Abstract
The peripheral nervous system (PNS) is subject to a wide range of structural and functional insults including direct damage to axons, loss of myelin, and progressive deficits in saltatory conduction. Drugs that damage the PNS often result in neuropathies that impact the structure and function of targeted nerves. In most cases, both sensory and motor neurons are affected with damage initially evident in the distal extremities. Drug-induced neuropathies are potentially reversible following cessation of treatment, but early stages of neuropathy can be subclinical and asymptomatic making diagnosis difficult. Nerve biopsy is highly validated and provides definitive evidence of nerve injury and corresponding severity; however, it is limited in some respects and electrophysiological measures can complement histopathological assessments and provide a functional measure of potential toxicity. In a drug development setting, nerve conduction assessments are valuable to monitor nerve function longitudinally if nerve damage is suspected or confirmed, and importantly, can be used to monitor progression and/or recovery of a drug-induced neuropathy. This review will summarize the methodology used in nerve conduction assessments as well as discuss data interpretation and considerations for use in nonclinical species. Finally, the use of nerve conduction assessments in nonclinical drug development is discussed.
Introduction
Peripheral neuropathy can be a debilitating condition with significant effects on daily function and quality of life due to motor and/or sensory deficits. Neuropathy can develop due to underlying disease or upon exposure to toxic chemicals or drugs that damage the peripheral nervous system (PNS). Histopathological assessment of nerve biopsy is often utilized to diagnose a neuropathy; however, electrophysiological measures, such as nerve conduction assessments, can be useful in conjunction with histopathological analysis when nerve damage is suspected or confirmed. The scope of this review will focus on nerve conduction assessments to evaluate drug-induced peripheral neuropathies in nonclinical species within the context of nonclinical safety assessments during drug development.
Anatomy of the PNS
The PNS is a complex network of nerves located outside of the brain and spinal cord, which functions to transmit signals from the central nervous system (CNS) to the rest of the body or vice versa. A single nerve encapsulates a bundle of axons projecting from neuronal cell bodies and enables the transmission of electrochemical impulses along those axons. Nerves are generally classified by function and are motor, sensory, autonomic, or mixed. Axons within a nerve are typically referred to as “nerve fibers,” which can be myelinated or unmyelinated and vary in size ranging from 2 to 20 µM and 0.2 to 3 µM diameter, respectively. 1 Myelination of a nerve fiber in the PNS is maintained via Schwann cells, which enables impulses to travel rapidly via saltatory conduction, whereas unmyelinated or thinly myelinated nerves have slower conduction velocities. 2,3
Motor nerve fibers carry impulses from the spinal cord to skeletal muscle. Motor neuron cell bodies reside in the ventral gray matter of the spinal cord with efferent axonal projections that synapse on the neuromuscular junction of the muscle fibers it innervates. 4 Sensory nerve fibers are responsible for carrying sensory information (ie, proprioception, pain, touch, vibration, etc) from the PNS to the CNS. Sensory neurons are bipolar, with neuron cell bodies residing in the dorsal root ganglion with afferent projections from the periphery and efferent projections to the CNS. 5 The autonomic nervous system has both sympathetic and parasympathetic components with neuron cell bodies located in the lateral horn of the spinal cord and brain stem/sacral spinal cord, respectively. Autonomic nerve fibers control involuntary or semivoluntary functions of smooth muscle such as heart rate and blood pressure. 6 The enteric division of the autonomic nervous system innervates the gastrointestinal tract and functions to maintain appropriate gastric motility and secretion. 7 The peripheral spinal nerves are classified as mixed nerves, as they contain motor, sensory, and autonomic nerve fibers.
