Abstract
Quantitative assessment of epidermal nerve fibers (ENFs) has become a widely used clinical tool for the diagnosis of small fiber neuropathies such as diabetic neuropathy and human immunodeficiency virus–associated sensory neuropathy (HIV-SN). To model and investigate the pathogenesis of HIV-SN using simian immunodeficiency virus (SIV)-infected Asian macaques, we adapted the skin biopsy and immunostaining techniques currently employed in human patients and then developed two unbiased image analysis techniques for quantifying ENF in macaque footpad skin. This report provides detailed descriptions of these tools and techniques for ENF assessment in macaques and outlines important experimental considerations that we have identified in the course of our long-term studies. Although initially developed for studies of HIV-SN in the SIV-infected macaque model, these methods could be readily translated to a range of studies involving peripheral nerve degeneration and neurotoxicity in nonhuman primates as well as preclinical investigations of agents aimed at neuroprotection and regeneration.
Keywords
Introduction
Transmission of afferent sensory information from the periphery to the central nervous system (CNS) is accomplished by a variety of sensory nerve types, ranging from the large caliber, thickly myelinated fibers that convey the senses of innocuous touch and limb position to the small diameter thinly myelinated Aδ fibers and unmyelinated C-fibers that transmit the sensations of temperature and pain (McGlone and Reilly 2010). Small fiber neuropathies (SFNs) are defined as conditions that selectively or predominantly affect the Aδ or C-fibers (Hoeijmakers et al. 2012). The clinical manifestations of SFN typically involve a spectrum of positive and negative sensory abnormalities that may include neuropathic pain, paresthesias (spontaneous tingling or burning sensations), hyperalgesia, and numbness (Hoeijmakers et al. 2012; McArthur 2012; Gibbons 2014). The etiologies associated with SFN are numerous and include common metabolic disorders such as diabetes mellitus (Russell and Zilliox 2014) and hyperlipidemia (Morkavuk and Leventoglu 2013), infectious diseases such as HIV (Kaku and Simpson 2014) and herpesviral infections (Steiner 2012), as well as rare hereditary (Brouwer et al. 2014) and autoimmune conditions (Oomatia et al. 2014). Relevant to neurotoxicology, several pharmaceutical compounds, including chemotherapeutic agents (Cavaletti et al. 2010) and antiretroviral drugs (Cherry et al. 2003; Ellis et al. 2008; Dorsey et al. 2015), have frequently been associated with peripheral neurotoxicity leading to SFN. For some patients, these detrimental side effects can be dose limiting and negatively impact their quality of life (Hong, Tian, and Wu 2014). Thus, it is imperative to assess the peripheral neurotoxicity of new drugs during preclinical testing (Authier et al. 2009).
The diagnosis and study of SFN have historically been challenging (McArthur 2012). Standard nerve conduction studies are relatively insensitive to alterations in small diameter nerve fibers and are often normal in patients with SFN (Herrmann et al. 1999; Gonzales-Duarte, Ronbinson-Papp, and Simpson 2008; Hoeijmakers et al. 2012). Similarly, morphologic examination of peripheral nerve tissue, such as the sural or sciatic nerves, may not reflect early changes to small fibers that may begin in distal regions (Herrmann et al. 1999). In recent years, visualization and assessment of epidermal nerve fibers (ENFs) in skin biopsies has become a standard clinical tool to diagnose SFN caused by a wide variety of conditions (Ebenezer et al. 2007; Lauria et al. 2010). Unlike quantitative sensory testing that relies on patient responses and is subject to bias (Hlubocky et al. 2009), ENF analysis is objective and can be applied to animal models (Lauria et al. 2005). Skin biopsy is also a relatively quick technique that is applicable to longitudinal sampling in contrast to performing sural or sciatic nerve biopsies (David 2008). In several conditions, including HIV infection, loss of ENF density has been shown to correlate strongly with severity of neuropathic symptoms (Zhou et al. 2007; Obermann et al. 2008).
In this report, we describe the development and implementation of skin biopsy as a tool to study sensory nerve alterations in simian immunodeficiency virus (SIV)-infected macaques. Previous studies by our group have shown that SIV-infected pigtailed macaques (Macaca nemestrina) develop morphologic and functional lesions of the peripheral nervous system (PNS) that closely parallel those seen in HIV patients with neuropathy (Laast et al. 2011). These include inflammation, glial activation, and viral replication in the lumbar spinal cord (Mangus et al. 2014) and dorsal root ganglia as well as decreased conduction velocity in isolated C-fibers from the sural nerve (Laast et al. 2011). Because skin biopsy and ENF quantification are currently the gold standard to diagnose and monitor SFN in HIV patients, we sought to adapt this technique for use in macaques. In addition to pigtailed macaques, we have evaluated ENF of uninfected and SIV-infected rhesus macaques (Macaca mullata), allowing for comparison of normative and experimental data between macaque species. Through these studies, we have learned valuable lessons regarding skin sample collection and processing, quantitative image analysis of ENF, and selection of study animals. These lessons are highly applicable to any study evaluating the PNS of nonhuman primates, including preclinical testing of potentially neurotoxic compounds or novel drugs aimed at the treatment of SFN.
