotic bodies (arrows). Inset: the surface of the glandular epithelium becomes positive for PAS staining (PAS-hematoxylin stain). (E) Uterine glands in zone III: the glandular epithelium consists of clear to weakly eosinophilic columnar cells. Apoptotic figures are present (arrow). Inset: a number of PAS-positive granules in the glandular cells (arrowhead, PAS-hematoxylin stain). (F) Vagina: the mucosa is composed of 6 to 8 layers of epithelial cells. Inset: the mucosal surface layers become positive for PAS staining (PAS-hematoxylin stain). If otherwise not specified, the staining represents hematoxylin and eosin staining. AF = atretic follicle; rC = regressing corpus luteum.
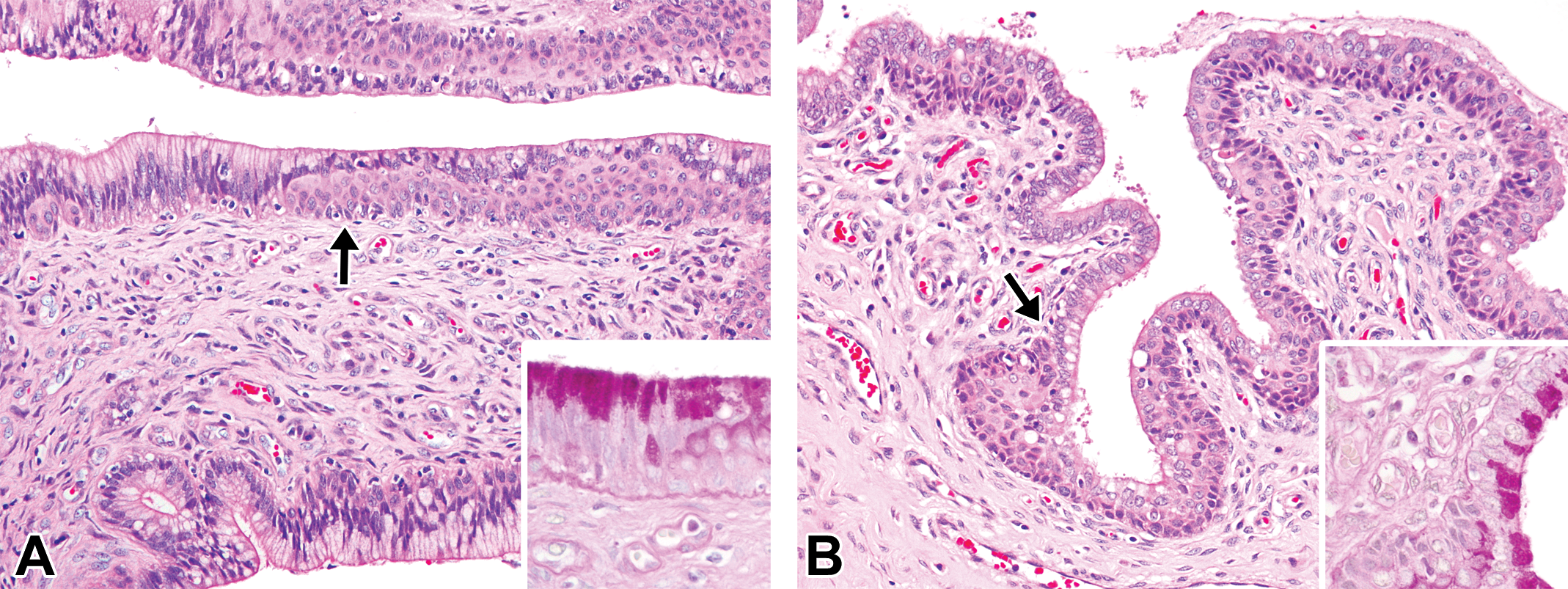
Photomicrographs of the cervix in the different phases of the estrous cycle in microminipigs. (A) Transition area between the endocervix and the ectocervix in the follicular/proliferation phase, consisting of highly columnar epithelial cells with abundant clear cytoplasm and a layer of basal squamous cells covered by cuboidal to columnar cells. Inset: a higher magnification of the area indicated by the arrow. Highly columnar cells contain large amounts of periodic acid–Schiff (PAS)-positive material. PAS-hematoxylin staining. (B) Transition area between the endocervix and the ectocervix in the mid-luteal phase, consisting of columnar surface epithelium and a basal layer of squamous cells. Inset: a higher magnification of the area indicated by the arrow. Columnar epithelial cells containing PAS-positive material. PAS-hematoxylin staining. If otherwise not specified, the staining represents hematoxylin and eosin staining.