Toxic Neuropathy
Peripheral neuropathy is a debilitating condition involving the damage and/or degeneration of nerves in the PNS. Neuropathy can develop due to underlying disease or upon exposure to toxic chemicals or drugs that target peripheral nerve structure, function, and/or support mechanisms (ie, Schwann cells, scavenger systems, etc) and can include direct damage to axons, loss of myelin, and progressive deficits in saltatory conduction. Many different mechanisms have been shown to contribute to the development of toxic neuropathy depending on the pharmacology of the toxin, including oxidative stress, mitochondrial dysfunction, immune-mediated factors, inflammation, cytoskeleton disruption, and disruption of axonal transport. 8 –11
Depending on the mechanism, location, and degree of nerve injury, the physiological reaction of the nerve can vary. 1,12 Injury that is targeted at the level of the cell body is termed “neuronopathy,” damage localized to the axon is an “axonopathy,” and injury to the myelin sheath is termed “myelinopathy.” Neuronopathies can be limited to motor, sensory, or autonomic neurons or may involve any combination or nerve types. Axonopathies can impact a range of nerve fiber types, but toxicant-induced axonopathies typically affect large-diameter fibers at the distal axon. 1,8,9,11 Regardless of the site of injury (cell body, axon, or myelin), end-stage neuropathies are characterized by both demyelination and axonal disintegration. 1
Neuropathies that are characterized by degeneration of the distal axon typically retain viability of the cell body for some time, and the peripheral component of the nerve fiber can usually regenerate if the toxicant is removed. 11 Wallerian degeneration is a complex active process of cellular and molecular events resulting in axonal fragmentation and degeneration distal to the site of the initial lesion. This mechanism is necessary to facilitate successful remyelination and repair of the distal nerve via Schwann cells. 13 On the other hand, chronic denervation and/or decreased axonal maintenance by Schwann cells can result in irreversible nerve injury. 13 –15 Thus, drug-induced neuropathies may be reversible with cessation of treatment, but this is not always the case, as is observed with some chemotherapeutic agents, 10 and reversibility/recovery is likely dependent on multiple factors including duration of dosing, location (eg, distal vs proximal), and/or mechanism of the nerve injury, genetic factors, and/or variability in individual nerve repair capacities and scavenger systems. There are several useful published reviews of the animal models used for peripheral neuropathy assessments, which are beyond the scope of the current review. 13,16 –18
Histopathological assessment of PNS morphology via nerve biopsy is the gold standard assessment to confirm diagnosis of peripheral neuropathy as it is highly validated and provides definitive evidence of injury and corresponding severity. However, histopathology is limited in some aspects, for example, there is only a narrow window of time where dead and dying cells can be captured microscopically, after which there is no evidence of axon organelles and potentially any evidence of the presence of axon may disappear. 12 Furthermore, histopathological assessment affords only a snapshot in time rather than an ability to monitor progression. Thus, electrophysiological measures of nerve function can be used to complement histopathological assessments where nerve damage is suspected or confirmed. 19
Nerve Conduction Studies
Depending on the degree of damage and the specific types of nerves affected, clinical presentation of neuropathy can include motor and/or sensory deficits such as muscle cramps, paresthesia, weakness, tingling, numbness, or pain in the extremities. 8,11,20 In the early stages of nerve injury, neuropathies are subclinical and asymptomatic, and therefore, if PNS toxicity is suspected, electrophysiological tests could be conducted to diagnose early signs of nerve damage as well as to monitor progression and recovery. 21 A common electrodiagnostic test to identify and characterize nerve injury is a nerve conduction study, which measures various electrophysiological parameters of the nerve and can typically differentiate between neuropathies primarily affecting axons versus those primarily affecting myelin.
General Methodology and Measurements
Nonclinical nerve conduction assessments are minimally invasive, typically utilize subcutaneous needle electrodes, and include one or more stimulating electrode(s), a recording electrode, and a reference or grounding electrode. Supramaximal electrical stimulation of the nerve can be achieved via a constant voltage square pulse. Generally, a nerve is stimulated by placing the stimulating electrode over a portion of the nerve and an action potential is recorded at a site some distance away via the recording electrode. The parameters that are measured for the compound muscle action potential or sensory nerve action potential (SNAP) include the amplitude (in voltage), latency (in milliseconds), duration (in milliseconds), and area (in millivolts milliseconds) of the action potential (Figure 1). Nerve conduction velocity (NCV; measured in meters/second) can be calculated by dividing the distance between the stimulating and recording electrode by the action potential onset latency.