Material and Method
Animals
In total, 117 pigtailed macaques (M. nemestrina) and 22 rhesus macaques (M. mulatta) were evaluated in this study. Pigtailed macaques had a mean age of 4 years (range 1–9 years) and were male; rhesus macaques had a mean age of 12 years (range 7–15 years). The rhesus macaque group included 3 males and 19 females. For SIV studies, macaques were inoculated intravenously with a combination of the neurovirulent clone SIV/17E-Fr and the immunosuppressive viral swarm SIV/DeltaB670 as previously described (Clements et al. 2011). Age-matched uninfected macaques served as procedural controls (i.e., control animals). At study end points, animals were euthanized and whole body perfused with room temperature (RT) sterile saline. This removes blood from vasculature, thus allowing for quantification of virus and other factors, such as cytokines, in tissues with minimal blood contamination. For the pigtailed macaques, which demonstrate an accelerated, predictable SIV disease progression (Mankowski, Clements, and Zink 2002), specific end points were predetermined according to stage of disease (acute phase at 7 and 10 days postinfection [dpi], asymptomatic phase at 21, 35, 42, and 56 dpi, and terminal AIDS at 84 dpi). Because the course of disease progression is more variable in rhesus macaques, these animals were euthanized when 2 or more AIDS-defining criteria were met (mean length of infection 213 days; Beck et al. 2015). Macaques were pair housed in facilities accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International, and all procedures were performed according to principles set forth by the Johns Hopkins University Institutional Animal Care and Use Committee and the National Research Council’s Guide for the Care and Use of Laboratory Animals. Macaques were fed a complete commercial diet (Harlan Laboratories, Cambridgeshire, UK) and provided with daily enrichment including fresh fruits and vegetables.
Skin Biopsy Collection and Fixation
Skin biopsies for ENF analysis in human patients are typically obtained from an area of the distal leg above the lateral malleolus (Hoeijmakers et al. 2012). However, the high density of hair follicles and adnexa present in haired macaque skin results in variation among sections and obscures visualization of ENF. We therefore obtained skin samples from the glabrous (nonhaired), skin of the metatarsal footpad between the third and fourth digits. To obtain preinfection biopsies, which were utilized for longitudinal ENF studies, animals were anesthetized with ketamine hydrochloride (15–20 mg/kg), and 3-mm punch biopsies were collected from the left metatarsal footpad. Biopsy sites were then covered with antibiotic ointment, followed by a sterile nonadherent dressing and elastic bandaging, which animals typically removed themselves within 24 hr. No systemic antibiotics or analgesics were needed postoperatively, and the procedure was generally well tolerated. Postinfection biopsies were obtained from the right metatarsal footpad at necropsy. The biopsy sample was handled carefully using fine-tipped forceps to grasp only the deep dermal fat and avoid any crush artifact to the epidermis.
Skin biopsies were immersed in Zamboni’s fixative (Newcomer Supply, Middleton, WI) and fixed for 12 to 24 hr at 4°C. Compared to other standard fixatives, such as formalin and paraformaldehyde, we have found that Zamboni’s better preserves antigenicity in the ENF, and it is stable for long periods at RT. After fixation, sections were rinsed with 0.08 M Sorensen’s phosphate buffer (prepared from 0.4 M Sorenson’s buffer: 7.176 g Na3PO4 monobasic monohydrate and 49.4 g Na3PO4 dibasic anhydrous dissolved in 1 L distilled water, pH 7.6). Fixed sections were transferred to a cryprotective buffer (20% glycerol in 0.08 M Sorensen’s phosphate buffer) and stored at 4°C until processing (typically within 1 week).
Immunohistochemistry
Protein gene product 9.5 (PGP9.5) is widely expressed in neuronal cell bodies and axons, including ENFs (McCarthy et al. 1995). Although PGP9.5 antibodies can be used in paraffin-embedded tissues, cryosectioning reduces tissue shrinkage and allows for the production of consistent, thick sections, which are necessary when implementing stereological methods. For these studies, cryoprotected skin samples were cut perpendicular to the epidermis at a thickness of 50 μm using a freezing sliding microtome (Microm HM 440E, GMI, Ramsey, MN). Each biopsy yielded approximately 48 to 50 sections, from which 4 sections were chosen for immunostaining. Sections were selected at roughly fixed intervals (e.g., 10th, 20th, 30th, and 40th sections) to be representative of the whole biopsy. Individual sections were transferred to 96-well plates and immunostained for the pan-axonal marker PGP9.5 using a free-floating section method as previously described (McCarthy et al. 1995). All staining procedures were conducted at RT, and all incubations were performed with gentle agitation on an orbital shaker (Mini Orbital Shaker, VWR, Radnor, PA). Sections were first bleached with 0.25% potassium permanganate for 5 min followed by 5.0% oxalic acid for 2 min to reduce melanin pigmentation. The sections were blocked for 10 min with a solution of 1.0% Triton X-100 (Sigma, St. Louis, MO) with 0.5% powdered milk (Bio Rad, Hercules, CA) and 5% normal goat serum (Vector, Burlingame, CA) in Tris-buffered saline (250 mM Tris base, 250 mM NaCl, pH 7.4), then incubated with the primary anti-PGP9.5 antibody overnight (product #7863-1004; rabbit polyclonal, 1:10,000, ABD Serotec, Oxford, UK). The following day, sections were incubated with a biotinylated secondary antibody (product #BA-1000; goat antirabbit, 1:100, Vector) for 1 hr and treated with 1% hydrogen peroxide in 30% methanol/phosphate buffered saline for 30 min to quench endogenous peroxidase activity. Sections were then incubated for 1 hr in avidin/biotin complex solution (product #PK-6100; ABC, Vector) and developed for 5 to 8 min using the Vector SG peroxidase substrate kit (product #SK-4700), which produces dark blue-gray chromogenic staining. The stained sections were mounted on chrome alum-gelatin subbed slides, air dried for 30 min at RT, and rinsed in RT tap water for 1 min to remove dried salt residues. Finally, slides were lightly counterstained with dilute eosin (1%) for 1 min and coverslipped with Permount (Fisher Scientific, Pittsburgh, PA). This procedure results in final section thickness ranging from 35 to 40 μm.
Quantitative ENF Assessment
Measurement of ENF density in human skin biopsies is typically achieved by manually counting all PGP9.5-stained ENFs that cross the dermal–epidermal junction and dividing this number by the linear length of the skin section in millimeters (McCarthy et al. 1995). Although this method is highly sensitive and reproducible in human skin samples (A. G. Smith et al. 2004) and has also been utilized for peripheral neuropathy studies in feline immunodeficiency virus–infected cats (J. M. Kennedy et al. 2004), the remarkably rich sensory innervation of the macaque footpad makes manual counting of individual fibers cumbersome and poorly reproducible. Thus, we developed two quantitative image analysis techniques, one based on ENF density and another on ENF length, to facilitate ENF assessment in our macaque skin samples. For both techniques, slides were blinded prior to analysis to reduce operator bias.