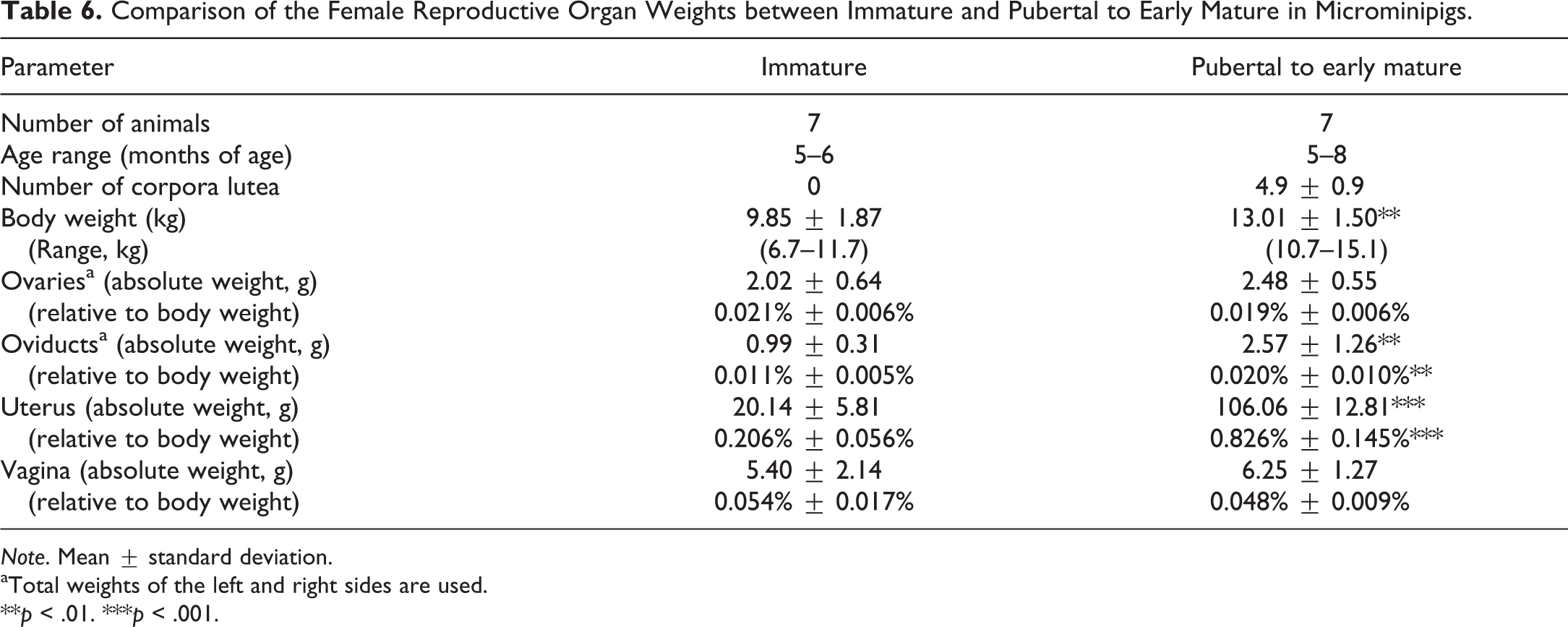
Comparison of the Female Reproductive Organ Weights between Immature and Pubertal to Early Mature Microminipigs
As shown in Table 6, there were significant differences in the mean body weight (
Comparison of the Female Reproductive Organ Weights between Immature and Pubertal to Early Mature in Microminipigs.
aTotal weights of the left and right sides are used.
**
Discussion
The present histological study is the first to describe the developmental and estrous cycle–dependent changes in the female reproductive organs of microminipigs aged 0 to 26 months. Our comprehensive histological investigations have set the stage for defining sexual maturity and the incidence of spontaneous pathological changes and revealed the practical criteria for determining each phase of the estrous cycle in the reproductive organs of female microminipigs.