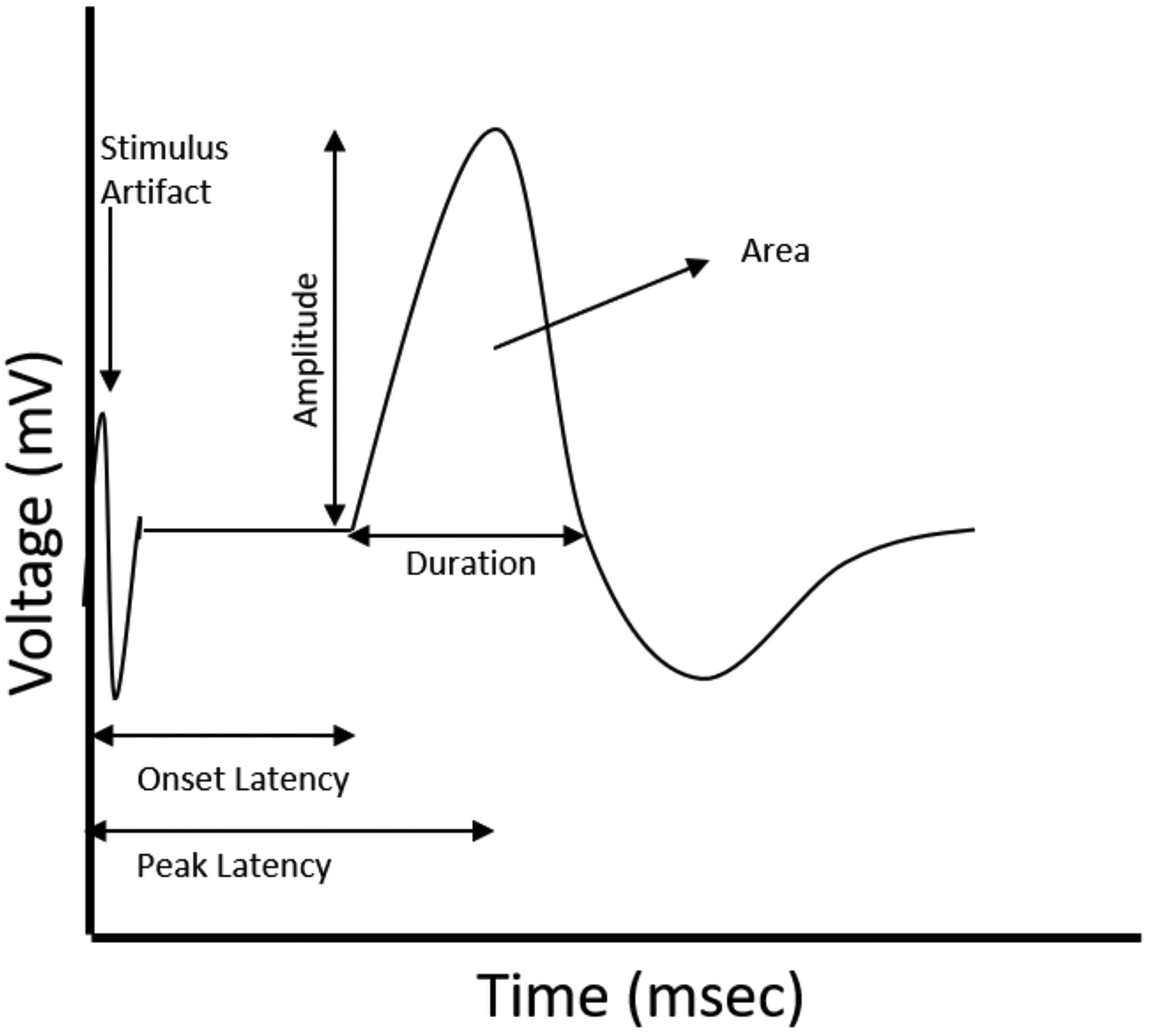
Schematic representing the components of a nerve action potential waveform, including the amplitude, latency (onset and peak), duration, and area of the action potential.