Measurement of ENF Density
The method used to determine ENF density in macaque footpad samples was adapted from the confocal “optical sectioning” technique described by W. R. Kennedy et al. (2005). It combines the thick sections required to visualize linear objects in tissues with standard, two-dimensional image anaylsis tools to determine the percentage of epidermal area occupied by PGP9.5-stained nerve fibers. Using a bright-field microscope equipped with a Z-axis motor (Carl Zeiss, Oberkochen, Germany) and IP Lab software (Scanalytics, Campbell, CA, version 3.9), Z-stack images were obtained from adjacent, nonoverlapping, 400× fields for each immunostained skin section. To reduce operator bias during image acquisition, the stage was moved in a systematic grid-like manner to collect nonoverlapping images encompassing the entire epidermal region on the section (typically 10–15 fields). Section thickness (Z distance) was measured at each imaging field by the operator focusing on the top and bottom of the tissue. Serial Z-stack images for each field were collected at 0.5-μm intervals throughout the Z-axis and then collapsed into a single image. For each collapsed image, the epidermal region of interest (ROI) was defined by tracing along the dermal–epidermal junction and the stratum corneum using the paint function. Using iVision software (BioVision Technologies, Exton, PA, version 4.0.14), collapsed images were then binarized by setting the threshold at the right-most inflection point in the image intensity histogram, and PGP9.5 immunoreactivity was measured as percentage of the ROI (%ROI) area occupied by positively stained pixels. To adjust for minor variations in skin section thickness, results were normalized to the thickness of each sample by dividing the %ROI value by the average Z distance of the section.
Measurement of ENF Length
An advantage of quantifying length rather than density of linear structures in tissue sections is the ability to employ nonbiased, software-guided stereologic image analysis techniques. These methods are based on counting random intersections of the object of interest, such as an axon or blood vessel, with a geometric probe that is superimposed over the image of the tissue (Mouton et al. 2002). Additionally, stereologic algorithms provide an estimate of the total length of objects in a specimen by systematic random sampling (SRS), which saves time compared to analyzing an entire specimen. To measure length of ENFs in macaque footpad samples, we used the Stereo Investigator space ball probe (MBF Biosciences, Williston, VT, version 9), similar to the method described by Ebenezer and colleagues (2011) and (2012). Three, 50-μm thick, PGP9.5-immunostained skin sections were evaluated per animal. For each section, an ROI in the epidermis, extending from the dermal–epidermal junction to the stratum corneum, was traced under a Nikon 4×/0.2 Plan Apo objective on a Nikon Eclipse E600 light microscope equipped with a motorized stage (see Figure 1, upper panel for illustration). The Stereo Investigator software then created an SRS grid of rectangular counting frames encompassing the entire ROI and moved the slide to random sampling sites via the motorized stage. At each sampling site, section thickness was measured