The most frequently used histological features for confirming sexual maturity in females of all species are the presence of corpora lutea and the evidence of cyclicity (Halpern et al. 2016). The developmental states of sexual maturity are commonly categorized into 3 stages: immature, pubertal, and mature. The key histological features of each stage are as follows: immature, no evidence of ovulation and no onset of cyclicity; pubertal, evidence of ovulation and/or no onset of cyclicity; and mature, evidence of ovulation from multiple cycles and current cyclicity (Vidal 2017). The identification of hyalinized regressing corpora lutea reveals regular cyclicity in minipigs, because they are recognized as regressing corpora lutea derived from the cycle preceding the previous cycle (Tortereau, Howroyd, and Lorentsen 2013). In the present study, no animals showed any evidence of ovulation until 5 months of age, and the animals with the earliest onset of ovulation presented corpora lutea at 5 and 6 months of age. Then, all females possessed hyalinized regressing corpora lutea as well as corpora lutea from 8 months of age. Since the duration of the estrous cycle was estimated at approximately 21 days in microminipigs, the presence of hyalinized regressing corpora lutea at 8 months of age suggested that the first ovulation had occurred around 7 months of age at the latest. Of note, there were variations between individuals with regard to the onset of puberty, which were consistent with published data (Tortereau, Howroyd, and Lorentsen 2013; Peter et al. 2016). According to our observations, the stages of sexual maturity in female microminipigs were identified as the immature state under 5 months of age, the pubertal state from 5 to 7 months of age, and the mature state at 8 months of age and over.
Previous studies have indicated that the onset of puberty in pigs is affected by various environmental factors such as housing, management, diet, and the age of the first boar contact (Brooks and Cole 1970; Mavrogenis and Robison 1976; Kirkwood and Hughes 1979; Klindt, Yen, and Christenson 1999). In the present study, 1 of the 5 animals (20%) at 5 months of age and 2 of the 5 animals (40%) at 6 months of age showed the histological features of puberty, whereas the rest (70%) at 5 and 6 months of age were immature. Similar observations have been made in Göttingen minipigs: the onset of puberty was observed at 3.7 to 4.2 or 6.1 to 6.5 months of age when female minipigs were housed with a boar, and only 50% of the females showed evidence of puberty by 6.5 months of age when they were not housed with a boar (Tortereau, Howroyd, and Lorentsen 2013; Peter et al. 2016). All the females in the present study were bred, raised, housed, fed, and introduced to boars (a few minutes every day) under the same conditions, so that the differential developmental responses to sexual maturation in these microminipigs are caused only by simple individual variation. However, we speculated that females might mature faster if they had been housed in the same room with a boar throughout the day and night. Further studies are needed to confirm the age of onset for puberty and sexual maturity, considering the environmental effects on the development of female reproductive systems.
Age, body weight, and organ weights in both males and females, as well as cyclicity in females, are standard parameters required for the assessment of sexual maturity (Halpern et al. 2016). Precise data have been reported in Göttingen minipigs, for which increases in body and uterine weights were indicative of maturity (Tortereau, Howroyd, and Lorentsen 2013). In female microminipigs of approximately 5 to 8 months of age, there were statistically significant differences in body weights between immature and pubertal to mature animals, which were correlated with changes in oviduct and uterine weights. Thus, the body weight of female microminipigs can help to predict the stage of sexual maturity, even during daily management. Besides the obviously visible ovarian corpus luteum, the weights of oviducts and uteri can also provide a reliable indication of maturity at necropsy.
Mesonephric duct remnants, including paraovarian cysts, were frequently observed around oviducts in female microminipigs at various ages. In minipigs, few studies have reported the presence of remnants derived from embryonic elements. To our knowledge, a few cases of paraovarian cysts have been reported in minipigs (Ilha et al. 2010; Erneholm et al. 2016). Cystic structures may be encountered at multiple sites in the female reproductive organs, and such structures are believed to be congenital; thus, they are seldom recorded in toxicity studies (Dixon et al. 2014). Since their incidence seems to decrease with age in microminipigs, the importance of mesonephric duct remnants for toxicology studies may be minimal. Polyovular follicles may occur due to the abnormal oocyte separation during the formation of primordial follicles. These are found with low frequency in rodents and are commonly encountered in beagles and young macaques (Koering 1983; Cline et al. 2008; Sato et al. 2012; Dixon et al. 2014; Vidal 2017). However, to our knowledge, the incidence of polyovular follicles in minipigs is still undetermined. In domestic pigs, the number of polyovular follicles is higher in the ovaries of gilts than in the ones of sows (Stankiewicz, Blaszczyk, and Udala 2009). Since such follicles were not observed in females after 8 months of age, the presence of polyovular follicles might indicate an immature state of follicular development in microminipigs.
In addition, several spontaneous findings in the female reproductive organs of minipigs have been reported (Jeppesen and Skydsgaard 2015; Helke et al. 2016a, 2016b). The most common findings were the mineralization that occurred in the ovaries of Göttingen and Yucatan minipigs, while a similar lesion was also observed in microminipigs. Follicular and hemorrhagic cysts in the ovary and inflammatory cell infiltration in the vagina were reported in Göttingen minipigs, but no such lesions were observed in the present study. Because the number of examined animals was limited in this study, further investigation is necessary to determine the accurate incidence and the extent of spontaneous findings in female microminipigs.