Motor responses are typically in the millivolt (mV) range and are larger than sensory responses, which are typically in the microvolt range (µV). Thus, motor nerve assessments are technically easier due to a lower signal to noise ratio. However, because of the anatomy of specific nerves, there are technical considerations that must be taken into account to record a primarily sensory versus a primarily motor NCV. For example, the peripheral spinal nerves are mixed nerves made up of both myelinated and unmyelinated axons that carry motor, sensory, and autonomic signals. Peripheral branches of the spinal nerves are either primarily sensory (eg, cutaneous nerve branches innervating the skin) or mixed (eg, nerve branches innervating a specific muscle containing not only axons of primary motoneurons but also afferent sensory projections).
To measure the conduction velocity of a primarily sensory nerve, one should place the stimulating electrode on the distal portion of the sensory branch and the recording electrode proximal to the stimulating electrode (ie, before the branch). Sensory NCV is then calculated by dividing the distance between the 2 electrodes by the latency of the stimulus to reach the recording electrode (Figure 2).
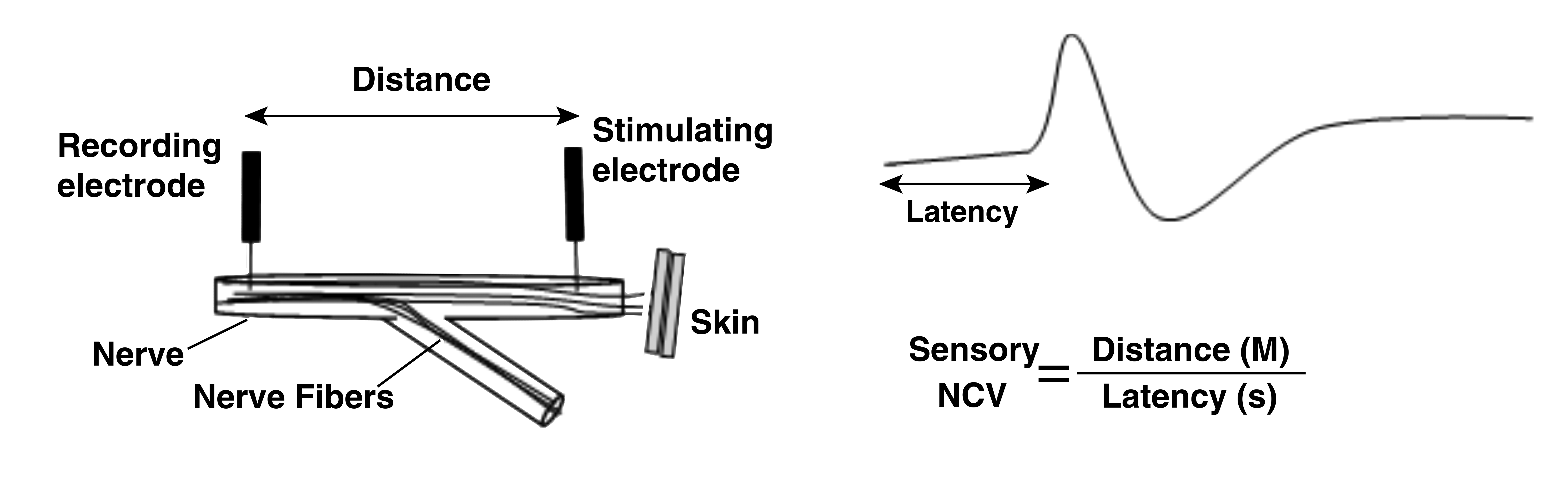
A schematic representing measurement of a sensory NCV. The nerve is stimulated on the distal portion of the nerve and the action potential recorded from the proximal portion of the nerve. Nerve conduction velocity (M/s) is calculated as the distance between the stimulating and recording electrode (M) divided by the onset latency of the action potential(s). NCV indicates nerve conduction velocity.
Measurement of motor nerve conduction is more complex, and the electrophysiologist must monitor the response of the muscle fibers to ensure that the response is primarily a result of motor axon stimulation. Furthermore, when recording a stimulus at the level of the muscle, there are conduction delays at the neuromuscular junction and throughout the muscle that confound the measured latency of the stimulus to reach the nerve terminus. To circumvent this and obtain an accurate measure of motor NCV, one can place the distal recording electrode on the belly of the muscle and 2 stimulating electrodes some distance apart proximal to the recording electrode. In this scenario, 2 stimulus latencies will be recorded with the stimulus from the electrode closest to the recording electrode having the shorter latency. Each latency represents the time for the stimulus to reach the muscle, and by subtracting the 2 latencies, one can ascertain the time it took for the stimulus to travel between the 2 stimulating electrodes. Thus, motor NCV can be calculated by dividing the difference in the distance between the 2 stimulating electrodes by the difference between the 2 latencies (Figure 3).