by focusing on the top and bottom of the tissue using a Nikon 60×/1.4 oil Plan Apo objective. Total ENF length was estimated by counting intersections of nerve fibers with a hemispheric probe that had a 30-μm radius and 2-μm guard zones (to account for tissue irregularity at the edges of the sections). Only nerve fibers that were clearly within the epidermis (single, finely beaded fibers visualized within the epidermal ROI contour) were counted (Figure 1, lower panel). All stereology measurements were obtained using Stereo Investigator DAT files, and results were expressed as the estimated total length of PGP9.5-stained ENFs.
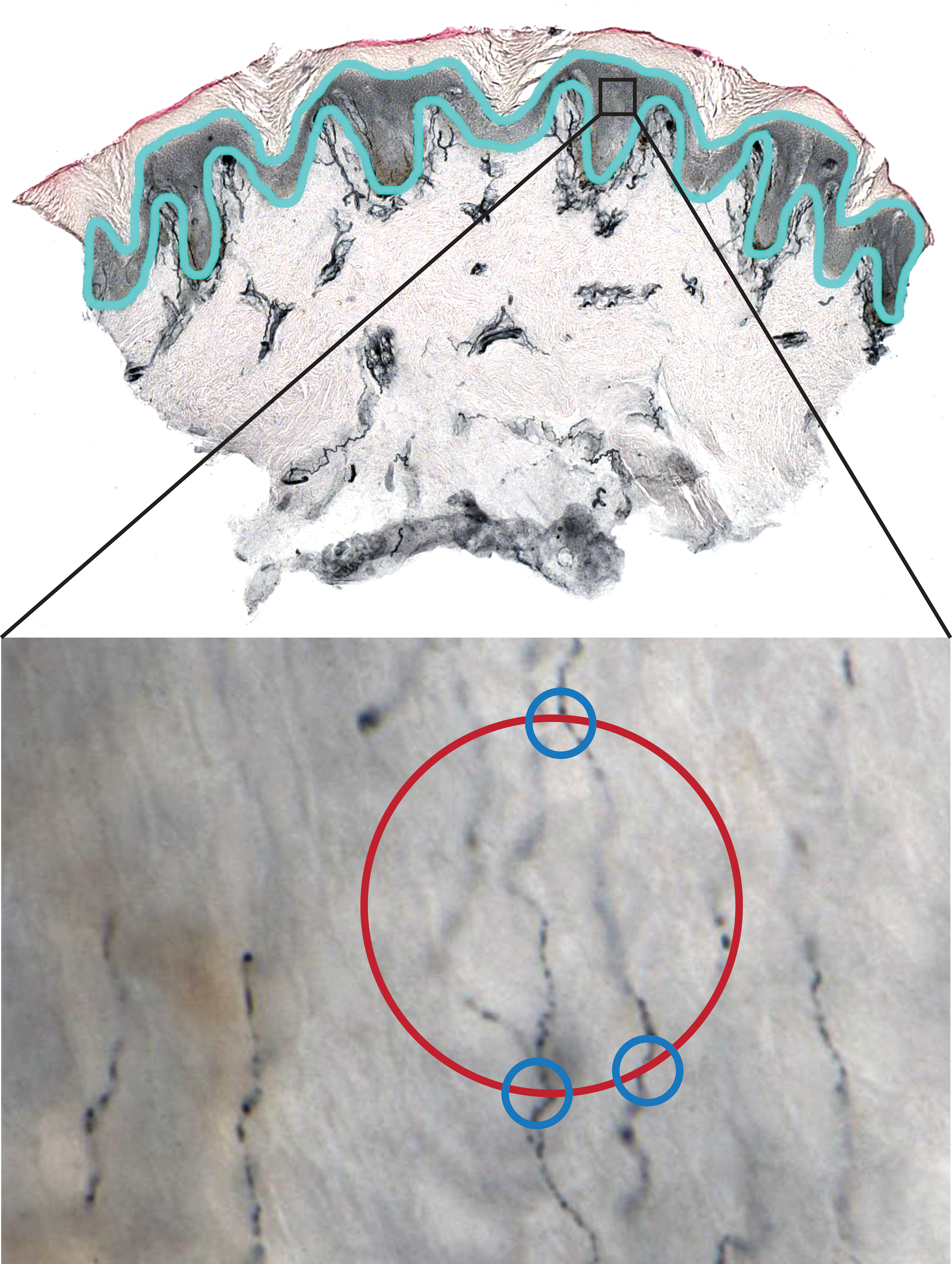
Illustration of the stereologic technique for measuring epidermal nerve fiber (ENF) length in a footpad skin biopsy. As depicted in the upper panel, the epidermis is traced (light blue outline) in a PGP9.5-stained skin section under low magnification. This serves as the region of interest (ROI). The Stereo Investigator software then systematically selects a subset of fields within the ROI for measuring ENF (represented by the black square). After switching to high magnification, as shown in the lower panel (60× oil objective), the probe is moved through the Z-axis of the tissue by a motorized stage and points of intersection between ENF and a hemispheric probe (represented by red circle) are marked by the user (blue circles). Intersections are only counted when the nerve fiber is in sharp focus at the point of crossing.
Statistics
All statistical analyses were performed using Prism software (GraphPad, San Diego, CA, version 5.0d) and nonparametric methods. The Mann–Whitney U test was used for 2-group comparisons, and the Spearman correlation coefficient was used to analyze relationships between continuous variables. Statistical inferences were considered significant when p < .05.
Results
Two ENF Quantification Methods Yield Strongly Correlative Results
In our SIV/macaque studies, we have found that ENFs can be effectively visualized in footpad biopsies by immunostaining thick cryosections with the pan-neuronal marker PGP9.5. Using these immunostained sections, 2 different image analysis techniques, one assessing nerve fiber density and the other measuring fiber length, were developed and found to be effective means of objectively quantifying ENF. Furthermore, when both quantification methods were applied to the same skin samples, we found a strong, direct correlation between the results (Figure 2), showing that either technique could serve as a valid tool in other macaque ENF studies. Once optimized, the time required to perform these techniques is similar (roughly 60–90 min per animal), and choice of methods would likely depend on available imaging equipment and software.
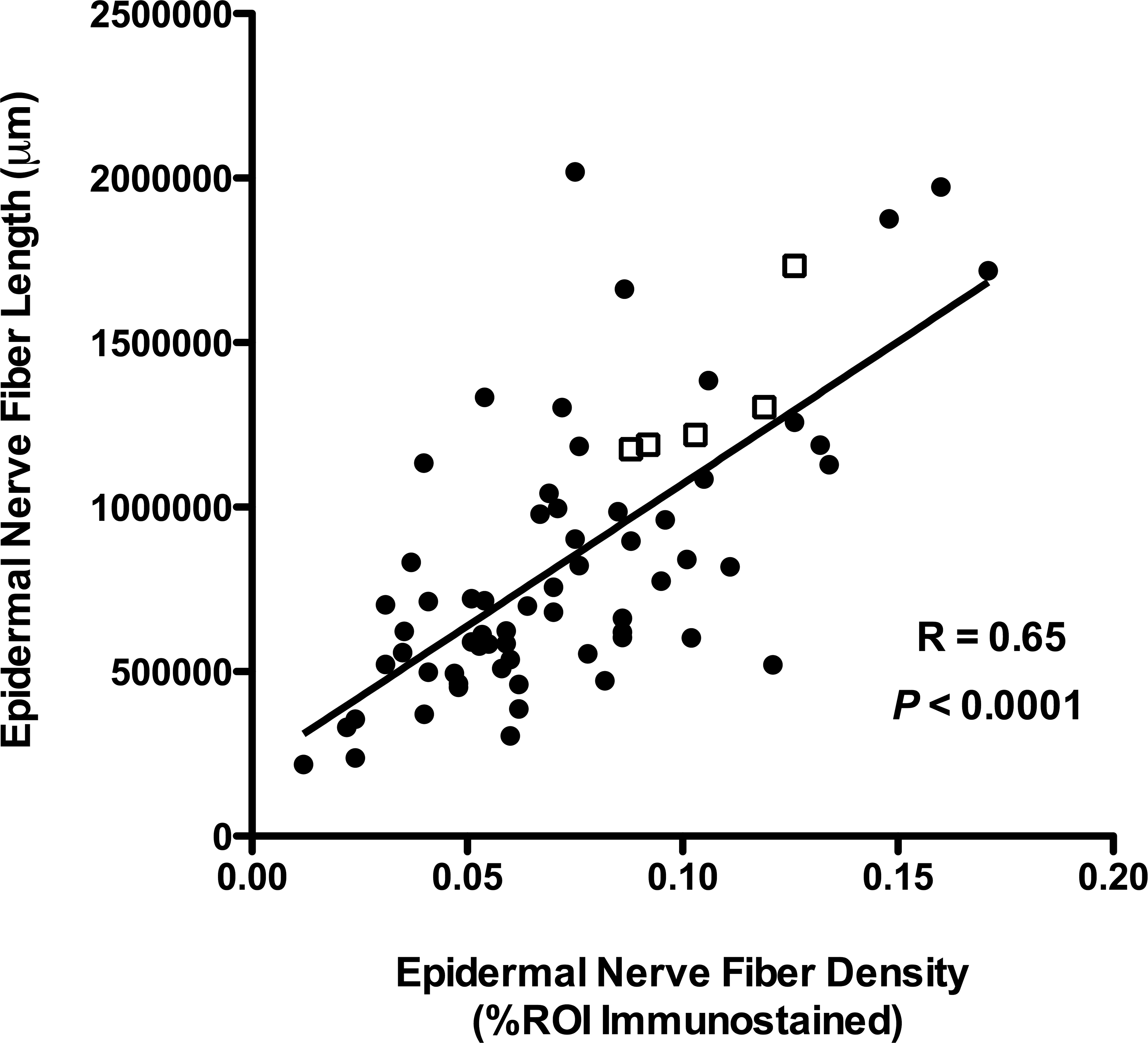
Two epidermal nerve fiber quantification methods yield strongly correlative results. Two different image analysis techniques, one measuring nerve fiber density and the other measuring fiber length, were developed. When both quantification methods were applied to the same skin samples from uninfected control (open squares) and simian immunodeficiency virus-infected pigtailed macaques (solid circles), there was a strong direct correlation between the techniques.