In regulatory toxicity studies, it is important for toxicologic pathologists to evaluate the female reproductive tract tissues by taking into account normal cyclicity and to understand the morphologic features representative of each phase for each reproductive organ, so that deviations from normal can be detected (Dixon et al. 2014). These alterations can be recognized by histological examination, as a disturbance in the coordinated morphology of the components of the tract or as the abnormal appearance of a particular region (Westwood 2008). Table 7 shows the summary of the histological features of female reproductive organs during the normal estrous cycle in microminipigs. The cyclic changes within the reproductive organs are mainly regulated by the hormones, so that the hormonal perturbation will affect the morphologic appearance of the various portions of the reproductive tract (Halpern et al. 2016). The hormone profiles in mature microminipigs during the estrous cycle have been previously reported (Noguchi et al. 2015). The plasma progesterone concentration starts to rise at 2 to 3 days after the luteinizing hormone (LH) surge and reaches a plateau on day 6 after the LH surge. Then, progesterone levels begin to decrease 14 days after the LH surge and become the lowest at 16 days after the LH surge and thereafter (Noguchi et al. 2015). Considering that ovulation in microminipigs occurs within 48 hr after the LH peak and before the end of the standing heat (Noguchi et al. 2015), the F/P, ovulation, and LL phases in this study correspond to the stages with the lowest plasma progesterone levels, and the EL and ML phases correspond to the rising and plateau stages of plasma progesterone levels. Progesterone is produced by corpora lutea and has various functions during the estrous cycle, such as stimulating the secretion from the uterine glands and acting as an antagonistic agent against oviductal ciliogenesis and endometrial mitogenesis (Basha, Bazer, and Roberts 1979; Brenner et al. 1983). The cyclic features observed in the EL and ML phases in microminipigs, such as the highly columnar uterine glandular cells, the prevalence of nonciliated epithelial cells in the oviducts, and the reduced number of mitotic figures in the uterine luminal epithelium, appear to be well correlated with the low levels of the plasma progesterone concentration, as previously described. On the other hand, the plasma concentration of estradiol-17β starts to rise at 16 to 17 days after the LH surge, becoming the highest before the LH surge, and then decreasing to a low level at 4 days after the LH surge and thereafter (Noguchi et al. 2015). Estradiol-17β is produced by the granulosa cells in the large mature follicles (Guthrie, Bolt, and Cooper 1993), causes edema of the oviducts and endometrium, and increases the secretion of mucus (Keys and King 1988; Walter and Bavdek 1997). Besides the edema present in the oviducts and uterus, PAS-positive reactions were observed in the vaginal epithelium and on the surface of the epithelium in the oviducts and uterus in the LL, F/P, and ovulation phases. These appeared to be generated by estradiol-17β, because these features were correlated with the stage of high estradiol-17β concentration. Since the histological features in microminipigs during the estrous cycle are apparently shifting in response to the cyclic hormonal changes, the histological alterations observed in the present study may provide a clue for future toxicity studies investigating hormonal disturbances.
Summary of the Histological Features of Female Reproductive Organs during the Estrous Cycle in Microminipigs.