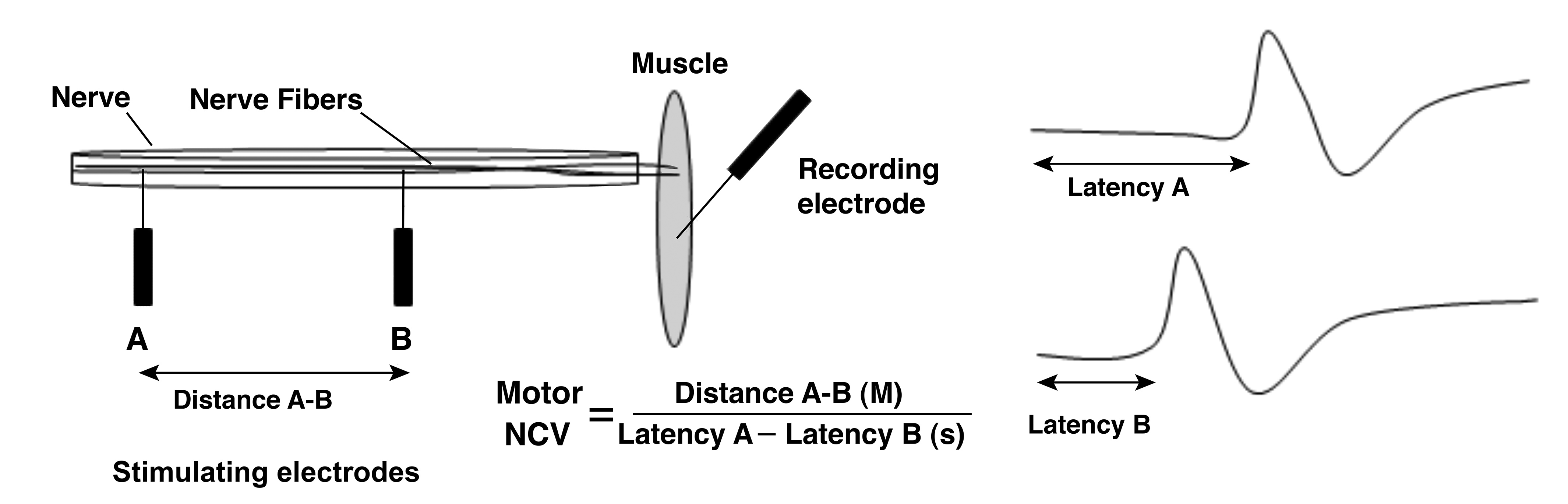
A schematic representing measurement of a motor NCV. The nerve is stimulated at 2 sites on the proximal portion of the nerve and the action potential recorded from the distal portion of the nerve. Nerve conduction velocity (M/s) is calculated as the distance between the 2 stimulating electrodes (M) divided by the difference in the onset latency of each action potential(s). NCV indicates nerve conduction velocity.
Interpretation/Examples
Generally, the amplitude of the action potential reflects the number and synchrony of responding fibers, whereas conduction velocity reflects the speed at which the electrical stimulus travels along the nerve. Action potential amplitude is sensitive to changes in axon density and/or diameter and NCV is sensitive to changes in myelination status, as myelinated axons conduct electrical impulses faster than nonmyelinated axons. 3 Therefore, the specific morphologies of the nerve action potential can vary depending on the stage/severity of the peripheral neuropathy and mechanism of nerve injury (Figure 4).