Species Differences in ENF Loss during SIV Infection
The species of macaque used in our group’s SIV work is based on the desired phenotypic outcome for each study. Since most of our studies have aimed to model the neurologic consequences of HIV infection, the vast majority of our study animals are pigtailed macaques, which more quickly and consistently develop SIV-associated CNS and PNS disease (Mankowski, Clements, and Zink 2002). Smaller numbers of rhesus macaques have been used in studies involving HIV-associated cardiac dysfunction, as this species survives longer following SIV infection and more readily develops functional cardiac abnormalities (Kelly et al. 2012). We have collected normative ENF data on uninfected control groups of both pigtailed (Figure 3A) and rhesus (Figure 3B) macaques. While the median ENF density among control rhesus was lower than that of pigtailed macaques, this difference did not reach statistical significance (p = .081, data not shown). However, in the context of SIV infection, only infected pigtailed macaques exhibited a significant decline in ENF density compared to control animals (p = .0033; Figure 3A). There was no significant difference in ENF density between rhesus macaques euthanized at terminal time points and control animals (p = .26; Figure 3B). This finding demonstrates the potential for considerable variation in host responses between closely related primate species and underscores the need for careful species selection in studies evaluating the PNS.
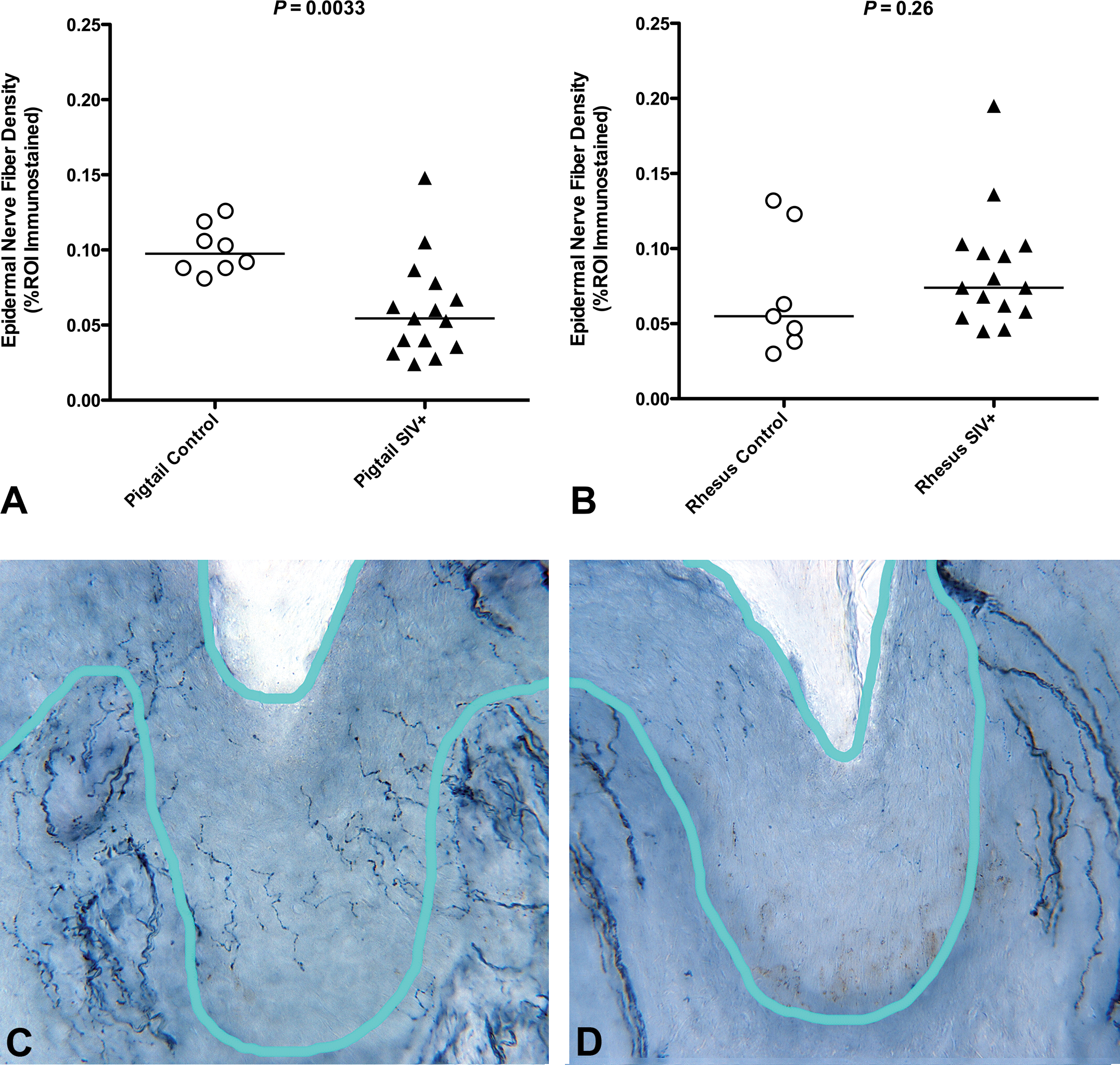
Species differences in epidermal nerve fiber (ENF) loss during simian immunodeficiency virus (SIV) infection. Pigtailed macaques exhibited significant decline in ENF density at 84 days post-SIV infection (A; p = .0033), but there was no significant difference in ENF density between rhesus macaques euthanized at terminal SIV time points versus control animals (B; p = .26, Mann–Whitney U test). Photomicrographs of skin biopsies from control (C) and SIV-infected (D) pigtailed macaques demonstrate a marked decrease in the number of thin, finely beaded PGP9.5-stained nerve fibers in the epidermis. The light blue line delineates the region of interest for assessing epidermal nerve fibers. Bundles of thicker PGP9.5-stained dermal nerve fibers can be seen below the dermoepidermal junction.