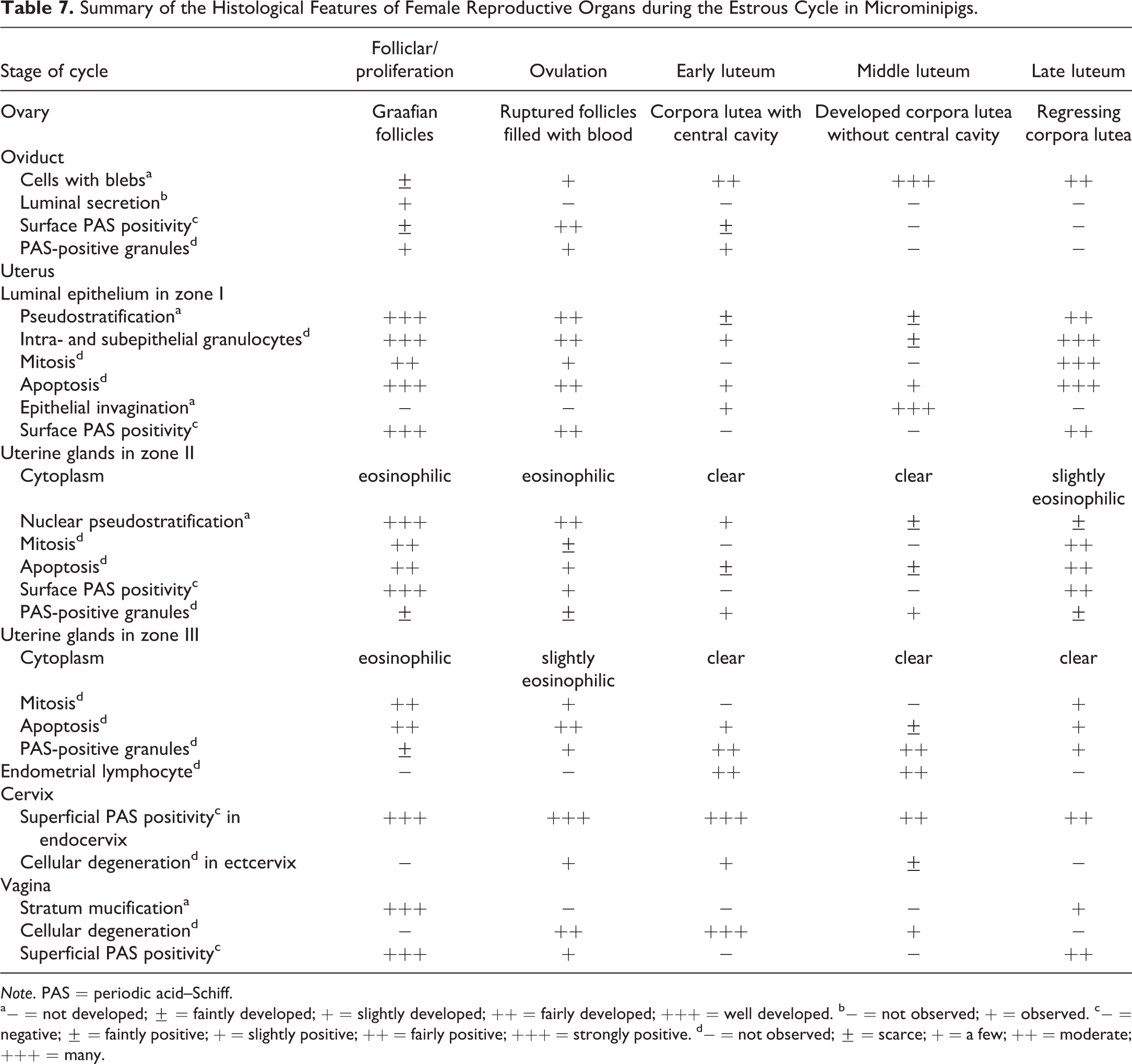
a− = not developed; ± = faintly developed; + = slightly developed; ++ = fairly developed; +++ = well developed. b− = not observed; + = observed. c− = negative; ± = faintly positive; + = slightly positive; ++ = fairly positive; +++ = strongly positive. d− = not observed; ± = scarce; + = a few; ++ = moderate; +++ = many.
The present histological evaluation revealed that female microminipigs reached puberty at approximately 5 months of age and sexual maturity at 8 months of age. The onset of puberty correlated well with the increase in body weight and was also indicated by a remarkable increase in the oviduct and uterine weights. The morphological changes occurring during the estrous cycle were similar to those reported in domestic pigs and minipigs. Moreover, in addition to the ovaries and the uterus, the oviducts and vagina in sexually mature microminipigs showed consistent cyclic changes after 8 months of age. Therefore, female microminipigs from 6 to 7 months of age were suitable for the assessment of reproductive organs, since they were sexually mature by the end of the 4-week and 13-week toxicology studies. In this study, we used several measurements such as the weights of the oviducts and vagina and PAS-H staining that is not routinely evaluated in toxicity studies (Sellers et al. 2007). However, these proved useful for the purposes of characterizing the features of female reproductive organs. We predict that our results will be valuable for future toxicity studies in microminipigs. Since a limited number of animals have been investigated so far, further studies of the morphological manifestations in response to toxic injury are necessary to fully understand the function of the reproductive organs in microminipigs.
Footnotes
Acknowledgments
The authors thank Mr. Toshiaki Fukumura (Shizuoka Prefectural Research Institute of Animal Industry, Swine and Poultry Research Center) for the care of the animals used in this study.
Author Contribution
Authors contributed to conception or design (AK); data acquisition, analysis, or interpretation (MO, SE, TY, MS); drafting the manuscript (AK); and critically revising the manuscript (MO, SE, TY, MS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