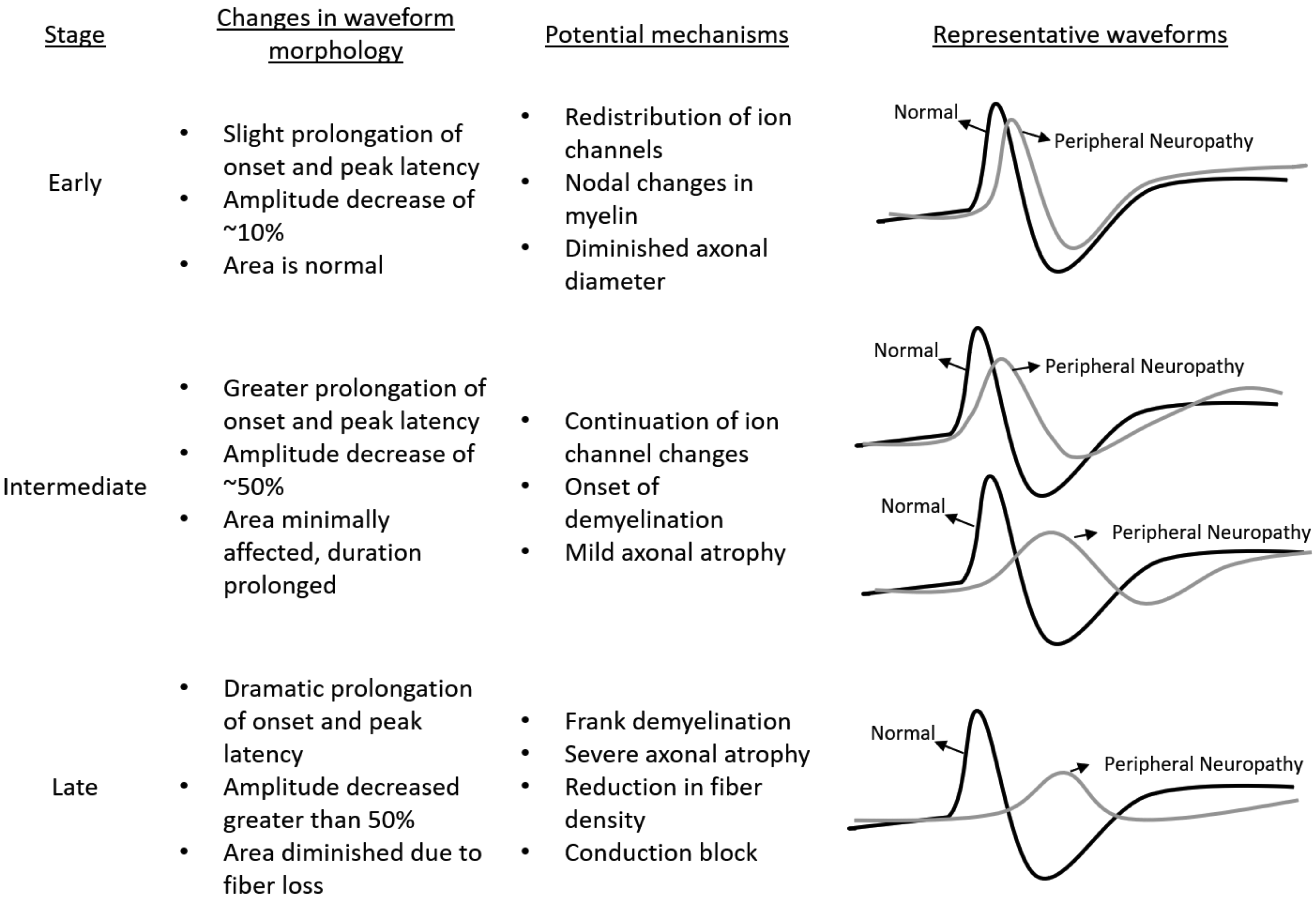
Example nerve action potential waveform morphologies and corresponding potential mechanisms of nerve injury in early-, intermediate-, and late-stage neuropathy.
One of the most important strengths of full nerve electrophysiology is the opportunity to document changes over time in the same subject. It is thus possible to document progressive worsening or improvement over time in a targeted nerve. Histopathology provides detailed, accurate, and localized measures of nerve structural integrity at a single time point. In contrast, standard electrophysiology can document changes (worsening or improvement) in nerve function in the same subject over an extended time period.