Pigtailed Macaques of U.S. and Indonesian Origin Have Different Baseline ENF Length
In addition to considering potential difference in ENF response between species, we examined whether pigtailed macaques from different originating countries had comparable ENF status prior to entering SIV studies. Interestingly, we found that pigtailed macaques imported from Indonesia had significantly lower baseline ENF length than macaques born in the United States (p = .0048; Figure 4). Whether this observation results from genetic or environmental differences between Indonesian and U.S. pigtailed macaque populations is currently unclear. However, this finding suggests that uniformity of animal origin may be a critical factor when designing studies evaluating ENF status of primates.
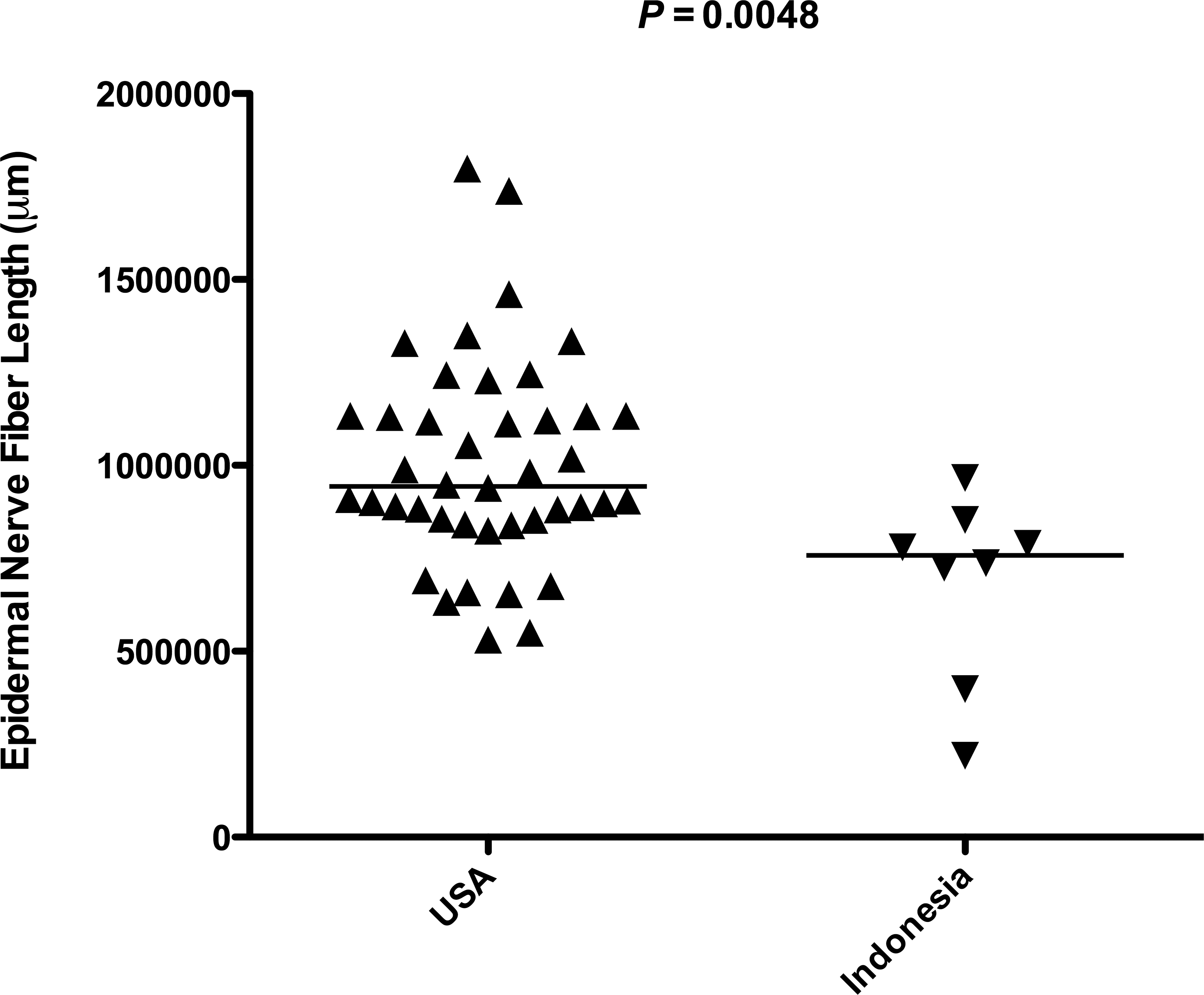
Differences in baseline epidermal nerve fiber (ENF) length based on country of origin. When comparing baseline ENF length of pigtailed macaques originating from different countries, animals from the United States had significantly higher baseline ENF than animals born in Indonesia (p = .0048, Mann–Whitney U test).
Measuring Change from Baseline Allows Longitudinal Assessment of ENF Loss
Skin biopsies are a minimally invasive and repeatable tool for assessing the status of peripheral sensory nerves in humans and animal models and therefore can be used for longitudinal assessment of individual subjects. In our more recent macaque studies, we collected preinfection footpad biopsies and quantified baseline ENF lengths that were then compared to values obtained from necropsy skin samples. Among pigtailed macaques, we found that SIV-infected animals euthanized during terminal disease (84 dpi) had a median decrease in ENF length of nearly 5 × 104 μm from their baseline value and that this change was significantly different from that observed among uninfected controls that were also euthanized at 84 days postmock infection (p = .0028; Figure 5). We believe that this longitudinal manner of ENF assessment produces more reliable data than cross-sectional sampling and measurement and plan to utilize this method in future studies of SIV neuropathy.
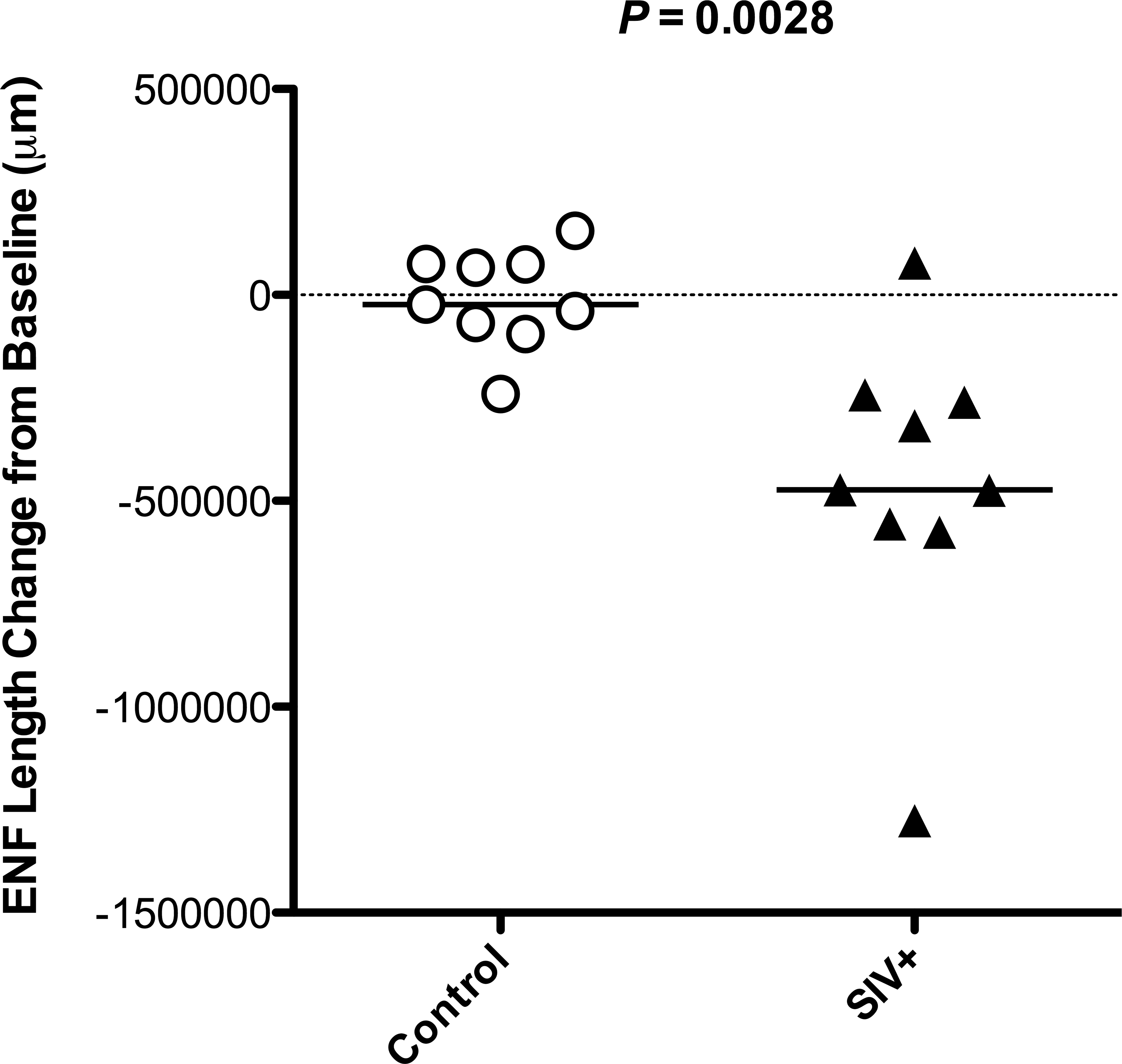
Measuring change from baseline allows longitudinal assessment of epidermal nerve fiber (ENF) loss. Footpad skin biopsies were collected from pigtailed macaques at the time of inoculation (simian immunodeficiency virus [SIV] or mock) and at necropsy, 84 dpi. ENF length measurements obtained from each animal’s necropsy skin samples were compared to measurement from their preinfection samples, and results were plotted as “change from baseline.” SIV-infected pigtailed macaques had a median decrease of approximately 5 × 104 μm, which was significantly different from the median change observed in uninfected animals, which was close to zero (p = .0028, Mann–Whitney U test).