Although alterations in the nerve action potential via nerve conduction assessment alone may detect a deficit in peripheral nerve function, it is important to confirm the presence and severity of structural nerve damage via histopathological evaluation. There are a number of reports that document a strong correlation between behavioral, electrophysiological, and histopathological end points in nonclinical studies investigating toxic neuropathy. 22,23 For example, chemotherapy-induced peripheral neuropathy (CIPN) is a well-documented toxic neuropathy associated with most anticancer drugs, and manifestation of this toxicity typically presents as a sensory neuropathy. The mechanisms leading to CIPN are not fully understood but are thought to involve DNA damage, oxidative stress, mitochondrial toxicity, and/or ion channel remodeling. 10 In one study, 22 pharmacologically diverse chemotherapeutics such as paclitaxel (taxane), cisplatin (platinum analog), vincristine (vinca alkaloid), and bortezomib (proteasome inhibitor) were characterized via behavior, electrophysiology, and histopathology following subacute administration in C57Bl/6 mice. Behavioral assessments indicated that all chemotherapeutics tested resulted in mechanical allodynia and gait alterations. The SNAP amplitude decreased ∼30% to 50% from baseline for all chemotherapeutics tested, which is consistent with the decrease in axon density and mean axon diameter noted in the histopathology assessment. Furthermore, NCV was reduced to ∼90% of control in mice administered cisplatin and paclitaxel but was not affected by vincristine or bortezomib, which is consistent with the lack of an effect on myelin thickness. However, the ratio of axon diameter to myelin was slightly increased in cisplatin-treated animals, suggesting a hypomyelination status, which may explain the slight reduction in NCV in these animals. 22
It is important to note, however, that electrophysiological end points do not always correlate with visible structural alterations and some neuropathies are not detected by standard nerve conduction end points. For example, mechanisms primarily affecting transmembrane currents via alteration of ion channel dynamics could affect NCV without a histopathological correlate. 19 On the other hand, there may be instances where structural nerve damage is present in the absence of a functional nerve conduction deficit, as can be observed in small-fiber neuropathy. 24 –26 Small-fiber neuropathy primarily affects the small-diameter myelinated and unmyelinated nerve fibers; however, standard nerve conduction tests evaluate the function of large nerve fibers and are generally insensitive to neuropathies affecting only small-diameter fibers. Clinical presentation of small-fiber neuropathy can include symptoms of painful burning in the extremities (in a stocking glove distribution), decreased pain and thermal thresholds, and autonomic dysfunction. 25,26 Diagnosis of a small-fiber neuropathy can involve quantitative sensory testing (to detect altered sensory thresholds), evaluation of autonomic function, and skin biopsy for epidermal nerve fiber density assessment. 24,25 Small-fiber neuropathies can progress to involve damage to the large-diameter fibers, and thus, standard nerve conduction assessment should be conducted to fully characterize the extent of the neuropathy. Therefore, if peripheral nerve damage is suspected, it is important to select the appropriate behavioral, electrophysiological, and histopathological assessments to adequately characterize the potential toxicity.
Advantages, Limitations, and Considerations
There are many advantages to nerve conduction assessments. Electrophysiological tests of nerve function are quantitative and noninvasive. Nerve conduction studies can be measured longitudinally to monitor progression and/or recovery of functional deficits. Also, there are comparable techniques available across not only nonclinical species but also humans.
Nonetheless, it is important to remember that nerve conduction studies have some limitations that must be taken into account when interpreting whether or not a toxicity exists. Electrophysiological assessments measure the properties of the entire nerve rather than individual nerve fibers, and there are redundancies in nerve organization that make subtle, subclinical deficits difficult to detect. As such, it is the largest and fastest conducting fibers within the nerve that are actually represented in the NCV calculation. Therefore, if only a subset of axons are affected, or if the neuropathy is purely a small-fiber neuropathy, motor and sensory NCVs may be normal even with the underlying pathology. In addition, nerve conduction assessments only evaluate the portion of the nerve between the stimulating and recording electrodes and thus any abnormal functionality outside of this small area could be overlooked. Therefore, it is important for the electrophysiologist to carefully select the location for electrodiagnostic assessment.