Discussion
When studying an animal model of human disease or toxicity, a priority is to employ experimental outcome measures similar to those currently used in clinical settings. Over the past decade, assessment of sensory nerve fibers in skin biopsies has emerged as a sensitive indicator of early damage to distal unmeylinated nerve fibers and has supplanted older methods, such as sural nerve morphometry and electrodiagnostic studies, in the clinical diagnosis of HIV-associated sensory neuropathy (SN) and other SFNs. In line with this advancement, our group has worked to develop and implement techniques to measure progressive ENF loss in SIV-infected macaques. These techniques have proven to be key assets in our investigations of the pathogenesis of SIV-SN (Laast et al. 2011; Mangus et al. 2014; Dorsey et al. 2014). While distal ENF loss has been widely reported in rodent models of metabolic and toxic neuropathies, such as diabetic- and chemotherapy-induced peripheral neuropathy (Lauria et al. 2005; Siau, Xiao, and Bennett 2006; Drel et al. 2006; Höke and Ray 2014), published data on the ENF responses of nonhuman primates outside of SIV research are scarce. We propose that broader application of the techniques described herein for quantitative ENF analysis in Asian macaques could greatly advance preclinical studies of peripheral neurodegeneration and neurotoxicity in nonhuman primates.
Our work with both pigtailed and rhesus macaques has also revealed important experimental variables that need to be considered when planning ENF studies in primates. First, we found that there was no significant difference in footpad ENF density between uninfected control macaques of the 2 species, despite a substantial age difference between the groups. However, only pigtailed macaques exhibited significant ENF loss during the course of SIV infection. ENF density in SIV-infected rhesus macaques with terminal disease was not significantly different from controls. In contrast, Lakritz and colleagues (2015) recently reported significant ENF decline in SIV-infected rhesus macaques that were also depleted of CD8+ cells including lymphocytes and natural killer cells. This discrepancy in experimental outcome between closely related macaque species emphasizes the importance of host immune responses in animal studies of peripheral neuropathy. Rodent studies have also revealed marked differences in the susceptibility of various mouse strains to peripheral neuropathy (Höke 2012). For example, in a study investigating the sensitivity of different inbred mouse strains to paclitaxel-induced neuropathy, S. B. Smith and colleagues (2004) showed that DBA/2J mice exhibited especially robust mechanical hypersensitivity after exposure to paclitaxel, while the C57BL/6J mice were relatively resistant to this change. The influence of genetic background has also been shown to be a critical variable in mouse models of diabetic neuropathy (Sullivan et al. 2008).
Second, when evaluating pigtailed macaques originating from different countries, we found that animals from Indonesia had significantly lower baseline ENF length than animals born in the United States. Although a relatively low number of Indonesian animals were included in this analysis, the robust difference was nonetheless evident in our data set. Whether this observation is related to genetic or environmental factors is not clear. Significant differences in ENF density have also been reported in people of distinct geographic origins (Shikuma et al. 2013). A recent study investigating ENF density as a marker of neuropathy risk in Thai HIV patients showed that very few subjects in that cohort developed neuropathic signs or symptoms despite use of stavudine, a known neurotoxic antiretroviral drug that is still often used in developing countries (Shikuma et al. 2015). While several factors likely contributed to this finding, the authors note that HIV seronegative Thai subjects have higher ENF density compared to published U.S. control values and that this higher baseline ENF density may provide Thai subjects with greater “ENF reserve.” Together, our findings suggest that both species and geographic origin are important variables to consider when planning studies of peripheral neuropathy in Asian macaques. While these variables are potential confounding factors in preclinical studies of peripheral neuropathy in primates, they could ultimately be informative in future research aimed at identifying “neuroprotective” host genes and environmental factors. Furthermore, these findings should encourage investigators to exercise caution when comparing macaque ENF values among different laboratories, especially regarding control data, as animal source and environment may affect the results.
Finally, we have shown that by obtaining footpad skin biopsies prior to SIV infection, each animal’s ENF measurement at necropsy can then be compared to its own preinfection ENF measurement. With this longitudinal sampling method, we were able to demonstrate that SIV-infected pigtailed macaques experienced a significantly greater loss of distal ENF length compared to control animals at 84 dpi or postmock infection. By using “change-from-baseline” data rather than single, cross-sectional ENF measurements, each macaque serves as its own control, which helps account for individual variation in baseline ENF values. Longitudinal sampling could be of great benefit for ENF studies facilitating use of smaller groups of animals or when uniformity of animals (age, origin, etc.) is not feasible. In addition, longitudinal ENF studies can be used to monitor ENF regeneration, as demonstrated by Ebenezer and colleagues in SIV-infected pigtailed macaques (2009).
Given its extensive distribution and anatomic complexity, systematic sampling and assessment of the PNS can be challenging, especially in large animal models. Skin biopsy and subsequent ENF analysis provide relatively simple methods for obtaining information about the status of peripheral sensory neurons and can be performed objectively with little specialized training. Moreover, loss of ENF density is a more sensitive indicator of early damage to small unmeylinated nerves than traditional histologic or morphometric nerve analyses. Our group’s efforts to evaluate ENF in the context of SIV-associated SN has led to the development of reliable tools and unbiased, quantitative techniques that could be applied to a wide range of peripheral neuropathy and neurotoxicity studies in primate species. Additionally, skin biopsies are relatively nontraumatic and well tolerated by humans and animals. Thus, they are repeatable and can be used for longitudinal assessment of individuals, both in the clinic and in the context of drug safety studies.
Footnotes
Author Contributions
Authors contributed to conception or design (LM, JD, JM); data acquisition, analysis, or interpretation (LM, JD, RW, GE, PH, VL, JM); drafting the manuscript (LM); and critically revising the manuscript (LM, JD, RW, GE, PH, VL, JM). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were supported by NIH NS055651, NIH MH070306, NIH NS077869, NIH NS089482, NIH ORIP P40 OD013117, NIH ORIP T32 OD011089, and NIH R25MH08066108.