Additional factors that affect electrophysiological parameters and should be considered when designing a nerve conduction study is the temperature, size, and age of the subjects. Temperature affects NCV nonlinearly in rats, 27 dogs, 28,29 and humans, 30 and thus, it is important to understand the thermal effects of any compound or drug candidate under investigation in order to reliably measure conduction velocity. Size and age of the subject can also affect electrophysiological measures of nerve function, for example, baseline values for distal latency and measurement of nerve length differed depending on dog weight, 31 and peripheral nerve function (action potential amplitude and conduction velocity) declines with age in mice 32 as well as in humans. 33 It is important to have size and age-matched comparator groups, as well as baseline measurements of nerve function in a nerve conduction study, particularly if there will be multiple time points measured.
Use of Nerve Conduction Studies in Nonclinical Drug Development
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use M3(R2) on nonclinical safety studies for the conduct of human clinical trials and marketing authorization for pharmaceuticals 34 and Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers 35 describe the regulatory guidance around nonclinical safety studies to support clinical trials and setting of the maximum recommended starting dose for first-in-human clinical trials. Electrophysiological assessment of nerve function is not a standard assessment in nonclinical toxicity studies, and therefore, nerve conduction studies may only be conducted prior to the observation of a structural nerve injury via histopathological assessment in a nonclinical toxicity study if there is a theoretical or known risk for toxic neuropathy based on the primary and/or secondary pharmacology of the compound being evaluated.
The majority of nerve conduction assessments in nonclinical dug development are conducted retrospectively following an observation of peripheral nerve injury in a histopathology assessment in a standard toxicity study. As such, nerve conduction assessment would then be conducted in the most sensitive toxicity species, which is typically a rodent or nonrodent (dog or nonhuman primate) species. In these cases, alterations in nerve conduction can potentially provide a functional correlate to the histopathology observation, a measure of recovery or reversibility of the functional deficit as well as identification of a monitorable end point to detect the emergence of similar toxicity in clinical trials and thus reduce patient risk. A longitudinal assessment (either in a nonclinical or clinical study) allows for subjects to be monitored prior to administration of a drug, over long periods of drug administration and additionally once the drug is withdrawn. Such a study design provides a comprehensive view of the time course of toxicity and well as worsening and/or recovery of the insult once the offending toxin is removed. However, it is important to recognize that electrophysiology and histopathology do not always correlate, and interpretations of dissociated electrophysiological and histopathological data must be carefully considered.
Conclusions and Future Directions
Nerve conduction assessment is a noninvasive, quantitative measure of nerve function that can be used to detect and/or monitor the progression and/or recovery of functional nerve deficits across nonclinical species and humans. As with any application, there are considerations specific to the physiology of the nervous system as well as the technique itself that should be considered when designing a nerve conduction study and interpreting the results. Most notably, electrophysiological nerve assessment measures the function of the entire nerve rather than individual nerve fibers and as such may not detect minor, subclinical structural alterations, injury to a subset of axons within a nerve, or neuropathies that purely affect small fibers. Furthermore, nerve conduction assessment only allows deficits contained within the portion of the nerve studied to be detected and therefore requires an experienced electrophysiologist to determine the appropriate location to conduct electrodiagnostic assessment.
A significant gap in knowledge exists regarding translation of drug-induced toxic effects of new chemical entities across nonclinical species and human, as these comparisons are often confounded by differences in methodology between laboratories, as well as interspecies differences in primary and/or secondary pharmacological target distribution, target function, and/or metabolic and pharmacokinetic profiles of the compound in question. In addition, the degree to which a nonclinical species can recover, or reverse, nerve injury may be largely dependent on specific genetic factors and/or variability in individual nerve repair capacities and scavenger systems. Thus, a generalized ranking of species sensitivity to drug-induced nerve injury cannot be deduced without side-by-side comparison of compounds across species, and therefore, dedicated studies comparing toxic effects of drugs on nerve function across species are needed.
Nonetheless, the range of options that are now available for scoring initial drug-induced toxic effects on NCV, the ability to document changes in sensitivity over time and length, and the support of novel means of tracing NCV have greatly enhanced our understanding and interpretation of nerve conduction parameters. Based to a large degree on animal studies and in parallel to the measures outlined above, there is now greatly improved set of procedures to document changes in all elements of nerve conduction in the clinical setting.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
